Published online Aug 28, 2025. doi: 10.4329/wjr.v17.i8.109146
Revised: May 25, 2025
Accepted: July 24, 2025
Published online: August 28, 2025
Processing time: 120 Days and 3.3 Hours
Ultrasound (US) can be a valuable tool for assessing arthritis associated with infla
A 40-year-old Caucasian woman with psoriatic arthritis (PsA) and ulcerative colitis (UC) reported experiencing finger pain, knee arthritis, and bloody diarrhea. She showed a high Disease Activity index for PsA score and a severe Mayo score. She began treatment with adalimumab. Over the course of six months, the 18 MHz US procedure was performed on her joints and a 3.5 MHz US on her in
More research is needed to find out the importance of using US in patients with PsA and UC as this could improve treatment strategies.
Core Tip: This case study highlights a promising ultrasound (US) use in the simultaneous assessment of joint and intestinal inflammation in a patient diagnosed with psoriatic arthritis (PsA) and ulcerative colitis (UC). By employing high-frequency US (18 MHz) to evaluate dactylitis as well as abdominal US (3.5 MHz) to detect colonic inflammation, the findings un
- Citation: Mendonça JA, de Aquino JLB. Does ultrasound detect joint and intestinal changes in psoriatic arthritis and ulcerative colitis after immunobiological treatment: A case report. World J Radiol 2025; 17(8): 109146
- URL: https://www.wjgnet.com/1949-8470/full/v17/i8/109146.htm
- DOI: https://dx.doi.org/10.4329/wjr.v17.i8.109146
Psoriasis (PsO) is associated with several manifestations, including joint involvement and systemic conditions such as uveitis and inflammatory bowel disease (IBD). Patients with PsO, psoriatic arthritis (PsA), and spondyloarthritis have a 1-4 times higher risk and a 1-6 times higher risk of developing IBD compared to the general population. IBD includes primarily two main forms: (1) Crohn's disease (CD); and (2) Ulcerative colitis (UC). Both CD and UC are chronic dis
The pathophysiology underlying the hypothesis of "arthritogenic bowel" involves genetic factors. A significant cause is the high prevalence of the human leukocyte antigen (HLA)-B27 antigen, found in approximately 25% of individuals with spondyloarthritis and 78% of those with IBD. Other genetic factors, such as HLA-DRB1 and HLA-B44, have also been identified, along with non-histocompatibility factors like variations in the CARD15 gene, which increases the risk of sacroiliitis in CD[4,5]. These conditions are supported by transcriptomic analysis, which identifies CD 64+ myeloid cells as a source of interleukin (IL)-23 stimulation in the intestine. The situation is exacerbated by epigenetic alterations th
Regarding AS, few reports detailing this disorder association with IBD are available. However, one study indicates a significant reduction in microbiota among AS patients, demonstrating dysbiosis similar to that seen in IBD patients, with a comparable bacterial profile. Nevertheless, stronger evidence is needed to provide more evidence for this association[7].
In recent years, high-resolution ultrasound (US), particularly Doppler technology in US, has proven beneficial in diagnosing and monitoring rheumatological diseases helping to evaluate synovitis and enthesopathies, especially in cases of subclinical disease activity. Advances in understanding intestinal imaging using US techniques have also allowed for the demonstration of pathological changes associated with IBD[8,9]. US can help characterize intestinal inflammation by highlighting hypoechogenicity and thickening of the inflamed intestinal wall, while Doppler assesses vascular changes due to intense neovascularization, inflammatory infiltration, small artery thrombosis, and dilation of arteries and veins that are manifestations of IBD[9,10].
This clinical case emphasizes the importance of US for assessing joint ill manifestations as well as detecting intestinal inflammation and helping monitoring treatments.
Two patients, 51 ± 11 years of age, were diagnosed with PsA and UC through colonoscopy. The intestinal US examinations performed revealed signs of inflammation while remaining blinded to their clinical conditions. One patient was female and the other was male. The female patient, a businesswoman, completed US treatment and follow-up, allowing her to be included in the study. In contrast, the male patient was excluded due to lack of follow-up on his intestinal US procedure.
The female patient had been diagnosed with PsO on her scalp and elbows eight years before.
After experiencing skin lesions for four years, she began to develop inflammatory joint pain while opening and closing her fingers, along with arthritis in her left knee. Three years after these joint complaints started, she experienced over 20 episodes of bloody diarrhea and was subsequently diagnosed with UC. Her father had been diagnosed with PsA 20 years earlier.
Before starting the adalimumab treatment, the patient’s exams indicated she had: (1) Mild PsO according to the Psoriasis Area and Severity Index (PASI); (2) High score on the Disease Activity Index for PsA (DAPSA); and (3) Severe Mayo score (Table 1). The clinical evaluation was conducted using the PASI, which measures the severity of PsO based on four criteria: (1) Crythema; (2) Induration; (3) Scaling; and (4) The extent of the affected area. The severity of skin involvement is categorized based on the surface area affected (BSA) as follows: (1) No PsO (0 BSA); (2) Mild (< 10 BSA); (3) Moderate (10-20 BSA); and (4) Severe (> 20 BSA)[11].
| Before | After | |
| Psoriasis Area and Severity Index | 8.3 | 2.0 |
| Disease Activity Index for Psoriatic Arthritis | 29.16 | 0 |
| Mayo score | 12 | 0 |
| Paratenonitis | Present | Absent |
| Fecal calprotectin | 5984 mg/kg | 600 mg/kg |
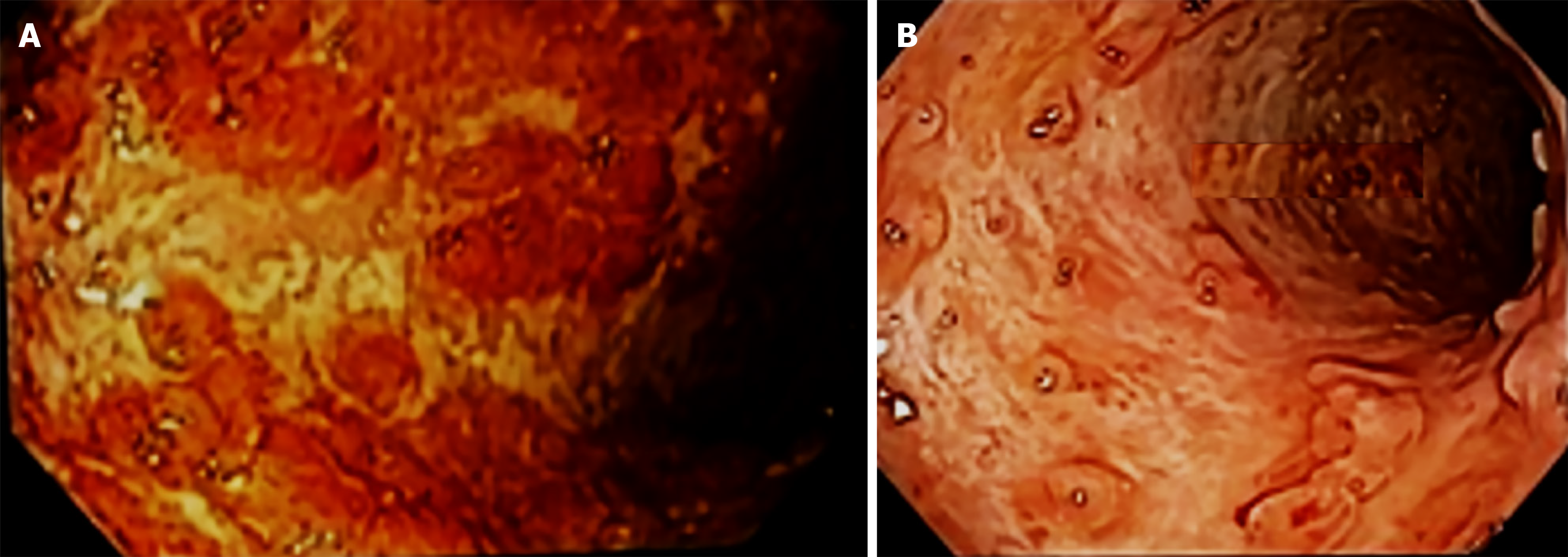
| Colonoscopy | Intense inflammation | Remission |
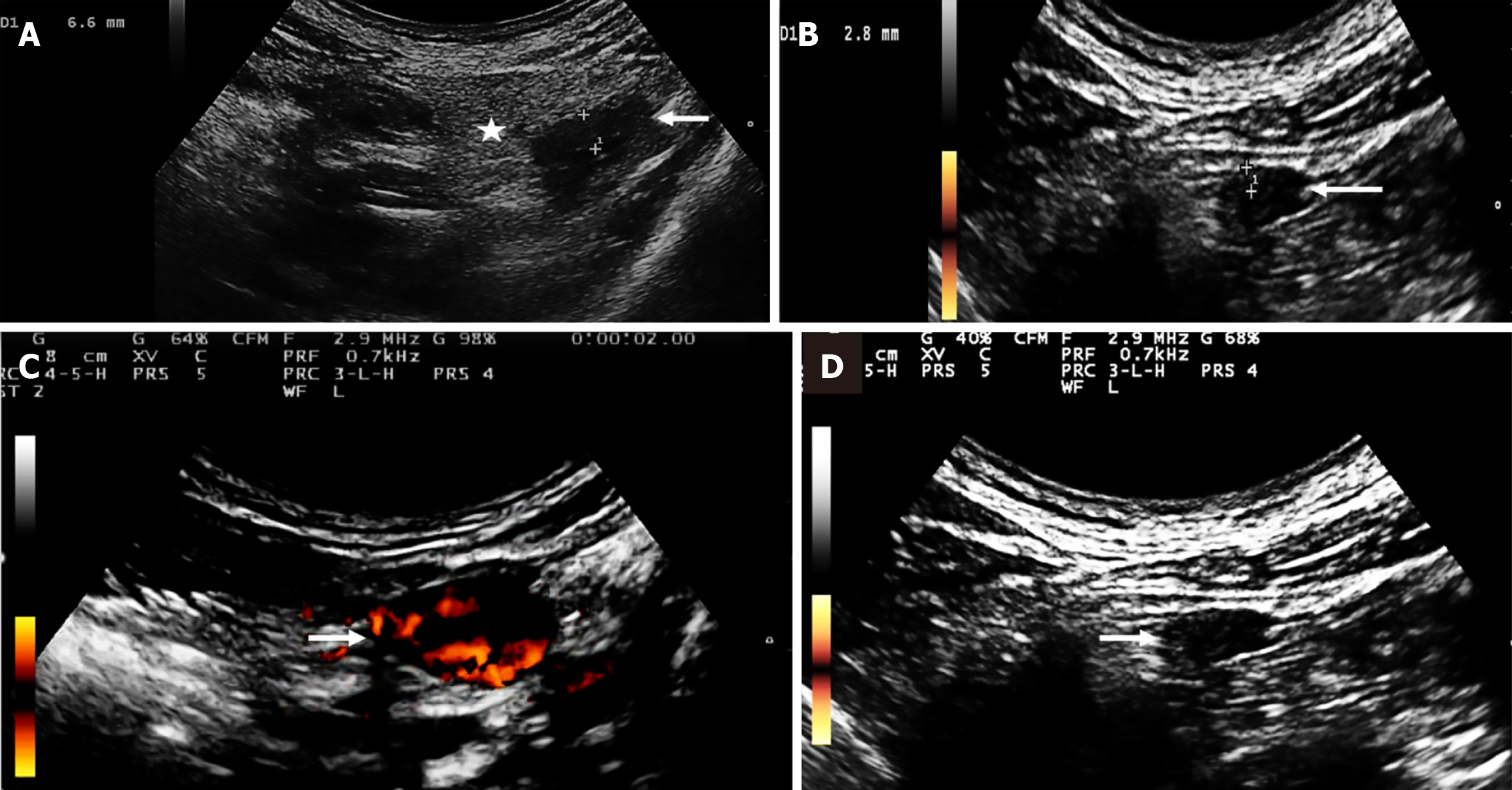
| Intestinal wall thickness | 6.6 mm | 2.8 mm |
| Degree of thickening of perienteric fat | 2 | 0 |
| Doppler degree in the intestinal wall | 3 | 0 |
To assess arthritis, the DAPSA score was used. This score includes the number of painful and swollen joints (28 in total), the patient’s visual analogue pain scale (ranging from 0 to 10), the physician’s assessment, and the inflammatory marker C-reactive protein. Disease activity is classified into four categories: (1) High (≥ 28); (2) Moderate (15-28); (3) Low (5-14); and (4) In remission (≤ 4)[12-14].
The patient’s laboratory exams showed a negative rheumatoid factor, a fecal calprotectin level of 5984 mg/kg[15], serology testing for hepatitis B, C, human immunodeficiency virus, and syphilis, a negative Mantoux test, and no anomalies on the chest X-ray.
The patient gave her consent by signing the Informed Consent Form for the US evaluation and other procedures condu
When there is a link between arthritis and IBD, the concept of an arthritogenic bowel becomes particularly important. This connection emphasizes the need for a multidisciplinary approach, which is associated with better clinical outcomes. These outcomes include higher rates of sustained remission, reduced hospitalizations stay and reduced surgical procedures, as well as an overall improved quality of life as reported by patients. Additionally, a multidisciplinary approach encourages collaborative clinical decision-making, humanizes patient care, and enhances the integration of support, education, and research within specialized centers. Therefore, it is crucial to promote and incorporate multidisciplinary consultations as a standard practice for managing patients with UC, especially in complex clinical situations within tertiary care centers and IBD referral units[18,19].
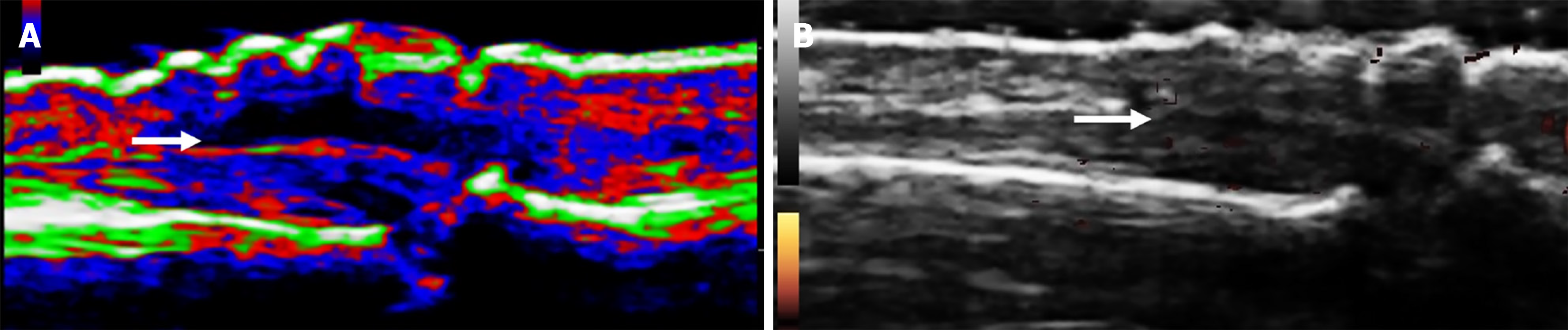
The fingers’ joints examination revealed findings consistent with dactylitis, which was attributed to significant paratenonitis in the second and third proximal interphalangeal joints of the right hand, characterized by effusion. A bowel US showed 6.6 mm edema in the sigmoid colon wall and indicated grade 2 mesenteric fat and grade 3 Doppler signal on the Limberg scale. Additionally, a colonoscopy revealed severe UC extending to the hepatic flexure and mild stenosis of the transverse colon, with a Mayo score 3 and 7 UC Disease Activity Index Score.
The treatment consisted of 100 mg adalimumab and azathioprine during six months.
All clinical, US, laboratory, and colonoscopy parameters showed improvement, indicating preserved mucosa with a distinct vascular pattern, with no edema, erosions, or ulcerations, and no signs of inflammatory activity (Table 1, Figures 1, 2 and 3).
High-resolution B-mode and Doppler ultrasonography effectively translate clinical and often subclinical dactylitis into observable conditions such as synovitis, tenosynovitis, enthesitis, and inflammation of both the subcutaneous tissue and the periextensor tendon. Recently, a dactylitis score scale has been developed for patients with PsA, as this condition can be challenging to detect during clinical examinations. Patients frequently exhibit US abnormalities without accompanying inflammatory signs. The clinical significance of dactylitis is highlighted by its role as a marker of disease aggressiveness and poor prognosis in affected patients. An US study found that 35.7% of PsO patients exhibited subclinical dactylitis compared to a control group, along with a 30% occurrence of moderate peritendinitis of the fingers’ extensor tendon[18,20,21].
The use of US in UC has proven beneficial as it helps to identify structural damage to the large intestine a damage which is an indication of an active inflammatory process in the bowel. Accuracy of US in assessing the bowel inflammation extent and severity, as well as in monitoring therapeutic responses has been evidenced[19].
The TNF plays a crucial role in the pathogenesis of immune-mediated inflammatory diseases; hence, inhibiting TNF is vital for managing severe and progressive inflammatory conditions. This intervention can lead to rapid and sustained clinical remission, enhance quality of life, and prevent damage to other target organs. Adalimumab, the first fully human monoclonal antibody targeting TNF, has demonstrated efficacy and tolerability in patients with various extensive inflammatory diseases, including PsA, plaque PsO, and IBD such as CD and UC. Accurate mapping of intestinal manifestations in patients with PsA is crucial, as it allows for more precise treatment approaches. Adjustments in the therapeutic classes and the potential increase of the anti-TNF drug dosage may be necessary for effectively managing intestinal inflammation, since differences in dosing requirements compared to arthritis treatment exist. Research indicates that a higher dosage of adalimumab achieves sustained efficacy in inducing remission of intestinal inflammation, reporting a 25% success rate in moderate to severe cases over four years[22,23].
However, the limitations of US in UC affect its effectiveness in the assessments, as inflammation primarily affects the mucosa. The technique is better suited for identifying changes in deeper bowel layers, as seen in CD. Visualizing the rectum can be challenging, and mild or microscopic inflammation may not be detected, the tissues often appearing normal. In cases of remission, the bowel wall thickness typically returns to normal, further decreasing US sensitivity. Factors such as flatulence, obesity, and operator’s skill can also impact image quality. Therefore, while US may have some use, it cannot replace colonoscopy for confirming remission or for monitoring[19].
Our study emphasizes the importance of joint and intestinal mapping when treating patients with a humanized mo
We are grateful to the patients for their participation in this exploratory and innovative investigation of joint and bowel imaging.
| 1. | Bolt JW, van Ansenwoude CMJ, Hammoura I, van de Sande MG, van Baarsen LGM. Translational Research Studies Unraveling the Origins of Psoriatic Arthritis: Moving Beyond Skin and Joints. Front Med (Lausanne). 2021;8:711823. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 2. | Ono K, Kishimoto M, Deshpande GA, Fukui S, Kawaai S, Sawada H, Matsuura M, Rodriguez VR, Proft F, Tada K, Tamura N, Taniguchi Y, Hirata A, Kameda H, Tsuji S, Kaneko Y, Dobashi H, Okano T, Haji Y, Morita A, Okada M, Komagata Y, Medina CL, Molto A, Dougados M, Hisamatsu T, Tomita T, Kaname S. Clinical characteristics of patients with spondyloarthritis and inflammatory bowel disease versus inflammatory bowel disease-related arthritis. Rheumatol Int. 2022;42:1751-1766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 3. | Schreiber S, Colombel JF, Feagan BG, Reich K, Deodhar AA, McInnes IB, Porter B, Das Gupta A, Pricop L, Fox T. Incidence rates of inflammatory bowel disease in patients with psoriasis, psoriatic arthritis and ankylosing spondylitis treated with secukinumab: a retrospective analysis of pooled data from 21 clinical trials. Ann Rheum Dis. 2019;78:473-479. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 145] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 4. | Danve A. Thoracic Manifestations of Ankylosing Spondylitis, Inflammatory Bowel Disease, and Relapsing Polychondritis. Clin Chest Med. 2019;40:599-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 5. | Atreya R, Abreu MT, Krueger JG, Eyerich K, Greving C, Hammaker D, Stoveken B, Hartman J, Leppard K, Sarabia I, Wertheimer J, Deming J, Kohler K, Li H, Freeman T, Hart A, Keyes B, Ritchlin C, Mcinnes IB, Allez M, Fourie A, Sachen K. P165 Guselkumab binding to CD64+ IL-23–producing myeloid cells enhances potency for neutralizing IL-23 signaling. J Crohns Colitis. 2024;18:i465-i465. [DOI] [Full Text] |
| 6. | Bourgonje AR, Ungaro RC, Mehandru S, Colombel JF. Targeting the Interleukin 23 Pathway in Inflammatory Bowel Disease. Gastroenterology. 2025;168:29-52.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 18] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 7. | Breban M. Gut microbiota and inflammatory joint diseases. Joint Bone Spine. 2016;83:645-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 8. | Balint PV, Terslev L, Aegerter P, Bruyn GAW, Chary-Valckenaere I, Gandjbakhch F, Iagnocco A, Jousse-Joulin S, Möller I, Naredo E, Schmidt WA, Wakefield RJ, D'Agostino MA; OMERACT Ultrasound Task Force members. Reliability of a consensus-based ultrasound definition and scoring for enthesitis in spondyloarthritis and psoriatic arthritis: an OMERACT US initiative. Ann Rheum Dis. 2018;77:1730-1735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 166] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 9. | Nassef MA, Botros SM, Adel Ghaffar MK. The update of ultrasound techniques in diagnosis of inflammatory bowel disease. Egypt J Radiol Nucl Med. 2014;45:289-294. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 10. | Huber MK, Valim V, Serrano ÉV, Mendonça JA, Lourenço RB, Espírito Santo TMR, Nordal H, de Fátima Bissoli M, de Oliveira Gavi MBR. Prevalence of spondyloarthritis in inflammatory bowel disease according ASAS and ultrassonography and its correlation with plasma calprotectin. Adv Rheumatol. 2024;64:27. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 11. | Mattei PL, Corey KC, Kimball AB. Psoriasis Area Severity Index (PASI) and the Dermatology Life Quality Index (DLQI): the correlation between disease severity and psychological burden in patients treated with biological therapies. J Eur Acad Dermatol Venereol. 2014;28:333-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 218] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 12. | Schoels MM, Aletaha D, Alasti F, Smolen JS. Disease activity in psoriatic arthritis (PsA): defining remission and treatment success using the DAPSA score. Ann Rheum Dis. 2016;75:811-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 344] [Cited by in RCA: 321] [Article Influence: 35.7] [Reference Citation Analysis (0)] |
| 13. | Hackett S, Coates LC. Outcome measures in psoriatic arthritis: Where next? Musculoskeletal Care. 2022;20 Suppl 1:S22-S31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 14. | Lewis JD, Chuai S, Nessel L, Lichtenstein GR, Aberra FN, Ellenberg JH. Use of the noninvasive components of the Mayo score to assess clinical response in ulcerative colitis. Inflamm Bowel Dis. 2008;14:1660-1666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 733] [Cited by in RCA: 711] [Article Influence: 41.8] [Reference Citation Analysis (0)] |
| 15. | Mahdipour M, Shafaghi A, Mansour-ghanaei F, Hojati A, Joukar F, Mavaddati S. Fecal calprotectin role in diagnosis of ulcerative colitis and treatment follow-up. J Coloproctol. 2019;39:115-120. [DOI] [Full Text] |
| 16. | Bruyn GA, Iagnocco A, Naredo E, Balint PV, Gutierrez M, Hammer HB, Collado P, Filippou G, Schmidt WA, Jousse-Joulin S, Mandl P, Conaghan PG, Wakefield RJ, Keen HI, Terslev L, D'Agostino MA; OMERACT Ultrasound Working Group. OMERACT Definitions for Ultrasonographic Pathologies and Elementary Lesions of Rheumatic Disorders 15 Years On. J Rheumatol. 2019;46:1388-1393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 161] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 17. | Frias-Gomes C, Torres J, Palmela C. Intestinal Ultrasound in Inflammatory Bowel Disease: A Valuable and Increasingly Important Tool. GE Port J Gastroenterol. 2022;29:223-239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 18. | Mendonca J, Heloísa Ferreira W, Gutierrez M. AB0481 Subclinical Dactylitis in Patients with Psoriasis: Role of Ultrasound. Ann Rheum Dis. 2024;83:1507-1508. [DOI] [Full Text] |
| 19. | Nagarajan KV, Bhat N. Intestinal ultrasound in inflammatory bowel disease: New kid on the block. Indian J Gastroenterol. 2024;43:160-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 20. | Naredo E, D'Agostino MA, Terslev L, Pineda C, Miguel MI, Blasi J, Bruyn GA, Kortekaas MC, Mandl P, Nestorova R, Szkudlarek M, Todorov P, Vlad V, Wong P, Bakewell C, Filippucci E, Zabotti A, Micu M, Vreju F, Mortada M, Mendonça JA, Guillen-Astete CA, Olivas-Vergara O, Iagnocco A, Hanova P, Tinazzi I, Balint PV, Aydin SZ, Kane D, Keen H, Kaeley GS, Möller I. Validation and incorporation of digital entheses into a preliminary GLobal OMERACT Ultrasound DActylitis Score (GLOUDAS) in psoriatic arthritis. Ann Rheum Dis. 2024;83:1060-1071. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 21. | Felbo SK, Østergaard M, Sørensen IJ, Terslev L. Which ultrasound lesions contribute to dactylitis in psoriatic arthritis and their reliability in a clinical setting. Clin Rheumatol. 2021;40:1061-1067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 306] [Article Influence: 51.0] [Reference Citation Analysis (0)] |
| 23. | Calvo Moya M, González Lama Y, Ruíz Antorán B, Omella Usieto I, El Hajra Martinez I, Santos Pérez E, Menchén Viso B, Matallana Royo V, González Partida I, de Lucas Tellez de Meneses R, Bella Castillo P, González Rodriguez M, Vera Mendoza MI. Treatment With Adalimumab 80 mg Every Other Week in Inflammatory Bowel Disease: Results of Treatment Intensification in Clinical Practice. Crohns Colitis 360. 2023;5:otac051. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |