Published online Aug 28, 2025. doi: 10.4329/wjr.v17.i8.107732
Revised: May 4, 2025
Accepted: July 18, 2025
Published online: August 28, 2025
Processing time: 153 Days and 8.7 Hours
Photon-counting detector (PCD) CT represents a transformative advancement in radiological imaging, offering superior spatial resolution, enhanced contrast-to-noise ratio, and reduced radiation dose compared with the conventional energy-integrating detector CT.
To evaluate PCD CT in oncologic imaging, focusing on its role in tumor detection, staging, and treatment response assessment.
We performed a systematic PubMed search from January 1, 2017 to December 31, 2024, using the keywords “photon-counting CT”, “cancer”, and “tumor” to ide
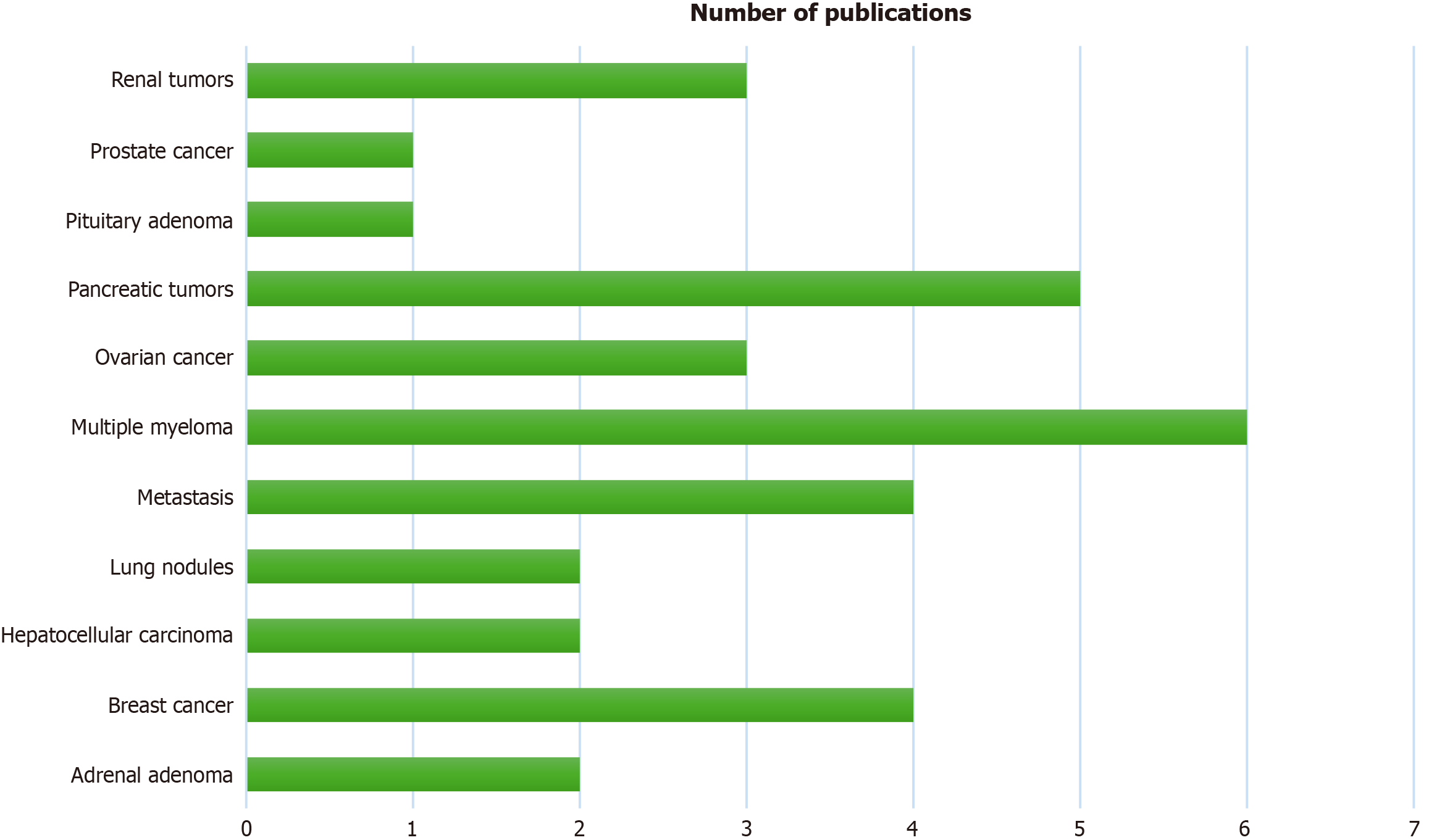
Key findings highlighted the advantages of PCD CT in imaging renal masses, adrenal adenomas, ovarian cancer, breast cancer, prostate cancer, pancreatic tumors, hepatocellular carcinoma, metastases, multiple myeloma, and lung cancer. Additionally, PCD CT has demonstrated improved lesion characterization and enhanced diagnostic accuracy in oncology. Despite its promising capabilities challenges related to data processing, storage, and accessibility remain.
As PCD CT technology evolves, its integration into routine oncologic imaging has the potential to significantly enhance cancer diagnosis and patient management.
Core Tip: Photon-counting detector (PCD) CT represents a paradigm shift in oncologic imaging, offering superior spatial resolution, reduced radiation dose, and enhanced material decomposition compared with conventional CT. This study highlighted the potential of PCD CT in tumor detection, characterization, and therapy response assessment, underscoring its role in advancing precision oncology. By leveraging energy-resolved photon detection, PCD CT enhances lesion conspicuity and quantification, paving the way for improved diagnostic accuracy and personalized treatment strategies.
- Citation: Perera Molligoda Arachchige AS, Dashiell A, Jesuraj AS, D’Urso AI, Fiore B, Cattaneo M, Pierzynska E, Szydelko S, Centini FR, Verma Y. Applications of photon-counting CT in oncologic imaging: A systematic review. World J Radiol 2025; 17(8): 107732
- URL: https://www.wjgnet.com/1949-8470/full/v17/i8/107732.htm
- DOI: https://dx.doi.org/10.4329/wjr.v17.i8.107732
Photon-counting detector (PCD) CT represents a groundbreaking advancement in radiological imaging, offering significant improvements over traditional CT modalities. Traditional CT uses energy-integrating detectors (EIDs), which sum the total energy deposited by X-ray photons. This process inherently reduces spectral information and limits spatial resolution and image quality, especially in patients with a high body mass index. Unlike traditional EIDs, PCD CT utilizes PCDs, which individually capture each X-ray photon and its energy, enabling enhanced spatial resolution, contrast-to-noise ratio, and spectral imaging capabilities while reducing radiation dose. Beyond the hardware innovation PCD CT integrates novel image reconstruction techniques such as virtual non-contrast (VNC) imaging, which allows reconstruction of unenhanced images from contrast-enhanced scans, eliminating the need for multiple phases, and quantum iterative reconstruction (QIR), which enhances image quality by reducing noise while preserving spatial detail. This technological evolution aims to address several inherent limitations of conventional CT, providing a more refined imaging tool for clinical practice[1].
The clinical potential of PCD CT has been under investigation for over a decade with early experimental systems demonstrating promising results. However, the technology initially faced challenges related to detector performance under high X-ray flux rates. A major milestone was achieved on September 29, 2021 when the Food and Drug Administration cleared the first PCD CT scanner developed by Siemens Healthineers for clinical use. This regulatory approval marked a turning point, providing real-world validation of the advantages of PCD CT[2]. This milestone marked the beginning of broader clinical adoption with over 50 installations worldwide and a sharp rise in scientific publications (over 500 in the last year) demonstrating increasing interest and application.
Among the various clinical domains where PCD CT is making strides, oncologic imaging is emerging as a key area of interest. The superior spectral resolution, improved lesion characterization, and reduced radiation dose of PCD CT offer distinct advantages in oncologic applications, particularly in tumor detection, staging, and treatment response assessment[3,4]. However, the literature is currently fragmented, spanning diverse tumor types and protocols. This systematic review aimed to consolidate these findings by evaluating trends, identifying strengths and limitations of PCD CT in oncologic imaging, and presenting visual comparisons to clarify its clinical value relative to conventional CT.
A systematic search was conducted for the period of January 1, 2017 to December 31, 2024 using PubMed to identify relevant literature on the applications of PCD CT in oncologic imaging. The search strategy included the following keywords: (“photon-counting CT”) AND (“cancer”) and (“photon-counting CT”) AND (“tumor”). Studies were considered eligible for inclusion if they were experimental studies involving human subjects or human phantom studies. Non-experimental reports such as scoping reviews, narrative reviews, systematic reviews, and meta-analyses along with editorials, letters, commentaries, non-English studies, and animal studies were excluded.
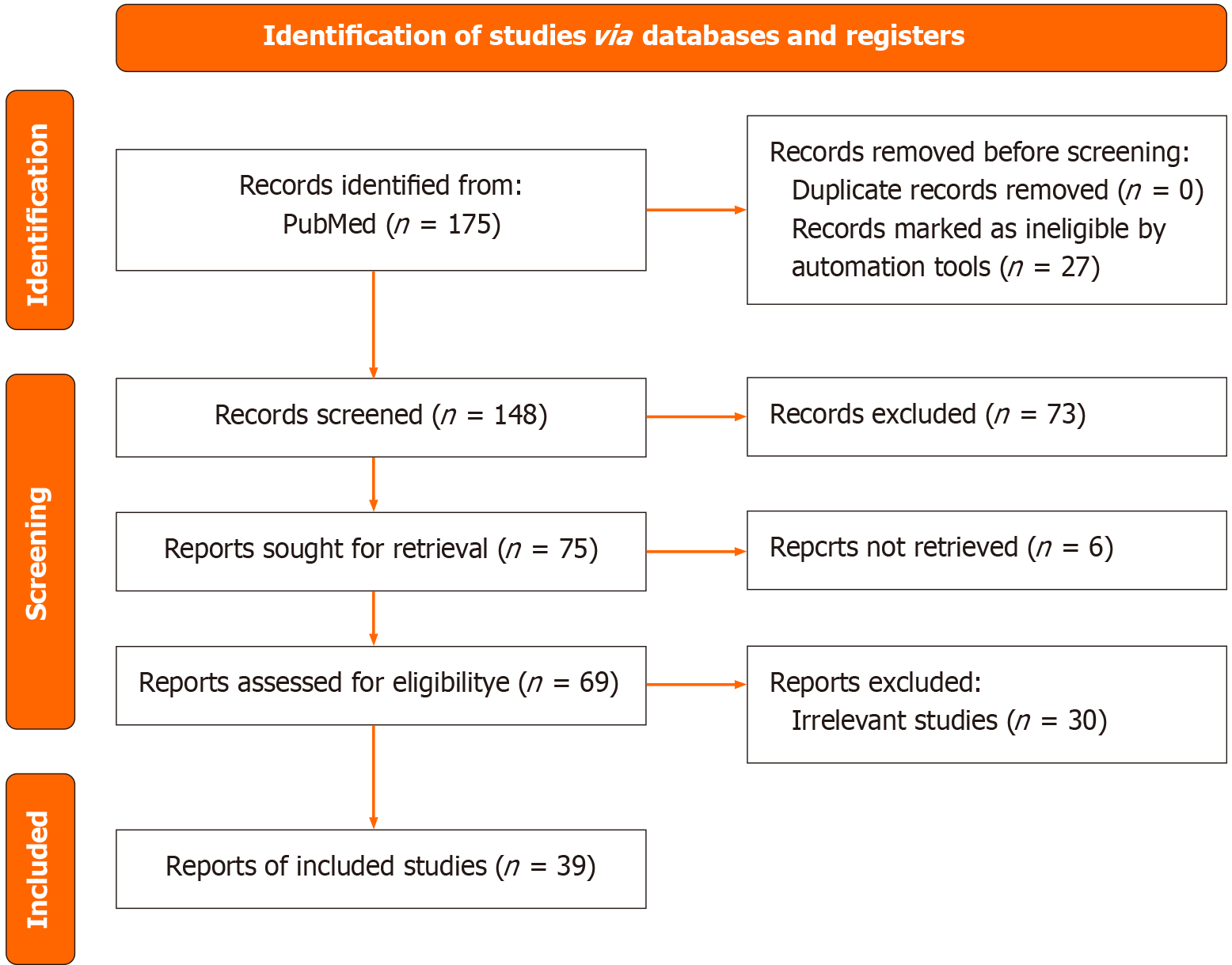
The study selection process followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines[5]. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram (Figure 1) outlines the screening process, including study identification, screening, eligibility assessment, and final inclusion. Initially, 175 studies were identified from PubMed. After the removal of 27 ineligible studies using automation tools, 148 studies remained for screening based on title and abstract. At this stage 73 studies were excluded, and the remaining 75 reports were sought for retrieval. Six reports could not be retrieved, leading to a full-text assessment of 69 studies. Following this assessment, 30 studies were excluded due to irrelevance, resulting in a final inclusion of 39 studies.
Data extraction focused on key study characteristics, including study type (comparison vs non-comparison), the first author’s country of origin, and the application of PCD CT in oncologic imaging. A formal risk of bias assessment was not conducted as the review primarily included experimental studies without direct intervention comparisons, thereby minimizing the risk of systematic bias in study design. Data synthesis was performed using a qualitative approach to summarize trends in the literature and the various applications of PCD CT in oncologic imaging.
This systematic review was not registered in PROSPERO because it did not involve a meta-analysis or a formal risk-of-bias assessment, both of which are key requirements for PROSPERO registration (Figure 1).
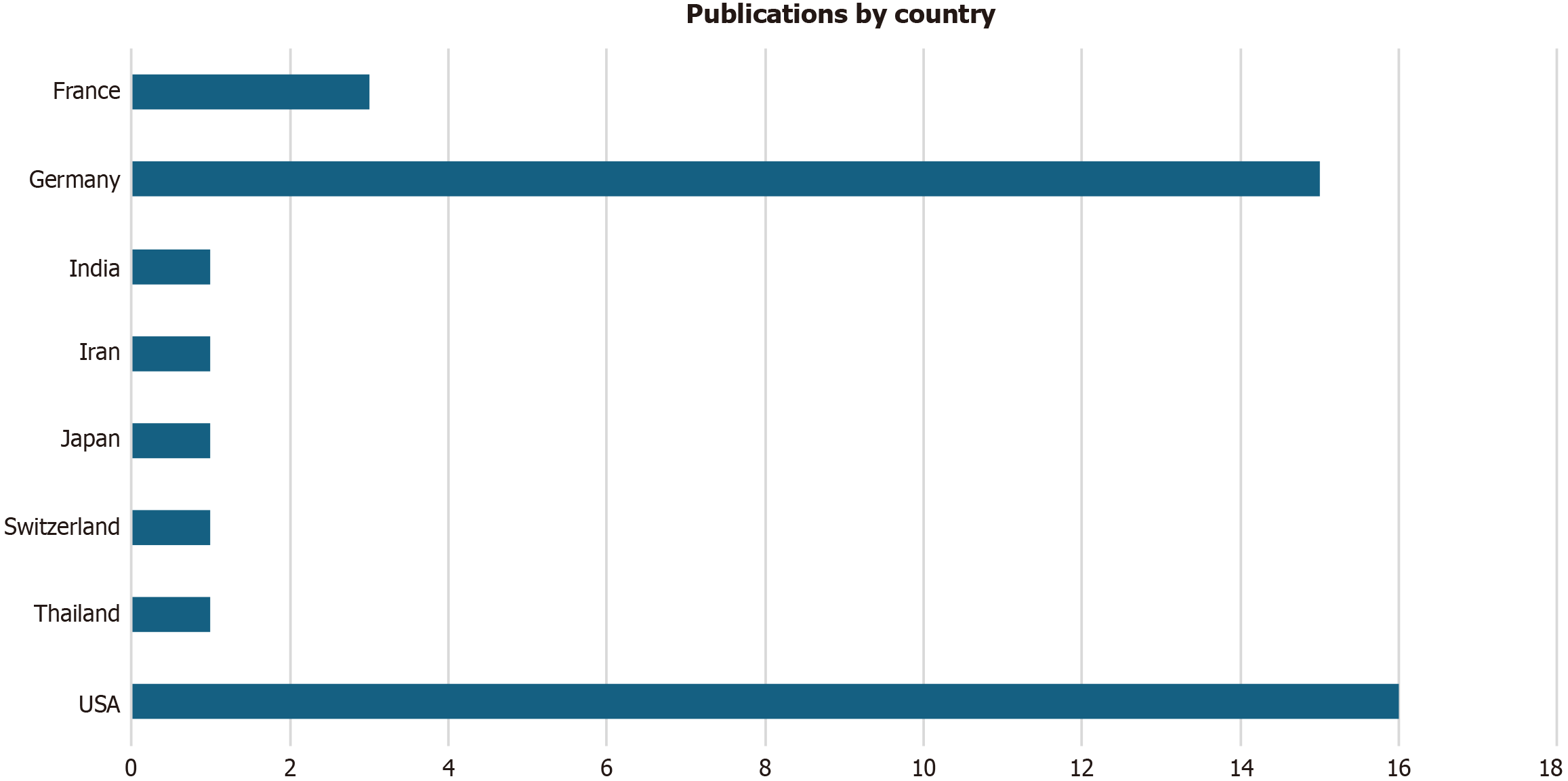
The present systematic review included 39 studies with key trends illustrated in the form of charts. Figure 2 highlights concentrated research efforts in high-resource settings with the United States contributing the most published studies
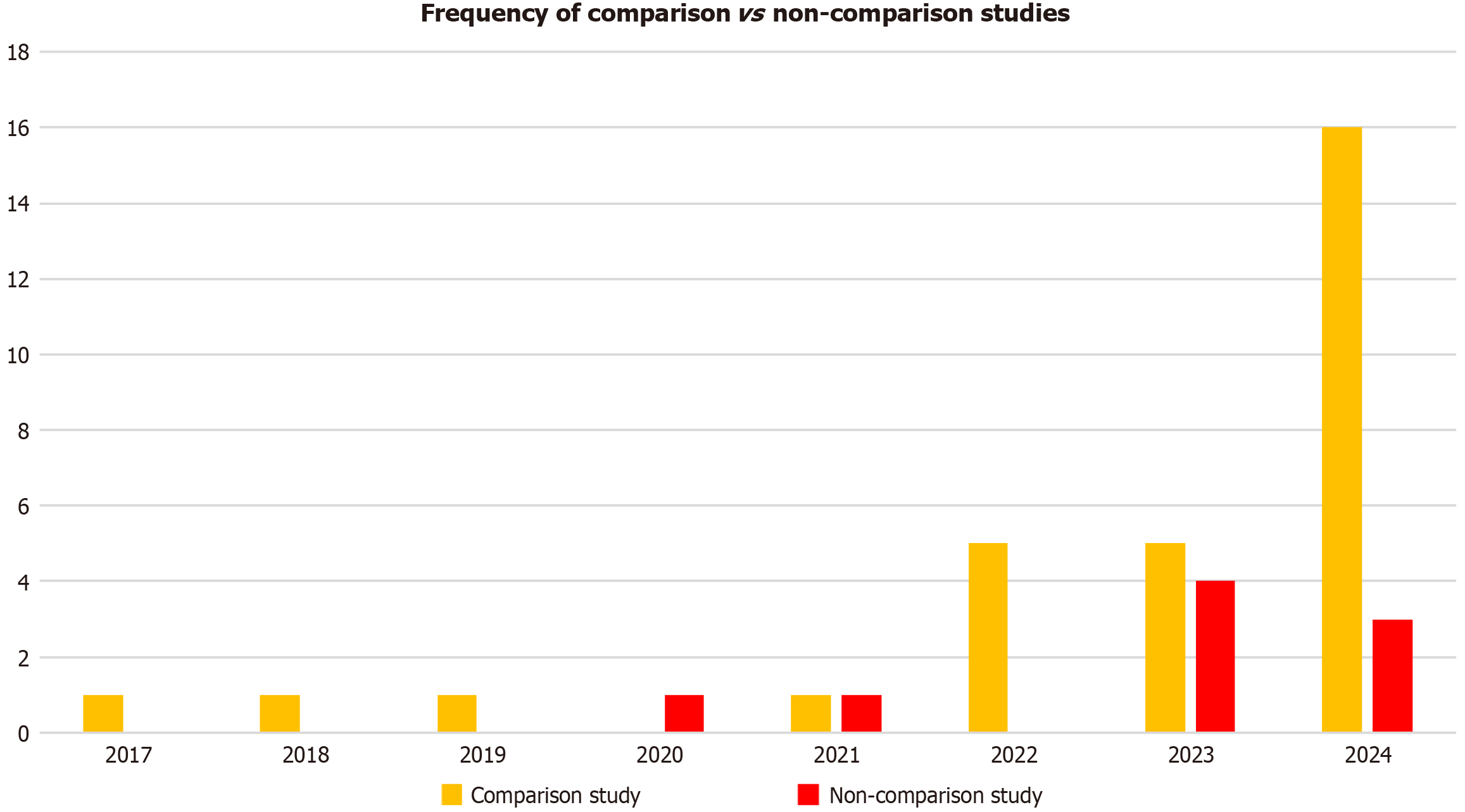
Figure 4 illustrates study design trends from 2017-2024, and our analyses demonstrated a marked increase in com
This systematic review highlighted the evolving role of PCD CT in oncologic imaging, showcasing its advantages over conventional CT in terms of spatial resolution, spectral imaging, and radiation dose reduction. The increasing number of experimental studies in recent years reflects the growing interest in integrating PCD CT into oncologic workflows with applications spanning tumor detection, characterization, and treatment monitoring.
In the following sections we discussed the key applications of PCD CT in oncologic imaging, examining how this technology enhances diagnostic accuracy, improves lesion differentiation, and influences clinical decision-making. Furthermore, we explored current limitations and future directions that may shape the widespread adoption of PCD CT in oncology.
PCD CT is comparable in performance with MRI in detecting and characterizing renal cell carcinoma and other renal masses, particularly in terms of inter-reader reliability and sensitivity. This capability is especially beneficial in the detection of benign renal cysts vs renal neoplasms in which the ability of PCD CT to differentiate based on iodine concentration (IC) has proven effective. Specifically, studies have demonstrated that renal neoplasms exhibit higher IC values compared with benign cysts with maximum and mean IC values offering excellent discriminatory power (area under the curve = 0.983 and 0.992, respectively) for heterogeneous lesions[6]. Moreover, the single-phase imaging capability of PCD CT reduces scan complexity and radiation exposure, an important consideration in clinical practice. However, challenges remain with virtual monochromatic imaging (VMI) at low energy levels (e.g., 40 keV), which may misclassify non-enhancing cysts as enhancing ones. Higher VMI levels (70 keV and above) appear optimal for minimizing such misclassification and ensuring accurate lesion characterization[7]. Furthermore, the use of VNC images derived from PCD CT has shown promise in reliable cyst detection, potentially replacing non-contrast imaging to reduce radiation dose[8].
The application of PCD CT in adrenal lesion characterization, particularly in differentiating adrenal adenomas from metastases, has been explored with promising results. VNC reconstructions, both conventional and new algorithms, show significant potential in distinguishing adrenal adenomas, which have lower CT values, from metastases, which typically present with higher CT values[9]. However, while VNC reconstruction offers a useful diagnostic tool, it tends to overestimate CT values compared with true non-contrast images, suggesting that adjusted threshold values may be necessary for optimal diagnostic accuracy. Studies have recommended thresholds of 26 Hounsfield unit (HU) instead of the traditional 10 HU for accurate lesion differentiation. Further research with larger cohorts is necessary to refine these thresholds and confirm the clinical utility of VNC reconstructions in routine practice[10].
In the context of ovarian cancer, PCD CT has emerged as a novel tool for non-invasive real-time monitoring and dia
PCD CT has demonstrated superior diagnostic performance in breast cancer staging compared with traditional mammography, particularly in terms of tumor conspicuity and classification of locoregional disease. Studies show that PCD CT outperforms traditional mammography, especially in dense breasts, providing higher sensitivity and specificity for detecting ductal carcinoma in situ and T classification of tumors[12]. Furthermore, the ability of PCD CT to detect metastatic lymph nodes and multifocal breast cancer suggests its potential as a complementary modality to breast MRI in certain clinical scenarios. In addition the higher diagnostic accuracy and inter-reader agreement observed with PCD CT could lead to more confident clinical decision-making and improved patient outcomes.
While traditional CT imaging is limited in prostate cancer management due to its reliance on size-based assessments, PCD CT offers a promising alternative by providing better tissue characterization through spectral imaging. This capability enables more accurate evaluation of prostate cancer and its metastases with the added benefit of reduced radiation exposure and improved radiotherapy treatment planning. However, despite these advantages technical challenges remain. Specifically, image quality can degrade in pelvic imaging due to motion artifacts from bowel peristalsis, beam hardening near the bladder and rectum, and high attenuation from surrounding bony structures. These issues can compromise lesion conspicuity and spectral accuracy, limiting the reliability of PCD CT in certain diagnostic and staging scenarios. As a result the clinical adoption of PCD CT in routine prostate cancer imaging remains cautious, pending further technical refinements and validation in large-scale studies[13].
In the case of pituitary tumors, particularly in the context of Cushing’s disease, dynamic contrast-enhanced PCD CT has shown potential in improving adenoma identification. The ability of PCD CT to provide high-resolution imaging of the pituitary gland, coupled with its ability to delineate the peripheral margins of adenomas, offers a distinct advantage in presurgical planning. This technique when combined with advanced image reconstruction protocols provides enhanced sensitivity in detecting hypoenhancing lesions, which are often difficult to visualize on conventional imaging modalities like MRI[14].
PCD CT offers significant improvements over conventional EID CT in detecting and staging pancreatic ductal adenocarcinoma. Low keV VMI derived from PCD CT enhance tumor conspicuity, especially in the portal venous phase, which aids earlier detection and diagnostic confidence[15]. Studies show that PCD CT improves tumor delineation and reduces radiation dose compared to EID CT with substantial inter-reader agreement for vascular involvement in pancreatic ductal adenocarcinoma, particularly in critical arteries and veins such as the celiac artery and superior mesenteric artery. These improvements in image quality and diagnostic accuracy are critical for surgical planning[16,17].
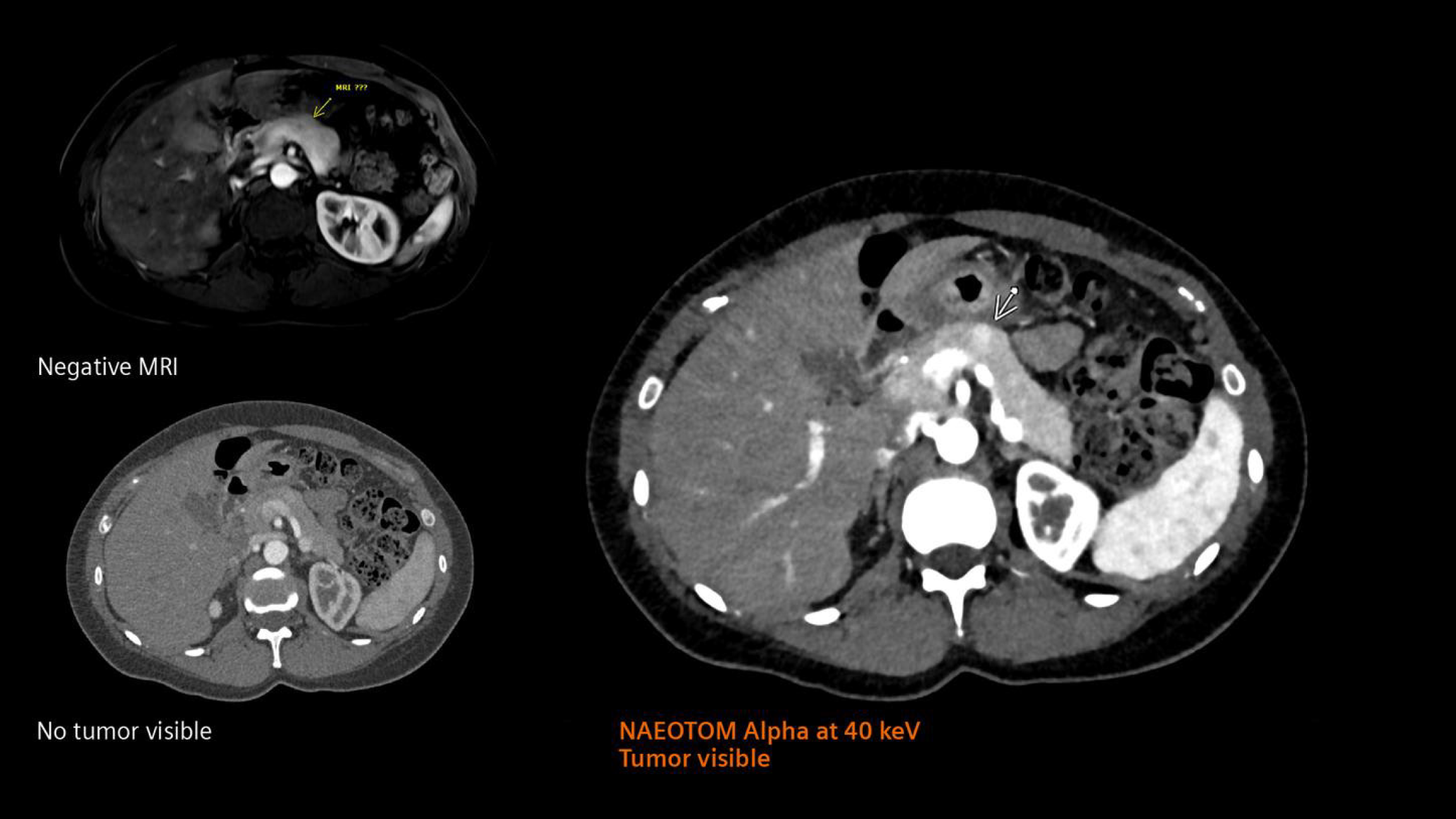
Additionally, PCD CT excels in detecting small pancreatic lesions, such as side-branch intraductal papillary mucinous neoplasms, which are challenging to detect on conventional CT. In a prevalence study PCD CT demonstrated higher cyst detection rates (4.9% vs 3.0%, P = 0.017) and detected previously overlooked cysts, further highlighting its potential in early tumor detection and surveillance[18]. An example of this high-precision visualization is shown in Figure 5[19], which demonstrates a superior depiction of a pancreatic insulinoma using PCD CT when compared with conventional CT and MRI.
PCD CT combined with QIR provides superior image quality for diagnosing hepatocellular carcinoma in patients with cirrhosis. Higher QIR levels (e.g., QIR-4) significantly reduce image noise, improving the contrast-to-noise ratio by up to 50%. Soft reconstruction kernels with a sharpness level of 36 offer the best balance between noise suppression and lesion visualization, making them ideal for spectral imaging applications in oncologic settings[20,21]. These advancements in PCD CT reconstruction parameters improve lesion conspicuity and may enhance diagnostic confidence in routine clinical practice (Figure 5).
PCD CT has demonstrated significant advantages in oncologic imaging, particularly in detecting bone metastases. It provides improved lesion characterization with reduced inter-reader variability, helping assess tumor size and structural integrity. PCD CT also offers substantial radiation dose reduction compared with dual-source (DS) CT while maintaining or improving image quality[22,23]. In peritoneal metastasis assessment spectral PCD CT has improved contrast resolution, especially with low-energy virtual monoenergetic images, aiding in better differentiation between malignant and benign lesions[23,24].
Several studies have compared the diagnostic impact of PCD CT vs EID CT in the context of multiple myeloma. Baffour et al[25] showed that whole-body low-dose PCD CT in ultra-high-resolution mode with and without a deep learning noise reduction algorithm was superior in viewing multiple myeloma lesions compared with EID CT[25].
In a prospective study involving 7 patients with monoclonal gammopathy, it was demonstrated that PCD CT offered significant benefits for multiple myeloma imaging compared with EID CT with both imaging modalities performed under standardized protocols. Specifically, PCD CT delivered an 83% reduction in radiation dose compared with EID CT, and PCD CT identified 79% of osteolytic lesions compared with 64% detected with EID CT; moreover, PCD CT de
Similarly, a study involving 9 patients with stage III myeloma who underwent imaging of the lower lumbar spine and pelvis showed that PCD CT identified 21% more osteolytic bone lesions than EID CT with improved sensitivity for smaller lesions[27].
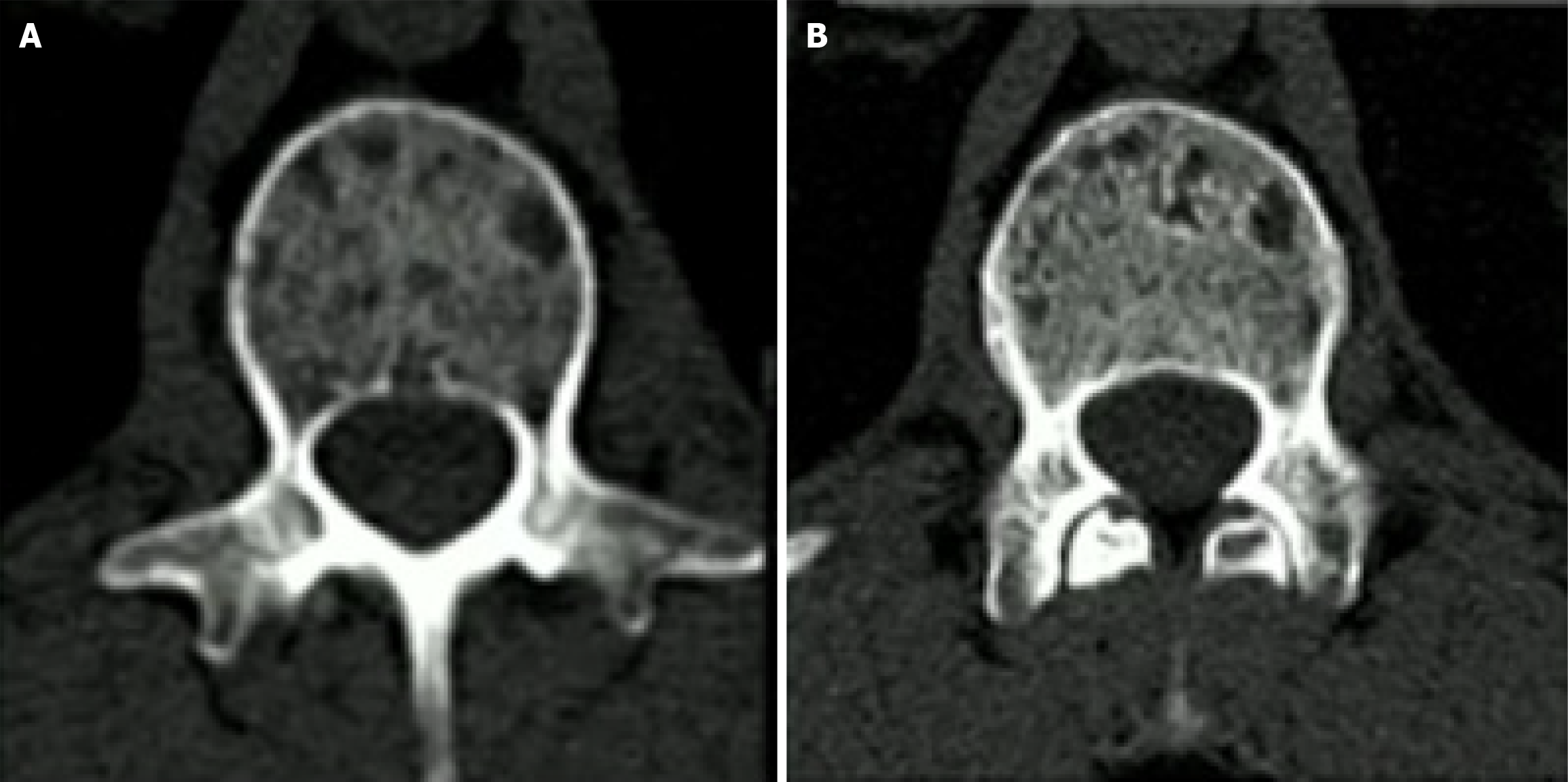
Compared with EIDs, PCDs significantly increase the spatial resolution of bone microstructure in patients with multiple myeloma. Subjective and objective image quality comparison of bone microstructure and disease-related abnormalities in multiple myeloma patients using a first-generation DS-PCD CT and a second-generation DS-EID CT showed that overall quality as well as detectability and sharpness in the delineation of lytic bone lesions were superior for DS-PCD CT compared with DS-EID CT (P < 0.0001; Figure 6)[19]. The quantitative metric for bone microstructure in the femoral head showed significantly better visualization of trabeculae in DS-PCD CT compared with DS-EID CT
Symons et al[28] evaluated the feasibility of using a whole-body PCD CT scanner for low dose lung cancer screening compared with a conventional EID CT system and showed that PCD CT provides better HU stability for lung, ground-glass, and emphysema equivalent forms at lower radiation dose settings with better reproducibility than EID CT. Additionally, PCD CT showed up to 10% less noise and 11% higher contrast-to-noise ratio at 0.75 mGy for both 100 and 80 kVp. Thus, PCD CT technology may help reduce radiation exposure in lung cancer screening while maintaining diagnostic quality[30,31].
Similarly, another study evaluated the potential of PCD CT for dose reduction in pulmonary nodule visualization for human readers as well as for computer-aided detection (CAD) studies. At the half radiation dose, sensitivity remained 100% for the human reader and CAD studies in PCD CT. At the quarter radiation dose, PCD CT achieved the same results as EID CT at the standard radiation dose setting (93%, P = 1.00) in human reading studies. Thus, CAD algorithms can be effective even at ultra-low doses[32].
This systematic review had several limitations. First, trends in the literature may have changed in 2025, potentially influencing the relevance and applicability of our findings. As the field of PCD CT continues to evolve, newer studies published beyond our search period may provide additional insights into its oncologic applications. Second, our study was limited to PubMed as the sole database for literature retrieval. While this decision was made to ensure that included studies met the rigorous indexing criteria of PubMed, particularly given the clinical nature of our review, it may have led to the omission of relevant studies from other databases such as Web of Science, Google Scholar, EMBASE, and Scopus. As a result some valuable contributions from non-PubMed-indexed journals may not have been considered in our analysis. Future systematic reviews incorporating multiple databases may provide a more comprehensive overview of the expanding role of PCD CT in oncologic imaging.
Despite all the benefits arising from the ultra-high resolution, some limitations need to be addressed mainly linked to the large amount of data extracted, amounting to approximately 16-fold of that of standard CT and giving rise to problems of storage and extreme data volumes to be processed[32]. PCD CT is poised to significantly enhance oncologic imaging by providing unprecedented image quality, reducing radiation doses, and improving diagnostic accuracy. As the technology continues to evolve and become more widely adopted, it is likely to play a central role in pushing the borders of cancer imaging, ultimately leading to better patient outcomes.
| 1. | Hsieh SS, Leng S, Rajendran K, Tao S, McCollough CH. Photon Counting CT: Clinical Applications and Future Developments. IEEE Trans Radiat Plasma Med Sci. 2021;5:441-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 92] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 2. | United States Food and Drug Administration. FDA Clears First Major Imaging Device Advancement for Computed Tomography in Nearly a Decade. [cited 3 July 2025]. Available from: https://www.fda.gov/news-events/press-announcements/fda-clears-first-major-imaging-device-advancement-computed-tomography-nearly-decade. |
| 3. | Perera Molligoda Arachchige AS, Catapano F, Lisi C, El Choueiri J, Pellicanò F, Figliozzi S, Politi LS, Francone M. Trends in Clinical Cardiac Photon-Counting Detector CT Research: A Comprehensive Bibliometric Analysis. Diagnostics (Basel). 2025;15:504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 4. | Perera Molligoda Arachchige AS, Verma Y. Role of photon-counting computed tomography in pediatric cardiovascular imaging. World J Clin Pediatr. 2025;14:99288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 5. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44932] [Cited by in RCA: 40536] [Article Influence: 10134.0] [Reference Citation Analysis (2)] |
| 6. | Tóth A, Chamberlin JH, Mendez S, Varga-Szemes A, Hardie AD. Iodine quantification of renal lesions: Preliminary results using spectral-based material extraction on photon-counting CT. J Clin Imaging Sci. 2024;14:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 7. | Schade KA, Mergen V, Sartoretti T, Alkadhi H, Euler A. Pseudoenhancement in Cystic Renal Lesions - Impact of Virtual Monoenergetic Images of Photon-Counting Detector CT on Lesion Classification. Acad Radiol. 2023;30 Suppl 1:S305-S313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 8. | Rau S, Rau A, Stein T, Hagar MT, Faby S, Bamberg F, Weiss J. Value of virtual non-contrast images to identify uncomplicated cystic renal lesions: photon-counting detector CT vs. dual-energy integrating detector CT. Radiol Med. 2024;129:669-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 9. | Bette S, Risch F, Canalini L, Becker J, Leithner EV, Huber A, Haerting M, Jehs B, Wollny C, Schwarz F, Tehlan K, Scheurig-Muenkler C, Wendler T, Kroencke T, Decker JA. Diagnostic performance of photon-counting detector CT for differentiation between adrenal adenomas and metastases. Eur Radiol. 2024;34:5944-5953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 10. | Lennartz S, Schoenbeck D, Kröger JR, Borggrefe J, Henning Niehoff J. Photon-counting CT Material Decomposition: Initial Experience in Assessing Adrenal Adenoma. Radiology. 2023;306:202-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 11. | Kumar D, Moghiseh M, Chitcholtan K, Mutreja I, Lowe C, Kaushik A, Butler A, Sykes P, Anderson N, Raja A. LHRH conjugated gold nanoparticles assisted efficient ovarian cancer targeting evaluated via spectral photon-counting CT imaging: a proof-of-concept research. J Mater Chem B. 2023;11:1916-1928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 12. | Neubauer J, Wilpert C, Gebler O, Taran FA, Pichotka M, Stein T, Molina-Fuentes MF, Weiss J, Juhasz-Böss I, Bamberg F, Windfuhr-Blum M, Neubauer C. Diagnostic Accuracy of Contrast-Enhanced Thoracic Photon-Counting Computed Tomography for Opportunistic Locoregional Staging of Breast Cancer Compared With Digital Mammography: A Prospective Trial. Invest Radiol. 2024;59:489-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 13. | O'Connell J, Schwartz F, Morris T, and Dunning C, Harris TC, Ferguson D, Myronakis M, Sodickson A, Berbeco RI. Initial Examination of Commercial Photon Counting CT for Prostate Planning. Int J Radiat Oncol Biol Phys. 2024;120:e172. [DOI] [Full Text] |
| 14. | Mark IT, Van Gompel J, Bancos I, Nagelschneider AA, Johnson DR, Bathla G, Madhavan AA, Weber NM, Yu L. Back to the Future: Dynamic Contrast-Enhanced Photon-Counting Detector CT for the Detection of Pituitary Adenoma in Cushing Disease. AJNR Am J Neuroradiol. 2024;45:743-746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 15. | Woeltjen MM, Niehoff JH, Roggel R, Michael AE, Gerdes B, Surov A, Borggrefe J, Kroeger JR. Pancreatic cancer in photon-counting CT: Low keV virtual monoenergetic images improve tumor conspicuity. Eur J Radiol. 2024;173:111374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 16. | Decker JA, Becker J, Härting M, Jehs B, Risch F, Canalini L, Wollny C, Scheurig-Muenkler C, Kroencke T, Schwarz F, Bette S. Optimal conspicuity of pancreatic ductal adenocarcinoma in virtual monochromatic imaging reconstructions on a photon-counting detector CT: comparison to conventional MDCT. Abdom Radiol (NY). 2024;49:103-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 17. | Kim J, Mabud T, Huang C, Lloret Del Hoyo J, Petrocelli R, Vij A, Dane B. Inter-reader agreement of pancreatic adenocarcinoma resectability assessment with photon counting versus energy integrating detector CT. Abdom Radiol (NY). 2024;49:3149-3157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 18. | Khanungwanitkul K, Schwartz FR. Photon-Counting CT Shows Side-Branch Intraductal Papillary Mucinous Neoplasm-Pancreatic Duct Connection. Radiology. 2024;312:e240611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 19. | Siemens Healthineers. The NAEOTOM Alpha class. [cited 7 July 2025]. Available from: https://www.siemens-healthineers.com/computed-tomography/naeotom. |
| 20. | Graafen D, Stoehr F, Halfmann MC, Emrich T, Foerster F, Yang Y, Düber C, Müller L, Kloeckner R. Quantum iterative reconstruction on a photon-counting detector CT improves the quality of hepatocellular carcinoma imaging. Cancer Imaging. 2023;23:69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 21. | Graafen D, Müller L, Halfmann MC, Stoehr F, Foerster F, Düber C, Yang Y, Emrich T, Kloeckner R. Soft Reconstruction Kernels Improve HCC Imaging on a Photon-Counting Detector CT. Acad Radiol. 2023;30 Suppl 1:S143-S154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 22. | Wehrse E, Sawall S, Klein L, Glemser P, Delorme S, Schlemmer HP, Kachelrieß M, Uhrig M, Ziener CH, Rotkopf LT. Potential of ultra-high-resolution photon-counting CT of bone metastases: initial experiences in breast cancer patients. NPJ Breast Cancer. 2021;7:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 23. | Hagen F, Walder L, Fritz J, Gutjahr R, Schmidt B, Faby S, Bamberg F, Schoenberg S, Nikolaou K, Horger M. Image Quality and Radiation Dose of Contrast-Enhanced Chest-CT Acquired on a Clinical Photon-Counting Detector CT vs. Second-Generation Dual-Source CT in an Oncologic Cohort: Preliminary Results. Tomography. 2022;8:1466-1476. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 24. | Grange R, Si-Mohamed S, Kepenekian V, Boccalini S, Glehen O, Rousset P. Spectral photon-counting CT: Hype or hope for colorectal peritoneal metastases imaging? Diagn Interv Imaging. 2024;105:118-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 25. | Baffour FI, Huber NR, Ferrero A, Rajendran K, Glazebrook KN, Larson NB, Kumar S, Cook JM, Leng S, Shanblatt ER, McCollough CH, Fletcher JG. Photon-counting Detector CT with Deep Learning Noise Reduction to Detect Multiple Myeloma. Radiology. 2023;306:229-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 43] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 26. | Schwartz FR, Vinson EN, Spritzer CE, Colglazier R, Samei E, French RJ, Said N, Waldman L, McCrum E. Prospective Multireader Evaluation of Photon-counting CT for Multiple Myeloma Screening. Radiol Imaging Cancer. 2022;4:e220073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 27. | Grözinger M, Wennmann M, Sawall S, Wehrse E, Sedaghat S, Neelsen C, Bauer F, Goldschmidt H, Weru V, Ziener CH, Kopp-Schneider A, Schlemmer HP, Rotkopf LT. Detection of myeloma-associated osteolytic bone lesions with energy-integrating and photon-counting detector CT. Radiologie (Heidelb). 2024;64:24-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 28. | Winkelmann MT, Hagen F, Le-Yannou L, Weiss J, Riffel P, Gutjahr R, Faby S, Nikolaou K, Horger M. Myeloma bone disease imaging on a 1st-generation clinical photon-counting detector CT vs. 2nd-generation dual-source dual-energy CT. Eur Radiol. 2023;33:2415-2425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 29. | Symons R, Cork TE, Sahbaee P, Fuld MK, Kappler S, Folio LR, Bluemke DA, Pourmorteza A. Low-dose lung cancer screening with photon-counting CT: a feasibility study. Phys Med Biol. 2017;62:202-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 83] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 30. | Jungblut L, Euler A, Landsmann A, Englmaier V, Mergen V, Sefirovic M, Frauenfelder T. Pulmonary nodule visualization and evaluation of AI-based detection at various ultra-low-dose levels using photon-counting detector CT. Acta Radiol. 2024;65:1238-1245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 31. | Perera Molligoda Arachchige AS. MRI versus CT in Detecting Pulmonary Nodules. Radiology. 2022;304:E51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 32. | Flohr T, Petersilka M, Henning A, Ulzheimer S, Ferda J, Schmidt B. Photon-counting CT review. Phys Med. 2020;79:126-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 302] [Article Influence: 60.4] [Reference Citation Analysis (0)] |