Published online Jun 28, 2021. doi: 10.4329/wjr.v13.i6.171
Peer-review started: February 7, 2021
First decision: May 6, 2021
Revised: May 15, 2021
Accepted: June 23, 2021
Article in press: June 23, 2021
Published online: June 28, 2021
Processing time: 138 Days and 5.3 Hours
The role of radiology and the radiologist have evolved throughout the coronavirus disease-2019 (COVID-19) pandemic. Early on, chest computed tomography was used for screening and diagnosis of COVID-19; however, it is now indicated for high-risk patients, those with severe disease, or in areas where polymerase chain reaction testing is sparsely available. Chest radiography is now utilized mainly for monitoring disease progression in hospitalized patients showing signs of worsening clinical status. Additionally, many challenges at the operational level have been overcome within the field of radiology throughout the COVID-19 pandemic. The use of teleradiology and virtual care clinics greatly enhanced our ability to socially distance and both are likely to remain important mediums for diagnostic imaging delivery and patient care. Opportunities to better utilize of imaging for detection of extrapulmonary manifestations and complications of COVID-19 disease will continue to arise as a more detailed under
Core Tip: The role of both radiology and the radiologist has evolved throughout the coronavirus disease-2019 (COVID-19) pandemic. Detecting extrapulmonary manifestations as well as complications of COVID-19 disease with imaging remain important areas for further research. The development of COVID-19 severity scoring systems and standardized reporting methods has begun to lay the foundations for artificial intelligence systems. Furthermore, teleradiology and virtual care clinics were important components of the response to the COVID-19 pandemic and will remain important mediums for diagnostic imaging delivery and patient care. Finally, the emergence of point of care ultrasound is an exciting yet underexplored area of imaging applications for COVID-19.
- Citation: Pezzutti DL, Wadhwa V, Makary MS. COVID-19 imaging: Diagnostic approaches, challenges, and evolving advances. World J Radiol 2021; 13(6): 171-191
- URL: https://www.wjgnet.com/1949-8470/full/v13/i6/171.htm
- DOI: https://dx.doi.org/10.4329/wjr.v13.i6.171
In December 2019, the first reports of respiratory-related infections with a novel coronavirus emerged[1]. This virus, now known as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and the disease it causes, coronavirus disease-2019 (COVID-19), then quickly spread across the globe and the World Health Organization officially announced a global pandemic on March 11, 2020[2]. At the time of this writing, there have been more than 100000000 confirmed cases and 2200000 deaths worldwide from COVID-19. It is now the third leading cause of death within the United States, behind cancer and cardiovascular disease, accounting for nearly 27000000 cases in the United States and more than 440000 deaths in the United States[3]. In individuals over the age of 35, it is the leading cause of death in the United States[4].
From the pandemic’s inception, radiological imaging has played a critical role in the diagnosis and management of COVID-19. However, the role of imaging has evolved throughout the pandemic. The purpose of this review is to address the evolving role of imaging in the diagnosis and management of COVID-19. In addition, we aim to discuss the current state of COVID-19 severity scoring systems in imaging, the operational challenges and preparedness response to COVID-19 in regard to the field of radiology, as well as the opportunities and future directions of radiological imaging in COVID-19.
At the onset of the pandemic, the use of imaging, specifically chest radiography (CXR) and chest computed tomography (CT), served primarily as diagnostic and screening tools for COVID-19[5]. This is because the development of real time-polymerase chain reaction (RT-PCR) assays was still in process and the availability of these tests was not yet widespread[6,7]. Chest CT was shown to be more sensitive than CXR (95% vs 69%), but the use of either was not solely dependent on their sensitivities, but also the availability of each technology and the risk of subsequent exposure to other staff during each study[5]. Furthermore, early reports suggested a high sensitivity for chest CT for patients with COVID-19, and thus argued for its use as a screening tool[8]. In contrast, a later study conducted by Bernheim et al[9], revealed that in a study of 121 patients that examined CT findings in patients within two days of symptom onset, all but one patient tested positive with RT-PCR but had negative chest CT findings.
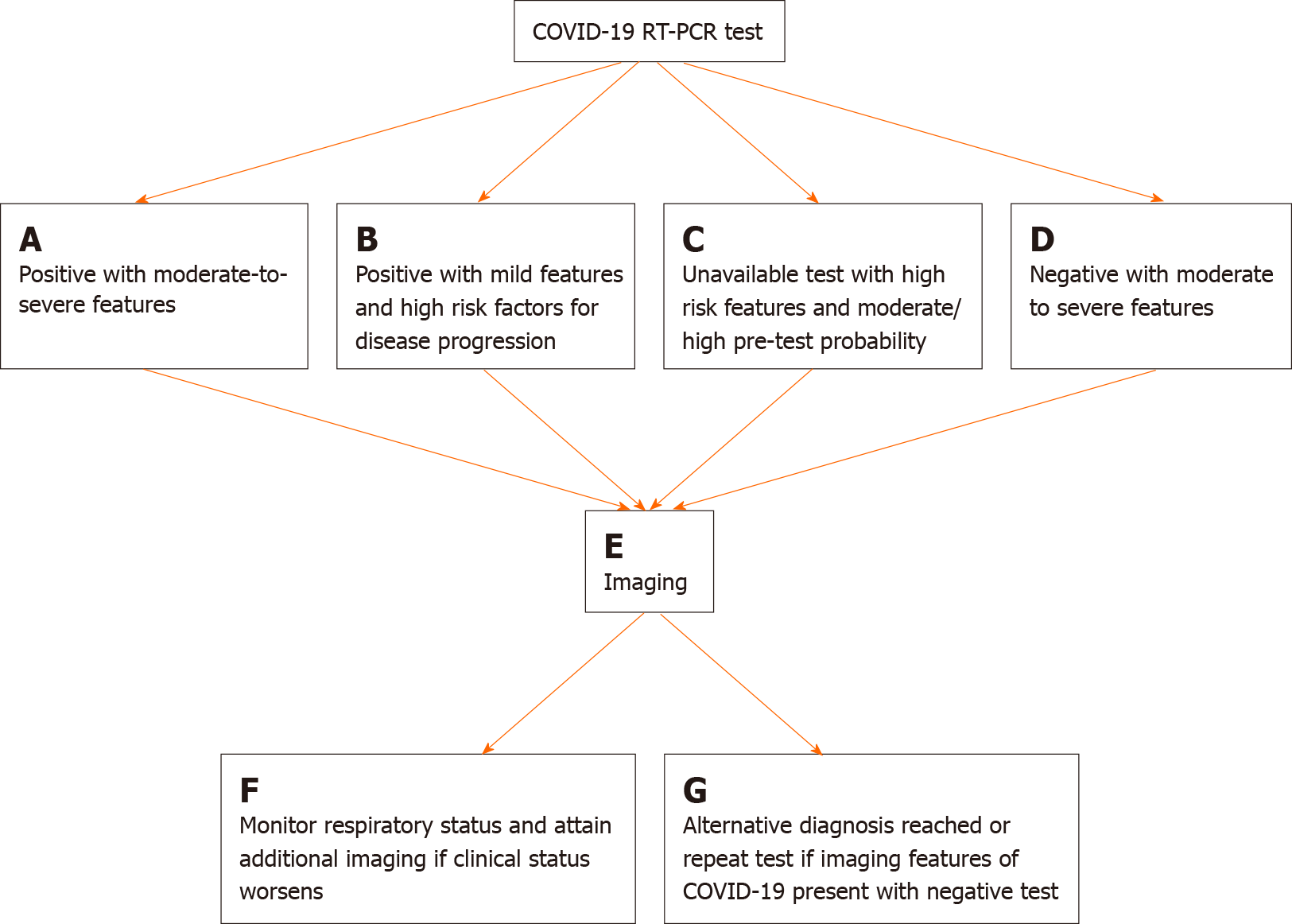
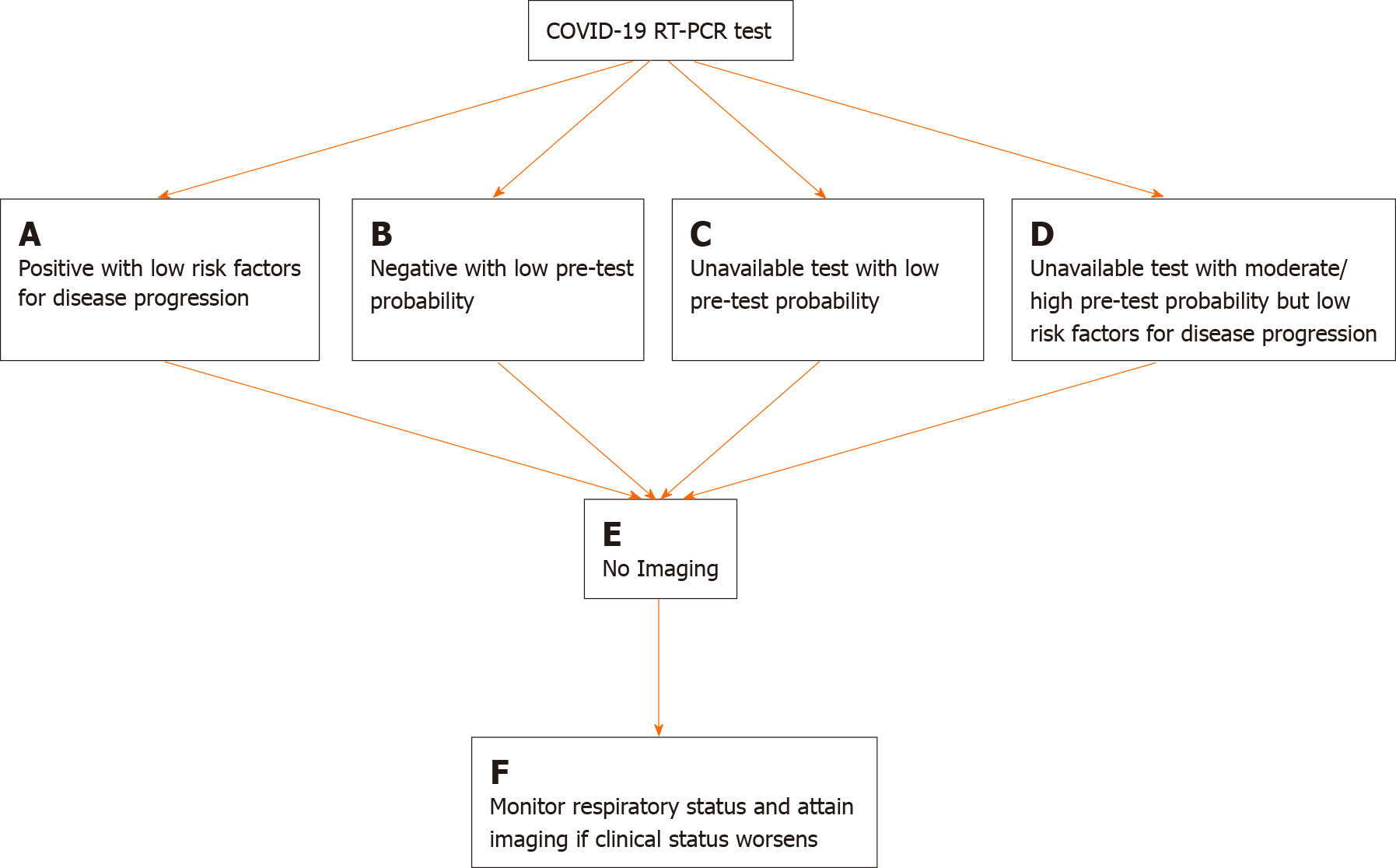
As testing kits became more widely available across communities, the role of imaging as a primary diagnostic and screening tool for COVID-19 became secondary to RT-PCR[7]. Although still a topic of debate, the Fleischner Society released a consensus statement indicating various scenarios where imaging in COVID-19 patients may be utilized and the radiologist be called upon, with each scenario being dependent upon the severity of disease, the pre-test probability, and the availability of resources [i.e., personal protective equipment (PPE), testing kits, staff, etc.].
These scenarios where imaging is indicated in patients with confirmed or suspected COVID-19 are specific for CXR and chest CT and include the following: (1) patients with a positive RT-PCR test or high pre-test probability in the absence of a RT-PCR test with evidence of risk factors for disease progression (i.e., > 65 years of age, immunocompromised, comorbidities such as diabetes mellitus, hypertension, chronic lung disease, cardiovascular disease); (2) patients with moderate to severe features of COVID-19, regardless of RT-PCR test results; and (3) patients with moderate-to-severe symptoms within a high prevalence of disease environment and with limited testing resources, regardless of a RT-PCR result[7]. A modified algorithm based on the Fleischner Society’s recommendations for imaging in the workup and management of COVID-19 disease is detailed in Figures 1 and 2.
Radiologists and medical imaging in general have been important not only in the screening and diagnosis of patients with COVID-19, but also in monitoring disease progression, predicting prognosis, monitoring treatment response, and determining disease severity[10]. In addition to CXR and chest CT, the use of imaging modalities such as ultrasound, magnetic resonance imaging (MRI), and 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography (PET) have been reported in the management of COVID-19 patients. Hereafter, the various imaging modalities used for COVID-19 patients, along with their indications, advantages and disadvantages, and the various features of disease present on each is described.
CXR is widely known to be a cost-effective and easily accessible imaging modality and is thus often used initially in the assessment of a patient with suspected COVID-19. The overall sensitivity of CXR is 69%-74%, with a lower sensitivity at earlier points in the course of the disease[1]. It is also easily portable, which makes it useful in patients that are immobile or bedridden. CXR can also be used to easily monitor disease progression and may be employed for those patients who are showing signs of worsening clinical status in the hospital[11]. The utility of CXR is also in evaluating for alternative diagnoses which may present with symptoms similar to COVID-19.
Pulmonary imaging findings on CXR are similar to those found on CT, and are often bilateral, posterior and peripheral, with a predominance in the lower lung fields. The most commonly reported interstitial abnormalities are reticular and reticulonodular patterns and the most commonly reported alveolar findings include hazy pulmonary opacities similar to the ground-glass opacities (GGOs) identified on CT. These can be accompanied with or without consolidation[12,13]. Progression of disease can be identified by the pulmonary opacities becoming more diffuse and thickening of the interstitial markings. The most severe signs of disease are present 10-12 d after symptom onset[14].
Chest CT for COVID-19 has been shown to have a sensitivity of 94% and a specificity of 37%, with a positive predictive value of 1.5%-30.7% and a negative predictive value of 95.4%–99.8%[15]. Thus, in areas of low prevalence with COVID-19, using CT will equate to an increased number of false positives[1]. However, because of this high sensitivity, its use may be warranted in the setting of a high prevalence of disease and a negative or unavailable PCR test[16].
The high sensitivity of chest CT also allows for radiologists to detect COVID-19 disease on those patients receiving CT for other indications. This allows for early detection and containment in an otherwise asymptomatic patient, which are thought to be 18%-33% of those infected with SARS-CoV-2[17-19]. Furthermore, CT can be used in the evaluation of certain complications from COVID-19 that may not be discernible on CXR, such as pulmonary thromboembolism, lung abscesses, acute respiratory distress syndrome (ARDS), myocarditis, and acute lung edema[20,21]. Despite the augmented sensitivity, higher resolution and improved clarity in identifying both pulmonary and extrapulmonary manifestations with CT, it is important to note that the use of CT leads to involvement of more staff within the hospital and the use of more personal protective equipment, leading to increased cost and heightened risk of spread to hospital employees[22].
The two most commonly identified pulmonary findings on chest CT are GGOs and reticular opacities, typically with bilateral involvement and a multifocal pattern in a peripheral, sub-pleural, and posterior distribution[9,15,23-26]. Consolidations are also frequently observed, and can be present alone or alongside GGOs, in which case they are known as “mixed lesions”. Finally, GGOs with superimposed intralobular lines and interlobular septal thickening–known as the crazy paving pattern–is a common finding on chest CT[27]. Other notable findings of disease include adjacent pleural thickening, intralobular septal thickening, air bronchograms, reverse halo sign, and a variant of the reverse halo sign known as the bullseye sign[28-30]. Findings such as pleural effusions and lymphadenopathy are normally absent[24].
The CT findings of patients with COVID pneumonia are dynamic and progress through a series of four stages marked from the time of symptom onset[31]. The early phase (0-4 d) is characterized mainly by the emergence of GGOs. The progressive phase (5-8 d) is characterized by an increase in the size and number of GGOs with the gradual transformation of GGOs into multifocal, consolidative areas and the development of a crazy-paving pattern. The peak stage (9-13 d) is characterized by more extensive lung involvement and the presence of more dense consolidations. Following the peak stage, an absorption stage can be identified where consolidations are slowly reabsorbed and fibrotic bands, a sign of repaired lungs, begin to appear[25,31]. Evidence of lung abnormalities persist long beyond symptom resolution, with one study reporting 94% of patients having residual CT findings 25 d following symptom onset[32]. Over the course of recovery, it is common to observe traction bronchiectasis, as well as peribronchovascular thickening[33].
Ultrasound is well known to have advantages in the realm of medical imaging in that it does not expose patients to ionizing radiation and is easily accessible in terms of its portability and the ability to perform bedside examinations[34]. In the age of a pandemic, being able to perform ultrasound at the bedside offers advantages in that one does not need to transfer the patient to another part of the hospital and risk spreading the virus to other members in the hospital, especially other patients. This also frees up those staff involved in the transfer of the patient to attend to their other responsibilities. Furthermore, ultrasound is well known to be extremely affordable, results are available instantly, and it can be performed on patients who otherwise should not be exposed to radiation (i.e., pregnant patients)[22,34-36].
Common findings identified on lung ultrasound in patients with COVID-19 include non-specific findings such as the presence of B-line artifacts, an irregularly thickened pleura, and sub-pleural consolidations. B-line artifacts are vertically oriented hyper-echoic artifacts that originate from the pleura or from areas of consolidation. These lines indicate accumulation of fluid in the pulmonary interstitial space or alveoli[12,37]. A-lines can then be seen in the recovery phase of the disease[38].
Despite showing promise as a useful imaging modality in COVID-19 patients, the main evidence for the use of ultrasound come from small case series, tutorials, and opinion articles, and there are no large-scale studies examining its utility. The current guidelines from the major radiological societies for lung imaging in COVID-19 state no official role for the use of ultrasound and it is mainly an investigational tool at this time[39]. The use of point of care ultrasound (POCUS) will be further discussed in a later section.
In imaging of COVID-19 patients, 18F-FDG PET has a high sensitivity but a poor specificity and has mainly been reported as an imaging modality that incidentally detects evidence of COVID-19 disease[23,40]. For example, a report of a patient who underwent 18F-FDG PET/CT for suspected recurrence of non-small cell lung cancer, was noted to have incidental foci consistent with GGO in the lower lobes of the lungs bilaterally, determined to likely be related to an acute-inflammatory process. The patient then tested positive for SARS-CoV-2, and clinically deteriorated and required intensive-care unit (ICU) level care. This example illustrates the importance of radiologists maintaining a high-level of suspicion for incidental findings noted on PET imaging, as early detection of the virus can lead to improved clinical outcomes, especially in those highly susceptible to severe complications[41].
In addition to aiding in early detection, 18F-FDG PET has also shown promise in predicting the severity of a lesion and the length of time it will take to heal by correlating 18F-FDG uptake with erythrocyte sedimentation rates[42]. Furthermore, the use of PET imaging may be beneficial in further understanding the neurological complications initiated by infection with SARS-CoV-2. For example, it may aid in identifying which brain regions are affected, which cells in the brain are involved, and also could aid in selecting patients at risk of developing neurological complications[43]. Nevertheless, the cost, exposure to excess radiation, prolonged acquisition time, and involvement of multiple lines of medical staff and consumption of an unnecessary amount of PPE argue against the use of PET imaging for diagnostic purposes[44]. Further research is warranted to examine the utility of PET imaging in assessing functionality of lesions and predicting disease severity.
MRI of the chest does not provide additional findings in patients with COVID-19 when compared with CT in terms of pulmonary manifestations. It is less widely available, more expensive, and has an increased imaging acquisition time[23,45,46]. The primary indications for the use of MRI in patients with COVID-19 are in the evaluation of complications involving the neurological and cardiac systems, such as acute necrotizing encephalopathy or myocarditis[1]. Furthermore, for patient groups in whom exposure to ionizing radiation should be avoided (i.e., young children, pregnant patients), MRI may be a viable option[47].
Being able to quantitatively capture the severity of disease in COVID-19 patients via imaging provides clinicians with a method of identifying and managing patients with severe disease in situations where rapid triage is a necessity. The application of pre-COVID-19 severity scores has been utilized and novel scoring systems have also been developed specifically for patients with COVID-19[48]. The details of various scoring systems developed in both CXR and Chest CT are outlined below. Although it is unclear as to which scoring method is the most optimal, it is imperative that radiological departments around the world adopt a system and consistently use it, while also ensuring their results can be consistently reproduced[48].
As previously stated, CXR has a low sensitivity early in the COVID-19 disease course, but in the emergency setting and for patients in the ICU, it can be used to monitor rapid progression of lung involvement in later stages of the disease[48]. The severe acute respiratory infection chest radiography severity scoring system was developed in 2015 with the original intent for it to be used by the non-radiologist clinicians to examine patients with acute respiratory processes[49]. Yoon et al[50] reported the use of this scoring system in the assessment of pulmonary involvement in COVID-19 patients[48]. The Radiographic Assessment of Lung Edema classification system was developed in March of 2020 by Wong et al [15], but it was adapted based on a similar score created by Warren et al in 2018. The only score to date created solely for patients with COVID-19 were proposed by Borghesi in March of 2020[51]. The median score of patients from the original study was 6.5 and the CXR score in patients who died was significantly higher than those discharged from the hospital[51]. A summary of the various features of each chest radiographic scoring system is detailed in Table 1.
| Chest Radiography Scoring Systems for COVID-19 | |||
| Characteristics of Chest Radiography Scoring Systems | Severe Acute Respiratory Infection[48] | Radiographic Assessment of Lung Edema[49] | Chest X-ray Score[51] |
| Division of Lungs | None | 2 lungs | 6 zones (3 zones each lung) |
| Methodology of Score Calculation | Entire lungs scored as one | Each lung scored and totaled | Each zone scored and totaled |
| Characteristics Scored | Various radiographic findings | 1GGOs or consolidation | Various radiographic findings |
| Scoring Scale | 1 = normal; 2 = patchy/hyperinflation/bronchial wall thickening; 3 = focal consolidation; 4 = multifocal consolidation; 5 = diffuse alveolar change | 1 ≤ 25%; 2 = 25%-50%; 3 = 50%-75%; 4 ≥ 75% | 0 = no abnormalities; 1 = interstitial infiltrates; 2 = interstitial (predominant) & alveolar infiltrates; 3 = interstitial & alveolar (predominant) infiltrates |
| Designed Specifically for COVID-19 Disease | No | No | Yes |
The use of CT imaging is highly effective at detecting COVID-19 early on in the disease course, with a sensitivity of up to 95%[15]. Thus, early on in the pandemic, clinicians pursued a severity scoring system for patients with COVID-19 based on CT findings[48]. The chest computed tomography severity score was developed by Yang et al[52] in March of 2020 as an adaptation from a previously used method during the SARS epidemic of 2005, and was used to rapidly identify those patients in need of hospital admission on initial presentation. The total severity score (TSS), also developed in March of 2020 by Li et al[53], was designed to examine the relationship between imaging findings and clinical presentation in patients with COVID-19. The chest computed tomography score was published by Li et al[54] in March of 2020 and is similar to the TSS. A summary of the various features of each chest CT scoring system is detailed in Table 2.
| Chest CT Scoring Systems for COVID-19 | |||
| Characteristics of Chest CT Scoring Systems | Chest Computed Tomography Severity Score[52] | Total Severity Score [53] | Chest Computed Tomography Score [54] |
| Division of Lungs | 20 regions for each lung | 5 lobes | 5 lobes |
| Methodology of Score Calculation | Each region scored & amount totaled | Each lobe scored and amount totaled | Each lobe scored and amount totaled |
| Characteristics Scored | Amount of opacification | % of disease in each lobe (1GGOs, mixed GGOs, consolidation) | % of disease in each lobe (no specific features) |
| Scoring Scale | 0 = 0%; 1 = 1%-50%; 2 = 51%-100% | 0 = 0%; 1 = 1%-25%; 2 = 26%-50%; 3 = 51%-75%; 4 = 76%-100% | 0 = 0%; 1 ≤ 5%; 2 = 5%-25%; 3 = 26%-49%; 4 = 50%-75%; 5 ≥ 75% |
| Sensitivity & Specificity | 83% & 94% | 83% & 100% | 80% & 82% |
| Lowest Score for Severe COVID-19 Cases | 19.5 | 7.5 | 7 |
| Designed Specifically for COVID-19 Disease | Yes | Yes | Yes |
One of the main challenges in COVID-19 imaging lies in its differential diagnosis on chest CT. This is due to the fact many of the findings on CT can be observed in other categories of disease, which include the following: inspiration/motion artifacts, trauma, other alveolar infectious etiologies (both viral and non-viral), as well as various interstitial and vascular pathologies[55]. While it is not possible to arrive at a diagnosis for COVID-19 based on imaging features alone, it is crucial to understand its features and their overlap with other infectious and non-infectious causes. Furthermore, it is also critical to always consider the epidemiological history of the patient and their symptoms in addition the objective laboratory and imaging findings prior to making a diagnosis[33]. Nonetheless, the various infectious/non-infectious mimickers of COVID-19 pneumonia follow hereafter.
GGOs may be one of the most common manifestations on CT of COVID-19 pneumonia; however, shallow inspiration, motion artifact from cardiac pulsation, and fibrotic bands/sub segmental atelectasis may present as a GGO-like appearance[56]. Traumatic lung findings, such as pulmonary contusion, can lead to the appearance of nodular opacities or large areas of consolidation based on the degree of trauma. Although these findings are present in COVID-19 pneumonia, appropriate clinical assessment of the patient will help to differentiate a trauma related finding from COVID-19 pneumonia[57]. Furthermore, pulmonary edema may present as diffuse or patchy GGOS, but with a central predominance and will change with positioning because of the gravitational predominance. Additionally, pulmonary edema is often accompanied by pleural effusions, an uncommon finding in COVID-19 pneumonia.
Viral causes of pneumonia that can present similarly to COVID-19 pneumonia include influenza virus, parainfluenza virus, adenovirus virus, respiratory syncytial virus, and other coronaviruses[33,58]. The typical findings for these viral infections include those involving the interstitium and non-unique findings such as GGOs, peribronchovascular thickening, centrilobular opacities, “tree-in-bud” pattern, and patchy consolidations[58]. However, some findings that more often support COVID-19 pneumonia specifically include the presence of GGOs in a peripheral and sub-pleural distribution, a reverse halo sign, and vascular enlargement[59]. It is important to note that while these findings may be typical in COVID-19 infection, they are not completely unique to the disease. For example, the reverse halo sign has been described in tuberculosis and various fungal infections, and the sub-pleural distribution of opacities and crazy-paving pattern can also be observed in other coronaviruses such as SARS and Middle East respiratory syndrome virus[60].
Bacterial pneumonias can also be included on the differential diagnosis when examining a chest CT for COVID-19; however, bacterial pneumonia commonly causes a lobar/bronchopneumonia and pleural effusions, both of which are atypical findings in COVID-19 pneumonia[61]. Pneumocystis jiroveci pneumonia commonly presents as GGOs with a crazy-paving pattern in the immunosuppressed host. However, these can typically be observed in the upper lobes and in a central distribution, helping to distinguish it from COVID-19 pneumonia[62]. Other fungal infections such as candidiasis, cryptococcosis, and coccidiomycosis tend to cause lymphadenopathy and cavitation, both uncommon findings in patients with COVID[63].
Neoplasms may also be included in the differential of a suspected COVID-19 patient since the presence of focal GGOs and/or rounded opacities may be features of both[64]. For completeness, additional pathologies to be included in the differential diagnosis include organizing pneumonia, pulmonary alveolar proteinosis, sarcoidosis, pulmonary infarction, various interstitial lung diseases, vasculitides, and aspiration pneumonia[33,55,58,65].
It is important to consider the logistical challenges that radiological imaging has faced throughout the COVID-19 pandemic and understand how those challenges have been dealt with systematically. Because COVID-19 is spread through person-to-person contact and/or respiratory droplets, and one of the most effective methods to prevent spread of the virus is through social distancing, radiology departments around the world were required to come up with efficient and safe protocols to keep staff and patients safe while imaging patients with suspected COVID-19[66]. In general, for radiology departments to continue to safely operate during the COVID-19 pandemic, constant communication concerning the number of positive cases in the department, the available amount of PPE, the currently quarantined staff, as well as scheduling for previously cancelled non-urgent imaging and a daily analysis of labor costs vs. staffing and the available work shift slots are all vital tasks[67]. Many of the specific examples of adjustments made with in departments in response to operational challenges prompted by virus are detailed below.
One of the initial steps taken by many departments to provide safety for those working in the radiology department has been to limit the number of onsite radiologists and ensure a reserve of radiologists at home with less potential exposure to COVID-19. Additionally, any in-person meetings that could be held on a virtual platform were transitioned appropriately[68]. Furthermore, radiologist workstations have been spread out amongst areas in the hospital with extra availability, and the option of remote interpretation has become more widely employed[68]. Finally, many non-urgent imaging examinations such as low-dose CT for lung cancer screening and screening mammography were postponed at one point or another to decrease the volume of patients in and out of the radiology imaging rooms[69]. This latter adjustment led to a significant decrease in outpatient imaging (i.e., In July 2020, an 87% reduction overall in outpatient imaging was observed, with a 93% reduction in mammography specifically)[70].
Patients with suspected or confirmed COVID-19 are now often dedicated to isolated imaging rooms for both chest radiography and CT, and dedicated pathways to these rooms through the hospital have been created to limit contact with unnecessary staff and other patients[71,72]. Moreover, non-essential items within these dedicated COVID-19 imaging rooms are removed to ensure more effective and efficient sanitization sessions[71]. To ensure staff safety during imaging acquisition of patients with suspected or confirmed COVID-19, protocols have been developed for each type of imaging. In chest radiographs, one method reported used two separate radio
Interventional radiology (IR) also has faced several unique challenges throughout the COVID-19 pandemic. At the most basic level, IR suites were forced to redesign their layouts to provide maximal containment of disease before, during, and after patient transmission, as well as minimal transmission through fomite exposure[76,77]. Furthermore, adjustments in case prioritization often via a tier-based system became a necessity to balance both risk of infection with appropriately timed delivery of care to non-COVID-19 patients[78]. The volume of cases in IR was also impacted by the COVID-19 pandemic. For example, according to a survey administered to IR departments in Canada in May of 2020, 50% of respondents reported a decreased demand for acute IR services, which correlated with a simultaneous decrease in emergency department admissions[79,80]. Elective IR procedures were also noted to be reduced as a necessary measure to ensure maximum risk reduction in terms of viral spread[81]. While the overall volume of cases within IR declined as a result of the pandemic and many procedures within IR were documented to decrease in volume, venous IR procedures actually increased in volume[82]. It is possible that this rise could be attributed in part to the association of COVID-19 with venous thromboembolism, as well as the predominantly sedentary lifestyle of the general public in the face of stay-at-home orders and social distancing policies[83].
While SARS-CoV-2 primarily causes respiratory related illness, it has also been demonstrated to manifest pathologically in the cardiac, neurologic, gastrointestinal, genitourinary, vascular, and dermatological systems[38,84]. It is hypothesized that the complex pathophysiology the virus induces, which involves a heightened immune response, coagulation system dysfunction, and severe hypoxia, contribute to its induction of multiorgan system disease processes[84]. Additionally, its mechanism of cell entry via the angiotensin-cell converting enzyme II receptor, which is distributed widely throughout human tissues, may also explain these multisystem manifestations. Briefly, each organ system impacted by COVID-19 and the associated pathologies and the relevant imaging used to assess these pathologies will be described. The reader may reference Table 3 for a comparison of the various imaging modalities used in COVID-19 disease and their associated findings/features characterized by organ system (both pulmonary and extrapulmonary).
| Characteristic Features of COVID-19 by Imaging Modality | ||||
| Organ Systems Impacted by COVID-19 | 1CT | Ultrasound | Magnetic resonance imaging | Chest radiography |
| Pulmonary | 2GGOs and reticular opacities; consolidations; 3crazy paving pattern; multifocal and bilateral in a peripheral, sub-pleural, and posterior distribution | 4B-line artifacts; irregularly thickened pleura; sub-pleural consolidations | Similar to CT | Interstitial reticular and reticulonodular patterns; alveolar hazy pulmonary opacities (equivalent to GGOs on CT); consolidations; multifocal & bilateral in a peripheral, sub-pleural, and posterior distribution |
| Cardiac | Cardiac thromboembolism | Pericardial effusion | Myocarditis; pericardial effusion | |
| Neurological | Stroke (ischemic/thromboembolic) | Venous sinus thrombosis | Stroke (ischemic/thromboembolic); venous sinus thrombosis, hyper-intensities | |
| Gastrointestinal | Wall thickening; edema; fluid filled intestinal lumen; mucosal hyper-enhancement; mesenteric vascular thrombi/ischemia | Portal vein thrombosis | ||
| Genitourinary | Perinephric fat stranding | Renal vein/artery thrombosis | ||
In terms of cardiac dysfunction, COVID-19 disease has been reported to induce myocardial injury, arrythmias (i.e., atrial fibrillation), arterial/venous thromboemboli, cardiomyopathies, myocarditis, and cardiogenic shock[85,86]. Cardiac MRI is the ideal imaging modality to detect cardiac abnormalities in COVID-19 patients and the use of CT angiography is also important in surveying for coagulation related pathologies such as cardiac thrombosis[87,88]. Neurologically, COVID-19 has been reported to be associated with acute stroke, encephalopathy, epilepsy, altered mental status, hypogeusia, hyposmia, and anosmia[89,90]. The use of non-contrast CT and/or non-enhanced MRI can be used to detect areas of infarct or venous sinus thrombosis related to COVID-19[91]. Furthermore, T2-weighted fluid-attenuated-inversion recovery imaging can detect areas of hyper-intensity, one of the most common locations being unilaterally in the mesial temporal lobe[92,93].
Disturbance of the permeability of the small and large intestine caused by SARS-CoV-2 is thought to be one of the mechanisms for gastrointestinal symptoms in COVID-19 patients[94]. In fact, up to 40% of COVID-19 patients present with abdominal symptoms such as acute abdominal pain, nausea/vomiting, and diarrhea, although these symptoms may be a result of referred pain due to the basilar distribution of COVID-19 infection[83,95]. If these symptoms construct the primary chief complaint of the patient, an abdominopelvic CT is indicated; clinicians should include COVID-19 on the differential diagnosis if bilateral ground glass opacities are observed at the lung bases[95]. CT findings of the gastrointestinal tract in COVID-19 patients include wall thickening, edema, fluid filled intestinal lumen, and mucosal hyper-enhancement[96]. Furthermore, the use of CT angiography is beneficial for assessing for mesenteric arterial or venous thrombi and ischemia, a common finding in the COVID-19 patients given the pathophysiology of the disease involves coagulation system dysfunction[97]. Doppler ultrasound may also be employed in situations where portal vein thrombosis is suspected[97].
Other abdominal symptoms are a result of infection of the liver, which is the second most common organ to be involved in COVID-19 infection behind the lungs, and can be adequately assessed with CT, MRI, or ultrasound[98]. It is thought that the virus infects the cholangiocytes of the liver and not the hepatocytes themselves, and thus abnormality in liver function tests or gall bladder enzymes (found in 53% of COVID-19 patients) should prompt the use of these imaging modalities to assess the hepatobiliary system[38].
The genitourinary system, and in particular, the kidneys, are commonly injured among critically ill COVID-19 patients (20%-40%) as a result of infarction and inflammation[99]. The use of ultrasound is first line for evaluating suspected renal vascular involvement as allergies to contrast and renal insufficiency preclude the use of CT with contrast[100]. Other notable extrapulmonary manifestations of COVID-19 that don’t necessarily maintain a role for imaging but are included here for completeness sake include dermatologic pathologies such as COVID toes (frostbite-like toes or “pseudochilbain”) and maculopapular eruptions[101]. These are the result of microvascular thrombosis and often appear in more severe cases[102].
A number of complications from COVID-19 infection can arise and the use of imaging in the detection and monitoring of each may improve patient outcomes and overall survival[103]. The presence of pleural effusions, multiple lung nodules, tree-in-bud opacities, and lymphadenopathy–all uncommon findings in isolated COVID-19 pneumonia–on imaging should raise a suspicion for a bacterial superinfection, a complication reported in 14% of patients in the ICU[104,105]. ARDS, a severe complication in COVID-19 patients more common in patients in critical condition, presents clinically as marked arterial de-oxygenation and respiratory failure and can be confirmed by CT imaging that shows diffuse bilateral areas of GGOs[106]. Pulmonary emboli have been reported to arise in 13% of COVID-19 patients, with the majority of cases also occurring in critically ill patients[103]. Clinical suspicion of this complication should prompt the use of CT-angiography for confirmation and determination of the clinical treatment course[107]. Interestingly, because COVID-19 can lead to both macro- and micro-vascular complications, it is possible that those in the recovery phase of COVID-19 may develop chronic thromboembolic disease (CTED) or chronic thromboembolic pulmonary hypertension (CTEPH)[108]. The work-up of suspected CTED and CTEPH should be pursued with ventilation/perfusion scintigraphy over CT as it more sensitive in detection[109].
Because of the wide variety of disease COVID-19 can cause, it is critical for radiologists to understand the pathophysiology that leads to multiorgan system dysfunction so that complications are recognized more regularly and the detection of one complication prompts a thorough search for others[38]. As more research is pursued and our understanding of the pathophysiology and manifestations of COVID-19 disease evolve, so too will the role of imaging in the detection, diagnosis, and monitoring of disease progression in the extrapulmonary manifestations and complications in COVID-19 patients[38,110].
Teleradiology is a subset of telemedicine that involves the interpretation of diagnostic imaging at a site that is remote from where that image was acquired[111]. It can be categorized as intramural–the radiologist interpreting the imaging works for the institution where the image was taken–or extramural–the radiologist interpreting the imaging works for a group or practice that is not a part of the institution where the image was acquired[112]. Teleradiology was originally used in the 1990s to provide intramural emergency radiology access from remote sites[113]. However, advances in technology and demand from market forces quickly propelled its growth and utility in a variety of aspects of diagnostic imaging[114]. In fact, the global teleradiology market is projected to reach $ 8.2 billion in size by the year 2024[113,115]. During the COVID-19 pandemic, teleradiology has unsurprisingly become an important asset for the field of radiology. It allows for isolation of radiologists from suspected or confirmed COVID-19 patients in the clinical setting and a reduction in the number of staff in the hospital[75]. Additionally, a teleradiology infrastructure that is properly organized and staffed can allow for enhanced preparedness in surges in imaging as a result of COVID-19 patient influxes[78]. With these benefits in mind, it is important to consider the challenges facing teleradiology in aspects such as licensing and credentialing, technology and systems along with their integration, and staffing models[115]. Addressing these will allow for a more robust integration of teleradiology into everyday clinical practice and will improve the response and handling of the COVID-19 pandemic and future pandemics alike.
The use of teleradiology in strengthening our response to surges in imaging, as well as reducing the risk of viral spread via providing the opportunity to socially distance, are not the only aspects of virtual care in the field of radiology that has played a role in the COVID-19 pandemic. IR clinics, for example, have made an effort to transition to virtual appointments to reduce the spread of COVID-19. A recent survey of 122 patients from an Interventional Neuroradiology clinic demonstrated additional benefits in having a virtual clinic beyond social distancing[116]. The study found that virtual clinics are not only more efficient, but also are preferred among patients and physicians in the non-urgent setting[116]. This illustrates an important shift in the delivery of care for patients that not only reduces the transmission of COVID-19, but also delivers more efficient and preferred care[116-118].
A standardized reporting system known as the COVID-19 Reporting and Data System was developed by the Dutch Radiological Society in April of 2020[119]. This task was pursued to promote a form of standardized communication in regard to COVID-19 CT imaging disease findings and improve communication between radiologists and referring physicians[59,119,120]. The Radiological Society of North America also developed a consensus on standardized reporting for COVID-19 imaging findings[121]. A comparison of the two systems can be found in Table 4.
| Standardized CT Imaging Reporting Systems for COVID-19 | ||
| Characteristics of the Reporting System | Coronavirus Disease 2019 Reporting and Data System[118]-Dutch Radiological Society | Consensus Statement on Reporting Chest CT Findings for COVID-19-Radiological Society of North America[120] |
| Type of Reporting System | Quantitative | Qualitative |
| Components & Relationship Between Both Reporting Systems | 0 = inadequate or suboptimal imaging | No equivalent |
| 1 = very low suspicion for COVID-19 with findings of non-infectious etiology | Negative for pneumonia = no CT features to suggest pneumonia | |
| 2 = low suspicion of COVID-19 with infectious findings not typical for COVID-19 | Atypical appearance = absence of typical or indeterminate features & the presence of lobar or segmental consolidation, but no GGOs or centrilobular nodules | |
| 3 = equivocal scan with common findings of COVID-19 | No equivalent | |
| 4 = high suspicion of COVID-19 with typical features that overlap with other viral pneumonias | Indeterminate appearance = absence of typical features and the presence of multifocal, diffuse, or unilateral GGOs with or without consolidation in a non-specific distribution | |
| 5 = very high suspicion of COVID-19 with typical findings of disease in typical locations | Typical appearance = peripheral and bilateral GGOs with or without consolidations/crazy paving pattern | |
| 6 = RT-PCR positive COVID-19 | No equivalent | |
| Inter-observer agreement | Absolute agreement between 68.2% of observers; > 80% observer agreement on COVID-19 being low to very low or high to very high | No data |
Structured reporting aids in the radiologists’ recognition of certain disease patterns, decreases the variability in radiological reporting, and provides more certainty for findings that are likely a result of COVID-19 disease. Furthermore, standardized reporting systems such as these lead to improved educational and research-oriented projects, improve selection of cutoff points that clinicians utilize for clinical management, and enhance the specificity of CT imaging for COVID-19[122]. These systems and future systems alike will continue to improve and their adoption, implementation and utilization among radiologists around the world will be vital for optimal patient care and future clinician education.
Aside from RT-PCR, one of the main point of care diagnostic imaging tools that is showing promise for COVID-19 patients is POCUS. POCUS has begun to gain traction in the medical community for the diagnosis and subsequent management of COVID-19 patients as it offers many benefits. For example, it is quick, affordable, requires no ionizing radiation, can be done at the bedside, and it addresses many of the same clinical questions that chest radiography and CT scans address[123]. In fact, lung ultrasound has been shown to be more sensitive than traditional chest radiography in the detection of infections involving the lower respiratory tract[124,125]. POCUS can not only be useful in the initial diagnosis of COVID-19, but it is also useful in following disease progression and monitoring for many of the associated complications[34]. These include evaluating for ARDS, cardiogenic pulmonary edema, pericardial and pleural effusions, determining ventricular function, assessing for a pneumothorax, screening for deep vein thromboses, assessing adequate lung recruitment during mechanical ventilation, predicting the efficacy of prone positioning, aiding in weaning of patients on mechanical ventilation[126-130].
A recent study from Italy proposed a standardized acquisition protocol and scoring system for lung ultrasound in COVID-19 patients[36]. The acquisition protocol requires scanning of 14 areas (3 posterior, 2 lateral and 2 anterior) for 10 s. The scoring procedure is as follows: 0 = the pleural line is continuous and horizontal artifacts (A-lines) are present; 1 = the pleural line is indented, and vertical areas of white are visible; 2 = the pleural line is broken and below the breaking point are darker areas with corresponding white areas beneath, indicating areas of consolidation; 3 = the pleural line is broken, and the scanned area shows dense and diffuse white lung with or without darker areas of consolidation[36]. Additionally, a United States study developed a 6-zone protocol that emphasizes provider safety, image time acquisition, and focuses mostly on the posterior and lateral fields[131].
It is important to consider the logistical adjustments that need to be made when using POCUS in COVID-19 patients. For example, acquiring video loops instead of static images decreases image acquisition time and thus exposure time. Additionally, POCUS examinations should be performed in pairs with one healthcare provider coming in contact with the patient as to minimize transmission[132]. Furthermore, properly disinfecting machines and the associated equipment and materials involved per manufacturer specific guidelines is critical to ensuring safe use of POCUS in the management of patients with COVID-19[22,133-135].
There are many limitations to the use of POCUS in COVID-19 patients and the evidence supporting its use to date. For example, many of the studies conducted thus far were during a period of high prevalence of disease, which likely influences the diagnostic accuracy of POCUS[136]. Furthermore, inter-operator reproducibility of POCUS on COVID-19 patients is not known. This is especially valuable information as ultrasound is heavily operator dependent and inexperienced providers may not achieve optimal images[137]. However, in general, while there are limited studies and none that are prospective in nature in regard to POCUS in COVID-19, POCUS has many features that offer clinicians valuable data while managing COVID-19 patients[138,139]. More research is needed to better understand the role it plays in managing COVID-19 patients.
Another exciting avenue of research in COVID-19 imaging involves the use of artificial intelligence (AI). One manner through which AI can be utilized in the field of radiology is to help gather and integrate large data sets from disconnected sources that can then be used to create models that aid in predicting diagnosis of disease[140]. Using AI to do this, for example, with imaging findings related to COVID-19, is the most effective method to ensure an expeditious development of these models[19]. These data sets should include not only imaging data, but also the radiology reports and the clinical information such as symptoms and laboratory data[141]. It is also important to note that the widespread use of standardized reporting of COVID-19 imaging findings is necessary for the development of deep learning networks following data set acquisition, which can eventually assist in the detection of COVID-19 based on imaging features and other clinically relevant data[20].
One of the main uses of AI in imaging for the detection of COVID-19 applies to CXR and CT[142]. Several studies have demonstrated the use of AI models that accurately differentiate COVID-19 from community-acquired-pneumonia on based on the differences in their associated imaging features on both CXR and CT[143-145]. Aside from detection and differentiation of disease from other similar presentations, AI models have been developed to assess severity of infection and predict clinical outcomes based on the amount of opacities present, vascular changes, and other pertinent imaging findings[146]. Despite these promising advances, there remains much more room for improvement in the homogeneity of COVID-19 imaging sets and also in the detection and prediction of complications from COVID-19, which contribute significantly to mortality in COVID-19 patients[142,147].
The role of radiology and the radiologist have evolved throughout the COVID-19 pandemic, but both have always remained important in the diagnosis and subsequent management of patients with COVID-19 disease. Many challenges at the operational level have been overcome within the field of radiology and the current rising trend in teleradiology offers an opportunity for better preparedness during the remaining duration of the COVID-19 pandemic and future pandemics alike. Opportunities to better utilize of imaging for detection of extrapulmonary manifestations and complications of COVID-19 disease will arise as a more detailed understanding of the pathophysiology of the virus continues to be uncovered and identification of predisposing risk factors for complication development continue to be better understood. Furthermore, unidentified advancements in areas such as standardized imaging reporting, POCUS and AI offer exciting discovery pathways that will inevitably lead to improved care for patients with COVID-19.
Manuscript source: Invited manuscript
Specialty type: Radiology, nuclear medicine and medical imaging
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Tumminello G S-Editor: Zhang H L-Editor: A P-Editor: Li X
| 1. | Meirelles GSP. COVID-19: a brief update for radiologists. Radiol Bras. 2020;53:320-328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 2. | Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W; China Novel Coronavirus Investigating and Research Team. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020;382:727-733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18987] [Cited by in RCA: 17614] [Article Influence: 3522.8] [Reference Citation Analysis (0)] |
| 3. | Johns Hopkins University. COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). 2020. [cited 7 Feb 2021]. Available from: https://publichealthupdate.com/jhu/. |
| 4. | Woolf SH, Chapman DA, Lee JH. COVID-19 as the Leading Cause of Death in the United States. JAMA. 2021;325:123-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 174] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 5. | Tan BS, Dunnick NR, Gangi A, Goergen S, Jin ZY, Neri E, Nomura CH, Pitcher RD, Yee J, Mahmood U. RSNA International Trends: A Global Perspective on the COVID-19 Pandemic and Radiology in Late 2020. Radiology. 2021;299:E193-E203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 6. | Jiang MD, Zu ZY, Schoepf UJ, Savage RH, Zhang XL, Lu GM, Zhang LJ. Current Status of Etiology, Epidemiology, Clinical Manifestations and Imagings for COVID-19. Korean J Radiol. 2020;21:1138-1149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 7. | Rubin GD, Ryerson CJ, Haramati LB, Sverzellati N, Kanne JP, Raoof S, Schluger NW, Volpi A, Yim JJ, Martin IBK, Anderson DJ, Kong C, Altes T, Bush A, Desai SR, Goldin J, Goo JM, Humbert M, Inoue Y, Kauczor HU, Luo F, Mazzone PJ, Prokop M, Remy-Jardin M, Richeldi L, Schaefer-Prokop CM, Tomiyama N, Wells AU, Leung AN. The Role of Chest Imaging in Patient Management During the COVID-19 Pandemic: A Multinational Consensus Statement From the Fleischner Society. Chest. 2020;158:106-116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 447] [Cited by in RCA: 486] [Article Influence: 97.2] [Reference Citation Analysis (0)] |
| 8. | Ai T, Yang Z, Hou H, Zhan C, Chen C, Lv W, Tao Q, Sun Z, Xia L. Correlation of Chest CT and RT-PCR Testing for Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology. 2020;296:E32-E40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3614] [Cited by in RCA: 3283] [Article Influence: 656.6] [Reference Citation Analysis (0)] |
| 9. | Bernheim A, Mei X, Huang M, Yang Y, Fayad ZA, Zhang N, Diao K, Lin B, Zhu X, Li K, Li S, Shan H, Jacobi A, Chung M. Chest CT Findings in Coronavirus Disease-19 (COVID-19): Relationship to Duration of Infection. Radiology. 2020;295:200463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1728] [Cited by in RCA: 1593] [Article Influence: 318.6] [Reference Citation Analysis (1)] |
| 10. | Kwee TC, Kwee RM. Chest CT in COVID-19: What the Radiologist Needs to Know. Radiographics. 2020;40:1848-1865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 263] [Cited by in RCA: 250] [Article Influence: 50.0] [Reference Citation Analysis (0)] |
| 11. | Roshkovan L, Chatterjee N, Galperin-Aizenberg M, Gupta N, Shah R, Barbosa EM Jr, Simpson S, Cook T, Nachiappan A, Knollmann F, Litt H, Desjardins B, Jha S, Panebianco N, Baston C, Thompson JC, Katz SI. The Role of Imaging in the Management of Suspected or Known COVID-19 Pneumonia. A Multidisciplinary Perspective. Ann Am Thorac Soc. 2020;17:1358-1365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 12. | Floridi C, Fogante M, Agostini A, Borgheresi A, Cellina M, Natella R, Bruno F, Cozzi D, Maggialetti N, Palumbo P, Miele V, Carotti M, Giovagnoni A. Radiological diagnosis of Coronavirus Disease 2019 (COVID-19): a Practical Guide. Acta Biomed. 2020;91:51-59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 13. | Gravell RJ, Theodoreson MD, Buonsenso D, Curtis J. Radiological manifestations of COVID-19: key points for the physician. Br J Hosp Med (Lond). 2020;81:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Wong MD, Thai T, Li Y, Liu H. The role of chest computed tomography in the management of COVID-19: A review of results and recommendations. Exp Biol Med (Maywood). 2020;245:1096-1103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 15. | Kim H, Hong H, Yoon SH. Diagnostic Performance of CT and Reverse Transcriptase Polymerase Chain Reaction for Coronavirus Disease 2019: A Meta-Analysis. Radiology. 2020;296:E145-E155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 354] [Cited by in RCA: 365] [Article Influence: 73.0] [Reference Citation Analysis (0)] |
| 16. | Kovács A, Palásti P, Veréb D, Bozsik B, Palkó A, Kincses ZT. The sensitivity and specificity of chest CT in the diagnosis of COVID-19. Eur Radiol. 2021;31:2819-2824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 106] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 17. | Laghi A, Grassi R. Italian Radiology's Response to the COVID-19 Outbreak. J Am Coll Radiol. 2020;17:699-700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 18. | Li M. Chest CT features and their role in COVID-19. Radiol Infect Dis. 2020;7:51-54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 19. | Kaufman AE, Naidu S, Ramachandran S, Kaufman DS, Fayad ZA, Mani V. Review of radiographic findings in COVID-19. World J Radiol. 2020;12:142-155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 34] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 20. | Salehi S, Abedi A, Balakrishnan S, Gholamrezanezhad A. Coronavirus disease 2019 (COVID-19) imaging reporting and data system (COVID-RADS) and common lexicon: a proposal based on the imaging data of 37 studies. Eur Radiol. 2020;30:4930-4942. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 105] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 21. | Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS; China Medical Treatment Expert Group for Covid-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382:1708-1720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19202] [Cited by in RCA: 18852] [Article Influence: 3770.4] [Reference Citation Analysis (7)] |
| 22. | Gandhi D, Ahuja K, Grover H, Sharma P, Solanki S, Gupta N, Patel L. Review of X-ray and computed tomography scan findings with a promising role of point of care ultrasound in COVID-19 pandemic. World J Radiol. 2020;12:195-203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 12] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 23. | Fields BKK, Demirjian NL, Dadgar H, Gholamrezanezhad A. Imaging of COVID-19: CT, MRI, and PET. Semin Nucl Med. 2021;51:312-320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 24. | Hani C, Trieu NH, Saab I, Dangeard S, Bennani S, Chassagnon G, Revel MP. COVID-19 pneumonia: A review of typical CT findings and differential diagnosis. Diagn Interv Imaging. 2020;101:263-268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 231] [Cited by in RCA: 263] [Article Influence: 52.6] [Reference Citation Analysis (0)] |
| 25. | Pontone G, Scafuri S, Mancini ME, Agalbato C, Guglielmo M, Baggiano A, Muscogiuri G, Fusini L, Andreini D, Mushtaq S, Conte E, Annoni A, Formenti A, Gennari AG, Guaricci AI, Rabbat MR, Pompilio G, Pepi M, Rossi A. Role of computed tomography in COVID-19. J Cardiovasc Comput Tomogr. 2021;15:27-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 69] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 26. | Shi H, Han X, Jiang N, Cao Y, Alwalid O, Gu J, Fan Y, Zheng C. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis. 2020;20:425-434. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2493] [Cited by in RCA: 2309] [Article Influence: 461.8] [Reference Citation Analysis (0)] |
| 27. | Bao C, Liu X, Zhang H, Li Y, Liu J. Coronavirus Disease 2019 (COVID-19) CT Findings: A Systematic Review and Meta-analysis. J Am Coll Radiol. 2020;17:701-709. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 294] [Cited by in RCA: 269] [Article Influence: 53.8] [Reference Citation Analysis (0)] |
| 28. | Nagpal P, Narayanasamy S, Vidholia A, Guo J, Shin KM, Lee CH, Hoffman EA. Imaging of COVID-19 pneumonia: Patterns, pathogenesis, and advances. Br J Radiol. 2020;93:20200538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 29. | Song F, Shi N, Shan F, Zhang Z, Shen J, Lu H, Ling Y, Jiang Y, Shi Y. Emerging 2019 Novel Coronavirus (2019-nCoV) Pneumonia. Radiology. 2020;297:E346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 30. | McLaren TA, Gruden JF, Green DB. The bullseye sign: A variant of the reverse halo sign in COVID-19 pneumonia. Clin Imaging. 2020;68:191-196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 31. | Pan F, Ye T, Sun P, Gui S, Liang B, Li L, Zheng D, Wang J, Hesketh RL, Yang L, Zheng C. Time Course of Lung Changes at Chest CT during Recovery from Coronavirus Disease 2019 (COVID-19). Radiology. 2020;295:715-721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1617] [Cited by in RCA: 1758] [Article Influence: 351.6] [Reference Citation Analysis (0)] |
| 32. | Wang Y, Dong C, Hu Y, Li C, Ren Q, Zhang X, Shi H, Zhou M. Temporal Changes of CT Findings in 90 Patients with COVID-19 Pneumonia: A Longitudinal Study. Radiology. 2020;296:E55-E64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 493] [Cited by in RCA: 588] [Article Influence: 117.6] [Reference Citation Analysis (0)] |
| 33. | Fan L, Li D, Xue H, Zhang L, Liu Z, Zhang B, Yang W, Xie B, Duan X, Hu X, Cheng K, Peng L, Yu N, Song L, Chen H, Sui X, Zheng N, Liu S, Jin Z. Progress and prospect on imaging diagnosis of COVID-19. Chin J Acad Radiol. 2020;1-10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 34. | Kulkarni S, Down B, Jha S. Point-of-care lung ultrasound in intensive care during the COVID-19 pandemic. Clin Radiol. 2020;75:710.e1-710.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 35. | Soldati G, Smargiassi A, Inchingolo R, Buonsenso D, Perrone T, Briganti DF, Perlini S, Torri E, Mariani A, Mossolani EE, Tursi F, Mento F, Demi L. Is There a Role for Lung Ultrasound During the COVID-19 Pandemic? J Ultrasound Med. 2020;39:1459-1462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 298] [Cited by in RCA: 311] [Article Influence: 62.2] [Reference Citation Analysis (0)] |
| 36. | Soldati G, Smargiassi A, Inchingolo R, Buonsenso D, Perrone T, Briganti DF, Perlini S, Torri E, Mariani A, Mossolani EE, Tursi F, Mento F, Demi L. Proposal for International Standardization of the Use of Lung Ultrasound for Patients With COVID-19: A Simple, Quantitative, Reproducible Method. J Ultrasound Med. 2020;39:1413-1419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 380] [Cited by in RCA: 421] [Article Influence: 84.2] [Reference Citation Analysis (0)] |
| 37. | Revzin MV, Raza S, Warshawsky R, D'Agostino C, Srivastava NC, Bader AS, Malhotra A, Patel RD, Chen K, Kyriakakos C, Pellerito JS. Multisystem Imaging Manifestations of COVID-19, Part 1: Viral Pathogenesis and Pulmonary and Vascular System Complications. Radiographics. 2020;40:1574-1599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (2)] |
| 38. | Revzin MV, Raza S, Srivastava NC, Warshawsky R, D'Agostino C, Malhotra A, Bader AS, Patel RD, Chen K, Kyriakakos C, Pellerito JS. Multisystem Imaging Manifestations of COVID-19, Part 2: From Cardiac Complications to Pediatric Manifestations. Radiographics. 2020;40:1866-1892. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 39. | Ghosh S, Deshwal H, Saeedan MB, Khanna VK, Raoof S, Mehta AC. Imaging algorithm for COVID-19: A practical approach. Clin Imaging. 2021;72:22-30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 40. | Maurea S, Mainolfi CG, Bombace C, Annunziata A, Attanasio L, Petretta M, Del Vecchio S, Cuocolo A. FDG-PET/CT imaging during the Covid-19 emergency: a southern Italian perspective. Eur J Nucl Med Mol Imaging. 2020;47:2691-2697. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 41. | Polverari G, Arena V, Ceci F, Pelosi E, Ianniello A, Poli E, Sandri A, Penna D. 18F-Fluorodeoxyglucose Uptake in Patient With Asymptomatic Severe Acute Respiratory Syndrome Coronavirus 2 (Coronavirus Disease 2019) Referred to Positron Emission Tomography/Computed Tomography for NSCLC Restaging. J Thorac Oncol. 2020;15:1078-1080. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 42. | Qin C, Liu F, Yen TC, Lan X. 18F-FDG PET/CT findings of COVID-19: a series of four highly suspected cases. Eur J Nucl Med Mol Imaging. 2020;47:1281-1286. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 205] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 43. | Fontana IC, Bongarzone S, Gee A, Souza DO, Zimmer ER. PET Imaging as a Tool for Assessing COVID-19 Brain Changes. Trends Neurosci. 2020;43:935-938. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 44. | Guedj E, Million M, Dudouet P, Tissot-Dupont H, Bregeon F, Cammilleri S, Raoult D. 18F-FDG brain PET hypometabolism in post-SARS-CoV-2 infection: substrate for persistent/delayed disorders? Eur J Nucl Med Mol Imaging. 2021;48:592-595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 120] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 45. | Ates OF, Taydas O, Dheir H. Thorax Magnetic Resonance Imaging Findings in Patients with Coronavirus Disease (COVID-19). Acad Radiol. 2020;27:1373-1378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 46. | Yang S, Zhang Y, Shen J, Dai Y, Ling Y, Lu H, Zhang R, Ding X, Qi H, Shi Y, Zhang Z, Shan F. Clinical Potential of UTE-MRI for Assessing COVID-19: Patient- and Lesion-Based Comparative Analysis. J Magn Reson Imaging. 2020;52:397-406. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 47. | Torkian P, Rajebi H, Zamani T, Ramezani N, Kiani P, Akhlaghpoor S. Magnetic resonance imaging features of coronavirus disease 2019 (COVID-19) pneumonia: The first preliminary case series. Clin Imaging. 2021;69:261-265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 48. | Wasilewski PG, Mruk B, Mazur S, Półtorak-Szymczak G, Sklinda K, Walecki J. COVID-19 severity scoring systems in radiological imaging - a review. Pol J Radiol. 2020;85:e361-e368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 49. | Taylor E, Haven K, Reed P, Bissielo A, Harvey D, McArthur C, Bringans C, Freundlich S, Ingram RJ, Perry D, Wilson F, Milne D, Modahl L, Huang QS, Gross D, Widdowson MA, Grant CC; SHIVERS Investigation Team. A chest radiograph scoring system in patients with severe acute respiratory infection: a validation study. BMC Med Imaging. 2015;15:61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 50. | Yoon SH, Lee KH, Kim JY, Lee YK, Ko H, Kim KH, Park CM, Kim YH. Chest Radiographic and CT Findings of the 2019 Novel Coronavirus Disease (COVID-19): Analysis of Nine Patients Treated in Korea. Korean J Radiol. 2020;21:494-500. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 431] [Cited by in RCA: 374] [Article Influence: 74.8] [Reference Citation Analysis (0)] |
| 51. | Borghesi A, Maroldi R. COVID-19 outbreak in Italy: experimental chest X-ray scoring system for quantifying and monitoring disease progression. Radiol Med. 2020;125:509-513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 263] [Article Influence: 52.6] [Reference Citation Analysis (0)] |
| 52. | Yang W, Sirajuddin A, Zhang X, Liu G, Teng Z, Zhao S, Lu M. The role of imaging in 2019 novel coronavirus pneumonia (COVID-19). Eur Radiol. 2020;30:4874-4882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 177] [Article Influence: 35.4] [Reference Citation Analysis (0)] |
| 53. | Li K, Fang Y, Li W, Pan C, Qin P, Zhong Y, Liu X, Huang M, Liao Y, Li S. CT image visual quantitative evaluation and clinical classification of coronavirus disease (COVID-19). Eur Radiol. 2020;30:4407-4416. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 458] [Cited by in RCA: 455] [Article Influence: 91.0] [Reference Citation Analysis (0)] |
| 54. | Li K, Wu J, Wu F, Guo D, Chen L, Fang Z, Li C. The Clinical and Chest CT Features Associated With Severe and Critical COVID-19 Pneumonia. Invest Radiol. 2020;55:327-331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 870] [Cited by in RCA: 796] [Article Influence: 159.2] [Reference Citation Analysis (0)] |
| 55. | Bakhshayeshkaram M, Haseli S, Iranpour P. Radiological Mimickers of COVID-19 Pneumonia: A Pictorial Review. Tanaffos. 2020;19:100-107. [PubMed] |
| 56. | Lynch DA. Ground glass attenuation on CT in patients with idiopathic pulmonary fibrosis. Chest. 1996;110:312-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 57. | Thoongsuwan N, Kanne JP, Stern EJ. Spectrum of blunt chest injuries. J Thorac Imaging. 2005;20:89-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 58. | Duzgun SA, Durhan G, Demirkazik FB, Akpinar MG, Ariyurek OM. COVID-19 pneumonia: the great radiological mimicker. Insights Imaging. 2020;11:118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 59. | Bai HX, Hsieh B, Xiong Z, Halsey K, Choi JW, Tran TML, Pan I, Shi LB, Wang DC, Mei J, Jiang XL, Zeng QH, Egglin TK, Hu PF, Agarwal S, Xie FF, Li S, Healey T, Atalay MK, Liao WH. Performance of Radiologists in Differentiating COVID-19 from Non-COVID-19 Viral Pneumonia at Chest CT. Radiology. 2020;296:E46-E54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 726] [Cited by in RCA: 726] [Article Influence: 145.2] [Reference Citation Analysis (0)] |
| 60. | Marchiori E, Zanetti G, Escuissato DL, Souza AS Jr, de Souza Portes Meirelles G, Fagundes J, Souza CA, Hochhegger B, Marom EM, Godoy MCB. Reversed halo sign: high-resolution CT scan findings in 79 patients. Chest. 2012;141:1260-1266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 69] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 61. | Miller WT Jr, Mickus TJ, Barbosa E Jr, Mullin C, Van Deerlin VM, Shiley KT. CT of viral lower respiratory tract infections in adults: comparison among viral organisms and between viral and bacterial infections. AJR Am J Roentgenol. 2011;197:1088-1095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 86] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 62. | Fujii T, Nakamura T, Iwamoto A. Pneumocystis pneumonia in patients with HIV infection: clinical manifestations, laboratory findings, and radiological features. J Infect Chemother. 2007;13:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 90] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 63. | Orlowski HLP, McWilliams S, Mellnick VM, Bhalla S, Lubner MG, Pickhardt PJ, Menias CO. Imaging Spectrum of Invasive Fungal and Fungal-like Infections. Radiographics. 2017;37:1119-1134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 90] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 64. | Chung M, Bernheim A, Mei X, Zhang N, Huang M, Zeng X, Cui J, Xu W, Yang Y, Fayad ZA, Jacobi A, Li K, Li S, Shan H. CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV). Radiology. 2020;295:202-207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1827] [Cited by in RCA: 1693] [Article Influence: 338.6] [Reference Citation Analysis (0)] |
| 65. | McStay R, Johnstone A, Hare SS, Jacob J, Nair A, Rodrigues JCL, Edey A, Robinson G. COVID-19: looking beyond the peak. Challenges and tips for radiologists in follow-up of a novel patient cohort. Clin Radiol. 2021;76:74.e1-74.e14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 66. | Fallucchi F, Faravelli M, Quercia S. Fair allocation of scarce medical resources in the time of COVID-19: what do people think? J Med Ethics. 2021;47:3-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 67. | Seghers VJ, Desai NK, Masand PM, Nasir S, Foster TL, Indiero DA, Johnson TD, Huisman TAGM. After the initial COVID-19 surge: a phased radiology departmental re-opening plan. Pediatr Radiol. 2020;50:1551-1559. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 68. | Prabhakar AM, Glover M 4th, Schaefer PW, Brink JA. Academic Radiology Departmental Operational Strategy Related to the Coronavirus Disease 2019 (COVID-19) Pandemic. J Am Coll Radiol. 2020;17:730-733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 69. | Rosen MP, Norbash A, Kruskal J, Meltzer CC, Yee J, Thrall J. Impact of Coronavirus Disease 2019 (COVID-19) on the Practice of Clinical Radiology. J Am Coll Radiol. 2020;17:1096-1100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 70. | Naidich JJ, Boltyenkov A, Wang JJ, Chusid J, Hughes D, Sanelli PC. Impact of the Coronavirus Disease 2019 (COVID-19) Pandemic on Imaging Case Volumes. J Am Coll Radiol. 2020;17:865-872. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 125] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 71. | Wan YL, Schoepf UJ, Wu CC, Giovagnoli DP, Wu MT, Hsu HH, Chang YC, Yang CT, Cherng WJ. Preparedness and Best Practice in Radiology Department for COVID-19 and Other Future Pandemics of Severe Acute Respiratory Infection. J Thorac Imaging. 2020;35:239-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 72. | Zanardo M, Martini C, Monti CB, Cattaneo F, Ciaralli C, Cornacchione P, Durante S. Management of patients with suspected or confirmed COVID-19, in the radiology department. Radiography (Lond). 2020;26:264-268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 73. | Kooraki S, Hosseiny M, Raman SS, Myers L, Gholamrezanezhad A. Coronavirus Disease 2019 (COVID-19) Precautions: What the MRI Suite Should Know. J Am Coll Radiol. 2020;17:830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 74. | Stogiannos N, Fotopoulos D, Woznitza N, Malamateniou C. COVID-19 in the radiology department: What radiographers need to know. Radiography (Lond). 2020;26:254-263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 75. | Mossa-Basha M, Medverd J, Linnau KF, Lynch JB, Wener MH, Kicska G, Staiger T, Sahani DV. Policies and Guidelines for COVID-19 Preparedness: Experiences from the University of Washington. Radiology. 2020;296:E26-E31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 76. | H'ng MW, Tan BP, Punamiya S. Setting Up an Interventional Radiology Suite in an Infectious Diseases Centre: Lessons from the Novel Coronavirus (COVID-19) Outbreak. Ann Acad Med Singap. 2020;49:684-689. [PubMed] |
| 77. | Lamparello NA, Choi S, Charalel R, Lee KS, Kesselman A, Scherer K, Harnain CM, Browne WF, Shiffman M, Holzwanger DJ, Pua BB. Transforming Positive Pressure IR Suites to Treat COVID-19 Patients. J Vasc Interv Radiol. 2020;31:1496-1499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 78. | Orsi MA, Oliva AG, Cellina M. Radiology Department Preparedness for COVID-19: Facing an Unexpected Outbreak of the Disease. Radiology. 2020;295:E8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 79. | Lazzerini M, Barbi E, Apicella A, Marchetti F, Cardinale F, Trobia G. Delayed access or provision of care in Italy resulting from fear of COVID-19. Lancet Child Adolesc Health. 2020;4:e10-e11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 708] [Cited by in RCA: 882] [Article Influence: 176.4] [Reference Citation Analysis (0)] |
| 80. | Patel NR, El-Karim GA, Mujoomdar A, Mafeld S, Jaberi A, Kachura JR, Tan KT, Oreopoulos GD. Overall Impact of the COVID-19 Pandemic on Interventional Radiology Services: A Canadian Perspective. Can Assoc Radiol J. 2020;846537120951960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 81. | Mujoomdar A, Graham T, Baerlocher MO, Soulez G. The Canadian Association for Interventional Radiology (CAIR) and Canadian Association of Radiologists (CAR) Guidelines for Interventional Radiology Procedures for Patients With Suspected or Confirmed COVID-19. Can Assoc Radiol J. 2020;71:514-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 82. | Marone EM, Rinaldi LF. Upsurge of deep venous thrombosis in patients affected by COVID-19: Preliminary data and possible explanations. J Vasc Surg Venous Lymphat Disord. 2020;8:694-695. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 83. | Zhang L, Feng X, Zhang D, Jiang C, Mei H, Wang J, Zhang C, Li H, Xia X, Kong S, Liao J, Jia H, Pang X, Song Y, Tian Y, Wang B, Wu C, Yuan H, Zhang Y, Li Y, Sun W, Zhu S, Wang S, Xie Y, Ge S, Zhang L, Hu Y, Xie M. Deep Vein Thrombosis in Hospitalized Patients With COVID-19 in Wuhan, China: Prevalence, Risk Factors, and Outcome. Circulation. 2020;142:114-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 317] [Article Influence: 63.4] [Reference Citation Analysis (0)] |
| 84. | Behzad S, Aghaghazvini L, Radmard AR, Gholamrezanezhad A. Extrapulmonary manifestations of COVID-19: Radiologic and clinical overview. Clin Imaging. 2020;66:35-41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 176] [Cited by in RCA: 178] [Article Influence: 35.6] [Reference Citation Analysis (0)] |
| 85. | Kang Y, Chen T, Mui D, Ferrari V, Jagasia D, Scherrer-Crosbie M, Chen Y, Han Y. Cardiovascular manifestations and treatment considerations in COVID-19. Heart. 2020;106:1132-1141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 252] [Cited by in RCA: 262] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 86. | Rojulpote C, Gonuguntla K, Patil S, Bhattaru A, Bravo PE. COVID-19 and the Heart. Colomb Med (Cali). 2020;51:e4320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 87. | Citro R, Pontone G, Bellino M, Silverio A, Iuliano G, Baggiano A, Manka R, Iesu S, Vecchione C, Asch FM, Ghadri JR, Templin C. Role of multimodality imaging in evaluation of cardiovascular involvement in COVID-19. Trends Cardiovasc Med. 2021;31:8-16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 88. | Gupta A, Madhavan MV, Sehgal K, Nair N, Mahajan S, Sehrawat TS, Bikdeli B, Ahluwalia N, Ausiello JC, Wan EY, Freedberg DE, Kirtane AJ, Parikh SA, Maurer MS, Nordvig AS, Accili D, Bathon JM, Mohan S, Bauer KA, Leon MB, Krumholz HM, Uriel N, Mehra MR, Elkind MSV, Stone GW, Schwartz A, Ho DD, Bilezikian JP, Landry DW. Extrapulmonary manifestations of COVID-19. Nat Med. 2020;26:1017-1032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2419] [Cited by in RCA: 2043] [Article Influence: 408.6] [Reference Citation Analysis (2)] |
| 89. | Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, Chang J, Hong C, Zhou Y, Wang D, Miao X, Li Y, Hu B. Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4761] [Cited by in RCA: 4695] [Article Influence: 939.0] [Reference Citation Analysis (0)] |
| 90. | Wu Y, Xu X, Chen Z, Duan J, Hashimoto K, Yang L, Liu C, Yang C. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav Immun. 2020;87:18-22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1219] [Cited by in RCA: 1252] [Article Influence: 250.4] [Reference Citation Analysis (0)] |
| 91. | Egbert AR, Cankurtaran S, Karpiak S. Brain abnormalities in COVID-19 acute/subacute phase: A rapid systematic review. Brain Behav Immun. 2020;89:543-554. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 77] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 92. | Conklin J, Frosch MP, Mukerji S, Rapalino O, Maher M, Schaefer PW, Lev MH, Gonzalez RG, Das S, Champion SN, Magdamo C, Sen P, Harrold GK, Alabsi H, Normandin E, Shaw B, Lemieux J, Sabeti P, Branda JA, Brown EN, Westover MB, Huang SY, Edlow BL. Cerebral Microvascular Injury in Severe COVID-19. medRxiv. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 93. | Kremer S, Lersy F, de Sèze J, Ferré JC, Maamar A, Carsin-Nicol B, Collange O, Bonneville F, Adam G, Martin-Blondel G, Rafiq M, Geeraerts T, Delamarre L, Grand S, Krainik A, Caillard S, Constans JM, Metanbou S, Heintz A, Helms J, Schenck M, Lefèbvre N, Boutet C, Fabre X, Forestier G, de Beaurepaire I, Bornet G, Lacalm A, Oesterlé H, Bolognini F, Messié J, Hmeydia G, Benzakoun J, Oppenheim C, Bapst B, Megdiche I, Henry Feugeas MC, Khalil A, Gaudemer A, Jager L, Nesser P, Talla Mba Y, Hemmert C, Feuerstein P, Sebag N, Carré S, Alleg M, Lecocq C, Schmitt E, Anxionnat R, Zhu F, Comby PO, Ricolfi F, Thouant P, Desal H, Boulouis G, Berge J, Kazémi A, Pyatigorskaya N, Lecler A, Saleme S, Edjlali-Goujon M, Kerleroux B, Zorn PE, Matthieu M, Baloglu S, Ardellier FD, Willaume T, Brisset JC, Boulay C, Mutschler V, Hansmann Y, Mertes PM, Schneider F, Fafi-Kremer S, Ohana M, Meziani F, David JS, Meyer N, Anheim M, Cotton F. Brain MRI Findings in Severe COVID-19: A Retrospective Observational Study. Radiology. 2020;297:E242-E251. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 288] [Cited by in RCA: 305] [Article Influence: 61.0] [Reference Citation Analysis (0)] |
| 94. | Gu J, Han B, Wang J. COVID-19: Gastrointestinal Manifestations and Potential Fecal-Oral Transmission. Gastroenterology. 2020;158:1518-1519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 954] [Cited by in RCA: 949] [Article Influence: 189.8] [Reference Citation Analysis (1)] |
| 95. | Dane B, Brusca-Augello G, Kim D, Katz DS. Unexpected Findings of Coronavirus Disease (COVID-19) at the Lung Bases on Abdominopelvic CT. AJR Am J Roentgenol. 2020;215:603-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 96. | Bhayana R, Som A, Li MD, Carey DE, Anderson MA, Blake MA, Catalano O, Gee MS, Hahn PF, Harisinghani M, Kilcoyne A, Lee SI, Mojtahed A, Pandharipande PV, Pierce TT, Rosman DA, Saini S, Samir AE, Simeone JF, Gervais DA, Velmahos G, Misdraji J, Kambadakone A. Abdominal Imaging Findings in COVID-19: Preliminary Observations. Radiology. 2020;297:E207-E215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 227] [Article Influence: 45.4] [Reference Citation Analysis (0)] |
| 97. | Parry AH, Wani AH, Yaseen M. Acute Mesenteric Ischemia in Severe Coronavirus-19 (COVID-19): Possible Mechanisms and Diagnostic Pathway. Acad Radiol. 2020;27:1190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 98. | Xu L, Liu J, Lu M, Yang D, Zheng X. Liver injury during highly pathogenic human coronavirus infections. Liver Int. 2020;40:998-1004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 622] [Cited by in RCA: 574] [Article Influence: 114.8] [Reference Citation Analysis (0)] |
| 99. | Petrilli CM, Jones SA, Yang J, Rajagopalan H, O'Donnell L, Chernyak Y, Tobin KA, Cerfolio RJ, Francois F, Horwitz LI. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1590] [Cited by in RCA: 1814] [Article Influence: 362.8] [Reference Citation Analysis (1)] |
| 100. | Fabrizi F, Alfieri CM, Cerutti R, Lunghi G, Messa P. COVID-19 and Acute Kidney Injury: A Systematic Review and Meta-Analysis. Pathogens. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 54] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 101. | Galván Casas C, Català A, Carretero Hernández G, Rodríguez-Jiménez P, Fernández-Nieto D, Rodríguez-Villa Lario A, Navarro Fernández I, Ruiz-Villaverde R, Falkenhain-López D, Llamas Velasco M, García-Gavín J, Baniandrés O, González-Cruz C, Morillas-Lahuerta V, Cubiró X, Figueras Nart I, Selda-Enriquez G, Romaní J, Fustà-Novell X, Melian-Olivera A, Roncero Riesco M, Burgos-Blasco P, Sola Ortigosa J, Feito Rodriguez M, García-Doval I. Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020;183:71-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 783] [Cited by in RCA: 878] [Article Influence: 175.6] [Reference Citation Analysis (1)] |
| 102. | Manalo IF, Smith MK, Cheeley J, Jacobs R. A dermatologic manifestation of COVID-19: Transient livedo reticularis. J Am Acad Dermatol. 2020;83:700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 154] [Cited by in RCA: 153] [Article Influence: 30.6] [Reference Citation Analysis (0)] |
| 103. | Monpara JD, Sodha SJ, Gupta PK. COVID-19 associated complications and potential therapeutic targets. Eur J Pharmacol. 2020;886:173548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 104. | Bassetti M, Kollef MH, Timsit JF. Bacterial and fungal superinfections in critically ill patients with COVID-19. Intensive Care Med. 2020;46:2071-2074. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 77] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 105. | Jajodia A, Ebner L, Heidinger B, Chaturvedi A, Prosch H. Imaging in corona virus disease 2019 (COVID-19)-A Scoping review. Eur J Radiol Open. 2020;7:100237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 106. | Tang X, Du RH, Wang R, Cao TZ, Guan LL, Yang CQ, Zhu Q, Hu M, Li XY, Li Y, Liang LR, Tong ZH, Sun B, Peng P, Shi HZ. Comparison of Hospitalized Patients With ARDS Caused by COVID-19 and H1N1. Chest. 2020;158:195-205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 235] [Cited by in RCA: 236] [Article Influence: 47.2] [Reference Citation Analysis (0)] |
| 107. | Malas MB, Naazie IN, Elsayed N, Mathlouthi A, Marmor R, Clary B. Thromboembolism risk of COVID-19 is high and associated with a higher risk of mortality: A systematic review and meta-analysis. EClinicalMedicine. 2020;29:100639. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 424] [Cited by in RCA: 412] [Article Influence: 82.4] [Reference Citation Analysis (0)] |
| 108. | Gall H, Hoeper MM, Richter MJ, Cacheris W, Hinzmann B, Mayer E. An epidemiological analysis of the burden of chronic thromboembolic pulmonary hypertension in the USA, Europe and Japan. Eur Respir Rev. 2017;26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 147] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 109. | Gopalan D, Delcroix M, Held M. Diagnosis of chronic thromboembolic pulmonary hypertension. Eur Respir Rev. 2017;26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 109] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 110. | Azer SA. COVID-19: pathophysiology, diagnosis, complications and investigational therapeutics. New Microbes New Infect. 2020;37:100738. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 70] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 111. | Bashshur RL, Krupinski EA, Thrall JH, Bashshur N. The Empirical Foundations of Teleradiology and Related Applications: A Review of the Evidence. Telemed J E Health. 2016;22:868-898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 112. | European Society of Radiology (ESR). ESR white paper on teleradiology: an update from the teleradiology subgroup. Insights Imaging. 2014;5:1-8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 113. | Thrall JH. Teleradiology. Part I. History and clinical applications. Radiology. 2007;243:613-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 75] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 114. | Boland GW. Teleradiology coming of age: winners and losers. AJR Am J Roentgenol. 2008;190:1161-1162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 115. | Hanna TN, Steenburg SD, Rosenkrantz AB, Pyatt RS Jr, Duszak R Jr, Friedberg EB. Emerging Challenges and Opportunities in the Evolution of Teleradiology. AJR Am J Roentgenol. 2020;215:1411-1416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 116. | Lun R, Walker G, Daham Z, Ramsay T, Portela de Oliveira E, Kassab M, Fahed R, Quateen A, Lesiuk H, P Dos Santos M, Drake B. Transition to virtual appointments for interventional neuroradiology due to the COVID-19 pandemic: a survey of satisfaction. J Neurointerv Surg. 2020;12:1153-1156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 117. | Lam PW, Sehgal P, Andany N, Mubareka S, Simor AE, Ozaldin O, Leis JA, Daneman N, Chan AK. A virtual care program for outpatients diagnosed with COVID-19: a feasibility study. CMAJ Open. 2020;8:E407-E413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 118. | Robb JF, Hyland MH, Goodman AD. Comparison of telemedicine versus in-person visits for persons with multiple sclerosis: A randomized crossover study of feasibility, cost, and satisfaction. Mult Scler Relat Disord. 2019;36:101258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 70] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 119. | Prokop M, van Everdingen W, van Rees Vellinga T, Quarles van Ufford H, Stöger L, Beenen L, Geurts B, Gietema H, Krdzalic J, Schaefer-Prokop C, van Ginneken B, Brink M; COVID-19 Standardized Reporting Working Group of the Dutch Radiological Society. CO-RADS: A Categorical CT Assessment Scheme for Patients Suspected of Having COVID-19-Definition and Evaluation. Radiology. 2020;296:E97-E104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 646] [Cited by in RCA: 589] [Article Influence: 117.8] [Reference Citation Analysis (0)] |
| 120. | Vermani S, Kaushal A, Kaushal J. COVID-19 and the Radiology Department: What We Know So Far. SN Compr Clin Med. 2020;1-7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 121. | Simpson S, Kay FU, Abbara S, Bhalla S, Chung JH, Chung M, Henry TS, Kanne JP, Kligerman S, Ko JP, Litt H. Radiological Society of North America Expert Consensus Statement on Reporting Chest CT Findings Related to COVID-19. Endorsed by the Society of Thoracic Radiology, the American College of Radiology, and RSNA - Secondary Publication. J Thorac Imaging. 2020;35:219-227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 400] [Cited by in RCA: 563] [Article Influence: 112.6] [Reference Citation Analysis (0)] |
| 122. | Figliozzi S, Masci PG, Ahmadi N, Tondi L, Koutli E, Aimo A, Stamatelopoulos K, Dimopoulos MA, Caforio ALP, Georgiopoulos G. Predictors of adverse prognosis in COVID-19: A systematic review and meta-analysis. Eur J Clin Invest. 2020;50:e13362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 225] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 123. | Kumar S, Kumar A, Goel P, Vyas S, Baitha U, Wig N. Use of ultrasonography in COVID-19: Probing for success. J Family Med Prim Care. 2020;9:3863-3866. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 124. | Bitar ZI, Maadarani OS, El-Shably AM, Al-Ajmi MJ. Diagnostic accuracy of chest ultrasound in patients with pneumonia in the intensive care unit: A single-hospital study. Health Sci Rep. 2019;2:e102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 125. | Testa A, Soldati G, Copetti R, Giannuzzi R, Portale G, Gentiloni-Silveri N. Early recognition of the 2009 pandemic influenza A (H1N1) pneumonia by chest ultrasound. Crit Care. 2012;16:R30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 110] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 126. | Bikdeli B, Madhavan MV, Jimenez D, Chuich T, Dreyfus I, Driggin E, Nigoghossian C, Ageno W, Madjid M, Guo Y, Tang LV, Hu Y, Giri J, Cushman M, Quéré I, Dimakakos EP, Gibson CM, Lippi G, Favaloro EJ, Fareed J, Caprini JA, Tafur AJ, Burton JR, Francese DP, Wang EY, Falanga A, McLintock C, Hunt BJ, Spyropoulos AC, Barnes GD, Eikelboom JW, Weinberg I, Schulman S, Carrier M, Piazza G, Beckman JA, Steg PG, Stone GW, Rosenkranz S, Goldhaber SZ, Parikh SA, Monreal M, Krumholz HM, Konstantinides SV, Weitz JI, Lip GYH; Global COVID-19 Thrombosis Collaborative Group; Endorsed by the ISTH, NATF, ESVM, and the IUA; Supported by the ESC Working Group on Pulmonary Circulation and Right Ventricular Function. COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-Up: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75:2950-2973. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2178] [Cited by in RCA: 2190] [Article Influence: 438.0] [Reference Citation Analysis (0)] |
| 127. | Copetti R, Soldati G, Copetti P. Chest sonography: a useful tool to differentiate acute cardiogenic pulmonary edema from acute respiratory distress syndrome. Cardiovasc Ultrasound. 2008;6:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 362] [Cited by in RCA: 369] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 128. | Tusman G, Acosta CM, Costantini M. Ultrasonography for the assessment of lung recruitment maneuvers. Crit Ultrasound J. 2016;8:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 61] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 129. | Qian Z, Yang M, Li L, Chen Y. Ultrasound assessment of diaphragmatic dysfunction as a predictor of weaning outcome from mechanical ventilation: a systematic review and meta-analysis. BMJ Open. 2018;8:e021189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 54] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 130. | Wang XT, Ding X, Zhang HM, Chen H, Su LX, Liu DW; Chinese Critical Ultrasound Study Group (CCUSG). Lung ultrasound can be used to predict the potential of prone positioning and assess prognosis in patients with acute respiratory distress syndrome. Crit Care. 2016;20:385. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 65] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 131. | Duggan NM, Shokoohi H, Liteplo AS, Huang C, Goldsmith AJ. Best Practice Recommendations for Point-of-Care Lung Ultrasound in Patients with Suspected COVID-19. J Emerg Med. 2020;59:515-520. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 132. | Antúnez-Montes OY, Buonsenso D. Routine use of Point-of-Care lung ultrasound during the COVID-19 pandemic. Med Intensiva (Engl Ed). 2020;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 133. | World Federation for Ultrasound in Medicine and Biology Safety Committee (Jacques S Abramowicz; Iwaki Akiyama, David Evans, J Brian Fowlkes, Karel Marsal, Yusef Sayeed and Gail ter Haar), Abramowicz JS, Basseal JM. World Federation for Ultrasound in Medicine and Biology Position Statement: How to Perform a Safe Ultrasound Examination and Clean Equipment in the Context of COVID-19. Ultrasound Med Biol. 2020;46:1821-1826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 68] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 134. | Moore S, Gardiner E. Point of care and intensive care lung ultrasound: A reference guide for practitioners during COVID-19. Radiography (Lond). 2020;26:e297-e302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 135. | Sheth S, Fetzer D, Frates M, Needleman L, Middleton W, Jones J, Podrasky A, Mankowski Gettle L. Guidelines for Ultrasound in the Radiology Department During the COVID-19 Pandemic. Ultrasound Q. 2020;36:200-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 136. | Piscaglia F, Stefanini F, Cantisani V, Sidhu PS, Barr R, Berzigotti A, Chammas MC, Correas JM, Dietrich CF, Feinstein S, Huang P, Jenssen C, Kono Y, Kudo M, Liang P, Lyshchik A, Nolsøe C, Xie X, Tovoli F. Benefits, Open questions and Challenges of the use of Ultrasound in the COVID-19 pandemic era. The views of a panel of worldwide international experts. Ultraschall Med. 2020;41:228-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 137. | Vetrugno L, Bove T, Orso D, Barbariol F, Bassi F, Boero E, Ferrari G, Kong R. Our Italian experience using lung ultrasound for identification, grading and serial follow-up of severity of lung involvement for management of patients with COVID-19. Echocardiography. 2020;37:625-627. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 140] [Cited by in RCA: 153] [Article Influence: 30.6] [Reference Citation Analysis (0)] |
| 138. | Jackson K, Butler R, Aujayeb A. Lung ultrasound in the COVID-19 pandemic. Postgrad Med J. 2021;97:34-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 139. | Yau O, Gin K, Luong C, Jue J, Abolmaesumi P, Tsang M, Nair P, Tsang TSM. Point-of-care ultrasound in the COVID-19 era: A scoping review. Echocardiography. 2021;38:329-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 140. | Long JB, Ehrenfeld JM. The Role of Augmented Intelligence (AI) in Detecting and Preventing the Spread of Novel Coronavirus. J Med Syst. 2020;44:59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 141. | Li L, Qin L, Xu Z, Yin Y, Wang X, Kong B, Bai J, Lu Y, Fang Z, Song Q, Cao K, Liu D, Wang G, Xu Q, Fang X, Zhang S, Xia J. Using Artificial Intelligence to Detect COVID-19 and Community-acquired Pneumonia Based on Pulmonary CT: Evaluation of the Diagnostic Accuracy. Radiology. 2020;296:E65-E71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1260] [Cited by in RCA: 939] [Article Influence: 187.8] [Reference Citation Analysis (3)] |
| 142. | Shaikh F, Andersen MB, Sohail MR, Mulero F, Awan O, Dupont-Roettger D, Kubassova O, Dehmeshki J, Bisdas S. Current Landscape of Imaging and the Potential Role for Artificial Intelligence in the Management of COVID-19. Curr Probl Diagn Radiol. 2021;50:430-435. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 143. | Apostolopoulos ID, Mpesiana TA. Covid-19: automatic detection from X-ray images utilizing transfer learning with convolutional neural networks. Phys Eng Sci Med. 2020;43:635-640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 968] [Cited by in RCA: 848] [Article Influence: 169.6] [Reference Citation Analysis (0)] |
| 144. | Shi F, Wang J, Shi J, Wu Z, Wang Q, Tang Z, He K, Shi Y, Shen D. Review of Artificial Intelligence Techniques in Imaging Data Acquisition, Segmentation, and Diagnosis for COVID-19. IEEE Rev Biomed Eng. 2021;14:4-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 534] [Cited by in RCA: 526] [Article Influence: 131.5] [Reference Citation Analysis (0)] |
| 145. | Wang L, Lin ZQ, Wong A. COVID-Net: a tailored deep convolutional neural network design for detection of COVID-19 cases from chest X-ray images. Sci Rep. 2020;10:19549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1447] [Cited by in RCA: 955] [Article Influence: 191.0] [Reference Citation Analysis (0)] |
| 146. | Tang Z, Zhao W, Xie X, Zhong Z, Shi F, Ma T, Liu J, Shen D. Severity assessment of COVID-19 using CT image features and laboratory indices. Phys Med Biol. 2021;66:035015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 147. | Helms J, Kremer S, Merdji H, Clere-Jehl R, Schenck M, Kummerlen C, Collange O, Boulay C, Fafi-Kremer S, Ohana M, Anheim M, Meziani F. Neurologic Features in Severe SARS-CoV-2 Infection. N Engl J Med. 2020;382:2268-2270. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1741] [Cited by in RCA: 1894] [Article Influence: 378.8] [Reference Citation Analysis (0)] |