Peer-review started: October 26, 2020
First decision: November 16, 2020
Revised: November 20, 2020
Accepted: December 16, 2020
Article in press: December 16, 2020
Published online: January 28, 2021
Processing time: 93 Days and 3 Hours
Coronavirus disease-2019 (COVID-19), a respiratory tract infection caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has become a global health emergency and a threat the entire world. The COVID-19 shows a wide spectrum of clinical presentations, severity, and fatality rates. Although the fatal outcomes of the COVID-19 pandemic are evident in all age groups, the most devastating impact on the health consequences and death from COVID-19 are associated with older adults, especially older men. COVID-19 pandemic is affecting different countries in the world especially in the 65+ years age male group. In fact, several genes involved into the regulation of the immune system are strategically placed on the X-chromosome and trigger a gendered mediated antiviral fight. The aim of this study is to explore and exploit whether a relationship exists between male sex and COVID-19 mortality and the relationship is age dependent. Herein we discuss the possible role of physiological and immunological sex differences into the higher morbidity and mortality of SARS-CoV-2 between females and males. Deciphering gender differences in COVID-19 offers a window into the principles of immunity against SARS-CoV-2 infection and this information on ageing dependent gender disparity might contribute to our current understanding of COVID-19 infection and disease treatment.
Core Tip: (1) Older age, male sex and acute illness severity are associated with increased mortality risk; (2) Older age, underlying co-morbidities, social deprivation and ethnicity have been associated with worse outcomes from coronavirus disease-2019 (COVID-19); (3) Sex hormones might be implicated in the age-dependent and sex-specific severity of COVID-19; (4) Male sex hormones usually appear as immunosuppressants, whereas female sex hormones enhances the actions of humoral immunity; and (5) Female sex hormones exert a protective effect of COVID-19 severity on females through direct antiviral activity or immune-mediated mechanisms.
- Citation: Al-Bari MAA, Hossain S, Zahan MKE. Exploration of sex-specific and age-dependent COVID-19 fatality rate in Bangladesh population. World J Radiol 2021; 13(1): 1-18
- URL: https://www.wjgnet.com/1949-8470/full/v13/i1/1.htm
- DOI: https://dx.doi.org/10.4329/wjr.v13.i1.1
The world is facing a major public health crisis due to the epidemic of coronavirus infection named coronavirus disease-2019 (COVID-19) by the World Health Organization (WHO) caused by SARS-CoV-2 (amplified as severe acute respiratory syndrome coronavirus 2)[1,2]. SARS-CoV-2 infection epidemic originated from Wuhan city, Hubei, China, in December late 2019, has sporadically spread throughout the world. The SARS-CoV-2 that causes COVID-19 is a zoonotic pathogen, which can infect both human and animal. As of today, the 1 October 2020, WHO has reported that the epidemic has blown-out to more than 213 nations and areas with more than 33722075 confirmed cases, more than 1009270 confirmed expiries and more than 25492274 total salvages in around the world (https://covid19.who.int). Several millions of lives have been troubled due to compulsory isolations/quarantines. This epidemic has the power to overburden nationwide healthcare delivery systems and have main repercussions on international economy if SARS-CoV-2 proliferation and virulency power is not contained, or current treatments are not established. The infection is currently constituting a serious health, economic, social, and psychological effects on the whole world as the world is under lock down as a measure to curb the spread of the virus[3]. SARS-CoV-2 is primarily transmitted from person to person through respiratory airborne droplets produced when infected persons cough, sneeze, breathe deeply, or talk within a proximity to uninfected persons. With this emerging combat against this life-threatening virus, the WHO has taken several strategies to interrupt human contacts with others, segregate patients at preliminary stages, recognize and decrease spread from the animal source for minimizing the social and economic impact.
Coronaviruses belong to the family of Coronaviridae. SARS-CoV-2 is a beta-coronavirus like the two other viruses that have caused fatal infections over the last couple of decades: The SARS-CoV and the MERS-CoV (amplified as Middle East respiratory syndrome coronavirus). The SARS-CoV-2 is a non-segmented, enveloped, single-stranded, positive-sense RNA virus with a nucleocapsid. Analysis of the viral full genome sequencing has shown that the SARS-CoV-2 is phylogenetically close to the causative agent of a viral outbreak in 2002, SARS-CoV, with which it shares about 79% of its genome[1,2]. Since SARS-CoV-2 is hereditarily and anatomically related to SARS-CoV, it is appearing clear that it has its own exceptional properties that shared to the quick outspread around the world. Despite its similarity to SARS-CoV, its transmission efficiency and diagnostic methods are rather different. The coronavirus crown-like (“corona”) morphology is created by transmembrane spike glycoproteins (S proteins) which is essential for SARS-CoV-2 attachment and invasion into host cells via formation of homotrimers protruding from the viral surface[3]. The distinguishing factor of SARS-CoV-2 is probably the nucleotide changes in the S protein and its receptor-binding domain (RBD)[4]. The S proteins of SARS-CoV and SARS-CoV-2 show organizational homology and preserved ectodomains, so that previous approaches are applied to stop binding of SARS-CoV to its host cell receptor, angiotensin-converting enzyme 2 (ACE2) through a non-pH dependent endocytosis, since SARS-CoV-2 also employs ACE2 for cell entry[5,6]. In molecular modelling analysis, it has shown similarities between the RBDs of SARS-CoV and SARS-CoV-2 (also called S proteins), which are the most immunogenic part of the virus and probably bind the same ACE2 receptors in order to gain cell entry[7,8], thus suggesting that a similar pathogenic mechanism is involved in both viral infections. Interestingly, ACE2 receptors are not only expressed on alveolar epithelial type II cells, which represent 83% of all ACE2-expressing cells, but also on heart, kidney, endothelium, and gut cells[9]. Thus, ACE2 may create a therapeutical target to control the cell entry of SARS-CoV-2. For example, the clinically used antimalarial drugs chloroquine analogues such as hydroxy-chloroquine have been found to prevent terminal phosphorylation of ACE2 and to raise the pH in lysosomes. Moreover, the glycosylated S protein of SARS-CoV-2 is extremely immune-sensitive to the host, and murine polyclonal antibodies against S protein of SARS-Co-V effectively hinder S-mediated cell entry of SARS-CoV-2, suggesting that cross-neutralizing antibodies targeting preserved S epitopes can be provoked upon immunization[10].
Although SARS symptoms appear with MERS, and COVID-19, the assessed fatality rate of COVID-19 (2.3%) is considerably lesser than SARS (11.0%) and MERS (34.0%)[11,12]. In comparison with SARS and MERS, COVID-19 has outspread very quickly, possibly due to expanded globalization and modification of the virus in closely each environment[12,13]. Although SARS-CoV-2 is less lethal than SARS and MERS-CoV insofar as most patients affected with SARS-CoV-2 may progress from the asymptomatic state or to acute respiratory distress syndrome (ARDS) and septic shock in severe form of the disease. In major cases, coronavirus infected patients show a mild flu-like symptoms, in which the utmost general signs are fever and cough. However, a major portion of the patients (15.7%) who develop severe disease have increased difficulty in breathing because of pneumonia. However, COVID-19 may rapidly develop into SARS characterized by interstitial pneumonia and the rapid development of ARDS or septic shock in older people (> 60 year, up to 10%-20%), especially in those with underlying medical comorbidities, such as hypertension, diabetes, and pulmonary diseases[1,14]. It more interesting that female adults are excluded in the danger group, as small number cases of serious COVID-19 in female have been testified. This takes up questions concerning the molecular mechanisms of gender disparity linked to the COVID-19 sternness.
In some patients the SARS-CoV-2 may associated with terrible symptoms when it infects the lungs initiating a strong inflammatory response, a cytokine storm with extreme levels of acute-phase reactants[15,16]. This hyperinflammatory situation is categorized by increased levels of cytokines, including interleukin-6 (IL-6), monocyte chemoattractant protein 1 and granulocyte-colony stimulating factor as well as appeared with the macrophage activation syndrome like hyperferritinaemia. Here, we report the current understanding of SARS-CoV-2 such as its sociodemographic characteristics included age, sex, smoking, race/ethnicity and level of education as well as its clinical features, imparting the critical information for regulating our responses against the SARS-CoV-2 contagion. We also recapitulate the state-of-the-art inventions on targeting SARS-CoV-2 through a cellular point of interpretation. Understanding and elucidating of cellular and molecular mechanisms of gender disparity associated with the severity of COVID-19 may significantly advance our knowledge of the disease pathogenity, and thus provide to the health professionals as to how to well treat the ageing patients.
Objectives: The recent COVID-19 pandemic has appeared as a threat to global health. Though current evidence on the epidemiology of the disease is emerging, very little is known about the predictors of recovery. The current objective of the report is to describe the epidemiology of confirmed COVID-19 patients in the United States and Bangladesh and identify predictors of recovery. Data source: We have collected these data by using publicly available data for confirmed cases in the Coronavirus Disease 2019 (COVID-19)-Associated Hospitalization Surveillance Network (COVID-NET) from the Centers for Disease Control and Prevention (CDC), United States from March 07, 2020, to September 19, 2020 (https://gis.cdc.gov/grasp/COVIDNet/COVID19_3.html), and (https://gis.cdc.gov/grasp/COVIDNet/COVID19_5.html) as well as press release under Ministry of Health and Family Welfare (MOH&FW), Bangladesh (https://corona.gov.bd/press-release). Variables: We have undertaken descriptive analyses of cases stratified by sex, age group, demographic information (e.g., race, ethnicity) and clinical (medical) history (underlying health conditions). Statistical methods: Correlation analysis is performed among all predictors (sex, age group, race and ethnicity) with student’s t-test, statistical analysis accordingly.
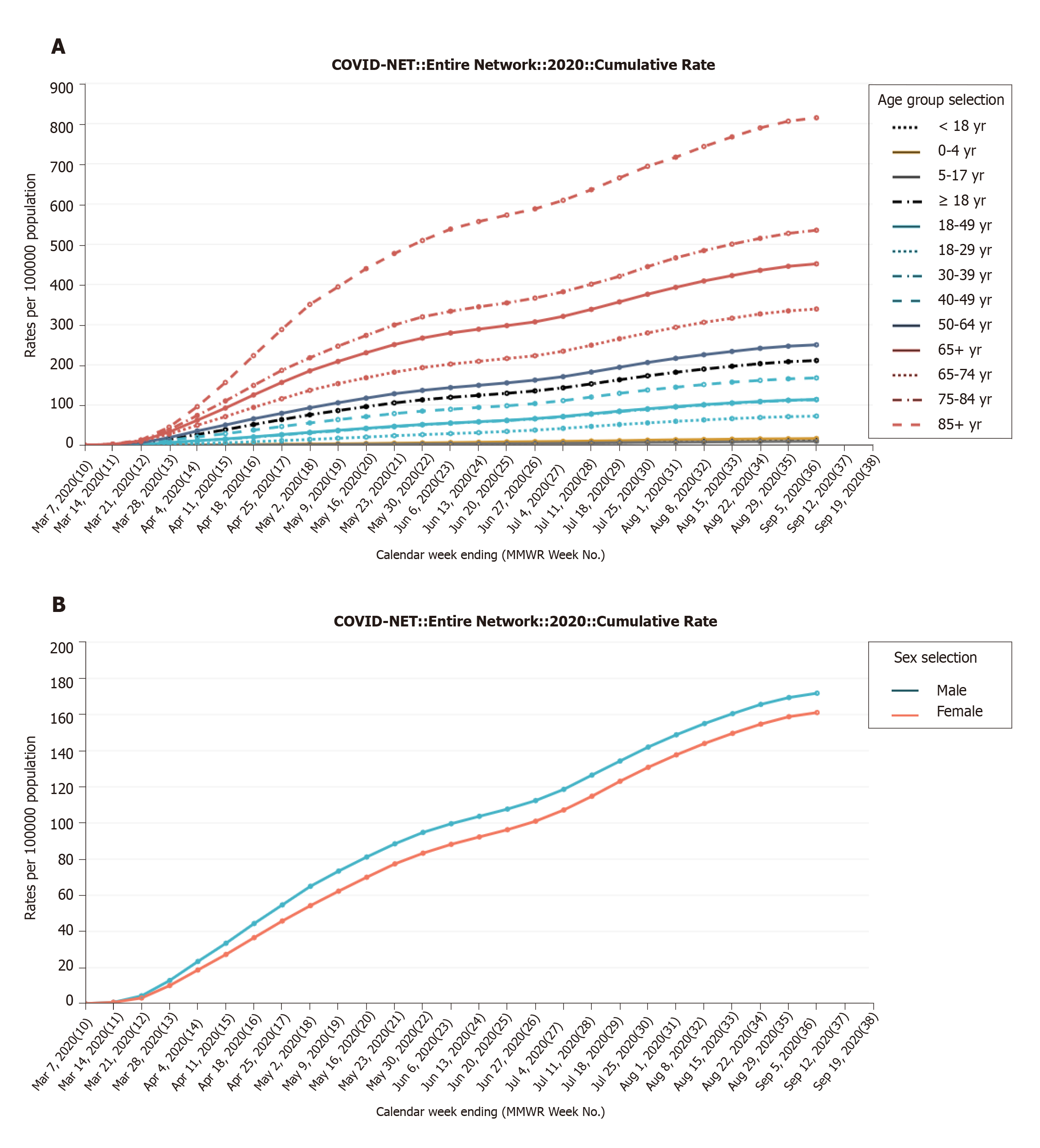
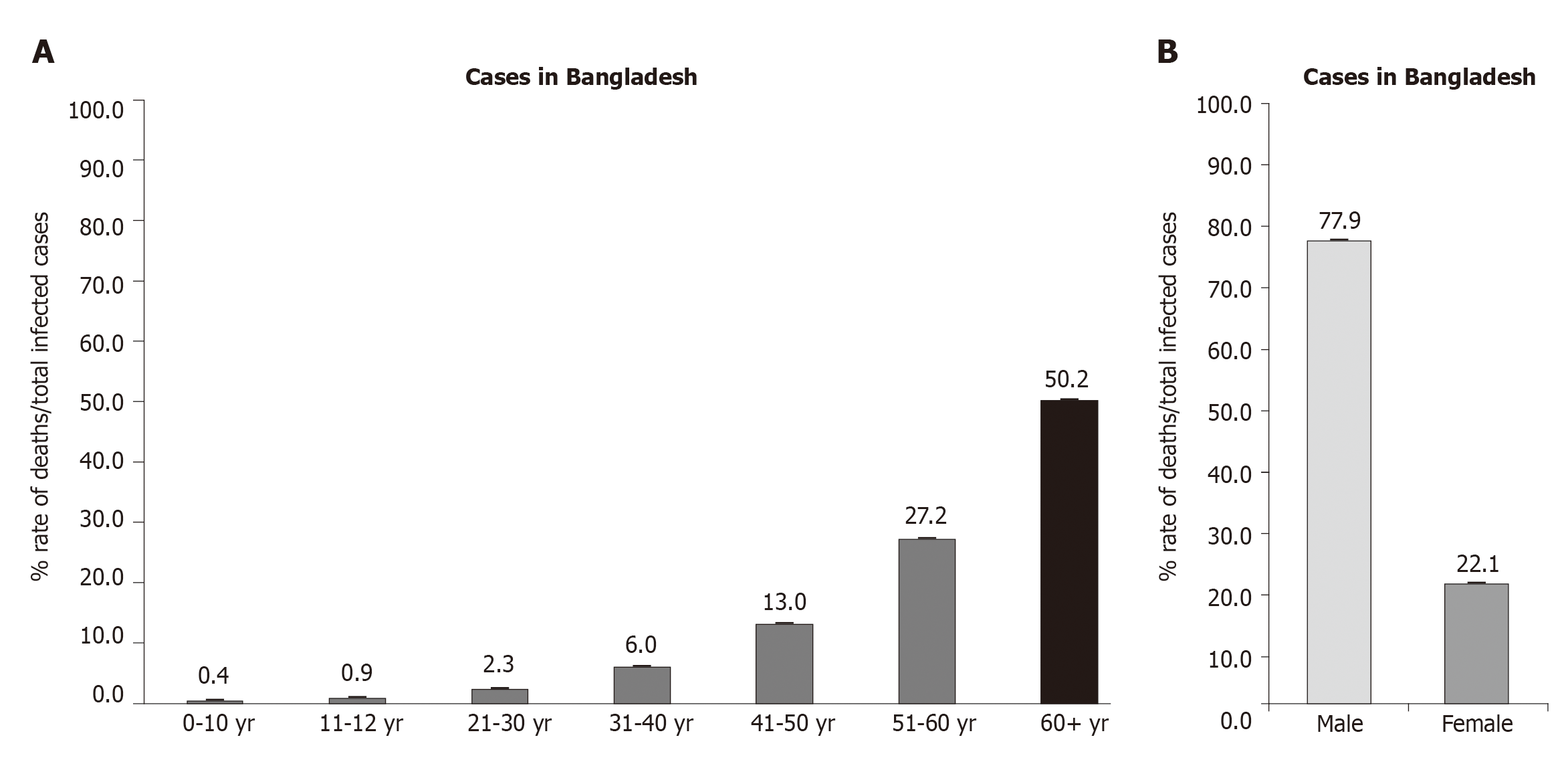
As shown in Figure 1A, the first case of COVID-19 in United States is confirmed on March 7, 2020. There are a small number cases of new infections for about a month. After two months, the figure abruptly has risen at May 30, 2020 (cumulative rate 5.9, 3, 51.6, 136.5 and 266.6 per 100000 population as 0 to 4 year, 5 to 17 year, 18 to 49 year, 50 to 64 year and 65+ year respectively), to reach the peak around end of June and early July (9.1, 4.3, 66.3, 162.0 and 306.9 per 100000 population as 0 to 4 year, 5 to 17 year, 18 to 49 year, 50 to 64 year and 65+ year respectively). It reached continually its peak on the 5 September with 16.8, 9.7, 113.8, 249.8 and 451.2 per 100000 population as 0 to 4 year, 5 to 17 year, 18 to 49 year, 50 to 64 year and 65+ year respectively confirmed cases. Similar case is found in Bangladesh that the rate of death per total infected cases (50.2%) is found in over 60-year-old patients (Figure 2A). The United States’ data indicate that mortality rate among younger age group patients with mildly disease is less prominent. This result is consistent with other report that younger patients less than 17 years have slighter COVID-19 severity, with practically no hospitalizations or expiries stated[17]. However, the mortality is higher among elderly patients particularly 65+ years old that is required for intensive care unit admission in hospital. These results are similar to the other reports that the elderly people (aged over 60) were at a high risk of developing into death based on a worldwide data (www.who.int)[17-19].
As in Figure 1B shows on United States data that on May 30, 2020 the curve shows that cumulative rate 94.8 and 83.3 per 100000 population as male and female respectively (adjusted ratio 1.134:1) and gradually reach the peak around end of June and early July (112.4 and 101 per 100000 population as male and female respectively (adjusted ratio 1.11:1). It reached continually its peak on the 5 September with 171.8 and 161.1 per 100000 population as male and female respectively confirmed cases (1.065:1). Interestingly, the prominent data is found in Bangladesh that the rate of death per total infected cases (77.9%) is found in male patients over female patients (22.1%) (Figure 2B). As shown in Figure 1B both adult males and females had similar recovery rates, and their difference is not statistically significant. However, in case of Bangladesh the rate of death in male patients is strongly statistically significant (P value < 0.0001). Regarding the sex proportion, there is an apparently indisputable outline that COVID-19 killed more males than females (Box 1). Unlike the fewer statement in the research from the Asian subcontinental areas such as China, South Korea, the data from the United States reflect the male sex is in danger for disease severity[20-22]. To assess the over-all situation about the world, the country-wise data[23], have found that the case-mortality rate among men is about 35% more inflated than women. The sex-disparity is consistent across age groups and regions.
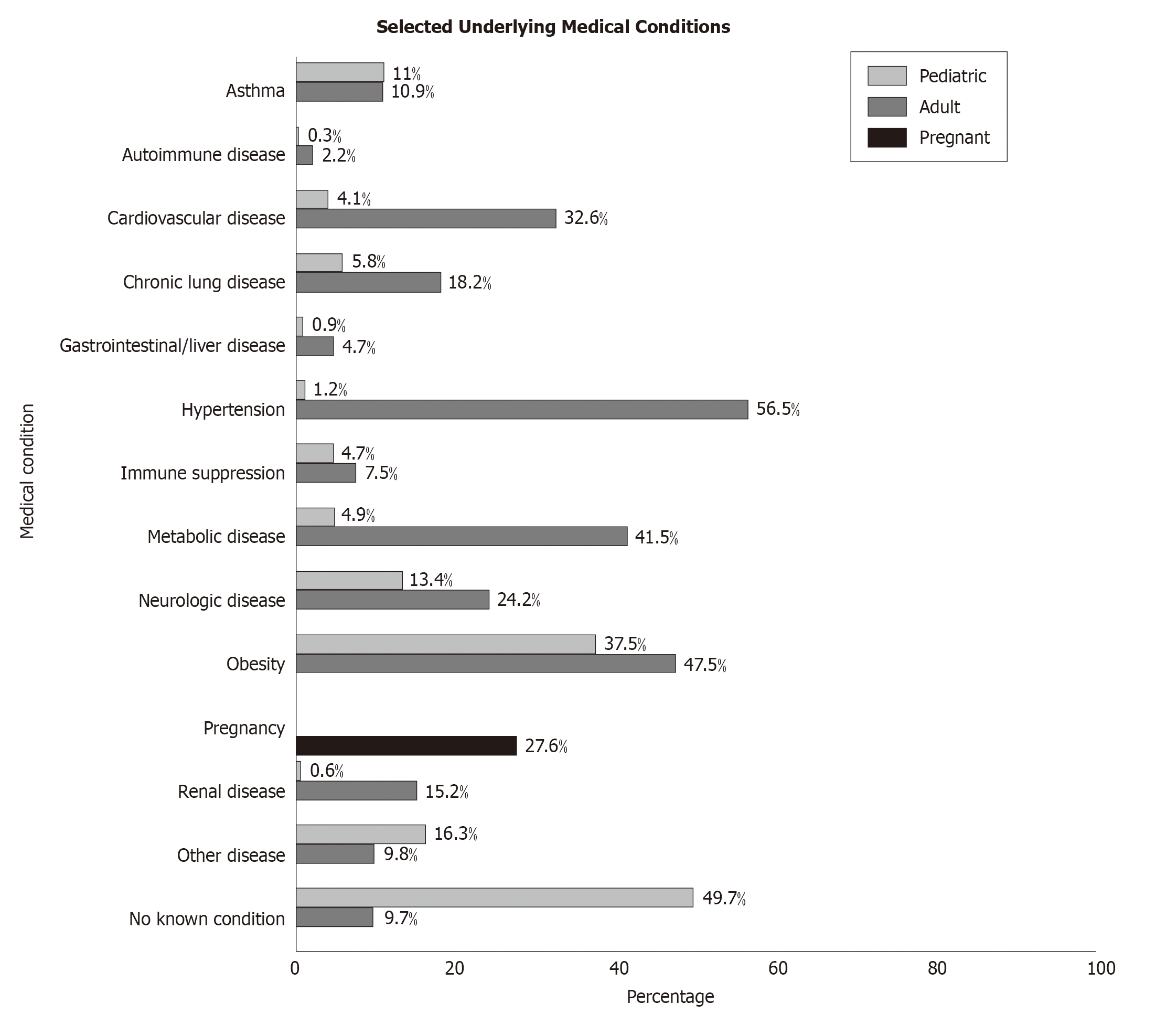
Findings from multiple reports also show that patients who are more than 65 years of age particularly male sex having a higher BMI value (> 35 kg/m2), co-morbidities such as hypertension, cardiovascular disease (CVD), chronic lung disease, metabolic disease, neurological disease, obesity, renal disease, diabetes, coronary disease, obstructive pulmonary disease, nicotine dependence, and heart failure have vital risk factors for developing COVID-19 complications[24,25] and a high mortality rate[26,27]. Among them, obesity is a critical risk factor which aggravates the COVID-19[28]. In consistent with these views, Figure 3 shows that adult patients are susceptible for COVID-19 having the following serious complications such as CVD (32.6%), chronic lung disease (18.2%), hypertension (56.5%), metabolic disease (41.5%), neurological disease (24.2%), obesity (47.5%) and renal disease (15.2%). In the paediatric cases, the percentages of the infection cases are quite less than the adults. However, in major cases paediatrics are infected with COVID-19 in unknown conditions (49.7%). Delayed hospitalization and microbial infections are also proposed greater danger factors for disease development[27]. Smoking history is also a probable danger issue for emerging severe complications[5].
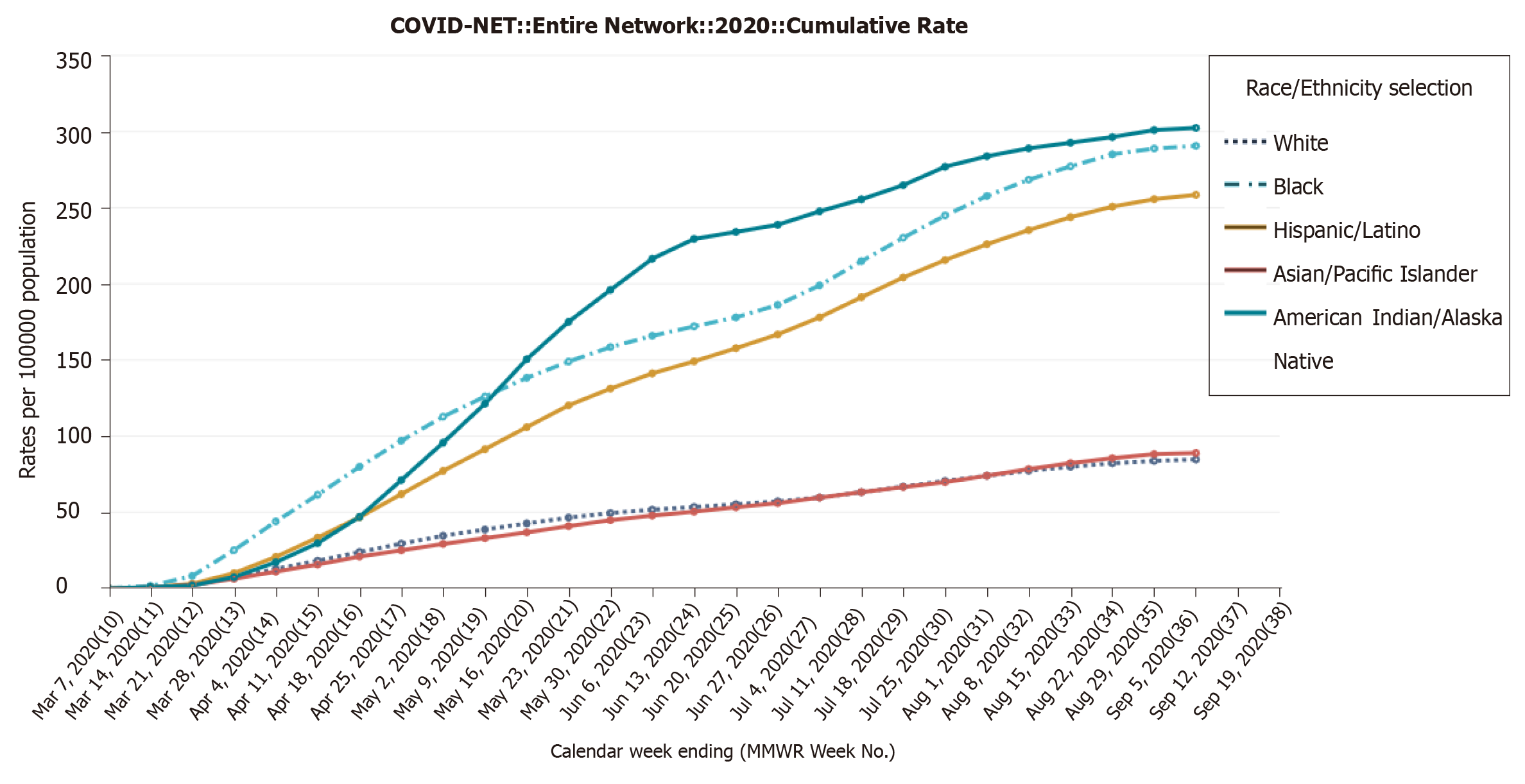
Baseline patient characteristics are also provided in Figure 4. Black patients are generally more susceptible than white patients with the age group (65+ years old. On May 30, 2020, the cumulative rates are 49.5, 158.5 and 196 per 100000 population as white, black and American Indian/Alaska Native respectively, to reach the peak around end of June and early July (57, 186.2 and 238.8 per 100000 population as white, black and American Indian/Alaska Native respectively). It reached continually its peak on the 5 September with 84.7, 290.6 and 302.4 per 100000 population as white, black and American Indian/Alaska Native respectively confirmed cases. In another report, black patients have a relative risk for hospitalization[29]. After correcting for gender, stage group, and comorbidities, black people have a 1.42 times higher danger of hospitalization for COVID-19 severity in comparison with white patients. The relative danger of death from COVID-19 infection is increased for males than for females in almost all age groups in all nations.
The COVID-19 pandemic is causing millions of deaths worldwide and it has become as an emerging threat to the public health globally. Although existing evidence on the epidemiology of the COVID-19 is emergent, a slight is identified about the predictors of salvage. Many countries throughout the world have experienced an unprecedented healthcare crisis caused by the SARS-CoV-2 infection[30,31]. Many parameters likely contribute to the etiology of the COVID-19 disease. The viral population and way of infection can elucidate why healthcare workforces are at a greater danger; diversities in the genome sequences of the viruses or the genome of the host-cells (i.e., patient’s genetic makeup) may consider for the variables detected among different countries and people. At the person level, personal immunity is also a vital forecaster of the disease prognosis, which can be reshuffled by age levels, gender, race, ethnicity as well as the presence of co-morbidities. Gender- and sex-determinants are also important for advising the endemic in interstellar and over time. To exemplify the status of this opinion, data on gender of the COVID-19 deaths in the United States and Bangladesh, recorded until 5th September 2020 were used to evaluate age- and sex-standardized figures in the United States and Bangladesh.
In comparison with disease occurrence, approximately similar distribution is detected among males and females at different age groups according to the WHO case-based surveillance system as of April 18, 2020[32]. However, from data on today in COVID-19, not only the progression of disease severity, but also mortality and fatality rates necessity to be clarified by age and, in addition by sex[33]. Preliminary data suggest that selective persons such as the elderly, males and people with comorbidities, including hypertension, diabetes and obesity, have slight COVID-19 consequences[34,35]. As the pandemic outspread over the United States during the last 4 mo, patterns of high-danger properties explained to emerge and data of poor consequences (specifically high case fatality) among racial and ethnic minorities[34].
Evidences suggest that male gender and aged persons are key factors connected to higher danger of severe events and death from COVID-19[36,37]. The enhancing mortality rate from COVID-19 for males (2.4 times) than for females is overall comparable to that originated in other coronaviruses during the past two years, including the SARS-CoV and the MERS-CoV[37-39]. The explanations for the sex differences in COVID-19 are perhaps multifaceted including variations in immune response, higher incidence of pre-existing disease, biological differences between the sexes such as high levels of androgens in men, differences in lifestyle such as smoking habits as well as differences in underlying comorbidities[40-42]. Male are commonly reported to have higher serious pathological conditions, such as CVDs, whereas females tend to have higher non-serious long-lasting disorders, such as skeletal and autoimmune hypersensitive diseases[43]. Thus, the risk of male death from COVID-19 may explain the comparatively more occurrence of causal comorbidities such as CVD, diabetes and chronic lung disease[44].
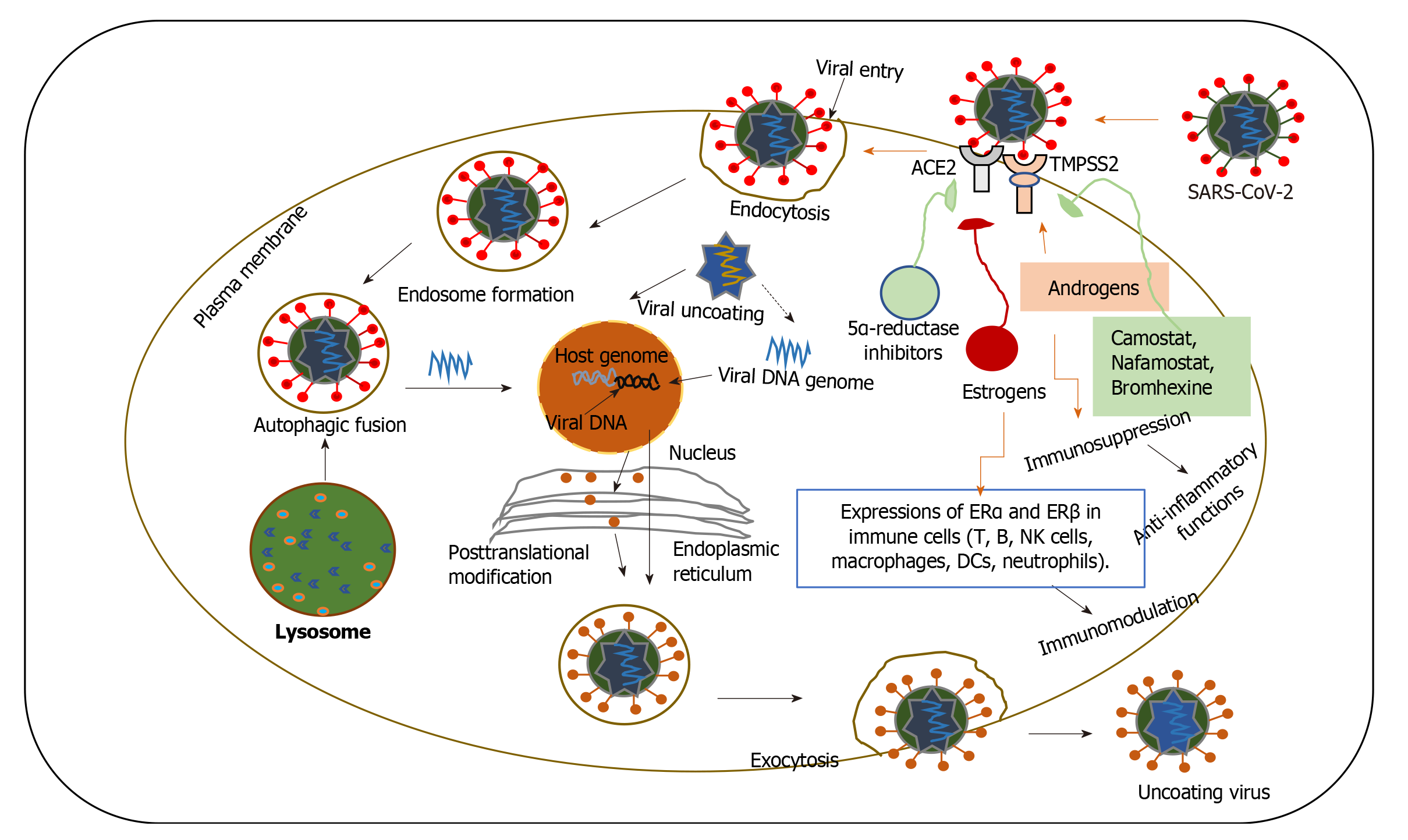
Mechanistically the age and gender differences in COVID-19 can be explained by the variable expression of an extracellular anchor represented by a cell-surface zinc peptidase, ACE2 which mediates SARS-CoV-2 binding and entry into cells[45,46]. Here the viral spike (S) protein is indeed a key determinant for transmissibility. Although ACE2 is pivotal for the entry point of the SARS-CoV-2, CD26 receptor also interacts with the S1 domain of the viral S protein and affects its virulence[47-49]. Since ACE2 receptor is abundantly expressed by pneumocytes in the lungs[48], SARS-CoV-2 infection and down-regulated ACE2 lead to higher the expression of angiotensin II (Ang II) that directly causes unregulated inflammatory lung damage[47-49]. Interestingly, ACE2 expression does not denote a completely capable of cell entry receptor as confirmed for SARS-CoV-2, until the cleavage at the S1/S2 and the S2′ site of the S protein operated by TMPRSS2 a 70 kDa membrane-anchored enzyme (type 2 transmembrane serine protease) in order to allow viral-cellular membrane fusion[50]. ACE2 is commonly accountable for altering Ang II into vasodilatory and low immune enhancing variants of angiotensin. Ang II specifically interacts with its type 1 receptors called angiotensin receptors (AT1Rs) in the lung to stimulate inflammation and vasoconstriction via induction of the NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) pathway, which enhances cytokine synthesis[6,51]. Low expression of ACE-2 levels and high Ang II expressions turn to enhance the permeability of pulmonary vessels which then consequences in inflammatory damage to the pulmonary tissues[52,53]. The primary culprit of severe COVID-19 is the cytokine storm resulting from an unchecked inflammatory response that damages the lung tissue and causing death in a substantial percentage of cases[54]. In the lungs, ACE2 down-regulation associates with the human ARDS via enhanced vascular permeability, increased lung oedema, neutrophil accumulation and worsened lung function[51,55,56]. Moreover, if SARS-CoV-2 causes sepsis, then ARDS occurrence exaggerates the edema, swelling and can cause of death[57]. Additionally, when COVID-19 infection occurs, the virus SARS-CoV-2 is internalized and stimulates TNF-α converting enzyme, ADAM17 (ADAM metallopeptidase domain 17)[58]. ADAM17 slashes the ACE2 receptors resulting them insensitive to the stimulation of renin–angiotensin–aldosterone system. This is eventually accountable for additional making of cytokines, which worsen the inflammation[59]. In the existence of pre-existing CVD, the cytokine storm can intensify underscoring diseases by infuriating pre-existing heart failure, causing suppression of myocardial activity, enhancing the oxygen demand/supply ratio and endothelial disfunction[59,60]. In this setting, ACE2 could denote the first variable to validate different effects of the infection between sexes.
ACE2 gene is located on the X chromosome (Xp22.2), in the Barr zone. The X chromosome in females (XX genotype) bring twofold as many X-linked genes (> 1000 genes) related to males (XY genotype). The X-linked gene expressions are equivalent between two sexes via X chromosome inactivation (XCI) process which transcriptionally deactivates one copy of the X chromosome. XCI is recognized during embryonic development and regularly preserved throughout the life[61,62]. However, a part of X-codified genes (almost 15%-23%) can discharge, fully or partly, from XCI and this privilege is suitable for those genes located in the pseudoautosomal regions (PAR) 1 and 2[32,63]. The ACE2 gene is located within PAR1 and the influence may not inevitably be an increased expression of ACE2 in women. Male susceptibility to COVID-19 infection may be additional boosted by X-linked inheritance of genetic pleomorphisms as loci of both androgen receptors (ARs) and ACE2 genes are positioned on the X chromosome[32]. Since ACE2 expression is originated in the testes (specially in Leydig cells)[5,64] serum luteinizing hormone (LH) level is significantly increased. As a result, the proportions of testosterone to LH and follicle stimulating hormone (FSH) to LH are pointedly diminished in males with COVID-19[5]. Thus, it is inevitability to evaluate gonadal role among patients who have improved from the SARS-CoV-2 infection, particularly in reproductive-aged men.
Another exciting finding related to coronaviruses resides on the co-expression of TMPRSS2 together with ACE2. TMPRSS2 is a critical factor in enabling cellular infection by SARS-CoV-2 for priming the viral S protein S1 domain and employing the S2 domain for viral infectivity[50,65,66]. Several speculations may strengthen the role of sex into the expression of TMPRSS2. TMPRSS2 is located on chromosome 21q22.3 and several AR elements are positioned upstream of the transcriptional promoter region[67,68]. Notably, AR activity seems to be required for the transcription of the TMPRSS2[5,69]. It is hypothesized that genetic variation of AR is associated with prostate cancer and androgenetic alopecia is also related to ethnic disparities in COVID-19 death[70]. Androgens powerfully upregulate the TMPRSS2 expression in prostate cancer cells[31,71] and they can also regulate the oncogenic ERG transcription factor (or, more rarely, other members of the ETS family) when the TMPRSS2:ERG fusion gene is formed due to somatic gene reshuffles in prostate cancers[32].
Sex hormones might be implicated in the age-dependent and sex-specific severity of COVID-19. Sex hormones, e.g., testosterone and oestrogen significantly affect immune responses in both sexes[36,72,73], a part of which are in straight connections between sex hormones and immune cells. Increasing evidence proposes that both sex hormones and hormone therapy could be beneficial in COVID-19 treatment through direct modulation of antiviral activity or immune regulation[32]. Several studies suggest that both high and low testosterone levels can favour severe COVID-19[32,74]. For example, high testosterone levels upregulate TMPRSS2, facilitating the entry of SARS-CoV-2 into host cells via ACE2 (Figure 5). A recent analysis supports the hypothesis that androgen-deprivation therapy (ADT) might protect men from SARS-CoV-2 infection[32,75]. An epidemiological data also provision that ADT provide a defensive role in COVID-19 patients with prostate cancer. A mode of clarification for this concept is connected to the viral entry facilitated by TMPRSS2[32,75]. Furthermore, upregulated testosterone expressions can also impart to the progress of microthrombi and venous thromboembolism, which are signs of severe COVID-19 patients[76]. In addition, the 5α-reductase (a well-known converting enzyme to testosterone) inhibitors (dutasteride) can be applicable in COVID-19, by suppressing the ACE2 expressions and the internalization of the spike receptor[32]. Contrarywise, other studies propose that the immune modifying properties of androgens can defend from the non-satisfactory cytokine storm of COVID-19. Preclinical data also recommend that camostat mesylate, which hinders the protease action of TMPRSS2, is able to hinder the entry of SARS-CoV-2 in lung epithelial cells[50]. Preclinical data showed that inhibitors of TMPRSS2 (such as camostat, nafamostat and bromhexine) and of 5α-reductase might be active against SARS-CoV-2[32,50]. Although the androgen-driven concept is fascinating, it remains obscure why younger males with COVID-19, who have greater testosterone levels in comparison to adult males, display diminished sternness and fatality rates[77]. Likewise, it would be unpredicted that aged males who have lesser testosterone levels display amplified sternness and fatality rates to COVID-19. Obesity is a well-known risk factor for CVDs and testosterone levels of obese males are reported to be distinctly lesser than in the non-obese people. Remarkably, the amount of dropping testosterone levels is interrelated to blood glucose levels and lipid profiles[78]. By inclining to obesity, lowered levels of male sex hormones, specifically testosterone, can possibly be involved in the advance of CVDs and COVID-19. Additional experimental and clinical studies are vital to categorize the underlying associations among testosterone levels, obesity and CVDs, and the basic mechanisms. Thus, it is vital to evaluate why-among males with COVID-19-younger age is powerfully defensive against adverse consequences. It is probable that testosterone has a defensive anti-inflammatory action in younger males.
Testosterone is reported to have anti-inflammatory functions via suppression of both the cellular and humoral immune systems[52,79]. Testosterone is reported to decrease IL-6 and tumor necrosis factor α (TNF-α) levels via suppression of the NF-κB pathway. Down-regulated testosterone expression, as can happen in aging males, has also been correlated with upregulated inflammatory cytokines including IL-6 and may trigger to high risk of pulmonary injury after pneumonia[52]. Androgens usually inhibit the inflammatory signals by reducing the action of the peripheral blood mononuclear cells, and the secretion of inflammatory factors and cytokines, such as IL-1β, IL-2, TNF-α[32,41]. Androgens may also endorse the release of inflammatory cytokines such as IL-10 and TGFβ (transforming growth factor-β) via AR signaling pathway[32]. These immune- oppressive actions of androgens could induce COVID-19 infection, but might also suppress the cytokine storm that exemplifies with the most COVID-19 severity.
For the most severe infections, females have been constantly found to stand a greater immune reply than do males. Generally, the women show more immune responses effectively to microorganisms by making greater quantities of interferons (IFN) and antibodies; though this defensive action mediated mainly by estrogen, is reduced in postmenopausal females[52]. In cases of coronaviruses, females have verified a steady survival benefit over males[52]. A large amount of authentication suggests that female sex hormones, particularly estrogens and progesterone might apply a protecting role on women via direct antiviral action or immune-protective effects, thus elucidating the greater COVID-19 sternness in post-menopausal females. For instance, expressions of estrogen receptors (abbreviated as ERα and ERβ) occur in a wide variety of immune cells (T, B, NK cells, DCs, macrophages, neutrophils). Additionally, sex hormones are proposed to provide dose-dependent action on immune cells[41,80]. Hereafter, age-related changes or menstrual-cycle dependent variations in the female sex hormone levels can affect the collaboration between sex hormones and immune cells. Unexpectedly, it is found that immune responses between both sexes deviate as age upsurges, although the hormonal levels lower with ages[80]. Estrogens can downregulate the expression of ACE2 mRNA in bronchial epithelial cells in vitro[81]. Beyond this mechanism, estrogens have also a potential favorable role related to their immune-modulating properties. Notably, testosterone can be transformed to estrogen in peripheral tissues via aromatase enzyme, which may provide an anti-inflammatory action. This observation suggests that estrogens can protect females from severe COVID-19 compared to men and that post-menopausal women[81]. Although estrogen has a multifaceted role in modifying the immune system, it is stated to have an anti-inflammatory action at regular biological levels in premenopausal females[82]. In general, inflammatory cytokines, such as IL-6, IL-8 and TNF-α are suppressed by periovulatory doses of estrogen, although minimal estradiol levels can enhance inflammatory factors, which can clarify the proinflammatory states suffered by postmenopausal women. Although postmenopausal women are described to have greater expressions of proinflammatory cytokines including IL-6; these cytokine expressions are suppressed by the application of hormone replacement therapy (HRT)[83]. Therefore, the NF-κB pathway activated by Ang II enhances cytokine production after SARS infection while the NF-κB pathway can be shut down by estrogen and this strategy might be relevant for COVID-19 treatment in female patients.
Progesterone and 17β-estradiol (E2) have distinct roles in modulating innate and adaptive immunity[72] based on concentration[81]. Low concentrations of E2 promote pro-inflammatory cytokine production and stimulate TH1 (T helper type 1) cells, whereas highly concentrated E2 suppresses cytokine secretion and enhances TH2-cell mediated humoral immunity (Figure 5). In general, progesterone stimulates anti-inflammatory effects and can indulge the CD4+ T skewness from TH1-cells to TH2-cell actions[84]. It has been suggested that a triggered TH2-cell mediated immune response to such as in patients with asthma, might protect against severe COVID-19[85]. Finally, current data propose that progesterone provides a straight antiviral action on SARS-CoV-2 via the modification of the Sigma receptors[86]. Moreover, MERS-CoV and SARS-CoV in vivo data also support that SERMs (selective estrogen receptor modulators) such tamoxifen and toremifene, may be applicable against COVID-19[87], although emphasizing the necessity of more investigations in patient treatment.
The complex variability of immune responses based on age and sex may also elucidate the age-dependent and sex-selective sternness of COVID-19[54,88]. Our immune system is composed of two distinct arms with different functions: Adaptive and innate immunity. The first line of defence, innate immunity acts against dangerous invaders like SARS-CoV-2 via capturing and deactivating pathogenic organisms and initiating inflammation. Classically, acute inflammatory responses lead to a quick accumulation of immune cells and macromolecules at the injurious sites for eliminating the aggressor. However, chronic inflammatory responses can lengthen to affect abundant cellular machineries. Aging phenomena have been correlated with such chronic stimulation of inborn immunity, linked to systemic strengthen in inflammation (called as “inflamm-aging”) that might be harmful for the body[89]. The cellular senescence modulates the pathogen clearance during infections, and this mode of action might impart to clarify the age-dependent COVID-19 severity[72]. Additionally, discrete immune responses are confirmed between the sexes, and can consequence in disparity occurrence and vulnerability of males and females to autoimmune diseases, tumours and infections[72]. Acquired immune cells are militarized when the inborn immunity is inadequate to defeat a hazard. Cell mediated immunity specifically B and T cells can eradicate a danger precisely by selectively binding with a certain threat (for example, a small fragment of protein or a part of antigen to SARS-CoV-2). In addition to chronic activation of innate immunity, adaptive immune functions decline with age[90].
The X chromosome of Drosophila melanogaster docks many genes encoding for innate signalling proteins. This can provide a probable clarification for the sex-specific differences into immunity against viral infections. However, Y chromosome encoded Sry expression decrease the immune response. It is supported that X chromosome is partly accountable for the over-active respondents of the female immunity. Hence the high incidences of auto-immune diseases may occur in women by contributing to the collapse of self-tolerance[91]. Moreover, the giant X chromosome comprises the greatest number of immune-correlated genes in the full genome[92], including genes that are involved in innate [e.g., PRRs (pattern recognition receptors), TLR7 and TLR8 and acquired immune responses (e.g., chemokine receptor CXCR3). Although inactivation of X chromosome has may preserve correspondent gene expression into the two sexes, a lower number of genes located in the intron regions can escape this mode. Therefore, the products of these genes are exposed in females and the PRRs, TLR7 and TLR8 are escaped from XCI region[73]. Upon ligand interaction, TLR7 dimerizes and activates MyD88 (myeloid differentiation primary response gene 88), MAPK (mitogen-activated protein kinase) cascades, NF-κB pathway as well as IRF (IFN regulatory factor) -7 and IRF-5 activation[93]. In humans, mRNA levels for IRF-5 associate with oestrogen receptor 1 (ER1) levels proposing a possible IRF-5 regulation by transcriptional ER1 level[94]. Besides, IL-6 has been claimed to be critically involved into the down-regulated host immune response of COVID-19 patients[95]. Finally, TLR7 may stimulate B cells to enhanced antibody production.
A current study supports that females with severe COVID-19 cases have a greater amount of serum SARS-CoV-2 IgG in comparison with males, and the production of IgG in the initial phases of contagion looks like to be vigorous in women than in men[96]. It is also discovered distinct sex variances in how the B cell change with age[80]. B cells (numbers and percentages) are lower in older men (> 65+ years)[97] supporting that some of these sex-variances are preserved transversely people. Reduction number of B cells in aged men might consequence in reduction of antibody supply that might weaken the ability of an individual to fight against infectious pathogens. A pilot study suggests that injection of plasma therapy from recovered patients that comprises antibodies are capable to counteract SARS-CoV-2 virus pointedly and upgraded the critically ill COVID-19 patients[98]. But, a biosafety issue is a spectacle called antibody-dependent enhancement (ADE), when non-counteracting antiviral antibodies initiate the entry into host cells thereby cumulative the SARS-CoV-2 infectivity[98].
It is also found accelerated age-related T cell function declines in men compared to women[80]. For example, incidences of naive T cells reduced with age, principally in CD8+ T cells in both sexes, although females had greater naive T cells in comparison with men in both young and aged persons[99]. Females have been observed to have higher thymic action in comparison with males in all ages[100], which may likely clarify sex-variances in naive T cells. Lymphocytopenia has been reported in severe cases of COVID-19[101] including severe decays in CD4+ and CD8+ T cells. Collected these data support that SARS-CoV-2 may weaken antiviral immunity pointedly and this weakening may have drastic outcomes for aged persons.
Irreparable process, human aging causes decrease in cognitive ability with the increase in age. There are many factors accelerating a person’s biological age such as diet, exercise, lifestyle and co-morbidities (hypertension, diabetes, obesity). With aging, changes in hematopoietic stem cell (HSC) pool contribute to the functional decline in both innate and adaptive immune systems. Somatic mutations in HSCs is more commonly found in aged persons, where consequence of a mutated HSC and its immune cell offspring is denoted as “clonal hematopoiesis”[102] and associated with COVID-19 morbidity. Mounting evidence support that cardiac comorbidities are common in COVID-19 patients and such patients are in greater risk of mortality. The danger of CVD is two times greater in persons with clonal hematopoiesis[102]. Abnormal clonal hematopoiesis can provoke pro-inflammatory cytokines such as IL-6, IL-1β and IL-8, and inflammatory signals in macrophages and mast cells[102]. Higher levels of cytokines cause a sustained confluency of innate immune cells and a decrease production of acquired immune cells, so that the outcome of clonal hematopoiesis may participate to deprived COVID-19 consequences in aged persons. It is also found that SARS-CoV-2 directly activates mast cells with the subsequent release of proinflammatory cytokines such as IL-1.
The association COVID-19 with age is long-established with aged patients being additional susceptible to die. Principally ACE2 receptors and CD26 are responsible for the increased age-related susceptibility of COVID-19 and both the receptors are highly expressed in senescent cells. Coronaviruses target both ACE2 receptors and CD26 and the overexpression of these receptors in older patients cause augmented fatality rate in COVID-19 patients[29,103]. Ageing, a progressive decline in tissue homeostasis is correlated with chronic inflammatory symptoms. Several factors such as abnormal immune function, cytokines production by senescent cells, NF-κB signaling pathway activation or a defective autophagy response may enhance the activation of inflammatory pathways (i.e., the NOD-like receptor 3 inflammasome). Mounting reports support that cytokines storm is aroused in patients with COVID-19 which is chiefly revealed by enhancing IL-2, IL-7, G-CSF (granulocyte colony stimulating factor), and TNF-α. Of all the cytokines, IL-6 has been observed to be interlinked to extremely severe SARS-CoV-2 infection owing due to amplified viral replication[104]. It is observed that the CD8+ counts in frail COVID-19 patients are dramatically decrease than that in normal patients. CD4+ and CD8+ T cells are also necessary for clearance of viruses during principal infection in the mucosa[105]. Cytotoxic CD8+ T cells can destroy virus mediated infected cells. Thus, frailty-associated decay in immune action may clarify the interlinked between ageing and higher adverse consequences.
The emerging COVID-19 pandemic as a global threat and public health challenges throughout the world. This report highlights the importance of multiple risk factors of disease severity and mortality such as old age, male sex, smoking, and comorbidities for the pathobiology and clinical landscape of COVID-19. Mounting evidence suggests that COVID-19 is a sex specific and aged influenced disease and it affects by a wide variety of variables fluctuating from genetic to socioeconomic factors. Therefore, in our considerations, we covered the emerging COVID-19 pandemic infection in the comprehensive and many-sided context of connections. Although it is endeavored to draw hypotheses about gender and ageing specific disparities of SARS-CoV-2 infection, gender equality and frailty should be given the first priority for further investigation to treat COVID-19 infection.
All of the authors are grateful to the Centers for Disease Control and Prevention (CDC), United States and Ministry of Health and Family Welfare (MOH&FW), Bangladesh for making this data publicly available.
Manuscript source: Invited manuscript
Corresponding Author's Membership in Professional Societies: Bangladesh Biosafety and Biosecurity Association, BBBA62.
Specialty type: Radiology, nuclear medicine and medical imaging
Country/Territory of origin: Bangladesh
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: El-Arabey AA S-Editor: Huang P L-Editor: A P-Editor: Yuan YY
| 1. | Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF, Shi ZL. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270-273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15248] [Cited by in RCA: 14106] [Article Influence: 2821.2] [Reference Citation Analysis (1)] |
| 2. | Wu F, Zhao S, Yu B, Chen YM, Wang W, Song ZG, Hu Y, Tao ZW, Tian JH, Pei YY, Yuan ML, Zhang YL, Dai FH, Liu Y, Wang QM, Zheng JJ, Xu L, Holmes EC, Zhang YZ. A new coronavirus associated with human respiratory disease in China. Nature. 2020;579:265-269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6893] [Cited by in RCA: 7487] [Article Influence: 1497.4] [Reference Citation Analysis (0)] |
| 3. | Vellingiri B, Jayaramayya K, Iyer M, Narayanasamy A, Govindasamy V, Giridharan B, Ganesan S, Venugopal A, Venkatesan D, Ganesan H, Rajagopalan K, Rahman PKSM, Cho SG, Kumar NS, Subramaniam MD. COVID-19: A promising cure for the global panic. Sci Total Environ. 2020;725:138277. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 394] [Cited by in RCA: 321] [Article Influence: 64.2] [Reference Citation Analysis (0)] |
| 4. | Kannan S, Shaik Syed Ali P, Sheeza A, Hemalatha K. COVID-19 (Novel Coronavirus 2019) - recent trends. Eur Rev Med Pharmacol Sci. 2020;24:2006-2011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 136] [Reference Citation Analysis (0)] |
| 5. | Białas AJ, Kumor-Kisielewska A, Górski P. Ageing, sex, obesity, smoking and COVID-19 - truths, myths and speculations. Adv Respir Med. 2020;88:335-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 6. | Groß S, Jahn C, Cushman S, Bär C, Thum T. SARS-CoV-2 receptor ACE2-dependent implications on the cardiovascular system: From basic science to clinical implications. J Mol Cell Cardiol. 2020;144:47-53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 102] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 7. | Yang J, Petitjean SJL, Koehler M, Zhang Q, Dumitru AC, Chen W, Derclaye S, Vincent SP, Soumillion P, Alsteens D. Molecular interaction and inhibition of SARS-CoV-2 binding to the ACE2 receptor. Nat Commun. 2020;11:4541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 309] [Cited by in RCA: 441] [Article Influence: 88.2] [Reference Citation Analysis (0)] |
| 8. | Edwards MJ, Becker KA, Gripp B, Hoffmann M, Keitsch S, Wilker B, Soddemann M, Gulbins A, Carpinteiro E, Patel SH, Wilson GC, Pöhlmann S, Walter S, Fassbender K, Ahmad SA, Carpinteiro A, Gulbins E. Sphingosine prevents binding of SARS-CoV-2 spike to its cellular receptor ACE2. J Biol Chem. 2020;295:15174-15182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 9. | Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng W, Bao L, Zhang B, Liu G, Wang Z, Chappell M, Liu Y, Zheng D, Leibbrandt A, Wada T, Slutsky AS, Liu D, Qin C, Jiang C, Penninger JM. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med. 2005;11:875-879. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2710] [Cited by in RCA: 2645] [Article Influence: 132.3] [Reference Citation Analysis (0)] |
| 10. | Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020; 181: 281-292. e6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4743] [Cited by in RCA: 6146] [Article Influence: 1229.2] [Reference Citation Analysis (0)] |
| 11. | Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020;323:1239-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11409] [Cited by in RCA: 11496] [Article Influence: 2299.2] [Reference Citation Analysis (0)] |
| 12. | Peeri NC, Shrestha N, Rahman MS, Zaki R, Tan Z, Bibi S, Baghbanzadeh M, Aghamohammadi N, Zhang W, Haque U. The SARS, MERS and novel coronavirus (COVID-19) epidemics, the newest and biggest global health threats: what lessons have we learned? Int J Epidemiol. 2020;49:717-726. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 947] [Cited by in RCA: 819] [Article Influence: 163.8] [Reference Citation Analysis (0)] |
| 13. | Nieman DC. Coronavirus disease-2019: A tocsin to our aging, unfit, corpulent, and immunodeficient society. J Sport Health Sci. 2020;9:293-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 80] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 14. | Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S, Huang H, Zhang L, Zhou X, Du C, Zhang Y, Song J, Wang S, Chao Y, Yang Z, Xu J, Zhou X, Chen D, Xiong W, Xu L, Zhou F, Jiang J, Bai C, Zheng J, Song Y. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med. 2020;180:934-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4960] [Cited by in RCA: 5515] [Article Influence: 1103.0] [Reference Citation Analysis (1)] |
| 15. | Fung SY, Yuen KS, Ye ZW, Chan CP, Jin DY. A tug-of-war between severe acute respiratory syndrome coronavirus 2 and host antiviral defence: lessons from other pathogenic viruses. Emerg Microbes Infect. 2020;9:558-570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 244] [Cited by in RCA: 262] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 16. | Ruscitti P, Berardicurti O, Barile A, Cipriani P, Shoenfeld Y, Iagnocco A, Giacomelli R. Severe COVID-19 and related hyperferritinaemia: more than an innocent bystander? Ann Rheum Dis. 2020;79:1515-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 17. | CDC COVID-19 Response Team. Severe Outcomes Among Patients with Coronavirus Disease 2019 (COVID-19) - United States, February 12-March 16, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:343-346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1690] [Cited by in RCA: 1512] [Article Influence: 302.4] [Reference Citation Analysis (0)] |
| 18. | COVID-19 National Emergency Response Center, Epidemiology and Case Management Team, Korea Centers for Disease Control and Prevention. Coronavirus Disease-19: The First 7,755 Cases in the Republic of Korea. Osong Public Health Res Perspect. 2020;11:85-90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 89] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 19. | COVID-19 Surveillance Group. Characteristics of COVID-19 patients dying in Italy: Report based on available data on March 26th, 2020 [cited 25 May 2020]. Available from: https://www.epicentro.iss.it/coronavirus/bollettino/Report-COVID-2019_26_marzo_eng.pdf. |
| 20. | Xie Y, Wang Z, Liao H, Marley G, Wu D, Tang W. Epidemiologic, clinical, and laboratory findings of the COVID-19 in the current pandemic: systematic review and meta-analysis. BMC Infect Dis. 2020;20:640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 82] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 21. | Das AK, Gopalan SS. Epidemiology of COVID-19 and Predictors of Recovery in the Republic of Korea. Pulm Med. 2020;2020: 7291698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 22. | Zheng H, Tan J, Zhang X, Luo A, Wang L, Zhu W, Hébert HL, Meng W, Smith BH, Colvin LA, Hu J. Impact of sex and age on respiratory support and length of hospital stay among 1792 patients with COVID-19 in Wuhan, China. Br J Anaesth. 2020;125:e378-e380. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 23. | Pareek M, Bangash MN, Pareek N, Pan D, Sze S, Minhas JS, Hanif W, Khunti K. Ethnicity and COVID-19: an urgent public health research priority. Lancet. 2020;395:1421-1422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 342] [Cited by in RCA: 296] [Article Influence: 59.2] [Reference Citation Analysis (0)] |
| 24. | Petrilli CM, Jones SA, Yang J, Rajagopalan H, O'Donnell L, Chernyak Y, Tobin KA, Cerfolio RJ, Francois F, Horwitz LI. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1590] [Cited by in RCA: 1814] [Article Influence: 362.8] [Reference Citation Analysis (1)] |
| 25. | Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323:1061-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14113] [Cited by in RCA: 14757] [Article Influence: 2951.4] [Reference Citation Analysis (0)] |
| 26. | Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054-1062. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17476] [Cited by in RCA: 18184] [Article Influence: 3636.8] [Reference Citation Analysis (0)] |
| 27. | Li X, Wang L, Yan S, Yang F, Xiang L, Zhu J, Shen B, Gong Z. Clinical characteristics of 25 death cases with COVID-19: A retrospective review of medical records in a single medical center, Wuhan, China. Int J Infect Dis. 2020;94:128-132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 249] [Cited by in RCA: 262] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 28. | El-Arabey AA, Abdalla M. Metformin and COVID-19: A novel deal of an old drug. J Med Virol. 2020;92:2293-2294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 29. | Vahidy FS, Nicolas JC, Meeks JR, Khan O, Pan A, Jones SL, Masud F, Sostman HD, Phillips R, Andrieni JD, Kash BA, Nasir K. Racial and ethnic disparities in SARS-CoV-2 pandemic: analysis of a COVID-19 observational registry for a diverse US metropolitan population. BMJ Open. 2020;10:e039849. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 152] [Cited by in RCA: 187] [Article Influence: 37.4] [Reference Citation Analysis (0)] |
| 30. | Carter P, Anderson M, Mossialos E. Health system, public health, and economic implications of managing COVID-19 from a cardiovascular perspective. Eur Heart J. 2020;41:2516-2518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 31. | Moula AI, Micali LR, Matteucci F, Lucà F, Rao CM, Parise O, Parise G, Gulizia MM, Gelsomino S. Quantification of Death Risk in Relation to Sex, Pre-Existing Cardiovascular Diseases and Risk Factors in COVID-19 Patients: Let's Take Stock and See Where We Are. J Clin Med. 2020;9:2685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 32. | Salvati L, Biagioni B, Vivarelli E, Parronchi P. A gendered magnifying glass on COVID-19. Clin Mol Allergy. 2020;18:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 33. | Bhopal R. Covid-19 worldwide: we need precise data by age group and sex urgently. BMJ. 2020;369:m1366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 34. | Goyal MK, Simpson JN, Boyle MD, Badolato GM, Delaney M, McCarter R, Cora-Bramble D. Racial and/or Ethnic and Socioeconomic Disparities of SARS-CoV-2 Infection Among Children. Pediatrics. 2020;146:e2020009951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 155] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 35. | Grasselli G, Zangrillo A, Zanella A, Antonelli M, Cabrini L, Castelli A, Cereda D, Coluccello A, Foti G, Fumagalli R, Iotti G, Latronico N, Lorini L, Merler S, Natalini G, Piatti A, Ranieri MV, Scandroglio AM, Storti E, Cecconi M, Pesenti A; COVID-19 Lombardy ICU Network. Baseline Characteristics and Outcomes of 1591 Patients Infected With SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA. 2020;323:1574-1581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3537] [Cited by in RCA: 3825] [Article Influence: 765.0] [Reference Citation Analysis (0)] |
| 36. | Cattrini C, Bersanelli M, Latocca MM, Conte B, Vallome G, Boccardo F. Sex Hormones and Hormone Therapy during COVID-19 Pandemic: Implications for Patients with Cancer. Cancers (Basel). 2020;12:2325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 54] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 37. | Karlberg J, Chong DS, Lai WY. Do men have a higher case fatality rate of severe acute respiratory syndrome than women do? Am J Epidemiol. 2004;159:229-231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 282] [Cited by in RCA: 298] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 38. | Matsuyama R, Nishiura H, Kutsuna S, Hayakawa K, Ohmagari N. Clinical determinants of the severity of Middle East respiratory syndrome (MERS): a systematic review and meta-analysis. BMC Public Health. 2016;16:1203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 90] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 39. | Leong HN, Earnest A, Lim HH, Chin CF, Tan C, Puhaindran ME, Tan A, Chen MI, Leo YS. SARS in Singapore--predictors of disease severity. Ann Acad Med Singap. 2006;35:326-331. [PubMed] |
| 40. | Bartz D, Chitnis T, Kaiser UB, Rich-Edwards JW, Rexrode KM, Pennell PB, Goldstein JM, O'Neal MA, LeBoff M, Behn M, Seely EW, Joffe H, Manson JE. Clinical Advances in Sex- and Gender-Informed Medicine to Improve the Health of All: A Review. JAMA Intern Med. 2020;180:574-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 138] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 41. | Klein SL, Flanagan KL. Sex differences in immune responses. Nat Rev Immunol. 2016;16:626-638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2321] [Cited by in RCA: 3909] [Article Influence: 434.3] [Reference Citation Analysis (0)] |
| 42. | Jin JM, Bai P, He W, Wu F, Liu XF, Han DM, Liu S, Yang JK. Gender Differences in Patients With COVID-19: Focus on Severity and Mortality. Front Public Health. 2020;8:152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1408] [Cited by in RCA: 1329] [Article Influence: 265.8] [Reference Citation Analysis (0)] |
| 43. | Scheel-Hincke LL, Möller S, Lindahl-Jacobsen R, Jeune B, Ahrenfeldt LJ. Cross-national comparison of sex differences in ADL and IADL in Europe: findings from SHARE. Eur J Ageing. 2020;17:69-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 44. | Global Health 50/50. Sex, gender and COVID-19: overview and resources 2020 [cited 27 June, 2020]. Available from: https://globalhealth5050.org/covid19. |
| 45. | Oudit GY, Pfeffer MA. Plasma angiotensin-converting enzyme 2: novel biomarker in heart failure with implications for COVID-19. Eur Heart J. 2020;41:1818-1820. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 58] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 46. | Bunyavanich S, Do A, Vicencio A. Nasal Gene Expression of Angiotensin-Converting Enzyme 2 in Children and Adults. JAMA. 2020;323:2427-2429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 563] [Cited by in RCA: 635] [Article Influence: 127.0] [Reference Citation Analysis (0)] |
| 47. | Wu C, Liu Y, Yang Y, Zhang P, Zhong W, Wang Y, Wang Q, Xu Y, Li M, Li X, Zheng M, Chen L, Li H. Analysis of therapeutic targets for SARS-CoV-2 and discovery of potential drugs by computational methods. Acta Pharm Sin B. 2020;10:766-788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1221] [Cited by in RCA: 1412] [Article Influence: 282.4] [Reference Citation Analysis (0)] |
| 48. | Gheblawi M, Wang K, Viveiros A, Nguyen Q, Zhong JC, Turner AJ, Raizada MK, Grant MB, Oudit GY. Angiotensin-Converting Enzyme 2: SARS-CoV-2 Receptor and Regulator of the Renin-Angiotensin System: Celebrating the 20th Anniversary of the Discovery of ACE2. Circ Res. 2020;126:1456-1474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1325] [Cited by in RCA: 1376] [Article Influence: 275.2] [Reference Citation Analysis (0)] |
| 49. | Vankadari N, Wilce JA. Emerging WuHan (COVID-19) coronavirus: glycan shield and structure prediction of spike glycoprotein and its interaction with human CD26. Emerg Microbes Infect. 2020;9:601-604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 388] [Cited by in RCA: 449] [Article Influence: 89.8] [Reference Citation Analysis (0)] |
| 50. | Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020; 181: 271-280. e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11946] [Cited by in RCA: 14245] [Article Influence: 2849.0] [Reference Citation Analysis (0)] |
| 51. | Jia H. Pulmonary Angiotensin-Converting Enzyme 2 (ACE2) and Inflammatory Lung Disease. Shock. 2016;46:239-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 227] [Article Influence: 25.2] [Reference Citation Analysis (0)] |
| 52. | Al-Lami RA, Urban RJ, Volpi E, Algburi AMA, Baillargeon J. Sex Hormones and Novel Corona Virus Infectious Disease (COVID-19). Mayo Clin Proc. 2020;95:1710-1714. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 95] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 53. | Gurwitz D. Angiotensin receptor blockers as tentative SARS-CoV-2 therapeutics. Drug Dev Res. 2020;81:537-540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 553] [Cited by in RCA: 608] [Article Influence: 121.6] [Reference Citation Analysis (0)] |
| 54. | Shi Y, Wang Y, Shao C, Huang J, Gan J, Huang X, Bucci E, Piacentini M, Ippolito G, Melino G. COVID-19 infection: the perspectives on immune responses. Cell Death Differ. 2020;27:1451-1454. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 920] [Cited by in RCA: 1002] [Article Influence: 200.4] [Reference Citation Analysis (0)] |
| 55. | Khan A, Benthin C, Zeno B, Albertson TE, Boyd J, Christie JD, Hall R, Poirier G, Ronco JJ, Tidswell M, Hardes K, Powley WM, Wright TJ, Siederer SK, Fairman DA, Lipson DA, Bayliffe AI, Lazaar AL. A pilot clinical trial of recombinant human angiotensin-converting enzyme 2 in acute respiratory distress syndrome. Crit Care. 2017;21:234. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 408] [Cited by in RCA: 469] [Article Influence: 58.6] [Reference Citation Analysis (0)] |
| 56. | Nicholls J, Peiris M. Good ACE, bad ACE do battle in lung injury, SARS. Nat Med. 2005;11:821-822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 57. | Park TK, Yang JH, Jeon K, Choi SH, Choi JH, Gwon HC, Chung CR, Park CM, Cho YH, Sung K, Suh GY. Extracorporeal membrane oxygenation for refractory septic shock in adults. Eur J Cardiothorac Surg. 2015;47:e68-e74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 72] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 58. | Santos RAS, Oudit GY, Verano-Braga T, Canta G, Steckelings UM, Bader M. The renin-angiotensin system: going beyond the classical paradigms. Am J Physiol Heart Circ Physiol. 2019;316:H958-H970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 211] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 59. | Wang K, Gheblawi M, Oudit GY. Angiotensin Converting Enzyme 2: A Double-Edged Sword. Circulation. 2020;142:426-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 213] [Article Influence: 42.6] [Reference Citation Analysis (0)] |
| 60. | Sun H, Ning R, Tao Y, Yu C, Deng X, Zhao C, Meng S, Tang F, Xu D. Risk Factors for Mortality in 244 Older Adults With COVID-19 in Wuhan, China: A Retrospective Study. J Am Geriatr Soc. 2020;68:E19-E23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 136] [Article Influence: 27.2] [Reference Citation Analysis (0)] |
| 61. | Schurz H, Salie M, Tromp G, Hoal EG, Kinnear CJ, Möller M. The X chromosome and sex-specific effects in infectious disease susceptibility. Hum Genomics. 2019;13:2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 254] [Cited by in RCA: 249] [Article Influence: 41.5] [Reference Citation Analysis (0)] |
| 62. | Márquez EJ, Trowbridge J, Kuchel GA, Banchereau J, Ucar D. The lethal sex gap: COVID-19. Immun Ageing. 2020;17:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 63. | Helena Mangs A, Morris BJ. The Human Pseudoautosomal Region (PAR): Origin, Function and Future. Curr Genomics. 2007;8:129-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 165] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 64. | Wang Z, Xu X. scRNA-seq Profiling of Human Testes Reveals the Presence of the ACE2 Receptor, A Target for SARS-CoV-2 Infection in Spermatogonia, Leydig and Sertoli Cells. Cells. 2020;9:920. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 400] [Cited by in RCA: 398] [Article Influence: 79.6] [Reference Citation Analysis (0)] |
| 65. | Liu PP, Blet A, Smyth D, Li H. The Science Underlying COVID-19: Implications for the Cardiovascular System. Circulation. 2020;142:68-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 509] [Cited by in RCA: 587] [Article Influence: 117.4] [Reference Citation Analysis (0)] |
| 66. | Alexander SPH, Armstrong JF, Davenport AP, Davies JA, Faccenda E, Harding SD, Levi-Schaffer F, Maguire JJ, Pawson AJ, Southan C, Spedding M. A rational roadmap for SARS-CoV-2/COVID-19 pharmacotherapeutic research and development: IUPHAR Review 29. Br J Pharmacol. 2020;177:4942-4966. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 67. | Stopsack KH, Mucci LA, Antonarakis ES, Nelson PS, Kantoff PW. TMPRSS2 and COVID-19: Serendipity or Opportunity for Intervention? Cancer Discov. 2020;10:779-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 287] [Article Influence: 57.4] [Reference Citation Analysis (0)] |
| 68. | Robinson EL, Alkass K, Bergmann O, Maguire JJ, Roderick HL, Davenport AP. Genes encoding ACE2, TMPRSS2 and related proteins mediating SARS-CoV-2 viral entry are upregulated with age in human cardiomyocytes. J Mol Cell Cardiol. 2020;147:88-91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 69. | Wambier CG, Goren A. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection is likely to be androgen mediated. J Am Acad Dermatol. 2020;83:308-309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 161] [Article Influence: 32.2] [Reference Citation Analysis (0)] |
| 70. | McCoy J, Wambier CG, Vano-Galvan S, Shapiro J, Sinclair R, Ramos PM, Washenik K, Andrade M, Herrera S, Goren A. Racial variations in COVID-19 deaths may be due to androgen receptor genetic variants associated with prostate cancer and androgenetic alopecia. Are anti-androgens a potential treatment for COVID-19? J Cosmet Dermatol. 2020;19:1542-1543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 71. | Gebhard C, Regitz-Zagrosek V, Neuhauser HK, Morgan R, Klein SL. Impact of sex and gender on COVID-19 outcomes in Europe. Biol Sex Differ. 2020;11:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 797] [Cited by in RCA: 732] [Article Influence: 146.4] [Reference Citation Analysis (0)] |
| 72. | Klein SL, Marriott I, Fish EN. Sex-based differences in immune function and responses to vaccination. Trans R Soc Trop Med Hyg. 2015;109:9-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 399] [Article Influence: 39.9] [Reference Citation Analysis (0)] |
| 73. | Ghosh S, Klein RS. Sex Drives Dimorphic Immune Responses to Viral Infections. J Immunol. 2017;198:1782-1790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 163] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 74. | Pozzilli P, Lenzi A. Commentary: Testosterone, a key hormone in the context of COVID-19 pandemic. Metabolism. 2020;108:154252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 117] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 75. | Montopoli M, Zumerle S, Vettor R, Rugge M, Zorzi M, Catapano CV, Carbone GM, Cavalli A, Pagano F, Ragazzi E, Prayer-Galetti T, Alimonti A. Androgen-deprivation therapies for prostate cancer and risk of infection by SARS-CoV-2: a population-based study (N = 4532). Ann Oncol. 2020;31:1040-1045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 353] [Cited by in RCA: 396] [Article Influence: 79.2] [Reference Citation Analysis (0)] |
| 76. | Walker RF, Zakai NA, MacLehose RF, Cowan LT, Adam TJ, Alonso A, Lutsey PL. Association of Testosterone Therapy With Risk of Venous Thromboembolism Among Men With and Without Hypogonadism. JAMA Intern Med. 2020;180:190-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 91] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 77. | Golan R, Scovell JM, Ramasamy R. Age-related testosterone decline is due to waning of both testicular and hypothalamic-pituitary function. Aging Male. 2015;18:201-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 78. | Cao J, Chen TM, Hao WJ, Li J, Liu L, Zhu BP, Li XY. Correlation between sex hormone levels and obesity in the elderly male. Aging Male. 2012;15:85-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 79. | Traish A, Bolanos J, Nair S, Saad F, Morgentaler A. Do Androgens Modulate the Pathophysiological Pathways of Inflammation? J Clin Med. 2018;7:549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 92] [Article Influence: 13.1] [Reference Citation Analysis (1)] |
| 80. | Márquez EJ, Chung CH, Marches R, Rossi RJ, Nehar-Belaid D, Eroglu A, Mellert DJ, Kuchel GA, Banchereau J, Ucar D. Sexual-dimorphism in human immune system aging. Nat Commun. 2020;11:751. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 348] [Article Influence: 69.6] [Reference Citation Analysis (0)] |
| 81. | Villa A, Rizzi N, Vegeto E, Ciana P, Maggi A. Estrogen accelerates the resolution of inflammation in macrophagic cells. Sci Rep. 2015;5:15224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 191] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 82. | Gaskins AJ, Wilchesky M, Mumford SL, Whitcomb BW, Browne RW, Wactawski-Wende J, Perkins NJ, Schisterman EF. Endogenous reproductive hormones and C-reactive protein across the menstrual cycle: the BioCycle Study. Am J Epidemiol. 2012;175:423-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 139] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 83. | Giefing-Kröll C, Berger P, Lepperdinger G, Grubeck-Loebenstein B. How sex and age affect immune responses, susceptibility to infections, and response to vaccination. Aging Cell. 2015;14:309-321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 402] [Cited by in RCA: 515] [Article Influence: 51.5] [Reference Citation Analysis (0)] |
| 84. | Piccinni MP, Giudizi MG, Biagiotti R, Beloni L, Giannarini L, Sampognaro S, Parronchi P, Manetti R, Annunziato F, Livi C. Progesterone favors the development of human T helper cells producing Th2-type cytokines and promotes both IL-4 production and membrane CD30 expression in established Th1 cell clones. J Immunol. 1995;155:128-133. [PubMed] |
| 85. | Liu S, Zhi Y, Ying S. COVID-19 and Asthma: Reflection During the Pandemic. Clin Rev Allergy Immunol. 2020;59:78-88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 141] [Cited by in RCA: 129] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 86. | Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM, O'Meara MJ, Rezelj VV, Guo JZ, Swaney DL, Tummino TA, Hüttenhain R, Kaake RM, Richards AL, Tutuncuoglu B, Foussard H, Batra J, Haas K, Modak M, Kim M, Haas P, Polacco BJ, Braberg H, Fabius JM, Eckhardt M, Soucheray M, Bennett MJ, Cakir M, McGregor MJ, Li Q, Meyer B, Roesch F, Vallet T, Mac Kain A, Miorin L, Moreno E, Naing ZZC, Zhou Y, Peng S, Shi Y, Zhang Z, Shen W, Kirby IT, Melnyk JE, Chorba JS, Lou K, Dai SA, Barrio-Hernandez I, Memon D, Hernandez-Armenta C, Lyu J, Mathy CJP, Perica T, Pilla KB, Ganesan SJ, Saltzberg DJ, Rakesh R, Liu X, Rosenthal SB, Calviello L, Venkataramanan S, Liboy-Lugo J, Lin Y, Huang XP, Liu Y, Wankowicz SA, Bohn M, Safari M, Ugur FS, Koh C, Savar NS, Tran QD, Shengjuler D, Fletcher SJ, O'Neal MC, Cai Y, Chang JCJ, Broadhurst DJ, Klippsten S, Sharp PP, Wenzell NA, Kuzuoglu-Ozturk D, Wang HY, Trenker R, Young JM, Cavero DA, Hiatt J, Roth TL, Rathore U, Subramanian A, Noack J, Hubert M, Stroud RM, Frankel AD, Rosenberg OS, Verba KA, Agard DA, Ott M, Emerman M, Jura N, von Zastrow M, Verdin E, Ashworth A, Schwartz O, d'Enfert C, Mukherjee S, Jacobson M, Malik HS, Fujimori DG, Ideker T, Craik CS, Floor SN, Fraser JS, Gross JD, Sali A, Roth BL, Ruggero D, Taunton J, Kortemme T, Beltrao P, Vignuzzi M, García-Sastre A, Shokat KM, Shoichet BK, Krogan NJ. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020;583:459-468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3145] [Cited by in RCA: 3181] [Article Influence: 636.2] [Reference Citation Analysis (0)] |
| 87. | Zhou Y, Hou Y, Shen J, Huang Y, Martin W, Cheng F. Network-based drug repurposing for novel coronavirus 2019-nCoV/SARS-CoV-2. Cell Discov. 2020;6:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1048] [Cited by in RCA: 1040] [Article Influence: 208.0] [Reference Citation Analysis (0)] |
| 88. | Scully EP, Haverfield J, Ursin RL, Tannenbaum C, Klein SL. Considering how biological sex impacts immune responses and COVID-19 outcomes. Nat Rev Immunol. 2020;20:442-447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 466] [Cited by in RCA: 611] [Article Influence: 122.2] [Reference Citation Analysis (0)] |
| 89. | Furman D, Campisi J, Verdin E, Carrera-Bastos P, Targ S, Franceschi C, Ferrucci L, Gilroy DW, Fasano A, Miller GW, Miller AH, Mantovani A, Weyand CM, Barzilai N, Goronzy JJ, Rando TA, Effros RB, Lucia A, Kleinstreuer N, Slavich GM. Chronic inflammation in the etiology of disease across the life span. Nat Med. 2019;25:1822-1832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1084] [Cited by in RCA: 2715] [Article Influence: 452.5] [Reference Citation Analysis (0)] |
| 90. | Goronzy JJ, Weyand CM. Successful and Maladaptive T Cell Aging. Immunity. 2017;46:364-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 246] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 91. | Libert C, Dejager L, Pinheiro I. The X chromosome in immune functions: when a chromosome makes the difference. Nat Rev Immunol. 2010;10:594-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 418] [Cited by in RCA: 525] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 92. | Bianchi I, Lleo A, Gershwin ME, Invernizzi P. The X chromosome and immune associated genes. J Autoimmun. 2012;38:J187-J192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 267] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 93. | Petes C, Odoardi N, Gee K. The Toll for Trafficking: Toll-Like Receptor 7 Delivery to the Endosome. Front Immunol. 2017;8:1075. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 124] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 94. | Griesbeck M, Ziegler S, Laffont S, Smith N, Chauveau L, Tomezsko P, Sharei A, Kourjian G, Porichis F, Hart M, Palmer CD, Sirignano M, Beisel C, Hildebrandt H, Cénac C, Villani AC, Diefenbach TJ, Le Gall S, Schwartz O, Herbeuval JP, Autran B, Guéry JC, Chang JJ, Altfeld M. Sex Differences in Plasmacytoid Dendritic Cell Levels of IRF5 Drive Higher IFN-α Production in Women. J Immunol. 2015;195:5327-5336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 193] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 95. | Coomes EA, Haghbayan H. Interleukin-6 in Covid-19: A systematic review and meta-analysis. Rev Med Virol. 2020;30:1-9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 493] [Cited by in RCA: 512] [Article Influence: 102.4] [Reference Citation Analysis (0)] |
| 96. | Zeng F, Dai C, Cai P, Wang J, Xu L, Li J, Hu G, Wang Z, Zheng F, Wang L. A comparison study of SARS-CoV-2 IgG antibody between male and female COVID-19 patients: A possible reason underlying different outcome between sex. J Med Virol. 2020;92:2050-2054. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 190] [Cited by in RCA: 204] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 97. | Piasecka B, Duffy D, Urrutia A, Quach H, Patin E, Posseme C, Bergstedt J, Charbit B, Rouilly V, MacPherson CR, Hasan M, Albaud B, Gentien D, Fellay J, Albert ML, Quintana-Murci L; Milieu Intérieur Consortium. Distinctive roles of age, sex, and genetics in shaping transcriptional variation of human immune responses to microbial challenges. Proc Natl Acad Sci USA. 2018;115:E488-E497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 157] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 98. | Shen C, Wang Z, Zhao F, Yang Y, Li J, Yuan J, Wang F, Li D, Yang M, Xing L, Wei J, Xiao H, Yang Y, Qu J, Qing L, Chen L, Xu Z, Peng L, Li Y, Zheng H, Chen F, Huang K, Jiang Y, Liu D, Zhang Z, Liu Y, Liu L. Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma. JAMA. 2020;323:1582-1589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1734] [Cited by in RCA: 1591] [Article Influence: 318.2] [Reference Citation Analysis (0)] |
| 99. | Olson NC, Doyle MF, Jenny NS, Huber SA, Psaty BM, Kronmal RA, Tracy RP. Decreased naive and increased memory CD4(+) T cells are associated with subclinical atherosclerosis: the multi-ethnic study of atherosclerosis. PLoS One. 2013;8:e71498. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 76] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 100. | Clave E, Araujo IL, Alanio C, Patin E, Bergstedt J, Urrutia A, Lopez-Lastra S, Li Y, Charbit B, MacPherson CR, Hasan M, Melo-Lima BL, Douay C, Saut N, Germain M, Trégouët DA, Morange PE, Fontes M, Duffy D, Di Santo JP, Quintana-Murci L, Albert ML, Toubert A; Milieu Intérieur Consortium. Human thymopoiesis is influenced by a common genetic variant within the TCRA-TCRD locus. Sci Transl Med. 2018;10:eaao2966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 101. | Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5228] [Cited by in RCA: 5778] [Article Influence: 1155.6] [Reference Citation Analysis (2)] |
| 102. | Jaiswal S, Natarajan P, Silver AJ, Gibson CJ, Bick AG, Shvartz E, McConkey M, Gupta N, Gabriel S, Ardissino D, Baber U, Mehran R, Fuster V, Danesh J, Frossard P, Saleheen D, Melander O, Sukhova GK, Neuberg D, Libby P, Kathiresan S, Ebert BL. Clonal Hematopoiesis and Risk of Atherosclerotic Cardiovascular Disease. N Engl J Med. 2017;377:111-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1338] [Cited by in RCA: 1850] [Article Influence: 231.3] [Reference Citation Analysis (0)] |
| 103. | Sargiacomo C, Sotgia F, Lisanti MP. COVID-19 and chronological aging: senolytics and other anti-aging drugs for the treatment or prevention of corona virus infection? Aging (Albany NY). 2020;12:6511-6517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 148] [Article Influence: 29.6] [Reference Citation Analysis (0)] |
| 104. | Franceschi C, Garagnani P, Parini P, Giuliani C, Santoro A. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat Rev Endocrinol. 2018;14:576-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1183] [Cited by in RCA: 1855] [Article Influence: 265.0] [Reference Citation Analysis (0)] |
| 105. | Ma Y, Hou L, Yang X, Huang Z, Yang X, Zhao N, He M, Shi Y, Kang Y, Yue J, Wu C. The association between frailty and severe disease among COVID-19 patients aged over 60 years in China: a prospective cohort study. BMC Med. 2020;18:274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |