INTRODUCTION
Computed tomography (CT) scanner has rapidly evolved from single slice to multislice CT (MSCT) which started from 4-slice systems in 1998 to the latest 256-slice and 320-slice CT systems. With smaller detector size and faster gantry rotation speed, spatial and temporal resolutions of the 64- and post-64 MSCT scanners have enabled coronary artery imaging a feasible and reliable clinical test. The technological advancements from 16- to 320-slice systems have progressed in a relatively uniform fashion with improved longitudinal (z-axis) volume coverage, decreased gantry rotation time, and smaller detector elements[1,2]. With the ability to acquire volume data, technological improvements in CT scanning also enable generation of 3D image processing such as multiplanar reformation, maximum intensity projection, surface-shaded display, and volume-rendering techniques, and these reconstructed visualizations have made coronary CT angiography (CCTA) an important component of medical imaging visualization in daily practice[3].
The purpose of this paper is to provide an overview of CCTA with a focus on the diagnostic accuracy and prognostic value in coronary artery disease. Technological developments of MSCT scanners are briefly discussed, while limitations and challenges of CCTA are highlighted.
TECHNOLOGICAL DEVELOPMENTS IN CCTA
Diagnostic performance of CCTA is closely related to technological improvements that occurred with each successive generation of MSCT scanners. The spatial and temporal resolution of MSCT scanners determine the diagnostic value of CCTA in coronary artery disease (CAD).
4-slice CT
In 1998, a 4-slice CT scanner was introduced by several manufacturers representing an obvious quantum leap in clinical performance[4,5]. Four detector “rows”corresponding to the 4 simultaneously collected slices fed data into four parallel data ”channels”, so that these 4-slice scanners were said to possess four data channels. These 4-slice scanners, however, were quite flexible with regard to how detector rows could be configured; groups of detector elements in the z-direction could be electronically linked to function as a single, longer detector, thus providing more flexibility in the section thickness of the four acquired slices[6,7]. Fundamental advantages of MSCT include substantially shorter acquisition times, retrospective creation of thinner or thicker sections from the same raw data, and improved three-dimensional rendering with diminished helical artifacts[4].
The main advantage is the increased volume coverage per unit time at high axial resolution and subsequent improved temporal resolution[4]. Four-slice scanners are the basic system for CCTA examination. With only 250 ms of temporal resolution from a gantry rotation of 500 ms CCTA with use of 4-slice CT requires longer longitudinal scan to cover the entire cardiac chamber and coronary arteries, thus, this may result in long breath-hold between 30 and 40 s which leads to breathing and motion artifacts, and also limits to patients with low heart rates[8].
16-slice CT
The installation of 16-slice CT scanners in 2002 provides 16 detector channels enabling simultaneously acquisition of 16 slices per gantry rotation[9]. In addition to simultaneously acquiring up to 16 slices, the detector arrays associated with 16-slice scanners were redesigned to allow thinner slices to be obtained as well. Note that in all of the models, the innermost 16 detector elements along the z-axis are half the size of the outermost elements, allowing simultaneous acquisition of 16 thin slices (from 0.5 mm to 0.75 mm thick, depending on the model and manufacturer). When the inner detectors were used to acquire submillimeter slices, the total acquired z-axis length and therefore the total width of the x-ray beam ranged from 8 mm for the Toshiba model to 12 mm for the Philips and Siemens scanners. Alternatively, the inner 16 elements could be linked in pairs for the acquisition of up to 16 thicker slices[10].
Sixteen-slice scanners have a slightly better spatial resolution and faster gantry rotation (420 ms) than that in 4-slice CT[11]. The major advantage of 16-slice scanners over 4-slice CT is the longer z-axis coverage (16 mm × 0.75 mm vs 4 mm × 1.0 mm), resulting in significantly shorter breath- hold and fewer motion artifacts[12-14]. The rotational speed of 16-slice scanners is only marginally faster, and adaptive multi-cycle reconstructions, which require a high number of detectors, cannot be applied because of heart rate variations. As a consequence of these factors, image quality with the 16-slice scanner is significantly improved, reducing the number of coronary segments with poor image quality[12-15].
CCTA became more clinically practical with 16-slice CT scanners using retrospective electrocardiogram (ECG) gating to capture cardiac motion plus the z-axis coverage[16]. However, cardiac motion and stair-step artifacts are the main challenge for this system. Therefore, there are a few steps that are suggested to overcome these problems, which include increasing the number of detector elements and the volume coverage along the z-axis of detector block. Moreover, increase in the sensitivity of detector material and application of iterative image reconstruction algorithms represents another approach to improve cardiac image quality[17,18]. During 2003 and 2004, manufacturers introduced different types of MSCT models with less than 16-slice scanners, but most commonly the introduction of more than 16-slice scanners represented the main direction for improving MSCT systems[9].
64-slice CT
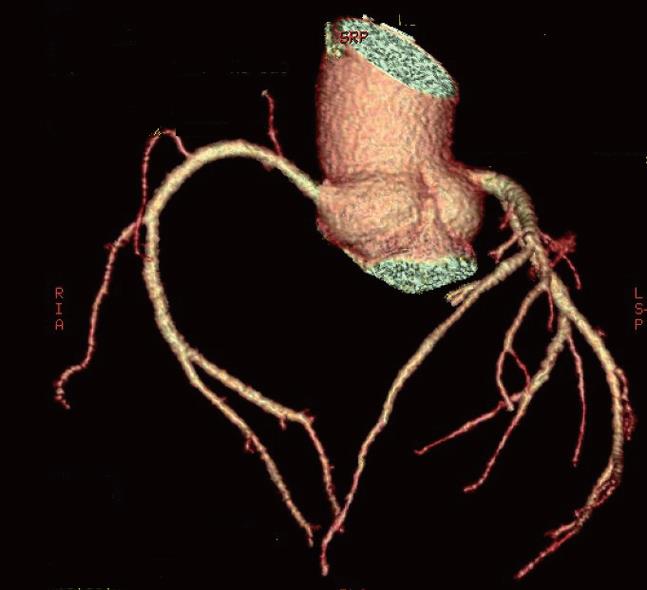
The 64-slice CT was first introduced with a single x-ray source mounted opposite to a 64-detector-array in the gantry unit. With gantry rotation times down to 0.33 s for 64-slice CT (0.375 s for 16-slice CT), temporal resolution for ECG-gated cardiac imaging is again markedly improved. The increased temporal resolution of 64-slice CT has the potential to improve the clinical strength of ECG-gated cardiac examinations at higher heart rates, thereby reducing the number of patients requiring heart rate control. In contrast to previous studies, high diagnostic accuracy has been achieved despite the presence of calcified coronary plaques. In addition, using 64-slice CT the scanning time is reduced to less than 15 s, allowing a decreased breath-hold time, better utilization of contrast medium with fewer enhancements of adjacent structures and a lower dose of applied contrast medium. Improvement of image quality has also been reported in the visualization of all coronary artery branches with high sensitivity and specificity achieved (Figure 1).
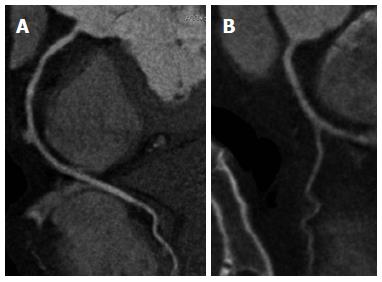
Figure 1 Three-dimensional volume rendering image acquired with 64-slice coronary computed tomography angiography demonstrates right and left coronary arteries without lumen stenosis.
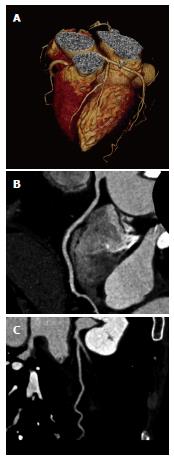
The new-generation dual-source MSCT (Somatom Definition FLASH; Siemens Medical Solution, Forchheim, Germany) which was introduced in late 2008 is equipped with two 64-detector row units, each with an alternating focal spot. The 360° gantry rotation time is 280 ms, translating to a temporal resolution of approximately 75 ms when the scanner operates with both x-ray tubes collecting data at the same energy (Figure 2). The vendor has proposed a high-pitch prospectively ECG-triggered scanning acquisition[19,20]. In single-source 64-slice CT, the maximum pitch used in CCTA is roughly between 0.2 and 0.5 for gapless image reconstruction. The pitch can be increased up to 3.4 in dual-source Siemens Definition Flash systems. For CCTA, the typical phase window required for a diagnostic quality examination regarding motion artifact is 10% of the R-R interval. The pitch required for multiphase acquisition ranges from 0.2 to 0.5, depending on the heart rate[21].
Figure 2 New-generation dual-source multislice computed tomography.
A: 3D volume rendering image acquired with dual-source coronary computed tomography angiography shows excellent visualisation of normal left coronary artery and its side branches; B, C: Curved planar reformation clearly shows the anatomical structure of right and left coronary arteries.
With the high-pitch acquisition mode, only one phase is acquired, which gradually increases with the z-axis table translation. The influence on image quality for different clinical scenarios and heart rates is evaluated with the second generation dual-source CT. Achenbach et al[22] demonstrated the feasibility of this new scanning method using second-generation dual-source CT. However, slow and regular heart rates are the prerequisite for this acquisition protocol that is prospectively triggered by ECG signal and is anticipated to scan the entire heart in 270 ms, with a pitch of up to 3.4[22]. Another potential advantage of dual-source CT is tissue characterization with both detector systems operating at different tube voltages known as dual-energy CT (DECT). Although this has not been extensively studied to date, the two x-ray beams of different energy spectra in theory could better demonstrate varying attenuation characteristics of different tissues[23,24]. Studies have shown the feasibility of using DECT for myocardial perfusion imaging of CAD. Ruzsics et al[23] compared DECT with SPECT to evaluate the diagnostic performance of DECT for imaging coronary artery morphology and assessing myocardial blood supply. In a group of 36 patients with suspected or known CAD, over 90% diagnostic accuracy was achieved with DECT for detecting any type of myocardial perfusion defect observed on SPECT. Nagao et al[25] used iodine map that is available with DECT to detect alterations in coronary flow during adenosine stress and rest. This is the first non-invasive method to provide a functional assessment of coronary artery flow using cardiac CT, although further studies are needed to confirm these early results.
DECT cardiac imaging can also be achieved with a single X-ray tube. GE Healthcare’s Discovery CT750 HD spectral imaging is based on fast kV switching-dynamic switching between 2 different energy levels of X-rays from view to view during a single rotation[26]. This allows for demonstration of different material densities as scatter plots, histograms and region of interest, thus, enabling myocardial perfusion analysis of cardiac function. Despite these promising results, however, large patient cohorts are needed to confirm the potential application of a single protocol for anatomic and myocardial perfusion assessment of CAD. The diagnostic accuracy of CCTA has been reported extensively in the literature ranging from the earlier studies using retrospectively ECG-gated protocols to the recent reports comparing prospective ECG-triggering and retrospective ECG-gating. In retrospectively ECG-gated CCTA, several studies on different types and generations of MSCT scanners were carried out with overall results showing that CCTA had moderate to high sensitivity of 86%-99% and high specificity of 89%-100% in patients with suspected CAD. Image quality of coronary artery visualization was impaired and suboptimal in a number of cases with 4-slice CT as the unassessable coronary segments could be as high as more than 20%[27]. With 16-and 64-slice CT, thinner detector rows increased the spatial resolution and further shortened the total scan time, resulting in improved diagnostic value of CCTA[12,14,15]. In particular, a very high negative predictive value of over 95% (96%-99%) has been reported in these studies indicating that CCTA can be used as a reliable screening tool for CAD[28,29]. Moreover, multicenter studies were also conducted on 64-slice CT scanner to investigate the diagnostic accuracy of CCTA with different risks of CAD prevalence. The results showed that high sensitivity (94%), specificity (83%) and negative predictive value (99%) was achieved in high risk patients with CAD (68%). Similarly, high diagnostic accuracy was also presented in low risk of CAD with sensitivity, specificity and negative predictive value being 94%, 83% and 99% in 25% of CAD prevalence; 85%, 90% and 83% in 56% of CAD prevalence, respectively[30-32]. A study on the high-pitch mode with dual source CT also resulted in high sensitivity, specificity and negative predictive value of 94%, 91% and 97% respectively[33]. Over the last few years, prospectively ECG-triggered CCTA is increasingly used in the diagnosis of CAD with promising results reported. The sensitivity (93.7%-100%), specificity (82.7%-97%) and negative predictive value (95%-98%) in the assessment of CAD were reported in multiple studies confirming the feasibility of this fast developing technique[28,29] (Figure 3).
Figure 3 Electrocardiogram-triggered coronary computed tomography angiography.
Prospectively ECG-triggered coronary computed tomography angiography shows a mixed plaque at the mid-segment of right coronary artery (A, arrows), and calcified plaques at the proximal segment of left anterior descending branch (B, arrows). ECG: Electrocardiogram.
128- and 256-slice CT
In late 2007, the 128-slice CT (Brilliance iCT; Philips Healthcare, Cleveland, OH) was introduced with a 128 mm × 0.625 mm detector row system with dual focal spot positions to double the number of slices within the 8-cm (width) z-axis gantry coverage. The iCT has a gantry rotation time of 270 ms, which translates to an approximate temporal resolution of 135 ms. Prospectively ECG-triggered CCTA typically covers the entire heart in two axial acquisitions over three heartbeats. During the diastole of the first heartbeat, the upper half of the heart is imaged. During the second heartbeat, the X-ray table translates 62.4 mm. Subsequently, the lower half of the heart is acquired during the diastole of the third heartbeat. The scanner is equipped with several radiation reduction capabilities, including a dynamic helical collimator and an adaptive axial collimator to reduce z-over scanning[34,35].
Second generation of 128-slice CT was introduced with dual-source which uses two x-ray tubes with opposing 64 detector arrays mounted 90° from each other. The main advantage of this system is that the temporal resolution is effectively halved because each x-ray tube/detector array system only needs to rotate half of the angle that would otherwise be required by a single-source system. The number of detector rows in the longitudinal axis (z-axis) and the number of slices of CT system are not interchangeable terms because multiple systems with an alternating focal spot allow the same z-axis coverage to be sampled twice, and thus the number of image slices generated is double the number of detector rows[20]. However, the volume coverage remains the same; for example, a 128-detector row scanner with two alternating z-focal spot positions can be referred to as 256-slice CT. It is important to specify the number of detector rows in z-axis, with or without alternating focal spot positions, and single versus dual source. A 128-slice dual-source CT also demonstrated high diagnostic accuracy of 93%, 94% and 97% corresponding to sensitivity, specificity and negative predictive value, respectively[33].
Most of the current studies using 128- and 256-slice CCTA focus on image quality and radiation dose reduction, while reports on the diagnostic performance are scarce[34-36]. Two recent studies have reported that 256-slice CCTA have high sensitivity (> 90% patient- and segment-based) and high diagnostic accuracy in patients with suspected CAD, with resultant very low radiation dose[37,38], althoughfurther research is needed to investigate the diagnostic performance of CCTA with use of these recent models based on a large cohort and multi-centre studies.
320-slice CT
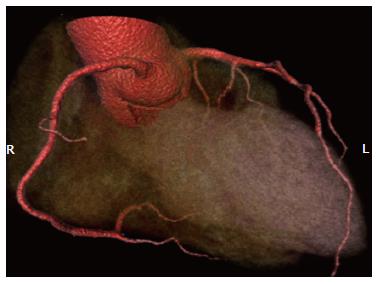
This hardware (Aquilion One Dynamic Volume CT; Toshiba Medical System, Japan) currently has the largest z-axis detector coverage. It was released shortly after experiments with a 256-detector row CT prototype[39-41]. Each detector element is 0.5 mm wide, yielding a maximum of 16-cm z-axis coverage (Figure 4). This configuration allows three-dimensional volumetric entire heart imaging during the diastole of one R-R interval. In 320-detector row CT, the entire heart is imaged with temporal uniformity. Furthermore, if the x-ray beam is turned on for a longer period, the scanner can capture the heart over one or more cardiac cycles. This has been described as four-dimensional CT or volumetric cine imaging[42]. The temporal resolution of CT scanner reflects the ability to freeze cardiac motion, thus producing motion-free images. The 320-detector scanner has a standard temporal resolution of approximately 175 ms, half of the gantry rotation times. For patients with higher heart rate (> 65 bpm) and contraindications to β-blockers, multi-segment reconstruction can be used at the expense of higher radiation dose. For example, in two-segment reconstruction, data required for image reconstruction are acquired over two cardiac cycles. Therefore, only data from 90° rotation during each of the two cardiac cycles are used, improving the effective temporal resolution by a factor of 2[43].
Figure 4 3D volume rendering image acquired with 320-slice coronary computed tomography angiography in a single heartbeat shows excellent visualisation of coronary arteries and side branches without artifacts.
Results using 320-slice CCTA are compared favourably to the studies using 64-slice and DSCT coronary angiography[44,45]. van Velzen et al[44] in their recent study reported sensitivity and specificity of 100% and 85% for 320-slice CCTA in 106 patients with acute chest pain admitted to the Emergency Department. Pellicia et al[45] in their prospective study consisting of 118 unselected consecutive patients with suspected CAD demonstrated the excellent results with 320-slice CT, with more than 90% of sensitivity, specificity, positive predictive value and negative predictive value achieved at the per-patient, per-vessel and per-segment analysis. These results indicate that 320-slice CT has the potential to broaden the use of CCTA to more patients, such as patients with atrial fibrillation.
Two recently reported systematic reviews and meta-analyses further confirmed the high diagnostic accuracy of 320-slice CCTA[46,47]. These results also revealed that negative predictive value of CCTA was close to 100%, indicating the high value of 320-slice CCTA for excluding coronary artery stenosis. However, it has to be recognized that diagnostic performance of 320-slice CCTA is similar to that of 64- and 128-slice for the determination of ≥ 50% coronary artery stenosis due to its limited temporal resolution, despite improved extended z-axis coverage.
DIAGNOSTIC VALUE OF CCTA: CURRENT STATUS AND CHALLENGES
Despite promising results having been achieved with CCTA in coronary artery disease, it suffers from some limitations which affect its diagnostic performance to some extent. Artifacts (motion-related or due to severe calcification) represent one of the common limitations, although this is less commonly seen in CCTA performed with latest post-64 slice CT. Heart rate comprises another issue which needs to be addressed in the cardiac imaging, and temporal resolution of CCTA is still inferior to that of invasive coronary angiography.
Artifacts
Imaging coronary arteries using coronary CT angiography requires high spatial and temporal resolution, good low-contrast resolution, intravascular contrast enhancement and a short scanning time. Image artifacts are always associated with the limitations of either temporal resolution, or noise or the reconstruction algorithm in the scanner system. Images artifacts are mainly demonstrated as blooming, streaks, partial volume and motion artifacts. All these artifacts can arise from technical, operator, and patient errors[48].
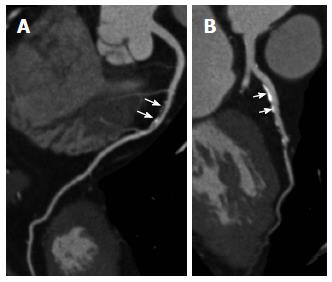
Stair-step artifact is the most common artifact that occurs in CCTA. Stair-step artifact occurs especially in patients with high heart rates, heart rate variability, and the presence of irregular or ectopic heart beats such as premature ventricular contractions and atrial fibrillation during image acquisition (Figure 5). It can be best recognized in a sagittal or coronal view. Therefore, beta-blockers should be used to lower the heart rate prior to the scan. Reducing this artifact is achieved by reconstructing the dataset at different phases of the cardiac cycle. In general, reconstructions for CCTA are performed in mid-diastole to late diastole (60%-70% of the R-R interval). However, because the duration of diastole decreases as the heart rate increases, an end-systolic phase reconstruction at 25%-35% of the R-R interval might be considered for image processing[48].
Figure 5 Prospectively electrocardiogram-triggered coronary computed tomography angiography and coronary arteries.
Prospectively ECG-triggered coronary computed tomography angiography curved planar reformatted images show right (A) and left (B) coronary arteries with blurred borders due to motion artifacts. ECG: Electrocardiogram.
Heart rate
Heart rate variability is another limitation for the prospectively ECG-triggered technique. Heart rate variability of > 5 beats/minute is considered not applicable for prospective triggering. Therefore, the scan has to be reverted into retrospective ECG gating technique if patients’ heart rate elevated or heart rate variability does not meet the requirement after β-blocker has been given[49]. However, precautions have to be taken in patients who are contraindicated to β-blockage agent. Alternatively, calcium channel blocker could be used in order to reduce the heart rate. The maximum of 15 mg of intravenous metoprolol (β-blocker) or 40 mg of intravenous diltiazem (calcium channel blocker) is recommended prior to the scan in order to control the heart rate[50,51].
However, the prospectively ECG-triggered technique in patients with higher heart rates still produces diagnostic images. CT scanner with higher detector arrays is an alternative option in patients with high or irregular heart rates. It has been reported that high diagnostic value could be achieved with 320-slice CCTA in the diagnosis of CAD, with image quality independent of heart rate[42]. The increased longitudinal coverage scan value (up to 160 mm) of 320-slice CT results in improved image quality within a wide range of heart rates; thus providing the opportunity to image patients with higher heart rates without requiring pre-examination beta-blockage[42,44,45].
Coronary CT angiography is most commonly performed in the spiral acquisition mode with continuous acquisition of data throughout the cardiac cycle. Multiple reconstruction parameters determine the quality of the reconstructed axial images. Images are usually reconstructed with a slice thickness of 0.5-0.6 mm, 50% overlap between images (0.4 mm increment), and a pixel matrix of 512 × 512. Although a thinner slice improves the resolution of the 3D dataset and the quality of reconstructed images, it comes at the cost of increased image noise, which can significantly limit the diagnostic assessment of the coronary arteries in patients with body mass index of greater than 30 kg/m2[52].
Temporal resolution
In single-source CT, improved temporal resolution is obtained at the expense of limited spiral pitch and correspondingly increased radiation dose to the patient. For a single segment reconstruction, the table has to travel slowly in order to ensure that each z-position of the heart is visualized by a detector slice during each phase of cardiac cycle. Therefore, the patient’s heart rate would determine the spiral pitch (if the heart rate goes up, the spiral pitch can be increased). Moreover, if multi-segment reconstructions are applied at higher heart rates to improve temporal resolution, the spiral pitch has to be reduced again. For example, each z-position of the heart has to be visualized by a detector slice during two consecutive heart beats in a 2-segment reconstruction; and three consecutive heart beats for a 3-segment reconstruction; and so on. In general, manufacturers of single-source CT scanners recommend an adaptive approach for ECG-gated cardiac scanning which the pitch of the ECG-gated spiral scan is kept constant at a relatively low value between 0.2 and 0.25. Therefore, more segments are used for image reconstruction at higher heart rates to improve temporal resolution[53,54].
Using a DSCT system, a temporal resolution of a quarter of the gantry rotation time is achieved, resulting in high temporal resolution of 75 ms, independent of the patient’s heart rate.This shows a significant improvement in cardiac imaging. However, the temporal resolution is still inferior to that of invasive coronary angiography, which is 10 ms, therefore, aggressive approaches such as heart rate control with the use of beta-blockers are necessary in CCTA examinations.
Excellent spatial resolution of 0.4 to 0.5 mm is achieved with latest CT models, however, this is still not comparable to that of invasive coronary angiography, with spatial resolution being 0.1 and 02 mm. Although CCTA enables excellent visualization of main and side coronary arteries, identification and characterization of coronary plaques[55,56], differentiation of lipid-rich content from fibrous component with CCTA remains difficult and challenging due to overlap in the attenuation values of lipid and fibrous tissue[57].
Severe calcifications
Vessel calcification poses a serious challenge to the accurate assessment of coronary artery lumen. Calcium deposits have CT attenuation which is similar to metal density and thus overwhelm the density of other tissues in the same voxel. Beam hardening is due to the attenuation of low-energy X-ray by very dense structures such as calcium. A higher energy beam, causing a darker appearance that can be mistaken for plaque, therefore penetrates adjacent pixels. All these effects can be modified, but not eliminated, by the smaller voxel size produced by the 64-slice scanner. The efficacy of this scanner in ameliorating imaging difficulties is shown in an overall sensitivity of 95% and specificity of 90% for the detection of angiographically significant stenosis even in the presence of high coronary calcium scores (Agatston score of > 400)[58] (Figure 6).
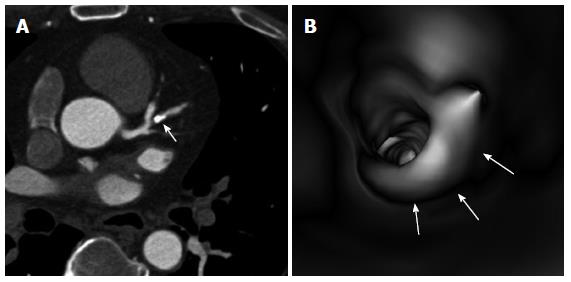
Figure 6 A severely calcified plaque is present in the left coronary artery (ramus intermedius) (A, arrow), with impression of more than 90% lumen stenosis on a 2D axial image.
Corresponding 3D virtual endoscopy views shows the intraluminal protrusion sign due to presence of plaque (B, arrows), but with less than 60% lumen stenosis.
A study by Brodoefel et al[58] compared overall calcium burden and studies the effects of calcium on image quality and diagnostic accuracy. Their results showed that dual-source CCTA was affected by calcification in terms of image quality and diagnostic value. Furthermore, a total of 100 (8.1%) segments that were considered non-diagnostic because of abundant calcification suggest that calcium burden remains a fundamental problem of coronary CT angiography and is certainly not addressed by exclusive increase of temporal resolution. In fact, from the linear regression analysis[59], there is a persistent threshold for adequate image quality at an Agatston score around 400. This is supported by reports from Diederichsen et al[59] and Chen et al[60] who also concluded that the specificity of CCTA was decreased significantly in patients with high calcium score > 400. However, Stolzmann et al[61] stated in their study that CCTA had high diagnostic accuracy despite the presence of heavy calcifications with sensitivity and specificity being 99% and 99% in patients with median CAC score < 316, and 98% and 99% in patients with median CAC score > 316. Despite these promising results, further studies on 256- and 320-slice CT are needed to evaluate the diagnostic performance of CCTA in patients with high coronary calcium score.
PROGNOSTIC VALUE OF CCTA
CCTA allows for visualization and characterization of coronary plaques, thus, it can detect non-obstructive and non-calcified plaques as well as plaques with positive modelling, both of which play an important role in the pathophysiology of acute myocardial infarction and may be indicative of vulnerable plaques. Studies based on single centre experiences have demonstrated that CCTA provides prognostic information for predictive adverse cardiac events in patients with known or suspected CAD[62-65]. Ostrom et al[66] demonstrated a correlation between mortality and the number of involved vessels for both nonobstructive and obstructive coronary lesions. Min et al[67] reported that coronary segments with presence of plaque, regardless of stenosis severity, had a particularly good correlation with patient survival.
Prospective large and multi-centre trials evaluating patients presenting to the emergency department with acute chest pain symptoms further confirmed the prognostic value of CCTA. In a 2-year follow-up of the ROMICAT trial, Schlett et al[68] evaluated the prognostic value of CCTA for major adverse cardiac events in 333 patients with a mean follow-up of 23 mo. Their results showed that in acute chest pain emergency patients, CCTA provided incremental prognostic value beyond clinical risk score in predicting major adverse cardiac events with absence of CAD leading to a 2-year cardiac events free warranty period, while coronary stenosis with regional wall motion abnormalities associated with highest risk of major cardiac events. Results from the international Coronary CT Angiography Evaluation For Clinical Outcomes: An International Multicenter Registry consisting of 20299 patients have further reaffirmed the predictive value of segmental plaque burden above and beyond the degree of stenosis[69]. A predictive score combining CCTA parameters with clinical information has been demonstrated to significantly improve prediction compared with well-established clinical risk scores.