INTRODUCTION
Peripheral artery disease (PAD) is caused by narrowing of the peripheral arteries typically due to atherosclerosis, which instigates restriction of blood flow to the distal extremities. Risk factors for developing atherosclerosis include smoking, diabetes mellitus, obesity (body mass index > 30), increasing age, hypertension, high cholesterol levels, raised homocysteine levels, and family history of cardiovascular disease[1,2].
PAD affects almost 200 million people globally[3] with patients either being asymptomatic or presenting with claudication, critical limb ischemia (CLI), or acute limb ischemia (ALI)[4,5]. It has been shown that PAD-affected patients have an increased risk of coronary artery disease (CAD) mortality, cardiovascular mortality, and all-cause mortality[6]. Additionally, when compared to patients with symptomatic CAD, patients presenting with CLI can demonstrate higher mortality rates[7]. Diabetes mellitus is another recognized risk factor for CLI correlated with increased lower-limb major amputation and mortality rates. A recent large retrospective analysis of the 2002–2015 National Inpatient Sample database demonstrated a significant increase in hospitalization for CLI for subjects with diabetes in the United States but a decreased in-hospital mortality over time, correlated with revascularization[8]. As a result, PAD management is critical to improve patients’ long-term survival and decrease mortality, with management varying depending on symptoms and severity of the disease. Aggressive risk factor modification is vital to improve cardiovascular risk. Lifestyle changes, pharmacotherapy, exercise therapy, endovascular, and/or surgical intervention are further available treatment options[9]. Notably, endovascular interventions for PAD treatment have significantly increased in recent years, significantly outnumbering surgical procedures[8,10]. Amongst other factors, this could be due to the high technical success, low complication rates, and shorter hospitalization stays observed with endovascular treatments but also because many PAD patients either do not have surgical options or are deemed unfit for surgery[11-13].
Over the past few years, several beneficial devices have been introduced in everyday clinical practice to improve outcomes of endovascular procedures used for the management of PAD. Yet, for the functional assessment of these outcomes, the same tests that have been used in the past decades [e.g., intraprocedural morphological assessment with digital subtraction angiography (DSA), ankle-brachial index (ABI), pressure measurements across the treated lesion] are still being employed. Nonetheless, these methods only allow an indirect evaluation of the outcomes of revascularization procedures for the treatment of PAD, while an objective quantification of limb perfusion is not feasible. For example, since angiography only demonstrates post-intervention vessel patency, it is unable to accurately estimate actual limb perfusion and hence is incapable of quantifying treatment outcome. Therefore, for the appropriate assessment of endovascular procedures in the management of PAD, there is a significant necessity for objectively measurable procedural outcomes of limb perfusion.
The purpose of this review is to familiarize readers with currently available perfusion-assessment methods and to evaluate possible prospects in quantifying and optimizing endovascular outcomes in the treatment of lower limb arterial ischemia.
STANDARD ARTERIAL PRESSURE MEASUREMENTS
Systolic blood pressure (SBP) measurements throughout the vascular tree offer valuable information in PAD diagnosis.
The ABI is a readily available, fast, inexpensive examination[14] that calculates the ratio produced by dividing the SBP measured at the ankle with the higher SBP measured at either arm. Ankle pressure and ABI should be calculated with the patient lying flat in the supine position after a 5 to 10-min rest[15]. Values most considered as indicative of PAD are those ≤ 0.90. More importantly, in patients with signs of ischemia, an ABI < 0.90 is considered diagnostic for PAD with sensitivity and specificity reaching 95% and 100% accordingly[16]. Furthermore, an ABI < 0.50 has been linked to more severe CAD and increased mortality[17]. On the other hand, values between 1.0 and 1.4 are considered normal, while values > 1.4 are attributed to non-compressible arteries and are therefore considered non-diagnostic[15,18]. As heavily calcified, non-compressible vessels are very common in patients with diabetes mellitus and chronic kidney disease; ankle pressures and ABI values are often untrustworthy and rendered non-diagnostic in such patients because they can be falsely increased[18,19].
Toe-brachial index - Toe systolic pressure
Great toe pressures and measurements of the toe-brachial index (TBI) can prove superior to non-diagnostic, high-value ABIs in these patient groups demonstrating severely calcified vessels, because the digital arteries are typically spared, thus being able to more accurately determine vascular disease[19,20]. A TBI ≤ 0.70 is abnormal and considered diagnostic of PAD[21], and a TBI < 0.11 is related to ischemic rest pain[22]. However, since toe pressure evaluation between studies demonstrates variable diagnostic accuracy, there has not yet been a widely accepted TBI or toe pressure threshold that can confirm the diagnosis of CLI[23].
Limitations
The ABI may not prove useful in the setting of severely calcified, non-compressible vessels. Moreover, false-negative ABI measurement can occur in up to 17% of diabetic patients and 24% of hemodialysis patients[24,25]. Nonetheless, there are several additional significant limitations to the employment of arterial pressure measurements. Amongst other considerations, some of the technical factors that should be thought through when calculating the ABI include correct cuff placement (2 cm above the medial malleolus)[26], use of the appropriate pressure cuff size (width should be at least 40% of the limb circumference)[27,28], use of a suitable Doppler probe (8-10 MHz), and correct probe-to-skin surface angle (45-60°)[15]. Additionally, the potential risk of thrombosis does not allow segmental pressure measurements over formerly placed stents or bypass grafts. Moreover, the utilization of arterial pressure measurements can be limited in patients with CLI since placing the cuff over an affected limb will often prove intolerable and painful. Nevertheless, in the presence of open wounds and ulcers, cuff placement may also pose the risk of contamination, rendering ankle or toe pressure measurements inapplicable depending on wound/ulcer location. In the same setting, arterial pressure measurements will not be applicable in cases of prior amputation.
Reed et al[29] showed that a post interventional ABI increase of ≥ 0.23 can augment wound healing and potentially reduce the necessity for a subsequent angioplasty, while a post endovascular revascularization TBI increase of ≥ 0.21 is correlated with reduced major adverse limb events and with improved wound healing. Also, Decrinis et al[30] found that a post-angioplasty ABI increase of 0.10 and 0.15 can predict no residual > 50% stenosis, with sensitivities of 79% and 67% and specificities of 92% and 100%, respectively. However, other studies show that ABI correlates poorly with Rutherford classification and the angiographic vessel runoff and that it may not adequately predict wound healing[31,32].
Notably, the ABI can only imply the approximate site of stenosis or occlusion, but it cannot determine its exact location since it could also reflect changes elsewhere in the arterial tree[33]. Due to this fact it cannot accurately differentiate graft failure from PAD progression, hence rendering its accuracy in predicting revascularization failure poor. Considering the above, ABI alone is not a dependable method of post-revascularization follow-up. Lastly, arterial pressure measurements such as ABI and TBI can only assess tibial or toe pressure, respectively, but are not able to quantify oxygenation and tissue perfusion. Hence, they only indirectly evaluate treatment outcomes.
Overall, post-procedural foot perfusion outcomes cannot be accurately quantified using ABI, as the existing data does not consider ABI a sufficient predictor of post-procedural results and relegate it a poor predictor of wound healing.
LASER DOPPLER SKIN PERFUSION PRESSURE MEASUREMENTS AND TRANSCUTANEOUS OXYGEN MONITORING
The first attempt to non-invasively assess post-revascularization limb perfusion was made using laser Doppler skin perfusion pressure (SPP) measurements and transcutaneous oxygen (TcPO2) monitoring.
Laser doppler skin perfusion pressure measurements
Laser Doppler measurement of SPP is a non-invasive technique that (by using a laser Doppler sensor) detects the movement of red blood cells (RBCs) following the slow release of the pressure occlusion cuff. In this way, SPP measurements calculate the capillary opening pressure and provide an indirect estimation of the microcirculatory flow status within the artery at skin level[34]. SPP is advantageous in that it is not altered by vascular calcifications[35]. Notably, SPP values > 30 mmHg after successful endovascular treatment with balloon angioplasty and/or stenting have been correlated with improved wound healing in a retrospective study of 113 consecutive CLI patients, with a sensitivity and a specificity of 81.4% and 69.2%, respectively[34]. On the other hand, if SPP values are < 30 mmHg, wound healing is doubtful[36]. More recently, in a large sub-analysis of 156 CLI patients included in a prospective multicenter registry, postprocedural SPP was significantly correlated with amputation-free survival, major adverse limb events, and wound healing at 1-year follow-up. Additionally, the authors reported that ABI did not correlate with clinical outcomes[37].
Transcutaneous oxygen monitoring
Transcutaneous oxygen (TcPO2) is a non-invasive test measuring oxygen concentration in the subcutaneous tissue 1 to 2 mm below the skin that can be performed at the bedside and does not use ionizing radiation. First, a conductive gel is applied over the area under examination, and subsequently, an electrode that can sense oxygen is placed on the affected limb. The electrodes heat the area beneath the skin, dilating the capillaries and allowing oxygen to readily flow to the skin, thus providing an optimal reading. The test serves as an indicator of oxygen and nutrients reaching the tissues through microcirculation[38], and since these are carried to tissues through the arteries, it can indirectly measure blood flow. Therefore, it can provide direct information about the endpoint of limb perfusion, which is tissue oxygenation. Normal TcPO2 values are between 50-70 mmHg.
Over the past years, several authors have investigated the role of TcPO2 in PAD patients, and sufficient data support its use both for PAD screening and post-revascularization success evaluation. Patients with CLI (ABI < 0.4) will almost always have TcPO2 values < 30 mmHg[39]. Moreover, it is a highly accurate and particularly valuable test in predicting wound/ulcer and amputation healing[40]. Specifically, values over 30 mmHg have been correlated with improved wound healing rates[41,42], while values < 30 mmHg have been associated with a reduced chance of wound healing[43,44]. Likewise, Andrews et al[45] found that a cut-off TcPO2 value of 38 mmHg had a sensitivity and specificity of 71% in predicting wound healing or failure.
When compared to the ABI, Pardo et al[38] found that an increase in TcPO2 measurements following endovascular procedures has more specificity and sensitivity compared to ABI, and hence may be a better alternative in the evaluation of angioplasty results.
In the setting of open wounds or prior amputations, where arterial pressure measurements are not applicable, TcPO2 and SPP measurements can be adequate alternatives. Additionally, since arterial pressure measurements cannot accurately identify the exact arterial disease location, these local perfusion tests may prove advantageous in angiosome-based revascularization[46,47].
Limitations
Despite published evidence suggesting the clinical utility of SPP and TcPO2, these techniques are not widely available and present several limitations. TcPO2 is only a skin perfusion marker, whose measurements are performed in a small area. TcPO2 results can be affected by numerous physiological, methodological, and technical issues. Perfusion assessment measures should be attained in a warm room to avoid cold-induced vessel constriction that could alter TcPO2 and SPP values[39]. Similarly, in patients exhibiting abnormal involuntary movements, reliable measurements will also not be feasible[48]. Other factors such as site selection, electrode equilibration, patient age, patient positioning, patient status before examination, pain, smoking and caffeine consumption, skin temperature changes, sympathetic tone, tissue edema, hyperkeratosis, cellulitis, and local skin integrity[35,39] may affect the accuracy of measurements, hence decreasing overall reliability. In addition, bony prominences, larger veins, or varicose veins should be avoided during cuff placement for SPP measurements, since they may produce artifacts[48].
Also, TcPO2 can calculate the partial pressure of oxygen adjacently to the wound but cannot determine actual measurements within the wound itself[49]. In the same manner, since measurements are relatively localized, values may not accurately represent total limb ischemia[39].
MATTERS THAT NEED TO BE ADDRESSED
The above-mentioned non-invasive techniques that are being employed in the assessment of limb perfusion estimate different perfusion characteristics, and each one of them has its advantages and disadvantages. For instance, ABI/TBI measures arterial pressures and hence cannot quantify tissue oxygenation and cannot provide sufficient information regarding wound healing and patient outcome. TcPO2 measures oxygen tension, while SPP measures capillary opening pressure. Although the latter methods are more reliable predictors of outcomes and offer a quantifiable outcome indicative of tissue perfusion, accurate tissue perfusion values cannot be obtained. Moreover, despite optimistic initial results, data regarding the role of SPP and TcPO2 in guiding or predicting endovascular treatment outcomes in PAD patients remain limited, and large prospective controlled studies are awaited.
NON-INVASIVE IMAGING STUDIES
Examples of non-invasive imaging studies include duplex ultrasound (DUS), computed tomography (CT) angiography (CTA), and magnetic resonance imaging angiography (MRA). DUS uses two modes of ultrasound, the B-mode that assesses vessel structure, and Doppler that evaluates blood flow velocity and direction. CTA/MRA provide information regarding the vessel’s anatomic relationship with other organs and provide information regarding vessel structure and morphology of stenosis. As a result, these methods can only measure lower extremity blood flow, and thus, are only surrogate tissue perfusion markers. Techniques that enable more accurate quantification of tissue perfusion include perfusion CT and arterial spin labeling (ASL) and blood oxygen level-dependent (BOLD) magnetic resonance imaging (MRI). However, these techniques are not employed regularly in the clinical evaluation of PAD.
CT perfusion
CT perfusion (CTP) has been readily employed in cerebral imaging, with its most well-known application being the non-invasive diagnosis of cerebral infarction/ischemia[50]. Similarly, Hur et al[51] demonstrated that CTP could be used to measure blood flow in lower limbs and also in the diagnosis of PAD by providing quantitative information regarding foot perfusion, while in the same study, color-coded perfusion map readings correlated well with both clinical and angiographic findings in PAD patients undergoing revascularization. Additionally, Sah et al[51] showed that CTP is feasible in PAD assessment and demonstrated a positive correlation between CTP blood flow, blood volume, and lesion length, and an inverse correlation between blood volume and ABI, suggesting that CTP may serve as a non-invasive technique, supplementing the diagnostic workup of PAD[52]. CTP examinations can be performed by contemporary CT scanners, as a supplement to CTA. Although CTP has the potential to quantify perfusion and monitor treatment response, it is a costly examination that necessitates the use of iodinated contrast and exposes patients to radiation.
Arterial spin labeling MRI
Arterial spin labeling (ASL) MRI is a non-invasive and non-ionizing perfusion measurement technique that allows measurements at the tissue level by exploiting the ability of MRI to magnetically label inflowing arterial blood before it enters the tissue under investigation (below the imaging slab)[53]. T1 signal differences between control and tag images are proportionate to blood flow and are used to produce perfusion maps[54]. With this technique, blood serves as an endogenous tracer, obviating the need for contrast agents.
Although initially developed and used to measure brain perfusion[55], ASL has found several clinical applications since. Notably, it is a reproducible, radiation-free technique that can successfully quantify peak exercise blood flow in calf muscles in patients experiencing claudication[56].
Limitations of ASL include low signal-to-noise ratio, relatively long scan acquisition times, patient movement artifacts, and errors owing to potentially slow blood flow, often seen in PAD/CLI patients.
Blood oxygen level–dependent MRI
BOLD MRI has been used for many years in functional MRI (fMRI) of the brain. This technique exploits the ability of fMRI to detect changes in the concentration of oxygenated and deoxygenated hemoglobin (Hb) due to a fundamental difference in their paramagnetic properties (deoxy Hb is paramagnetic while oxyHb is not). As a result, it uses the Hb saturation state as an endogenous tracer, hence also eliminating the need for contrast agents. Heavily T2* weighted sequences are used to detect such changes[57]. Even though BOLD imaging has been mostly used in fMRI to detect which parts of the brain demonstrate most activity, Bajwa et al[58] evaluated BOLD MRI as a clinical tool for the measurements of calf muscle perfusion in CLI patients. This study demonstrated statistically significant interuser and interscan reproducibility and found a significant correlation between MRI measurements and tissue vascularity found in muscle biopsy samples obtained in the scanned region.
Limitations of BOLD imaging include erroneous measurements potentially related to slow blood flow (as mentioned with ASL) often seen in PAD patients and susceptibility of T2* sequences to artifacts. However, when compared to ASL MRI, BOLD MRI seems to have a higher signal-to-noise ratio, faster acquisition times, and reduced motion artifacts[58].
Nonetheless, for the purposes of this review, the most important limitation of ASL and BOLD imaging is the fact that although they can be used to quantify post-procedural perfusion outcomes, they are impractical when physicians require real-time feedback during endovascular interventions.
INTRA-OPERATIVE TWO-DIMENSIONAL PERFUSION ANGIOGRAPHY
Neuro-interventional procedures associated with acute stroke treatment have effectively employed two-dimensional perfusion angiography (2D-PA) by using flat detector technology[59]. This application has improved acute stroke patient management by allowing cerebral blood volume (CBV) assessment and by predicting final infarct volume.
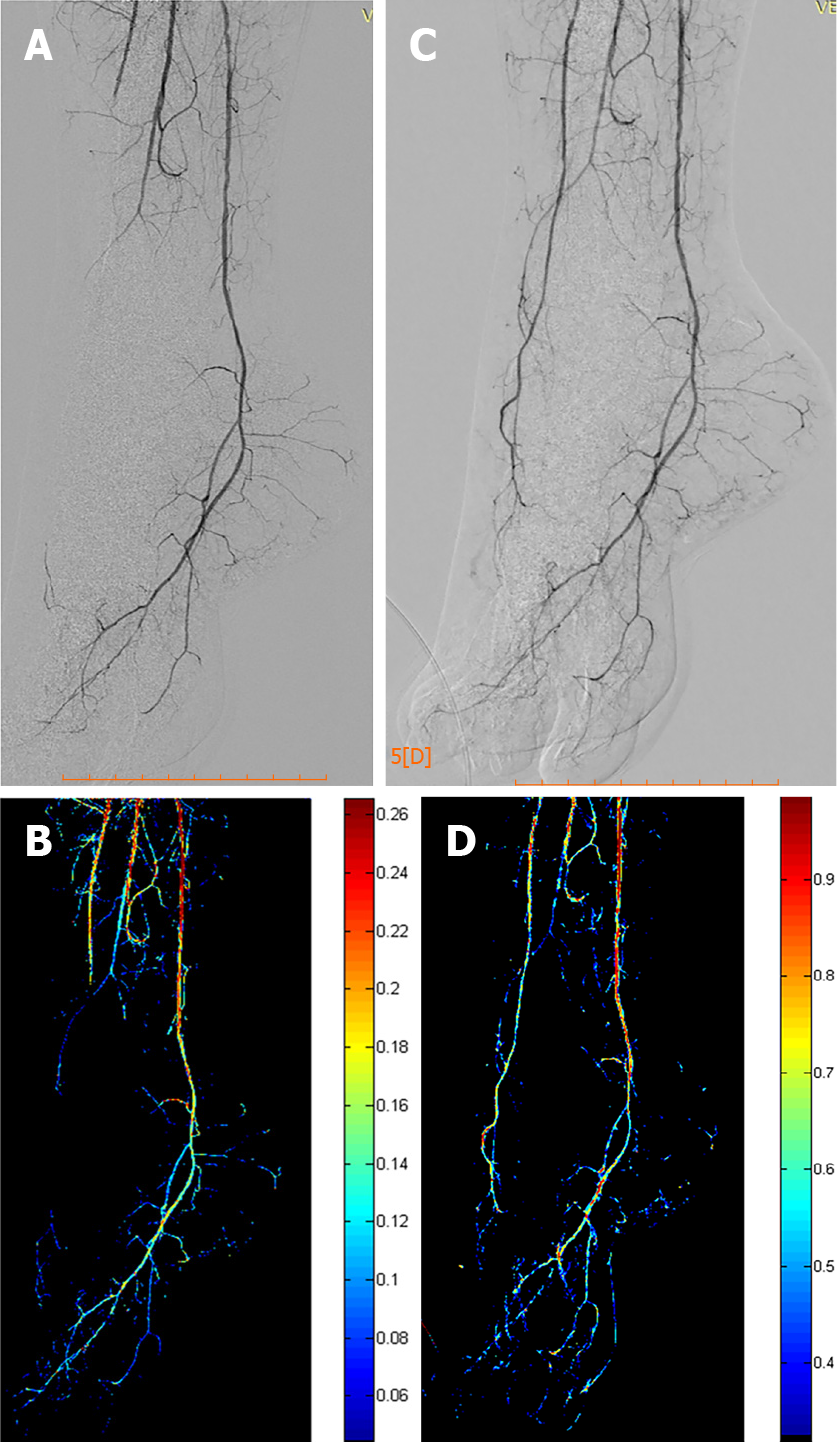
When applied in peripheral vessels, standard DSA is used, with a rate of three frames per second. The images that are attained are reconstructed with a post-processing software that calculates the changes in density per pixel before and after the endovascular intervention, and the results are then viewed on a dedicated workstation[60]. In a study conducted by Jens et al[60], the DSA data obtained during this technique were used to produce a time–density curve, which represented foot perfusion concerning time. Successful endovascular interventions performed on below the knee arteries in this study showcased improved perfusion curves, hence making functional imaging in CLI patients a more feasible goal. Moreover, another study conducted by Kagadis et al[61], using a custom-made, 2D perfusion DSA algorithm (Figure 1) demonstrated a post angioplasty mean transit time decrease that indicated increased tissue perfusion following successful revascularization. However, this study was limited mainly by the very small number of patients investigated.
Figure 1 Two-dimensional perfusion digital subtraction angiography algorithm.
A and B: Pre-procedural digital subtraction angiography (DSA) depicting a chronic total occlusion of the distal anterior tibial and pedal arteries with the respective perfusion blood volume (PBV) map; C and D: Post-procedural DSA after balloon angioplasty of the occlusion and corresponding post-procedural PBV map.
Overall, advantages of 2DPA include the fact that it does not require additional radiation exposure or additional contrast administration[62] and that it can be used instantly, while the procedure is being conducted to evaluate the interventional results[60]. This will allow physicians to make more informative and objective decisions on whether the undertaken treatment is sufficient or further improvements are required[62], while it could also aid in determining a meaningful, functional revascularization endpoint, beyond the anatomical endpoint assessed with standard intraprocedural DSA (less than 30% residual stenosis)[60]. However, acquisition of the appropriate software is required while obtained images are very susceptible to patient/limb movement-induced artifacts[60-62]. The fact that such perfusion software is currently incorporated in new Angiography systems (Siemens, Toshiba, Phillips) indicates that there is a recognized need to overcome standard anatomical imaging and progress to the era of real-time, functional, quantifiable imaging.
Nonetheless, additional studies are necessary to further validate this technique and determine its role in PAD/CLI-related interventions by comparing it to other available methods and correlating it with clinical outcomes.
IMPLANTABLE DEVICES - O2 MICRO-SENSORS
Implantable devices such as O2 micro biosensors may be advantageous because they can measure oxygen concentrations in tissues instead of vascular oxygenation. They can calculate tissue oxygenation in multiple areas and can potentially be utilized in monitoring tissues at risk, wounds, and reconstructive surgery. These phosphorescence devices are as small as 0.5 mm × 0.5 mm × 5 mm and can be integrated within the area/tissue of interest via a 16G or 18G needle[63]. Once positioned, these biocompatible hydrogel sensors remain in the body permanently, surpassing the foreign body response[64]. Subsequently, a reader is fixed on the skin above the microdevice, and then a LED source emits light into the skin above the sensor. Local tissue O2 levels will then be calculated by an external photodetector in the reader and can be continuously tracked. The first-in-man prospective, single-arm, observational study included 10 patients with CLI (four micro-oxygen sensors injected in the foot of each patient). The study demonstrated that injectable micro-oxygen sensors could effectively and safely calculate tissue O2 concentrations in CLI patients during their endovascular intervention and post-operatively for 28 days[63]. Moreover, a statistically significant postoperative increase in the concentration of oxygen was noted. Nonetheless, additional and larger studies are required to confirm the utility and applications of these devices.
MICROWAVE RADIOMETRY THERMOMETRY
Microwave radiometry (MWR) is a non-invasive technique allowing accurate temperature measurements of internal tissues. Based on the principle that radiation intensity is proportional to tissue temperature, this method detects internal tissue temperatures at microwave frequencies by using contrasting dielectric qualities of different tissues[65]. This technique uses an antenna containing a microwave and an infrared sensor, which is applied at a 90 angle over the tissue surface being examined, for an approximate duration of 8-10 s. To obtain measurements, the microwave emissions are converted into temperature by using a data processing unit and suitable software.
Tissue temperature measurements have previously been applied in the diagnosis of numerous entities[66] including the characterization of neoplastic tissue or inflammation of atherosclerotic plaques[67-69]. Guidelines for diabetic foot management suggest that pedal perfusion is evaluated by the combination of ABI, TBI, Doppler arterial waveforms, and TcPO2[70,71]. However, detecting the concomitant presence of vascular disease in diabetic foot ulcers remains a difficult and challenging task.
Spiliopoulos et al[72] conducted a study to investigate the ability of MWR thermometry to non-invasively differentiate CLI in patients with diabetic foot ulcers. This multicenter study including 80 patients demonstrated that the temperatures measured near ulcers were significantly lower in patients with CLI (either with or without diabetes) than in non-ischemic controls. Importantly, it showed that a cut-off temperature less than 31.8 °C was diagnostic of CLI with a sensitivity and specificity of 100.0% and 88.37%, respectively.
The above findings indicate that MWR thermometry could be employed in the differentiation of arterial ischemia in patients with diabetic foot ulcers. This thermometric technology is currently being studied for the quantifiable assessment of post revascularization foot perfusion to predict successful wound healing and initial data are awaited. Limitations of this modality include the fact that it needs to be performed in standard room temperatures and that in cases of overlying infection/inflammation, false-negative results may be obtained.
PERIPHERAL FRACTIONAL FLOW RESERVE
Fractional flow reserve (FFR) is an invasive method frequently employed in coronary artery revascularization to calculate pressure variations across a stenotic lesion[73,74]. It is a pressure ratio produced by dividing the mean pressure distal to the lesion with that proximal to the lesion. The cutoff FFR value is 0.80, with an FFR above 0.80 indicating non-significant stenosis and an FFR below 0.80 indicating a significant lesion[75]. Measurements are obtained via a small sensor/ transducer on the tip of the angiographic wire during pharmacologically induced hyperemia, which is achieved by injecting vasodilating agents. FFR allows the determination of whether certain stenosis is oxygen depriving, hence rendering it a valuable indicator of the stenosis’s functional severity and the necessity for coronary intervention[73].
Notably, according to a meta-analysis from Christou et al[76], the concordance of FFR when compared with non-invasive imaging conveyed a sensitivity and specificity of 76%, concluding that FFR results usually agree with non-invasive imaging studies, although their correlation can be deficient.
The use of FFR in PAD (peripheral FFR-pFFR) uses the same principles as the FFR in CAD. It has been recently employed in the evaluation of the physiological implications of iliac and superficial femoral artery stenosis[77,78] and also in CLI patients with severe below-the-knee arterial disease[79]. Hioki et al[77] found a significant association between pFFR and post-exercise ABI and noted a substantial pFFR improvement after endovascular procedures, while Banerjee et al[80] demonstrated an important correlation between walking impairment, pFFR, and rest and exercise ABI. Potential applications of pFFR include its intraoperative evaluation after apparently successful revascularization. Although FFR has been thoroughly investigated in CAD and is considered to be a valuable tool (particularly in the management of intermediate lesions), the use of pFFR has been limited, with available evidence-based data being sparse, and clinical efficacy remaining questionable warranting further investigation.
An advantage of FFR is that it is unaffected by heart rate and SBP changes and that it considers collateral flow[73]. The latter means that it could potentially render morphologically severe stenosis functionally insignificant. Moreover, FFR allows for simultaneous intraprocedural diagnosis and treatment with angioplasty and stenting. Importantly, FFR can have significant value in vessels with multiple stenotic lesions[81]. The pFFR can be obtained by 2D perfusion DSA and therefore implemented in real-time decision-making during peripheral angioplasty procedures. Nevertheless, its drawbacks include its invasive, time-consuming nature and the requirement for drug administration (vasodilating agents), which could cause important side effects[82].
INDOCYANINE GREEN FLUORESCENCE ANGIOGRAPHY
Indocyanine green (ICG) fluorescence angiography was initially used in retinal vessel imaging[83] and subsequently in the evaluation of tissue perfusion in various medical fields[84,85]. Following intravenous injection of the fluorescent ICG dye, the latter is activated by the use of near-infrared laser (approximately 780 nm)[84]. This procedure produces fluorescence that is subsequently detected and measured.
There is currently an insufficient number of studies that have applied this method in PAD patients. ICG fluorescence angiography perfusion measurements have been shown to predict the possibility of amputation healing[86] and have the potential to detect PAD patients with lower limb arterial occlusions who have developed extensive collaterals[87]. Additionally, it may permit real-time evaluation of flow in peripheral bypass and endovascular procedures[88]. In a retrospective study of 11 patients (13 peripheral bypass grafts), Yamamoto et al[89] performed quantitative near-infrared fluorescence angiography (NIR) using the fluorescence ICG to visualize blood perfusion and predict the patency of peripheral arterial bypass graft patency by measuring their fluorescence luminance intensities. Time-intensity curves of ICG opacification through the graft (Q graft) and distal host artery (Q distal) were assessed. Interestingly, increased Δ(Qgraft − Qdistal) and integral(Qgraft − Qdistal) quantitatively analyzed using NIR could predict anastomotic stenosis.
Igari et al[90] enabled peripheral blood circulation assessment during DSA in a retrospective study of ICG intraarterial DSA in 16 patients, (22 limbs) with PAD. The authors concluded that ICG evaluation during DSA might be used to assess peripheral blood flow during endovascular procedures.
ICG fluorescence angiography has the limitation of only being able to evaluate tissues up to 3 mm beneath the surface of the skin, hence only allowing microcirculation evaluation in the aforementioned regions. As a result, this technique may demonstrate inadequate efficacy in early PAD patients, because in this patient category microcirculation perfusion alterations will initially occur in deeper muscular tissue than at the skin, as seen in advanced CLI patients[84].
NEAR-INFRARED SPECTROSCOPY
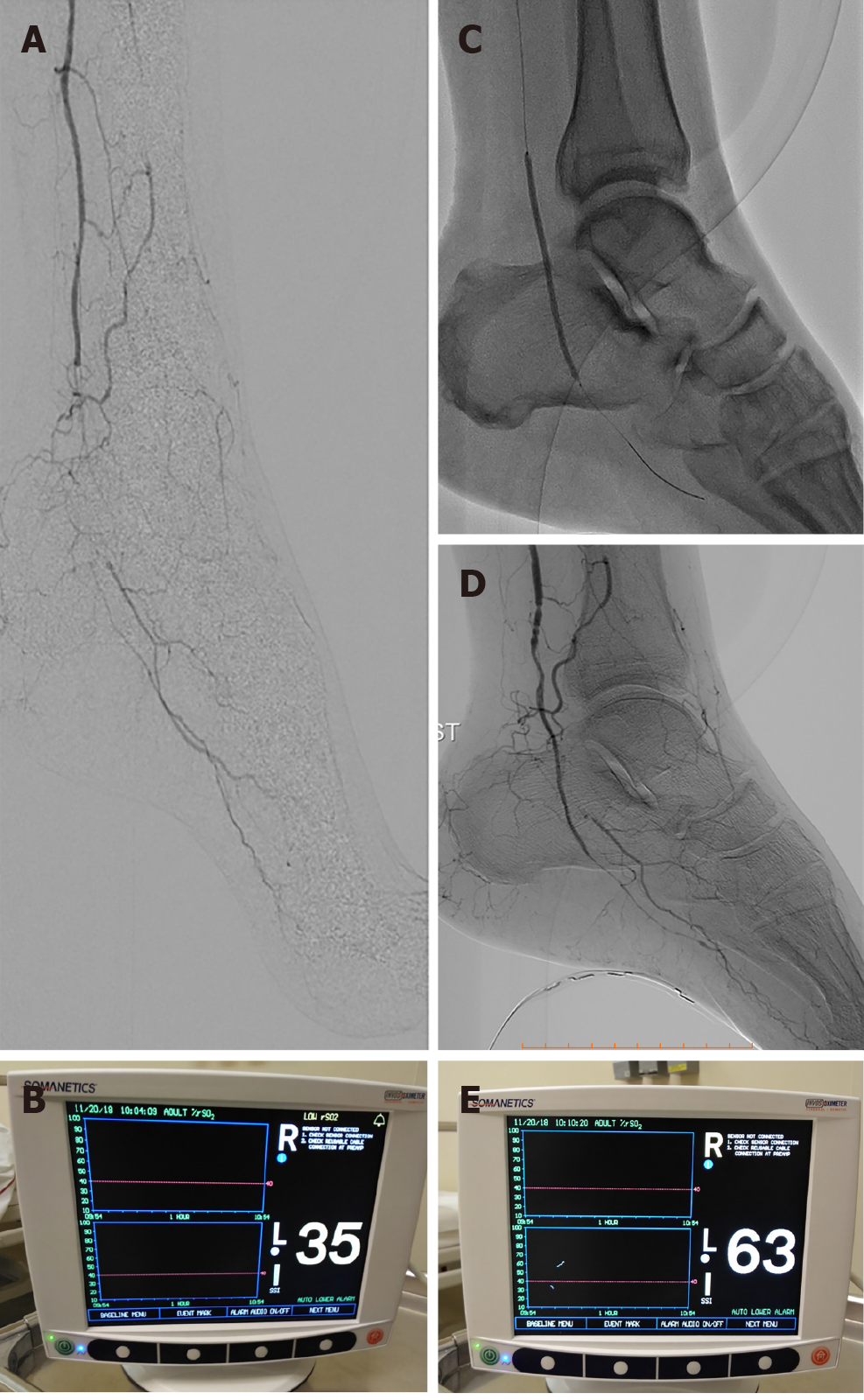
Near-infrared spectroscopy (NIRS) is a non-invasive method that employs near-infrared light to determine tissue oxygenation. It comprises a light source emitting near-infrared wavelengths and a detector that measures the reflected light after the latter has been absorbed and scattered by illuminated tissue being examined. According to the device being used, NIRS can illuminate tissue at depths between 1-3 cm. Variations in wavelength absorption reflect the concentration of oxygenated and deoxygenated hemoglobin, thus providing information regarding tissue oxygen saturation and, as a result, tissue perfusion (Figure 2)[91].
Figure 2 A case of 68-year-old male patient with insulin-dependent diabetes mellitus and advanced Rutherford-Becker class 6 gangrene of the left foot.
A and B: Digital subtraction angiography depicting a total occlusion of the distal posterior tibial artery and very low percentage value (35%) of regional hemoglobin oxygen saturation according to real-time near-infrared spectroscopy (NIRS) assessment of foot perfusion; C and D: Balloon angioplasty and final angiographic result demonstrating revascularization of the target occlusion. Note the NIRS electrode attached to the plantar surface of the treated foot; E: Immediately after revascularization, the percentage value of regional hemoglobin oxygen saturation was increased to 63%, demonstrating an 80% increase in foot tissue perfusion.
NIRS applications vary widely, including several utilizations in the field of medicine[92]. Amongst these, it has also been investigated in PAD where it appears to have the ability to be used as a complementary method in its diagnosis and evaluation of severity[93]. Importantly, studies from Mesquita et al[94] and Kagaya et al[95] revealed significantly lower tissue oxygen saturation values in PAD patients when compared to healthy patients, while a comparative study from Boezeman et al[96] found that using NIRS to monitor foot oxygenation (tissue oxygen saturation; StO2 in muscle tissue) in 14 CLI patients being subjected to EVT is a safe and feasible technique that can detect and record hemodynamic changes.
Limitations of NIRS include overlying infection/inflammation and the strong possibility of erroneous measurements when examining tissues with an abundance of overlaying fat[97]. Moreover, NIRS measurement results can be sensitive to oxygen delivery and extraction[98].
Nonetheless, additional studies are necessary to further clarify the role of NIRS in PAD, its intraprocedural applications, and its potential in predicting wound healing.
LASER-BASED IMAGING
Laser doppler imaging
Laser doppler imaging (LDI) is a non-invasive, non-ionizing, bedside-performed technique measuring blood cell speed and concentration. Similar to other methods, it also employs laser light, but in this instance, it is reflected and scattered by flowing blood cells, and then detected and converted into an electrical signal[99]. Subsequently, a color-coded perfusion image can be generated that may allow for a more accurate evaluation of the angiosome(s) associated with the disease[99]. Applied from a distance, LDI evaluates a larger area of skin by implementing a scanning motion that includes the whole leg[100]. Laser wavelength, scanning speed, and distance between scanner and area under examination can alter tissue penetration and hence should be unchanged to permit reproducibility of measurements and reliability in their comparison[101].
LDI has already found several clinical applications, including rheumatologic disorders, burn and dermal inflammation assessment, and assessment of wound healing and cutaneous ulceration[101]. The prospect of wound healing and cutaneous ulcer evaluation could prove valuable in PAD patients. Moreover, in the setting of PAD, quantifiable real-time increases in microcirculatory perfusion following EVT have been reported[102]. As a result, when using LDI, the operator can obtain real-time quantitative data regarding changes in target angiosome perfusion during the endovascular procedure, while it also has the potential to be proven valuable in the perioperative assessment.
Disadvantages of this technique include the low-depth penetration of laser light, the fact that measurements can be altered by temperature and vasoactive substance consumption, and the fact that measurements can be inconstant, rendering their serial comparison problematic[84].
Laser speckle contrast imaging
Laser speckle contrast imaging (LSCI) is a fast, inexpensive, and relatively simple imaging technique with the ability to produce 2D perfusion maps of large areas under investigation[103]. A typical LSCI comprises a laser light emitter and its diffuser, a camera with its lens, and appropriate processing software. When the tissue under investigation is illuminated with coherent laser light, it serves as a scattering medium and the backscatter produced causes a random interference pattern, which is called a speckle. This technique is based on the variations of backscattered light caused by the interaction of light with flowing RBCs[104]. LSCI is similar to laser Doppler in many ways, but it is still unclear if it measures flow or velocity[104]. Although originally used to measure retinal blood flow, LSCI has been employed in cerebral and skin perfusion assessment[105] and can generally be used in perfusion assessment of various tissues with many additional clinical applications (e.g., burn wound assessment and rheumatological disorders, to name a few)[104]. Moreover, LSCI has been shown useful in assessing diabetic foot ulcers owning to local ischemia[106], while - along with TcPO2 – it has also proven beneficial in the diagnosis of severe foot ischemia[107].
LSCI allows microvascular blood flow assessment owning to its high spatial resolution, while it has an added advantage of not necessitating direct contact with the tissue being examined to measure blood flow[105,107]. LSCI is mainly limited by its susceptibility to motion artifacts either due to patient breathing or movement[108]. As a result, addressing this issue is crucial in order to increase the usage of this technique in PAD/CLI patient management, where involuntary limb movements are often present.
Diffuse contrast speckle analysis
Diffuse contrast speckle analysis (DSCA) is a new non-invasive, non-ionizing optical modality that combines LSCI and diffuse correlation spectroscopy (DCS) in order to measure deep tissue blood flow by exploiting the sensitivity of the speckle contrast signal to RBCs movement[109]. DCS can probe deep tissue blood flow by monitoring light intensity fluctuations of the reflected diffuse speckle from tissue and can offer continuous blood flow monitoring, while LSCI (which has been analyzed above) is more suitable for shallow depths[109]. As a result, DSCA employs the deep tissue probing applications of DCS and the simple instrumentation and analysis of LSCI. The difference between DCS and DSCA is that with DSCA a specific region is imaged using a charge-couple device camera so that speckle contrast can be attained for a specific exposure time[110].
DSCA has found several applications in many fields since its development. Clinical applications include cerebral blood flow assessment[111], peripheral vascular health assessment for the evaluation of burns[112], and monitoring of foot tissue blood perfusion in diabetic patients with non-healing wound lesions undergoing vascular interventions[113].
Similar to other optical modalities, this technique has a limited depth penetration and, just as previously mentioned with LSCI, is susceptible to motion artifacts[111].
EMERGING DEVICES
Based on these latter technologies, novel perfusion devices have been available in the market, and others are currently under development. Recently, a real-time tissue perfusion device (PEDRA™ Xauron™ Perfusion System) has received Food and Drug Administration Breakthrough Device Designation for intraprocedural use during CLI treatment. It is a portable laser-based tissue monitoring system currently undergoing its first-in-man study. It provides real-time intraprocedural feedback on whether microcirculation has been improved post-intervention. As blood flows through the skin, the RBCs scatter the laser light, the amount of which is reflected on the monitor, hence quantifying perfusion. Data achieved with this system are claimed to be highly reproducible, however, further research on its clinical utility is required.
Additionally, the HyperView monitor (HyperMed Imaging; Memphis, TN, United States), which enables hyperspectral imaging for superficial tissue oximetry by measuring oxyhemoglobin, deoxyhemoglobin, and oxyhemoglobin saturation in superficial limb tissue, has recently received CE-Mark.
CONCLUSION
Current research validates the prospect of real-time, intraoperative, perfusion-guided revascularization techniques that will allow vascular experts to intraoperatively quantify and assess outcomes optimizing treatment. Such modalities have the potential to obviate misinterpretation of treatment outcomes and provide significantly improved clinical results. Also, novel four-dimensional angiography systems are expected to introduce state-of-the-art fusion imaging and real-time foot perfusion monitoring in everyday clinical practice soon. This will enable the use of novel tissue perfusion endpoints that could improve outcomes of endovascular treatment of arterial ischemia.
Therefore, the development of real-time tissue perfusion imaging and monitoring modalities that allow objective measurements and accurate quantification should be further investigated and validated by large, randomized trials.
Manuscript source: Invited manuscript
Specialty type: Radiology, nuclear medicine and medical imaging
Country/Territory of origin: Greece
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Taydas O S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Wu RR