Published online Aug 26, 2020. doi: 10.4330/wjc.v12.i8.373
Peer-review started: March 21, 2020
First decision: May 5, 2020
Revised: May 19, 2020
Accepted: July 19, 2020
Article in press: July 19, 2020
Published online: August 26, 2020
Processing time: 149 Days and 3.3 Hours
Heart failure (HF) is a complex clinical syndrome characterized by the activation of at least several neurohumoral pathways that have a common role in maintaining cardiac output and adequate perfusion pressure of target organs and tissues. The sympathetic nervous system (SNS) is upregulated in HF as evident in dysfunctional baroreceptor and chemoreceptor reflexes, circulating and neuronal catecholamine spillover, attenuated parasympathetic response, and augmented sympathetic outflow to the heart, kidneys and skeletal muscles. When these sympathoexcitatory effects on the cardiovascular system are sustained chronically they initiate the vicious circle of HF progression and become associated with cardiomyocyte apoptosis, maladaptive ventricular and vascular remodeling, arrhythmogenesis, and poor prognosis in patients with HF. These detrimental effects of SNS activity on outcomes in HF warrant adequate diagnostic and treatment modalities. Therefore, this review summarizes basic physiological concepts about the interaction of SNS with the cardiovascular system and highlights key pathophysiological mechanisms of SNS derangement in HF. Finally, special emphasis in this review is placed on the integrative and up-to-date overview of diagnostic modalities such as SNS imaging methods and novel laboratory biomarkers that could aid in the assessment of the degree of SNS activation and provide reliable prognostic information among patients with HF.
Core tip: Sympathetic nervous system activation is one of the key neurohumoral mechanisms that are operative in heart failure and is robustly associated with adverse myocardial remodeling, arrhythmias, sudden cardiac death, and overall poor prognosis in this population. Therefore, adequate diagnosis and quantification of the degree of upregulated sympathetic nervous system activity must be assessed by the clinician in every heart failure patient. A special emphasis must be put on adequate treatment by neurohumoral antagonists such as beta-blockers that will mitigate these adverse effects and improve outcomes. The adjunct use of advanced imaging methods and novel biomarkers might aid in clinical decision-making.
- Citation: Borovac JA, D'Amario D, Bozic J, Glavas D. Sympathetic nervous system activation and heart failure: Current state of evidence and the pathophysiology in the light of novel biomarkers. World J Cardiol 2020; 12(8): 373-408
- URL: https://www.wjgnet.com/1949-8462/full/v12/i8/373.htm
- DOI: https://dx.doi.org/10.4330/wjc.v12.i8.373
Heart failure (HF) is a complex clinical syndrome characterized by the symptoms such as breathlessness, fatigue and ankle edema and signs like elevated jugular venous pressure, lung crepitations during auscultation and peripheral edema[1]. The central hemodynamic consequence of HF is the inability of a heart to support required metabolic demands and perfusion of organs and tissues due to structural and/or functional cardiac abnormalities that predilect to decreased cardiac output (CO) and/or increased intracardiac filling pressures during the rest or exercise[2]. HF nowadays represents a relevant clinical entity and global pandemic that affects more than 26 million adults worldwide while its general prevalence in population accrues to about 2% with yearly incidence of approximately 0.2% in Western countries[3]. Projected burden of HF, assuming the stable incidence of this syndrome in persons ≥ 65 years was already surpassed by the actual burden of the HF in the United States and it is expected that more than 8 million people will have this condition in the United States by 2030[4,5]. This increase in HF prevalence observed worldwide might be attributed not necessarily to increased HF incidence but to phenomena such as advancing age of the population and increased comorbidity burden coupled with improved HF survival due to progress in HF treatment and diagnosis while the decreased incidence of HF according to some data might partially be the consequence of more efficacious treatment of acute coronary syndromes, lower severity of index HF events and improvements in HF primary prevention programs[5,6].
Despite the advancements in therapeutic management, HF is still characterized by the rather high morbidity and mortality rates and considerable healthcare expenditures while these outcomes appear to be strongly dependent on the region of the world, healthcare infrastructure and the level of quality/access to specialized HF care[5,7-10]. Of note, HF is at least as deadly or even deadlier than some of the common malignancies in both men and women. Among men, patients with HF had worse mortality outcomes than those with prostate and bladder cancer while among women those with HF had worse mortality outcomes than female patients suffering from breast cancer[11]. Survival after a diagnosis of HF has shown only modest improvement in the 21st century, with an increase in average survival rates between 6.4% to 7.2% during nearly two decades thus clearly indicating that our clinical efforts to improve outcomes in HF substantially lag behind advancements in other severe conditions such as cancer[12]. In support to this notion, a recent data from the United States nationwide temporal analysis showed that age-adjusted death rates for HF did not change significantly, in fact there even emerges a trend of the slight rise of HF-related mortality recently, after nearly 15 years of modest but gradual decline in HF-related mortality since the late nineties[13]. Similarly, recent longitudinal data acquired from the Framingham Heart Study and Cardiovascular Health Study showed that HF incidence was relatively stable for almost two decades and this was true for mortality outcomes as well (including cardiac death, non-cardiac death, and all-cause mortality)[14]. This study also showed that the incidence of heart failure with reduced ejection fraction (HFrEF) significantly declined whereas the incidence of heart failure with preserved ejection fraction (HFpEF) significantly increased over time in both sexes. Approximately 50% of community patients with HF nowadays have an HFpEF clinical phenotype while multimorbidity seems to be a stronger driver for HFpEF onset although it is a highly prevalent phenomenon in both HFrEF and HFpEF. Likewise, both phenotypes portend a comparable 5-year mortality[15-17]. Finally, the proportion of those dying of non-cardiovascular causes seems to be higher in HFpEF than HFrEF and this holds for non-cardiovascular-related 30-d readmissions that are more common among HFpEF compared with HFrEF patients[15,18].
Taken together, these recent trends strongly suggest that HF is a clinical entity that will continue to impose a significant burden on modern societies, urging for the advances in our understanding of its complex pathophysiology and development of new treatments. Equally important, the discovery and implementation of biomarkers that might aid in the diagnosis, prognosis, and risk stratification of patients with HF is required in a contemporary clinical practice[19]. For these reasons, aims of the present review are to provide recent updates regarding the HF pathophysiology with the special emphasis on novel biomarkers that might reflect the sympathetic nervous system (SNS) activation as one of the constituent neurohumoral pathways that are upregulated to preserve CO in the setting of a failing heart.
Any abnormality or combination of abnormalities that cause structural, mechanical, or electrical dysfunction of the heart carry the potential to induce HF. Most commonly HF is the consequence of the myocyte injury caused by coronary artery disease, uncontrolled arterial hypertension and diabetes mellitus, however, adverse myocardial remodeling can be triggered and sustained by valvular dysfunction, tachyarrhythmias (especially atrial fibrillation/flutter), interatrial and interventricular conduction abnormalities or pulmonary disorders such as chronic obstructive pulmonary disease or pulmonary arterial hypertension[20]. Less common etiologies include cardiomyopathies, myocarditis, infections, systemic toxins, and cardiotoxic drugs that are nowadays increasingly used in various chemotherapeutic regimens[21,22]. At least several pathophysiological mechanisms are at play in the setting of failing myocardium such as increased hemodynamic overload, ventricular dysfunction due to subclinical or overt ischemia, pathologic ventricular remodeling, upregulated neurohumoral activation, impaired intracellular calcium cycling and accelerated apoptosis of cardiac myocytes, imbalance in the formation and breakdown of the extracellular matrix, and various genetic predilections[2].
Clinically, a majority of patients with HF have both systolic and the diastolic dysfunction occurring at the same time and these two pathophysiological mechanisms often overlap but even in the isolation of each other, they cause a similar degree of HF signs and symptoms[23,24]. For the didactic purposes, in the systolic dysfunction, the primary pathomorphological substrate is the loss of functional myocardium (primary myocyte injury) most commonly due to ischemic disease and myocardial infarction causing impaired contractility and insufficient emptying of the ventricles consequently leading to increased left ventricular (LV) end-diastolic and end-systolic volumes and rise in end-diastolic pressure (LVEDP) within the left ventricle further decreasing stroke volume and left ventricular ejection fraction (LVEF)[25,26]. An increase in LVEDP might retrogradely increase left atrial (LA) pressure which consequently increases pressure in the pulmonary circulation, and if this cascade progresses even further can induce right heart failure, congestive hepatopathy and affect portal and peripheral circulation thus altogether precipitating fluid extravasation leading to pulmonary and/or splanchnic and peripheral congestion.
On the other hand, in diastolic dysfunction, the contractile ability of the heart might be preserved, however, functional mechanisms that are responsible for the adequate filling of the heart are impaired. It is estimated that up to 50% of patients presenting with signs and symptoms of HF have normal or near-normal LVEF but exhibit abnormalities predominantly in diastolic function[27,28]. Even more, those with normal LVEF by conventional transthoracic echocardiography and verified diastolic dysfunction can often have subclinical contractile dysfunction that is captured only by the means of myocardial deformation studies such as LV global longitudinal strain and speckle tracking techniques or advanced cardiac imaging methods such as cardiac magnetic resonance[29-31]. These filling abnormalities may occur due to impairments in early diastolic relaxation of the LV (an active energy-consuming process) and/or increased stiffness and rigidity of the LA and LV (a passive process independent of energy) coupled with reduced arterial compliance in both major vessels such as the aorta and peripheral arteries[32,33]. Among patients with HFpEF both processes of active relaxation and increased passive stiffness are impaired and are predominant pathophysiological mechanisms leading to diastolic dysfunction[34,35]. These abnormalities altogether act synergistically to produce a rise in the LVEDP thus causing significant venous congestion in HFpEF patients that is as severe as among those with HFrEF[36]. Of note, significant increase in passive LV stiffness is propagated by aberancies in collagen-dependent and titin-dependent deposition cellular pathways[35]. Similarly, longstanding elevated ventricular pressures further perpetuate LA dilation that is clinically detected as an increased LA volume at rest and reduced LA filling during submaximal exercise[37]. Also, peripheral oxygen extraction is blunted in HFpEF resulting in exercise intolerance while reduced peak oxygen uptake and increased perfusion/ventilation mismatch carry important prognostic information and assist in the selection of patients that might require advanced HF interventions such as heart transplantation or deployment of ventricular assist devices[38-41].
A complex interaction of highly prevalent comorbidities such as salt-sensitive hypertension, obesity, diabetes mellitus, metabolic syndrome, iron deficiency, chronic obstructive pulmonary disease and, atrial fibrillation (AF), combined with natural pathophysiological effects of aging can give rise to systemic proinflammatory state that affects coronary microvasculature and endothelium by upregulating cytokine-mediated inflammation pathways[42,43]. In this proposed pathophysiologic scheme, pioneered by Paulus and Tschöpe[44] in 2013, endothelial inflammation of coronary microvasculature acts as a central transitioning mechanism by which synergistic effects of comorbidities are translated onto heart thus causing secondary myocyte injury that ultimately leads to structural and functional alterations of the myocardium in HFpEF[44]. According to the postulated model, coronary microvascular endothelial inflammation reduces nitric oxide (NO) bioavailability and cyclic guanosine monophosphate (cGMP) content and reduces protein kinase G activity in adjacent cardiomyocytes thus highlighting NO-cGMP-PKG signaling pathway disruption as the key culprit in HFpEF pathophysiology. This disruption leads to the onset of cardiac hypertrophy and increased resting tension (Fpassive) of cardiomyocytes due to hypophosphorylation of titin and increased myocardial nitrosative/oxidative stress[45-47]. Furthermore, hypophosphorylation of constitutive myofilament proteins and increased calcium sensitivity of sarcomeres causes increased LV stiffness and abnormal relaxation contributing to HFpEF onset while these derangements are not present in normal myocardium[48]. Graziani et al[49] also proposed that microvascular dysfunction is the common pathophysiological pathway contributing to both microvascular angina and HFpEF[49]. Of note, endothelial dysfunction represents a pathological vascular phenotype of all systemic arteries that encompasses damaging effects of vasoconstrictive, prothrombotic and proinflammatory substances and mediators on the endothelial vascular lining and diminished repairability of endothelium thus further acting as an independent pathobiological driver of atherosclerosis and overt cardiovascular disease[50-52].
Furthermore, a novel pathophysiological concept of endothelial-to-mesenchymal transition has been recently proposed describing a process by which endothelial cells undergo a series of molecular events that lead to a loss of their endothelial properties and a consequent shift in phenotype toward mesenchymal cells such as myofibroblasts, smooth muscle cells, and osteoblasts[53]. Accumulation of these cells promotes plaque formation and atherosclerosis by secreting proinflammatory cytokines and metalloproteinases and increasing extracellular matrix and collagen deposition thereby affecting the structure and function of cardiac valves, native vein grafts that are used in coronary artery bypass graft surgery and inducing interstitial cardiac fibrosis, diastolic dysfunction, endocardial fibroelastosis and contributing even to the development of pulmonary arterial hypertension[54-59]. From the molecular perspective, it seems that activation of transforming growth factor-beta plays a key role in the initiation of endothelial-to-mesenchymal transition cascade and tissue fibrosis through its interaction with SMAD-2/3/4 and SLUG signaling pathways[57,60,61]. Furthermore, endothelial cells in which the EndoMT pathway was experimentally activated had significantly elevated secretion of proinflammatory cytokines such as interleukin-6, interleukin-8 and tumor necrosis factor-alpha thus likely representing an integrative pathophysiological cross-talk between fibrosis and inflammation[59,62]. In summary, EndoMT might be the key link in interaction between inflammation, endothelial dysfunction, and chronic cardiac fibrosis, and thus might become a viable target for novel therapeutic solutions for cardiovascular disease[63,64]. Altogether, these converging and mutually complementary pathophysiological mechanisms may contribute to a net effect of stiffening of cardiac myocytes and overt interstitial fibrosis thus directly inducing myocardial dysfunction during diastole and subsequent HF development.
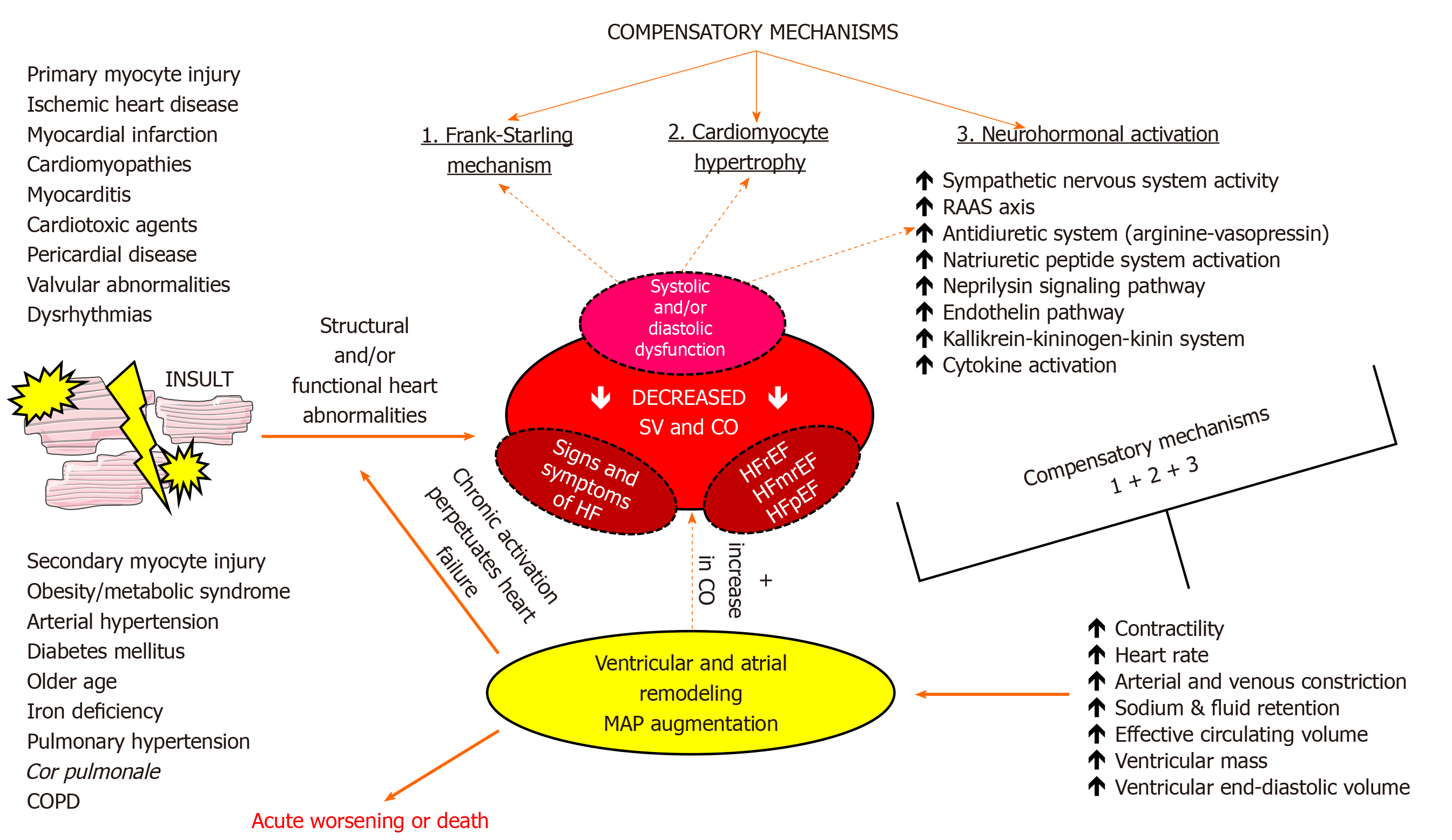
In order to maintain adequate tissue perfusion in the setting of the failing heart, several compensatory mechanisms are activated to increase CO via the Frank-Starling mechanism, increased ventricular volume and wall thickness through the process of ventricular remodeling and augmenting mean arterial pressure (MAP) by activating several neurohormonal pathways and cytokine systems[21]. These compensatory mechanisms are initially able to compensate for impaired myocardial function, however, they inflict deleterious effects on cardiac structure and function if chronically activated leading to further worsening of HF and progressive clinical deterioration of a patient. Neurohumoral systems that are upregulated act to promote beneficial short-term changes in heart, kidneys, and vasculature to maintain cardiovascular homeostasis[65]. They encompass the activation of the renin-angiotensin-aldosterone system (RAAS), arginine-vasopressin (antidiuretic) system, kallikrein-kininogen-kinin system, activation of natriuretic peptides system, neprilysin signaling pathway, endothelin pathway, and cytokine systems[66-70]. Finally, the upregulation of adrenergic/SNS pathways and blunted responsiveness of the parasympathetic nervous system (PNS), also collectively known as autonomic nervous system (ANS) imbalance, is one of the hallmark neurohumoral disturbances that are operative in HF and is of central interest in this review[71,72]. The summary of the most common etiologies, pathophysiological effects, and compensatory mechanisms in HF is shown in Figure 1.
It is worth of brief mentioning that evidence-based treatments such as angiotensin-converting enzyme (ACE) inhibitors, angiotensin receptor blockers, beta-blockers, mineralocorticoid receptor antagonists, angiotensin receptor neprilysin inhibitors, ivabradine, sodium/glucose cotransporter 2 inhibitors, digoxin, and cardiac resynchronization therapy (CRT) devices are developed around our understanding of compensatory and maladaptive mechanisms in HF[1,73]. Importantly, these treatment modalities were found successful in reducing mortality rates and hospitalizations in patients with HFrEF, however, no evidence-based pharmacologic treatments with clear beneficial effects on these endpoints were observed in patients with HFpEF while current guidelines stipulate symptom control with diuretics and efficacious management of comorbidities such as arterial hypertension, AF, obesity, and diabetes in this population[1,74].
Recent European Society of Cardiology and Heart Failure Association expert panel issued a scientific position statement in which ANS imbalance is recognized as an important contributor to cardiac disease progression and is designated as a prognostic parameter and a therapeutic target in HF by the means of novel pharmacologic and/or device therapies[75]. Furthermore, heart and brain are in bidirectional interaction meaning that depressed cardiac function affects cerebral structures and functional capacity while dysregulation of neuro-cardiac reflexes significantly affects the cardiovascular system thus aggravating and further sustaining the progression of HF[76].
In the advent of our understanding of HF, this syndrome was largely perceived as a hemodynamic disorder thus all treatment strategies were primarily directed toward the correction of hemodynamic abnormalities. However, since hemodynamic derangements could not fully explain the progression and long-term effects of the disease, a neurohormonal hypothesis was developed in which neurohumoral mechanisms encompassing RAAS and SNS activation were emphasized as independent drivers of cardiac dysfunction and progression of HF[77].
SNS activation is a fundamental physiological response to stress conditions (also known as the fight-or-flight response) such as hypovolemia, hypoglycemia, hypoxia or cardiovascular dysfunction[78]. SNS activity can modify and induce a wide spectrum of potent hemodynamic effects such as an increase in heart rate (positive chronotropic effect), augmentation of cardiac contractility (positive inotropic effect), accelerated cardiac relaxation (positive lusitropy), enhanced (shortened) atrioventricular conduction (positive dromotropy), reduced venous capacitance and peripheral vasoconstriction of resistance and cutaneous vessels[71,79]. The actions of SNS are dominantly mediated by secreted neurotransmitters such as norepinephrine (NE) that is released by sympathetic nerve terminals and, to a lesser degree, by the adrenal medulla and by epinephrine (EPI) that is chiefly released into peripheral circulation by the adrenal medulla.
Peripheral target organs are regulated by the two major sets of neurons serially connected to control the motor outflow of the SNS: (1) Preganglionic neurons that originate in the brainstem or the spinal cord; and (2) Postganglionic neurons that are part of sympathetic ganglia that are located outside of the central nervous system (CNS). Intrathoracic and extracardiac ganglia including stellate ganglia, middle cervical ganglia, and T2-T4 thoracic ganglia modulate the sympathetic outflow to the heart while sympathetic afferent impulses are carried through the dorsal root ganglia and reach the spinal cord, brain stem, and higher CNS centers. Cardiac sympathetic nerve fibers innervate myocardium at the subepicardial level, follow the path of major coronary arteries and are a predominant autonomic component in the ventricular tissue while parasympathetic nerve fibers, along with vagus nerve, run through subendocardium crossing the atrioventricular groove and are significantly more abundant in the atrial than ventricular myocardium thus exerting negative chronotropic effect with minimal effects on cardiac contractility[79]. Furthermore, sympathetic innervation has a relatively higher density in the anatomical areas around sinoatrial node and coronary sinus while its density gradually increases from the base of the ventricle to the apex (positive base-to-apex gradient)[80,81]. Intrinsic cardiac ganglia are located epicardially and receive innervation from post-ganglionic sympathetic and pre-ganglionic parasympathetic connections while most of sympathetic efferent and parasympathetic preganglionic fibers exhibit a large degree of intermixing thus most of the nerves reaching the heart in the mediastinum have mixed fibers (both sympathetic and parasympathetic components)[82].
The degree of SNS activation and sympathetic outflow to the heart and peripheral circulation, under physiological conditions, is regulated by a complex integration of autonomic cardiovascular reflexes. These reflexes include arterial baroreflexes, cardiopulmonary mechanosensitive reflexes, cardiac chemoreflexes, peripheral and central chemoreceptor reflexes, pulmonary stretch reflexes, cardio-cardiac reflexes and reflexes that are afferently projected from skeletal muscles[72]. All of these reflexes have a common role in fine-tuning and maintaining adequate heart rate, mean arterial blood pressure, vascular tone, ventilation, and respiratory drive in response to various hemodynamic changes[83]. These reflexes are listed with a summary of their function and potential impairment in HF (Table 1). According to modern pathophysiological findings, any depression of ventricular systolic function (irrespective of the underlying etiology) is augmenting cardiac reflex sympathoexcitation in chronic HF but might also be a leading culprit for the acute HF onset[72]. For example, a physiological response to the sudden increase in the cardiac filling pressures should act to vasodilate venous capacitance vessels to accommodate for excessive fluid, however, paradoxical sympathetic discharge in HF instead causes vasoconstriction of venous pool (mainly splanchnic circulation) and redistributes fluid to cardiopulmonary pool thus precipitating congestion and causing dyspnea. For this reason, it could be that rapid increase in the effective circulating volume from the mobilization of fluid from the splanchnic bed is the dominant driving force behind increased central venous pressure and congestion encountered during HF decompensation episode and might depend on an external fluid gain to a lesser degree[84,85]. Finally, cardiovascular-low threshold polymodal receptors are sensory endings localized in all cardiac chambers and large thoracic vessels that detect both mechanical and chemical stimuli and act in positive-feedback fashion with stimulatory effects on SNS[86].
| Type of neurally-mediated cardiovascular reflex | Proposed mechanism of action | Pathophysiological consequence in heart failure |
| Arterial baroreceptor reflexes | In HF acts as a response to perceived reduction in stroke volume or diastolic blood pressure; It is implicated that reduced carotid sinus and aortic arch afferent nerve firing as a response to systolic stretch disinhibits efferent sympathetic discharge; This reflex is impaired in terms of heart rate control, however, efferent sympathetic nerve activity might be preserved in human HF, even in advance stage | ↓ Reduced reflex vagal response; ↓ reduced heart rate variability; ↑ increased cardiac NE spillover; ←→no change in renal NE spillover; ↑mean sympathetic discharge to peripheral muscles is increased |
| Cardiac chemosensitive reflexes | Myocardial ischemia and reperfusion elicits increased sympathoexcitatory response by chemically (reactive oxygen species) stimulating sympathetic afferent fibers in both anterior and infero-posterior regions of the left ventricle; Platelet activation and local release of serotonin (5-HT) through a 5HT3 receptor mechanism and regional changes in pH from lactic acid stimulate sympathetic afferents in myocardium; Cardiac sympathetic afferent reflex is enhanced in HF and acts in the positive-feedback fashion | ↑ Increased shift and predominance of sympathetic efferent discharge; ↓ parasympathetic depletion; ↑ sympathetic activation; ↑ increased blood pressure; ↑ adverse left-ventricular remodeling; ↑ increased propensity for malignant arrhythmias and sudden cardiac death |
| Cardiopulmonary mechanosensitive reflexes | Normally elicited by the stretch of unmyelinated afferents sensitive to mechanical input, located intracardially and within pulmonary veins; It is implicated that impairement of this reflex decreases efferent sympathoinhibition to periphery; Cardiac-specific myelinated afferent are responsible for observed sympathoexcitatory effects characterized by the increased local cardiac NE spillover due to increased filling pressures (e.g. ↑ high LA pressure); Bezold-Jarisch reflex – mediated by nonmyelinated vagal afferent pathways – acts in sympathoinhibitory fashion and promotes reflex bradycardia, vasodilation and hypotension | ↓ Reduced cardiopulmonary reflex regulation of central sympathetic outflow to peripheral tissues (dominantly skeletal muscles); ↑ paradoxical excitation and increase in sympathetic outflow in the setting of high LA pressure |
| Cardio-cardiac reflexes | Coronary occlusion elicits the activity of preganglionic fibers in left thoracic sympathetic ramus communicans (T3) and increases discharge towards heart via efferent sympathetic innervation | ↑ Increased myocardial oxygen consumption; ↑ facilitation of malignant arrhythmias; ←→ might also have a protective effect in sense that they augment contractility, therefore, opposing ventricular dilatation and/or impending cardiogenic shock |
| Peripheral and central chemoreceptor reflexes | These receptors monitor partial pressures of oxygen and CO2 within arterial vessels and close to heart and escalate afferent sensory discharge according to changes; Peripheral chemoreceptors – dominantly respond to hypoxia; Central chemoreceptors – dominantly respond to hypercapnia; Peripheral and central receptor chemosensitivity is significantly increased in HF and is linked to augmented MSNA | ↑ Increased ventilation; ↑ increased sympathetic outflow; ↑ increased heart rate and systolic blood pressure; ↓ suppressed inhibition of sympathetic outflow that is mediated by arterial baroreflexes; ↑ increased peripheral and central chemoreflex-mediated sympathoexcitation is linked to poor 4-yr survival in HF patients |
| Pulmonary stretch receptor reflex | Fast and shallow breathing (high respiratory rate and low tidal volume) decreases stimulation of sympathoinhibitory reflex that is initated with lung stretch; HF patients with such breathing had increased MSNA burst frequency or amplitude; There is a correlation between decrease in resting tidal volume and attenuated sympathoinhibitory effect of lung inflation reflex with increased sympathoexcitation | ↓ Decreased the resting tidal volume; ↓ attenuated sympathoinhibitory effect of lung inflation reflex |
| Reflexes originating from skeletal muscles | Autonomic responses of skeletal muscles during exercise are modulated by skeletal ergo-receptors in order to optimize muscle work; HF patients had augmented afferent reflexes originating from skeletal muscles | ↑ Increase in the efferent ventilatory and sympathoneural responses to exercise |
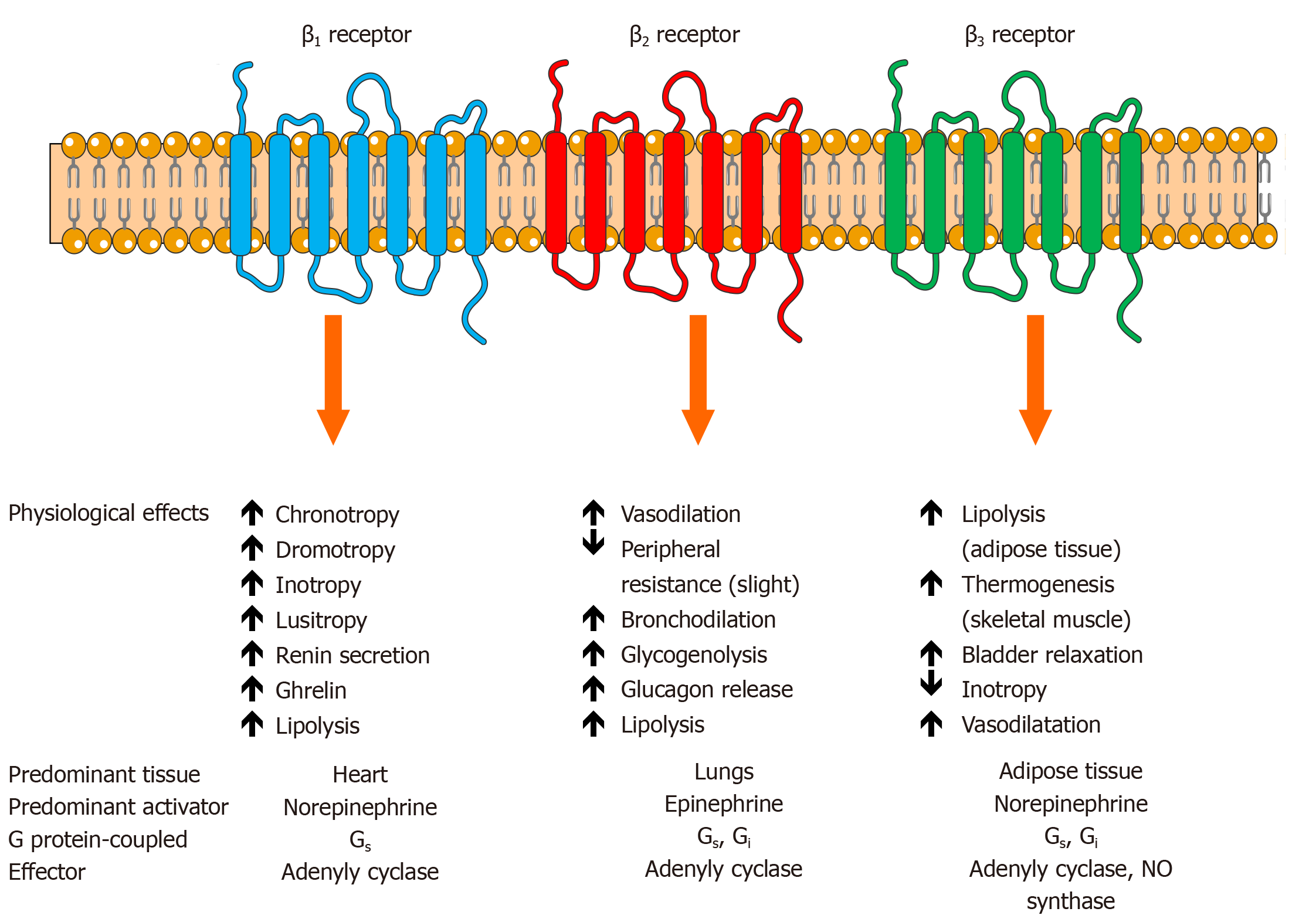
In terms of prinicipal neurotransmitters that propagate the effects of SNS, NE is ejected in the synaptic cleft upon the stimulation from stellate ganglions via post-ganglionic fibers thus activating adrenergic receptors (ARs) in the heart and physiologically augmenting contractile strength, chronotropy, dromotropy and increasing mean arterial perfusion pressure. About 80% to 90% of released NE is reuptaken by the noradrenaline transporter 1 which is a monoamine transporter that clears NE from sympathetic nerve terminals/chromaffin cells while about 10% to 20% of remaining NE content is spilled into circulation[87,88]. This NE turnover and metabolism can be evaluated with imaging methods such as scintigraphy by using radiolabelled guanethidine analogs of NE[89,90]. Similarly, sympathetic fibers that innervate the adrenal gland stimulate chromaffin cells in the adrenal medulla that act as modified post-ganglionic fibers to release catecholamines in response to stressors or exercise. This efflux of catecholamines from adrenal medulla is predominantly comprised of EPI (about 80%) while NE makes up the remaining 20% with small amounts of dopamine being released into peripheral circulation as well[91]. EPI and NE bind to specific ARs that are proteins embedded within the cell membrane with 7 transmembrane structures coupled to heterotrimeric G proteins. A total of two classes of ARs (alpha- and beta-adrenergic receptors) with 9 subtypes have been identified thus far: three α1 receptors, three α2 receptors and three β receptors (β1, β2, and β3)[92]. A healthy human heart mostly consists of β1 (75%-80%) and β2 (20%-25%) adrenergic receptors and they represent the key effectors behind positive chronotropic and inotropic effects of catecholamines while β3 adrenergic receptors (comprising less than 5% of total beta-receptor density) have been postulated to exert negative inotropic effects through upregulation of nitric oxide synthase pathway in human ventricle[93-95]. It has been recently confirmed that β1 and α1B receptors are present in all ventricular cardiomyocytes[96]. Alpha-1 adrenergic receptors (α1) and alpha-2 adrenergic receptors (α2) are chiefly expressed in vascular smooth muscle cells proximal and distal to sympathetic nerve terminals, respectively, and their activation elicits vasoconstriction of peripheral arteriolar and venous vessels while in the brain stem they modulate sympathetic outflow[97]. A recent study by Becker et al[98] showed that activation of neuronal endothelin B receptors can increase arterial blood pressure mediated through α1-adrenergic receptor signaling showing that abnormalities of endothelin system have a cross-talk with adrenergic systems in hypertension and HF[98].
Beta-adrenergic receptors act as powerful regulators of cardiac output and upon acute stimulation by catecholamines they facilitate fight-or-flight response while their chronic stimulation results in maladaptive and pathologic cardiac remodeling[99-101]. Activation of β adrenergic receptors induces the activation of the stimulatory G protein (Gs) which further activates adenylyl cyclase leading to an increase in levels of intracellular cyclic adenosine monophosphate and activation of protein kinase A that phosphorylates several target proteins within the cardiomyocyte such as phospholamban, L-type calcium channels, troponin I, contractile proteins, and the cardiac ryanodine receptor and this mainly is the mechanism by which β1 receptors regulate cardiac contractility/relaxation and heart rate[99,102]. Furthermore, activation of β1 receptors results in apoptotic and maladaptive remodeling signaling in the heart via protein kinase A-independent pathway mediated by Ca2+/calmodulin-dependent protein kinase II[99]. On the other hand, β2 receptors are distributed widely in the lungs, kidneys and blood vessels and possess a distinct function from the β1 subtype as they are coupled both to Gs and inhibitory G protein (Gi) in cardiomyocytes and their activation enhances cardiac function and myocyte viability[103].
Finally, β3 adrenergic receptors have a relatively minimal expression in the heart and they mediate unique downstream cellular effects once activated by catecholamines as they are mostly expressed in white adipose tissue where they mobilize stored fatty acids and regulate the release of adipokines while in brown adipose tissue they stimulate adaptive nonshivering thermogenesis[104]. A study by Napp et al[105] showed that β3 adrenergic receptors were mostly expressed in the endothelium of failing myocardium thus negative inotropic effect was most likely elicited by the NO liberation from the cardiac endothelial cells while β3 stimulation itself seemed to deactivate rather than activate endothelial NOS[105]. A study by Dessy et al[106] showed that β3 receptors are abundantly expressed in the microvasculature of human coronary arteries in which their activation caused vasodilatation through NO-dependent pathway and vessel hyperpolarization[106]. Of note, the expression of β3 adrenergic receptors in diverse cardiovascular pathologies seems to be upregulated and resistant to desensitization while in normal heart their activation resulted with a moderate negative inotropic effect[107,108]. Similarly, in septic cardiomyopathy, functional β3 receptors were upregulated and they increased negative inotropic response to β3 agonists[109]. Furthermore, in the setting of HF, activation of these receptors conferred beneficial effects with respect to excitation-contraction coupling and electrophysiological and mechanical remodeling of cardiomyocytes while also mediating vasodilatative pathways when β1 and/or β2 receptors are inoperative[110]. In the clinical and translational realm, relevant studies confirmed these initial findings as they showed that the third-generation beta-blocker, nebivolol, exhibited agonistic action on β3 adrenergic receptors in human ventricle thus providing evidence that highly selective blockade of β1 receptors coupled with NO-dependent endothelial vasodilatation and neoangiogenesis in coronary microcirculation could improve cardiac energetics[111,112]. Taken together, in the HF context, β3 adrenergic stimulation might confer cardioprotection by attenuating excessive catecholaminergic stimulation mediated by β1 adrenoceptors thereby presenting an attractive therapeutic target. The physiological effects of beta-adrenergic receptors are summarized and shown in Figure 2.
Finally, the expression of β-adrenergic receptors is physiologically modulated through G protein-coupled receptor kinases (GRKs), β-arrestins and complex intracellular signalosome[113]. GRK family consists of seven different protein kinases that canonically recognize and phosphorylate agonist-activated G protein-coupled receptor signaling and initiate downstream β-arrestin-mediated cellular pathways[114]. β-arrestins have a crucial role in the desensitization of activated seven transmembrane receptors such as β-adrenergic receptors, and they are key mediators of receptor endocytosis, ubiquitylation, and G-protein-independent cellular signaling[115]. Therefore, it becomes obvious that the normal expression of GRKs is a cellular prerequisite to maintain physiological homeostasis regarding β-adrenergic receptor turnover by phosphorylation, degradation or clathrin-mediated receptor downregulation and internalization[116].
A chronic SNS overactivity is one of the key pathophysiological mechanisms that are operative in HF. In the acute phase, this upregulated SNS activity is an essential compensatory response initiated in order to counteract reduced contractility, however, in the long-term, it becomes a major contributor to cardiac dysfunction as it promotes maladaptive cardiac hypertrophy and cell death.
In a seminal study performed more than three decades ago, Swedberg et al[117] showed that patients with chronic HF (CHF) had significantly higher arterial and coronary sinus venous NE concentrations compared to patients without HF while the net myocardial NE release in patients with CHF was about 20 times higher than that in patients without CHF[117]. This was subsequently confirmed by Viquerat et al[118] demonstrating that endogenous plasma levels of NE and dopamine were significantly higher among patients with CHF compared to patients without CHF thus reflecting enhanced sympathetic activity in response to failing heart[118]. Such overt sympathetic activity in HF closely paralleled increases in pulmonary artery pressures while activation of noradrenergic neurons in the brain might also be an underlying CNS mechanism of generalized sympathoexcitatory response observed in HF[119,120]. In fact, it has been shown that the RAAS axis is the major regulator of the SNS activity in the brain via angiotensin II type 1 receptors[121]. This likely occurs due to the upregulated expression of angiotensin II type 1 receptors (promoting sympathoexcitation) and decreased expression of angiotensin II type 2 receptors (promoting sympathoinhibition) in the rostral ventrolateral medulla[122]. Likewise, historical studies showed that 24-h urinary excretion of NE, EPI, and their O-methylated metabolites – normetanephrine and metanephrine was significantly higher in patients with congestive HF and reflected functional disease severity as assessed by the New York Heart Association (NYHA) class[123,124]. Furthermore, there is not only a significant increase in circulating catecholamines but there is also an augmented neuronal NE spillover due to increased cardiac sympathetic nerve activity (SNA) while renal SNA nearly reached its maximum in the state of HF and showed to be an independent predictor of mortality in HF[125-127]. Recent research efforts demonstrated that NE spillover does not only depend on increases in SNA but it also partially occurs due to mechanisms controlling NE release and reuptake in the synapse and these mechanisms seem to be deranged in HF[128]. Increased NE spillover is in most cases paralleled by the reduced neuronal NE reuptake thus higher net concentrations of NE are present in the sympathetic synaptic cleft which further desensitizes myocardial β-adrenergic receptors[129,130]. A study by Hasking et al[131] further showed that cardiac and renal NE spillover in subjects with congestive HF was increased by 540% and 206%, respectively, compared to patients without HF while adrenomedullary-mediated EPI spillover was also markedly increased among these patients[131].
As previously mentioned, in a failing human heart, an important pathophysiological characteristic is a decreased sensitivity of β-adrenergic receptors to catecholamines while β-receptors are downregulated and decreased in their density and quantity[132]. For example, β1 adrenergic receptors are reduced up to 50% in HF and there is a 200% increase in Gi-mediated cellular pathways with concomitant significant upregulation of GRK2 activity (also known as β-adrenergic receptor kinase 1 or βARK1) that further promotes adrenergic receptor internalization[133]. Myocardial GRK2 activity and expression have been increased in the failing heart as shown in several studies[134]. Conversely, experimental inhibition of βARK1 resulted in a marked reversal of ventricular dysfunction[135]. Finally, a wide variability of HF phenotypes and different response to HF treatment might suggest variants and functional polymorphisms of beta and alpha-adrenergic receptor genes[79]. Some pharmacogenomic studies suggested that polymorphisms in β1-adrenergic receptors might affect susceptibility to HF such as Gly389 allele and Gly389 homozygotes; improved response to β-blocker treatment among Arg389 homozygotes while none of the candidate polymorphisms was an independent predictor of prognosis in HF[136,137]. Likewise, specific β2-adrenergic receptor polymorphisms were linked with lower myocardial infarction rate and improved reverse left ventricular remodeling among patients with HF[138,139].
From the structural perspective, catecholamine spillover is cardiotoxic and its overexpression promotes senescence and inflammation of cardiomyocytes, upregulates tumor suppressor p53 pathway, and production of adhesion molecules by endothelial cells and macrophages and mediates cardiac dysfunction[140]. Chronic and persistent stimulation by catecholamines in HF causes interstitial fibrosis, myocyte hypertrophy, oxidative stress, and impairs the responsiveness and function of cardiac β-adrenergic receptors[141]. Engelhardt and colleagues experimentally demonstrated that increased chronic stimulation of β1 adrenergic signaling resulted in a significant cardiomyocyte hypertrophy and apoptosis resulting in a marked loss of contractility and progressive reduction of LVEF with histological and functional deficits typical of HF[142]. Catecholamine toxicity and generalized autonomic storm also have an important pathophysiological role in causing acute stress-related cardiomyopathies such as Takotsubo cardiomyopathy, acute LV dysfunction associated with subarachnoid hemorrhage, pheochromocytoma, and exogenous catecholamine administration as well as acute LV dysfunction in critically ill[143]. Contrary to this, activation of β2 adrenergic receptors delivered an antiapoptotic signal to cardiac myocytes through Gi-dependent coupling to phosphoinositol 3-kinase[144]. Furthermore, it seems that the number of β2-adrenergic receptors does not change significantly in HF[145]. These findings suggest that a fine balance between proapoptotic and antiapoptotic pathways initiated by differential adrenergic signaling is of fundamental importance for physiological cardiomyocyte function[146].
Importantly, studies have shown that the activation of SNS in the course of heart failure exhibits specific temporal dynamics and regional sympathetic profile. Rundqvist et al[147] showed that a selective increase in cardiac NE spillover (defined as increased amounts of NE at neuroeffector junctions) in patients with mild-to-moderate CHF was higher for more than a three-fold compared to healthy subjects while total body and renal NE spillover, as well as sympathetic outflow to skeletal muscles, were not different in HF patients compared to healthy controls[147]. This study clearly showed that in the early stages of HF, selective increase in cardiac adrenergic drive precedes generalized sympathetic hyperactivity and outflow towards the periphery (skeletal muscles and kidneys) which is characteristic of advanced HF. In the early stages of HF, such cardiac sympathoexcitation might trigger ventricular arrhythmias and is associated with poor prognosis[126,148]. Furthermore, local cardiac NE spillover might be the first component required for further β-receptor downregulation and depletion, adverse myocardial remodeling, depletion of NE stores, and impairment in G-protein signaling pathways, as discussed earlier. This might further drive hemodynamic deterioration and progressive LV dysfunction. Even more, blunted response and withdrawal of parasympathetic cardiac control seem to precede sympathetic activation during the development of HF. In support of this claim, in the tachycardia-induced model of HF, Ishise et al[149] showed that parasympathetic withdrawal occurs rapidly and correlates with the decline in LV contractility and plasma NE increased gradually as LV diastolic function worsened while all of these changes recovered toward baseline values once pacing was ceased[149]. The proposed mechanism was that depressed contractility resulted in the attenuated stimulation to the carotid sinus baroreceptor which diminished vagal efferent activity towards the heart thus demonstrating parasympathetic tonic withdrawal. Together, these findings suggest that in the course of SNS dysfunction in HF, sympathovagal imbalance might occur earliest as evidenced in parasympathetic withdrawal while sympathetic hyperactivity likely first occurs at the cardiac level before it is propagated to peripheral tissues and organs as observed in the advanced stages in HF.
Furthermore, dysfunction of cardiac reflexes is a hallmark of SNS hyperactivity in HF and it occurs to a similar degree regardless of HF etiology (ischemic or nonischemic)[150,151]. There is a diminished baroreflex sensitivity in HF characterized by the marked suppression of inhibitory SNS reflexes such as arterial baroreceptor reflex while excitatory SNS reflexes such as those fired from peripheral chemoreceptors are enhanced[152]. Floras et al[153] showed that a failing heart reacts to increased cardiopulmonary filling pressures through responsive and sensitive arterial baroreflex that elicits potent sympathoexcitatory hemodynamic actions[153]. Furthermore, even among patients with mild CHF, an SNS-inhibiting baroreceptor function is already significantly impaired thus implying that baroreflex dysfunction might be one of the earliest constitutive phases in SNS activation during the natural course of CHF[154]. Reduction in baroreflex sensitivity is even more severe if obesity and arterial hypertension are present among HF patients[155]. Conversely, baroreflex activation therapy in HF, encompassing the deployment of a device electrically stimulating carotid sinus, succeeded in improving muscle sympathetic nervous activity and relevant clinical indices thus showing that modulation of autonomic balance in HF might improve relevant outcomes[156,157].
Collectively, these findings are of clinical relevance because ANS imbalance and predominance of sympathetic excitation cause electrophysiological perturbations in the vulnerable cardiac syncytium and can initiate arrhythmogenesis[158]. For example, simultaneous stimulation of both sympathetic and parasympathetic systems can trigger AF while increased sympathetic stimulation is a contributing culprit to initiation of ventricular fibrillation (VF) or ventricular tachycardias (VT) or sudden cardiac death (SCD)[159]. Beat-to-beat variability of ventricular action potential duration is increased with elevated sympathetic activity in HF patients and might precipitate ventricular arrhythmias while beta-blocker, bisoprolol, attenuated these effects[160]. It was previously shown by Brunner-La Rocca et al[161] that high cardiac sympathetic activity in HF was an independent risk factor for sudden death, especially if sympathetic innervation was intact[161]. Sympathetic denervation in the heart combined with the presence of high NE levels is tightly correlated to progression of HF and SCD[162]. From the other way around, stellate ganglion blockade was effective in the acute reduction of ventricular arrhythmia burden and suppression of electrical storm thus clinically validating the concept that attenuation of sympathetic outflow to the heart from sympathetic ganglia can indeed mitigate the risk of future arrhythmic events[163-165]. These clinical observations were inspired by the previous animal study demonstrating that spontaneous high-amplitude discharge activity from left stellate ganglion was strongly associated with the induction of malignant ventricular arrhythmias[166]. Modern state-of-the-art neuromodulation strategies that are capable of mitigating VT/VF and atrial arrhythmias are, therefore, focused on increasing parasympathetic drive and inhibiting sympathetic neurotransmission[167,168].
Finally, it should also be noted that the widespread SNS activation also affects the function of skeletal muscles and promotes exercise intolerance in HF. Of note, diminished exercise capacity in terms of reduced peak oxygen uptake is present among HF subjects and is related to increased efferent sympathetic traffic to skeletal muscles, compared to control subjects[169]. This study also showed that resting muscle SNA is inversely related to peak oxygen uptake thus suggesting that there is a peripheral neurogenic limit to exercise in HF. As later validated, this reduced exercise capacity in HF is more dependent on sympathetic outflow to skeletal muscles than to cardiac sympathetic outflow, as assessed by NE spillover[170]. Furthermore, a subsequent study showed that muscle SNA was significantly higher while peak oxygen uptake was significantly lower in patients with ischemic vs nonischemic cardiomyopathy[171]. The most recent clinical study also demonstrated that the α-adrenergic-mediated vasoconstriction in HFrEF patients elicited a marked decrease in exercising skeletal muscle blood flow thus contributing to reduced exercise capacity in this population[172]. Finally, HF patients present with a high degree of chronotropic incompetence and attenuated heart rate response to exercise which is partially due to postsynaptic desensitization of the β-adrenergic receptor pathways[173].
A noninvasive in vivo imaging modalities can assess sympathetic innervation of the heart and for these purposes single-photon emission computed tomography and positron emission tomography (PET) are used by employing radiolabeled analogs of NE. The myocardial uptake of these radioanalogs dominantly represents presynaptic nerve function and their density in the heart. The most commonly used single-photon emission computed tomography tracer is 123I-metaiodobenzylguanadine (123I-mIBG) while most common PET tracer in clinical use is 11C-hydroxyephedrine (11C-HED)[174,175].
Recent studies demonstrated that impaired myocardial sympathetic innervation and regional sympathetic denervation, as detected by the presence of 11C-HED by PET imaging, were independently associated with grade 2-3 diastolic dysfunction and contractile dysfunction and fibrotic burden among patients with HFpEF, respectively[176,177]. Similarly, data from prospective HF cohort studies demonstrated that diminished 123I-mIBG uptake quantified as the reduced heart-to-mediastinum uptake ratio (H/M, indicating neuronal function including uptake and release of 123I-mIBG) or increased myocardial 123I-mIBG washout rate (indicating higher adrenergic drive) were strong markers of abnormal myocardial sympathetic innervation and consistent predictors of poor prognosis among patients with HF[90,178-180]. Furthermore, the ADMIRE-HFX study confirmed that H/M remained as a significant and independent predictor of all-cause mortality and the composite endpoint of death or death-equivalent events among nearly thousand NYHA II-III HF subjects during the median of 24 mo follow-up[181].
An elegant study by Wakabayashi et al[182] exploring 123I-mIBG kinetics in terms of underlying HF etiology showed that 123I-mIBG activity provided independent long-term prognostic information for both ischemic and non-ischemic etiologies of HF with lower H/M values having a greater impact on cardiac death among patients with ischemic compared to non-ischemic cardiomyopathy[182]. In concordance with such findings among HF patients with ischemic cardiomyopathy, 11C-HED PET-based studies revealed that regional myocardial sympathetic denervation and volume of denervated myocardium accurately predicted the risk of sudden cardiac arrest thus clearly correlating SNS innervation abnormalities with future arrhythmogenic events[183,184]. Similar findings were confirmed by another research group showing that denervated myocardium quantified using PET strongly predicted the risk of sudden cardiac arrest, independent of LVEF, infarct volume and other clinical variables among HF patients with ischemic cardiomyopathy and with LVEF < 35% that were eligible for implantable cardioverter-defibrillator device for primary prevention[185]. Finally, the most recent study conducted among patients admitted for acute decompensated heart failure and prospectively enrolled in the OPAR registry demonstrated that patients with cardiac sympathetic nerve dysfunction, defined as low late H/M, had a significantly greater risk of future adverse cardiac events, irrespective of clinical phenotype based on the LVEF values[186]. This study also showed that even a mild impairment in cardiac contractility (as shown in borderline LVEF values represented in HFmrEF cohort) was associated with sympathetic nerve dysfunction and was independently linked to poor outcomes thus suggesting that use of beta-blocker therapy in patients with HFmrEF phenotype is a viable pharmacotherapeutic option, as also supported by expert consensus statement and data from a large meta-analysis[73,187].
Taken together, these studies suggest that non-invasive cardiac imaging with norepinephrine analogs provides a reliable estimation of cardiac sympathetic nerve activity and this activity is strongly associated with clinical outcomes, regardless of clinical phenotypes or if HF is of chronic or acute onset. Such findings validate the concept that SNS overactivity is an important pathophysiological target in HF that must be efficaciously treated to improve outcomes and prevent sudden cardiac death.
Heart rate variability (HRV) is an established and widely used noninvasive method for the assessment of autonomic modulation of heart rate. It uses electrocardiographic (ECG) signal to measure subtle variations in the beat-to-beat heart intervals and is considered as a surrogate parameter of the complex interaction between CNS and cardiovascular system[188,189]. These periodic oscillations in heart rate signals are transformed into different frequency areas and their relative intensity is reported as a numerical value[190]. Briefly, low-frequency power (LF) and high-frequency power (HF), as well as the LF/HF ratio, are the most commonly used parameters in HRV analysis[189,191]. In most of the studies, HF power is regarded as a surrogate of PNS activity while LF power is modulated by both SNS and PNS. Likewise, high LF power values are associated with increased sympathetic activity while the LF/HF ratio reflects global sympathetic/vagal balance[191]. Generally, decreased HRV is associated with various pathologies and decreased life expectancy in several studies[188].
Regarding cardiovascular diseases, depressed HRV has been associated with autonomic neuropathy, heart transplantation, congestive HF, MI, and other incident cardiac conditions[192,193]. Most data for low HRV and increased mortality have been corroborated from studies investigating populations with cardiovascular diseases such as post-MI patients, patients with HF and those experiencing SCD, and in contrast to this, such associations of HRV were historically more diluted when it comes to risk stratification among the general asymptomatic population[194]. However, a recent study by Hillebrand et al[195] showed that low HRV was associated with a 32%-45% increased risk of a first cardiovascular event in populations without known cardiovascular disease[195]. In a similar fashion, abnormal HRV parameters were independently associated with incident CHF in asymptomatic older adults[196].
In the setting of a failing heart, HRV is significantly reduced in most patients and associated with the high risk of death due to progressive HF, SCD and syndrome severity[197-199]. Ponikowski et al[200,201] demonstrated that depressed HRV on 24-h ambulatory ECG monitoring was an independent risk factor for poor prognosis in patients with CHF and was related to a higher risk of ventricular tachycardia[200,201]. Similar findings were also confirmed in patients hospitalized for decompensated HF[202]. An important study by Pousset et al[203] showed that a beta-blocker, bisoprolol, administered in a single dose of 5 mg per day managed to reduce heart rate and significantly increase HRV as per 24-h Holter ECG monitoring among patients with HF[203]. This effect was attributed to the increased parameters of parasympathetic activity in HF thus showing that increased vagal tone may be responsible for the protective effect of beta blockers and may provide prognostic implications in HF. Similarly, beta-blockers improved cardiac autonomic regulation during high sympathetic stress of decompensated HF[204].
However, the foundational framework that links low-frequency and high-frequency components of HRV with sympathetic and parasympathetic nervous system division was developed decades ago and this algorithm does not integrate findings and data on HRV that were gathered in the past 30 years thus might have certain limitations in clinical practice[205]. Another potential limit for the use of HRV in risk stratification of HF patients might lie in the fact that these parameters tend to be very low in most HF subjects, therefore, data dispersion might be small thus limiting survival regression models while many confounding non-neural factors might affect HRV values in HF[206]. The future of risk stratification of events in HF likely lies in the improvement of HRV spectral analyses algorithms and integration of HRV data with other biosignals acquired from novel HF devices, imaging methods, and laboratory biomarkers.
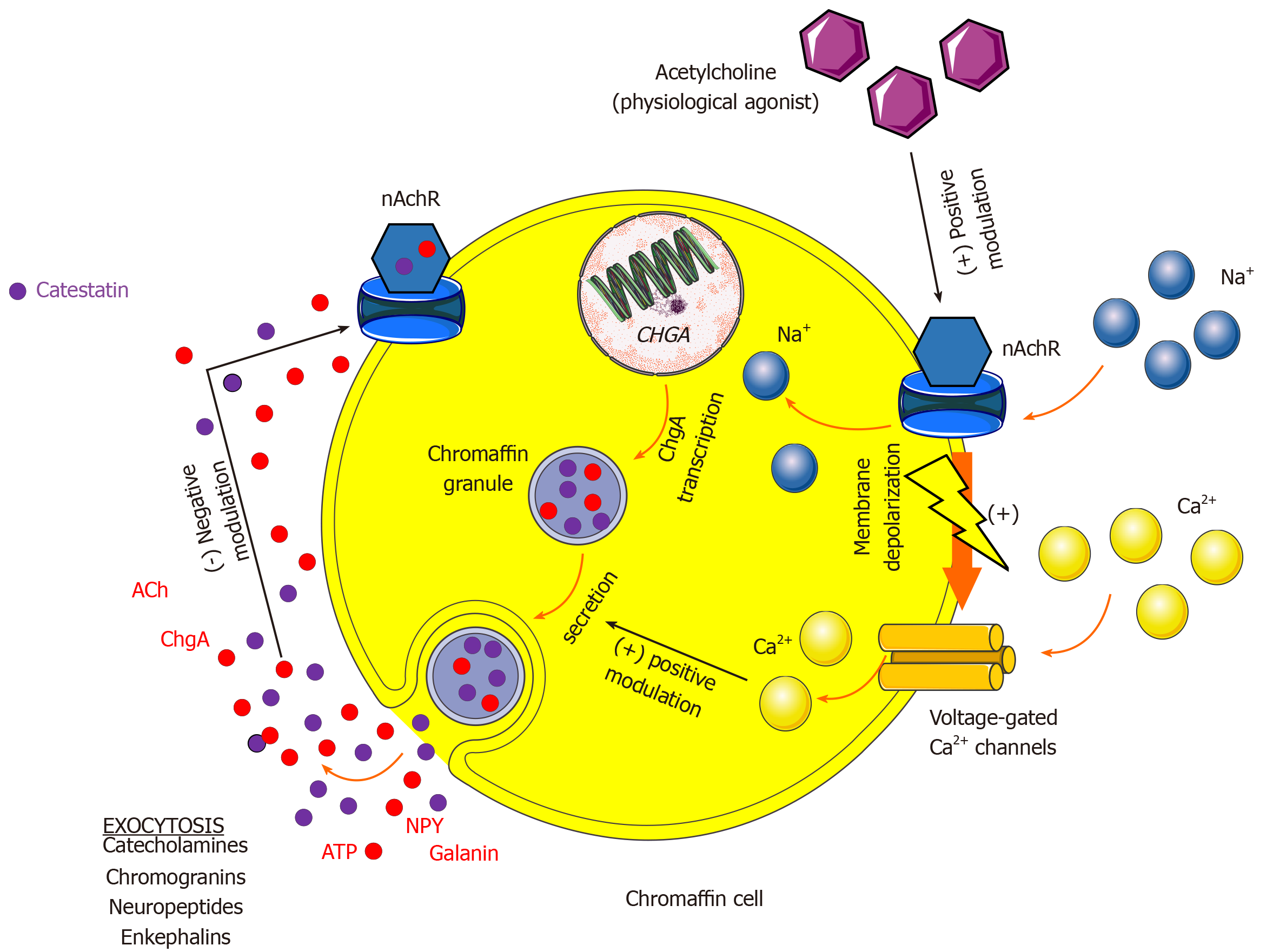
Laboratory biomarkers that can be measured in the peripheral circulation of HF patients can give us insight on underlying pathophysiological mechanisms that are occurring in patients with both acute and chronic HF. Since HF is a complex syndrome characterized by the high prevalence of comorbidities an integrated approach using multiple biomarkers could aid in the diagnosis, accurate risk stratification regarding mortality and future hospitalizations and perhaps enable optimal tailoring of pharmacotherapeutic and/or device therapies for the individual HF patient[207,208]. A wide array of novel biomarkers reflecting pathophysiological processes of myocardial stretch, matrix remodeling, myocyte injury, oxidative stress, inflammation, neurohumoral activation, and renal dysfunction are becoming increasingly studied and integrated into the process of care for HF patient and clinical decision-making[19]. The early adoption of these novel biomarkers in modern clinical practice has a great potential to complement traditional biomarkers that are regularly used in the workup of HF patients such as N-terminal prohormone of brain natriuretic peptide (NT-proBNP), brain natriuretic peptide (BNP), high sensitivity cardiac troponin (hs-cTn), soluble suppression of tumorigenicity 2 or C-reactive protein[19].
In this last section of the review, we will focus on both established and novel laboratory biomarkers that are implicated in the pathophysiology of SNS activation in HF and as such might be potentially used in clinical practice. The summary of pathophysiological effects, cellular mechanisms of action, circulating levels, and association of selected biomarkers with outcomes in HF is presented in Table 2.
| NE | NPY | GAL | ET-1 | CST | |
| Pathophysiological effects in heart failure or cardiovascular diseases | ↑ Promotes cardiac hypertrophy; ↑ promotes induction of fetal genes in myocardial remodeling; ↑ mediates and enhances apoptosis of cardiac myocytes in vitro; ↑ promotes arterial vasoconstriction; ↑ promotes tachyphylaxis; ↑ increased cardiac and renal spillover in HF; ↓ impaired oxygen utilization and exercise efficiency in patients with stable HF; ↑ increased sympathetic nerve activity and reduced clearance of norepinephrine | ↑ Vasoconstriction; ↑ promotes adverse cardiac remodeling; ↑ increased cardiac spillover; ↑ promotes angiogenesis; ↑ associated with increased platelet aggregation and adhesion following thrombosis; ↑ stimulates atherosclerosis; ↑ promotes vasoconstriction of coronary microvasculature; ↑ enhancing the NE-mediated effect of sympathetic discharge, associated with the increased incidence of ventricular arrhythmia; ↑ enhances inhibition of vagally-mediated ;bradycardia through Y2 receptors; ↑ potentiates arrhyhtmias following STEMI, despite beta-blocker therapy | ↓ Reduces cardiac cholinergic neurotransmission; ↓ reduces acetylcholine biovailability in the synapse junctions; ↓ reduces vagally-mediated bradycardia; ↑promotes antithrombotic phenotype on endocardial endothelial cells; ↑ increased cardioprotective activity against ischemia-reperfusion injury in H9C2 cardiomyoblasts in vitro | ↑ Promotes vasoconstriction (most potent vasoconstrictor in humans); ↑ promotes vascular and cardiac hypertrophy; ↓ decreases NE reuptake thus propagating adrenergic effects; ↓ reduces coronary flow; ↑ promotes inotropic and chronotropic responses in cardiomyocytes; ↑ promotes mitogenic actions; ↑ activation of endothelin-dependent pathways is observed in HF; ↑ correlates with hemodynamic impairment and severity of pulmonary hypertension in HF; ↑ promotes angiogenesis | ↓ Decreases arterial blood pressure (direct and indirect vasodilation); ↓ inhibits catecholamine release; ↓ decreases NPY and ATP release; ↓ attenuates cardiac inotropy and chronotropy; ↑ promotes angiogenesis; ↓ blunts atherosclerosis; ↓ reduces inflammation; ↓ reduces thrombogenicity; ↑ promotes VSMC proliferation; ↓ decreases arrhytmogenic events; ↓ decreases ventricular remodeling |
| Cellular mechanism | Activation of α and β adrenergic receptors (G protein-coupled) | Activation of G protein-coupled post-synaptic Y1-Y6 receptors (Y2 is also pre-synaptic) on sympathetic nerve endings | Activation of G protein-coupled receptors – GAL1R, GAL2R, GAL3R | Activation of endothelin A (ETA) and B (ETB) receptors(both G protein-coupled) | Acts on neuronal nicotinic acetylcholine receptor (nAchR) |
| Circulating levels in HF vs controls | ↑ Circulating plasma levels;↑ urinary excreted levels | ↑ Circulating plasma levels | ←→ Not significantly different plasma levels | ↑ Plasma levels;↑ renal tissue levels | ↑ Circulating plasma levels |
| Association with mortality and morbidity in HF | ↑ High NE levels were associated with significantly increased mortality and morbidity in patients with congestive HF; ↑ circulating NE levels positively correlate with HF syndrome severity | ↑ Elevated levels in coronary sinus were associated with composite endpoint of VAD implantation, death, and cardiac transplant among patients with stable chronic HF undergoing CRT implantation | Not established (no studies available) | ↑ Increased ET-1 levels associated with higher HF syndrome severity;↑ increased ET-1 levels associated with mortality in HF | ↑ Increased CST levels were independently associated with all-cause and cardiac mortality in patients with chronic HF; ↑ correlates with NYHA functional class |
As previously discussed, circulating plasma levels and urinary excretion of norepinephrine (NE) are significantly higher among patients with congestive HF compared to those without, reflecting elevated sympathetic drive[117,118,124]. A recent study by Matsushita et al[209] showed that the endogenous catecholamine surge might be the cause of urgently presenting acute HF by eliciting an abrupt and excessive rise in blood pressure leading to increased after-overload and volume-shift lung congestion[209]. A few decades ago, Cohn and colleagues showed that plasma NE has been independently related to subsequent risk of mortality among patients with chronic congestive HF and was also higher among those that had progressive HF compared to patients that died suddenly[129]. This was later confirmed in the V-HEFT II study that enrolled patients with congestive HF showing that plasma NE was an independent predictor of prognosis and plasma NE values > 900 pg/mL were associated with significantly greater mortality risk compared to lower NE tertiles[210]. In the longitudinal follow-up of patients with HF from the Val-HeFT trial, changes of BNP and NE from baseline to 4 and 12 mo post-discharge significantly correlated to changes in morbidity and mortality[211]. However, the administration of neurohormonal antagonists such as ACE inhibitors and beta-blockers in HF patients had variable and heterogeneous effects on circulating NE levels and there was a significant incongruency of these levels with endpoints such as mortality and reverse ventricular remodeling in a handful of relevant trials[212]. These data suggested that reducing NE levels might not be the appropriate goal of neurohumoral antagonists and that NE is not a feasible laboratory biomarker of choice when it comes to measuring response to HF-directed pharmacotherapy. Finally, the fact that circulating NE measurements require high-performance liquid chromatography is a significant limitation to its wide use in clinical practice and imposes several analytical challenges and physiological limitations thus making it likely impractical as a routine biomarker in HF[213].
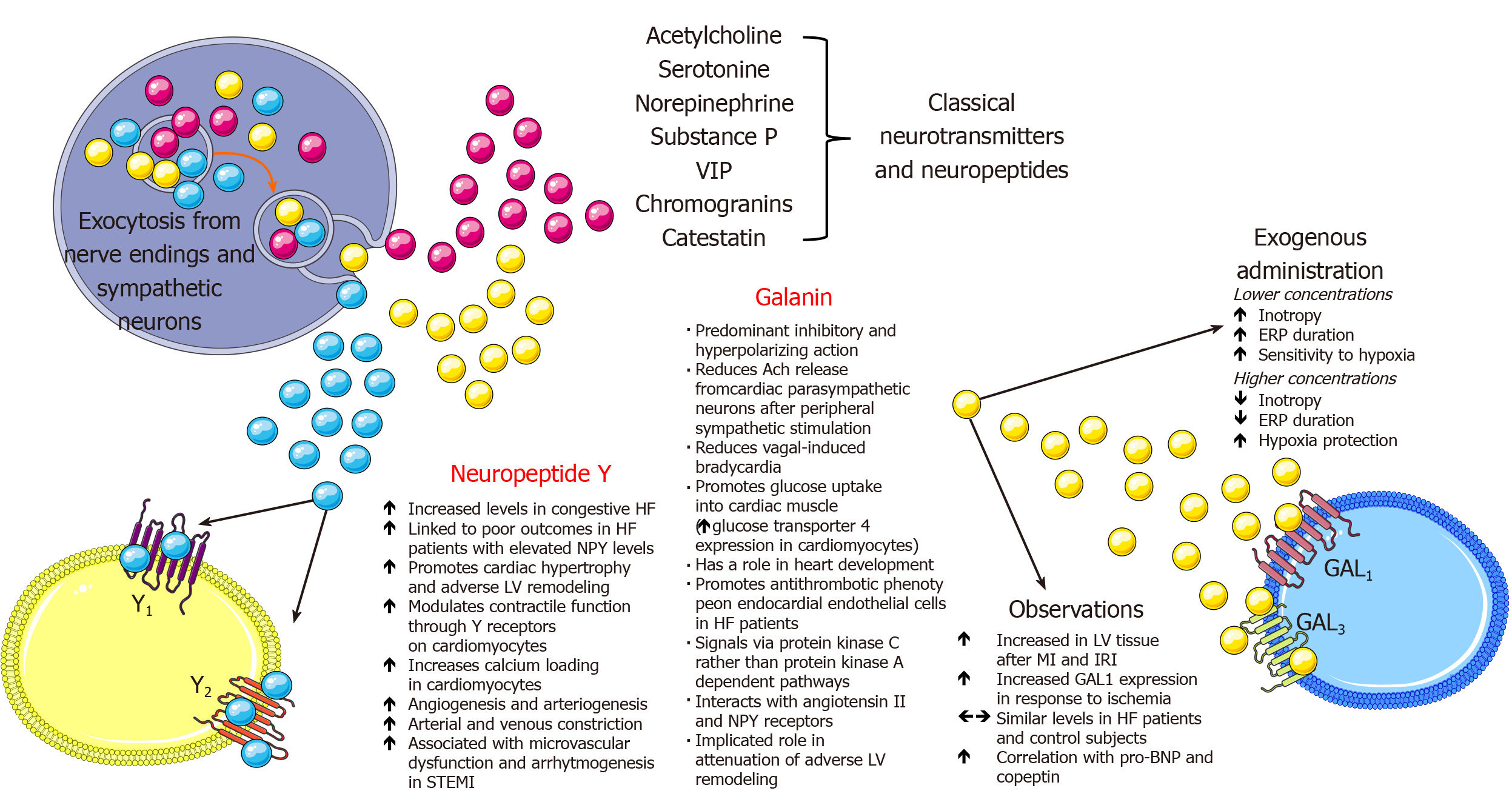
Neuropeptide Y (NPY) is a sympathetic co-transmitter with a longer half-life than NE and is widely distributed in the CNS and peripheral nervous system with pleiotropic physiological actions. In the cardiovascular system, NPY is co-released from cardiac sympathetic nerve terminals along with catecholamines (predominantly NE) and galanin[214]. These sympathetic nerves supply vasculature, cardiomyocytes and endocardial endothelial cells in the ventricle while NPY physiologically modulates cardiovascular function, potentiates pressor effects of angiotensin II, elicits arterial and venous constriction, blunts parasympathetic activity, augments cardiomyocyte calcium loading, participates in cardiomyocyte remodeling and promotes angiogenesis[215-223]. NPY and galanin have a direct ability to modulate vagus nerve to release acetylcholine and control heart rate while NPY plasma levels had a strong correlation with coronary microvascular function among patients with ST-elevation myocardial infarction[224]. Maisel and colleagues were the first to report on elevated levels of plasma NPY in patients with congestive HF and this was later confirmed in several subsequent studies[225-227].
In the recent clinical study by Ajijola et al[228], NPY was sampled from the coronary sinus (CS) among patients with stable CHF during the elective CRT device implantation[228]. Researchers sought to answer if NPY as a peptide involved in adrenergic signaling is associated with outcomes among patients with stable CHF. They found that patients with NPY CS levels > 130 pg/mL had significantly worse outcomes compared to those with lower NPY CS levels, even after adjusting for age, estimated glomerular filtration rate (eGFR), and LVEF (HR: 9.5, 95%CI: 2.92-30.5, P < 0.001) during the median follow-up of 28.8 mo while the composite endpoint consisted of death, ventricular assist device placement and cardiac transplant. Most of the signal from the composite endpoint was driven by death events and interestingly, CRT data at 6-mo follow-up showed that CS NPY levels did not significantly differ between CRT responders and non-responders (P = 0.76). Finally, immunohistochemical analyses revealed that sympathetic ganglia (stellate and middle cervical ganglion) of CHF patients contained less NPY compared to ganglia tissue obtained from healthy donors while no significant difference was observed in the NPY production between both groups as examined by the measured NPY mRNA levels. This study showed that CS NPY levels were elevated in stable CHF patients and associated with adverse outcomes and relevant clinical and laboratory characteristics while increased stellate ganglia sympathetic discharge was likely the culprit for these elevated levels.
Although CS NPY levels provided robust prognostic information among stable CHF patients, a problem in clinical practice arises in the peripheral venous sampling of NPY since those levels are not cardiac-specific and are mostly of hepatomesenteric origin since NPY has been identified as a stimulator to food intake[229]. In cardiac failure, there is an increase in resting NPY spillover within the myocardium, however, the net overflow of NPY to plasma was dominantly from hepatic circulation, but not the cardiac, forearm or cerebral circulations showing a marked difference in regional distribution of NPY content[230]. It has also been shown that sympathetic activation by exercise produced only a modest increase in cardiac NPY overflow without the concomitant change in arterial NPY concentrations finally concluding that plasma NPY concentrations are less sensitive than those of plasma NE in terms of quantifying SNS responses regulating the systemic circulation and cardiac hemodynamics in HF, as implied in some previous studies[225,230,231]. Finally, a recent preclinical study showed that NPY blockade by experimental Nur77 agent protected against adverse cardiac remodeling by limiting NPY-mediated signaling (NPY-NPY1R) in the cardiomyocytes[232]. Most important characteristics and effects of NPY are depicted in the Figure 3. In the future, antagonists of NPY receptors Y1 and Y2 might be a feasible therapeutic option in acute myocardial infarction but also during chronic HF and hypertension[224]. These pharmacotherapeutic options would complement beta-blockers and implantable vagus nerve stimulators to improve outcomes in patients with cardiovascular diseases[224].
Similarly to NE and NPY, galanin is an adrenergic co-transmitter with a short half-life (about 5 min) released from peripheral postganglionic neurons and is implicated in attenuation of cardiac cholinergic tonus after burst sympathetic activity thus contributing to autonomic imbalance and the pathophysiological phenomenon known as “sympathovagal crosstalk“[224,233]. This phenomenon can remain chronically activated and sustained even in the presence of beta-adrenergic blockade thereby it could be a valid therapeutic target in the spectrum of neurohumoral activation in HF[224]. Furthermore, nerve terminals of parasympathetic neurons in the heart express both galanin receptors and NPY receptors (NPY Y2) which, upon activation, reduce acetylcholine release[233]. During the prolonged sympathetic activation there is a release of a slowly diffusing co-transmitter galanin, together with NPY, that bind to these receptors and reduce cholinergic neurotransmission in the heart[234]. Furthermore, galanin through its receptors interacts with other neuropeptides such as NPY and angiotensin II and their receptors, namely Y1 and AT1 thus having a potential role in neurochemical modulation of central cardiovascular control[235].
A recent prospective case-control study in the clinical realm showed that unlike pro-BNP, copeptin and NPY, galanin levels were similar among patients with HF patients and control subjects while pro-BNP was the only significant determinant of galanin levels in HF patients[236]. Authors postulated that galanin most likely has a predominant paracrine modulatory function at the level of peripheral cardiac sympathetic nerves, therefore, its circulating levels in plasma might not reflect the degree of its local involvement in sympathovagal crosstalk. Finally, since natriuretic peptides promote catecholamine release from cardiac sympathetic neurons, authors suggested biological plausibility of their finding that galanin positively correlated with BNP[237]. On the other hand, galanin promoted anti-thrombotic phenotype on cultured endocardial endothelial cells from HF patients through attenuation of von Willebrand factor extrusion and multimer expression while this effect was not elicited by the NPY[238]. One preclinical study in the animal model of HF showed that galanin receptor type 1 agonist improved cardiac function and attenuated ventricular remodeling[239]. Most important characteristics and effects of galanin are depicted in Figure 3. Due to the scarcity of studies examining the role of galanin in HF, future preclinical and clinical studies are warranted to further elucidate its biological functions and its potential as a biomarker in HF.
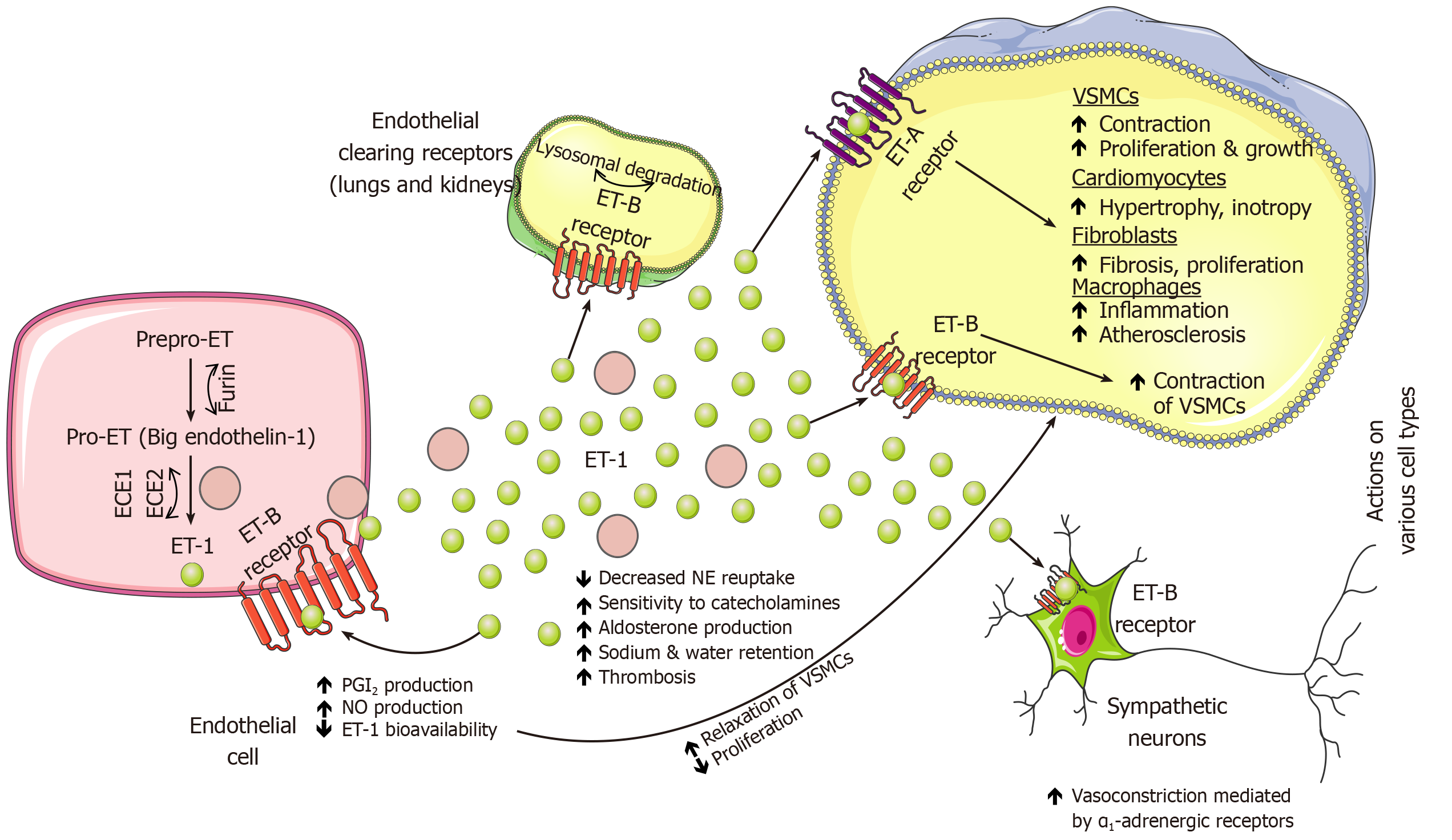
Endothelins represent a family of three similar 21 amino acid length peptides – endothelin 1 (ET-1), 2 (ET-2) and 3 (ET-3) of which ET-1 and ET-2 bind to G-protein coupled endothelin receptors A (ETA) and B (ETB) on vascular smooth muscle cells with equal affinity to both while ET-3 exhibits lower affinity for ETA relative to ETB receptor[240]. Of all endothelins, ET-1 is predominantly produced by vascular tissue, has inotropic, chemotactic and mitogenic properties, induces collagen synthesis by cardiac fibroblasts, and is biologically the most potent vasoconstrictor in the human cardiovascular system[241]. Furthemore, autocrine binding of ET-1 to ETB receptors promotes NO and prostaglandin release and consequent relaxation of vascular smooth muscle cells. ET-1 plays a role in neuronal development, growth, and function while biologically promoting vascular and cardiac hypertrophy, inflammatory responses and is an independent factor contributing to exacerbation of the cardiovascular disease[242-244]. The main source of ET-1 and its precursor, big endothelin-1 (BigET-1) are pulmonary vascular endothelial cells, therefore, elevated plasma levels of ET-1 or bigET-1 might closely reflect the degree of pulmonary endothelial dysfunction in HF while ET-1 was significantly overexpressed in the lungs of patients with pulmonary hypertension[245-247]. Stangl et al[246] demonstrated that in severe congestive HF lungs act as a producer while coronary and peripheral circulation act as consumers of BigET-1 and ET-1 while short-term vasodilator therapy decreased endothelins and restored pulmonary, coronary, and peripheral balance[246]. Endothelin receptors are also expressed in the CNS and central administration of endothelin modulated endocrine and cardiovascular regulation, behavior and MAP[248]. In the preclinical experiment, an injection of ET-1 in different regions of the brainstem of normotensive rats resulted in a differential response in heart rate, arterial blood pressure, and respiratory frequency indicating that endothelin has a modulatory role in cardiovascular function[249].
Previous studies showed that HF is associated with high levels of ET-1 in plasma and renal tissue and these levels correlated with syndrome severity, especially with the extent of pulmonary hypertension, and overall contributed to the progression of chronic HF[250-254]. In a preclinical study, infusion of tezosentan (ET-1 antagonist) significantly decreased MAP in both normal and HF animals and reduced cardiac sympathetic nerve activity (CSNA) in normal animals, however, no decrease was observed in HF animals[255]. Therefore, this study showed that endogenous levels of ET-1 contribute to the baseline levels of CSNA in healthy animals, however, this correlation was absent in experimentally induced HF. Contrary to this, a non-selective experimental ETA and ETB antagonist (TAK-044) suppressed sympathetic activity and improved arterial baroreflex function in rats with HF[256]. Similarly, the addition of ACE inhibitor to ETA receptor antagonist significantly improved cardiac failure after extensive MI in a rat model of congestive HF, compared with ACE inhibition monotherapy[257]. A cross-talk between the endothelin system and the adrenergic system has been demonstrated as activation of ETB receptors on sympathetic neurons caused an increase in arterial blood pressure through vasoconstriction mediated by α1-adrenergic receptors[98]. Sympathoexcitatory effects are also promoted through the interaction of ET-1 with ETA receptors as this resulted in cardiomyocyte hypertrophy through adrenergic signaling pathways and massive NE release while it also contributed to impaired responsiveness of renal mechanosensory nerves in congestive HF[258,259]. In the rat model of HF, endogenous ET-1 impaired NE reuptake through activation of ETA receptors while in a healthy heart ETA-mediated inhibition of NE reuptake was countered, but to a lesser degree, by the ETB-mediated silencing of NE release resulting in a net increase in left ventricular contractility suggesting that fine balance between NE reuptake and exocytotic release is modulated by endothelin signaling as it was also suggested in previous studies[260,261].
However, while endothelin pathway inhibition seemed promising in animal and preclinical models of HF, these observations did not translate to human clinical studies as ET-1 antagonist tezosentan did not improve symptoms or clinical outcomes in patients with acute HF although ET-1 levels were independently associated with short term in-hospital outcomes and 180-d mortality in patients hospitalized for acute HF, as demonstrated in ASCEND-HF substudy[262-264]. A predictive value of BigET-1 in patients with left ventricular dysfunction after AMI on the composite endpoint of cardiovascular death or hospitalization for worsening HF has been demonstrated in the subanalysis from EPHESUS study, however, neurohumoral antagonist – eplerenone seemed to have no significant effect in modifying BigET-1 levels at follow-up[265]. Authors proposed that levels of BigET-1 (as a precursor of ET-1) likely reflect the degree of ET-1 synthesis while BigET-1 is also a more feasible laboratory biomarker due to its longer half-life than that of ET-1[266]. This notion has been confirmed in a previous study that established how elevated plasma ET-1 levels in human CHF dominantly represent the elevation of Big-ET-1 while ET activity was not changed in CHF compared to a healthy state[267]. Furthermore, increased ET-1 levels were detected only in moderate or severe CHF and not among asymptomatic patients or those with mild CHF while plasma concentrations in range 5-40 pmol/L seemed to exhibit vasoactive effects[267,268]. Previous studies confirmed that BigET-1 provided prognostic information regarding the cardiovascular mortality during the 12-mo follow-up (HR: 1.42, 95%CI: 1.04-1.95, P = 0.03), all-cause mortality during the 23-mo follow-up (HR: 1.49, 95%CI: 1.20-1.84, P = 0.0003) and the composite endpoint of mortality and morbidity (HR: 1.43, 95%CI: 1.20-1.69, P < 0.001) at 23 mo, however, in the latter study BNP remained the strongest neurohormonal prognostic factor[269,270]. In the small study that enrolled patients with severe CHF, Big-ET-1 and ET-1 levels were higher at baseline than in patients with mild to moderate CHF or healthy subjects and were found as robust independent predictors of survival, even beyond natriuretic peptide levels[271].
When 32 studies with 18497 HF patients were summarized in the meta-analysis, it was shown that plasma ET-1 and its related peptides were associated with poor prognosis and mortality in diverse spectrum of HF populations[272]. On the other hand, a meta-analysis of randomized clinical trials showed that neurohumoral antagonism of ET receptors in HF patients improved cardiac output, pulmonary and systemic hemodynamics but had a modest effect on clinical outcomes[273]. Therefore, these data suggest that there is a significant discrepancy between these observations – on one hand, ET signaling has been consistently associated with poor outcomes and prognosis in HF and on the other hand, pharmacological targeting of these adverse pathways seems less impressive in improving outcomes.
Perhaps there is a need to fine-tune and identify which subgroups of HF patients would have the greatest benefit from drugs interfering with ET pathways. In that regard, ET-1 and its fragments have shown some potential as valuable biomarkers among HFpEF patients with pulmonary hypertension or pulmonary dysfunction as its levels were associated with the degree of pulmonary hemodynamic derangements, reduced functional reserve of the right ventricle, diminished cardiac output and impaired cardiac response to exercise and peak oxygen consumption[274,275]. Even in the general population, elevated plasma ET-1 levels were in strong relation with elevated pulmonary artery systolic pressures on the echocardiogram and correlated with mortality and incident HF[276].
Therefore, current data suggest that activation of the endothelin system may play an important role in the pathophysiology of pulmonary hypertension in HFpEF and that it might present a viable target and a step towards precision medicine approach in HFpEF[277]. Regarding the potential ET pathway inhibition in HFpEF, thus far there are limited but encouraging preliminary reports. In the preclinical murine model of HFpEF, dual ETA/ETB blockade by macitentan improved HFpEF by abrogating aldosterone-induced cardiomyocyte hypertrophy and reducing stiffness through decreased expression of type I collagen and titin n2B in the left ventricle[278]. In the clinical domain, in patients with HFpEF, ETA receptor antagonist sitaxsentan improved exercise tolerance, however, failed to decrease left ventricular mass or improve diastolic function while the study was not powered for mortality and rehospitalization analyses[279]. A small and prematurely stopped study showed that ET receptor blocker bosentan did not improve outcomes in HFpEF patients with pulmonary hypertension[280]. Therefore, due to the size and scarcity of available studies, a question whether ET-1 antagonists would improve outcomes in HFpEF yet remains to be answered by future and adequately powered randomized controlled trials. It is possible that neurohumoral biomarkers such as endothelin and its derivatives will enable us a more precise phenotyping of HFpEF patients to identify those that have a significantly impaired pulmonary function and that would receive the greatest benefit from ET pathway-oriented therapeutic interventions. The summary of synthesis, cellular effects, and pathophysiological implications of ET-1 are presented in Figure 4.
Catestatin (CST) is a product of precursor hormone chromogranin A (ChgA) and was isolated in 1997 by Mahata et al[281]. Its principal physiological action is the negative regulation of catecholamine release into circulation through the mechanism of non-competitive and reversible antagonism of neuronal nicotinic cholinergic receptors (nAChR)[281,282]. Its precursor molecule, ChgA, and other soluble secretory proteins are co-stored and co-released with catecholamines from vesicles in the neuroendocrine, endocrine and immune cells and sympathetic neurons thus have an important modulatory role of the adrenergic system[283]. Upon stimulation of chromaffin cells or sympathetic axons, a marked elevation of ChgA levels was detected[284]. Levels of ChgA are elevated in peripheral blood of patients with chronic HF and AMI and correlate with mortality and poor outcomes[285-287]. Even more, an intramyocardial production of ChgA is established in humans and was associated with negative inotropic and lusitropic effects on the mammalian heart thus providing evidence for neuroendocrine regulation of cardiac function by ChgA[288]. Furthermore, immunohistochemical biopsy studies showed that ChgA is co-localized with BNP in the dilated and hypertrophic left ventricle while ChgA levels correlated with end-diastolic left ventricular pressures[288].
CST is a 21 amino acid fragment derived from ChgA (ChgA352-372) and is secreted by neuroendocrine tissues and nerve endings while it is widely distributed in the secretory granules of skin, sensory organs, and myocardium[289]. Its most important physiological effect is the autocrine action on the chromaffin cells in the adrenal medulla and adrenergic neurons by which it modulates spillover of catecholamines (primarily NE) into peripheral circulation while concomitantly exhibiting paracrine and endocrine effects since it can be readily measured in the venous and arterial blood[290]. Furthermore, CST is potent regulator of arterial blood pressure since it exerts direct vasodilatative effect in humans in vivo, activates histamine release from mast cells and stimulates production of NO within endothelial cells[291-293]. In the chromaffin cell, ACh is a physiological agonist that, upon activation of ionotropic nAChR receptor, permits Na+ entry into the cell which further depolarizes cellular membrane and enables activation of voltage-gated Ca2+ channels and subsequent Ca2+ entry that mobilizes chromaffin granules and initiates exocytosis of several neurohormones, neuropeptides, and catecholamines[281,294]. Once secreted outside of the cell through the process of exocytosis, extracellular post-translational proteolytic processing of the ChgA molecule will release several bioactive peptides and CST that will ultimately bind nAChR receptors of chromaffin cells in autocrine fashion thus antagonizing ACh actions in the periphery as depicted in Figure 5[295].
In the perspective of previously discussed catecholamine storage vesicle neurotransmitters, Mahapatra and colleagues demonstrated that CST inhibited nicotinically triggered exocytotic release of several co-transmitters from chromaffin granules such as NPY, adenosine triphosphate, chromogranins and catecholamines thereby demonstrating that CST is a potent regulator of neuropeptide transmission in the sympathochromaffin system[296]. However, in the CNS, CST exhibits both sympathoexcitatory and procholinergic effects depending on the region of medulla where its expressed[297,298]. Of established cardiovascular effects, CST suppresses beta-adrenergic activation and acts in a negative inotropic and chronotropic fashion, stimulates angiogenesis and proliferation of vascular smooth muscle cells, decreases thrombogenicity of endothelial cells, suppresses atherosclerosis and inflammation while also exerts cardioprotective effects by abrogating cardiomyocyte ischemia-reperfusion injury[299-306]. A very recent study by Alam et al[307] showed that CST has a direct and independent inhibiting effect on hypertrophy elicited by NE in the cultured H9c2 cardiac myoblasts and that is involved in the regulation of a large number of fetal genes that are upregulated during the process of myocardial hypertrophy[307]. Furthermore, the same study showed that CST effectively blunted stimulative effects of NE and other mitogenic signals on β1 and β2 adrenergic receptors thus providing novel evidence that CST has a direct modulatory effect on adrenergic transmission at the level of adrenergic receptors. Similarly, in the model of rat heart, CST activated β2 and β3 adrenergic receptors thus upregulating the activity of eNOS and consequently increasing cyclic GMP and phosphodiesterase type 2 (PDE2) levels[308].
In line with its sympatholytic effects, chronic administration of exogenous CST improved autonomic function, shortened QT interval, and action potential duration and reduced the incidence of experimentally-induced ventricular arrhythmias in a rat model of myocardial infarction[309]. Similarly, in the rat model of hyperadrenergic hypertension, rats with ablated ChgA gene showed significantly longer QT interval, R-amplitude, and QRS time-voltage and this was accompanied by increased resting heart rate and QT variability thus demonstrating that arrhythmogenic ventricular assault develops in the status of low circulating CST levels[310]. These preclinical observations were clinically validated as elevated CST levels were an independent predictor of complicated malignant arrhythmias among AMI patients[311]. This observation might seem counterintuitive at first, however, it could be explained in the sense that CST levels reflect a compensatory response for the increased SNS activity and excess catecholamine discharge and are attempting to restore autonomic balance. Therefore, circulating CST levels likely “mirror“ biological catecholamine turnover and degree of sympathetic activity as CST co-localizes and is co-released with catecholamines and other neuropeptides. Finally, cardioprotective effects of CST are likely pathophysiologically overpowered by sympathetic discharge in conditions in which cardiovascular homeostasis is disrupted such as AMI or decompensated HF, despite the relatively high circulating CST levels.
There are only a few available studies that examined the role of CST in HF and investigated its prognostic role in this syndrome.
In the first study by Zhu et al[312] CST levels gradually decreased from stage A to C of HF while there was a significant difference between stage A and B in terms of CST concentrations with a cut-off value of 19.73 ng/mL providing 90% sensitivity and 50.9% specificity for the detection of B stage of HF[312]. This finding is of clinical relevance since stage B presumes structural cardiac disorder but without symptoms, while stage A assumes patients at high risk for developing HF but without functional or structural heart disorder. Therefore, this study showed that there is a utility for decreased CST levels implying structural heart disease among asymptomatic patients. In the study by Liu et al[313] performed in the similar setting, CST was found higher in patients with HF compared to control subjects and it positively correlated with functional syndrome burden as assessed by the NYHA class[313]. Furthermore, etiology of HF (ischemic or not) and NYHA class predicted plasma CST levels while BNP provided better area under the curve value than CST in terms of detecting moderate to severe HF diagnosis (area under the curve values of 0.831 and 0.626, respectively). Adding CST to BNP did not improve diagnostic accuracy.
Recently, Borovac et al[314] showed in CATSTAT-HF study, that patients with acutely decompensated HF had higher serum CST levels if they belonged to a higher NYHA functional class while circulating CST levels were significantly higher among patients with ischemic vs non-ischemic etiology of disease thus likely reflecting an augmented SNS neurohumoral activation in patients with ischemic etiology of the disease (in the study defined as those with the history of myocardial infarction)[314]. The same study revealed that CST levels did not differ in respect to LVEF phenotypes while CST levels independently correlated with NYHA class, waist-to-hip ratio, HbA1c, LDL cholesterol, non-HDL cholesterol, high sensitivity cardiac troponin I and the heart rate at admission and rest. Finally, higher CST levels were strongly associated with favorable echocardiographic profile as they positively correlated with smaller LV volumes and dimensions, as well as with decreased left ventricular mass and smaller dimensions of the left atrium and this finding clinically validates the concept that CST locally has cardioprotective effects, attenuates adverse ventricular remodeling and acts in antihypertrophic fashion, as these biological effects were postulated in a few earlier preclinical studies[307,315].
Finally, the prognostic value of CST as a biomarker was demonstrated among chronic HF patients. In the multivariate Cox regression analysis, plasma CST was an independent risk factor for all-cause death (HR: 1.84, 95%CI: 1.02-3.32, P = 0.042) and cardiac death (HR: 2.41, 95%CI: 1.26-4.62, P = 0.008), respectively, during the median follow-up of 52.5 mo[316]. If patients had both high CST and BNP levels during hospitalization the risk of all-cause death increased 3-fold while the risk of cardiac death increased 4-fold.
Based on these findings it is plausible that CST could be a reliable indirect marker of SNS activity and it is likely that high CST levels reflect advanced disease burden and high sympathoexcitatory profile of an individual HF patient. Furthermore, CST provides complementary prognostic information to natriuretic peptides in terms of mortality in HF and could aid in the risk stratification of chronic HF patients. However, since the latter finding is based on only one clinical study further large-scale studies are required to validate these findings and clarify the role of circulating CST levels in predicting HF prognosis. Finally, patients with elevated CST levels might be suitable candidates for the introduction or up-titration of sympatholytic agents such as beta-adrenergic blockers, however, the effects of neurohumoral antagonists on circulating CST levels are yet to be determined.
Figures presented in this manuscript are of author’s own design, however, they were constructed by using some of the illustration elements kindly provided by Servier. Servier Medical Art is licensed under a Creative Commons Attribution 3.0 Unported Licence.
Manuscript source: Invited manuscript
Specialty type: Cardiac and cardiovascular systems
Country/Territory of origin: Croatia
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Roysommuti S S-Editor: Zhang L L-Editor: A P-Editor: Li JH
| 1. | Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JG, Coats AJ, Falk V, González-Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, Parissis JT, Pieske B, Riley JP, Rosano GM, Ruilope LM, Ruschitzka F, Rutten FH, van der Meer P; Authors/Task Force Members; Document Reviewers. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2016;18:891-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4368] [Cited by in RCA: 4921] [Article Influence: 546.8] [Reference Citation Analysis (4)] |
| 2. | Braunwald E. Heart failure. JACC Heart Fail. 2013;1:1-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 476] [Cited by in RCA: 561] [Article Influence: 46.8] [Reference Citation Analysis (1)] |
| 3. | Savarese G, D'Amario D. Sex Differences in Heart Failure. Adv Exp Med Biol. 2018;1065:529-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 4. | Benjamin EJ, Blaha MJ, Chiuve SE, Cushman M, Das SR, Deo R, de Ferranti SD, Floyd J, Fornage M, Gillespie C, Isasi CR, Jiménez MC, Jordan LC, Judd SE, Lackland D, Lichtman JH, Lisabeth L, Liu S, Longenecker CT, Mackey RH, Matsushita K, Mozaffarian D, Mussolino ME, Nasir K, Neumar RW, Palaniappan L, Pandey DK, Thiagarajan RR, Reeves MJ, Ritchey M, Rodriguez CJ, Roth GA, Rosamond WD, Sasson C, Towfighi A, Tsao CW, Turner MB, Virani SS, Voeks JH, Willey JZ, Wilkins JT, Wu JH, Alger HM, Wong SS, Muntner P; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2017 Update: A Report From the American Heart Association. Circulation. 2017;135:e146-e603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6484] [Cited by in RCA: 6391] [Article Influence: 798.9] [Reference Citation Analysis (0)] |
| 5. | Savarese G, Lund LH. Global Public Health Burden of Heart Failure. Card Fail Rev. 2017;3:7-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1147] [Cited by in RCA: 1766] [Article Influence: 220.8] [Reference Citation Analysis (0)] |
| 6. | Conrad N, Judge A, Tran J, Mohseni H, Hedgecott D, Crespillo AP, Allison M, Hemingway H, Cleland JG, McMurray JJV, Rahimi K. Temporal trends and patterns in heart failure incidence: a population-based study of 4 million individuals. Lancet. 2018;391:572-580. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 605] [Cited by in RCA: 873] [Article Influence: 124.7] [Reference Citation Analysis (0)] |
| 7. | Lesyuk W, Kriza C, Kolominsky-Rabas P. Cost-of-illness studies in heart failure: a systematic review 2004-2016. BMC Cardiovasc Disord. 2018;18:74. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 296] [Article Influence: 42.3] [Reference Citation Analysis (0)] |
| 8. | Ambrosy AP, Fonarow GC, Butler J, Chioncel O, Greene SJ, Vaduganathan M, Nodari S, Lam CSP, Sato N, Shah AN, Gheorghiade M. The global health and economic burden of hospitalizations for heart failure: lessons learned from hospitalized heart failure registries. J Am Coll Cardiol. 2014;63:1123-1133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1248] [Cited by in RCA: 1570] [Article Influence: 142.7] [Reference Citation Analysis (0)] |
| 9. | Taylor CJ, Ryan R, Nichols L, Gale N, Hobbs FR, Marshall T. Survival following a diagnosis of heart failure in primary care. Fam Pract. 2017;34:161-168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 10. | Dokainish H, Teo K, Zhu J, Roy A, AlHabib KF, ElSayed A, Palileo-Villaneuva L, Lopez-Jaramillo P, Karaye K, Yusoff K, Orlandini A, Sliwa K, Mondo C, Lanas F, Prabhakaran D, Badr A, Elmaghawry M, Damasceno A, Tibazarwa K, Belley-Cote E, Balasubramanian K, Islam S, Yacoub MH, Huffman MD, Harkness K, Grinvalds A, McKelvie R, Bangdiwala SI, Yusuf S; INTER-CHF Investigators. Global mortality variations in patients with heart failure: results from the International Congestive Heart Failure (INTER-CHF) prospective cohort study. Lancet Glob Health. 2017;5:e665-e672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 245] [Article Influence: 30.6] [Reference Citation Analysis (0)] |
| 11. | Mamas MA, Sperrin M, Watson MC, Coutts A, Wilde K, Burton C, Kadam UT, Kwok CS, Clark AB, Murchie P, Buchan I, Hannaford PC, Myint PK. Do patients have worse outcomes in heart failure than in cancer? A primary care-based cohort study with 10-year follow-up in Scotland. Eur J Heart Fail. 2017;19:1095-1104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 210] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 12. | Taylor CJ, Ordóñez-Mena JM, Roalfe AK, Lay-Flurrie S, Jones NR, Marshall T, Hobbs FDR. Trends in survival after a diagnosis of heart failure in the United Kingdom 2000-2017: population based cohort study. BMJ. 2019;364:l223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 201] [Cited by in RCA: 321] [Article Influence: 53.5] [Reference Citation Analysis (0)] |
| 13. | Mensah GA, Wei GS, Sorlie PD, Fine LJ, Rosenberg Y, Kaufmann PG, Mussolino ME, Hsu LL, Addou E, Engelgau MM, Gordon D. Decline in Cardiovascular Mortality: Possible Causes and Implications. Circ Res. 2017;120:366-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 486] [Cited by in RCA: 576] [Article Influence: 72.0] [Reference Citation Analysis (0)] |
| 14. | Tsao CW, Lyass A, Enserro D, Larson MG, Ho JE, Kizer JR, Gottdiener JS, Psaty BM, Vasan RS. Temporal Trends in the Incidence of and Mortality Associated With Heart Failure With Preserved and Reduced Ejection Fraction. JACC Heart Fail. 2018;6:678-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 306] [Cited by in RCA: 360] [Article Influence: 51.4] [Reference Citation Analysis (1)] |
| 15. | Dunlay SM, Roger VL, Redfield MM. Epidemiology of heart failure with preserved ejection fraction. Nat Rev Cardiol. 2017;14:591-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 559] [Cited by in RCA: 999] [Article Influence: 124.9] [Reference Citation Analysis (0)] |
| 16. | Shah KS, Xu H, Matsouaka RA, Bhatt DL, Heidenreich PA, Hernandez AF, Devore AD, Yancy CW, Fonarow GC. Heart Failure With Preserved, Borderline, and Reduced Ejection Fraction: 5-Year Outcomes. J Am Coll Cardiol. 2017;70:2476-2486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 445] [Cited by in RCA: 756] [Article Influence: 94.5] [Reference Citation Analysis (1)] |
| 17. | Henning RJ. Diagnosis and treatment of heart failure with preserved left ventricular ejection fraction. World J Cardiol. 2020;12:7-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 18. | Goyal P, Loop M, Chen L, Brown TM, Durant RW, Safford MM, Levitan EB. Causes and Temporal Patterns of 30-Day Readmission Among Older Adults Hospitalized With Heart Failure With Preserved or Reduced Ejection Fraction. J Am Heart Assoc. 2018;7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 33] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 19. | Ibrahim NE, Januzzi JL. Established and Emerging Roles of Biomarkers in Heart Failure. Circ Res. 2018;123:614-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 213] [Article Influence: 35.5] [Reference Citation Analysis (0)] |
| 20. | Ziaeian B, Fonarow GC. Epidemiology and aetiology of heart failure. Nat Rev Cardiol. 2016;13:368-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 791] [Cited by in RCA: 1263] [Article Influence: 140.3] [Reference Citation Analysis (0)] |
| 21. | Kemp CD, Conte JV. The pathophysiology of heart failure. Cardiovasc Pathol. 2012;21:365-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 368] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 22. | Cuomo A, Rodolico A, Galdieri A, Russo M, Campi G, Franco R, Bruno D, Aran L, Carannante A, Attanasio U, Tocchetti CG, Varricchi G, Mercurio V. Heart Failure and Cancer: Mechanisms of Old and New Cardiotoxic Drugs in Cancer Patients. Card Fail Rev. 2019;5:112-118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 23. | De Keulenaer GW, Brutsaert DL. Systolic and diastolic heart failure are overlapping phenotypes within the heart failure spectrum. Circulation. 2011;123:1996-2004; discussion 2005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 103] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 24. | Aziz F, Tk LA, Enweluzo C, Dutta S, Zaeem M. Diastolic heart failure: a concise review. J Clin Med Res. 2013;5:327-334. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 25. | Federmann M, Hess OM. Differentiation between systolic and diastolic dysfunction. Eur Heart J. 1994;15 Suppl D:2-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 26. | Bloom MW, Greenberg B, Jaarsma T, Januzzi JL, Lam CSP, Maggioni AP, Trochu JN, Butler J. Heart failure with reduced ejection fraction. Nat Rev Dis Primers. 2017;3:17058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 147] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 27. | Ouzounian M, Lee DS, Liu PP. Diastolic heart failure: mechanisms and controversies. Nat Clin Pract Cardiovasc Med. 2008;5:375-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 91] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 28. | Yoon S, Eom GH. Heart failure with preserved ejection fraction: present status and future directions. Exp Mol Med. 2019;51:1-9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 29. | Buggey J, Alenezi F, Yoon HJ, Phelan M, DeVore AD, Khouri MG, Schulte PJ, Velazquez EJ. Left ventricular global longitudinal strain in patients with heart failure with preserved ejection fraction: outcomes following an acute heart failure hospitalization. ESC Heart Fail. 2017;4:432-439. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 54] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 30. | DeVore AD, McNulty S, Alenezi F, Ersboll M, Vader JM, Oh JK, Lin G, Redfield MM, Lewis G, Semigran MJ, Anstrom KJ, Hernandez AF, Velazquez EJ. Impaired left ventricular global longitudinal strain in patients with heart failure with preserved ejection fraction: insights from the RELAX trial. Eur J Heart Fail. 2017;19:893-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 130] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 31. | Nagueh SF, Chang SM, Nabi F, Shah DJ, Estep JD. Cardiac Imaging in Patients With Heart Failure and Preserved Ejection Fraction. Circ Cardiovasc Imaging. 2017;10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 32. | Zile MR, Brutsaert DL. New concepts in diastolic dysfunction and diastolic heart failure: Part II: causal mechanisms and treatment. Circulation. 2002;105:1503-1508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 545] [Cited by in RCA: 506] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 33. | Borlaug BA. The pathophysiology of heart failure with preserved ejection fraction. Nat Rev Cardiol. 2014;11:507-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 400] [Cited by in RCA: 525] [Article Influence: 47.7] [Reference Citation Analysis (0)] |
| 34. | Zile MR, Baicu CF, Gaasch WH. Diastolic heart failure--abnormalities in active relaxation and passive stiffness of the left ventricle. N Engl J Med. 2004;350:1953-1959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1116] [Cited by in RCA: 1100] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 35. | Zile MR, Baicu CF, Ikonomidis JS, Stroud RE, Nietert PJ, Bradshaw AD, Slater R, Palmer BM, Van Buren P, Meyer M, Redfield MM, Bull DA, Granzier HL, LeWinter MM. Myocardial stiffness in patients with heart failure and a preserved ejection fraction: contributions of collagen and titin. Circulation. 2015;131:1247-1259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 414] [Cited by in RCA: 520] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 36. | Van Aelst LNL, Arrigo M, Placido R, Akiyama E, Girerd N, Zannad F, Manivet P, Rossignol P, Badoz M, Sadoune M, Launay JM, Gayat E, Lam CSP, Cohen-Solal A, Mebazaa A, Seronde MF. Acutely decompensated heart failure with preserved and reduced ejection fraction present with comparable haemodynamic congestion. Eur J Heart Fail. 2018;20:738-747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 120] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 37. | Reddy YNV, Andersen MJ, Obokata M, Koepp KE, Kane GC, Melenovsky V, Olson TP, Borlaug BA. Arterial Stiffening With Exercise in Patients With Heart Failure and Preserved Ejection Fraction. J Am Coll Cardiol. 2017;70:136-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 208] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 38. | Dhakal BP, Malhotra R, Murphy RM, Pappagianopoulos PP, Baggish AL, Weiner RB, Houstis NE, Eisman AS, Hough SS, Lewis GD. Mechanisms of exercise intolerance in heart failure with preserved ejection fraction: the role of abnormal peripheral oxygen extraction. Circ Heart Fail. 2015;8:286-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 346] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 39. | Malhotra R, Bakken K, D'Elia E, Lewis GD. Cardiopulmonary Exercise Testing in Heart Failure. JACC Heart Fail. 2016;4:607-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 298] [Article Influence: 33.1] [Reference Citation Analysis (0)] |
| 40. | Reddy YNV, Olson TP, Obokata M, Melenovsky V, Borlaug BA. Hemodynamic Correlates and Diagnostic Role of Cardiopulmonary Exercise Testing in Heart Failure With Preserved Ejection Fraction. JACC Heart Fail. 2018;6:665-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 152] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 41. | Banning AP, Lewis NP, Northridge DB, Elborn JS, Hendersen AH. Perfusion/ventilation mismatch during exercise in chronic heart failure: an investigation of circulatory determinants. Br Heart J. 1995;74:27-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 47] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 42. | D'Amario D, Migliaro S, Borovac JA, Restivo A, Vergallo R, Galli M, Leone AM, Montone RA, Niccoli G, Aspromonte N, Crea F. Microvascular Dysfunction in Heart Failure With Preserved Ejection Fraction. Front Physiol. 2019;10:1347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 90] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 43. | Gevaert AB, Boen JRA, Segers VF, Van Craenenbroeck EM. Heart Failure With Preserved Ejection Fraction: A Review of Cardiac and Noncardiac Pathophysiology. Front Physiol. 2019;10:638. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 94] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 44. | Paulus WJ, Tschöpe C. A novel paradigm for heart failure with preserved ejection fraction: comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J Am Coll Cardiol. 2013;62:263-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1983] [Cited by in RCA: 2591] [Article Influence: 215.9] [Reference Citation Analysis (0)] |
| 45. | Franssen C, Chen S, Unger A, Korkmaz HI, De Keulenaer GW, Tschöpe C, Leite-Moreira AF, Musters R, Niessen HW, Linke WA, Paulus WJ, Hamdani N. Myocardial Microvascular Inflammatory Endothelial Activation in Heart Failure With Preserved Ejection Fraction. JACC Heart Fail. 2016;4:312-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 276] [Cited by in RCA: 403] [Article Influence: 40.3] [Reference Citation Analysis (0)] |
| 46. | van Heerebeek L, Hamdani N, Falcão-Pires I, Leite-Moreira AF, Begieneman MP, Bronzwaer JG, van der Velden J, Stienen GJ, Laarman GJ, Somsen A, Verheugt FW, Niessen HW, Paulus WJ. Low myocardial protein kinase G activity in heart failure with preserved ejection fraction. Circulation. 2012;126:830-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 337] [Cited by in RCA: 400] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 47. | Kötter S, Gout L, Von Frieling-Salewsky M, Müller AE, Helling S, Marcus K, Dos Remedios C, Linke WA, Krüger M. Differential changes in titin domain phosphorylation increase myofilament stiffness in failing human hearts. Cardiovasc Res. 2013;99:648-656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 98] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 48. | Hamdani N, Bishu KG, von Frieling-Salewsky M, Redfield MM, Linke WA. Deranged myofilament phosphorylation and function in experimental heart failure with preserved ejection fraction. Cardiovasc Res. 2013;97:464-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 184] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 49. | Graziani F, Varone F, Crea F, Richeldi L. Treating heart failure with preserved ejection fraction: learning from pulmonary fibrosis. Eur J Heart Fail. 2018;20:1385-1391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 50. | Leucker TM, Jones SP. Endothelial dysfunction as a nexus for endothelial cell-cardiomyocyte miscommunication. Front Physiol. 2014;5:328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 51. | Gevaert AB, Lemmens K, Vrints CJ, Van Craenenbroeck EM. Targeting Endothelial Function to Treat Heart Failure with Preserved Ejection Fraction: The Promise of Exercise Training. Oxid Med Cell Longev. 2017;2017:4865756. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 52. | Gimbrone MA, García-Cardeña G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ Res. 2016;118:620-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1448] [Cited by in RCA: 2352] [Article Influence: 261.3] [Reference Citation Analysis (0)] |
| 53. | Kovacic JC, Dimmeler S, Harvey RP, Finkel T, Aikawa E, Krenning G, Baker AH. Endothelial to Mesenchymal Transition in Cardiovascular Disease: JACC State-of-the-Art Review. J Am Coll Cardiol. 2019;73:190-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 313] [Cited by in RCA: 417] [Article Influence: 69.5] [Reference Citation Analysis (0)] |
| 54. | Wirrig EE, Yutzey KE. Conserved transcriptional regulatory mechanisms in aortic valve development and disease. Arterioscler Thromb Vasc Biol. 2014;34:737-741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 55. | Spillmann F, Miteva K, Pieske B, Tschöpe C, Van Linthout S. High-density lipoproteins reduce endothelial-to-mesenchymal transition. Arterioscler Thromb Vasc Biol. 2015;35:1774-1777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 63] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 56. | Murdoch CE, Chaubey S, Zeng L, Yu B, Ivetic A, Walker SJ, Vanhoutte D, Heymans S, Grieve DJ, Cave AC, Brewer AC, Zhang M, Shah AM. Endothelial NADPH oxidase-2 promotes interstitial cardiac fibrosis and diastolic dysfunction through proinflammatory effects and endothelial-mesenchymal transition. J Am Coll Cardiol. 2014;63:2734-2741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 162] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 57. | Cooley BC, Nevado J, Mellad J, Yang D, St Hilaire C, Negro A, Fang F, Chen G, San H, Walts AD, Schwartzbeck RL, Taylor B, Lanzer JD, Wragg A, Elagha A, Beltran LE, Berry C, Feil R, Virmani R, Ladich E, Kovacic JC, Boehm M. TGF-β signaling mediates endothelial-to-mesenchymal transition (EndMT) during vein graft remodeling. Sci Transl Med. 2014;6:227ra34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 324] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 58. | Xu X, Friehs I, Zhong Hu T, Melnychenko I, Tampe B, Alnour F, Iascone M, Kalluri R, Zeisberg M, Del Nido PJ, Zeisberg EM. Endocardial fibroelastosis is caused by aberrant endothelial to mesenchymal transition. Circ Res. 2015;116:857-866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 99] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 59. | Good RB, Gilbane AJ, Trinder SL, Denton CP, Coghlan G, Abraham DJ, Holmes AM. Endothelial to Mesenchymal Transition Contributes to Endothelial Dysfunction in Pulmonary Arterial Hypertension. Am J Pathol. 2015;185:1850-1858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 259] [Article Influence: 25.9] [Reference Citation Analysis (0)] |
| 60. | Jiang Y, Zhou X, Hu R, Dai A. TGF-β1-induced SMAD2/3/4 activation promotes RELM-β transcription to modulate the endothelium-mesenchymal transition in human endothelial cells. Int J Biochem Cell Biol. 2018;105:52-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 61. | Piera-Velazquez S, Mendoza FA, Jimenez SA. Endothelial to Mesenchymal Transition (EndoMT) in the Pathogenesis of Human Fibrotic Diseases. J Clin Med. 2016;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 210] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 62. | Yoshimura A, Wakabayashi Y, Mori T. Cellular and molecular basis for the regulation of inflammation by TGF-beta. J Biochem. 2010;147:781-792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 314] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 63. | Cho JG, Lee A, Chang W, Lee MS, Kim J. Endothelial to Mesenchymal Transition Represents a Key Link in the Interaction between Inflammation and Endothelial Dysfunction. Front Immunol. 2018;9:294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 211] [Article Influence: 30.1] [Reference Citation Analysis (0)] |
| 64. | Hulshoff MS, Xu X, Krenning G, Zeisberg EM. Epigenetic Regulation of Endothelial-to-Mesenchymal Transition in Chronic Heart Disease. Arterioscler Thromb Vasc Biol. 2018;38:1986-1996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 65. | Hartupee J, Mann DL. Neurohormonal activation in heart failure with reduced ejection fraction. Nat Rev Cardiol. 2017;14:30-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 431] [Article Influence: 47.9] [Reference Citation Analysis (0)] |
| 66. | Volpe M, Carnovali M, Mastromarino V. The natriuretic peptides system in the pathophysiology of heart failure: from molecular basis to treatment. Clin Sci (Lond). 2016;130:57-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 211] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 67. | Jhund PS, McMurray JJ. The neprilysin pathway in heart failure: a review and guide on the use of sacubitril/valsartan. Heart. 2016;102:1342-1347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 115] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 68. | Orsborne C, Chaggar PS, Shaw SM, Williams SG. The renin-angiotensin-aldosterone system in heart failure for the non-specialist: the past, the present and the future. Postgrad Med J. 2017;93:29-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 45] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 69. | Goldsmith SR. The role of vasopressin in congestive heart failure. Cleve Clin J Med. 2006;73 Suppl 3:S19-S23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 37] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 70. | Lee CS, Tkacs NC. Current concepts of neurohormonal activation in heart failure: mediators and mechanisms. AACN Adv Crit Care. 2008;19:364-85; quiz 386-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 71. | Zhang DY, Anderson AS. The sympathetic nervous system and heart failure. Cardiol Clin. 2014;32:33-45, vii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 134] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 72. | Floras JS, Ponikowski P. The sympathetic/parasympathetic imbalance in heart failure with reduced ejection fraction. Eur Heart J. 2015;36:1974-182b. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 193] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 73. | Seferovic PM, Ponikowski P, Anker SD, Bauersachs J, Chioncel O, Cleland JGF, de Boer RA, Drexel H, Ben Gal T, Hill L, Jaarsma T, Jankowska EA, Anker MS, Lainscak M, Lewis BS, McDonagh T, Metra M, Milicic D, Mullens W, Piepoli MF, Rosano G, Ruschitzka F, Volterrani M, Voors AA, Filippatos G, Coats AJS. Clinical practice update on heart failure 2019: pharmacotherapy, procedures, devices and patient management. An expert consensus meeting report of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2019;21:1169-1186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 438] [Article Influence: 73.0] [Reference Citation Analysis (0)] |
| 74. | Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi JL, Johnson MR, Kasper EK, Levy WC, Masoudi FA, McBride PE, McMurray JJ, Mitchell JE, Peterson PN, Riegel B, Sam F, Stevenson LW, Tang WH, Tsai EJ, Wilkoff BL; American College of Cardiology Foundation; American Heart Association Task Force on Practice Guidelines. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;62:e147-e239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4116] [Cited by in RCA: 4654] [Article Influence: 387.8] [Reference Citation Analysis (1)] |
| 75. | van Bilsen M, Patel HC, Bauersachs J, Böhm M, Borggrefe M, Brutsaert D, Coats AJS, de Boer RA, de Keulenaer GW, Filippatos GS, Floras J, Grassi G, Jankowska EA, Kornet L, Lunde IG, Maack C, Mahfoud F, Pollesello P, Ponikowski P, Ruschitzka F, Sabbah HN, Schultz HD, Seferovic P, Slart RHJA, Taggart P, Tocchetti CG, Van Laake LW, Zannad F, Heymans S, Lyon AR. The autonomic nervous system as a therapeutic target in heart failure: a scientific position statement from the Translational Research Committee of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2017;19:1361-1378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 117] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 76. | Doehner W, Ural D, Haeusler KG, Čelutkienė J, Bestetti R, Cavusoglu Y, Peña-Duque MA, Glavas D, Iacoviello M, Laufs U, Alvear RM, Mbakwem A, Piepoli MF, Rosen SD, Tsivgoulis G, Vitale C, Yilmaz MB, Anker SD, Filippatos G, Seferovic P, Coats AJS, Ruschitzka F. Heart and brain interaction in patients with heart failure: overview and proposal for a taxonomy. A position paper from the Study Group on Heart and Brain Interaction of the Heart Failure Association. Eur J Heart Fail. 2018;20:199-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 142] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 77. | Packer M. The neurohormonal hypothesis: a theory to explain the mechanism of disease progression in heart failure. J Am Coll Cardiol. 1992;20:248-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 736] [Cited by in RCA: 718] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 78. | de Lucia C, Piedepalumbo M, Paolisso G, Koch WJ. Sympathetic nervous system in age-related cardiovascular dysfunction: Pathophysiology and therapeutic perspective. Int J Biochem Cell Biol. 2019;108:29-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 79. | Triposkiadis F, Karayannis G, Giamouzis G, Skoularigis J, Louridas G, Butler J. The sympathetic nervous system in heart failure physiology, pathophysiology, and clinical implications. J Am Coll Cardiol. 2009;54:1747-1762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 622] [Cited by in RCA: 681] [Article Influence: 42.6] [Reference Citation Analysis (1)] |
| 80. | Angelakos ET, King MP, Millard RW. Regional distribution of catecholamines in the hearts of various species. Ann N Y Acad Sci. 1969;156:219-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 54] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 81. | Pierpont GL, DeMaster EG, Reynolds S, Pederson J, Cohn JN. Ventricular myocardial catecholamines in primates. J Lab Clin Med. 1985;106:205-210. [PubMed] |
| 82. | Janes RD, Brandys JC, Hopkins DA, Johnstone DE, Murphy DA, Armour JA. Anatomy of human extrinsic cardiac nerves and ganglia. Am J Cardiol. 1986;57:299-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 156] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 83. | Heesch CM. Reflexes that control cardiovascular function. Am J Physiol. 1999;277:S234-S243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 39] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 84. | Burchell AE, Sobotka PA, Hart EC, Nightingale AK, Dunlap ME. Chemohypersensitivity and autonomic modulation of venous capacitance in the pathophysiology of acute decompensated heart failure. Curr Heart Fail Rep. 2013;10:139-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 85. | Fallick C, Sobotka PA, Dunlap ME. Sympathetically mediated changes in capacitance: redistribution of the venous reservoir as a cause of decompensation. Circ Heart Fail. 2011;4:669-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 240] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 86. | Malliani A, Montano N. Emerging excitatory role of cardiovascular sympathetic afferents in pathophysiological conditions. Hypertension. 2002;39:63-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 118] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 87. | Leineweber K, Wangemann T, Giessler C, Bruck H, Dhein S, Kostelka M, Mohr FW, Silber RE, Brodde OE. Age-dependent changes of cardiac neuronal noradrenaline reuptake transporter (uptake1) in the human heart. J Am Coll Cardiol. 2002;40:1459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 88. | Torres GE, Gainetdinov RR, Caron MG. Plasma membrane monoamine transporters: structure, regulation and function. Nat Rev Neurosci. 2003;4:13-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 685] [Cited by in RCA: 674] [Article Influence: 30.6] [Reference Citation Analysis (0)] |
| 89. | Rengo G, Pagano G, Vitale DF, Formisano R, Komici K, Petraglia L, Parisi V, Femminella GD, de Lucia C, Paolillo S, Cannavo A, Attena E, Pellegrino T, Dellegrottaglie S, Memmi A, Trimarco B, Cuocolo A, Filardi PP, Leosco D, Ferrara N. Impact of aging on cardiac sympathetic innervation measured by 123I-mIBG imaging in patients with systolic heart failure. Eur J Nucl Med Mol Imaging. 2016;43:2392-2400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 90. | Jacobson AF, Senior R, Cerqueira MD, Wong ND, Thomas GS, Lopez VA, Agostini D, Weiland F, Chandna H, Narula J; ADMIRE-HF Investigators. Myocardial iodine-123 meta-iodobenzylguanidine imaging and cardiac events in heart failure. Results of the prospective ADMIRE-HF (AdreView Myocardial Imaging for Risk Evaluation in Heart Failure) study. J Am Coll Cardiol. 2010;55:2212-2221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 672] [Cited by in RCA: 652] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 91. | de Diego AM, Gandía L, García AG. A physiological view of the central and peripheral mechanisms that regulate the release of catecholamines at the adrenal medulla. Acta Physiol (Oxf). 2008;192:287-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 86] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 92. | Philipp M, Hein L. Adrenergic receptor knockout mice: distinct functions of 9 receptor subtypes. Pharmacol Ther. 2004;101:65-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 113] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 93. | Rozec B, Gauthier C. beta3-adrenoceptors in the cardiovascular system: putative roles in human pathologies. Pharmacol Ther. 2006;111:652-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 134] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 94. | Rockman HA, Koch WJ, Lefkowitz RJ. Seven-transmembrane-spanning receptors and heart function. Nature. 2002;415:206-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 718] [Cited by in RCA: 689] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 95. | Gauthier C, Leblais V, Kobzik L, Trochu JN, Khandoudi N, Bril A, Balligand JL, Le Marec H. The negative inotropic effect of beta3-adrenoceptor stimulation is mediated by activation of a nitric oxide synthase pathway in human ventricle. J Clin Invest. 1998;102:1377-1384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 285] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 96. | Myagmar BE, Flynn JM, Cowley PM, Swigart PM, Montgomery MD, Thai K, Nair D, Gupta R, Deng DX, Hosoda C, Melov S, Baker AJ, Simpson PC. Adrenergic Receptors in Individual Ventricular Myocytes: The Beta-1 and Alpha-1B Are in All Cells, the Alpha-1A Is in a Subpopulation, and the Beta-2 and Beta-3 Are Mostly Absent. Circ Res. 2017;120:1103-1115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 97] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 97. | Reid JL. Alpha-adrenergic receptors and blood pressure control. Am J Cardiol. 1986;57:6E-12E. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 57] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 98. | Becker BK, Speed JS, Powell M, Pollock DM. Activation of neuronal endothelin B receptors mediates pressor response through alpha-1 adrenergic receptors. Physiol Rep. 2017;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 99. | Woo AY, Xiao RP. β-Adrenergic receptor subtype signaling in heart: from bench to bedside. Acta Pharmacol Sin. 2012;33:335-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 130] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 100. | Ali DC, Naveed M, Gordon A, Majeed F, Saeed M, Ogbuke MI, Atif M, Zubair HM, Changxing L. β-Adrenergic receptor, an essential target in cardiovascular diseases. Heart Fail Rev. 2020;25:343-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 101. | Madamanchi A. Beta-adrenergic receptor signaling in cardiac function and heart failure. Mcgill J Med. 2007;10:99-104. [PubMed] |
| 102. | Baruscotti M, Barbuti A, Bucchi A. The cardiac pacemaker current. J Mol Cell Cardiol. 2010;48:55-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 94] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 103. | Santulli G, Iaccarino G. Pinpointing beta adrenergic receptor in ageing pathophysiology: victim or executioner? Evidence from crime scenes. Immun Ageing. 2013;10:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 65] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 104. | Collins S, Cao W, Robidoux J. Learning new tricks from old dogs: beta-adrenergic receptors teach new lessons on firing up adipose tissue metabolism. Mol Endocrinol. 2004;18:2123-2131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 134] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 105. | Napp A, Brixius K, Pott C, Ziskoven C, Boelck B, Mehlhorn U, Schwinger RH, Bloch W. Effects of the beta3-adrenergic agonist BRL 37344 on endothelial nitric oxide synthase phosphorylation and force of contraction in human failing myocardium. J Card Fail. 2009;15:57-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 106. | Dessy C, Moniotte S, Ghisdal P, Havaux X, Noirhomme P, Balligand JL. Endothelial beta3-adrenoceptors mediate vasorelaxation of human coronary microarteries through nitric oxide and endothelium-dependent hyperpolarization. Circulation. 2004;110:948-954. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 121] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 107. | Dessy C, Balligand JL. Beta3-adrenergic receptors in cardiac and vascular tissues emerging concepts and therapeutic perspectives. Adv Pharmacol. 2010;59:135-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 73] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 108. | Imbrogno S, Angelone T, Adamo C, Pulerà E, Tota B, Cerra MC. Beta3-adrenoceptor in the eel (Anguilla anguilla) heart: negative inotropy and NO-cGMP-dependent mechanism. J Exp Biol. 2006;209:4966-4973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 109. | Moniotte S, Belge C, Sekkali B, Massion PB, Rozec B, Dessy C, Balligand JL. Sepsis is associated with an upregulation of functional beta3 adrenoceptors in the myocardium. Eur J Heart Fail. 2007;9:1163-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 37] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 110. | Gauthier C, Rozec B, Manoury B, Balligand JL. Beta-3 adrenoceptors as new therapeutic targets for cardiovascular pathologies. Curr Heart Fail Rep. 2011;8:184-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 35] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 111. | Rozec B, Erfanian M, Laurent K, Trochu JN, Gauthier C. Nebivolol, a vasodilating selective beta(1)-blocker, is a beta(3)-adrenoceptor agonist in the nonfailing transplanted human heart. J Am Coll Cardiol. 2009;53:1532-1538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 112. | Dessy C, Saliez J, Ghisdal P, Daneau G, Lobysheva II, Frérart F, Belge C, Jnaoui K, Noirhomme P, Feron O, Balligand JL. Endothelial beta3-adrenoreceptors mediate nitric oxide-dependent vasorelaxation of coronary microvessels in response to the third-generation beta-blocker nebivolol. Circulation. 2005;112:1198-1205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 164] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 113. | de Lucia C, Eguchi A, Koch WJ. New Insights in Cardiac β-Adrenergic Signaling During Heart Failure and Aging. Front Pharmacol. 2018;9:904. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 199] [Cited by in RCA: 205] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 114. | Gurevich VV, Gurevich EV. GPCR Signaling Regulation: The Role of GRKs and Arrestins. Front Pharmacol. 2019;10:125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 444] [Cited by in RCA: 389] [Article Influence: 64.8] [Reference Citation Analysis (0)] |
| 115. | Shukla AK, Xiao K, Lefkowitz RJ. Emerging paradigms of β-arrestin-dependent seven transmembrane receptor signaling. Trends Biochem Sci. 2011;36:457-469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 374] [Cited by in RCA: 356] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 116. | Sato PY, Chuprun JK, Schwartz M, Koch WJ. The evolving impact of g protein-coupled receptor kinases in cardiac health and disease. Physiol Rev. 2015;95:377-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 119] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 117. | Swedberg K, Viquerat C, Rouleau JL, Roizen M, Atherton B, Parmley WW, Chatterjee K. Comparison of myocardial catecholamine balance in chronic congestive heart failure and in angina pectoris without failure. Am J Cardiol. 1984;54:783-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 118] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 118. | Viquerat CE, Daly P, Swedberg K, Evers C, Curran D, Parmley WW, Chatterjee K. Endogenous catecholamine levels in chronic heart failure. Relation to the severity of hemodynamic abnormalities. Am J Med. 1985;78:455-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 106] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 119. | Kaye DM, Lambert GW, Lefkovits J, Morris M, Jennings G, Esler MD. Neurochemical evidence of cardiac sympathetic activation and increased central nervous system norepinephrine turnover in severe congestive heart failure. J Am Coll Cardiol. 1994;23:570-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 211] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 120. | Aggarwal A, Esler MD, Lambert GW, Hastings J, Johnston L, Kaye DM. Norepinephrine turnover is increased in suprabulbar subcortical brain regions and is related to whole-body sympathetic activity in human heart failure. Circulation. 2002;105:1031-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 121. | Zucker IH, Schultz HD, Patel KP, Wang W, Gao L. Regulation of central angiotensin type 1 receptors and sympathetic outflow in heart failure. Am J Physiol Heart Circ Physiol. 2009;297:H1557-H1566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 78] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 122. | Gao L, Wang WZ, Wang W, Zucker IH. Imbalance of angiotensin type 1 receptor and angiotensin II type 2 receptor in the rostral ventrolateral medulla: potential mechanism for sympathetic overactivity in heart failure. Hypertension. 2008;52:708-714. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 98] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 123. | Mäurer W, Tschada R, Manthey J, Ablasser A, Kübler W, Delius W, Gerlach E, Grobecker H, Kübler W. Catecholamines in Patients with Heart Failure. Delius W, Gerlach E, Grobecker H, Kübler W. Catecholamines and the Heart. Berlin, Heidelberg: Springer 1981; 236-246. [DOI] [Full Text] |
| 124. | Chidsey CA, Braunwald E, Morrow AG. Catecholamine Excretion and Cardiac Stores of Norepinephrine in Congestive Heart Failure. Am J Med. 1965;39:442-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 436] [Cited by in RCA: 397] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 125. | Ramchandra R, Hood SG, Denton DA, Woods RL, McKinley MJ, McAllen RM, May CN. Basis for the preferential activation of cardiac sympathetic nerve activity in heart failure. Proc Natl Acad Sci USA. 2009;106:924-928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 73] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 126. | Kaye DM, Lefkovits J, Jennings GL, Bergin P, Broughton A, Esler MD. Adverse consequences of high sympathetic nervous activity in the failing human heart. J Am Coll Cardiol. 1995;26:1257-1263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 497] [Cited by in RCA: 495] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 127. | Ramchandra R, Barrett CJ. Regulation of the renal sympathetic nerves in heart failure. Front Physiol. 2015;6:238. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 128. | Ramchandra R, Hood SG, Xing D, Lambert GW, May CN. Mechanisms underlying the increased cardiac norepinephrine spillover in heart failure. Am J Physiol Heart Circ Physiol. 2018;315:H340-H347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 129. | Cohn JN, Levine TB, Olivari MT, Garberg V, Lura D, Francis GS, Simon AB, Rector T. Plasma norepinephrine as a guide to prognosis in patients with chronic congestive heart failure. N Engl J Med. 1984;311:819-823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2411] [Cited by in RCA: 2236] [Article Influence: 54.5] [Reference Citation Analysis (0)] |
| 130. | Chidsey CA, Harrison DC, Braunwald E. Augmentation of the plasma nor-epinephrine response to exercise in patients with congestive heart failure. N Engl J Med. 1962;267:650-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 302] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 131. | Hasking GJ, Esler MD, Jennings GL, Burton D, Johns JA, Korner PI. Norepinephrine spillover to plasma in patients with congestive heart failure: evidence of increased overall and cardiorenal sympathetic nervous activity. Circulation. 1986;73:615-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 665] [Cited by in RCA: 634] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 132. | Bristow MR, Ginsburg R, Minobe W, Cubicciotti RS, Sageman WS, Lurie K, Billingham ME, Harrison DC, Stinson EB. Decreased catecholamine sensitivity and beta-adrenergic-receptor density in failing human hearts. N Engl J Med. 1982;307:205-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1753] [Cited by in RCA: 1590] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 133. | Freedman NJ, Lefkowitz RJ. Anti-beta(1)-adrenergic receptor antibodies and heart failure: causation, not just correlation. J Clin Invest. 2004;113:1379-1382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 81] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 134. | Mangmool S, Parichatikanond W, Kurose H. Therapeutic Targets for Treatment of Heart Failure: Focus on GRKs and β-Arrestins Affecting βAR Signaling. Front Pharmacol. 2018;9:1336. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 135. | Shah AS, White DC, Emani S, Kypson AP, Lilly RE, Wilson K, Glower DD, Lefkowitz RJ, Koch WJ. In vivo ventricular gene delivery of a beta-adrenergic receptor kinase inhibitor to the failing heart reverses cardiac dysfunction. Circulation. 2001;103:1311-1316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 125] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 136. | Liu WN, Fu KL, Gao HY, Shang YY, Wang ZH, Jiang GH, Zhang Y, Zhang W, Zhong M. β1 adrenergic receptor polymorphisms and heart failure: a meta-analysis on susceptibility, response to β-blocker therapy and prognosis. PLoS One. 2012;7:e37659. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 137. | Biolo A, Clausell N, Santos KG, Salvaro R, Ashton-Prolla P, Borges A, Rohde LE. Impact of beta1-adrenergic receptor polymorphisms on susceptibility to heart failure, arrhythmogenesis, prognosis, and response to beta-blocker therapy. Am J Cardiol. 2008;102:726-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 53] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 138. | Metaxa S, Missouris C, Mavrogianni D, Miliou A, Oikonomou E, Toli E, Kormali L, Vlismas A, Drakakis P, Tousoulis D. Polymorphism Gln27Glu of β2 Adrenergic Receptors in Patients with Ischaemic Cardiomyopathy. Curr Vasc Pharmacol. 2018;16:618-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 139. | Pezzali N, Curnis A, Specchia C, Carubelli V, Covolo L, Donato F, Auricchio A, Regoli F, Metra M. Adrenergic receptor gene polymorphism and left ventricular reverse remodelling after cardiac resynchronization therapy: preliminary results. Europace. 2013;15:1475-1481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 140. | Katsuumi G, Shimizu I, Yoshida Y, Hayashi Y, Ikegami R, Suda M, Wakasugi T, Nakao M, Minamino T. Catecholamine-Induced Senescence of Endothelial Cells and Bone Marrow Cells Promotes Cardiac Dysfunction in Mice. Int Heart J. 2018;59:837-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 141. | Bonnefont-Rousselot D, Mahmoudi A, Mougenot N, Varoquaux O, Le Nahour G, Fouret P, Lechat P. Catecholamine effects on cardiac remodelling, oxidative stress and fibrosis in experimental heart failure. Redox Rep. 2002;7:145-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 142. | Engelhardt S, Hein L, Wiesmann F, Lohse MJ. Progressive hypertrophy and heart failure in beta1-adrenergic receptor transgenic mice. Proc Natl Acad Sci USA. 1999;96:7059-7064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 567] [Cited by in RCA: 552] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 143. | Richard C. Stress-related cardiomyopathies. Ann Intensive Care. 2011;1:39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 144. | Chesley A, Lundberg MS, Asai T, Xiao RP, Ohtani S, Lakatta EG, Crow MT. The beta(2)-adrenergic receptor delivers an antiapoptotic signal to cardiac myocytes through G(i)-dependent coupling to phosphatidylinositol 3'-kinase. Circ Res. 2000;87:1172-1179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 290] [Cited by in RCA: 299] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 145. | Liggett SB, Tepe NM, Asai T, Lorenz JN, Canning AM, Jantz TD, Mitarai S, Yatani A, Dorn GW 2nd. Early and delayed consequences of beta(2)-adrenergic receptor overexpression in mouse hearts: critical role for expression level. Circulation. 2000;101:1707-1714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 269] [Cited by in RCA: 248] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 146. | Communal C, Singh K, Sawyer DB, Colucci WS. Opposing effects of beta(1)- and beta(2)-adrenergic receptors on cardiac myocyte apoptosis : role of a pertussis toxin-sensitive G protein. Circulation. 1999;100:2210-2212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 391] [Cited by in RCA: 391] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 147. | Rundqvist B, Elam M, Bergmann-Sverrisdottir Y, Eisenhofer G, Friberg P. Increased cardiac adrenergic drive precedes generalized sympathetic activation in human heart failure. Circulation. 1997;95:169-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 190] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 148. | Meredith IT, Broughton A, Jennings GL, Esler MD. Evidence of a selective increase in cardiac sympathetic activity in patients with sustained ventricular arrhythmias. N Engl J Med. 1991;325:618-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 303] [Cited by in RCA: 305] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 149. | Ishise H, Asanoi H, Ishizaka S, Joho S, Kameyama T, Umeno K, Inoue H. Time course of sympathovagal imbalance and left ventricular dysfunction in conscious dogs with heart failure. J Appl Physiol (1985). 1998;84:1234-1241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 68] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 150. | Ferguson DW, Berg WJ, Roach PJ, Oren RM, Mark AL. Effects of heart failure on baroreflex control of sympathetic neural activity. Am J Cardiol. 1992;69:523-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 157] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 151. | Grassi G, Seravalle G, Bertinieri G, Turri C, Stella ML, Scopelliti F, Mancia G. Sympathetic and reflex abnormalities in heart failure secondary to ischaemic or idiopathic dilated cardiomyopathy. Clin Sci (Lond). 2001;101:141-146. [PubMed] |
| 152. | Ferguson DW, Abboud FM, Mark AL. Selective impairment of baroreflex-mediated vasoconstrictor responses in patients with ventricular dysfunction. Circulation. 1984;69:451-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 154] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 153. | Floras JS. Arterial baroreceptor and cardiopulmonary reflex control of sympathetic outflow in human heart failure. Ann N Y Acad Sci. 2001;940:500-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 154. | Grassi G, Seravalle G, Cattaneo BM, Lanfranchi A, Vailati S, Giannattasio C, Del Bo A, Sala C, Bolla GB, Pozzi M. Sympathetic activation and loss of reflex sympathetic control in mild congestive heart failure. Circulation. 1995;92:3206-3211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 289] [Cited by in RCA: 293] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 155. | Grassi G, Seravalle G, Quarti-Trevano F, Dell'Oro R, Bolla G, Mancia G. Effects of hypertension and obesity on the sympathetic activation of heart failure patients. Hypertension. 2003;42:873-877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 82] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 156. | Gronda E, Seravalle G, Brambilla G, Costantino G, Casini A, Alsheraei A, Lovett EG, Mancia G, Grassi G. Chronic baroreflex activation effects on sympathetic nerve traffic, baroreflex function, and cardiac haemodynamics in heart failure: a proof-of-concept study. Eur J Heart Fail. 2014;16:977-983. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 140] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 157. | Gronda E, Francis D, Zannad F, Hamm C, Brugada J, Vanoli E. Baroreflex activation therapy: a new approach to the management of advanced heart failure with reduced ejection fraction. J Cardiovasc Med (Hagerstown). 2017;18:641-649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 158. | Shen MJ, Zipes DP. Role of the autonomic nervous system in modulating cardiac arrhythmias. Circ Res. 2014;114:1004-1021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 469] [Cited by in RCA: 595] [Article Influence: 54.1] [Reference Citation Analysis (0)] |
| 159. | Franciosi S, Perry FKG, Roston TM, Armstrong KR, Claydon VE, Sanatani S. The role of the autonomic nervous system in arrhythmias and sudden cardiac death. Auton Neurosci. 2017;205:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 105] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 160. | Porter B, Bishop MJ, Claridge S, Behar J, Sieniewicz BJ, Webb J, Gould J, O'Neill M, Rinaldi CA, Razavi R, Gill JS, Taggart P. Autonomic Modulation in Patients with Heart Failure Increases Beat-to-Beat Variability of Ventricular Action Potential Duration. Front Physiol. 2017;8:328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 161. | Brunner-La Rocca HP, Esler MD, Jennings GL, Kaye DM. Effect of cardiac sympathetic nervous activity on mode of death in congestive heart failure. Eur Heart J. 2001;22:1136-1143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 165] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 162. | Fukuda K, Kanazawa H, Aizawa Y, Ardell JL, Shivkumar K. Cardiac innervation and sudden cardiac death. Circ Res. 2015;116:2005-2019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 302] [Article Influence: 30.2] [Reference Citation Analysis (0)] |
| 163. | Fudim M, Boortz-Marx R, Ganesh A, Waldron NH, Qadri YJ, Patel CB, Milano CA, Sun AY, Mathew JP, Piccini JP. Stellate ganglion blockade for the treatment of refractory ventricular arrhythmias: A systematic review and meta-analysis. J Cardiovasc Electrophysiol. 2017;28:1460-1467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 81] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 164. | Meng L, Tseng CH, Shivkumar K, Ajijola O. Efficacy of Stellate Ganglion Blockade in Managing Electrical Storm: A Systematic Review. JACC Clin Electrophysiol. 2017;3:942-949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 116] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 165. | Fudim M, Qadri YJ, Waldron NH, Boortz-Marx RL, Ganesh A, Patel CB, Podgoreanu MV, Sun AY, Milano CA, Tong BC, Harpole DH, Mathew JP, Piccini JP. Stellate Ganglion Blockade for the Treatment of Refractory Ventricular Arrhythmias. JACC Clin Electrophysiol. 2020;6:562-571. [PubMed] |
| 166. | Zhou S, Jung BC, Tan AY, Trang VQ, Gholmieh G, Han SW, Lin SF, Fishbein MC, Chen PS, Chen LS. Spontaneous stellate ganglion nerve activity and ventricular arrhythmia in a canine model of sudden death. Heart Rhythm. 2008;5:131-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 153] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 167. | Wu P, Vaseghi M. The autonomic nervous system and ventricular arrhythmias in myocardial infarction and heart failure. Pacing Clin Electrophysiol. 2020;43:172-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 168. | Waldron NH, Fudim M, Mathew JP, Piccini JP. Neuromodulation for the Treatment of Heart Rhythm Disorders. JACC Basic Transl Sci. 2019;4:546-562. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 169. | Notarius CF, Ando S, Rongen GA, Floras JS. Resting muscle sympathetic nerve activity and peak oxygen uptake in heart failure and normal subjects. Eur Heart J. 1999;20:880-887. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 57] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 170. | Notarius CF, Azevedo ER, Parker JD, Floras JS. Peak oxygen uptake is not determined by cardiac noradrenaline spillover in heart failure. Eur Heart J. 2002;23:800-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 171. | Notarius CF, Spaak J, Morris BL, Floras JS. Comparison of muscle sympathetic activity in ischemic and nonischemic heart failure. J Card Fail. 2007;13:470-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 172. | Barrett-O'Keefe Z, Lee JF, Ives SJ, Trinity JD, Witman MAH, Rossman MJ, Groot HJ, Sorensen JR, Morgan DE, Nelson AD, Stehlik J, Richardson RS, Wray DW. α-Adrenergic receptor regulation of skeletal muscle blood flow during exercise in heart failure patients with reduced ejection fraction. Am J Physiol Regul Integr Comp Physiol. 2019;316:R512-R524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 173. | Colucci WS, Ribeiro JP, Rocco MB, Quigg RJ, Creager MA, Marsh JD, Gauthier DF, Hartley LH. Impaired chronotropic response to exercise in patients with congestive heart failure. Role of postsynaptic beta-adrenergic desensitization. Circulation. 1989;80:314-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 257] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 174. | Boutagy NE, Sinusas AJ. Imaging of the Cardiac Sympathetic Nervous System Has Potential Value in the Evaluation of Patients with Heart Failure with Preserved Ejection Fraction. J Nucl Med. 2017;58:781-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 175. | Matsunari I, Aoki H, Nomura Y, Takeda N, Chen WP, Taki J, Nakajima K, Nekolla SG, Kinuya S, Kajinami K. Iodine-123 metaiodobenzylguanidine imaging and carbon-11 hydroxyephedrine positron emission tomography compared in patients with left ventricular dysfunction. Circ Cardiovasc Imaging. 2010;3:595-603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 55] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 176. | Aikawa T, Naya M, Obara M, Manabe O, Tomiyama Y, Magota K, Yamada S, Katoh C, Tamaki N, Tsutsui H. Impaired Myocardial Sympathetic Innervation Is Associated with Diastolic Dysfunction in Heart Failure with Preserved Ejection Fraction: 11C-Hydroxyephedrine PET Study. J Nucl Med. 2017;58:784-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 177. | Aikawa T, Naya M, Obara M, Oyama-Manabe N, Manabe O, Magota K, Ito YM, Katoh C, Tamaki N. Regional interaction between myocardial sympathetic denervation, contractile dysfunction, and fibrosis in heart failure with preserved ejection fraction: 11C-hydroxyephedrine PET study. Eur J Nucl Med Mol Imaging. 2017;44:1897-1905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 178. | Shah AM, Bourgoun M, Narula J, Jacobson AF, Solomon SD. Influence of ejection fraction on the prognostic value of sympathetic innervation imaging with iodine-123 MIBG in heart failure. JACC Cardiovasc Imaging. 2012;5:1139-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 179. | Nakata T, Nakajima K, Yamashina S, Yamada T, Momose M, Kasama S, Matsui T, Matsuo S, Travin MI, Jacobson AF. A pooled analysis of multicenter cohort studies of (123)I-mIBG imaging of sympathetic innervation for assessment of long-term prognosis in heart failure. JACC Cardiovasc Imaging. 2013;6:772-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 161] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 180. | Verberne HJ, Brewster LM, Somsen GA, van Eck-Smit BL. Prognostic value of myocardial 123I-metaiodobenzylguanidine (MIBG) parameters in patients with heart failure: a systematic review. Eur Heart J. 2008;29:1147-1159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 200] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 181. | Narula J, Gerson M, Thomas GS, Cerqueira MD, Jacobson AF. ¹²³I-MIBG Imaging for Prediction of Mortality and Potentially Fatal Events in Heart Failure: The ADMIRE-HFX Study. J Nucl Med. 2015;56:1011-1018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 182. | Wakabayashi T, Nakata T, Hashimoto A, Yuda S, Tsuchihashi K, Travin MI, Shimamoto K. Assessment of underlying etiology and cardiac sympathetic innervation to identify patients at high risk of cardiac death. J Nucl Med. 2001;42:1757-1767. [PubMed] |
| 183. | Fallavollita JA, Heavey BM, Luisi AJ, Michalek SM, Baldwa S, Mashtare TL, Hutson AD, Dekemp RA, Haka MS, Sajjad M, Cimato TR, Curtis AB, Cain ME, Canty JM. Regional myocardial sympathetic denervation predicts the risk of sudden cardiac arrest in ischemic cardiomyopathy. J Am Coll Cardiol. 2014;63:141-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 292] [Cited by in RCA: 320] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 184. | Fallavollita JA, Dare JD, Carter RL, Baldwa S, Canty JM. Denervated Myocardium Is Preferentially Associated With Sudden Cardiac Arrest in Ischemic Cardiomyopathy: A Pilot Competing Risks Analysis of Cause-Specific Mortality. Circ Cardiovasc Imaging. 2017;10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 185. | Cain ME. Impact of denervated myocardium on improving risk stratification for sudden cardiac death. Trans Am Clin Climatol Assoc. 2014;125:141-53; discussion 153. [PubMed] |
| 186. | Seo M, Yamada T, Tamaki S, Watanabe T, Morita T, Furukawa Y, Kawasaki M, Kikuchi A, Kawai T, Abe M, Nakamura J, Yamamoto K, Kayama K, Kawahira M, Tanabe K, Kimura T, Ueda K, Sakamoto D, Sakata Y, Fukunami M. Prognostic significance of cardiac I-123-metaiodobenzylguanidine imaging in patients with reduced, mid-range, and preserved left ventricular ejection fraction admitted for acute decompensated heart failure: a prospective study in Osaka Prefectural Acute Heart Failure Registry (OPAR). Eur Heart J Cardiovasc Imaging. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 187. | Cleland JGF, Bunting KV, Flather MD, Altman DG, Holmes J, Coats AJS, Manzano L, McMurray JJV, Ruschitzka F, van Veldhuisen DJ, von Lueder TG, Böhm M, Andersson B, Kjekshus J, Packer M, Rigby AS, Rosano G, Wedel H, Hjalmarson Å, Wikstrand J, Kotecha D. Beta-blockers in Heart Failure Collaborative Group. Beta-blockers for heart failure with reduced, mid-range, and preserved ejection fraction: an individual patient-level analysis of double-blind randomized trials. Eur Heart J. 2018;39:26-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 399] [Cited by in RCA: 467] [Article Influence: 66.7] [Reference Citation Analysis (0)] |
| 188. | Ernst G. Heart-Rate Variability-More than Heart Beats? Front Public Health. 2017;5:240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 165] [Cited by in RCA: 230] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 189. | Shaffer F, McCraty R, Zerr CL. A healthy heart is not a metronome: an integrative review of the heart's anatomy and heart rate variability. Front Psychol. 2014;5:1040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 673] [Cited by in RCA: 1022] [Article Influence: 92.9] [Reference Citation Analysis (0)] |
| 190. | Rajendra Acharya U, Paul Joseph K, Kannathal N, Lim CM, Suri JS. Heart rate variability: a review. Med Biol Eng Comput. 2006;44:1031-1051. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1509] [Cited by in RCA: 1326] [Article Influence: 69.8] [Reference Citation Analysis (0)] |
| 191. | Shaffer F, Ginsberg JP. An Overview of Heart Rate Variability Metrics and Norms. Front Public Health. 2017;5:258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1840] [Cited by in RCA: 3141] [Article Influence: 392.6] [Reference Citation Analysis (0)] |
| 192. | Stys A, Stys T. Current clinical applications of heart rate variability. Clin Cardiol. 1998;21:719-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 193. | Tsuji H, Larson MG, Venditti FJ, Manders ES, Evans JC, Feldman CL, Levy D. Impact of reduced heart rate variability on risk for cardiac events. The Framingham Heart Study. Circulation. 1996;94:2850-2855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1201] [Cited by in RCA: 1230] [Article Influence: 42.4] [Reference Citation Analysis (0)] |
| 194. | Sessa F, Anna V, Messina G, Cibelli G, Monda V, Marsala G, Ruberto M, Biondi A, Cascio O, Bertozzi G, Pisanelli D, Maglietta F, Messina A, Mollica MP, Salerno M. Heart rate variability as predictive factor for sudden cardiac death. Aging (Albany NY). 2018;10:166-177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 180] [Cited by in RCA: 178] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 195. | Hillebrand S, Gast KB, de Mutsert R, Swenne CA, Jukema JW, Middeldorp S, Rosendaal FR, Dekkers OM. Heart rate variability and first cardiovascular event in populations without known cardiovascular disease: meta-analysis and dose-response meta-regression. Europace. 2013;15:742-749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 320] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 196. | Patel VN, Pierce BR, Bodapati RK, Brown DL, Ives DG, Stein PK. Association of Holter-Derived Heart Rate Variability Parameters With the Development of Congestive Heart Failure in the Cardiovascular Health Study. JACC Heart Fail. 2017;5:423-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 197. | Nolan J, Batin PD, Andrews R, Lindsay SJ, Brooksby P, Mullen M, Baig W, Flapan AD, Cowley A, Prescott RJ, Neilson JM, Fox KA. Prospective study of heart rate variability and mortality in chronic heart failure: results of the United Kingdom heart failure evaluation and assessment of risk trial (UK-heart). Circulation. 1998;98:1510-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 800] [Cited by in RCA: 813] [Article Influence: 30.1] [Reference Citation Analysis (0)] |
| 198. | Palacios M, Friedrich H, Götze C, Vallverdú M, de Luna AB, Caminal P, Hoyer D. Changes of autonomic information flow due to idiopathic dilated cardiomyopathy. Physiol Meas. 2007;28:677-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 199. | La Rovere MT, Pinna GD, Maestri R, Mortara A, Capomolla S, Febo O, Ferrari R, Franchini M, Gnemmi M, Opasich C, Riccardi PG, Traversi E, Cobelli F. Short-term heart rate variability strongly predicts sudden cardiac death in chronic heart failure patients. Circulation. 2003;107:565-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 613] [Cited by in RCA: 607] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 200. | Ponikowski P, Anker SD, Chua TP, Szelemej R, Piepoli M, Adamopoulos S, Webb-Peploe K, Harrington D, Banasiak W, Wrabec K, Coats AJ. Depressed heart rate variability as an independent predictor of death in chronic congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am J Cardiol. 1997;79:1645-1650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 333] [Cited by in RCA: 319] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 201. | Ponikowski P, Anker SD, Amadi A, Chua TP, Cerquetani E, Ondusova D, O'Sullivan C, Adamopoulos S, Piepoli M, Coats AJ. Heart rhythms, ventricular arrhythmias, and death in chronic heart failure. J Card Fail. 1996;2:177-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 202. | Aronson D, Mittleman MA, Burger AJ. Measures of heart period variability as predictors of mortality in hospitalized patients with decompensated congestive heart failure. Am J Cardiol. 2004;93:59-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 50] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 203. | Pousset F, Copie X, Lechat P, Jaillon P, Boissel JP, Hetzel M, Fillette F, Remme W, Guize L, Le Heuzey JY. Effects of bisoprolol on heart rate variability in heart failure. Am J Cardiol. 1996;77:612-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 84] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 204. | Aronson D, Burger AJ. Effect of beta-blockade on heart rate variability in decompensated heart failure. Int J Cardiol. 2001;79:31-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 48] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 205. | Hayano J, Yuda E. Pitfalls of assessment of autonomic function by heart rate variability. J Physiol Anthropol. 2019;38:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 192] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 206. | Lombardi F, Mortara A. Heart rate variability and cardiac failure. Heart. 1998;80:213-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 207. | Sarhene M, Wang Y, Wei J, Huang Y, Li M, Li L, Acheampong E, Zhengcan Z, Xiaoyan Q, Yunsheng X, Jingyuan M, Xiumei G, Guanwei F. Biomarkers in heart failure: the past, current and future. Heart Fail Rev. 2019;24:867-903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 83] [Article Influence: 16.6] [Reference Citation Analysis (2)] |
| 208. | Iqbal N, Wentworth B, Choudhary R, Landa Ade L, Kipper B, Fard A, Maisel AS. Cardiac biomarkers: new tools for heart failure management. Cardiovasc Diagn Ther. 2012;2:147-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 209. | Matsushita M, Shirakabe A, Kobayashi N, Okazaki H, Shibata Y, Goda H, Shigihara S, Asano K, Tani K, Kiuchi K, Okajima F, Hata N, Asai K, Shimizu W. Mechanisms of Urgently Presenting Acute Heart Failure. Int Heart J. 2020;61:316-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 210. | Francis GS, Cohn JN, Johnson G, Rector TS, Goldman S, Simon A. Plasma norepinephrine, plasma renin activity, and congestive heart failure. Relations to survival and the effects of therapy in V-HeFT II. The V-HeFT VA Cooperative Studies Group. Circulation. 1993;87:VI40-VI48. [PubMed] |
| 211. | Anand IS, Fisher LD, Chiang YT, Latini R, Masson S, Maggioni AP, Glazer RD, Tognoni G, Cohn JN; Val-HeFT Investigators. Changes in brain natriuretic peptide and norepinephrine over time and mortality and morbidity in the Valsartan Heart Failure Trial (Val-HeFT). Circulation. 2003;107:1278-1283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 639] [Cited by in RCA: 686] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 212. | Givertz MM, Braunwald E. Neurohormones in heart failure: predicting outcomes, optimizing care. Eur Heart J. 2004;25:281-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 213. | Hjemdahl P. Plasma catecholamines--analytical challenges and physiological limitations. Baillieres Clin Endocrinol Metab. 1993;7:307-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 90] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 214. | Habecker BA, Anderson ME, Birren SJ, Fukuda K, Herring N, Hoover DB, Kanazawa H, Paterson DJ, Ripplinger CM. Molecular and cellular neurocardiology: development, and cellular and molecular adaptations to heart disease. J Physiol. 2016;594:3853-3875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 76] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 215. | Tan CMJ, Green P, Tapoulal N, Lewandowski AJ, Leeson P, Herring N. The Role of Neuropeptide Y in Cardiovascular Health and Disease. Front Physiol. 2018;9:1281. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 142] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 216. | Herring N, Lokale MN, Danson EJ, Heaton DA, Paterson DJ. Neuropeptide Y reduces acetylcholine release and vagal bradycardia via a Y2 receptor-mediated, protein kinase C-dependent pathway. J Mol Cell Cardiol. 2008;44:477-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 71] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 217. | Heredia Mdel P, Delgado C, Pereira L, Perrier R, Richard S, Vassort G, Bénitah JP, Gómez AM. Neuropeptide Y rapidly enhances [Ca2+]i transients and Ca2+ sparks in adult rat ventricular myocytes through Y1 receptor and PLC activation. J Mol Cell Cardiol. 2005;38:205-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 54] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 218. | Tatemoto K. Neuropeptide Y: complete amino acid sequence of the brain peptide. Proc Natl Acad Sci USA. 1982;79:5485-5489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 760] [Cited by in RCA: 737] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 219. | Alasvand M, Javanmard SH, Rashidi B, Khazaei M. Myocardial capillary density after neuropeptide Y antagonist administration in normal and high-fat diet C57BL6 mice. Adv Biomed Res. 2016;5:165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 220. | Saraf R, Mahmood F, Amir R, Matyal R. Neuropeptide Y is an angiogenic factor in cardiovascular regeneration. Eur J Pharmacol. 2016;776:64-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 221. | Jacques D, D'Orléans-Juste P, Magder S, Bkaily G. Neuropeptide Y and its receptors in ventricular endocardial endothelial cells. Can J Physiol Pharmacol. 2017;95:1224-1229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 222. | Feng Q, Lambert ML, Callow ID, Arnold JM. Venous neuropeptide Y receptor responsiveness in patients with chronic heart failure. Clin Pharmacol Ther. 2000;67:292-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 223. | Feng QP, Sun XY, Hedner T. Cardiovascular responses and interactions by neuropeptide Y in rats with congestive heart failure. Blood Press. 1996;5:312-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 224. | Shanks J, Herring N. Peripheral cardiac sympathetic hyperactivity in cardiovascular disease: role of neuropeptides. Am J Physiol Regul Integr Comp Physiol. 2013;305:R1411-R1420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 225. | Feng QP, Hedner T, Andersson B, Lundberg JM, Waagstein F. Cardiac neuropeptide Y and noradrenaline balance in patients with congestive heart failure. Br Heart J. 1994;71:261-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 226. | Ullman B, Lindvall K, Lundberg JM, Sigurdsson A, Swedberg K. Response of plasma neuropeptide Y and noradrenaline to dynamic exercise and ramipril treatment in patients with congestive heart failure. Clin Physiol. 1994;14:123-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 227. | Maisel AS, Scott NA, Motulsky HJ, Michel MC, Boublik JH, Rivier JE, Ziegler M, Allen RS, Brown MR. Elevation of plasma neuropeptide Y levels in congestive heart failure. Am J Med. 1989;86:43-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 86] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 228. | Ajijola OA, Chatterjee NA, Gonzales MJ, Gornbein J, Liu K, Li D, Paterson DJ, Shivkumar K, Singh JP, Herring N. Coronary Sinus Neuropeptide Y Levels and Adverse Outcomes in Patients With Stable Chronic Heart Failure. JAMA Cardiol. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 229. | Morton GJ, Schwartz MW. The NPY/AgRP neuron and energy homeostasis. Int J Obes Relat Metab Disord. 2001;25 Suppl 5:S56-S62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 166] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 230. | Morris MJ, Cox HS, Lambert GW, Kaye DM, Jennings GL, Meredith IT, Esler MD. Region-specific neuropeptide Y overflows at rest and during sympathetic activation in humans. Hypertension. 1997;29:137-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 72] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 231. | Dubois-Randé JL, Comoy E, Merlet P, Benvenuti C, Carville C, Hittinger L, Macquin-Mavier I, Bohuon C, Castaigne A. Relationship among neuropeptide Y, catecholamines and haemodynamics in congestive heart failure. Eur Heart J. 1992;13:1233-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 232. | Medzikovic L, van Roomen C, Baartscheer A, van Loenen PB, de Vos J, Bakker ENTP, Koenis DS, Damanafshan A, Creemers EE, Arkenbout EK, de Vries CJM, de Waard V. Nur77 protects against adverse cardiac remodelling by limiting neuropeptide Y signalling in the sympathoadrenal-cardiac axis. Cardiovasc Res. 2018;114:1617-1628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 233. | Smith-White MA, Iismaa TP, Potter EK. Galanin and neuropeptide Y reduce cholinergic transmission in the heart of the anaesthetised mouse. Br J Pharmacol. 2003;140:170-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 234. | Herring N, Cranley J, Lokale MN, Li D, Shanks J, Alston EN, Girard BM, Carter E, Parsons RL, Habecker BA, Paterson DJ. The cardiac sympathetic co-transmitter galanin reduces acetylcholine release and vagal bradycardia: implications for neural control of cardiac excitability. J Mol Cell Cardiol. 2012;52:667-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 74] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 235. | Díaz-Cabiale Z, Parrado C, Narváez M, Millón C, Puigcerver A, Fuxe K, Narváez JA. Neurochemical modulation of central cardiovascular control: the integrative role of galanin. Exp Suppl. 2010;102:113-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 236. | Özkaramanlı Gür D, Sağbaş M, Akyüz A, Güzel S, Alpsoy Ş, Güler N. Role of sympathetic cotransmitter galanin on autonomic balance in heart failure: an active player or a bystander? Anatol J Cardiol. 2017;18:281-288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 237. | Chan NY, Robador PA, Levi R. Natriuretic peptide-induced catecholamine release from cardiac sympathetic neurons: inhibition by histamine H3 and H4 receptor activation. J Pharmacol Exp Ther. 2012;343:568-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 238. | Tyrrell C, Toyooka A, Khan F, Thornburg KL, Mudd JO, Hasan W. The neuropeptide galanin promotes an anti-thrombotic phenotype on endocardial endothelial cells from heart failure patients. Auton Neurosci. 2017;206:35-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 239. | Chen A, Li M, Song L, Zhang Y, Luo Z, Zhang W, Chen Y, He B. Effects of the Galanin Receptor Antagonist M40 on Cardiac Function and Remodeling in Rats with Heart Failure. Cardiovasc Ther. 2015;33:288-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 240. | Davenport AP, Hyndman KA, Dhaun N, Southan C, Kohan DE, Pollock JS, Pollock DM, Webb DJ, Maguire JJ. Endothelin. Pharmacol Rev. 2016;68:357-418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 616] [Cited by in RCA: 571] [Article Influence: 63.4] [Reference Citation Analysis (0)] |
| 241. | Agapitov AV, Haynes WG. Role of endothelin in cardiovascular disease. J Renin Angiotensin Aldosterone Syst. 2002;3:1-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 110] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 242. | Dhaun N, Webb DJ. Endothelins in cardiovascular biology and therapeutics. Nat Rev Cardiol. 2019;16:491-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 156] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 243. | Koseki C, Imai M, Hirata Y, Yanagisawa M, Masaki T. Autoradiographic distribution in rat tissues of binding sites for endothelin: a neuropeptide? Am J Physiol. 1989;256:R858-R866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 47] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 244. | Lehmann LH, Stanmore DA, Backs J. The role of endothelin-1 in the sympathetic nervous system in the heart. Life Sci. 2014;118:165-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 245. | Lepailleur-Enouf D, Egidy G, Philippe M, Louedec L, Henry J, Mulder P, Michel J. Pulmonary endothelinergic system in experimental congestive heart failure. Cardiovasc Res. 2001;49:330-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 246. | Stangl K, Dschietzig T, Richter C, Laule M, Stangl V, Tanis E, Baumann G, Felix SB. Pulmonary release and coronary and peripheral consumption of big endothelin and endothelin-1 in severe heart failure: acute effects of vasodilator therapy. Circulation. 2000;102:1132-1138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 247. | Giaid A, Yanagisawa M, Langleben D, Michel RP, Levy R, Shennib H, Kimura S, Masaki T, Duguid WP, Stewart DJ. Expression of endothelin-1 in the lungs of patients with pulmonary hypertension. N Engl J Med. 1993;328:1732-1739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1346] [Cited by in RCA: 1219] [Article Influence: 38.1] [Reference Citation Analysis (0)] |
| 248. | Lim K, van den Buuse M, Head GA. Effect of Endothelin-1 on Baroreflexes and the Cardiovascular Action of Clonidine in Conscious Rabbits. Front Physiol. 2016;7:321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 249. | Mosqueda-Garcia R, Inagami T, Appalsamy M, Sugiura M, Robertson RM. Endothelin as a neuropeptide. Cardiovascular effects in the brainstem of normotensive rats. Circ Res. 1993;72:20-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 64] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 250. | Friedrich EB, Muders F, Luchner A, Dietl O, Riegger GA, Elsner D. Contribution of the endothelin system to the renal hypoperfusion associated with experimental congestive heart failure. J Cardiovasc Pharmacol. 1999;34:612-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 251. | Sakai S, Miyauchi T, Kobayashi M, Yamaguchi I, Goto K, Sugishita Y. Inhibition of myocardial endothelin pathway improves long-term survival in heart failure. Nature. 1996;384:353-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 414] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 252. | Mulder P, Richard V, Derumeaux G, Hogie M, Henry JP, Lallemand F, Compagnon P, Macé B, Comoy E, Letac B, Thuillez C. Role of endogenous endothelin in chronic heart failure: effect of long-term treatment with an endothelin antagonist on survival, hemodynamics, and cardiac remodeling. Circulation. 1997;96:1976-1982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 225] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 253. | Yamauchi-Kohno R, Miyauchi T, Hoshino T, Kobayashi T, Aihara H, Sakai S, Yabana H, Goto K, Sugishita Y, Murata S. Role of endothelin in deterioration of heart failure due to cardiomyopathy in hamsters: increase in endothelin-1 production in the heart and beneficial effect of endothelin-A receptor antagonist on survival and cardiac function. Circulation. 1999;99:2171-2176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 67] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 254. | Cody RJ, Haas GJ, Binkley PF, Capers Q, Kelley R. Plasma endothelin correlates with the extent of pulmonary hypertension in patients with chronic congestive heart failure. Circulation. 1992;85:504-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 444] [Cited by in RCA: 409] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 255. | Abukar Y, May CN, Ramchandra R. Role of endothelin-1 in mediating changes in cardiac sympathetic nerve activity in heart failure. Am J Physiol Regul Integr Comp Physiol. 2016;310:R94-R99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 256. | Tsuchiyama Y, Kasamatsu K, Hano T, Nishio I. Acute effect of endothelin AB antagonist on sympathetic outflow in conscious rats with heart failure. Circ J. 2002;66:841-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 257. | Fraccarollo D, Bauersachs J, Kellner M, Galuppo P, Ertl G. Cardioprotection by long-term ET(A) receptor blockade and ACE inhibition in rats with congestive heart failure: mono- versus combination therapy. Cardiovasc Res. 2002;54:85-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 258. | Lehmann LH, Rostosky JS, Buss SJ, Kreusser MM, Krebs J, Mier W, Enseleit F, Spiger K, Hardt SE, Wieland T, Haass M, Lüscher TF, Schneider MD, Parlato R, Gröne HJ, Haberkorn U, Yanagisawa M, Katus HA, Backs J. Essential role of sympathetic endothelin A receptors for adverse cardiac remodeling. Proc Natl Acad Sci USA. 2014;111:13499-13504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 259. | Kopp UC, Cicha MZ, Jones SY. Activation of endothelin A receptors contributes to impaired responsiveness of renal mechanosensory nerves in congestive heart failure. Can J Physiol Pharmacol. 2010;88:622-629. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 260. | Backs J, Bresch E, Lutz M, Kristen AV, Haass M. Endothelin-1 inhibits the neuronal norepinephrine transporter in hearts of male rats. Cardiovasc Res. 2005;67:283-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 261. | Khalil RA. Modulators of the vascular endothelin receptor in blood pressure regulation and hypertension. Curr Mol Pharmacol. 2011;4:176-186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 262. | McMurray JJ, Teerlink JR, Cotter G, Bourge RC, Cleland JG, Jondeau G, Krum H, Metra M, O'Connor CM, Parker JD, Torre-Amione G, van Veldhuisen DJ, Lewsey J, Frey A, Rainisio M, Kobrin I; VERITAS Investigators. Effects of tezosentan on symptoms and clinical outcomes in patients with acute heart failure: the VERITAS randomized controlled trials. JAMA. 2007;298:2009-2019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 284] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 263. | O'Connor CM, Gattis WA, Adams KF, Hasselblad V, Chandler B, Frey A, Kobrin I, Rainisio M, Shah MR, Teerlink J, Gheorghiade M; Randomized Intravenous TeZosentan Study-4 Investigators. Tezosentan in patients with acute heart failure and acute coronary syndromes: results of the Randomized Intravenous TeZosentan Study (RITZ-4). J Am Coll Cardiol. 2003;41:1452-1457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 109] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 264. | Perez AL, Grodin JL, Wu Y, Hernandez AF, Butler J, Metra M, Felker GM, Voors AA, McMurray JJ, Armstrong PW, Starling RC, O'Connor CM, Tang WH. Increased mortality with elevated plasma endothelin-1 in acute heart failure: an ASCEND-HF biomarker substudy. Eur J Heart Fail. 2016;18:290-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 265. | Olivier A, Girerd N, Michel JB, Ketelslegers JM, Fay R, Vincent J, Bramlage P, Pitt B, Zannad F, Rossignol P; EPHESUS Investigators. Combined baseline and one-month changes in big endothelin-1 and brain natriuretic peptide plasma concentrations predict clinical outcomes in patients with left ventricular dysfunction after acute myocardial infarction: Insights from the Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study (EPHESUS) study. Int J Cardiol. 2017;241:344-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 266. | Plumpton C, Haynes WG, Webb DJ, Davenport AP. Measurement of C-terminal fragment of big endothelin-1: a novel method for assessing the generation of endothelin-1 in humans. J Cardiovasc Pharmacol. 1995;26 Suppl 3:S34-S36. [PubMed] |
| 267. | Wei CM, Lerman A, Rodeheffer RJ, McGregor CG, Brandt RR, Wright S, Heublein DM, Kao PC, Edwards WD, Burnett JC. Endothelin in human congestive heart failure. Circulation. 1994;89:1580-1586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 399] [Cited by in RCA: 377] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 268. | McMurray JJ, Ray SG, Abdullah I, Dargie HJ, Morton JJ. Plasma endothelin in chronic heart failure. Circulation. 1992;85:1374-1379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 336] [Cited by in RCA: 327] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 269. | Jankowska EA, Filippatos GS, von Haehling S, Papassotiriou J, Morgenthaler NG, Cicoira M, Schefold JC, Rozentryt P, Ponikowska B, Doehner W, Banasiak W, Hartmann O, Struck J, Bergmann A, Anker SD, Ponikowski P. Identification of chronic heart failure patients with a high 12-month mortality risk using biomarkers including plasma C-terminal pro-endothelin-1. PLoS One. 2011;6:e14506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 270. | Masson S, Latini R, Anand IS, Barlera S, Judd D, Salio M, Perticone F, Perini G, Tognoni G, Cohn JN; Val-HeFT investigators. The prognostic value of big endothelin-1 in more than 2,300 patients with heart failure enrolled in the Valsartan Heart Failure Trial (Val-HeFT). J Card Fail. 2006;12:375-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 53] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 271. | Van Beneden R, Gurné O, Selvais PL, Ahn SA, Robert AR, Ketelslegers JM, Pouleur HG, Rousseau MF. Superiority of big endothelin-1 and endothelin-1 over natriuretic peptides in predicting survival in severe congestive heart failure: a 7-year follow-up study. J Card Fail. 2004;10:490-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 59] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 272. | Zhang CL, Xie S, Qiao X, An YM, Zhang Y, Li L, Guo XB, Zhang FC, Wu LL. Plasma endothelin-1-related peptides as the prognostic biomarkers for heart failure: A PRISMA-compliant meta-analysis. Medicine (Baltimore). 2017;96:e9342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 273. | Xiong B, Nie D, Cao Y, Zou Y, Yao Y, Tan J, Qian J, Rong S, Wang C, Huang J. Clinical and Hemodynamic Effects of Endothelin Receptor Antagonists in Patients With Heart Failure. Int Heart J. 2017;58:400-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 274. | Chowdhury MA, Moukarbel GV, Gupta R, Frank SM, Anderson AM, Liu LC, Khouri SJ. Endothelin 1 Is Associated with Heart Failure Hospitalization and Long-Term Mortality in Patients with Heart Failure with Preserved Ejection Fraction and Pulmonary Hypertension. Cardiology. 2019;143:124-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 275. | Obokata M, Kane GC, Reddy YNV, Melenovsky V, Olson TP, Jarolim P, Borlaug BA. The neurohormonal basis of pulmonary hypertension in heart failure with preserved ejection fraction. Eur Heart J. 2019;40:3707-3717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 276. | Jankowich MD, Wu WC, Choudhary G. Association of Elevated Plasma Endothelin-1 Levels With Pulmonary Hypertension, Mortality, and Heart Failure in African American Individuals: The Jackson Heart Study. JAMA Cardiol. 2016;1:461-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 277. | Omland T. Targeting the endothelin system: a step towards a precision medicine approach in heart failure with preserved ejection fraction? Eur Heart J. 2019;40:3718-3720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 278. | Valero-Munoz M, Li S, Wilson RM, Boldbaatar B, Iglarz M, Sam F. Dual Endothelin-A/Endothelin-B Receptor Blockade and Cardiac Remodeling in Heart Failure With Preserved Ejection Fraction. Circ Heart Fail. 2016;9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 65] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 279. | Zile MR, Bourge RC, Redfield MM, Zhou D, Baicu CF, Little WC. Randomized, double-blind, placebo-controlled study of sitaxsentan to improve impaired exercise tolerance in patients with heart failure and a preserved ejection fraction. JACC Heart Fail. 2014;2:123-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 63] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 280. | Koller B, Steringer-Mascherbauer R, Ebner CH, Weber T, Ammer M, Eichinger J, Pretsch I, Herold M, Schwaiger J, Ulmer H, Grander W. Pilot Study of Endothelin Receptor Blockade in Heart Failure with Diastolic Dysfunction and Pulmonary Hypertension (BADDHY-Trial). Heart Lung Circ. 2017;26:433-441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 67] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 281. | Mahata SK, O'Connor DT, Mahata M, Yoo SH, Taupenot L, Wu H, Gill BM, Parmer RJ. Novel autocrine feedback control of catecholamine release. A discrete chromogranin a fragment is a noncompetitive nicotinic cholinergic antagonist. J Clin Invest. 1997;100:1623-1633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 316] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 282. | Taupenot L, Harper KL, O'Connor DT. The chromogranin-secretogranin family. N Engl J Med. 2003;348:1134-1149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 632] [Cited by in RCA: 625] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 283. | Mahata SK, Corti A. Chromogranin A and its fragments in cardiovascular, immunometabolic, and cancer regulation. Ann N Y Acad Sci. 2019;1455:34-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 59] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 284. | Takiyyuddin MA, Brown MR, Dinh TQ, Cervenka JH, Braun SD, Parmer RJ, Kennedy B, O'Connor DT. Sympatho-adrenal secretion in humans: factors governing catecholamine and storage vesicle peptide co-release. J Auton Pharmacol. 1994;14:187-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 285. | Ceconi C, Ferrari R, Bachetti T, Opasich C, Volterrani M, Colombo B, Parrinello G, Corti A. Chromogranin A in heart failure; a novel neurohumoral factor and a predictor for mortality. Eur Heart J. 2002;23:967-974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 171] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 286. | Omland T, Dickstein K, Syversen U. Association between plasma chromogranin A concentration and long-term mortality after myocardial infarction. Am J Med. 2003;114:25-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 51] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 287. | Estensen ME, Hognestad A, Syversen U, Squire I, Ng L, Kjekshus J, Dickstein K, Omland T. Prognostic value of plasma chromogranin A levels in patients with complicated myocardial infarction. Am Heart J. 2006;152:927.e1-927.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 53] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 288. | Pieroni M, Corti A, Tota B, Curnis F, Angelone T, Colombo B, Cerra MC, Bellocci F, Crea F, Maseri A. Myocardial production of chromogranin A in human heart: a new regulatory peptide of cardiac function. Eur Heart J. 2007;28:1117-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 124] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 289. | Mahata SK, Mahata M, Fung MM, O'Connor DT. Catestatin: a multifunctional peptide from chromogranin A. Regul Pept. 2010;162:33-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 88] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 290. | Mahata SK, Kiranmayi M, Mahapatra NR. Catestatin: A Master Regulator of Cardiovascular Functions. Curr Med Chem. 2018;25:1352-1374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 291. | Fung MM, Salem RM, Mehtani P, Thomas B, Lu CF, Perez B, Rao F, Stridsberg M, Ziegler MG, Mahata SK, O'Connor DT. Direct vasoactive effects of the chromogranin A (CHGA) peptide catestatin in humans in vivo. Clin Exp Hypertens. 2010;32:278-287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 71] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 292. | Krüger PG, Mahata SK, Helle KB. Catestatin (CgA344-364) stimulates rat mast cell release of histamine in a manner comparable to mastoparan and other cationic charged neuropeptides. Regul Pept. 2003;114:29-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 63] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 293. | Kiranmayi M, Chirasani VR, Allu PK, Subramanian L, Martelli EE, Sahu BS, Vishnuprabu D, Kumaragurubaran R, Sharma S, Bodhini D, Dixit M, Munirajan AK, Khullar M, Radha V, Mohan V, Mullasari AS, Naga Prasad SV, Senapati S, Mahapatra NR. Catestatin Gly364Ser Variant Alters Systemic Blood Pressure and the Risk for Hypertension in Human Populations via Endothelial Nitric Oxide Pathway. Hypertension. 2016;68:334-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 294. | Kennedy BP, Mahata SK, O'Connor DT, Ziegler MG. Mechanism of cardiovascular actions of the chromogranin A fragment catestatin in vivo. Peptides. 1998;19:1241-1248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 106] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 295. | Troger J, Theurl M, Kirchmair R, Pasqua T, Tota B, Angelone T, Cerra MC, Nowosielski Y, Mätzler R, Troger J, Gayen JR, Trudeau V, Corti A, Helle KB. Granin-derived peptides. Prog Neurobiol. 2017;154:37-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 54] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 296. | Mahapatra NR, Mahata M, Mahata SK, O'Connor DT. The chromogranin A fragment catestatin: specificity, potency and mechanism to inhibit exocytotic secretion of multiple catecholamine storage vesicle co-transmitters. J Hypertens. 2006;24:895-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 297. | Gaede AH, Pilowsky PM. Catestatin in rat RVLM is sympathoexcitatory, increases barosensitivity, and attenuates chemosensitivity and the somatosympathetic reflex. Am J Physiol Regul Integr Comp Physiol. 2010;299:R1538-R1545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 298. | Gaede AH, Pilowsky PM. Catestatin, a chromogranin A-derived peptide, is sympathoinhibitory and attenuates sympathetic barosensitivity and the chemoreflex in rat CVLM. Am J Physiol Regul Integr Comp Physiol. 2012;302:R365-R372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 299. | Bassino E, Fornero S, Gallo MP, Ramella R, Mahata SK, Tota B, Levi R, Alloatti G. A novel catestatin-induced antiadrenergic mechanism triggered by the endothelial PI3K-eNOS pathway in the myocardium. Cardiovasc Res. 2011;91:617-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 300. | Imbrogno S, Garofalo F, Cerra MC, Mahata SK, Tota B. The catecholamine release-inhibitory peptide catestatin (chromogranin A344-363) modulates myocardial function in fish. J Exp Biol. 2010;213:3636-3643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 301. | Bassino E, Fornero S, Gallo MP, Gallina C, Femminò S, Levi R, Tota B, Alloatti G. Catestatin exerts direct protective effects on rat cardiomyocytes undergoing ischemia/reperfusion by stimulating PI3K-Akt-GSK3β pathway and preserving mitochondrial membrane potential. PLoS One. 2015;10:e0119790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 302. | Theurl M, Schgoer W, Albrecht K, Jeschke J, Egger M, Beer AG, Vasiljevic D, Rong S, Wolf AM, Bahlmann FH, Patsch JR, Wolf D, Schratzberger P, Mahata SK, Kirchmair R. The neuropeptide catestatin acts as a novel angiogenic cytokine via a basic fibroblast growth factor-dependent mechanism. Circ Res. 2010;107:1326-1335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 78] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 303. | Guo X, Zhou C, Sun N. The neuropeptide catestatin promotes vascular smooth muscle cell proliferation through the Ca2+-calcineurin-NFAT signaling pathway. Biochem Biophys Res Commun. 2011;407:807-812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 304. | Chen H, Liu D, Ge L, Wang T, Ma Z, Han Y, Duan Y, Xu X, Liu W, Yuan J, Liu J, Li R, Du R. Catestatin prevents endothelial inflammation and promotes thrombus resolution in acute pulmonary embolism in mice. Biosci Rep. 2019;39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 305. | Chen Y, Wang X, Yang C, Su X, Yang W, Dai Y, Han H, Jiang J, Lu L, Wang H, Chen Q, Jin W. Decreased circulating catestatin levels are associated with coronary artery disease: The emerging anti-inflammatory role. Atherosclerosis. 2019;281:78-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 306. | Chu SY, Peng F, Wang J, Liu L, Meng L, Zhao J, Han XN, Ding WH. Catestatin in defense of oxidative-stress-induced apoptosis: A novel mechanism by activating the beta2 adrenergic receptor and PKB/Akt pathway in ischemic-reperfused myocardium. Peptides. 2020;123:170200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 307. | Alam MJ, Gupta R, Mahapatra NR, Goswami SK. Catestatin reverses the hypertrophic effects of norepinephrine in H9c2 cardiac myoblasts by modulating the adrenergic signaling. Mol Cell Biochem. 2020;464:205-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 308. | Angelone T, Quintieri AM, Pasqua T, Gentile S, Tota B, Mahata SK, Cerra MC. Phosphodiesterase type-2 and NO-dependent S-nitrosylation mediate the cardioinhibition of the antihypertensive catestatin. Am J Physiol Heart Circ Physiol. 2012;302:H431-H442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 309. | Wang D, Liu T, Shi S, Li R, Shan Y, Huang Y, Hu D, Huang C. Chronic Administration of Catestatin Improves Autonomic Function and Exerts Cardioprotective Effects in Myocardial Infarction Rats. J Cardiovasc Pharmacol Ther. 2016;21:526-535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 310. | Dev NB, Mir SA, Gayen JR, Siddiqui JA, Mustapic M, Vaingankar SM. Cardiac electrical activity in a genomically "humanized" chromogranin a monogenic mouse model with hyperadrenergic hypertension. J Cardiovasc Transl Res. 2014;7:483-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 311. | Pei Z, Ma D, Ji L, Zhang J, Su J, Xue W, Chen X, Wang W. Usefulness of catestatin to predict malignant arrhythmia in patients with acute myocardial infarction. Peptides. 2014;55:131-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 312. | Zhu D, Wang F, Yu H, Mi L, Gao W. Catestatin is useful in detecting patients with stage B heart failure. Biomarkers. 2011;16:691-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 313. | Liu L, Ding W, Li R, Ye X, Zhao J, Jiang J, Meng L, Wang J, Chu S, Han X, Peng F. Plasma levels and diagnostic value of catestatin in patients with heart failure. Peptides. 2013;46:20-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 314. | Borovac JA, Glavas D, Susilovic Grabovac Z, Supe Domic D, D'Amario D, Bozic J. Catestatin in Acutely Decompensated Heart Failure Patients: Insights from the CATSTAT-HF Study. J Clin Med. 2019;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 315. | Meng L, Ye XJ, Ding WH, Yang Y, Di BB, Liu L, Huo Y. Plasma catecholamine release-inhibitory peptide catestatin in patients with essential hypertension. J Cardiovasc Med (Hagerstown). 2011;12:643-647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 316. | Peng F, Chu S, Ding W, Liu L, Zhao J, Cui X, Li R, Wang J. The predictive value of plasma catestatin for all-cause and cardiac deaths in chronic heart failure patients. Peptides. 2016;86:112-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |