Published online May 26, 2020. doi: 10.4330/wjc.v12.i5.167
Peer-review started: December 23, 2019
First decision: February 20, 2020
Revised: March 21, 2020
Accepted: March 26, 2020
Article in press: March 26, 2020
Published online: May 26, 2020
Processing time: 154 Days and 5.9 Hours
Coarctation of the aorta (CoA) is a relatively common congenital cardiac defect often causing few symptoms and therefore can be challenging to diagnose. The hallmark finding on physical examination is upper extremity hypertension, and for this reason, CoA should be considered in any young hypertensive patient, justifying measurement of lower extremity blood pressure at least once in these individuals. The presence of a significant pressure gradient between the arms and legs is highly suggestive of the diagnosis. Early diagnosis and treatment are important as long-term data consistently demonstrate that patients with CoA have a reduced life expectancy and increased risk of cardiovascular complications. Surgical repair has traditionally been the mainstay of therapy for correction, although advances in endovascular technology with covered stents or stent grafts permit nonsurgical approaches for the management of older children and adults with native CoA and complications. Persistent hypertension and vascular dysfunction can lead to an increased risk of coronary disease, which, remains the greatest cause of long-term mortality. Thus, blood pressure control and periodic reassessment with transthoracic echocardiography and three-dimensional imaging (computed tomography or cardiac magnetic resonance) for should be performed regularly as cardiovascular complications may occur decades after the intervention.
Core tip: Coarctation of the aorta (CoA) is a common congenital cardiac defect with few symptoms and difficult to diagnose. Early diagnosis and treatment are important as long-term data demonstrate reduced life expectancy and increased risk of cardiovascular complications. Surgical repair has traditionally been the mainstay of therapy for correction, although advances in endovascular technology with covered stents and stent grafts permit nonsurgical approaches for the management of children and adults with native CoA and complications. Persistent hypertension and vascular dysfunction can lead to an increased risk of coronary disease, which remains the greatest cause of long-term mortality.
- Citation: Agasthi P, Pujari SH, Tseng A, Graziano JN, Marcotte F, Majdalany D, Mookadam F, Hagler DJ, Arsanjani R. Management of adults with coarctation of aorta. World J Cardiol 2020; 12(5): 167-191
- URL: https://www.wjgnet.com/1949-8462/full/v12/i5/167.htm
- DOI: https://dx.doi.org/10.4330/wjc.v12.i5.167
Coarctation of the aorta (CoA) is one of the most common congenital cardiac anomalies causing a narrowing of the proximal descending[1,2] varying within a range of arch abnormalities from a discrete narrowing to a long segment of arch hypoplasia[2]. It was first described by Thiene[3] in 1760 and accounts for 4%-6% of all congenital heart defects, with an incidence is of 3-4 cases per 10000 live births[4,5]. Males are commonly affected than females with a ratio between 1.27:1 and 1.74:1, respectively[1,6-8].
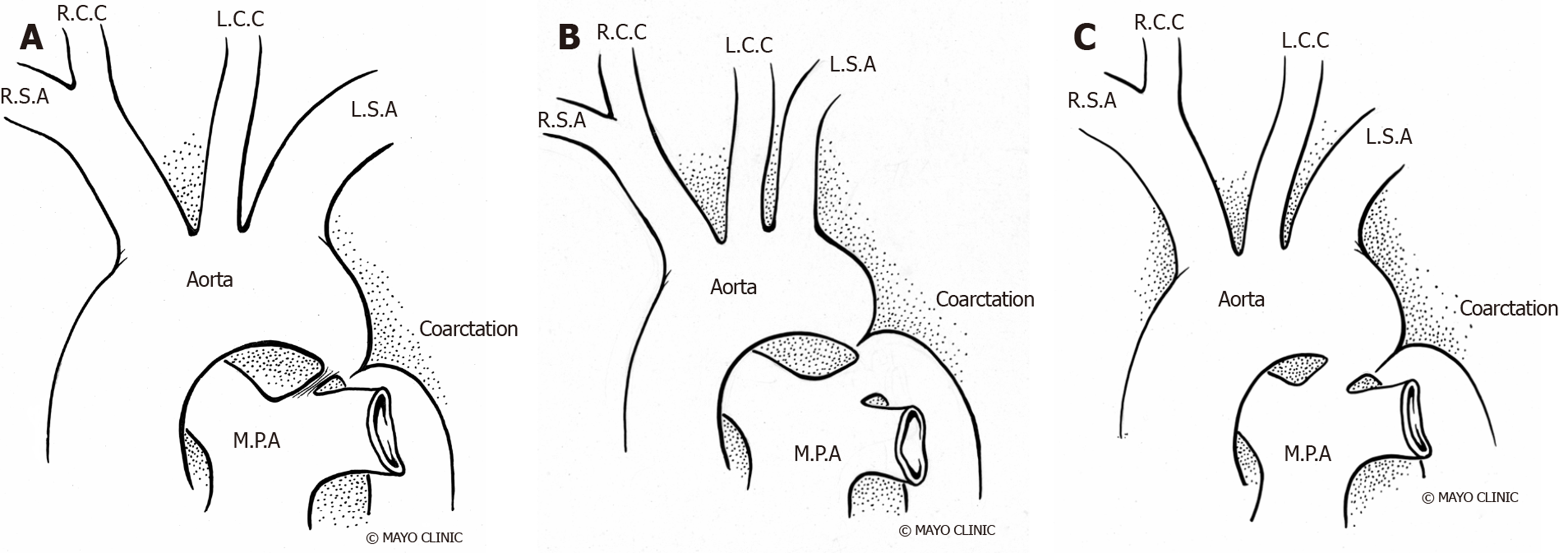
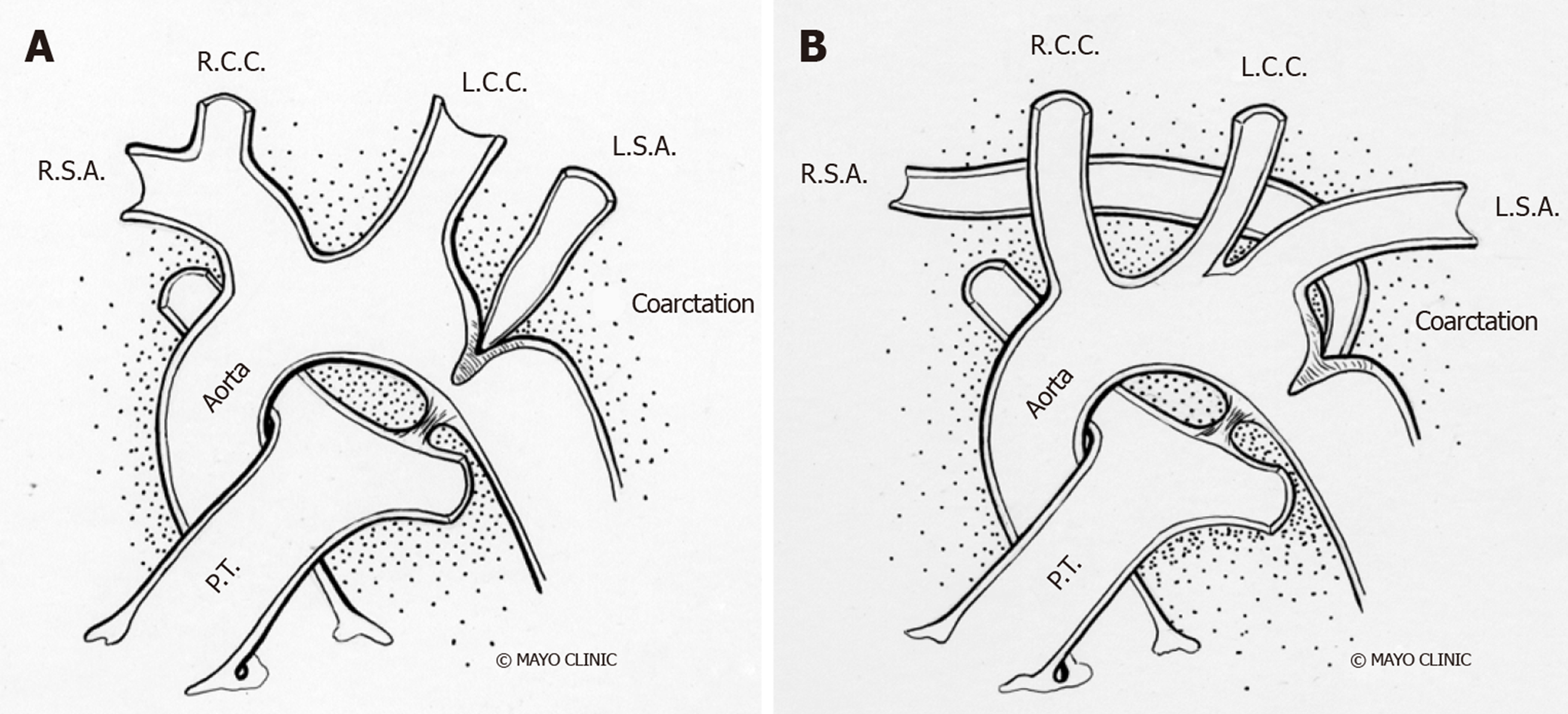
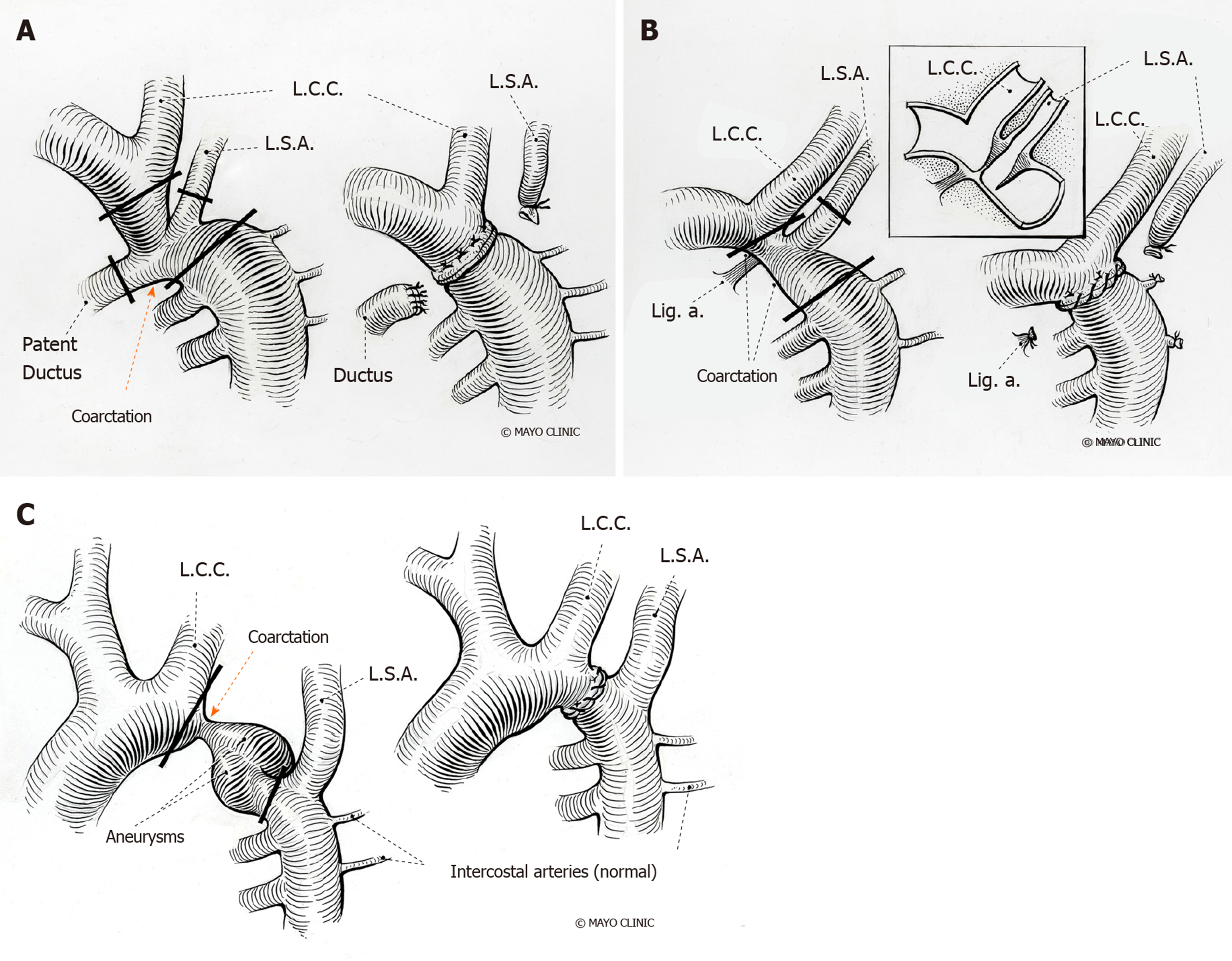
The narrowing of CoA is classically situated between the left subclavian artery and the ligamentum arteriosum at the aortic isthmus. CoA can also occur in atypical locations, between the transverse aortic arch and the bifurcation of the abdominal aorta[9]. Traditionally, CoA was classified as pre-ductal, juxta-ductal, and post-ductal based on its relationship with the ductus arteriosus (Figure 1). However, the degree of narrowing of the aorta and the association with arch vessels are regarded as more clinically relevant[10]. Approximately 40%-80% of patients have associated transverse arch hypoplasia, which varies in severity and must be accurately defined prior to planned intervention[11-13]. Although the ratio of the narrowed segment’s diameter to that of the distal descending aorta was formerly used to define the degree of CoA[14], arch hypoplasia is currently defined by z-scores of -2 or lower[15]. Rarely, we can observe tubular hypoplasia representing a combination of small arch diameter and increased length between segments of the arch[16]. CoA ranges in severity from complete aortic luminal atresia to a mild, precisely defined posterior shelf like lesion, and it needs to be differentiated from an interrupted aortic arch, in which there is a true discontinuity of the aortic walls (Figure 1)[17].
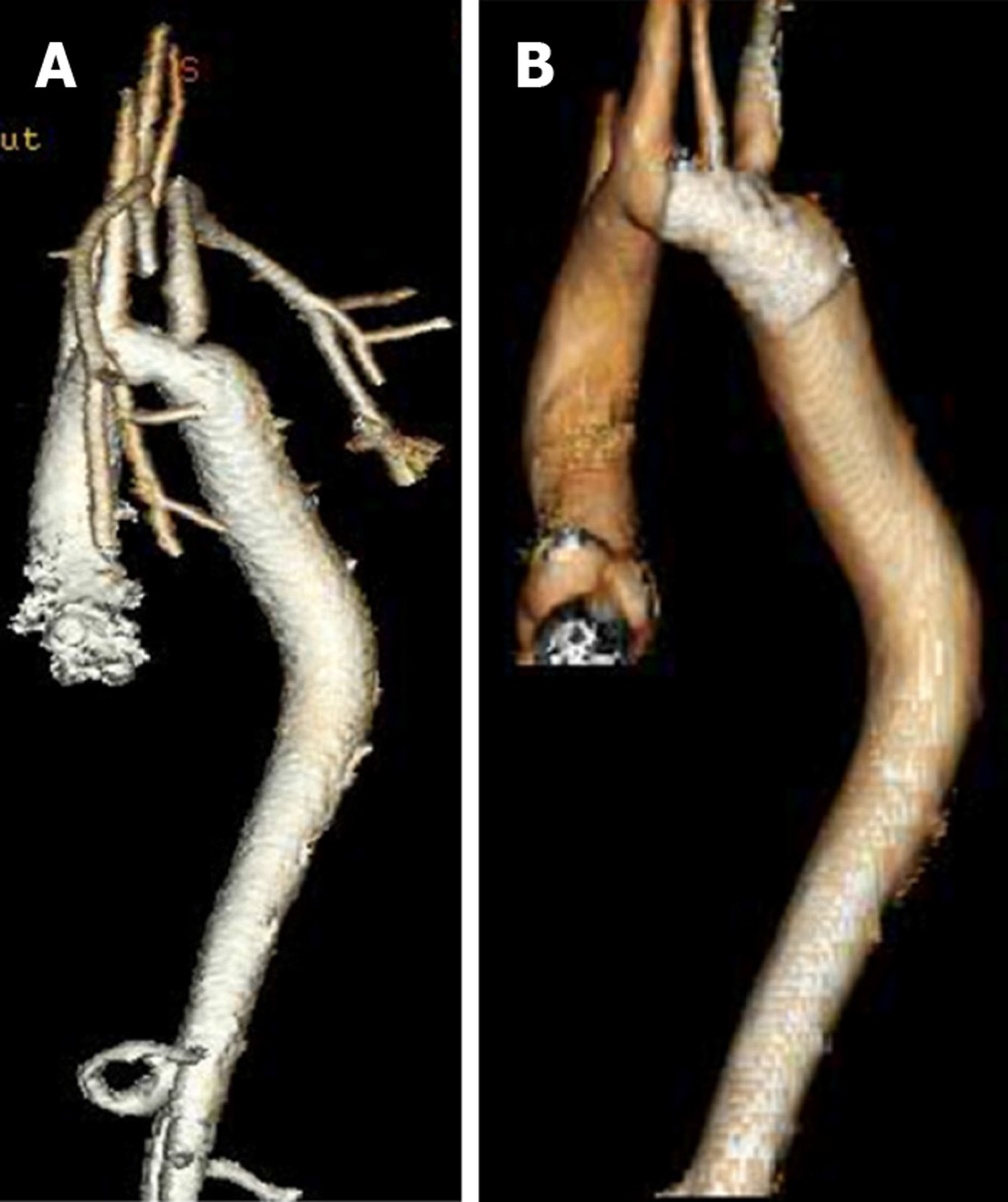
Studies have shown evidence of a common molecular pathogenic mechanism for the co-existence of CoA with other left heart obstructive pathologies such as aortic stenosis and hypoplastic left heart syndrome[18-20]. Although 75% of patients with CoA have a co-existent bicuspid aortic valve (BAV)[21,22], in patients with BAV and previous repaired CoA, the probability of requiring surgery for the aortic valve or ascending aorta is lower than in isolated BAV[23] with a slower progression of ascending aorta dilation on serial echocardiography[24]. Higher prevalence of CoA in BAV patients justify the need for routine screening and arch imaging for CoA in patients with BAV[25]. Other associated cardiac anomalies include left heart obstructive lesions like cor triatriatum, parachute mitral valve and discrete subaortic stenosis, referred as Shone syndrome as well as ventricular septal defect, patent ductus arteriosus, double outlet right ventricle, transposition of great arteries, atrio-ventricular canal defects[22,26-28] and rarely, aberrant arch vessels (Figure 2).
Chromosomal abnormalities like Turner’s syndrome (23 X0) can be associated with multiple congenital extra-cardiac anomalies (short stature, wide set nipples, webbed neck and infertility), aortic aneurysm, an 18% incidence of coarctation[29] and increased risk of aortic dissection[30-32]. CoA is also associated with a number of syndromes like PHACES (posterior fossa malformations, hemangiomas, arterial anomalies, cardiac defects, eye abnormalities, sternal cleft, and supra-umbilical raphe) syndrome[33], Williams-Beuren syndrome[34], Alagille syndrome[35], and Noonan syndrome[36]. Cerebral artery aneurysms and various anomalies of the pulmonary and aortic arch vessels have been associated with CoA[37].
Abnormal histology of the arterial wall adjacent and distal to CoA site often observed with medial degeneration noted in pre- and post-stenotic specimens leading to increased incidence of aortic dissection and aneurysm. Hence, CoA is considered a generalized arteriopathy[38]. The ductal tissue with in-folding of aortic media is observed in the tissue ridge, which is often noted extending from the posterior aortic wall and protruding into aortic lumen, on examination of the lesions of the coarctation[39].
The underlying pathogenesis is not completely understood. However, the most commonly accepted theories include hemodynamic, ductal hypothesis, and abnormal genetic mutations. Hemodynamic theory states that reduced anterograde intrauterine blood flow to the fetal arch leads to its underdevelopment[40,41]. Ductal hypothesis postulates the migration of ductal tissue into the wall of the fetal thoracic aorta[42-45]. The NOTCH1 gene, which plays an important role in cardiovascular development, and several other genes have been implicated in the etiology of CoA[38]. Increasing evidence of genetic contribution to CoA has been documented[46,47] with siblings having a 0.5% risk of CoA at birth and a 1% risk of congenital heart disease[48]. In the development of the aorta, vascular endothelial growth factor (VEGF) plays a crucial role, acting as a chemoattractant and stimulating angioblast migration toward the midline[2]. Studies have shown that targeted VEGF disruption leads to significant impairment of aortic formation[49] with increased pre-coarctation collagen and decreased smooth muscle content when compared to the post-coarctation aorta or proximal aorta in young transplant donors[50]. Mechanical models indicate that blood flow abnormalities, defective endothelial cell migration, and excessive deposition of aortic duct tissue at the aortic isthmus can lead to coarctation[51]. Coarctation can also be acquired in inflammatory diseases of aorta, such as Takayasu arteritis[52], and also in severe atherosclerosis[53]. Environmental factors such as chemical exposures, particularly solvents have been suggested to have a possible role in development of CoA and studies noting the geographical variations in CoA also suggest the same[54].
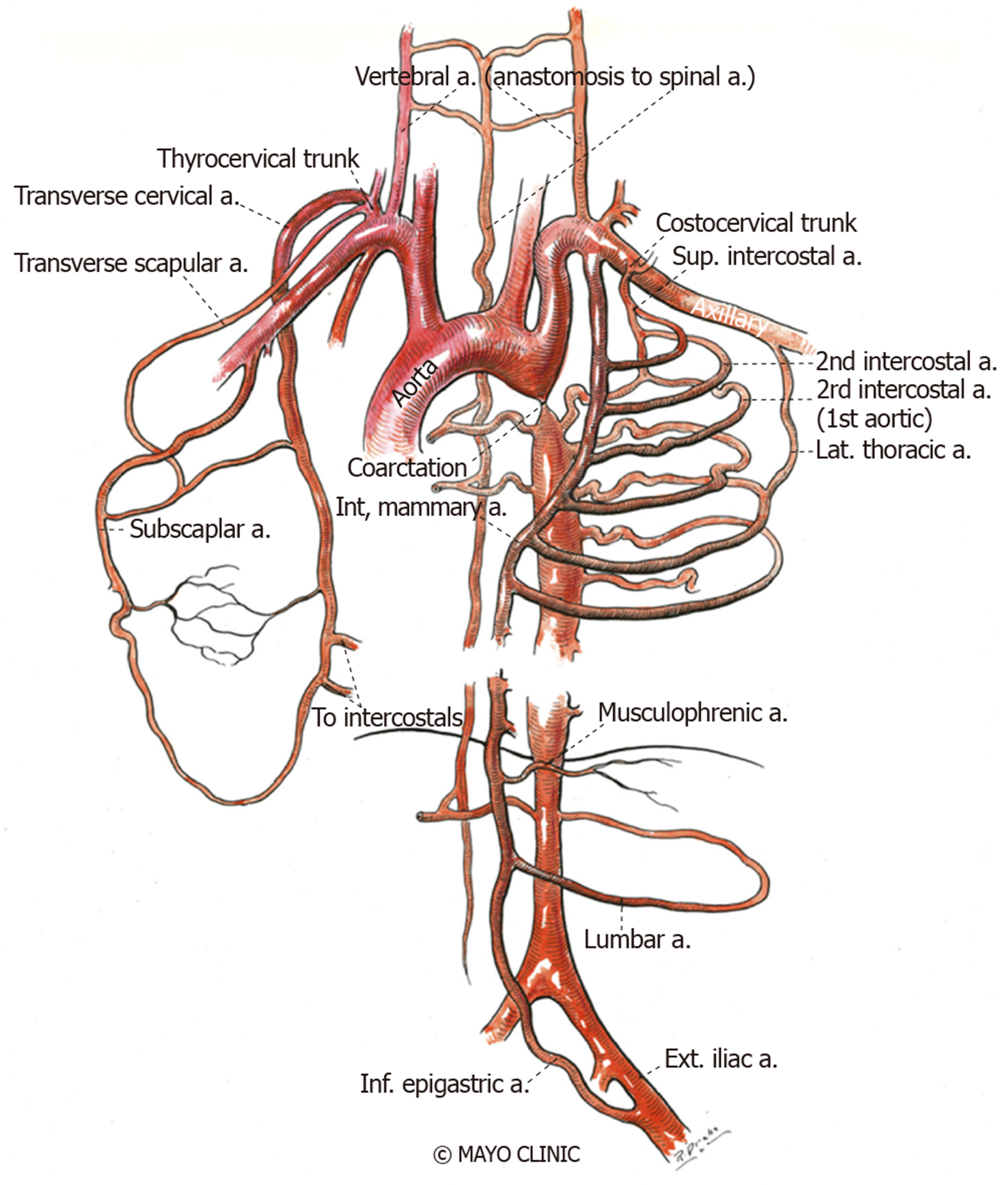
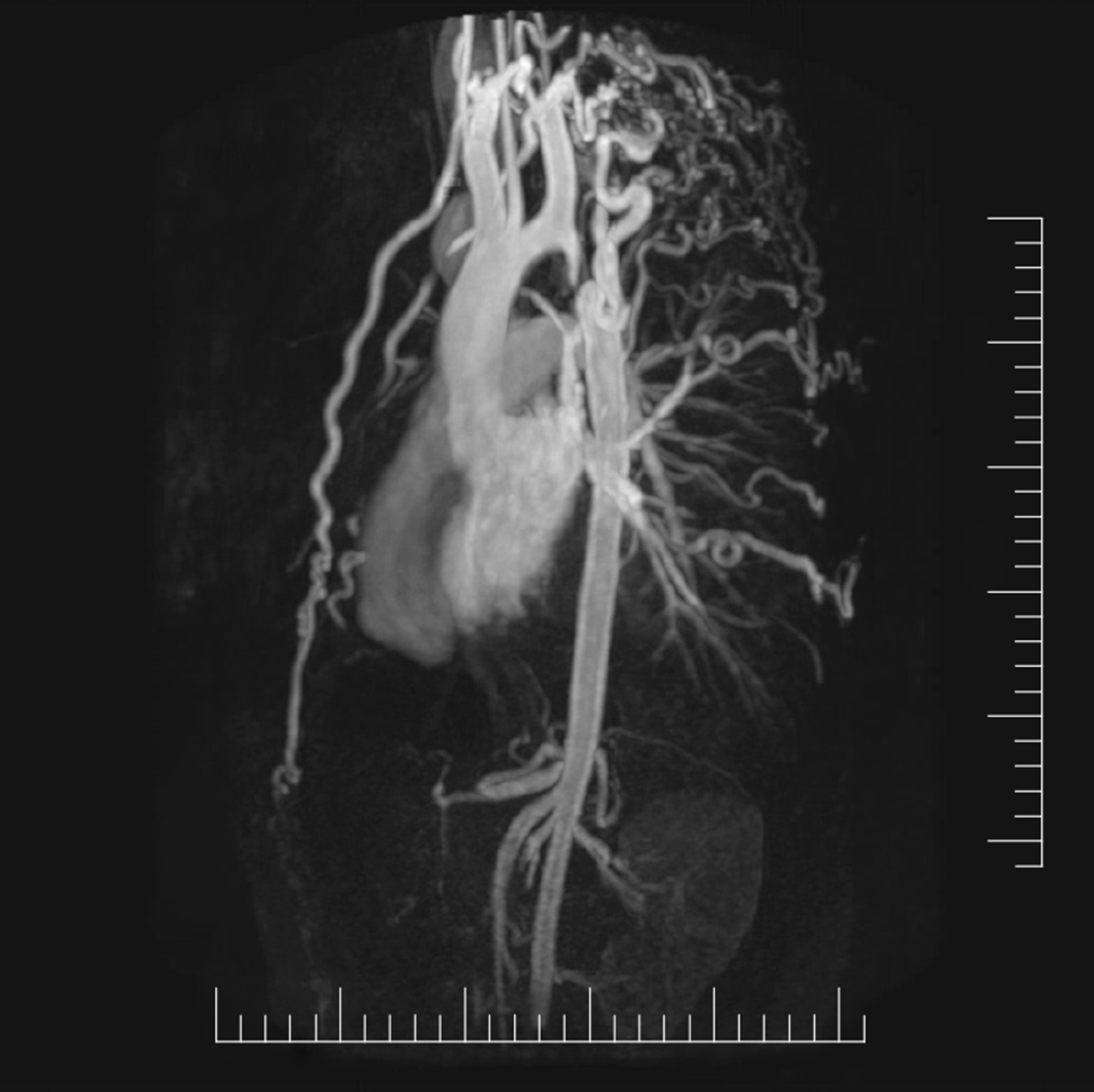
Narrowing of the aorta causes an increase in left ventricular afterload and reduced lower body perfusion, resulting in activation of the renin-angiotensin-aldosterone system and subsequent upper body hypertension. Compensatory mechanisms ensue, including left ventricular hypertrophy, pre- and post- stenotic vessel dilation and development of collateral flow in the intercostal, internal mammary and scapular vessels (Figure 3 and Figure 4). Flow disturbances in the aorta caused by the coarctation can increase the risk of endocarditis, especially when associated with congenital heart disease, such as bicuspid aortic valve[55]. The clinical presentation may vary from a critically ill neonate with heart failure to an asymptomatic hypertensive adult based on the balance between the extent of aortic narrowing and the above compensatory mechanisms[56]. Moreover, there may be an increased risk of aortic aneurysm or dissection and cerebral berry aneurysm development depending on the possible existence of underlying intrinsic aortopathy and hypertension[38].
Natural history is mainly obtained from hospital post-mortem records and from case series prior to the availability of surgical correction first performed in 1948[57]. The prognosis of CoA depends on the hemodynamic severity and is generally poor without intervention. Historical data shows that patients who survived beyond infancy died at a mean age of 34 years with a 75% mortality rate by age 43 years from congestive heart failure, aortic dissection or rupture, endocarditis, endarteritis, intracranial bleed and myocardial infarction[39]. Studies suggest a less than 5% chance of developing hypertension by early adulthood in patients repaired in infancy versus 25%-33% in those operated after the age of one year[58-61].
A substantial number of asymptomatic subjects with aortic coarctation are not detected until adult life, underestimating the incidence at birth[5]. The age of presentation and manifestations depend on the severity of narrowing, relationship with arch vessels, and collateral vessel formation. Therefore, the clinical presentation of coarctation differs significantly in pediatric patients and adults. In the fetus and neonate coarctation may be suspected because of right ventricular enlargement associated with decreased left ventricular flow and greater flow through the ductus arteriosus. Infants may remain asymptomatic when there is associated patent ductus arteriosus. However, after ductal closure, severe CoA results in heart failure and/or shock from acute left ventricular pressure overload. Clinical presentation in children and adolescents is typically through lower extremity fatigue and exertional dyspnea. Presentation in adults is relatively rare, 10.3% of patients with CoA being diagnosed after the age of 40 years[62]. In virtually all cases, CoA presents with upper extremity hypertension and lower blood pressure in the lower extremities with delayed femoral pulses. Pulse and blood pressure reading patterns will vary with the origin of the subclavian arteries relative to CoA site. The classic bilateral upper extremity hypertension occurs when the left subclavian artery is proximal to the coarctation. Occasionally, left subclavian originates distal to the coarctation, resulting in unequal arm blood pressure readings with diminished left brachial pulse and pressure. Rarely, when both the right and left subclavian originate distal to the coarctation as in anomalous origin of the subclavian artery from the descending aorta, pulses and pressures in all four extremities are equally decreased. Despite the blood pressure variability in the extremities, regional blood flow is usually maintained by autoregulatory mechanisms causing vasoconstriction in hypertensive areas and vasodilation in hypotensive areas[63]. Several theories support the etiology of hypertension: Imbalance within the autonomic nervous system[64], impaired vascular function[65], and hyperactivation of the renin-angiotensin system[66,67], which are the most widely accepted. According to the renin-angiotensin system theory, lower body hypoperfusion results in activation of the renin-angiotensin-aldosterone system and consequent upper body hypertension. Symptoms and complications secondary to hypertension include headaches, epistaxis, exercise intolerance, angina, shortness of breath due to left ventricular dysfunction, heart failure and ruptured cerebral artery aneurysms[37,68]. Other potential mechanisms for hypertension in CoA include endothelial dysfunction, reduced arterial compliance, and blunted baroreceptor function[69].
Patients can also present with colder and fatigued lower extremities, claudication, and abdominal angina. Occasionally, an initial presentation of CoA may include dissection and/or aneurysm/pseudoanuerysm of the aorta and branch vessels, including the spinal and intercostal arteries[70,71]. Complications such as premature coronary atherosclerosis, cerebrovascular events, left ventricular systolic dysfunction, and endocarditis can occur. Timely recognition of CoA and risk factor modification and treatment improves survival but life expectancy remains lower compared to normal individuals Analysis of 80 adolescents who underwent surgical repair of CoA in childhood did not find intracranial aneurysms, implying the possibility of aneurysms developing with age and preferentially in the presence of hypertension[72]. Late hypertension may be associated with residual or recurrent obstruction[2]. Hence, ambulatory blood pressure monitoring use can help in early diagnosis of late hypertension[73]. Blood pressure is usually 10%-20% higher in the lower extremities due to wave amplification. An upper to lower extremity pressure gradient of 10 mmHg should raise suspicion of coarctation, and a gradient of 35 mmHg or greater is considered highly specific for CoA[74]. Occasionally, claudication can be noted due to the ischemia of the lower limb. Cardiac auscultation may demonstrate a harsh systolic murmur in the left sternal border with radiation to the inter-scapular region in the back. In the suprasternal notch, an associated thrill may also be palpable, and occasionally, a left ventricular lift can be observed of there is left ventricular pressure or volume overload. The development of arterial collaterals is suggested by continuous murmur, especially in patients with long-standing unrepaired coarctation and may diminish the pressure gradient.
Initial workup includes blood pressure measurements in all four extremities. Clinical diagnosis is usually based upon the characteristic findings on physical examination such as delayed or diminished femoral pulses, systolic hypertension in the upper extremities, and low or undetectable blood pressure in lower extremities. Confirmation is usually done by imaging techniques; echocardiography being the most widely used initially but three-dimensional techniques like contrast enhanced computed tomography (CT) or cardiovascular magnetic resonance imaging (MRI) are the most cost-effective.
Patients with CoA may have a normal Electrocardiogram (EKG) or show the evidence of left ventricular hypertrophy and dilatation from long-standing left ventricular pressure overload. Ischemic changes may also be found infrequently[38].
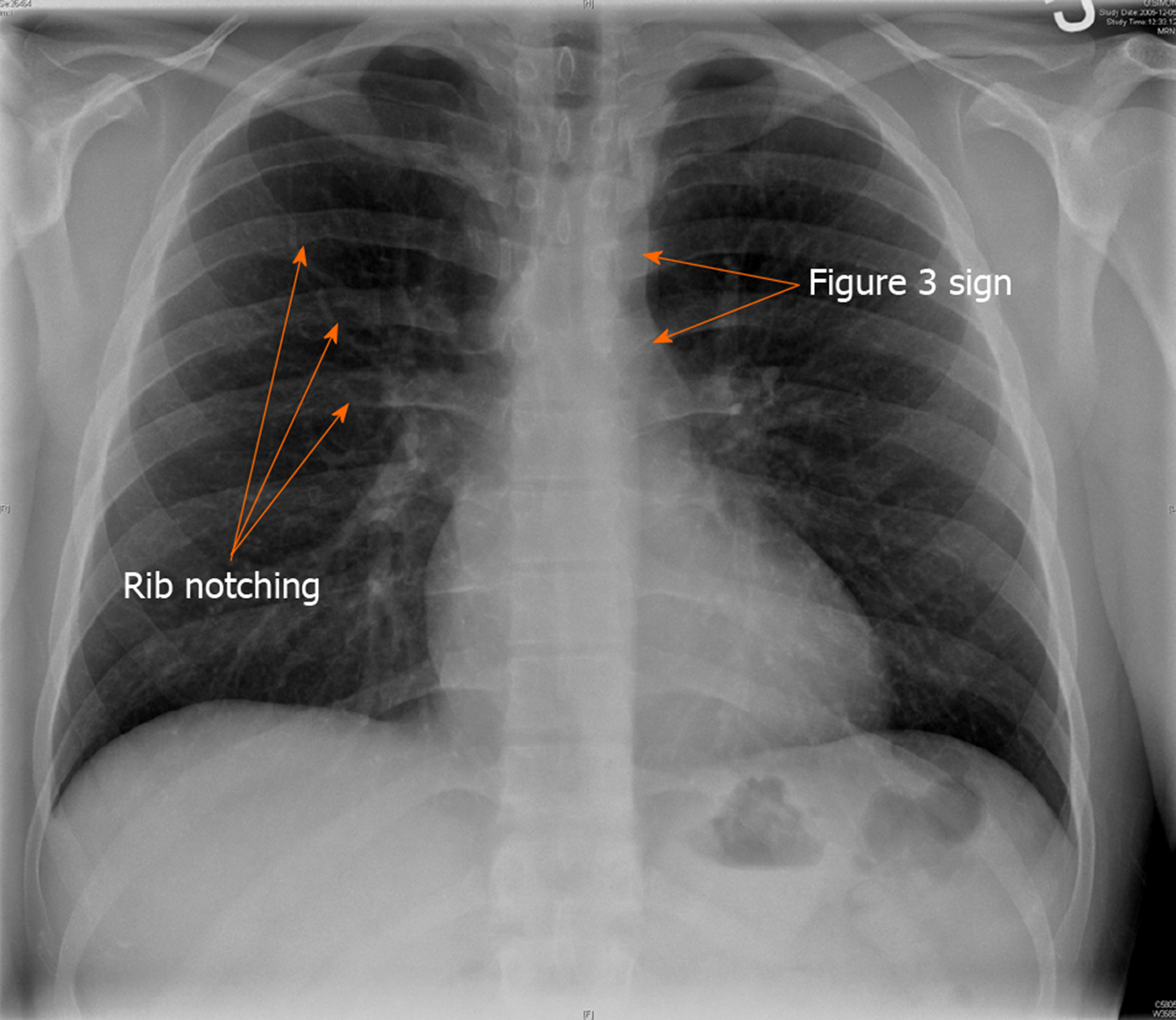
Chest radiograph may show a normal cardiac contour or can be mildly enlarged. A characteristic finding of “Figure of 3” beneath the aortic notch suggests the narrowing of the descending aorta at the level of coarctation and dilatation pre and post coarctation (Figure 5). Bilateral inferior rib notching may also be seen in the third to eighth ribs suggesting the presence of dilated intercostal collateral arteries[68].
Transthoracic echocardiography (TTE) is the imaging modality most often used in the assessment of cardiac disease but has limitations in evaluating extra cardiac structures and collateral circulation. TTE can help confirm suspected CoA, assessing pressure gradient severity and providing diagnosis of other associated cardiac and valvular abnormalities, most commonly, left-sided obstructive lesions (subvalvular and supravalvular aortic stenosis parachute mitral valve and cor-triatriatum) but especially ruling out the presence of a bicuspid aortic valve.
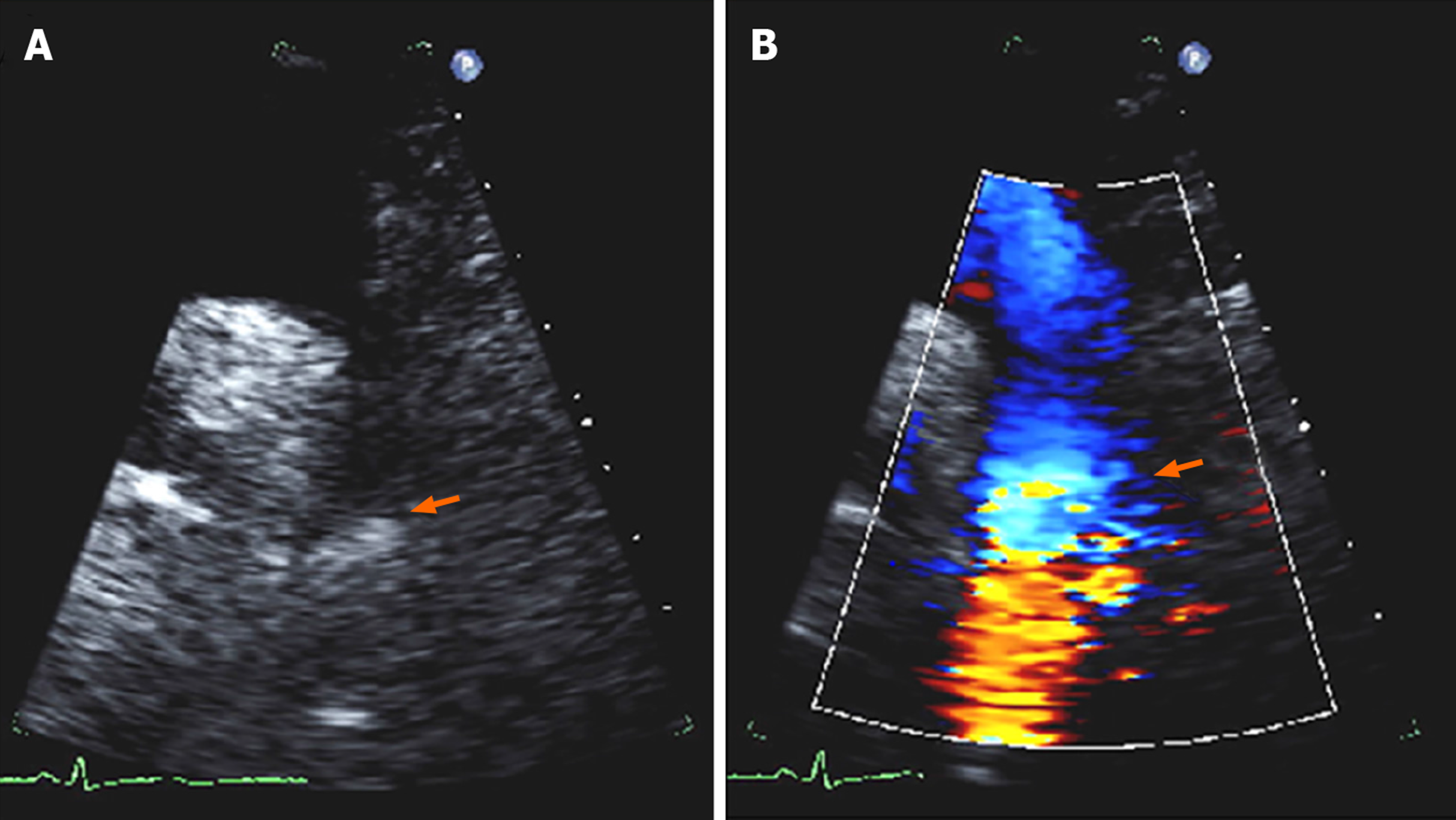
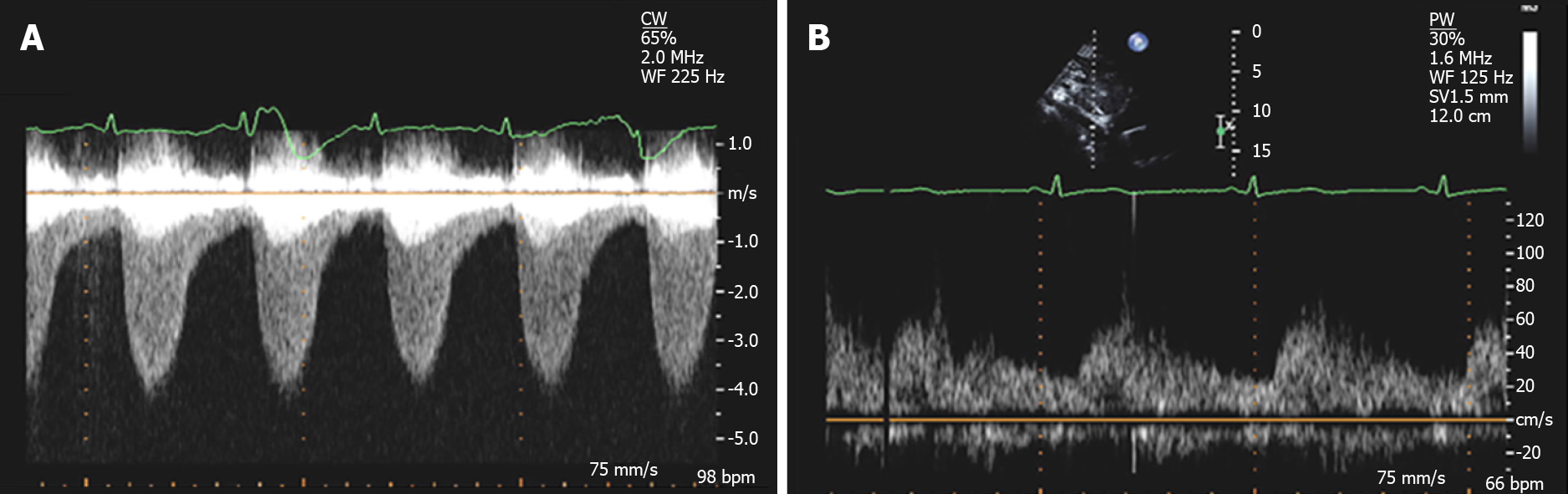
The presence of any associated aortopathy can be evaluated by following the serial measurements of aortic root and ascending dimensions. Long axis (candy cane) view demonstrates a focal area of narrowing of the proximal descending thoracic aorta distal to the origin of subclavian artery and color flow Doppler demonstrates associated flow turbulence (Figure 6). The severity of CoA can be estimated using continuous-wave Doppler, by calculating the pressure gradient across the narrowed area, although appropriate correction for velocity proximal to the site of coarctation is important to avoid overestimation of the true gradient[1]. The mean systolic gradient often provides an excellent estimate of coarctation gradient. The severity of CoA can also be estimated by calculating the ratio of the maximal velocity across the coarctation in the suprasternal view to the peak velocity in the abdominal aorta in the subcostal view[75]. If transverse arch hypoplasia is present, the proximal velocity increases as well; therefore, the systolic pressure gradient should be calculated with the Bernoulli equation[76]. Diastolic flow persistence with velocities over 1 m/s is a characteristic finding in CoA (Figure 7). Sometimes, in the presence of collateral blood flow, pressure gradients across the coarctation, may be less severe than expected[77]. Severe obstruction, eccentric gradient, or long, tortuous vessels may also affect the Doppler gradient by potentially under or overestimating the degree of severity and echo derived gradient.
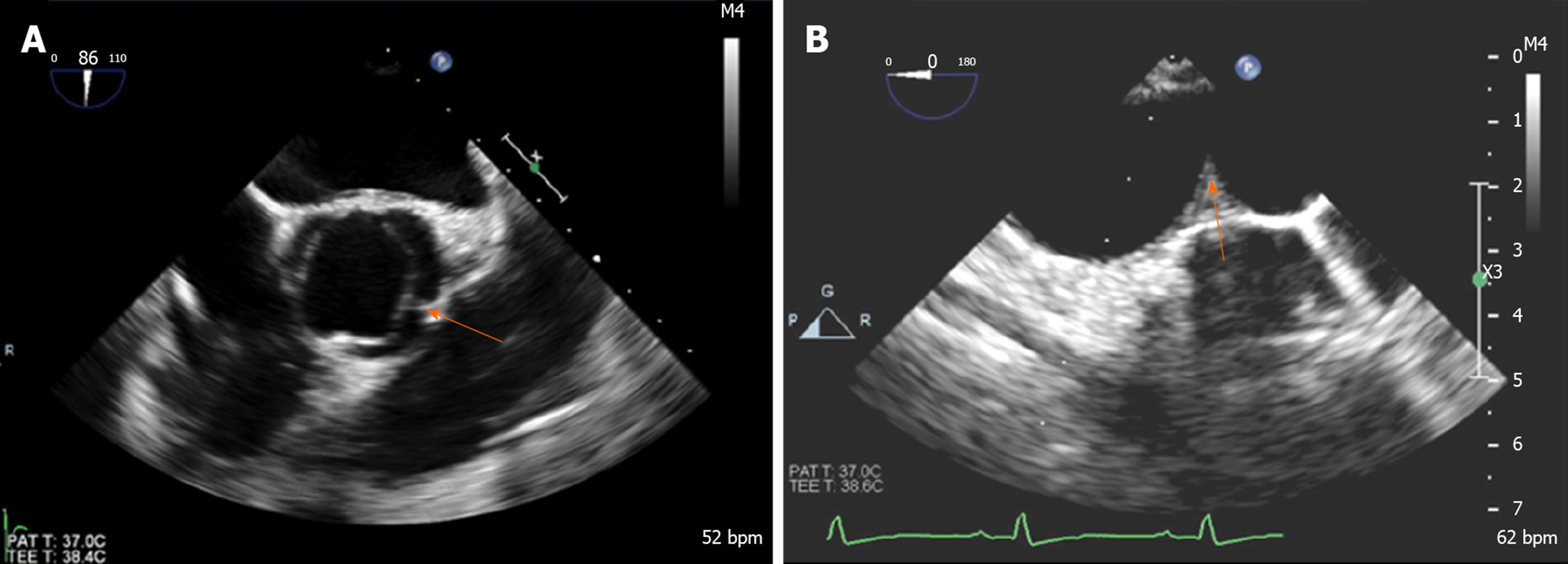
Subcostal views are used to assess the distal thoracic and upper abdominal aorta. In healthy individuals without obstruction, pulse wave Doppler in the abdominal aorta shows a rapid systolic upstroke, short deceleration time, followed by a brief early diastolic flow reversal and little anterograde flow throughout diastole. The presence of coarctation causes a delay (> 50 ms from the EKG R wave) and reduction in systolic upstroke velocity (< 55 cm/s) with anterograde diastolic flow persistence and loss of early diastolic flow reversal[2,78] (Figure 7). Prolonged diastolic flow and reduced peak systolic velocity are sensitive indicators of aortic obstruction. Transesophageal echocardiography (Figure 8) can provide accurate imaging of the descending aorta, but has limited acoustic windows in the arch because trachea-bronchial shadowing and is not widely used in the diagnosis of CoA.
Cardiac magnetic resonance imaging (CMR) demonstrates superior visualization of the thoracic aorta when compared to echocardiography and can clearly define the location and severity of CoA, collaterals, branching patterns, and CoA pressure gradients[75,79,80]. Gadolinium-enhanced magnetic resonance angiography, provides excellent flow-independent resolution of vascular structures and is helpful in providing anatomic information in adults prior to surgical or catheter-based intervention, and is also recommended for follow-up imaging[81]. CMR is generally preferred over cardiovascular CT for serial imaging because it lacks ionizing radiation[82]. However, CMR is more susceptible to metallic artifacts than CT, leading to difficulties in the assessment of vessel lumen patency, identifying re-coarctation, aneurysm, or fracture stented patients[68].
Phase-contrast flow imaging is useful to quantify the flow volumes and velocity[83] and to determine the degree of collateral flow. Collateral flow joining the descending aorta is identified by the increase in flow by 30% or more from proximal to distal aorta[84]. Four-dimensional flow MRI is an emergent tool in evaluating the hemodynamic significance of collateral blood flow[85]. CMR should be routinely applied in adult patients who previously had surgical or interventional treatment of coarctation in childhood.
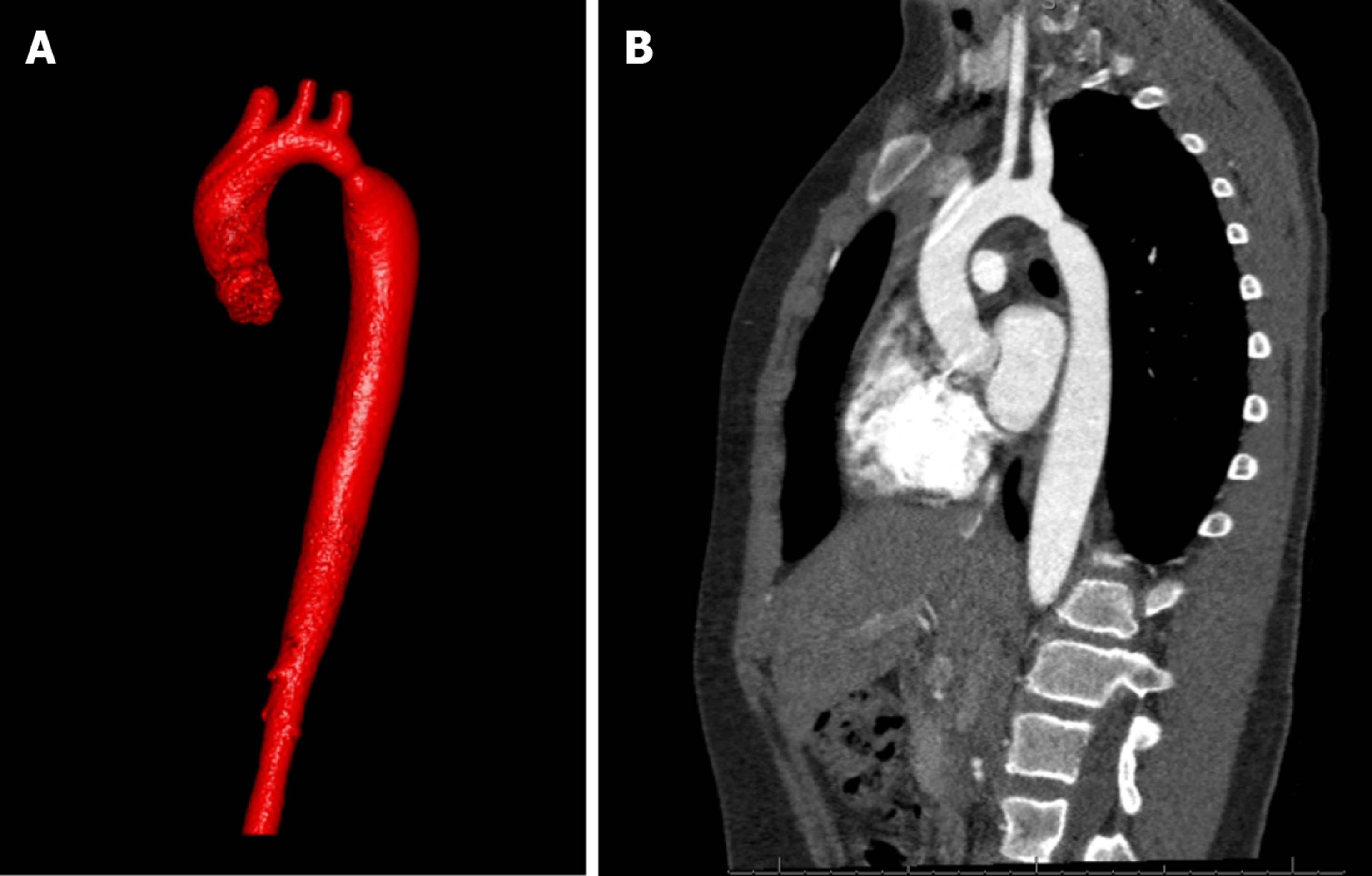
Computed tomographic angiography (CTA) also provides invaluable information in the diagnosis and management of patients with CoA. CTA has better spatial resolution, shorter acquisition times, causes less claustrophobia than CMR and allows for the presence of metallic implants as well as previous stent implantation without a signal noise artifact[68]. Similar to CMR, it can be performed to visualize the CoA segment, any aneurysms distal to CoA, hypoplasia of aortic arch, re-coarctation post repair, to follow serial aortic dimensions, and to demonstrate associated vascular anomalies (Figure 9 and Figure 10). However, it requires ionizing radiation and contrast, and cannot provide hemodynamic information such as peak pressure gradient and the degree of collateral circulation[38]. However, dose saving algorithms reduce the radiation exposure.
Cardiac catheterization and angiography (CA) is the gold standard in assessing the pressure gradients across the CoA and provides high-resolution images of the aorta[38]. Coarctation is classically defined as a catheterization-measured peak systolic gradient > 20 mmHg. Hemodynamic assessment should include coarctation gradient assessment, right and left heart catheterization including assessment of left ventricular end diastolic pressure. Aortic angiography is obtained to determine the site, severity of obstruction and associated vascular abnormalities[86].
Indications for intervention (surgical or transcatheter intervention) in coarctation of aorta are: (1) Peak to peak coarctation gradient ≥ 20 mmHg: This gradient can be measured as a difference between systolic blood pressures from the upper and lower extremities, from catheterization data in which peak pressure distal to the coarctation is subtracted from peak pressure proximal to the coarctation, or with echocardiography. However, the resting gradient may be lower in significant left ventricular systolic dysfunction due to the low forward stroke volume[81]; (2) Radiographic evidence of significant collateral flow; (3) Coarctation-attributed systemic hypertension; (4) Coarctation-attributed heart failure; and (5) Exercise limitations from limited lower extremity blood flow (i.e., claudication).
Various surgical techniques have been utilized for CoA repair.
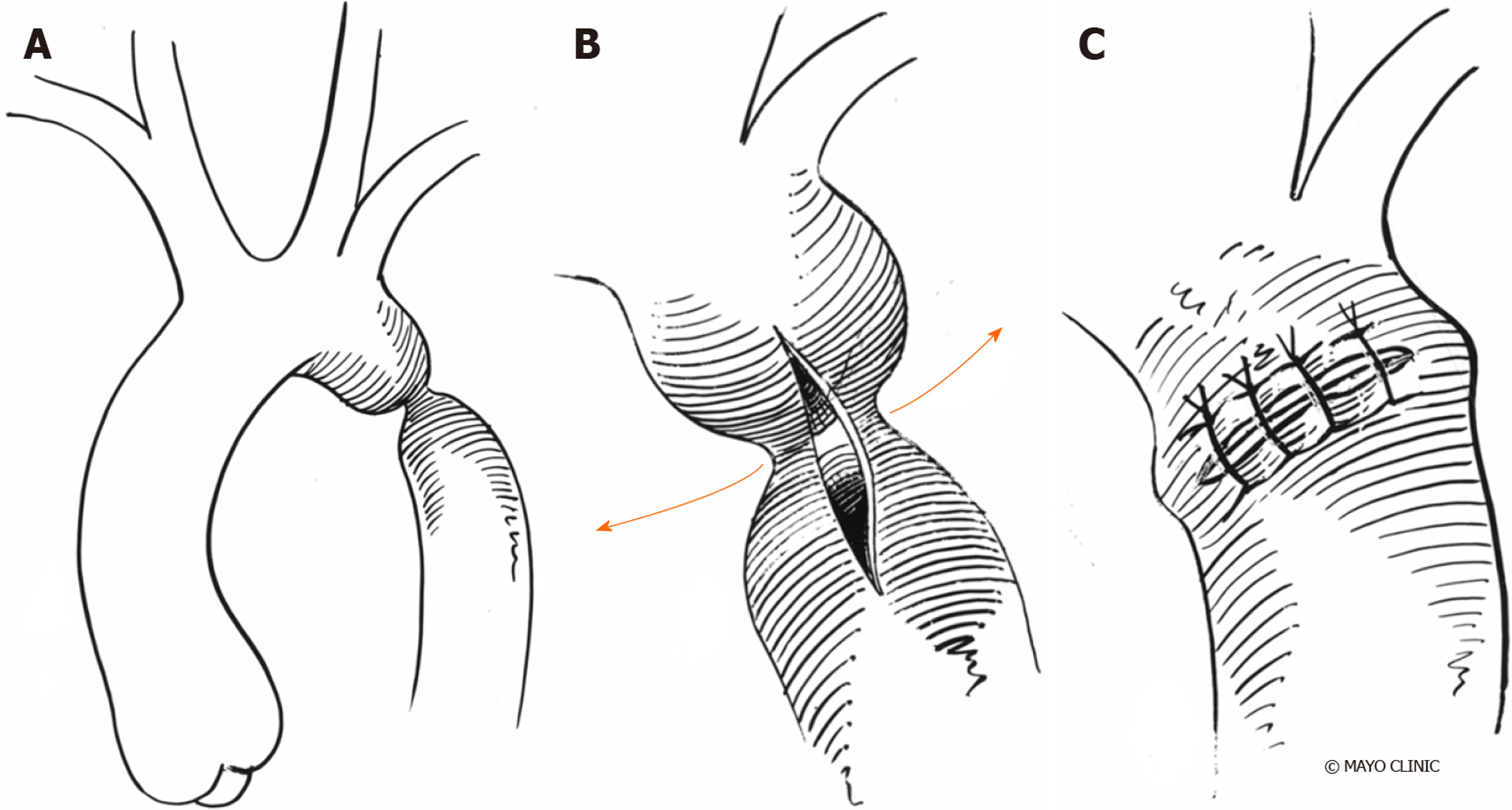
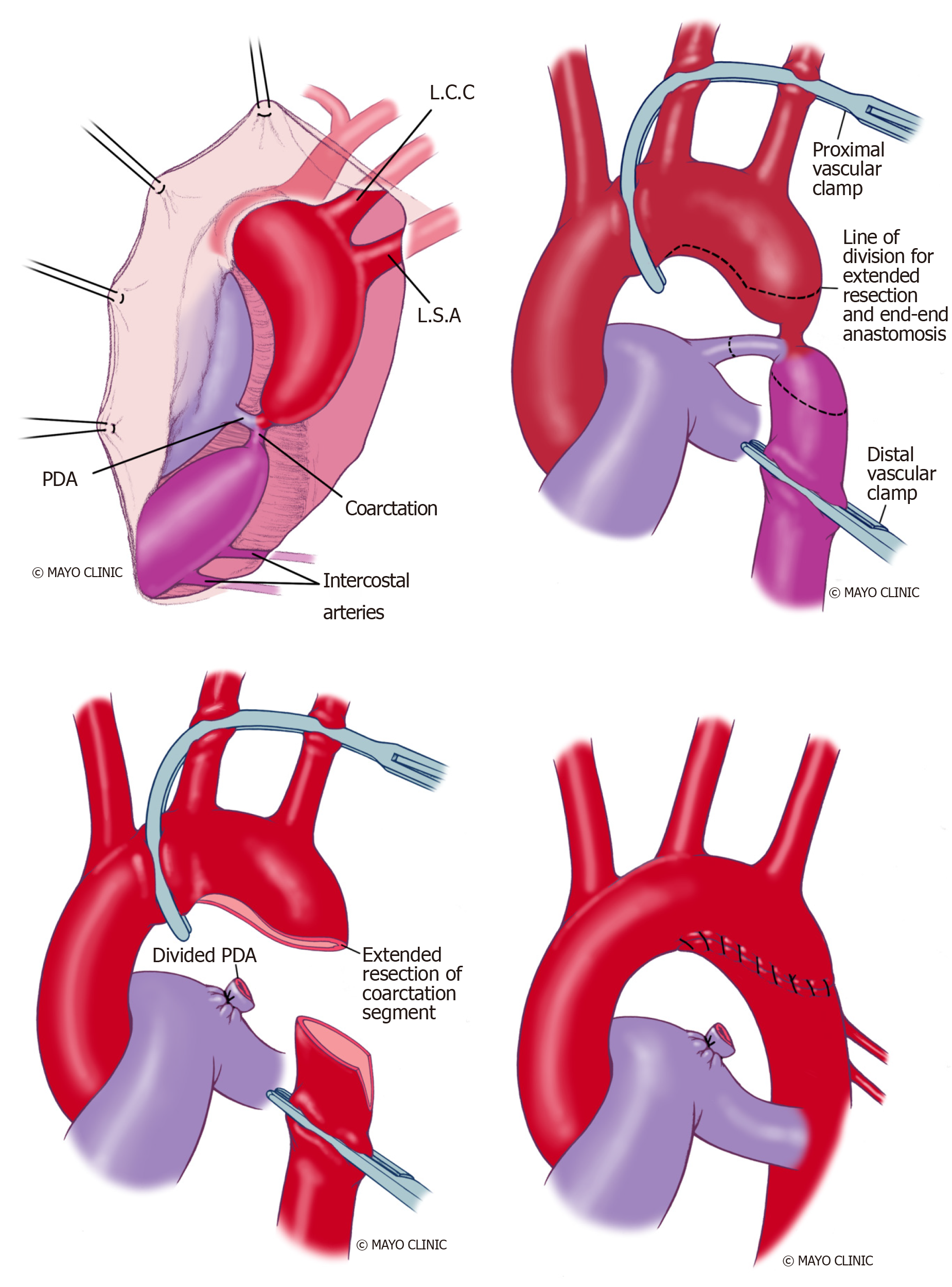
Resection with end-to-end anastomosis: Crafoord[87] and Gross[88] described the first successful surgical intervention via a left lateral thoracotomy in 1945. The intervention involves resection of the coarcted segment followed by direct suture anastomosis of the transected ends (Figure 11). The incidence of re-coarctation was comparatively high following the initial repair, with the incidence rates of 41% to 51%[89-92] and highest in neonates.
Prosthetic patch aortoplasty: Vosschulte first described the usage of a prosthetic patch to augment the aorta (Figure 12)[93]. Dacron grafts showed to have a lower rate of re-coarctation[94], but had a high incidence of aortic aneurysm formation (20%-40%) and are no longer performed[95] despite patch material evolution from Dacron to polytetrafluoroethylene (PTFE), which, reduced aneurysm formation to 7%, but increased the rate of re-coarctation to 25%[96].
Subclavian flap aortoplasty: Waldhausen et al[97] first described the technique in 1966. A flap is generated from the subclavian artery, which is ligated close to the left vertebral artery origin by an incision extending down onto aortic isthmus and across the coarcted segment. Re-coarctation rates are close to 3% in older children, but up to 23% in neonates[98-100]. Sacrificing the subclavian artery might cause claudication in the left arm in the long term, although it usually does not result in left arm ischemia[99].
Extended end-to-end anastomosis: Amato et al[101] first described the technique in 1977 and is currently frequently applied. The aortic arch is clamped proximally, including the subclavian artery and distally below the coarcted segment (Figure 13). An end-end anastomosis of the aortic arch, which has been opened on its inferior aspect and the descending aorta, is performed after the division of ductus arteriosus. This technique showed lower re-coarctation rates of 4%-13% and low perioperative mortality[102].
Interposition graft: The resection and graft interposition were first described by Gross in 1951[88,103,104] and is the preferred approach in adults. A tube graft of either aortic homograft or Dacron is sewn into the aorta, after the cross-clamping of the aorta and resection of the coarcted segment. It is useful in patients with long-segment CoA; however, it requires longer cross-clamp time and does not grow with the patient thus being unsuitable for pediatric patients[2]. A surgical follow-up study described a high prevalence of dilatation of interposition grafts to 150% of the original diameter, with most dilatation occurring in the first year after the procedure[105]. Dilatation was most pronounced in knotted (Gelseal and Gelsoft; Vaskutek Ltd., Inchinnan, Renfrewshire, Scotland) grafts compared with woven (Gelweave; Vascutek Ltd., Renfrewshire, United Kingdom) grafts[105]. Hence, routine serial arch imaging is recommended for patients after surgical repair. Another study reported no peri-operative mortality or re-coarctation events during a mean follow-up of 10 years ± 7.6 years[106].
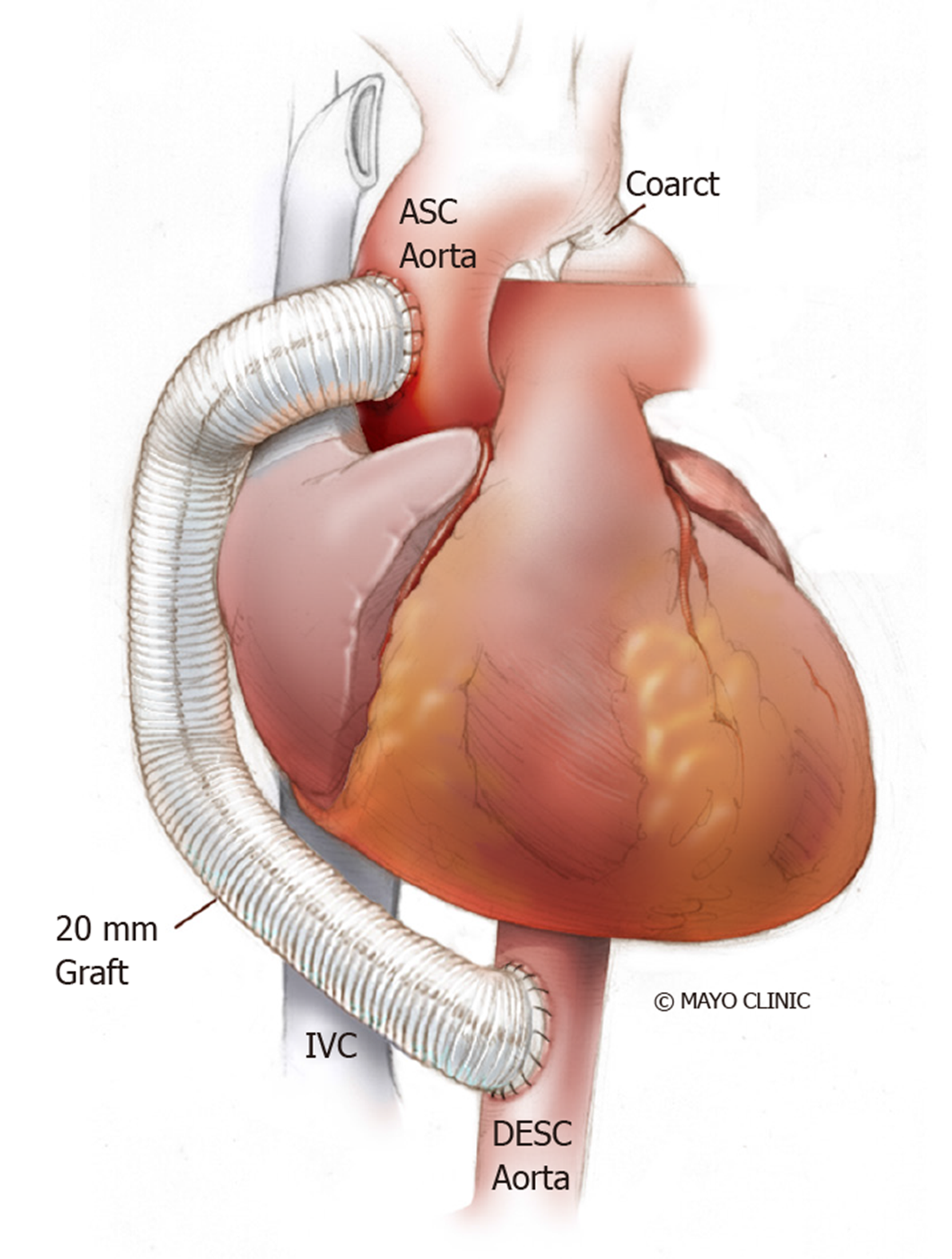
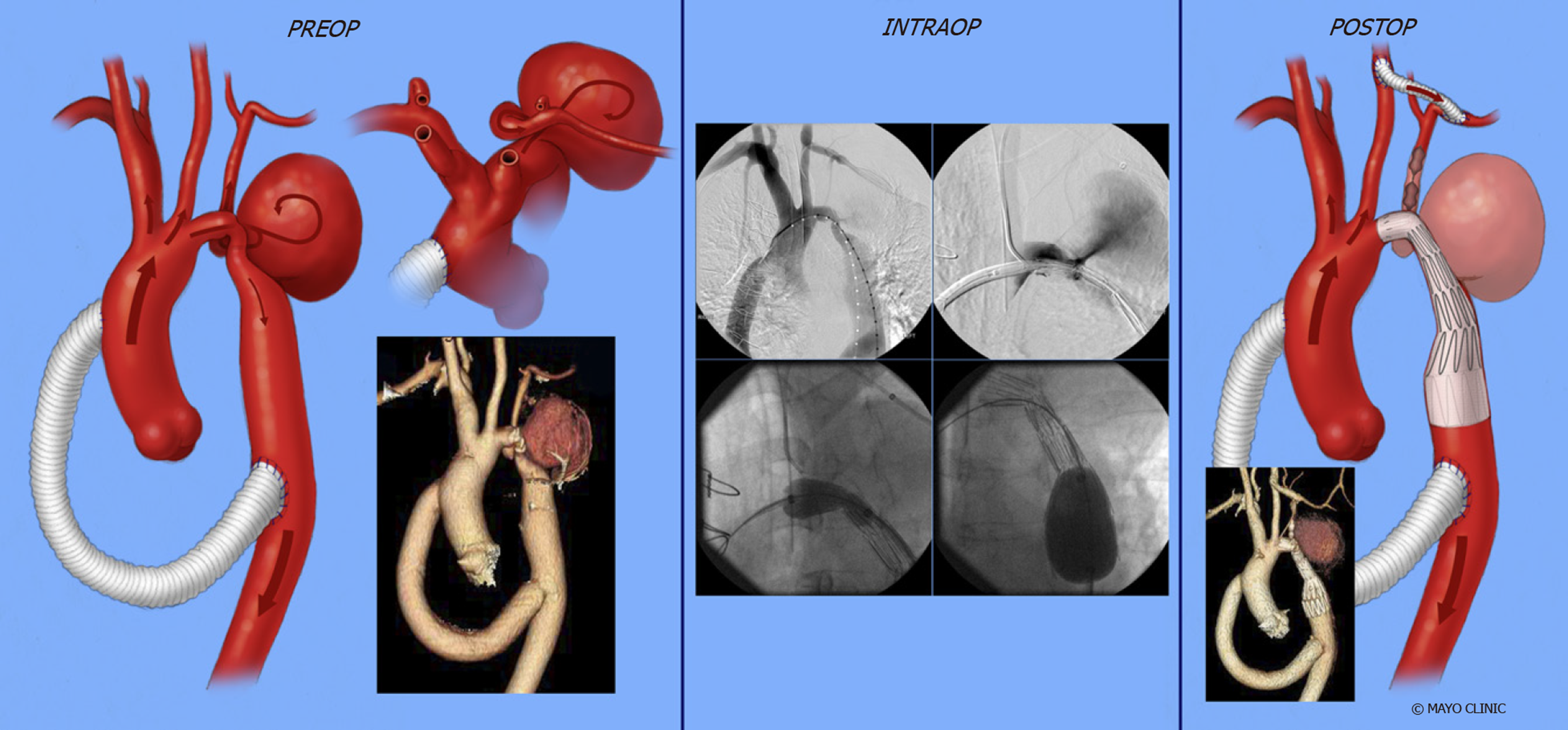
Extra-anatomical correction: This procedure is possible when concomitant cardiac procedures such as coronary artery bypass grafting or aortic valve replacement need to be addressed[107]. It is performed in adults through median sternotomy with cardiopulmonary bypass support and provides additional blood flow to the distal aorta leaving the stenosed aorta in situ[108]. A prosthetic conduit is anastomosed proximally to the ascending aorta or distally to the subclavian artery[109], bypassing the coarcted segment. A study of 80 patients who underwent ascending-to-descending aortic bypass (Figure 14) followed for 7 years ± 6 years showed improvement in blood pressure, no early deaths and no paraplegia or stroke. Late deaths occurred in 6%, while 4% required re-intervention (for peri-prosthetic regurgitation and mitral valve replacement)[110]. Some have noted anastomotic stenoses and pseudoanuerysm formation.
When considering surgery, the prognostic benefit and age-related risks for procedural complications need to be discussed. A study assessing re-coarctation 20 years ± 7 years after surgical repair in infancy showed moderate-to-severe re-coarctation in 34% of patients highlighting that re-coarctation is common in follow-up patients after surgical repair of CoA[111]. However, surgery might still be the preferred option in the setting of significant arch hypoplasia or long coarctation segments[112].
Transcatheter procedures play an important and evolving role in the repair of native CoA and the treatment of re-coarctation or aneurysm formation after the initial repair.
Balloon angioplasty: Transcatheter balloon angioplasty was first described by Singer et al[113] in 1982, and became widely used over the following two decades. A balloon catheter is advanced up to the obstructed segment and inflated to induce intimal tear and limited medial tear in the arterial wall. There remains a potential risk for later aneurysm formation with this approach, especially when treating a native CoA[38]. Short-term results were satisfactory in infants and children, but long-term outcomes less favorable, with re-coarctation rates of up to 80%[114-117].
A follow-up study of 99 patients who underwent balloon angioplasty for re-coarctation showed that re-intervention was required in 28 patients[118]. Comparison of balloon angioplasty with surgery in 80 patients aged 12 years ± 10 years, showed increased aortic complications during follow-up, with 26% requiring re-intervention for re-coarctation and 21% for aneurysm formation[119]. Hence, this approach may be reasonable to stabilize critically ill infants as a bridge to surgery and an alternative for mild coarctation in adolescents. A recent meta-analysis showed that balloon dilation is less likely to achieve treatment success as measured by the proportion of patients achieving a blood pressure gradient ≤ 20 mmHg when compared to stenting. Odds ratio for re-coarctation in patients undergoing balloon dilation versus stenting was 7.01 and 3.340 for aortic wall injuries[120].
Stent implantation: Stent implantation has become the mainstay of interventional treatment for CoA, with a reduction in clinically significant residual gradient and aortic wall abnormalities when compared with balloon angioplasty alone[121]. It improves luminal diameter, results in minimal residual peak pressure gradient and sustained hemodynamic benefit. Additionally, it prevents vascular recoil resulting in re-coarctation and can tack intimal flaps to the aortic wall allowing the wall to heal, reducing the risks of dissection and aneurysm[38]. Balloon-expandable intravascular stents have emerged as the preferred treatment option for patients with native CoA and re-coarctation (Figure 15)[122].
Stent placement is considered a reasonable treatment option in patients weighing more than 30 kg, as children weighing less than 30 kg may need repeated interventions for stent expansion in the face of growth with the potential injury risk to the femoral arteries during vascular access for stent delivery[123,124]. Studies support the safety and efficacy of stent placement for both native and recurrent coarctation[125-127]. Stent placement was successful in 99% in the Coarctation of the Aorta Stent Trial (COAST) I trial in which 105 children and adults underwent stent implantation for the treatment of native CoA and re-coarctation[127]. Nine patients required re-intervention for aneurysms during the first two years, and 10 patients required re-intervention after two years. Another study of 578 CoA patients who underwent stent placement showed a 98% implantation success rate, which was defined as a reduction in the pressure gradient to < 20 mmHg or a ratio of post-stent coarctation to descending aorta of > 0.8[125,128].
Stents can be classified based on the design as closed-cell, open-cell, and hybrid design, or as bare metal or covered. Cheatham-platinum (CP) stent, Palmaz series, Genesis XD and Intra Stent are some of the stents commonly used. Rarely, formula or Vallejo stents are used in small children as a bridge to anticipated surgery. In closed-cell design, all internal inflection points of the structural components are connected, whereas they are not in open-cell design. Open-cells have an advantage of longitudinal flexibility of the cells due to the absence of connections[38]. Stents can be made of different metals such as stainless steel, platinum-iridium alloy, and chromium-cobalt alloy. Covered stents usually have a polytetrafluoroethylene sleeve that can exclude aneurysms from the circulation and may prevent endoleaks within the sleeved section of the stent. Deployment of a second covered stent may be of utility in the acute management of aortic rupture at the time of stenting[129]. Premounted covered stents are potentially very valuable for rapid deployment in the event of significant aortic wall injury. Some studies have suggested routine use of covered stents in face of more severe degree of coarctation and in older patients.
Previous studies suggested that bare metal stents might have a higher risk of aneurysm development[95,130,131] but a more recent study compared did not observe this difference[132]. Serial re-dilation of purposely under-deployed, covered stents is a strategy considered in growing children and when there is a concern of aortic rupture by deployment to the size of the normal aorta[133,134]. Table 1 summarizes the major studies that used covered stents in the treatment of CoA.
| Ref. | Patients (n) | Age of patients (yr) | Native coarctation (n) | Aneurysm (n) | Presenting gradient (mmHg) | Post stenting gradient (mmHg) | Aortic complications | Re-coarctation/ Serial re-dilatation |
| Tzifa et al[190] | 30 | 28 ± 18 | 14 | 8 | 36 ± 20 | 4 ± 4 | Dissection, spontaneously resolved (n = 1) | 4 (serial dilatation) |
| Butera et al[191] | 33 | 6-66 | 20 | 2 | 20-75, median 39 | 0-12, median 0 | - | 1 |
| Bruckheimer et al[133] | 22 | 8-39 | 22 | 0 | 29 ± 9 | 7 ± 7 | Aortic tear treated with second covered stent (n = 1) | 9 (serial dilatation) |
| Kenny et al[192] | 37 | 9-65 | 13 | 7 | 10-60, median 26 | 0-20, median 4 | Aortic rupture, surgery required (n = 1) | - |
| Tanous et al[129] | 22 | 39 ± 14 | 14 | 6 | 29 ± 17 | 3 ± 5 | Aortic tear treated with second covered stent (n = 1) | 3 |
Re-coarctation: Factors predisposing to re-coarctation include: (1) Patient age < 30 days[135]; (2) Isthmic hypoplasia[136]; and (3) Coarctation segment diameter of 3.5 mm or less and 6 mm or less prior to angioplasty and post-intervention respectively[135]. Persistent post-procedural hypertension, post-procedure peak pressure gradient of ≥ 20 mmHg, or collaterals usually warrant re-intervention[137-139].
Aneurysm: Aneurysm development is dependent on abnormal aortic tissue at the site of CoA. The pathological aorta consists of a medial layer with fragmented elastic fibers, fewer smooth muscles, and a higher amount of ground substance, unlike a pseudoanuerysm which, forms between the tunica media and tunica adventitia[140]. Aneurysm can arise proximal or distal to the coarctation and occasionally from the origin of branch vessels such as the left subclavian artery (Figure 16). Incomplete removal of the pathological tissue during surgical repair[141,142] or balloon angioplasty also favors the development of an aneurysm. The distal aortic arch aneurysm formation is highest after subclavian flap and lowest with end-to-end anastomosis[143]. Balloon angioplasty is reported to have a higher incidence of aneurysm formation compared to surgery by Shaddy et al[144], while other studies reported a lower incidence at catheterization after balloon angioplasty[145,146]. Stents were shown to have a lower likelihood of aneurysm formation (as low as in 5% of patients) due to reducing trauma to the vessel wall through the dispersion of force[147-149].
Late systemic hypertension: Hypertension is frequently found and a great concern post CoA repair. Age at repair is perhaps the most important predictor[59,150,151]. O’Sullivan et al[187] showed a prevalence of hypertension of 30% in patients repaired at a mean age of 0.2 years, at a mean follow-up of 12 years after repair. An abnormal structure and function of the pre-coarctation arterial conduits is the pathological mechanism thought to be involved. Arterial wall compliance is diminished, and rigidity is increased due to more collagen and less smooth muscle mass[50,152-155]. Other factors contributing to hypertension in post-coarctectomy patients are reduced baroreceptor sensitivity and residual aortic arch gradients[156-158]. Hence, patients with persistent hypertension after the repair may need long-term anti-hypertensive treatment.
Surgical treatment for re-intervention: Surgical repair is mandatory when there is a persistent endoleak or prosthetic infection after stent graft placement. The mortality rate in surgical repair for re-intervention is higher than the native CoA repair and ranging from 1%-3%, to 5%-10% in patients with significant comorbidities, although guidelines recommend an endovascular approach initially for re-coarctation[159].
Perioperative complications are often aggravated by dense adhesions including revision for bleeding (2%), pulmonary-associated complications such as pneumothorax and pleural effusion (8%), and trauma of the thoracic duct or the esophagus (0%-4%)[160,161]. Other complications are lower body ischemia and spinal cord trauma with subsequent paralysis.
Endovascular treatment for re-intervention: Guidelines recommend an inter-ventional approach as the initial treatment strategy for re-coarctation[138,139,162,163]. Balloon angioplasty is the preferred treatment option for recurrent coarctation after previous surgical repair, although it has a high rate of re-coarctation after native CoA repair[159]. The scar tissue has a lower tendency for vascular remodeling and aortic wall injury occurring in only 1%-2% of cases[118,164]. Studies have shown good outcomes with balloon angioplasty of re-coarctation[164-171], with a high procedural success rate and lower complication rate when compared to surgical repair[145,172]. The procedural success rate ranges between 80%-100%[118,125,126,173], and a simple re-dilation often leads to satisfying results when re-coarctation occurs[174].
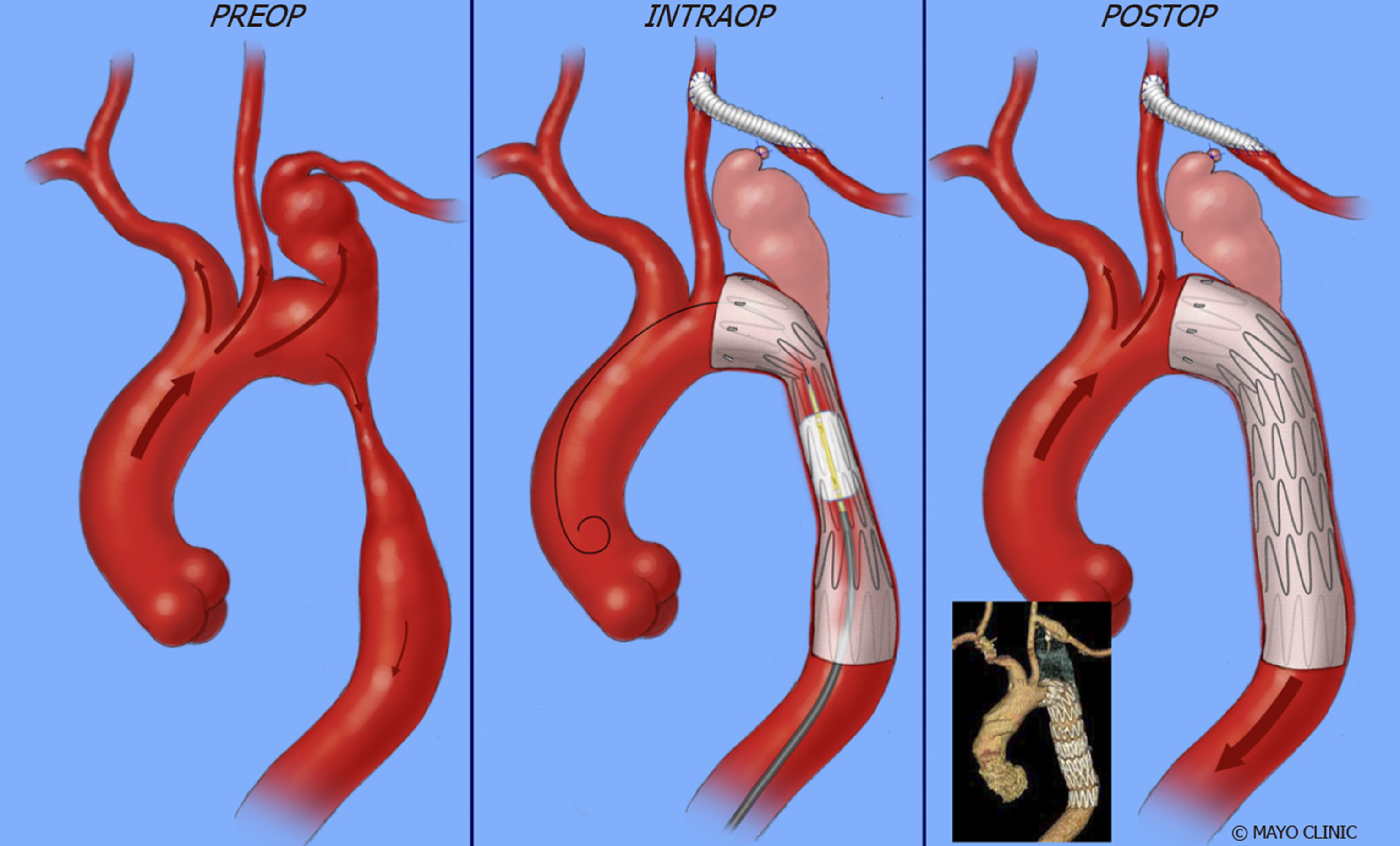
First described in 1991, stent graft placement is an alternative approach to balloon angioplasty[175] and has been used in treating re-coarctation. The procedural success rates ranges between 94%-97%[149,176]. Figure 17 demonstrates exclusion of aneurysm of proximal descending thoracic aorta by deploying a covered stent across the aneurysm.
A number of studies have described the outcomes of interventions in CoA patients. The Congenital Cardiovascular Interventional Study Consortium trial and other studies[125,177-180] showed a successful outcome following stent implantation in children and adults, with a 98% success rate of reducing peak pressure gradient to less than 20 mmHg. The COAST[127] trial, which assessed the safety and efficacy of CP stents in children and adults with native or re-coarctation showed no deaths, serious adverse events or need for surgical interventional during the two-year follow-up. Complication rates were low, including aneurysms (5.7%) and stent fracture (11%). Many other studies support a low rate of complications and mortality[177,181]. The COAST II trial studied 158 patients with native and re-coarctation, showing a success rate of 92% with covered CP stent[127]. However, there was no significant difference between bare metal and covered stents in a randomized trial of 120 patients[132]. A study by Forbes et al[182] showed a lower rate of acute complications after stent implantation compared to balloon angioplasty or surgery, similarly to another study by Carr et al[183].
Patients with CoA have a reduced life expectancy and increased risk of morbidity after intervention, despite good long-term survival rates. Follow-up protocols after intervention vary by institution but most commonly, routine visits at 3, 6, and 12 mo in the first year are followed by annual visits, obtaining blood pressure in all limbs and EKG at each visit with consideration for echocardiograms for concerns about restenosis or ventricular function. CTA or CMR can be done at 6-12 mo after the intervention to detect late complications such as aneurysm and repeated at five years or less depending on the findings. Perhaps the most important and prevalent long-term complication is chronic hypertension, occurring in 35%-68% of patients[58,184-185]. Furthermore, exercise-induced hypertension occurs in over one-third of normotensive patients at rest[186]. Follow-up should include ambulatory 24 h blood pressure measurement and exercise testing, as exercise-induced hypertension can predict future systemic hypertension[58]. Usually, normotensive patients at rest and with exercise lead normal lives without restriction, excluding extensive static sports. Patients with chronic hypertension, residual obstruction, or other complications should avoid strenuous sports and heavy exercises[162].
Patients with CoA should be evaluated before pregnancy for appropriate counseling and undergo arch imaging to assess aneurysm formation that might result in complications related to the increased cardiac output during pregnancy. Possible re-coarctation should be managed before conception to avoid uncontrolled upper body hypertension and possible reduced placental perfusion. Case studies have reported aortic rupture and dissection occurring with pregnancy after CoA repair but remain rare. However, there is a greater risk of developing hypertension during pre-gnancy[187]. A study reported acceptable outcomes of pregnancy in patients after CoA repair with no significant cardiovascular complications during pregnancy and delivery[188]. Another study showed that serious complications were uncommon in women with a hemodynamically significant gradient (≥ 20 mmHg) after repair[189]. However, women with unrepaired and repaired CoA are at increased risk of arterial hypertension, re-coarctation, aortic dissection or rupture, and rupture of a cerebral aneurysm during pregnancy and delivery.
Coarctation of the aorta is a relatively common congenital cardiac defect that often has few symptoms and therefore can be difficult to diagnose. The hallmark physical exam finding is upper extremity hypertension, and for this reason the diagnosis of coarctation of the aorta should be considered in any patient with systemic hypertension with assessment of a lower extremity blood pressure. The presence of a significant arm to leg gradient is highly suggestive of the diagnosis. Early diagnosis and treatment are important as long-term data consistently demonstrate that patients with CoA continue to have a reduced life expectancy and increased risk of cardiovascular complications. Surgical repair has traditionally been the mainstay of therapy for correction but advances in endovascular technology with covered stents and stent grafts now permit nonsurgical approaches for the management of children and adults with native CoA and complications. Persistent hypertension and vascular dysfunction can lead to an increased risk of coronary disease, which remains the greatest cause of long-term mortality. Thus, blood pressure control and periodic reassessment with TTE and CMR for evaluation of the heart and aorta should be performed regularly as cardiovascular complications may occur decades after the intervention.
Manuscript source: Invited manuscript
Specialty type: Cardiac and cardiovascular systems
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): A, A
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Li YH, IyngkaranP, Spiliopoulos S S-Editor: Wang YQ L-Editor: A E-Editor: Liu JH
| 1. | Reller MD, Strickland MJ, Riehle-Colarusso T, Mahle WT, Correa A. Prevalence of congenital heart defects in metropolitan Atlanta, 1998-2005. J Pediatr. 2008;153:807-813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 952] [Cited by in RCA: 799] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 2. | Salahuddin A, Chan a, Zaidi AN. The Adult with Coarctation of the Aorta. Congenital Heart Disease. 2018;. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 3. | Thiene G. [G. B. Morgagni: De sedibus et causis morborum per anatomen indagatis]. G Ital Cardiol. 1985;15:558-560. [PubMed] |
| 4. | Grech V. Diagnostic and surgical trends, and epidemiology of coarctation of the aorta in a population-based study. Int J Cardiol. 1999;68:197-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 5. | Hoffman JI, Kaplan S. The incidence of congenital heart disease. J Am Coll Cardiol. 2002;39:1890-1900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3680] [Cited by in RCA: 3718] [Article Influence: 161.7] [Reference Citation Analysis (1)] |
| 6. | Ferencz C, Rubin JD, McCarter RJ, Brenner JI, Neill CA, Perry LW, Hepner SI, Downing JW. Congenital heart disease: prevalence at livebirth. The Baltimore-Washington Infant Study. Am J Epidemiol. 1985;121:31-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 708] [Cited by in RCA: 609] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 7. | Campbell M, Polani PE. The aetiology of coarctation of the aorta. Lancet. 1961;1:463-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 49] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 8. | Hoffman JI. Incidence of congenital heart disease: II. Prenatal incidence. Pediatr Cardiol. 1995;16:155-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 129] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 9. | Sebastià C, Quiroga S, Boyé R, Perez-Lafuente M, Castellà E, Alvarez-Castells A. Aortic stenosis: spectrum of diseases depicted at multisection CT. Radiographics. 2003;23 Spec No:S79-S91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 40] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 10. | Abbruzzese PA, Aidala E. Aortic coarctation: an overview. J Cardiovasc Med (Hagerstown). 2007;8:123-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 11. | Conte S, Lacour-Gayet F, Serraf A, Sousa-Uva M, Bruniaux J, Touchot A, Planché C. Surgical management of neonatal coarctation. J Thorac Cardiovasc Surg. 1995;109:663-74; discussion 674-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 113] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 12. | Hager A, Schreiber C, Nutzl S, Hess J. Mortality and restenosis rate of surgical coarctation repair in infancy: a study of 191 patients. Cardiology. 2009;112:36-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 33] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 13. | Lacour-Gayet F, Bruniaux J, Serraf A, Chambran P, Blaysat G, Losay J, Petit J, Kachaner J, Planché C. Hypoplastic transverse arch and coarctation in neonates. Surgical reconstruction of the aortic arch: a study of sixty-six patients. J Thorac Cardiovasc Surg. 1990;100:808-816. [PubMed] |
| 14. | Kimura-Hayama ET, Meléndez G, Mendizábal AL, Meave-González A, Zambrana GF, Corona-Villalobos CP. Uncommon congenital and acquired aortic diseases: role of multidetector CT angiography. Radiographics. 2010;30:79-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 58] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 15. | Elgamal MA, McKenzie ED, Fraser CD. Aortic arch advancement: the optimal one-stage approach for surgical management of neonatal coarctation with arch hypoplasia. Ann Thorac Surg. 2002;73:1267-1272; discussion 1272-1273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 56] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Matsui H, Adachi I, Uemura H, Gardiner H, Ho SY. Anatomy of coarctation, hypoplastic and interrupted aortic arch: relevance to interventional/surgical treatment. Expert Rev Cardiovasc Ther. 2007;5:871-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 17. | Nance JW, Ringel RE, Fishman EK. Coarctation of the aorta in adolescents and adults: A review of clinical features and CT imaging. J Cardiovasc Comput Tomogr. 2016;10:1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 18. | McBride KL, Riley MF, Zender GA, Fitzgerald-Butt SM, Towbin JA, Belmont JW, Cole SE. NOTCH1 mutations in individuals with left ventricular outflow tract malformations reduce ligand-induced signaling. Hum Mol Genet. 2008;17:2886-2893. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 172] [Cited by in RCA: 148] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 19. | McBride KL, Zender GA, Fitzgerald-Butt SM, Koehler D, Menesses-Diaz A, Fernbach S, Lee K, Towbin JA, Leal S, Belmont JW. Linkage analysis of left ventricular outflow tract malformations (aortic valve stenosis, coarctation of the aorta, and hypoplastic left heart syndrome). Eur J Hum Genet. 2009;17:811-819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 65] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 20. | Elzenga NJ, Gittenberger-de Groot AC. Coarctation and related aortic arch anomalies in hypoplastic left heart syndrome. Int J Cardiol. 1985;8:379-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 21] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 21. | Smith DE, Matthews MB. Aortic valvular stenosis with coarctation of the aorta, with special reference to the development of aortic stenosis upon congenital bicuspid valves. Br Heart J. 1955;17:198-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 22. | Becker AE, Becker MJ, Edwards JE. Anomalies associated with coarctation of aorta: particular reference to infancy. Circulation. 1970;41:1067-1075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 129] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 23. | Tzemos N, Therrien J, Yip J, Thanassoulis G, Tremblay S, Jamorski MT, Webb GD, Siu SC. Outcomes in adults with bicuspid aortic valves. JAMA. 2008;300:1317-1325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 417] [Cited by in RCA: 405] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 24. | Zhao Q, Shi K, Yang ZG, Diao KY, Xu HY, Liu X, Guo YK. Predictors of aortic dilation in patients with coarctation of the aorta: evaluation with dual-source computed tomography. BMC Cardiovasc Disord. 2018;18:124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 25. | Dijkema EJ, Leiner T, Grotenhuis HB. Diagnosis, imaging and clinical management of aortic coarctation. Heart. 2017;103:1148-1155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 106] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 26. | Anderson RH, Lenox CC, Zuberbuhler JR. Morphology of ventricular septal defect associated with coarctation of aorta. Br Heart J. 1983;50:176-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 48] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Shinebourne EA, Tam AS, Elseed AM, Paneth M, Lennox SC, Cleland WP. Coarctation of the aorta in infancy and childhood. Br Heart J. 1976;38:375-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 83] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 28. | Shone JD, Sellers RD, Anderson RC, Adams P, Lillehei CW, Edwards JE. The developmental complex of "parachute mitral valve," supravalvular ring of left atrium, subaortic stenosis, and coarctation of aorta. Am J Cardiol. 1963;11:714-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 430] [Cited by in RCA: 346] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 29. | Cramer JW, Bartz PJ, Simpson PM, Zangwill SD. The spectrum of congenital heart disease and outcomes after surgical repair among children with Turner syndrome: a single-center review. Pediatr Cardiol. 2014;35:253-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 30. | Bondy CA. Aortic dissection in Turner syndrome. Curr Opin Cardiol. 2008;23:519-526. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 105] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 31. | Eckhauser A, South ST, Meyers L, Bleyl SB, Botto LD. Turner Syndrome in Girls Presenting with Coarctation of the Aorta. J Pediatr. 2015;167:1062-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 32. | Wong SC, Burgess T, Cheung M, Zacharin M. The prevalence of turner syndrome in girls presenting with coarctation of the aorta. J Pediatr. 2014;164:259-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 33. | Arora SS, Plato BM, Sattenberg RJ, Downs RK, Remmel KS, Heidenreich JO. Adult presentation of PHACES syndrome. Interv Neuroradiol. 2011;17:137-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 34. | Kammache I, Acar P, Kreitmann B, Fraisse A. Williams-Beuren syndrome: an unusual cause of neonatal and infantile coarctation. J Thorac Cardiovasc Surg. 2010;140:e80-e81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 35. | Agarwal V, Ramnarine I, Corno AF, Pozzi M. Recurrent coarctation in a patient with Alagille syndrome. Interact Cardiovasc Thorac Surg. 2005;4:514-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 36. | Marino B, Digilio MC, Toscano A, Giannotti A, Dallapiccola B. Congenital heart diseases in children with Noonan syndrome: An expanded cardiac spectrum with high prevalence of atrioventricular canal. J Pediatr. 1999;135:703-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 164] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 37. | Benyounes N, Blanc R, Boissonnet H, Piotin M. Subarachnoid hemorrhage revealing aortic coarctation in a young man. Neuroradiology. 2011;53:931-932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 38. | Alkashkari W, Albugami S, Hijazi ZM. Management of Coarctation of The Aorta in Adult Patients: State of The Art. Korean Circ J. 2019;49:298-313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 39. | Campbell M. Natural history of coarctation of the aorta. Br Heart J. 1970;32:633-640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 392] [Cited by in RCA: 357] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 40. | Rudolph AM, Heymann MA, Spitznas U. Hemodynamic considerations in the development of narrowing of the aorta. Am J Cardiol. 1972;30:514-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 330] [Cited by in RCA: 265] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 41. | Hutchins GM. Coarctation of the aorta explained as a branch-point of the ductus arteriosus. Am J Pathol. 1971;63:203-214. [PubMed] |
| 42. | Ho SY, Anderson RH. Coarctation, tubular hypoplasia, and the ductus arteriosus. Histological study of 35 specimens. Br Heart J. 1979;41:268-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 129] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 43. | Russell GA, Berry PJ, Watterson K, Dhasmana JP, Wisheart JD. Patterns of ductal tissue in coarctation of the aorta in the first three months of life. J Thorac Cardiovasc Surg. 1991;102:596-601. [PubMed] |
| 44. | Wielenga G, Dankmeijer J. Coarctation of the aorta. J Pathol Bacteriol. 1968;95:265-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 45. | Krediet P. An Hypothesis of the Development of Coarctation in Man. Acta Morphol Neerl Scand. 1965;6:207-212. [PubMed] |
| 46. | Sehested J. Coarctation of the aorta in monozygotic twins. Br Heart J. 1982;47:619-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 47. | Wessels MW, Berger RM, Frohn-Mulder IM, Roos-Hesselink JW, Hoogeboom JJ, Mancini GS, Bartelings MM, Krijger Rd, Wladimiroff JW, Niermeijer MF, Grossfeld P, Willems PJ. Autosomal dominant inheritance of left ventricular outflow tract obstruction. Am J Med Genet A. 2005;134A:171-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 52] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 48. | Boon AR, Roberts DF. A family study of coarctation of the aorta. J Med Genet. 1976;13:420-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 49. | Carmeliet P, Ferreira V, Breier G, Pollefeyt S, Kieckens L, Gertsenstein M, Fahrig M, Vandenhoeck A, Harpal K, Eberhardt C, Declercq C, Pawling J, Moons L, Collen D, Risau W, Nagy A. Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature. 1996;380:435-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2997] [Cited by in RCA: 2871] [Article Influence: 99.0] [Reference Citation Analysis (0)] |
| 50. | Sehested J, Baandrup U, Mikkelsen E. Different reactivity and structure of the prestenotic and poststenotic aorta in human coarctation. Implications for baroreceptor function. Circulation. 1982;65:1060-1065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 120] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 51. | Nguyen L, Cook SC. Coarctation of the Aorta: Strategies for Improving Outcomes. Cardiol Clin. 2015;33:521-530, vii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 52. | Pagni S, Denatale RW, Boltax RS. Takayasu's arteritis: the middle aortic syndrome. Am Surg. 1996;62:409-412. [PubMed] |
| 53. | Sheikhzadeh A, Giannitsis E, Gehl HB, Maring C, Stierle U. Acquired thromboatheromatous coarctation of the aorta: acquired coarctation of the aorta. Int J Cardiol. 1999;69:87-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 54. | McBride KL, Marengo L, Canfield M, Langlois P, Fixler D, Belmont JW. Epidemiology of noncomplex left ventricular outflow tract obstruction malformations (aortic valve stenosis, coarctation of the aorta, hypoplastic left heart syndrome) in Texas, 1999-2001. Birth Defects Res A Clin Mol Teratol. 2005;73:555-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 55] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 55. | Doshi AR, Chikkabyrappa S. Coarctation of Aorta in Children. Cureus. 2018;10:e3690. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 56. | Rhodes JF, Hijazi ZM, Sommer RJ. Pathophysiology of congenital heart disease in the adult, part II. Simple obstructive lesions. Circulation. 2008;117:1228-1237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 57. | Jenkins NP, Ward C. Coarctation of the aorta: natural history and outcome after surgical treatment. QJM. 1999;92:365-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 118] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 58. | Bocelli A, Favilli S, Pollini I, Bini RM, Ballo P, Chiappa E, Zuppiroli A. Prevalence and long-term predictors of left ventricular hypertrophy, late hypertension, and hypertensive response to exercise after successful aortic coarctation repair. Pediatr Cardiol. 2013;34:620-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 59. | Cohen M, Fuster V, Steele PM, Driscoll D, McGoon DC. Coarctation of the aorta. Long-term follow-up and prediction of outcome after surgical correction. Circulation. 1989;80:840-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 492] [Cited by in RCA: 452] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 60. | Clarkson PM, Nicholson MR, Barratt-Boyes BG, Neutze JM, Whitlock RM. Results after repair of coarctation of the aorta beyond infancy: a 10 to 28 year follow-up with particular reference to late systemic hypertension. Am J Cardiol. 1983;51:1481-1488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 151] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 61. | Seirafi PA, Warner KG, Geggel RL, Payne DD, Cleveland RJ. Repair of coarctation of the aorta during infancy minimizes the risk of late hypertension. Ann Thorac Surg. 1998;66:1378-1382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 74] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 62. | Liberthson RR, Pennington DG, Jacobs ML, Daggett WM. Coarctation of the aorta: review of 234 patients and clarification of management problems. Am J Cardiol. 1979;43:835-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 126] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 63. | Tobian L. A viewpoint concerning the enigma of hypertension. Am J Med. 1972;52:595-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 64. | Johnson D, Perrault H, Vobecky SJ, Trudeau F, Delvin E, Fournier A, Davignon A. Resetting of the cardiopulmonary baroreflex 10 years after surgical repair of coarctation of the aorta. Heart. 2001;85:318-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 65. | de Divitiis M, Rubba P, Calabrò R. Arterial hypertension and cardiovascular prognosis after successful repair of aortic coarctation: a clinical model for the study of vascular function. Nutr Metab Cardiovasc Dis. 2005;15:382-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 66. | Pedersen TA, Pedersen EB, Munk K, Hjortdal VE, Emmertsen K, Andersen NH. High pulse pressure is not associated with abnormal activation of the renin-angiotensin-aldosterone system in repaired aortic coarctation. J Hum Hypertens. 2015;29:268-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 67. | Martinez-Maldonado M, Delafontaine P, Anwar A, Fath KA, Pimentel JL, Bernstein KE, Luo C, Wang S, Benabe JE. Aortic and renal regulation of the renin-angiotensin system in interrenal aortic coarctation. Trans Assoc Am Physicians. 1993;106:120-127. [PubMed] |
| 68. | Torok RD, Campbell MJ, Fleming GA, Hill KD. Coarctation of the aorta: Management from infancy to adulthood. World J Cardiol. 2015;7:765-775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 101] [Article Influence: 10.1] [Reference Citation Analysis (3)] |
| 69. | Kenny D, Polson JW, Martin RP, Paton JF, Wolf AR. Hypertension and coarctation of the aorta: an inevitable consequence of developmental pathophysiology. Hypertens Res. 2011;34:543-547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 54] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 70. | Aoun SG, El Ahmadieh TY, Soltanolkotabi M, Ansari SA, Marden FA, Batjer HH, Bendok BR. Ruptured spinal artery aneurysm associated with coarctation of the aorta. World Neurosurg. 2014;81:441.e17-441.e22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 71. | Luo ZQ, Lai YQ, Zhu JM, Li JH, Liu KY, Zhou QW. Intercostal artery aneurysm associated with coarctation of aorta in an adult. J Card Surg. 2010;25:719-720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 72. | Donti A, Spinardi L, Brighenti M, Faccioli L, Leoni C, Fabi M, Trossello MP, Gargiulo GD, Bonvicini M. Frequency of Intracranial Aneurysms Determined by Magnetic Resonance Angiography in Children (Mean Age 16) Having Operative or Endovascular Treatment of Coarctation of the Aorta (Mean Age 3). Am J Cardiol. 2015;116:630-633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 73. | Urbina E, Alpert B, Flynn J, Hayman L, Harshfield GA, Jacobson M, Mahoney L, McCrindle B, Mietus-Snyder M, Steinberger J, Daniels S; American Heart Association Atherosclerosis, Hypertension, and Obesity in Youth Committee. Ambulatory blood pressure monitoring in children and adolescents: recommendations for standard assessment: a scientific statement from the American Heart Association Atherosclerosis, Hypertension, and Obesity in Youth Committee of the council on cardiovascular disease in the young and the council for high blood pressure research. Hypertension. 2008;52:433-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 362] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 74. | Engvall J, Sonnhag C, Nylander E, Stenport G, Karlsson E, Wranne B. Arm-ankle systolic blood pressure difference at rest and after exercise in the assessment of aortic coarctation. Br Heart J. 1995;73:270-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 75. | Teien DE, Wendel H, Björnebrink J, Ekelund L. Evaluation of anatomical obstruction by Doppler echocardiography and magnetic resonance imaging in patients with coarctation of the aorta. Br Heart J. 1993;69:352-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 76. | Marx GR, Allen HD. Accuracy and pitfalls of Doppler evaluation of the pressure gradient in aortic coarctation. J Am Coll Cardiol. 1986;7:1379-1385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 63] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 77. | Houston AB, Simpson IA, Pollock JC, Jamieson MP, Doig WB, Coleman EN. Doppler ultrasound in the assessment of severity of coarctation of the aorta and interruption of the aortic arch. Br Heart J. 1987;57:38-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 54] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 78. | Silvilairat S, Cetta F, Biliciler-Denktas G, Ammash NM, Cabalka AK, Hagler DJ, O'Leary PW. Abdominal aortic pulsed wave Doppler patterns reliably reflect clinical severity in patients with coarctation of the aorta. Congenit Heart Dis. 2008;3:422-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 79. | Nielsen JC, Powell AJ, Gauvreau K, Marcus EN, Prakash A, Geva T. Magnetic resonance imaging predictors of coarctation severity. Circulation. 2005;111:622-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 95] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 80. | Muzzarelli S, Meadows AK, Ordovas KG, Hope MD, Higgins CB, Nielsen JC, Geva T, Meadows JJ. Prediction of hemodynamic severity of coarctation by magnetic resonance imaging. Am J Cardiol. 2011;108:1335-1340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 81. | Stout KK, Daniels CJ, Aboulhosn JA, Bozkurt B, Broberg CS, Colman JM, Crumb SR, Dearani JA, Fuller S, Gurvitz M, Khairy P, Landzberg MJ, Saidi A, Valente AM, Van Hare GF. 2018 AHA/ACC Guideline for the Management of Adults With Congenital Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;73:e81-e192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 606] [Article Influence: 86.6] [Reference Citation Analysis (0)] |
| 82. | Greenberg SB, Balsara RK, Faerber EN. Coarctation of the aorta: diagnostic imaging after corrective surgery. J Thorac Imaging. 1995;10:36-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 83. | Shepherd B, Abbas A, McParland P, Fitzsimmons S, Shambrook J, Peebles C, Brown I, Harden S. MRI in adult patients with aortic coarctation: diagnosis and follow-up. Clin Radiol. 2015;70:433-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 84. | Goldberg A, Jha S. Phase-contrast MRI and applications in congenital heart disease. Clin Radiol. 2012;67:399-410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 85. | Hope MD, Meadows AK, Hope TA, Ordovas KG, Saloner D, Reddy GP, Alley MT, Higgins CB. Clinical evaluation of aortic coarctation with 4D flow MR imaging. J Magn Reson Imaging. 2010;31:711-718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 105] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 86. | Marek J, Skovránek J, Hucín B, Chaloupecký V, Tax P, Reich O, Samánek M. Seven-year experience of noninvasive preoperative diagnostics in children with congenital heart defects: comprehensive analysis of 2,788 consecutive patients. Cardiology. 1995;86:488-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 87. | Crafoord C. Congenital coarctation of the aorta and its surgical treatment. J Thorac Surg. 1945;14:347-361. |
| 88. | Gross RE, Hufnagel CA. Coarctation of the aorta: experimental studies regarding its srugical correciton. N Engl J Med. 1945;233:287-293. [RCA] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 202] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 89. | Williams WG, Shindo G, Trusler GA, Dische MR, Olley PM. Results of repair of coarctation of the aorta during infancy. J Thorac Cardiovasc Surg. 1980;79:603-608. [PubMed] |
| 90. | Kappetein AP, Zwinderman AH, Bogers AJ, Rohmer J, Huysmans HA. More than thirty-five years of coarctation repair. An unexpected high relapse rate. J Thorac Cardiovasc Surg. 1994;107:87-95. [PubMed] |
| 91. | Hesslein PS, McNamara DG, Morriss MJ, Hallman GL, Cooley DA. Comparison of resection versus patch aortoplasty for repair of coarctation in infants and children. Circulation. 1981;64:164-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 56] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 92. | Ziemer G, Jonas RA, Perry SB, Freed MD, Castaneda AR. Surgery for coarctation of the aorta in the neonate. Circulation. 1986;74:I25-I31. [PubMed] |
| 93. | Vossschulte K. [Plastic surgery of the isthmus in aortic isthmus stenosis]. Thoraxchirurgie. 1957;4:443-450. [PubMed] |
| 94. | Venturini A, Perna AM, Bianchi G. Repair of coarctation of the thoracic aorta without resection. Patch graft aortoplasty. Follow-up study of 46 cases. J Cardiovasc Surg (Torino). 1978;19:49-54. [PubMed] |
| 95. | Tanous D, Benson LN, Horlick EM. Coarctation of the aorta: evaluation and management. Curr Opin Cardiol. 2009;24:509-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 71] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 96. | Walhout RJ, Lekkerkerker JC, Oron GH, Hitchcock FJ, Meijboom EJ, Bennink GB. Comparison of polytetrafluoroethylene patch aortoplasty and end-to-end anastomosis for coarctation of the aorta. J Thorac Cardiovasc Surg. 2003;126:521-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 61] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 97. | Waldhausen JA, Nahrwold DL. Repair of coarctation of the aorta with a subclavian flap. J Thorac Cardiovasc Surg. 1966;51:532-533. [PubMed] |
| 98. | Beekman RH, Rocchini AP, Behrendt DM, Bove EL, Dick M 2nd, Crowley DC, Snider AR, Rosenthal A. Long-term outcome after repair of coarctation in infancy: subclavian angioplasty does not reduce the need for reoperation. J Am Coll Cardiol. 1986;8:1406-1411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 67] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 99. | Pandey R, Jackson M, Ajab S, Gladman G, Pozzi M. Subclavian flap repair: review of 399 patients at median follow-up of fourteen years. Ann Thorac Surg. 2006;81:1420-1428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 100. | Barreiro CJ, Ellison TA, Williams JA, Durr ML, Cameron DE, Vricella LA. Subclavian flap aortoplasty: still a safe, reproducible, and effective treatment for infant coarctation. Eur J Cardiothorac Surg. 2007;31:649-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 27] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 101. | Amato JJ, Rheinlander HF, Cleveland RJ. A method of enlarging the distal transverse arch in infants with hypoplasia and coarctation of the aorta. Ann Thorac Surg. 1977;23:261-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 79] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 102. | Thomson JD, Mulpur A, Guerrero R, Nagy Z, Gibbs JL, Watterson KG. Outcome after extended arch repair for aortic coarctation. Heart. 2006;92:90-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 103. | van Son JA, Daniëls O, Lacquet LK. [Current viewpoints concerning the surgical treatment of aortic coarctation in infants and children]. Ned Tijdschr Geneeskd. 1990;134:1682-1688. [PubMed] |
| 104. | van Heurn LW, Wong CM, Spiegelhalter DJ, Sorensen K, de Leval MR, Stark J, Elliott MJ. Surgical treatment of aortic coarctation in infants younger than three months: 1985 to 1990. Success of extended end-to-end arch aortoplasty. J Thorac Cardiovasc Surg. 1994;107:74-85; discussion 85-86. [PubMed] |
| 105. | Troost E, Gewillig M, Daenen W, Meyns B, Bogaert J, Van Deyk K, Budts W. Behaviour of polyester grafts in adult patients with repaired coarctation of the aorta. Eur Heart J. 2009;30:1136-1141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 106. | Yousif A, Kloppenburg G, Morshuis WJ, Heijmen R. Repair of adult aortic coarctation by resection and interposition grafting. Interact Cardiovasc Thorac Surg. 2016;23:526-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 107. | Bartoccioni S, Giombolini C, Fiaschini P, Martinelli G, Fedeli C, Di Lazzaro D, Lanzillo G, Possati G. Aortic coarctation, aortic valvular stenosis, and coronary artery disease: combined one-stage surgical therapy operation. J Card Surg. 1995;10:594-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 108. | Heinemann MK, Ziemer G, Wahlers T, Köhler A, Borst HG. Extraanatomic thoracic aortic bypass grafts: indications, techniques, and results. Eur J Cardiothorac Surg. 1997;11:169-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 50] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 109. | Grinda JM, Macé L, Dervanian P, Folliguet TA, Neveux JY. Bypass graft for complex forms of isthmic aortic coarctation in adults. Ann Thorac Surg. 1995;60:1299-1302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 110. | Said SM, Burkhart HM, Dearani JA, Connolly HM, Schaff HV. Ascending-to-descending aortic bypass: a simple solution to a complex problem. Ann Thorac Surg. 2014;97:2041-2047; discussion 2047-2048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 111. | Puranik R, Tsang VT, Puranik S, Jones R, Cullen S, Bonhoeffer P, Hughes ML, Taylor AM. Late magnetic resonance surveillance of repaired coarctation of the aorta. Eur J Cardiothorac Surg. 2009;36:91-95; discussion 95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 112. | Wilson W, Osten M, Benson L, Horlick E. Evolving trends in interventional cardiology: endovascular options for congenital disease in adults. Can J Cardiol. 2014;30:75-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 113. | Singer MI, Rowen M, Dorsey TJ. Transluminal aortic balloon angioplasty for coarctation of the aorta in the newborn. Am Heart J. 1982;103:131-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 180] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 114. | Rothman A, Galindo A, Evans WN, Collazos JC, Restrepo H. Effectiveness and safety of balloon dilation of native aortic coarctation in premature neonates weighing < or = 2,500 grams. Am J Cardiol. 2010;105:1176-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 115. | Suárez de Lezo J, Pan M, Romero M, Segura J, Pavlovic D, Ojeda S, Algar J, Ribes R, Lafuente M, Lopez-Pujol J. Percutaneous interventions on severe coarctation of the aorta: a 21-year experience. Pediatr Cardiol. 2005;26:176-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 59] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 116. | Rao PS, Jureidini SB, Balfour IC, Singh GK, Chen SC. Severe aortic coarctation in infants less than 3 months: successful palliation by balloon angioplasty. J Invasive Cardiol. 2003;15:202-208. [PubMed] |
| 117. | Fiore AC, Fischer LK, Schwartz T, Jureidini S, Balfour I, Carpenter D, Demello D, Virgo KS, Pennington DG, Johnson RG. Comparison of angioplasty and surgery for neonatal aortic coarctation. Ann Thorac Surg. 2005;80:1659-1664; discussion 1664-1665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 118. | Reich O, Tax P, Bartáková H, Tomek V, Gilík J, Lisy J, Radvansky J, Matejka T, Tláskal T, Svobodová I, Chaloupecky V, Skovránek J. Long-term (up to 20 years) results of percutaneous balloon angioplasty of recurrent aortic coarctation without use of stents. Eur Heart J. 2008;29:2042-2048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 119. | Rodés-Cabau J, Miró J, Dancea A, Ibrahim R, Piette E, Lapierre C, Jutras L, Perron J, Tchervenkow CI, Poirier N, Dahdah NS, Houde C. Comparison of surgical and transcatheter treatment for native coarctation of the aorta in patients > or = 1 year old. The Quebec Native Coarctation of the Aorta study. Am Heart J. 2007;154:186-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 56] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 120. | Salcher M, Naci H, Law TJ, Kuehne T, Schubert S, Kelm M; Cardioproof Consortium. Balloon Dilatation and Stenting for Aortic Coarctation: A Systematic Review and Meta-Analysis. Circ Cardiovasc Interv. 2016;9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 121. | Pedra CA, Fontes VF, Esteves CA, Pilla CB, Braga SL, Pedra SR, Santana MV, Silva MA, Almeida T, Sousa JE. Stenting vs. balloon angioplasty for discrete unoperated coarctation of the aorta in adolescents and adults. Catheter Cardiovasc Interv. 2005;64:495-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 71] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 122. | Trent MS, Parsonnet V, Shoenfeld R, Brener BJ, Eisenbud DE, Novick AS, Campbell AY, Ferrara-Ryan M, Villanueva A. A balloon-expandable intravascular stent for obliterating experimental aortic dissection. J Vasc Surg. 1990;11:707-717. [PubMed] |
| 123. | Mohan UR, Danon S, Levi D, Connolly D, Moore JW. Stent implantation for coarctation of the aorta in children <30 kg. JACC Cardiovasc Interv. 2009;2:877-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 39] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 124. | Thanopoulos BD, Giannakoulas G, Giannopoulos A, Galdo F, Tsaoussis GS. Initial and six-year results of stent implantation for aortic coarctation in children. Am J Cardiol. 2012;109:1499-1503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 125. | Forbes TJ, Moore P, Pedra CA, Zahn EM, Nykanen D, Amin Z, Garekar S, Teitel D, Qureshi SA, Cheatham JP, Ebeid MR, Hijazi ZM, Sandhu S, Hagler DJ, Sievert H, Fagan TE, Ringwald J, Du W, Tang L, Wax DF, Rhodes J, Johnston TA, Jones TK, Turner DR, Pass R, Torres A, Hellenbrand WE. Intermediate follow-up following intravascular stenting for treatment of coarctation of the aorta. Catheter Cardiovasc Interv. 2007;70:569-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 112] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 126. | Forbes TJ, Garekar S, Amin Z, Zahn EM, Nykanen D, Moore P, Qureshi SA, Cheatham JP, Ebeid MR, Hijazi ZM, Sandhu S, Hagler DJ, Sievert H, Fagan TE, Ringewald J, Du W, Tang L, Wax DF, Rhodes J, Johnston TA, Jones TK, Turner DR, Pedra CA, Hellenbrand WE; Congenital Cardiovascular Interventional Study Consortium (CCISC). Procedural results and acute complications in stenting native and recurrent coarctation of the aorta in patients over 4 years of age: a multi-institutional study. Catheter Cardiovasc Interv. 2007;70:276-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 171] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 127. | Meadows J, Minahan M, McElhinney DB, McEnaney K, Ringel R; COAST Investigators*. Intermediate Outcomes in the Prospective, Multicenter Coarctation of the Aorta Stent Trial (COAST). Circulation. 2015;131:1656-1664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 107] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 128. | Kische S, D'Ancona G, Stoeckicht Y, Ortak J, Elsässer A, Ince H. Percutaneous treatment of adult isthmic aortic coarctation: acute and long-term clinical and imaging outcome with a self-expandable uncovered nitinol stent. Circ Cardiovasc Interv. 2015;8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 129. | Tanous D, Collins N, Dehghani P, Benson LN, Horlick EM. Covered stents in the management of coarctation of the aorta in the adult: initial results and 1-year angiographic and hemodynamic follow-up. Int J Cardiol. 2010;140:287-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 58] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 130. | Bruckheimer E, Birk E, Santiago R, Dagan T, Esteves C, Pedra CA. Coarctation of the aorta treated with the Advanta V12 large diameter stent: acute results. Catheter Cardiovasc Interv. 2010;75:402-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 131. | Chakrabarti S, Kenny D, Morgan G, Curtis SL, Hamilton MC, Wilde P, Tometzki AJ, Turner MS, Martin RP. Balloon expandable stent implantation for native and recurrent coarctation of the aorta--prospective computed tomography assessment of stent integrity, aneurysm formation and stenosis relief. Heart. 2010;96:1212-1216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 61] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 132. | Sohrabi B, Jamshidi P, Yaghoubi A, Habibzadeh A, Hashemi-Aghdam Y, Moin A, Kazemi B, Ghaffari S, Abdolahzadeh Baghayi MR, Mahmoody K. Comparison between covered and bare Cheatham-Platinum stents for endovascular treatment of patients with native post-ductal aortic coarctation: immediate and intermediate-term results. JACC Cardiovasc Interv. 2014;7:416-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 133. | Bruckheimer E, Dagan T, Amir G, Birk E. Covered Cheatham-Platinum stents for serial dilation of severe native aortic coarctation. Catheter Cardiovasc Interv. 2009;74:117-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 134. | Butera G, Gaio G, Carminati M. Redilation of e-PTFE covered CP stents. Catheter Cardiovasc Interv. 2008;72:273-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 135. | Rao PS, Thapar MK, Kutayli F, Carey P. Causes of recoarctation after balloon angioplasty of unoperated aortic coarctation. J Am Coll Cardiol. 1989;13:109-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 81] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 136. | Fletcher SE, Nihill MR, Grifka RG, O'Laughlin MP, Mullins CE. Balloon angioplasty of native coarctation of the aorta: midterm follow-up and prognostic factors. J Am Coll Cardiol. 1995;25:730-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 112] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 137. | Warnes CA, Williams RG, Bashore TM, Child JS, Connolly HM, Dearani JA, del Nido P, Fasules JW, Graham TP, Hijazi ZM, Hunt SA, King ME, Landzberg MJ, Miner PD, Radford MJ, Walsh EP, Webb GD. ACC/AHA 2008 Guidelines for the Management of Adults with Congenital Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to develop guidelines on the management of adults with congenital heart disease). Circulation. 2008;118:e714-e833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 663] [Cited by in RCA: 636] [Article Influence: 37.4] [Reference Citation Analysis (0)] |
| 138. | Feltes TF, Bacha E, Beekman RH, Cheatham JP, Feinstein JA, Gomes AS, Hijazi ZM, Ing FF, de Moor M, Morrow WR, Mullins CE, Taubert KA, Zahn EM; American Heart Association Congenital Cardiac Defects Committee of the Council on Cardiovascular Disease in the Young; Council on Clinical Cardiology; Council on Cardiovascular Radiology and Intervention; American Heart Association. Indications for cardiac catheterization and intervention in pediatric cardiac disease: a scientific statement from the American Heart Association. Circulation. 2011;123:2607-2652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 477] [Cited by in RCA: 550] [Article Influence: 39.3] [Reference Citation Analysis (0)] |
| 139. | Silversides CK, Kiess M, Beauchesne L, Bradley T, Connelly M, Niwa K, Mulder B, Webb G, Colman J, Therrien J. Canadian Cardiovascular Society 2009 Consensus Conference on the management of adults with congenital heart disease: outflow tract obstruction, coarctation of the aorta, tetralogy of Fallot, Ebstein anomaly and Marfan's syndrome. Can J Cardiol. 2010;26:e80-e97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 145] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 140. | Pourmoghadam KK, Velamoor G, Kneebone JM, Patterson K, Jones TK, Lupinetti FM. Changes in protein distribution of the aortic wall following balloon aortoplasty for coarctation. Am J Cardiol. 2002;89:91-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 141. | Isner JM, Donaldson RF, Fulton D, Bhan I, Payne DD, Cleveland RJ. Cystic medial necrosis in coarctation of the aorta: a potential factor contributing to adverse consequences observed after percutaneous balloon angioplasty of coarctation sites. Circulation. 1987;75:689-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 119] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 142. | Moodie DS. Aortic dissection and coarctation. Curr Opin Cardiol. 1990;5:649-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 143. | von Kodolitsch Y, Aydin MA, Koschyk DH, Loose R, Schalwat I, Karck M, Cremer J, Haverich A, Berger J, Meinertz T, Nienaber CA. Predictors of aneurysmal formation after surgical correction of aortic coarctation. J Am Coll Cardiol. 2002;39:617-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 146] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 144. | Shaddy RE, Boucek MM, Sturtevant JE, Ruttenberg HD, Jaffe RB, Tani LY, Judd VE, Veasy LG, McGough EC, Orsmond GS. Comparison of angioplasty and surgery for unoperated coarctation of the aorta. Circulation. 1993;87:793-799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 105] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 145. | Rao PS, Galal O, Smith PA, Wilson AD. Five- to nine-year follow-up results of balloon angioplasty of native aortic coarctation in infants and children. J Am Coll Cardiol. 1996;27:462-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 136] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 146. | Fawzy ME, Sivanandam V, Galal O, Dunn B, Patel A, Rifai A, von Sinner W, Al Halees Z, Khan B. One- to ten-year follow-up results of balloon angioplasty of native coarctation of the aorta in adolescents and adults. J Am Coll Cardiol. 1997;30:1542-1546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 47] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 147. | Mullen MJ. Coarctation of the aorta in adults: do we need surgeons? Heart. 2003;89:3-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 148. | Zabal C, Attie F, Rosas M, Buendía-Hernández A, García-Montes JA. The adult patient with native coarctation of the aorta: balloon angioplasty or primary stenting? Heart. 2003;89:77-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 92] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 149. | Hamdan MA, Maheshwari S, Fahey JT, Hellenbrand WE. Endovascular stents for coarctation of the aorta: initial results and intermediate-term follow-up. J Am Coll Cardiol. 2001;38:1518-1523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 153] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 150. | Daniels SR. Repair of coarctation of the aorta and hypertension: does age matter? Lancet. 2001;358:89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 151. | Toro-Salazar OH, Steinberger J, Thomas W, Rocchini AP, Carpenter B, Moller JH. Long-term follow-up of patients after coarctation of the aorta repair. Am J Cardiol. 2002;89:541-547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 232] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 152. | Gidding SS, Rocchini AP, Moorehead C, Schork MA, Rosenthal A. Increased forearm vascular reactivity in patients with hypertension after repair of coarctation. Circulation. 1985;71:495-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 65] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 153. | Pelech AN, Kartodihardjo W, Balfe JA, Balfe JW, Olley PM, Leenen FH. Exercise in children before and after coarctectomy: hemodynamic, echocardiographic, and biochemical assessment. Am Heart J. 1986;112:1263-1270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 35] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 154. | Trojnarska O, Szczepaniak-Chicheł L, Mizia-Stec K, Gabriel M, Bartczak A, Grajek S, Gąsior Z, Kramer L, Tykarski A. Vascular remodeling in adults after coarctation repair: impact of descending aorta stenosis and age at surgery. Clin Res Cardiol. 2011;100:447-455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 155. | de Divitiis M, Pilla C, Kattenhorn M, Zadinello M, Donald A, Leeson P, Wallace S, Redington A, Deanfield JE. Vascular dysfunction after repair of coarctation of the aorta: impact of early surgery. Circulation. 2001;104:I165-I170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 194] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 156. | Beekman RH, Katz BP, Moorehead-Steffens C, Rocchini AP. Altered baroreceptor function in children with systolic hypertension after coarctation repair. Am J Cardiol. 1983;52:112-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 79] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 157. | Simsolo R, Grunfeld B, Gimenez M, Lopez M, Berri G, Becú L, Barontini M. Long-term systemic hypertension in children after successful repair of coarctation of the aorta. Am Heart J. 1988;115:1268-1273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 158. | Weber HS, Cyran SE, Grzeszczak M, Myers JL, Gleason MM, Baylen BG. Discrepancies in aortic growth explain aortic arch gradients during exercise. J Am Coll Cardiol. 1993;21:1002-1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 159. | Beckmann E, Jassar AS. Coarctation repair-redo challenges in the adults: what to do? J Vis Surg. 2018;4:76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 160. | Zoghbi J, Serraf A, Mohammadi S, Belli E, Lacour Gayet F, Aupecle B, Losay J, Petit J, Planché C. Is surgical intervention still indicated in recurrent aortic arch obstruction? J Thorac Cardiovasc Surg. 2004;127:203-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 161. | Früh S, Knirsch W, Dodge-Khatami A, Dave H, Prêtre R, Kretschmar O. Comparison of surgical and interventional therapy of native and recurrent aortic coarctation regarding different age groups during childhood. Eur J Cardiothorac Surg. 2011;39:898-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 162. | Baumgartner H, Bonhoeffer P, De Groot NM, de Haan F, Deanfield JE, Galie N, Gatzoulis MA, Gohlke-Baerwolf C, Kaemmerer H, Kilner P, Meijboom F, Mulder BJ, Oechslin E, Oliver JM, Serraf A, Szatmari A, Thaulow E, Vouhe PR, Walma E; Task Force on the Management of Grown-up Congenital Heart Disease of the European Society of Cardiology (ESC); Association for European Paediatric Cardiology (AEPC); ESC Committee for Practice Guidelines (CPG). ESC Guidelines for the management of grown-up congenital heart disease (new version 2010). Eur Heart J. 2010;31:2915-2957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1602] [Cited by in RCA: 1555] [Article Influence: 103.7] [Reference Citation Analysis (1)] |
| 163. | Warnes CA, Williams RG, Bashore TM, Child JS, Connolly HM, Dearani JA, Del Nido P, Fasules JW, Graham TP, Hijazi ZM, Hunt SA, King ME, Landzberg MJ, Miner PD, Radford MJ, Walsh EP, Webb GD. ACC/AHA 2008 guidelines for the management of adults with congenital heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Develop Guidelines on the Management of Adults With Congenital Heart Disease). Developed in Collaboration With the American Society of Echocardiography, Heart Rhythm Society, International Society for Adult Congenital Heart Disease, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2008;52:e143-e263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 993] [Cited by in RCA: 1016] [Article Influence: 63.5] [Reference Citation Analysis (0)] |
| 164. | Yetman AT, Nykanen D, McCrindle BW, Sunnegardh J, Adatia I, Freedom RM, Benson L. Balloon angioplasty of recurrent coarctation: a 12-year review. J Am Coll Cardiol. 1997;30:811-816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 93] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 165. | Hellenbrand WE, Allen HD, Golinko RJ, Hagler DJ, Lutin W, Kan J. Balloon angioplasty for aortic recoarctation: results of Valvuloplasty and Angioplasty of Congenital Anomalies Registry. Am J Cardiol. 1990;65:793-797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 138] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 166. | Hijazi ZM, Fahey JT, Kleinman CS, Hellenbrand WE. Balloon angioplasty for recurrent coarctation of aorta. Immediate and long-term results. Circulation. 1991;84:1150-1156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 83] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 167. | Anjos R, Qureshi SA, Rosenthal E, Murdoch I, Hayes A, Parsons J, Baker EJ, Tynan M. Determinants of hemodynamic results of balloon dilation of aortic recoarctation. Am J Cardiol. 1992;69:665-671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 41] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 168. | Witsenburg M, The SH, Bogers AJ, Hess J. Balloon angioplasty for aortic recoarctation in children: initial and follow up results and midterm effect on blood pressure. Br Heart J. 1993;70:170-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 169. | Magee AG, Brzezinska-Rajszys G, Qureshi SA, Rosenthal E, Zubrzycka M, Ksiazyk J, Tynan M. Stent implantation for aortic coarctation and recoarctation. Heart. 1999;82:600-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 97] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 170. | Harrison DA, McLaughlin PR, Lazzam C, Connelly M, Benson LN. Endovascular stents in the management of coarctation of the aorta in the adolescent and adult: one year follow up. Heart. 2001;85:561-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 105] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 171. | Kpodonu J, Ramaiah VG, Rodriguez-Lopez JA, Diethrich EB. Endovascular management of recurrent adult coarctation of the aorta. Ann Thorac Surg. 2010;90:1716-1720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 172. | Brown ML, Burkhart HM, Connolly HM, Dearani JA, Hagler DJ, Schaff HV. Late outcomes of reintervention on the descending aorta after repair of aortic coarctation. Circulation. 2010;122:S81-S84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 44] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 173. | Saxena A. Recurrent coarctation: interventional techniques and results. World J Pediatr Congenit Heart Surg. 2015;6:257-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 174. | Brown JW, Ruzmetov M, Hoyer MH, Rodefeld MD, Turrentine MW. Recurrent coarctation: is surgical repair of recurrent coarctation of the aorta safe and effective? Ann Thorac Surg. 2009;88:1923-30; discussion 1930-1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 53] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 175. | O'Laughlin MP, Perry SB, Lock JE, Mullins CE. Use of endovascular stents in congenital heart disease. Circulation. 1991;83:1923-1939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 351] [Cited by in RCA: 319] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 176. | Marshall AC, Perry SB, Keane JF, Lock JE. Early results and medium-term follow-up of stent implantation for mild residual or recurrent aortic coarctation. Am Heart J. 2000;139:1054-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 104] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 177. | Hartman EM, Groenendijk IM, Heuvelman HM, Roos-Hesselink JW, Takkenberg JJ, Witsenburg M. The effectiveness of stenting of coarctation of the aorta: a systematic review. EuroIntervention. 2015;11:660-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 178. | Bulbul ZR, Bruckheimer E, Love JC, Fahey JT, Hellenbrand WE. Implantation of balloon-expandable stents for coarctation of the aorta: implantation data and short-term results. Cathet Cardiovasc Diagn. 1996;39:36-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 179. | Pilla CB, Fontes VF, Pedra CA. Endovascular stenting for aortic coarctation. Expert Rev Cardiovasc Ther. 2005;3:879-890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 180. | Golden AB, Hellenbrand WE. Coarctation of the aorta: stenting in children and adults. Catheter Cardiovasc Interv. 2007;69:289-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 83] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 181. | Yang L, Chua X, Rajgor DD, Tai BC, Quek SC. A systematic review and meta-analysis of outcomes of transcatheter stent implantation for the primary treatment of native coarctation. Int J Cardiol. 2016;223:1025-1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 182. | Forbes TJ, Kim DW, Du W, Turner DR, Holzer R, Amin Z, Hijazi Z, Ghasemi A, Rome JJ, Nykanen D, Zahn E, Cowley C, Hoyer M, Waight D, Gruenstein D, Javois A, Foerster S, Kreutzer J, Sullivan N, Khan A, Owada C, Hagler D, Lim S, Canter J, Zellers T; CCISC Investigators. Comparison of surgical, stent, and balloon angioplasty treatment of native coarctation of the aorta: an observational study by the CCISC (Congenital Cardiovascular Interventional Study Consortium). J Am Coll Cardiol. 2011;58:2664-2674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 217] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 183. | Carr JA. The results of catheter-based therapy compared with surgical repair of adult aortic coarctation. J Am Coll Cardiol. 2006;47:1101-1107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 105] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 184. | Canniffe C, Ou P, Walsh K, Bonnet D, Celermajer D. Hypertension after repair of aortic coarctation--a systematic review. Int J Cardiol. 2013;167:2456-2461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 102] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 185. | Choudhary P, Canniffe C, Jackson DJ, Tanous D, Walsh K, Celermajer DS. Late outcomes in adults with coarctation of the aorta. Heart. 2015;101:1190-1195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 84] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 186. | O'Sullivan J. Late hypertension in patients with repaired aortic coarctation. Curr Hypertens Rep. 2014;16:421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 187. | Krieger EV, Landzberg MJ, Economy KE, Webb GD, Opotowsky AR. Comparison of risk of hypertensive complications of pregnancy among women with versus without coarctation of the aorta. Am J Cardiol. 2011;107:1529-1534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 188. | Vriend JW, Drenthen W, Pieper PG, Roos-Hesselink JW, Zwinderman AH, van Veldhuisen DJ, Mulder BJ. Outcome of pregnancy in patients after repair of aortic coarctation. Eur Heart J. 2005;26:2173-2178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 77] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 189. | Beauchesne LM, Connolly HM, Ammash NM, Warnes CA. Coarctation of the aorta: outcome of pregnancy. J Am Coll Cardiol. 2001;38:1728-1733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 150] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 190. | Tzifa A, Ewert P, Brzezinska-Rajszys G, Peters B, Zubrzycka M, Rosenthal E, Berger F, Qureshi SA. Covered Cheatham-platinum stents for aortic coarctation: early and intermediate-term results. J Am Coll Cardiol. 2006;47:1457-1463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 115] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 191. | Butera G, Piazza L, Chessa M, Negura DG, Rosti L, Abella R, Delogu A, Condoluci C, Magherini A, Carminati M. Covered stents in patients with complex aortic coarctations. Am Heart J. 2007;154:795-800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 48] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 192. | Kenny D, Margey R, Turner MS, Tometzki AJ, Walsh KP, Martin RP. Self-expanding and balloon expandable covered stents in the treatment of aortic coarctation with or without aneurysm formation. Catheter Cardiovasc Interv. 2008;72:65-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (1)] |