Published online Dec 10, 2015. doi: 10.4239/wjd.v6.i17.1337
Peer-review started: July 24, 2015
First decision: August 16, 2015
Revised: October 30, 2015
Accepted: November 17, 2015
Article in press: November 25, 2015
Published online: December 10, 2015
Processing time: 141 Days and 22 Hours
AIM: To assess the performance of the Finnish Diabetes Risk Score (FINDRISC) questionnaire for detecting and predicting type 2 diabetes mellitus (DM2) in a Colombian population.
METHODS: This is a longitudinal observational study conducted in Floridablanca, Colombia. Adult subjects (age ≥ 35 years) without known diabetes, were included. A modified version of FINDRISC was completed, and the glycemia values from all the subjects were collected from the hospital’s database. Firstly, a cross-sectional analysis was performed and then, the subsample of prediabetic participants was followed for diabetes incidence.
RESULTS: A total of 772 subjects were suitable for the study. The overall prevalence of undiagnosed DM2 was 2.59%, and the incidence of DM2 among the prediabetic participants was 7.5 per 100 person-years after a total of 265257 person-years follow-up. The FINDRISC at baseline was significantly associated with undiagnosed and incident DM2. The area under receiver operating characteristics curve of the FINDRISC score for detecting undiagnosed DM2 in both men and women was 0.7477 and 0.7175, respectively; and for predicting the incidence of DM2 among prediabetics was 71.99% in men and 67.74% in women.
CONCLUSION: The FINDRISC questionnaire is a useful screening tool to identify cross-sectionally unknown DM2 and to predict the incidence of DM2 among prediabetics in the Colombian population.
Core tip: The prevalence of type 2 diabetes mellitus (DM2) is rapidly increasing worldwide, and unfortunately its diagnosis is being made when vascular complications are already exhibited. Since laboratory-based diagnostic methods are costly, the International Diabetes Federation suggests to do an early detection of undiagnosed DM2 patients, and to identify individuals at risk for developing DM2 by simple risk-scoring questionnaires. The present study assesses the performance of the Finnish Diabetes Risk Score questionnaire in a Colombian population, and aims to establish the specific cutoff values for detecting subjects at increased risk of undiagnosed DM2 and for predicting the incidence of DM2 in prediabetic individuals.
- Citation: Gomez-Arbelaez D, Alvarado-Jurado L, Ayala-Castillo M, Forero-Naranjo L, Camacho PA, Lopez-Jaramillo P. Evaluation of the Finnish Diabetes Risk Score to predict type 2 diabetes mellitus in a Colombian population: A longitudinal observational study. World J Diabetes 2015; 6(17): 1337-1344
- URL: https://www.wjgnet.com/1948-9358/full/v6/i17/1337.htm
- DOI: https://dx.doi.org/10.4239/wjd.v6.i17.1337
The prevalence of type 2 diabetes mellitus (DM2) is rapidly increasing worldwide[1]. Recently, it has been estimated that the number of adults with DM2 will increase between 2010 and 2030 by 69% and 20% in developing and industrialized countries, respectively[1]. In Colombia the overall prevalence of DM2 is 5.51%, however significant differences are observed and in some regions the prevalence rises to 8.2%[2]. The increase in the prevalence of DM2 across the world has become an important public health concern given that it is a major risk factor for death and numerous nonfatal complications. Hence, this situation will form a large burden to the patients, their families, and the health care system[3].
Several studies have demonstrated that DM2 could be prevented, and its complications can be limited when a timely and appropriate intervention is started[4,5]. However, in the majority of cases detection is delayed and at the time of diagnosis many patients already exhibit signs of microvascular and macrovascular complications[6]. Remarkably, there has been proposed that Latin American population has an increased vulnerability for developing macrovascular diseases at glycemia levels lower than the internationally established cut points for DM2[7,8]. Thus, it is clinically important to do an early detection of undiagnosed DM2 patients, and to identify individuals at risk for developing DM2 to implement intensive preventive interventions.
The diagnosis of DM2 is obtained by increased values of fasting plasma glucose (FPG), oral glucose tolerance test (OGTT) and/or glycated hemoglobin (HbA1C)[9], but the determination of biochemical variables is costly, and this impairs its use as a large scale screening tool. Conversely, simple risk-scoring questionnaires are useful and constitute a more cost-effective DM2 screening approach[10]. Hence, the international diabetes federation has recommended performing a blood test to detect possible diabetes when a high risk score has been obtained[11].
A number of predictive risk-scoring models for DM2 are currently available for clinical use[12-14]. But, most require specific blood test results and this limits its widespread use from a public health perspective. Meanwhile, the Finnish Diabetes Risk Score (FINDRISC)[15] has demonstrated to be a simple, fast, inexpensive, noninvasive, and reliable tool to identify individuals at high risk for DM2. Therefore, the FINDRISC has been internationally assessed in several countries, including Colombia[16-20]. However, in our country the ability of FINDRISC to predict the development of incident DM2 was not evaluated, and the specific risk scores for predicting undiagnosed and incident DM2 in our population were not established. Hence, this study aims to assess the performance of the FINDRISC score in detecting undiagnosed DM2 in the general population and to predict incident DM2 among prediabetics. Moreover, the study aims to establish the specific cutoff values for identifying increased risk of undiagnosed DM2 in the general population and incident DM2 among prediabetics in Colombia.
A longitudinal observational study has been conducted. Between June 1, 2012 and October 31, 2012 adult subjects (age ≥ 35 years) who attended the general practitioner for any reason at the ambulatory service of the Ophthalmological Foundation of Santander - FOSCAL in Floridablanca, Colombia, were involved in the screening. People with known diabetes mellitus (type 1 or 2) were not recruited. Any acute illness, pregnancy in women, and currently use of metformin or other glucose-modifying prescription drugs, were also considered as exclusion criteria.
The subjects were asked to complete a modified version of the FINDRISC score[15], which evaluates eight variables that are clearly correlated with the risk of DM2: Age, body mass index (BMI), waist circumference (WC), current antihypertensive medication, frequency of fruit and vegetable consumption, physical activity, personal history of high blood glucose, and family history of DM2. Variables are scored according to the risk that they may confer, resulting in a range of 0-26 total points. We adjusted the WC cutoffs points according to the previously described values that confer an increased cardio-metabolic risk in the Colombian population[21]. As a result, the WC has been scored as follows: Men: < 90 cm, the score is 0; 90-98 cm, the score is 3; > 98 cm, the score is 4. Women: < 80 cm, the score is 0; 80-88 cm, the score is 3; > 88 cm, the score is 4.
General practitioners performed the anthropometric measurements. Weight and height were measured with light clothing and no shoes with calibrated scales and a wall-mounted stadiometers, respectively, while participants were asked to stand erect with their head positioned in the Frankfort horizontal plane. WC was measured midway between the lowest rib and the iliac crest using an anthropometric tape. BMI was calculated by dividing body weight by the square of height [BMI = weight (kg)/height (m)2].
Laboratory tests (FPG, OGTT and HbA1C) were collected directly from the hospital’s database. Only those tests taken within the two months previous or after the survey were valid for the study. At least one of the tests: FPG, OGTT or HbA1c, should be present to consider the patient as suitable for the study. Classification of glucometabolic state was based on the American Diabetes Association (ADA) criteria[9]. The diagnosis of DM2 was established when FPG ≥ 126 mg/dL, OGTT ≥ 200 mg/dL and/or HbA1c ≥ 6.5%. Prediabetes was diagnosed by the presence of impaired FPG (≥ 100 mg/dL to < 126 mg/dL), impaired OGTT (≥ 140 mg/dL to < 200 mg/dL) and/or impaired HbA1c (≥ 5.7% to ≤ 6.4%).
Thereafter, the subsample of baseline prediabetic participants was followed for diabetes incidence in real life settings. The updated glucometabolic tests (FPG, OGTT and HbA1C) were also collected from the hospital’s database, and DM2 incident cases were diagnosed according to the ADA criteria[9].
The study protocol was in accordance with the Declaration of Helsinki, and the health research ethics board of the Ophthalmological Foundation of Santander - FOSCAL approved all study procedures. The subjects expressed their interest in participating in the study before they were included. As there were no interventions directly related to the study written informed consent was not required.
Descriptive statistics were computed for variables of interests, and included mean values and standard deviations of continuous variables and absolute and relative frequencies of categorical factors. Normality of distribution was checked for continuous variables using the Shapiro-Wilk test and by graphical methods. Wilconcox Rank Sum test was used to investigate the differences in continuous variables. Testing for differences in categorical variables was accomplished using the Pearson’s χ2 test.
Moreover, we used unconditional multivariate logistic regression models to assess the associations between the FINDRISC score and undiagnosed and incident DM2. These analyses were adjusted for potential confounders, such as gender and age. We re-coded the FINDRISC into tertiles and compared the risk of DM2 in each tertile with the lowest category of risk (reference group).
To assess performance of the FINDRISC score with respect to undiagnosed and incident DM2, receiver operating characteristics (ROC) curves, sensitivity, specificity, positive predictive values and negative predictive values (NPV) were calculated. The maximum values of the Youden’s index[22] were used as a criterion for selecting the optimum cut-off points. All statistical analysis was carried out by a biomedical statistician using Stata statistical software, release 12.0 (Stata Corporation, College Station, TX, United States). A P < 0.05 was considered statistically significant.
A total of 772 subjects were suitable for the study, of which 544 (70.47%) were women. The overall mean age was 58.34 ± 12.07 years, the overall prevalence of undiagnosed DM2 was 2.59% (95%CI: 1.46-3.71), and the prevalence of prediabetes was 24.09% (95%CI: 21.06-27.11) (Table 1). Baseline demographic, anthropometric and metabolic characteristics of the study population are presented in Table 1, and the prevalences of FINDRISC questionnaire components are presented in Table 2.
| Variables | Total (n = 772) | Men (n = 228) | Women (n = 544) |
| 1Age, yr | 58.34 (12.07) | 58.63 (12.67) | 58.22 (11.82) |
| 1BMI, kg/m2 | 27.36 (4.56) | 27.08 (4.56) | 27.47 (4.56) |
| 1Waist circumference, cm | 91.91 (10.47) | 97.05 (10.88) | 89.78 (9.52)2 |
| 1Fasting plasma glucose, mg/dL | 94.41 (12.60) | 96.17 (13.94) | 93.68 (11.94)2 |
| 1,4Oral glucose tolerance test, mg/dL | 107.89 (32.82) | 111.11 (36.55) | 106.23 (30.75) |
| 1,4HbA1c, % | 6.49 (1.27) | 6.39 (1.24) | 6.56 (1.31) |
| Undiagnosed DM2, n(%) | 20 (2.59) | 6 (2.63) | 14 (2.57) |
| Prediabetes, n(%) | 186 (24.09) | 61 (26.75) | 125 (22.98) |
| FINDRISC score | 11.84 (4.80) | 11.00 (4.71) | 12.18 (4.80)3 |
| Variables | Total (n = 772) | Men (n = 228) | Women (n = 544) |
| Age (yr) | |||
| < 45 | 106 (13.73) | 35 (15.35) | 71 (13.05) |
| 45-54 | 207 (26.81) | 59 (25.88) | 148 (27.21) |
| 55-64 | 203 (26.30) | 51 (22.37) | 152 (27.94) |
| > 64 | 256 (33.16) | 83 (36.40) | 173 (31.80) |
| BMI (kg/m2) | |||
| < 25 | 250 (32.38) | 75 (32.89) | 175 (32.17) |
| 25-30 | 331 (42.88) | 105 (46.05) | 226 (41.54) |
| > 30 | 191 (24.74) | 48 (21.05) | 143 (26.29) |
| WC (cm) | |||
| M: < 90; W: < 80 | 121 (15.67) | 50 (21.93) | 71 (13.05)2 |
| M: 90-98; W: 80-88 | 272 (35.23) | 84 (36.84) | 188 (34.56) |
| M: > 98; W: > 88 | 379 (49.09) | 94 (41.23) | 285 (52.39) |
| PA (30 min/d) | |||
| Yes | 362 (46.95) | 112 (49.12) | 250 (46.04) |
| No | 409 (53.05) | 116 (50.88) | 293 (53.96) |
| Vegetables - fruits | |||
| Daily | 433 (56.09) | 112 (49.12) | 321 (59.01)1 |
| No daily | 339 (43.91) | 116 (50.88) | 223 (40.99) |
| Hypertension | |||
| Without medication | 442 (57.25) | 136 (59.65) | 306 (56.25) |
| With medication | 330 (42.75) | 92 (40.35) | 238 (43.75) |
| Hyperglycemia antecedent | |||
| No | 634 (82.12) | 199 (87.28) | 435 (79.96)1 |
| Yes | 138 (17.88) | 29 (12.72) | 109 (20.04) |
| Familiar antecedents DM2 | |||
| No | 473 (61.27) | 147 (64.47) | 326 (59.93) |
| Grandparents | 72 (9.33) | 26 (11.40) | 46 (8.46) |
| Parents | 227 (29.40) | 55 (24.12) | 172 (31.62) |
The FINDRISC score was positively associated with undiagnosed DM2 (Table 3). The risk of DM2 increased with increasing tertiles of FINDRISC. Compared with participants in the lowest tertile, the risk of DM2 was 5.69 times higher for those in the highest tertile (OR = 5.69, 95%CI: 1.56-20.67).
| Undiagnosed DM2 (FPG≥126 mg/dL and/or OGTT≥200 mg/dL and/or HbA1c≥6.5%) | |||||||||
| 1st tertile | 2nd tertile | 3rd tertile | |||||||
| OR | 95%CI | P value | OR | 95%CI | P value | OR | 95%CI | P value | |
| Unadjusted | 1.0 | - | - | 2.45 | 0.60-9.92 | 0.207 | 5.69 | 1.56-20.67 | 0.008 |
| Gender-adjusted | 1.0 | - | - | 2.49 | 0.61-10.10 | 0.199 | 5.88 | 1.60-21.54 | 0.007 |
| Age- and gender-adjusted | 1.0 | - | - | 2.28 | 0.55-9.35 | 0.252 | 4.93 | 1.28-18.92 | 0.020 |
A total of 20 incident cases of DM2 (10.75%, 7 men and 13 women) among baseline prediabetic individuals occurred during 265.257 (82.234 men and 182.923 women) person-years of follow-up. The overall incidence of subsequent DM2 development was 7.5 (95%CI: 4.9-11.7) per 100 person-years. The incidence rates were slightly higher in men (8.5, 95%CI: 4.0-17.8, per 100 person-years) than in women (7.1, 95%CI: 4.1-12.2, per 100 person-years), but the difference was not statistically significant. Likewise, compared with participants in the lowest tertile of FINDRISC score at baseline, the risk of incident DM2 was 5.31 times higher for those in the highest tertile (HR = 5.31, 95%CI: 1.15-24.43) (Table 4).
| Incident DM2 (FPG≥126 mg/dL and/or OGTT≥200 mg/dL and/or HbA1c≥6.5%) | |||||||||
| 1st tertile | 2nd tertile | 3rd tertile | |||||||
| HR | 95%CI | P value | HR | 95%CI | P value | HR | 95%CI | P value | |
| Unadjusted | 1.00 | - | - | 3.67 | 0.77-17.36 | 0.101 | 5.31 | 1.15-24.43 | 0.032 |
| Gender-adjusted | 1.00 | - | - | 3.93 | 0.81-18.89 | 0.087 | 5.75 | 1.22-27.03 | 0.027 |
| Age- and gender-adjusted | 1.00 | - | - | 3.52 | 0.70-17.53 | 0.124 | 4.81 | 0.93-24.85 | 0.061 |
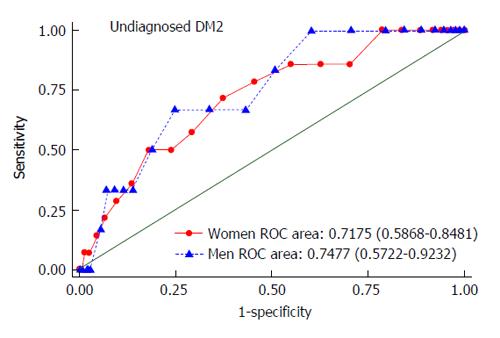
The area under ROC curve (AUROC) of the FINDRISC score for detecting undiagnosed DM2 in both men and women was 0.7477 (95%CI: 0.5722-0.9232) and 0.7175 (95%CI: 0.5868-0.8481), respectively (Figure 1). The performance assessment of the FINDRISC score for identifying individuals at risk of undiagnosed DM2 is shown in Table 5. At the cutoff value of 14 in both men (sensitivity = 66.7%; NPV = 98.8%) and women (sensitivity = 71.4%; NPV = 98.8%), the Youden’s index was the highest for undiagnosed DM2 (0.419 in men and 0.340 in women).
| FINDRISC score | Undiagnosed DM2 (FPG≥126 mg/dL and/or OGTT≥200 mg/dL and/or HbA1c≥6.5%) | ||||
| Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Youden's index | |
| Cutoff value ≥ 11 | |||||
| Men | 83.3 | 49.1 | 4.2 | 99.1 | 0.324 |
| Women | 85.7 | 37.2 | 3.5 | 99.0 | 0.228 |
| Cutoff value ≥ 12 | |||||
| Men | 66.7 | 56.8 | 4.0 | 98.4 | 0.234 |
| Women | 85.7 | 44.9 | 3.9 | 99.2 | 0.306 |
| Cutoff value ≥ 13 | |||||
| Men | 66.7 | 66.2 | 5.1 | 98.7 | 0.328 |
| Women | 78.6 | 54.3 | 4.4 | 99.0 | 0.329 |
| Cutoff value ≥ 14 | |||||
| Men | 66.7 | 75.2 | 6.8 | 98.8 | 0.419 |
| Women | 71.4 | 62.6 | 4.8 | 98.8 | 0.340 |
| Cutoff value ≥ 15 | |||||
| Men | 50.0 | 81.1 | 6.7 | 98.4 | 0.310 |
| Women | 57.1 | 70.7 | 4.9 | 98.4 | 0.278 |
| Cutoff value ≥ 16 | |||||
| Men | 33.3 | 86.0 | 6.1 | 98.0 | 0.193 |
| Women | 50.0 | 76.2 | 5.3 | 98.3 | 0.262 |
| Cutoff value ≥ 17 | |||||
| Men | 33.3 | 88.3 | 7.1 | 98.0 | 0.216 |
| Women | 50.0 | 81.9 | 6.8 | 98.4 | 0.318 |
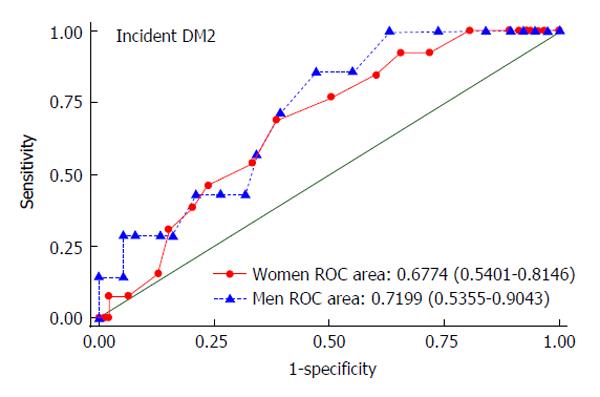
The ROC curve for the incidence of DM2 among the prediabetic subsample by FINDRISC is shown in Figure 2. The AUROC curve were 0.7199 (95%CI: 0.5355-0.9043) in men and 0.6774 (95%CI: 0.5401-0.8146) in women. Given a Youden’s index of 0.383 in men and 0.305 in women, FINDRISC cutoff values for incident DM2 were calculated to be 13 (sensitivity = 85.7%; NPV = 95.2%) in men and 16 (sensitivity = 69.2%; NPV = 93.4%) in women (Table 6).
| FINDRISC score | Incident DM2 (FPG≥126 mg/dL and/or OGTT≥200 mg/dL and/or HbA1c≥6.5%) | ||||
| Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Youden's index | |
| Cutoff value ≥ 12 | |||||
| Men | 85.7 | 44.7 | 22.2 | 94.4 | 0.304 |
| Women | 92.3 | 28.0 | 15.2 | 96.3 | 0.202 |
| Cutoff value ≥ 13 | |||||
| Men | 85.7 | 52.6 | 25.0 | 95.2 | 0.383 |
| Women | 92.3 | 34.4 | 16.4 | 97.0 | 0.267 |
| Cutoff value ≥ 14 | |||||
| Men | 71.4 | 60.5 | 25.0 | 92.0 | 0.319 |
| Women | 84.6 | 39.8 | 16.4 | 94.9 | 0.244 |
| Cutoff value ≥ 15 | |||||
| Men | 57.1 | 65.8 | 23.5 | 89.3 | 0.229 |
| Women | 76.9 | 49.5 | 17.5 | 93.9 | 0.263 |
| Cutoff value ≥ 16 | |||||
| Men | 42.9 | 68.4 | 20.0 | 86.7 | 0.112 |
| Women | 69.2 | 61.3 | 20.0 | 93.4 | 0.305 |
| Cutoff value ≥ 17 | |||||
| Men | 42.9 | 73.7 | 23.0 | 87.5 | 0.165 |
| Women | 53.9 | 66.7 | 18.4 | 91.2 | 0.205 |
| Cutoff value ≥ 18 | |||||
| Men | 42.9 | 79.0 | 27.3 | 88.2 | 0.218 |
| Women | 46.2 | 76.3 | 21.4 | 91.0 | 0.224 |
The current study assessed the performance of the FINDRISC questionnaire, and demonstrates that this can work reasonably well as screening tool, detecting cross-sectionally undiagnosed DM2 in the general population, and longitudinally incident DM2 among individuals with prediabetes in Colombia. Our findings show a positive association between the FINDRISC score, undiagnosed DM2 in the general population and incident DM2 among prediabetic subjects. Moreover, in our population the FINDRISC score had an AUROC of 0.7477 in men and 0.7175 in women for identifying individuals at increased risk of undiagnosed DM2, and an AUROC of 0.7199 in men and 0.6774 in women for predicting incident DM2, which is comparable to that obtained in other similar studies[18,19]. Using an optimal cutoff value of greater or equal to 14 both in men and women, this screening tool had good performance in identifying undiagnosed DM2. Meanwhile, FINDRISC cutoff values of 13 in men and 16 in women at baseline were defined as optimal to predict DM2 in the subsample of prediabetic individuals.
DM2 is a strong, independent risk factor for cardiovascular disease and death[3,23], and many epidemiologic analyses have identified a progressive relationship between prediabetes and these outcomes[24,25]. Therefore, identifying individuals with undiagnosed DM2 and detecting individuals at risk for developing DM2 is essential to lead this target population the preventive actions, minimizing human and economic costs of diabetic complications[9,11]. However, there are almost no symptoms of prediabetes or DM2 and as a consequence its detection is often delayed and at the time of diagnosis advanced complications are frequently present. It is estimated that approximately one-third of all people with DM2 may be undiagnosed[1,2]. Hence, using a simple and valid questionnaire, such as the FINDRISC score, as a preliminary screening method followed with more invasive and accurate diagnosis in primary care constitute a cost-effective and practical method with a potentially high national impact in terms of public health.
The efficiency of risk scores may vary between populations, and therefore, these should be validated in each population before use. The FINDRISC questionnaire was developed in Finland[15], and has been validated in other populations studied so far[16-20], demonstrating to be a simple and inexpensive tool that can identify those at high risk of having abnormalities in the glucose metabolism. Certainly, a previous study had already demonstrated the usefulness of FINDRISC to identify cross-sectionally people with glucose metabolism disorders in Colombia[20], however in that study the cutoff level to identify patients at risk was chosen arbitrary. In that study, people with 13 or more FINDRISC points were screened with an OGTT, and they found this cutoff point to be useful to identify people with glucose metabolism disorders. Furthermore, this previous study did not assess the ability of FINDRISC in predicting incident DM2.
It is worth highlighting that there are several methodological differences between the several studies that have validated the FINDRISC questionnaire; modified or shortened versions have been used, in some studies the plasmatic glycemia tests have been only performed in subjects with a particular score, and in other cases prediabetes or metabolic syndrome has been also considered as an outcome. Therefore, the present study should be compared to these previously conducted studies with caution.
An important aspect of the present study is the assessment of the capacity of FINDRISC in the short-term prediction of incident DM2 among prediabetic individuals. To our knowledge, this is the first longitudinal validation of the FINDRISC questionnaire conducted in Latin America, while just other one study of similar characteristics was conducted in Europe. Soriguer et al[19] reported that in Spanish subjects with prediabetes the best prediction of risk of incident DM2 was found in those subjects with a FINDRISC score ≥ 9. It should be noted that gender-specific cutoff values were not estimated in this previous study making thus comparisons difficult with our study.
There are some study limitations that warrant consideration. First, the participants were drawn from a unique healthcare center in Floridablanca and, thus, the results may not be applicable to the rest of Colombia. Second, the number of participants was relatively small, although the results were similar to those of other studies in different populations and larger cohorts[16-20], which supports the validity of our present findings. Third, in most cases the diagnosis of DM2 was based on only one plasmatic value (FPG, OGTT or HbA1c when performed), not two as recommended, and this may cause a serious bias in respect the definition of the main outcome event underestimating the true prevalence/incidence of DM2. However, diagnosis based on a single test result is accepted in epidemiological studies. In addition, it is noteworthy that this study was conducted in real life conditions, and the Colombian health system restricts the use of OGTT and HbA1c in patients with FPG < 100 mg/dL. Fourth, in our study most respondents were women, and this situation may partly be explained by behavioral habits, women are more likely to participate in completing questionnaires, they visit their physician more often, and usually pay more attention to their health.
This study has also considerable strengths. First, the diagnosis of DM2 was not self-reported. Second, all the analyzed subjects have available at least one plasmatic glycemia value. Third, the age distribution of the participants was wide and included the vast majority of the high-risk population.
In conclusion, the present study has assessed the performance of the FINDRISC questionnaire, demonstrating this as a useful screening tool to identify unknown DM2 in a cross-section of the Colombian population, and to predict incident DM2 among prediabetics. Moreover, we have demonstrated that a cutoff value greater or equal to 14 is the more appropriate detecting any previously undiagnosed DM2 in our population, both in men and women. Meanwhile, the optimal cutoff values for predict incident DM2 in prediabetic individuals were 13 in men and 16 in women.
The prevalence of type 2 diabetes mellitus (DM2) is increasing worldwide, but unfortunately its diagnosis is being made, in a high percentage, when vascular complications are already present. Therefore, the International Diabetes Federation recommends identification of persons at risk using simple risk-scoring questionnaires. Thus, this study aims to assess the performance of the Finnish Diabetes Risk Score (FINDRISC) questionnaire for detecting and predicting DM2 in a Colombian population.
The FINDRISC questionnaire has been assessed and validated in several countries, including Colombia. However, in the authors’ country the ability of FINDRISC to predict the development of incident DM2 has not been evaluated.
In the present study, the FINDRISC score demonstrated to be a useful screening tool to identify unknown DM2 in a cross-section of the Colombian population, and to predict incident DM2 among prediabetics.
Using a simple and valid questionnaire, such as the FINDRISC score, as a preliminary screening method for DM2 followed by more invasive and accurate diagnostic exams in primary care, constitute a cost-effective and practical method with a potentially high national impact in terms of public health.
The FINDRISC is a simple, fast, inexpensive, noninvasive, and reliable tool to identify individuals at high risk for DM2.
This is a good observational study. Authors assessed the performance of the FINDRISC questionnaire in a Colombian population to detect undiagnosed DM2 in the general population and to predict incident DM2 among prediabetics. The study design is clear and the results are well to present. It demonstrated that the modified FINDRISC questionnaire is a good tool to detect undiagnosed DM2 and to predict incident DM2 in Colombian population.
P- Reviewer: Hssan M, Zhao JB S- Editor: Kong JX L- Editor: A E- Editor: Liu SQ
| 1. | Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87:4-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4438] [Cited by in RCA: 4379] [Article Influence: 291.9] [Reference Citation Analysis (4)] |
| 2. | López-Jaramillo P, Gómez-Arbeláez D, Camacho PA, Álvarez J, Molina DI, Sánchez G, Arcos E, Narváez C, García H, Pérez M. Prevalencia, conocimiento, tratamiento y control de la diabetes mellitus tipo 2 en Colombia. Resultados del estudio PURE-Colombia. Rev Colomb Cardiol. 2013;20:16. |
| 3. | Narayan KM, Gregg EW, Fagot-Campagna A, Engelgau MM, Vinicor F. Diabetes--a common, growing, serious, costly, and potentially preventable public health problem. Diabetes Res Clin Pract. 2000;50 Suppl 2:S77-S84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 166] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 4. | Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346:393-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13206] [Cited by in RCA: 12431] [Article Influence: 540.5] [Reference Citation Analysis (1)] |
| 5. | Tuomilehto J, Lindström J, Eriksson JG, Valle TT, Hämäläinen H, Ilanne-Parikka P, Keinänen-Kiukaanniemi S, Laakso M, Louheranta A, Rastas M. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med. 2001;344:1343-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7148] [Cited by in RCA: 6609] [Article Influence: 275.4] [Reference Citation Analysis (0)] |
| 6. | Harris MI, Klein R, Welborn TA, Knuiman MW. Onset of NIDDM occurs at least 4-7 yr before clinical diagnosis. Diabetes Care. 1992;15:815-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 856] [Cited by in RCA: 809] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 7. | López-Jaramillo P, Velandia-Carrillo C, Gómez-Arbeláez D, Aldana-Campos M. Is the present cut-point to define type 2 diabetes appropriate in Latin-Americans? World J Diabetes. 2014;5:747-755. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 8. | López-Jaramillo P, Gómez-Arbeláez D, López-López J, López-López C, Martínez-Ortega J, Gómez-Rodríguez A, Triana-Cubillos S. The role of leptin/adiponectin ratio in metabolic syndrome and diabetes. Horm Mol Biol Clin Investig. 2014;18:37-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 198] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 9. | American Diabetes Association. Standards of medical care in diabetes--2014. Diabetes Care. 2014;37 Suppl 1:S14-S80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2830] [Cited by in RCA: 3016] [Article Influence: 274.2] [Reference Citation Analysis (0)] |
| 10. | Schwarz PE, Li J, Lindstrom J, Tuomilehto J. Tools for predicting the risk of type 2 diabetes in daily practice. Horm Metab Res. 2009;41:86-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 139] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 11. | International Diabetes Federation Guideline Development Group. Global guideline for type 2 diabetes. Diabetes Res Clin Pract. 2014;104:1-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 347] [Article Influence: 31.5] [Reference Citation Analysis (1)] |
| 12. | Buijsse B, Simmons RK, Griffin SJ, Schulze MB. Risk assessment tools for identifying individuals at risk of developing type 2 diabetes. Epidemiol Rev. 2011;33:46-62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 222] [Cited by in RCA: 204] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 13. | Griffin SJ, Little PS, Hales CN, Kinmonth AL, Wareham NJ. Diabetes risk score: towards earlier detection of type 2 diabetes in general practice. Diabetes Metab Res Rev. 2000;16:164-171. [PubMed] |
| 14. | Heikes KE, Eddy DM, Arondekar B, Schlessinger L. Diabetes Risk Calculator: a simple tool for detecting undiagnosed diabetes and pre-diabetes. Diabetes Care. 2008;31:1040-1045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 154] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 15. | Lindström J, Tuomilehto J. The diabetes risk score: a practical tool to predict type 2 diabetes risk. Diabetes Care. 2003;26:725-731. [PubMed] |
| 16. | Franciosi M, De Berardis G, Rossi MC, Sacco M, Belfiglio M, Pellegrini F, Tognoni G, Valentini M, Nicolucci A. Use of the diabetes risk score for opportunistic screening of undiagnosed diabetes and impaired glucose tolerance: the IGLOO (Impaired Glucose Tolerance and Long-Term Outcomes Observational) study. Diabetes Care. 2005;28:1187-1194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1160] [Cited by in RCA: 1235] [Article Influence: 56.1] [Reference Citation Analysis (0)] |
| 17. | Makrilakis K, Liatis S, Grammatikou S, Perrea D, Stathi C, Tsiligros P, Katsilambros N. Validation of the Finnish diabetes risk score (FINDRISC) questionnaire for screening for undiagnosed type 2 diabetes, dysglycaemia and the metabolic syndrome in Greece. Diabetes Metab. 2011;37:144-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 121] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 18. | Saaristo T, Peltonen M, Lindström J, Saarikoski L, Sundvall J, Eriksson JG, Tuomilehto J. Cross-sectional evaluation of the Finnish Diabetes Risk Score: a tool to identify undetected type 2 diabetes, abnormal glucose tolerance and metabolic syndrome. Diab Vasc Dis Res. 2005;2:67-72. [PubMed] |
| 19. | Soriguer F, Valdés S, Tapia MJ, Esteva I, Ruiz de Adana MS, Almaraz MC, Morcillo S, García Fuentes E, Rodríguez F, Rojo-Martinez G. [Validation of the FINDRISC (FINnish Diabetes RIsk SCore) for prediction of the risk of type 2 diabetes in a population of southern Spain. Pizarra Study]. Med Clin (Barc). 2012;138:371-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 54] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 20. | Barengo NC, Acosta T, Arrieta A, Ricaurte C, Mayor D, Tuomilehto JO; the DEMOJUAN study group. Screening for people with glucose metabolism disorders within the framework of the DEMOJUAN project (DEMOnstration area for primary prevention of type 2 diabetes, JUAN Mina and Barranquilla, Colombia). Diabetes Metab Res Rev. 2013;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 21. | Perez M, Casas JP, Cubillos-Garzón LA, Serrano NC, Silva F, Morillo CA, López-Jaramillo P. Using waist circumference as a screening tool to identify Colombian subjects at cardiovascular risk. Eur J Cardiovasc Prev Rehabil. 2003;10:328-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 23. | O’Donnell MJ, Xavier D, Liu L, Zhang H, Chin SL, Rao-Melacini P, Rangarajan S, Islam S, Pais P, McQueen MJ. Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): a case-control study. Lancet. 2010;376:112-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2026] [Cited by in RCA: 2179] [Article Influence: 145.3] [Reference Citation Analysis (0)] |
| 24. | Selvin E, Steffes MW, Zhu H, Matsushita K, Wagenknecht L, Pankow J, Coresh J, Brancati FL. Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults. N Engl J Med. 2010;362:800-811. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1161] [Cited by in RCA: 1093] [Article Influence: 72.9] [Reference Citation Analysis (0)] |
| 25. | Gerstein HC, Swedberg K, Carlsson J, McMurray JJ, Michelson EL, Olofsson B, Pfeffer MA, Yusuf S. The hemoglobin A1c level as a progressive risk factor for cardiovascular death, hospitalization for heart failure, or death in patients with chronic heart failure: an analysis of the Candesartan in Heart failure: Assessment of Reduction in Mortality and Morbidity (CHARM) program. Arch Intern Med. 2008;168:1699-1704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 176] [Article Influence: 10.4] [Reference Citation Analysis (0)] |