Peer-review started: September 23, 2014
First decision: November 13, 2014
Revised: November 27, 2014
Accepted: December 3, 2014
Article in press: December 10, 2014
Published online: February 15, 2015
Processing time: 161 Days and 20 Hours
Despite improvements in glucose, lipids and blood pressure control, vascular complications remain the most important cause of morbidity and mortality in patients with type 1 diabetes. For that reason, there is a need to identify additional risk factors to utilize in clinical practice or translate to novel therapies to prevent vascular complications. Reduced insulin sensitivity is an increasingly recognized component of type 1 diabetes that has been linked with the development and progression of both micro- and macrovascular complications. Adolescents and adults with type 1 diabetes have reduced insulin sensitivity, even when compared to their non-diabetic counterparts of similar adiposity, serum triglycerides, high-density lipoprotein cholesterol, level of habitual physical activity, and in adolescents, pubertal stage. Reduced insulin sensitivity is thought to contribute both to the initiation and progression of macro- and microvascular complications in type 1 diabetes. There are currently clinical trials underway examining the benefits of improving insulin sensitivity with regards to vascular complications in type 1 diabetes. Reduced insulin sensitivity is an increasingly recognized component of type 1 diabetes, is implicated in the pathogenesis of vascular complications and is potentially an important therapeutic target to prevent vascular complications. In this review, we will focus on the pathophysiologic contribution of insulin sensitivity to vascular complications and summarize related ongoing clinical trials.
Core tip: Adolescents and adults with type 1 diabetes have reduced insulin sensitivity compared to their non-diabetic counterparts. Reduced insulin sensitivity is implicated in the development and progression of micro and macrovascular complications in type 1 diabetes. Clinical trials are underway investigating insulin sensitivity as a therapeutic target to prevent vascular complications in type 1 diabetes. Methods are needed to identify which patients with type 1 diabetes would benefit from treatment of insulin resistance and translation of this to clinical practice.
- Citation: Bjornstad P, Snell-Bergeon JK, Nadeau KJ, Maahs DM. Insulin sensitivity and complications in type 1 diabetes: New insights. World J Diabetes 2015; 6(1): 8-16
- URL: https://www.wjgnet.com/1948-9358/full/v6/i1/8.htm
- DOI: https://dx.doi.org/10.4239/wjd.v6.i1.8
The public health burden of type 1 diabetes (T1D), a disease affecting approximately 1.4 million people in the United States and 30 million globally[1], is progressively increasing, largely due to the prevalence of the associated macro- and microvascular complications[1-3]. Macrovascular disease in the form of coronary artery disease (CAD) is the major cause of morbidity and mortality in patients with T1D[3-7]. Annually, up to 1%-2% of young adults (28-38 years of age) with T1D develop CAD[3-5,8,9]. By their mid-forties, over 70% of men and 50% of women with T1D develop measureable coronary artery calcification (CAC) by computed tomography (CT) scan[3-5,10,11] - a marker of significant atherosclerotic plaque burden. In addition to macrovascular disease, microvascular disease continues to cause morbidity and mortality; for example, diabetic nephropathy remains the leading cause of end-stage renal disease in the United States[12-14], and diabetic retinopathy, another form of microvascular disease, is the single most common cause of new-onset blindness[15].
Despite significant improvements in conventional risk factors (e.g., blood pressure, glucose and lipid control) during the past two decades, vascular complications remain a major concern in T1D[14,16-19]. There is a need for improved methods to identify people at risk for, and prevent the development and progression of, these complications.
Adolescents and adults with T1D demonstrate reduced insulin sensitivity, even when compared to non-diabetic counterparts of similar adiposity, body fat distribution, serum triglycerides, high-density lipoprotein cholesterol, level of habitual physical activity, and in adolescents, pubertal stage[17,20-25]. In addition to the insulin resistance documented historically in adolescents with very poor glycemic control[22], more recently significant insulin resistance has been documented in adolescents and adults with T1D, despite modern advances in technology and better glycemic control[20]. Increasing rates of obesity in T1D most likely also contribute to impaired insulin sensitivity[26]. Moreover, a subset of participants in The Diabetes Control and Complications Trial (DCCT) in the intensive treatment arm experienced greater weight gain and worse CVD profiles, which may suggest that lowering HbA1c is not without potential negative effects[27].
The role of insulin sensitivity in the development and progression of macro-[28-30] and microvascular com-plications[31] in T1D is increasingly recognized. Prospective studies are needed to test the hypothesis that reduced insulin sensitivity is a unifying risk factor for the development of both micro- and macrovascular complications. However, there is increasing evidence implicating reduced insulin sensitivity in the path-ogenesis of vascular complications in T1D[26,32]. It is therefore important to better understand the role of insulin sensitivity in the development of micro- and macrovascular complications in T1D to enable us to intervene early in the pathophysiologic course to halt or slow progression. Accordingly, in this review, we examine the current evidence addressing insulin sensitivity and vascular complications in T1D.
Historically, when glycemic control was poorer, reduced insulin sensitivity in people with T1D was thought to be solely related to adiposity and high HbA1c[22,33], but recent data have challenged this assumption and suggest that reduced insulin sensitivity cannot simply be explained by excess weight or poor glycemic control[20,21]. In fact, insulin resistance in multiple tissues has recently been documented in T1D subjects with glycemic control much improved from the pre-DCCT era (7.5% ± 0.9% and 8.6% ± 1.6% in adults and adolescents) and with BMI similar to that of non-diabetic individuals[20,21]. These data suggest that resistance to insulin’s action on glucose utilization, hepatic glucose release and non-esterified fatty acid suppression is also mediated by factors beyond prevailing adiposity or glycemia[20,21].
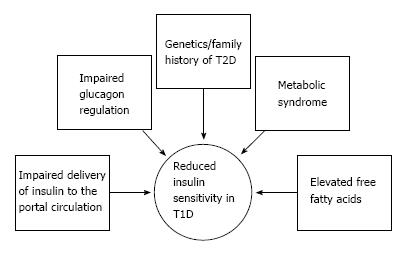
The exact mechanism of reduced insulin sensitivity in T1D is poorly understood. Various hypotheses exist to explain reduced insulin sensitivity in T1D[34,35], including prolonged peripheral exposure to supraphysiologic levels of exogenous insulin, weight gain in part caused by intensive insulin therapy, and genetic and environmental factors that contribute to type 2 diabetes (T2D)[26,34,35] (Figure 1). Another proposed mechanism includes lack of delivery of insulin to the portal circulation, causing reduced insulin delivery to the liver, and subsequent lower hepatic IGF-1 production, and lower feedback inhibition to the pituitary leading to higher growth hormone levels, which are known to induce insulin resistance[36] (Figure 1). Abnormal glucagon regulation leading to increased hepatic glucose output has also been implicated in insulin resistance[36]. Another possible mechanistic pathway linking reduced insulin sensitivity to vascular complications in T1D is via insulin’s effects on overall non-essential fatty acid (NEFA) exposure and lipotoxicity in development of macro- and microangiopathy[20,34,35,37]. Finally, ectopically accumulated fat and its catabolites have been proposed to induce insulin resistance via the effects of various signaling pathways including MAPK, protein kinase C, IkB kinases, S6 kinases and ER stress on GLUT4, although the role of ectopic fat remains controversial[38-40] (Figure 1).
There are also robust data demonstrating that subjects with T1D with greater degrees of insulin resistance and/or family history of T2D are at greater risk of micro- and macrovascular complications[41-44] (Figure 1). Furthermore, a high prevalence of metabolic syndrome (38% in men and 40% in women) has been reported in adults with T1D[45]. A close relation between serum uric acid (SUA), insulin sensitivity and metabolic syndrome in non-diabetic subjects has been shown[46]. Conversely, in adolescents and adults with T1D we demonstrated very weak associations between SUA and estimated insulin sensitivity[47].
Although insulin resistance is recognized as an important component of vascular complications in T1D, none of the guidelines make specific recommendations about when or how to test for insulin resistance. The insulinopenic nature of T1D prohibits the use of the IV glucose tolerance test (IVGTT) and oral glucose tolerance test (OGTT)-based methods to estimate insulin sensitivity. The gold standard measurement of insulin sensitivity in T1D, glucose disposal rate (GDR) from a hyperinsulinemic-euglycemic clamp, is too cumbersome for use in routine clinical care, but newer insulin sensitivity estimation equations (estimated GDR, eGDR), demonstrate strong agreement with measured insulin sensitivity and offer promise in the clinical setting[11]. Insulin sensitivity prediction equations from the SEARCH Study (eIS-SEARCH)[11], the Pittsburgh Epidemiology of Diabetes Complications Study (eIS-EDC)[48], and the Coronary Artery Calcification in Type 1 diabetes study (eIS-CACTI)[20,31,47] are available, with others currently under development. The eIS-CACTI model includes waist circumference, daily insulin dose per kg body weight, triglycerides and DBP: exp(4.1075 - 0.01299*waist(cm) - 1.05819 *insulin dose [daily units per kg) - 0.00354*triglycerides (mg/dL) - 0.00802*DBP (mm Hg)]. The eIS-CACTI explains 63% of the variance in the GDR in hyperinsulinemic-euglycemic clamp studies, and has been validated in adolescent and adult cohorts with and without T1D[20,31,47]. There remains a need for a commonly accepted measure to estimate insulin sensitivity to be used in the clinical setting to identify patients who would benefit from therapies to improve insulin sensitivity.
The association between insulin sensitivity and micro-vascular complications is increasingly recognized, but it is not a recent discovery. In 1968, Martin et al[49] showed that microvascular complications were associated with insulin resistance in long-standing T1D subjects. In 1993, Yip et al[50] explored insulin resistance as an underlying factor in T1D and found reduced insulin sensitivity, measured by GDR, in a small group with microalbuminuria, while Orchard et al[48] later demonstrated that eGDR predicted overt nephropathy in T1D subjects in the EDC cohort. Finally, we and others have reported that higher estimated insulin sensitivity at baseline predicts lower odds of developing diabetic retinopathy, proliferative diabetic retinopathy and diabetic neuropathy independent of other established risk factors[32,48,51]. We have also previously demonstrated that reduced estimated insulin sensitivity predicted incident microalbuminuria and rapid glomerular filtration decline by cystatin C over 6 years[31] and that increased insulin sensitivity predicted regression of albuminuria in adults with T1D[32], similar to data in the EDC study[48,52].
Mechanisms underlying insulin sensitivity and renal pathology remain unclear, but the association between reduced insulin sensitivity and hemodynamic changes in the kidney is increasingly recognized[13]. A growing body of data demonstrates that insulin resistance is associated with an elevation of glomerular hydrostatic pressure causing increased renal vascular permeability and ultimately glomerular hyperfiltration[13,53-60]. Another possible mechanistic pathway linking insulin resistance to diabetic nephropathy is via effects on overall non-esterified fatty acid exposure and lipotoxicity, leading to the development of angiopathy[36].
Markers of reduced insulin sensitivity in adults with T1D have also been linked with increased risk of diabetic retinopathy and proliferative diabetic retinopathy in the EURODIAB Prospective Complications Study[61]. However, the investigators in EURODIAB included surrogates for insulin sensitivity including triglycerides and waist-to-hip ratio rather than measured or validated estimates of insulin sensitivity[61]. In the DCCT/EDIC study, higher estimated insulin sensitivity using the Pittsburgh eGDR equation was associated with a lower risk for retinopathy in both the conventionally treated and intensively treated groups, though not independent of HbA1c[30]. Some studies in adults with T2D demonstrate an association between reduced measured insulin sensitivity and diabetic retinopathy[62,63], but there is a need for more data in T1D. There are also limited data on insulin sensitivity and diabetic neuropathy in T1D, with only a single small study showing an association between estimated insulin sensitivity and diabetic neuropathy in adults with T1D[64]. In contrast, the association between reduced insulin sensitivity and diabetic neuropathy is well recognized in adults with T2D[65-67].
In the general population, insulin resistance has been implicated as an important contributor to accelerated atherosclerosis[68,69]. In the CACTI study, a longitudinal cohort study of adults with T1D designed to investigate the determinants of early and accelerated atherosclerosis in T1D, insulin sensitivity independently predicted CAC[20,70]. Data from EDC also demonstrated strong associations between insulin sensitivity and coronary artery disease in adults with T1D[30]. In the DCCT, excess weight gain was associated with insulin resistance, as well as more extensive atherosclerosis during EDIC[43]. Moreover, we have also previously shown associations between insulin resistance and cardiac and exercise dysfunction in adolescents with T1D[21]. Renal health, which is known to be associated with better insulin sensitivity, is also strongly associated with higher peak exercise capacity in adolescents with T1D[52]. Finally, we have shown that higher estimated insulin sensitivity in adolescents with T1D is inversely associated with CVD risk factors[44].
Modification of insulin sensitivity therefore holds promise as a therapeutic target to reduce vascular complications in T1D, since both life style changes (diet and exercise) and drugs such as metformin can improve insulin sensitivity in other populations. Metformin is an inexpensive and well tolerated medication. The most common adverse effects associated with metformin are gastrointestinal, with anemia due to vitamin B12 malabsorption and lactic acidosis being rare events when used in T2D[71]. In T2D, metformin is widely considered to protect against cardiovascular complications[71,72]. In contrast, metformin is not advocated in any major national or international guidelines for the management of T1D[72]. In a recent meta-analysis of randomized trials, Vella et al[72] found that metformin therapy in T1D was associated with reduced insulin-dose requirements but no clear evidence for an improvement in glycemic control was demonstrated[72]. Moreover, Nadeau et al[73] recently showed that low-dose metformin decreased total daily insulin doses in adolescents with T1D, likely representing improvement in insulin sensitivity[73]. Metformin is also associated with reduced LDL cholesterol[74], precursors of advanced glycation end production[75,76] and a decrease in blood pressure[77,78] in adults with T1D and T2D. However, there are currently insufficient data on vascular outcomes and their surrogates to routinely recommend metformin therapy in adolescents and adults with T1D for improving cardiovascular outcomes. For that reason, there is a need for well-designed randomized clinical trials of sufficient size and duration to show clinical benefit of metformin. Another important consideration is the significant variation observed in adults with T2D in response to metformin[71]. The inter-individual variation in metformin may also apply to patients with T1D. Genetic variation may be one of the important determinants of this inter-individual variation, and improved understanding of underlying genes and pathways has the potential to improve the effect of metformin on insulin sensitivity[71].
The BARI-2D study showed no benefit of an insulin sensitizing strategy compared to insulin provision on diabetic nephropathy in older adults with coronary artery disease T2D[79]. In contrast to most contemporary cohorts with T1D, the BARI-2D trial included older adults with T2D, with most participants already having both hypertension and hyperlipidemia. It is plausible that the vascular injuries in older adults with T2D and longstanding pathology may not be responsive to changes in insulin sensitivity. A hypothesis is that early intervention prior to establishment of vascular lesions may result in significant delay of clinical pathology as suggested by the concept of “metabolic memory” in the DCCT-EDIC study[80-84]. Also, clinical cardiovascular disease typically does not manifest until older ages; for example, it took 17 years of follow-up for the benefits of intensive management to manifest in DCCT[27]. Improvements in outcomes due to adjunctive therapy in the era of intensive management may be more subtle. Furthermore, long-term studies in children are lacking[85].
The REducing With MetfOrmin Vascular Adverse Lesions in Type 1 Diabetes (REMOVAL, NCT01483560) study is an ongoing double-blind randomized clinical trial with metformin to improve insulin sensitivity in adults with T1D in an attempt to prevent vascular complications, and the Effects of Metformin on Cardiovascular Function in Adolescents With Type 1 Diabetes (EMERALD, NCT01808690) is an ongoing double-blind randomized clinical trial with metformin to evaluate if metformin will improve tissue-specific insulin resistance in T1D adolescents using the hyperinsulinemic-euglycemic clamp technique, as well as improve vascular, cardiac, exercise and muscle mitochondrial function (Table 1). The effect of Metformin on Vascular and Mitochondrial Function in Type 1 Diabetes (MeT1, NCT01813929) study is also underway which seeks to measure the effect of improving insulin sensitivity on vascular function and compliance, and mitochondrial function in adults with T1D. Metformin Therapy for Overweight Adolescents with Type 1 Diabetes (NCT01881828) being performed in the T1D Exchange clinic network seeks to evaluate the efficacy and safety of the use of metformin in addition to standard insulin therapy in overweight and obese children and adolescents, age 12 - < 20 years, with type 1 diabetes for at least 1 year. Furthermore, the insulin Clamp Ancillary Study for Assessment of Insulin Resistance (NCT02045290) is an associated study underway to evaluate if metformin will improve tissue-specific insulin resistance in obese T1D adolescents using hyperinsulinemic-euglycemic clamp technique (Table 1). Smaller metformin trials are also underway. Additional studies are also needed to assess the impact of other drugs that influence insulin sensitivity in T2D, including glucagon-like peptide-1 analogues, dipeptidyl peptidase-4 inhibitor, sodium glucose co-transporter 2 inhibitors, thiazolidinediones and bromocriptine, which may have similarly beneficial effects in T1D.
| Clinical trial name(s) | Description |
| Metformin Vascular Adverse Lesions in Type 1 Diabetes (REMOVAL) trial (NCT01483560) | Double-blind RCT with metformin to improve insulin sensitivity in subjects with T1D in an attempt to prevent vascular complications |
| Effects of Metformin On Cardiovascular Function In Adolescents with Type 1 Diabetes (EMERALD) study (NCT01808690) | Double-blind RCT with metformin to evaluate if metformin will improve tissue-specific insulin resistance in T1D adolescents using the hyperinsulinemic-euglycemic clamp technique, as well as improve vascular, cardiac, exercise and muscle mitochondrial function |
| Insulin Clamp Ancillary Study for Assessment of Insulin Resistance (NCT02045290) | Assess if metformin will improve tissue-specific insulin resistance in type 1 diabetes using hyperinsulinemic euglycemic clamps |
| Metformin Therapy for Overweight Adolescents With Type 1 Diabetes (NCT01881828) | Evaluate the efficacy and safety of the use of metformin in addition to standard insulin therapy in overweight and obese children and adolescents, age 12 - < 20 yr, with type 1 diabetes for at least 1 yr |
| Effect of Metformin on Vascular and Mitochondrial Function in Type 1 Diabetes (MeT1, NCT01813929) | Measure the effect of insulin sensitivity vascular function and compliance, and mitochondrial function in T1D |
Diet and exercise are important modifiable risk factors in the development of insulin resistance and vascular complications in T1D[85]. We have previously reported that adults with T1D consume higher levels of fat and saturated fat than their non-diabetic peers, and that the high intake of fat is associated with risk factors for coronary heart disease[86,87]. Although studies suggest that nutrition influences vascular complications in adults with T1D[85,88], it remains inconclusive whether insulin resistance or specific nutrients are responsible for this association. The ADA and ISPAD both emphasize incorporation of fruits, vegetables, whole grains, and low-fat food choices[89,90], but further studies related to insulin sensitivity are required. Adolescents with T1D also appear to be more sedentary and less fit than their non-diabetic counterparts[91]. Higher physical fitness among youth with T1D is associated with lower HbA1c[85,91]. In a study of overweight and sedentary nondiabetic children, 3 mo of aerobic training provided dose-response benefits for insulin resistance compared with usual physical activity[92]. Moreover, small T1D studies have demonstrated increased fitness and decreased total daily insulin dosage with aerobic and strength training, compared with normal daily activities[93]. Finally, higher levels of fitness reduced the mortality risk associated with diabetes and CVD in older adult men with diabetes, but those with T1D vs T2D were not analyzed separately[94]. Moreover, higher levels of energy expenditure (due to a more active lifestyle) were found to be associated with increased cardiorespiratory fitness in a small study of T1D adults, but not necessarily better glycemic control[95]. Therefore, while physical activity appears very promising to improve insulin resistance and reduce CVD, definitive trials in T1D are still required.
One of the major challenges in preventing vascular complications in T1D relates to the accurate identification of high risk patients at an early stage when pathology may be amenable to intervention. A promising potential therapeutic target is insulin sensitivity. Reduced insulin sensitivity is well documented in both adolescents and adults with T1D, and is thought to contribute both to the initiation and progression of macro- and microvascular complications. Novel insulin sensitivity equations derived from easily identified risk factors (e.g., waist circumference and insulin dose) may allow the clinician to estimate insulin sensitivity in the clinical setting. Interventions to improve insulin sensitivity, including diet, exercise and insulin sensitizing medications, are potential therapies to supplement conventional therapies in reducing vascular complications in T1D, but further study is required. Finally, translation of insulin sensitivity into clinical practice as a therapeutic target to reduce vascular complications requires investment in adequately powered clinical trials designed to capture important long-term vascular outcomes.
P- Reviewer: Liu JG S- Editor: Ji FF L- Editor: A E- Editor: Lu YJ
| 1. | Forlenza GP, Rewers M. The epidemic of type 1 diabetes: what is it telling us? Curr Opin Endocrinol Diabetes Obes. 2011;18:248-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 77] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 2. | Stark Casagrande S, Fradkin JE, Saydah SH, Rust KF, Cowie CC. The prevalence of meeting A1C, blood pressure, and LDL goals among people with diabetes, 1988-2010. Diabetes Care. 2013;36:2271-2279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 529] [Cited by in RCA: 572] [Article Influence: 47.7] [Reference Citation Analysis (0)] |
| 3. | Libby P, Nathan DM, Abraham K, Brunzell JD, Fradkin JE, Haffner SM, Hsueh W, Rewers M, Roberts BT, Savage PJ. Report of the National Heart, Lung, and Blood Institute-National Institute of Diabetes and Digestive and Kidney Diseases Working Group on Cardiovascular Complications of Type 1 Diabetes Mellitus. Circulation. 2005;111:3489-3493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 202] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 4. | Krolewski AS, Kosinski EJ, Warram JH, Leland OS, Busick EJ, Asmal AC, Rand LI, Christlieb AR, Bradley RF, Kahn CR. Magnitude and determinants of coronary artery disease in juvenile-onset, insulin-dependent diabetes mellitus. Am J Cardiol. 1987;59:750-755. [PubMed] |
| 5. | Olson JC, Edmundowicz D, Becker DJ, Kuller LH, Orchard TJ. Coronary calcium in adults with type 1 diabetes: a stronger correlate of clinical coronary artery disease in men than in women. Diabetes. 2000;49:1571-1578. [PubMed] |
| 6. | de Ferranti SD, de Boer IH, Fonseca V, Fox CS, Golden SH, Lavie CJ, Magge SN, Marx N, McGuire DK, Orchard TJ. Type 1 diabetes mellitus and cardiovascular disease: a scientific statement from the American Heart Association and American Diabetes Association. Circulation. 2014;130:1110-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 251] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 7. | Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Blaha MJ, Dai S, Ford ES, Fox CS, Franco S. Heart disease and stroke statistics--2014 update: a report from the American Heart Association. Circulation. 2014;129:e28-e292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3027] [Cited by in RCA: 3579] [Article Influence: 325.4] [Reference Citation Analysis (0)] |
| 8. | Pambianco G, Costacou T, Ellis D, Becker DJ, Klein R, Orchard TJ. The 30-year natural history of type 1 diabetes complications: the Pittsburgh Epidemiology of Diabetes Complications Study experience. Diabetes. 2006;55:1463-1469. [PubMed] |
| 9. | Secrest AM, Becker DJ, Kelsey SF, Laporte RE, Orchard TJ. Cause-specific mortality trends in a large population-based cohort with long-standing childhood-onset type 1 diabetes. Diabetes. 2010;59:3216-3222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 237] [Cited by in RCA: 239] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 10. | Alman AC, Maahs DM, Rewers MJ, Snell-Bergeon JK. Ideal cardiovascular health and the prevalence and progression of coronary artery calcification in adults with and without type 1 diabetes. Diabetes Care. 2014;37:521-528. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 11. | Duca LM, Bergman BC, Kinney G, Maahs D, Nadeau K, Rewers M, Schauer IE, Sippl RM, Snell-Bergeon J. Validating Insulin Sensitivity Prediction Equations in Type 1 Diabetes. USA: American Diabetes Association 2014; Abstract. |
| 12. | Collins AJ, Foley RN, Herzog C, Chavers B, Gilbertson D, Ishani A, Kasiske B, Liu J, Mau LW, McBean M. US Renal Data System 2010 Annual Data Report. Am J Kidney Dis. 2011;57:A8, e1-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 261] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 13. | Bjornstad P, Cherney D, Maahs DM. Early Diabetic Nephropathy in Type 1 Diabetes - New Insights. Current Opinion in Endocrinology, Diabetes & Obesity. 2014;21:279-286. [RCA] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 89] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 14. | Marshall SM. Diabetic nephropathy in type 1 diabetes: has the outlook improved since the 1980s? Diabetologia. 2012;55:2301-2306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 44] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 15. | Fong DS, Aiello L, Gardner TW, King GL, Blankenship G, Cavallerano JD, Ferris FL, Klein R. Diabetic retinopathy. Diabetes Care. 2003;26:226-229. [PubMed] |
| 16. | Orchard TJ, Secrest AM, Miller RG, Costacou T. In the absence of renal disease, 20 year mortality risk in type 1 diabetes is comparable to that of the general population: a report from the Pittsburgh Epidemiology of Diabetes Complications Study. Diabetologia. 2010;53:2312-2319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 232] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 17. | Snell-Bergeon JK, Hokanson JE, Jensen L, MacKenzie T, Kinney G, Dabelea D, Eckel RH, Ehrlich J, Garg S, Rewers M. Progression of coronary artery calcification in type 1 diabetes: the importance of glycemic control. Diabetes Care. 2003;26:2923-2928. [PubMed] |
| 18. | Bjornstad P, Wadwa RP. Risks and benefits of statin use in young people with type 1 diabetes. Curr Diab Rep. 2014;14:499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 19. | Nathan DM, Zinman B, Cleary PA, Backlund JY, Genuth S, Miller R, Orchard TJ. Modern-day clinical course of type 1 diabetes mellitus after 30 years’ duration: the diabetes control and complications trial/epidemiology of diabetes interventions and complications and Pittsburgh epidemiology of diabetes complications experience (1983-2005). Arch Intern Med. 2009;169:1307-1316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 537] [Cited by in RCA: 455] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 20. | Schauer IE, Snell-Bergeon JK, Bergman BC, Maahs DM, Kretowski A, Eckel RH, Rewers M. Insulin resistance, defective insulin-mediated fatty acid suppression, and coronary artery calcification in subjects with and without type 1 diabetes: The CACTI study. Diabetes. 2011;60:306-314. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 183] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 21. | Nadeau KJ, Regensteiner JG, Bauer TA, Brown MS, Dorosz JL, Hull A, Zeitler P, Draznin B, Reusch JEB. Insulin Resistance in Adolescents with Type 1 Diabetes and Its Relationship to Cardiovascular Function. J ClinEndocr Metab. 2010;95:513-521. [RCA] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 234] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 22. | Amiel SA, Sherwin RS, Simonson DC, Lauritano AA, Tamborlane WV. Impaired insulin action in puberty. A contributing factor to poor glycemic control in adolescents with diabetes. N Engl J Med. 1986;315:215-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 664] [Cited by in RCA: 644] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 23. | Heptulla RA, Stewart A, Enocksson S, Rife F, Ma TY, Sherwin RS, Tamborlane WV, Caprio S. In situ evidence that peripheral insulin resistance in adolescents with poorly controlled type 1 diabetes is associated with impaired suppression of lipolysis: a microdialysis study. Pediatr Res. 2003;53:830-835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 24. | Williams KV, Erbey JR, Becker D, Arslanian S, Orchard TJ. Can clinical factors estimate insulin resistance in type 1 diabetes? Diabetes. 2000;49:626-632. [PubMed] |
| 25. | Levy-Marchal C, Arslanian S, Cutfield W, Sinaiko A, Druet C, Marcovecchio ML, Chiarelli F. Insulin resistance in children: consensus, perspective, and future directions. J Clin Endocrinol Metab. 2010;95:5189-5198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 292] [Cited by in RCA: 282] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 26. | Cleland SJ, Fisher BM, Colhoun HM, Sattar N, Petrie JR. Insulin resistance in type 1 diabetes: what is ‘double diabetes’ and what are the risks? Diabetologia. 2013;56:1462-1470. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 166] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 27. | Nathan DM, Cleary PA, Backlund JY, Genuth SM, Lachin JM, Orchard TJ, Raskin P, Zinman B. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med. 2005;353:2643-2653. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3867] [Cited by in RCA: 3436] [Article Influence: 171.8] [Reference Citation Analysis (0)] |
| 28. | Orchard TJ, Olson JC, Erbey JR, Williams K, Forrest KY, Smithline Kinder L, Ellis D, Becker DJ. Insulin resistance-related factors, but not glycemia, predict coronary artery disease in type 1 diabetes: 10-year follow-up data from the Pittsburgh Epidemiology of Diabetes Complications Study. Diabetes Care. 2003;26:1374-1379. [PubMed] |
| 29. | Soedamah-Muthu SS, Chaturvedi N, Toeller M, Ferriss B, Reboldi P, Michel G, Manes C, Fuller JH. Risk factors for coronary heart disease in type 1 diabetic patients in Europe: the EURODIAB Prospective Complications Study. Diabetes Care. 2004;27:530-537. [PubMed] |
| 30. | Kilpatrick ES, Rigby AS, Atkin SL. Insulin resistance, the metabolic syndrome, and complication risk in type 1 diabetes: “double diabetes” in the Diabetes Control and Complications Trial. Diabetes Care. 2007;30:707-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 317] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 31. | Bjornstad P, Snell-Bergeon JK, Rewers M, Jalal D, Chonchol MB, Johnson RJ, Maahs DM. Early diabetic nephropathy: a complication of reduced insulin sensitivity in type 1 diabetes. Diabetes Care. 2013;36:3678-3683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 51] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 32. | Bjornstad P, Maahs DM, Johnson RJ, Rewers M, Snell-Bergeon JK. Estimated insulin sensitivity predicts regression of albuminuria in Type 1 diabetes. Diabet Med. 2014;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 33. | Yki-Järvinen H, Koivisto VA. Natural course of insulin resistance in type I diabetes. N Engl J Med. 1986;315:224-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 222] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 34. | Perseghin G, Lattuada G, Danna M, Sereni LP, Maffi P, De Cobelli F, Battezzati A, Secchi A, Del Maschio A, Luzi L. Insulin resistance, intramyocellular lipid content, and plasma adiponectin in patients with type 1 diabetes. Am J Physiol Endocrinol Metab. 2003;285:E1174-E1181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 130] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 35. | Houstis N, Rosen ED, Lander ES. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature. 2006;440:944-948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1770] [Cited by in RCA: 1895] [Article Influence: 99.7] [Reference Citation Analysis (0)] |
| 36. | Aronoff SL, Berkowitz K, Shreiner B, Want L. Glucose Metabolism and Regulation: Beyond Insulin and Glucagon. Diabetes Spectrum. 2004;17:183-190. |
| 37. | Bergman BC, Howard D, Schauer IE, Maahs DM, Snell-Bergeon JK, Eckel RH, Perreault L, Rewers M. Features of hepatic and skeletal muscle insulin resistance unique to type 1 diabetes. J Clin Endocrinol Metab. 2012;97:1663-1672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 83] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 38. | Ye J. Role of insulin in the pathogenesis of free fatty acid-induced insulin resistance in skeletal muscle. Endocr Metab Immune Disord Drug Targets. 2007;7:65-74. [PubMed] |
| 39. | Wullaert A, van Loo G, Heyninck K, Beyaert R. Hepatic tumor necrosis factor signaling and nuclear factor-kappaB: effects on liver homeostasis and beyond. Endocr Rev. 2007;28:365-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 180] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 40. | Kaneto H, Matsuoka TA, Katakami N, Kawamori D, Miyatsuka T, Yoshiuchi K, Yasuda T, Sakamoto K, Yamasaki Y, Matsuhisa M. Oxidative stress and the JNK pathway are involved in the development of type 1 and type 2 diabetes. Curr Mol Med. 2007;7:674-686. [PubMed] |
| 41. | Erbey JR, Kuller LH, Becker DJ, Orchard TJ. The association between a family history of type 2 diabetes and coronary artery disease in a type 1 diabetes population. Diabetes Care. 1998;21:610-614. [PubMed] |
| 42. | Roglic G, Colhoun HM, Stevens LK, Lemkes HH, Manes C, Fuller JH. Parental history of hypertension and parental history of diabetes and microvascular complications in insulin-dependent diabetes mellitus: the EURODIAB IDDM Complications Study. Diabet Med. 1998;15:418-426. [PubMed] [DOI] [Full Text] |
| 43. | Purnell JQ, Zinman B, Brunzell JD. The effect of excess weight gain with intensive diabetes mellitus treatment on cardiovascular disease risk factors and atherosclerosis in type 1 diabetes mellitus: results from the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study (DCCT/EDIC) study. Circulation. 2013;127:180-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 204] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 44. | Specht BJ, Wadwa RP, Snell-Bergeon JK, Nadeau KJ, Bishop FK, Maahs DM. Estimated insulin sensitivity and cardiovascular disease risk factors in adolescents with and without type 1 diabetes. J Pediatr. 2013;162:297-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 45. | Thorn LM, Forsblom C, Fagerudd J, Thomas MC, Pettersson-Fernholm K, Saraheimo M, Wadén J, Rönnback M, Rosengård-Bärlund M, Björkesten CG. Metabolic syndrome in type 1 diabetes: association with diabetic nephropathy and glycemic control (the FinnDiane study). Diabetes Care. 2005;28:2019-2024. [PubMed] |
| 46. | Rathmann W, Funkhouser E, Dyer AR, Roseman JM. Relations of hyperuricemia with the various components of the insulin resistance syndrome in young black and white adults: the CARDIA study. Coronary Artery Risk Development in Young Adults. Ann Epidemiol. 1998;8:250-261. [PubMed] |
| 47. | Bjornstad P, Snell-Bergeon JK, McFann K, Wadwa RP, Rewers M, Rivard CJ, Jalal D, Chonchol MB, Johnson RJ, Maahs DM. Serum uric acid and insulin sensitivity in adolescents and adults with and without type 1 diabetes. J Diabetes Complications. 2014;28:298-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 48. | Orchard TJ, Chang YF, Ferrell RE, Petro N, Ellis DE. Nephropathy in type 1 diabetes: a manifestation of insulin resistance and multiple genetic susceptibilities? Further evidence from the Pittsburgh Epidemiology of Diabetes Complication Study. Kidney Int. 2002;62:963-970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 169] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 49. | Martin FI, Stocks AE. Insulin sensitivity and vascular disease in insulin-dependent diabetics. Br Med J. 1968;2:81-82. [PubMed] |
| 50. | Yip J, Mattock MB, Morocutti A, Sethi M, Trevisan R, Viberti G. Insulin resistance in insulin-dependent diabetic patients with microalbuminuria. Lancet. 1993;342:883-887. [PubMed] |
| 51. | Bjornstad P, Maahs DM, Cherney DZ, Cree-Green M, West A, Pyle L, Nadeau KJ. Insulin sensitivity is an important determinant of renal health in adolescents with type 2 diabetes. Diabetes Care. 2014;37:3033-3039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 52. | Bjornstad P, Cree Green M, Baumgartner A, Maahs D, Cherney D, Pyle L, Regensteiner JG, Reusch JE, Nadeau K. Renal function is associated with peak exercise capacity in adolescents with type 1 diabetes. Diabetes Care. 2014;In Press. |
| 53. | Catalano C, Muscelli E, Quiñones Galvan A, Baldi S, Masoni A, Gibb I, Torffvit O, Seghieri G, Ferrannini E. Effect of insulin on systemic and renal handling of albumin in nondiabetic and NIDDM subjects. Diabetes. 1997;46:868-875. [PubMed] |
| 54. | Cohen AJ, McCarthy DM, Stoff JS. Direct hemodynamic effect of insulin in the isolated perfused kidney. Am J Physiol. 1989;257:F580-F585. [PubMed] |
| 55. | Tucker BJ, Anderson CM, Thies RS, Collins RC, Blantz RC. Glomerular hemodynamic alterations during acute hyperinsulinemia in normal and diabetic rats. Kidney Int. 1992;42:1160-1168. [PubMed] |
| 56. | Cherney DZ, Miller JA, Scholey JW, Nasrallah R, Hébert RL, Dekker MG, Slorach C, Sochett EB, Bradley TJ. Renal hyperfiltration is a determinant of endothelial function responses to cyclooxygenase 2 inhibition in type 1 diabetes. Diabetes Care. 2010;33:1344-1346. [PubMed] |
| 57. | Cherney DZ, Perkins BA, Soleymanlou N, Maione M, Lai V, Lee A, Fagan NM, Woerle HJ, Johansen OE, Broedl UC. Renal hemodynamic effect of sodium-glucose cotransporter 2 inhibition in patients with type 1 diabetes mellitus. Circulation. 2014;129:587-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 847] [Cited by in RCA: 980] [Article Influence: 81.7] [Reference Citation Analysis (0)] |
| 58. | Cherney DZ, Reich HN, Jiang S, Har R, Nasrallah R, Hébert RL, Lai V, Scholey JW, Sochett EB. Hyperfiltration and effect of nitric oxide inhibition on renal and endothelial function in humans with uncomplicated type 1 diabetes mellitus. Am J Physiol Regul Integr Comp Physiol. 2012;303:R710-R718. [PubMed] |
| 59. | Cherney DZ, Scholey JW, Sochett E, Bradley TJ, Reich HN. The acute effect of clamped hyperglycemia on the urinary excretion of inflammatory cytokines/chemokines in uncomplicated type 1 diabetes: a pilot study. Diabetes Care. 2011;34:177-180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 60. | Cherney DZ, Sochett EB. Evolution of renal hyperfiltration and arterial stiffness from adolescence into early adulthood in type 1 diabetes. Diabetes Care. 2011;34:1821-1826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 61. | Chaturvedi N, Sjoelie AK, Porta M, Aldington SJ, Fuller JH, Songini M, Kohner EM. Markers of insulin resistance are strong risk factors for retinopathy incidence in type 1 diabetes. Diabetes Care. 2001;24:284-289. [PubMed] |
| 62. | Parvanova A, Iliev I, Filipponi M, Dimitrov BD, Vedovato M, Tiengo A, Trevisan R, Remuzzi G, Ruggenenti P. Insulin resistance and proliferative retinopathy: a cross-sectional, case-control study in 115 patients with type 2 diabetes. J Clin Endocrinol Metab. 2004;89:4371-4376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 63. | Anan F, Takayuki M, Takahashi N, Nakagawa M, Eshima N, Saikawa T, Yoshimatsu H. Diabetic retinopathy is associated with insulin resistance and cardiovascular autonomic dysfunction in type 2 diabetic patients. Hypertens Res. 2009;32:299-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 64. | Chillarón JJ, Goday A, Flores-Le-Roux JA, Benaiges D, Carrera MJ, Puig J, Cano-Pérez JF, Pedro-Botet J. Estimated glucose disposal rate in assessment of the metabolic syndrome and microvascular complications in patients with type 1 diabetes. J Clin Endocrinol Metab. 2009;94:3530-3534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 84] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 65. | Cho YN, Lee KO, Jeong J, Park HJ, Kim SM, Shin HY, Hong JM, Ahn CW, Choi YC. The role of insulin resistance in diabetic neuropathy in Koreans with type 2 diabetes mellitus: a 6-year follow-up study. Yonsei Med J. 2014;55:700-708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 66. | Lee KO, Nam JS, Ahn CW, Hong JM, Kim SM, Sunwoo IN, Moon JS, Na SJ, Choi YC. Insulin resistance is independently associated with peripheral and autonomic neuropathy in Korean type 2 diabetic patients. Acta Diabetol. 2012;49:97-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 67. | Lehtinen JM, Niskanen L, Hyvönen K, Siitonen O, Uusitupa M. Nerve function and its determinants in patients with newly-diagnosed type 2 (non-insulin-dependent) diabetes mellitus and in control subjects--a 5-year follow-up. Diabetologia. 1993;36:68-72. [PubMed] |
| 68. | Howard G, O’Leary DH, Zaccaro D, Haffner S, Rewers M, Hamman R, Selby JV, Saad MF, Savage P, Bergman R. Insulin sensitivity and atherosclerosis. The Insulin Resistance Atherosclerosis Study (IRAS) Investigators. Circulation. 1996;93:1809-1817. [PubMed] |
| 69. | Ginsberg HN. Insulin resistance and cardiovascular disease. J Clin Invest. 2000;106:453-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 811] [Cited by in RCA: 836] [Article Influence: 33.4] [Reference Citation Analysis (0)] |
| 70. | Rodrigues TC, Haarhues MD, Kinney GL, Rewers M, Snell-Bergeon JK, Veyna AM. Obesity and coronary artery calcium in diabetes: the coronary artery calcification in type 1 diabetes (CACTI) study. Diabetes Technol Ther. 2011;13:991. |
| 71. | Pawlyk AC, Giacomini KM, McKeon C, Shuldiner AR, Florez JC. Metformin pharmacogenomics: current status and future directions. Diabetes. 2014;63:2590-2599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 108] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 72. | Vella S, Buetow L, Royle P, Livingstone S, Colhoun HM, Petrie JR. The use of metformin in type 1 diabetes: a systematic review of efficacy. Diabetologia. 2010;53:809-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 143] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 73. | Nadeau KJ, Chow K, Alam S, Lindquist K, Campbell S, McFann K, Klingensmith G, Walravens P. Effects of low dose metformin in adolescents with type I diabetes mellitus: a randomized, double-blinded placebo-controlled study. Pediatr Diabetes. 2014;Apr 3; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 74. | DeFronzo RA, Goodman AM. Efficacy of metformin in patients with non-insulin-dependent diabetes mellitus. The Multicenter Metformin Study Group. N Engl J Med. 1995;333:541-549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 937] [Cited by in RCA: 880] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 75. | Beisswenger PJ, Howell SK, Touchette AD, Lal S, Szwergold BS. Metformin reduces systemic methylglyoxal levels in type 2 diabetes. Diabetes. 1999;48:198-202. [PubMed] |
| 76. | Nagi DK, Yudkin JS. Effects of metformin on insulin resistance, risk factors for cardiovascular disease, and plasminogen activator inhibitor in NIDDM subjects. A study of two ethnic groups. Diabetes Care. 1993;16:621-629. [PubMed] |
| 77. | Giugliano D, Quatraro A, Consoli G, Minei A, Ceriello A, De Rosa N, D’Onofrio F. Metformin for obese, insulin-treated diabetic patients: improvement in glycaemic control and reduction of metabolic risk factors. Eur J Clin Pharmacol. 1993;44:107-112. [PubMed] |
| 78. | Landin K, Tengborn L, Smith U. Treating insulin resistance in hypertension with metformin reduces both blood pressure and metabolic risk factors. J Intern Med. 1991;229:181-187. [PubMed] |
| 79. | August P, Hardison RM, Hage FG, Marroquin OC, McGill JB, Rosenberg Y, Steffes M, Wall BM, Molitch M. Change in albuminuria and eGFR following insulin sensitization therapy versus insulin provision therapy in the BARI 2D study. Clin J Am Soc Nephrol. 2014;9:64-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 80. | Effect of Intensive Diabetes Therapy on the Progression of Diabetic Retinopathy in Patients with Type 1 Diabetes: 18 Years of Follow-up in the DCCT/EDIC. Diabetes. 2014;Sep 9; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 212] [Cited by in RCA: 230] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 81. | de Boer IH, Rue TC, Cleary PA, Lachin JM, Molitch ME, Steffes MW, Sun W, Zinman B, Brunzell JD, White NH. Long-term renal outcomes of patients with type 1 diabetes mellitus and microalbuminuria: an analysis of the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications cohort. Arch Intern Med. 2011;171:412-420. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 281] [Cited by in RCA: 255] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 82. | de Boer IH, Sun W, Cleary PA, Lachin JM, Molitch ME, Steffes MW, Zinman B. Intensive diabetes therapy and glomerular filtration rate in type 1 diabetes. N Engl J Med. 2011;365:2366-2376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 402] [Cited by in RCA: 419] [Article Influence: 29.9] [Reference Citation Analysis (0)] |
| 83. | Group DR. The Effect of Intensive Treatment of Diabetes on the Development and Progression of Long-Term Complications in Insulin-Dependent Diabetes Mellitus. N Engl J Med. 1993;329:977-986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17510] [Cited by in RCA: 16280] [Article Influence: 508.8] [Reference Citation Analysis (4)] |
| 84. | Purnell JQ, Hokanson JE, Marcovina SM, Steffes MW, Cleary PA, Brunzell JD. Effect of excessive weight gain with intensive therapy of type 1 diabetes on lipid levels and blood pressure: results from the DCCT. Diabetes Control and Complications Trial. JAMA. 1998;280:140-146. [PubMed] |
| 85. | Maahs DM, Daniels SR, de Ferranti SD, Dichek HL, Flynn J, Goldstein BI, Kelly AS, Nadeau KJ, Martyn-Nemeth P, Osganian SK. Cardiovascular disease risk factors in youth with diabetes mellitus: a scientific statement from the american heart association. Circulation. 2014;130:1532-1558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 130] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 86. | Bishop FK, Maahs DM, Snell-Bergeon JK, Ogden LG, Kinney GL, Rewers M. Lifestyle risk factors for atherosclerosis in adults with type 1 diabetes. Diab Vasc Dis Res. 2009;6:269-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 87. | Snell-Bergeon JK, Chartier-Logan C, Maahs DM, Ogden LG, Hokanson JE, Kinney GL, Eckel RH, Ehrlich J, Rewers M. Adults with type 1 diabetes eat a high-fat atherogenic diet that is associated with coronary artery calcium. Diabetologia. 2009;52:801-809. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 71] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 88. | Albu JB, Heilbronn LK, Kelley DE, Smith SR, Azuma K, Berk ES, Pi-Sunyer FX, Ravussin E. Metabolic changes following a 1-year diet and exercise intervention in patients with type 2 diabetes. Diabetes. 2010;59:627-633. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 74] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 89. | Standards of medical care in diabetes--2014. Diabetes Care. 2014;37 Suppl 1:S14-S80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2830] [Cited by in RCA: 3016] [Article Influence: 274.2] [Reference Citation Analysis (0)] |
| 90. | Acerini C, Craig ME, de Beaufort C, Maahs DM, Hanas R. Introduction to ISPAD Clinical Practice Consensus Guidelines 2014 Compendium. Pediatr Diabetes. 2014;15 Suppl 20:1-3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 91. | Robertson K, Riddell MC, Guinhouya BC, Adolfsson P, Hanas R. Exercise in children and adolescents with diabetes. Pediatr Diabetes. 2014;15 Suppl 20:203-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 92. | Davis CL, Pollock NK, Waller JL, Allison JD, Dennis BA, Bassali R, Meléndez A, Boyle CA, Gower BA. Exercise dose and diabetes risk in overweight and obese children: a randomized controlled trial. JAMA. 2012;308:1103-1112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 153] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 93. | D’hooge R, Hellinckx T, Van Laethem C, Stegen S, De Schepper J, Van Aken S, Dewolf D, Calders P. Influence of combined aerobic and resistance training on metabolic control, cardiovascular fitness and quality of life in adolescents with type 1 diabetes: a randomized controlled trial. Clin Rehabil. 2011;25:349-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 69] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 94. | McAuley P, Myers J, Emerson B, Oliveira RB, Blue CL, Pittsley J, Froelicher VF. Cardiorespiratory fitness and mortality in diabetic men with and without cardiovascular disease. Diabetes Res Clin Pract. 2009;85:e30-e33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 95. | Valletta JJ, Chipperfield AJ, Clough GF, Byrne CD. Daily energy expenditure, cardiorespiratory fitness and glycaemic control in people with type 1 diabetes. PLoS One. 2014;9:e97534. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |