Published online Dec 15, 2022. doi: 10.4239/wjd.v13.i12.1168
Peer-review started: July 20, 2022
First decision: September 4, 2022
Revised: September 20, 2022
Accepted: October 27, 2022
Article in press: October 27, 2022
Published online: December 15, 2022
Processing time: 148 Days and 4.9 Hours
Cardiovascular outcome trials have demonstrated cardiovascular safety of glimepiride (a sulfonylureas) against dipeptidyl peptidase-4 inhibitor linagliptin. Gliclazide (another newer sulfonylureas) has shown similar glycemic efficacy and 50% decreased risk of hypoglycemia compared to glimepiride.
Considering the absence of cardiovascular outcome trials for gliclazide, we decided to conduct a systematic review of the literature to assess the car-diovascular (CV) safety by assessing the risk for major adverse CV events and hypoglycemia risk of gliclazide vs linagliptin in patients with type 2 diabetes (T2D).
This systematic review followed the current Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines to analyze all the clinical studies published from 2008 that compared the two drugs in patients with T2D with no risk of CV disease (CVD). We included only evidence designated high quality by the Oxford Center for Evidence-based Medicine-Levels of Evidence.
Eight clinical studies were included in the narrative descriptive analysis (gliclazide: 5 and linagliptin: 3). The CV safety of gliclazide in the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation trial and of linagliptin in the Cardiovascular and Renal Microvascular Outcome Study With Linagliptin (CARMELINA) and CARdiovascular Outcome study of LINAgliptin vs glimepiride in patients with T2D (CAROLINA) trials were excluded from the comparative analysis as these trials demonstrated CV and hypoglycemia benefits in patients at high risk of CVD. However, since these are landmark trials, they were discussed in brief to show the CV benefits and low hypoglycemia risk of gliclazide and linagliptin. We did not find any study comparing gliclazide with linagliptin. Hence, direct comparison of their major adverse CV events and hypoglycemia risk could not be carried out. However, the literature meeting the inclusion criteria showed that both drugs were effective in achieving the desired glycemic control and had low major adverse CV events and hypoglycemia risk in adult patients with no history of CVD.
Gliclazide can be considered an effective and safe glucose-lowering drug in T2D patients with no established CVD but at high risk of CVD due to their T2D status. Future randomized controlled trials comparing gliclazide with linagliptin or dipeptidyl peptidase-4 inhibitors can confirm these findings.
Core Tip: This systematic review showed the lack of high-quality evidence and head-to head trials comparing the cardiovascular safety and hypoglycemia risk of gliclazide (a sulfonylurea) vs linagliptin (dipeptidyl peptidase-4 inhibitor) in adults with type 2 diabetes and no cardiovascular disease. While dipeptidyl peptidase-4 inhibitors have been proven to be cardiovascular neutral, sulfonylureas like gliclazide are commonly prescribed and recommended glucose-lowering drugs in low resource settings. Hence, it is important to establish the cardiovascular safety and hypoglycemia risk of gliclazide vs linagliptin to highlight that gliclazide may be a cost-effective yet safe treatment option for patients with type 2 diabetes.
- Citation: Mohan V, Wangnoo S, Das S, Dhediya R, Gaurav K. Comparison of gliclazide vs linagliptin on hypoglycemia and cardiovascular events in type 2 diabetes mellitus: A systematic review. World J Diabetes 2022; 13(12): 1168-1183
- URL: https://www.wjgnet.com/1948-9358/full/v13/i12/1168.htm
- DOI: https://dx.doi.org/10.4239/wjd.v13.i12.1168
Type 2 diabetes (T2D), characterized by chronic hyperglycemia and impaired insulin secretion, is often associated with disease-related microvascular and macrovascular complications and treatment-related complications like hypoglycemia[1,2]. Consequently, patients with T2D are at an increased risk for cardiovascular (CV) complications and hypoglycemia. Hence, glucose-lowering drugs (GLDs) should not have CV complications and higher hypoglycemic episodes (HE) as adverse effects (AEs) and should ideally provide CV benefits or neutrality[1,2].
Sulfonylureas (SUs) are the most prescribed T2D pharmacotherapy, especially in resource limited settings[3]. Apart from their cost benefit, Sus have an exceptional glycemic efficacy with average glycosylated hemoglobin (HbA1c) reduction by 1%-2%, good safety profile and gastrointestinal tolerability[3]. However, hypoglycemia, weight gain and decreasing efficacy over time are the main concerns with SUs due to their insulinotropic mechanism of action[3-5]. On the other hand, newer oral GLDs like dipeptidyl peptidase-4 (DPP4) inhibitors and sodium-glucose cotransporter-2 (SGLT2) inhibitors provide comparably less glycemic control than SUs (average HbA1c reduction 0.5%-0.8%), are costlier than SUs and often need to be combined with SUs to achieve the required glycemic control[3].
However, since, the time of their inception into T2D treatment regime, SUs have been subjected to criticism for CV safety[3,6]. The CV safety of SUs has been derived from small, inadequately powered randomized controlled trials (RCTs) and observational studies[3]. However, formal cardiovascular outcome trials (CVOTs) are not available for SUs[3,6].
Then, in 2008, the United States Food and Drug Administration mandated the assessment of CV safety of newer GLDs[7]. Hence, large multinational, CVOTs of newer oral GLDs like DPP4 inhibitors[8-12] and SGLT2 inhibitors[13-15] were conducted and showed their CV benefits. DPP4 inhibitors and SGLT2 inhibitors proved to be costly options in resource limited settings because of the chronic disease nature of T2D and because most patients pay from their pocket for the treatment[16,17].
Despite their unquestionable glucose lowering efficacy, current diabetes guidelines no longer favors the use of SUs because of CV safety concerns except when cost is an issue[3,6]. SUs have been recommended as the add-on of choice after metformin for adequate glycemic control in resource limited settings by the World Health Organization (WHO) Guidelines, the Research Society for the Study of Diabetes in India/Endocrine Society of India (RSSDI-ESI) (2020) guidelines from India[18,19], the International Task Force (ITF) Consensus[20] and the International Diabetes Federation (IDF)[21]. The ITF recommends glimepiride and gliclazide modified release (MR) as the SU of choice to be added to metformin, while the IDF gave equal importance to SUs (except glibenclamide/glyburide), a DPP4 inhibitor or an SGLT2 inhibitor[20,21].
The American Diabetes Association (ADA) (2021) guidelines recommend various add-on pharmacotherapies for T2D patients poorly controlled on metformin, including DPP4 inhibitors, SGLT2 inhibitors and SUs[22]. The American Diabetes Association guidelines recommend T2D patients with CV and renal morbidities should ideally be prescribed SGLT2 inhibitors or glucagon-like peptide-1 (GLP-1) agonists as the next oral GLDs after metformin[22]. However, the choice of add-on therapy in patients without CV risk is not clear.
Of the various DPP4 inhibitors used in T2D, landmark linagliptin trials have demonstrated CV safety and safety against HE in T2D patients with a high risk of CV disease (CVD)[8,9]. On the other hand, a landmark non-CVOT trial in patients with high CV risk showed that high intensity gliclazide treatment conferred low CV risk[23].
Many systematic reviews (SRs) and/or meta-analyses (MAs) have assessed the efficacy and safety [hypoglycemia and major adverse cardiovascular events (MACE; CV death, nonfatal myocardial infarction/ischemia/acute coronary syndrome or nonfatal stroke)] of SUs vs DPP4 inhibitors with mixed results[24-28]. These SRs and meta-analyses identified a need for RCTs comparing individual SUs with a DPP4 inhibitor. Hence, this SR was carried out to assess the CV safety and hypoglycemia risk of gliclazide vs linagliptin in T2D patients, both in monotherapy and as add-on to metformin setting.
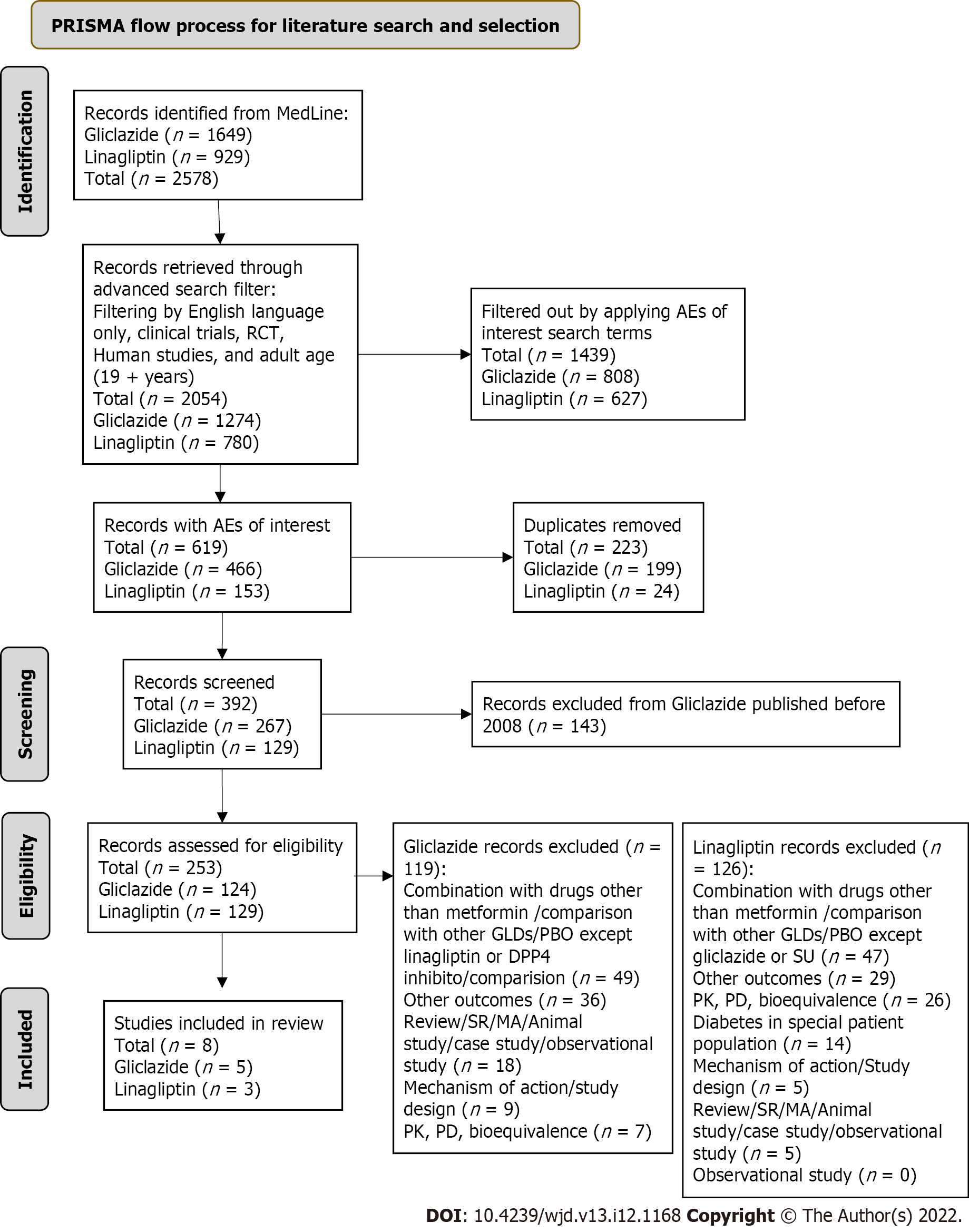
The MEDLINE database was searched on September 9, 2021 for records on gliclazide or linagliptin with no filter added. This retrieved 2578 records. An advanced search filter was then applied to filter by English language only, clinical trials, RCT, human studies and adult age (19 + years). These filters retrieved 2054 records. The records were further filtered by applying adverse events of interest: hypoglycemia, low blood sugar, myocardial infarction/myocardial ischemia (MI), transient ischemic attack, CV death and stroke. This retrieved 615 records; 223 duplicates were removed and the remaining 392 records were screened. It was seen that linagliptin records were available from 2008 onwards only. Hence, to standardize the time period for the entire literature search, gliclazide records published before 2008 were removed. The remaining 248 records were assessed for eligibility. After excluding records that did not meet the eligibility criteria as mentioned in Table 1, eight records were included (5 for gliclazide and 3 for linagliptin). The details of the literature search and study selection are outlined in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart (Figure 1). Google Scholar was searched for any additional manuscripts that were missed on MEDLINE. This retrieved no additional records as per study selection criteria.
| Inclusion criteria | Exclusion criteria |
| Age 19 yr and < 70 yr; Male and Female; type 2 diabetes | Age below 19 yr or ≥ 70 yr; type 1 diabetes; no diabetes |
| Human studies: Any race, ethnicity | Clinical trials evaluating gliclazide or linagliptin in patients with specific comorbidites including CVD1 |
| Randomized clinical trials on safety of: | Review articles, systematic reviews and meta-analysis, network meta-analysis, pooled analysis of trials, case studies, non-randomized trials |
| -Gliclazide monotherapy versus linagliptin monotherapy | |
| -Gliclazide + metformin versus linagliptin + metformin | |
| Randomized clinical trials on safety of: | Pharmacokinetic, pharmacodynamic and bioequivalence study; retrospective chart review; observational real-world study; case study; trials studying mechanism of action of gliclazide or linagliptin; literature reporting only study design; trial summaries and implications; animal studies; preclinical studies |
| -Gliclazide versus DPP4 inhibitors | |
| -Linagliptin versus sulfonylureas | |
| Randomized clinical trials on gliclazide or linagliptin monotherapy evaluating the following outcomes: | Clinical trials evaluating gliclazide or linagliptin versus PBO |
| -Hypoglycemia or low blood sugar | Clinical trials evaluating gliclazide or linagliptin in combination with other GLDs except metformin |
| -Occurrence of 3 point major adverse cardiovascular events (3P-MACE): Cardiovascular death, nonfatal myocardial infarction/ischemia/acute coronary syndrome, or nonfatal stroke (transient ischemic attack included) | Clinical trials evaluating gliclazide or linagliptin versus other GLDs except: (1) DPP4 inhibitors for gliclazide; and (2)sulfonylureas for linagliptin |
| Clinical trials evaluating other glycemic, cardiac, cardiovascular outcomes than those of interest; other outcomes (e.g., microvascular complications) |
Two independent reviewers used the current PRISMA guidelines for SRs[29,30] to independently carry out the literature search on the same day. Any conflict in the number of records at identification, screening, eligibility and inclusion were mutually discussed and resolved by consensus. We do note that the protocol for this systematic review has not been published.
As shown by the PRISMA flow chart, there were many articles regarding both gliclazide and linagliptin. Hence, we included only high-quality evidence. RCTs were designated the highest quality by the Oxford Center for Evidence-based Medicine-Levels of Evidence[31] followed by a randomized design of any type. Hence, we included only randomized studies. Placebo controlled studies were not included as there were no gliclazide vs placebo studies. The main reason for this could be that trials in the initial trajectory of drug development were missed by standardizing the study period from 2008 onwards. Additionally, studies comparing gliclazide or linagliptin with metformin were also not included because both drugs have a known and comparable efficacy and safety profile vs metformin.
Further, risk of bias assessment was independently carried out by two researchers who assessed the scientific quality of the records using the Cochrane Collaboration’s tool for risk of bias assessment[32]. The Cochrane Risk of Bias tool assesses seven domains of bias and stratifies the risk of bias as low, high and unclear risk. Discrepancies between reviewers at any stage were resolved by discussion and consensus.
All the studies clearly defined and reported the outcomes of interest (hypoglycemia and MACE) and clearly mentioned all the CVDs that were assessed as exclusion criteria. Only one gliclazide trial[33] did not have any CVD as an exclusion criteria. The trials clearly explained the randomization schedule and were largely double-blind studies. The number of participants for which the outcomes of interest were reported was clearly stated.
However, most studies were not designed to report the outcome of interest (hypoglycemia and MACE) as their main primary and/or secondary endpoint. These outcomes of interest were primarily reported as AEs or safety endpoints.
The systematic literature search (Figure 1) did not retrieve any head-to-head trials comparing gliclazide ± metformin with linagliptin ± metformin. Hence, direct comparison of their outcomes was not possible. The gliclazide and linagliptin trials that met the inclusion criteria could not be compared to reach a statistical analysis due to various reasons. The studies captured for the two drugs were heterogeneous with respect to study design and duration, the outcomes of interest being evaluated as primary or secondary or safety (as AE) endpoints or as incident findings, definition of outcomes [e.g., definition of hypoglycemia-cut off blood glucose (BG) level] and the statistical method used for analysis. The study population of the various studies differed in age, ethnicity and patient profile (e.g., treatment naïve or after failure of SU). Hence, a meta-analysis or a network meta-analysis could not be carried out. Therefore, key outcomes were described in a narrative manner for each drug separately, with due consideration given to the PRISMA checklist[29].
This section aimed to include RCTs that compared gliclazide vs linagliptin or a DPP4 inhibitor in a monotherapy setting or compared gliclazide as an add-on to metformin vs linagliptin/DPP4 inhibitor as add-on to metformin.
Gliclazide vs linagliptin or DPP4 inhibitors: There were no records comparing gliclazide with linagliptin. One study compared gliclazide with vildagliptin, a DPP4 inhibitor[34] (Table 2). Foley et al[34] compared the efficacy and safety of 2 years of monotherapy with vildagliptin vs gliclazide in 1092 drug-naïve patients with T2D having HbA1c of 7.5%-11.0%. In this vildagliptin non-inferiority trial, the vildagliptin group had a lower incidence of grade 1 hypoglycemia than the gliclazide group (0.7% vs 1.7%).
| Ref. | Primary study objective | Study design | Study population | CVD excluded | Number of participants | Study duration | Endpoint (hypoglycemia) | Hypoglycemia definition | Hypoglycemia results | Endpoint (MACE) | MACE definition | MACE results |
| Gliclazide vs DPP4 inhibitor (vildagliptin) | ||||||||||||
| Foley et al[34], 2009 | Compare the efficacy and safety of vildagliptin vs gliclazide | Randomized, multicenter, double-blind, active-controlled study | Drug-naïve patients with T2D, HbA1c of 7.5%-11.0% | CHF NYHA class III or IV, ECG abnormalities | 1092 | 104 wk | AEs were safety endpoints | Grade 1 hypoglycemic events per week: symptoms suggestive of low blood glucose confirmed by SMBG measurement of < 3.1 mmol/L plasma glucose equivalent not requiring the assistance of another party; Grade 2 hypoglycemic event (requiring the assistance of another party) or if there were 3 or more asymptomatic glucose values < 3.1 mmol/L per week | Grade 1 hypoglycemia: 4 patients (0.7%) in the vildagliptin group and 14 (1.7%) in the gliclazide group. ≥ 2 HEs: 2 patients in the gliclazide group and none in vildagliptin group No grade 2 HEs in either group | - | - | - |
| Gliclazide + metformin vs DPP4 inhibitor (vildagliptin) + metformin | ||||||||||||
| Vianna et al[35], 2018 (Part of BoneGlic Trial) | Compare the effects on glycemic variability and bone metabolism | Single center, randomized, controlled, open-label (blinded to the observer) | Postmenopausal Brazilian women with T2D and treated with a stable metformin dose for ≤ 3 mo | CV complications | 56 (42 randomized) | 2-wk pre-randomization period followed by 24 wk | As AE | Major hypoglycemia: glucagon, carbohydrates administration by another person or other resuscitative measures; minor hypoglycemia: BG ≤ 3.9 mmol/L with or without typical symptoms or hypoglycemia symptoms without BG test | No differences from baseline in time to hypoglycemia (% of time ≤ 3.9 mmol/L) | As SAE | Vildagliptin: 1 hemorrhagic stroke gliclazide MR group: 1 death due to AMI, the investigator did not consider the SAEs to be related to the study medications | |
| No major hypoglycemia | ||||||||||||
| Minor hypoglycemia events: 7 in the gliclazide; 2 in the vildagliptin group (P = 0.062) | ||||||||||||
| Hassanein et al[36], 2014 (STEADFAST study) | HE during Ramadan | Multiregional, randomized double-blind | Patients fasting during Ramadan | CHF (NYHA class III or IV); other significant CV history within 6 mo | 557 | 4-wk Ramadan period | Primary | Hypoglycemia: Low BG symptoms with or without confirmatory, SMBG measurement < 3.9 mmol/L; PGE or asymptomatic SMBG < 3.9 mmol/L PGE; confirmed hypoglycemia: symptomatic/asymptomatic SMBG measurement < 3.9 mmol/L; PGE and severe HE requiring assistance from another party irrespective of whether SMBG value was available or not | Confirmed and/or severe HE during Ramadan: vildagliptin vs glicalzide was 3.0% vs 7.0% (P = 0.039; one-sided test); HEs: vildagliptin vs gliclazide was 6.0% and 8.7% (P = 0.173) | - | - | - |
| Filozof and Gautier[37], 2010 | Demonstrate non-inferiority of vildagliptin compared with gliclazide, as an add-on therapy | Randomized, double-blind, active-controlled | T2D uncontrolled with metformin | Serious cardiac conditions (torsades de pointes, sustained and clinically relevant VT or VF, PCI ≤ 3 mo, MI, CABG, unstable angina, or stroke ≤ 6 mo and CHF requiring pharmacological treatment, 2nd- or 3rd-degree AV block or prolonged QTc) | 1007 | 52 wk | AE | Symptoms suggestive of hypoglycemia and confirmed by SMBG < 3.1 mmol/L | HE vildagliptin vs gliclazide (6 vs 11 events) | - | - | - |
Two patients in the gliclazide group and none in the vildagliptin group had ≥ 2 HEs[34]. Though the baseline HbA1c values were slightly higher in the group treated with gliclazide vs the vildagliptin group (HbA1c of 8.7% ± 0.1% vs 8.5% ± 0.1%), the mean HbA1c reduction from baseline to week 104 was -0.5% and -0.6% in the vildagliptin vs gliclazide group[34]. The study could not show the non-inferiority of the DPP4 inhibitor over gliclazide.
Gliclazide + metformin vs linagliptin/DPP4 inhibitors + metformin: There were no records comparing gliclazide + metformin with linagliptin + metformin. Vianna et al[35] compared the glycemic variability of gliclazide MR and vildagliptin and their effect on bone metabolism. This study was the single center part of the BoneGlic Trial, which reported hypoglycemia and MACE as AEs in 42 postmenopausal Brazilian women with T2D and treated with a stable metformin dose for ≤ 3 mo. The study found no difference in time to hypoglycemia and the number of HEs in both the groups (P = 0.062). The investigator did not consider MACE events (Table 2) to be related to study drugs.
The study also found that the gliclazide MR group had a significantly longer time within the target BG range [> 3.9 mmol/L and ≤ 10.0 mmol/L (> 70.27 mg/dL and ≤ 180.18 mg/dL)] and a significantly lower percentage of time with BG > 10 mmol/L (180.18 mg/dL) (P = 0.038 and P = 0.029). In comparison, time within the target BG was insignificantly increased and percentage of time with BG > 10 mmol/L (180.18 mg/dL) was insignificantly lower in the vildagliptin group (P = 0.111 and P = 0.133, respectively). However there were no differences between gliclazide and the DPP4 inhibitor for both the parameters[35].
The STEADFAST study conducted on 557 T2D patients fasting during the holy month of Ramadan found that both gliclazide and vildagliptin as add-on therapy was safe[36]. However, confirmed and/or severe HEs during Ramadan were significantly higher (Table 2) in the glicalzide group[36]. The HEs observed with gliclazide were lower than reported from observational studies. The authors of the STEADFAST study concluded that HEs with gliclazide could be avoided through frequent patient-physician contacts and Ramadan-focused advice[36].
A vildagliptin non-inferiority trial in patients with T2D uncontrolled with metformin demonstrated that as an add-on to metformin, vildagliptin was non-inferior to gliclazide in achieving glycemic control (95% confidence interval: 0.11%-0.20%)[37]. However, more patients in the vildagliptin group discontinued treatment due to an unsatisfactory effect compared with the gliclazide group (n = 22 vs 13, respectively). HEs were lower in the vildagliptin group vs the gliclazide group (6 events vs 11 events)[37].
All three trials[35-37] comparing gliclazide + metformin with DPP4 inhibitor + metformin described in this section were specific to a patient population (post-menopausal women) or in special situation (fasting during Ramadan). Therefore, these trials did not meet the strict inclusion criteria of this narrative synthesis. They were included because there were no other trials retrieved that compared gliclazide with a DPP4 inhibitor as an add-on therapy. The results on these trials may have been influenced by the patient population or the fasting state of the patients.
This section aimed to include randomized trials that compared linagliptin vs gliclazide/SU in a monotherapy setting or compared linagliptin as add-on to metformin vs gliclazide/SU as add-on to metformin.
Linagliptin vs gliclazide or SUs: There were no studies comparing linagliptin with gliclazide or another SU. The landmark “CARdiovascular Outcome study of LINAgliptin vs glimepiride in patients with type 2 diabetes” (CAROLINA)[9] trial and studies[38,39] trial did not meet the inclusion criteria of the narrative synthesis as the study primarily focused on the cardiac and renal patient population. Therefore, other studies[38,39] analyzing the outcomes of interest from the CAROLINA trial were also not included in the narrative synthesis. However, this non-inferiority of linagliptin to glimepiride trial merits discussion as it compared linagliptin with an SU, glimepiride. The trial is covered under the excluded trial section.
However, a study by Barnett et al[40] in “metformin contraindicated” T2D patients compared linagliptin 5 mg once daily with placebo for 18 wk and then compared linagliptin with glimepiride after weeks 18 for 34 wk. The study defined hypoglycemia according to the 2005 American Diabetes Association guidelines[41]. The linagliptin group experienced less hypoglycemia [≤ 70 mg/dL (≤ 3.9 mmol/L)] (2.2% vs 7.8%) and clinical event committee confirmed CV events (0.7% vs 1.6%) than the glimepiride group[40]. However, the difference did not reach clinical significance and more patients in the linagliptin group discontinued treatment due to an AE.
Linagliptin + metformin vs gliclazide/SU + metformin: The literature search did not retrieve any linagliptin + metformin vs gliclazide/SU + metformin studies meeting the inclusion criteria.
The literature search did not retrieve any gliclazide vs placebo studies meeting the inclusion criteria. The main reason for this could be that trials in the initial trajectory of drug development were missed by standardizing the study period from 2008 onwards. Also, there were no trials comparing gliclazide ± metformin with linagliptin ± metformin. Hence, this section aimed to include trials evaluating gliclazide alone or gliclazide + metformin without a comparator and linagliptin alone or linagliptin + metformin without a comparator. These trials were then assessed separately to see if the outcomes of interest could be compared.
Gliclazide ± metformin: Only one trial met the inclusion criteria and is detailed in Table 3. The multicenter, randomized, parallel-group “Diamicron MR in NIDDM: Assessing Management and Improving Control” (DINAMIC 1)[33] trial compared the efficacy, tolerability and acceptability of gliclazide MR for T2D management in the self-monitoring of BG (SMBG) vs non SMBG group. HEs were reported as a safety outcome and were classified as follows: Grade 1, suspected mild hypoglycemia; grade 2, suspected moderate hypoglycemia; grade 3, suspected severe hypoglycemia with need of third-party assistance; and grade 4, suspected severe hypoglycemia with need of medical assistance. In 610 T2D patients (aged 40-80 years) followed up for 6 mo, 8.7% patients in the SMBG group had a total of 51 HEs and 7.0% of patients in the non-SMBG group had a total of 66 HEs. There were no severe (grade 3 or 4) HEs in any group.
| Ref./treatment | Primary study objective | Study design | Study population | CVD excluded | Number of participants | Study duration | Endpoint (hypoglycemia) | Hypoglycemia definition | Hypoglycemia results | Endpoint (MACE) | MACE definition | MACE results |
| Barnett et al[33], 2008/DINAMIC 1/Gliclazide | Compare the efficacy, tolerability and acceptability of gliclazide in SMBG vs non-SMBG group | Multicenter randomized parallel-group | T2D patients managed on diet alone | Not mentioned | 610 | 6 mo | Safety endpoint (AE) | Grade 1: Suspected mild hypoglycemia | SMBG group: 8.7% patients had 51 HE: symptomatic (27), asymptomatic (11), SMBG-confirmed (11) and non-graded (2) | - | - | - |
| Grade 2: Suspected moderate hypoglycemia | Non-SMBG group: 7.0% patients had 66 HE: Symptomatic (66) and non-graded (2). Two HE-related withdrawals | |||||||||||
| Grade 3: Suspected severe hypoglycemia with need of third party assistance | No grade 3 or 4 symptoms | |||||||||||
| Grade 4: Suspected severe hypoglycemia with need of medical assistance | Symptoms suggestive of nocturnal hypoglycemia: SMBG group: 3 and non-SMBG group: 7 | |||||||||||
| Ross et al[42], 2015/Linagliptin/metformin vs linagliptin monotherapy | Change from baseline in HbA1c | Randomized, double-blind, active-controlled, parallel group, multinational | Newly diagnosed (≤ 12 mo) T2D and marked hyperglycemia (≥ 8.5 and ≤ 12.0%) | ACS, stroke or TIA < 3 mo | 316 | 24 wk | Safety endpoint (AE) | Severe hypoglycemia: Requiring assistance from another person to administer carbohydrate or other resuscitative action | Linagliptin/metformin: 1.9% of patients and linagliptin: 3.2% of patients no severe hypoglycemia | - | - | No deaths |
Symptoms suggestive of nocturnal hypoglycemia were experienced by 3 and 7 patients in the SMBG vs non-SMBG, respectively. Two patients withdrew from the study because of hypoglycemia, and both were in the non-SMBG group. The study highlighted the importance of SMBG in T2D management.
Linagliptin ± metformin: Only one trial met the inclusion criteria and is detailed in Table 3. This study compared linagliptin + metformin with only linagliptin and hence was included. Ross et al[42] conducted a randomized study to evaluate the efficacy and safety of initial treatment with linagliptin/metformin combination in newly diagnosed T2D patients with marked hyperglycemia. Hypoglycemia occurred in 1.9% of patients in the linagliptin/metformin and 3.2% of patients in the linagliptin group. No severe HEs was reported[42]. At week 24, there was a significant reduction in HbA1c from baseline in the linagliptin/metformin vs linagliptin group (P < 0.0001 for treatment difference)[42]. Target HbA1c of < 7.0% was achieved by 61% of patients in the linagliptin/metformin arm and 40% of patients in the linagliptin arm[42].
Other studies of linagliptin + metformin[43-45] compared the combination with either metformin or with placebo and hence were not included.
Some landmark and important gliclazide and linagliptin trials were excluded from the narrative synthesis due to the applied exclusion criteria. However, given their importance in the drug trajectory, they require a special mention to obtain a clear picture regarding the HE and MACE AEs associated with gliclazide and linagliptin.
Action in diabetes and vascular disease, Preterax and Diamicron Modified Release Controlled Evaluation trial: Gliclazide studies retrieved during the literature search that reported MACE as an outcome were the “Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation” (ADVANCE)[23] trial and its analyses[46-53]. However, the ADVANCE trial and its analyses were excluded from the narrative synthesis because the ADVANCE trial compared high intensity glucose control (with gliclazide) with standard glucose control (with other SUs). Also, in the high intensity group, those not achieving the targeted HbA1c with highest gliclazide dose were further given metformin, thiazolidinediones, acarbose or insulin as add-on therapy[23]. Comparison studies of gliclazide vs other GLDs (except DPP4 inhibitors) and studies analyzing gliclazide in combinations with other GLDs (except metformin) were excluded from the analysis.
Additionally, the ADVANCE trial recruited patients at high CV risk[23,54]. Patients with a history of stroke, MI, unstable angina, transient ischemic attack and coronary or peripheral vascularization met the inclusion criteria for the study[23,54]. Thus, the ADVANCE trial evaluated the MACE outcome in patients at high risk for MACE. However, the ADVANCE trial also recruited patients with no history of CVD but at high risk of MACE as they had T2D for ≥ 10 years or were ≥ 65-years-old.
The primary macrovascular endpoint of the ADVANCE trial was a composite of CV endpoints (death from CV causes, nonfatal MI or nonfatal stroke). Individual CV endpoints were evaluated as secondary endpoints[23,54]. The trial also evaluated microvascular endpoints both as a composite and individual endpoint[23,54]. During the 5-year follow-up there were no significant effects of the type of glucose control on major macrovascular events[23].
Hypoglycemia was a secondary endpoint of the ADVANCE trial. It was defined as a BG level of < 2.8 mmol/L (< 50.5 mg/dL) or the presence of typical symptoms and signs of hypoglycemia without other apparent causes. Patients with transient dysfunction of the central nervous system requiring external help for treatment were considered to have severe hypoglycemia. During the 5-year follow-up severe hypoglycemia was uncommon. However, it was significantly more common in the intensive-control than standard-control group (2.7% vs 1.5%)[23].
Cardiovascular and Renal Microvascular Outcome Study With Linagliptin trial: The other study of linagliptin vs placebo that reported both HE and MACE as outcomes was the landmark “Cardiovascular and Renal Microvascular Outcome Study With Linagliptin” (CARMELINA) trial. This study was excluded from the narrative synthesis because it evaluated HE and MACE in 6979 T2D patients with high CV and renal risk[8]. However, given that this was a landmark trial, it is discussed in the excluded linagliptin studies section.
This study evaluated HE and MACE in 6979 T2D patients with high CV and renal risk[8]. The trial, designed as a non-inferiority trial of linagliptin vs placebo, assessed the first occurrence of the composite of MACE as a primary endpoint and hypoglycemia was assessed as an AE. The outcomes of interest were well defined according to predefined criteria. After a median follow-up of 2.2 years, MACE occurred in 12.4% and 12.1% in the linagliptin and placebo groups, respectively, and the difference was statistically significant[8]. The frequency of confirmed HEs including severe hypoglycemia in the linagliptin vs placebo group was 15.9% vs 16.4%. HE in the placebo group was due to rescue medications that were allowed to control hyperglycemia[8].
CAROLINA trial: In the CAROLINA trial, 6042 subjects with T2D and HbA1c of 6.5%-8.5% who were at high CV risk (had established CV disease and renal impairment but not end stage renal disease) were randomized to linagliptin at 5 mg/d (n = 3028) vs glimepiride at doses of 1-4 mg/d (n = 3014)[9]. After a mean follow-up of 6.3 years, the primary outcome of the trial (MACE) occurred in 11.8% of subjects in the linagliptin arm vs 12% of subjects in the glimepiride arm, and the difference was statistically significant[9]. At least one HE occurred in 10.6% vs 37.7% of participants in the linagliptin vs glimepiride group, respectively[9].
There were no CVOT trials for gliclazide. The landmark ADVANCE trial[23] compared two levels of glycemic control, intensive (HbA1c < 6.5%) vs standard (managed with oral GLD according to local practice). It was not a CV safety trial of gliclazide, but the trial did show that the primary endpoint of the composite of microvascular and macrovascular events was significantly reduced by 18.1% in the intensive control gliclazide arm.
On the other hand, CV safety of linagliptin has been demonstrated by two RCTs, namely the CARMELINA[8] (vs placebo) and the CAROLINA[9] (vs glimepiride, a SU) trials. These dual randomized CVOT linagliptin trials in T2D patients (CARMELINA[8] and CAROLINA[9]) showed that linagliptin was non-inferior to placebo and glimepiride, respectively, for the composite of MACE.
This CV safety of gliclazide in the ADVANCE trial and of linagliptin in the CARMELINA and CAROLINA trials was demonstrated in patients at high risk of CVD. Hence, gliclazide and linagliptin can be considered as oral GLD that can be given safely in T2D patients with CVD or at high risk of CVD.
In this context, the two RCTs comparing gliclazide with vildagliptin, a DPP4 inhibitor[34,35], were not powered to assess hypoglycemia and MACE as outcomes. Instead, they reported these as AEs. However, neither trial reported a significant difference in CV safety and/or HE incidence between gliclazide and vildagliptin. In this context, it is important to note that linagliptin and vildagliptin belong to two different classes of DPP4 inhibitors[55]. Hence, it is important to compare gliclazide with linagliptin.
Also, all SUs do not have the same CV risks. SUs like glyburide/glibenclamide inhibit an ATP-sensitive potassium channel in the heart and pancreas and are therefore associated with increased CV risk as compared to gliclazide, which selectively inhibits ATP-sensitive potassium channels only in the pancreas[56]. The CARMELINA trial compared linagliptin with glimepiride. However, the double-blind head-to-head comparison GUIDE study showed that compared to glimepiride, gliclazide had a better safety profile and resulted in 50% fewer HEs[2]. The frequency of CV AEs was similar in both glimepiride and gliclazide groups and judged by the investigator as not related to the treatment[2].
Literature was searched using only free resources such as MEDLINE and Google scholar. Hence, the SR is likely to have missed some important articles on the paid sites. The strict inclusion and exclusion criteria is likely to have filtered out important RCTs and real-world studies that could have added value to the CV and hypoglycemia profile of these two drugs. This SR was also limited by its reporting style of narrative synthesis. However, as explained under the “Narrative synthesis of data” section, there were no trials comparing gliclazide and linagliptin. Hence, gliclazide and linagliptin studies were independently assessed for the outcomes of interest. For most studies included in the narrative synthesis, except the CARMELINA[8], ADVANCE[23] and Diamicron MR in NIDDM: Assessing Management and Improving Control 1 study[33], hypoglycemia, MI and other CV events were reported as cause of exclusion from the study or withdrawal from study and non-inclusion in analysis. Hence, these trials looked at outcome of interest in patients, not at risk of CV and renal events.
Filtering of gliclazide trials by the year (2008) resulted in inclusion of trials in the later trajectory of gliclazide compared to linagliptin trials that were in the earlier stage of drug trajectory. This resulted in exclusion of five randomized gliclazide clinical trials that reported the outcomes of interest in the initial drug trajectory[2,57-60] These included trials compared various gliclazide formulations[57,60] and trials comparing gliclazide with other SUs such as the GUIDE Study[2] and with thiazolidinediones (QUARTET Study Group)[58]. However, none of these RCTs included a DPP4 inhibitor as a comparator. Hence, their exclusion did not affect the narrative synthesis.
All the records included in this study were RCTs or a factorial randomized design. Hence, quality of records included was good.
Although, the head-to-head comparative clinical data between gliclazide and linagliptin is lacking, both the drugs have shown effective glycemic control along with CV safety in patients with T2D. In resource limited settings, SUs are commonly used as the first add-on therapy after metformin because of cost constraints. In these settings, there is a need to compare modern Sus like gliclazide, which have a cardiac-sparing action, with drugs with established CV safety in CVOT such as DPP4 inhibitors. Future RCTs may confirm the comparative CV outcomes between gliclazide and linagliptin and other DPP4 inhibitors.
Type 2 diabetes (T2D) patients are at increased cardiovascular and treatment-related hypoglycemia risk. Various guidelines recommend dipeptidyl peptidase-4 (DPP4) inhibitors as the first add-on therapy to metformin in T2D due to their confirmed cardiovascular benefits demonstrated through cardiovascular outcome trials. However, in resource limited countries like India, newer sulfonylureas, like gliclazide and glimepiride, are the most commonly used glucose-lowering drugs in T2D due to their low cost. Gliclazide and glimepiride have similar glycemic efficacy, but gliclazide has a 50% lower hypoglycemia risk.
A landmark cardiovascular outcome trial demonstrated the cardiovascular safety of glimepiride against linagliptin (a DPP4 inhibitor). However, the cardiovascular safety of gliclazide vs linagliptin has not been established through cardiovascular outcome trials. If the cardiovascular safety and lower hypoglycemia risk of gliclazide is established vs linagliptin, it will help physicians prescribe it with assurance of safety for their patients.
To assess the cardiovascular safety and hypoglycemia risk of gliclazide as compared to linagliptin (and other DPP4 inhibitors). The objective was to assess whether gliclazide was as safe as the guideline recommended DPP4 inhibitor (linagliptin) in providing cardiovascular safety and lowering hypoglycemia risk in T2D. This systematic review was likely to help provide assurance regarding cardiovascular and hypoglycemia safety of gliclazide in T2D as compared to costlier DPP4 inhibitors.
This systematic review followed the current Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines to analyze all the clinical studies published from 2008 through the present that compared the cardiovascular safety and hypoglycemia risk of the two drugs in patients with T2D with no cardiovascular disease. Using keywords such as “linagliptin”, “Gliclazide”, “hypoglycemia”, “myocardial infarction”, and “cardiovascular death”, we searched the databases MEDLINE and Google Scholar. Two independent reviewers assessed the trials included using the current Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines for systematic reviews. We included only evidence designated high quality by the Oxford Center for Evidence-based Medicine-Levels of Evidence. The primary outcomes compared were major adverse cardiovascular events and hypoglycemia risk.
We could not find any trial comparing gliclazide with linagliptin, either as monotherapy or as add-on therapy to metformin. The cardiovascular safety of gliclazide in the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) trial and of linagliptin in the Cardiovascular and Renal Microvascular Outcome Study With Linagliptin (CARMELINA) and CARdiovascular Outcome study of LINAgliptin vs glimepiride in patients with T2D (CAROLINA) trials were excluded from the comparative analysis as these trials demonstrated cardiovascular and hypoglycemia benefits in patients at high risk of cardiovascular disease. However, since these are landmark trials, their results are important and hence described in detail as a separate section. The final analysis included five gliclazide and three linagliptin trials (total eight studies) that individually studied the outcomes of interest in T2D patients with no established cardiovascular disease. Statistical comparisons of the results were not possible as the trials had different designs, different definitions of major adverse cardiovascular events and hypoglycemia and were conducted in different patient populations. Hence, no direct comparisons were possible. The trials were therefore described individually, and their results were compared through narrative synthesis. We assessed that both drugs were effective in achieving the desired glycemic control and had low major adverse cardiovascular events and hypoglycemia risk in adult patients with no cardiovascular disease.
Gliclazide can be considered as an effective and safe glucose-lowering drug in T2D patients with no established cardiovascular disease but at high risk of cardiovascular disease due to their T2D status.
Future randomized controlled trials comparing gliclazide with linagliptin or DPP4 inhibitors can add value to the findings of this systematic review.
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work and have given final approval for the version to be published. The authors thank Dr. Punit Srivastava and Dr. Kokil Mathur of Mediception Science Pvt. Ltd (www.mediception.com) for providing medical writing support in the preparation of this manuscript.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country/Territory of origin: India
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): E
P-Reviewer: Feng JF, China; Gluvic Z, Serbia; Ma JH, China; Su G, China S-Editor: Chen YL L-Editor: Filipodia P-Editor: Chen YL
| 1. | Kadowaki T, Yamamoto F, Taneda Y, Naito Y, Clark D, Lund SS, Okamura T, Kaku K. Effects of anti-diabetes medications on cardiovascular and kidney outcomes in Asian patients with type 2 diabetes: a rapid evidence assessment and narrative synthesis. Expert Opin Drug Saf. 2021;20:707-720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 2. | Schernthaner G, Grimaldi A, Di Mario U, Drzewoski J, Kempler P, Kvapil M, Novials A, Rottiers R, Rutten GE, Shaw KM. GUIDE study: double-blind comparison of once-daily gliclazide MR and glimepiride in type 2 diabetic patients. Eur J Clin Invest. 2004;34:535-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 167] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 3. | John M, Kalra S, Nair T. Modern sulphonylureas and cardiovascular adverse effects: Will CAROLINA put an end to the controversy? Indian Heart J. 2020;72:312-315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (3)] |
| 4. | Gallwitz B, Rosenstock J, Patel S, von Eynatten M, Hehnke U, Mehlburger L, Dugi KA, Woerle HJ. Regardless of the degree of glycaemic control, linagliptin has lower hypoglycaemia risk than all doses of glimepiride, at all time points, over the course of a 2-year trial. Diabetes Obes Metab. 2015;17:276-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 5. | Mishriky BM, Cummings DM, Tanenberg RJ. The efficacy and safety of DPP4 inhibitors compared to sulfonylureas as add-on therapy to metformin in patients with Type 2 diabetes: A systematic review and meta-analysis. Diabetes Res Clin Pract. 2015;109:378-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 6. | Ghosh S, Mukhopadhyay P, Pandey P, Chatterjee P, Pandit K. Cardiovascular safety of Glimepiride: An indirect comparison from CAROLINA and CARMELINA. Diab Vasc Dis Res. 2020;17:1479164120973653. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Schnell O, Cos X, Cosentino F, Forst T, Giorgino F, Heersprink HJL, Kosiborod M, Wanner C, Standl E. Report from the CVOT Summit 2020: new cardiovascular and renal outcomes. Cardiovasc Diabetol. 2021;20:75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 8. | Rosenstock J, Perkovic V, Johansen OE, Cooper ME, Kahn SE, Marx N, Alexander JH, Pencina M, Toto RD, Wanner C, Zinman B, Woerle HJ, Baanstra D, Pfarr E, Schnaidt S, Meinicke T, George JT, von Eynatten M, McGuire DK; CARMELINA Investigators. Effect of Linagliptin vs Placebo on Major Cardiovascular Events in Adults With Type 2 Diabetes and High Cardiovascular and Renal Risk: The CARMELINA Randomized Clinical Trial. JAMA. 2019;321:69-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 679] [Cited by in RCA: 784] [Article Influence: 130.7] [Reference Citation Analysis (0)] |
| 9. | Rosenstock J, Kahn SE, Johansen OE, Zinman B, Espeland MA, Woerle HJ, Pfarr E, Keller A, Mattheus M, Baanstra D, Meinicke T, George JT, von Eynatten M, McGuire DK, Marx N; CAROLINA Investigators. Effect of Linagliptin vs Glimepiride on Major Adverse Cardiovascular Outcomes in Patients With Type 2 Diabetes: The CAROLINA Randomized Clinical Trial. JAMA. 2019;322:1155-1166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 372] [Cited by in RCA: 397] [Article Influence: 66.2] [Reference Citation Analysis (0)] |
| 10. | Scirica BM, Bhatt DL, Braunwald E, Steg PG, Davidson J, Hirshberg B, Ohman P, Frederich R, Wiviott SD, Hoffman EB, Cavender MA, Udell JA, Desai NR, Mosenzon O, McGuire DK, Ray KK, Leiter LA, Raz I; SAVOR-TIMI 53 Steering Committee and Investigators. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med. 2013;369:1317-1326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2804] [Cited by in RCA: 2571] [Article Influence: 214.3] [Reference Citation Analysis (0)] |
| 11. | Green JB, Bethel MA, Armstrong PW, Buse JB, Engel SS, Garg J, Josse R, Kaufman KD, Koglin J, Korn S, Lachin JM, McGuire DK, Pencina MJ, Standl E, Stein PP, Suryawanshi S, Van de Werf F, Peterson ED, Holman RR; TECOS Study Group. Effect of Sitagliptin on Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2015;373:232-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1831] [Cited by in RCA: 1872] [Article Influence: 187.2] [Reference Citation Analysis (0)] |
| 12. | White WB, Cannon CP, Heller SR, Nissen SE, Bergenstal RM, Bakris GL, Perez AT, Fleck PR, Mehta CR, Kupfer S, Wilson C, Cushman WC, Zannad F; EXAMINE Investigators. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N Engl J Med. 2013;369:1327-1335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1927] [Cited by in RCA: 1895] [Article Influence: 157.9] [Reference Citation Analysis (0)] |
| 13. | Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, Mattheus M, Devins T, Johansen OE, Woerle HJ, Broedl UC, Inzucchi SE; EMPA-REG OUTCOME Investigators. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med. 2015;373:2117-2128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7124] [Cited by in RCA: 8248] [Article Influence: 824.8] [Reference Citation Analysis (1)] |
| 14. | Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, Shaw W, Law G, Desai M, Matthews DR; CANVAS Program Collaborative Group. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N Engl J Med. 2017;377:644-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4498] [Cited by in RCA: 5372] [Article Influence: 671.5] [Reference Citation Analysis (0)] |
| 15. | Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Silverman MG, Zelniker TA, Kuder JF, Murphy SA, Bhatt DL, Leiter LA, McGuire DK, Wilding JPH, Ruff CT, Gause-Nilsson IAM, Fredriksson M, Johansson PA, Langkilde AM, Sabatine MS; DECLARE–TIMI 58 Investigators. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2019;380:347-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4652] [Cited by in RCA: 4225] [Article Influence: 704.2] [Reference Citation Analysis (0)] |
| 16. | Tiwaskar M. INSIGHTS (INSITES) on DPP 4 Inhibitors (Gliptins) for Diabetes Management in India. J Assoc Physicians India. 2019;67:11-12. [PubMed] |
| 17. | S S, Elizabeth AA, L A. Cost Analysis of SGLT2 Inhibitors in patients with type 2 Diabetes in India. Res J of Pharm and Tech. 2020;13:5861-5865. [DOI] [Full Text] |
| 18. | Roglic G, Norris SL. Medicines for Treatment Intensification in Type 2 Diabetes and Type of Insulin in Type 1 and Type 2 Diabetes in Low-Resource Settings: Synopsis of the World Health Organization Guidelines on Second- and Third-Line Medicines and Type of Insulin for the Control of Blood Glucose Levels in Nonpregnant Adults With Diabetes Mellitus. Ann Intern Med. 2018;169:394-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 65] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 19. | Chawla R, Madhu SV, Makkar BM, Ghosh S, Saboo B, Kalra S; RSSDI-ESI Consensus Group. RSSDI-ESI Clinical Practice Recommendations for the Management of Type 2 Diabetes Mellitus 2020. Indian J Endocrinol Metab. 2020;24:1-122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 70] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 20. | Kalra S, Bahendeka S, Sahay R, Ghosh S, Md F, Orabi A, Ramaiya K, Al Shammari S, Shrestha D, Shaikh K, Abhayaratna S, Shrestha PK, Mahalingam A, Askheta M, A Rahim AA, Eliana F, Shrestha HK, Chaudhary S, Ngugi N, Mbanya JC, Aye TT, Latt TS, Akanov ZA, Syed AR, Tandon N, Unnikrishnan AG, Madhu SV, Jawa A, Chowdhury S, Bajaj S, Das AK. Consensus Recommendations on Sulfonylurea and Sulfonylurea Combinations in the Management of Type 2 Diabetes Mellitus - International Task Force. Indian J Endocrinol Metab. 2018;22:132-157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 59] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 21. | IDF. IDF Clinical Practice Recommendations for Managing Type 2 Diabetes in Primary CareGuidelines. September 28, 2021. [cited 3 September 2022]. Available from: https://www.idf.org/e-library/guidelines/128-idf-clinical-practice-recommendations-for-managing-type-2-diabetes-in-primary-care.html. [RCA] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 119] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 22. | American Diabetes Association. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes-2021. Diabetes Care. 2021;44:S111-S124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 582] [Cited by in RCA: 683] [Article Influence: 170.8] [Reference Citation Analysis (0)] |
| 23. | Szyndler A. [Commentary to the articles: Kaplan NM. Vascular outcome in type 2 diabetes: an ADVANCE? Kardiol Pol. 2007;65:1527-9; discussion 1530. [PubMed] |
| 24. | Zhou JB, Bai L, Wang Y, Yang JK. The benefits and risks of DPP4-inhibitors vs sulfonylureas for patients with type 2 diabetes: accumulated evidence from randomised controlled trial. Int J Clin Pract. 2016;70:132-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | Yu OH, Yin H, Azoulay L. The combination of DPP-4 inhibitors versus sulfonylureas with metformin after failure of first-line treatment in the risk for major cardiovascular events and death. Can J Diabetes. 2015;39:383-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 26. | Eriksson JW, Bodegard J, Nathanson D, Thuresson M, Nyström T, Norhammar A. Sulphonylurea compared to DPP-4 inhibitors in combination with metformin carries increased risk of severe hypoglycemia, cardiovascular events, and all-cause mortality. Diabetes Res Clin Pract. 2016;117:39-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 27. | Foroutan N, Muratov S, Levine M. Safety and efficacy of dipeptidyl peptidase-4 inhibitors vs sulfonylurea in metformin-based combination therapy for type 2 diabetes mellitus: Systematic review and meta-analysis. Clin Invest Med. 2016;39:E48-E62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 28. | Loh HH, Yee A, Loh HS, Sukor N, Kamaruddin NA. Comparative studies of dipeptidyl peptidase 4 inhibitor vs sulphonylurea among Muslim Type 2 diabetes patients who fast in the month of Ramadan: A systematic review and meta-analysis. Prim Care Diabetes. 2016;10:210-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 29. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44932] [Cited by in RCA: 39563] [Article Influence: 9890.8] [Reference Citation Analysis (2)] |
| 30. | Page MJ, Moher D, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, McKenzie JE. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ. 2021;372:n160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4127] [Cited by in RCA: 4593] [Article Influence: 1148.3] [Reference Citation Analysis (0)] |
| 31. | OCEBM Levels of Evidence Working Group; Howick J, Chalmers (James Lind Library) I, et al. The Oxford 2011 Levels of Evidence. Oxford Centre for Evidence-Based Medicine. 2011. [cited September 20, 2021]. Available from: https://www.cebm.ox.ac.uk/resources/levels-of-evidence/ocebm-levels-of-evidence. [DOI] [Full Text] |
| 32. | Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA; Cochrane Bias Methods Group; Cochrane Statistical Methods Group. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18487] [Cited by in RCA: 24683] [Article Influence: 1763.1] [Reference Citation Analysis (3)] |
| 33. | Barnett AH, Krentz AJ, Strojek K, Sieradzki J, Azizi F, Embong M, Imamoglu S, Perusicová J, Uliciansky V, Winkler G. The efficacy of self-monitoring of blood glucose in the management of patients with type 2 diabetes treated with a gliclazide modified release-based regimen. A multicentre, randomized, parallel-group, 6-month evaluation (DINAMIC 1 study). Diabetes Obes Metab. 2008;10:1239-1247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 30] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 34. | Foley JE, Sreenan S. Efficacy and safety comparison between the DPP-4 inhibitor vildagliptin and the sulfonylurea gliclazide after two years of monotherapy in drug-naïve patients with type 2 diabetes. Horm Metab Res. 2009;41:905-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 87] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 35. | Vianna AGD, Lacerda CS, Pechmann LM, Polesel MG, Marino EC, Faria-Neto JR. A randomized controlled trial to compare the effects of sulphonylurea gliclazide MR (modified release) and the DPP-4 inhibitor vildagliptin on glycemic variability and control measured by continuous glucose monitoring (CGM) in Brazilian women with type 2 diabetes. Diabetes Res Clin Pract. 2018;139:357-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 36. | Hassanein M, Abdallah K, Schweizer A. A double-blind, randomized trial, including frequent patient-physician contacts and Ramadan-focused advice, assessing vildagliptin and gliclazide in patients with type 2 diabetes fasting during Ramadan: the STEADFAST study. Vasc Health Risk Manag. 2014;10:319-326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 37. | Filozof C, Gautier JF. A comparison of efficacy and safety of vildagliptin and gliclazide in combination with metformin in patients with Type 2 diabetes inadequately controlled with metformin alone: a 52-week, randomized study. Diabet Med. 2010;27:318-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 110] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 38. | Espeland MA, Pratley RE, Rosenstock J, Kadowaki T, Seino Y, Zinman B, Marx N, McGuire DK, Andersen KR, Mattheus M, Keller A, Weber M, Johansen OE. Cardiovascular outcomes and safety with linagliptin, a dipeptidyl peptidase-4 inhibitor, compared with the sulphonylurea glimepiride in older people with type 2 diabetes: A subgroup analysis of the randomized CAROLINA trial. Diabetes Obes Metab. 2021;23:569-580. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 39. | Kadowaki T, Wang G, Rosenstock J, Yabe D, Peng Y, Kanasaki K, Mu Y, Mattheus M, Keller A, Okamura T, Johansen OE, Marx N. Effect of linagliptin, a dipeptidyl peptidase-4 inhibitor, compared with the sulfonylurea glimepiride on cardiovascular outcomes in Asians with type 2 diabetes: subgroup analysis of the randomized CAROLINA® trial. Diabetol Int. 2021;12:87-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 40. | Barnett AH, Patel S, Harper R, Toorawa R, Thiemann S, von Eynatten M, Woerle HJ. Linagliptin monotherapy in type 2 diabetes patients for whom metformin is inappropriate: an 18-week randomized, double-blind, placebo-controlled phase III trial with a 34-week active-controlled extension. Diabetes Obes Metab. 2012;14:1145-1154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 41. | Workgroup on Hypoglycemia; American Diabetes Association. Defining and reporting hypoglycemia in diabetes: a report from the American Diabetes Association Workgroup on Hypoglycemia. Diabetes Care. 2005;28:1245-1249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 867] [Cited by in RCA: 879] [Article Influence: 44.0] [Reference Citation Analysis (0)] |
| 42. | Ross SA, Caballero AE, Del Prato S, Gallwitz B, Lewis-D'Agostino D, Bailes Z, Thiemann S, Patel S, Woerle HJ, von Eynatten M. Initial combination of linagliptin and metformin compared with linagliptin monotherapy in patients with newly diagnosed type 2 diabetes and marked hyperglycaemia: a randomized, double-blind, active-controlled, parallel group, multinational clinical trial. Diabetes Obes Metab. 2015;17:136-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 43. | Haak T, Meinicke T, Jones R, Weber S, von Eynatten M, Woerle HJ. Initial combination of linagliptin and metformin improves glycaemic control in type 2 diabetes: a randomized, double-blind, placebo-controlled study. Diabetes Obes Metab. 2012;14:565-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 117] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 44. | Haak T, Meinicke T, Jones R, Weber S, von Eynatten M, Woerle HJ. Initial combination of linagliptin and metformin in patients with type 2 diabetes: efficacy and safety in a randomised, double-blind 1-year extension study. Int J Clin Pract. 2013;67:1283-1293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 45. | Ji L, Zinman B, Patel S, Ji J, Bailes Z, Thiemann S, Seck T. Efficacy and safety of linagliptin co-administered with low-dose metformin once daily versus high-dose metformin twice daily in treatment-naïve patients with type 2 diabetes: a double-blind randomized trial. Adv Ther. 2015;32:201-215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 46. | van Steen SC, Woodward M, Chalmers J, Li Q, Marre M, Cooper ME, Hamet P, Mancia G, Colagiuri S, Williams B, Grobbee DE, DeVries JH; ADVANCE Collaborative Group. Haemoglobin glycation index and risk for diabetes-related complications in the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) trial. Diabetologia. 2018;61:780-789. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 47. | Tian J, Ohkuma T, Cooper M, Harrap S, Mancia G, Poulter N, Wang JG, Zoungas S, Woodward M, Chalmers J. Effects of Intensive Glycemic Control on Clinical Outcomes Among Patients With Type 2 Diabetes With Different Levels of Cardiovascular Risk and Hemoglobin A1c in the ADVANCE Trial. Diabetes Care. 2020;43:1293-1299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 48. | de Galan BE, Zoungas S, Chalmers J, Anderson C, Dufouil C, Pillai A, Cooper M, Grobbee DE, Hackett M, Hamet P, Heller SR, Lisheng L, MacMahon S, Mancia G, Neal B, Pan CY, Patel A, Poulter N, Travert F, Woodward M; ADVANCE Collaborative group. Cognitive function and risks of cardiovascular disease and hypoglycaemia in patients with type 2 diabetes: the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) trial. Diabetologia. 2009;52:2328-2336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 164] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 49. | van der Leeuw J, Visseren FL, Woodward M, van der Graaf Y, Grobbee DE, Harrap S, Heller S, Mancia G, Marre M, Poulter N, Zoungas S, Chalmers J. Estimation of individual beneficial and adverse effects of intensive glucose control for patients with type 2 diabetes. Diabetologia. 2016;59:2603-2612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 50. | Zoungas S, Chalmers J, Kengne AP, Pillai A, Billot L, de Galan B, Marre M, Neal B, Harrap S, Poulter N, Patel A. The efficacy of lowering glycated haemoglobin with a gliclazide modified release-based intensive glucose lowering regimen in the ADVANCE trial. Diabetes Res Clin Pract. 2010;89:126-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 51. | Zoungas S, Patel A, Chalmers J, de Galan BE, Li Q, Billot L, Woodward M, Ninomiya T, Neal B, MacMahon S, Grobbee DE, Kengne AP, Marre M, Heller S; ADVANCE Collaborative Group. Severe hypoglycemia and risks of vascular events and death. N Engl J Med. 2010;363:1410-1418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1067] [Cited by in RCA: 1099] [Article Influence: 73.3] [Reference Citation Analysis (0)] |
| 52. | Hirakawa Y, Arima H, Zoungas S, Ninomiya T, Cooper M, Hamet P, Mancia G, Poulter N, Harrap S, Woodward M, Chalmers J. Impact of visit-to-visit glycemic variability on the risks of macrovascular and microvascular events and all-cause mortality in type 2 diabetes: the ADVANCE trial. Diabetes Care. 2014;37:2359-2365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 277] [Article Influence: 25.2] [Reference Citation Analysis (0)] |
| 53. | Lee AK, Woodward M, Wang D, Ohkuma T, Warren B, Sharrett AR, Williams B, Marre M, Hamet P, Harrap S, Mcevoy JW, Chalmers J, Selvin E. The Risks of Cardiovascular Disease and Mortality Following Weight Change in Adults with Diabetes: Results from ADVANCE. J Clin Endocrinol Metab. 2020;105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 54. | ADVANCE Management Committee. Study rationale and design of ADVANCE: action in diabetes and vascular disease--preterax and diamicron MR controlled evaluation. Diabetologia. 2001;44:1118-1120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 150] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 55. | Gallwitz B. Clinical Use of DPP-4 Inhibitors. Front Endocrinol (Lausanne). 2019;10:389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 220] [Article Influence: 36.7] [Reference Citation Analysis (0)] |
| 56. | Abdelmoneim AS, Eurich DT, Gamble JM, Johnson JA, Seubert JM, Qiu W, Simpson SH. Risk of acute coronary events associated with glyburide compared with gliclazide use in patients with type 2 diabetes: a nested case-control study. Diabetes Obes Metab. 2014;16:22-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 57. | Drouin P. Diamicron MR once daily is effective and well tolerated in type 2 diabetes: a double-blind, randomized, multinational study. J Diabetes Complications. 2000;14:185-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 58. | Charbonnel BH, Matthews DR, Schernthaner G, Hanefeld M, Brunetti P; QUARTET Study Group. A long-term comparison of pioglitazone and gliclazide in patients with Type 2 diabetes mellitus: a randomized, double-blind, parallel-group comparison trial. Diabet Med. 2005;22:399-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 106] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 59. | Shimabukuro M, Higa N, Takasu N. Comparison of the antioxidant and vascular effects of gliclazide and glibenclamide in Type 2 diabetic patients: a randomized crossover study. J Diabetes Complications. 2006;20:179-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 60. | Lu CH, Chang CC, Chuang LM, Wang CY, Jiang YD, Wu HP. Double-blind, randomized, multicentre study of the efficacy and safety of gliclazide-modified release in the treatment of Chinese type 2 diabetic patients. Diabetes Obes Metab. 2006;8:184-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |