Published online Mar 15, 2019. doi: 10.4239/wjd.v10.i3.154
Peer-review started: February 18, 2019
First decision: February 19, 2019
Revised: March 10, 2019
Accepted: March 11, 2019
Article in press: March 11, 2019
Published online: March 15, 2019
Processing time: 25 Days and 17.4 Hours
Type 2 diabetes (T2D) is a disorder characterized by chronic inflated blood glucose levels (hyperglycemia), at first due to insulin resistance and unregulated insulin secretion but with tendency towards global spreading. The gut microbiota is recognized to have an influence on T2D, although surveys have not formed a clear overview to date. Because of the interactions between gut microbiota and host homeostasis, intestinal bacteria are believed to play a large role in various diseases, including metabolic syndrome, obesity and associated disease. In this review, we highlight the animal and human studies which have elucidated the roles of metformin, α-glucosidase inhibitors, glucagon-like peptide-1 agonists, peroxisome proliferator-activated receptors γ agonists, inhibitors of dipeptidyl peptidase-4, sodium/glucose cotransporter inhibitors, and other less studied medications on gut microbiota. This review is dedicated to one of the most widespread diseases, T2D, and the currently used antidiabetic drugs and most promising new findings. In general, the gut microbiota has been shown to have an influence on host metabolism, food consumption, satiety, glucose homoeostasis, and weight gain. Altered intestinal microbiota composition has been noticed in cardiovascular diseases, colon cancer, rheumatoid arthritis, T2D, and obesity. Therefore, the main effect of antidiabetic drugs is on the microbiome composition, basically increasing the short-chain fatty acids-producing bacteria, responsible for losing weight and suppressing inflammation.
Core tip: Gut microbiota was found to have an influence on host metabolism, food consumption, satiety, glucose homoeostasis, and weight gain. Altered intestinal microbiota composition has been noticed in cardiovascular diseases, colon cancer, rheumatoid arthritis, type 2 diabetes, and obesity. Therefore, the main effect of antidiabetic drugs is on the microbiome composition, basically increasing the short-chain fatty acids-producing bacteria, responsible for losing weight and suppressing the inflammation.
- Citation: Kyriachenko Y, Falalyeyeva T, Korotkyi O, Molochek N, Kobyliak N. Crosstalk between gut microbiota and antidiabetic drug action. World J Diabetes 2019; 10(3): 154-168
- URL: https://www.wjgnet.com/1948-9358/full/v10/i3/154.htm
- DOI: https://dx.doi.org/10.4239/wjd.v10.i3.154
Over the last few decades, diseases related to metabolic processes, such as type 2 diabetes mellitus (T2D), obesity, dyslipidemia, hypertension and cardiovascular diseases (CVD) have become the main health problems around the world[1]. T2D is a disorder characterized by chronic inflated blood glucose levels (hyperglycemia), at first due to insulin resistance and unregulated insulin secretion but with a tendency towards global spreading. Environmental and genetic factors’ contribution are known, but sedentary life style and dietary habits are not the least constituents. Influence of gut microbiota on T2D is also recognized[2-4], although findings differ between surveys. Because of the interactions between gut microbiota and host homeostasis, gut bacteria are thought to play a great role in diseases, including metabolic syndrome[5-7].
The composition and richness of the gut microbiota is modulated by diet, host health, age, ethnicity and genetics, and thus are unique and highly variable among individuals[3,4]. Turnbaugh et al[8] suggest that there is a “core gut microbiome” that could be responsible for proper gut functioning. That core gut microbial profile predominantly consists of bacteria, which belong to the Gram-positive Firmicutes and the Gram-negative Bacteroidetes[5]. Nevertheless, the increase of intestinal Firmicutes/Bacteroidetes ratio is observed in both obesity and during consumption of energy-rich diets in humans and animal models[5,9,10]. Similar to obesity outcomes, T2D induces a dysbiosis, mainly by reduction in butyrate-producing bacteria[11,12] and in Akkermansia muciniphila, which is now considered a biomarker for glucose intolerance[4].
The bacterial phylotypes found to be correlated with weight are associated with the phyla Firmicutes (2 families and 11 genera), Bacteroidetes (1 family and 2 genera) and Tenericutes (1 family and 1 genus)[3,13]. Among them are five genera affiliated with an increase in weight, including Erysipelotrichaceae incertae sedis, Marvinbryantia, Roseburia, Candidatus arthromitus, and Parabacteroides[3,13]. The phylotypes associated with weight loss were of the genera Lactobacillus, Turicibacter, Anaerostipes, Coprococcus, Blautia, Oscillibacter, and Clostridium[3,13]. For instance, Vrieze et al[14] showed that obesity was associated with modifications in the abundance, diversity, and metabolic function of the gut microbiota, mostly represented as a higher quantity of Firmicutes and a reduced abundance of Bacteroidetes in animal experiments.
Furthermore, one main function of the gut microbiota is to devastate nondigestible carbohydrates into short-chain fatty acids (SCFAs), mostly propionate, acetate and butyrate[15]. Lines of evidence have suggested that intestinal microbiota and SCFAs exert positive effects on glucose-lowering agents in T2D. Glucose-lowering agents can also alter gut microbiota[16], thus meliorating glucose metabolism and energy balance; they also have an influence on the production of SCFAs, thereby providing beneficial effects[12]. Perhaps mechanisms may also affect gene expression, levels of inflammatory cytokines, and the regulation of SCFA synthesis. Furthermore, gut microbiota may attenuate side effects caused by glucose-lowering agents, which is an advantage for diabetic patients. It has even been suggested that human gut microbiota express some enzymes which are capable of binding to and transforming a wide spectrum of bioactive substances[17,18].
Orally-taken medicines reach the gastrointestinal tract and encounter the intestinal microbiota. It has been shown that microbiota-encoded enzymes have the ability to metabolize xenobiotics and to impact the pharmacogenetics of drugs and their bioavailability[19,20]. Accordingly, the gut microbiota may have an influence on drug effectiveness.
Moreover, supplementation with probiotic strains and their combination with nutraceuticals has been demonstrated to provide health benefits in obesity and associated diseases in both animal[21,22] and human studies[23-25].
In this review, we will focus on gut microbiota alterations in obese and T2D patients and its response to currently used antidiabetic drugs (Figure 1). Below, we highlight the animal and human research that has begun to elucidate the role of metformin (1,1-dimetylbiguanide hydrochloride), alpha-glucosidase inhibitors (α-GIs), glucagon-like peptide-1 (GLP-1) agonists, peroxisome proliferator-activated receptors (PPARs) activators, inhibitors of dipeptidyl peptidase-4 (DPP-4) and sodium/glucose cotransporter (SGLT-2), and other less studied medications on gut microbiota.
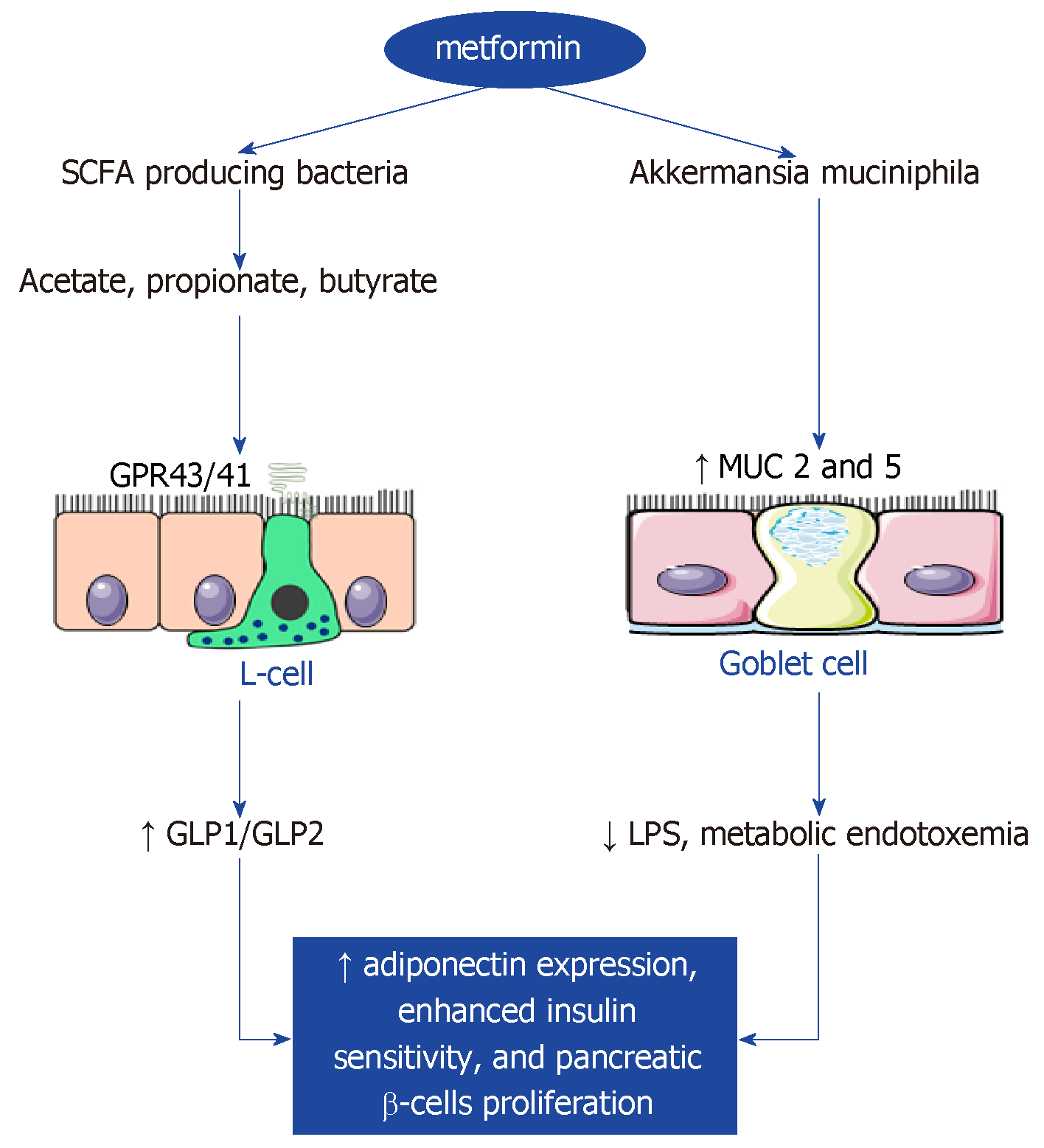
Metformin is the most used nonmetabolizable compound from the biguanide class that patients take orally. It is currently the drug of choice recommended by the American Diabetes Association and the European Association for the Study of Diabetes. Metformin has blood glucose-lowering and insulin sensitizing effects and inhibits liver glucose production. Also, this drug modulates the incretin pathway by improving the expression of GLP-1 receptor in the pancreatic islets and raising plasma levels of GLP-1[5,26,27]. A recent study suggested that inhibition of mitochondrial glycerophosphate dehydrogenase, an enzyme in the glycerophosphate shuttle, could be the main system involved in the metformin-induced inhibition of gluconeogenesis[28].
Treatment with metformin also alters bile acid recirculation[6], suggesting that the primary actions of metformin could be in the gut[11]; however, the absorption of metformin mainly occurs in the small intestine. Moreover, T2D patient treated with metformin can experience improvement in their lipid levels, which would contribute to the reduction of chronic micro- and macrovascular complications. Most of metformin’s pleiotropic effects are predetermined by adenosine monophosphate–activated protein kinase (AMPK) activation in the skeletal muscle and liver[29]. In addition, AMPK activation is known to upregulate autophagic activity through direct phosphorylation of unc-51-like kinase and Beclin 1, key molecules involved in the initiation of autophagy; consequently, metformin can magnify autophagy[30]. Autophagy is valuable for nutrient supply in the case of energy deficiency, has a significant impact on body metabolism, and is also essential for the proper turnover of organelles, such as mitochondria and the endoplasmic reticulum[28]. These organelles play critical roles in pancreatic β-cell physiology and insulin sensitivity. Although, a global increase in autophagic activity is likely to improve the metabolic profile under metabolic stress conditions[28,31], which might be related to attenuation of the chronic low-grade tissue inflammation associated with obesity[28,32].
Metformin treatment is accompanied by the enrichment of SCFA-producing bacteria, such as Blautia, Bacteroides, Butyricoccus, Bifidobacterium, Prevotella, Megasphaera, Butyrivibrio[33] or Phascolarctobacterium, and has positive effects on the Proteobacteria phylum as well as the Allobaculum and Lactobacillus genera[5,32]. Additionally, the composition of the phylum Verrucomicrobia in a high-fat diet (HFD)-Met-treated experimental group was notably raised. Metformin treatment was also shown to have an effect on the gut microbiota in mice on normal diet. Furthermore, the families Rikenellaceae, Ruminococcaceae, and Verrucomicrobiaceae, as well as Alistipes spp., Akkermansia spp., and Clostridium spp., were found to be more abundant with normal diet plus metformin treatment than in the control experimental group[34].
At the genera level, an increase of Escherichia and a decrease of Intestinibacter has been detected in a metformintreated group[33]. The number of positive connections among microbial genera, especially those within Proteobacteria and Firmicutes, was also found to be increased after 2 mo of metformin treatment[33]. After 4 mo of treatment with metformin, there were significantly larger increases in fecal concentrations of lactate and a trend toward a larger increase in fecal concentrations of succinate[33]. In addition, Shin et al[35] showed significant differences in the abundance of Firmicutes and Bacteroidetes and gut microbiota composition between metformin-treated and non-treated mice but only under HFD conditions. Correspondingly, Lee et al[34] observed that metformin caused a decrease in bacterial diversity in mice on HFD.
Recently, the degradation of mucin was reported in mice on HFD[36] and suggested as possibly related to metabolic disorders. After metformin treatment in female mice, but not in male mice, expression of the genes MUC2 and MUC5 in the small intestine became remarkably increased[34]. This is a beneficial effect of the drug, because gastrointestinal mucins produced by goblet cells protect the underlying epithelium from pathogens. Moreover, female hormones are known to exert a protective effect against metabolic disorders[37] and to be involved in lipid and glucose metabolism. Therefore, the observed differences in the gut microbiota between male and female mice during metformin treatment might be caused by differences in hormone levels, which might be associated with metabolic phenotypes[34].
Nevertheless, the abundance of Akkermansia, which are mucin-degrading bacteria, is positively correlated with the quantity of goblet cells. Studies have highlighted that metformin multiplies the number of goblet cells, irrespective of diet[5,35]. Another study showed that treatment with A. muciniphila, which was identified from enterotype gut microbiota, improved metabolic parameters[34,36]. Albeit, approximately 30% of patients report experiencing side effects with metformin, including diarrhea, nausea, vomiting, bloating, and lactic acidosis.
The α-GIs are oral hypoglycemic antidiabetic drugs that postpone the digestion of carbohydrates such as disaccharides and starch in the small intestine, and which reduce postprandial hyperglycemia[5] and delay the absorption of glucose, thereby managing blood glucose levels and related complications. To this class belong acarbose, voglibose (also naturally occurring in Streptomyces) and miglitol. Thus, α-GIs alter the nutrient sources of bacteria by segregating complex carbohydrates.
Acarbose delays the enzymatic carbohydrates decaying in the small intestine and thereby diminish postprandial hyperglycemia. Clinical studies have shown that acarbose significantly enhances glycemic control and lowers known CVD risk factors, including triglycerides’ levels, body mass index, insulin levels, and systolic blood pressure[38-40]. The prominent mechanisms for this cardiovascular protective function are only partially understood, but they can be attributed to the ability of acarbose to neutralize oxidative stress by increasing H2 production in the gastrointestinal tract[38,41]. Panwar et al[42] found that Lactobacillus strains exert effects of glucosidase-inhibitors and regulate blood glucose responses to carbohydrates in vivo. As mentioned above, SCFAs play a crucial role in diabetes. In individuals with impaired glucose tolerance, acarbose was found to increase serum butyrate levels. The underlying mechanism for this effect might be that acarbose increases the fermentation of insoluble fibers in the colon. Interestingly, oral supplementation of butyrate was found to improve insulin sensitivity and increase energy expenditure by enhancing mitochondrial work in mice[38,43]. To minimize the known gastrointestinal side effects of acarbose (e.g., flatulence, diarrhea, or abdominal cramps), the drug was administered in small proportions. Zhang et al[39] compared treatment for T2D with acarbose and metformin and showed that both treatments notably increased GLP-1 concentration and decreased glucagon after 24 wk. However, additional benefits of acarbose besides its antidiabetic effect remain unknown.
Acarbose is effective in lowering blood glucose level in patients with T2D by delaying the digestion of complex carbohydrates through the inhibition of pancreatic α-amylase and a variety of α-glucosides[38]. Later, microbiota will ferment these carbohydrates, which will alter the composition of the intestinal microbiota. The features of gut microbiota in patients with prediabetes (before treatment) are genera abundance of Bacteroides (belonging to Bacteroidetes) and Faecalibacterium (belonging to Firmicutes). The most plentiful phyla include Firmicutes (68.53% of all reads), Bacteroidetes (27.85% of all reads), Proteobacteria (1.98% of all reads), and Actinobacteria (0.98% of all reads)[38].
After the treatment with acarbose, five genera, including Lactobacillus and Dialister, flourished. In response to acarbose, Lactobacillaceae, Ruminococcaceae and Veillonellaceae increased and six genera, including Butyricicoccus, Phascolarctobacterium and Ruminococcus, decreased. Likewise, many of the operational taxonomic units that greatly increased in response to acarbose belong to SCFA-producing taxa, such as Faecalibacterium, Prevotella, and Lactobacillus[38]. Some species of Megasphaera also thrived following acarbose treatment. They can transform carbohydrates into SCFAs, including butyrate, formate, acetate, valerate, and caproate, a process which is valuable for Lactobacillus development[38]. Consequently, the effect of acarbose on body weight might be related to reorganized microbiota structure. It is supposed that SCFAs such as acetate, butyrate and propionate and their concentrations are prognostic of lifespan[38]. Thus, the increased levels of SCFAs in acarbose-treated mice may lead to the beneficial effect on the lifespan[44]. However, studies have shown that the weight loss in female mice on acarbose was more dramatic than in males, while the longevity effect is much stronger in males[45].
Another α-GI, voglibose, changes dysbiosis in diet-induced obese mice[46]. These changes could increase the production of bile acid metabolites and have an advantageous systemic outcome. Specifically, scientists have found favorable effects of voglibose on several cardiovascular end-points, as it improves glycemic control in mice with cardiac overpressure[47]. Voglibose has anti-obesity effects on diet-induced obese mice. Possibly, the effects of incretins in voglibose, activation of neuroendocrine linked to leptin, and inducement of the genes responsible for magnified energy metabolism cause the reduction in energy intake and improvement of mitochondrial function[48]. The reduction in food intake is possibly derived from increased GLP-1 levels due to voglibose supplementation or from direct modulation of hypothalamic genes which lead to the satiety response.
Miglitol shortens the intestinal transit time and suppresses histological and molecular markers of inflammation, for which concentrations are elevated by a high-fat and high-glucose diet and which shifts with the increases in Erysipelotrichaceae and Coriobacteriaceae induced by the energy-rich diet[49]. Miglitol is able to alter human gut microbiota because of the transit time reduction.
The development of nonalcoholic steatohepatitis (NASH) could be dependent on the gut environment as well. As α-GIs change the gut environment, they might also protect against NASH development, because of its sensibility to changes in the gut environment[49]. NASH is characterized by hepatocellular lipid accumulation along with inflammation and fibrosis that is a precondition for oxidative stress, inflammatory cytokines, and endotoxins. There is an essential need for therapeutic interventions considering that NASH can lead to cirrhosis and liver cancer. In addition, acarbose was also demonstrated to have a protective effect against NASH development in HFD-induced obese rats[50]. However, the underlying mechanisms should be further investigated.
Miglitol was shown to restrain the accumulation of lipid droplets and in-flammatory cell infiltration, and to lead to a decrease in the numbers of ballooning hepatocytes as well as to stoppage of the activation of stellate cells, which plays a role in liver fibrosis[49].
The administration of an α-GI has been found to increase the levels of butyric acid in the intestines of healthy individuals. Indeed, the administration of butyric acid was demonstrated to suppress intestinal inflammation in mice[51]. These findings suggest that miglitol administration increases the butyric acid level in the intestine and suppresses colon inflammation.
Human gut bacterium Blaubia (Ruminococcus) obeum expresses enzymes, such as α-glucosidases (Ro-αG1), which have specific crystal structures with free active site(s) to bind and interact with volatile substrates. Therefore, the proposed theory is that α-GIs (acarbose, voglibose, miglitol) can affect the bacterial Ro-αG1 in human gut and exert positive effects or create adverse gastrointestinal symptoms[52]. The α-GIs bind to the active site of Ro-αG1 and change the enzyme’s activity. Acarbose was found to slightly inhibit the gut bacterial α-glucosidases as well as other currently used α-GIs.
Intestinal endocrine cells (L cells) respond to food ingestion by secreting GLP-1, an incretin hormone[53]. This hormone can intensify glucose-induced insulin from pancreatic β-cells and suppress glucagon secretion; in addition, it can protect pancreatic β-cells from apoptosis and promote β-cell proliferation. Together, incretins are liable for 50%–60% of postprandial insulin secretion. In addition, GLP-1 plays critical roles in gastrointestinal motility as well as in metabolism; moreover, GLP-1 can possibly suppress gastrointestinal motility, thus affecting the absorption of digested food[54]. However, the natural GLP-1 is degraded rapidly, primarily through enzymatic destruction by DPP-4. Therefore, another pharmaceutical approach to treat T2D is to increase GLP-1 function, either by the administration of GLP-1 peptide mimetics or suppressing its degradation by DPP-4. GLP-1 expression could be stimulated by binding of SCFAs and secondary bile acids (lithocholic acid and deoxycholic acid) with the G-coupled protein receptor FFAR2 (formerly GPR43)[53]. Many studies have shown that satiety and glucose homoeostasis are modulated by the gut microbiota that induces the secretion of GLP-1[55,56].
The body weight control induced by GLP-1 is maintained by reduced food intake and inhibition of appetite and gastric emptying[57]. However, restricted dietary intake supplemented with GLP-1 results in more significant weight loss. Interestingly, the microbial diversity after GLP-1 increment seemed to be dependent on the glycemic state of the mice studied. In normoglycemic mice treated with liraglutide and saxagliptin, bacterial variety significantly decreased, while in transiently hyperglycemic mice it rose to the normal level[13]. Of late, scientists have proposed liraglutide as a prospective anti-obesity drug because of its additional impact on weight loss in obese and diabetic individuals. The daily injection of liraglutide has been shown to significantly improve glucose tolerance and insulin tolerance in diabetic rats rather than in nondiabetic rats. Although, it was found to alter the microbial composition in both simple obese and diabetic obese rats. The genera Candidatus, Roseburia, Arthromitus and Marvinbryantia may promote weight gain, while the genera Coprococcus and Lactobacillus are associated with weight loss[58].
Liraglutide administration has been shown to decrease the relative abundance of all of the obesity-related phylotypes (such as Romboutsia, Ruminiclostridium, and Erysipelotrichaceae) and to enrich the lean-related genera Blautia and Coprococcus[59]. After liraglutide intervention, the abundance of Firmicutes was also found to tend towards decrease in obese rats, whereas the finding was contrary when the study was carried out in human volunteers under field conditions without restriction[60]. Patients with long duration of T2D show a significantly reduced Akkermensia variety. After comparison of the gut microbiota of subjects receiving a GLP-1 agonist and metformin, higher Akkermansia abundances were detected in the liraglutide-treated patients[61]. At first, the genus Akkermansia and some genera in the family Christensenellaceae increased prominently under liraglutide, unlike that seen under metformin, with the latter of which leading to a greater expansion of Dorea and Sutterella genera.
GLP-1 receptors are placed on neurons innervating the portal vein, on β cells of the pancreas, and the central nervous system[62]. GLP-1 after its release can affect afferent neurons innervating the gastrointestinal tract which signal to the caudal brainstem or enteric neurons, and/or they can enter the circulation to functionate centrally, or on peripheral targets to regulate metabolic disorders[59,63]. Thus, the weight-loss and glucose-controlling effects of liraglutide are possibly mediated by the gut–brain axis. In addition, the GLP-1 analog liraglutide reduces visceral hypersensitivity and acts as a sort of pain-killer[64]. ROSE-010, another GLP-1 analog, has been shown to diminish visceral pain in patients suffering from irritable bowel syndrome[65].
Long-term HFD intake has been shown to result in a lack of energy substrates, reduced acetylcholine synthesis, membrane deterioration and oxidative stress, and consequently is valid in intestinal myenteric neurons loss in mice[66]. Grasset et al[67] revealed that gut microbiota dysbiosis causes the loss of enteric neurons, attenuated nitric oxide production and following GLP-1 resistance. Nitric oxide produced by nuclear nitric oxide synthase has been generally expected to have a protective effect on enteric neurons that is greater than its damaging effect. The GLP-1 secretagogue l-arginine oral administration has also been shown to improve glucose tolerance by influencing GLP-1R signaling[68]. L-arginine is a substrate of nuclear nitric oxide synthase which improves GLP-1 sensitivity in HFD-fed mice and increases the glucose-induced insulin secretion. In consequence, l-arginine supplementation could have beneficial effects on postprandial GLP-1 response and GLP-1 sensitivity in patients with T2D.
Protein CD26 present on the lymphocyte cell surface was first described to have a proteolytic activity in 1966 and was later named as DPP-4. The potential substrates of DPP-4 activity are gut hormones, like incretins, GLP-1 and gastric inhibitory polypeptide, neuropeptides, chemokines, and dietary proteins[69]. Through the cleavage of key hormones and peptides, the DPP-4 activity influences behavioral, intestinal and metabolic disorders[70]. DPP-4 can enhance the agonistic activity of gut hormones, like neuropeptide Y and peptide YY (PYY), by cleaving off the N-terminal dipeptide. As PYY has an influence on the ileal and colonic brake of digestion and on the induction of satiety via activation of hypothalamic Y2 receptors, these effects could be enhanced by DPP-4[71]. However, the capacity of DPP-4 in the regulation of satiety has not yet been fully elucidated.
An experiment on Dpp-4 knockout mice showed an increased GLP-1 level and improved glucose tolerance associated with it. This effect was achieved in humans by administration of DPP-4 inhibitors, antidiabetic agents that maintain incretins in their active form[72]. Moreover, Ahmed et al[73] revealed that overweight and obese patients, in comparison to normal-weight patients, have increased DPP-4 activity, which decreases the activity of GLP-1. The administration of prebiotics led to decreased DPP-4 activity, which was explained by double concentration in the active form of GLP-1[74].
Some studies have acknowledged that some commensal bacteria, such as Prevotella or Lactobacillus, can express human DPP-4 homologs[70,75]. Two groups of scientists published findings of DPP-4 expression being higher in gnotobiotic mice colonized with feces of a lean subject that in germ-free mice, which strongly indicates that their intestinal microbiota produced DPP-4-like activity[70,76]. The overall literature affirms that DPP-4 encoded by the gut microbiota could compose an innovative mechanism to alter protein digestion, host metabolism, and behavior.
The novel type of DPP-4 inhibitors are sitagliptin, saxagliptin and vildagliptin, which are administrated orally. Sitagliptin increases insulin and suppresses glucagon secretion. In patients with T2D, therapies which include DPP-4 inhibitors promote healing of colitis and diminish depression symptoms. DPP-4 was proposed as a possible target for treating autoimmune diseases, including inflammatory bowel disease as it has an influence on the immune system, particularly on T cell function[77]. Obese dams with adverse pregnancy outcomes have been reported as having decreased Lactobacillus spp. compared with dams with normal litters. Secondly, maternal obesity and reduced fertility are related to bad pregnancy outcomes, gestational diabetes, and preeclampsia[78]. Sitagliptin or prebiotic consumption during pregnancy could normalize gestational weight gain, increase Bifidobacterium spp., reduce fasting glucose levels, and possibly alleviate pregnancy termination associated with maternal obesity while improving offspring metabolic health and composition of the intestinal microbiome[79].
Saxagliptin, another DPP-4 inhibitor, appears to act only on a small target group of gut microbes, mainly on the Firmicutes/Bacteroides ratio. It has been shown to enhance the development of the genus Lactobacillus within the class Lactobacillaceae, the genera Allobaculum and Turicibacter within class Erysipelotrichaceae and suppresses the genus Bacteroides within the class Bacteroidaceae, and the genus Prevotella within the class Prevotellaceae. As compared to the GLP-1 agonist liraglutide, saxagliptin reduced the enrichment of the genus Blautia and of the genus Coprococcus. Moreover, despite the fact of similar food intake reduction, saxagliptin had a neutral effect on body weight, with subjects on liraglutide having significantly lower weight regardless of their glycemic control[13].
Another DPP-4 inhibitor, vildagliptin, affects the gut microbiota composition and its metabolic activity. Vildagliptin has been shown to reduce the fasting blood glucose and HbA1c levels. In obese murine models, vildagliptin reduced Ruminococcaceae, such as the genera Oscillibacter, Ruminiclostridium_6, Anaerotruncus, and Ruminococcaceae_UCG_007, as well as the families Planococcaceae, Christensenellaceae, and Prevotellaceae. The enriched phylotypes were of the Streptococcaceae family, the genera Bacteroides, and the family Bacteroidaceae. In general, it modified the Firmicutes/Bacteroides ratio, reduced DPP-4 activity in the portal vein and increased the concentration of active GLP-1, improving gastrointestinal function according to AMPs’ expression restoration and the depth of the crypts in the ileum[80]. Vildagliptin also has a potential effect on inflammation, due to reduction of TLR and cytokine expression. Zhang et al[80] suggested that vildagliptin enriches SCFA-producing bacteria and ameliorates gastrointestinal health and could ultimately mediate their beneficial effects on the host, especially in diabetes.
The DPP-4 inhibitor PKF-275-055 improves glucose/cholesterol metabolism, decreases Firmicutes/Bacteroidetes ratio, and drops mass gain and mesenteric adipose accumulation. Moreover, mice on HFD with PKF-275-055 treatment showed enriched butyrate-producing Rumminococcus and of the acetogen Dorea compared to the control group[81].
The sodium/glucose cotransporters SGLT1 and SGLT2 are generally expressed in the small intestine and are regulated by a sodium gradient created by Na+/K+ ATPase. SGLT1 transports glucose and galactose across the apical membrane of enterocytes, whereas SGLT2, and at some extent SGLT1, reabsorbs glucose in the renal tubule. SGLT2-selective inhibitors are a new class of treatment for T2D[82]. SGLT2 differs from other antidiabetic medications because it ameliorates vascular function and thus has advantageous effects on CVD. Among the numerous T2D consequences, CVD is the most widespread and dramatic. Data characterize the gut microbiota as an important regulator of vascular function[83]. Indeed, people with diabetes have vascular dysfunction and heightened risk of CVD. Distinct signs of diabetes-related CVD are arterial stiffness, endothelial dysfunction, and vascular smooth muscle functional disorder. Reduction in endothelium-independent dilation is present in T2D patients and is a marker of cardiovascular complications.
After 8 wk of treatment with a selective SGLT2 inhibitor dapagliflozin, diabetic mice showed lower arterial stiffness and blood glucose level, and improvements in endothelial and vascular smooth muscle dysfunctions compared to nontreated diabetic mice. In addition, reductions in circulating inflammatory markers, such as MCP-1, IL-1β and IL-6, and hyperglycemia improvement were detected[84]. Animals with diabetes treated with dapagliflozin showed decreased Firmicutes/Bacteroidetes ratio and Oscillospira, and increased Akkermansia muciniphila.
The dual SGLT1/2 inhibitor has been shown to reduce blood glucose levels and HbA1c and to increase total GLP1 in mice fed a high-sucrose diet[85]. The higher doses of SGLT1/2 inhibitor accelerated body weight gain and increased Bacteroidetes and decreased Firmicutes quantity, but the Akkermansia spp. was not modified[82]. SGLT1/2 inhibitors or SGLT2-selective drugs like canagliflozin guarantee intestinal SGLT1 inhibition in T2D[86]. They enhance GLP-1 and PYY secretion and delay the glucose excursion after carbohydrate intake. The dual SGLT1/2 inhibitor LX4211/sotagliflozin, in clinical testing, has the same effects as the medicine described directly above[87]. However, SGLT1 cannot be completely inhibited as long as changed Na+ homeostasis and elevated colonic carbohydrates can lead to the opposite gastrointestinal effects and diarrhea.
The PPARs belong to the nuclear receptor family of regulatory factors that are ligand-activated transcription factors. There are two PPARγ isoforms: PPARγ1 and PPARγ2. They form a heterodimeric complex with the retinoid X receptor, which binds to PPAR-responsive elements and then regulates transcription. Its main functions are linked to maintaining homeostasis in the intestine, inducing adipocyte growth and differentiation[88], cellular apoptosis[89], regulation of genes involved in glucose and lipid metabolism[90], and inflammatory responses. PPARγ is expressed mainly in several tissues such as of the lungs, breast, ovaries, placenta and at most in the colon, where it regulates colonocyte metabolism and cell cycle[91].
PPARγ is considered to have anti-inflammatory effects and to be a molecular target for cancer chemoprevention[92]. It also enhances insulin sensitivity and regulates the genes involved in hypertension and contributing to atherosclerosis. There are lines of evidence indicating reduction of intestinal inflammation and colon cancer development and T2D[93] by specific PPARγ agonists.
Nepelska et al[91] engineered a colonic epithelial HT-29-PPARγ reporter cell line to control the influence of bacterial metabolites on transcriptional activity of PPARγ. Two main metabolites of intestinal bacteria, butyrate and propionate, were linked to activation of PPARγ transcriptional activity. Notwithstanding, phylogenetic affiliation of the strains were found to not rigorously correspond to reporter gene activities, among them the most general stimulating effect was noticed for Firmicutes and Fusobacteria, while Actinobacteria exerted moderate or no modulation.
The strongest potential of PPARγ activation is exerted on Roseburia hominis, Roseburia intestinalis ,and Fusobacterium naviforme. These are well-known producers of butyrate, therefore the response pattern of PPARγ reporter cells is exposed to the composition of organic acids of conditioned media. Gene regulation in intestinal epithelial cells is known to be regulated by the SCFAs, especially butyrate[94]. Acetate negatively affected the PPARγ reporter system, demonstrating a reverse correlation. Moreover, butyrate and propionate stimulated PPARγ activity, even at such low concentrations as 0.5 mM. Acetate, however, showed an insignificant activation starting from 8 mM, and lactate did not affect the activity but was cytotoxic from 2 mM, which lead to cell detachment[95]. In general, high concentrations of all organic acids, especially acetic and lactic, have a deleterious effect on the viability of cells, possibly because of decrease in pH.
Some species can activate the expression of PPARγ target genes even without presence of butyrate and propionate in their conditioned media. To them belong Atopobium parvulum and Prevotella copri. These bacteria were shown to increase ANGPTL4 and ADRP expression in HT-29 cells. Their underlying mechanisms are probably different, because stimulation with conditioned media of A. parvulum showed its influence after 6 h аnd P. copri in 12 h[91]. The induction of ANGPTL4 by the PPARγ-specific ligand troglitazone was weaker in either case. In addition, A. parvulum and P. copri promote PPARγ phosphorylation via ERK1/2[91]. Studies confirmed that bacterial upregulation of PPARγ in enteral epithelial cells occurs by phosphorylation[96]. Though, high levels of P. copri and A. parvulum in the intestine has been connected to arthritis[97] and linked to periodontitis[98] accordingly.
However, the elevated risk of cardiovascular ischemic events is affiliated with the use of such PPARγ ligands as rosiglitazone. PPARγ agonists are used in clinics despite the fact that they have serious adverse effects, such as heart failure, weight gain, and increased bone fracture[99]. At present, natural products could be found as a source of drugs[100].
Fish oil has established favorable effects in diabetes, CVDs, autoimmune inflammatory diseases, and inflammatory bowel diseases. Neschen et al[101] revealed advantageous effects of fish oil on glucose and lipid metabolism, improvement of insulin sensitivity, and reduction of triglycerides. Additionally, the n-3 polyunsaturated fatty acids of fish oil, eicosapentaenoic acid and docosahexaenoic acid, are endogenous ligands for PPAR; consequently, even small changes in their structures affect PPAR activation. These fatty acids adjust the insulin-sensitizing, anti-inflammatory and lipid-lowering properties of fish oil[102].
Over the past 10 yr, an increasing body of literature has suggested that the gut microbiota plays a crucial role in the host immune system, modulation of inflammatory processes, extraction of energy from the host diet, and alterations of human gene expression, and is considered to make an important impact on obesity/insulin resistance development. Several mechanisms that contribute to explaining the link between altered gut microbiota and pathogenesis of insulin resistance have been described[7]. They control the fermentation and absorption of dietary polysaccharides to produce SCFAs, which may explain their importance in the regulation of fat accumulation. SCFAs can stimulate the secretion of GLP-1 and GLP-2, thus increasing insulin and adiponectin expression, which might contribute to enhanced insulin sensitivity and pancreatic ß-cells proliferation[103,104]. Another mechanism by which the microbiome may contribute to insulin resistance is compromised gut barrier function with an increased intestinal permeability, accumulation of lipopolysaccharide, and metabolic endotoxemia development[105].
Lactobacillus and Bifidobacterium are commonly used as probiotics and are the most studied strains in the treatment and prevention of obesity-associated disorders[15]. Moreover, several potential bacterial candidates, such as Saccharomyces cerevisiae var. boulardii, Parabacteroides goldsteinii, Enterobacter halii or Akkermansia muciniphila, have been identified and innovative mechanisms of action overriding their beneficial effects for insulin resistance/obesity have been elucidated[106,107].
The abundance of Akkermansia muciniphila, which is a mucin-degrading bacterium that resides in the mucus layer, has been found to be decreased in obese/T2D and inversely correlated with body weight in both rodents and humans[34]. Metformin treatment[35,36], consumption of oligofructose[108], dietary concord grape polyphenols[109], and gastric bypass surgery in humans[110] and mice[111] leads to a marked increase in A. muciniphila abundance with subsequent weight loss and reversed metabolic disorders, including fat-mass gain, metabolic endotoxemia, adipose tissue inflammation, and insulin resistance[34].
F. prausnitzii plays an important role in preserving the gut barrier and controlling inflammation and T2D progression[112]. A traditional Chinese berberine-containing herbal formula given to T2D patients[112,113] changed the gut microbiota by increasing F. prausnitzii, which was negatively correlated with fasting blood glucose, HbA1c and postprandial blood glucose levels, and positively correlated with homoeostasis model assessment of β-cell function (commonly known as the HOMA-B).
Parabacteroides goldsteinii is a commensal bacterium with reduced level in HFD-fed mice. Oral treatment of HFD-fed mice with live P. goldsteinii reduced obesity and was found to be associated with increased adipose tissue thermogenesis, enhanced intestinal integrity, and reduced levels of inflammation and insulin resistance[106].
Identifying the most important microbiota-related metabolic pathways could lead to the development of integrated strategies using new prebiotics or beneficial bacterial strains to prevent and treat these metabolic disorders in the near future[112].
This review is dedicated to one of the most widespread diseases, diabetes, and the currently used antidiabetic drugs and possible promising new findings in this field. The gut microbiota has been found to have an influence on host metabolism, food consumption, satiety, glucose homoeostasis, and weight gain. Altered intestinal microbiota composition has been noticed in CVDs, colon cancer, rheumatoid arthritis, diabetes, and obesity. Therefore, the main effect of antidiabetic drugs is thought to be on the microbiome composition, basically increasing the SCFA-producing bacteria responsible for losing weight and suppressing inflammation. Scientists have found that some drugs for T2D also elicit favorably effects on several cardiovascular end points and have anticancer as well as anti-aging effects. However, further detailed experimental and clinical investigations should be conducted to gain a deeper understanding of antidiabetic drugs’ functions.
Manuscript source: Invited manuscript
Specialty type: Endocrinology and metabolism
Country of origin: Ukraine
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Das U S- Editor: Wang JL L- Editor: Filipodia E- Editor: Wu YXJ
| 1. | Mykhalchyshyn G, Kobyliak N, Bodnar P. Diagnostic accuracy of acyl-ghrelin and it association with non-alcoholic fatty liver disease in type 2 diabetic patients. J Diabetes Metab Disord. 2015;14:44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 2. | Qin J, Li Y, Cai Z, Li S, Zhu J, Zhang F, Liang S, Zhang W, Guan Y, Shen D, Peng Y, Zhang D, Jie Z, Wu W, Qin Y, Xue W, Li J, Han L, Lu D, Wu P, Dai Y, Sun X, Li Z, Tang A, Zhong S, Li X, Chen W, Xu R, Wang M, Feng Q, Gong M, Yu J, Zhang Y, Zhang M, Hansen T, Sanchez G, Raes J, Falony G, Okuda S, Almeida M, LeChatelier E, Renault P, Pons N, Batto JM, Zhang Z, Chen H, Yang R, Zheng W, Li S, Yang H, Wang J, Ehrlich SD, Nielsen R, Pedersen O, Kristiansen K, Wang J. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490:55-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3971] [Cited by in RCA: 4832] [Article Influence: 371.7] [Reference Citation Analysis (1)] |
| 3. | Karlsson FH, Tremaroli V, Nookaew I, Bergström G, Behre CJ, Fagerberg B, Nielsen J, Bäckhed F. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature. 2013;498:99-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1766] [Cited by in RCA: 2148] [Article Influence: 179.0] [Reference Citation Analysis (0)] |
| 4. | Zhang X, Shen D, Fang Z, Jie Z, Qiu X, Zhang C, Chen Y, Ji L. Human gut microbiota changes reveal the progression of glucose intolerance. PLoS One. 2013;8:e71108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 481] [Cited by in RCA: 634] [Article Influence: 52.8] [Reference Citation Analysis (0)] |
| 5. | Montandon SA, Jornayvaz FR. Effects of Antidiabetic Drugs on Gut Microbiota Composition. Genes (Basel). 2017;8:E250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 97] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 6. | Napolitano A, Miller S, Nicholls AW, Baker D, Van Horn S, Thomas E, Rajpal D, Spivak A, Brown JR, Nunez DJ. Novel gut-based pharmacology of metformin in patients with type 2 diabetes mellitus. PLoS One. 2014;9:e100778. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 223] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 7. | Kobyliak N, Virchenko O, Falalyeyeva T. Pathophysiological role of host microbiota in the development of obesity. Nutr J. 2016;15:43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 104] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 8. | Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE, Sogin ML, Jones WJ, Roe BA, Affourtit JP, Egholm M, Henrissat B, Heath AC, Knight R, Gordon JI. A core gut microbiome in obese and lean twins. Nature. 2009;457:480-484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6397] [Cited by in RCA: 5669] [Article Influence: 354.3] [Reference Citation Analysis (1)] |
| 9. | Woting A, Blaut M. The Intestinal Microbiota in Metabolic Disease. Nutrients. 2016;8:202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 209] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 10. | Murphy EF, Cotter PD, Healy S, Marques TM, O'Sullivan O, Fouhy F, Clarke SF, O'Toole PW, Quigley EM, Stanton C, Ross PR, O'Doherty RM, Shanahan F. Composition and energy harvesting capacity of the gut microbiota: relationship to diet, obesity and time in mouse models. Gut. 2010;59:1635-1642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 647] [Cited by in RCA: 709] [Article Influence: 47.3] [Reference Citation Analysis (0)] |
| 11. | Brunkwall L, Orho-Melander M. The gut microbiome as a target for prevention and treatment of hyperglycaemia in type 2 diabetes: from current human evidence to future possibilities. Diabetologia. 2017;60:943-951. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 231] [Cited by in RCA: 252] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 12. | Forslund K, Hildebrand F, Nielsen T, Falony G, Le Chatelier E, Sunagawa S, Prifti E, Vieira-Silva S, Gudmundsdottir V, Pedersen HK, Arumugam M, Kristiansen K, Voigt AY, Vestergaard H, Hercog R, Costea PI, Kultima JR, Li J, Jørgensen T, Levenez F, Dore J; MetaHIT consortium, Nielsen HB, Brunak S, Raes J, Hansen T, Wang J, Ehrlich SD, Bork P, Pedersen O. Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature. 2015;528:262-266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1208] [Cited by in RCA: 1513] [Article Influence: 151.3] [Reference Citation Analysis (0)] |
| 13. | Wang L, Li P, Tang Z, Yan X, Feng B. Structural modulation of the gut microbiota and the relationship with body weight: compared evaluation of liraglutide and saxagliptin treatment. Sci Rep. 2016;6:33251. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 112] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 14. | Vrieze A, Holleman F, Zoetendal EG, de Vos WM, Hoekstra JB, Nieuwdorp M. The environment within: how gut microbiota may influence metabolism and body composition. Diabetologia. 2010;53:606-613. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 208] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 15. | Kobyliak N, Falalyeyeva T, Boyko N, Tsyryuk O, Beregova T, Ostapchenko L. Probiotics and nutraceuticals as a new frontier in obesity prevention and management. Diabetes Res Clin Pract. 2018;141:190-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 16. | Gu Y, Wang X, Li J, Zhang Y, Zhong H, Liu R, Zhang D, Feng Q, Xie X, Hong J, Ren H, Liu W, Ma J, Su Q, Zhang H, Yang J, Wang X, Zhao X, Gu W, Bi Y, Peng Y, Xu X, Xia H, Li F, Xu X, Yang H, Xu G, Madsen L, Kristiansen K, Ning G, Wang W. Analyses of gut microbiota and plasma bile acids enable stratification of patients for antidiabetic treatment. Nat Commun. 2017;8:1785. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 215] [Cited by in RCA: 298] [Article Influence: 37.3] [Reference Citation Analysis (0)] |
| 17. | Kobyliak N, Abenavoli L, Falalyeyeva T, Beregova T. Efficacy of Probiotics and Smectite in Rats with Non-Alcoholic Fatty Liver Disease. Ann Hepatol. 2018;17:153-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 18. | Kobyliak N, Falalyeyeva T, Bodnar P, Beregova T. Probiotics Supplemented with Omega-3 Fatty Acids are More Effective for Hepatic Steatosis Reduction in an Animal Model of Obesity. Probiotics Antimicrob Proteins. 2017;9:123-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 19. | Jourova L, Anzenbacher P, Anzenbacherova E. Human gut microbiota plays a role in the metabolism of drugs. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2016;160:317-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 54] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 20. | Yoo HH, Kim IS, Yoo DH, Kim DH. Effects of orally administered antibiotics on the bioavailability of amlodipine: gut microbiota-mediated drug interaction. J Hypertens. 2016;34:156-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 90] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 21. | Kobyliak N, Falalyeyeva T, Virchenko O, Mykhalchyshyn G, Bodnar P, Spivak M, Yankovsky D, Beregova T, Ostapchenko L. Comparative experimental investigation on the efficacy of mono- and multiprobiotic strains in non-alcoholic fatty liver disease prevention. BMC Gastroenterol. 2016;16:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 22. | Kobyliak N, Falalyeyeva T, Beregova T, Spivak M. Probiotics for experimental obesity prevention: focus on strain dependence and viability of composition. Endokrynol Pol. 2017;68:659-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 23. | Kobyliak N, Falalyeyeva T, Mykhalchyshyn G, Kyriienko D, Komissarenko I. Effect of alive probiotic on insulin resistance in type 2 diabetes patients: Randomized clinical trial. Diabetes Metab Syndr. 2018;12:617-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 127] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 24. | Kobyliak N, Abenavoli L, Falalyeyeva T, Mykhalchyshyn G, Boccuto L, Kononenko L, Kyriienko D, Komisarenko I, Dynnyk O. Beneficial effects of probiotic combination with omega-3 fatty acids in NAFLD: a randomized clinical study. Minerva Med. 2018;109:418-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 81] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 25. | Kobyliak N, Abenavoli L, Mykhalchyshyn G, Kononenko L, Boccuto L, Kyriienko D, Dynnyk O. A Multi-strain Probiotic Reduces the Fatty Liver Index, Cytokines and Aminotransferase levels in NAFLD Patients: Evidence from a Randomized Clinical Trial. J Gastrointestin Liver Dis. 2018;27:41-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 177] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 26. | Pernicova I, Korbonits M. Metformin--mode of action and clinical implications for diabetes and cancer. Nat Rev Endocrinol. 2014;10:143-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 785] [Cited by in RCA: 942] [Article Influence: 85.6] [Reference Citation Analysis (0)] |
| 27. | Viollet B, Guigas B, Sanz Garcia N, Leclerc J, Foretz M, Andreelli F. Cellular and molecular mechanisms of metformin: an overview. Clin Sci (Lond). 2012;122:253-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1054] [Cited by in RCA: 1224] [Article Influence: 94.2] [Reference Citation Analysis (0)] |
| 28. | Hur KY, Lee MS. New mechanisms of metformin action: Focusing on mitochondria and the gut. J Diabetes Investig. 2015;6:600-609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 121] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 29. | Devaraj S, Venkatachalam A, Chen X. Metformin and the Gut Microbiome in Diabetes. Clin Chem. 2016;62:1554-1555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 30. | Kim J, Cheon H, Jeong YT, Quan W, Kim KH, Cho JM, Lim YM, Oh SH, Jin SM, Kim JH, Lee MK, Kim S, Komatsu M, Kang SW, Lee MS. Amyloidogenic peptide oligomer accumulation in autophagy-deficient β cells induces diabetes. J Clin Invest. 2014;124:3311-3324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 140] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 31. | Lim YM, Lim H, Hur KY, Quan W, Lee HY, Cheon H, Ryu D, Koo SH, Kim HL, Kim J, Komatsu M, Lee MS. Systemic autophagy insufficiency compromises adaptation to metabolic stress and facilitates progression from obesity to diabetes. Nat Commun. 2014;5:4934. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 150] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 32. | Zhang X, Zhao Y, Xu J, Xue Z, Zhang M, Pang X, Zhang X, Zhao L. Modulation of gut microbiota by berberine and metformin during the treatment of high-fat diet-induced obesity in rats. Sci Rep. 2015;5:14405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 370] [Cited by in RCA: 475] [Article Influence: 47.5] [Reference Citation Analysis (0)] |
| 33. | Wu H, Esteve E, Tremaroli V, Khan MT, Caesar R, Mannerås-Holm L, Ståhlman M, Olsson LM, Serino M, Planas-Fèlix M, Xifra G, Mercader JM, Torrents D, Burcelin R, Ricart W, Perkins R, Fernàndez-Real JM, Bäckhed F. Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug. Nat Med. 2017;23:850-858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 824] [Cited by in RCA: 1137] [Article Influence: 142.1] [Reference Citation Analysis (0)] |
| 34. | Lee H, Ko G. Effect of metformin on metabolic improvement and gut microbiota. Appl Environ Microbiol. 2014;80:5935-5943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 293] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 35. | Shin NR, Lee JC, Lee HY, Kim MS, Whon TW, Lee MS, Bae JW. An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut. 2014;63:727-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1031] [Cited by in RCA: 1190] [Article Influence: 108.2] [Reference Citation Analysis (0)] |
| 36. | Everard A, Belzer C, Geurts L, Ouwerkerk JP, Druart C, Bindels LB, Guiot Y, Derrien M, Muccioli GG, Delzenne NM, de Vos WM, Cani PD. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc Natl Acad Sci U S A. 2013;110:9066-9071. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2639] [Cited by in RCA: 3318] [Article Influence: 276.5] [Reference Citation Analysis (0)] |
| 37. | Wu BN, O'Sullivan AJ. Sex differences in energy metabolism need to be considered with lifestyle modifications in humans. J Nutr Metab. 2011;2011:391809. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 93] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 38. | Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M; STOP-NIDDM Trail Research Group. Acarbose for prevention of type 2 diabetes mellitus: the STOP-NIDDM randomised trial. Lancet. 2002;359:2072-2077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1820] [Cited by in RCA: 1636] [Article Influence: 71.1] [Reference Citation Analysis (0)] |
| 39. | Zhang X, Fang Z, Zhang C, Xia H, Jie Z, Han X, Chen Y, Ji L. Effects of Acarbose on the Gut Microbiota of Prediabetic Patients: A Randomized, Double-blind, Controlled Crossover Trial. Diabetes Ther. 2017;8:293-307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 136] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 40. | Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M; STOP-NIDDM Trial Research Group. Acarbose treatment and the risk of cardiovascular disease and hypertension in patients with impaired glucose tolerance: the STOP-NIDDM trial. JAMA. 2003;290:486-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1238] [Cited by in RCA: 1120] [Article Influence: 50.9] [Reference Citation Analysis (0)] |
| 41. | Suzuki Y, Sano M, Hayashida K, Ohsawa I, Ohta S, Fukuda K. Are the effects of alpha-glucosidase inhibitors on cardiovascular events related to elevated levels of hydrogen gas in the gastrointestinal tract? FEBS Lett. 2009;583:2157-2159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 59] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 42. | Panwar H, Calderwood D, Grant IR, Grover S, Green BD. Lactobacillus strains isolated from infant faeces possess potent inhibitory activity against intestinal alpha- and beta-glucosidases suggesting anti-diabetic potential. Eur J Nutr. 2014;53:1465-1474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 43. | Gao Z, Yin J, Zhang J, Ward RE, Martin RJ, Lefevre M, Cefalu WT, Ye J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes. 2009;58:1509-1517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1337] [Cited by in RCA: 1581] [Article Influence: 98.8] [Reference Citation Analysis (0)] |
| 44. | Koh A, De Vadder F, Kovatcheva-Datchary P, Bäckhed F. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell. 2016;165:1332-1345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2533] [Cited by in RCA: 4217] [Article Influence: 468.6] [Reference Citation Analysis (0)] |
| 45. | Byron JS, Richard AM, Ericsson C, Harrison DC, Strong R, Schmidt TM. Changes in the gut microbiota and fermentation products associated with enhanced longevity in acarbose-treated mice. 2018;bioRxiv:311456. [DOI] [Full Text] |
| 46. | Do HJ, Lee YS, Ha MJ, Cho Y, Yi H, Hwang YJ, Hwang GS, Shin MJ. Beneficial effects of voglibose administration on body weight and lipid metabolism via gastrointestinal bile acid modification. Endocr J. 2016;63:691-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 47. | Liao Y, Takashima S, Zhao H, Asano Y, Shintani Y, Minamino T, Kim J, Fujita M, Hori M, Kitakaze M. Control of plasma glucose with alpha-glucosidase inhibitor attenuates oxidative stress and slows the progression of heart failure in mice. Cardiovasc Res. 2006;70:107-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 48. | Do HJ, Jin T, Chung JH, Hwang JW, Shin MJ. Voglibose administration regulates body weight and energy intake in high fat-induced obese mice. Biochem Biophys Res Commun. 2014;443:1110-1117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 49. | Kishida Y, Okubo H, Ohno H, Oki K, Yoneda M. Effect of miglitol on the suppression of nonalcoholic steatohepatitis development and improvement of the gut environment in a rodent model. J Gastroenterol. 2017;52:1180-1191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 50. | Lieber CS, Leo MA, Mak KM, Xu Y, Cao Q, Ren C, Ponomarenko A, DeCarli LM. Acarbose attenuates experimental non-alcoholic steatohepatitis. Biochem Biophys Res Commun. 2004;315:699-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 51. | Vieira EL, Leonel AJ, Sad AP, Beltrão NR, Costa TF, Ferreira TM, Gomes-Santos AC, Faria AM, Peluzio MC, Cara DC, Alvarez-Leite JI. Oral administration of sodium butyrate attenuates inflammation and mucosal lesion in experimental acute ulcerative colitis. J Nutr Biochem. 2012;23:430-436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 221] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 52. | Tan K, Tesar C, Wilton R, Jedrzejczak RP, Joachimiak A. Interaction of antidiabetic α-glucosidase inhibitors and gut bacteria α-glucosidase. Protein Sci. 2018;27:1498-1508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 53. | Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 2006;368:1696-1705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2787] [Cited by in RCA: 2844] [Article Influence: 149.7] [Reference Citation Analysis (1)] |
| 54. | Amato A, Cinci L, Rotondo A, Serio R, Faussone-Pellegrini MS, Vannucchi MG, Mulè F. Peripheral motor action of glucagon-like peptide-1 through enteric neuronal receptors. Neurogastroenterol Motil. 2010;22:664-e203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 93] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 55. | Aoki R, Kamikado K, Suda W, Takii H, Mikami Y, Suganuma N, Hattori M, Koga Y. A proliferative probiotic Bifidobacterium strain in the gut ameliorates progression of metabolic disorders via microbiota modulation and acetate elevation. Sci Rep. 2017;7:43522. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 160] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 56. | Vettorazzi JF, Ribeiro RA, Borck PC, Branco RC, Soriano S, Merino B, Boschero AC, Nadal A, Quesada I, Carneiro EM. The bile acid TUDCA increases glucose-induced insulin secretion via the cAMP/PKA pathway in pancreatic beta cells. Metabolism. 2016;65:54-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 77] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 57. | Vilsbøll T, Christensen M, Junker AE, Knop FK, Gluud LL. Effects of glucagon-like peptide-1 receptor agonists on weight loss: systematic review and meta-analyses of randomised controlled trials. BMJ. 2012;344:d7771. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 737] [Cited by in RCA: 660] [Article Influence: 50.8] [Reference Citation Analysis (0)] |
| 58. | Tilg H, Moschen AR. Microbiota and diabetes: an evolving relationship. Gut. 2014;63:1513-1521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 519] [Cited by in RCA: 560] [Article Influence: 50.9] [Reference Citation Analysis (0)] |
| 59. | Zhao L, Chen Y, Xia F, Abudukerimu B, Zhang W, Guo Y, Wang N, Lu Y. A Glucagon-Like Peptide-1 Receptor Agonist Lowers Weight by Modulating the Structure of Gut Microbiota. Front Endocrinol (Lausanne). 2018;9:233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 94] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 60. | Schwiertz A, Taras D, Schäfer K, Beijer S, Bos NA, Donus C, Hardt PD. Microbiota and SCFA in lean and overweight healthy subjects. Obesity (Silver Spring). 2010;18:190-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1670] [Cited by in RCA: 1787] [Article Influence: 119.1] [Reference Citation Analysis (0)] |
| 61. | Wang Z, Saha S, Van Horn S. Gut microbiome differences between metformin‐ and liraglutide‐treated T2DM subjects. Endocrinol Diab Metab. 2018;1:e9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 61] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 62. | Pyke C, Heller RS, Kirk RK, Ørskov C, Reedtz-Runge S, Kaastrup P, Hvelplund A, Bardram L, Calatayud D, Knudsen LB. GLP-1 receptor localization in monkey and human tissue: novel distribution revealed with extensively validated monoclonal antibody. Endocrinology. 2014;155:1280-1290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 491] [Cited by in RCA: 626] [Article Influence: 56.9] [Reference Citation Analysis (0)] |
| 63. | Heppner KM, Kirigiti M, Secher A, Paulsen SJ, Buckingham R, Pyke C, Knudsen LB, Vrang N, Grove KL. Expression and distribution of glucagon-like peptide-1 receptor mRNA, protein and binding in the male nonhuman primate (Macaca mulatta) brain. Endocrinology. 2015;156:255-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 149] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 64. | Nozu T, Miyagishi S, Kumei S, Nozu R, Takakusaki K, Okumura T. Glucagon-like peptide-1 analog, liraglutide, improves visceral sensation and gut permeability in rats. J Gastroenterol Hepatol. 2018;33:232-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 65. | Hellström PM, Hein J, Bytzer P, Björnssön E, Kristensen J, Schambye H. Clinical trial: the glucagon-like peptide-1 analogue ROSE-010 for management of acute pain in patients with irritable bowel syndrome: a randomized, placebo-controlled, double-blind study. Aliment Pharmacol Ther. 2009;29:198-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 71] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 66. | Voss U, Sand E, Olde B, Ekblad E. Enteric neuropathy can be induced by high fat diet in vivo and palmitic acid exposure in vitro. PLoS One. 2013;8:e81413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 67. | Grasset E, Puel A, Charpentier J, Collet X, Christensen JE, Tercé F, Burcelin R. A Specific Gut Microbiota Dysbiosis of Type 2 Diabetic Mice Induces GLP-1 Resistance through an Enteric NO-Dependent and Gut-Brain Axis Mechanism. Cell Metab. 2017;25:1075-1090.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 186] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 68. | Clemmensen C, Smajilovic S, Smith EP, Woods SC, Bräuner-Osborne H, Seeley RJ, D'Alessio DA, Ryan KK. Oral L-arginine stimulates GLP-1 secretion to improve glucose tolerance in male mice. Endocrinology. 2013;154:3978-3983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 61] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 69. | Seino Y, Fukushima M, Yabe D. GIP and GLP-1, the two incretin hormones: Similarities and differences. J Diabetes Investig. 2010;1:8-23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 350] [Cited by in RCA: 498] [Article Influence: 45.3] [Reference Citation Analysis (0)] |
| 70. | Olivares M, Schüppel V, Hassan AM, Beaumont M, Neyrinck AM, Bindels LB, Benítez-Páez A, Sanz Y, Haller D, Holzer P, Delzenne NM. The Potential Role of the Dipeptidyl Peptidase-4-Like Activity From the Gut Microbiota on the Host Health. Front Microbiol. 2018;9:1900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 71. | Holzer P, Reichmann F, Farzi A. Neuropeptide Y, peptide YY and pancreatic polypeptide in the gut-brain axis. Neuropeptides. 2012;46:261-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 330] [Cited by in RCA: 353] [Article Influence: 27.2] [Reference Citation Analysis (0)] |
| 72. | Zhong J, Gong Q, Goud A, Srinivasamaharaj S, Rajagopalan S. Recent Advances in Dipeptidyl-Peptidase-4 Inhibition Therapy: Lessons from the Bench and Clinical Trials. J Diabetes Res. 2015;2015:606031. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 73. | Ahmed RH, Huri HZ, Muniandy S, Al-Hamodi Z, Al-Absi B, Alsalahi A, Razif MF. Altered circulating concentrations of active glucagon-like peptide (GLP-1) and dipeptidyl peptidase 4 (DPP4) in obese subjects and their association with insulin resistance. Clin Biochem. 2017;50:746-749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 74. | Cani PD, Neyrinck AM, Maton N, Delzenne NM. Oligofructose promotes satiety in rats fed a high-fat diet: involvement of glucagon-like Peptide-1. Obes Res. 2005;13:1000-1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 270] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 75. | Fteita D, Musrati AA, Könönen E, Ma X, Gürsoy M, Peurla M, Söderling E, Sintim HO, Gürsoy UK. Dipeptidyl peptidase IV and quorum sensing signaling in biofilm-related virulence of Prevotella aurantiaca. Anaerobe. 2017;48:152-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 76. | Stengel A, Goebel-Stengel M, Teuffel P, Hofmann T, Buße P, Kobelt P, Rose M, Klapp BF. Obese patients have higher circulating protein levels of dipeptidyl peptidase IV. Peptides. 2014;61:75-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 77. | Klemann C, Wagner L, Stephan M, von Hörsten S. Cut to the chase: a review of CD26/dipeptidyl peptidase-4's (DPP4) entanglement in the immune system. Clin Exp Immunol. 2016;185:1-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 321] [Article Influence: 35.7] [Reference Citation Analysis (0)] |
| 78. | Diouf I, Charles MA, Thiebaugeorges O, Forhan A, Kaminski M, Heude B; EDEN Mother–Child Cohort Study Group. Maternal weight change before pregnancy in relation to birthweight and risks of adverse pregnancy outcomes. Eur J Epidemiol. 2011;26:789-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 79. | Paul HA, Bomhof MR, Vogel HJ, Reimer RA. Diet-induced changes in maternal gut microbiota and metabolomic profiles influence programming of offspring obesity risk in rats. Sci Rep. 2016;6:20683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 142] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 80. | Zhang Q, Xiao X, Li M, Yu M, Ping F, Zheng J, Wang T, Wang X. Vildagliptin increases butyrate-producing bacteria in the gut of diabetic rats. PLoS One. 2017;12:e0184735. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 81. | Ryan PM, Patterson E, Carafa I, Mandal R. Metformin and a DPP-4 Inhibitor Differentially Modulate the Microbiome and Metabolome of Metabolic Syndrome Mice. Can J Diabetes. 2018;42:S40. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 82. | Du F, Hinke SA, Cavanaugh C, Polidori D, Wallace N, Kirchner T, Jennis M, Lang W, Kuo GH, Gaul MD, Lenhard J, Demarest K, Ajami NJ, Liang Y, Hornby PJ. Potent Sodium/Glucose Cotransporter SGLT1/2 Dual Inhibition Improves Glycemic Control Without Marked Gastrointestinal Adaptation or Colonic Microbiota Changes in Rodents. J Pharmacol Exp Ther. 2018;365:676-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 83. | Battson ML, Lee DM, Jarrell DK, Hou S, Ecton KE, Weir TL, Gentile CL. Suppression of gut dysbiosis reverses Western diet-induced vascular dysfunction. Am J Physiol Endocrinol Metab. 2018;314:E468-E477. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 84. | Lee DM, Battson ML, Jarrell DK, Hou S, Ecton KE, Weir TL, Gentile CL. SGLT2 inhibition via dapagliflozin improves generalized vascular dysfunction and alters the gut microbiota in type 2 diabetic mice. Cardiovasc Diabetol. 2018;17:62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 184] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 85. | Kuo GH, Gaul MD, Liang Y, Xu JZ, Du F, Hornby P, Xu G, Qi J, Wallace N, Lee S, Grant E, Murray WV, Demarest K. Synthesis and biological evaluation of benzocyclobutane-C-glycosides as potent and orally active SGLT1/SGLT2 dual inhibitors. Bioorg Med Chem Lett. 2018;28:1182-1187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 86. | Mudaliar S, Polidori D, Zambrowicz B, Henry RR. Sodium-Glucose Cotransporter Inhibitors: Effects on Renal and Intestinal Glucose Transport: From Bench to Bedside. Diabetes Care. 2015;38:2344-2353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 181] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 87. | Zambrowicz B, Ding ZM, Ogbaa I, Frazier K, Banks P, Turnage A, Freiman J, Smith M, Ruff D, Sands A, Powell D. Effects of LX4211, a dual SGLT1/SGLT2 inhibitor, plus sitagliptin on postprandial active GLP-1 and glycemic control in type 2 diabetes. Clin Ther. 2013;35:273-285.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 63] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 88. | Lefterova MI, Lazar MA. New developments in adipogenesis. Trends Endocrinol Metab. 2009;20:107-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 617] [Cited by in RCA: 667] [Article Influence: 41.7] [Reference Citation Analysis (0)] |
| 89. | Yoshizawa K, Cioca DP, Kawa S, Tanaka E, Kiyosawa K. Peroxisome proliferator-activated receptor gamma ligand troglitazone induces cell cycle arrest and apoptosis of hepatocellular carcinoma cell lines. Cancer. 2002;95:2243-2251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 58] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 90. | Grimaldi PA. Peroxisome proliferator-activated receptors as sensors of fatty acids and derivatives. Cell Mol Life Sci. 2007;64:2459-2464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 60] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 91. | Nepelska M, de Wouters T, Jacouton E, Béguet-Crespel F, Lapaque N, Doré J, Arulampalam V, Blottière HM. Commensal gut bacteria modulate phosphorylation-dependent PPARγ transcriptional activity in human intestinal epithelial cells. Sci Rep. 2017;7:43199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 92. | Brown PH, Lippman SM. Chemoprevention of breast cancer. Breast Cancer Res Treat. 2000;62:1-17. [PubMed] |
| 93. | Anghel SI, Wahli W. Fat poetry: a kingdom for PPAR gamma. Cell Res. 2007;17:486-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 111] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 94. | Alex S, Lange K, Amolo T, Grinstead JS, Haakonsson AK, Szalowska E, Koppen A, Mudde K, Haenen D, Al-Lahham S, Roelofsen H, Houtman R, van der Burg B, Mandrup S, Bonvin AM, Kalkhoven E, Müller M, Hooiveld GJ, Kersten S. Short-chain fatty acids stimulate angiopoietin-like 4 synthesis in human colon adenocarcinoma cells by activating peroxisome proliferator-activated receptor γ. Mol Cell Biol. 2013;33:1303-1316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 230] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 95. | Ortuño Sahagún D, Márquez-Aguirre AL, Quintero-Fabián S, López-Roa RI, Rojas-Mayorquín AE. Modulation of PPAR-γ by Nutraceutics as Complementary Treatment for Obesity-Related Disorders and Inflammatory Diseases. PPAR Res. 2012;2012:318613. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 37] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 96. | Are A, Aronsson L, Wang S, Greicius G, Lee YK, Gustafsson JA, Pettersson S, Arulampalam V. Enterococcus faecalis from newborn babies regulate endogenous PPARgamma activity and IL-10 levels in colonic epithelial cells. Proc Natl Acad Sci U S A. 2008;105:1943-1948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 115] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 97. | Scher JU, Sczesnak A, Longman RS, Segata N, Ubeda C, Bielski C, Rostron T, Cerundolo V, Pamer EG, Abramson SB, Huttenhower C, Littman DR. Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis. Elife. 2013;2:e01202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1133] [Cited by in RCA: 1440] [Article Influence: 120.0] [Reference Citation Analysis (0)] |
| 98. | Kumar PS, Griffen AL, Barton JA, Paster BJ, Moeschberger ML, Leys EJ. New bacterial species associated with chronic periodontitis. J Dent Res. 2003;82:338-344. [PubMed] |
| 99. | Rizos CV, Elisaf MS, Mikhailidis DP, Liberopoulos EN. How safe is the use of thiazolidinediones in clinical practice? Expert Opin Drug Saf. 2009;8:15-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 119] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 100. | Newman DJ, Cragg GM. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J Nat Prod. 2012;75:311-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3569] [Cited by in RCA: 3178] [Article Influence: 244.5] [Reference Citation Analysis (0)] |
| 101. | Neschen S, Morino K, Dong J, Wang-Fischer Y, Cline GW, Romanelli AJ, Rossbacher JC, Moore IK, Regittnig W, Munoz DS, Kim JH, Shulman GI. n-3 Fatty acids preserve insulin sensitivity in vivo in a peroxisome proliferator-activated receptor-alpha-dependent manner. Diabetes. 2007;56:1034-1041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 173] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 102. | Puglisi MJ, Hasty AH, Saraswathi V. The role of adipose tissue in mediating the beneficial effects of dietary fish oil. J Nutr Biochem. 2011;22:101-108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 86] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 103. | Kim MH, Kang SG, Park JH, Yanagisawa M, Kim CH. Short-chain fatty acids activate GPR41 and GPR43 on intestinal epithelial cells to promote inflammatory responses in mice. Gastroenterology. 2013;145:396-406.e1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 576] [Cited by in RCA: 771] [Article Influence: 64.3] [Reference Citation Analysis (0)] |
| 104. | Puddu A, Sanguineti R, Montecucco F, Viviani GL. Evidence for the gut microbiota short-chain fatty acids as key pathophysiological molecules improving diabetes. Mediators Inflamm. 2014;2014:162021. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 229] [Cited by in RCA: 226] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 105. | Cani PD, Amar J, Iglesias MA, Poggi M, Knauf C, Bastelica D, Neyrinck AM, Fava F, Tuohy KM, Chabo C, Waget A, Delmée E, Cousin B, Sulpice T, Chamontin B, Ferrières J, Tanti JF, Gibson GR, Casteilla L, Delzenne NM, Alessi MC, Burcelin R. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56:1761-1772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4095] [Cited by in RCA: 4566] [Article Influence: 253.7] [Reference Citation Analysis (1)] |
| 106. | Wu TR, Lin CS, Chang CJ, Lin TL, Martel J, Ko YF, Ojcius DM, Lu CC, Young JD, Lai HC. Gut commensal Parabacteroides goldsteinii plays a predominant role in the anti-obesity effects of polysaccharides isolated from Hirsutella sinensis. Gut. 2019;68:248-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 371] [Cited by in RCA: 548] [Article Influence: 91.3] [Reference Citation Analysis (0)] |
| 107. | Cani PD, de Vos WM. Next-Generation Beneficial Microbes: The Case of Akkermansia muciniphila. Front Microbiol. 2017;8:1765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 507] [Cited by in RCA: 699] [Article Influence: 87.4] [Reference Citation Analysis (0)] |
| 108. | Everard A, Lazarevic V, Derrien M, Girard M, Muccioli GG, Neyrinck AM, Possemiers S, Van Holle A, François P, de Vos WM, Delzenne NM, Schrenzel J, Cani PD. Responses of gut microbiota and glucose and lipid metabolism to prebiotics in genetic obese and diet-induced leptin-resistant mice. Diabetes. 2011;60:2775-2786. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 755] [Cited by in RCA: 824] [Article Influence: 58.9] [Reference Citation Analysis (0)] |
| 109. | Roopchand DE, Carmody RN, Kuhn P, Moskal K, Rojas-Silva P, Turnbaugh PJ, Raskin I. Dietary Polyphenols Promote Growth of the Gut Bacterium Akkermansia muciniphila and Attenuate High-Fat Diet-Induced Metabolic Syndrome. Diabetes. 2015;64:2847-2858. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 449] [Cited by in RCA: 491] [Article Influence: 49.1] [Reference Citation Analysis (0)] |
| 110. | Zhang H, DiBaise JK, Zuccolo A, Kudrna D, Braidotti M, Yu Y, Parameswaran P, Crowell MD, Wing R, Rittmann BE, Krajmalnik-Brown R. Human gut microbiota in obesity and after gastric bypass. Proc Natl Acad Sci U S A. 2009;106:2365-2370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1323] [Cited by in RCA: 1402] [Article Influence: 87.6] [Reference Citation Analysis (1)] |
| 111. | Liou AP, Paziuk M, Luevano JM, Machineni S, Turnbaugh PJ, Kaplan LM. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci Transl Med. 2013;5:178ra41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 643] [Cited by in RCA: 711] [Article Influence: 59.3] [Reference Citation Analysis (0)] |
| 112. | Blandino G, Inturri R, Lazzara F, Di Rosa M, Malaguarnera L. Impact of gut microbiota on diabetes mellitus. Diabetes Metab. 2016;42:303-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 170] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 113. | Xu J, Lian F, Zhao L, Zhao Y, Chen X, Zhang X, Guo Y, Zhang C, Zhou Q, Xue Z, Pang X, Zhao L, Tong X. Structural modulation of gut microbiota during alleviation of type 2 diabetes with a Chinese herbal formula. ISME J. 2015;9:552-562. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 269] [Cited by in RCA: 337] [Article Influence: 30.6] [Reference Citation Analysis (1)] |