Published online Jan 15, 2019. doi: 10.4239/wjd.v10.i1.16
Peer-review started: August 24, 2018
First decision: October 5, 2018
Revised: November 8, 2018
Accepted: December 12, 2018
Article in press: December 13, 2018
Published online: January 15, 2019
Processing time: 145 Days and 8.8 Hours
Diabetes affects approximately 30 million persons in the United States. Diabetes ketoacidosis is one of the most serious and acute complications of diabetes. At the time of presentation and during treatment of diabetic ketoacidosis (DKA), several metabolic and electrolyte derangements can ultimately result in respiratory compromise. Most commonly, hypokalemia, hypomagnesemia and hypophosphatemia can eventually lead to respiratory muscles failure. Furthermore, tachypnea, hyperpnea and more severely, Kussmaul breathing pattern can develop. Also, hydrostatic and non-hydrostatic pulmonary edema can occur secondary to volume shifts into the extracellular space and secondary to increased permeability of the pulmonary capillaries. The presence of respiratory failure in patients with DKA is associated with higher morbidity and mortality. Being familiar with the causes of respiratory compromise in DKA, and how to treat them, may represent better outcomes for patients with DKA.
Core tip: Several electrolyte and metabolic derangements associated with diabetic ketoacidosis (DKA) and its treatment can affect the respiratory system. Since respiratory failure in DKA is associated with increased morbidity and mortality, the recognition and treatment of those derangements have the potential to improve outcomes in DKA.
- Citation: Gallo de Moraes A, Surani S. Effects of diabetic ketoacidosis in the respiratory system. World J Diabetes 2019; 10(1): 16-22
- URL: https://www.wjgnet.com/1948-9358/full/v10/i1/16.htm
- DOI: https://dx.doi.org/10.4239/wjd.v10.i1.16
Diabetes ketoacidosis (DKA) is one of the most serious and acute complications of diabetes. It is characterized by moderate hyperglycemia (blood glucose usually between 250 mg/dL and 800 mg/dL at presentation), metabolic acidosis, and presence of serum ketones with an elevated anion gap[1]. It represents an extreme in the spectrum of hyperglycemia and presentation of complicated diabetes.
Diabetes affects approximately 30 million persons in the United States[2]. Since 2009, there has been an increase of around 6% of hospitalizations due to DKA (from 19.5 to 30.2 per 1000 persons). However, the in-hospital mortality has declined at an annual average rate of 6.8% (from 1.1% to 0.4%)[2].
The presence of DKA is accompanied by several electrolytes, metabolic and acid-base derangements that affect the respiratory system. Depletion of ions, such as potassium and phosphate, affect the respiratory muscles leading to acute respiratory failure[3]. Reduction in colloid osmotic pressure increases lung water content, leading to noncardiogenic pulmonary edema and decrease in lung compliance[4,5]. As a compensatory mechanism, the presence of metabolic acidosis will cause hyperventilation[6].
Respiratory failure in DKA has been associated with increased morbidity and mortality[3,7]. In this review, we analyze the common electrolytes, metabolic and acid-base abnormalities seen in DKA, their association with respiratory failure and its management.
Potassium, magnesium and phosphorous are intracellular ions which serum concentrations decrease as a direct consequence of hyperglycemia and ketoacidosis (potassium), or as a consequence of the correction of acidosis with insulin (magnesium and phosphorous). A major goal in the treatment of DKA is to closely monitor these ions concentrations as DKA is corrected. Also, replace them on a timely fashion in order to prevent them from reaching critically low values. The clinical significance of their deficit is discussed below.
Patients being admitted for DKA usually have a total body potassium deficit that averages 300 to 600 mEq[8]. Osmotic diuresis is provoked by the hyperglycemia resulting from lack of insulin. In an attempt to maintain osmolality, the kidneys will retain sodium ions at the expense of potassium ions[9]. Furthermore, when acidosis is present, hydrogen ions from the bicarbonate nucleus will be reabsorbed at the expenditure of potassium[10].
The gastrointestinal tract is also responsible for potassium loss in DKA. The body will try to maintain osmotic pressure at the cost of tissue and serum electrolytes. An acute hyperkalemia will happen when potassium shifts into the extracellular fluid (ECF), causing gastric cells to preserve hydrogen ions concentration. Consequently, nausea, vomit and diarrhea will occur, promoting even more potassium loss[11,12]. However, due to a shift of potassium from intracellular fluid into ECF caused by hyper osmolality and insulin deficiency, only 5% of patients with DKA will present with hypokalemia[8,13].
When potassium levels fall below 2.5 mg/dL, severe ascending muscular weakness can occur[14]. The muscular weakness can affect the respiratory muscles causing acute respiratory failure[15], and requirement of mechanical ventilation[16]. Aggressive potassium replacement should start once serum potassium concentration reaches a value of 3.3 mEq/L[13].
At presentation of DKA, the levels of serum magnesium are usually normal. Excessive amounts of magnesium are excreted during acidosis, secondary to insulin deficiency[17]. As the acidosis gets corrected, magnesium levels fall, reaching their nadir within the first 25 h of acidosis correction[18,19].
Hypomagnesemia, defined as having a serum magnesium concentration below 1.6 mg/dL (0.66 mmol/L), usually doesn’t lead to clinically significant symptoms until serum levels fall below 1.2 mg/dL (0.5 mmol/L)[20].
Magnesium regulates intracellular calcium levels, influencing smooth muscle tone[21]. Because of its role in regulating smooth muscle tone, magnesium deficiency has been associated with systemic hypertension, neuromuscular excitability, bronchoconstriction, coronary vasospasm and seizures[22].
Muscular weakness and tetany associated with hypomagnesemia can affect the respiratory muscles, impairing ventilation in patients who are spontaneously breathing and delaying extubation of mechanically ventilated patients[22,23]. Empirical magnesium replacement has been associated with improvement of respiratory muscle power in patients with DKA[23].
When treating patients with DKA, clinicians should aim to keep magnesium levels at normal range, since hypomagnesemia is associated with weakness of the respiratory muscles.
Acidosis causes potassium shifts into the ECF and hyperglycemia causes phosphaturia by osmotic diuresis, which will ultimately lead to hypophosphatemia. However, DKA patients will present with normal phosphorous concentration due to the shift into the ECF associated with ECF volume concentration[24]. The true state of phosphate equilibrium is revealed with volume expansion[24,25].
Severe hypophosphatemia (< 1 mg/dL) is associated to the depletion of high-energy phosphate compounds in muscles, causing muscular weakness and rhabdomyolysis[3,26].
Acute muscular weakness caused by hypophosphatemia in DKA has been associated with hypercapnic respiratory failure and prolonged mechanical ventilation in critically ill patients[26-28].
Routine replacement of phosphorous in patients who presented with DKA is not beneficial and has been associated with worsening hypomagnesemia and causing hypocalcemia[29,30]. However, if serum phosphate concentration falls below 1 mg/dL, or if hypophosphatemia is associated with cardiac dysfunction or respiratory depression, it should be replaced[28,31,32].
The presence of metabolic acidosis will normally generate a respiratory response. The reduction of serum bicarbonate and pH will result in hyperventilation and reduction in carbon dioxide (CO2), partially preventing further fall in pH and bicarbonate concentration. Respiratory compensation for metabolic acidosis will cause the arterial CO2 to decrease by 1.2 mmHg for each 1 meq/L fall in the serum bicarbonate[33].
The respiratory response usually begins within 30 min of metabolic acidosis onset, and is generally complete within 12-24 h However, a lag in respiratory compensation can occur when respiratory acidosis develops quickly; more than 4 meq/L of bicarbonate decrease in less than 6-12 h[34,35].
There is a limit to the lungs’ ability to compensate for metabolic acidosis. Even with serum bicarbonate concentrations below 6 meq/L, CO2 levels cannot fall lower than 8-12 mmHg[34]. Furthermore, the duration of the respiratory compensation is limited by respiratory muscle fatigue[33,36].
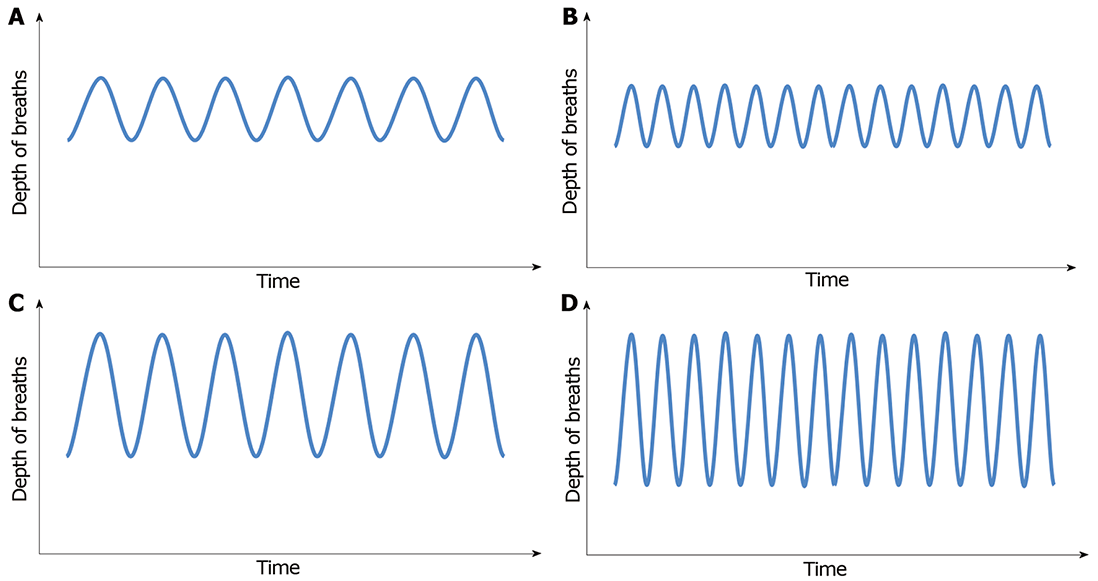
Initially, patients will develop tachypnea, which is increased respiratory rate, leading to decrease in CO2 concentration. With progression of acidosis, respiratory pattern evolves to hyperpnea, which is increased tidal volume, and ultimately, patients will develop a deep, fast and agonal pattern of breathing, named Kussmaul’s respiration (Figure 1A-D)[34,37].
Once patients with DKA develop Kussmaul’s respiration, they are reaching the point of respiratory muscles fatigue, and mechanical ventilation should be considered[34,38-40]. Furthermore, patients in DKA are severely “air hungry” prior to intubation, and are at higher risk to develop acute respiratory distress syndrome (ARDS)[3,41] due to hyperpnea. Mechanical ventilation in these patients is particularly delicate, since a lung protective strategy, with low tidal volumes and controlled transpulmonary pressures, should be maintained, while attempting to increase minute-ventilation until metabolic acidosis is completely corrected[42,43].
There are two types of pulmonary edema that have been described in patients with DKA: One associated with elevated pulmonary venous pressure and another associated with increased pulmonary capillary permeability. The diagnosis is made based on clinical findings of dyspnea, an A-a gradient on arterial blood gas and chest image showing bilateral pulmonary infiltrates.
Also known as hydrostatic pulmonary edema, it is usually existent at presentation of DKA, is corrected during the treatment of DKA and is more common in patients with concomitant renal failure[44-47]. The occurrence of circulatory overload and pulmonary edema with elevated pulmonary venous pressure is a result of the acute shift of an abundant volume of fluid into the extracellular compartment. This fluid shift happens as a consequence of solute accumulation in the extracellular compartment secondary to hyperglycemia[44]. Therefore, correction of hyperglycemia shifts fluid back into cells, also correcting hydrostatic pulmonary edema. However, some patients might require hemodialysis and mechanical ventilation.
The degree of fluid shift and, consequently, the likelihood of developing hydrostatic pulmonary edema during a DKA episode are determined by the severity of hyperglycemia and by the volume status prior to the development of DKA[47]. The amount of fluid transferred from the cells into the extracellular space is directly proportional to the changes in serum glucose concentration[48]. The patients’ volume status at the time of hyperglycemia onset is also a determinant of the volume that will shift into the extracellular space. Patients with baseline low extremity edema and/or anasarca have been shown to shift larger amounts of fluid and have a higher incidence of pulmonary edema, than those patients who are euvolemic when becoming hyperglycemic[49].
Even though hydrostatic pulmonary edema has been described more commonly in patients with advanced renal disease, there are several cases reported in patients with DKA who developed pulmonary edema without having renal dysfunction. Several cases have been reported of takotsubo cardiomyopathy happening in the setting of DKA and causing pulmonary edema[50,51]. There are also reports of myocardial dysfunction secondary to severe acidosis and electrolyte abnormalities[52].
Also known as non-hydrostatic pulmonary edema, this type of pulmonary edema is caused by changes at the histological level of the alveolar epithelium. In diabetic patients, there is thickening of the alveolar epithelium and pulmonary capillary basal membrane, corroborating the presence of pulmonary microangiopathy[53,54].
ARDS can develop during the course of DKA or during its treatment[3], and it is more frequent and severe than hydrostatic pulmonary edema[54,55]. The mechanism of ARDS in DKA is not completely understood. The most accepted explanation is activation of lymphocytes and release of cytokines, especially interleukin-1, which serum levels are much higher during treatment of DKA[56-58].
The treatment of non-hydrostatic pulmonary edema in DKA is supportive. Focus should be on treating DKA and its exacerbating factor, early intubation and protective lung ventilation.
In DKA, respiratory failure is caused by several electrolytes, metabolic and cardiac and lung end-organ damage. Developing respiratory failure during DKA onset or treatment is associated with high mortality. Early recognition and treatment of the risk factors for the development of respiratory failure have the potential to decrease morbi-mortality of patients with DKA.
Manuscript source: Invited manuscript
Specialty type: Endocrinology and metabolism
Country of origin: United States
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Hamaguchi M, Hamasaki H, Serhiyenko VA S- Editor: Ji FF L- Editor: A E- Editor: Wu YXJ
| 1. | Kitabchi AE, Umpierrez GE, Miles JM, Fisher JN. Hyperglycemic crises in adult patients with diabetes. Diabetes Care. 2009;32:1335-1343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1090] [Cited by in RCA: 1181] [Article Influence: 73.8] [Reference Citation Analysis (3)] |
| 2. | Benoit SR, Zhang Y, Geiss LS, Gregg EW, Albright A. Trends in Diabetic Ketoacidosis Hospitalizations and In-Hospital Mortality - United States, 2000-2014. MMWR Morb Mortal Wkly Rep. 2018;67:362-365. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 212] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 3. | Konstantinov NK, Rohrscheib M, Agaba EI, Dorin RI, Murata GH, Tzamaloukas AH. Respiratory failure in diabetic ketoacidosis. World J Diabetes. 2015;6:1009-1023. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 14] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (2)] |
| 4. | Brun-Buisson CJ, Bonnet F, Bergeret S, Lemaire F, Rapin M. Recurrent high-permeability pulmonary edema associated with diabetic ketoacidosis. Crit Care Med. 1985;13:55-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 39] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 5. | Sprung CL, Rackow EC, Fein IA. Pulmonary edema: A complication of diabetic ketoacidosis. Chest. 1980;77:687-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 6. | Whited L, Graham DD. Abnormal Respirations. StatPearls. Treasure Island (FL): StatPearls Publishing. 2018;. [PubMed] |
| 7. | Freire AX, Umpierrez GE, Afessa B, Latif KA, Bridges L, Kitabchi AE. Predictors of intensive care unit and hospital length of stay in diabetic ketoacidosis. J Crit Care. 2002;17:207-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 42] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 8. | Adrogué HJ, Lederer ED, Suki WN, Eknoyan G. Determinants of plasma potassium levels in diabetic ketoacidosis. Medicine (Baltimore). 1986;65:163-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 97] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 9. | Kitabchi AE, Nyenwe EA. Hyperglycemic crises in diabetes mellitus: diabetic ketoacidosis and hyperglycemic hyperosmolar state. Endocrinol Metab Clin North Am. 2006;35:725-751, viii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 102] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 10. | Usman A. Initial Potassium Replacement in Diabetic Ketoacidosis: The Unnoticed Area of Gap. Front Endocrinol (Lausanne). 2018;9:109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 11. | Schaefer TJ, Wolford RW. Disorders of potassium. Emerg Med Clin North Am. 2005;23:723-747, viii-viix. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 78] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 12. | Umpierrez GE, Kitabchi AE. Diabetic ketoacidosis: Risk factors and management strategies. Treat Endocrinol. 2003;2:95-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 124] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 13. | Murthy K, Harrington JT, Siegel RD. Profound hypokalemia in diabetic ketoacidosis: a therapeutic challenge. Endocr Pract. 2005;11:331-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 33] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 14. | Comi G, Testa D, Cornelio F, Comola M, Canal N. Potassium depletion myopathy: a clinical and morphological study of six cases. Muscle Nerve. 1985;8:17-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 15. | Haddad S, Arabi Y, Shimemeri AA. Hypokalemic paralysis mimicking Guillain-Barré syndrome and causing acute respiratory failure. Middle East J Anaesthesiol. 2004;17:891-897. [PubMed] |
| 16. | Valsamis EM, Smith I, De Sousa A. An unusual cause of ventilatory failure in motor neurone disease. Respir Med Case Rep. 2017;21:59-61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 17. | Wacker WE, Parisi AF. Magnesium metabolism. N Engl J Med. 1968;278:712-717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 135] [Article Influence: 2.4] [Reference Citation Analysis (2)] |
| 18. | Bauza J, Ortiz J, Dahan M, Justiniano M, Saenz R, Vélez M. Reliability of serum magnesium values during diabetic ketoacidosis in children. Bol Asoc Med P R. 1998;90:108-112. [PubMed] |
| 19. | Ionescu-Tîrgovişte C, Bruckner I, Mihalache N, Ionescu C. Plasma phosphorus and magnesium values during treatment of severe diabetic ketoacidosis. Med Interne. 1981;19:63-68. [PubMed] |
| 20. | Swaminathan R. Magnesium metabolism and its disorders. Clin Biochem Rev. 2003;24:47-66. [PubMed] |
| 21. | Laurant P, Touyz RM. Physiological and pathophysiological role of magnesium in the cardiovascular system: implications in hypertension. J Hypertens. 2000;18:1177-1191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 105] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 22. | Tong GM, Rude RK. Magnesium deficiency in critical illness. J Intensive Care Med. 2005;20:3-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 120] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 23. | Dhingra S, Solven F, Wilson A, McCarthy DS. Hypomagnesemia and respiratory muscle power. Am Rev Respir Dis. 1984;129:497-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 24. | Kebler R, McDonald FD, Cadnapaphornchai P. Dynamic changes in serum phosphorus levels in diabetic ketoacidosis. Am J Med. 1985;79:571-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 55] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 25. | Shen T, Braude S. Changes in serum phosphate during treatment of diabetic ketoacidosis: predictive significance of severity of acidosis on presentation. Intern Med J. 2012;42:1347-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 26. | Ditzel J, Lervang HH. Disturbance of inorganic phosphate metabolism in diabetes mellitus: clinical manifestations of phosphorus-depletion syndrome during recovery from diabetic ketoacidosis. Diabetes Metab Syndr Obes. 2010;3:319-324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 27. | Federspiel CK, Itenov TS, Thormar K, Liu KD, Bestle MH. Hypophosphatemia and duration of respiratory failure and mortality in critically ill patients. Acta Anaesthesiol Scand. 2018;62:1098-1104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 28. | Alsumrain MH, Jawad SA, Imran NB, Riar S, DeBari VA, Adelman M. Association of hypophosphatemia with failure-to-wean from mechanical ventilation. Ann Clin Lab Sci. 2010;40:144-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 29. | Fisher JN, Kitabchi AE. A randomized study of phosphate therapy in the treatment of diabetic ketoacidosis. J Clin Endocrinol Metab. 1983;57:177-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 116] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 30. | Winter RJ, Harris CJ, Phillips LS, Green OC. Diabetic ketoacidosis. Induction of hypocalcemia and hypomagnesemia by phosphate therapy. Am J Med. 1979;67:897-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 66] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 31. | Agarwal A, Yadav A, Gutch M, Consul S, Kumar S, Prakash V, Gupta AK, Bhattacharjee A. Prognostic factors in patients hospitalized with diabetic ketoacidosis. Endocrinol Metab. 2016;31:424-432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 32. | Belke F, Augst D, Konrad K, Finetti C. Rhabdomyolysis as a rare complication of diabetic ketoacidosis in a twelve-year-old boy. Neuropediatrics. 2016;47-P04-17. [DOI] [Full Text] |
| 33. | Adrogué HJ, Madias NE. Secondary responses to altered acid-base status: the rules of engagement. J Am Soc Nephrol. 2010;21:920-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 34. | Pierce NF, Fedson DS, Brigham KL, Mitra RC, Sack RB, Mondal A. The ventilatory response to acute base deficit in humans. Time course during development and correction of metabolic acidosis. Ann Intern Med. 1970;72:633-640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 35. | Wiederseiner JM, Muser J, Lutz T, Hulter HN, Krapf R. Acute metabolic acidosis: Characterization and diagnosis of the disorder and the plasma potassium response. J Am Soc Nephrol. 2004;15:1589-1596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 36. | Choi HS, Kwon A, Chae HW, Suh J, Kim DH, Kim HS. Respiratory failure in a diabetic ketoacidosis patient with severe hypophosphatemia. Ann Pediatr Endocrinol Metab. 2018;23:103-106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 37. | Minagar A, Weiner WJ. Adolf Kussmaul and his respiratory sign. J Med Biogr. 2001;9:181-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 38. | Manifold CA, Davids N, Villers LC, Wampler DA. Capnography for the nonintubated patient in the emergency setting. J Emerg Med. 2013;45:626-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 39. | Parke ML. The ventilatory management of a metabolic acidosis: A case study. Can J Respir Ther. 2012;48:6-12. |
| 40. | Singhi SC, Mathew JL, Jindal A. Clinical pearls in respiratory diseases. Indian J Pediatr. 2011;78:603-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 41. | Wiesel S, Catella P, Siddiqui F, D EL-S. Acute respiratory distress syndrome secondary to influenza a (H1N1 2009) complicated by diabetic ketoacidosis: A fatal case of swine flu. Am J Respir Crit Care Med. 2017;195:A6025. |
| 42. | Mora Carpio AL, Mora JI. Ventilation, Ventilator Management. StatPearls. Treasure Island (FL): StatPearls Publishing. 2018;. [PubMed] |
| 43. | Neto AS, Simonis FD, Barbas CS, Biehl M, Determann RM, Elmer J, Friedman G, Gajic O, Goldstein JN, Linko R, Pinheiro de Oliveira R, Sundar S, Talmor D, Wolthuis EK, Gama de Abreu M, Pelosi P, Schultz MJ; PROtective Ventilation Network Investigators. Lung-Protective Ventilation With Low Tidal Volumes and the Occurrence of Pulmonary Complications in Patients Without Acute Respiratory Distress Syndrome: A Systematic Review and Individual Patient Data Analysis. Crit Care Med. 2015;43:2155-2163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 178] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 44. | Axelrod L. Response of congestive heart failure to correction of hyperglycemia in the presence of diabetic nephropathy. N Engl J Med. 1975;293:1243-1245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 45. | Blicker J, Herd AM, Talbot J. Diabetic ketoacidosis in the dialysis-dependent patient: two case reports and recommendations for treatment. CJEM. 2004;6:281-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 46. | Catalano C, Fabbian F, Di Landro D. Acute pulmonary oedema occurring in association with diabetic ketoacidosis in a diabetic patient with chronic renal failure. Nephrol Dial Transplant. 1998;13:491-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 47. | Kaldany A, Curt GA, Estes NM, Weinrauch LA, Christlieb AR, D’Elia JA. Reversible acute pulmonary edema due to uncontrolled hyperglycemia in diabetic individuals with renal failure. Diabetes Care. 1982;5:506-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 48. | Tzamaloukas AH, Ing TS, Siamopoulos KC, Rohrscheib M, Elisaf MS, Raj DS, Murata GH. Body fluid abnormalities in severe hyperglycemia in patients on chronic dialysis: review of published reports. J Diabetes Complications. 2008;22:29-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 49. | Tzamaloukas AH, Ing TS, Siamopoulos KC, Rohrscheib M, Elisaf MS, Raj DS, Murata GH. Body fluid abnormalities in severe hyperglycemia in patients on chronic dialysis: theoretical analysis. J Diabetes Complications. 2007;21:374-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 50. | Chandorkar A, Codolosa JN, Lippmann ML, Pressman GS, Sta Cruz JP. Recurrent right ventricular Takotsubo cardiomyopathy in a patient with recurrent aspiration. Echocardiography. 2014;31:E240-E242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 51. | Wu WT, Hsu PC, Huang HL, Chen YC, Chien SC. A case of Takotsubo cardiomyopathy precipitated by thyroid storm and diabetic ketoacidosis with poor prognosis. Acta Cardiol Sin. 2014;30:574-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 52. | Christodoulidou M, Selmi F. Severe diabetic ketoacidosis leading to cardiac failure, pulmonary oedema and spinal cord oedema resulting in tetraplegia. BMJ Case Rep. 2012;2012:bcr2012006769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 53. | Sandler M. Is the lung a ‘target organ’ in diabetes mellitus? Arch Intern Med. 1990;150:1385-1388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 75] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 54. | Powner D, Snyder JV, Grenvik A. Altered pulmonary capillary permeability complicating recovery from diabetic ketoacidosis. Chest. 1975;68:253-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 55. | Fernandes Júnior CJ, Hidal JT, Barbas CS, Akamine N, Knobel E. Noncardiogenic pulmonary edema complicating diabetic ketoacidosis. Endocr Pract. 1996;2:379-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 56. | Eisenhut M. Interleukin-1 and the constellation of pulmonary oedema, and cerebral infarctions and oedema in diabetic ketoacidosis. Diabet Med. 2006;23:1386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 57. | Hoffman WH, Burek CL, Waller JL, Fisher LE, Khichi M, Mellick LB. Cytokine response to diabetic ketoacidosis and its treatment. Clin Immunol. 2003;108:175-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 101] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 58. | Hoffman WH, Helman SW, Passmore G. Acute activation of peripheral lymphocytes during treatment of diabetic ketoacidosis. J Diabetes Complications. 2001;15:144-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (1)] |