Published online May 15, 2022. doi: 10.4251/wjgo.v14.i5.973
Peer-review started: April 20, 2021
First decision: August 19, 2021
Revised: September 17, 2021
Accepted: April 1, 2022
Article in press: April 1, 2022
Published online: May 15, 2022
Processing time: 384 Days and 19.9 Hours
Ephrin-A1 is a protein that in humans is encoded by the EFNA1 gene. The ephrins and EPH-related receptors comprise the largest subfamily of receptor protein-tyrosine kinases which play an indispensable role in normal growth and development or in the pathophysiology of various tumors. The role of EFNA1 in tumorigenesis and development is complex and depends on the cell type and microenvironment which in turn affect the expression of EFNA1. This article reviews the expression, prognostic value, regulation and clinical significance of EFNA1 in gastrointestinal tumors.
Core Tip: Ephrin-A1, a protein that in humans is encoded by the EFNA1 gene, is the ligand of EphA2. Studies have shown that the EphA2 receptor and its ligand ephrin-A1 are expressed in a variety of malignant tumors and the interaction between the two promotes the migration of tumor vascular endothelial cells. In addition, studies have shown that EFNA1 widely affects tumor growth through enhancing tumor angiogenesis, malignant cell events and invasiveness. EFNA1 is also up-regulated in gastrointestinal tumors and is closely related to the prognosis of gastrointestinal tumors. Therefore, this article reviews the expression, prognostic value, regulation and clinical significance of EFNA1 in gastrointestinal tumors.
- Citation: Chu LY, Huang BL, Huang XC, Peng YH, Xie JJ, Xu YW. EFNA1 in gastrointestinal cancer: Expression, regulation and clinical significance. World J Gastrointest Oncol 2022; 14(5): 973-988
- URL: https://www.wjgnet.com/1948-5204/full/v14/i5/973.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v14.i5.973
Current theory suggests that a tumor is an "organ" that contains a diverse collection of cells. Different cells sense changes in the external environment through signaling molecules on the surface of cell membrane or plasma membrane. It regulates a series of biological behaviors, such as tumor occurrence, development, invasion and metastasis[1]. Receptor tyrosine kinases (RTKs) can directly transmit external information to the nucleus and are key molecules in the signal transduction pathways through which cells convert external stimuli into biological behavior. The Eph (erythropoietin-producing hepatoma-amplified sequence) receptor family is the largest known family of RTKs[2]. By interacting with its ephrin ligands, Eph receptors regulate physiological and pathological processes, including the formation of tissues and organs, signal transmission of the nervous system, angiogenesis and cell-to-cell adhesion[3]. Studies have shown that the EphA2 receptor and its ligand ephrin-A1 are expressed in a variety of malignant tumors and the interaction between the two promotes the migration of tumor vascular endothelial cells[4]. Therefore, in recent years, the role of ephrins in the occurrence and development of tumors has become a hot topic in cancer research.
Studies have shown that EFNA1 widely affects tumor growth through enhancing tumor angiogenesis[5,6], malignant cell events[7,8] and invasiveness[9-11]. It is up-regulated in gastrointestinal tumors (such as esophageal cancer (EC)[12], colorectal cancer (CRC)[13], and hepatocellular carcinoma (HCC)[14]) and is closely related to the prognosis of gastrointestinal tumors[12-16]. This article summarizes the research progress on EFNA1 in terms of gene composition, protein structure, expression, regulation and biological effects. On this basis, the role of EFNA1 in tumors and its regulatory mechanisms are described in detail as well as its potential clinical significance in gastric cancer (GC), HCC, CRC, EC and some common gastrointestinal cancers.
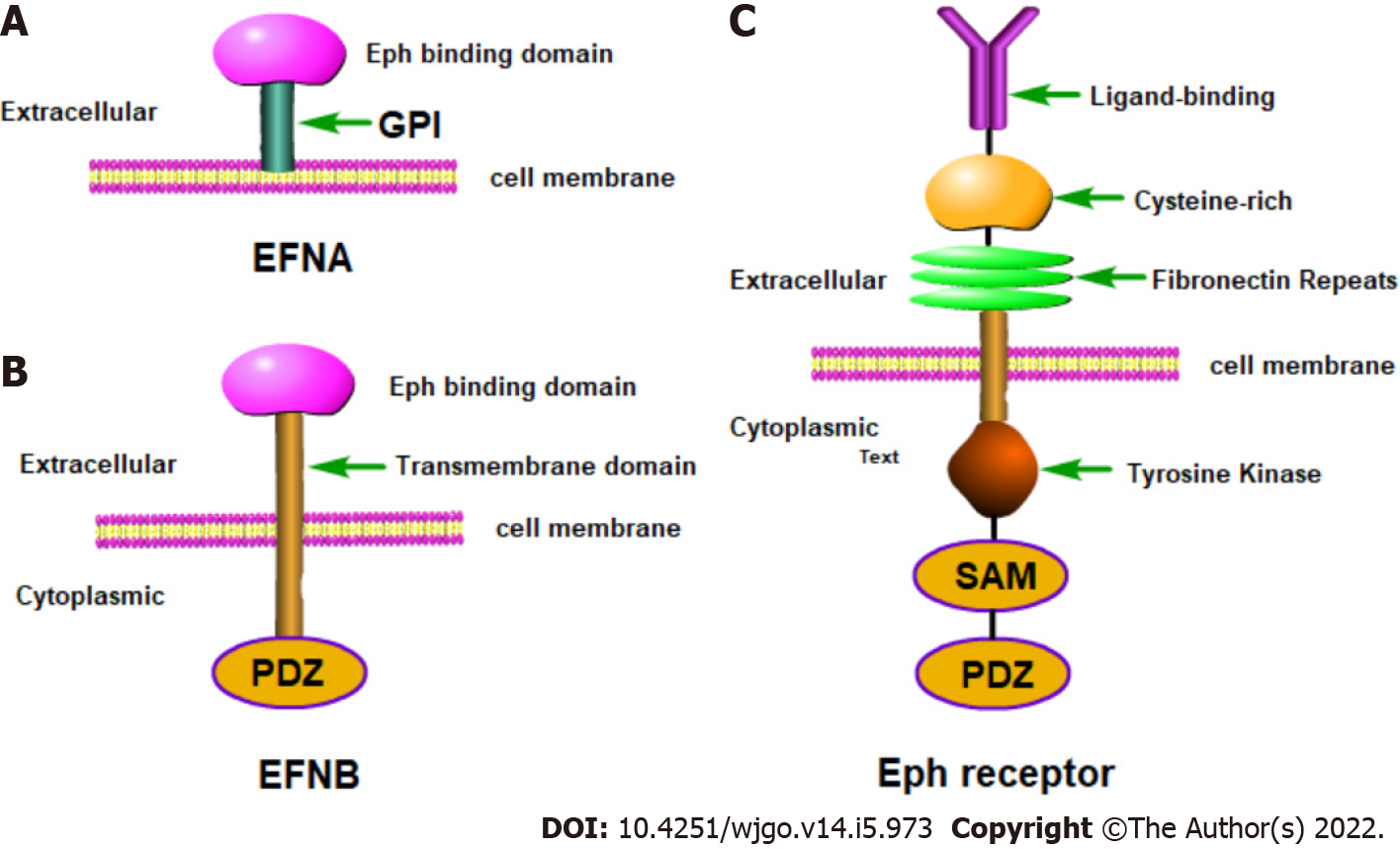
The Eph family contains 14 tyrosine kinase receptors[17] and is the largest known RTK family. The Eph receptor is located on the cell membrane and can directly receive stimulation from the external environment. Eph receptors can also be divided into two categories: A and B, where EphA is comprised of 8 members and EphB is comprised of six members. Eph receptors contain a typical transmembrane structure and belong to transmembrane proteins[18,19]. The typical Eph family receptor structure involves an extracellular domain consisting of a globular domain, a unique cysteine-rich motif and two fibronectin type III motifs. The extracellular domain and the intracellular domain are connected by a short transmembrane domain. The intracellular membrane region is relatively conserved and includes the domain with tyrosine kinase activity, a sterile alpha motif domain and a C-terminal postsynaptic density protein, discs large, zonula occludens (PDZ) domain[20]. Ephrin ligands are divided into two subclasses according to the way they attach to the membrane. Type A ephrins are firmly anchored to the cell membrane with the aid of glycosylphosphatidylinositol (GPI) and include five members (ephrins A1-A5). Type B ephrins are transmembrane proteins[18,19] and include three members (ephrins B1-B3). Ephrin-B contains a PDZ-binding region and there is also a conserved tyrosine residue that can be phosphorylated. Ephrin-A is rather special in that it only contains a receptor-binding region which is coupled to the cell membrane through a GPI anchor. This structure also leads to the specificity of ephrin-A signal transduction (Figure 1).
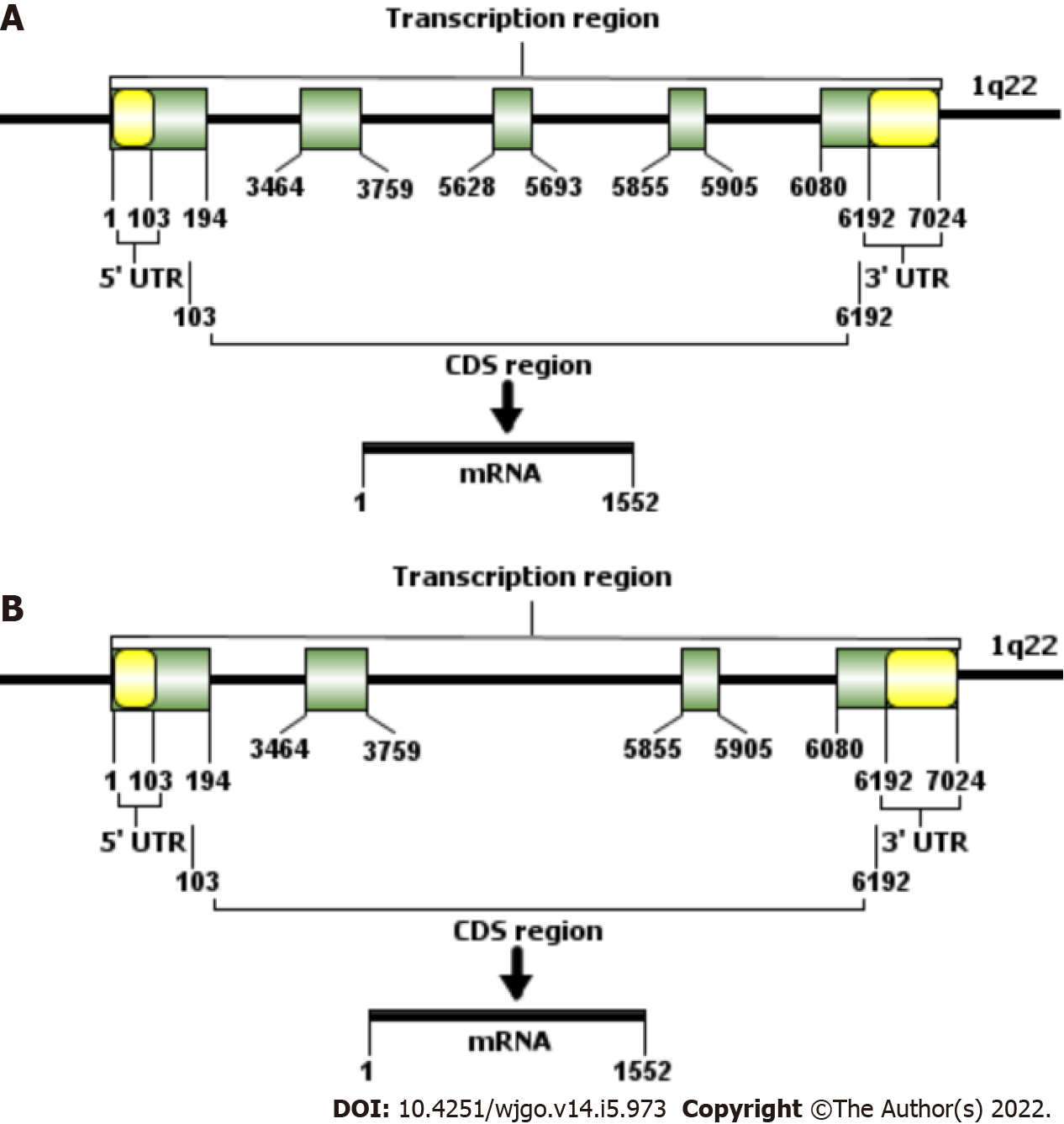
Ephrin-A1 was first discovered in 1990 as a soluble protein produced by human umbilical vein endothelial cells (HUVECs) in response to treatment with tumor necrosis factor (TNF). However, it was not confirmed until 1994 to be a ligand for EphA2 which had been considered an independent RTK kinase before then[21,22]. Ephrin-A1 is a single-chain protein molecule containing 205 amino acid residues, has a molecular weight of 22-KD and is a membrane-coupled ligand protein. The EFNA1 gene, encoding ephrine-A1, is located on chromosome Iq22[20,23]. EFNA1 is 7024 bp in length and contains 5 exons (Table 1 and Figure 2). The length of exon 1 is 194 bp and includes the entire 5' untranslated region (5'UTR). Exons 2 and 3 are 295 bp and 65 bp, respectively, and encode most of the amino acid sequence of the central junction domain. The C-terminus of ephrin-A1 is encoded by exon 4 and the first half of exon 5 (the latter half is the 3' untranslated region (3' UTR)). As early as 1996, a study by Daniel et al[24] found that soluble ephrin-A1 can induce HUVECs cultured in vitro to form capillary-like structures, suggesting that ephrin-A1 has the potential to promote angiogenesis.
| Gene name (known as) | Position and length | Exon number | Encoding mRNA and protein | 5’UTR | CDS | 3’UTR |
| EFNA1 (ephrin-A1; B61; EFL1; GMAN; ECKLG) | 1q22; 7024bp | 5 (1..194, 3464..3759, 5682..5693, 5855..5905,6082..7024) | NM_004428.3, 1552bp, NM_182685.2, 1486 bp; NP_004419.2 , 205aa, NP_872626.1, 183 aa | 1..103 | 103..194, 3464..3759, 5682..5693, 5855..5905,6082..7024 | 6192..7024 |
The binding of Eph receptor to ephrin ligand is very complicated. The same Eph receptor can bind different ephrins and the same ligand can also interact with multiple Eph receptors. EphA2 is the most common receptor for ephrin-A1. The signal transduction by the EphA2 receptor and ephrin-A1 is unique in that they can mediate two-way signal transmission. They can act as receptors or ligands for each other and transmit signals to the cells in which they are located. At present, the signal transmitted by the EphA2 receptor is usually referred to as forward signaling, and the intracellular signal transduction mediated by ephrin-A1 is called the reverse signaling[25,26]. For EphA2 to be activated by ephrin-A1, it must form oligomers in a ligand-dependent manner, indicating that the activation of the EphA2 receptor depends on the interaction between it and the ephrin-A1 Ligand[27]. When EphA2 is activated through ephrin-A1 binding, the tyrosines in their intracellular regions are phosphorylated to form a binding site for another protein, ultimately resulting in the signal transduction complex.
Ephrin is up-regulated in various subtypes of tumor tissues and the up-regulation is closely related to tumor growth[28]. Among the ephrins, ephrin-A1 is highly expressed in human gastrointestinal cancers such as GC, CRC, and EC, as well as HCC. The degree of up-regulation of its expression is closely related to the malignancy of the tumor, metastatic potential and prognosis of the patient[13,29]. We summarize the expression of ephrin-A1 in gastrointestinal cancers and its prognostic value in Table 2.
| Tumor type | Sample type | Expression | Methods | Prognosis value | Notes | Ref. |
| Gastric cancer | Tissues | Increased | RT-PCR | (-) | EFNA1 expression is related to GC stage, depth of invasion, lymph node metastasis and recurrence | Nakamura et al[30], 2005 |
| Tissues | Increased | IHC | Poor DSS | (-) | Miyazaki et al[32], 2013 | |
| Tissues | Increased | IHC, RT-PCR | Poor DSS | EFNA1 expression is related to TNM and lymph node metastasis | Yuan et al[33], 2009 | |
| Tissues | Increased | RT-PCR | (-) | SNP (rs12904G>A) can regulate the expression of EFNA1 and is related to GC susceptibility | Li et al[34], 2014 | |
| Tissues | Increased | (-) | (-) | EFNA1 expression increase the susceptibility of GC | Zhu et al[35], 2015 | |
| Tissues | Increased | (-) | (-) | EFNA1 expression may be related to GC susceptibility | Lee et al[36], 2015 | |
| Tissues | Increased | IHC, RT-PCR | Poor DSS | GMAN up-regulates the expression of EFNA1 and promotes the transfer of GC | Zhou et al[37], 2019 | |
| Colorectal cancer | Cells | (-) | (-) | (-) | EFNA1 overexpression can inhibit the growth of HT29 cells | Potla et al[42], 2002 |
| Tissues | Increased | IHC, RT-PCR | (-) | The expression of EFNA1 promotes the development of rectal adenocarcinoma to rectal cancer | Shi et al[43], 2012 | |
| Tissues | Increased | (-) | (-) | EFNA1 may be used as a diagnostic biomarker for the characteristics of CRC. In addition, the rs12904G/A variant is related to the susceptibility to CRC | Mao et al[44], 2013 | |
| Cells | (-) | (-) | (-) | Eck and B61 are co-expressed in the same cell, suggesting the existence of an autocrine loop | Rosenberg et al[46], 1997 | |
| Tissues | Increased | RT-PCR | Poor DSS | Decreased survival | Kataoka et al[47], 2004 | |
| Cells | Reduced | (-) | Poor DSS | EFNA1 can be used as a prognostic marker for CRC | Robertis et al[48], 2017 | |
| Tissues | Increased | RT-PCR | Poor DSS | EFNA1 is an independent prognostic factor for CRC | Yamamoto et al[49], 2013 | |
| Serum | Increased | IHC, QRT-PCR | Poor DSS | EFNA1 may be used for the identification of CRC | Lip et al[50], 2008 | |
| Hepatocellular carcinoma | Tissues | Increased | RT-PCR | Poor DSS | The high expression of EFNA1 protein is related to histological differentiation, portal vein tumor thrombus and lymph node metastasis | Zhang et al[54], 2007 |
| Tissues | Increased | RT-PCR | Poor DSS | EFNA1 is an independent prognostic factor of HCC | Wada et al[55], 2014 | |
| Tissues | Increased | IHC | Poor DSS | EFNA1 is involved in the mechanism of AFP induction in HCC | Lida et al[57], 2005 | |
| Tissues, Serum | Increased | IHC, RT-PCR | Poor DSS | The expression of EFNA1 is positively correlated with AFP | Cui et al[58], 2010 | |
| Esophageal cancer | Tissues | Increased | IHC, RT-PCR | Poor DSS | Decreased survival | Xu et al[59], 2005 |
| Tissues | Increased | (-) | Poor DSS | Decreased survival | Chen et al[60], 2019 | |
| Cells | Increased | RT-PCR | (-) | High expression of EFNA1 decreased the viability of ESCC cells | Yang et al[61], 2015 |
As a tumor-related secreted protein, ephrin-A1 is highly expressed in most GC tissues and cells. Further studies have found that there is a positive correlation between the expression level of EFNA1 and the degree of malignancy of GC[30-41]. EFNA1 is highly expressed in GC tissues but is low or not expressed in benign GC lesions, and its expression surges with increases in malignancy[30]. Overexpression of ephrin-A1 in GC tumors was reported for 57% of patients in one study and 72.7% of patients in another study, and the overexpression of ephrin-A1 was significantly related to TNM staging and lymph node metastasis[31]. Studies by Miyazaki et al[32] found that EFNA1 is highly expressed in GC, and its high expression may be related to the occurrence, development, invasion and metastasis of GC. EFNA1 expression increases with both clinical stage and lymph node metastasis and decreases in the degree of tissue differentiation, which indicates the malignant degree of GC. Yuan et al[33] studied 176 cases of human GC and found that EFNA1 mRNA and protein are highly expressed in GC, suggesting a pre-transcriptional regulatory mechanism in GC. In addition, the study also found that EFNA1 is greatly expressed in the highly invasive cancer cell line AGS compared with moderately invasive cancer cell lines, suggesting that high expression of ephrin-A1 is related to a more aggressive behavior. These results suggest that EFNA1 plays an important role in progression and metastasis after human GC resection.
Genetic variation of miRNA binding sites may change the susceptibility of individuals to many cancers. Li et al[34] selected 525 GC patients and 501 controls, and selected 3 miRNA binding-site single nucleotide polymorphisms (SNPs) from 30 untranslated regions (UTRs) of GC-related genes to study their relationship with GC susceptibility. It was found that rs12904 in the EFNA1 gene was significantly related to the risk of GC. In addition, luciferase detection showed that EFNA1 mRNA is the target of hsamiR-200c, and expression of the rs12904G>A isoform resulted in a change of luciferase expression. In summary, these findings indicate that the miR-200c binding site containing the SNP (rs12904G>A) can regulate the expression of EFNA1 and is related to GC susceptibility[34-36]. Zhuo et al[37] found that a lncRNA, GMAN, was increased in GC tissues and was associated with GC metastasis and decreased survival rates. GMAN regulates the translation of EFNA1 mRNA by competitively binding antisense GMAN RNA, thereby affecting the invasion and metastasis of GC cells; and up-regulation of GMAN is associated with a poor prognosis of GC.
EFNA1 is highly expressed in most CRC tissues and cells. In recent years, studies based on the relationship between EFNA1 and CRC have shown that it plays an important role in CRC cell growth, invasion, metastasis and angiogenesis[42-52]. Potla et al[42] found that overexpression of EFNA1 can promote the growth of HT29 CRC cells. Ephrin-A1 activates EphA2 to weaken the connections between tumor cells, resulting in increased adhesion of tumor cells to the extracellular matrix (ECM) and enhanced invasion into the matrix. All of these are important characteristics of tumor cells for acquiring the ability to invade and metastasize. Shi et al[43] selected 14 genes through a literature analysis and compared their expression in rectal cancer tissues and para-cancerous tissues, as well as rectal adenomas and cancer tissues. Among them, the gene copy number and mRNA expression of EFNA1 increased in the progression from adenoma to cancer, indicating that EFNA1 may be a driving gene to promote rectal cancer. Studies have also evaluated the genetic association between EFNA1 polymorphisms and susceptibility to CRC. The results showed that, compared with the normal control group, expression of EFNA1 in CRC is increased, suggesting that EFNA1 is involved in the occurrence of CRC and may be used as a diagnostic biomarker for CRC. In addition, it was also found that the rs12904G/A variant is significantly associated with a lower risk of CRC compared with the AA genotype[44,45]. A study by Rosenberg et al[46] showed that the CRC epithelial cell line Caco-2 simultaneously expresses ephrin-A1 (B61) and its receptor EphA2 (Eck). The ephrin-A1 and EphA2 are co-localized in the same cell and play a role in the development, migration and barrier function of CRC epithelial cells helping to maintain the homeostasis and continuity of the epithelial barrier.
Kataoka et al[47] detected the expression of EFNA1 in CRC specimens and found that 62.5% (25/37) expressed ephrin-A1 to a greater extent which correlated with low survival rate and poor prognosis. Overexpression of EFNA1 in CRC stages I and II is more significant than in stages III and IV, and overexpression in tumors < 5 cm is greater than that in tumors > 5 cm. This data suggests an importance of EFNA1 in the early stages of CRC progression. However, the prognostic role of EFNA1 in CRC patients is still controversial. Robertis et al[48] reported that low expression of EFNA1 in CRC cells is indicative of poor patient prognosis, including poor disease-free survival, cancer-specific survival and progression-free survival. However, two other gene chip analyses showed that the prognosis of patients with high EFNA1 expression is worse than that of patients with low expression[49,50]. In addition, multivariate analysis showed that EFNA1 expression is an independent prognostic factor of CRC[49,50]. Therefore, a large sample, multi-center clinical study is needed to verify the prognostic value of EFNA1 in CRC.
EFNA1 is widely expressed in HCC tissues[53-58]. Its expression is lowest in normal liver tissues, increases in liver cirrhosis tissues and is further increased in HCC tissues[54,57,58]. Existing studies have shown that the expression of EFNA1 is related to HCC tissue differentiation and lymph node metastasis. In addition, overexpression of EFNA1 indicates poor prognosis[14,54]. Cox multivariate analysis showed that EFNA1 is an independent prognostic factor of HCC, suggesting that the expression of EFNA1 may be a useful indicator for predicting the high risk of recurrence after radical resection of HCC[55].
In HCC, ephrin-A1 is closely related to expression of alpha-fetoprotein (AFP) and can indicate poor prognosis in patients with AFP[57,58]. A study by Lida et al[57] showed that ephrin-A1 induces the expression of genes related to the cell cycle (p21), angiogenesis, and cell-cell interaction (Rho, integrins, and matrix metalloproteinases) in HCC cells, and these ephrin-A1-induced genes are also activated in HCC tissues overexpressing AFP. Part of the reason for the poor prognosis of HCC patients with AFP is the expression of ephrin-A1 which induces the expression of tumor cell growth, angiogenesis, invasion and metastasis-related genes. In addition, Cui et al[58] found that the frequency of EFNA1 expression in HCC tissues is higher than that of AFP (91% and 45%, respectively). In HCC cell lines and tissues, ephrin-A1 is positively correlated with AFP expression. In terms of secreted proteins, ephrin-A1 is detected in the supernatant of most primary HCC cell lines and it was clearly found that serum ephrin-A1 Levels in HCC patients are elevated. This suggests that EFNA1 can be used as a useful serum marker to measure the development and progress of HCC.
At present, there are few studies on EFNA1 in EC. Existing studies have confirmed that EFNA1 is highly expressed in esophageal squamous cell carcinoma (ESCC) tissues and cells, and is indicative of a relatively poor prognosis[59-61]. Xu et al[59] used immunohistochemistry and reverse transcription-polymerase chain reaction (RT-PCR) to analyze the expression of EFNA1 protein and mRNA in ESCC tissue. The results showed that 84.4% (146/173) sample positively expressed and 15.6% (27/173) sample negatively expressed EFNA1. In addition to overall survival, EFNA1 protein expressions were significantly associated with histological grade, number of lymph node metastasis and clinical stage for patients with ESCC in the univariate analysis. In addition, studies have also shown that ephrin-A1 and EphA2 often co-localize in the tumor area and vascular endothelial cells in ESCC, and their expression is related to co-localization[59]. A study by Chen et al[60] showed that the expression level of EFNA1 in ESCC tissues is higher than that in normal tissues. Survival analysis showed that EFNA1 expression is associated with shorter overall survival. Regarding the expression of EFNA1 in ESCC and its prognostic role, more studies are needed to further confirm these results.
EFNA1 is differentially-expressed in many gastrointestinal cancers and high expression of EFNA1 may have an important function in the formation of the malignant phenotype of gastrointestinal cancers[28-61]. The effects of differential EFNA1 expression on gastrointestinal cancers are mainly manifested in the following aspects.
Ephrin-A1 exerts an inhibitory effect on the growth of GC, CRC, HCC and ESCC cells. Both anchorage-dependent and anchorage-independent growth of tumor cells overexpressing EphA2 was observed to be reduced by treatment with ephrin-A1-Fc, an ephrin-A1 fused to the Fc domain of IgG[30,62]. The EphA2 receptor is activated by its ligand ephrin-A1, triggering the down-regulation of the total expression of EphA2 in GC cells resulting in a net inhibition of the proliferation of GC cells[33]. Potla et al[42] found that in three-dimensional spheroid cultures of HT29 colon cancer cells, an increase of EFNA1 expression reduces the growth of tumor cells. Shi et al[43] reported that the expression of EFNA1 mRNA increases in the progression from rectal adenoma to rectal cancer. In addition, a recent study conducted by Yamamoto et al[49] showed that EFNA1 is an independent prognostic factor for CRC and its loss of function is related to decreased proliferation, invasion and migration of CRC cell lines.
Eph/Ephrin can also regulate the effects of other growth factors on cell growth. Miao et al[63] reported that when EphA2 is activated by ephrin-A1, the Ras/Erk pathway can be inhibited to reduce cell growth induced by platelet-derived growth factor, vascular endothelial growth factor (VEGF) and epidermal growth factor. In addition, the overexpression of EFNA1 is related to the growth and proliferation of gastrointestinal cancer cells and may play the role of a cell growth factor or growth promoting factor[64]. Therefore, in a sense, EFNA1 can be considered as a potential growth factor[65] and its abnormal expression in cancers can affect tumor growth and formation.
Malignant tumor cells often exhibit low cell adhesion which can be due to a lack of cadherin function. Ephrin-A1 has been shown to recruit the Src family kinase Fyn into lipid rafts which is followed by redistribution of vinculin, activation of the mitogen-activated protein kinase pathway, protein tyrosine phosphorylation and increased cell-substrate adhesion[66,67]. In addition, studies have shown that the amount of ephrin-A1 determines the extent of EphA2-dependent, integrin-mediated cell adhesion[68]. In cancer cells lacking cadherin, cell-to-cell contact is reduced. Therefore, EphA2 cannot bind to ephrin-A1 attached to the adjacent cell membrane and cannot undergo tyrosine phosphorylation which facilitates cancer cell detachment from surrounding cells leading to cancer cell spread and increased invasion.
Studies have shown that cadherin can significantly affect the expression and subcellular localization of ephrin-A1/EphA2, and ephrin-A1/EphA2 in turn can also regulate the function of cadherin[69]. EphA2 promotes tumor growth by enhancing the adhesion of tumor cells to the extracellular matrix increasing anchorage-independent growth and angiogenesis[70]. The specific mechanism may be related to the dysfunction of the cadherin glycoprotein in the phosphorylation or distribution of EphA2 at the sites of cell contact[71].
EFNA1 not only plays a role in normal physiological processes but also plays an important role in pathological processes such as tumor formation[72,73]. It has been reported that ephrin-A1 and EphA2 are up-regulated in most gastrointestinal tumors and this up-regulation is related to tumor formation and tumor migration[73-75]. Microarray analysis of 220 CRC samples and RT-PCR analysis of 146 CRC samples showed that loss of ephrin-A1 after siRNA knockdown decreases cell proliferation, invasion and migration. Expression of EFNA1 is a high-risk indicator for predicting recurrence and cancer-related death after radical resection of CRC[49]. Leguchi et al[76] showed that when tumor cells treated with PBS or ephrin-A1-Fc are injected into mice, tumor cells in the lungs can be detected, but that ephrin-A1-Fc treatment increased lung permeability and enhanced tumor metastasis, whereas neutralization by anti- ephrin-A1 antibody reduced the effect.
The regulation of Eph/Ephrin on cancer cell migration is mainly through its influence on the function of integrins. Miao et al[77] showed that when EphA2 is activated, it can inactivate integrin function, inhibit cell spreading, migration and integrin-dependent cell adhesion. They also found that when EphA2 is activated with ephrin-A1, EphA2 can quickly recruit the tyrosine phospholipase SHP2, which can dephosphorylate focal adhesion kinase (FAK) and paxillin, leading to the dissociation of the EphA2 and FAK complex[77,78]. Other data also indicate that the activation of ephrin-A1 can generally increase the adhesion of cells to the extracellular matrix and promote cell migration[79-81].
Tumor angiogenesis is a common pathological phenomenon in carcinogenesis and directly regulates the pathological process of tumor growth, invasion and metastasis. Tumor angiogenesis can bring nutrients and oxygen necessary for tumor cell growth and discharge metabolic waste. At the same time, new blood vessels can be used as a metastasis channel to mediate distant metastasis of tumors[82]. Angiogenesis is regulated by a variety of pro-angiogenic factors and anti-angiogenic factors. Currently, five major protein families are considered to be key regulators of tumor angiogenesis, namely VEGF and its receptor family, angiopoietin and the TIE receptor family, Notch receptor family, Eph/ephrin family and Slit ligand/Robo receptor family[1,83]. Among them, ephrin-A1 and its main receptor EphA2, as the main members of the Eph/ephrin family, are not only significantly expressed in a variety of malignant tumors but are also closely related to normal and tumor angiogenesis.
In 2000, Ogawa et al[84] first reported that ephrin-A1/EphA2 plays an important role in tumor angiogenesis, showing that overexpression of ephrin-A1 in tumor cells promotes tumor angiogenesis, whereas down-regulation of ephrin-A1 expression inhibits tumor cell-induced endothelial cell migration and reduces microvascular density. Functional changes such as migration of vascular endothelial cells, play a key role in tumor angiogenesis. Ephrin-A1 is mainly expressed in tumor cells while EphA2 is mainly expressed in tumor blood vessels. Therefore, it is speculated that tumor cells expressing ephrin-A1 have the effect of attracting endothelial cells expressing EphA2 leading to formation of new blood vessels and angiogenesis. EphA2 expressed on the surface of endothelial cells is a key component in the regulation of angiogenesis. Blocking EphA2 can limit the migration of endothelial cells, vascular reorganization and VEGF-induced angiogenesis.
EphA2 can promote the migration of tumor vascular endothelium and ephrin-A1 has been confirmed to act as a chemical inducer in the process of vascular remodeling[85], suggesting that the interaction between the two in tumor cells and vascular endothelial cells is jointly involved in tumor angiogenesis[85,86]. Combination of the two can promote the migration of tumor vascular endothelial cells and promote the formation of capillary-like structures in tissues and endothelial cells by affecting the cytoskeleton, matrix adhesion and/or cell adhesion. Inhibition of EphA2 activation also reduces tumor angiogenesis, further supporting an important role for EphA2 in tumor neovascularization, invasion and metastasis[85-87]. Pandy et al[85] confirmed that ephrin-A1, not fibroblast growth factors, specifically regulates TNF-α-induced angiogenesis in mice in vivo. This suggests that the induction of ephrin-A1 and subsequent activation of its receptor EphA2 may regulate angiogenesis mediated by TNF-α.
Ogawa et al[84] found that ephrin-A1 and EphA2 are stably expressed in some endothelial cells within gastrointestinal tumors including EC and CRC. In CRC, the expression of ephrin-A1/EphA2 is up-regulated in tumor areas with higher blood vessel density. In small volume CRC tumors (< 5 cm), the expression of ephrin-A1 and EphA2 is higher[47,88]. Liu et al[89] used the microvessel density (MVD) method to label tumor blood vessels with CD34 and directly observe and quantify tumor angiogenesis as well as observe tumor invasion and metastasis. The results of the study showed that MVD in GC tissue is higher than that in adjacent tissues and normal gastric mucosa. MVD increases with the decrease of GC differentiation and increases in infiltration depth, lymph node metastasis and tumor diameter and it is closely related to increased tumor malignancy and metastasis. It is also positively correlated with the expression of EphA2 and ephrin-A1. This suggests that ephrin-A1 may play a role in promoting vascularization and play an important role in the formation of blood vessels in GC.
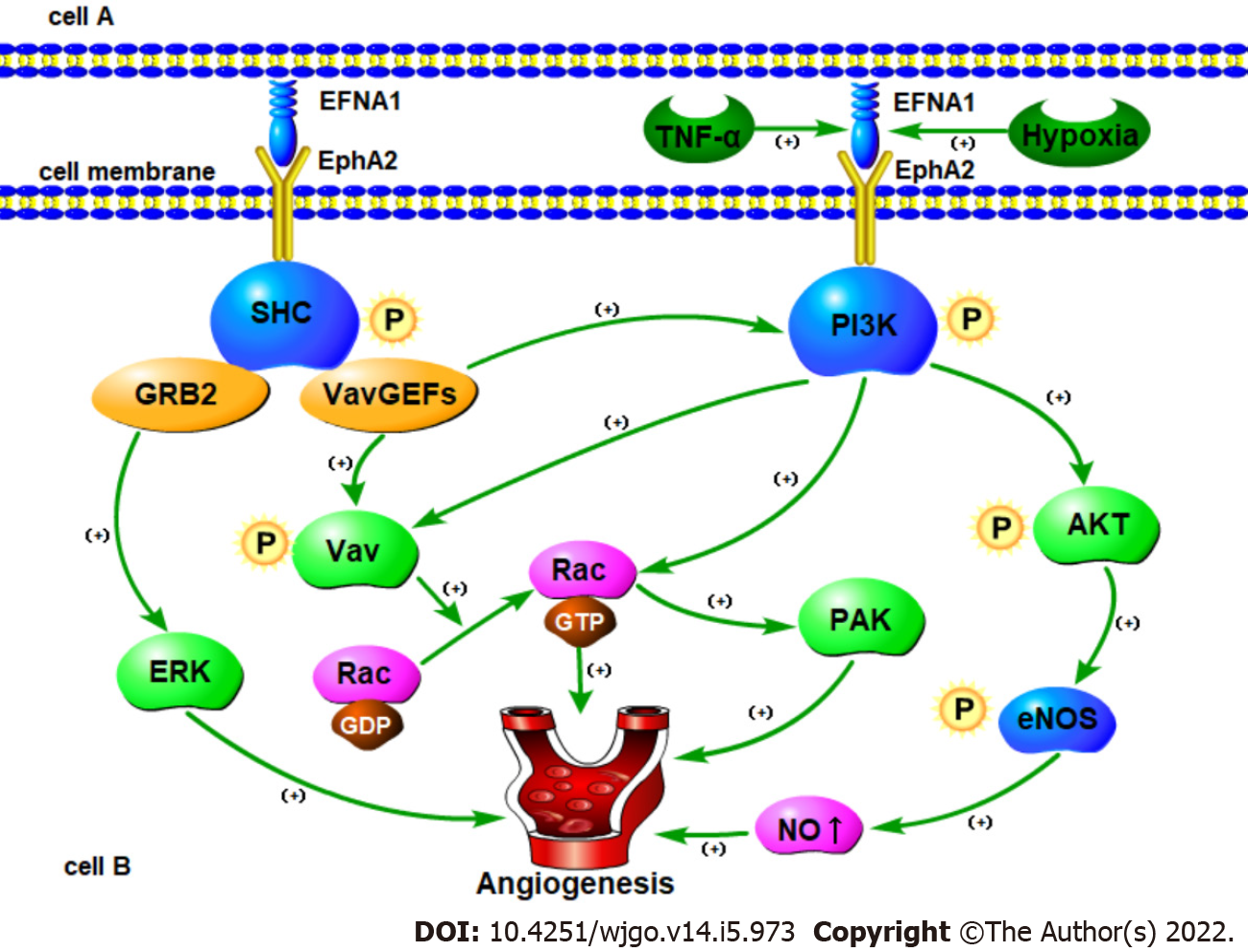
There is sufficient experimental evidence to show that EphA2 activation on endothelial cells is necessary for ephrin-A1 to exert its angiogenic effect in vitro and in vivo[90]. The mechanism by which EFNA1 induces angiogenesis is not fully understood. So far, only a few studies have shed light on the molecular mechanism of ephrin-A1-induced angiogenesis. Based on this, we summarize the possible mechanism by which ephrin-A1 promotes tumor angiogenesis (Figure 3).
EFNA1 can be activated via the ERK1/2 pathway through EphA2 and promote the proliferation, migration and angiogenesis of HUVECs[91,92]. Activation of EphA2 by ephrin-A1 can promote the migration of endothelial cells and the formation of capillary structures by regulating the morphology, migration, adhesion and proliferation of vascular endothelial cells. Interaction between the two has also been confirmed to induce angiogenesis in vivo[93]. For example, ephrin-A1-Fc can increase the adhesion of HUVECs by activating integrins and promoting vascular function[94].
Pratt et al[95] have shown that ephrin-A1-mediated stimulation of EphA2 receptor tyrosine kinase can transmit signals from the cell membrane through MAP kinase. These signals are transmitted to the nucleus by inducing the transcription of Elk-1 and are transmitted back to the cell membrane through the destabilization of the cell's attachment to the ECM. In addition, studies have shown that the biochemical mechanism of EphA2 signaling involves the activation-dependent interaction between tyrosine phosphorylation of EphA2 and SHC adaptor protein. SHC in turn bridges EphA2 to GRB2 which contributes to ERK kinase activation and nuclear translocation.
In different types of cells, growth factors and cytokines can induce the expression of EFNA ligands. Ephrin-A1 was the first EFNA ligand identified and shown to be an immediate early gene product induced by TNF-α in cultured HUVECs[21]. Unlike other angiogenic factors induced by TNF-α[96,97], Cheng et al[98] showed that EFNA1 induction does not require NF-kB or p42/44 MAPK signaling, but rather activation of the JNK and p38MAPK signaling pathways[99]. Both of these pathways have been shown to regulate actin reorganization and cell migration in endothelial cells[100,101]. Therefore, regulating the expression of EFNA1 by p38 MAPK and JNK is consistent with the role of EFNA1 in endothelial cell migration and blood vessel assembly. In addition, Hess et al[102] showed that TNF-α can up-regulate the expression of EFNA1 by acting on HUVECs leading to increased phosphorylation of EphA2 resulting in increased angiogenesis and enhanced cell chemotaxis. Phosphorylation of EphA2 caused by ephrin-A1 can activate phosphatidylinositol 3-hydroxy kinase (PI3K) and up-regulate Rac1 activity thereby causing endothelial cell migration to increase and promote angiogenesis[102].
In addition to TNF-α, ephrin-A1 is also induced by lipopolysaccharide[103], interleukin-1β[21,103], and VEGF in HUVECs and microvascular endothelial cells[98]. The study of Cheng et al[98] showed that similar to TNF-α, VEGF induces ephrin-A1 as an immediate early gene product. Blocking EphA receptor signaling inhibits VEGF-induced endothelial cell survival, migration, in vitro sprouting and in vivo angiogenesis indicating that EphA receptor activation is necessary for VEGF-induced angiogenesis[98]. Ojima[104] and Chen et al[105] showed that soluble ephrin-A1-Fc can promote the tube formation and migration of HUVECs, while EphA2-Fc can antagonize the interaction between EphA2 and ephrin-A1 thereby reducing VEGF-induced endothelial cell migration and proliferation.
Studies have shown that EFNA1 stimulates endothelial cell migration and assembly in culture[84,106], while EphA2 receptor-dependent endothelial cell migration and assembly require activation of Rac1 GTPase[107]. In addition, Vav2 and/or Vav3 are required for ephrin-A1-induced endothelial cell migration/assembly and Rac1 activation[107,108]. Therefore, Hunter et al[108] studied ephrin-A1 and Vav and found that when ephrin-A1 binds to EphA2, EphA2 is phosphorylated by tyrosine. Activated EphA2 can directly recruit Vav-GEFs through the SH2 region so that the Vav protein can be phosphorylated and activated directly or indirectly. In addition, by recruiting p85, EphA2 receptors can also up-regulate phosphatidylinositol-3,4,5-trisphosphate levels through the PH domain and enhance Vav-GEF activity. The activated Vav-GEFs subsequently increase Rac1-GTP levels and promote endothelial cell migration and angiogenesis.
The promotion and inhibition of ephrin-A1 on the same signal pathway has also been observed in different cell or tumor types. It is well known that endothelial nitric oxide synthase (eNOS) and NO play a key role in endothelial cell migration and angiogenesis[109]. There is ample evidence that eNOS is mainly expressed in tumor vascular endothelial cells, and the NO produced by it plays a direct role in tumor angiogenesis induced by various angiogenic factors[110,111]. Hypoxia is one of the most common and important features in the tumor microenvironment which helps induce a variety of angiogenic factors[112].
Therefore, Song et al[113] explored the mechanism of EFNA1 regulating angiogenesis by observing the effect of hypoxia on the expression and secretion of ephrin-A1 in tumor cells and the possible relationship between EFNA1 and eNOS/NO in tumor angiogenesis. Studies have shown that the upregulation of membrane-bound ephrin-A1 induced by hypoxia may interact with EphA2 receptors on endothelial cells in the tumor microenvironment and induce eNOS phosphorylation and increase NO production through PI3K/AKT-dependent pathways thereby promoting tumor angiogenesis. These results show that the PI3K/AKT/eNOS signaling cascade may be a common pathway for hypoxia-induced ephrin-A1-dependent angiogenesis.
Studies have shown that in the vasculature, stimulating vascular smooth muscle cells with ephrin-A1 can inhibit cell proliferation through the inactivation of Rac1 and p21-activated kinase (PAK)[107]. Therefore, ephrin-A1 stimulation leads to inactivation of Rac1 and inhibition of cell proliferation in smooth muscle cells of the blood vessel wall leading to a loss of blood vessels. On the contrary, ephrin-A1 activates Rac1 and induces cell migration and blood vessel assembly of endothelial cells and promotes the sprouting and branching of new capillaries from existing blood vessels[107,114].
However, another study using rat vascular smooth muscle cells showed that ephrin-A1-mediated morphological changes are related to the inhibition of Rac1 and PAK1 activity and are antagonized by the expression of a constitutively-active Rac mutant[115]. The use of siRNA to inhibit the synthesis of Rac1 enhanced the ephrin-A1-induced inhibition of proliferation. Sphingosine-1-phosphate (S1P), a lipid mediator known to inhibit Rac activation in vascular smooth muscle cells, amplifies the effect of ephrin-A1. In conclusion, the authors emphasized the role of the Rac/PAK pathway in ephrin-A1-mediated cell proliferation inhibition. In this way, ephrin-A1 alone or in synergy with S1P can participate in vascular instability which is a prerequisite for angiogenesis[107,115].
EFNA1 is widely expressed in gastrointestinal cancer tissues, especially in highly aggressive cancer cells, suggesting that ephrin-A1 can be used as an important surface marker of gastrointestinal cancer cells and has potential diagnostic and prognostic value. The close relationship between EFNA1 and the occurrence and development of gastrointestinal cancers has been confirmed which could represent a breakthrough in the search for new cancer treatment drugs.
Yang et al[61] found that EFNA1 is involved in the resistance of ESCC cells to Photofrin-mediated photodynamic therapy (PDT). EFNA1 is up-regulated in PDT-resistant ESCC cells and simultaneous incubation with oligomeric ephrin-A1 and soluble ephrin-A1 leads to significant resistance of ESCC cells to Photofrin-PDT[61]. These findings suggest that in ESCC, ephrin-A1 may be an attractive research direction and target for PDT resistance.
Studies have shown that in CRC, the combination of ephrin-A1-Fc and EphA2 can make EphA2 phosphorylated, and the complex formed moves into the cell and gradually degrades, thereby achieving the effect of inhibiting tumor progression[30]. In addition, the overexpression of EphA2 in CRC leads to resistance to chemotherapy[48] and the activation of EphA2 after ephrin-A1 treatment restores the efficacy of cetuximab against CRC cells[116]. These studies show that the combination of ephrin-A1 and cetuximab in tumor treatment provides a method for reversing CRC chemotherapy resistance but more preclinical and clinical studies are needed for confirmation.
Aiming at the specific binding between the G-H loop of ephrin-A1 and the ligand binding domain of EphA2[117], investigators have screened for small molecule antagonists that can selectively block Eph receptors thereby preventing the activation of EphA2[118]. For example, lithocholic acid (LCA), as a small molecule compound, can compete to hinder the binding of ephrin-A1 and EphA2. Its role is to interact with the G-H loop of ephrin-A1 and hinder the binding of ephrin-A1 to its receptor[119]. In addition, anti-EphA2 antibody and EphA2-Fc fusion protein have also been used to block the activation of EphA2,and significant anti-tumor angiogenesis effects have been observed in vitro and in vivo[120-122]. The activation of EphA2 receptors in tumor cells can block the activation of some important oncogenes[123,124] and ephrin-A1-Fc is currently the most widely used EphA2 receptor agonist. Duggineni et al[125] have designed and synthesized peptide molecules that can functionally bind to ephrin-A1 based on the characteristics of the ephrin-A1-binding domain. Such peptides can be expected to become new drugs for tumor suppression, targeted therapy and tumor imaging.
In summary, EFNA1 plays an important role in the occurrence, development and angiogenesis of gastrointestinal tumors and its mechanism of promoting angiogenesis has also been studied in depth. However, the research on EFNA1 and pancreatic cancer is still in the initial exploration stage. In future work, the clinical application of EFNA1 in pancreatic cancer still needs more experiments and clinical studies to conduct a comprehensive verification of the system. In addition, the specific molecular mechanism of EFNA1 in tumor progression is still poorly understood, and many aspects remain to be explored.
Rac/PAK, PI3K/AKT, ERK and other pathways are involved in tumor angiogenesis mediated by EFNA1/EphA2. EFNA1 is expressed in tumor cells and tumor-related blood vessels. Current research mainly focuses on the function and mechanism of EFNA1 in tumor cells and vascular endothelial cells. Tumors are dependent on angiogenesis but there are few reports on whether ephrin-A1 on the surface of tumor cells is related to EphA2 receptors on the surface of vascular endothelial cells or how they interact.
Ephrin-A1 has always been considered a GPI-coupled membrane-coupled ligand and its activation requires cell-to-cell contact. However, in 2008, Wykosky et al[126] found that ephrin-A1 can be secreted from malignant glioma cells and breast cancer cells into the cell supernatant and still retain its ability to activate EphA2. This suggests that ephrin-A1 derived from tumor cells not only acts on adjacent vascular endothelial cells to induce angiogenesis through a paracrine mechanism, but may also act on distant blood vessels to promote angiogenesis.
Hypoxia and inflammation are two major characteristics of the tumor microenvironment. Accompanied by many pathological processes, such as tumor occurrence, development, invasion, metastasis and angiogenesis, they also regulate the expression and function of tumor-related proteins. Studies have found that in solid tumors with hypoxia due to ischemia, the expression of EFNA1 can be significantly upregulated[127]. Vihanto et al[128] also found, using a rat skin hypoxia model, that the expression of ephrin and Eph receptors in skin epithelial cells increases under hypoxic conditions. If it is possible to clarify the effect of hypoxia on the expression of EFNA1 in gastrointestinal tumor cells, especially the effect on the secretion of soluble EFNA1, it may further reveal the function of EFNA1 in gastrointestinal tumors.
Research on EFNA1 in gastrointestinal tumor formation, tumor cell apoptosis and angiogenesis are still in its infancy. Further analysis and study of its signal transduction mechanisms in gastrointestinal tumors will help clarify the mechanism of tumor progression, invasion and metastasis, and provide a more reliable theoretical basis for tumor therapy.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Oncology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Farouk S, Egypt; Kałuzińska Ż, Poland S-Editor: Wang LL L-Editor: Filipodia P-Editor: Wang LL
| 1. | Ziyad S, Iruela-Arispe ML. Molecular mechanisms of tumor angiogenesis. Genes Cancer. 2011;2:1085-1096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 148] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 2. | Himanen JP, Rajashankar KR, Lackmann M, Cowan CA, Henkemeyer M, Nikolov DB. Crystal structure of an Eph receptor-ephrin complex. Nature. 2001;414:933-938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 257] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 3. | Arvanitis D, Davy A. Eph/ephrin signaling: networks. Genes Dev. 2008;22:416-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 249] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 4. | Cheng N, Brantley DM, Liu H, Lin Q, Enriquez M, Gale N, Yancopoulos G, Cerretti DP, Daniel TO, Chen J. Blockade of EphA receptor tyrosine kinase activation inhibits vascular endothelial cell growth factor-induced angiogenesis. Mol Cancer Res. 2002;1:2-11. [PubMed] |
| 5. | Ma TT, Wang L, Wang JL, Liu YJ, Chen YC, He HJ, Song Y. Hypoxia-Induced Cleavage Of Soluble ephrinA1 From Cancer Cells Is Mediated By MMP-2 And Associates With Angiogenesis In Oral Squamous Cell Carcinoma. Onco Targets Ther. 2019;12:8491-8499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 6. | Brantley-Sieders DM, Fang WB, Hwang Y, Hicks D, Chen J. Ephrin-A1 facilitates mammary tumor metastasis through an angiogenesis-dependent mechanism mediated by EphA receptor and vascular endothelial growth factor in mice. Cancer Res. 2006;66:10315-10324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 106] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 7. | Genander M, Frisén J. Ephrins and Eph receptors in stem cells and cancer. Curr Opin Cell Biol. 2010;22:611-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 121] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 8. | Khodayari N, Mohammed KA, Lee H, Kaye F, Nasreen N. MicroRNA-302b targets Mcl-1 and inhibits cell proliferation and induces apoptosis in malignant pleural mesothelioma cells. Am J Cancer Res. 2016;6:1996-2009. [PubMed] |
| 9. | Salem AF, Gambini L, Udompholkul P, Baggio C, Pellecchia M. Therapeutic Targeting of Pancreatic Cancer via EphA2 Dimeric Agonistic Agents. Pharmaceuticals (Basel). 2020;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 10. | Hamaoka Y, Negishi M, Katoh H. EphA2 is a key effector of the MEK/ERK/RSK pathway regulating glioblastoma cell proliferation. Cell Signal. 2016;28:937-945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 57] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 11. | Miao H, Gale NW, Guo H, Qian J, Petty A, Kaspar J, Murphy AJ, Valenzuela DM, Yancopoulos G, Hambardzumyan D, Lathia JD, Rich JN, Lee J, Wang B. EphA2 promotes infiltrative invasion of glioma stem cells in vivo through cross-talk with Akt and regulates stem cell properties. Oncogene. 2015;34:558-567. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 134] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 12. | Miyazaki T, Kato H, Fukuchi M, Nakajima M, Kuwano H. EphA2 overexpression correlates with poor prognosis in esophageal squamous cell carcinoma. Int J Cancer. 2003;103:657-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 163] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 13. | Hao Y, Li G. Role of EFNA1 in tumorigenesis and prospects for cancer therapy. Biomed Pharmacother. 2020;130:110567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 14. | Ieguchi K. Eph as a target in inflammation. Endocr Metab Immune Disord Drug Targets. 2015;15:119-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 15. | Shi L, Itoh F, Itoh S, Takahashi S, Yamamoto M, Kato M. Ephrin-A1 promotes the malignant progression of intestinal tumors in Apc(min/+) mice. Oncogene. 2008;27:3265-3273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 16. | Hong HN, Won YJ, Shim JH, Kim HJ, Han SH, Kim BS, Kim HS. Cancer-associated fibroblasts promote gastric tumorigenesis through EphA2 activation in a ligand-independent manner. J Cancer Res Clin Oncol. 2018;144:1649-1663. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 17. | Luo H, Wan X, Wu Y, Wu J. Cross-linking of EphB6 resulting in signal transduction and apoptosis in Jurkat cells. J Immunol. 2001;167:1362-1370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 58] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 18. | Ferluga S, Hantgan R, Goldgur Y, Himanen JP, Nikolov DB, Debinski W. Biological and structural characterization of glycosylation on ephrin-A1, a preferred ligand for EphA2 receptor tyrosine kinase. J Biol Chem. 2013;288:18448-18457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 19. | Kullander K, Klein R. Mechanisms and functions of Eph and ephrin signalling. Nat Rev Mol Cell Biol. 2002;3:475-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 893] [Cited by in RCA: 907] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 20. | Wykosky J, Debinski W. The EphA2 receptor and ephrinA1 Ligand in solid tumors: function and therapeutic targeting. Mol Cancer Res. 2008;6:1795-1806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 247] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 21. | Holzman LB, Marks RM, Dixit VM. A novel immediate-early response gene of endothelium is induced by cytokines and encodes a secreted protein. Mol Cell Biol. 1990;10:5830-5838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 40] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 22. | Lindberg RA, Hunter T. cDNA cloning and characterization of eck, an epithelial cell receptor protein-tyrosine kinase in the eph/elk family of protein kinases. Mol Cell Biol. 1990;10:6316-6324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 86] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 23. | Xiao T, Xiao Y, Wang W, Tang YY, Xiao Z, Su M. Targeting EphA2 in cancer. J Hematol Oncol. 2020;13:114. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 146] [Article Influence: 29.2] [Reference Citation Analysis (0)] |
| 24. | Daniel TO, Stein E, Cerretti DP, St John PL, Robert B, Abrahamson DR. ELK and LERK-2 in developing kidney and microvascular endothelial assembly. Kidney Int Suppl. 1996;57:S73-S81. [PubMed] |
| 25. | Luxey M, Jungas T, Laussu J, Audouard C, Garces A, Davy A. Eph:ephrin-B1 forward signaling controls fasciculation of sensory and motor axons. Dev Biol. 2013;383:264-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 26. | Allen-Sharpley MR, Cramer KS. Coordinated Eph-ephrin signaling guides migration and axon targeting in the avian auditory system. Neural Dev. 2012;7:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Bocharov EV, Mayzel ML, Volynsky PE, Goncharuk MV, Ermolyuk YS, Schulga AA, Artemenko EO, Efremov RG, Arseniev AS. Spatial structure and pH-dependent conformational diversity of dimeric transmembrane domain of the receptor tyrosine kinase EphA1. J Biol Chem. 2008;283:29385-29395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 94] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 28. | Hafner C, Schmitz G, Meyer S, Bataille F, Hau P, Langmann T, Dietmaier W, Landthaler M, Vogt T. Differential gene expression of Eph receptors and ephrins in benign human tissues and cancers. Clin Chem. 2004;50:490-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 233] [Cited by in RCA: 263] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 29. | Ieguchi K, Maru Y. Roles of EphA1/A2 and ephrin-A1 in cancer. Cancer Sci. 2019;110:841-848. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 54] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 30. | Nakamura R, Kataoka H, Sato N, Kanamori M, Ihara M, Igarashi H, Ravshanov S, Wang YJ, Li ZY, Shimamura T, Kobayashi T, Konno H, Shinmura K, Tanaka M, Sugimura H. EPHA2/EFNA1 expression in human gastric cancer. Cancer Sci. 2005;96:42-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 110] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 31. | Katoh Y, Katoh M. Comparative integromics on Ephrin family. Oncol Rep. 2006;15:1391-1395. [PubMed] |
| 32. | Miyazaki K, Inokuchi M, Takagi Y, Kato K, Kojima K, Sugihara K. EphA4 is a prognostic factor in gastric cancer. BMC Clin Pathol. 2013;13:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 43] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 33. | Yuan WJ, Ge J, Chen ZK, Wu SB, Shen H, Yang P, Hu B, Zhang GW, Chen ZH. Over-expression of EphA2 and EphrinA-1 in human gastric adenocarcinoma and its prognostic value for postoperative patients. Dig Dis Sci. 2009;54:2410-2417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 34. | Li Y, Nie Y, Cao J, Tu S, Lin Y, Du Y, Li Y. G-A variant in miR-200c binding site of EFNA1 alters susceptibility to gastric cancer. Mol Carcinog. 2014;53:219-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 35. | Zhu H, Yang M, Zhang H, Chen X, Yang X, Zhang C, Qin Q, Cheng H, Sun X. Genome-wide association pathway analysis to identify candidate single nucleotide polymorphisms and molecular pathways for gastric adenocarcinoma. Tumour Biol. 2015;36:5635-5639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 36. | Lee JH, Kim Y, Choi JW, Kim YS. Genetic variants and risk of gastric cancer: a pathway analysis of a genome-wide association study. Springerplus. 2015;4:215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 37. | Zhuo W, Liu Y, Li S, Guo D, Sun Q, Jin J, Rao X, Li M, Sun M, Jiang M, Xu Y, Teng L, Jin Y, Si J, Liu W, Kang Y, Zhou T. Long Noncoding RNA GMAN, Up-regulated in Gastric Cancer Tissues, Is Associated With Metastasis in Patients and Promotes Translation of Ephrin A1 by Competitively Binding GMAN-AS. Gastroenterology. 2019;156:676-691.e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 196] [Article Influence: 32.7] [Reference Citation Analysis (0)] |
| 38. | Bennett BD, Wang Z, Kuang WJ, Wang A, Groopman JE, Goeddel DV, Scadden DT. Cloning and characterization of HTK, a novel transmembrane tyrosine kinase of the EPH subfamily. J Biol Chem. 1994;269:14211-14218. [PubMed] |
| 39. | Yan SX, Wang DG, Chen YJ, Liu HC, Xu YC. Expression of EphA2 and Ephrin a1 in gastric cancer tissues with helicobacter pylori infection and its relationship with distant metastasis. Medicine and Philosophy. 2016;37:63-66. [DOI] [Full Text] |
| 40. | Kiyokawa E, Takai S, Tanaka M, Iwase T, Suzuki M, Xiang YY, Naito Y, Yamada K, Sugimura H, Kino I. Overexpression of ERK, an EPH family receptor protein tyrosine kinase, in various human tumors. Cancer Res. 1994;54:3645-3650. [PubMed] |
| 41. | Coffman KT, Hu M, Carles-Kinch K, Tice D, Donacki N, Munyon K, Kifle G, Woods R, Langermann S, Kiener PA, Kinch MS. Differential EphA2 epitope display on normal vs malignant cells. Cancer Res. 2003;63:7907-7912. [PubMed] |
| 42. | Potla L, Boghaert ER, Armellino D, Frost P, Damle NK. Reduced expression of EphrinA1 (EFNA1) inhibits three-dimensional growth of HT29 colon carcinoma cells. Cancer Lett. 2002;175:187-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 43. | Shi ZZ, Zhang YM, Shang L, Hao JJ, Zhang TT, Wang BS, Liang JW, Chen X, Zhang Y, Wang GQ, Wang MR. Genomic profiling of rectal adenoma and carcinoma by array-based comparative genomic hybridization. BMC Med Genomics. 2012;5:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 44. | Mao YY, Jing FY, Jin MJ, Li YJ, Ding Y, Guo J, Wang FJ, Jiang LF, Chen K. rs12904 polymorphism in the 3'UTR of EFNA1 is associated with colorectal cancer susceptibility in a Chinese population. Asian Pac J Cancer Prev. 2013;14: 5037-5041. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 45. | Simonian M, Mosallaei M, Khosravi S, Salehi R. rs12904 polymorphism in the 3'-untranslated region of ephrin A1 Ligand and the risk of sporadic colorectal cancer in the Iranian population. J Cancer Res Ther. 2019;15:15-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 46. | Rosenberg IM, Göke M, Kanai M, Reinecker HC, Podolsky DK. Epithelial cell kinase-B61: an autocrine loop modulating intestinal epithelial migration and barrier function. Am J Physiol. 1997;273:G824-G832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 36] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 47. | Kataoka H, Igarashi H, Kanamori M, Ihara M, Wang JD, Wang YJ, Li ZY, Shimamura T, Kobayashi T, Maruyama K, Nakamura T, Arai H, Kajimura M, Hanai H, Tanaka M, Sugimura H. Correlation of EPHA2 overexpression with high microvessel count in human primary colorectal cancer. Cancer Sci. 2004;95:136-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 81] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 48. | De Robertis M, Loiacono L, Fusilli C, Poeta ML, Mazza T, Sanchez M, Marchionni L, Signori E, Lamorte G, Vescovi AL, Garcia-Foncillas J, Fazio VM. Dysregulation of EGFR Pathway in EphA2 Cell Subpopulation Significantly Associates with Poor Prognosis in Colorectal Cancer. Clin Cancer Res. 2017;23:159-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 72] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 49. | Yamamoto H, Tei M, Uemura M, Takemasa I, Uemura Y, Murata K, Fukunaga M, Ohue M, Ohnishi T, Ikeda K, Kato T, Okamura S, Ikenaga M, Haraguchi N, Nishimura J, Mizushima T, Mimori K, Doki Y, Mori M. Ephrin-A1 mRNA is associated with poor prognosis of colorectal cancer. Int J Oncol. 2013;42:549-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 50. | Lips EH, van Eijk R, de Graaf EJ, Oosting J, de Miranda NF, Karsten T, van de Velde CJ, Eilers PH, Tollenaar RA, van Wezel T, Morreau H. Integrating chromosomal aberrations and gene expression profiles to dissect rectal tumorigenesis. BMC Cancer. 2008;8:314. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 51. | Xiong Y, Li KX, Wei H, Jiao L, Yu SY, Zeng L. Eph/ephrin signalling serves a bidirectional role in lipopolysaccharide-induced intestinal injury. Mol Med Rep. 2018;18:2171-2181. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 52. | Brantley-Sieders DM, Chen J. Eph receptor tyrosine kinases in angiogenesis: from development to disease. Angiogenesis. 2004;7:17-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 113] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 53. | Sepehri Z, Kiani Z, Kohan F, Alavian SM, Ghavami S. Toll like receptor 4 and hepatocellular carcinoma; A systematic review. Life Sci. 2017;179:80-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 54. | Zhang G, Zhang SJ, Zhao YF, Wu Y, Li Z, Wang JX. Expression and clinical significance of Ephrin-A1 in primary hepatocellular carcinoma. Zhonghua Waikexue Zazhi. 2007;45:499-502. [PubMed] |
| 55. | Wada H, Yamamoto H, Kim C, Uemura M, Akita H, Tomimaru Y, Hama N, Kawamoto K, Kobayashi S, Eguchi H, Umeshita K, Doki Y, Mori M, Nagano H. Association between ephrin-A1 mRNA expression and poor prognosis after hepatectomy to treat hepatocellular carcinoma. Int J Oncol. 2014;45:1051-1058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 56. | Yu HT, Guo PY, Xie XZ, Chen G. The effect of regulated EphA1/EphrinA1 signaling axis on endothelial progenitor cells to promote their angiogenesis potency in hepatocellular carcinoma. Wenzhou Yixueyuan Xuebao. 2019;49:791-796. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 57. | Iida H, Honda M, Kawai HF, Yamashita T, Shirota Y, Wang BC, Miao H, Kaneko S. Ephrin-A1 expression contributes to the malignant characteristics of {alpha}-fetoprotein producing hepatocellular carcinoma. Gut. 2005;54:843-851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 74] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 58. | Cui XD, Lee MJ, Yu GR, Kim IH, Yu HC, Song EY, Kim DG. EFNA1 Ligand and its receptor EphA2: potential biomarkers for hepatocellular carcinoma. Int J Cancer. 2010;126:940-949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 47] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 59. | Xu F, Zhong W, Li J, Shanshen Z, Cui J, Nesland JM, Suo Z. Predictive value of EphA2 and EphrinA-1 expression in oesophageal squamous cell carcinoma. Anticancer Res. 2005;25:2943-2950. [PubMed] |
| 60. | Chen FF, Zhang SR, Peng H, Chen YZ, Cui XB. Integrative genomics analysis of hub genes and their relationship with prognosis and signaling pathways in esophageal squamous cell carcinoma. Mol Med Rep. 2019;20:3649-3660. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 61. | Yang PW, Chiang TH, Hsieh CY, Huang YC, Wong LF, Hung MC, Tsai JC, Lee JM. The effect of ephrin-A1 on resistance to Photofrin-mediated photodynamic therapy in esophageal squamous cell carcinoma cells. Lasers Med Sci. 2015;30:2353-2361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 62. | Wykosky J, Gibo DM, Stanton C, Debinski W. EphA2 as a novel molecular marker and target in glioblastoma multiforme. Mol Cancer Res. 2005;3:541-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 224] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 63. | Miao H, Wei BR, Peehl DM, Li Q, Alexandrou T, Schelling JR, Rhim JS, Sedor JR, Burnett E, Wang B. Activation of EphA receptor tyrosine kinase inhibits the Ras/MAPK pathway. Nat Cell Biol. 2001;3:527-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 278] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 64. | Easty DJ, Hill SP, Hsu MY, Fallowfield ME, Florenes VA, Herlyn M, Bennett DC. Up-regulation of ephrin-A1 during melanoma progression. Int J Cancer. 1999;84:494-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 65. | Tuzi NL, Gullick WJ. eph, the largest known family of putative growth factor receptors. Br J Cancer. 1994;69:417-421. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 92] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 66. | Davy A, Gale NW, Murray EW, Klinghoffer RA, Soriano P, Feuerstein C, Robbins SM. Compartmentalized signaling by GPI-anchored ephrin-A5 requires the Fyn tyrosine kinase to regulate cellular adhesion. Genes Dev. 1999;13:3125-3135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 242] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 67. | Huai J, Drescher U. An ephrin-A-dependent signaling pathway controls integrin function and is linked to the tyrosine phosphorylation of a 120-kDa protein. J Biol Chem. 2001;276:6689-6694. [RCA] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 132] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 68. | Pasquale EB. Eph receptor signalling casts a wide net on cell behaviour. Nat Rev Mol Cell Biol. 2005;6:462-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 764] [Cited by in RCA: 836] [Article Influence: 41.8] [Reference Citation Analysis (0)] |
| 69. | Orsulic S, Kemler R. Expression of Eph receptors and ephrins is differentially regulated by E-cadherin. J Cell Sci. 2000;113:1793-1802. [PubMed] |
| 70. | Lu C, Shahzad MM, Wang H, Landen CN, Kim SW, Allen J, Nick AM, Jennings N, Kinch MS, Bar-Eli M, Sood AK. EphA2 overexpression promotes ovarian cancer growth. Cancer Biol Ther. 2008;7:1098-1103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 71. | Zantek ND, Azimi M, Fedor-Chaiken M, Wang B, Brackenbury R, Kinch MS. E-cadherin regulates the function of the EphA2 receptor tyrosine kinase. Cell Growth Differ. 1999;10:629-638. [PubMed] |
| 72. | Surawska H, Ma PC, Salgia R. The role of ephrins and Eph receptors in cancer. Cytokine Growth Factor Rev. 2004;15:419-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 263] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 73. | Dodelet VC, Pasquale EB. Eph receptors and ephrin ligands: embryogenesis to tumorigenesis. Oncogene. 2000;19:5614-5619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 230] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 74. | Chen J, Zhuang G, Frieden L, Debinski W. Eph receptors and Ephrins in cancer: common themes and controversies. Cancer Res. 2008;68:10031-10033. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 75. | Wimmer-Kleikamp SH, Lackmann M. Eph-modulated cell morphology, adhesion and motility in carcinogenesis. IUBMB Life. 2005;57:421-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 51] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 76. | Ieguchi K, Tomita T, Omori T, Komatsu A, Deguchi A, Masuda J, Duffy SL, Coulthard MG, Boyd A, Maru Y. ADAM12-cleaved ephrin-A1 contributes to lung metastasis. Oncogene. 2014;33:2179-2190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 61] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 77. | Miao H, Burnett E, Kinch M, Simon E, Wang B. Activation of EphA2 kinase suppresses integrin function and causes focal-adhesion-kinase dephosphorylation. Nat Cell Biol. 2000;2:62-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 413] [Cited by in RCA: 431] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 78. | Hynes RO. Integrins: versatility, modulation, and signaling in cell adhesion. Cell. 1992;69:11-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7430] [Cited by in RCA: 7404] [Article Influence: 224.4] [Reference Citation Analysis (0)] |
| 79. | Davy A, Robbins SM. Ephrin-A5 modulates cell adhesion and morphology in an integrin-dependent manner. EMBO J. 2000;19:5396-5405. [RCA] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 175] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 80. | Meyer S, Hafner C, Guba M, Flegel S, Geissler EK, Becker B, Koehl GE, Ors öE, Landthaler M, Vogt T. Ephrin-B2 overexpression enhances integrin-mediated ECM-attachment and migration of B16 melanoma cells. Int J Oncol. 2005;27:1197-1206. [RCA] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 81. | Huynh-Do U, Vindis C, Liu H, Cerretti DP, McGrew JT, Enriquez M, Chen J, Daniel TO. Ephrin-B1 transduces signals to activate integrin-mediated migration, attachment and angiogenesis. J Cell Sci. 2002;115:3073-3081. [PubMed] |
| 82. | Folkman J. Role of angiogenesis in tumor growth and metastasis. Semin Oncol. 2002;29:15-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1845] [Cited by in RCA: 1937] [Article Influence: 84.2] [Reference Citation Analysis (0)] |
| 83. | Carmeliet P, Jain RK. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011;473:298-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3471] [Cited by in RCA: 4027] [Article Influence: 287.6] [Reference Citation Analysis (0)] |
| 84. | Ogawa K, Pasqualini R, Lindberg RA, Kain R, Freeman AL, Pasquale EB. The ephrin-A1 Ligand and its receptor, EphA2, are expressed during tumor neovascularization. Oncogene. 2000;19:6043-6052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 287] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 85. | Pandey A, Shao H, Marks RM, Polverini PJ, Dixit VM. Role of B61, the ligand for the Eck receptor tyrosine kinase, in TNF-alpha-induced angiogenesis. Science. 1995;268:567-569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 282] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 86. | Pasquale EB. Eph-ephrin bidirectional signaling in physiology and disease. Cell. 2008;133:38-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 903] [Cited by in RCA: 998] [Article Influence: 58.7] [Reference Citation Analysis (0)] |
| 87. | Brantley-Sieders DM, Fang WB, Hicks DJ, Zhuang G, Shyr Y, Chen J. Impaired tumor microenvironment in EphA2-deficient mice inhibits tumor angiogenesis and metastatic progression. FASEB J. 2005;19:1884-1886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 104] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 88. | Ieguchi K, Tomita T, Takao T, Omori T, Mishima T, Shimizu I, Tognolini M, Lodola A, Tsunoda T, Kobayashi S, Wada S, Maru Y. Analysis of ADAM12-Mediated Ephrin-A1 Cleavage and Its Biological Functions. Int J Mol Sci. 2021;22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 89. | Liu H, Guo JW, Liu JH, Zuo LF. EphA2/EphrinA1 expressions in human gastric carcinoma and their relationship with angiogenesis. ACTA academiae medicinae militaris tertiae. 2008;30:2183-2186. [DOI] [Full Text] |
| 90. | Chu M, Zhang C. Inhibition of angiogenesis by leflunomide via targeting the soluble ephrin-A1/EphA2 system in bladder cancer. Sci Rep. 2018;8:1539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 33] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 91. | Tang FY, Chiang EP, Shih CJ. Green tea catechin inhibits ephrin-A1-mediated cell migration and angiogenesis of human umbilical vein endothelial cells. J Nutr Biochem. 2007;18:391-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 92. | Vaught D, Brantley-Sieders DM, Chen J. Eph receptors in breast cancer: roles in tumor promotion and tumor suppression. Breast Cancer Res. 2008;10:217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 73] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 93. | Saik JE, Gould DJ, Keswani AH, Dickinson ME, West JL. Biomimetic hydrogels with immobilized ephrinA1 for therapeutic angiogenesis. Biomacromolecules. 2011;12:2715-2722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 52] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 94. | Moon JJ, Lee SH, West JL. Synthetic biomimetic hydrogels incorporated with ephrin-A1 for therapeutic angiogenesis. Biomacromolecules. 2007;8:42-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 67] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 95. | Pratt RL, Kinch MS. Activation of the EphA2 tyrosine kinase stimulates the MAP/ERK kinase signaling cascade. Oncogene. 2002;21:7690-7699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 110] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 96. | Boyle EM, Jr. , Kovacich JC, Canty TG, Jr., Morgan EN, Chi E, Verrier ED, Pohlman TH. Inhibition of nuclear factor-kappa B nuclear localization reduces human E-selectin expression and the systemic inflammatory response. Circulation. 1998;98:II282-288. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 97. | Yoshida S, Ono M, Shono T, Izumi H, Ishibashi T, Suzuki H, Kuwano M. Involvement of interleukin-8, vascular endothelial growth factor, and basic fibroblast growth factor in tumor necrosis factor alpha-dependent angiogenesis. Mol Cell Biol. 1997;17:4015-4023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 456] [Cited by in RCA: 485] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 98. | Cheng N, Brantley DM, Chen J. The ephrins and Eph receptors in angiogenesis. Cytokine Growth Factor Rev. 2002;13:75-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 231] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 99. | Cheng N, Chen J. Tumor necrosis factor-alpha induction of endothelial ephrin A1 expression is mediated by a p38 MAPK- and SAPK/JNK-dependent but nuclear factor-kappa B-independent mechanism. J Biol Chem. 2001;276:13771-13777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 60] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 100. | Shi CS, Leonardi A, Kyriakis J, Siebenlist U, Kehrl JH. TNF-mediated activation of the stress-activated protein kinase pathway: TNF receptor-associated factor 2 recruits and activates germinal center kinase related. J Immunol. 1999;163:3279-3285. [PubMed] |
| 101. | Rousseau S, Houle F, Landry J, Huot J. p38 MAP kinase activation by vascular endothelial growth factor mediates actin reorganization and cell migration in human endothelial cells. Oncogene. 1997;15:2169-2177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 656] [Cited by in RCA: 666] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 102. | Hess AR, Margaryan NV, Seftor EA, Hendrix MJ. Deciphering the signaling events that promote melanoma tumor cell vasculogenic mimicry and their link to embryonic vasculogenesis: role of the Eph receptors. Dev Dyn. 2007;236:3283-3296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 65] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 103. | Ivanov AI, Steiner AA, Scheck AC, Romanovsky AA. Expression of Eph receptors and their ligands, ephrins, during lipopolysaccharide fever in rats. Physiol Genomics. 2005;21:152-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 104. | Ojima T, Takagi H, Suzuma K, Oh H, Suzuma I, Ohashi H, Watanabe D, Suganami E, Murakami T, Kurimoto M, Honda Y, Yoshimura N. EphrinA1 inhibits vascular endothelial growth factor-induced intracellular signaling and suppresses retinal neovascularization and blood-retinal barrier breakdown. Am J Pathol. 2006;168:331-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 62] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 105. | Chen J, Hicks D, Brantley-Sieders D, Cheng N, McCollum GW, Qi-Werdich X, Penn J. Inhibition of retinal neovascularization by soluble EphA2 receptor. Exp Eye Res. 2006;82:664-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 51] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 106. | Bergers G, Benjamin LE. Tumorigenesis and the angiogenic switch. Nat Rev Cancer. 2003;3:401-410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2432] [Cited by in RCA: 2465] [Article Influence: 112.0] [Reference Citation Analysis (0)] |
| 107. | Brantley-Sieders DM, Caughron J, Hicks D, Pozzi A, Ruiz JC, Chen J. EphA2 receptor tyrosine kinase regulates endothelial cell migration and vascular assembly through phosphoinositide 3-kinase-mediated Rac1 GTPase activation. J Cell Sci. 2004;117:2037-2049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 160] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 108. | Hunter SG, Zhuang G, Brantley-Sieders D, Swat W, Cowan CW, Chen J. Essential role of Vav family guanine nucleotide exchange factors in EphA receptor-mediated angiogenesis. Mol Cell Biol. 2006;26:4830-4842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 103] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 109. | Fukumura D, Gohongi T, Kadambi A, Izumi Y, Ang J, Yun CO, Buerk DG, Huang PL, Jain RK. Predominant role of endothelial nitric oxide synthase in vascular endothelial growth factor-induced angiogenesis and vascular permeability. Proc Natl Acad Sci U S A. 2001;98:2604-2609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 738] [Cited by in RCA: 771] [Article Influence: 32.1] [Reference Citation Analysis (0)] |
| 110. | Gallo O, Masini E, Morbidelli L, Franchi A, Fini-Storchi I, Vergari WA, Ziche M. Role of nitric oxide in angiogenesis and tumor progression in head and neck cancer. J Natl Cancer Inst. 1998;90:587-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 316] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 111. | Fukumura D, Kashiwagi SS, Jain RK. The role of nitric oxide in tumour progression. Nat Rev Cancer. 2006;6:521-34. [RCA] [DOI] [Full Text] [Cited by in Crossref: 923] [Cited by in RCA: 954] [Article Influence: 50.2] [Reference Citation Analysis (0)] |
| 112. | Acker T, Plate KH. Role of hypoxia in tumor angiogenesis-molecular and cellular angiogenic crosstalk. Cell Tissue Res. 2003;314:145-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 45] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 113. | Song Y, Zhao XP, Song K, Shang ZJ. Ephrin-A1 is up-regulated by hypoxia in cancer cells and promotes angiogenesis of HUVECs through a coordinated cross-talk with eNOS. PLoS One. 2013;8:e74464. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 114. | Zhuang G, Hunter S, Hwang Y, Chen J. Regulation of EphA2 receptor endocytosis by SHIP2 Lipid phosphatase via phosphatidylinositol 3-Kinase-dependent Rac1 activation. J Biol Chem. 2007;282:2683-2694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 116] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 115. | Deroanne C, Vouret-Craviari V, Wang B, Pouysségur J. EphrinA1 inactivates integrin-mediated vascular smooth muscle cell spreading via the Rac/PAK pathway. J Cell Sci. 2003;116:1367-1376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 97] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 116. | Cuyàs E, Queralt B, Martin-Castillo B, Bosch-Barrera J, Menendez JA. EphA2 receptor activation with ephrin-A1 Ligand restores cetuximab efficacy in NRAS-mutant colorectal cancer cells. Oncol Rep. 2017;38:263-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 117. | Himanen JP, Goldgur Y, Miao H, Myshkin E, Guo H, Buck M, Nguyen M, Rajashankar KR, Wang B, Nikolov DB. Ligand recognition by A-class Eph receptors: crystal structures of the EphA2 Ligand-binding domain and the EphA2/ephrin-A1 complex. EMBO Rep. 2009;10:722-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 106] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 118. | Noberini R, Koolpe M, Peddibhotla S, Dahl R, Su Y, Cosford ND, Roth GP, Pasquale EB. Small molecules can selectively inhibit ephrin binding to the EphA4 and EphA2 receptors. J Biol Chem. 2008;283:29461-29472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 113] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 119. | Giorgio C, Hassan Mohamed I, Flammini L, Barocelli E, Incerti M, Lodola A, Tognolini M. Lithocholic acid is an Eph-ephrin ligand interfering with Eph-kinase activation. PLoS One. 2011;6:e18128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 66] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 120. | Kiewlich D, Zhang J, Gross C, Xia W, Larsen B, Cobb RR, Biroc S, Gu JM, Sato T, Light DR, Heitner T, Willuda J, Vogel D, Monteclaro F, Citkowicz A, Roffler SR, Zajchowski DA. Anti-EphA2 antibodies decrease EphA2 protein levels in murine CT26 colorectal and human MDA-231 breast tumors but do not inhibit tumor growth. Neoplasia. 2006;8:18-30. [RCA] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 121. | Brantley DM, Cheng N, Thompson EJ, Lin Q, Brekken RA, Thorpe PE, Muraoka RS, Cerretti DP, Pozzi A, Jackson D, Lin C, Chen J. Soluble Eph A receptors inhibit tumor angiogenesis and progression in vivo. Oncogene. 2002;21:7011-7026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 242] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 122. | Jackson D, Gooya J, Mao S, Kinneer K, Xu L, Camara M, Fazenbaker C, Fleming R, Swamynathan S, Meyer D, Senter PD, Gao C, Wu H, Kinch M, Coats S, Kiener PA, Tice DA. A human antibody-drug conjugate targeting EphA2 inhibits tumor growth in vivo. Cancer Res. 2008;68:9367-9374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 121] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 123. | Tandon M, Vemula SV, Sharma A, Ahi YS, Mittal S, Bangari DS, Mittal SK. EphrinA1-EphA2 interaction-mediated apoptosis and FMS-like tyrosine kinase 3 receptor ligand-induced immunotherapy inhibit tumor growth in a breast cancer mouse model. J Gene Med. 2012;14:77-89. [RCA] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 124. | Fang WB, Brantley-Sieders DM, Hwang Y, Ham AJ, Chen J. Identification and functional analysis of phosphorylated tyrosine residues within EphA2 receptor tyrosine kinase. J Biol Chem. 2008;283:16017-16026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 102] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 125. | Duggineni S, Mitra S, Lamberto I, Han X, Xu Y, An J, Pasquale EB, Huang Z. Design and Synthesis of Potent Bivalent Peptide Agonists Targeting the EphA2 Receptor. ACS Med Chem Lett. 2013;4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 126. | Wykosky J, Palma E, Gibo DM, Ringler S, Turner CP, Debinski W. Soluble monomeric EphrinA1 is released from tumor cells and is a functional ligand for the EphA2 receptor. Oncogene. 2008;27:7260-7273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 97] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 127. | Bishop-Bailey D. Tumour vascularisation: a druggable target. Curr Opin Pharmacol. 2009;9:96-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 128. | Vihanto MM, Plock J, Erni D, Frey BM, Frey FJ, Huynh-Do U. Hypoxia up-regulates expression of Eph receptors and ephrins in mouse skin. FASEB J. 2005;19:1689-1691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 107] [Article Influence: 5.4] [Reference Citation Analysis (0)] |