Published online Sep 15, 2021. doi: 10.4251/wjgo.v13.i9.1132
Peer-review started: February 28, 2021
First decision: April 19, 2021
Revised: May 2, 2021
Accepted: July 5, 2021
Article in press: July 5, 2021
Published online: September 15, 2021
Processing time: 193 Days and 20.7 Hours
Hepatocellular carcinoma (HCC) is one of the most prevalent cancers and one of the main causes of cancer-related deaths worldwide. Most HCCs develop in an inflammatory microenvironment, and mounting evidence emphasizes the importance of immune aspects in hepatocarcinogenesis. In normal physiology, both innate and adaptive immune responses are responsible for eliminating malignantly transformed cells, thus preventing the development of liver cancer. However, in the setting of impaired natural killer cells and exhaustion of T cells, HCC can develop. The immunogenic features of HCC have relevant clinical implications. There is a large number of immune markers currently being studied for the early detection of liver cancer, which would be critical in order to improve surveillance programs. Moreover, novel immunotherapies have recently been proven to be effective, and the combination of atezolizumab and bevacizumab is currently the most effective treatment for advanced HCC. It is expected that in the near future different subgroups of patients will benefit from specific immunotherapy. The better we understand the immune aspects of HCC, the greater the benefit to patients through surveillance aiming for early detection of liver cancer, which allows for curative treatments, and, in cases of advanced disease, through the selection of the best possible therapy for each individual.
Core Tip: Recent developments in the field of hepatocellular carcinoma have drawn attention to the immune aspects of this neoplasm. Understanding the immune features of hepatocarcinogenesis is instrumental in order to comprehend current advances in immune markers and immunotherapy. Intensifying research on immune markers may improve surveillance and allow for precision medicine. Immunotherapy is already first-line treatment for advanced hepatocellular carcinoma.
- Citation: Mattos ÂZ, Debes JD, Boonstra A, Vogel A, Mattos AA. Immune aspects of hepatocellular carcinoma: From immune markers for early detection to immunotherapy. World J Gastrointest Oncol 2021; 13(9): 1132-1143
- URL: https://www.wjgnet.com/1948-5204/full/v13/i9/1132.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v13.i9.1132
Liver cancer has a global incidence of 11.6/100000 individuals (905677 new cases in 2020) and a mortality rate of 10.7/100000 individuals (830180 deaths in 2020), which places it sixth among all malignant neoplasms regarding incidence and second concerning mortality worldwide[1]. Moreover, liver cancer accounted for 12.5 million disability-adjusted life years across the globe in 2019[2]. Also, it is estimated that the age-standardized incidence rate of liver cancer will increase between 2017 and 2030 from 11.80 to 14.08 per 100000 individuals[3]. Hepatocellular carcinoma (HCC) is responsible for the vast majority of primary liver cancers[4,5]. HCC develops most frequently in patients with cirrhosis, and, while the most important causes of liver disease in patients with HCC are still chronic hepatitis B and C, non-alcoholic fatty liver disease is continuously growing in importance[6].
Current guidelines recommend surveillance for HCC with semiannual ultrasound in high-risk individuals, particularly in patients with cirrhosis[7-10]. The American Association for the Study of Liver Diseases makes it optional to add alpha-fetoprotein (AFP) to the surveillance program and it also recommends surveying high-risk individuals with hepatitis B who do not have cirrhosis (patients with African ancestry, Asian males over 40 years of age or females over 50 years of age, and patients with a family history of HCC)[7,8]. Regarding surveillance for patients without cirrhosis, the European Association for the Study of the Liver (EASL) also targets people with advanced liver fibrosis (Metavir fibrosis stage ≥ F3) and individuals with hepatitis B and a PAGE-B score ≥ 10[9]. Patients with cirrhosis presenting with liver nodules ≥1 cm on ultrasound should be referred for diagnostic evaluation of HCC with either a multiphasic computed tomography scan or magnetic resonance imaging[7-9]. Unfortunately, different studies demonstrate that only half of cases with HCC are diagnosed while patients are under surveillance, despite the association between diagnosis in a surveillance program and better prognosis[11-13]. HCC management depends on the stage of the disease. Table 1 shows the most commonly recommended therapeutic options according to the Barcelona Clinic Liver Cancer staging system at the time of publication of the current guidelines, in 2018[7-9].
Since then, many advances in the field of HCC have occurred. Several of these advances are directly related to the immune aspects of this disease, as it must be highlighted that HCC usually develops in an inflammatory and immunogenic background, such as that of viral hepatitis. The aim of this article is to review the immune characteristics of hepatocarcinogenesis, as well as the impact of the immune features of HCC on its early detection and on the novel systemic therapies against it.
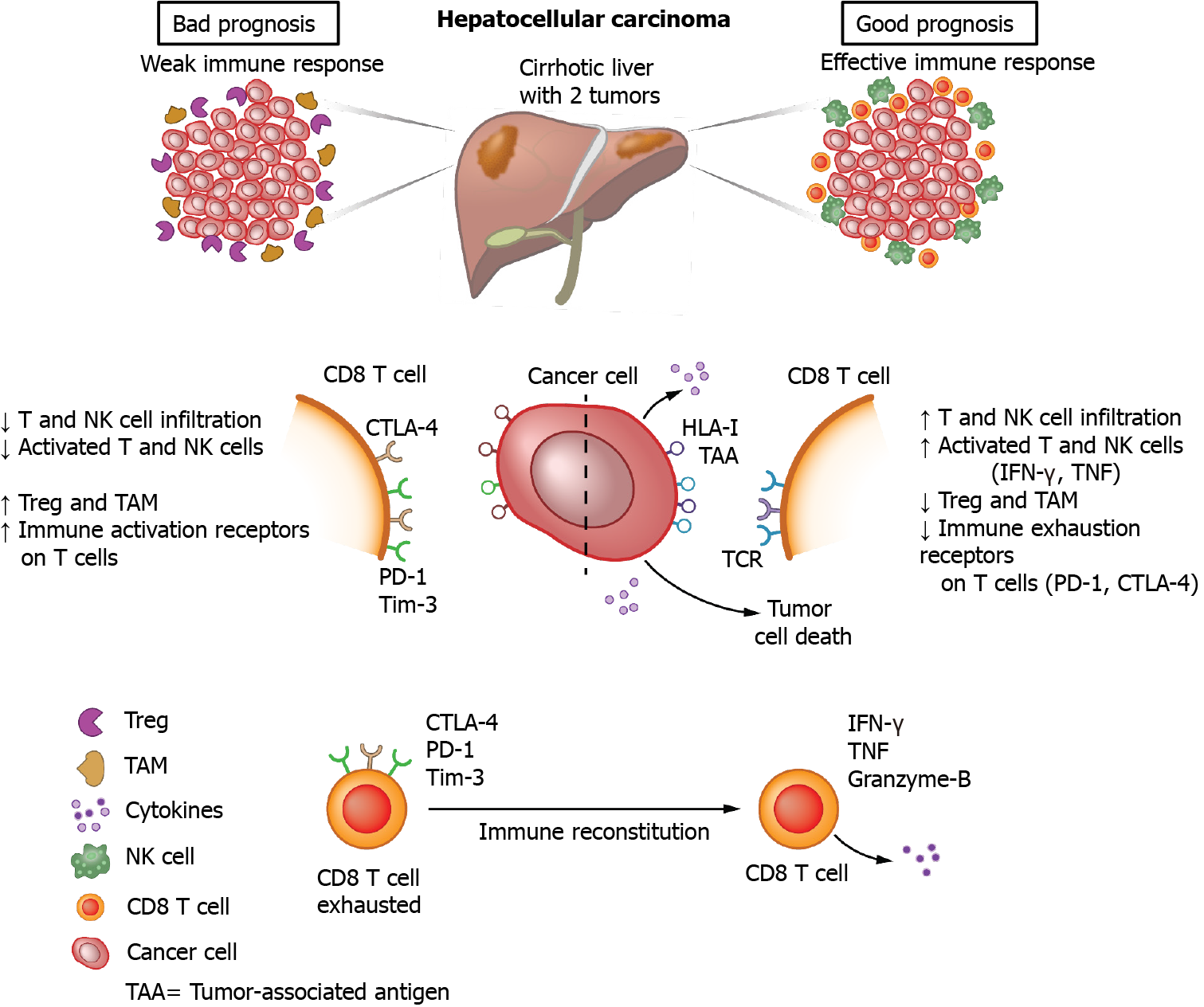
Similar to many other tumors, HCC is infiltrated by a wide variety of immune cells that shape the microenvironment of the tumor. Tumor-associated macrophages (TAM) are considered major players in the inflammation process and in dampening the anti-tumor response. These macrophages are of the so-called M2-type, meaning that they express immunomodulatory cytokines such as interleukin (IL)-10 and transforming growth factor-β (TGF-β) capable of inhibiting anti-tumor immunity[14,15]. In addition, TAM express low levels of pro-inflammatory cytokines, and are poor antigen presenters. Consequently, the presence of M2 macrophages has been associated with poor clinical outcomes in patients with HCC[16]. In recent years, more data has become available on the importance of TAM in promoting tumor cell proliferation, angiogenesis, and metastasis[17]. Via the release of chemokines, regulatory T cells are attracted towards the tumor, which contributes to impairment of the T cell response at the tumor site. Recent developments in transcriptomic profiling of the HCC material by single cell RNA sequencing clearly revealed the presence of multiple macrophage subsets in the tumor at distinct states, which were also found to be associated with disease progression[18,19].
Besides TAM, other cells of the innate immune system, such as natural killer (NK) cells, and cells of the adaptive immune system, such as CD8+ T cells, are also detected in the tumor tissue and are able to kill the transformed cells by the release of cytolytic enzymes, such as perforins and granzymes. Perforin acts by making pores in the cellular membrane, while granzymes are serine proteases which, among other activities, cleave and activate caspase-dependent apoptotic pathways thereby killing the cells. Recognition of the tumor by NK cells occurs via germ-line encoded receptors that are triggered when their stimulatory and inhibitory signals are out of balance due to their encounter with virus-infected or tumor cells[20]. CD8+ T cells, on the other hand, recognize the tumor via the interaction of specific receptors on T cells with a major histocompatibility complex class I (MHC-I) molecule present on tumor cells that harbors tumor-associated antigens (TAA). A number of classical tumor antigens have been described for HCC, including cancer-testis antigens (such as melanoma-associated gene-A1 — MAGE-A1 and New York-esophageal squamous cell carcinoma-1 — NY-ESO-1 proteins), and oncofetal antigens (such as AFP and glypican-3 — GPC-3)[21,22]. Antigen-specific CD8+ T cell responses directed against all four of these TAA have been readily observed in the blood of more than 50% of HCC patients[23], and found to be expressed exclusively or at much higher levels in HCC tumor tissue than in tumor-free liver tissue[22].
HCC is an immunogenic tumor, and this has been convincingly demonstrated by the fact that extensive immune cell infiltration is seen in HCC tissue, predominantly consisting of T cells. It has been reported that increased T cell infiltrates in the tumor are associated with improved overall survival (OS) in HCC and with lower tumor recurrence following resection[23,24]. Also, higher numbers of infiltrating NK cells in the neoplastic tissue have been shown to be associated with better survival of patients. In the same study, patients with advanced-stage HCC exhibited not only lower numbers of NK cells, but these cells were also functionally impaired, with lower production of interferon-γ and tumor necrosis factor (TNF)[25]. The impairment of NK cell function is, at least in part, ascribed to downregulation or enhanced shedding of activating receptors or their ligands, as has been reported for the interaction between the receptor NK group 2 member D (NKG2D) on NK cells and its ligands MHC-I-related chain A (MICA) and UL16-binding protein-1 (ULBP1) on tumor cells[26,27]. In addition, increased expression of the inhibitory ligand NK group 2 member A (NKG2A) on NK cells has been reported in HCC and has been shown to induce IL-10, which may further impair NK cell activity[28].
Despite the importance of NK cells in immune surveillance and progression of HCC, most studies on the immunology of HCC have focused on T cell responses, and especially on CD8+ T cell responses. Due to the wide availability of reagents to detect TAA-specific CD8+ T cells using MHC-I multimers, studies on CD8+ T cells have outnumbered those on TAA-specific CD4+ T cells. As mentioned above, the tumor-immune microenvironment of HCC is characterized by an abundance of tumor-infiltrating T cells and, although TAA have been identified during recognition of the tumor, the responses are too weak to eliminate the tumor and the effects are dampened by diverse immunosuppressive mechanisms in the tumor environment. It is crucially important to gain in-depth knowledge on these suppressive mechanisms, as shifting the balance by improving and restoring the quality of the immune response in the tumor environment may strongly benefit treatment outcomes for HCC patients[19]. However, the immunosuppressive mechanisms affecting the function of tumor-specific T cells are complex and diverse. Indeed, numerous suppressive immune populations able to inhibit T cell responses have been described to be present at higher numbers in the tumor environment of the liver as compared to the tumor-free liver. These cells include classical forkhead box P3 (FoxP3) positive regulatory T cells, IL-10 producing regulatory cells, myeloid derived suppressor cells and TAM[29-31]. Besides increased numbers, enhanced expression of inhibitory molecules and release of suppressive cytokines, such as IL-10 and TGF-β, have also been reported for these cells[32].
In addition to these suppressive immune cells, it has long been recognized that continuous tumor antigen exposure for prolonged periods of time leads to a differentiation program in T cells which gradually switches off specific activities of the affected cell, such as the capacity to perform cytotoxicity, to produce cytokines or to proliferate. These so-called exhausted T cells are characterized by overexpression of inhibitory receptors, including programmed cell death protein 1 (PD-1), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), lymphocyte-activation gene 3 (LAG-3) and T cell immunoglobulin and mucin domain-containing protein 3 (Tim-3)[33]. Importantly, higher numbers of exhausted CD8+ T cells have been reported to be associated with poor prognosis in HCC. The inhibitory intracellular signals are delivered to the T cell upon encountering its ligands on the tumor cell. This interaction is complex and diverse due to the high number of different inhibitory receptors known and the different levels of expression depending on the individual patient, but also on the activation state of the T cell and its antigen-specificity. However, mouse studies and in vitro studies have convincingly shown that blockade of inhibitory receptors can restore T cell function and lead to a reduction in tumor size[33,34].
In HCC patients, enhanced fractions of CD8+ T cells expressing PD-1, CTLA-4, LAG-3 and Tim-3 were observed in the tumor as compared to tumor-free tissue and blood of the same patient, with higher expression found on TAA-specific as compared to non-TAA-specific CD8+ T cells[35]. In vitro blockade of PD-1, CTLA-4 or LAG-3 using monoclonal antibodies increased the proliferation and cytokine production of CD8+ T cells isolated from the tumor upon polyclonal stimulation, and combinations of PD-1 blockade with any of the other antibodies further enhanced the CD8+ T cell activities[35]. Soluble PD-1 and PD ligand-1 (PD-L1) can be detected in serum, but no significant associations of soluble PD-1 or soluble PD-L1 with either intra-tumoral PD-L1 expression or the numbers of CD8+ T cells in the tumor have been determined[36]. Importantly, it has been suggested that, within the PD-1 expressing CD8+ T cell population, subgroups can be identified in which HCC with a discrete subgroup of cells that express high levels of PD-1 are more aggressive than HCC without such a population of cells, and that the levels of PD-1 play a role in the response to anti-PD-1 blockade[37]. The immune mechanisms involved in hepatocarcinogenesis are depicted in Figure 1.
Regardless of the underlying liver disease, chronic inflammation is a common denominator present in more than 90% of patients with HCC[38]. Indeed, local activation of intrahepatic cell populations can trigger coordinated processes followed by immune cell infiltration. It is with these concepts in mind that a broad array of inflammatory molecules, including cytokines and chemokines, have been investigated as potential biomarkers to predict early development of HCC, and to understand the mechanisms involved in HCC formation[39-41].
Peripheral immune markers represent an attractive option as they are easily measured in plasma or serum with relatively low interface technology. There are, however, several concerning issues when using immune markers for the detection of HCC. The first one is the poor reproducibility in measuring immune analytes by different platforms. This is mainly based on the variable expression level of these analytes at any given time in the organ system (serum or plasma), as well as collection and storage variability of samples. The second issue is that of the underlying liver disease. Immune markers that show promise for early detection of HCC in individuals with hepatitis B might not be reliable in individuals with HCC secondary to alcoholic liver disease due the differently activated immune pathways. Finally, a major issue lies in immunosurveillance, the recognition of tumor cells by leucocytes, which has been well-described for a variety of tumors and plays an important role during oncogenesis[42]. In this regard, it is difficult to distinguish the modulation of immune markers secondary to the underlying liver disease from that associated with a response to HCC formation. Nonetheless, some data suggest that, in the setting of chronic viral hepatitis, a hyperimmune environment due to the continuous presence of a virus in the liver over the years could pose a hyperreactive immune response with larger modulations of immune markers measurable in blood during the transition from a liver nodule to HCC[43,44].
A multitude of individual immune markers has been studied with the goal of early recognition of HCC. Among the best studied immune markers are: Osteopontin (OPN), growth differentiation factor 15 (GDF15), vascular endothelial growth factor (VEGF) and TGF-β. Other immune markers, not described here, have been studied for recognition of HCC at different stages, such as IL-6, IL-10, monocyte chemoattractant protein 1, and fibroblast growth factor 2, among others.
OPN has been examined as an early HCC marker by several research groups. OPN mediates a large array of different biological functions in the immune system and has been extensively studied in a variety of cancers[45,46]. Increased serum and plasma levels of OPN in individuals with HCC compared to those with liver cirrhosis or controls have been reported by multiple studies[47-50]. Most of the studies showing a high area under the receiver operating characteristic curve (AUROC) for OPN have been performed in Asian cohorts. However, more recent studies with West-African and European cohorts, including the EPIC study, have also shown promising findings[51].
GDF15 has been shown to modulate intrinsic pathways in inflammation, ischemia and several different cancers[52]. A Chinese study looking at GDF15 in predominantly viral hepatitis-related HCC showed elevated levels of this immune marker in those with early HCC compared to controls, with an AUROC of 0.84[53].
VEGF is an angiogenic factor related to vascular endothelial formation. A small retrospective Japanese study showed increased serum VEGF levels in hepatitis C-related HCC. The diagnostic performance of VEGF in this study was better than that of AFP[54]. Nonetheless, a later study performed in Egypt also in hepatitis C-related HCC did not detect serum VEGF differences between the HCC and the control groups[55]. Both studies were relatively small, and larger cohorts to further clarify these ambivalent results are needed. A more recent longitudinal study from our group identified serum VEGF as one of 12 immune mediators able to predict HCC development in individuals with hepatitis C[43]. However, this evaluation was also performed in a small cohort and in co-measurement with other immune analytes.
TGF-β regulates a variety of inflammatory processes and has been implicated in modulation of cell proliferation, differentiation and survival[56]. Previous studies have shown that serum levels of TGF-β are associated with HCC development, as well as early detection[49,57]. Nevertheless, most of these studies are small, mainly in individuals with hepatitis B or C, and several of them have shown improvement in the detection of HCC only when in conjunction with another analyte[58].
Most of the studied immune biomarkers show some degree of limitation in their role to detect early HCC. Recently, further attention has been placed on multiple immune-analyte detection rather than on individual measurements. Our group found a series of co-measured immune analytes to be associated with the future development of HCC in a cohort of individuals with hepatitis C, even when the cancer occurred up to two years later. The C-statistic for correct prediction of HCC was >0.90 for four of these markers (monokine-induced gamma interferon, IL-2, TNF-related apoptosis-inducing ligand and A proliferation-inducing ligand), and >0.80 for the rest. However, this study was performed in a small and specific cohort, and larger studies will be needed to evaluate the use of these and other immune markers in HCC detection[43].
Considering the clear role of the immune system in the genesis and progression of HCC, attempting to treat HCC with immunotherapy has been a reasonable approach evaluated by many groups. However, the complex interactions among the immune system, the underlying liver disease and the tumor make it challenging to develop effective therapies. This helps explain why only now immunotherapy is becoming part of the therapeutic armamentarium for HCC.
Until recently, sorafenib was the only drug approved for advanced HCC. It was the first medication with proven efficacy for this stage of the disease and it was the standard of care for over 10 years[59]. After many phase III failures, additional multitargeted tyrosine kinase inhibitors such as lenvatinib, regorafenib and cabozantinib and the anti-VEGF receptor antibody ramucirumab have been approved for both first- and second-line treatment in the last four years[60].
During the same time, the first promising results for immunotherapy in HCC were published and specifically the data for nivolumab in the phase II CheckMate-040 study provided compelling evidence that at least a subgroup of HCC patients is sensitive to immune-oncology (IO) based therapies[61]. A high disease control rate and overall response rate (ORR) were observed across all investigated subgroups, including “poor-prognosis patients” with impaired liver function (Child-Pugh B), extrahepatic tumor burden, or in patients after treatment with sorafenib. Based on this study, nivolumab was approved in the United States before the publication of the pivotal CheckMate-459 trial. The latter study investigated nivolumab in comparison to the standard of care (sorafenib) in the first-line setting, but failed to show a statistically significant improvement in OS, despite an OS of 16.4 mo, which had not been achieved in a phase III trial by that time. The response rate of just 15% confirmed the observation from the phase II study that immunotherapy alone has clinically meaningful efficacy only in a subset of patients[62].
Similar to nivolumab, promising data were initially reported for pembrolizumab in second-line HCC therapy in the phase II KEYNOTE-224 study, which led to its approval in the United States[63]. However, the subsequent KEYNOTE-240 pivotal trial also failed to reach the significance level for the predefined primary endpoints [an improvement in OS and/or progression-free survival (PFS) compared to placebo] due to the dual endpoints and several predefined interim analyses[64]. The response rate in both studies was 17% and 18.4% respectively, with a remarkable OS of 12.9 and 13.9 mo, which also stands out in comparison to non-IO phase III studies in the second-line setting[63,64]. Overall, despite good phase-II data, neither nivolumab nor pembrolizumab met their primary endpoints in the pivotal studies, and, accordingly, there is no approval for both drugs in Europe for advanced HCC.
To date, there is not a definitive correlation between clinical efficacy of IO and specific biomarkers. Initial evidence suggests that higher PD-L1 expression might correlate with improved survival under treatment with nivolumab[65]. On the other hand, despite initial suggestion that the wingless-related integration site (Wnt)/β-catenin pathway might convey resistance to IO-based therapies, these findings have not yet been confirmed in any of the prospective clinical trials[66].
Due to the lack of clinically meaningful biomarkers at the time, there were early efforts to develop IO-based combination therapies. The combination of nivolumab plus ipilimumab has already shown a superior OS in patients with different cancer types, including advanced melanoma and advanced non-small-cell lung cancer, compared to nivolumab monotherapy. In the CheckMate-040 study, three combinations were evaluated and revealed a consistently high ORR, as well as a promising OS in the second-line setting[67]. Based on these results, the United States Food and Drug Administration (FDA) approved nivolumab plus ipilimumab as second-line treatment for patients with advanced HCC previously treated with sorafenib. Currently, the CheckMate-9DW study is evaluating the efficacy of nivolumab plus ipilimumab compared to sorafenib or lenvatinib in first-line therapy.
Overexpression of VEGF has been implicated in the development and progression of liver cancer, and previous phase II studies have shown modest anti-tumor efficacy of bevacizumab as monotherapy in advanced HCC[68]. Additionally, there is increasing evidence that anti-VEGF therapies can enhance anti-PD-1 and anti-PD-L1 efficacy by reversing VEGF-mediated immunosuppression and promoting T-cell infiltration in tumors. Based on these observations, there was a rationale to evaluate the combination of the PD-L1 inhibitor atezolizumab with the VEGF-inhibitor bevacizumab in advanced HCC. In a relatively large phase Ib study for various tumor entities, bevacizumab in combination with atezolizumab achieved an impressive response rate of 36% (37/104 patients) in the HCC subgroup. The disease control rate was 71%, with a PFS of 7.3 mo and an OS of 17.1 mo[69]. The results were subsequently confirmed in the phase III IMbrave 150 trial in the first-line setting[70]. Of note, the OS at 12 mo was 67.2% among patients receiving bevacizumab plus atezolizumab and 54.6% among those receiving sorafenib, with a hazard ratio for death of 0.58 (P < 0.001). Moreover, the confirmed objective response was higher with the combination therapy than with sorafenib [27.3% vs 11.9% by Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 and 33.2% vs 13.3% by modified RECIST (mRECIST), P < 0.0001]. In addition to the significantly higher clinical activity, the side-effect profile and the quality-of-life evaluation were also considered favorable[70]; thus, the combination of atezolizumab and bevacizumab was approved by the FDA and the European Medicines Agency for the first-line therapy of HCC and it will become the next standard of care in first-line therapy for advanced HCC.
Currently, several ongoing phase III trials are evaluating the efficacy of IO/IO combinations and combinations of checkpoint inhibitors with tyrosine kinase inhibitors based on promising phase I and II studies. In agreement with findings for other solid tumors[71], the combination of lenvatinib and pembrolizumab, for instance, achieved a very high disease control rate of 88%, with an ORR of 46% by mRECIST and 36% by RECIST 1.1 as first-line therapy against unresectable HCC in the phase Ib KEYNOTE-524 study. The high ORR translated into an encouraging PFS of 9.3 mo, which is the longest PFS reported so far in any IO-based study in HCC, and an OS of 22 mo[72]. Based on these findings, an FDA breakthrough therapy designation was granted to the combination in 2019. However, due to the approval of atezolizumab and bevacizumab, the FDA denied approval of pembrolizumab and lenvatinib as frontline therapy in 2020. Now, the efficacy of the combination is being evaluated in comparison to lenvatinib in the phase III LEAP-002 trial (NCT03713593).
The progressive knowledge on the immune aspects of HCC is leading to important clinically applicable breakthroughs. Novel immunotherapies against advanced HCC are now available, resulting in increased survival, and many more are under development. Research on immune markers must be intensified, in order to identify tools for early detection of HCC, allowing for curative treatments to be offered to a larger proportion of patients, as well as to recognize which subgroups of individuals with advanced HCC will benefit the most from each systemic therapy.
Manuscript source: Invited manuscript
Specialty type: Oncology
Country/Territory of origin: Brazil
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Yang J S-Editor: Gao CC L-Editor: Webster JR P-Editor: Xing YX
| 1. | World Health Organization. Global Cancer Observatory - GLOBOCAN 2020. [cited 17 January 2021]. In: World Health Organization [Internet]. Available from: https://gco.iarc.fr/. |
| 2. | GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396:1204-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11327] [Cited by in RCA: 9637] [Article Influence: 1927.4] [Reference Citation Analysis (35)] |
| 3. | Liu Z, Xu K, Jiang Y, Cai N, Fan J, Mao X, Suo C, Jin L, Zhang T, Chen X. Global trend of aetiology-based primary liver cancer incidence from 1990 to 2030: a modelling study. Int J Epidemiol. 2021;50:128-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 80] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 4. | McGlynn KA, Petrick JL, El-Serag HB. Epidemiology of Hepatocellular Carcinoma. Hepatology. 2021;73 Suppl 1:4-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 820] [Cited by in RCA: 1346] [Article Influence: 336.5] [Reference Citation Analysis (1)] |
| 5. | Arnold M, Abnet CC, Neale RE, Vignat J, Giovannucci EL, McGlynn KA, Bray F. Global Burden of 5 Major Types of Gastrointestinal Cancer. Gastroenterology. 2020;159:335-349.e15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 857] [Cited by in RCA: 1232] [Article Influence: 246.4] [Reference Citation Analysis (0)] |
| 6. | Moon AM, Singal AG, Tapper EB. Contemporary Epidemiology of Chronic Liver Disease and Cirrhosis. Clin Gastroenterol Hepatol. 2020;18:2650-2666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 805] [Cited by in RCA: 720] [Article Influence: 144.0] [Reference Citation Analysis (0)] |
| 7. | Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR, Zhu AX, Murad MH, Marrero JA. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67:358-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2107] [Cited by in RCA: 3031] [Article Influence: 433.0] [Reference Citation Analysis (3)] |
| 8. | Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, Abecassis MM, Roberts LR, Heimbach JK. Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2018;68:723-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2121] [Cited by in RCA: 3243] [Article Influence: 463.3] [Reference Citation Analysis (1)] |
| 9. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018;69:182-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5593] [Cited by in RCA: 6064] [Article Influence: 866.3] [Reference Citation Analysis (3)] |
| 10. | Vogel A, Cervantes A, Chau I, Daniele B, Llovet JM, Meyer T, Nault JC, Neumann U, Ricke J, Sangro B, Schirmacher P, Verslype C, Zech CJ, Arnold D, Martinelli E; ESMO Guidelines Committee. Hepatocellular carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29:iv238-iv255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 595] [Cited by in RCA: 723] [Article Influence: 103.3] [Reference Citation Analysis (0)] |
| 11. | Fassio E, Díaz S, Santa C, Reig ME, Martínez Artola Y, Alves de Mattos A, Míguez C, Galizzi J, Zapata R, Ridruejo E, de Souza FC, Hernández N, Pinchuk L; Multicenter Group for Study of Hepatocarcinoma in Latin America; Asociación Latinoamericana para el Estudio del Hígado (ALEH). Etiology of hepatocellular carcinoma in Latin America: a prospective, multicenter, international study. Ann Hepatol. 2010;9:63-69. [PubMed] |
| 12. | Debes JD, Chan AJ, Balderramo D, Kikuchi L, Gonzalez Ballerga E, Prieto JE, Tapias M, Idrovo V, Davalos MB, Cairo F, Barreyro FJ, Paredes S, Hernandez N, Avendaño K, Diaz Ferrer J, Yang JD, Carrera E, Garcia JA, Mattos AZ, Hirsch BS, Gonçalves PT, Carrilho FJ, Roberts LR. Hepatocellular carcinoma in South America: Evaluation of risk factors, demographics and therapy. Liver Int. 2018;38:136-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 62] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 13. | Appel-da-Silva MC, Miozzo SA, Dossin IA, Tovo CV, Branco F, de Mattos AA. Incidence of hepatocellular carcinoma in outpatients with cirrhosis in Brazil: A 10-year retrospective cohort study. World J Gastroenterol. 2016;22:10219-10225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 14. | Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010;11:889-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2390] [Cited by in RCA: 2921] [Article Influence: 194.7] [Reference Citation Analysis (0)] |
| 15. | Solinas G, Germano G, Mantovani A, Allavena P. Tumor-associated macrophages (TAM) as major players of the cancer-related inflammation. J Leukoc Biol. 2009;86:1065-1073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 960] [Cited by in RCA: 1031] [Article Influence: 64.4] [Reference Citation Analysis (0)] |
| 16. | Ding T, Xu J, Wang F, Shi M, Zhang Y, Li SP, Zheng L. High tumor-infiltrating macrophage density predicts poor prognosis in patients with primary hepatocellular carcinoma after resection. Hum Pathol. 2009;40:381-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 187] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 17. | Tian Z, Hou X, Liu W, Han Z, Wei L. Macrophages and hepatocellular carcinoma. Cell Biosci. 2019;9:79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 114] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 18. | Zhang Q, He Y, Luo N, Patel SJ, Han Y, Gao R, Modak M, Carotta S, Haslinger C, Kind D, Peet GW, Zhong G, Lu S, Zhu W, Mao Y, Xiao M, Bergmann M, Hu X, Kerkar SP, Vogt AB, Pflanz S, Liu K, Peng J, Ren X, Zhang Z. Landscape and Dynamics of Single Immune Cells in Hepatocellular Carcinoma. Cell. 2019;179:829-845.e20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 763] [Cited by in RCA: 1007] [Article Influence: 167.8] [Reference Citation Analysis (0)] |
| 19. | Lim CJ, Lee YH, Pan L, Lai L, Chua C, Wasser M, Lim TKH, Yeong J, Toh HC, Lee SY, Chan CY, Goh BK, Chung A, Heikenwälder M, Ng IO, Chow P, Albani S, Chew V. Multidimensional analyses reveal distinct immune microenvironment in hepatitis B virus-related hepatocellular carcinoma. Gut. 2019;68:916-927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 250] [Article Influence: 41.7] [Reference Citation Analysis (0)] |
| 20. | Morvan MG, Lanier LL. NK cells and cancer: you can teach innate cells new tricks. Nat Rev Cancer. 2016;16:7-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 712] [Cited by in RCA: 877] [Article Influence: 97.4] [Reference Citation Analysis (0)] |
| 21. | Mizukoshi E, Nakamoto Y, Arai K, Yamashita T, Sakai A, Sakai Y, Kagaya T, Honda M, Kaneko S. Comparative analysis of various tumor-associated antigen-specific t-cell responses in patients with hepatocellular carcinoma. Hepatology. 2011;53:1206-1216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 148] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 22. | Sideras K, Bots SJ, Biermann K, Sprengers D, Polak WG, IJzermans JN, de Man RA, Pan Q, Sleijfer S, Bruno MJ, Kwekkeboom J. Tumour antigen expression in hepatocellular carcinoma in a low-endemic western area. Br J Cancer. 2015;112:1911-1920. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 38] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 23. | Flecken T, Schmidt N, Hild S, Gostick E, Drognitz O, Zeiser R, Schemmer P, Bruns H, Eiermann T, Price DA, Blum HE, Neumann-Haefelin C, Thimme R. Immunodominance and functional alterations of tumor-associated antigen-specific CD8+ T-cell responses in hepatocellular carcinoma. Hepatology. 2014;59:1415-1426. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 288] [Cited by in RCA: 298] [Article Influence: 27.1] [Reference Citation Analysis (0)] |
| 24. | Unitt E, Marshall A, Gelson W, Rushbrook SM, Davies S, Vowler SL, Morris LS, Coleman N, Alexander GJ. Tumour lymphocytic infiltrate and recurrence of hepatocellular carcinoma following liver transplantation. J Hepatol. 2006;45:246-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 201] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 25. | Wu Y, Kuang DM, Pan WD, Wan YL, Lao XM, Wang D, Li XF, Zheng L. Monocyte/macrophage-elicited natural killer cell dysfunction in hepatocellular carcinoma is mediated by CD48/2B4 interactions. Hepatology. 2013;57:1107-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 215] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 26. | Jinushi M, Takehara T, Tatsumi T, Hiramatsu N, Sakamori R, Yamaguchi S, Hayashi N. Impairment of natural killer cell and dendritic cell functions by the soluble form of MHC class I-related chain A in advanced human hepatocellular carcinomas. J Hepatol. 2005;43:1013-1020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 178] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 27. | Kamimura H, Yamagiwa S, Tsuchiya A, Takamura M, Matsuda Y, Ohkoshi S, Inoue M, Wakai T, Shirai Y, Nomoto M, Aoyagi Y. Reduced NKG2D ligand expression in hepatocellular carcinoma correlates with early recurrence. J Hepatol. 2012;56:381-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 98] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 28. | Sun C, Xu J, Huang Q, Huang M, Wen H, Zhang C, Wang J, Song J, Zheng M, Sun H, Wei H, Xiao W, Sun R, Tian Z. High NKG2A expression contributes to NK cell exhaustion and predicts a poor prognosis of patients with liver cancer. Oncoimmunology. 2017;6:e1264562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 209] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 29. | Fu J, Xu D, Liu Z, Shi M, Zhao P, Fu B, Zhang Z, Yang H, Zhang H, Zhou C, Yao J, Jin L, Wang H, Yang Y, Fu YX, Wang FS. Increased regulatory T cells correlate with CD8 T-cell impairment and poor survival in hepatocellular carcinoma patients. Gastroenterology. 2007;132:2328-2339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 598] [Cited by in RCA: 693] [Article Influence: 38.5] [Reference Citation Analysis (0)] |
| 30. | Chew V, Lai L, Pan L, Lim CJ, Li J, Ong R, Chua C, Leong JY, Lim KH, Toh HC, Lee SY, Chan CY, Goh BKP, Chung A, Chow PKH, Albani S. Delineation of an immunosuppressive gradient in hepatocellular carcinoma using high-dimensional proteomic and transcriptomic analyses. Proc Natl Acad Sci USA. 2017;114:E5900-E5909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 185] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 31. | Bayik D, Lauko AJ, Roversi GA, Serbinowski E, Acevedo-Moreno LA, Lanigan C, Orujov M, Lo A, Alban TJ, Kim A, Silver DJ, Nagy LE, Brown JM, Allende DS, Aucejo FN, Lathia JD. Hepatobiliary malignancies have distinct peripheral myeloid-derived suppressor cell signatures and tumor myeloid cell profiles. Sci Rep. 2020;10:18848. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 32. | Pedroza-Gonzalez A, Zhou G, Vargas-Mendez E, Boor PP, Mancham S, Verhoef C, Polak WG, Grünhagen D, Pan Q, Janssen H, Garcia-Romo GS, Biermann K, Tjwa ET, IJzermans JN, Kwekkeboom J, Sprengers D. Tumor-infiltrating plasmacytoid dendritic cells promote immunosuppression by Tr1 cells in human liver tumors. Oncoimmunology. 2015;4:e1008355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 85] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 33. | McLane LM, Abdel-Hakeem MS, Wherry EJ. CD8 T Cell Exhaustion During Chronic Viral Infection and Cancer. Annu Rev Immunol. 2019;37:457-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 645] [Cited by in RCA: 1283] [Article Influence: 213.8] [Reference Citation Analysis (0)] |
| 34. | Willimsky G, Schmidt K, Loddenkemper C, Gellermann J, Blankenstein T. Virus-induced hepatocellular carcinomas cause antigen-specific local tolerance. J Clin Invest. 2013;123:1032-1043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 35. | Zhou G, Sprengers D, Boor PPC, Doukas M, Schutz H, Mancham S, Pedroza-Gonzalez A, Polak WG, de Jonge J, Gaspersz M, Dong H, Thielemans K, Pan Q, IJzermans JNM, Bruno MJ, Kwekkeboom J. Antibodies Against Immune Checkpoint Molecules Restore Functions of Tumor-Infiltrating T Cells in Hepatocellular Carcinomas. Gastroenterology. 2017;153:1107-1119.e10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 322] [Article Influence: 40.3] [Reference Citation Analysis (0)] |
| 36. | Chang B, Huang T, Wei H, Shen L, Zhu D, He W, Chen Q, Zhang H, Li Y, Huang R, Li W, Wu P. The correlation and prognostic value of serum levels of soluble programmed death protein 1 (sPD-1) and soluble programmed death-ligand 1 (sPD-L1) in patients with hepatocellular carcinoma. Cancer Immunol Immunother. 2019;68:353-363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 97] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 37. | Kim HD, Song GW, Park S, Jung MK, Kim MH, Kang HJ, Yoo C, Yi K, Kim KH, Eo S, Moon DB, Hong SM, Ju YS, Shin EC, Hwang S, Park SH. Association Between Expression Level of PD1 by Tumor-Infiltrating CD8+ T Cells and Features of Hepatocellular Carcinoma. Gastroenterology. 2018;155:1936-1950.e17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 202] [Article Influence: 28.9] [Reference Citation Analysis (0)] |
| 38. | El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132:2557-2576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3846] [Cited by in RCA: 4267] [Article Influence: 237.1] [Reference Citation Analysis (2)] |
| 39. | Ibrahim GH, Mahmoud MA, Aly NM. Evaluation of circulating Transforming growth factor-beta1, Glypican-3 and Golgi protein-73 mRNAs expression as predictive markers for hepatocellular carcinoma in Egyptian patients. Mol Biol Rep. 2013;40:7069-7075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 40. | Marra F, Tacke F. Roles for chemokines in liver disease. Gastroenterology. 2014;147:577-594.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 507] [Cited by in RCA: 628] [Article Influence: 57.1] [Reference Citation Analysis (0)] |
| 41. | Parikh ND, Mehta AS, Singal AG, Block T, Marrero JA, Lok AS. Biomarkers for the Early Detection of Hepatocellular Carcinoma. Cancer Epidemiol Biomarkers Prev. 2020;29:2495-2503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 87] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 42. | Guillerey C, Smyth MJ. NK Cells and Cancer Immunoediting. Curr Top Microbiol Immunol. 2016;395:115-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 49] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 43. | Debes JD, van Tilborg M, Groothuismink ZMA, Hansen BE, Schulze Zur Wiesch J, von Felden J, de Knegt RJ, Boonstra A. Levels of Cytokines in Serum Associate With Development of Hepatocellular Carcinoma in Patients With HCV Infection Treated With Direct-Acting Antivirals. Gastroenterology. 2018;154:515-517.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 91] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 44. | Jiang R, Tan Z, Deng L, Chen Y, Xia Y, Gao Y, Wang X, Sun B. Interleukin-22 promotes human hepatocellular carcinoma by activation of STAT3. Hepatology. 2011;54:900-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 258] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 45. | O'Brien ER, Garvin MR, Stewart DK, Hinohara T, Simpson JB, Schwartz SM, Giachelli CM. Osteopontin is synthesized by macrophage, smooth muscle, and endothelial cells in primary and restenotic human coronary atherosclerotic plaques. Arterioscler Thromb. 1994;14:1648-1656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 207] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 46. | Higashi A, Dohi Y, Uraoka N, Sentani K, Uga S, Kinoshita H, Sada Y, Kitagawa T, Hidaka T, Kurisu S, Yamamoto H, Yasui W, Kihara Y. The Potential Role of Inflammation Associated with Interaction between Osteopontin and CD44 in a Case of Pulmonary Tumor Thrombotic Microangiopathy Caused by Breast Cancer. Intern Med. 2015;54:2877-2880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 47. | Ge T, Shen Q, Wang N, Zhang Y, Ge Z, Chu W, Lv X, Zhao F, Zhao W, Fan J, Qin W. Diagnostic values of alpha-fetoprotein, dickkopf-1, and osteopontin for hepatocellular carcinoma. Med Oncol. 2015;32:59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 48. | Shang S, Plymoth A, Ge S, Feng Z, Rosen HR, Sangrajrang S, Hainaut P, Marrero JA, Beretta L. Identification of osteopontin as a novel marker for early hepatocellular carcinoma. Hepatology. 2012;55:483-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 247] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 49. | da Costa AN, Plymoth A, Santos-Silva D, Ortiz-Cuaran S, Camey S, Guilloreau P, Sangrajrang S, Khuhaprema T, Mendy M, Lesi OA, Chang HK, Oh JK, Lee DH, Shin HR, Kirk GD, Merle P, Beretta L, Hainaut P. Osteopontin and latent-TGF β binding-protein 2 as potential diagnostic markers for HBV-related hepatocellular carcinoma. Int J Cancer. 2015;136:172-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 50. | Zhu M, Zheng J, Wu F, Kang B, Liang J, Heskia F, Zhang X, Shan Y. OPN is a promising serological biomarker for hepatocellular carcinoma diagnosis. J Med Virol. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 51. | Duarte-Salles T, Misra S, Stepien M, Plymoth A, Muller D, Overvad K, Olsen A, Tjønneland A, Baglietto L, Severi G, Boutron-Ruault MC, Turzanski-Fortner R, Kaaks R, Boeing H, Aleksandrova K, Trichopoulou A, Lagiou P, Bamia C, Pala V, Palli D, Mattiello A, Tumino R, Naccarati A, Bueno-de-Mesquita HB, Peeters PH, Weiderpass E, Quirós JR, Agudo A, Sánchez-Cantalejo E, Ardanaz E, Gavrila D, Dorronsoro M, Werner M, Hemmingsson O, Ohlsson B, Sjöberg K, Wareham NJ, Khaw KT, Bradbury KE, Gunter MJ, Cross AJ, Riboli E, Jenab M, Hainaut P, Beretta L. Circulating Osteopontin and Prediction of Hepatocellular Carcinoma Development in a Large European Population. Cancer Prev Res (Phila). 2016;9:758-765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 52. | Wischhusen J, Melero I, Fridman WH. Growth/Differentiation Factor-15 (GDF-15): From Biomarker to Novel Targetable Immune Checkpoint. Front Immunol. 2020;11:951. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 213] [Cited by in RCA: 325] [Article Influence: 65.0] [Reference Citation Analysis (0)] |
| 53. | Liu X, Chi X, Gong Q, Gao L, Niu Y, Cheng M, Si Y, Wang M, Zhong J, Niu J, Yang W. Association of serum level of growth differentiation factor 15 with liver cirrhosis and hepatocellular carcinoma. PLoS One. 2015;10:e0127518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 83] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 54. | Mukozu T, Nagai H, Matsui D, Kanekawa T, Sumino Y. Serum VEGF as a tumor marker in patients with HCV-related liver cirrhosis and hepatocellular carcinoma. Anticancer Res. 2013;33:1013-1021. [PubMed] |
| 55. | Zekri AR, Bahnassy AA, Alam El-Din HM, Morsy HM, Shaarawy S, Moharram NZ, Daoud SS. Serum levels of β-catenin as a potential marker for genotype 4/hepatitis C-associated hepatocellular carcinoma. Oncol Rep. 2011;26:825-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 56. | Massagué J. TGFbeta in Cancer. Cell. 2008;134:215-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2725] [Cited by in RCA: 3136] [Article Influence: 184.5] [Reference Citation Analysis (0)] |
| 57. | Hussein YM, Morad FE, Gameel MA, Emam WA, El Sawy WH, El Tarhouny SA, Bayomy ES, Raafat N. MAGE-4 gene m-RNA and TGF in blood as potential biochemical markers for HCC in HCV-infected patients. Med Oncol. 2012;29:3055-3062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 58. | Lu WQ, Qiu JL, Huang ZL, Liu HY. Enhanced circulating transforming growth factor beta 1 is causally associated with an increased risk of hepatocellular carcinoma: a mendelian randomization meta-analysis. Oncotarget. 2016;7:84695-84704. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 59. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Häussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J; SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9016] [Cited by in RCA: 10271] [Article Influence: 604.2] [Reference Citation Analysis (2)] |
| 60. | Llovet JM, Kelley RK, Villanueva A, Singal AG, Pikarsky E, Roayaie S, Lencioni R, Koike K, Zucman-Rossi J, Finn RS. Hepatocellular carcinoma. Nat Rev Dis Primers. 2021;7:6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4432] [Cited by in RCA: 3887] [Article Influence: 971.8] [Reference Citation Analysis (3)] |
| 61. | El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling TH Rd, Meyer T, Kang YK, Yeo W, Chopra A, Anderson J, Dela Cruz C, Lang L, Neely J, Tang H, Dastani HB, Melero I. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389:2492-2502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3278] [Cited by in RCA: 3314] [Article Influence: 414.3] [Reference Citation Analysis (1)] |
| 62. | Yau T, Park JW, Finn RS, Cheng AL, Mathurin P, Edeline J, Kudo M, Han KH, Harding JJ, Merle P, Rosmorduc O, Wyrwicz L, Schott E, Choo SP, Kelley RK, Begic D, Chen G, Neely J, Anderson J, Sangro B. CheckMate 459: A randomized, multi-center phase III study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Ann Oncol. 2019;30 Suppl 5:874-875. [RCA] [DOI] [Full Text] [Cited by in Crossref: 426] [Cited by in RCA: 433] [Article Influence: 72.2] [Reference Citation Analysis (0)] |
| 63. | Zhu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmer D, Verslype C, Zagonel V, Fartoux L, Vogel A, Sarker D, Verset G, Chan SL, Knox J, Daniele B, Webber AL, Ebbinghaus SW, Ma J, Siegel AB, Cheng AL, Kudo M; KEYNOTE-224 investigators. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 2018;19:940-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1184] [Cited by in RCA: 1900] [Article Influence: 271.4] [Reference Citation Analysis (0)] |
| 64. | Finn RS, Ryoo BY, Merle P, Kudo M, Bouattour M, Lim HY, Breder V, Edeline J, Chao Y, Ogasawara S, Yau T, Garrido M, Chan SL, Knox J, Daniele B, Ebbinghaus SW, Chen E, Siegel AB, Zhu AX, Cheng AL; KEYNOTE-240 investigators. Pembrolizumab As Second-Line Therapy in Patients With Advanced Hepatocellular Carcinoma in KEYNOTE-240: A Randomized, Double-Blind, Phase III Trial. J Clin Oncol. 2020;38:193-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1365] [Cited by in RCA: 1343] [Article Influence: 268.6] [Reference Citation Analysis (0)] |
| 65. | Sangro B, Melero I, Wadhawan S, Finn RS, Abou-Alfa GK, Cheng AL, Yau T, Furuse J, Park JW, Boyd Z, Tang HT, Shen Y, Tschaika M, Neely J, El-Khoueiry A. Association of inflammatory biomarkers with clinical outcomes in nivolumab-treated patients with advanced hepatocellular carcinoma. J Hepatol. 2020;73:1460-1469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 309] [Article Influence: 61.8] [Reference Citation Analysis (0)] |
| 66. | Harding JJ, Nandakumar S, Armenia J, Khalil DN, Albano M, Ly M, Shia J, Hechtman JF, Kundra R, El Dika I, Do RK, Sun Y, Kingham TP, D'Angelica MI, Berger MF, Hyman DM, Jarnagin W, Klimstra DS, Janjigian YY, Solit DB, Schultz N, Abou-Alfa GK. Prospective Genotyping of Hepatocellular Carcinoma: Clinical Implications of Next-Generation Sequencing for Matching Patients to Targeted and Immune Therapies. Clin Cancer Res. 2019;25:2116-2126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 448] [Article Influence: 64.0] [Reference Citation Analysis (0)] |
| 67. | Yau T, Kang YK, Kim TY, El-Khoueiry AB, Santoro A, Sangro B, Melero I, Kudo M, Hou MM, Matilla A, Tovoli F, Knox JJ, Ruth He A, El-Rayes BF, Acosta-Rivera M, Lim HY, Neely J, Shen Y, Wisniewski T, Anderson J, Hsu C. Efficacy and Safety of Nivolumab Plus Ipilimumab in Patients With Advanced Hepatocellular Carcinoma Previously Treated With Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 2020;6:e204564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 908] [Cited by in RCA: 971] [Article Influence: 194.2] [Reference Citation Analysis (0)] |
| 68. | Siegel AB, Cohen EI, Ocean A, Lehrer D, Goldenberg A, Knox JJ, Chen H, Clark-Garvey S, Weinberg A, Mandeli J, Christos P, Mazumdar M, Popa E, Brown RS Jr, Rafii S, Schwartz JD. Phase II trial evaluating the clinical and biologic effects of bevacizumab in unresectable hepatocellular carcinoma. J Clin Oncol. 2008;26:2992-2998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 381] [Cited by in RCA: 396] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 69. | Lee MS, Ryoo BY, Hsu CH, Numata K, Stein S, Verret W, Hack SP, Spahn J, Liu B, Abdullah H, Wang Y, He AR, Lee KH; GO30140 investigators. Atezolizumab with or without bevacizumab in unresectable hepatocellular carcinoma (GO30140): an open-label, multicentre, phase 1b study. Lancet Oncol. 2020;21:808-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 434] [Cited by in RCA: 411] [Article Influence: 82.2] [Reference Citation Analysis (0)] |
| 70. | Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Kudo M, Breder V, Merle P, Kaseb AO, Li D, Verret W, Xu DZ, Hernandez S, Liu J, Huang C, Mulla S, Wang Y, Lim HY, Zhu AX, Cheng AL; IMbrave150 Investigators. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med. 2020;382:1894-1905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2542] [Cited by in RCA: 4704] [Article Influence: 940.8] [Reference Citation Analysis (2)] |
| 71. | Taylor MH, Lee CH, Makker V, Rasco D, Dutcus CE, Wu J, Stepan DE, Shumaker RC, Motzer RJ. Phase IB/II Trial of Lenvatinib Plus Pembrolizumab in Patients With Advanced Renal Cell Carcinoma, Endometrial Cancer, and Other Selected Advanced Solid Tumors. J Clin Oncol. 2020;38:1154-1163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 158] [Cited by in RCA: 317] [Article Influence: 63.4] [Reference Citation Analysis (0)] |
| 72. | Finn RS, Ikeda M, Zhu AX, Sung MW, Baron AD, Kudo M, Okusaka T, Kobayashi M, Kumada H, Kaneko S, Pracht M, Mamontov K, Meyer T, Kubota T, Dutcus CE, Saito K, Siegel AB, Dubrovsky L, Mody K, Llovet JM. Phase Ib Study of Lenvatinib Plus Pembrolizumab in Patients With Unresectable Hepatocellular Carcinoma. J Clin Oncol. 2020;38:2960-2970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 661] [Cited by in RCA: 877] [Article Influence: 175.4] [Reference Citation Analysis (0)] |