Published online Mar 15, 2018. doi: 10.4251/wjgo.v10.i3.82
Peer-review started: December 21, 2017
First decision: January 15, 2018
Revised: February 5, 2018
Accepted: March 6, 2018
Article in press: March 6, 2018
Published online: March 15, 2018
Processing time: 83 Days and 19 Hours
To investigated the association between adenoma detection rate (ADR) and sessile serrated ADR (SSADR) and significant predictors for sessile serrated adenomas (SSA) detection.
This study is a retrospective, single-center analysis. Total colonoscopies performed by the gastroenterologists at the University of Tokyo Hospital between January and December 2014 were retrospectively identified. Polyps were classified as low-grade or high-grade adenoma, cancer, SSA, or SSA with cytological dysplasia, and the prevalence of each type of polyp was investigated. Predictors of adenoma and SSA detection were examined using logistic generalized estimating equation models. The association between ADR and SSADR for each gastroenterologist was investigated by calculating a correlation coefficient weighted by the number of each gastroenterologist’s examination.
A total of 3691 colonoscopies performed by 35 gastroenterologists were assessed. Overall, 978 (26.5%) low- and 84 (2.2%) high-grade adenomas, 81 (2.2%) cancers, 66 (1.8%) SSAs, and 2 (0.1%) SSAs with cytological dysplasia were detected. Overall ADR was 29.5% (men 33.2%, women 23.8%) and overall SSADR was 1.8% (men 1.7%, women 2.1%). In addition, 672 low-grade adenomas (68.8% of all the detected low-grade adenomas), 58 (69.9%) high-grade adenomas, 29 (34.5%) cancers, 52 (78.8%) SSAs, and 2 (100%) SSAs with cytological dysplasia were found in the proximal colon. Adenoma detection was the only significant predictor of SSA detection (adjusted OR: 2.53, 95%CI: 1.53-4.20; P < 0.001). The correlation coefficient between ADR and SSADR weighted by the number of each gastroenterologist’s examinations was 0.606 (P < 0.001).
Our results demonstrated that ADR is correlated to SSADR. In addition, patients with adenomas had a higher prevalence of SSAs than those without adenomas.
Core tip: Sessile serrated adenomas (SSAs) are difficult to detect and are associated with interval colorectal cancer (CRC). To reduce interval CRC and CRC death, SSA detection is important, and evaluation of the sessile serrated adenoma detection rate (SSADR) is crucial. In Western countries, there have been some reports showing the correlation of adenoma detection rate (ADR) and SSADR. However, in Asian countries, little is known about the correlation between ADR and SSADR. We investigated the association between ADR and SSADR and significant predictors for SSA detection in Japanese population. We found that ADR is correlated with SSADR, and patients with adenomas have a higher prevalence of SSAs than those without adenomas.
- Citation: Ohki D, Tsuji Y, Shinozaki T, Sakaguchi Y, Minatsuki C, Kinoshita H, Niimi K, Ono S, Hayakawa Y, Yoshida S, Yamada A, Kodashima S, Yamamichi N, Hirata Y, Ushiku T, Fujishiro M, Fukayama M, Koike K. Sessile serrated adenoma detection rate is correlated with adenoma detection rate. World J Gastrointest Oncol 2018; 10(3): 82-90
- URL: https://www.wjgnet.com/1948-5204/full/v10/i3/82.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v10.i3.82
Colorectal cancer (CRC) is one of the major causes of cancer mortality in the world[1]. Incidence of CRC has been increasing in Japan, and it is now the second leading cause of cancer-related death[2]. Colonoscopy currently plays a central role in CRC screening[3-5]. Total colonoscopy has been shown to reduce the risk of death from CRC by removing precancerous adenomas[5]. Total colonoscopy and detection of adenomas are imperative for preventing CRC. The adenoma detection rate (ADR) has been reported to be an excellent quality indicator of total colonoscopy[6,7]. ADR is also associated with the risk of interval CRC and death[8,9].
However, there have been some reports indicating that total colonoscopy is less effective in reducing the risk of cancer in the proximal colon[10,11]. The presence of sessile serrated adenomas (SSAs) in the right colon, which would progress via the serrated pathway to CRC, is thought to be a potential reason. A serrated pathway is an alternative pathway in which serrated polyps replace the traditional adenoma as precursor lesions to CRC[12]. CRCs derived from serrated pathways account for 20%-30% of all CRCs[13,14]. SSAs are usually flat or sessile, and are occasionally covered by a mucous cap[13]. They are difficult to detect because of their subtle morphology, and even when detected, are often incompletely resected. In addition, some SSAs are reported to progress to invasive cancer in a short period of time[15,16]. Therefore, SSAs are thought to be strongly associated with interval CRC[16,17]. To reduce interval CRC and CRC-related death, detection of SSAs is important, and evaluation of the SSA detection rate (SSADR) is crucial. Recently, there have been few reports suggesting that SSADR is associated with ADR[17,18]. However, to the best of our knowledge, there has been no report in Asian countries showing a correlation between ADR and SSADR. In this context, we investigated the association between ADR and SSADR with significant predictors for SSA detection in total colonoscopy screening or surveillance in the Japanese population.
This study is a retrospective, single-center analysis. We extracted data on total colonoscopies performed at the University of Tokyo Hospital between January and December 2014 by reviewing electronic medical records. All total colonoscopies performed by gastroenterologists were included in this analysis. Indications for total colonoscopy were classified as surveillance total colonoscopy, positive fecal occult blood test, screening for other symptoms (e.g., abdominal pain, anemia, and chronic diarrhea), and others. The following colonoscopies were excluded: repeated examinations during the study period and referral colonoscopies for endoscopic mucosal resection/endoscopic submucosal dissection (Figure 1). All gastroenterologists involved in this study had more than 5 years of experience in total colonoscopy.
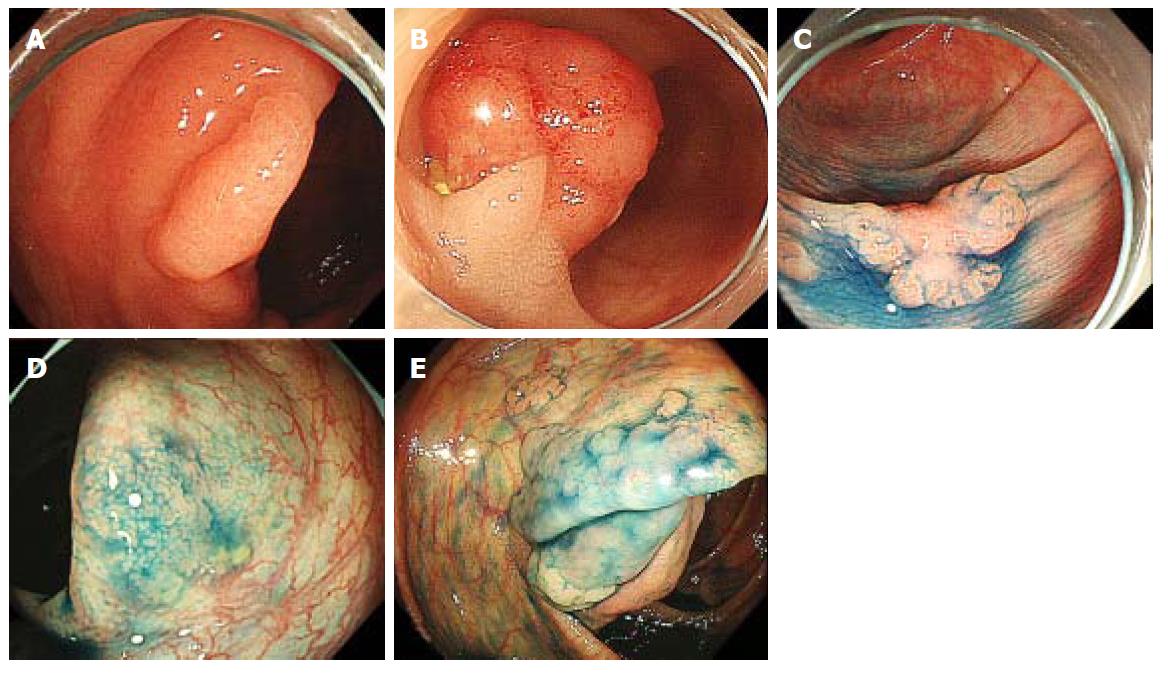
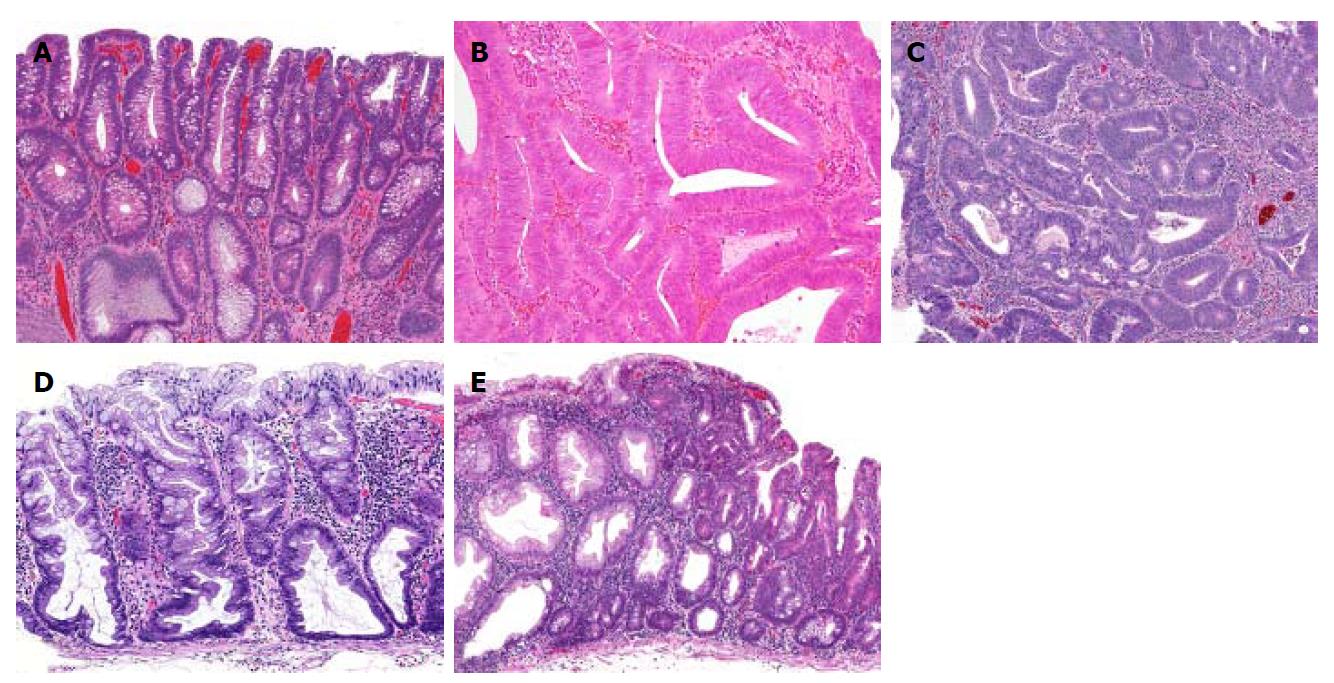
In this study, we classified the pathology of each resected polyp into the following categories: low- or high-grade adenoma, cancer (including intramucosal cancer), SSA, or SSA with cytological dysplasia (Figures 2 and 3). Polyps that were resected but not histologically evaluated, and endoscopically detected polyps that were not resected, were determined to be non-neoplastic. The histological definition for SSAs was in accordance with the definition of the Japanese Society for Cancer of the Colon and Rectum[19]. SSAs had two or more of the following features in more than 10% of the serrated area: (1) Dilated crypt; (2) irregularly branching crypt; and/or (3) dilation of the base of the crypt which often has a boot, L, or inverted T shape. SSA with cytological dysplasia was defined as a dysplastic area, similar to conventional adenoma[19,20]. In our institution, the comprehensive retrospective analysis of each patient’s medical record was approved by our ethics committee (No. 2058); this study is included in that category. The present study was performed in accordance with the Declaration of Helsinki.
The bowel preparation method in our institution was as follows: (1) 10 mL of 0.75% sodium picosulfate the day before endoscopy; and (2) 2-4 L of polyethylene glycol (Niflec: EA Pharma, Tokyo, Japan) on the morning of the endoscopy.
Video processor unit EVIS LUCERA SPECTRUM or EVIS LUCERA ELITE (Olympus Corporation, Tokyo, Japan) and single-channel lower gastrointestinal endoscope (PCF-Q260AZI, PCF-Q260AI, PCF-PQL, CF-240AI; Olympus Co.) were used. The choice of the endoscope was left to the discretion of each endoscopist.
Almost all colonoscopies were performed without sedation, but in some special cases where patients could not tolerate the colonoscopy procedure, conscious sedation using diazepam with or without pentazocine was administered.
The polyp detection rate and location of each polyp were investigated. The proximal colon was defined as the area proximal to the splenic flexure (transverse colon, ascending colon, and cecum), while the distal colon was defined as the area distal to the splenic flexure (descending colon, sigmoid colon, and rectum). ADR was calculated as described in previous literature[6,21]: the proportion of colonoscopies where at least one colorectal low- or high-grade adenoma or cancer was detected. SSADR was calculated in the same way: the proportion of colonoscopies where at least one SSA or SSA with cytological dysplasia was detected.
Factors possibly related to adenoma detection and SSA detection was assessed: (1) Patients’ age; (2) patients’ sex; (3) years of colonoscopy experience of the endoscopist; (4) withdrawal time; (5) cecal intubation rate; and (6) bowel cleansing level. Withdrawal time was defined as the time from identification of cecum to identification of anus in colonoscopy cases where no polyps were removed. The bowel cleansing level was classified as “adequate” or “non-adequate” according to the ASGE/ACG task force recommendations. “Adequate” was defined as the examination allowed for the detection of polyps > 5 mm in size[6,22].
Characteristics of patients were summarized and compared between the presence (+) or absence (-) of adenoma detection using t-test or chi-squared test. Gastroenterologists’ experience and their average withdrawal time that was calculated after excluding polypectomy were also summarized. Predictors of ADR were examined using logistic generalized estimating equation models, which explain the adenoma detection probability of each total colonoscopy by patient-and gastroenterologist-level variables. We used robust sandwich variance estimators that specified each gastroenterologist as a cluster to compute 95% confidence intervals (CI) and P-values. Predictors of SSADR were similarly examined, but adenoma detection of corresponding total colonoscopy was added as a predictor. The bivariate association of SSADR and ADR of each gastroenterologist were illustrated by a scatter plot and correlation coefficient that were weighted by the number of performed total colonoscopies. All analyses were conducted using SAS version 9.4 (Cary, NC, United States).
A total of 4253 colonoscopies were performed by gastrenterologists during the study period. Overall, 562 colonoscopies were excluded based on the predetermined criteria, and 3691 colonoscopies were included in the analysis (Figure 1). Baseline characteristics of colonoscopies are shown in Table 1. Adequate bowel cleansing and cecal intubation rate were observed in 3585 (97.1%) cases and 3636 (98.5%) cases, respectively.
| Total (n = 3691) | |
| Age, mean ± SD (yr) | 63.5 ± 13.3 |
| Sex: Male (%) | 2224 (60.3) |
| Adequate bowel cleansing (%) | 3585 (97.1) |
| Cecal intubation rate (%) | 3636 (98.5) |
| Indications for colonoscopy (%) | |
| Surveillance | 1314 (35.6) |
| Fecal occult blood test | 538 (14.6) |
| Screening for other symptoms | 544 (14.7) |
| Others | 1295 (35.1) |
Baseline characteristics of gastroenterologists are shown in Table 2. All gastroenterologists had at least 5 years of colonoscopy experience; 16 (45.7%) gastroenterologists performed more than 100 cases a year.
| n = 35 | |
| Sex: Male (%) | 25/35 (71.4) |
| Years of experience in colonoscopy (%) | |
| 5-9 | 24/35 (68.6) |
| 10-14 | 6/35 (17.1) |
| ≥ 15 | 5/35 (14.3) |
| Number of colonoscopies performed (%) | |
| ≤ 100 | 19/35 (54.3) |
| 100-200 | 10/35 (28.6) |
| ≥ 200 | 6/35 (17.1) |
| Withdrawal time: Mean (SD), m | 10.1 ± 6.9 |
Low- and high-grade adenomas, and cancers were found in 978 (26.5%) cases, 84 (2.2%) cases and 81 (2.2%) cases, respectively. Overall ADR was 29.5% (men 33.2%, women 23.8%). SSAs and SSAs with cytological dysplasia were found in 66 (1.8%) cases and 2 (0.1%) cases, respectively. Overall SSADR was 1.8% (men 1.7%, women 2.1%).
The location of each polyp was also investigated. Altogether, 672 low-grade adenomas (68.8% of all the detected low-grade adenomas), 58 (69.9%) high-grade adenomas, 29 (34.5%) cancers, 52 (78.8%) SSAs, and 2 (100%) SSAs with cytological dysplasia were found in the proximal colon.
Univariable and multivariable analyses were performed to evaluate factors associated with adenoma detection (Table 3). In our institution, the cecal intubation rate was almost 100%, but could not be used in the analyses. Mean withdrawal time was 10 min, and there were only 2 gastroenterologists whose withdrawal time was less than 6 min. According to the scatter diagram plotting each endoscopist’s ADR against their mean withdrawal time, as previously reported[7], the recommended ADR level of 25%[6] corresponded to a withdrawal time of 8 min. All factors, except for years of colonoscopy experience, were significantly associated with adenoma detection in both analyses with a 5% significance level. Being a woman (adjusted OR: 0.61, 95%CI: 0.54-0.70; P < 0.001) and those with non-adequate bowel cleansing (adjusted OR: 0.32, 95%CI: 0.19-0.52; P < 0.001) had a statistically inverse relationship with adenoma detection. Mean withdrawal time ≥ 8 min had statistically significant correlation with adenoma detection (adjusted OR: 1.77, 95%CI: 1.28-2.46; P < 0.001).
| Variable | Univariable model | Multivariable model | ||
| OR (95%CI) | P | OR (95%CI) | P | |
| Patient-level variable | ||||
| Age (yr) | 1.02 (1.02, 1.03) | < 0.001 | 1.02 (1.02, 1.03) | < 0.001 |
| Female | 0.63 (0.55, 0.71) | < 0.001 | 0.61 (0.54, 0.70) | < 0.001 |
| Non-adequate bowel cleansing | 0.36 (0.22, 0.57) | < 0.001 | 0.32 (0.19, 0.52) | < 0.001 |
| Endoscopist-level variable | ||||
| Endoscopist's experiment (yr) | 0.98 (0.94, 1.02) | 0.36 | 0.99 (0.96, 1.02) | 0.55 |
| Mean withdrawal time ≥ 8 min (vs < 8 min) | 1.72 (1.23, 2.41) | 0.0015 | 1.77 (1.28, 2.46) | < 0.001 |
Univariable and multivariable analyses were performed to evaluate factors associated with SSA detection (Table 4). Both analyses revealed that adenoma detection was the only significant predictor for SSA detection (adjusted OR: 2.53, 95%CI: 1.53-4.20; P < 0.001). Mean withdrawal time ≥ 8 min tended to be associated with SSA detection, but was not statistically significant (adjusted OR 1.53; 95%CI: 0.62-3.75; P = 0.35).
| Variable | Univariable model | Multivariable model | ||
| OR (95%CI) | P | OR (95%CI) | P | |
| Patient-level variable | ||||
| Adenoma detection (vs none) | 2.44 (1.45, 4.09) | < 0.001 | 2.53 (1.53, 4.20) | < 0.001 |
| Age (yr) | 0.99 (0.98, 1.01) | 0.27 | 0.99 (0.98, 1.00) | 0.07 |
| Female | 1.28 (0.77, 2.11) | 0.34 | 1.40 (0.85, 2.29) | 0.19 |
| Non-adequate bowel cleansing | 0.50 (0.07, 3.47) | 0.48 | 0.60 (0.08, 4.28) | 0.61 |
| Endoscopist-level variable | ||||
| Endoscopist's experiment (yr) | 0.99 (0.89, 1.10) | 0.86 | 1.00 (0.91, 1.09) | 0.96 |
| Mean withdrawal time ≥ 8 min (vs < 8 min) | 1.74 (0.70, 4.29) | 0.23 | 1.53 (0.62, 3.75) | 0.35 |
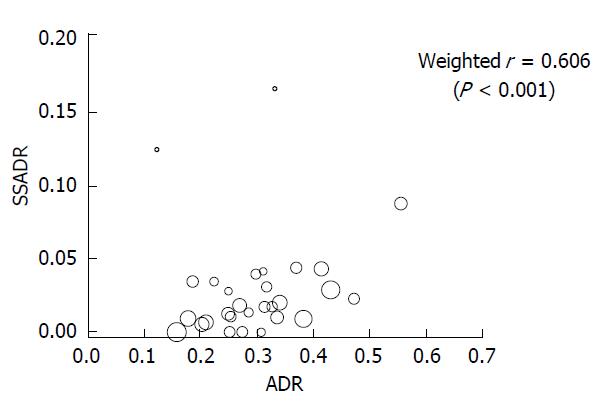
As for the correlation between ADR and SSADR, a scatter diagram of ADR and SSADR is shown in Figure 4. The correlation coefficient between ADR and SSADR weighted by the number of each gastroenterologist’s examinations was 0.606 (P < 0.001).
In the present study, a relatively strong association between ADR and SSADR was observed. Some reports have described the correlation of ADR and SSADR in Western countries patients[17,18]; however, to our knowledge, the prevalence of SSAs or SSADR in Asian populations has not yet been fully investigated and appropriate SSADR has not been determined. Therefore, our study holds importance, as it is the first report to demonstrate the correlation between SSADR and ADR in Asian populations.
There is controversy regarding the prevalence of SSAs, which differs among previously published studies, varying from 2%-10%[13,17,18,23,24]. In our institution, the prevalence of SSAs was approximately 2%, which is lower than previously reported results in Western populations. Each endoscopist’s cognitive capability to detect SSAs may differ in degree. Payne et al[25] reported that the prevalence of SSAs varied among endoscopy centers. In addition, Abdeljawad K et al[26] reported that a review of pathology slides by an experienced gastrointestinal pathologist increased the prevalence of SSAs, and the prevalence of SSAs increased over the study period, suggesting that each endoscopist improved his detection skills over time. However, the gastroenterologist’s ADR in this study was approximately 30%, which is within the standard of quality indicators for colonoscopy specified by ASGE[6]. Therefore, the quality of the present study is assured. The quality of the pathological evaluation was also high, because the experienced gastrointestinal pathologist (U.T.), who was acquainted with the definition of the Japanese Society for Cancer of the Colon and Rectum, reassessed the pathology slides. As previously mentioned, the prevalence of SSAs in Asian populations has not been determined, as there may be a difference between races. It is mandatory to investigate the true prevalence of SSAs in Asian populations in the future.
The factors associated with SSA detection were investigated, and our study demonstrated that adenoma detection at the patient level was the only independent significant factor associated with SSA detection. Previous reports have shown that when a patient presented with serrated lesions, especially SSAs, he/she was also more likely to have advanced neoplasia[23,27-29]. These results were compatible with previous reports and suggested that ADR is correlated with SSADR.
A withdrawal time of ≥ 8 min was not a statistically significant factor for SSA detection, although it was significantly related to adenoma detection. However, considering that ADR and SSADR are correlated, a longer duration of inspection seems to improve ADR and SSADR. In this study, the total number of SSAs was quite small. This may be a reason why a significant association between withdrawal time and SSA detection was not found.
We acknowledge that there were several limitations in our study. First, this study was a retrospective single center study, and the number of SSA cases was small. Second, there were many cases of total colonoscopy surveillance in the present study in addition to total colonoscopy screening. As previously stated, the target ADR should be changed according to patient risk[30]. However, factors associated with adenoma detection in this study were similar to those in previous reports. Moreover, Anderson JC reported that the serrated polyp detection rate was similar for screening or surveillance indications, suggesting that both indications could be used to derive the serrated polyp detection rate in practice[31].
Rex et al[32] has also recently reported that using overall ADR to calculate ADR from screening, surveillance, and diagnostic colonoscopies would be just as effective as a screening-only ADR. Taking this into account, the current findings can be applied to clinical practice to some extent. Finally, the ratio of adequate bowel cleansing in this study was much higher than in previous studies. The ASGE guidelines recommend that the quality of bowel cleansing should be evaluated after retained fluid or stool has been suctioned[6]. In our institution, if fluid and stool were retained, gastroenterologists suctioned as much as possible to identify polyps ≥ 5 mm in size. Such cases were considered adequate in our study, and therefore, the ratio of the “adequate” level was high.
In conclusion, our study suggests that ADR is correlated with SSADR. In addition, patients with adenomas may have a higher prevalence of SSAs than those without adenomas. A large-scale prospective study will be needed to validate these findings.
Sessile serrated adenomas (SSAa) are difficult to detect and strongly associated with interval colorectal cancer (CRC). It is necessary to investigate the factors which influence SSA detection and to evaluate the SSA detection rate (SSADR).
In Western countries, some reports have described the correlation of ADR and SSADR. However, to the best of our knowledge, there has been no report in Asian countries showing a correlation between ADR and SSADR. In this context, we investigated the association between ADR and SSADR with significant predictors for SSA detection in total colonoscopy screening or surveillance in the Japanese population.
The main objectives were as follows; the prevalence of each polyp (low-grade or high-grade adenoma, cancer, SSA, or SSA with cytological dysplasia), each gastoroenterologist’s ADR and SSADR, the association between ADR and SSADR for each gastroenterologist and predictors of adenoma and SSA detection.
Total colonoscopies performed by the gastroenterologists at the University of Tokyo Hospital between January and December 2014 were retrospectively identified. The prevalence of each type of polyp was investigated. Predictors of adenoma and SSA detection were examined using logistic generalized estimating equation models. The association between ADR and SSADR for each gastroenterologist was investigated by calculating a correlation coefficient weighted by the number of each gastroenterologist’s examination.
A total of 3691 colonoscopies by 35 gastroenterologists were assessed. 978 low grade adenomas (26.5%), 84 high grade adenomas (2.2%), 81 cancers (2.2%), 66 SSAs (1.8%) and 2 SSAs with cytological dysplasia (0.1%) were detected. Adenoma detection was the only significant predictor of SSA detection (adjusted OR: 2.53, 95%CI: 1.53-4.20; P < 0.001). The correlation coefficient between ADR and SSADR weighted by the number of each gastroenterologist’s examinations was 0.606 (P < 0.001).
Our study suggests that ADR is correlated with SSADR. Some reports have described the correlation of ADR and SSADR in Western countries patients; however, to our knowledge, the prevalence of SSAs or SSADR in Asian populations has not yet been fully investigated and appropriate SSADR has not been determined. Therefore, our study holds importance, as it is the first report to demonstrate the correlation between SSADR and ADR in Asian populations. In addition, patients with adenomas may have a higher prevalence of SSAs than those without adenomas.
This study was a retrospective single center study, and the number of SSA cases was small. Therefore, a large-scale prospective study will be needed to validate these findings.
This paper was presented at the Digestive Disease Week (DDW), May 21-24, 2016, in San Diego, United States of America.
Manuscript source: Unsolicited manuscript
Specialty type: Oncology
Country of origin: Japan
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C, C
Grade D (Fair): D
Grade E (Poor): 0
P- Reviewer: Ahluwalia NK, De Silva AP, Dutta AK, Herszenyi L, Teramoto-Matsubara OT S- Editor: Cui LJ L- Editor: A E- Editor: Li RF
| 1. | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12135] [Cited by in RCA: 12991] [Article Influence: 1443.4] [Reference Citation Analysis (2)] |
| 2. | Matsuda A, Matsuda T, Shibata A, Katanoda K, Sobue T, Nishimoto H; Japan Cancer Surveillance Research Group. Cancer incidence and incidence rates in Japan in 2008: a study of 25 population-based cancer registries for the Monitoring of Cancer Incidence in Japan (MCIJ) project. Jpn J Clin Oncol. 2014;44:388-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 170] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 3. | Lieberman DA, Rex DK, Winawer SJ, Giardiello FM, Johnson DA, Levin TR. Guidelines for colonoscopy surveillance after screening and polypectomy: a consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2012;143:844-857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1367] [Cited by in RCA: 1445] [Article Influence: 111.2] [Reference Citation Analysis (0)] |
| 4. | European Colorectal Cancer Screening Guidelines Working Group, von Karsa L, Patnick J, Segnan N, Atkin W, Halloran S, Lansdorp-Vogelaar I, Malila N, Minozzi S, Moss S, Quirke P, Steele RJ, Vieth M, Aabakken L, Altenhofen L, Ancelle-Park R, Antoljak N, Anttila A, Armaroli P, Arrossi S, Austoker J, Banzi R, Bellisario C, Blom J, Brenner H, Bretthauer M, Camargo Cancela M, Costamagna G, Cuzick J, Dai M, Daniel J, Dekker E, Delicata N, Ducarroz S, Erfkamp H, Espinàs JA, Faivre J, Faulds Wood L, Flugelman A, Frkovic-Grazio S, Geller B, Giordano L, Grazzini G, Green J, Hamashima C, Herrmann C, Hewitson P, Hoff G, Holten I, Jover R, Kaminski MF, Kuipers EJ, Kurtinaitis J, Lambert R, Launoy G, Lee W, Leicester R, Leja M, Lieberman D, Lignini T, Lucas E, Lynge E, Mádai S, Marinho J, Maučec Zakotnik J, Minoli G, Monk C, Morais A, Muwonge R, Nadel M, Neamtiu L, Peris Tuser M, Pignone M, Pox C, Primic-Zakelj M, Psaila J, Rabeneck L, Ransohoff D, Rasmussen M, Regula J, Ren J, Rennert G, Rey J, Riddell RH, Risio M, Rodrigues V, Saito H, Sauvaget C, Scharpantgen A, Schmiegel W, Senore C, Siddiqi M, Sighoko D, Smith R, Smith S, Suchanek S, Suonio E, Tong W, Törnberg S, Van Cutsem E, Vignatelli L, Villain P, Voti L, Watanabe H, Watson J, Winawer S, Young G, Zaksas V, Zappa M, Valori R. European guidelines for quality assurance in colorectal cancer screening and diagnosis: overview and introduction to the full supplement publication. Endoscopy. 2013;45:51-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 204] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 5. | Zauber AG, Winawer SJ, O’Brien MJ, Lansdorp-Vogelaar I, van Ballegooijen M, Hankey BF, Shi W, Bond JH, Schapiro M, Panish JF. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012;366:687-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1952] [Cited by in RCA: 2286] [Article Influence: 175.8] [Reference Citation Analysis (2)] |
| 6. | Rex DK, Schoenfeld PS, Cohen J, Pike IM, Adler DG, Fennerty MB, Lieb JG 2nd, Park WG, Rizk MK, Sawhney MS, Shaheen NJ, Wani S, Weinberg DS. Quality indicators for colonoscopy. Gastrointest Endosc. 2015;81:31-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 836] [Article Influence: 83.6] [Reference Citation Analysis (0)] |
| 7. | Barclay RL, Vicari JJ, Doughty AS, Johanson JF, Greenlaw RL. Colonoscopic withdrawal times and adenoma detection during screening colonoscopy. N Engl J Med. 2006;355:2533-2541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 911] [Cited by in RCA: 951] [Article Influence: 50.1] [Reference Citation Analysis (0)] |
| 8. | Corley DA, Levin TR, Doubeni CA. Adenoma detection rate and risk of colorectal cancer and death. N Engl J Med. 2014;370:2541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 72] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 9. | Kaminski MF, Regula J, Kraszewska E, Polkowski M, Wojciechowska U, Didkowska J, Zwierko M, Rupinski M, Nowacki MP, Butruk E. Quality indicators for colonoscopy and the risk of interval cancer. N Engl J Med. 2010;362:1795-1803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1287] [Cited by in RCA: 1468] [Article Influence: 97.9] [Reference Citation Analysis (0)] |
| 10. | Brenner H, Hoffmeister M, Arndt V, Stegmaier C, Altenhofen L, Haug U. Protection from right- and left-sided colorectal neoplasms after colonoscopy: population-based study. J Natl Cancer Inst. 2010;102:89-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 409] [Cited by in RCA: 419] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 11. | Baxter NN, Warren JL, Barrett MJ, Stukel TA, Doria-Rose VP. Association between colonoscopy and colorectal cancer mortality in a US cohort according to site of cancer and colonoscopist specialty. J Clin Oncol. 2012;30:2664-2669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 271] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 12. | Yamane L, Scapulatempo-Neto C, Reis RM, Guimarães DP. Serrated pathway in colorectal carcinogenesis. World J Gastroenterol. 2014;20:2634-2640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 68] [Cited by in RCA: 83] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 13. | Rex DK, Ahnen DJ, Baron JA, Batts KP, Burke CA, Burt RW, Goldblum JR, Guillem JG, Kahi CJ, Kalady MF. Serrated lesions of the colorectum: review and recommendations from an expert panel. Am J Gastroenterol. 2012;107:1315-1329; quiz 1314, 1330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 825] [Cited by in RCA: 830] [Article Influence: 63.8] [Reference Citation Analysis (0)] |
| 14. | Saiki H, Nishida T, Yamamoto M, Hayashi S, Shimakoshi H, Shimoda A, Amano T, Sakamoto A, Otake Y, Sugimoto A. Frequency of coexistent carcinoma in sessile serrated adenoma/polyps and traditional serrated adenomas removed by endoscopic resection. Endosc Int Open. 2016;4:E451-E458. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 15. | Oono Y, Fu K, Nakamura H, Iriguchi Y, Yamamura A, Tomino Y, Oda J, Mizutani M, Takayanagi S, Kishi D. Progression of a sessile serrated adenoma to an early invasive cancer within 8 months. Dig Dis Sci. 2009;54:906-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 100] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 16. | Omori K, Yoshida K, Tamiya S, Daa T, Kan M. Endoscopic Observation of the Growth Process of a Right-Side Sessile Serrated Adenoma/Polyp with Cytological Dysplasia to an Invasive Submucosal Adenocarcinoma. Case Rep Gastrointest Med. 2016;2016:6576351. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 17. | Ross WA, Thirumurthi S, Lynch PM, Rashid A, Pande M, Shafi MA, Lee JH, Raju GS. Detection rates of premalignant polyps during screening colonoscopy: time to revise quality standards? Gastrointest Endosc. 2015;81:567-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 18. | Zorzi M, Senore C, Da Re F, Barca A, Bonelli LA, Cannizzaro R, de Pretis G, Di Furia L, Di Giulio E, Mantellini P. Detection rate and predictive factors of sessile serrated polyps in an organised colorectal cancer screening programme with immunochemical faecal occult blood test: the EQuIPE study (Evaluating Quality Indicators of the Performance of Endoscopy). Gut. 2017;66:1233-1240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 19. | Yao T, Sugai T, Iwashita A, Fujimori T, Kushima R, Nobuki M, Mitomi H, Ajioka Y, Konishi F. Histopathological characteristics and diagnostic criteria of SSA/P. Project research ‘‘potential of cancerization of colorectal serrated lesions’’ of Japanese Society for Cancer of the Colon and Rectum. Stomach Intest. 2011;46:442-448. |
| 20. | Wada R, Morimoto T, Inayoshi T. Pathological features of the sessile serrated adenoma/polyp with special references of its carcinogenesis. Med Mol Morphol. 2014;47:123-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 21. | Rex DK, Petrini JL, Baron TH, Chak A, Cohen J, Deal SE, Hoffman B, Jacobson BC, Mergener K, Petersen BT. Quality indicators for colonoscopy. Am J Gastroenterol. 2006;101:873-885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 532] [Cited by in RCA: 561] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 22. | Rex DK, Bond JH, Winawer S, Levin TR, Burt RW, Johnson DA, Kirk LM, Litlin S, Lieberman DA, Waye JD. S. Multi-Society Task Force on Colorectal Cancer. Quality in the technical performance of colonoscopy and the continuous quality improvement process for colonoscopy: recommendations of the U.S. Multi-Society Task Force on Colorectal Cancer. Am J Gastroenterol. 2002;97:1296-1308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 691] [Cited by in RCA: 723] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 23. | IJspeert JE, de Wit K, van der Vlugt M, Bastiaansen BA, Fockens P, Dekker E. Prevalence, distribution and risk of sessile serrated adenomas/polyps at a center with a high adenoma detection rate and experienced pathologists. Endoscopy. 2016;48:740-746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 73] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 24. | Rex DK, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, Kaltenbach T, Levin TR, Lieberman D, Robertson DJ. Colorectal cancer screening: Recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;86:18-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 116] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 25. | Payne SR, Church TR, Wandell M, Rösch T, Osborn N, Snover D, Day RW, Ransohoff DF, Rex DK. Endoscopic detection of proximal serrated lesions and pathologic identification of sessile serrated adenomas/polyps vary on the basis of center. Clin Gastroenterol Hepatol. 2014;12:1119-1126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 124] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 26. | Abdeljawad K, Vemulapalli KC, Kahi CJ, Cummings OW, Snover DC, Rex DK. Sessile serrated polyp prevalence determined by a colonoscopist with a high lesion detection rate and an experienced pathologist. Gastrointest Endosc. 2015;81:517-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 135] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 27. | Álvarez C, Andreu M, Castells A, Quintero E, Bujanda L, Cubiella J, Salas D, Lanas Á, Carballo F, Morillas JD, Hernández C, Jover R, Sarasqueta C, Enriquéz-Navascués JM, Hernández V, Estévez P, Macenlle R, Sala T, Balaguer F, Pellisé M, Moreira L, Gil I, Peris A, González-Rubio F, Ferrández A, Poves C, Ponce M, Grau J, Serradesanferm A, Ono A, Cruzado J, Pérez-Riquelme F, Alonso-Abreu I, Carrillo-Palau M, Santander C, Díaz Tasende J, Herreros A, Cacho G, Barranco LE, Bessa X; ColonPrev study investigators. Relationship of colonoscopy-detected serrated polyps with synchronous advanced neoplasia in average-risk individuals. Gastrointest Endosc. 2013;78:333-341.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 53] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 28. | Gao Q, Tsoi KK, Hirai HW, Wong MC, Chan FK, Wu JC, Lau JY, Sung JJ, Ng SC. Serrated polyps and the risk of synchronous colorectal advanced neoplasia: a systematic review and meta-analysis. Am J Gastroenterol. 2015;110:501-509; quiz 510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 66] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 29. | Schreiner MA, Weiss DG, Lieberman DA. Proximal and large hyperplastic and nondysplastic serrated polyps detected by colonoscopy are associated with neoplasia. Gastroenterology. 2010;139:1497-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 204] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 30. | Sanaka MR, Rai T, Navaneethan U, Gohel TD, Podugu A, Thota PN, Lopez R, Kiran RP, Burke CA. Adenoma detection rate in high-risk patients differs from that in average-risk patients. Gastrointest Endosc. 2016;83:172-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 31. | Anderson JC, Butterly LF, Weiss JE, Robinson CM. Providing data for serrated polyp detection rate benchmarks: an analysis of the New Hampshire Colonoscopy Registry. Gastrointest Endosc. 2017;85:1188-1194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 115] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 32. | Rex DK, Ponugoti PL. Calculating the adenoma detection rate in screening colonoscopies only: Is it necessary? Can it be gamed? Endoscopy. 2017;49:1069-1074. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 59] [Article Influence: 7.4] [Reference Citation Analysis (0)] |