Published online Jun 8, 2015. doi: 10.4254/wjh.v7.i10.1337
Peer-review started: August 1, 2014
First decision: September 16, 2014
Revised: February 8, 2015
Accepted: March 5, 2015
Article in press: March 9, 2015
Published online: June 8, 2015
Processing time: 307 Days and 6.6 Hours
There is controversy regarding some aspects of hepatitis C virus (HCV) infection-associated liver steatosis, and their relationship with body fat stores. It has classically been found that HCV, especially genotype 3, exerts direct metabolic effects which lead to liver steatosis. This supports the existence of a so called viral steatosis and a metabolic steatosis, which would affect HCV patients who are also obese or diabetics. In fact, several genotypes exert metabolic effects which overlap with some of those observed in the metabolic syndrome. In this review we will analyse the pathogenic pathways involved in the development of steatosis in HCV patients. Several cytokines and adipokines also become activated and are involved in “pure” steatosic effects, in addition to inflammation. They are probably responsible for the evolution of simple steatosis to steatohepatitis, making it difficult to explain why such alterations only affect a proportion of steatosic patients.
Core tip: Chronic hepatitis C virus (HCV) infection can lead to steatosis and steatohepatitis. Increased liver triglyceride synthesis is mediated by several transcription factors such as sterol regulatory element-binding protein (SREBP) whose expression is enhanced, in turn, by HCV core protein. Chronic HCV infection is also associated with insulin resistance that seems to be selective because although it activates systemic lipolysis, it increases triglyceride synthesis within the liver. This is due to the stimulatory effect of insulin on SREBP. It remains to be answered why not all patients with HCV infection and steatosis develop steatohepatitis despite early cytokine activation and metabolic derangements.
- Citation: González-Reimers E, Quintero-Platt G, Rodríguez-Gaspar M, Alemán-Valls R, Pérez-Hernández O, Santolaria-Fernández F. Liver steatosis in hepatitis C patients. World J Hepatol 2015; 7(10): 1337-1346
- URL: https://www.wjgnet.com/1948-5182/full/v7/i10/1337.htm
- DOI: https://dx.doi.org/10.4254/wjh.v7.i10.1337
Non-alcoholic steatohepatitis is an increasingly common situation, in which fat-laden hepatocytes trigger an inflammatory response which may evolve to liver cirrhosis[1] and hepatocarcinoma[2]. Diabetes and obesity are the most important causes, since insulin deficiency and/or resistance alter the mobilization of fatty acids from adipose tissue to liver, the oxidative pathways, and lipid trafficking between liver and peripheral tissues. Steatosis and steatohepatitis are also observed in chronic hepatitis C virus (HCV) infection. Although HCV by itself-especially genotype 3a- may lead to liver steatosis, obesity and concomitant alcohol abuse are main factors involved[3]. The “two hit theory” sustains that cytokine activation and increased lipid peroxidation contribute to the evolution of liver steatosis to more advanced stages of steatohepatitis[4]. Throughout this manuscript we will show that cytokine activation may already exist, at least theoretically, in early stages of the disease (simple steatosis), but not all the patients showing simple steatosis develop steatohepatitis.
The outstanding role played by some genotype specific HCV viral proteins, which either have a direct steatogenic effect or induce insulin resistance, explains why some HCV infected individuals show liver steatosis in the absence of obesity and has led to the concept that there are two main pathogenetic mechanisms of steatosis in these patients: the so-called “metabolic” steatosis and “viral” steatosis. In the present paper we will revise the main pathways leading to steatosis in these HCV patients. However, viral and non-viral dependent pathways are intermingled, so we will not treat them separately. Also, although the objective of this review is only to revise mechanisms leading to steatosis, and not steatohepatitis, many pathways involved in simple steatosis are already able to trigger inflammation, which is the hallmark of steatohepatitis, so a precise limit between both clinicopathological stages is lacking. We will comment only those aspects of proinflammatory cytokines involved in the pathogenesis of “pure” steatosis.
As mentioned above, patients infected by genotype 3a HCV develop liver steatosis even in the absence of obesity[5], a finding which supports a direct cytopathic and steatogenic effect of this precise genotype[6]. Recent research has shown that this viral effect depends on several mechanisms which will be commented in this review. HCV genotype 3a up-regulates the expression of fatty acid synthase[7]. There are also data which suggest that in chronic hepatitis secondary to HCV there is decreased mitochondrial β-oxidation, possibly due to mitochondrial damage[8]. In addition, HCV impairs export of very low density lipoprotein (VLDL) particles from the liver to peripheral tissues, by several mechanisms. Hepatocyte release of HCV particles utilises the same pathway used in VLDL export, and HCV mediates inhibition of the microsomal triglyceride transfer protein (MTP), a molecule involved in export of intrahepatocytary triglycerides[9].
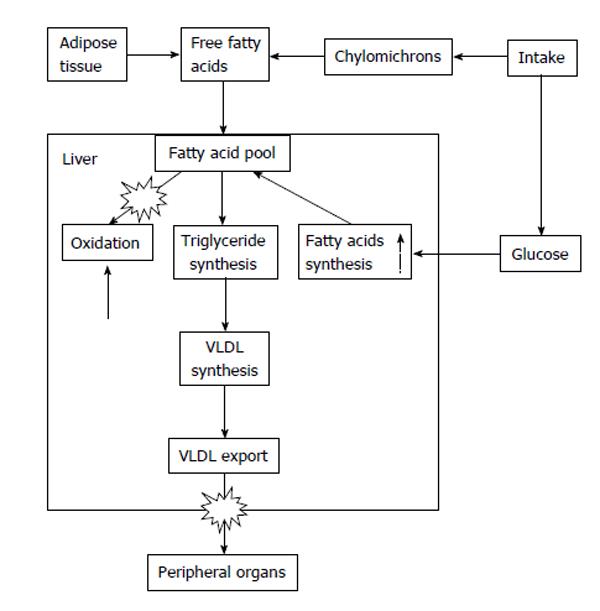
In addition, several viral proteins of diverse genotypes interfere with insulin signalling, leading to insulin resistance. Insulin resistance is the hallmark of obesity, but in HCV infection, patients do not necessarily have to be overweight for them to develop insulin resistance: despite the principal role of obesity and associated insulin resistance on liver steatosis, this lesion may develop in the face of a normal body mass index (BMI). Therefore, at any given load of fatty acids, HCV infected hepatocytes up-regulate synthesis of more fatty acids, impair β-oxidation of the available fatty acids, and impede the export of triglycerides (Figure 1).
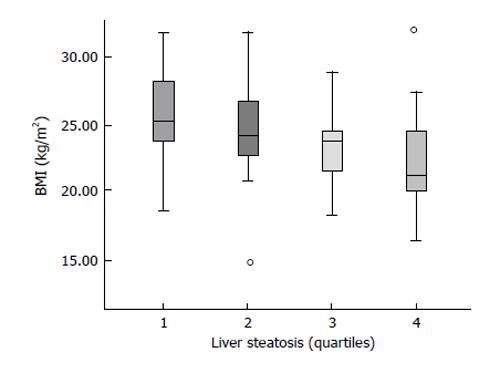
Fat mobilization is a necessary condition to develop liver steatosis, and liver steatosis is more intense the greater the BMI[10], also in HCV patients (Figure 2). During fasting-a situation characterized by low insulin levels-, fatty acids are released by the adipocyte and reach the liver, where they are taken up by liver cells and are destined to be used either as fuel, as a source of ketone bodies or they can be combined again with glycerol to be re-esterified as triglycerides. Triglycerides coupled with apoproteins and cholesterol form the so called VLDL which are then exported to peripheral tissues.
A situation with some features similar to those observed during fasting may take place in conditions accompanied by insulin resistance: in adipose tissue insulin fails to suppress lipolysis, so that an increased amount of free fatty acids reaches the liver. But in fasting, insulin levels are low, whereas in situations of insulin resistance, insulin levels are usually high. High insulin levels, even in a situation of insulin resistance and not-supressed lipolysis, still enhance liver triglyceride synthesis, but not adipocyte synthesis of triglyceride. Liver triglyceride synthesis implies esterification of glycerol with fatty acids. These fatty acids may derive from adipose tissue, from ingested fat, and also from ingested carbohydrates, the latter constituting the amount synthesized “de novo” by the liver. In insulin-resistant patients with non-alcoholic fatty liver disease the rate of de novo lipid synthesis is increased. Donnelly et al[11] showed that 26% of the triglycerides stored in the liver of 9 obese subjects with non-alcoholic fatty liver disease derived from de novo lipogenesis, in contrast with the 5% contribution (in the fasted state) observed among normal individuals[12]. This increased de novo liver lipogenesis in insulin-resistance situations is accompanied by a reduced triglyceride synthesis within the adipocyte, due to decreased availability of glycerol 3 phosphate, which is in turn due to an insulin-resistance-mediated decrease in glucose uptake[13].
Several transcription factors are involved in increased liver lipid synthesis[14]. These are:
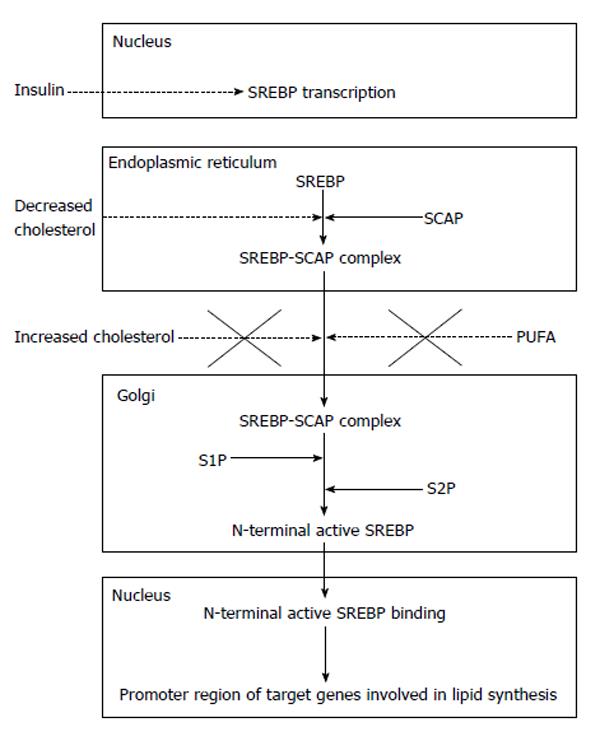
(1) Sterol regulatory element-binding proteins (SREBP), especially the SREBP-1c. SREBP-1c enhances transcription of genes required for fatty acid synthesis and predominates in the liver[15]. When cells become depleted in cholesterol, a protein called SREBP cleavage-activating protein (SCAP) binds to SREBP and transports it from the endoplasmic reticulum to the Golgi apparatus. In the Golgi apparatus there are two proteases (site 1 protease or S1P, and site 2 protease or S2P) which act sequentially to release the N-terminal active form of SREBP, which enters the nucleus and binds to a sterol responsive element in the enhancer/promoter region of the target genes (for instance, fatty acid synthase), activating transcription. The movement of the SREBP-SCAP complex from the endoplasmic reticulum to the Golgi apparatus is supressed by high intracellular cholesterol levels; therefore, the SRBEP-SCAP system can be viewed as a sensor of cholesterol levels in the hepatocyte[16], although this inhibitory action affects SREBP-2. Inhibition of SREBP-1 processing requires the presence of polyunsaturated fatty acids in addition to cholesterol[17] (Figure 3).
SREBP-2 is also present in liver and other organs, and is more specifically involved in cholesterol synthesis. However, when expressed at higher than normal levels, each of the three SREBP isoforms can activate both cholesterol and fatty acid synthesis[18].
SREBP-1a is expressed only at low levels in liver, but in studies performed on genetically engineered mice, overexpression of SRBEP 1a led to a 26-fold increase in fatty liver synthesis and a massive liver steatosis[19].
SREBP transcription is strongly stimulated by insulin[20], whereas glucagon exerts an inhibitory effect. Over-expression of SREBP-1c may lead to an excessive synthesis of fatty acids (and cholesterol and triglycerides) within the liver cell, ultimately leading to liver steatosis[14,21]. Liver X-activated receptors (LXR) α and β are involved in SREBP-1c transcription. These are nuclear receptors that heterodimerize with retinoid X receptors after binding to a ligand. In the case of SREBP, they bind to a LXR response element in the promoter region of the SREBP-1 gene and activate SRBP-1c transcription[22]. Thus, glucagon, insulin, and LXR are classical modulators of SREBP-1c transcription.
The activity of SREBP-1c is increased in several situations in which liver steatosis ensues, such as alcoholism (acetaldehyde enhances its transcription[23]), high tumor necrosis factor alpha (TNF-α) levels[24], or HCV infection[25]. HCV core protein enhances both gene expression of SRBP and transcriptional activity of this molecule[26].
On the other hand, SRBP-1 is inactivated by sirtuin-1 (SIRT-1), a molecule whose activity is modulated by several variables, including ethanol and HCV core protein, among others[27]. Sirtuins are involved in the modulation of transcription factor activity by deacetylation of proteins. Biological activity of sirtuins depends on nicotinamide adenine dinucleotide (NAD) availability[28]. Specifically, sirtuin1 deacetylates and inhibitis SREBP-1c activity, therefore decreasing fat synthesis. Ethanol inhibitis sirtuin-1 activity, therefore increasing the lipogenic effect of SREBP 1-c[26]. HCV exerts a similar effect[29].
Sirtuin activity is coupled to that of AMP-activated kinase (AMPK), an enzyme that phosphorylates and, thus, inhibits, acetyl-CoA carboxylase, interrupting the formation of malonyl-CoA, a key step for fatty acids synthesis, and preserving cellular content in NAD[30]. Sirtuin activates AMPK acting on serine/threonine kinase 11, also known as liver kinase B1, a process which leads to an increase in cellular NAD availability, which favours SIRT-1 activity[31]. This reciprocally regulated circuit leads to inhibition of SREBP activity and fatty acids synthesis. It is important to keep in mind that ethanol metabolism consumes NAD, theoretically opposing to sirtuin activation.
Endocannabinoids are also involved in enhanced expression of SREBP-1c[32]. Cannabinoid agonists are orexigenic, and animal models support a role of endocannabinoids on diet-induced liver steatosis[33]. Daily cannabis consumption aggravates steatosis in HCV patients[34]. Conversely, HCV infection may up-regulate cannabinoid receptor 1 expression[35].
(2) However, although cholesterol synthesis seems to depend almost entirely on SREBP activity, suppression of the SREBPs machinery reduces fatty acid synthesis by only 30%[36]. Another transcription factor involved in liver steatosis is carbohydrate response element binding protein (ChREBP), whose activity is induced by a high carbohydrate diet, insulin[37] and ethanol[38]. It increases the expression of both lipogenic enzymes (such as fatty acid synthase) and glycolytic ones[39]. The effect of HCV on ChREBP is not known, to our knowledge.
(3) Peroxisome proliferator-activated receptor (PPAR)-γ is another master transcription factor involved in fat metabolism. Increased activity of PPAR-γ is associated with an increase in lipid synthesis[40] and is seen in patients with liver steatosis. It upregulates genes involved in lipid synthesis, increasing the activity of mediators such as SREBP-1, fatty acid synthase and acetyl coenzyme a carboxylase, all of them leading to increased hepatocyte lipid content.
Interestingly, PPAR-γ is related to increased expression of genes that regulate the synthesis of adipose differentiation related protein[41], which functions to coat lipid droplets within liver cells[42]. It has been shown that HCV core protein increases the transcriptional activity of PPARgamma, although it exerted no effect on PPARgamma gene expression[43].
Synthesis of triglycerides is a complex process, in which several enzymes participate. During fasting, the increased flux of fatty acids to the liver increases the translocation of lipin-1, a protein with dual activity on fatty acid metabolism[44]. Lipin proteins translocate from the cytosol to the endoplasmic reticulum where they show phosphatidate phosphatase (PAP 1) activity. This enzymatic activity transforms diacylglycerol 3 phosphate into diacylglycerol (DAG), which serves as substrate for triglyceride and phopholipid synthesis. Fatty acids are added to the DAG molecule through the action of acyl coenzyme A:diacylglycerol acyltransferase to form triglycerides[45].
In addition to its PAP 1 activity, lipin also translocates to the nucleus, where it enhances expression of genes involved in fatty acid oxidation[46]. This requires interaction with PPAR-α and PPAR-γ coactivator 1α, forming a physical complex. This leads to decreased intracellular levels of fatty acids which defends the cell from the damaging effect of these molecules[47,48].
Consistent with its effect on free fatty acids, insulin stimulates the activity of lipin-1 by unknown mechanisms, and obesity-related insulin resistance down-regulates lipin gene expression[49]. PAP 1 activity is enhanced in ethanol-induced liver steatosis[50], but lipin deficiency may exacerbate ethanol-associated liver steatosis-perhaps by impairment of fatty acid oxidation[51]. Concordantly, ethanol up-regulates lipin-1 gene expression[52]. Liver lipin is also regulated by SIRT-1[53], a molecule whose activity is inhibited by ethanol. However, to our knowledge, the effect of HCV on lipin proteins has not been analysed.
Fatty acid oxidation takes place mainly in the mitochondria, although in a small proportion it also includes microsomal ω- and peroxisomal β-oxidation. Improper fatty acid oxidation may also contribute to liver steatosis. As mentioned earlier, AMPK stimulates hepatic fatty acid oxidation and ketogenesis, since it lowers malonyl-CoA liver content, thereby permitting fatty acid transport to the mitochondria, where they suffer oxidation[30]. Ethanol exerts an inhibitory effect on AMPK[54] and HCV also downregulates AMPK[29]. In a study performed on 30 patients infected with HCV it was found that mitochondrial β-oxidation of fatty acids was impaired and that this impairment was related to serum levels of HCV core protein[8]. Therefore, both in alcoholic and non alcoholic fatty liver disease impaired fatty acid oxidation plays an crucial role, without the need of accompanying mechanisms. However, insulin resistance and proinflammatory cytokines also exert major effects on this mechanism.
Normally, insulin activates acetyl CoA-carboxylase, leading to the formation of malonyl-CoA, which inhibits mitochondrial fatty acid oxidation; it also strongly inhibits gluconeogenesis by blocking key enzymes such as phosphoenolpyruvate carboxykinase and glucose 6 phosphatase. Additionally, it inhibits lipolysis and promotes glycogen synthesis and de novo fat synthesis using carbohydrates as substrate. Finally, it favours SREBP and ChREBP transcription, as was mentioned above. Therefore, it exerts lipogenic effects on the liver cell[55]. Insulin action takes place after binding to a specific receptor, which, upon activation, leads to the phosphorylation of a series of inactive kinases called insulin responsive substrates (IRS), transforming them into active ones. Some final effects of this complex cascade of kinases include phosphorylation of transcription factors, such as forkhead box protein (FOX)O1 and FOXA2, among others[56]. Phosphorylated FOXO1 is unable to activate transcription of key enzymes involved in gluconeogenesis, such as phosphenolpyruvate carboxy-kinase or glucose 6 phosphatase, and thus, liver production of glucose is blocked[57]. Another transcription factor-FOXA2- is involved in hepatic fatty acid oxidation[58].
In states of insulin resistance, insulin fails to phosphorylate FOXO1 and therefore, it fails to block gluconeogenesis. Therefore, fasting hyperglycaemia is observed despite hyperinsulinism and lipolysis is also activated, leading to an increase in the fatty acid load to the liver. However, the expected decrease in liver triglyceride synthesis is not observed. This is interpreted as a result of the stimulatory effect of insulin on SREBP-1c, favouring triglyceride synthesis. Therefore, insulin resistance is selective[59]: the lack of inhibition of gluconeogenesis leads to hyperglycaemia and in turn hyperglycaemia leads to increased insulin secretion, but SREBP activity is enhanced, leading to increased triglyceride synthesis. Other factors that are mentioned below may possibly aid in explaining this paradox.
In normal conditions, insulin not only has an adipogenic effect on the hepatocyte but it also limits VLDL secretion[60]. This effect is mainly dependent on an insulin-derived increased rate of degradation of apoprotein (apo) B but it is also due to the inhibition of apo B 100 synthesis. This preserves the triglycerides stored in the hepatocytes from utilization in the postprandrial state, so that they do not compete with the exogenous fatty acids. Apo B synthesis is a necessary step for VLDL formation. Newly synthesized apo B translocates into the endoplasmic reticulum and encounters MTP, among other chaperone proteins[61]. Importantly, FOXO1 enhances MTP expression[62]. This may explain why in conditions associated with insulin resistance the postprandrial decrease in VLDL secretion does not take place, and why hypertriglyceridemia constitutes a feature of the metabolic syndrome. As mentioned earlier, HCV is able to modulate MTP activation, directly promoting steatosis[9,63]. A recently described orphan receptor protein (orphan receptor small heterodimer partner) also plays an important role in the development of liver steatosis, although precise mechanisms are still unknown[64]. It possibly represses transcriptional activation of MTP; HCV increases its expression[65].
HCV also directly provokes insulin resistance. In HCV infection, insulin resistance is more closely related to viral load than to obesity, supporting a direct effect of HCV on insulin metabolism[66]. In fact, diabetes is more frequently observed among HCV patients[67]. The mechanisms involved in insulin resistance seem to be genotype-specific. It has been shown that HCV non-structural protein 5A (NS5A) is able to phosphorylate serine residues of IRS-1, thereby interfering with the post receptor downstream cascade of insulin action[68]. In accordance with this fact, treatment of HCV patients with pegylated interferon and ribavirin reduces insulin resistance assessed by homeostasis model for assessment[69]. Moreover, NS5A protein also exerts direct lipogenic effect through activation of LXRs[70].
In addition to the effect of non-structural proteins, it has been recently shown that HCV 1 and 4 core proteins are able to alter the degradation of IRS-1 and IRS-2 in a pathway dependent on suppressor of cytokine signalling 3 (SOCS3), thus also altering insulin signalling[71,72] by stimulating ubiquitination and subsequent degradation of IRS. Moreover, Pazienza et al[73], in 2007, showed that core protein of genotype 3a promoted IRS degradation by down-regulation of PPAR γ and up-regulation of SOCS7, whereas the core protein of genotype 1b activated the mammalian target of rapamycin (mTOR). Activation of mTOR leads to insulin resistance[74], but also exerts a direct effect on SREBP-1, leading to increased lipid synthesis. Some authors believe that the main mediator of increased lipogenesis in conditions characterized by insulin resistance is mTOR, acting on SREBP[75].
We have seen that steatosis ultimately depends upon the fatty acids pool, mainly derived from adipose tissue. It is also important to keep in mind that fat is not only a source of free fatty acids, but also a source of pro-inflammatory and anti-inflammatory cytokines which are able to modulate the circulation of free fatty acids from fat tissue to liver and again from liver to peripheral tissues[76]. These cytokines are also involved in some key steps of the progressive liver damage observed in individuals affected by steatohepatitis. To add more complexity to this scenario, recent research has shown that fat tissue is not homogeneous. Trunk fat is associated with increased insulin resistance and vascular risk[77], whereas leg fat exerts opposite effects[78]. These differences probably reflect the secretion of a different cytokine profile. In general, trunk fat has a “negative” cytokine profile: it secretes less adiponectin, a “protective” cytokine, but more TNF and interleukin (IL)-6 than in the gynoid profile of fat distribution (fat around the hips and legs which is associated with increased production of adiponectin)[79]. Therefore, it is important to analyse the diverse fat compartments when studying the influence of these cytokines on liver steatosis. The cytokine profile associated with hepatitis C-liver steatosis, and the potential role of these cytokines on liver fat deposition is controversial[80-82] as well as their relationships with histological changes in chronic HCV infection.
Although cytokines are definitely involved in the inflammatory process which marks the evolution from steatosis to steatohepatitis, they also play a role in simple steatosis, both by aggravating it directly, or by affecting the metabolic axis which controls fatty acid trafficking. For instance, TNF-α is involved in SREBP activation[24]. On the contrary adiponectin activates fatty acid oxidation[83,84], but decreases the activity of fatty acid synthase and acetylcoenzyme A carboxylase[85]. Moreover, it inhibits liver production of TNF[86]. By upregulation of AMP activated protein kinase activity it also influences other pathways involved in lipid metabolism decreasing SREBP-1 and up-regulating PPAR-α in several tissues[87], an effect which is shared by IL-6[88]. PPAR-α is a transcription factor for several genes involved in the transport, oxidation, and export of free fatty acids[89,90]. It can be viewed as a sensor of intracellular free fatty acids, since it becomes activated by intracellular free fatty acids. PPAR-α deficiency promotes the development of fatty liver, and its activity is altered by classic factors involved in liver steatosis, such as ethanol consumption and HCV infection. Chronic ethanol feeding inhibits PPAR-α function due to the effect of acetaldehyde, which inhibits transcriptional activation of PPAR-α[91]. HCV causes down-regulation of PPAR-α[92], and is able to inhibit its activity by inducing repression of PPAR-α signaling by micro RNA-27b[93].
Leptin, another fat-derived cytokine, may promote fibrogenesis through up-regulation of transforming growth factor-β[94], but it also protects the liver from fat accumulation by lowering the expression of SREBP-1[95]. These nearly opposite effects may explain, perhaps, disparate findings in relation to leptin levels in chronic HCV infection[96]. Increased leptin levels, but also normal[97] or even decreased ones[98] have been reported in chronic HCV infection and leptin may[99] or may not be related to liver steatosis[96,100] in chronic HCV infection.
Increased trunk fat is not the only factor responsible for increased cytokine secretion in HCV infection. Increased reactive oxygen species (ROS)-which also directly impair mitochondrial oxidation of fatty acids[101] activate nuclear factor kappa B (NFκB), a key transcription factor for the expression of cytokines[102] such as TNF-α or IL-6, among others. In addition to the many proinflammatory effects of TNF-α, it also causes insulin resistance and liver steatosis by inhibiting IRS[103]. Excessive ROS production depends on the intracellular effect of HCV. NS3 and 5A are able to activate mitochondrial ROS production by altering calcium trafficking at the endoplasmic reticulum membrane[104]. This altered calcium influx also triggers increased transcription of STAT-3 and NFκB, leading to increased cytokine production which closes a positive feed-back loop. In addition, NS3 and NS 5A are also able to stimulate toll-like receptor-4, in a way similarly to that caused by the lipopolysaccharide in the initial stages of alcoholic hepatitis[105]. Furthermore, LXR, which can be directly activated by HCV, regulates a set of genes that encode proinflammatory mediators[43].
Liver steatosis is a very complex process, in which many proteins and enzymes are involved. As shown, viral proteins may affect several of the metabolic pathways leading to simple steatosis, including cytokine activation. Indeed, cytokine production takes place even at early stages, and, among many other questions outlined in this review, it remains to be answered why, despite early cytokine activation, only some patients evolve to steatohepatitis, a key step in the progression of HCV-induced liver damage.
P- Reviewer: Dehghani SM, Pavlidis C, Snyder N
S- Editor: Gong XM L- Editor: A E- Editor: Liu SQ
| 1. | Bugianesi E, Leone N, Vanni E, Marchesini G, Brunello F, Carucci P, Musso A, De Paolis P, Capussotti L, Salizzoni M. Expanding the natural history of nonalcoholic steatohepatitis: from cryptogenic cirrhosis to hepatocellular carcinoma. Gastroenterology. 2002;123:134-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1036] [Cited by in RCA: 1018] [Article Influence: 44.3] [Reference Citation Analysis (0)] |
| 2. | Hui E, Xu A, Bo Yang H, Lam KS. Obesity as the common soil of non-alcoholic fatty liver disease and diabetes: Role of adipokines. J Diabetes Investig. 2013;4:413-425. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 3. | Woreta TA, Sutcliffe CG, Mehta SH, Brown TT, Higgins Y, Thomas DL, Torbenson MS, Moore RD, Sulkowski MS. Incidence and risk factors for steatosis progression in adults coinfected with HIV and hepatitis C virus. Gastroenterology. 2011;140:809-817. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 4. | Lonardo A, Adinolfi LE, Loria P, Carulli N, Ruggiero G, Day CP. Steatosis and hepatitis C virus: mechanisms and significance for hepatic and extrahepatic disease. Gastroenterology. 2004;126:586-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 356] [Cited by in RCA: 338] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 5. | Adinolfi LE, Gambardella M, Andreana A, Tripodi MF, Utili R, Ruggiero G. Steatosis accelerates the progression of liver damage of chronic hepatitis C patients and correlates with specific HCV genotype and visceral obesity. Hepatology. 2001;33:1358-1364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 778] [Cited by in RCA: 774] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 6. | Kumar D, Farrell GC, Fung C, George J. Hepatitis C virus genotype 3 is cytopathic to hepatocytes: Reversal of hepatic steatosis after sustained therapeutic response. Hepatology. 2002;36:1266-1272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 256] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 7. | Jackel-Cram C, Babiuk LA, Liu Q. Up-regulation of fatty acid synthase promoter by hepatitis C virus core protein: genotype-3a core has a stronger effect than genotype-1b core. J Hepatol. 2007;46:999-1008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 120] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 8. | Sato C, Saito T, Misawa K, Katsumi T, Tomita K, Ishii R, Haga H, Okumoto K, Nishise Y, Watanabe H. Impaired mitochondrial β-oxidation in patients with chronic hepatitis C: relation with viral load and insulin resistance. BMC Gastroenterol. 2013;13:112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 9. | Perlemuter G, Sabile A, Letteron P, Vona G, Topilco A, Chrétien Y, Koike K, Pessayre D, Chapman J, Barba G. Hepatitis C virus core protein inhibits microsomal triglyceride transfer protein activity and very low density lipoprotein secretion: a model of viral-related steatosis. FASEB J. 2002;16:185-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 419] [Cited by in RCA: 428] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 10. | Goossens N, Negro F. The impact of obesity and metabolic syndrome on chronic hepatitis C. Clin Liver Dis. 2014;18:147-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 11. | Donnelly KL, Smith CI, Schwarzenberg SJ, Jessurun J, Boldt MD, Parks EJ. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest. 2005;115:1343-1351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2112] [Cited by in RCA: 2599] [Article Influence: 130.0] [Reference Citation Analysis (0)] |
| 12. | Timlin MT, Parks EJ. Temporal pattern of de novo lipogenesis in the postprandial state in healthy men. Am J Clin Nutr. 2005;81:35-42. [PubMed] |
| 13. | Tamura S, Shimomura I. Contribution of adipose tissue and de novo lipogenesis to nonalcoholic fatty liver disease. J Clin Invest. 2005;115:1139-1142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 14. | Anderson N, Borlak J. Molecular mechanisms and therapeutic targets in steatosis and steatohepatitis. Pharmacol Rev. 2008;60:311-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 297] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 15. | Shimomura I, Shimano H, Horton JD, Goldstein JL, Brown MS. Differential expression of exons 1a and 1c in mRNAs for sterol regulatory element binding protein-1 in human and mouse organs and cultured cells. J Clin Invest. 1997;99:838-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 589] [Cited by in RCA: 610] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 16. | Horton JD, Goldstein JL, Brown MS. SREBPs: activators of the complete program of cholesterol and fatty acid synthesis in the liver. J Clin Invest. 2002;109:1125-1131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 188] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 17. | Hannah VC, Ou J, Luong A, Goldstein JL, Brown MS. Unsaturated fatty acids down-regulate srebp isoforms 1a and 1c by two mechanisms in HEK-293 cells. J Biol Chem. 2001;276:4365-4372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 344] [Cited by in RCA: 353] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 18. | Shimomura I, Shimano H, Korn BS, Bashmakov Y, Horton JD. Nuclear sterol regulatory element-binding proteins activate genes responsible for the entire program of unsaturated fatty acid biosynthesis in transgenic mouse liver. J Biol Chem. 1998;273:35299-35306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 293] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 19. | Shimano H, Horton JD, Hammer RE, Shimomura I, Brown MS, Goldstein JL. Overproduction of cholesterol and fatty acids causes massive liver enlargement in transgenic mice expressing truncated SREBP-1a. J Clin Invest. 1996;98:1575-1584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 651] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 20. | Shimomura I, Bashmakov Y, Ikemoto S, Horton JD, Brown MS, Goldstein JL. Insulin selectively increases SREBP-1c mRNA in the livers of rats with streptozotocin-induced diabetes. Proc Natl Acad Sci USA. 1999;96:13656-13661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 587] [Cited by in RCA: 601] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 21. | Shimano H, Horton JD, Shimomura I, Hammer RE, Brown MS, Goldstein JL. Isoform 1c of sterol regulatory element binding protein is less active than isoform 1a in livers of transgenic mice and in cultured cells. J Clin Invest. 1997;99:846-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 633] [Cited by in RCA: 677] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 22. | Baranowski M. Biological role of liver X receptors. J Physiol Pharmacol. 2008;59 Suppl 7:31-55. [PubMed] |
| 23. | You M, Fischer M, Deeg MA, Crabb DW. Ethanol induces fatty acid synthesis pathways by activation of sterol regulatory element-binding protein (SREBP). J Biol Chem. 2002;277:29342-29347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 382] [Cited by in RCA: 415] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 24. | Endo M, Masaki T, Seike M, Yoshimatsu H. TNF-alpha induces hepatic steatosis in mice by enhancing gene expression of sterol regulatory element binding protein-1c (SREBP-1c). Exp Biol Med (Maywood). 2007;232:614-621. [PubMed] |
| 25. | Bose SK, Kim H, Meyer K, Wolins N, Davidson NO, Ray R. Forkhead box transcription factor regulation and lipid accumulation by hepatitis C virus. J Virol. 2014;88:4195-4203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 51] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 26. | Kim KH, Hong SP, Kim K, Park MJ, Kim KJ, Cheong J. HCV core protein induces hepatic lipid accumulation by activating SREBP1 and PPARgamma. Biochem Biophys Res Commun. 2007;355:883-888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 108] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 27. | Everitt H, Hu M, Ajmo JM, Rogers CQ, Liang X, Zhang R, Yin H, Choi A, Bennett ES, You M. Ethanol administration exacerbates the abnormalities in hepatic lipid oxidation in genetically obese mice. Am J Physiol Gastrointest Liver Physiol. 2013;304:G38-G47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 53] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 28. | Ponugoti B, Kim DH, Xiao Z, Smith Z, Miao J, Zang M, Wu SY, Chiang CM, Veenstra TD, Kemper JK. SIRT1 deacetylates and inhibits SREBP-1C activity in regulation of hepatic lipid metabolism. J Biol Chem. 2010;285:33959-33970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 447] [Cited by in RCA: 438] [Article Influence: 29.2] [Reference Citation Analysis (0)] |
| 29. | Yu JW, Sun LJ, Liu W, Zhao YH, Kang P, Yan BZ. Hepatitis C virus core protein induces hepatic metabolism disorders through down-regulation of the SIRT1-AMPK signaling pathway. Int J Infect Dis. 2013;17:e539-e545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 47] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 30. | Shirwany NA, Zou MH. AMPK: a cellular metabolic and redox sensor. A minireview. Front Biosci (Landmark Ed). 2014;19:447-474. [PubMed] |
| 31. | Hou X, Xu S, Maitland-Toolan KA, Sato K, Jiang B, Ido Y, Lan F, Walsh K, Wierzbicki M, Verbeuren TJ. SIRT1 regulates hepatocyte lipid metabolism through activating AMP-activated protein kinase. J Biol Chem. 2008;283:20015-20026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 590] [Cited by in RCA: 650] [Article Influence: 38.2] [Reference Citation Analysis (0)] |
| 32. | Alswat KA. The role of endocannabinoids system in fatty liver disease and therapeutic potentials. Saudi J Gastroenterol. 2013;19:144-151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 33. | Osei-Hyiaman D, Liu J, Zhou L, Godlewski G, Harvey-White J, Jeong WI, Bátkai S, Marsicano G, Lutz B, Buettner C. Hepatic CB1 receptor is required for development of diet-induced steatosis, dyslipidemia, and insulin and leptin resistance in mice. J Clin Invest. 2008;118:3160-3169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 357] [Cited by in RCA: 381] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 34. | Hézode C, Zafrani ES, Roudot-Thoraval F, Costentin C, Hessami A, Bouvier-Alias M, Medkour F, Pawlostky JM, Lotersztajn S, Mallat A. Daily cannabis use: a novel risk factor of steatosis severity in patients with chronic hepatitis C. Gastroenterology. 2008;134:432-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 124] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 35. | Sun LJ, Yu JW, Wan L, Zhang XY, Shi YG, Chen MY. Endocannabinoid system activation contributes to glucose metabolism disorders of hepatocytes and promotes hepatitis C virus replication. Int J Infect Dis. 2014;23:75-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 36. | Pai JT, Guryev O, Brown MS, Goldstein JL. Differential stimulation of cholesterol and unsaturated fatty acid biosynthesis in cells expressing individual nuclear sterol regulatory element-binding proteins. J Biol Chem. 1998;273:26138-26148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 174] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 37. | Denechaud PD, Dentin R, Girard J, Postic C. Role of ChREBP in hepatic steatosis and insulin resistance. FEBS Lett. 2008;582:68-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 106] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 38. | Ronis MJ, Mercer K, Suva LJ, Vantrease J, Ferguson M, Hogue WR, Sharma N, Cleves MA, Blackburn ML, Badger TM. Influence of fat/carbohydrate ratio on progression of fatty liver disease and on development of osteopenia in male rats fed alcohol via total enteral nutrition (TEN). Alcohol. 2014;48:133-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 39. | Dentin R, Denechaud PD, Benhamed F, Girard J, Postic C. Hepatic gene regulation by glucose and polyunsaturated fatty acids: a role for ChREBP. J Nutr. 2006;136:1145-1149. [PubMed] |
| 40. | Schadinger SE, Bucher NL, Schreiber BM, Farmer SR. PPARgamma2 regulates lipogenesis and lipid accumulation in steatotic hepatocytes. Am J Physiol Endocrinol Metab. 2005;288:E1195-E1205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 312] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 41. | Motomura W, Inoue M, Ohtake T, Takahashi N, Nagamine M, Tanno S, Kohgo Y, Okumura T. Up-regulation of ADRP in fatty liver in human and liver steatosis in mice fed with high fat diet. Biochem Biophys Res Commun. 2006;340:1111-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 139] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 42. | Bickel PE, Tansey JT, Welte MA. PAT proteins, an ancient family of lipid droplet proteins that regulate cellular lipid stores. Biochim Biophys Acta. 2009;1791:419-440. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 551] [Cited by in RCA: 521] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 43. | Lima-Cabello E, García-Mediavilla MV, Miquilena-Colina ME, Vargas-Castrillón J, Lozano-Rodríguez T, Fernández-Bermejo M, Olcoz JL, González-Gallego J, García-Monzón C, Sánchez-Campos S. Enhanced expression of pro-inflammatory mediators and liver X-receptor-regulated lipogenic genes in non-alcoholic fatty liver disease and hepatitis C. Clin Sci (Lond). 2011;120:239-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 113] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 44. | Reue K, Brindley DN. Thematic Review Series: Glycerolipids. Multiple roles for lipins/phosphatidate phosphatase enzymes in lipid metabolism. J Lipid Res. 2008;49:2493-2503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 152] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 45. | Yen CL, Stone SJ, Koliwad S, Harris C, Farese RV. Thematic review series: glycerolipids. DGAT enzymes and triacylglycerol biosynthesis. J Lipid Res. 2008;49:2283-2301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 890] [Cited by in RCA: 818] [Article Influence: 48.1] [Reference Citation Analysis (0)] |
| 46. | Finck BN, Gropler MC, Chen Z, Leone TC, Croce MA, Harris TE, Lawrence JC, Kelly DP. Lipin 1 is an inducible amplifier of the hepatic PGC-1alpha/PPARalpha regulatory pathway. Cell Metab. 2006;4:199-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 477] [Cited by in RCA: 458] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 47. | Bechmann LP, Kocabayoglu P, Sowa JP, Sydor S, Best J, Schlattjan M, Beilfuss A, Schmitt J, Hannivoort RA, Kilicarslan A. Free fatty acids repress small heterodimer partner (SHP) activation and adiponectin counteracts bile acid-induced liver injury in superobese patients with nonalcoholic steatohepatitis. Hepatology. 2013;57:1394-1406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 188] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 48. | Abdel-aleem S, Badr M, Perez-Tamayo RA, Anstadt MP, Lowe JE. Stimulation of myocyte insulin-responsive glucose transporters by the inhibition of fatty acid oxidation. Diabetes Res. 1993;22:11-19. [PubMed] |
| 49. | Croce MA, Eagon JC, LaRiviere LL, Korenblat KM, Klein S, Finck BN. Hepatic lipin 1beta expression is diminished in insulin-resistant obese subjects and is reactivated by marked weight loss. Diabetes. 2007;56:2395-2399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 62] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 50. | Reue K, Zhang P. The lipin protein family: dual roles in lipid biosynthesis and gene expression. FEBS Lett. 2008;582:90-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 162] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 51. | Hu M, Yin H, Mitra MS, Liang X, Ajmo JM, Nadra K, Chrast R, Finck BN, You M. Hepatic-specific lipin-1 deficiency exacerbates experimental alcohol-induced steatohepatitis in mice. Hepatology. 2013;58:1953-1963. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 52. | Hu M, Wang F, Li X, Rogers CQ, Liang X, Finck BN, Mitra MS, Zhang R, Mitchell DA, You M. Regulation of hepatic lipin-1 by ethanol: role of AMP-activated protein kinase/sterol regulatory element-binding protein 1 signaling in mice. Hepatology. 2012;55:437-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 114] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 53. | Yin H, Hu M, Liang X, Ajmo JM, Li X, Bataller R, Odena G, Stevens SM, You M. Deletion of SIRT1 from hepatocytes in mice disrupts lipin-1 signaling and aggravates alcoholic fatty liver. Gastroenterology. 2014;146:801-811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 178] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 54. | García-Villafranca J, Guillén A, Castro J. Ethanol consumption impairs regulation of fatty acid metabolism by decreasing the activity of AMP-activated protein kinase in rat liver. Biochimie. 2008;90:460-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 97] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 55. | Denechaud PD, Girard J, Postic C. Carbohydrate responsive element binding protein and lipid homeostasis. Curr Opin Lipidol. 2008;19:301-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 56. | Saltiel AR, Kahn CR. Insulin signalling and the regulation of glucose and lipid metabolism. Nature. 2001;414:799-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3583] [Cited by in RCA: 3653] [Article Influence: 152.2] [Reference Citation Analysis (0)] |
| 57. | Rui L. Energy metabolism in the liver. Compr Physiol. 2014;4:177-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 951] [Cited by in RCA: 1428] [Article Influence: 129.8] [Reference Citation Analysis (0)] |
| 58. | Wolfrum C, Stoffel M. Coactivation of Foxa2 through Pgc-1beta promotes liver fatty acid oxidation and triglyceride/VLDL secretion. Cell Metab. 2006;3:99-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 140] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 59. | Brown MS, Goldstein JL. Selective versus total insulin resistance: a pathogenic paradox. Cell Metab. 2008;7:95-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 694] [Cited by in RCA: 743] [Article Influence: 43.7] [Reference Citation Analysis (0)] |
| 60. | Sparks JD, Sparks CE, Adeli K. Selective hepatic insulin resistance, VLDL overproduction, and hypertriglyceridemia. Arterioscler Thromb Vasc Biol. 2012;32:2104-2112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 165] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 61. | Wang Y, Tran K, Yao Z. The activity of microsomal triglyceride transfer protein is essential for accumulation of triglyceride within microsomes in McA-RH7777 cells. A unified model for the assembly of very low density lipoproteins. J Biol Chem. 1999;274:27793-27800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 107] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 62. | Kamagate A, Qu S, Perdomo G, Su D, Kim DH, Slusher S, Meseck M, Dong HH. FoxO1 mediates insulin-dependent regulation of hepatic VLDL production in mice. J Clin Invest. 2008;118:2347-2364. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 137] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 63. | Mirandola S, Bowman D, Hussain MM, Alberti A. Hepatic steatosis in hepatitis C is a storage disease due to HCV interaction with microsomal triglyceride transfer protein (MTP). Nutr Metab (Lond). 2010;7:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 44] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 64. | Huang J, Iqbal J, Saha PK, Liu J, Chan L, Hussain MM, Moore DD, Wang L. Molecular characterization of the role of orphan receptor small heterodimer partner in development of fatty liver. Hepatology. 2007;46:147-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 123] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 65. | Wu C, Gilroy R, Taylor R, Olyaee M, Abdulkarim B, Forster J, O’Neil M, Damjanov I, Wan YJ. Alteration of hepatic nuclear receptor-mediated signaling pathways in hepatitis C virus patients with and without a history of alcohol drinking. Hepatology. 2011;54:1966-1974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 66. | Del Campo JA, Romero-Gómez M. Steatosis and insulin resistance in hepatitis C: a way out for the virus? World J Gastroenterol. 2009;15:5014-5019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 42] [Cited by in RCA: 48] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 67. | White DL, Ratziu V, El-Serag HB. Hepatitis C infection and risk of diabetes: a systematic review and meta-analysis. J Hepatol. 2008;49:831-844. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 325] [Cited by in RCA: 302] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 68. | Parvaiz F, Manzoor S, Iqbal J, McRae S, Javed F, Ahmed QL, Waris G. Hepatitis C virus nonstructural protein 5A favors upregulation of gluconeogenic and lipogenic gene expression leading towards insulin resistance: a metabolic syndrome. Arch Virol. 2014;159:1017-1025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 69. | Jung HJ, Kim YS, Kim SG, Lee YN, Jeong SW, Jang JY, Lee SH, Kim HS, Kim BS. The impact of pegylated interferon and ribavirin combination treatment on lipid metabolism and insulin resistance in chronic hepatitis C patients. Clin Mol Hepatol. 2014;20:38-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 70. | García-Mediavilla MV, Pisonero-Vaquero S, Lima-Cabello E, Benedicto I, Majano PL, Jorquera F, González-Gallego J, Sánchez-Campos S. Liver X receptor α-mediated regulation of lipogenesis by core and NS5A proteins contributes to HCV-induced liver steatosis and HCV replication. Lab Invest. 2012;92:1191-1202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 71. | Moucari R, Asselah T, Cazals-Hatem D, Voitot H, Boyer N, Ripault MP, Sobesky R, Martinot-Peignoux M, Maylin S, Nicolas-Chanoine MH. Insulin resistance in chronic hepatitis C: association with genotypes 1 and 4, serum HCV RNA level, and liver fibrosis. Gastroenterology. 2008;134:416-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 375] [Cited by in RCA: 360] [Article Influence: 21.2] [Reference Citation Analysis (1)] |
| 72. | Neuschwander-Tetri BA. Hepatitis C virus-induced insulin resistance: not all genotypes are the same. Gastroenterology. 2008;134:619-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 73. | Pazienza V, Clément S, Pugnale P, Conzelman S, Foti M, Mangia A, Negro F. The hepatitis C virus core protein of genotypes 3a and 1b downregulates insulin receptor substrate 1 through genotype-specific mechanisms. Hepatology. 2007;45:1164-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 177] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 74. | Fisher TL, White MF. Signaling pathways: the benefits of good communication. Curr Biol. 2004;14:R1005-R1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 75. | Laplante M, Sabatini DM. An emerging role of mTOR in lipid biosynthesis. Curr Biol. 2009;19:R1046-R1052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 430] [Cited by in RCA: 482] [Article Influence: 32.1] [Reference Citation Analysis (0)] |
| 76. | Hung CH, Lee CM, Chen CH, Hu TH, Jiang SR, Wang JH, Lu SN, Wang PW. Association of inflammatory and anti-inflammatory cytokines with insulin resistance in chronic hepatitis C. Liver Int. 2009;29:1086-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 77. | Maury E, Ehala-Aleksejev K, Guiot Y, Detry R, Vandenhooft A, Brichard SM. Adipokines oversecreted by omental adipose tissue in human obesity. Am J Physiol Endocrinol Metab. 2007;293:E656-E665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 149] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 78. | Snijder MB, Flyvbjerg A, Stehouwer CD, Frystyk J, Henry RM, Seidell JC, Heine RJ, Dekker JM. Relationship of adiposity with arterial stiffness as mediated by adiponectin in older men and women: the Hoorn Study. Eur J Endocrinol. 2009;160:387-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 79. | Wu H, Qi Q, Yu Z, Sun Q, Wang J, Franco OH, Sun L, Li H, Liu Y, Hu FB. Independent and opposite associations of trunk and leg fat depots with adipokines, inflammatory markers, and metabolic syndrome in middle-aged and older Chinese men and women. J Clin Endocrinol Metab. 2010;95:4389-4398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 80. | Ashour E, Samy N, Sayed M, Imam A. The relationship between serum adiponectin and steatosis in patients with chronic hepatitis C genotype-4. Clin Lab. 2010;56:103-110. [PubMed] |
| 81. | Cua IH, Hui JM, Bandara P, Kench JG, Farrell GC, McCaughan GW, George J. Insulin resistance and liver injury in hepatitis C is not associated with virus-specific changes in adipocytokines. Hepatology. 2007;46:66-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 107] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 82. | Tiftikci A, Atug O, Yilmaz Y, Eren F, Ozdemir FT, Yapali S, Ozdogan O, Celikel CA, Imeryuz N, Tozun N. Serum levels of adipokines in patients with chronic HCV infection: relationship with steatosis and fibrosis. Arch Med Res. 2009;40:294-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 83. | Yamauchi T, Kamon J, Minokoshi Y, Ito Y, Waki H, Uchida S, Yamashita S, Noda M, Kita S, Ueki K. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med. 2002;8:1288-1295. [PubMed] |
| 84. | Chen H, Zhang L, Li X, Li X, Sun G, Yuan X, Lei L, Liu J, Yin L, Deng Q. Adiponectin activates the AMPK signaling pathway to regulate lipid metabolism in bovine hepatocytes. J Steroid Biochem Mol Biol. 2013;138:445-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 61] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 85. | Matsunami T, Sato Y, Ariga S, Sato T, Shimomura T, Kashimura H, Hasegawa Y, Yukawa M. Regulation of synthesis and oxidation of fatty acids by adiponectin receptors (AdipoR1/R2) and insulin receptor substrate isoforms (IRS-1/-2) of the liver in a nonalcoholic steatohepatitis animal model. Metabolism. 2011;60:805-814. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 86. | Masaki T, Chiba S, Tatsukawa H, Yasuda T, Noguchi H, Seike M, Yoshimatsu H. Adiponectin protects LPS-induced liver injury through modulation of TNF-alpha in KK-Ay obese mice. Hepatology. 2004;40:177-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 312] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 87. | Yoon MJ, Lee GY, Chung JJ, Ahn YH, Hong SH, Kim JB. Adiponectin increases fatty acid oxidation in skeletal muscle cells by sequential activation of AMP-activated protein kinase, p38 mitogen-activated protein kinase, and peroxisome proliferator-activated receptor alpha. Diabetes. 2006;55:2562-2570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 395] [Cited by in RCA: 426] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 88. | Hong F, Radaeva S, Pan HN, Tian Z, Veech R, Gao B. Interleukin 6 alleviates hepatic steatosis and ischemia/reperfusion injury in mice with fatty liver disease. Hepatology. 2004;40:933-941. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 160] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 89. | Chen L, Yang G. PPARs Integrate the Mammalian Clock and Energy Metabolism. PPAR Res. 2014;2014:653017. [PubMed] |
| 90. | Ip E, Farrell GC, Robertson G, Hall P, Kirsch R, Leclercq I. Central role of PPARalpha-dependent hepatic lipid turnover in dietary steatohepatitis in mice. Hepatology. 2003;38:123-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 382] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 91. | Fischer M, You M, Matsumoto M, Crabb DW. Peroxisome proliferator-activated receptor alpha (PPARalpha) agonist treatment reverses PPARalpha dysfunction and abnormalities in hepatic lipid metabolism in ethanol-fed mice. J Biol Chem. 2003;278:27997-28004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 225] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 92. | Yamaguchi A, Tazuma S, Nishioka T, Ohishi W, Hyogo H, Nomura S, Chayama K. Hepatitis C virus core protein modulates fatty acid metabolism and thereby causes lipid accumulation in the liver. Dig Dis Sci. 2005;50:1361-1371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 81] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 93. | Singaravelu R, Chen R, Lyn RK, Jones DM, O’Hara S, Rouleau Y, Cheng J, Srinivasan P, Nasheri N, Russell RS. Hepatitis C virus induced up-regulation of microRNA-27: a novel mechanism for hepatic steatosis. Hepatology. 2014;59:98-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 88] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 94. | Wang J, Leclercq I, Brymora JM, Xu N, Ramezani-Moghadam M, London RM, Brigstock D, George J. Kupffer cells mediate leptin-induced liver fibrosis. Gastroenterology. 2009;137:713-723. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 158] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 95. | Myers MG, Cowley MA, Münzberg H. Mechanisms of leptin action and leptin resistance. Annu Rev Physiol. 2008;70:537-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 705] [Cited by in RCA: 757] [Article Influence: 44.5] [Reference Citation Analysis (0)] |
| 96. | Kukla M, Mazur W, Bułdak RJ, Zwirska-Korczala K. Potential role of leptin, adiponectin and three novel adipokines--visfatin, chemerin and vaspin--in chronic hepatitis. Mol Med. 2011;17:1397-1410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 62] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 97. | Giannini E, Ceppa P, Botta F, Mastracci L, Romagnoli P, Comino I, Pasini A, Risso D, Lantieri PB, Icardi G. Leptin has no role in determining severity of steatosis and fibrosis in patients with chronic hepatitis C. Am J Gastroenterol. 2000;95:3211-3217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 98. | Testa R, Franceschini R, Giannini E, Cataldi A, Botta F, Fasoli A, Tenerelli P, Rolandi E, Barreca T. Serum leptin levels in patients with viral chronic hepatitis or liver cirrhosis. J Hepatol. 2000;33:33-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 73] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 99. | Myers RP, Messous D, Poynard T, Imbert-Bismut F. Association between leptin, metabolic factors and liver histology in patients with chronic hepatitis C. Can J Gastroenterol. 2007;21:289-294. [PubMed] |
| 100. | Crespo J, Rivero M, Fábrega E, Cayón A, Amado JA, García-Unzeta MT, Pons-Romero F. Plasma leptin and TNF-alpha levels in chronic hepatitis C patients and their relationship to hepatic fibrosis. Dig Dis Sci. 2002;47:1604-1610. [PubMed] |
| 101. | Begriche K, Massart J, Robin MA, Bonnet F, Fromenty B. Mitochondrial adaptations and dysfunctions in nonalcoholic fatty liver disease. Hepatology. 2013;58:1497-1507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 375] [Cited by in RCA: 462] [Article Influence: 38.5] [Reference Citation Analysis (0)] |
| 102. | Sonnenberg GE, Krakower GR, Kissebah AH. A novel pathway to the manifestations of metabolic syndrome. Obes Res. 2004;12:180-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 153] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 103. | Sheikh MY, Choi J, Qadri I, Friedman JE, Sanyal AJ. Hepatitis C virus infection: molecular pathways to metabolic syndrome. Hepatology. 2008;47:2127-2133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 177] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 104. | Gong G, Waris G, Tanveer R, Siddiqui A. Human hepatitis C virus NS5A protein alters intracellular calcium levels, induces oxidative stress, and activates STAT-3 and NF-kappa B. Proc Natl Acad Sci USA. 2001;98:9599-9604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 485] [Cited by in RCA: 499] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 105. | Choi SH, Park KJ, Ahn BY, Jung G, Lai MM, Hwang SB. Hepatitis C virus nonstructural 5B protein regulates tumor necrosis factor alpha signaling through effects on cellular IkappaB kinase. Mol Cell Biol. 2006;26:3048-3059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 2.0] [Reference Citation Analysis (0)] |