Published online Mar 27, 2014. doi: 10.4254/wjh.v6.i3.137
Revised: November 5, 2013
Accepted: January 15, 2014
Published online: March 27, 2014
Processing time: 172 Days and 4.7 Hours
Liver innervation comprises sympathetic, parasympathetic and peptidergic nerve fibers, organized as either afferent or efferent nerves with different origins and roles. Their anatomy and physiology have been studied in the past 30 years, with different results published over time. Hepatocytes are the main cell population of the liver, making up almost 80% of the total liver volume. The interaction between hepatocytes and nerve fibers is accomplished through a wealth of neurotransmitters and signaling pathways. In this short review, we have taken the task of condensing the most important data related to how the nervous system interacts with the liver and especially with the hepatocyte population, how it influences their metabolism and functions, and how different receptors and transmitters are involved in this complex process.
Core tip: Liver innervation comprises sympathetic, parasympathetic and peptidergic nerve fibers, organized as either afferent or efferent nerves with different origins and roles. Their anatomy and physiology have been studied in the past 30 years, with different results published over time. Hepatocytes are the main cell population of the liver, making up almost 80% of the total liver volume. The interaction between hepatocytes and nerve fibers is accomplished through a wealth of neurotransmitters and signaling pathways.
- Citation: Streba LAM, Vere CC, Ionescu AG, Streba CT, Rogoveanu I. Role of intrahepatic innervation in regulating the activity of liver cells. World J Hepatol 2014; 6(3): 137-143
- URL: https://www.wjgnet.com/1948-5182/full/v6/i3/137.htm
- DOI: https://dx.doi.org/10.4254/wjh.v6.i3.137
Innervation of the liver comprises efferent and afferent nerves containing sympathetic, parasympathetic and peptidergic fibers. Sympathetic nerve fibers derive from splanchnic nerves and the parasympathetic counterparts have a vagal origin. Fibers derived from splanchnic, vagus and sometimes the phrenic nerves enter the liver through the hilum, together with the hepatic artery, portal vein and bile duct. Some nerve fibers do not accompany hepatic vessels and enter the liver via the small omentum or the hepatic vein. Sympathetic and parasympathetic nerves form two separate plexus but communicate with each other: the anterior plexus placed around the hepatic artery, consisting of nerve fibers with their origin in the celiac ganglion and posterior vagus nerve, and the posterior plexus located around the portal vein and bile duct, formed by fibers from the celiac ganglion and the right vagus[1]. Nervous fibers which are distributed to the hepatic parenchyma derive from a corresponding nervous plexus and their intrahepatic distribution differ according to species[2,3].
In the human liver, nerve endings are located in the hepatic lobules[4], which consists of hepatocytes and non-parenchymal cells. Unlike hepatocytes, which occupy almost 80% of liver volume and have numerous functions, non-parenchymal liver cells occupy only 6.5% of the liver, although representing 40% of total liver cells[5].
Hepatocytes are arranged as cellular cords with a radial disposition that converges towards the centrilobular vein, being separated by sinusoidal capillaries. Between hepatocyte cell cords and sinusoid capillaries there is an interstitial space, a perisinusoidal called a Disse space. This space is formed by a fine network of reticulin fibers, a support for the sinusoids, non myelinated nerve fibers and mesenchymal type cells[6]. Non-parenchymal cells are located in the liver sinusoidal compartment. The hepatic sinusoidal wall consists of three cell types: sinusoidal endothelial cells (SECs), Kupffer cells (KCs) and hepatic stellate cells (HSCs)[5]. Most nerve endings from intralobular spaces are located in Disse spaces[4,7-12], where they make close contact with HSCs, SECs and hepatocytes[7,8,10].
Hepatocytes serve multiple functions, such as synthesis, storage, metabolism and transformation of carbohydrates, amino acids, proteins, lipids, vitamins and detoxification, conjugation and excretion of exo- and endo-genous substances. During liver regeneration, hepatocytes initiate cell proliferation, maintain metabolic function of the liver, secrete interleukin-6 (IL-6), proteases, protease inhibitors and hepatocyte growth factor[13].
The liver receives both sympathetic and parasympathetic nerve fibers; however, the innervation that hepatocytes receive varies by species. Thus, in the cat, rabbit, guinea pig liver as well as primate liver, it appears that nerve endings are connected to all hepatocytes, unlike rats and mice in which only hepatic cells in the portal region appear to be in contact with intrahepatic nerve endings[14].
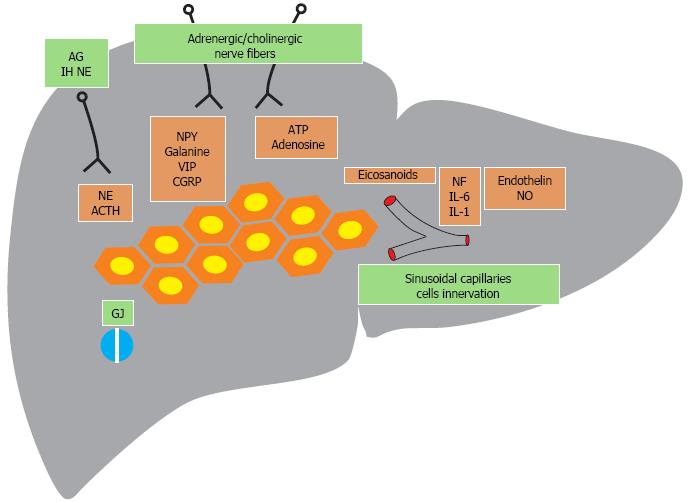
Nerve fiber communication with hepatocytes can be accomplished by several mechanisms (Figure 1): (1) hepatocyte direct innervation mediated by norepinephrine and acetylcholine, neuropeptides [neuropeptide Y, galanine (NPY), vasoactive intestinal peptide (VIP), calcitonin gene-related peptide (CGRP), etc.], purines [adenosine triphosphate or adenosine (ATP)]; (2) signals intercellular transmission using gap type junctions; and (3) sinusoidal capillary cells innervation which interact with hepatocytes through eicosanoids (prostaglandins, leukotrienes), cytokines (necrotic factor, IL-6, IL-1) and other chemical mediators (endothelin, nitric oxide).
Nerve transmission to hepatocytes is achieved through neurotransmitters such as norepinephrine and acetylcholine, neuropeptides, such as NPY, galanine, VIP and CGRP, or purine derivatives as ATP and adenosine.
The liver is stimulated by norepinephrine and epinephrine released from intrahepatic nerve endings but also derived through the blood from the adrenal glands. Catecholamines act in the liver on α1-, α2- and β2-adrenergic receptors[15-17]. Norepinephrine is removed from the site of action by intrahepatic nerve ending uptake, being degraded by liver cells and diffused through the vascular bed[1].
Experiments on rat liver have shown that stimulation of the autonomic nerve plexus around the hepatic artery and portal vein causes increased production of glucose and lactate[1], urate and allantoin formation[17], decreased ketogenesis[18], increased ureogenesis and ammonia uptake[19], as well as increased oxygen utilization[20,21]. Also, hepatic nerve stimulation leads to decreased[16,17,20,22,23] and redistributed intrahepatic flow[21], as well as raised noradrenaline levels in the hepatic vein[15-18]. All these effects of hepatic nerves are only possible in the presence of extracellular calcium[22,24].
NPY, galanin, SP, CGRP, VIP and purine derivatives (ATP, adenosine) act as neurotransmitters, both in adrenergic and cholinergic nerve fibers, as well as in the related hepatic nerves. These neurotransmitters are released locally and are involved in regulating hepatic microcirculation. NPY and ATP act as vasoconstrictors, while VIP, CGRP, SP and adenosine produce vasodilation[14].
Some neurotransmitters also have a metabolic function. Thus, sympathetic hepatic nerve stimulation causes the release of noradrenaline but also of galanin[25], suggesting that galanin potentiates the action of norepinephrine to stimulate hepatic glucose production under stress[14].
Yamamoto et al[26] revealed the metabolic activity of ATP, which potentiates the action of hepatic sympathetic nerves suppression action on the formation of ketone bodies in the liver, the effect probably being due to ATP interaction with norepinephrine[14].
Intrahepatic innervation varies by species. In some species, most hepatocytes are not directly innervated but there is an indirect mechanism for transmitting nervous inflow. One such mechanism is the intercellular communication carried out between adjacent hepatocytes via specific channels known as gap type junctions (GJ), which allow the passage of ions and small molecules[14].
GJ density is different among species[27]. Thus, hepatic GJ are more numerous in rats and mice compared to rabbits and guinea pigs[14].
GJ are membrane channels that allow intercellular communication between neighboring cells. GJ consists of two hemichannels, one hemichannel belonging to each of the two adjacent cells. A hemichannel consists of six subunits or connexins. Connexin 32 (Cx32) is the major protein component expressed in murine hepatocytes. Cx32 plays an essential role in signal propagation induced by the norepinephrine released from sympathetic nerve endings in hepatocytes[28].
GJ ensure the transmission of information to neighboring cells, achieving functional integration of hepatocytes, and thus functioning as a body and not as a mere cluster of cells[28].
GJ have an important role in transmitting nervous impulses from sympathetic nerve endings in parenchymal and non-parenchymal cells of the liver, in some species of mammals. There is an inverse relationship between sympathetic nerve fiber density and number of intrahepatic GJ[14]. Research on rat liver, which contains numerous GJ[15,29], showed that sympathetic nerves innervate only a part of parenchymal and non-parenchymal cells of the periportal area[15]. Metabolic and hemodynamic effects of hepatic sympathetic nerves are achieved by α1 receptor stimulation[17,23,30], are abolished by prostanoids synthesis inhibitors, and are mimicked by prostaglandins but not by thromboxanes[31]. Norepinephrine, as well as F2α prostaglandin, stimulates the release of glucose from isolated hepatocytes with increased inositol 1,4,5-triphosphate and glycogen formation[32].
Sending signals through GJ is involved in the metabolic effects of sympathetic nerves[14]. The norepinephrine released from sympathetic nerve endings binds to α1 receptors of parenchymal and non-parenchymal cells. The release of glucose from the proximal parenchymal cells from the periportal region under the action of norepinephrine initiates a signal which propagates through GJ to distal parenchymal cells, which in turn releases glucose. Norepinephrine stimulates contraction of proximal non-parenchymal cells (sphincters), reducing the flow in the sinusoid capillaries. It also causes the release of prostaglandins (PG) into the Disse space. PG, in turn, bind to prostanoid receptors of parenchymal and non-parenchymal cells. PG released from non-parenchymal cells are rapidly degraded in the liver[32] and so do not reach the distal cells. PG increase glucose release in the periportal parenchymal cells and perhaps initiate a signal that propagates to the other cells by GJ. PG stimulate contraction of proximal non-parenchymal cells, thus reducing the flow to sinusoid capillaries[15,33].
Sinusoidal capillaries are located between the cell cords and have a 9 to 12 micrometers diameter. The sinusoidal capillary wall is discontinuous and is formed out of a basement membrane and a capillary endothelium. Sinusoidal endothelium consists of flattened endothelial cells and phagocytic Kupffer cells in a ratio of 5/3. In addition to these two main types of cells in the sinusoid capillaries, there are also stellate cells and lymphocytes[6]. Capillary walls have an important role in the regulation of the sinusoidal microcirculation[34,35].
Endothelial cells are flat and have elongated, hyperchromic nuclei and reduced cytoplasm. Junctional complexes are lacking. There are very small spaces of 0.5 micrometers between the cells. The apical membrane of the endothelial cells has transcytoplasmic fenestrations which are small holes arranged in nests with a diameter of 100 nm, with a role in controlling cholesterol, lipoprotein and vitamin A metabolism. In the cytoplasm of endothelial cells, a small number of cell organelles and pinocytosis vesicles were revealed. Endothelial cells produce prostaglandins, endothelin, IL-1 and IL-2[6,13].
Hepatic stellate cells (Ito cells, lipocytes) are located in the Disse space in small niches, among hepatocytes. They are in contact with the liver cells and through their microvilli and cytoplasmic processes have contacts with endothelial cell microvilli[6]. Well developed organelles and lipid droplets were found in the HSC cytoplasm. HSCs store vitamin A, containing factors related to retinoid acid and retinol, and produce extracellular matrix. Under normal conditions, HSCs have a deposit function, while in pathological ones it transforms into myofibroblastic type cells[6,13,36,37].
Research has shown that the distribution of intrahepatic innervation varies by species[29,38-43]. Guinea pig, cat and tupaia have an intralobular innervation similar to the human one[11,12,29,40], in contrast to mice and rats where it differs from the human[39,41-43].
In human liver, nerve endings are located in the Disse space[4,11,12], closely connected to hepatocytes and non-parenchymal cells, particularly HSCs[7,8,10,11].
Non-parenchymal cells are the only ones that can synthesize eicosanoids (prostaglandins, thromboxanes and leukotrienes) from arachidonic acid released from phospholipids by the action of phospholipase A2 and converted to prostaglandins and thromboxanes via the cyclooxygenase path and leukotrienes via the lipoxygenase path[1]. Experiments on perfused rat liver have shown that the synthesis and secretion of prostanoids in non-parenchymal liver cells is influenced by a number of physiological stimuli, pathological and chemical. These stimuli also determine an increased release of glucose and lactate, as well as increased vascular resistance in the liver[1]. Of these stimuli, the most important are: extracellular nucleotides[44], nucleosides[45], zymosan[46,47], endotoxins[48], aggregates of immunoglobulins[49], anaphylatoxins[50,51], phorbol esters and calcium ionophores[44]. Norepinephrine and/or other chemical mediators released from nerve endings can stimulate the formation of prostanoids in non-parenchymal liver cells. Prostanoids, in turn, can modulate hepatocyte metabolism[1].
Of eicosanoids, only PG, without thromboxanes and leukotrienes, play a role in the events triggered by nerve stimulation[1].
PG participation in the chain of events initiated by nerve stimuli in the liver depends on hepatocellular receptors for PG. Research conducted so far confirms the existence of these receptors[1].
HSCs are indirectly involved in nerve fiber communication with hepatocytes, through PG. Noradrenaline may lead, by means of α1-adrenergic receptors, to increased synthesis of PG in the HSC, and PG, in turn, stimulate glycogenolysis in the hepatocytes. Unlike KC, producing predominantly PGD2, HSC secret PGF 2 alpha released in increased amounts compared with PGD2, as a result of sympathetic stimulation[52].
Intrahepatic nerve fiber terminations, often containing vesicles which contain neurotransmitters like substance P (SP) and vasoactive intestinal peptide (VIP), are closely related to HSC. It is considered that HSC that surround CECS, forming sinusoidal capillary walls, have a role in the contraction and relaxation of sinusoidal walls, thus intervening in the regulation of the sinusoidal microcirculation[4].
HSC contraction is stimulated by a number of substances such as endothelin-1 (ET-1), angiotensin II, norepinephrine, prostaglandin F2, thromboxane A2 and thrombin. In contrast, vasoactive substances such as acetylcholine, VIP, nitric oxide (NO), carbon monoxide, prostaglandin E2 and adrenomedulin produce HSC relaxation[4].
ET-1 produces contraction of HSCs through ET receptor stimulation on autocrine or paracrine pathways. HSC contraction appears to be related to the increase of intracellular Ca2 + and inositol phosphate. For the sinusoid microcirculation control role of the HSC, the presence in the HSC cytoplasm of α smooth muscle actin, which is a contractile protein, also stands, such that the contraction of the HSC can be compared with that of smooth muscle cells in the vessel wall structure. On the other hand, prostaglandin E2, adrenomedulin and other vasoactive substances determine HSC relaxation by increasing intracellular cAMP. In addition, HSC produces NO and inhibits contractility by an autocrine mechanism linked to NO[8].
Of the mentioned vasoactive substances, ET-1 and NO have an important role in the regulation of sinusoidal microcirculation. ET is a peptide consisting of 21 amino acids with a strong vasoconstrictive effect on the smooth muscle fibers. It has three isoforms, ET-1, ET-2 and ET-3[4,53].
Two receptors have been identified for ET: ETA and ETB, both belonging to the superfamily of G-protein-coupled receptors[54]. ETA receptor has a higher affinity for ET-1 and ET-2 than for ET-3, while the ETB receptor has a similar affinity for all three isoforms of ET. ETA receptors stimulation increases intracellular cAMP levels, whereas ETB receptor stimulation leads to inhibition of the adenylate cyclase system[55]. Also, ETB receptor stimulation activates Ca2+-dependent NOS[56]. Douglas et al[57] described two ETB receptor subtypes: ETB1 and ETB2. Stimulation of ETA and ETB1 receptors causes contraction of smooth muscle fibers, while ETB2 receptor stimulation causes dilation by increased synthesis of NO[58].
ET-1 receptors from intralobular spaces predominate in the juxtaportal region. About 35% of ET-1 receptors are located in the HSCs, a smaller number are located in the CECS and KCs[59]. Both ETA and ETB receptors are found in the liver. All cells of the sinusoidal capillary walls have ETB receptors but only HSCs have ETA receptors. Mallat et al[60] have identified 20% ETA receptors and 80% ETB receptors on activated HSCs.
NO is synthesized from L-arginine by the NO synthetase path (NOS). Three NOS isoforms have been identified: two are calcium-dependent, one produced by the neurons (nNOS or NOS I) and other by the vascular endothelial cells (eNOS or NOS III), and one calcium-independent isoform, cytokine-induced (iNOS or NOS II). Rat HSCs shrink under ET-1 or SP action and relax, causing vasodilatation, under NO action produced by HSC under the influence of IL-1[4,61].
In normal liver, sinusoidal contraction is inhibited by ETA receptor antagonists[62,63] but, according to some authors, not by ETB receptors antagonists[4,62-64]. Other researchers believe, however, that ETB receptor stimulation results in constriction of sinusoid capillaries[65]. It is possible that this discrepancy between the results obtained by different authors is due to coupling ETB receptors with NOS, which masks the vasoconstrictor effect[63,66].
The various subtypes of endothelin have different effects on hepatic microcirculation. The relationship between NO and endothelin is extremely important in the control of vascular tone[63].
Liu et al[58] have shown that ET-1 binding to ETB receptor leads to eNOS activation on the Akt phosphorylation path, thus reducing the phosphorylation of eNOS and NO synthesis. The same researchers highlighted the crucial role of βγ subunits of the G protein in triggering endothelin/NO reactions. The stimuli which regulate the ET expression and vascular sensitivity to ET also adjust the NOS and heme oxygenase-1 activity. Both enzymes catalyze the production of substances which, by guanylate cyclase activation, produce vasodilation[67].
Liver innervation is one of the most complex control systems in the human body; therefore, a better understanding of its inner workings is of paramount importance for developing future therapies and procedures for ameliorating the metabolic function of the liver. Being able to manipulate nerve impulses and synaptic mediators can possibly allow direct control over the functions of hepatocytes. Direct acting agents with excellent control over specific liver functions could become a reality, with direct implications for drug therapy, surgery or liver transplant.
P- Reviewers: Boin IFSF, Kitamura T, Serra PA S- Editor: Gou SX L- Editor: Roemmele A E- Editor: Zhang DN
| 1. | Gardemann A, Püschel GP, Jungermann K. Nervous control of liver metabolism and hemodynamics. Eur J Biochem. 1992;207:399-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 2. | Shimazu T, Usami M. Further studies on the mechanism of phosphorylase activation in rabbit liver in response to splanchnic nerve stimulation. J Physiol. 1982;329:231-242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 97] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 3. | Reilly FD, McCuskey PA, McCuskey RS. Intrahepatic distribution of nerves in the rat. Anat Rec. 1978;191:55-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 94] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Ueno T, Bioulac-Sage P, Balabaud C, Rosenbaum J. Innervation of the sinusoidal wall: regulation of the sinusoidal diameter. Anat Rec A Discov Mol Cell Evol Biol. 2004;280:868-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 44] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 5. | Kmieć Z. Cooperation of liver cells in health and disease. Adv Anat Embryol Cell Biol. 2001;161:III-XIII, 1-151. [PubMed] |
| 6. | Crişan M, Mureşan V. Anatomia microscopică a ficatului. Grigorescu M, editor. Bucuresti: Naţională M 2004; 24-34. |
| 7. | Ueno T, Inuzuka S, Torimura T, Sakata R, Sakamoto M, Gondo K, Aoki T, Tanikawa K, Tsutsumi V. Distribution of substance P and vasoactive intestinal peptide in the human liver: light and electron immunoperoxidase methods of observation. Am J Gastroenterol. 1991;86:1633-1637. [PubMed] |
| 8. | Ueno T, Tanikawa K. Intralobular innervation and lipocyte contractility in the liver. Nutrition. 1997;13:141-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 9. | Ito T, Shibasaki S. Electron microscopic study on the hepatic sinusoidal wall and the fat-storing cells in the normal human liver. Arch Histol Jpn. 1968;29:137-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 72] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 10. | Bioulac-Sage P, Lafon ME, Saric J, Balabaud C. Nerves and perisinusoidal cells in human liver. J Hepatol. 1990;10:105-112. [PubMed] |
| 11. | Akiyoshi H, Gonda T, Terada T. A comparative histochemical and immunohistochemical study of aminergic, cholinergic and peptidergic innervation in rat, hamster, guinea pig, dog and human livers. Liver. 1998;18:352-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 60] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 12. | Peinado MA, del Moral ML, Jiménez A, Rodrigo J, Esteban FJ. The nitrergic autonomic innervation of the liver. Auton Neurosci. 2002;99:67-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | Zheng ZY, Weng SY, Yu Y. Signal molecule-mediated hepatic cell communication during liver regeneration. World J Gastroenterol. 2009;15:5776-5783. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 22] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 14. | Shimazu T. Progress and perspective of neuro-hepatology. Shimazu T, editor. Eastleigh: John Libbey & Company Ltd 1996; 3-13. |
| 15. | Seseke FG, Gardemann A, Jungermann K. Signal propagation via gap junctions, a key step in the regulation of liver metabolism by the sympathetic hepatic nerves. FEBS Lett. 1992;301:265-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 16. | Gardemann A, Strulik H, Jungermann K. Different accessibility from the artery and the portal vein of alpha- and beta-receptors involved in the sympathetic nerve action on glycogenolysis and hemodynamics in perfused rat liver. Biol Chem Hoppe Seyler. 1989;370:47-54. [PubMed] |
| 17. | Ulken V, Püschel GP, Jungermann K. Increase in glucose and lactate output and perfusion resistance by stimulation of hepatic nerves in isolated perfused rat liver: role of alpha 1-, alpha 2-, beta 1- and beta 2-receptors. Biol Chem Hoppe Seyler. 1991;372:401-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 18. | Beuers U, Beckh K, Jungermann K. Control of ketogenesis in the perfused rat liver by the sympathetic innervation. Eur J Biochem. 1986;158:19-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 19. | Ballé C, Jungermann K. Control of urea production, glutamine release and ammonia uptake in the perfused rat liver by the sympathetic innervation. Eur J Biochem. 1986;158:13-18. [PubMed] |
| 20. | Beckh K, Hartmann H, Jungermann K, Scholz R. Regulation of oxygen consumption in perfused rat liver: decrease by alpha-sympathetic nerve stimulation and increase by the alpha-agonist phenylephrine. Pflugers Arch. 1984;401:104-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 36] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 21. | Ji S, Beckh K, Jungermann K. Regulation of oxygen consumption and microcirculation by alpha-sympathetic nerves in isolated perfused rat liver. FEBS Lett. 1984;167:117-122. [PubMed] |
| 22. | Hartmann H, Beckh K, Jungermann K. Direct control of glycogen metabolism in the perfused rat liver by the sympathetic innervation. Eur J Biochem. 1982;123:521-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 138] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 23. | Gardemann A, Strulik H, Jungermann K. Nervous control of glycogenolysis and blood flow in arterially and portally perfused liver. Am J Physiol. 1987;253:E238-E245. [PubMed] |
| 24. | Athari A, Jungermann K. Role of extracellular calcium in the metabolic and hemodynamic actions of sympathetic nerve stimulation, noradrenaline and prostaglandin F2 alpha in perfused rat liver. Differential inhibition by nifedipine and verapamil. Biochem Int. 1990;20:13-23. [PubMed] |
| 25. | Kowalyk S, Veith R, Boyle M, Taborsky GJ. Liver releases galanin during sympathetic nerve stimulation. Am J Physiol. 1992;262:E671-E678. [PubMed] |
| 26. | Yamamoto T, Iwai M, Kimura S, Shimazu T. The mechanism of action of hepatic sympathetic nerves on ketone-body output from perfused rat liver. The effect of the interaction of noradrenaline with ATP on the release of beta-hydroxybutyrate. Eur J Biochem. 1995;234:466-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 27. | Shimazu T, Iwai M; The hypothalamus, sympathetic nerves and regulation of peripheral glucose metabolism. In: New functional aspects of the suprachiasmatic nucleus of the hypothalamus. Nakagawa H, Oomura Y, Nagai K, editors. London: John Libbey: 93-103. . |
| 28. | Nelles E, Bützler C, Jung D, Temme A, Gabriel HD, Dahl U, Traub O, Stümpel F, Jungermann K, Zielasek J. Defective propagation of signals generated by sympathetic nerve stimulation in the liver of connexin32-deficient mice. Proc Natl Acad Sci USA. 1996;93:9565-9570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 308] [Cited by in RCA: 297] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 29. | Forssmann WG, Ito S. Hepatocyte innervation in primates. J Cell Biol. 1977;74:299-313. [PubMed] |
| 30. | Jungermann K. [Regulation of liver functions by autonomic hepatic nerves]. Naturwissenschaften. 1989;76:547-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 31. | Iwai M, Jungermann K. Possible involvement of eicosanoids in the actions of sympathetic hepatic nerves on carbohydrate metabolism and hemodynamics in perfused rat liver. FEBS Lett. 1987;221:155-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 44] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 32. | Athari A, Jungermann K. Direct activation by prostaglandin F2 alpha but not thromboxane A2 of glycogenolysis via an increase in inositol 1,4,5-trisphosphate in rat hepatocytes. Biochem Biophys Res Commun. 1989;163:1235-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 33. | Jungermann K, Katz N. Functional specialization of different hepatocyte populations. Physiol Rev. 1989;69:708-764. [PubMed] |
| 34. | Ueno T, Sata M, Sakata R, Torimura T, Sakamoto M, Sugawara H, Tanikawa K. Hepatic stellate cells and intralobular innervation in human liver cirrhosis. Hum Pathol. 1997;28:953-959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 35. | Geerts A. History, heterogeneity, developmental biology, and functions of quiescent hepatic stellate cells. Semin Liver Dis. 2001;21:311-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 548] [Cited by in RCA: 562] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 36. | Eng FJ, Friedman SL. Fibrogenesis I. New insights into hepatic stellate cell activation: the simple becomes complex. Am J Physiol Gastrointest Liver Physiol. 2000;279:G7-G11. [PubMed] |
| 37. | MacSween RNM, Scothorne RJ. Developmental anatomy and normal structure. Pathology of the liver. 4th ed. London: Churchill Livingston 2001; 1-66. |
| 38. | Barja F, Mathison R. Adrenergic and peptidergic (substance P and vasoactive intestinal polypeptide) innervation of the rat portal vein. Blood Vessels. 1982;19:263-272. [PubMed] |
| 39. | Barja F, Mathison R. Sensory innervation of the rat portal vein and the hepatic artery. J Auton Nerv Syst. 1984;10:117-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 40. | Ohata M. Electron microscope study on the innervation of guinea pig liver--proposal of sensory nerve terminals in the hepatic parenchyme. Arch Histol Jpn. 1984;47:149-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 41. | Sasaki Y, Kamada T, Hayashi N, Sato N, Kasahara A, Fusamoto H, Shiosaka S, Tohyama M, Shiotani Y. Immunohistochemical distribution of glucagon, substance P and vasoactive intestinal polypeptide in hepatic vasculature of the rat. Hepatology. 1984;4:1184-1189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 42. | Ito Y, Magari S, Sakanaka M. Immunoelectron-microscopic localization of peptidergic nerve fibers around lymphatic capillaries in the rat liver. Arch Histol Cytol. 1990;53 Suppl:199-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 43. | Fehér E, Fodor M, Fehér J. Ultrastructural localization of somatostatin- and substance P-immunoreactive nerve fibers in the feline liver. Gastroenterology. 1992;102:287-294. [PubMed] |
| 44. | Tran-Thi TA, Häussinger D, Gyufko K, Decker K. Stimulation of prostaglandin release by Ca2+-mobilizing agents from the perfused rat liver. A comparative study on the action of ATP, UTP, phenylephrine, vasopressin and nerve stimulation. Biol Chem Hoppe Seyler. 1988;369:65-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 45. | vom Dahl S, Wettstein M, Gerok W, Häussinger D. Stimulation of release of prostaglandin D2 and thromboxane B2 from perfused rat liver by extracellular adenosine. Biochem J. 1990;270:39-44. [PubMed] |
| 46. | Dieter P, Altin JG, Decker K, Bygrave FL. Possible involvement of eicosanoids in the zymosan and arachidonic-acid-induced oxygen uptake, glycogenolysis and Ca2+ mobilization in the perfused rat liver. Eur J Biochem. 1987;165:455-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 47. | Dieter P, Altin JG, Bygrave FL. Possible involvement of prostaglandins in vasoconstriction induced by zymosan and arachidonic acid in the perfused rat liver. FEBS Lett. 1987;213:174-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 48. | Casteleijn E, Kuiper J, Van Rooij HC, Kamps JA, Koster JF, Van Berkel TJ. Endotoxin stimulates glycogenolysis in the liver by means of intercellular communication. J Biol Chem. 1988;263:6953-6955. [PubMed] |
| 49. | Buxton DB, Fisher RA, Briseno DL, Hanahan DJ, Olson MS. Glycogenolytic and haemodynamic responses to heat-aggregated immunoglobulin G and prostaglandin E2 in the perfused rat liver. Biochem J. 1987;243:493-498. [PubMed] |
| 50. | Püschel GP, Oppermann M, Muschol W, Götze O, Jungermann K. Increase of glucose and lactate output and decrease of flow by human anaphylatoxin C3a but not C5a in perfused rat liver. FEBS Lett. 1989;243:83-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 51. | Muschol W, Püschel GP, Hülsmann M, Jungermann K. Eicosanoid-mediated increase in glucose and lactate output as well as decrease and redistribution of flow by complement-activated rat serum in perfused rat liver. Eur J Biochem. 1991;196:525-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 52. | Puschel GP, Neuschafer-Rube F, de Vries C, Hanecke K, Nolte A, Kirchner C, Schroder A, Schestag F, Athari A, Jungermann K. Characterization and molecular cloning of rat hepatocyte prostaglandin receptors possibly involved in the nerve stimulation-dependent increase in hepatic glucose output. Shimazu T, editor. Eastleigh: John Libbey & Company Ltd 1996; 87-94. |
| 53. | Yanagisawa M, Kurihara H, Kimura S, Tomobe Y, Kobayashi M, Mitsui Y, Yazaki Y, Goto K, Masaki T. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature. 1988;332:411-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8220] [Cited by in RCA: 7981] [Article Influence: 215.7] [Reference Citation Analysis (0)] |
| 54. | Huggins JP, Pelton JT, Miller RC. The structure and specificity of endothelin receptors: their importance in physiology and medicine. Pharmacol Ther. 1993;59:55-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 129] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 55. | Eguchi S, Hirata Y, Imai T, Marumo F. Endothelin receptor subtypes are coupled to adenylate cyclase via different guanyl nucleotide-binding proteins in vasculature. Endocrinology. 1993;132:524-529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 56. | Tsukahara H, Ende H, Magazine HI, Bahou WF, Goligorsky MS. Molecular and functional characterization of the non-isopeptide-selective ETB receptor in endothelial cells. Receptor coupling to nitric oxide synthase. J Biol Chem. 1994;269:21778-21785. [PubMed] |
| 57. | Douglas SA, Meek TD, Ohlstein EH. Novel receptor antagonists welcome a new era in endothelin biology. Trends Pharmacol Sci. 1994;15:313-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 54] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 58. | Liu S, Premont RT, Kontos CD, Huang J, Rockey DC. Endothelin-1 activates endothelial cell nitric-oxide synthase via heterotrimeric G-protein betagamma subunit signaling to protein jinase B/Akt. J Biol Chem. 2003;278:49929-49935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 116] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 59. | Gondo K, Ueno T, Sakamoto M, Sakisaka S, Sata M, Tanikawa K. The endothelin-1 binding site in rat liver tissue: light- and electron-microscopic autoradiographic studies. Gastroenterology. 1993;104:1745-1749. [PubMed] |
| 60. | Mallat A, Fouassier L, Préaux AM, Mavier P, Lotersztajn S. Antiproliferative effects of ET-1 in human liver Ito cells: an ETB- and a cyclic AMP-mediated pathway. J Cardiovasc Pharmacol. 1995;26 Suppl 3:S132-S134. [PubMed] |
| 61. | Sakamoto M, Uen T, Nakamura T, Hashimoto O, Sakata R, Kin M, Ogata R, Kawaguch T, Torimura T, Sata M. Estrogen upregulates nitric oxide synthase expression in cultured rat hepatic sinusoidal endothelial cells. J Hepatol. 2001;34:858-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 62. | Zhang JX, Bauer M, Clemens MG. Vessel- and target cell-specific actions of endothelin-1 and endothelin-3 in rat liver. Am J Physiol. 1995;269:G269-G277. [PubMed] |
| 63. | Clemens MG, Zhang JX. Regulation of sinusoidal perfusion: in vivo methodology and control by endothelins. Semin Liver Dis. 1999;19:383-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 73] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 64. | Wang HG, Shibamoto T, Miyahara T. Endothelin-1 selectively contracts portal vein through both ETA and ETB receptors in isolated rabbit liver. Am J Physiol. 1997;273:G1036-G1043. [PubMed] |
| 65. | Ito Y, Katori M, Majima M, Kakita A. Constriction of mouse hepatic venules and sinusoids by endothelins through ETB receptor subtype. Int J Microcirc Clin Exp. 1996;16:250-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 66. | Bauer M, Bauer I, Sonin NV, Kresge N, Baveja R, Yokoyama Y, Harding D, Zhang JX, Clemens MG. Functional significance of endothelin B receptors in mediating sinusoidal and extrasinusoidal effects of endothelins in the intact rat liver. Hepatology. 2000;31:937-947. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 77] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 67. | Suematsu M, Wakabayashi Y, Ishimura Y. Gaseous monoxides: a new class of microvascular regulator in the liver. Cardiovasc Res. 1996;32:679-686. [PubMed] |