Published online Aug 27, 2025. doi: 10.4254/wjh.v17.i8.108443
Revised: May 26, 2025
Accepted: July 14, 2025
Published online: August 27, 2025
Processing time: 135 Days and 4.1 Hours
Adrenal ectopia is a rare condition in which adrenal tissue is found in an abn
In this case report, we present a 53-year-old male with ectopic adrenal tissue in the liver, mimicking a potential hepatic malignancy. Based on computed tomo
This case report underscores the importance of considering adrenal ectopia in the differential diagnosis of liver lesions, especially when imaging findings suggest malignancy.
Core Tip: This rare case describes ectopic adrenal tissue in the liver mimicking malignancy in a 53-year-old male with adrenal adenoma. Imaging revealed a hepatic lesion prompting resection; histopathology confirmed adrenal ectopia, excluding malignancy. It underscores adrenal ectopia as a critical differential in liver masses, particularly with adrenal disorders, to prevent misdiagnosis. Multidisciplinary evaluation integrating imaging and histology is essential. The case enhances understanding of adrenal ectopia’s pathophysiology and highlights its consideration in adrenal-hepatic pathologies, optimizing diagnostic accuracy and treatment outcomes.
- Citation: Qin MQ, Zhao YP, Xie JP. Ectopic adrenal gland in the liver leading to a misdiagnosis of hepatocellular carcinoma: A case report. World J Hepatol 2025; 17(8): 108443
- URL: https://www.wjgnet.com/1948-5182/full/v17/i8/108443.htm
- DOI: https://dx.doi.org/10.4254/wjh.v17.i8.108443
Adrenal ectopia is a rare condition in which adrenal tissue is found in an abnormal location, often posing diagnostic challenges. Although generally considered benign, it can present as hepatic or other organ lesions, mimicking malignant tumors. The exact prevalence of adrenal ectopia is difficult to determine; however, studies suggest that it occurs in up to 50% of pediatric autopsies[1], with fewer cases reported in adults. The condition is often discovered incidentally during imaging studies conducted for unrelated diseases. One study found ectopic adrenal tissue in 13 out of 1120 patients who underwent surgical exploration of the groin[2].
The clinical significance of adrenal ectopia lies in its potential for misdiagnosis, particularly when located in organs such as the liver, lungs, or retroperitoneum, where malignant lesions are common. In the liver, ectopic adrenal tissue can closely resemble hepatocellular carcinoma (HCC) or metastatic disease, potentially leading to unnecessary aggressive treatments such as surgery or chemotherapy. Consequently, a high index of suspicion is essential to avoid misdiagnosis and ensure appropriate management. This case report underscores the importance of considering adrenal ectopia in the differential diagnosis of liver lesions, especially when imaging findings suggest malignancy.
A low echo mass in the left adrenal gland detected during a routine checkup three years ago.
A 53-year-old male was admitted to the hospital due to “a low echo mass in the left adrenal gland detected during a routine checkup three years ago”. The mass, approximately 1 cm × 1 cm in size, was initially diagnosed as an adenoma and was asymptomatic. The patient underwent regular annual follow-ups with no significant changes in lesion size.
His medical history included hyperlipidemia and hyperuricemia but no history of hypertension or hypokalemia.
Smoking and drinking for 20 years.
On admission, his physical examination revealed no notable findings, and the preliminary diagnosis remained adrenal adenoma.
Laboratory tests revealed an abnormal prothrombin time (40.38 mAU/mL), while routine blood tests, liver and kidney function tests, adrenal cortical function tests, and tumor markers such as alpha-fetoprotein were within normal ranges.
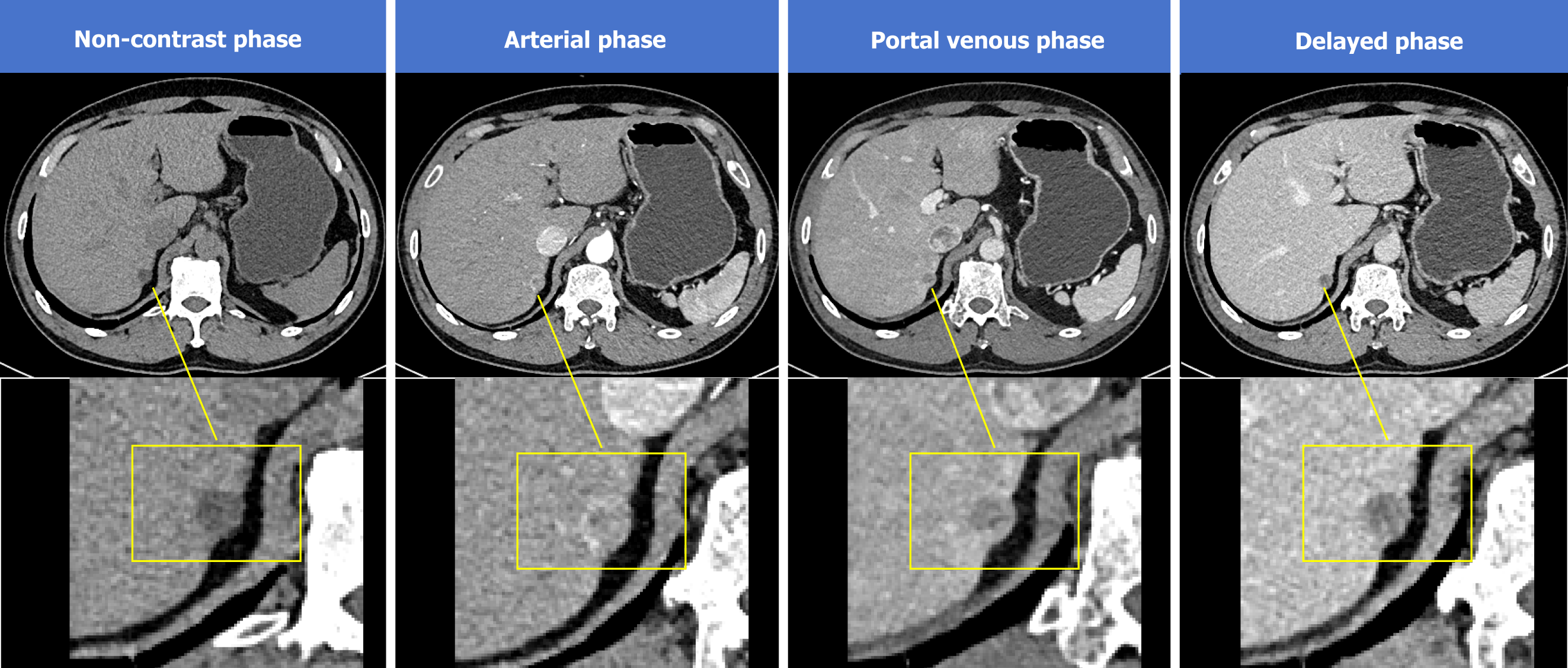
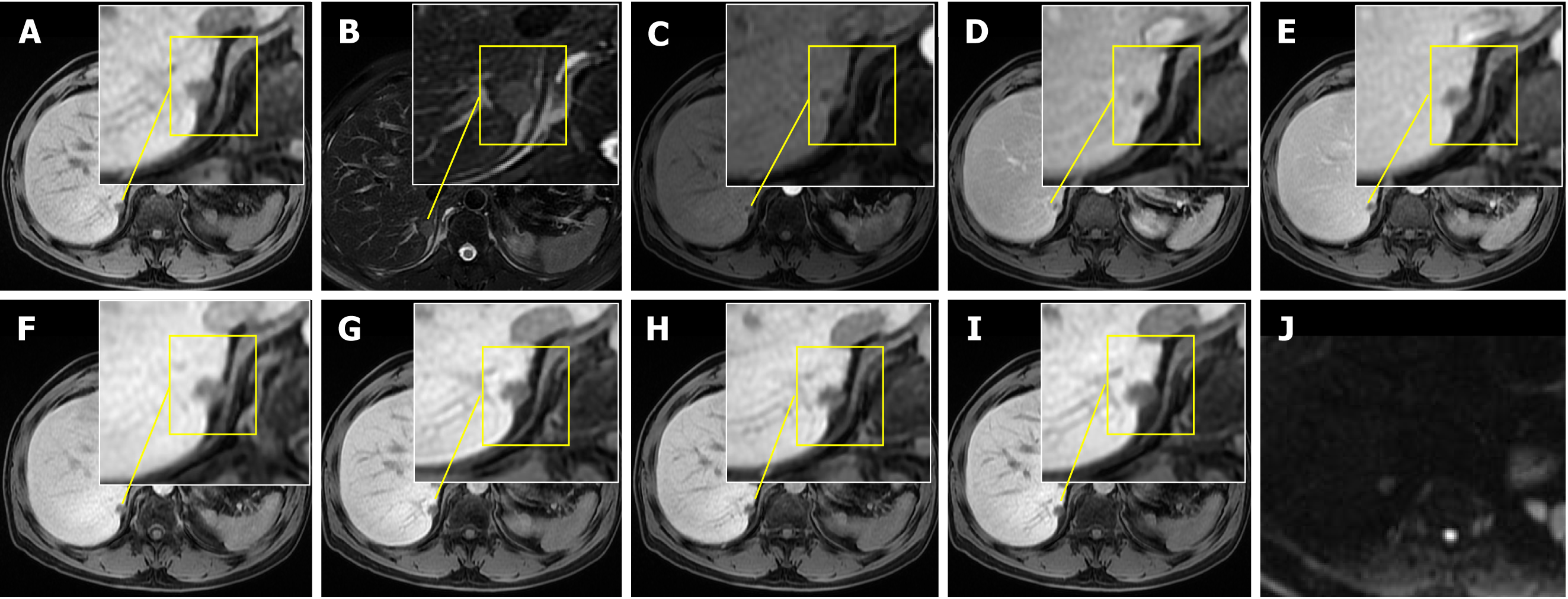
Abdominal-enhanced computed tomography (CT) scans revealed a hypodense nodule close to the posterior aspect of segment 7 of the liver, displaying heterogeneous enhancement in the arterial phase and marked hypo-enhancement in the delayed phase, suggestive of a malignant hepatic lesion (Figure 1). Additionally, multiple low- to isodense nodules were observed in the left adrenal gland, leading to a diagnosis of multiple adenomas. Abdominal magnetic resonance imaging (MRI) further confirmed the presence of the hepatic nodule and adrenal lesions, indicating a malignant hepatic tumor and adrenal adenomas (Figure 2).
HCC (CNLC stage Ia); adenoma of the left adrenal gland.
Based on CT and MRI findings, which suggested a malignant liver lesion and a left adrenal adenoma, the patient un
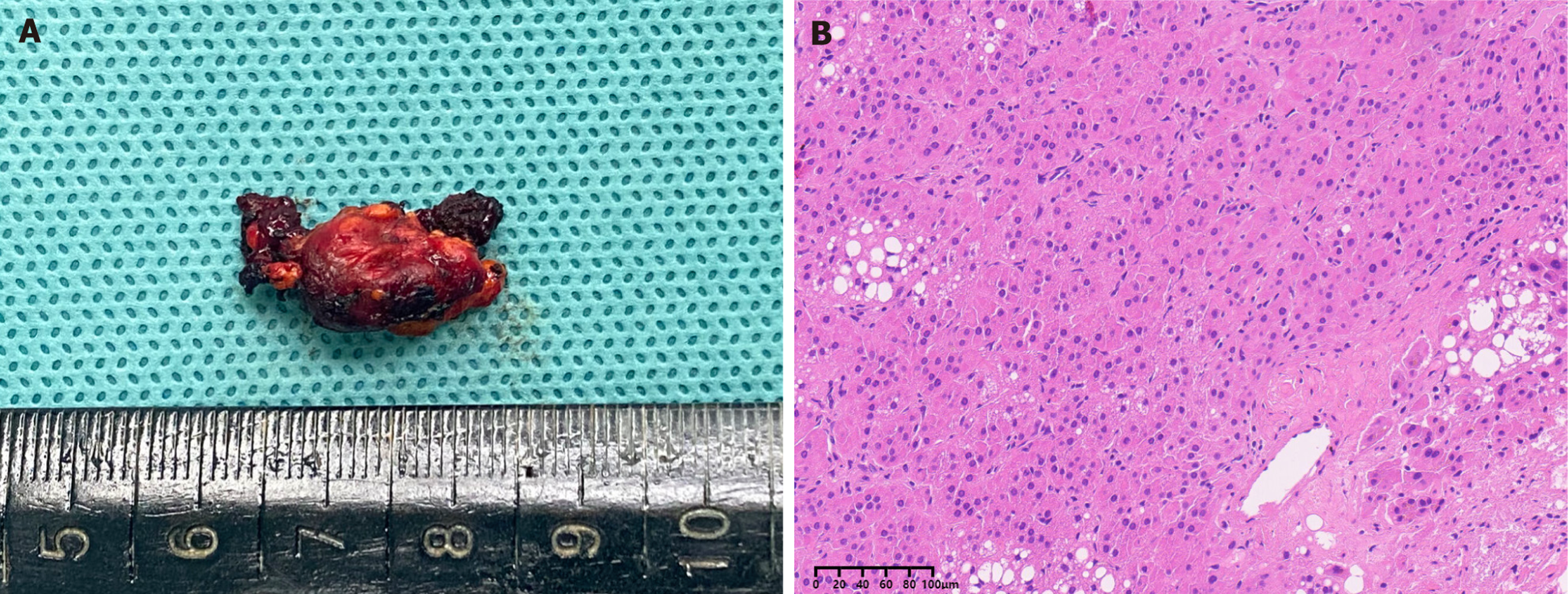
Intraoperatively, no signs of liver cirrhosis were observed. Dissection of the right adrenal gland revealed that the nodule had infiltrated the hepatic parenchyma, confirming the presence of ectopic adrenal tissue. Frozen section pathology revealed proliferating adrenal tissue. Postoperative histopathological analysis confirmed that the liver lesion consisted of proliferating adrenal tissue with no malignant features (Figure 3). Histology revealed small nests of cells, with approximately 50% eosinophilic cytoplasm and 50% clear cytoplasm, consistent with adrenal tissue. Immunohistochemical staining was positive for steroidogenic factor-1, melan-A, and inhibin-A, with partial positivity for synaptophysin. The Ki67 proliferation index was approximately 2%. Hepatocyte staining was positive at the periphery of normal liver tissue, and special staining demonstrated positive reticular fibers surrounding the cell nests. The patient recovered smoothly and was discharged 10 days postoperatively.
Adrenal ectopia refers to the presence of adrenal tissue in an abnormal location. It can be categorized as either adrenal residual tissue or adrenal cortical heterotopia, depending on whether its embryological origin aligns with the normal anatomical location[3]. The underlying mechanisms of adrenal ectopia remain unclear. The adrenal gland has dual embryological origins: The cortex derives from the mesoderm of the urogenital ridge, while the medulla originates from the neural crest[4]. During embryonic development, failure of the cortical tissue to separate from the peritoneal meso
Additionally, when multiple rudiments or cortical tissue are invaded by medullary components, ectopic adrenal tissue may develop[1]. However, simple developmental migration defects alone cannot fully explain cases of ectopic adrenal tissue found in distant locations, such as the pituitary or lungs[5]. Several pathological mechanisms are believed to contribute to adrenal ectopia, including mitotic cell proliferation, induced differentiation, migration factors, and apoptosis. These abnormalities reflect cellular proliferation and migration disruptions, leading to ectopic adrenal tissue forming in unusual locations[6].
Adrenal ectopia can occur at any age, though some ectopic tissue may atrophy or disappear with aging. It is most commonly found in children, with approximately 50% of neonatal and pediatric autopsies revealing ectopic adrenal tissue[1]. A study reported 15 cases of incidentally discovered ectopic adrenal tissue during surgery, with patients ranging from 10 to 69 years of age and an average age of 46 years[7]. Adrenal ectasia is most frequently found in the kidneys or tissues surrounding the gonads and ovaries[8,9]. However, adrenal ectopia in the liver is rare and is typically reported in isolated case studies[6,10].
Hepatic ectopic adrenal tissue is typically non-functional (as seen in this case), with insidious and non-specific clinical manifestations. Approximately 80% of reported cases are incidental findings on imaging, with no hormonal abnormalities or abdominal symptoms. Rare functional cases may present with hypertension, hypokalemia, or virilization, but such manifestations are more common in primary adrenal tumors. Notably, the lesion in this case and previous reports were located subcapsularly in the right hepatic lobe (segment VII), possibly related to embryonic migration pathways of adrenal cortical tissue. While this anatomical feature is not pathognomonic, it may raise suspicion for ectopic adrenal tissue.
On CT imaging, hepatic ectopic adrenal adenomas typically appear as solitary, round, hypodense nodules with well-defined borders. They exhibit heterogeneous enhancement in the arterial phase (“wash-in”) and significant attenuation in the portal and delayed phases (“wash-out”). Intralesional fat deposition (low CT values) may be observed. The CT features in this case align with literature (Figure 1). However, approximately 30% of HCCs may mimic this “wash-in/wash-out” pattern, necessitating additional discriminators.
The MRI characteristics of hepatic ectopic adrenal tissue are as follows: First, the lesion typically demonstrates hypointensity on T1-weighted imaging, marked signal loss on opposed-phase sequences (indicating intracellular lipid content), hyperintensity on T2-weighted imaging, and restricted diffusion on diffusion-weighted imaging (Figure 2). Furthermore, the enhancement pattern is characterized by predominant peripheral arterial enhancement with delayed capsular enhancement (as observed in a prior case study, which may mimic the “pseudocapsule” appearance of HCC. However, unlike HCC, which is frequently associated with cirrhosis and elevated alpha-fetoprotein, this entity lacks such clinical correlates. Critically, the signal dropout on opposed-phase chemical shift imaging serves as a hallmark feature of adrenal-origin tumors, providing a key discriminator against lipid-poor HCCs and metastatic lesions.
This case highlights a key diagnostic challenge: Misidentifying an adrenal ectopic nodule in the liver as a primary or metastatic hepatic malignancy (specifically HCC) or as hepatic adenoma (Table 1)[11-13]. The morphological changes and biological behavior of adrenal adenomas can be variable, often requiring immunohistochemistry analysis for accurate diagnosis. The steroidogenic factor-1 is considered the most reliable and specific biomarker of adrenocortical origin[14,15].
| Feature | Hepatic ectopic adrenal tissue | Hepatocellular carcinoma | Metastatic tumors | Hepatic adenoma |
| Clinical context | Asymptomatic, normal AFP[1,5] | Cirrhosis, HBV/HCV history, elevated AFP[1,5] | Known primary malignancy (e.g., colorectal cancer), elevated CEA/CA19-9 | Long-term oral contraceptive use in females, hemorrhage/necrosis |
| Lesion characteristics | Solitary, subcapsular (common in segment VII) | Solitary/multifocal, cirrhotic background | Multiple nodules with rim enhancement (“target sign”)[5,10] | Solitary, well-defined margins |
| CT/MRI enhancement | Heterogeneous arterial enhancement, rapid washout (“wash-in/wash-out”) | Homogeneous arterial enhancement, pseudocapsule | Peripheral arterial enhancement, delayed filling | Homogeneous arterial enhancement, persistent portal/delayed phase enhancement |
| Fat deposition | Common (signal dropout on opposed-phase imaging) | Rare (except steatotic variants) | Absent | Occasional hemorrhage/necrosis |
| DWI restriction | Mild restriction (moderate ADC values) | Marked restriction (low ADC values) | Severe restriction (very low ADC)[5,10] | Variable (depends on hemorrhage) |
| Pathologic markers | SF1+, melan-A+, inhibin-A+[1,5] | HepPar-1+, Glypican-3+[1,5] | Matches primary tumor (e.g., CK20+/CDX2+ for colorectal origin) | β-catenin nuclear+ (specific subtypes) |
In this case, imaging studies suggested malignancy, prompting further investigations and ultimately leading to a partial hepatectomy. The preoperative misinterpretation of the liver lesion as malignant resulted in an invasive surgery that might have been avoided had adrenal ectopia been considered earlier in the diagnostic process. This experience is an essential lesson for clinicians, particularly those in hepatobiliary surgery and oncology. It underscores the necessity of a comprehensive differential diagnosis when evaluating unusual liver lesions, especially when imaging findings suggest malignancy but lack definitive features of primary liver cancer or metastases. Although adrenal ectopia is rare and may be unable to make immediate diagnostic considerations, recognizing its possibility is crucial to preventing unnecessary procedures and treatments.
Although rare, adrenal ectopia should be considered in the differential diagnosis of hepatic lesions, particularly when they mimic malignancy. This case offers valuable insights into the importance of careful preoperative evaluation and emphasizes the need to consider adrenal ectopia when encountering unusual hepatic lesions. A thorough understanding of the pathophysiology of adrenal ectopia and its potential to resemble malignancy is crucial for clinicians to avoid diagnostic errors and ensure appropriate management.
We thank Dr. Di Tang (Associate Chief Physician) and Dr. Gang Deng (Associate Chief Physician) from the Department of General Surgery, The Seventh Affiliated Hospital of Sun Yat-sen University for performing the surgical procedures. We also extend our gratitude to all participants involved in this study.
| 1. | Anderson JR, Ross AH. Ectopic adrenal tissue in adults. Postgrad Med J. 1980;56:806-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 59] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 2. | Mendez R, Tellado MG, Somoza I, Liras J, Sanchez-Abuin A, Pais E, Vela D. Ectopic adrenal tissue in the spermatic cord in pediatric patients: surgical implications. Int Braz J Urol. 2006;32:202-7; discussion 207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 43] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 3. | Rizkalla CN, Tretiakova M. What's new in adrenal gland pathology: WHO 5th edition for adrenal cortex. J Pathol Transl Med. 2024;58:201-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 4. | Schechter DC. Aberrant adrenal tissue. Ann Surg. 1968;167:421-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 88] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Tarçın G, Ercan O. Emergence of Ectopic Adrenal Tissues-What are the Probable Mechanisms? J Clin Res Pediatr Endocrinol. 2022;14:258-266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 6. | Vestfrid MA. Ectopic adrenal cortex in neonatal liver. Histopathology. 1980;4:669-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 7. | Sensu S, Keser SH, Gurbuz Y, Barisik NO, Gul AE. Clinicopathological Evaluation of 15 Ectopic Adrenal Tissues. Arch Iran Med. 2021;24:301-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 8. | Uğur Kılınç AN, Bayramoğlu Z, Ünlü Y, Keçeli AM, Dönmez Mİ. Incidental ectopic adrenal cortical tissue: Retrospective analysis of 16 patients. J Pediatr Urol. 2021;17:258.e1-258.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 9. | Chai J, Feng R, Yu X, Ke C. Ectopic adrenal tissue in ovary: A rare case. Asian J Surg. 2024;S1015-9584(24)01524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 10. | Chai J, Feng R, Ke C. Ectopic adrenal tissue in the liver: A rare case. Asian J Surg. 2024;S1015-9584(24)02428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 11. | Yu H, He Y. Hepatic adrenal adenoma-rare tumor on right lobe of liver: a case report and literature review. BMC Surg. 2020;20:128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Zhuang BW, Wang W, Xie XY. A Rare Case of Liver Tumor. Gastroenterology. 2019;157:e5-e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 13. | Sugiyama T, Tajiri T, Hiraiwa S, Inomoto C, Kajiwara H, Kojima S, Tobita K, Nakamura N. Hepatic adrenal rest tumor: Diagnostic pitfall and proposed algorithms to prevent misdiagnosis as lipid-rich hepatocellular carcinoma. Pathol Int. 2015;65:95-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 14. | Mete O, Asa SL, Giordano TJ, Papotti M, Sasano H, Volante M. Immunohistochemical Biomarkers of Adrenal Cortical Neoplasms. Endocr Pathol. 2018;29:137-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 15. | Mete O, Erickson LA, Juhlin CC, de Krijger RR, Sasano H, Volante M, Papotti MG. Overview of the 2022 WHO Classification of Adrenal Cortical Tumors. Endocr Pathol. 2022;33:155-196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 165] [Article Influence: 55.0] [Reference Citation Analysis (0)] |