Published online Nov 27, 2021. doi: 10.4254/wjh.v13.i11.1629
Peer-review started: February 23, 2021
First decision: June 4, 2021
Revised: June 8, 2021
Accepted: October 11, 2021
Article in press: October 11, 2021
Published online: November 27, 2021
Processing time: 273 Days and 16.7 Hours
Hepatic resection is the gold standard for patients affected by primary or metastatic liver tumors but is hampered by the risk of post-hepatectomy liver failure. Despite recent impro
Core Tip: Hepatic resection is the gold standard for patients affected by liver tumors but is hampered by the risk of post-hepatectomy liver failure. We herein review the literature on the rational, the preliminary assessment, the advantages as well as the limits of each existing technique for preparing the liver for major hepatectomy.
- Citation: Del Basso C, Gaillard M, Lainas P, Zervaki S, Perlemuter G, Chagué P, Rocher L, Voican CS, Dagher I, Tranchart H. Current strategies to induce liver remnant hypertrophy before major liver resection. World J Hepatol 2021; 13(11): 1629-1641
- URL: https://www.wjgnet.com/1948-5182/full/v13/i11/1629.htm
- DOI: https://dx.doi.org/10.4254/wjh.v13.i11.1629
Hepatic resection is the gold standard for patients affected by primary or metastatic liver tumors but is hampered by the risk of post hepatectomy liver failure (PHLF). Indeed, PHLF is considered the most frightening complication of liver surgery, representing a major source of severe morbidity and mortality[1]. Despite recent improve
The liver has a unique capacity of preserving its volume due to regeneration. The atrophy-hypertrophy phenomenon is a prime example of the liver’s pathophysiologic (atrophy) and restorative (hypertrophy) response to injury[2]. It occurs whenever there is impairment of bile or blood flow: the liver reacts with atrophy of the region concerned and with compensatory hypertrophy of the less or not impaired regions, resulting in characteristic gross deformity of the organ and, in some instances, in rotation of the liver around a virtual hilar axis[3]. The mechanisms that induce cellular division are complex and based on different inflammatory cytokines. The Hepatocyte Growth Factor (HGF) seems to be the main mitogenic factor and its role has been established in liver regeneration[4].
The first case of in vivo human hepatic regeneration was described by Pack et al[5] in 1962. Starting from animal models in the first half of the 20th century, it was recognized that liver regeneration could also be induced by portal vein ligation (PVL)[6]. In 1986, the first cases of percutaneous transhepatic portal vein embolization (PVE) were performed before liver resection in the setting of hepatocellular carcinoma[7], and a few years later Makuuchi et al[8] reported the utility of PVE in promoting FLR hypertrophy prior to hepatic resection in patients with hilar cholangiocarcinoma. Since those initial reports, preoperative PVE has been established as the standard procedure for obtaining FRL hypertrophy, increasing the eligibility of patients for major hepatectomy as well as improving postoperative outcomes and safety. However, concerns regarding the insufficient increase of FLR and/or concomitant tumoral progression after PVE have led to the development of recent alternative techniques to push further the limits of liver surgery.
The aim of this article is to review the techniques available for preparing the liver for major hepatectomy, and to depict their advantages and limitations.
The liver’s unique capacity for regeneration was first recorded in the legend of Prometheus in Greek mythology and it represents the basis of the treatment of many liver diseases. Regeneration of the liver is a pathophysiological process, embracing both hypertrophy (increase in cell size or protein content in the prereplicative phase) and hyperplasia (increase in cell numbers). Both events can take place independently[9]. The mechanisms of liver regeneration have mainly been studied after extensive hepatectomy. The players of regeneration following the different techniques exposed in this article are thought to be similar to those after hepatectomy, but the precise mechanism remains unknown. Basically, the regeneration process is a cytokine- and growth-factor-mediated pathway. The main cytokine-mediated pathways include members of the innate immune system, tumor necrosis factor (TNF)α and interleukin (IL)-6, and growth-factor-mediated pathways are regulated by HGF and transforming growth factor (TGF)α[10]. It is a multi-step process, starting from the “priming” of hepatocytes, the moment they acquire replicative capacity, followed by the proliferative step in which an adequate cell mass is attained, and a termination stage in which liver cell proliferation is ended once the necessary functional mass has been reached[11]. Proliferation of hepatocytes advances from periportal to pericentral areas of the lobule, as a wave of mitoses[12]. Proliferation of biliary epithelial cells occurs a little later than hepatocytes. The particularity of liver regeneration is that replacement of the lost hepatic mass is not mediated by selected stem cells proliferation but it entirely depends on mature adult hepatocytes and other hepatic cell types. Concerning the time interval, as far as we know, normal liver weight is reestablished within 8-15 d in humans[13].
Although morbidity and mortality after liver surgery have improved over the past 10 years, PHLF is still reported in up to 8%, ranging from 1.2% to 32%, and depends on the patient’s condition and functional reserve of the liver before resection[1]. Different definitions of PHLF are available. In 2011, the International Study Group of Liver Surgery (ISGLS) defined PHLF as “a post-operatively acquired deterioration in the ability of the liver to maintain its synthetic, excretory, and detoxifying functions, which are characterized by an increased International Normalized Ratio (INR) and concomitant hyperbilirubinemia on or after postoperative day 5”[14]. It is worth pointing out that severe PHLF is associated with a mortality rate of 54%.
A related syndrome that results in a transient but sometimes fatal form of liver failure has been described following liver transplantation (LT) but also after extensive liver resection. This is the so-called Small For Size Syndrome (SFSS). In 2005, Dahm et al[15] defined SFSS as a graft to recipient weight ratio < 0.8% alongside two of the following for three consecutive days; bilirubin > 100 mmol/L, INR > 2 and encephalopathy grade 3 or 4. In this definition, SFSS is a clinical syndrome characterized by post-operative liver dysfunction, prolonged cholestasis and coagulopathy, portal hypertension and ascites. It can lead to a higher rate of hemorrhage, sepsis and gastrointestinal bleeding[16]. The key point of SFSS is the presence of portal hypertension and intra-hepatic portal congestion as the underlying cause of liver failure[17].
Despite improvements in surgical and postoperative management, parameters determining the degree of possible hepatectomy remain largely uncertain. Different patient related and surgical factors have to be considered to decrease PHLF incidence. Surgical factors include the extent of resection and volume of FLR, duration of intraoperative liver ischemia during portal pedicle clamping, duration of surgery and the need for blood transfusion. The risk of PHLF is highly influenced by the quality of underlying liver parenchyma. The type of underlying liver parenchyma is frequently assessed by preoperative liver biopsy, but noninvasive methods, such as liver stiffness, are now available. For example, liver stiffness measurement by transient elastography (Fibroscan) predicts persistent hepatic decompensation in patients undergoing resection for hepatocellular carcinoma[18].
It is generally thought that the minimal functional liver mass needed for adequate postoperative liver function is estimated to be 20%-25% in patients with normal liver parenchyma, whereas those with chemotherapy-induced liver injury require a FLR volume of approximately 30%, while those with cirrhosis at least a 40% minimal functional liver mass[19]. Therefore, standardized FLR volume can be easily evaluated by a tridimensional computed tomography (CT) reconstruction method, as FLR/ estimated total liver volume[20]. Estimated total liver volume is generally calculated using a formula based on body surface area[21].
In addition to volume, estimation of FLR function is an important factor. Typical biochemical parameters, such as liver function tests, albumin, and clotting factors must be evaluated. The old but effective Child-Turcotte-Pugh score, which was introduced in 1964, still represents a simple system for grading liver function[22]. The model for end-stage liver disease score, which is mainly used in liver transplantation, can also predict the survival rate of cirrhotic patients to better select ideal candidates for surgery[23]. A recent study also showed that mean serum level of hyaluronic acid can be a useful tool, especially when liver biopsy is not feasible[24].
Dynamic tests of liver function can also be used. The most well-known is indocyanine green (ICG) clearance. ICG is a water soluble, inert, fluorescent tricarbocyanine dye with protein binding close to 95% (mainly, alpha1- and beta-lipoproteins and albumin), a hepatic extraction rate above 70%, and is almost completely excreted in its unchanged form by the liver. ICG elimination can be expressed as ICG plasma disappearance rate (ICGPDR) or retention rate at 15 min (ICGR15), reflecting liver function. Use of the ICG test for patient selection has been shown to decrease postoperative mortality[25].
In recent years, there have been several attempts to assess hepatobiliary magnetic resonance imaging (MRI) as a tool to predict liver dysfunction. Since it was first described in 1991 by Weinmann et al[26], MRI has been showed to provide both global and segmental liver function information, and postoperative remnant liver function thanks to the measurement of liver signal intensity in the hepatobiliary phase.
Liver function evaluation by nuclear medicine techniques is also more and more used. Dynamic 99mTc-mebrofenin hepatobiliary scintigraphy has been used to provide quantitative information on total and regional liver function. The hepatic uptake of 99mTc-mebrofenin is similar to the uptake of organic anions such as bilirubin[27]. This technique efficiently estimates the risk of postoperative liver failure especially in patients with uncertain quality of liver parenchyma[28]. The 99m Tc-GSA is another recently proposed agent that is not affected by hyperbilirubinemia and can be used for liver function assessment in cholestatic patients[29]. Finally, the LiMAx test allows real-time in vivo determination of liver Cytochrome P450 1A2 (CYP1A2) activity. The CYP1A2 is not influenced by cholestasis or drugs and is ubiquitous in liver parenchyma. Intravenous administration of 13C methacetin, a substance exclusively metabolized by CYP1A2, with continuous real-time breath analysis represents the basis of the LiMAx test[30].
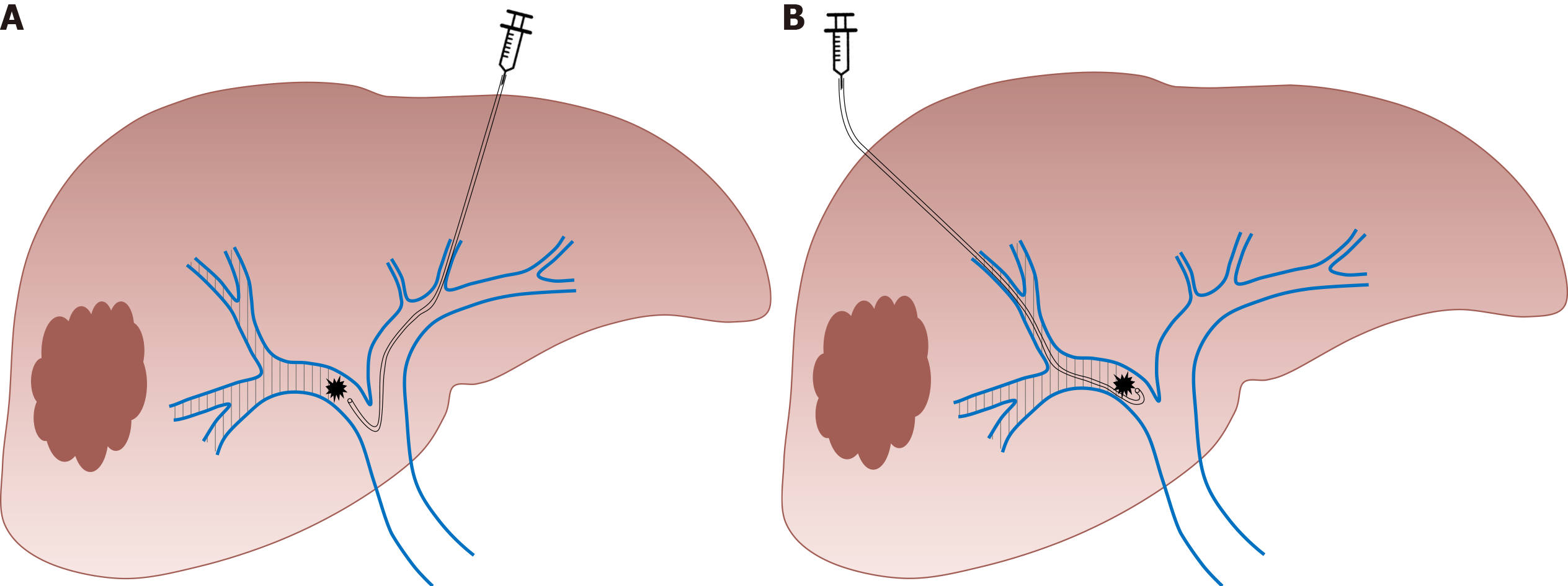
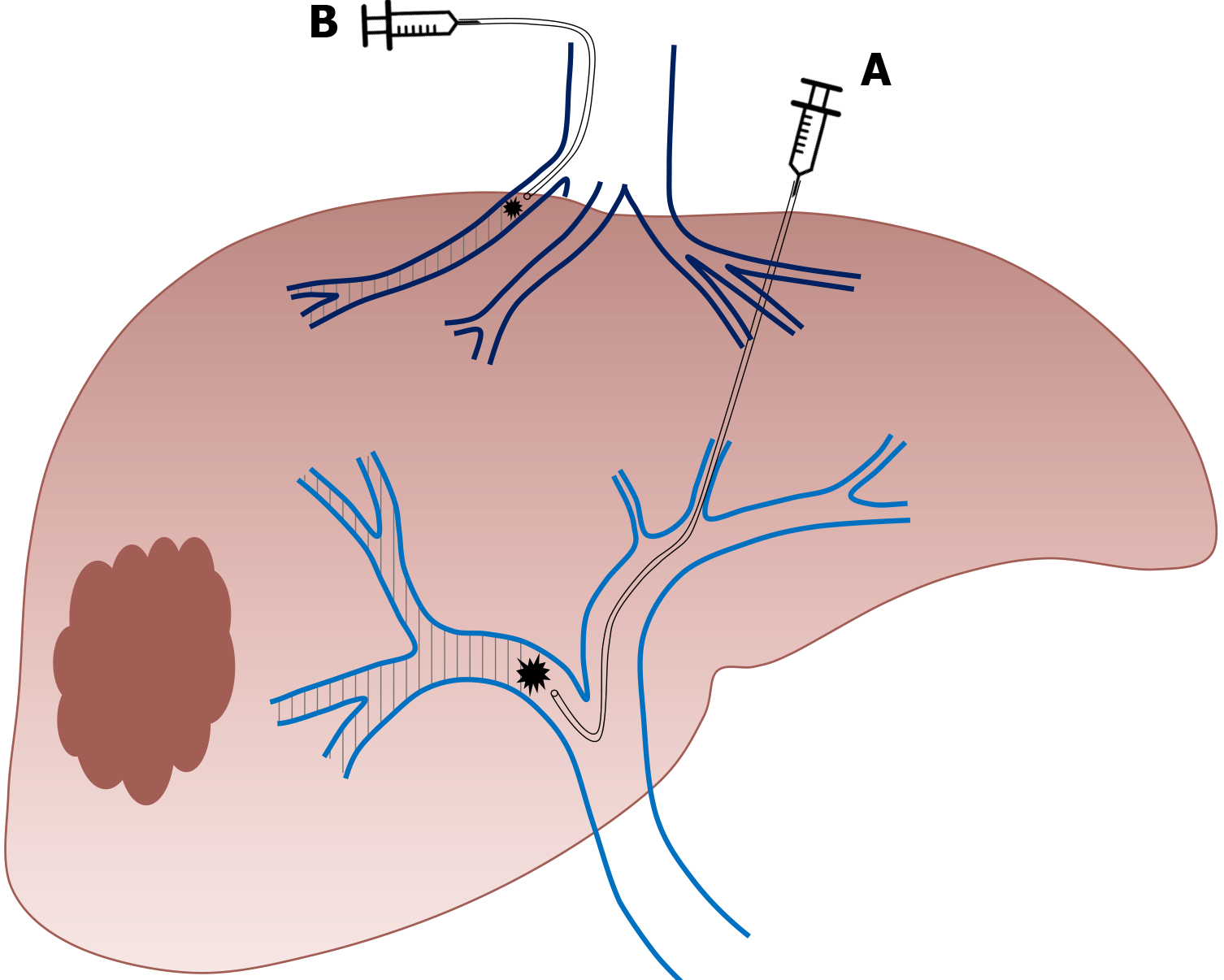
Since the first report in 1986, PVE has progressively become the gold standard for inducing liver hypertrophy with satisfying safety and efficacy[31]. Initially described by laparotomy, the portal system access is now obtained by percutaneous puncture of the portal vein. According to the operator’s preference, an ipsilateral or contralateral approach can be chosen, in reference to the segment bearing the tumor. The ipsilateral approach has the main advantage of protecting the FLR from injury[2] whereas the contralateral approach facilitates embolization[32]. Irrespective of the approach chosen, PVE is performed in a retrograde manner (Figure 1). Many embolic materials have been used for PVE without significant differences in terms of hypertrophy. Embolic materials include fibrin glue, N-butyl-2-cyanoacrylate and ethiodized oil, gelatin sponge and thrombin, coils, microparticles [e.g., polyvinyl alcohol (PVA) particles or tris-acryl gelatin microspheres] and absolute alcohol[33]. A non‐absorbable material is generally used. However, interesting results were reported with the use of an absorbable powder material (Gelfoam® powder, Pfizer, New York, USA) that lasts approximately 2 wk, leading to temporary PVE. In an animal model, this method showed efficient and stable liver regeneration[34]. These results were confirmed in a limited preliminary series in clinical practice[35] and a prospective study is undergoing (EMBORES study, NCT02945059). One of the advantages of temporary PVE is that it can theoretically be repeated several times to boost more liver hypertrophy, as has been suggested in an animal model[36].
PVE is successfully performed in more than 90% of cases[37]. A computed tomography scan with volumetric evaluation is generally performed between 4 and 8 wk after embolization. PVE induces a FLR hypertrophy than can reach 40%[37], with a low 2% morbidity rate and no mortality in the vast majority of studies[37-39]. PVE is considered an efficient method, allowing successful hepatectomy in more than 70% of cases[37,38,40].
Contraindications to PVE are extensive portal thrombus and important portal hypertension[41]. Another potential limit of PVE is the risk of tumor growth during the 4 to 8 wk separating PVE and liver surgery. In addition, several authors have suggested that PVE itself could promote tumor growth within the embolized liver[42-45]. Among others, these reasons have led to the development of alternative strategies.
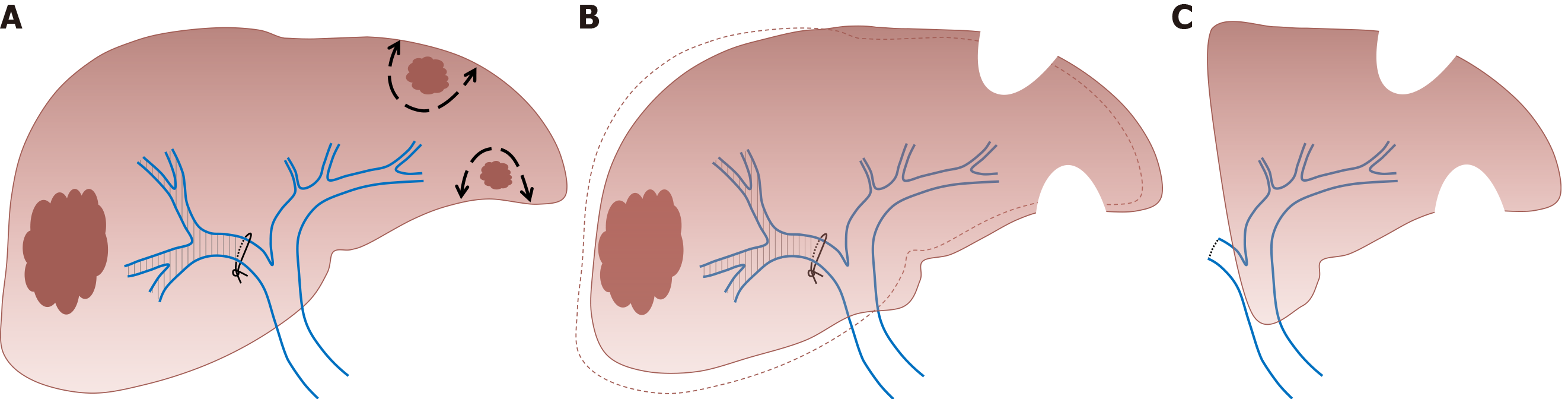
As it requires a surgical procedure with portal pedicles dissection, PVL is nowadays mainly indicated in the setting of two-stage hepatectomy (TSH) for the treatment of bilobar liver disease[46,47]. In the TSH strategy, the first surgical step includes tumoral clearance of the FLR (usually by parenchymal spearing resections or locoregional treatment like radiofrequency ablation) and concomitant PVL that allows FLR growth. In the second step, after liver regeneration (approximately 4 to 8 wk later), major liver resection is performed (usually right or right extended hepatectomy) (Figure 2). Similarly, PVL can be performed for the management of patients presenting synchronous colorectal metastases or neuroendocrine tumors[47]. The first surgical step associates colorectal resection with PVL, followed by major liver surgery in the second procedure. However, many centers have adopted PVE (performed by the percutaneous approach after FLR clearance or colorectal resection) for two-step procedures, avoiding portal pedicle dissection and facilitating the second procedure[48].
It was initially suggested that PVE resulted in superior FLR growth compared to PVL[49] as in theory PVE allows distal portal obstruction which decreases the possibility of intrahepatic collateral development. Several studies demonstrated that the results are globally similar[50,51]. In fact, the debate concerning the efficiency of PVL compared to PVE is no longer relevant. PVL requires a surgical procedure and can appear as an alternative to PVE only when a two-step surgery is planned. In other cases, percutaneous PVE is clearly a simpler and better tolerated approach.
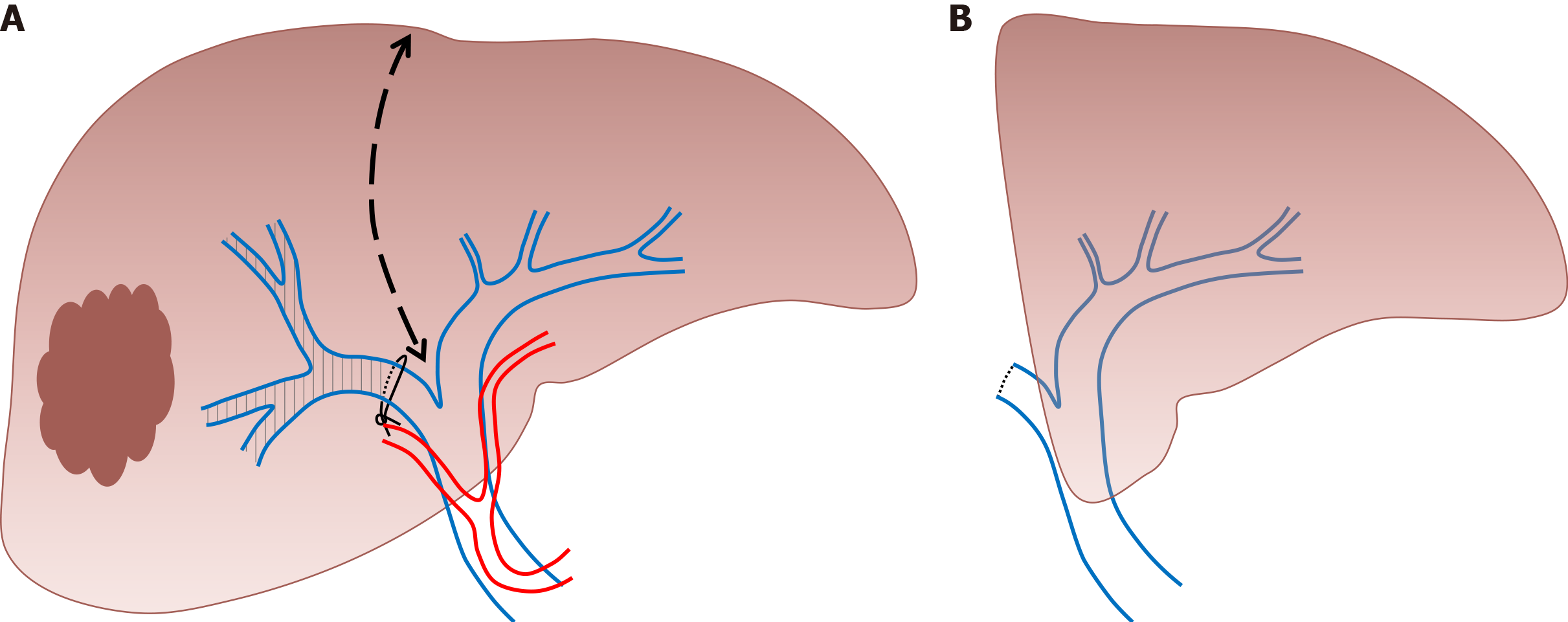
The aim of this alternative strategy, described by Schnizbauer et al[52] in 2012, is to induce rapid and massive liver hypertrophy, to allow liver surgery in a short period of time in patients with initially very limited FRL volume. The first step of the associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) procedure consists of performing PVL and an in situ splitting of the liver parenchyma, leaving the hepatic artery, bile duct, and hepatic vein intact until the subsequent operation. This first surgical step can be associated with tumoral clearance of the FRL. During the second operation (that can be performed one to two weeks later) the remaining hepatic artery, bile duct, and hepatic vein are divided and the liver specimen is extracted (Figure 3).
The first report demonstrated a morbidity rate of 44% and a mortality rate of 12%[52], and triggered an intense debate on the safety of this procedure, limiting its promotion worldwide. The morbi-mortality rate decreased with experience but remains high, with approximately 40% of major postoperative complications and 9% of mortality[53]. Nevertheless, the ALPPS technique induces more than 65% of FLR growth in approximately 7 days[52-55] and the second procedure is feasible in more than 90% of cases[56]. The main advantage of the ALPPS procedure is the rapid increase in FLR volume in a short interval and therefore a shorter interval between the two stages. Although the volumetric results of this technique are impressive, several authors suggested that FLR volume hypertrophy is not correlated to functional improvement[57,58] which could partly explain the high morbidity of the procedure. Besides, concerns have been raised by some authors regarding potentially poorer oncological results comparing to the classical TSH[59]. The results of a meta-analysis comparing ALPPS to TSH showed that the extent of FLR increase was not different between the two groups[60]. The time needed to reach final liver volume was shorter in ALPPS than in the TSH approach[60]. In this meta-analysis, ALPPS was associated with a higher incidence of major and overall morbidity and mortality compared to TSH[60]. However, in a recent randomized controlled trial, Hasselgren et al[61] observed similar morbidity between ALPPS and classical TSH and an improved survival in the ALPPS group.
To decrease complication rate, a variety of technical modifications have been proposed such as partial-ALPPS, mini-ALPPS, tourniquet-ALPPS, hybrid-ALPPS, microwave ablation-assisted ALPPS and radiofrequency ablation-assisted ALPPS. Huang et al[62] suggested in a systematic review that a partial ALPPS technique in which only partial parenchymal sparing is performed during the first surgical step could achieve lower morbidity and mortality rates, reaching the same FLR hypertrophy rate as ALPPS in non-cirrhotic patients.
Although PVE remains the gold standard for FLR hypertrophy, two concerns persist with this approach: An insufficient contralateral hypertrophy, particularly in patients with underlying liver disease (steatosis, fibrosis or cirrhosis), and the eventuality of tumor progression while waiting for the non-embolized liver to hypertrophy. In particular, portal flow interruption may induce a compensatory increase in arterial blood flow of embolized segments and result in a paradoxical growth of tumors vascularized by arterial blood flow. In this context, it has been postulated that the addition of trans-arterial embolization (TAE) or trans-arterial chemoembolization (TACE) would produce more rapid and extensive FLR growth (by obtaining obliteration of intrahepatic arterioportal shunts) and may help to counteract the stimulating effect on tumor growth[63]. Therefore, hepatocellular carcinomas, which are tumors particularly vascularized by arterial blood flow and develop generally in underlying pathological liver parenchyma, are the main target of this combined strategy[64].
During TAE, a catheter is directly inserted via either the common femoral or left radial artery and an intra-arterial injection of a combination of microspheres and PVA particles is performed in the arterial branches of the segments to be resected. During TACE, an intra-arterial injection of a cytotoxic drug is performed such as doxorubicin, epirubicin, idarubicin, mitomycin C, or cisplatin, that is emulsified in ethiodized oil (Lipiodol® Ultra-Fluid, Guerbet). This is followed by intra-arterial injection of an embolic agent, such as gelatin sponge, PVA particles, or microspheres[65] (Figure 4). TACE can also be performed using recently developed drug-eluting beads (DEB) that allow the slow release of chemotherapeutic agents, and increase ischemia intensity and duration[65].
A sequential approach, with a time interval of a few days, is recommended to limit the risk of nontumoral liver ischemic necrosis[66] and TAE is mostly performed before PVE[66,67]. Although the number of patients reported in studies that evaluated this approach is limited, observed FLR hypertrophy is generally superior to that observed after isolated PVE. For example, Yoo et al[68] reported a statistically significant increase of 7.3% and 5.8% in FLR (over the total liver volume) for sequential TACE/PVE and isolated PVE, respectively.
An important elevation of transaminases is generally observed after this sequential approach without important clinical consequences. In the largest series reporting this approach, Peng et al[64] reported 29 procedures without deaths and only one complication and 27 patients (93%) underwent subsequent hepatectomy. Post-hepatectomy morbidity and mortality among these patients was 27.5% and 6.9%, respectively.
Theoretical contraindications of this method include extensive portal thrombus, important portal hypertension or previous biliary surgery (biliodigestive anastomosis) which exposes the patient to hepatic abscess formation after arterial embolization.
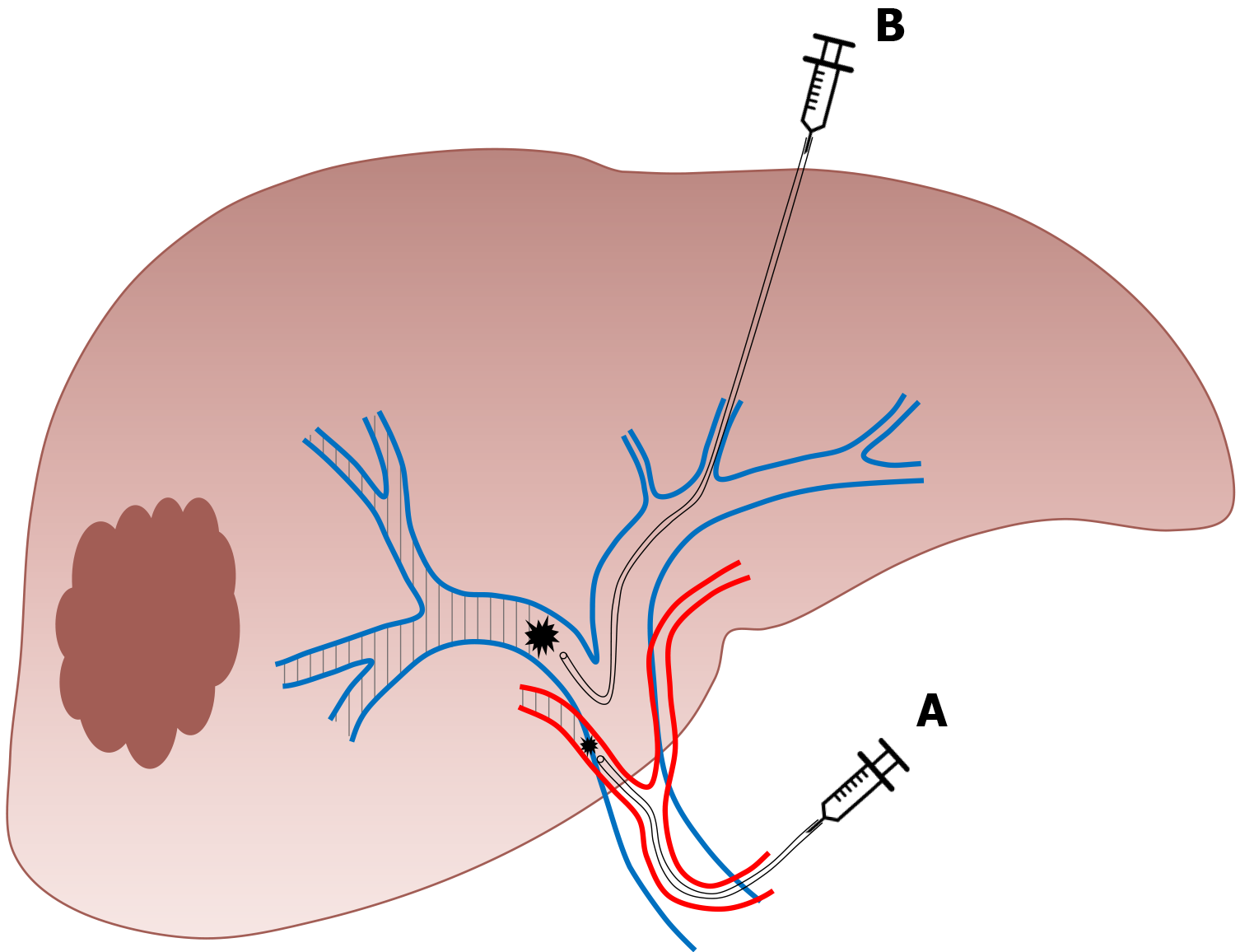
This technique consists of performing conventional PVE and ipsilateral hepatic vein obstruction (Figure 5). By associating hepatic vein embolization, the aim is to eliminate any residual portal vein flow and reduce hepatic artery inflow which can further encourage liver regeneration. Initially described as a sequential approach in which hepatic vein embolization is secondarily performed in case of insufficient FLR growth after PVE, it was demonstrated that both procedures (portal and hepatic vein embolization) can be performed simultaneously[69,70]. This novel approach is particularly interesting as it allows important liver regeneration with good tolerance. Although no study comparing ALPPS to LVD is available, it has been suggested that LVD could overcome the limits of ALPPS, abolishing the necessity of two major surgical interventions in close sequence.
Firstly, PVE is performed as previously described. For hepatic vein embolization, a vascular plug is placed in the proximal part of the hepatic vein to avoid migration of embolization agent. The vein is then embolized with a mixture of ethiodized oil and N- butyl cyanoacrylate[71]. The term “extended LVD” is used for concomitant embolization of the right and middle hepatic vein with the right portal branch[57].
The results of this approach on FLR increase are superior to those observed after isolated PVE. In a recent large comparative study, Laurent et al[71] observed a FLR volume increase of 28.9% after PVE compared to 61.2% after LVD (P < 0.0001). In this study, LVD allowed surgery in 86.4% of patients and no PHLF was reported. Kobayashi et al[72] observed similar results with a superior FLR hypertrophy after LVD compared to PVE (35% vs 24%, P = 0.034). In addition, the tolerance of LVD seems to be similar to the tolerance of isolated PVE[71,72].
This recent approach is derived from trans-arterial radioembolization with yttrium-90[73]. In radiation lobectomy (RL), radioembolization of both the tumor and the non-tumoral liver parenchyma that will be secondarily resected is performed, which requires higher radiation doses[74,75]. This technique allows concomitant tumoral control and FLR increase. One major advantage of this approach is that it could be carried out in patients with portal vein thrombosis[75].
The procedure is well-tolerated[74] with transient moderate adverse events. Results in terms of FLR volume growth are very similar to those observed after PVE. Vouche et al[74] reported 45% of FLR hypertrophy and observed a correlation between the presence of a portal vein thrombosis and FLR growth. However, series reporting major liver resection after RL are scarce[76,77]. Andel et al[77] recently reported 10 major hepatectomies in patients that were initially treated with RL for insufficient functional FLR. The RL allowed a 41% increase in FLR volume with 84% of FLR function increase (evaluated on scintigraphy). All resections were performed without major intraoperative problems. Only one patient developed a serious complication not directly related to the liver surgery and other complications were mild.
Careful initial evaluation of FLR volume and function is crucial before planning major liver resection. When required, several approaches are now available to decrease the risk of PHLF (Table 1) and thus postoperative mortality. Although PVE remains the gold standard, recent techniques that are derived from PVE might play an increasingly important role in future years.
| Approach | Indication | Advantage | Disadvantage |
| PVE | Insufficient FLR volume | Percutaneous approach | Contraindicated in patients with extensive portal thrombus and important portal hypertension; Could promote tumoral growth within the embolized liver |
| PVL and two-stage hepatectomy | Insufficient FLR volume and treatment of bilobar liver disease | PVL is performed during the first surgical step (tumoral clearance of the FLR) | Surgical procedure; Morbidity |
| Associating liver partition and PVL for staged hepatectomy | Insufficient FLR volume +/- treatment of bilobar liver disease | Liver surgery is performed in a short period of time (15 d); First surgical step (PVL and in situ splitting of the liver parenchyma) can be associated with tumoral clearance of the FLR | Surgical procedure; Morbidity |
| Sequential trans arterial embolization and PVE | Insufficient FLR volume in patients with hepatocellular carcinoma | Percutaneous approachMay help to counteract the stimulating effect of PVE on tumor growth | Sequential approach (two procedures) is recommended to limit the risk of nontumoral liver ischemic necrosis; Contraindicated in patients with extensive portal thrombus, important portal hypertension or previous biliary surgery (biliodigestive anastomosis) |
| Liver venous deprivation | Insufficient FLR volume | Percutaneous approach | Contraindicated in patients with extensive portal thrombus and important portal hypertension; Could promote tumoral growth within the embolized liver |
| RL | Insufficient FLR volume | Percutaneous approachConcomitant tumoral control and FLR increaseCan be carried out in patients with portal vein thrombosis | Data reporting liver resection after RL is scarce |
Provenance and peer review: Invited article; Externally peer reviewed.
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: France
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Jin Y S-Editor: Wang LL L-Editor: Webster JR P-Editor: Wang LL
| 1. | Schreckenbach T, Liese J, Bechstein WO, Moench C. Posthepatectomy liver failure. Dig Surg. 2012;29:79-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 103] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 2. | Black DM, Behrns KE. A scientist revisits the atrophy-hypertrophy complex: hepatic apoptosis and regeneration. Surg Oncol Clin N Am. 2002;11:849-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Kim RD, Kim JS, Watanabe G, Mohuczy D, Behrns KE. Liver regeneration and the atrophy-hypertrophy complex. Semin Intervent Radiol. 2008;25:92-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 54] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 4. | Fajardo-Puerta AB, Mato Prado M, Frampton AE, Jiao LR. Gene of the month: HGF. J Clin Pathol. 2016;69:575-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 5. | Pack GT, Islami AH, Hubbard JC, Brasfield RD. Regeneration of human liver after major hepatectomy. Surgery. 1962;52:617-623. [PubMed] |
| 6. | Honjo I, Suzuki T, Ozawa K, Takasan H, Kitamura O. Ligation of a branch of the portal vein for carcinoma of the liver. Am J Surg. 1975;130:296-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 88] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 7. | Kinoshita H, Sakai K, Hirohashi K, Igawa S, Yamasaki O, Kubo S. Preoperative portal vein embolization for hepatocellular carcinoma. World J Surg. 1986;10:803-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 310] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 8. | Makuuchi M, Thai BL, Takayasu K, Takayama T, Kosuge T, Gunvén P, Yamazaki S, Hasegawa H, Ozaki H. Preoperative portal embolization to increase safety of major hepatectomy for hilar bile duct carcinoma: a preliminary report. Surgery. 1990;107:521-527. [PubMed] |
| 9. | Palmes D, Spiegel HU. Animal models of liver regeneration. Biomaterials. 2004;25:1601-1611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 103] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 10. | Taub R. Liver regeneration: from myth to mechanism. Nat Rev Mol Cell Biol. 2004;5:836-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1091] [Cited by in RCA: 1175] [Article Influence: 56.0] [Reference Citation Analysis (0)] |
| 11. | Markiewski MM, DeAngelis RA, Lambris JD. Liver inflammation and regeneration: two distinct biological phenomena or parallel pathophysiologic processes? Mol Immunol. 2006;43:45-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 69] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 12. | Rabes HM. Kinetics of hepatocellular proliferation as a function of the microvascular structure and functional state of the liver. Ciba Found Symp. 1977;31-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 13. | Michalopoulos GK. Liver regeneration. J Cell Physiol. 2007;213:286-300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1246] [Cited by in RCA: 1158] [Article Influence: 64.3] [Reference Citation Analysis (0)] |
| 14. | Rahbari NN, Garden OJ, Padbury R, Brooke-Smith M, Crawford M, Adam R, Koch M, Makuuchi M, Dematteo RP, Christophi C, Banting S, Usatoff V, Nagino M, Maddern G, Hugh TJ, Vauthey JN, Greig P, Rees M, Yokoyama Y, Fan ST, Nimura Y, Figueras J, Capussotti L, Büchler MW, Weitz J. Posthepatectomy liver failure: a definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery. 2011;149:713-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1224] [Cited by in RCA: 1725] [Article Influence: 123.2] [Reference Citation Analysis (0)] |
| 15. | Dahm F, Georgiev P, Clavien PA. Small-for-size syndrome after partial liver transplantation: definition, mechanisms of disease and clinical implications. Am J Transplant. 2005;5:2605-2610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 444] [Cited by in RCA: 472] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 16. | Tucker ON, Heaton N. The 'small for size' liver syndrome. Curr Opin Crit Care. 2005;11:150-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 134] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 17. | Riddiough GE, Christophi C, Jones RM, Muralidharan V, Perini MV. A systematic review of small for size syndrome after major hepatectomy and liver transplantation. HPB (Oxford). 2020;22:487-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 18. | Rajakannu M, Cherqui D, Ciacio O, Golse N, Pittau G, Allard MA, Antonini TM, Coilly A, Sa Cunha A, Castaing D, Samuel D, Guettier C, Adam R, Vibert E. Liver stiffness measurement by transient elastography predicts late posthepatectomy outcomes in patients undergoing resection for hepatocellular carcinoma. Surgery. 2017;162:766-774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 19. | Adams RB, Aloia TA, Loyer E, Pawlik TM, Taouli B, Vauthey JN; Americas Hepato-Pancreato-Biliary Association; Society of Surgical Oncology; Society for Surgery of the Alimentary Tract. Selection for hepatic resection of colorectal liver metastases: expert consensus statement. HPB (Oxford). 2013;15:91-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 224] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 20. | Shindoh J, Truty MJ, Aloia TA, Curley SA, Zimmitti G, Huang SY, Mahvash A, Gupta S, Wallace MJ, Vauthey JN. Kinetic growth rate after portal vein embolization predicts posthepatectomy outcomes: toward zero liver-related mortality in patients with colorectal liver metastases and small future liver remnant. J Am Coll Surg. 2013;216:201-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 236] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 21. | Vauthey JN, Abdalla EK, Doherty DA, Gertsch P, Fenstermacher MJ, Loyer EM, Lerut J, Materne R, Wang X, Encarnacion A, Herron D, Mathey C, Ferrari G, Charnsangavej C, Do KA, Denys A. Body surface area and body weight predict total liver volume in Western adults. Liver Transpl. 2002;8:233-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 464] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 22. | Child CG, Turcotte JG. Surgery and portal hypertension. Major Probl Clin Surg. 1964;1:1-85. [PubMed] |
| 23. | Kong FH, Miao XY, Zou H, Xiong L, Wen Y, Chen B, Liu X, Zhou JJ. End-stage liver disease score and future liver remnant volume predict post-hepatectomy liver failure in hepatocellular carcinoma. World J Clin Cases. 2019;7:3734-3741. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 23] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 24. | Gudowska M, Gruszewska E, Panasiuk A, Cylwik B, Flisiak R, Świderska M, Szmitkowski M, Chrostek L. Hyaluronic acid concentration in liver diseases. Clin Exp Med. 2016;16:523-528. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 25. | Imamura H, Sano K, Sugawara Y, Kokudo N, Makuuchi M. Assessment of hepatic reserve for indication of hepatic resection: decision tree incorporating indocyanine green test. J Hepatobiliary Pancreat Surg. 2005;12:16-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 255] [Article Influence: 12.8] [Reference Citation Analysis (1)] |
| 26. | Weinmann HJ, Schuhmann-Giampieri G, Schmitt-Willich H, Vogler H, Frenzel T, Gries H. A new lipophilic gadolinium chelate as a tissue-specific contrast medium for MRI. Magn Reson Med. 1991;22:233-7; discussion 242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 181] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 27. | de Graaf W, van Lienden KP, Dinant S, Roelofs JJ, Busch OR, Gouma DJ, Bennink RJ, van Gulik TM. Assessment of future remnant liver function using hepatobiliary scintigraphy in patients undergoing major liver resection. J Gastrointest Surg. 2010;14:369-378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 205] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 28. | Erdogan D, Heijnen BH, Bennink RJ, Kok M, Dinant S, Straatsburg IH, Gouma DJ, van Gulik TM. Preoperative assessment of liver function: a comparison of 99mTc-Mebrofenin scintigraphy with indocyanine green clearance test. Liver Int. 2004;24:117-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 88] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 29. | Cieslak KP, Runge JH, Heger M, Stoker J, Bennink RJ, van Gulik TM. New perspectives in the assessment of future remnant liver. Dig Surg. 2014;31:255-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 30. | Stockmann M, Lock JF, Riecke B, Heyne K, Martus P, Fricke M, Lehmann S, Niehues SM, Schwabe M, Lemke AJ, Neuhaus P. Prediction of postoperative outcome after hepatectomy with a new bedside test for maximal liver function capacity. Ann Surg. 2009;250:119-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 224] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 31. | Shinkawa H, Takemura S, Tanaka S, Kubo S. Portal Vein Embolization: History and Current Indications. Visc Med. 2017;33:414-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 32. | Li D, Madoff DC. Portal vein embolization for induction of selective hepatic hypertrophy prior to major hepatectomy: rationale, techniques, outcomes and future directions. Cancer Biol Med. 2016;13:426-442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 33. | Madoff DC, Abdalla EK, Vauthey JN. Portal vein embolization in preparation for major hepatic resection: evolution of a new standard of care. J Vasc Interv Radiol. 2005;16:779-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 147] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 34. | Lainas P, Boudechiche L, Osorio A, Coulomb A, Weber A, Pariente D, Franco D, Dagher I. Liver regeneration and recanalization time course following reversible portal vein embolization. J Hepatol. 2008;49:354-362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 47] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 35. | Tranchart H, Catherine L, Maitre S, Lainas P, De Laveaucoupet J, Dagher I. Efficient liver regeneration following temporary portal vein embolization with absorbable gelatin sponge powder in humans. J Vasc Interv Radiol. 2015;26:507-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 36. | Tranchart H, Koffi GM, Gaillard M, Lainas P, Poüs C, Gonin P, Nguyen TH, Dubart-Kupperschmitt A, Dagher I. Liver regeneration following repeated reversible portal vein embolization in an experimental model. Br J Surg. 2016;103:1209-1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 37. | van Lienden KP, van den Esschert JW, de Graaf W, Bipat S, Lameris JS, van Gulik TM, van Delden OM. Portal vein embolization before liver resection: a systematic review. Cardiovasc Intervent Radiol. 2013;36:25-34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 345] [Cited by in RCA: 333] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 38. | Abulkhir A, Limongelli P, Healey AJ, Damrah O, Tait P, Jackson J, Habib N, Jiao LR. Preoperative portal vein embolization for major liver resection: a meta-analysis. Ann Surg. 2008;247:49-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 474] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 39. | Isfordink CJ, Samim M, Braat MNGJA, Almalki AM, Hagendoorn J, Borel Rinkes IHM, Molenaar IQ. Portal vein ligation vs portal vein embolization for induction of hypertrophy of the future liver remnant: A systematic review and meta-analysis. Surg Oncol. 2017;26:257-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 40. | Wajswol E, Jazmati T, Contractor S, Kumar A. Portal Vein Embolization Utilizing N-Butyl Cyanoacrylate for Contralateral Lobe Hypertrophy Prior to Liver Resection: A Systematic Review and Meta-Analysis. Cardiovasc Intervent Radiol. 2018;41:1302-1312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 41. | May BJ, Madoff DC. Portal vein embolization: rationale, technique, and current application. Semin Intervent Radiol. 2012;29:81-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 73] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 42. | Hoekstra LT, van Lienden KP, Doets A, Busch OR, Gouma DJ, van Gulik TM. Tumor progression after preoperative portal vein embolization. Ann Surg. 2012;256:812-7; discussion 817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 115] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 43. | Hoekstra LT, van Lienden KP, Verheij J, van der Loos CM, Heger M, van Gulik TM. Enhanced tumor growth after portal vein embolization in a rabbit tumor model. J Surg Res. 2013;180:89-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 44. | Maggiori L, Bretagnol F, Sibert A, Paradis V, Vilgrain V, Panis Y. Selective portal vein ligation and embolization induce different tumoral responses in the rat liver. Surgery. 2011;149:496-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 45. | Pamecha V, Levene A, Grillo F, Woodward N, Dhillon A, Davidson BR. Effect of portal vein embolisation on the growth rate of colorectal liver metastases. Br J Cancer. 2009;100:617-622. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 105] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 46. | Adam R, Laurent A, Azoulay D, Castaing D, Bismuth H. Two-stage hepatectomy: A planned strategy to treat irresectable liver tumors. Ann Surg. 2000;232:777-785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 587] [Cited by in RCA: 536] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 47. | Kianmanesh R, Farges O, Abdalla EK, Sauvanet A, Ruszniewski P, Belghiti J. Right portal vein ligation: a new planned two-step all-surgical approach for complete resection of primary gastrointestinal tumors with multiple bilateral liver metastases. J Am Coll Surg. 2003;197:164-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 116] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 48. | Jaeck D, Oussoultzoglou E, Rosso E, Greget M, Weber JC, Bachellier P. A two-stage hepatectomy procedure combined with portal vein embolization to achieve curative resection for initially unresectable multiple and bilobar colorectal liver metastases. Ann Surg. 2004;240:1037-49; discussion 1049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 395] [Cited by in RCA: 365] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 49. | Broering DC, Hillert C, Krupski G, Fischer L, Mueller L, Achilles EG, Schulte am Esch J, Rogiers X. Portal vein embolization vs portal vein ligation for induction of hypertrophy of the future liver remnant. J Gastrointest Surg. 2002;6:905-13; discussion 913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 145] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 50. | Aussilhou B, Lesurtel M, Sauvanet A, Farges O, Dokmak S, Goasguen N, Sibert A, Vilgrain V, Belghiti J. Right portal vein ligation is as efficient as portal vein embolization to induce hypertrophy of the left liver remnant. J Gastrointest Surg. 2008;12:297-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 122] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 51. | Capussotti L, Muratore A, Baracchi F, Lelong B, Ferrero A, Regge D, Delpero JR. Portal vein ligation as an efficient method of increasing the future liver remnant volume in the surgical treatment of colorectal metastases. Arch Surg. 2008;143:978-82; discussion 982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 118] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 52. | Schnitzbauer AA, Lang SA, Goessmann H, Nadalin S, Baumgart J, Farkas SA, Fichtner-Feigl S, Lorf T, Goralcyk A, Hörbelt R, Kroemer A, Loss M, Rümmele P, Scherer MN, Padberg W, Königsrainer A, Lang H, Obed A, Schlitt HJ. Right portal vein ligation combined with in situ splitting induces rapid left lateral liver lobe hypertrophy enabling 2-staged extended right hepatic resection in small-for-size settings. Ann Surg. 2012;255:405-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 837] [Cited by in RCA: 933] [Article Influence: 71.8] [Reference Citation Analysis (0)] |
| 53. | Sandström P, Røsok BI, Sparrelid E, Larsen PN, Larsson AL, Lindell G, Schultz NA, Bjørnbeth BA, Isaksson B, Rizell M, Björnsson B. ALPPS Improves Resectability Compared With Conventional Two-stage Hepatectomy in Patients With Advanced Colorectal Liver Metastasis: Results From a Scandinavian Multicenter Randomized Controlled Trial (LIGRO Trial). Ann Surg. 2018;267:833-840. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 228] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 54. | Schadde E, Ardiles V, Robles-Campos R, Malago M, Machado M, Hernandez-Alejandro R, Soubrane O, Schnitzbauer AA, Raptis D, Tschuor C, Petrowsky H, De Santibanes E, Clavien PA; ALPPS Registry Group. Early survival and safety of ALPPS: first report of the International ALPPS Registry. Ann Surg. 2014;260:829-36; discussion 836. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 352] [Cited by in RCA: 349] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 55. | Knoefel WT, Gabor I, Rehders A, Alexander A, Krausch M, Schulte am Esch J, Fürst G, Topp SA. In situ liver transection with portal vein ligation for rapid growth of the future liver remnant in two-stage liver resection. Br J Surg. 2013;100:388-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 136] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 56. | Schadde E, Schnitzbauer AA, Tschuor C, Raptis DA, Bechstein WO, Clavien PA. Systematic review and meta-analysis of feasibility, safety, and efficacy of a novel procedure: associating liver partition and portal vein ligation for staged hepatectomy. Ann Surg Oncol. 2015;22:3109-3120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 98] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 57. | Deshayes E, Schadde E, Piron L, Quenet F, Guiu B. Extended Liver Venous Deprivation Leads to a Higher Increase in Liver Function that ALPPS in Early Assessment: A comment to "Sparrelid E et al”. Dynamic Evaluation of Liver Volume and Function in Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy. J Gastrointest Surg. 2017;21:1754-1755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 58. | Sparrelid E, Jonas E, Tzortzakakis A, Dahlén U, Murquist G, Brismar T, Axelsson R, Isaksson B. Dynamic Evaluation of Liver Volume and Function in Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy. J Gastrointest Surg. 2017;21:967-974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 59. | Adam R, Imai K, Castro Benitez C, Allard MA, Vibert E, Sa Cunha A, Cherqui D, Baba H, Castaing D. Outcome after associating liver partition and portal vein ligation for staged hepatectomy and conventional two-stage hepatectomy for colorectal liver metastases. Br J Surg. 2016;103:1521-1529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 88] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 60. | Moris D, Ronnekleiv-Kelly S, Kostakis ID, Tsilimigras DI, Beal EW, Papalampros A, Dimitroulis D, Felekouras E, Pawlik TM. Operative Results and Oncologic Outcomes of Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy (ALPPS) Versus Two-Stage Hepatectomy (TSH) in Patients with Unresectable Colorectal Liver Metastases: A Systematic Review and Meta-Analysis. World J Surg. 2018;42:806-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 69] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 61. | Hasselgren K, Røsok BI, Larsen PN, Sparrelid E, Lindell G, Schultz NA, Bjørnbeth BA, Isaksson B, Larsson AL, Rizell M, Björnsson B, Sandström P. ALPPS Improves Survival Compared With TSH in Patients Affected of CRLM: Survival Analysis From the Randomized Controlled Trial LIGRO. Ann Surg. 2021;273:442-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 62. | Huang HC, Bian J, Bai Y, Lu X, Xu YY, Sang XT, Zhao HT. Complete or partial split in associating liver partition and portal vein ligation for staged hepatectomy: A systematic review and meta-analysis. World J Gastroenterol. 2019;25:6016-6024. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 5] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 63. | Kim D, Cornman-Homonoff J, Madoff DC. Preparing for liver surgery with "Alphabet Soup": PVE, ALPPS, TAE-PVE, LVD and RL. Hepatobiliary Surg Nutr. 2020;9:136-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 64. | Peng PD, Hyder O, Bloomston M, Marques H, Corona-Villalobos C, Dixon E, Pulitano C, Hirose K, Schulick RD, Barroso E, Aldrighetti L, Choti M, Shen F, Kamel I, Geschwind JF, Pawlik TM. Sequential intra-arterial therapy and portal vein embolization is feasible and safe in patients with advanced hepatic malignancies. HPB (Oxford). 2012;14:523-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 65. | Raoul JL, Forner A, Bolondi L, Cheung TT, Kloeckner R, de Baere T. Updated use of TACE for hepatocellular carcinoma treatment: How and when to use it based on clinical evidence. Cancer Treat Rev. 2019;72:28-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 423] [Article Influence: 60.4] [Reference Citation Analysis (0)] |
| 66. | Ogata S, Belghiti J, Farges O, Varma D, Sibert A, Vilgrain V. Sequential arterial and portal vein embolizations before right hepatectomy in patients with cirrhosis and hepatocellular carcinoma. Br J Surg. 2006;93:1091-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 184] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 67. | Aoki T, Imamura H, Hasegawa K, Matsukura A, Sano K, Sugawara Y, Kokudo N, Makuuchi M. Sequential preoperative arterial and portal venous embolizations in patients with hepatocellular carcinoma. Arch Surg. 2004;139:766-774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 137] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 68. | Yoo H, Kim JH, Ko GY, Kim KW, Gwon DI, Lee SG, Hwang S. Sequential transcatheter arterial chemoembolization and portal vein embolization vs portal vein embolization only before major hepatectomy for patients with hepatocellular carcinoma. Ann Surg Oncol. 2011;18:1251-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 94] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 69. | Hwang S, Lee SG, Ko GY, Kim BS, Sung KB, Kim MH, Lee SK, Hong HN. Sequential preoperative ipsilateral hepatic vein embolization after portal vein embolization to induce further liver regeneration in patients with hepatobiliary malignancy. Ann Surg. 2009;249:608-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 134] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 70. | Le Roy B, Perrey A, Fontarensky M, Gagnière J, Abergel A, Pereira B, Lambert C, Boyer L, Pezet D, Chabrot P, Buc E. Combined Preoperative Portal and Hepatic Vein Embolization (Biembolization) to Improve Liver Regeneration Before Major Liver Resection: A Preliminary Report. World J Surg. 2017;41:1848-1856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 71. | Laurent C, Fernandez B, Marichez A, Adam JP, Papadopoulos P, Lapuyade B, Chiche L. Radiological Simultaneous Portohepatic Vein Embolization (RASPE) Before Major Hepatectomy: A Better Way to Optimize Liver Hypertrophy Compared to Portal Vein Embolization. Ann Surg. 2020;272:199-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 72. | Kobayashi K, Yamaguchi T, Denys A, Perron L, Halkic N, Demartines N, Melloul E. Liver venous deprivation compared to portal vein embolization to induce hypertrophy of the future liver remnant before major hepatectomy: A single center experience. Surgery. 2020;167:917-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 73. | Vilgrain V, Pereira H, Assenat E, Guiu B, Ilonca AD, Pageaux GP, Sibert A, Bouattour M, Lebtahi R, Allaham W, Barraud H, Laurent V, Mathias E, Bronowicki JP, Tasu JP, Perdrisot R, Silvain C, Gerolami R, Mundler O, Seitz JF, Vidal V, Aubé C, Oberti F, Couturier O, Brenot-Rossi I, Raoul JL, Sarran A, Costentin C, Itti E, Luciani A, Adam R, Lewin M, Samuel D, Ronot M, Dinut A, Castera L, Chatellier G; SARAH Trial Group. Efficacy and safety of selective internal radiotherapy with yttrium-90 resin microspheres compared with sorafenib in locally advanced and inoperable hepatocellular carcinoma (SARAH): an open-label randomised controlled phase 3 trial. Lancet Oncol. 2017;18:1624-1636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 620] [Cited by in RCA: 594] [Article Influence: 74.3] [Reference Citation Analysis (0)] |
| 74. | Vouche M, Lewandowski RJ, Atassi R, Memon K, Gates VL, Ryu RK, Gaba RC, Mulcahy MF, Baker T, Sato K, Hickey R, Ganger D, Riaz A, Fryer J, Caicedo JC, Abecassis M, Kulik L, Salem R. Radiation lobectomy: time-dependent analysis of future liver remnant volume in unresectable liver cancer as a bridge to resection. J Hepatol. 2013;59:1029-1036. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 219] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 75. | Malhotra A, Liu DM, Talenfeld AD. Radiation Segmentectomy and Radiation Lobectomy: A Practical Review of Techniques. Tech Vasc Interv Radiol. 2019;22:49-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 76. | Lewandowski RJ, Donahue L, Chokechanachaisakul A, Kulik L, Mouli S, Caicedo J, Abecassis M, Fryer J, Salem R, Baker T. (90) Y radiation lobectomy: Outcomes following surgical resection in patients with hepatic tumors and small future liver remnant volumes. J Surg Oncol. 2016;114:99-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 85] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 77. | Andel D, Dassen MG, Reinders-Hut MTM, Peters NA, Kranenburg OW, Lam MGEH, Hagendoorn J, Rinkes IHMB. Surgical outcomes of major hepatectomy following "radiation lobectomy" for hepatic malignancies and insufficiently functional future liver remnant: initial experience. Br J Surg. 2020;107:e609-e610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |