Published online Aug 27, 2019. doi: 10.4254/wjh.v11.i8.613
Peer-review started: March 26, 2019
First decision: June 16, 2019
Revised: July 3, 2019
Accepted: July 16, 2019
Article in press: July 17, 2019
Published online: August 27, 2019
Processing time: 153 Days and 16.3 Hours
Porto sinusoidal vascular liver disease (PSVD) and portal vein thrombosis (PVT) are distinct vascular liver diseases characterized, respectively, by an intrahepatic and a prehepatic obstacle to the flow in the liver portal system. PVT may also occur as a complication of the natural history of PSVD, especially if a prothrombotic condition coexists. In other cases, it is associated to local and systemic pro-thrombotic conditions, even if its cause remains unknown in up to 25% despite an active search. In our opinion, the presence of PSVD should be suspected in patients with PVT especially in those with PVT “sine causa” and the active search of this condition should be included in their diagnostic work-out. However, sometimes the diagnosis of pre-existing PSVD is very hard. Biopsy cannot be fully discriminant as similar histological data have been described in both conditions. Liver stiffness may help as it has been shown to be higher in PSVD than in “pure” PVT, due to the presence of sclerosis in the portal venous radicles observable in PSVD patients. Nevertheless, comparing liver stiffness between PVT and PSVD has until now been restricted to very limited series of patients. In conclusion, even if it is still totally hypothetical, our point of view may have clinical consequences, especially when deciding to perform a liver biopsy in patients with a higher liver stiffness and suspending the anticoagulation in patients with PVT and no detectable prothrombotic factors.
Core tip: Porto sinusoidal vascular liver disease (PSVD) and chronic portal vein thrombosis (PVT) are vascular liver diseases. This review aims to discuss the possibility that (PSVD) should be suspected in patients with PVT and to analyse the possible diagnostic tools able to differentiate between these two conditions. Moreover, the review focuses on the possible and relevant clinical consequences of missing a diagnosis of PSVD in patients with PVT.
- Citation: Gioia S, Nardelli S, Ridola L, d’Amati G, Riggio O. Is porto sinusoidal vascular disease to be actively searched in patients with portal vein thrombosis? World J Hepatol 2019; 11(8): 613-618
- URL: https://www.wjgnet.com/1948-5182/full/v11/i8/613.htm
- DOI: https://dx.doi.org/10.4254/wjh.v11.i8.613
Porto sinusoidal vascular liver disease (PSVD) is a vascular liver disease characterized by portal hypertension in the absence of cirrhosis and other causes of liver disease[1]. Contrary to the Eastern World and Developing Countries, in the Western World PSVD is considered infrequent. However, a low grade of suspicion can contribute, at least partially, to the low frequency of PSVD in Western Countries. Indeed, in the last years, the deepening of the knowledge on PSVD and its clinical presentation led to the recognition of an increasing number of cases and to the awareness that, in the past, some patients affected by PSVD were misdiagnosed and classified as affected by cryptogenic cirrhosis[2]. Moreover, it has been recently shown that a number of disorders and drugs (Table 1) are associated with PSVD[1,2], and this observation suggests that an active search of signs of portal hypertension in patients affected by those diseases could lead to the identification of PSVD patients.
| Thrombophilia |
| Myeloproliferative neoplasm |
| Protein S or C deficiency |
| Antiphospholipid antibodies |
| Lupus anticoagulant |
| Factor V Leiden |
| Prothrombin mutation |
| Hematologic disease |
| Myeloproliferative neoplasm (polycythemia vera, chronic myelogenous leukaemia, essential thrombocythemia) |
| Myeloid metaplasia |
| Lymphoproliferative conditions (Hodgkin’s disease, non-Hodgkin’s lymphoma, chronic lymphocytic leukaemia and multiple myeloma) |
| Spherocytosis |
| Genetic disorders |
| Cystic fibrosis |
| Adams Oliver syndrome |
| Turner‘s disease |
| Autoimmune disease |
| Rheumatoid arthritis |
| Systemic lupus erythematosus |
| Systemic sclerosis |
| Scleroderma |
| Gut diseases |
| Celiac disease |
| Inflammatory bowel disease |
| Drug and toxics |
| Oxaliplatin |
| Azathioprine |
| 6-thioguanine |
| Arsenic |
| Busulfan |
| Cytosine arabinoside |
| Cyclophosphamide |
| Bleomycin |
| Chlorambucil |
| Doxyrubicin |
| Carmustine |
| Acquired and congenital immunodeficiency |
| Human immunodeficiency virus |
| Primary antibody-deficiency syndrome |
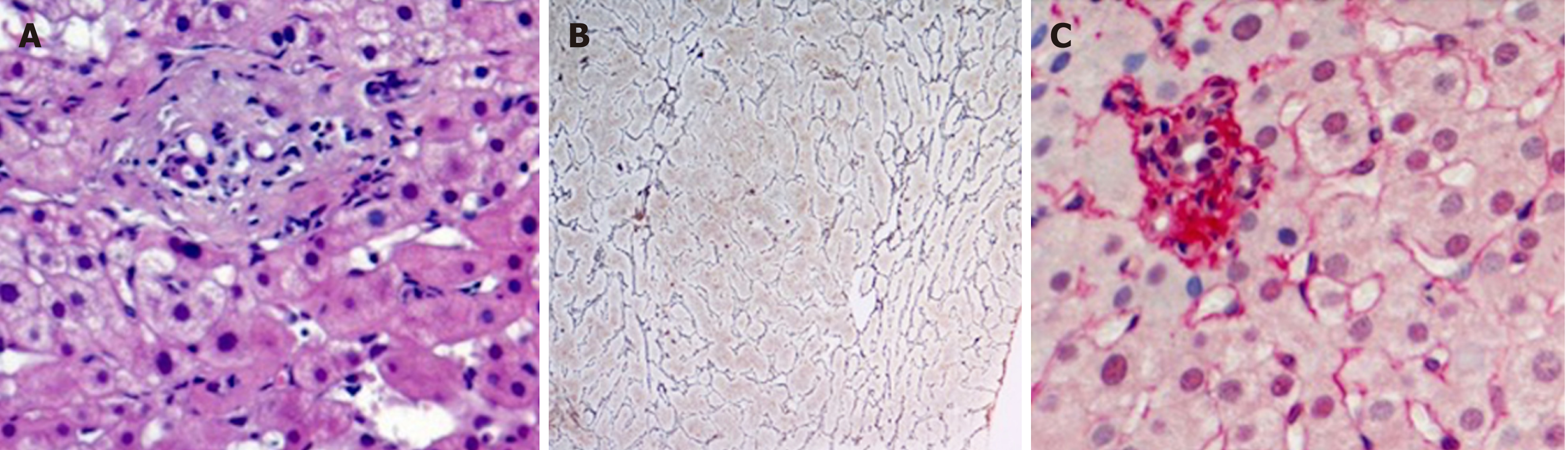
This review aims to discuss the possibility that PSVD should be suspected in patients with PVT. In fact, a particular relationship exists between PSVD and PVT. In a considerable number of patients[3], about 40%, PSVD is associated to prothrombotic conditions, including myeloproliferative disorders. The prothrombotic state may represent a pathogenic factor leading the occlusion of the small branches of the portal vein, contributing to the so called obliterative portal venopathy (Figure 1A), which is considered the primary histological lesion of PSVD[4]. In turn, in patients with PSVD, the prothrombotic state may favour, together with the low portal flow, the development of an extrahepatic portal vein thrombosis. PVT actually occurs in up to 40% of PSVD patients[5,6], especially if a prothrombotic condition coexists. Whether the anticoagulant therapy is useful for the prevention of portal vein thrombosis or to improve the natural history of porto sinusoidal vascular liver disease idiopathic non-cirrhotic portal hypertension is still unknown[7].
The strong relationship between PSVD, hypercoagulability and incidence of PVT led to the hypothesis that a subset of patients with acute or chronic PVT could be affected by a pre-existing, undiagnosed PSVD. Actually, despite an active search[8] of local or systemic predisposing conditions (Table 2), the cause of PVT is not recognizable in up to 25% of the patients. Moreover, in patients in whom a PVT is diagnosed, the evidence of a prothrombotic state usually leads to the diagnosis of a PVT secondary to that condition without taking into consideration the possibility of a pre-existing and undiagnosed PSVD. The possibility of missing a diagnosis of PSVD in a patient with PVT, especially if acute, may have relevant clinical consequences.
| Systemic prothrombotic conditions | Recommended checks |
| Inherited | Resistance to activated C protein/molecular biology for |
| Factor V Leiden | G1691A polymorphism |
| Prothrombin gene mutation | Molecular biology for G20210A polymorphism |
| Protein C, protein S and antithrombin deficiency | Ratio with F II, V, VII or X after correction for vitamin K deficiency; family survey (recommended) |
| Acquired | |
| Antiphospholipid syndrome | Anticardiolipin ELISA; LLAC |
| Myeloproliferative neoplasm (MPN) | JAK2 and CALR mutations; osteomidullary biopsy |
| Paroxysomal nocturnal hemoglobinuria | Flow cytometry (deficient cells of CD55 and CD59) |
| Oral contraceptive use | Anamnesis |
| Obesity | BMI > 30 kg/m2 |
| CMV infection | CMV IgG, CMV IgM |
| Pregnancy | Beta-HCG, anamnesis |
| Celiac disease | Anti-transglutaminase IgA/IgG |
| Local prothrombotic conditions | Anamnesis and radiological examination |
| Surgery and trauma | |
| Umbilical cannulation | |
| Splenectomy, cholecystectomy | |
| Hepatic resection | |
| Abdominal trauma | |
| Bariatric surgery | |
| Portosystemic shunts including TIPS | Anamnesis and radiological examination |
| Inflammatory and infectious diseases | |
| Neonatal omphalitis | |
| Appendicitis, diverticulitis, pancreatitis | |
| Inflammatory bowel disease | |
| Cholecystitis, cholangitis | |
| Other | |
| Cirrhosis | |
| Porto sinusoidal vascular liver disease | |
Unfortunately, the distinction between these two conditions is not so easy and well defined, and the usefulness of liver biopsy to establish a differential diagnosis is questionable. In fact, while liver biopsy is adequate to exclude the presence of cirrhosis in patients with portal hypertension, the specific diagnostic features of PSVD are much more difficult to recognise[2]. More to the point, the histological distinction between the modification of the liver structure occurring in patients with PSVD (an intrahepatic cause of low portal perfusion) or PVT (in which the low portal perfusion is due to extrahepatic obstruction) may be difficult. Studies reporting a direct comparison of the histology features of PSVD and PVT are rare, mainly because the patients with PVT are not usually submitted to liver biopsy. Moreover, some modifications of liver histology, such as nodular regeneration and sinusoidal dilatation, could be due to compensatory adaptation to the reduction of portal flow that is common to both conditions. Verheij et al[4] compared liver biopsies from 70 patients with PSVD and 23 patients with PVT and observed that phlebosclerosis (i.e. obliterative portal venopathy), nodular regeneration and portal tract remnants (Figure 1A-C) were significantly more frequent in patients with PSVD than in those with PVT, while portal vein dilatation was more frequently observed in patients with PVT. These results are in line with the hypothesis proposed by Wanless, which considers PSVD a microvascular disorder resulting from injury to the small portal vein branches, whereas PVT is, by definition, a macrovascular disorder. However, the applicability of the above described observations to the distinction of PVT secondary or not to PSVD in the single patient is arguable. In fact, histological features, such as sinusoidal dilatation, para-portal shunts and increased portal vessels, although with different prevalence, are actually observed in both conditions. Moreover, the inclusion of patients affected by a pre-existing PSVD in the PVT group in the study by Verheji et al[4] cannot be completely ruled out. Consequently, due to this existing bias, it is hard to really differentiate the first and the second group on the basis of histology.
Indeed, luminal narrowing and disappearance or sclerosis of the portal venous radicles are typical changes observed in the fibrotic portal tract of PSVD patients[9]. Moreover, according to the endothelial-mesenchymal transition theory proposed by Sato et al[10], which would explain in part the pathogenesis of the obliterative portal venopathy, an endothelial dysfunction is present in patients with PSVD. The vascular endothelial cells of portal venules acquire myofibroblastic features and start to synthesize type I collagen. Thus, as a consequence of the presence of a certain degree of liver fibrosis in patients with PSVD, it can be hypothesised that the use of liver stiffness measurement would be helpful to distinguish patients with PSVD and patients with PVT. The latter patients, at least theoretically, would be characterized by a lower liver stiffness. A higher liver stiffness was actually observed in a group of 22 patients affected by regenerative nodular hyperplasia in comparison with 13 patients with PVT[11,12].
In patients with PVT, especially if acute, the correct identification of a pre-existing PSVD as the condition predisposing to PVT has relevant practical therapeutic implications. In fact, even in the absence of consistent clinical studies, in patients with PSVD, a lifelong anticoagulation is strongly suggested in presence of a prothrombotic condition and/or a portal vein thrombosis[13]. On the contrary, current guidelines on non-cirrhotic PVT do not suggest prolonging anticoagulation treatment lifelong in the absence of a prothrombotic state and/or extension of the thrombosis to mesenteric and splenic veins[13]. Thus, in a patient with PVT and an underlying undiagnosed PSVD without a prothrombotic state and without extension of the thrombosis to mesenteric and splenic veins, the anticoagulant therapy could be erroneously stopped once the PVT is resolved. However, as in PSVD, the slowing down of the portal flow is probably the principal condition leading to PVT, and as this condition is irreversible, the stop of anticoagulation may induce the rapid relapse of PVT.
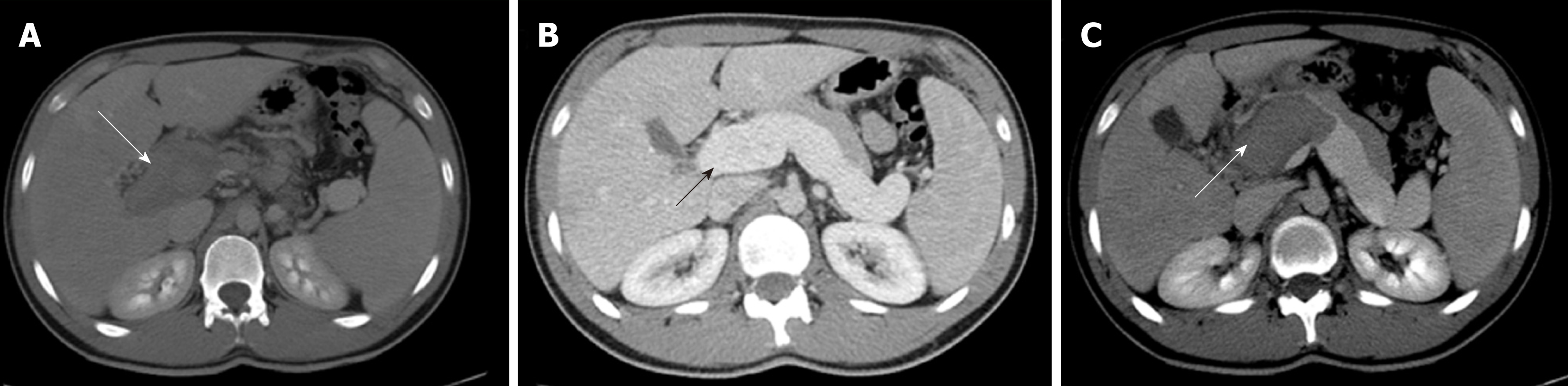
Figure 2 reports the computed tomography scan obtained in a 17-year-old male patient referred to our ward for acute portal vein thrombosis. Because of the negativity of any predisposing cause of portal vein thrombosis, including acquired and congenital prothrombotic states, he was treated with oral anticoagulation for only 1 year, and complete portal vein recanalization was documented at the computed tomography scan. However, after the withdrawal of anticoagulation, the patient had a rapid relapse of PVT with the extension of the thrombosis to splenic vein. The patient was firstly treated with low-molecular-weight-heparin and after with systemic thrombolysis with alteplase reaching the complete resolution of the thrombosis. With suspicion of porto sinusoidal vascular liver disease, the patient was submitted to liver biopsy, and finally the diagnosis of PSVD was made by observing perivenular and perisinusoidal fibrosis, sinusoidal dilatation, para-portal shunts and nodular regenerative hyperplasia.
In conclusion, PSVD is probably a not so rare condition that should be actively searched not only by the hepatologists in the patients with signs of portal hypertension but also by the specialists who manage the patients affected by diseases associated with PSVD. Among the conditions associated with PSVD, PVT could be also included, and the suspicion of a pre-existing and missed PSVD should arise in any patient with PVT included (and maybe especially) in those affected by a prothrombotic state. However, more studies are needed to clarify the distinctive features of PSVD and PVT both histologically and elastographically, and new studies are needed to clarify the indications and the benefits of anticoagulant therapy in patients affected by PSVD.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Italy
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Garbuzenko DV S-Editor: Ma YJ L-Editor: Filipodia E-Editor: Ma YJ
| 1. | Schouten JN, Garcia-Pagan JC, Valla DC, Janssen HL. Idiopathic noncirrhotic portal hypertension. Hepatology. 2011;54:1071-1081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 222] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 2. | Riggio O, Gioia S, Pentassuglio I, Nicoletti V, Valente M, d'Amati G. Idiopathic noncirrhotic portal hypertension: current perspectives. Hepat Med. 2016;8:81-88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 3. | Hillaire S, Bonte E, Denninger MH, Casadevall N, Cadranel JF, Lebrec D, Valla D, Degott C. Idiopathic non-cirrhotic intrahepatic portal hypertension in the West: a re-evaluation in 28 patients. Gut. 2002;51:275-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 206] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 4. | Verheij J, Schouten JN, Komuta M, Nevens F, Hansen BE, Janssen HL, Roskams T. Histological features in western patients with idiopathic non-cirrhotic portal hypertension. Histopathology. 2013;62:1083-1091. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 5. | Gioia S, Nardelli S, Pasquale C, Pentassuglio I, Nicoletti V, Aprile F, Merli M, Riggio O. Natural history of patients with non cirrhotic portal hypertension: Comparison with patients with compensated cirrhosis. Dig Liver Dis. 2018;50:839-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 52] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 6. | Siramolpiwat S, Seijo S, Miquel R, Berzigotti A, Garcia-Criado A, Darnell A, Turon F, Hernandez-Gea V, Bosch J, Garcia-Pagán JC. Idiopathic portal hypertension: natural history and long-term outcome. Hepatology. 2014;59:2276-2285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 138] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 7. | Valla DC, Cazals-Hatem D. Vascular liver diseases on the clinical side: definitions and diagnosis, new concepts. Virchows Arch. 2018;473:3-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 8. | DeLeve LD, Valla DC, Garcia-Tsao G; American Association for the Study Liver Diseases. Vascular disorders of the liver. Hepatology. 2009;49:1729-1764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 739] [Cited by in RCA: 652] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 9. | Guido M, Alves VAF, Balabaud C, Bathal PS, Bioulac-Sage P, Colombari R, Crawford JM, Dhillon AP, Ferrell LD, Gill RM, Hytiroglou P, Nakanuma Y, Paradis V, Quaglia A, Rautou PE, Theise ND, Thung S, Tsui WMS, Sempoux C, Snover D, van Leeuwen DJ; International Liver Pathology Study Group. Histology of portal vascular changes associated with idiopathic non-cirrhotic portal hypertension: nomenclature and definition. Histopathology. 2019;74:219-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Sato Y, Nakanuma Y. Role of endothelial-mesenchymal transition in idiopathic portal hypertension. Histol Histopathol. 2013;28:145-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 11. | Vuppalanchi R, Mathur K, Pyko M, Samala N, Chalasani N. Liver Stiffness Measurements in Patients with Noncirrhotic Portal Hypertension-The Devil Is in the Details. Hepatology. 2018;68:2438-2440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 12. | Gioia S, Nardelli S, Riggio O. Letter to the Editor: Liver Stiffness in Noncirrhotic Portal Hypertension: The Devil Is in the Diagnosis. Hepatology. 2019;70:444-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Vascular diseases of the liver. J Hepatol. 2016;64:179-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 428] [Cited by in RCA: 532] [Article Influence: 59.1] [Reference Citation Analysis (0)] |