Published online Jul 26, 2020. doi: 10.4252/wjsc.v12.i7.585
Peer-review started: December 31, 2019
First decision: April 2, 2020
Revised: May 27, 2020
Accepted: May 29, 2020
Article in press: May 29, 2020
Published online: July 26, 2020
Processing time: 208 Days and 3.8 Hours
Stem cells play a key role in tissue regeneration due to their self-renewal and multidirectional differentiation, which are continuously regulated by signals from the extracellular matrix (ECM) microenvironment. Therefore, the unique biological and physical characteristics of the ECM are important determinants of stem cell behavior. Although the acellular ECM of specific tissues and organs (such as the skin, heart, cartilage, and lung) can mimic the natural microenvironment required for stem cell differentiation, the lack of donor sources restricts their development. With the rapid development of adipose tissue engineering, decellularized adipose matrix (DAM) has attracted much attention due to its wide range of sources and good regeneration capacity. Protocols for DAM preparation involve various physical, chemical, and biological methods. Different combinations of these methods may have different impacts on the structure and composition of DAM, which in turn interfere with the growth and differentiation of stem cells. This is a narrative review about DAM. We summarize the methods for decellularizing and sterilizing adipose tissue, and the impact of these methods on the biological and physical properties of DAM. In addition, we also analyze the application of different forms of DAM with or without stem cells in tissue regeneration (such as adipose tissue), repair (such as wounds, cartilage, bone, and nerves), in vitro bionic systems, clinical trials, and other disease research.
Core tip: Decellularized adipose matrix (DAM) is widely used in soft tissue regeneration because it has unique biological and physical properties and can provide a natural microenvironment for the growth and differentiation of stem cells. There have been many studies on DAM, and our objective is to comprehensively describe the preparation, characterization and application of DAM from the perspective of stem cells. We also describe the problems that still need to be solved in DAM research and possible future developments.
- Citation: Yang JZ, Qiu LH, Xiong SH, Dang JL, Rong XK, Hou MM, Wang K, Yu Z, Yi CG. Decellularized adipose matrix provides an inductive microenvironment for stem cells in tissue regeneration. World J Stem Cells 2020; 12(7): 585-603
- URL: https://www.wjgnet.com/1948-0210/full/v12/i7/585.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v12.i7.585
Soft tissue defects caused by trauma, tumors, and aging are often seen in clinical work, and tissue regeneration is undoubtedly one of the biggest challenges. Stem cell therapy has always played an important role in the field of regenerative medicine[1-3]. Stem cells achieve tissue metabolism and regeneration of post-traumatic defects through two unique attributes: (1) The ability to self-renew in the process of symmetric division; and (2) The ability to multidirectionally differentiate in the process of asymmetric division[4]. Although stem cells play an important role in soft tissue regeneration, risks and challenges also exist. Stem cells often require extensive expansion in vitro, which increases the risk of shortened telomeres, impaired function, and contamination[5]. It is common for stem cells to fail to stabilize in the recipient region after implantation, leading to a poor survival.
Therefore, from the application perspective of tissue regeneration, what stem cells need more is a natural biomaterial scaffold. It can provide stem cells with a microenvironment for growth and support for their colonization, adhesion, proliferation, and differentiation[6-10]. The dynamic and specific microenvironment of stem cell proliferation and differentiation is called a niche. The main component of the niche is the extracellular matrix (ECM), which can dynamically regulate the behavior of stem cells and provide extracellular clues for stem cell recognition[6,11]. The ECM is composed of various collagens, glycoproteins, and growth factors and seems to be a static network structure, but it is actually in a process of continuous remodeling with dynamic interaction with stem cells[12,13]. Generally, stem cell proliferation and differentiation are accompanied by changes in the ECM structure. For example, stem cells bind to matrix protein residues to change local conformation[14,15], or stem cell remodeling reveals hidden binding sites of the ECM to promote self-adhesion and proliferation[16,17].
Despite the advances in bionic technology and the rapid development of polymer materials science, there is still a huge challenge to fully simulate the biological properties of the ECM. Most artificial scaffolds fail to meet the requirements of biologically active vectors due to their lack of the ability to induce stem cell differentiation and the potential for dynamic interaction with cells[18-20]. Therefore, the acellular matrix of the target tissue/organ is an ideal bioactive scaffold. A cell-free, natural ECM scaffold can be obtained through a previously developed protocol. It is characterized by a rich biomolecular and unique three-dimensional (3D) structure that can play a key role even if the acellular matrix differs from the anatomical region of the donor site[21].
At present, there are many studies on the use of xenogeneic and allogeneic acellular matrix for different hosts[22,23]. The risk of immunogenic residues limits the application of xenogeneic tissues[24,25]. Human allogeneic tissues may be the most desirable source of the ECM. Adipose tissue comes from a wide variety of sources, and lipoaspirate is largely discarded every year as medical waste. With the rapid development of adipose tissue engineering, many researchers have tried to develop better acellular solutions to obtain decellularized adipose matrix (DAM)[26-28]. DAM continues to integrate with surrounding soft tissues and plays an important role in the entire regeneration process of the recipient area[29].
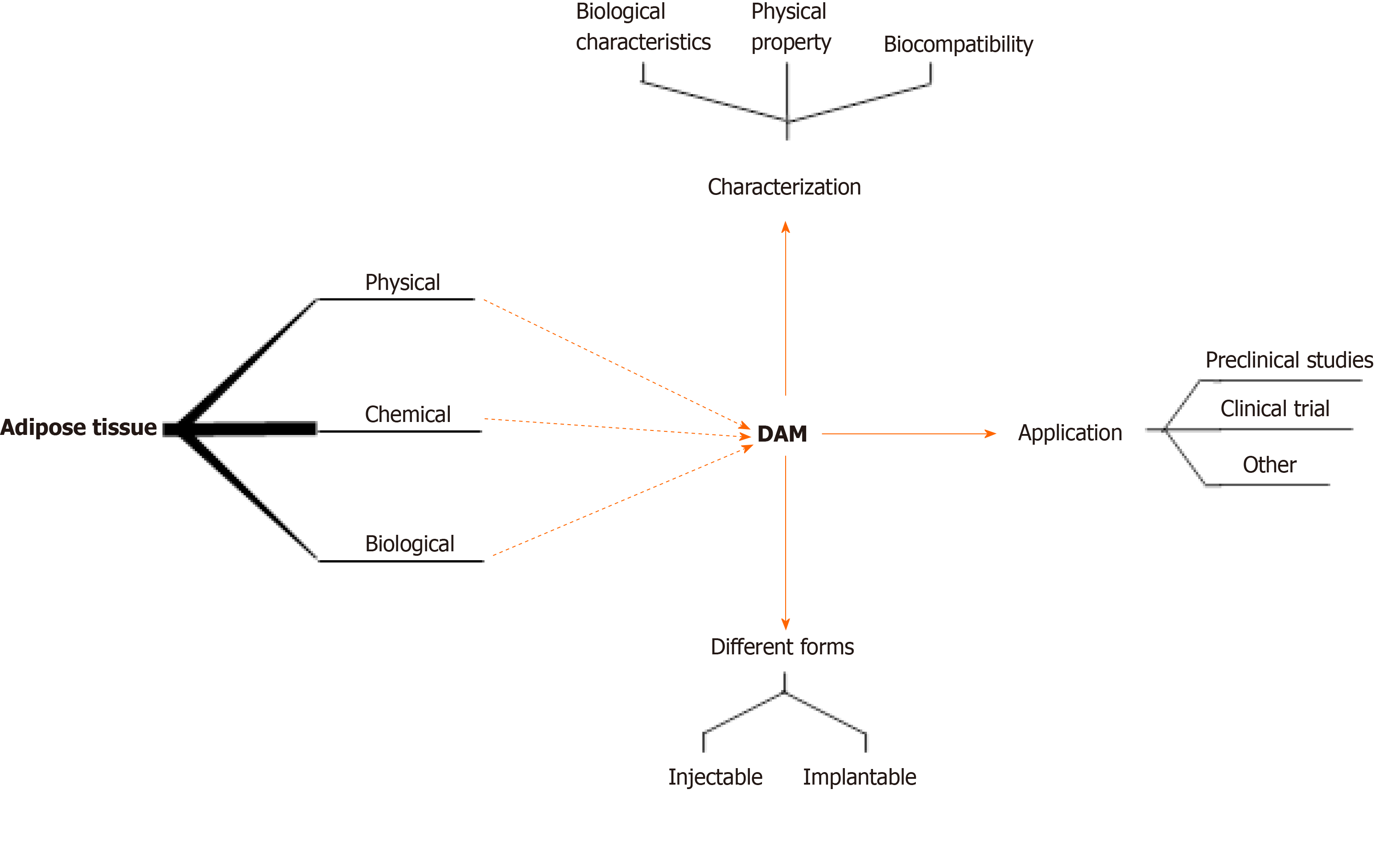
Currently, there are many protocols for the preparation of DAM. Different preparation methods have different effects on key components of DAM, and further affect the growth of stem cells and regeneration of soft tissues[30-34]. This review outlines the importance of DAM to provide an inductive microenvironment for stem cells in tissue regeneration. In particular, considering the DAM for tissue engineering purposes, the different decellularization methods used are fully described (Figure 1). In addition, the problems that still need to be addressed with regard to DAM are also described, as well as possible future developments of these emerging bioscaffold materials.
A literature search was conducted using the PubMed Advanced Search Builder. An advanced search was performed using “decellularized adipose tissue OR adipose-derived matrix OR acellular adipose matrix OR decellularized adipose matrix” as the title elements, and identified 236 studies. After further analysis and evaluation on whether the title and abstract involve fat-derived ECM and whether the article is written in English, a total of 75 studies were included.
The ECM is a 3D complex network structure composed of various collagens and glycosaminoglycans (GAGs), and provides effective biological information for the growth and differentiation of stem cells, and enables cell-cell and cell-ECM dynamic interaction through the establishment of a natural ecological microenvironment[35]. Stem cells continue to reshape the microenvironment created by the ECM, while the reshaped the ECM also constantly changes the behavior of stem cells[13]. This can keep the growth of stem cells in equilibrium with the degradation of the ECM and play a continuous and stable role in the entire tissue regeneration process[36]. At present, the ECM of various tissues including the skin[37], cartilage[38], bone[39], tendon[40,41], skeletal muscle[42,43], blood vessels[44,45], nerves[35,46], cornea[47], heart valves[48,49], myocardium[50,51], lung[52,53], liver[54,55], kidney[56,57], small intestine[58], and bladder[59] has been widely used in clinical or preclinical research in various fields.
In recent decades, the DAM extracted from a large amount of waste adipose tissue has aroused interest among researchers because of its abundant sources and excellent potential in soft tissue regeneration[60]. A large amount of adipose tissue can be obtained by using the developed method of degreasing and decellularization[26]. The DAM, which provides a natural microenvironment for the growth of stem cells [especially adipose-derived stem cells (ASCs)], has the following characteristics. First, the complex structure is composed of collagens I[22,61,62], IV[26,61,63,64], and VI[65], laminin[22,26,61,62,66,67], fibronectin[34,68], elastin[28], GAGs[22,28,62,63,69], and other biologically active macromolecules. Fibrillar collagen and glycoproteins provide structural stretch resistance and resilience[70], and play an important role in the entire dynamic remodeling process of stem cells. Second, the structure contains growth factors such as vascular endothelial growth factor (VEGF)[22,63,69,71], basic fibroblast growth factor (bFGF)[22,63,71], and transforming growth factor (TGF)-β[23], which are associated with specific ECM domains or proteins and play an irreplaceable role in the entire process of soft tissue regeneration[72,73].
In addition, there are different names about DAM, including decellularized adipose tissue[26,74-77], adipose-derived matrix[23,24], and acellular adipose matrix[78]. For the convenience of explanation, this article collectively names DAM from adipose tissue of different sources (including human, pig, mouse, etc.).
There have been many studies on DAM (Table 1). Different preparation methods result in the retention or loss of DAM key components to varying degrees, and affect the growth of stem cells and regeneration status of soft tissues[65,79]. The goal of DAM preparation is to remove all immunogenic components (such as nucleic acids and fragments) from all cells, while retaining the biologically active components of the ECM (including collagens, proteins, growth factors, and GAGs) and suitable 3D structure and mechanical properties, to provide host stem cell growth and differentiation microenvironment after transplantation[80]. However, all current decellularization schemes will inevitably cause different degrees of damage to the structure and composition of DAM[60].
| Decellularization methods | Sterilization methods | Refs. | ||
| Physical treatments | Chemical treatments | Biological treatments | ||
| Freeze–thaw, 3 cycles (-80 °C to 37 °C) | 99.9% isopropanol | 0.25% trypsin/0.1% EDTA 15000 U DNase, 12.5 mg RNase, 2000 U lipase | 70% ethanol/1% penicillin and streptomycin/UV light/1% antibiotic/antimycotic | [26,30,74,75,83,84,96,101,102] |
| 0.05% trypsin-EDTA 100 U/mL benzonase | 70% ethanol/1% penicillin and streptomycin | [63,66] | ||
| 0.05% trypsin 500 U/mL benzonase | 0.1% peracetic acid in 4% ethanol | [61] | ||
| 99.9% isopropanol 1 mol/L NaCl | 1 mmol/L EDTA + Lysis buffer (1% tergitol type NP-40, 0.1% SDS, 5 mmol/L EDTA, 0.4 mol/L NaCl, 50 mmol/L Tris-HCl pH 8, 1 mmol/L PMSF) | 70% ethanol/1% penicillin and streptomycin | [65] | |
| 100% isopropanol | 0.25% trypsin-EDTA; 1 mL DNase + 1 mL RNase + 2 mL lipase | 70% ethanol/1% penicillin and streptomycin | [135] | |
| 0.5 mol/L NaCl/1 mol/L NaCl/isopropanol/Triton X-100 | 0.25% trypsinEDTA | 1% penicillin and streptomycin | [69,71,93] | |
| Freeze-thaw, 3 cycles (-80 °C to 37 °C) + ultrasonic | 0.5% SDS + 100% ethanol | — | 100% ethanol | [25] |
| Freeze-thaw, 35 cycles (-80 °C to 37 °C) + homogenization, 5 min (12000 r/min) | 1% Triton X100 + 100% isopropanol + 1 mol/L NaCl | 100 U/mL DNase 100 μg/mL RNase | — | [68] |
| Freezethaw, 4 cycles (-80 °C to 37 °C) + ultrasonic | 96% ethanol 0.5% SDS | 0.05% trypsin/0.05 mmol/L EDTA + DNase | — | [87] |
| Freezethaw, 5 cycles (-80 °C to 37 °C) | Isopropanol | 0.25% trypsin/0.1% EDTA DNase I + RNase A | Ethylene oxide | [136] |
| Freeze-thaw, 5 cycles (liquid nitrogen to 37 °C) | 99.9% isopropanol | 0.05% trypsin-EDTA 20 ng/mL DNase I + 20 ng/mL RNase | 1% penicillin and streptomycin | [67] |
| Freezethaw, 4 or 5 cycles (liquid nitrogen to Room temperature) | 0.5 mol/L acetic acid | — | — | [88] |
| Freezethaw, 35 cycles (liquid nitrogen to Room temperature) | 0.1% SDS | 0.05% trypsin + 0.05% EDTA + 20 ng/mL DNase I + 20 ng/mL RNase | 1% penicillin and streptomycin | [89] |
| Freezethaw, 618 cycles (liquid nitrogen to room temperature) | 0.1% sodium azide + 1 mol/L NaCl + 4% sodium deoxycholate | 2000 K units DNase | — | [78] |
| 1% Triton X-100 | 2000 K units DNase | |||
| Homogenization, 5 min (12000 rpm) | 1 mol/L NaCl/0.5% SDS | 0.2% DNase + 200 μg/mL RNase | — | [27,28,81] |
| — | — | — | [104,137] | |
| 0.5% SDS + 100% isopropanol | — | — | [138] | |
| Homogenization, 5 min + ultrasonic | — | 0.25% Pancreatin | Ethylene oxide | [79] |
| Homogenization, 3 min (12000 r/min) | SDS | — | — | [90] |
| 4 mol/L urea | 4 mol/L Gu | Ethylene oxide | [100,139] | |
| Homogenization (twice) | 2 mol/L urea+70% ethanol | 2 U/mL dispase II + 4 mol/L GuHCl | Dialysis against chloroform | [23,24] |
| Homogenization | 2 mol/L urea buffer | — | 70% ethanol/1% antibiotic/antimycotic solution | [65] |
| Constant stirring | 1% SDS or 2.5 mmol/L sodium deoxycholate | 2.5 mmol/L sodium deoxycholate + 500 U porcine lipase + 500 U porcine colipase | 365 nm UV light | [62,85] |
| Constant stirring | 1% SDS | 2.5 mmol/L sodium deoxycholate + 100 μg/mL lipase + 50 ng/mL colipase; 50 μg/mL DNase + 50 μg/mL RNase | Ethylene oxide | [86] |
| Mechanical processing | 0.1%, 1%, 3%, or 5% Peracetic acid + 1% Triton X-100 | 600 U DNase | — | [31,94] |
| 3% Triton X100 + 4% sodium deoxycholate + 4% ethanol/0.1% peracetic acid + 100% n-propanol | 0.02% trypsin + 0.05% EDTA | 4% ethanol + 0.1% peracetic acid | [22] | |
| SCCO2 (180 bar) | Ethanol | — | SC-CO2 | [91] |
| — | 1% sodium dodecylsulfate + 100% isopropanol | 2.5 mmol/L sodium deoxycholate + 500 U lipase + 500 U colipase | 5000 IU penicillin and 5 mg/mL streptomycin | [32] |
| 0.5% SDS + isopropanol + 0.1% peracetic acid + 4% ethanol | — | 0.1% peracetic acid+4% ethanol | [92,103] | |
| 1% Triton X-100 | 10, 20 and 100 IU/mL DNase I | — | [82] | |
| 1-propanol | Sodium deoxycholate | Peracetic acid | [33] | |
| Organic solvent + surfactant/ethanol-based solution | — | Peracetic acid | [34,64] | |
The current effective decellularization protocol is achieved by a combination of physical, chemical, and enzymatic methods (Table 2). Usually, the first step is to destroy the cell membrane components by physical means (freezethaw cycle[26] and homogenization[28,81]). Second, chemical methods include the use of detergents[33]/nondetergents[82] to dissolve cytoplasmic and nuclear components and alcohols (such as isopropanol[26,83,84]) to remove lipid residues. Finally, cell residues and degraded nucleic acid fragments are removed by enzymatic methods[28,68,81] (including DNase and RNase). The above steps can be combined with continuous mechanical stirring and shaking, to shorten the action time of reagents, improve efficiency, and reduce structural damage[62,85,86]. In addition, in order to avoid the immune response caused by the residues of chemical and enzymatic substances, thorough washing at each step is essential[26]. Flynn et al[26] was the first to prepare complete DAM through the above-mentioned comprehensive method. After 5 d of nondetergent solution, DAM was finally obtained with a high retention rate (30%-45% of the original amount). After a series of characterizations, components such as collagens I and IV and laminin of DAM, which are important for adipogenesis and stem cell proliferation, are retained[26]. This method has been widely used and improved by many researchers subsequently.
| Agent/method | Function or advantages | Impact or disadvantages |
| Physical | ||
| Freezing thawing | Ice crystals destroy cell membranes | Ice crystals also destroy the continuity of |
| Preserve component integrity | DAM composition and microstructure | |
| Reduce immune response | ||
| Homogenization | Fully destroy the cell membrane structure and promote dissociation from basement membrane | Mechanical shear forces break the microstructure and component continuity |
| Constant stirring | Cleave the cell membrane | Stirring forces destroy microstructure |
| Full exposure accelerates the effect of chemical agents | Mechanical properties are affected | |
| Mechanical processing | Promote cell membrane rupture and release from the basement membrane | Pressure directly destroys microstructure; ultrastructure and basement membrane integrity are destroyed |
| SC-CO2 treatment | Supercritical inert gas penetrates tissues to remove cell residues/sterilization | Entrainer may reduce structural composition; supercritical pressure may destroy the structure |
| Ultrasonic | Ultrasonically break cell membrane | - |
| Chemical | ||
| Hypotonic/hypertonic solutions | Dissociate DNA from proteins; Osmotic pressure ruptures cell membranes | Little influence on the structure and composition of DAM |
| Alcohols | ||
| Isopropanol | Cell dehydration, cell membrane lysis | DAM protein components are precipitated; destruction of ultrastructure; degreasing alone has poor effect |
| Ethanol | Effectively remove lipid residue | |
| Acids and bases | ||
| Acetic acid | Hydrolyze biomolecules to remove residual nucleic acids; little effect on the structure; better retention of GAGs components | Some collagen components are destroyed and removed; reduced strength of DAM; collagen, growth factors, and GAGs are damaged |
| Peracetic acid | Little effect on the structure and composition of DAM | |
| Nonionic detergents | ||
| Triton X-100 | Disturbing DNA–protein, lipid–lipid, and lipid–protein associations; moderate effect/stable in solution | Destruction of ultrastructure; remove GAGs |
| Agent/ Methods | Function or advantage | Impact or disadvantage |
| Ionic detergents | ||
| SDS | Effectively remove cellular nucleic acid components/destruction of cell membrane phospholipids and lipoproteins/dissolving antigen and eliminating immune complexes | Disturbing protein–protein association; growth factor removal; destroy ultrastructure, GAGs ingredients; residue of the reagent causes cytotoxicity |
| Sodium deoxycholate | ||
| Triton X-200 | ||
| Biologics | ||
| Trypsin | Cleavage of the C-side peptide bond of Arg and Lys | Remove fibronectin, elastin, and GAGs components; damage degree of DAM composition and microstructure is highly time-dependent |
| Nucleases (DNase, RNase) | Cleavage nucleotides sequence | Difficult to remove residue from DAM; residual effects on host recellarization; causes host immune response |
| Lipase and colipase | Remove residual lipids | Destruction of ultrastructure; removes GAGs; efficiency of lipid removal is low |
| EDTA | Dissociation of metal ions plays a supporting role in tissue decellularization | Destruction of protein–protein linkages; poor application alone |
The physical treatment method has had the following improvements. First, the number of cycles is increased based on three freezethaw cycles[67,68,78,87-89] or the freezing temperature is reduced from -80 to -196 °C (liquid nitrogen)[67,78,88,89] or adding ultrasonic treatment[25,87] during the freeze-thaw process. According to the research, within a certain range (1-5 times), increasing the number of freezethaw cycles will not have much effect on the microstructure of DAM, and cell debris and residues cannot be removed only by freezethaw treatment. With the increase in the number of freeze-thaw cycles (6-18 times), the microstructure of DAM is damaged[78]. Second, Choi et al[28,81] changed the freezethaw treatment to homogenization. The homogenization can quickly and fully damage the cell membrane structure, but the longterm effect of mechanical shear force destroys the microstructure of DAM and results in the loss of specific components (such as laminin)[28,81]. Subsequently, Kim et al[90] reduced the time of homogenization, to protect the integrity of DAM structure and composition[23,24]. Third, Young et al[62] replaced freezethaw and homogenization with continuous stirring or mechanical pressing, with the aim of accelerating chemical and enzymatic surface contact in the later stages to shorten reaction time[31,86]. Fourth, Wang et al[91] tried to use the advanced technology of supercritical carbon dioxide (SC-CO2), and only used ethanol as an entrainer to decellularize and degrease adipose tissue to obtain DAM. Finally, Pati et al[92] abandoned the physical processing steps and directly used chemical and enzymatic methods to obtain DAM[33,34,64].
Chemical methods also have different application modifications. Alcohol, acid/base, or ionic/nonionic detergents affect the structure and composition of DAM to varying degrees. Hypertonic saline dissociates DNA from proteins in a gentle way to achieve decellularization and has little effect on the microstructure and composition of DAM[69,71,89,93]. Although the types and concentrations of alcohols are not the same, there seems to be no significant difference in lipid removal[25]. Alcohols such as isopropanol, n-propanol, and ethanol are superior to lipase in removing lipids from tissues. They can remove lipids in a short period of time, but at the same time, they can also denature the protein components of DAM (such as collagen and LN) and destroy the ultrastructure[22]. Therefore, caution should be exercised when using them. Acidic reagents can hydrolyze the biomolecules of tissues, acetic acid may cause damage to certain collagen components and reduce the structural stability of DAM[88], while peracetic acid is a commonly used disinfectant and can also be used as a decellularizing agent because it can gently remove residual nucleic acids. It has little effect on the composition and structure of DAM[22,31,92,94]. In general, ionic detergents (including SDS and sodium deoxycholate) are more effective in removing cellular components than nonionic detergents (such as Triton X-100), but they also damage the ultrastructure of DAM, and more GAGs and growth factor components are also removed[95]. The comprehensive application of multiple chemical agents may aggravate the loss of DAM components (such as GAGs and collagen) and destruction of the structure (including mechanical properties)[60].
Nuclease, trypsin, lipase, and EDTA are widely used as biological reagents. The removal of cell debris and residual lipids or degradation of nucleic acid fragments is their main functions. It is also difficult to use enzymes alone to completely remove cell residues. In addition, the residues of enzyme reagents may further affect the growth and differentiation of stem cells, and even cause an adverse immune response in the host. Nucleases (DNase, RNase, etc.) cleave nucleotide sequences after cell membrane rupture and help remove nucleic acid residues[83,84,96-99]. Trypsin and a chelator (such as EDTA) are often used in combination. Trypsin can efficiently remove cell residues and destroy collagen, elastin, GAGs, and other components, and the damage to the structure and components of DAM also increases with the time of action (time dependent)[83,84,93,96-99]; EDTA helps DAM proteins dissociate from cells. These two reagents have a poor effect when used alone, and only when combined can they play a synergistic role, and EDTA can reduce trypsin digestion time and reduce tissue damage[22,67,89]. Lipase and co-lipase are often used in combination to remove residual lipids[32,62,85]. In addition, Poon et al[23] used guanidine alone or combined with hydrochloric acid to remove lipid residues, and the growth factors detected in DAM were well retained[24,100].
After preliminary preparation, it is important to sterilize the prepared DAM when conducting in vivo or in vitro experiments. This mainly removes bacteria and viruses. At present, the methods for sterilizing biological scaffolds mainly include alcohols, acids, ethylene oxide, UV irradiation, and SC-CO2. The prepared DAM is usually stored in a 1% penicillin and streptomycin solution at 4°C[96-98,101,102], and then sterilized with 70% ethanol solution. Some researchers use 100% ethanol alone to sterilize biological scaffolds[25]. Four percent ethanol solution and 0.1% peracetic acid are often used in combination for sterilization, with significant effect[61,92,103], and they also have little effect on the structure and composition of DAM. Wang et al[79] used ethylene oxide for sterilization, but the effect on the microstructure of DAM is unclear. However, there is no doubt that residual reagents after ethylene oxide treatment may cause adverse host reactions and affect the function of the biological scaffold after implantation. In addition, Young et al[62,85] used UV radiation for sterilization. During the sterilization process, the collagen component of DAM may be partially denatured, which may accelerate degradation of the stent material in the body[62,85]. More research is needed. As an innovative method, SC-CO2 is applied to the decellularization of adipose tissue, and it sterilizes biological materials[91]. The specific impact on biological materials needs further comparative research.
Just as researchers have developed different preparation schemes, there is currently no uniform standard procedure for characterizing DAM materials. It is impractical to remove all cellular residues, but quantitative analysis of residual cellular components (such as phospholipids and double-stranded DNA) is possible. At present, characterization of DAM generally includes: Simple evaluation of the general effects of decellularization and degreasing of materials using simple histological staining and electron microscopy (EM); and DNA quantification, biochemical analysis, and mechanical stress testing to further evaluate various aspects of DAM. This section provides a brief summary.
For detection of cell residues, the first approach is histological staining and biochemical analysis. Simple histological staining including hematoxylin and eosin and oil red O staining to roughly check whether the nuclear and lipid components are removed[26,28,31,62,104]. Immunohistochemical staining includes DAPI and Hoechst staining to determine the presence of visible nucleic acid and cellular component residues. This is followed by further biochemical tests, including DNA quantification and reverse transcription-polymerase chain reaction analysis. Gilbert et al[105] have suggested the criteria for acellular matrix: DNA content < 50 ng/mg dry weight double-stranded DNA and DNA fragment length < 200 bp. This standard may be one of the most important for the application of biological materials, because hindering the further growth and differentiation of stem cells and causing adverse host reactions may be directly related to DNA residues.
In terms of detecting the structure and composition of DAM, the microstructure and structural stability of DAM are first detected by scanning electron microscopy (SEM) and mechanical stress testing[26,92]. As mentioned above, the effect of microstructure on stem cells may be crucial, where stiffness is a key indicator[106]. The process of stem cells responding to their environment after sensing external forces is called mechanical transduction. All types of stem cells have the ability to sense the structure and stiffness of DAM[11]. Cell morphology, skeleton, and migration can interact with DAM in the short term. The more important effects of proliferation and differentiation are long-term[106]. The porosity and 3D microstructure of DAM were observed by SEM[26]. Mechanical stress tests include Young modulus, storage modulus, and loss modulus, which are used to comprehensively evaluate the mechanical integrity, elasticity and rheological properties of materials[103]. The compression mechanical test of DAM was carried out with a universal testing machine. The sample was compressed to 50% of the initial height at a low constant rate. The compressive modulus was calculated using a linear region of stress-strain curve[103]. Perea-Gil et al[107] used the atomic force microscopy to determine the mechanical properties of decellularized myocardium tissue samples, such as stiffness and Young's modulus. This is followed by further analysis of its composition by staining and biochemical analysis. Masson trichrome staining can quickly and easily detect gross collagen components. Immuno-histochemical staining can detect components such as types I, IV, and VI collagen, laminin, fibronectin, and elastin in more detail[26,62]. However, there is currently no effective detection for the quality of these proteins in DAM. The specific contents of DAM (such as TGF-β and VEGF) and GAGs can be accurately detected and analyzed by ELISA[33].
In terms of biocompatibility, coculture of DAM with mesenchymal stem cells (mainly ASCs) to detect the adhesion and proliferation of stem cells on the material is required[30,69,82]. Flynn et al[26] verified the fat regeneration potential of the acellular matrix by detecting expression of adipogenic genes such as PPARγ and C/EBPα. They also found that the GAPDH activity of DAM differed when prepared from adipose tissue with different body mass index (BMI; BMI is inversely proportional to GAPDH activity)[26]. LIVE/DEAD analysis was performed by staining living and dead cells using a combination of Calcein and EthD-1[28,62]; Kokai et al[33] used calcein AM to further stain the cocultured material, and then used laser confocal imaging to show different colors to infer the depth of the stem cell infiltration of the scaffold material. SEM at different times shows the dynamic interaction process of stem cells and materials at the microscopic level. At the same time, the authors exposed ASCs to the adipose matrix for 21 d, and then used boron-dipyrromethene staining, followed by confocal imaging to observe the increase in lipid content. After transplanting DAM with/without stem cells into the subcutaneous tissue of animals, hematoxylin and eosin, Masson, and perilipin A immunofluorescence staining were used to observe adipose tissue regeneration in vivo[33].
After degreasing and decellularizing, DAM can be processed into different shapes of biological scaffolds and used with or without stem cells. It can be roughly divided into injectable and implantable types according to different usage methods.
The main advantages of injectable DAM are convenience and noninvasiveness, including powders and gels. DAM powder is digested into gel with pepsin, and then the pH is adjusted to the normal range with sodium hydroxide solution. During use and storage, the temperature should be controlled below 10 °C to prevent curing[62]. DAM (powder or gel) is usually absorbed to varying degrees after implantation. Some researchers have tried to use polymer crosslinking, which slows down the rate of stent degradation and enhances angiogenesis and fat induction[86].
The advantage of implantable DAM is that the structural integrity is preserved, including foam and sheets. Foam-like DAM is lyophilized into porous foam by dissolving with α-amylase, which has a milder effect than pepsin. Another type of bead foam is that the DAM solution is rapidly frozen after electrospray technology, and then freeze-dried at low temperature. Chemical crosslinking is avoided, and in vivo experiments have confirmed that foamed DAM has fat-forming ability and biocompatibility[83]. The DAM is cast in a superficial mold, and a sheet-like DAM is obtained after freeze-drying. Experiments have shown good mechanical integrity and multicellular compatibility[93].
DAM can also be combined with other artificial composite materials, such as methylcellulose (MC)[100], methacrylated glycol chitosan (MGC)[84], methacrylate chondroitin sulfate (MCS)[84], and polycaprolactone (PCL)[92], to be used as stem cell growth scaffolds. It has been shown in vitro that composites can effectively enhance host stem cell invasion and angiogenesis[32]. The use of PCL/DAM composites as bio-ink for 3D printing has boomed in recent years. This open porous structured scaffold has been verified in vitro to have better oxygen and nutrient exchange capacity than ordinary DAM gels[92,103].
At present, as a biological scaffold for tissue engineering, DAM is used alone[33] or in combination with stem cells[69] in various fields, including adipose tissue engineering, wound healing, nerve repair, cartilage and bone tissue engineering, and in vitro biomimetic system research.
DAM is the most widely studied as a filler for soft tissue defects. Stem cells are seeded on DAM and injected or transplanted into subcutaneous tissue, which provides a natural microenvironment for the growth of stem cells to further promote adipogenesis and angiogenesis. After coculture of DAM and ASCs, DAM can express the adipogenesis markers PPARγ and C/EBPα (major regulators of adipogenesis and differentiation) at high levels without exogenous adipogenesis induction compared to ordinary monolayer cultures such as Triplicate tissue culture polystyrene and Cell Aggregate[26]. Expression of these two genes plays a cross-regulatory role in the entire adipogenic differentiation and plays an important role in maintaining the transformation of adipocytes to mature phenotypes. After ASCs/stromal cells were seeded in DAM microcarriers and then cultured in a low-shear fine-tuning culture system for adipogenic culture, expression of the adipogenic genes PPARγ, C/EBPα, and LPL was higher than that of ordinary gelatin microcarriers[30]. This indicates that DAM plays an important role in mediating adipogenic differentiation of ASCs. After implanting DAM loaded with ASCs into the subcutaneous tissue of rats or nude mice[69], the implanted area showed significant recellularization and angiogenesis[32,108]. This shows that DAM plays an important role in supporting stem cell infiltration and tissue remodeling. Han et al[98] used ASCs for seeding on DAM bioscaffolds, and then implanted them into the subcutaneous tissue of rats. Cell tracking technology was used to verify that the new adipose tissue originated from the host[98], and ASC-seeded DAM contributed to fat formation by promoting neovascularization and modulating the inflammatory response. In addition, research on the combination of DAM and artificial composites is also developing. For example, light crosslinked MGC/MCS and DAM form a composite biological scaffold. In vitro studies showed that DAM can also enhance the viability, retention, and lipid accumulation of ASCs. MCS composites containing 5 wt% DAM were transplanted into the subcutaneous tissue of rats. After 12 wk, it was observed that DAM seeded with ASCs significantly increased regeneration of adipocytes[83,84]. ASCs were seeded in 3D printed PCL/DAM composite bioscaffolds and then implanted into the subcutaneous tissue of nude mice. The results after 12 wk showed that there were a reasonable number of mature adipocytes and functional blood vessels in the DAM area[92].
Clinically, deep burns or large skin trauma are usually treated by flap transfer surgery. Patients often have infections, fluid loss, and electrolyte disorders[109-112]. Lee et al[113] used DAM sheet scaffold dressing to treat full-thickness skin wounds on the back of rats. The results showed that the wound healing rate, epithelial formation rate, and microvascular density were significantly higher than those of ordinary wound dressings[113]. Woo et al[114] applied a double-layer dressing (the upper layer was made of titanium dioxide and chitosan film by electrospinning, and the lower layer was DAM) to a full-thickness wound in rats. It was showed that it can accelerate the induction of fresh granulation tissue regeneration and reduce epidermal scar formation. These results indicate that the components of DAM (such as collagen, laminin, fibronectin, and GAGs) and various growth factors (such as VEGF and bFGF) can promote regeneration of the ECM in the wound area, further recruit adipose stem cells, fibroblasts, and epithelial cells to accelerate tissue reconstruction and vascular regeneration[114].
Regeneration is difficult after nerve tissue damage. Lin et al[89] used DAM containing ASCs in a rat cavernous nerve injury model, and showed the best recovery of erectile function in rats with DAM seeded with stem cells, but the results did not reach statistical significance due to large differences. However, we also saw substantial recovery of erectile function and histological improvement associated with DAM seeded with ASCs, which has potential for clinical application in the future[89].
Cartilage is difficult to repair due to its nonvascular nature and long-term wear and tear. Cartilage-derived ECM has been used in research on cartilage regeneration[115,116]. The cartilage decellularized matrix seeded with ASCs can completely repair articular cartilage defects with hyaline cartilage. At the same time, the contents of GAGs and type II collagen and biomechanical properties have been proven to be comparable to those of natural cartilage[115]. Adipose-derived mesenchymal stem cells are also used for cartilage regeneration, which differentiates into chondrocytes and can produce important proteins required for articular cartilage (such as the mucus glycoprotein Lubricin)[117-119]. This alternative treatment has proven to be effective. However, due to limited resources, the prospect of clinical application is limited. Choi et al[81] have prepared an ECM/stem cell composite, which formed cartilage-like tissue after being cultured in cartilage induction medium for 45 d, and at the same time, the expression of cartilage-specific GAGs and type II collagen increased. This shows that DAM containing endogenous active factors can support cartilage differentiation of human ASCs and help with cartilage-specific glycoprotein and collagen synthesis[81], which has potential clinical value in the synthesis of cartilage-like tissue.
Bone has significant capacity of regeneration, but patients with large-scale bone defects need surgical autogenous bone transplantation, which causes damage and infection of the donor site[120-122]. Artificial composite scaffolds are poorly biocompatible and cannot support vascular regeneration and bone tissue growth[122], while the source of acellular bone tissue is insufficient to meet clinical needs[123,124]. Mohiuddin et al[125] used DAM hydrogels to treat C57BL/6 mice for critical size repair of femoral defects. The results showed that hydrogel can enhance expression of type I collagen and osteopontin, while the hydrogel-treated group significantly enhanced bone regeneration in vivo[125].
The composition and structure of DAM show that it can mimic the natural microenvironment of stem cells and even tumor cells in the body. Research shows that when seeding on DAM or chemically modified DAM[30,31,33,64,74,84,92,96,126], ASC[76,81,84,89,93,85,82,98,104,127], smooth muscle cells[90], umbilical vein endothelial cells[90], chondrocytes[90], and neuroblasts[25] can maintain high viability and excellent proliferation, indicating that DAM may become the ideal 3D culture system for the large-scale expansion of stem cells in vitro. Dunne et al[93] used DAM as a 3D cell culture system and established a human breast cancer in vitro bionic system to study the growth of breast cancer cell lines (MCF-7, BT474, and SKBR3) and the sensitivity of anticancer drugs (lapatinib and doxorubicin). This restored the original characteristics of breast cancer cell growth in vivo, and expression of adhesion molecules in tumors in vivo. This is undoubtedly beneficial to the screening and research of antitumor drugs[93].
Most studies on DAM were preclinical studies combined with stem cells. Recent research has shown that stem-cell-free DAM can also promote adipose tissue regeneration. For the first time, Kokai et al[33] applied DAM alone to a clinical trial. DAM prepared from cadaveric human adipose tissue was applied to the subcutaneous tissue of nude mice and the subcutaneous wrist dorsum of humans. After 24 wk in vivo, the material retention rate was 44% ± 16%, and the regeneration of adipocytes could be clearly observed by immunofluorescence assays, such as perilipin A. Clinical trials evaluated biocompatibility, volume retention, and soft tissue regeneration over a 16-wk period. There were wrist pain, redness, swelling, and itching at the initial stage during the observation period, which may have been related to the initial inflammation. At 16 wk, the average graft retention was about 47%. No inflammation or necrosis was observed in pathological observation, and adipose tissue was formed around the dilated vessels. Although the study had many limitations, the role of DAM in promoting adipose regeneration was verified[33].
As an important endocrine organ, adipose tissue is closely related to many metabolic diseases such as diabetes mellitus (DM). The specific mechanism of how the ECM is involved in regulating adipocyte metabolism is unclear. The relationship between DAM and DM has been the focus of research. The ECM of adipose tissue is closely related to metabolic diseases. Factors such as hypoxia, inflammation, and fibrosis of the ECM are related to insulin resistance and DM[128-132]. After collecting visceral and subcutaneous adipose tissue, Baker et al[99] used metabolic assays to measure glucose uptake, lipolysis, and adipogenesis in adipocytes in normal cell culture and 3D DAM culture. The results show that DAM with diabetes can cause metabolic dysfunction in adipocytes of non-DM patients; nondiabetic DAM can rescue metabolic dysfunction in adipocytes of DM patients. This indicates that the ECM is involved in regulating glucose uptake and lipolysis as a target for manipulating adipose tissue metabolism[99].
Autologous fat transplantation is used in the clinic to treat vocal cord paralysis. Although biocompatible, its unpredictable absorption rate is also a limitation[133,134]. Kim et al[100] used the MC/DAM composite hydrogel for rabbit vocal cord paralysis studies, and showed that the composite hydrogel group had no early absorption after 8 wk, and the physiological symmetry of vocal cord vibration returned to normal levels. The composite hydrogel overcomes the shortcoming of the indefinite absorption rate of autologous fat transplantation. Its good biocompatibility and positive functional recovery make it possible for clinical use as stable vocal cord enhancement laryngoplasty[100].
In the past 10 years, the preparation of DAM has been improved by different decellularized techniques. The material retains the main collagen components and most structural proteins and growth factors. This biologically active system can recruit host stem cells and mimic the growth microenvironment to promote the regeneration of soft tissues. Furthermore, DAM can be processed into different forms for different applications. For DAM, preliminary progress has been made in soft tissue regeneration and metabolic diseases. The combination of DAM with stem cells or growth factors has important value in preclinical studies such as wound healing, nerve repair, cartilage and bone tissue engineering, and bionic system. It is believed that with further exploration and research on DAM, it will play a major role in the field of stem cells and soft tissue regeneration.
However, residues of chemical and enzymatic reagents in the current preparation methods are still problems to be resolved. At the same time, the microstructural destruction and component loss (such as collagen and protein) caused by decellularized reagents and inefficient acellular technology are problems that require improvement. Will it be possible to develop a high-efficiency and high-retention decellularization technology based on physical methods to obtain a more complete DAM in the future? Considering this, a deep understanding of the cascade interaction in tissue regeneration, which is induced by DAM structural proteins and infiltrating host stem cells, is required.
The authors thank each of their colleagues at the Institute of Plastic Surgery, Xijing Hospital, Fourth Military Medical University for their full cooperation and support.
Manuscript source: Invited manuscript
Specialty type: Cell and tissue engineering
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Labusca L, Musumeci G S-Editor: Dou Y L-Editor: Wang TQ E-Editor: Xing YX
| 1. | Labusca L, Herea DD, Mashayekhi K. Stem cells as delivery vehicles for regenerative medicine-challenges and perspectives. World J Stem Cells. 2018;10:43-56. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 71] [Article Influence: 10.1] [Reference Citation Analysis (1)] |
| 2. | Tabatabaei Qomi R, Sheykhhasan M. Adipose-derived stromal cell in regenerative medicine: A review. World J Stem Cells. 2017;9:107-117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 76] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 3. | Tsuji W, Rubin JP, Marra KG. Adipose-derived stem cells: Implications in tissue regeneration. World J Stem Cells. 2014;6:312-321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 220] [Cited by in RCA: 269] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 4. | Fuchs E, Chen T. A matter of life and death: self-renewal in stem cells. EMBO Rep. 2013;14:39-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 133] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 5. | Bonab MM, Alimoghaddam K, Talebian F, Ghaffari SH, Ghavamzadeh A, Nikbin B. Aging of mesenchymal stem cell in vitro. BMC Cell Biol. 2006;7:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 572] [Cited by in RCA: 625] [Article Influence: 32.9] [Reference Citation Analysis (0)] |
| 6. | Lane SW, Williams DA, Watt FM. Modulating the stem cell niche for tissue regeneration. Nat Biotechnol. 2014;32:795-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 482] [Cited by in RCA: 425] [Article Influence: 38.6] [Reference Citation Analysis (0)] |
| 7. | Hynes RO. The extracellular matrix: not just pretty fibrils. Science. 2009;326:1216-1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2554] [Cited by in RCA: 2448] [Article Influence: 153.0] [Reference Citation Analysis (0)] |
| 8. | Trappmann B, Gautrot JE, Connelly JT, Strange DG, Li Y, Oyen ML, Cohen Stuart MA, Boehm H, Li B, Vogel V, Spatz JP, Watt FM, Huck WT. Extracellular-matrix tethering regulates stem-cell fate. Nat Mater. 2012;11:642-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1208] [Cited by in RCA: 1142] [Article Influence: 87.8] [Reference Citation Analysis (0)] |
| 9. | Zhang Z, Qu R, Fan T, Ouyang J, Lu F, Dai J. Stepwise Adipogenesis of Decellularized Cellular Extracellular Matrix Regulates Adipose Tissue-Derived Stem Cell Migration and Differentiation. Stem Cells Int. 2019;2019:1845926. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 10. | Ahmed M, Ffrench-Constant C. Extracellular Matrix Regulation of Stem Cell Behavior. Curr Stem Cell Rep. 2016;2:197-206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 120] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 11. | Gattazzo F, Urciuolo A, Bonaldo P. Extracellular matrix: a dynamic microenvironment for stem cell niche. Biochim Biophys Acta. 2014;1840:2506-2519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 789] [Cited by in RCA: 913] [Article Influence: 83.0] [Reference Citation Analysis (0)] |
| 12. | Lu P, Takai K, Weaver VM, Werb Z. Extracellular matrix degradation and remodeling in development and disease. Cold Spring Harb Perspect Biol. 2011;3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1129] [Cited by in RCA: 1552] [Article Influence: 110.9] [Reference Citation Analysis (0)] |
| 13. | Page-McCaw A, Ewald AJ, Werb Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat Rev Mol Cell Biol. 2007;8:221-233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2317] [Cited by in RCA: 2170] [Article Influence: 120.6] [Reference Citation Analysis (0)] |
| 14. | Ohashi T, Kiehart DP, Erickson HP. Dual labeling of the fibronectin matrix and actin cytoskeleton with green fluorescent protein variants. J Cell Sci. 2002;115:1221-1229. [PubMed] |
| 15. | Pankov R, Cukierman E, Katz BZ, Matsumoto K, Lin DC, Lin S, Hahn C, Yamada KM. Integrin dynamics and matrix assembly: tensin-dependent translocation of alpha(5)beta(1) integrins promotes early fibronectin fibrillogenesis. J Cell Biol. 2000;148:1075-1090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 385] [Cited by in RCA: 382] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 16. | Baneyx G, Baugh L, Vogel V. Coexisting conformations of fibronectin in cell culture imaged using fluorescence resonance energy transfer. Proc Natl Acad Sci USA. 2001;98:14464-14468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 146] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 17. | Baneyx G, Baugh L, Vogel V. Fibronectin extension and unfolding within cell matrix fibrils controlled by cytoskeletal tension. Proc Natl Acad Sci USA. 2002;99:5139-5143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 281] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 18. | Alhadlaq A, Tang M, Mao JJ. Engineered adipose tissue from human mesenchymal stem cells maintains predefined shape and dimension: implications in soft tissue augmentation and reconstruction. Tissue Eng. 2005;11:556-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 139] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 19. | Neubauer M, Hacker M, Bauer-Kreisel P, Weiser B, Fischbach C, Schulz MB, Goepferich A, Blunk T. Adipose tissue engineering based on mesenchymal stem cells and basic fibroblast growth factor in vitro. Tissue Eng. 2005;11:1840-1851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 89] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 20. | Fischbach C, Spruss T, Weiser B, Neubauer M, Becker C, Hacker M, Göpferich A, Blunk T. Generation of mature fat pads in vitro and in vivo utilizing 3-D long-term culture of 3T3-L1 preadipocytes. Exp Cell Res. 2004;300:54-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 81] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 21. | Porzionato A, Stocco E, Barbon S, Grandi F, Macchi V, De Caro R. Tissue-Engineered Grafts from Human Decellularized Extracellular Matrices: A Systematic Review and Future Perspectives. Int J Mol Sci. 2018;19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 227] [Article Influence: 32.4] [Reference Citation Analysis (0)] |
| 22. | Brown BN, Freund JM, Han L, Rubin JP, Reing JE, Jeffries EM, Wolf MT, Tottey S, Barnes CA, Ratner BD, Badylak SF. Comparison of three methods for the derivation of a biologic scaffold composed of adipose tissue extracellular matrix. Tissue Eng Part C Methods. 2011;17:411-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 157] [Article Influence: 11.2] [Reference Citation Analysis (3)] |
| 23. | Poon CJ, Pereira E Cotta MV, Sinha S, Palmer JA, Woods AA, Morrison WA, Abberton KM. Preparation of an adipogenic hydrogel from subcutaneous adipose tissue. Acta Biomater. 2013;9:5609-5620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 24. | Debels H, Gerrand YW, Poon CJ, Abberton KM, Morrison WA, Mitchell GM. An adipogenic gel for surgical reconstruction of the subcutaneous fat layer in a rat model. J Tissue Eng Regen Med. 2017;11:1230-1241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 25. | Roehm KD, Hornberger J, Madihally SV. In vitro characterization of acelluar porcine adipose tissue matrix for use as a tissue regenerative scaffold. J Biomed Mater Res A. 2016;104:3127-3136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 26. | Flynn LE. The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells. Biomaterials. 2010;31:4715-4724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 310] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 27. | Choi JS, Kim BS, Kim JY, Kim JD, Choi YC, Yang HJ, Park K, Lee HY, Cho YW. Decellularized extracellular matrix derived from human adipose tissue as a potential scaffold for allograft tissue engineering. J Biomed Mater Res A. 2011;97:292-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 111] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 28. | Choi YC, Choi JS, Kim BS, Kim JD, Yoon HI, Cho YW. Decellularized extracellular matrix derived from porcine adipose tissue as a xenogeneic biomaterial for tissue engineering. Tissue Eng Part C Methods. 2012;18:866-876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 93] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 29. | Guneta V, Zhou Z, Tan NS, Sugii S, Wong MTC, Choong C. Recellularization of decellularized adipose tissue-derived stem cells: role of the cell-secreted extracellular matrix in cellular differentiation. Biomater Sci. 2017;6:168-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 30. | Turner AE, Yu C, Bianco J, Watkins JF, Flynn LE. The performance of decellularized adipose tissue microcarriers as an inductive substrate for human adipose-derived stem cells. Biomaterials. 2012;33:4490-4499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 94] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 31. | Wu I, Nahas Z, Kimmerling KA, Rosson GD, Elisseeff JH. An injectable adipose matrix for soft-tissue reconstruction. Plast Reconstr Surg. 2012;129:1247-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 95] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 32. | Adam Young D, Bajaj V, Christman KL. Award winner for outstanding research in the PhD category, 2014 Society for Biomaterials annual meeting and exposition, Denver, Colorado, April 16-19, 2014: Decellularized adipose matrix hydrogels stimulate in vivo neovascularization and adipose formation. J Biomed Mater Res A. 2014;102:1641-1651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 33. | Kokai LE, Schilling BK, Chnari E, Huang YC, Imming EA, Karunamurthy A, Khouri RK, D'Amico RA, Coleman SR, Marra KG, Rubin JP. Injectable Allograft Adipose Matrix Supports Adipogenic Tissue Remodeling in the Nude Mouse and Human. Plast Reconstr Surg. 2019;143:299e-309e. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 71] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 34. | Giatsidis G, Succar J, Waters TD, Liu W, Rhodius P, Wang C, Nilsen TJ, Chnari E, Orgill DP. Tissue-Engineered Soft-Tissue Reconstruction Using Noninvasive Mechanical Preconditioning and a Shelf-Ready Allograft Adipose Matrix. Plast Reconstr Surg. 2019;144:884-895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 35. | Gulati AK. Evaluation of acellular and cellular nerve grafts in repair of rat peripheral nerve. J Neurosurg. 1988;68:117-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 113] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 36. | Rao Pattabhi S, Martinez JS, Keller TC. Decellularized ECM effects on human mesenchymal stem cell stemness and differentiation. Differentiation. 2014;88:131-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 48] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 37. | Feng X, Shen R, Tan J, Chen X, Pan Y, Ruan S, Zhang F, Lin Z, Zeng Y, Wang X, Lin Y, Wu Q. The study of inhibiting systematic inflammatory response syndrome by applying xenogenic (porcine) acellular dermal matrix on second-degree burns. Burns. 2007;33:477-479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 38. | Utomo L, Pleumeekers MM, Nimeskern L, Nürnberger S, Stok KS, Hildner F, van Osch GJ. Preparation and characterization of a decellularized cartilage scaffold for ear cartilage reconstruction. Biomed Mater. 2015;10:015010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 71] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 39. | Cheng CW, Solorio LD, Alsberg E. Decellularized tissue and cell-derived extracellular matrices as scaffolds for orthopaedic tissue engineering. Biotechnol Adv. 2014;32:462-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 261] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 40. | Long C, Galvez MG, Legrand A, Joubert LM, Wang Z, Chattopadhyay A, Chang J, Fox PM. Intratendinous Injection of Hydrogel for Reseeding Decellularized Human Flexor Tendons. Plast Reconstr Surg. 2017;139:1305e-1314e. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 41. | Martinello T, Bronzini I, Volpin A, Vindigni V, Maccatrozzo L, Caporale G, Bassetto F, Patruno M. Successful recellularization of human tendon scaffolds using adipose-derived mesenchymal stem cells and collagen gel. J Tissue Eng Regen Med. 2014;8:612-619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 42. | Urciuolo A, De Coppi P. Decellularized Tissue for Muscle Regeneration. Int J Mol Sci. 2018;19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 43. | Wilson K, Terlouw A, Roberts K, Wolchok JC. The characterization of decellularized human skeletal muscle as a blueprint for mimetic scaffolds. J Mater Sci Mater Med. 2016;27:125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 44. | Rodríguez-Rodríguez VE, Martínez-González B, Quiroga-Garza A, Reyes-Hernández CG, de la Fuente-Villarreal D, de la Garza-Castro O, Guzmán-López S, Elizondo-Omaña RE. Human Umbilical Vessels: Choosing the Optimal Decellularization Method. ASAIO J. 2018;64:575-580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 45. | Gui L, Muto A, Chan SA, Breuer CK, Niklason LE. Development of decellularized human umbilical arteries as small-diameter vascular grafts. Tissue Eng Part A. 2009;15:2665-2676. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 203] [Cited by in RCA: 188] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 46. | Haase SC, Rovak JM, Dennis RG, Kuzon WM, Cederna PS. Recovery of muscle contractile function following nerve gap repair with chemically acellularized peripheral nerve grafts. J Reconstr Microsurg. 2003;19:241-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 47. | Choi JS, Williams JK, Greven M, Walter KA, Laber PW, Khang G, Soker S. Bioengineering endothelialized neo-corneas using donor-derived corneal endothelial cells and decellularized corneal stroma. Biomaterials. 2010;31:6738-6745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 133] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 48. | VeDepo MC, Buse EE, Quinn RW, Williams TD, Detamore MS, Hopkins RA, Converse GL. Species-specific effects of aortic valve decellularization. Acta Biomater. 2017;50:249-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 49. | Cheung DY, Duan B, Butcher JT. Current progress in tissue engineering of heart valves: multiscale problems, multiscale solutions. Expert Opin Biol Ther. 2015;15:1155-1172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 99] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 50. | Oberwallner B, Brodarac A, Choi YH, Saric T, Anić P, Morawietz L, Stamm C. Preparation of cardiac extracellular matrix scaffolds by decellularization of human myocardium. J Biomed Mater Res A. 2014;102:3263-3272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 51. | Sánchez PL, Fernández-Santos ME, Costanza S, Climent AM, Moscoso I, Gonzalez-Nicolas MA, Sanz-Ruiz R, Rodríguez H, Kren SM, Garrido G, Escalante JL, Bermejo J, Elizaga J, Menarguez J, Yotti R, Pérez del Villar C, Espinosa MA, Guillem MS, Willerson JT, Bernad A, Matesanz R, Taylor DA, Fernández-Avilés F. Acellular human heart matrix: A critical step toward whole heart grafts. Biomaterials. 2015;61:279-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 125] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 52. | O'Neill JD, Anfang R, Anandappa A, Costa J, Javidfar J, Wobma HM, Singh G, Freytes DO, Bacchetta MD, Sonett JR, Vunjak-Novakovic G. Decellularization of human and porcine lung tissues for pulmonary tissue engineering. Ann Thorac Surg. 2013;96:1046-55; discussion 1055-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 186] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 53. | Bruzauskaite I, Raudoniute J, Denkovskij J, Bagdonas E, Meidute-Abaraviciene S, Simonyte V, Bironaite D, Siaurys A, Bernotiene E, Aldonyte R. Native matrix-based human lung alveolar tissue model in vitro: studies of the reparatory actions of mesenchymal stem cells. Cytotechnology. 2017;69:1-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 54. | Mattei G, Magliaro C, Pirone A, Ahluwalia A. Decellularized Human Liver Is Too Heterogeneous for Designing a Generic Extracellular Matrix Mimic Hepatic Scaffold. Artif Organs. 2017;41:E347-E355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 55. | Verstegen MMA, Willemse J, van den Hoek S, Kremers GJ, Luider TM, van Huizen NA, Willemssen FEJA, Metselaar HJ, IJzermans JNM, van der Laan LJW, de Jonge J. Decellularization of Whole Human Liver Grafts Using Controlled Perfusion for Transplantable Organ Bioscaffolds. Stem Cells Dev. 2017;26:1304-1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 67] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 56. | Song JJ, Guyette JP, Gilpin SE, Gonzalez G, Vacanti JP, Ott HC. Regeneration and experimental orthotopic transplantation of a bioengineered kidney. Nat Med. 2013;19:646-651. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 571] [Cited by in RCA: 578] [Article Influence: 48.2] [Reference Citation Analysis (0)] |
| 57. | Peloso A, Petrosyan A, Da Sacco S, Booth C, Zambon JP, OʼBrien T, Aardema C, Robertson J, De Filippo RE, Soker S, Stratta RJ, Perin L, Orlando G. Renal Extracellular Matrix Scaffolds From Discarded Kidneys Maintain Glomerular Morphometry and Vascular Resilience and Retains Critical Growth Factors. Transplantation. 2015;99:1807-1816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 57] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 58. | Patil PB, Chougule PB, Kumar VK, Almström S, Bäckdahl H, Banerjee D, Herlenius G, Olausson M, Sumitran-Holgersson S. Recellularization of acellular human small intestine using bone marrow stem cells. Stem Cells Transl Med. 2013;2:307-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 59. | Pokrywczynska M, Gubanska I, Drewa G, Drewa T. Application of bladder acellular matrix in urinary bladder regeneration: the state of the art and future directions. Biomed Res Int. 2015;2015:613439. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 48] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 60. | Banyard DA, Borad V, Amezcua E, Wirth GA, Evans GR, Widgerow AD. Preparation, Characterization, and Clinical Implications of Human Decellularized Adipose Tissue Extracellular Matrix (hDAM): A Comprehensive Review. Aesthet Surg J. 2016;36:349-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 45] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 61. | He Y, Lin M, Wang X, Guan J, Dong Z, Lu F, Xing M, Feng C, Li X. Optimized adipose tissue engineering strategy based on a neo-mechanical processing method. Wound Repair Regen. 2018;26:163-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 62. | Young DA, Ibrahim DO, Hu D, Christman KL. Injectable hydrogel scaffold from decellularized human lipoaspirate. Acta Biomater. 2011;7:1040-1049. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 157] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 63. | Lu Q, Li M, Zou Y, Cao T. Delivery of basic fibroblast growth factors from heparinized decellularized adipose tissue stimulates potent de novo adipogenesis. J Control Release. 2014;174:43-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 64. | Giatsidis G, Succar J, Haddad A, Lago G, Schaffer C, Wang X, Schilling B, Chnari E, Matsumine H, Orgill DP. Preclinical Optimization of a Shelf-Ready, Injectable, Human-Derived, Decellularized Allograft Adipose Matrix. Tissue Eng Part A. 2019;25:271-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 65. | Thomas-Porch C, Li J, Zanata F, Martin EC, Pashos N, Genemaras K, Poche JN, Totaro NP, Bratton MR, Gaupp D, Frazier T, Wu X, Ferreira LM, Tian W, Wang G, Bunnell BA, Flynn L, Hayes D, Gimble JM. Comparative proteomic analyses of human adipose extracellular matrices decellularized using alternative procedures. J Biomed Mater Res A. 2018;106:2481-2493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 66. | Zhang S, Lu Q, Cao T, Toh WS. Adipose Tissue and Extracellular Matrix Development by Injectable Decellularized Adipose Matrix Loaded with Basic Fibroblast Growth Factor. Plast Reconstr Surg. 2016;137:1171-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 46] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 67. | Song M, Liu Y, Hui L. Preparation and characterization of acellular adipose tissue matrix using a combination of physical and chemical treatments. Mol Med Rep. 2018;17:138-146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 68. | Zhao Y, Fan J, Bai S. Biocompatibility of injectable hydrogel from decellularized human adipose tissue in vitro and in vivo. J Biomed Mater Res B Appl Biomater. 2019;107:1684-1694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 69. | Wang L, Johnson JA, Zhang Q, Beahm EK. Combining decellularized human adipose tissue extracellular matrix and adipose-derived stem cells for adipose tissue engineering. Acta Biomater. 2013;9:8921-8931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 121] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 70. | Debelle L, Tamburro AM. Elastin: molecular description and function. Int J Biochem Cell Biol. 1999;31:261-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 370] [Cited by in RCA: 335] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 71. | Zhang Q, Johnson JA, Dunne LW, Chen Y, Iyyanki T, Wu Y, Chang EI, Branch-Brooks CD, Robb GL, Butler CE. Decellularized skin/adipose tissue flap matrix for engineering vascularized composite soft tissue flaps. Acta Biomater. 2016;35:166-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 77] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 72. | Ornitz DM. FGFs, heparan sulfate and FGFRs: complex interactions essential for development. Bioessays. 2000;22:108-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 73. | Harada M, Murakami H, Okawa A, Okimoto N, Hiraoka S, Nakahara T, Akasaka R, Shiraishi Y, Futatsugi N, Mizutani-Koseki Y, Kuroiwa A, Shirouzu M, Yokoyama S, Taiji M, Iseki S, Ornitz DM, Koseki H. FGF9 monomer-dimer equilibrium regulates extracellular matrix affinity and tissue diffusion. Nat Genet. 2009;41:289-298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 96] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 74. | Brown CF, Yan J, Han TT, Marecak DM, Amsden BG, Flynn LE. Effect of decellularized adipose tissue particle size and cell density on adipose-derived stem cell proliferation and adipogenic differentiation in composite methacrylated chondroitin sulphate hydrogels. Biomed Mater. 2015;10:045010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 75. | Yu C, Kornmuller A, Brown C, Hoare T, Flynn LE. Decellularized adipose tissue microcarriers as a dynamic culture platform for human adipose-derived stem/stromal cell expansion. Biomaterials. 2017;120:66-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 93] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 76. | Morissette Martin P, Shridhar A, Yu C, Brown C, Flynn LE. Decellularized Adipose Tissue Scaffolds for Soft Tissue Regeneration and Adipose-Derived Stem/Stromal Cell Delivery. Methods Mol Biol. 2018;1773:53-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 77. | Mohiuddin OA, Campbell B, Poche JN, Thomas-Porch C, Hayes DA, Bunnell BA, Gimble JM. Decellularized Adipose Tissue: Biochemical Composition, in vivo Analysis and Potential Clinical Applications. Adv Exp Med Biol. 2020;1212:57-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 78. | Sano H, Orbay H, Terashi H, Hyakusoku H, Ogawa R. Acellular adipose matrix as a natural scaffold for tissue engineering. J Plast Reconstr Aesthet Surg. 2014;67:99-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 79. | Wang JQ, Fan J, Gao JH, Zhang C, Bai SL. Comparison of in vivo adipogenic capabilities of two different extracellular matrix microparticle scaffolds. Plast Reconstr Surg. 2013;131:174e-187e. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 80. | Costa A, Naranjo JD, Londono R, Badylak SF. Biologic Scaffolds. Cold Spring Harb Perspect Med. 2017;7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 84] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 81. | Choi JS, Kim BS, Kim JD, Choi YC, Lee HY, Cho YW. In vitro cartilage tissue engineering using adipose-derived extracellular matrix scaffolds seeded with adipose-derived stem cells. Tissue Eng Part A. 2012;18:80-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 82. | Riis S, Hansen AC, Johansen L, Lund K, Pedersen C, Pitsa A, Hyldig K, Zachar V, Fink T, Pennisi CP. Fabrication and characterization of extracellular matrix scaffolds obtained from adipose-derived stem cells. Methods. 2020;171:68-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 83. | Yu C, Bianco J, Brown C, Fuetterer L, Watkins JF, Samani A, Flynn LE. Porous decellularized adipose tissue foams for soft tissue regeneration. Biomaterials. 2013;34:3290-3302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 129] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 84. | Cheung HK, Han TT, Marecak DM, Watkins JF, Amsden BG, Flynn LE. Composite hydrogel scaffolds incorporating decellularized adipose tissue for soft tissue engineering with adipose-derived stem cells. Biomaterials. 2014;35:1914-1923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 151] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 85. | Young DA, Choi YS, Engler AJ, Christman KL. Stimulation of adipogenesis of adult adipose-derived stem cells using substrates that mimic the stiffness of adipose tissue. Biomaterials. 2013;34:8581-8588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 193] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 86. | Kayabolen A, Keskin D, Aykan A, Karslıoglu Y, Zor F, Tezcaner A. Native extracellular matrix/fibroin hydrogels for adipose tissue engineering with enhanced vascularization. Biomed Mater. 2017;12:035007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 87. | van Dongen JA, Getova V, Brouwer LA, Liguori GR, Sharma PK, Stevens HP, van der Lei B, Harmsen MC. Adipose tissue-derived extracellular matrix hydrogels as a release platform for secreted paracrine factors. J Tissue Eng Regen Med. 2019;13:973-985. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 51] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 88. | Francis MP, Sachs PC, Madurantakam PA, Sell SA, Elmore LW, Bowlin GL, Holt SE. Electrospinning adipose tissue-derived extracellular matrix for adipose stem cell culture. J Biomed Mater Res A. 2012;100:1716-1724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 89. | Lin G, Albersen M, Harraz AM, Fandel TM, Garcia M, McGrath MH, Konety BR, Lue TF, Lin CS. Cavernous nerve repair with allogenic adipose matrix and autologous adipose-derived stem cells. Urology. 2011;77:1509.e1-1509.e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 2.5] [Reference Citation Analysis (2)] |
| 90. | Kim BS, Choi JS, Kim JD, Choi YC, Cho YW. Recellularization of decellularized human adipose-tissue-derived extracellular matrix sheets with other human cell types. Cell Tissue Res. 2012;348:559-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 91. | Wang JK, Luo B, Guneta V, Li L, Foo SEM, Dai Y, Tan TTY, Tan NS, Choong C, Wong MTC. Supercritical carbon dioxide extracted extracellular matrix material from adipose tissue. Mater Sci Eng C Mater Biol Appl. 2017;75:349-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 92. | Pati F, Ha DH, Jang J, Han HH, Rhie JW, Cho DW. Biomimetic 3D tissue printing for soft tissue regeneration. Biomaterials. 2015;62:164-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 239] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 93. | Dunne LW, Huang Z, Meng W, Fan X, Zhang N, Zhang Q, An Z. Human decellularized adipose tissue scaffold as a model for breast cancer cell growth and drug treatments. Biomaterials. 2014;35:4940-4949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 121] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 94. | Kochhar A, Wu I, Mohan R, Condé-Green A, Hillel AT, Byrne PJ, Elisseeff JH. A comparison of the rheologic properties of an adipose-derived extracellular matrix biomaterial, lipoaspirate, calcium hydroxylapatite, and cross-linked hyaluronic acid. JAMA Facial Plast Surg. 2014;16:405-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 95. | Du L, Wu X, Pang K, Yang Y. Histological evaluation and biomechanical characterisation of an acellular porcine cornea scaffold. Br J Ophthalmol. 2011;95:410-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 96. | Lin CY, Liu TY, Chen MH, Sun JS, Chen MH. An injectable extracellular matrix for the reconstruction of epidural fat and the prevention of epidural fibrosis. Biomed Mater. 2016;11:035010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 97. | Turner AE, Flynn LE. Design and characterization of tissue-specific extracellular matrix-derived microcarriers. Tissue Eng Part C Methods. 2012;18:186-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 98. | Han TT, Toutounji S, Amsden BG, Flynn LE. Adipose-derived stromal cells mediate in vivo adipogenesis, angiogenesis and inflammation in decellularized adipose tissue bioscaffolds. Biomaterials. 2015;72:125-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 118] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 99. | Baker NA, Muir LA, Washabaugh AR, Neeley CK, Chen SY, Flesher CG, Vorwald J, Finks JF, Ghaferi AA, Mulholland MW, Varban OA, Lumeng CN, O'Rourke RW. Diabetes-Specific Regulation of Adipocyte Metabolism by the Adipose Tissue Extracellular Matrix. J Clin Endocrinol Metab. 2017;102:1032-1043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 100. | Kim DW, Kim EJ, Kim EN, Sung MW, Kwon TK, Cho YW, Kwon SK. Human Adipose Tissue Derived Extracellular Matrix and Methylcellulose Hydrogels Augments and Regenerates the Paralyzed Vocal Fold. PLoS One. 2016;11:e0165265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 101. | Omidi E, Fuetterer L, Reza Mousavi S, Armstrong RC, Flynn LE, Samani A. Characterization and assessment of hyperelastic and elastic properties of decellularized human adipose tissues. J Biomech. 2014;47:3657-3663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 102. | Kuljanin M, Brown CFC, Raleigh MJ, Lajoie GA, Flynn LE. Collagenase treatment enhances proteomic coverage of low-abundance proteins in decellularized matrix bioscaffolds. Biomaterials. 2017;144:130-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 44] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 103. | Pati F, Jang J, Ha DH, Won Kim S, Rhie JW, Shim JH, Kim DH, Cho DW. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat Commun. 2014;5:3935. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1409] [Cited by in RCA: 1211] [Article Influence: 110.1] [Reference Citation Analysis (0)] |
| 104. | Choi JS, Kim BS, Kim JD, Choi YC, Lee EK, Park K, Lee HY, Cho YW. In vitro expansion of human adipose-derived stem cells in a spinner culture system using human extracellular matrix powders. Cell Tissue Res. 2011;345:415-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 105. | Gilbert TW, Freund JM, Badylak SF. Quantification of DNA in biologic scaffold materials. J Surg Res. 2009;152:135-139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 398] [Cited by in RCA: 352] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 106. | Smith LR, Cho S, Discher DE. Stem Cell Differentiation is Regulated by Extracellular Matrix Mechanics. Physiology (Bethesda). 2018;33:16-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 170] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 107. | Perea-Gil I, Uriarte JJ, Prat-Vidal C, Gálvez-Montón C, Roura S, Llucià-Valldeperas A, Soler-Botija C, Farré R, Navajas D, Bayes-Genis A. In vitro comparative study of two decellularization protocols in search of an optimal myocardial scaffold for recellularization. Am J Transl Res. 2015;7:558-573. [PubMed] |
| 108. | Choi YC, Choi JS, Woo CH, Cho YW. Stem cell delivery systems inspired by tissue-specific niches. J Control Release. 2014;193:42-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 109. | Lee M, Lee YK, Kim DH. The clinical result of arterialized venous free flaps for the treatment of soft tissue defect of the fingers. Medicine (Baltimore). 2019;98:e16017. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 110. | Elbanoby TM, Zidan SM, Elbatawy AM, Aly GM, Sholkamy K. Superficial temporal artery flap for reconstruction of complex facial defects: A new algorithm. Arch Plast Surg. 2018;45:118-127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 111. | Kim J, Yoon AP, Jones NF. Reverse Radial Forearm Flap to Provide Arterial Inflow to a Toe Transfer. Hand (N Y). 2017;12:154-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 112. | Pessoa Vaz M, Brandão C, Meireles R, Brito IM, Ferreira B, Pinheiro S, Zenha H, Ramos S, Diogo C, Teles L, Cabral L, Lima J. The role of microsurgical flaps in primary burn reconstruction. Ann Burns Fire Disasters. 2018;31:233-237. [PubMed] |
| 113. | Lee YJ, Baek SE, Lee S, Cho YW, Jeong YJ, Kim KJ, Jun YJ, Rhie JW. Wound-healing effect of adipose stem cell-derived extracellular matrix sheet on full-thickness skin defect rat model: Histological and immunohistochemical study. Int Wound J. 2019;16:286-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 114. | Woo CH, Choi YC, Choi JS, Lee HY, Cho YW. A bilayer composite composed of TiO2-incorporated electrospun chitosan membrane and human extracellular matrix sheet as a wound dressing. J Biomater Sci Polym Ed. 2015;26:841-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 57] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 115. | Kang H, Peng J, Lu S, Liu S, Zhang L, Huang J, Sui X, Zhao B, Wang A, Xu W, Luo Z, Guo Q. In vivo cartilage repair using adipose-derived stem cell-loaded decellularized cartilage ECM scaffolds. J Tissue Eng Regen Med. 2014;8:442-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 104] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 116. | Kun L, Yanhong Z, Chen X, Lianyong W, Qiang Y, Hongfa L, Binhong T. [Development and characterization of oriented scaffolds derived from cartilage extracellular matrix]. Hua Xi Kou Qiang Yi Xue Za Zhi. 2017;35:51-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 117. | Musumeci G, Lo Furno D, Loreto C, Giuffrida R, Caggia S, Leonardi R, Cardile V. Mesenchymal stem cells from adipose tissue which have been differentiated into chondrocytes in three-dimensional culture express lubricin. Exp Biol Med (Maywood). 2011;236:1333-1341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 118. | Musumeci G, Mobasheri A, Trovato FM, Szychlinska MA, Graziano AC, Lo Furno D, Avola R, Mangano S, Giuffrida R, Cardile V. Biosynthesis of collagen I, II, RUNX2 and lubricin at different time points of chondrogenic differentiation in a 3D in vitro model of human mesenchymal stem cells derived from adipose tissue. Acta Histochem. 2014;116:1407-1417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 119. | Szychlinska MA, Castrogiovanni P, Nsir H, Di Rosa M, Guglielmino C, Parenti R, Calabrese G, Pricoco E, Salvatorelli L, Magro G, Imbesi R, Mobasheri A, Musumeci G. Engineered cartilage regeneration from adipose tissue derived-mesenchymal stem cells: A morphomolecular study on osteoblast, chondrocyte and apoptosis evaluation. Exp Cell Res. 2017;357:222-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 120. | Zwingenberger S, Niederlohmann E, Vater C, Rammelt S, Matthys R, Bernhardt R, Valladares RD, Goodman SB, Stiehler M. Establishment of a femoral critical-size bone defect model in immunodeficient mice. J Surg Res. 2013;181:e7-e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 121. | Clough BH, McCarley MR, Gregory CA. A simple critical-sized femoral defect model in mice. J Vis Exp. 2015;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 122. | Hettwer W, Horstmann PF, Bischoff S, Güllmar D, Reichenbach JR, Poh PSP, van Griensven M, Gras F, Diefenbeck M. Establishment and effects of allograft and synthetic bone graft substitute treatment of a critical size metaphyseal bone defect model in the sheep femur. APMIS. 2019;127:53-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 123. | Zhang H, Yang L, Yang XG, Wang F, Feng JT, Hua KC, Li Q, Hu YC. Demineralized Bone Matrix Carriers and their Clinical Applications: An Overview. Orthop Surg. 2019;11:725-737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 97] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 124. | Skovrlj B, Guzman JZ, Al Maaieh M, Cho SK, Iatridis JC, Qureshi SA. Cellular bone matrices: viable stem cell-containing bone graft substitutes. Spine J. 2014;14:2763-2772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 125. | Mohiuddin OA, Campbell B, Poche JN, Ma M, Rogers E, Gaupp D, Harrison MAA, Bunnell BA, Hayes DJ, Gimble JM. Decellularized Adipose Tissue Hydrogel Promotes Bone Regeneration in Critical-Sized Mouse Femoral Defect Model. Front Bioeng Biotechnol. 2019;7:211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 126. | Zhu Y, Hideyoshi S, Jiang H, Matsumura Y, Dziki JL, LoPresti ST, Huleihel L, Faria GNF, Fuhrman LC, Lodono R, Badylak SF, Wagner WR. Injectable, porous, biohybrid hydrogels incorporating decellularized tissue components for soft tissue applications. Acta Biomater. 2018;73:112-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 42] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 127. | Klinger A, Kawata M, Villalobos M, Jones RB, Pike S, Wu N, Chang S, Zhang P, DiMuzio P, Vernengo J, Benvenuto P, Goldfarb RD, Hunter K, Liu Y, Carpenter JP, Tulenko TN. Living scaffolds: surgical repair using scaffolds seeded with human adipose-derived stem cells. Hernia. 2016;20:161-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 128. | Dankel SN, Svärd J, Matthä S, Claussnitzer M, Klöting N, Glunk V, Fandalyuk Z, Grytten E, Solsvik MH, Nielsen HJ, Busch C, Hauner H, Blüher M, Skurk T, Sagen JV, Mellgren G. COL6A3 expression in adipocytes associates with insulin resistance and depends on PPARγ and adipocyte size. Obesity (Silver Spring). 2014;22:1807-1813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 65] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 129. | Sun K, Park J, Gupta OT, Holland WL, Auerbach P, Zhang N, Goncalves Marangoni R, Nicoloro SM, Czech MP, Varga J, Ploug T, An Z, Scherer PE. Endotrophin triggers adipose tissue fibrosis and metabolic dysfunction. Nat Commun. 2014;5:3485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 257] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 130. | Bunney PE, Zink AN, Holm AA, Billington CJ, Kotz CM. Orexin activation counteracts decreases in nonexercise activity thermogenesis (NEAT) caused by high-fat diet. Physiol Behav. 2017;176:139-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 951] [Cited by in RCA: 866] [Article Influence: 108.3] [Reference Citation Analysis (0)] |
| 131. | Lackey DE, Burk DH, Ali MR, Mostaedi R, Smith WH, Park J, Scherer PE, Seay SA, McCoin CS, Bonaldo P, Adams SH. Contributions of adipose tissue architectural and tensile properties toward defining healthy and unhealthy obesity. Am J Physiol Endocrinol Metab. 2014;306:E233-E246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 86] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 132. | Divoux A, Tordjman J, Lacasa D, Veyrie N, Hugol D, Aissat A, Basdevant A, Guerre-Millo M, Poitou C, Zucker JD, Bedossa P, Clément K. Fibrosis in human adipose tissue: composition, distribution, and link with lipid metabolism and fat mass loss. Diabetes. 2010;59:2817-2825. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 418] [Cited by in RCA: 421] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 133. | Nishio N, Fujimoto Y, Hiramatsu M, Maruo T, Suga K, Tsuzuki H, Mukoyama N, Shimono M, Toriyama K, Takanari K, Kamei Y, Sone M. Computed tomographic assessment of autologous fat injection augmentation for vocal fold paralysis. Laryngoscope Investig Otolaryngol. 2017;2:459-465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 134. | Ricci Maccarini A, Stacchini M, Mozzanica F, Schindler A, Basile E, DE Rossi G, Woo P, Remacle M, Magnani M. Efficacy of trans-nasal fiberendoscopic injection laryngoplasty with centrifuged autologous fat in the treatment of glottic insufficiency due to unilateral vocal fold paralysis. Acta Otorhinolaryngol Ital. 2018;38:204-213. [PubMed] |
| 135. | Shridhar A, Gillies E, Amsden BG, Flynn LE. Composite Bioscaffolds Incorporating Decellularized ECM as a Cell-Instructive Component Within Hydrogels as In Vitro Models and Cell Delivery Systems. Methods Mol Biol. 2018;1577:183-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 136. | Wang X, Yu T, Chen G, Zou J, Li J, Yan J. Preparation and Characterization of a Chitosan/Gelatin/Extracellular Matrix Scaffold and Its Application in Tissue Engineering. Tissue Eng Part C Methods. 2017;23:169-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 137. | Choi JS, Yang HJ, Kim BS, Kim JD, Kim JY, Yoo B, Park K, Lee HY, Cho YW. Human extracellular matrix (ECM) powders for injectable cell delivery and adipose tissue engineering. J Control Release. 2009;139:2-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 124] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 138. | Tan QW, Zhang Y, Luo JC, Zhang D, Xiong BJ, Yang JQ, Xie HQ, Lv Q. Hydrogel derived from decellularized porcine adipose tissue as a promising biomaterial for soft tissue augmentation. J Biomed Mater Res A. 2017;105:1756-1764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 48] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 139. | Kim EJ, Choi JS, Kim JS, Choi YC, Cho YW. Injectable and Thermosensitive Soluble Extracellular Matrix and Methylcellulose Hydrogels for Stem Cell Delivery in Skin Wounds. Biomacromolecules. 2016;17:4-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 86] [Article Influence: 8.6] [Reference Citation Analysis (0)] |