Published online Jun 26, 2020. doi: 10.4252/wjsc.v12.i6.422
Peer-review started: February 29, 2020
First decision: April 25, 2020
Revised: May 8, 2020
Accepted: May 21, 2020
Article in press: May 21, 2020
Published online: June 26, 2020
Processing time: 116 Days and 20.3 Hours
Disabling hearing loss is the most common sensorineural disability worldwide. It affects around 466 million people and its incidence is expected to rise to around 900 million people by 2050, according to World Health Organization estimates. Most cases of hearing impairment are due to the degeneration of hair cells (HCs) in the cochlea, mechano-receptors that transduce incoming sound information into electrical signals that are sent to the brain. Damage to these cells is mainly caused by exposure to aminoglycoside antibiotics and to some anti-cancer drugs such as cisplatin, loud sounds, age, infections and genetic mutations. Hearing deficits may also result from damage to the spiral ganglion neurons that innervate cochlear HCs. Differently from what is observed in avian and non-mammalian species, there is no regeneration of missing sensory cell types in the adult mammalian cochlea, what makes hearing loss an irreversible process. This review summarizes the research that has been conducted with the aim of developing cell-based strategies that lead to sensory cell replacement in the adult cochlea and, ultimately, to hearing restoration. Two main lines of research are discussed, one directed toward the transplantation of exogenous replacement cells into the damaged tissue, and another that aims at reactivating the regenerative potential of putative progenitor cells in the adult inner ear. Results from some of the studies that have been conducted are presented and the advantages and drawbacks of the various approaches discussed.
Core tip: This review summarizes the various approaches that are being explored to establish cell-based therapies that tackle the irreversible loss of sensory cells in the adult cochlea. Advantages and disadvantages of the various approaches are discussed, based on published results, and some considerations are made on future perspectives, taking into account the new developments in the field.
- Citation: Durán-Alonso MB. Stem cell-based approaches: Possible route to hearing restoration? World J Stem Cells 2020; 12(6): 422-437
- URL: https://www.wjgnet.com/1948-0210/full/v12/i6/422.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v12.i6.422
Hearing loss is the most frequent sensorineural impairment in man; it affects over 6% of the world's population and its incidence is expected to rise to around 900 million people (10% of the population) by 2050 [World Health Organization (WHO) estimates, https://http://www.who.int/pbd/deafness/estimates/en/]; thus this disability poses very serious social and economic implications. Most cases of hearing impairment are due to degeneration of sensory hair cells (HCs), the mechano-receptors in the inner ear; damage to these cells may be caused by clinically administered drugs, such as aminoglycoside antibiotics and some chemotherapeutics (e.g., cisplatin), exposure to high levels of sound, aging, infections, some occupational hazards, genetic mutations. A smaller percentage of the cases are due to damage to the spiral ganglion neurons (SGNs) that innervate the HCs in the cochlea and act as the first relay in the transmission of the incoming sound information to the brain. Differently from the case in avian and other non-mammalian species, regeneration of missing cell types does not occur in the mammalian cochlea, and the ensuing hearing loss is thus permanent. Various approaches to cell regeneration are being explored, namely gene therapy, administration of survival factors and other biologically active molecules, and cell-based replacement strategies[1-5]. With regard to the latter studies, there are two main lines of research, one that is based on the introduction into the damaged cochlea of exogenous replacement cells, and another that aims at reactivating the regenerative potential of putative progenitor cells in the adult inner ear.
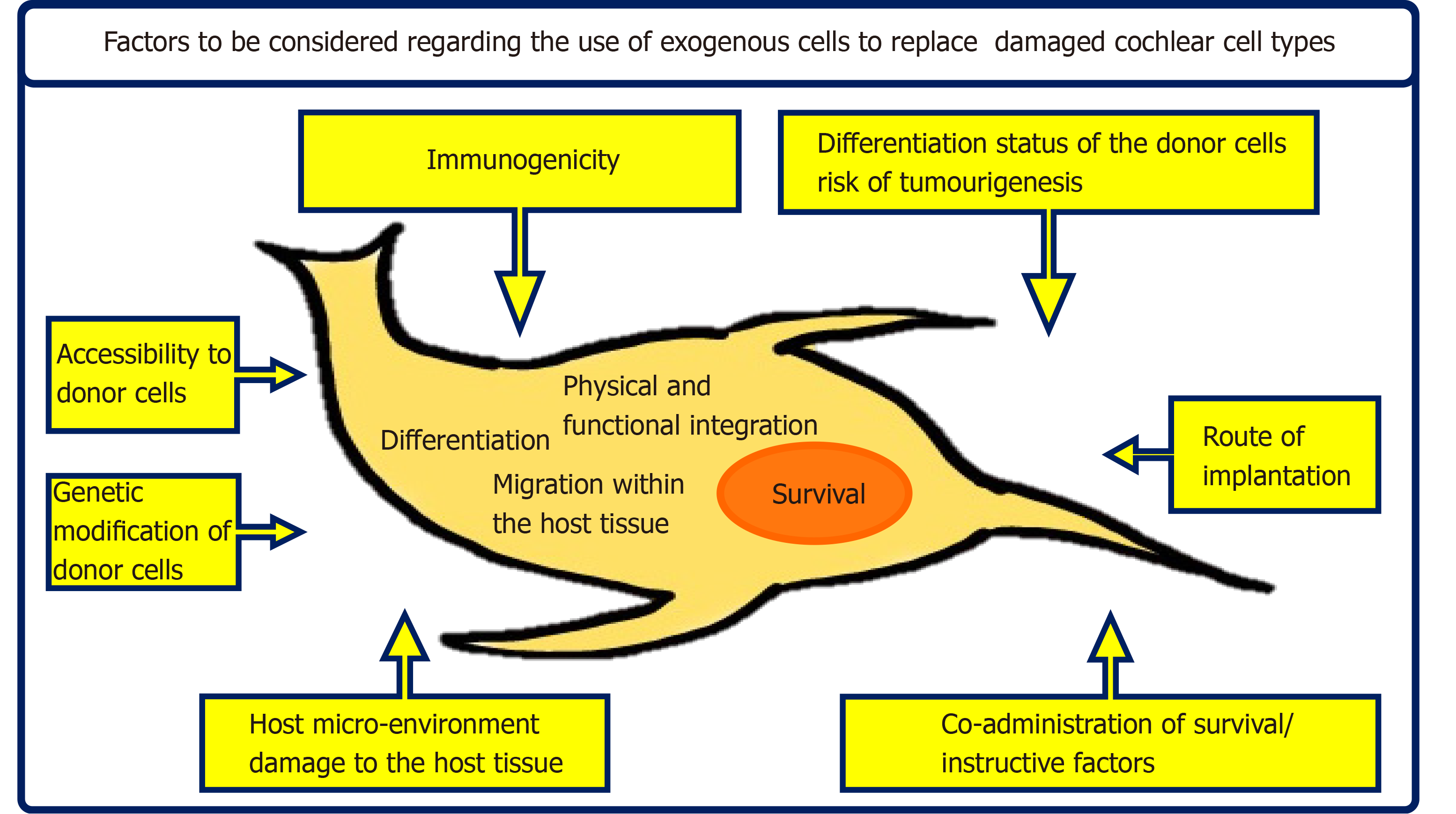
When considering the implantation of exogenous cells into the cochlea, a wide range of factors must be taken into account that have an important effect on the fate of the implanted cells and ultimately determine treatment outcome. Considerations such as the type of exogenous cells, their differentiation status and potential to differentiate towards other cell types, the selected route of implantation and the host micro-environment all play key roles in the survival and integration of the implanted cell population within the host tissue (Figure 1).
There are numerous studies reporting on the potential of different exogenous cell types to replace sensory cells in the damaged inner ear. Some of the work has focused on cells that, although coming from other tissues, may still share some relevant characteristics with otic cell types. Wei et al[6] demonstrated the capacity of ciliated ependymal cells, obtained from the forebrain germinal zone of adult mice, to get incorporated into streptomycin-treated cochlear explants, express markers typical of HCs and establish functional synapses with primary auditory neurons. In addition, the authors presented data indicating that neural stem cells (NSCs) isolated from the subventricular zone could be differentiated in vitro to neurons that established functional contacts with denervated HCs and with adult SGNs in corresponding co-cultures. In vivo work carried out by Hu et al[7] demonstrated significant migration of adult mouse NSCs transplanted into adult guinea pig cochleae to relevant locations such as the organ of Corti (OC), the spiral ganglion and the auditory nerve tract. However, NSC survival rates were very low; neomycin-induced damage to the cochlea and also Neurogenin2-transduction of the NSCs prior to transplantation improved differentiation of the transplanted NSCs towards a neuronal phenotype and increased NSC survival. Higher rates of cell survival were registered by Regala et al[8] following transplantation of murine adult NSCs into surgically damaged vestibulocochlear nerves in adult rats; importantly, the authors observed migration of murine cells to the brain stem in 50% of the transplanted animals. In another series of experiments, Edin et al[9] obtained SGN-like cells from human neural progenitors (NPs) by applying culture conditions used to maintain primary cultures of guinea pig and human SGNs; the cells developed morphological features and expressed an array of markers typical of SGNs.
Although NSCs from the subventricular zone could be used in autografts, thus overriding the need for immunosuppression, their isolation poses a series of technical issues. Therefore, other alternatives have been studied, such as the use of precursor cells that are resident in the olfactory epithelium[10,11], a readily accessible tissue. These precursors exhibit stem cell-like properties and express markers typical of otic HCs (MYOSIN VIIA, CALRETININ, ESPIN, PRESTIN) when co-cultured with cells from adult cochlea or when exposed to cochlea-conditioned medium[10]. Promising results were obtained by Xu et al[12], when implanting olfactory epithelium NSCs into the scala tympani of a rat model of noise-induced hearing loss; the implanted NSCs survived and migrated towards host spiral ganglion neurons, although they did not reach the OC. Yet the authors registered improved ABR results in NSC-treated deafened animals, compared to those from non-implanted deafened controls.
Notwithstanding, inner ear progenitors have been isolated from embryonic[13,14] and adult human tissues[15] that would be highly suitable donor cells; however, this is not a likely option, given their very low numbers. Interestingly, Stefan Heller and his group have shown that it is possible to isolate cochlear stem cells from post-mortem tissues without loss of their self-renewing and differentiation potential during the first 10 days following death[16].
Mesenchymal stem cells (MSCs) constitute another candidate donor cell source[17-21]. These are readily accessible cells with potential to differentiate into multiple lineages, which makes them highly desirable candidates for autologous cell-based therapies; importantly, they exert immunosuppressive properties[22] and it has been recently shown that transtympanic MSC administration into immunocompetent rat hosts does not elicit an inflammatory response[23]. Kondo et al[17] demonstrated the potential of murine MSCs (mMSCs) to acquire features of post-mitotic neurons following exposure to sonic hedgehog and retinoic acid (RA); the cells expressed a whole range of glutamatergic sensory neuron markers (SOX10, GATA3, GLUR4, VGLUT1, CALRETININ). An additional soluble protein present in hindbrain/somite/otocyst-conditioned medium and also in embryonic day 18 (E18) OC was required to induce the expression of additional sensory neuron markers (Brn3a, Neurogenin1, NeuroD). Further work by this group[24] demonstrated that Wnt signalling induces the expression of a whole array of sensory neuron markers through the up-regulation of T cell leukemia 3 (Tlx3), a transcription factor that promotes differentiation towards glutamatergic phenotypes. Infusion of Wnt1 was shown to increase the survival and engraftment rates of mMSCs implanted into the modiolus of ouabain-treated Mongolian gerbils[24]. Moreover, the implanted cells migrated throughout all the cochlear turns and reached the spiral ganglion; some of the cells adopted a clearly neuronal morphology and expressed neuronal markers.
In another series of experiments, Jeon et al[18] demonstrated that mMSCs could be differentiated to HC-like cells by exposing the mMSCs to a culture regime used to differentiate mouse embryonic stem cells (mESCs) into HC-like cells[25] and then over-expressing Math1, a well-known HC master gene[26]. mMSCs were differentiated to neurosensory progenitors (mMSC-NsPs) that expressed markers of early otic development, such as Otx2, Nestin, Sox2, Musashi, the early HC genes Math1, Brn3c and GATA3, and sensory neuronal markers such as TrkB and TrkC, among others. Transfection of mMSC-NsPs with Math1 induced expression of the HC genes MyosinVIIa and Espin, and other supporting cell (SC) and neuronal markers. Differentiation to a HC phenotype was promoted by co-culturing non-transfected mMSC-NsPs with E13 chick otocyst cells and also by injecting these cells into chick otocysts; of note, the injected cells integrated into chick otic epithelia, especially at sites of damage. Interestingly, when similar studies to those carried out by Kondo et al[17] and Jeon et al[18] were conducted on human MSCs (hMSCs), some differences were observed in the response of these human cells to differentiation cues, compared to that of their murine counterparts[19,20]. Therefore, Durán-Alonso et al[20] applied to hMSC cultures protocols that had been employed to direct human foetal auditory stem cells (hFASCs) from the cochleae of 9-11-week-old foetuses[27] towards HC and auditory neuron fates[28]. hMSCs were initially differentiated to neural progenitors (NPs); further treatment of hMSC-NPs resulted in induced expression of combinations of HC or SGN markers, depending on the culture regimes being applied. HC marker expression (ATOH1, MYOSINVIIa, BRN3C, CALRETININ) was only observed following treatment of hMSC-NPs that had been generated in suspension cultures, pointing to differences in the NP populations that had been obtained in floating and in adherent cultures. Unlike the results obtained by Jeon et al[18] on mMSCs, co-culture of hMSC-NPs with chick otocyst cells did not promote differentiation towards the HC lineage. Expression of sensory neuron markers (SOX2, GATA3, NGN1, ISLET1, NF200) could only be induced in hMSC-NP cultures that had undergone RA treatment, thought to render the cultures responsive to subsequent differentiation cues, such as bFGF. Additional experiments demonstrated that co-culture of 3D hMSC-NPs with murine cochlear explants promoted the expression of the neuronal marker NF-200 in these cells. Differently from the protocols applied by Durán-Alonso et al[20], based on the use of defined media containing specific growth factor combinations, Boddy et al[19] exposed hMSC cultures to media conditioned by hFASCs and observed sequential up-regulation of otic progenitor (PAX8, PAX2, SOX2), HC (ATOH1, BRN3C, MYOSINVIIA) and sensory neuron (NGN1, BRN3A) markers over time. A role was demonstrated for Wnt signalling at the early stages of otic induction. Additional work by Bas et al[21] demonstrated the capacity of human nasal MSCs to integrate into gentamicin-treated cochlear explants from post-natal rats, mostly in the spiral ganglion region; MSCs did not integrate into undamaged explants. Higher numbers of cells expressing βIII-TUBULIN were observed in cultures of damaged cochleae that had received hMSCs compared to those that had not been cultured with the human cells; over half of these neurons were hMSC-derived, indicating both differentiation of the exogenous cells and a protective effect on remaining SGNs. hMSC-derived neurons were excitable, and projected neurites towards the sensory epithelium, further promoted by Wnt signalling activation. In a different set of experiments, Schäck et al[29] explored the possibility to direct the differentiation of hMSCs to a glutamatergic neuron phenotype by conditionally expressing Ngn1, as already demonstrated by Reyes et al[30] on mESCs. hMSCs were refractory to adopting the desired fate as, although some glutamatergic neuronal markers were induced over time, their expression was not maintained once conditional expression of Ngn1 was halted[29].
In vivo survival and differentiation of transplanted stem cell types: Not-withstanding the valuable data obtained on the various types of exogenous cells mentioned above, the main advancements in the field have come from exploiting the great proliferative and multilineage differentiation potential offered by ESCs and induced pluripotent stem cells (iPSCs)[1,5,31]. Experiments have been carried out to investigate the influence of the host environment on the survival and differentiation of transplanted stem cells. Survival and induction of neuronal marker expression have been demonstrated at various timepoints following implantation into various in vivo animal models[8,32,33]. Additionally, some of these cells were seen to migrate to relevant locations such as the brain stem[8,32,33]; of note, work by Zhu et al[33] reported teratoma formation in a number of recipient cochleae following transplantation of murine iPSCs (miPSCs). Genetic modification of donor stem cells prior to their implantation in order to favour their in vivo survival and/or differentiation has also been carried out[30]. An example of this is the work carried out by Reyes et al[30]; transient Ngn1 expression in mECSs following their implantation into the scala tympani of kanamycin-treated guinea pigs resulted in increased migration and neuronal differentiation rates, compared to those of mESC controls[30].
In vitro differentiation. Effect of the otic micro-environment on differentiating cell types: Another line of work has pursued the in vitro differentiation of ESCs and iPSCs towards inner ear sensory cell types. A major breakthrough came from the observations by Stefan Heller’s group[25] that sequential incubation of mESC-derived embryoid bodies in serum-free medium (SFM) containing combinations of EGF, IGF-1 and bFGF resulted in the emergence of inner ear progenitor cells in the cultures; cells expressed markers that are seen during otic vesicle formation, such as Nestin, Otx2, Pax2, Bmp7 and Jagged1. In order to promote further maturation of these progenitor cells, the cultures were maintained in defined medium following withdrawal of growth factors; early markers Nestin, Pax2 and Bmp7 were downregulated, while the expression of HC genes such as Math1, Pou4f3, Jagged1, Myosin VIIa, Parvalbumin, AchRα9, p31Kip1 was induced; Espin was also expressed, indicative of stereociliary morphogenesis. Timing of expression and co-expression patterns of the various genes supported the hypothesis that cultures were mimicking in vivo inner ear developmental stages. Importantly, the authors demonstrated integration of inner ear progenitors into developing chick otic epithelia. Integration preferentially occurred in areas of the epithelium that had been damaged during surgery; progenitor cells that incorporated within HC-bearing regions up-regulated the HC marker MYOSIN VIIA and some developed F-actin-rich hair bundles that were labelled with an anti-ESPIN antibody, demonstrating an instructive role of the otic environment. The same group later developed a more elaborate step-wise approach to differentiate mESCs and miPSCs[34], where ectodermal induction was promoted at the expense of endoderm and mesoderm, and the formation of anterior ectoderm was favoured by the addition of IGF1; FGF combinations were then applied as the main otic inductive signals. Removal of the growth factors present in the medium[25] resulted in Math1 and MyosinVIIa expression; however , the cells did not present the typical HC morphology nor were hair bundle markers such as Espin detected. These features were only observed when the otic progenitor cells were grown on a layer of mitotically inactivated E18 chicken utricle stromal cells; the cells in these cultures exhibited stereociliary bundles and responded to mechanical stimulation in similar ways to those of immature HCs. Heller’s group then extended their studies to hESCs[35], applying to their cultures a modification of the treatment regime used for mESCs and miPSCs. The number of cells shown to express a combination of various HC markers was low and the cells resembled nascent HCs that did not further mature by increasing culture times but died instead. Additional studies were carried out[36] on monolayer cultures of hESCs and hiPSCs, in an attempt to better characterize the conditions required to obtain bona fide otic cell types from these cultures; this work identified retinoic acid as a critical factor for bFGF-induced expression of early otic markers in pre-placodal ectoderm cells. Nevertheless, no further differentiation of the cells was attained, indicating that the monolayer culture model lacked some of the factors found in aggregate cultures that promote the differentiation of otic progenitor cells. Supporting these findings, Abboud et al[37] obtained better results when applying an otic induction protocol [modified from (34)] to mESC cultures grown in floating conditions compared to cultures grown as monolayers. A greater proportion of the cells grown under non-adherent conditions expressed otic progenitor (PAX2, SIX1, EYA1, SOX2) and early HC markers (MYOSIN VIIA, POU4F3) following treatment, compared to adherent cultures. Following induction, floating cultures were partially dissociated and grafted into neomycin-damaged murine cochlear explants; a small number of these cells survived and integrated into the host tissue, preferentially in damaged areas of the OC, and expressed MYOSIN VIIA. Interestingly, this was not observed for any of the progenitors that had integrated outside the lesioned area. Notwithstanding, attempts have been made to conduct otic induction experiments on cell monolayers, rather than on three-dimensional cultures that are prone to higher variability[13,38]. Marcelo Rivolta’s group[13] obtained otic progenitor cells (expressing PAX8, SOX2, FOXG1, PAX2, NESTIN, SIX1 and GATA3) following 10-12-day-culture of hESC monolayers in SFM containing a combination of FGF3 and FGF10 or combinations of EGF, IGF-1 and bFGF factors[25]. The authors described two different types of colonies, large epithelioid colonies, composed of flat cells of large cytoplasm (otic epithelial progenitors, OEPs) and smaller colonies, formed by cells that presented denser chromatin and cytoplasmic projections (otic neural progenitors, ONPs). HC-like cells that co-expressed various HC marker combinations were obtained from OEPs following culture in SFM containing EGF and RA. A similar protocol was applied by Chen et al[39] to generate OEPs and ONPs from hiPSCs. OEPs were grown on mitomycin-treated chicken embryonic utricle stromal cells, in SFM containing EGF and RA, to yield rates of over 40% of cells that co-expressed HC markers (BRN3C, MYOSIN VIIA and ATOH1) and demonstrated some other characteristics of HCs such as the presence of mechano-transduction channels and some electrophysiological activity. In in vitro co-cultures of OEP-derived HC-like cells and SGNs from neonatal mice, SGNs extended neurites to the induced HC-like cells and formed active synapses. Additionally, OEPs were transplanted into the scala tympani of Slc26a4-null mice that present HC defects. At 4 wk post-transplantation some cells had migrated to the scala media and had integrated into the damaged epithelium, expressing MYOSIN VIIA and forming synaptic connections with native SGNs. The same protocol was applied by Azel Zine’s group[38] to obtain HC-like cells from hiPSCs; otic induction was significantly increased when the EGF/RA step was substituted by treatment with a Notch inhibitor, in agreement with accumulated evidence that Notch plays a key role in the differentiation of sensory otic lineages. Thus ATOH1 expression was much higher in cultures exposed to the Notch inhibitor and around 50% of the cells in these cultures expressed MYOSIN VIIA, as opposed to cultures grown in the presence of EGF/RA, where the percentage of cells expressing this HC marker did not reach 5%. Unfortunately, no hair bundle formation was detected on differentiating cells. Very importantly, hiPSC-derived otic progenitors could survive in an in vivo ototoxic damage model[40]. The cells were implanted into the cochlea of adult guinea pigs that had undergone amikacin treatment. Two weeks after implantation, surviving progenitors had engrafted within the damaged cochlear sensory epithelium and expressed MYOSIN VIIA; some expressed SOX2, pointing at their differentiation towards a SC type. Interestingly, those progenitors that had integrated outside the area of the OC did not express MYOSIN VIIA. Similar results were obtained when implanting murine otic progenitors into the same in vivo model.
In addition to the protocols discussed above, other HC induction protocols have been described. An example is provided by the work by Ouji and colleagues[41,42], based on the culture of mESC-derived embryoid bodies in medium conditioned by ST2 stromal cells. This treatment led to the induction of HC marker expression and the formation of stereocilia-like structures in some of the cells; additionally, some cells were shown to integrate into developing chick otocysts. A simpler method was developed by Ohnishi et al[43] in an attempt to eliminate the need for conditioned media, complex growth factor combinations, or the use of xenogeneic cells. They reported expression of MYOSIN VIIA and βIII TUBULIN proteins in hiPSC cultures that had been grown in defined medium, using bFGF as sole growth factor; stereocilia-like protrusions were observed in some MYOSIN VIIA-expressing cells. Although simpler than other methods, induction rates were extremely low.
Differently from the methods described above, Domingos Henrique’s group[44] directly programmed mESCs to become HCs by forcing the simultaneous expression of Gfi1, Pou4f3 and Atoh1 (GPA), coding for three key transcription factors in HC development. Theirs was an extremely fast and efficient induction protocol that in 8-12 days yielded large numbers of cells (54% ± 2%) that co-expressed various HC markers. Addition of RA or inhibition of the Notch pathway during GPA overexpression resulted in increased HC induction rates (84% ± 1% and 70% ± 2%, respectively). Some maturation of the MYOSIN VIIA+ cells were observed from d8 to d12, indicated by a decline in SOX2 expression and clear expression of the hair bundle proteins ESPIN and CADHERIN23 in membrane protrusions that did not reach the degree of organization found in normal HC stereociliary bundles. Nevertheless, FM1-43 incorporation experiments pointed at the presence of potentially functional mechano-transduction channels. Reyes et al[30] also resorted to genetic modification of mESCs in order to guide their differentiation in vitro and attained high rates of differentiation of mESCs to glutamatergic neurons through the transient expression of Ngn1 in the cultures.
ESCs and iPSCs have also been differentiated in vitro[13,45] and in vivo towards SGNs. Some of the work has consisted on generating stem cell-derived NPs that have then been implanted in the inner ear to promote their differentiation towards the SGN lineage. An example of this approach is the work carried out by Corrales et al[46], who grafted mESC-NPs into the cochlear nerve trunk of ouabain-treated gerbils. Implanted cells survived and demonstrated βIII TUBULIN and PERIPHERIN expression; interestingly, they extended processes towards the denervated HCs in the OC, indicating a role of the host environment as provider of survival, differentiation and guidance cues. Unfortunately, no functional recovery could be demonstrated. Coleman et al[47] implanted mESC-NPs into the scala tympani of chemically deafened guinea pigs, selecting a delivery route that was clinically more relevant than others previously used, such as direct injection into the auditory nerve. Transplanted cells were observed in the scala tympani of transplanted hosts at 4 wk post-transplantation. mESC-derived cells were also observed in Rosenthal’s canal, close to surviving endogenous SGNs, although their numbers were extremely low, indicating that delivery into the scala tympani was not an efficient route to direct exogenous cells to the Rosenthal’s canal. Sekiya et al[48] transplanted mESCs that had been exposed to the neuralizing activity of stromal cell-derived medium into the internal auditory meatal portion of the auditory nerve, aiming at minimizing the risk of damage to the cochlea and optimizing delivery to the target site. The group observed migration of the implanted cells along the damaged auditory nerve, into the Rosenthal´s canal and to the scala media. Interestingly, no significant migration was observed when mESC-NPs were implanted in intact auditory nerves; instead, the cells extended numerous neuritic processes along the nerve. These observations, together with the fact that implanted cells exhibited varying morphologies depending on their location, pointed at an interaction of the exogenous cells with local environmental cues. In another series of experiments, Okano et al[49] recorded higher rates of exogenous cell survival when implanting mESC-NPs in the modiolus of deafened guinea pigs, compared to their implantation in non-injured ears. Surviving cells differentiated to neurons that extended projections towards peripheral and central auditory targets. Interestingly, although synapse formation could not be demonstrated, some functional recovery was observed in some animals.
Work carried out by Albert Edge’s group on hESCs identified BMP4 as a critical molecule to differentiate hESCs towards SGNs[45]. When implanted in an in vivo gerbil model, hESC-derived NPs differentiated and engrafted in the auditory nerve trunk. The neurons extended projections to the sensory cochlear epithelium and towards the brain stem. Unfortunately, synapse formation could not be demonstrated. As mentioned above, hESC-NPs were also generated in Rivolta’s laboratory (otic neural progenitors, ONPs)[13]. ONPs were transplanted into the modiolus of ouabain-treated gerbils; implanted cells survived and formed an ectopic ganglion in the modiolus, with neurons that extended neurite projections to the OC. At 10 wk post-implantation some of the hESC-derived neurons had migrated from the ectopic ganglion to the Rosenthal’s canal and some cells were seen migrating towards the brainstem; SYNAPTOPHYSIN staining pointed to the establishment of synaptic connections of hESC-derived neurons with neurons in the cochlear nucleus. Importantly, functional tests carried out at 4 wk post-transplantation demonstrated an improvement in ABR thresholds of animals that had received ONPs; functional restoration correlated to the increase in neural density resulting from ONP transplantation. ONPs were also obtained from hiPSCs[39] and could be differentiated to neurons expressing combinations of sensory neuron and other neuronal markers (βIII-TUBULIN, BRN3A, NF200, NEUROD1, ISLET1). These neurons established active synapses in co-cultures with HC-like cells that were also generated from hiPSC cultures[39].
Survival of NPs derived from miPSCs has also been demonstrated in vivo[50], following transplantation into mouse cochleae; some of the surviving cells expressed the glutamatergic neuron marker VGLUT1 and were seen to project neurites towards cochlear HCs[50]. Differently to the approaches described above, Ishikawa et al[22] differentiated hiPSC-NPs to neurons in vitro, prior to their transplantation. Although the cultures contained a mixture of neuronal types and they were at various stages of maturation, around 95% of the cells expressed VGLUT1. The authors conducted parallel differentiation experiments on Matrigel-coated plates and on 3D collagen matrices, obtaining similar results. Implantation of 3D cultures into the scala tympani of normal hearing-competent guinea pigs demonstrated differentiation of hiPSC-NPs to glutamatergic neurons although there was a significant decline in the number of surviving exogenous cells during the first two weeks following transplantation. Based on the loss of Oct3/4 expression in differentiated cultures, the authors defended the safety of their approach, since one of the risks posed by the transplantation of undifferentiated cell types such as ESCs and iPSCs is their potential to give rise to tumours. Nevertheless, the risk of tumour formation by implanted cells cannot be completely eliminated, and thus efforts have also been made to obtain otic sensory cell-like cells from fully differentiated somatic cell types that may overcome this problem. In line with this argument, Durán-Alonso et al[51] applied to cultures of human fibroblasts the direct conversion protocol described by Costa et al[44]. Over-expression of the GPA combination of transcription factors induced the expression of HC markers MYOSIN VIIA, BRN3C and ESPIN. Despite good transduction rates and a strong increase in HC gene transcript expression, clear morphological changes and expression of a combination of HC proteins (MYOSIN VIIA, ANNEXIN A4, ESPIN) was only observed when transduced cells were cultured in SFM containing EGF and RA, as employed by Rivolta’s group on hFASCs[13,28]; however, cell polarization or formation of stereocilia-like protrusions were not observed. Transcriptomic analyses of these cultures indicated an enrichment of genes related to HC development and differentiation, together with genes involved in neuronal differentiation.
Organoids: Some recent work has resulted in the establishment of 3D inner ear organoid cultures[52-56], where differentiation of ESCs and iPSCs is conducted under culture conditions that sequentially recreate the stages leading to the development of various inner ear cell types in vivo. Thus the cultures are exposed to combinations of factors that activate and inhibit key signalling pathways to ultimately render the step-wise formation of non-neural ectoderm, pre-placodal and otic placodal epithelia, and otic vesicle epithelium that ultimately gives rise to HCs, SCs and sensory neurons[52,54,57]. Differentiating cultures are provided with added extracellular matrix proteins that support the self-organisation of the cells into biologically more relevant 3D cultures than those growing as monolayers on tissue culture plates. This arrangement yields clusters of HC-like cells that express an array of HC markers and exhibit basal-to-apical polarization, ESPIN-labelled hair bundles containing functional mechano-transduction channels, and a diversity of voltage-dependent currents. Neurons also emerge within these cultures that establish synaptic contacts with developing HCs[52,57]. Most of the work that has been carried out to date on inner ear organoids has focused on dissecting the identities and the complex interactions of the signalling pathways that regulate inner ear development[57-59], but the numerous advantages offered by these cultures, in terms of cellular complexity, cell phenotype maturation and numbers of induced cells make them ideal substrates for other important applications in the field[60,61]. At present there are important shortcomings to the use of inner ear organoids as sources for HC transplantation into the cochlea. A major hurdle is the fact that the applied protocols yield vestibular HC types[56,62]. Work is underway to elucidate what elements are missing in the current cultures that may yield cochlear HCs; Jeong et al[55] have recently described a series of modifications to the original method that result in the generation of various HC types in organoid cultures, some expressing cochlear HC markers. On the other hand, inner ear organoids may already constitute valid substrates to investigate SGN development and initiate studies towards a possible application in approaches to SGN regeneration. Perny et al[54] have modified initially published protocols, obtaining mESC-derived cultures that contain a large number of neurons that express a whole array of sensory otic neuron markers (GATA3, PROX1, ISLET1, p75, MAFB, PERIPHERIN) and display electrophysiological properties similar to those of SGNs. Very interestingly, characterization of the neurons present in these cultures indicates that these are not vestibular neurons but cochlear SGNs.
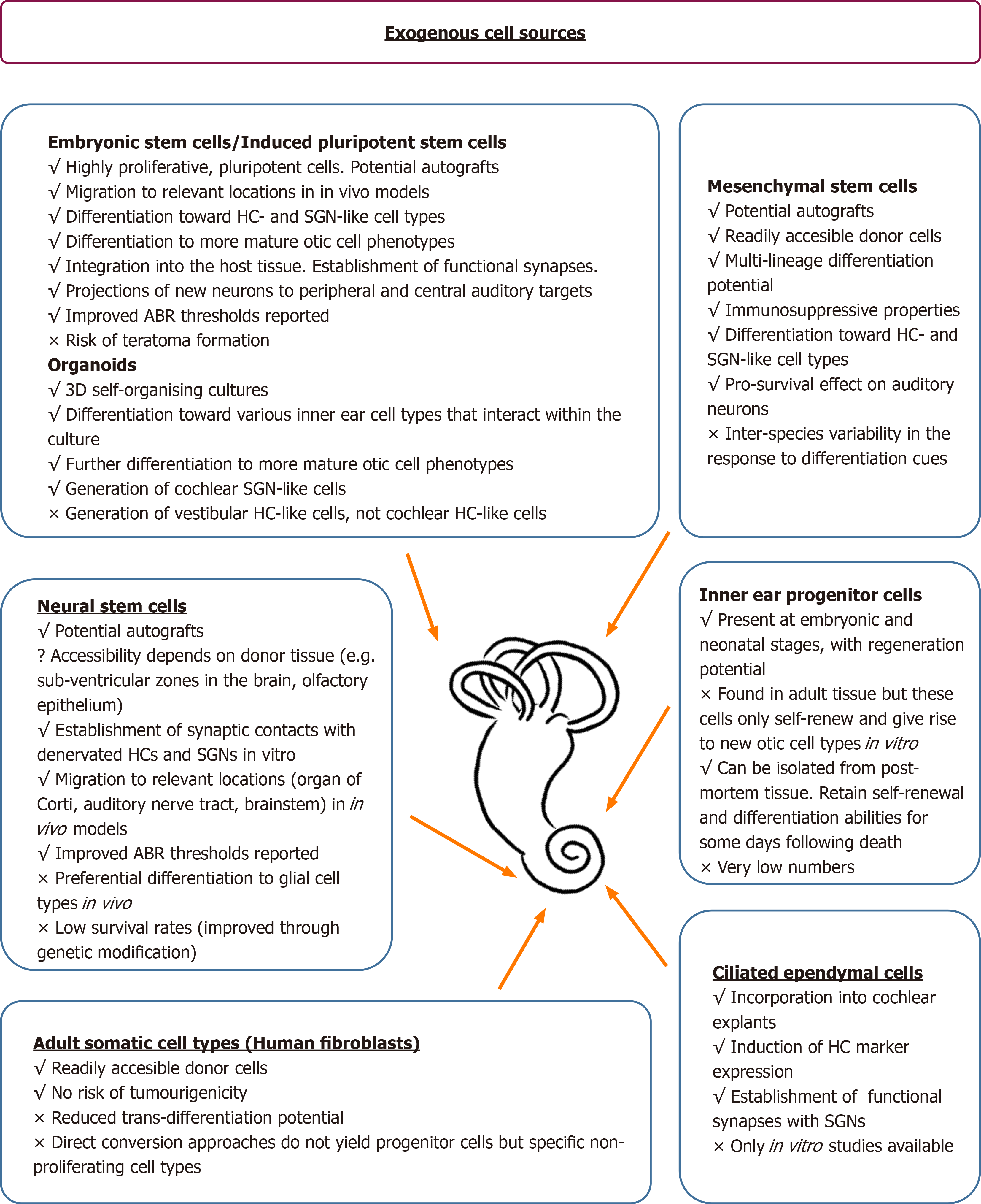
Summarizing what has been discussed above, a number of exogenous cell types have been evaluated for their potential to replace damaged cells in the adult mammalian cochlea, in a quest for a promising approach to hearing restoration. Some of these results are summarized in Figure 2.
Various populations of stem cell/progenitor cells have been described in the mammalian inner ear at embryonic and neonatal stages[14,27,63-65]. Initial studies on inner ear progenitors were based on the identification of cells in the inner ear that could self-renew in culture and give rise to different otic cell lineages when induced to differentiate[64,66,67]; otic progenitors have been identified in the vestibular sensory epithelium, the OC, the spiral ganglion and the stria vascularis[27,64,66,67]. On the other hand, since no regeneration takes place at later developmental stages following damage to HCs and/or SGNs, it has been traditionally accepted that there is no stem cell reservoir in the adult mammalian cochlea. This belief is supported by the fact that the features that characterize the progenitor cell populations that are present in the early stages of cochlear development are not encountered in more mature tissues[63,64,68]. Sphere-formation tests[63,66] indicate that there is a steep decline in the numbers of putative cochlear stem cell-like cells during the first three weeks following birth[63,64,68]; this has been associated to a decrease in the expression of stem and early otic markers like Nestin, Musashi1, Otx2, Mcm2, Pax2, Islet1[64], and the up-regulation of genes such as P27 in SCs from the OC of older organisms[63]. Work by Azel Zine’s group[69] demonstrated widespread expression of the stem/progenitor cell markers Gfap, ABCG2, SOX2 and JAGGED1 in cochlear SCs of P3 mice, and Nestin expression, which was mostly localized to inner phalangeal and border cells. Besides changes in the levels of expression of some of these genes, the authors unveiled a shift in the expression of Gfap and Abcg2 from the OC to SCs in the limbus area, and of Jagged1 to limbus interdental and Hensen cells; interestingly, these are areas where adenoviral-mediated Math1 overexpression has led to HC regeneration in adult cochleae, resulting in improved hearing function[70,71]. These and other observations add support to the notion that stem cell-like cells could still persist in the adult cochlea that have their regenerative potential curtailed by incoming signals from the surrounding tissue. Isolation from the post-mitotic cochlear epithelium of sphere-forming cells that can proliferate in vitro points at the presence of inhibitory signals originating from the cochlear tissue[63,64,66]. Moreover, despite the inability of the adult cochlea to regenerate damaged cell types, stem cells have been isolated from fully mature cochlear tissues that self-renew and give rise to differentiated otic lineages in vitro[15,16]. The numbers of these cells are very low; in addition, there are some technical difficulties associated to the study of cochlear tissues from adult mammalian models. As a result, the properties of various SC populations considered to be potential adult cochlear progenitor cells have been mostly explored on early post-natal animals, always bearing in mind that these models do not faithfully reproduce the conditions in the adult organ.
As already mentioned, a number of progenitor cells have been described in the embryonic cochlea[14,65,72,73]. Chen et al[27] isolated NESTIN+ SOX2+ PAX2+ otic progenitor cells from the cochleae of 10-week-old human foetuses; as discussed above, these cultures could be propagated as adherent cultures and presented two clearly different cell morphologies, which could be differentiated towards HC or SGN lineages. Roccio et al[14] later isolated a population of EPCAM+ CD311+ cells from the cochlea of 8-12-week-old foetuses; these cells proliferated in organoid cultures and could give rise to HCs. Other progenitor populations have been analysed at neonatal stages, soon after birth[65,69,73,74]; this is the case of SOX2+ NESTIN+ cells of the great epithelial ridge, considered to give rise to new inner border cells and inner phalangeal cells when these are damaged during the first two weeks following birth[73]; these progenitor cells are no longer present in the cochlea at the onset of hearing. A population of NESTIN+ cells that do not express HC or SC markers has been identified beneath the basilar membrane of post-natal animals[72,75]. These cells, called tympanic border cells, proliferate over the first 1-2 wk following birth; then, their proliferative potential declines, correlating with decreased Wnt signalling. Jan et al[72] observed migration of tympanic border cells into the OC and adjacent tissues during the first two weeks after birth; these cells then differentiated into HCs and various SC types. A small number of these cells are thought to remain in the adult cochlea, in a quiescent state. A good number of studies have been conducted in recent years to characterize a population of SCs that express the adult stem cell marker LGR5 (Leucine-rich repeat-containing G-protein coupled Receptor 5)[65,74,76,77]. These have been identified as HC progenitors in the neonatal cochlea[65,74,78], demonstrating some proliferative capacity and regenerating HCs in response to HC damage[65,74], Wnt signalling activation[65,79,80] and/or Notch signalling inhibition[81,82]. Efforts are being made to further unveil the signalling cascades that regulate the proliferation and HC regeneration abilities of LGR5-expressing cells[83-85], since expression of this stem cell marker persists in a reduced number of Deiter SCs in the adult cochlea[76]. However, most data have only been obtained on neonatal animal models[80-82,84]. The identification of a progenitor cell population in the cochlea and elucidating the reasons why these cells appear unable to regenerate the damaged tissue would be of the utmost importance; these progenitors would constitute an ideal cell source for regeneration therapies, placed at the appropriate location, and would override some of the main hurdles encountered by approaches that rely on the use of donor cells from exogenous sources (Figure 3).
While work is underway to better characterize the complex network of signalling cascades that regulate inner ear development in vivo, some promising results have been obtained when transplanting cells from exogenous tissues into the inner ear of animal models, demonstrating the formation of synaptic contacts between the implanted cells and HCs in the cochlear epithelium as well as with SGNs and neurons in areas of the brain stem[13,48,49]. There are many important questions to consider when contemplating this type of approach. Sufficient numbers of transplanted cells must survive, differentiate and integrate both physically and functionally into the damaged inner ear tissue in order to enable some degree of hearing restoration. Regeneration of HCs in the OC will convey survival of the donor cells in potassium-rich endolymph, a hostile medium to a large number of cell types; work is underway to establish protocols that increase the chances of survival of implanted cells in the scala media without compromising the internal conditions that are required for the correct functioning of the organ[86,87]. There is also the question of what cell type should be selected as donor cell and whether the cells should be transplanted as undifferentiated stem cells, partly differentiated progenitors, or fully differentiated cells. Stem cells and progenitor cells have been proposed as better candidates for transplantation than fully differentiated cells, on the argument that these cells should be more responsive to endogenous environmental cues. There are however concerns regarding their safety, in terms of a possible risk of tumour formation following implantation, and also the possibility that these cells may not differentiate to the desired cell type[22]; this is one of the hurdles encountered by NSC transplantation, when differentiation towards glial cell types often overrides neuronal differentiation. Improved survival and differentiation rates have been observed when using donor cells genetically engineered to express lineage-specific genes[7,30,42] and also when coupling exogenous cell transplantation with the administration of instructive factors[24,30]. One of the reasons to pursue the trans-differentiation of fully differentiated somatic cell types is the risk of teratoma formation by transplanted cells, as reported by Nishimura et al[88] following implantation of miPSC-NPs. A first report on the trans-differentiation of human fibroblasts to cells expressing HC markers constitutes a proof-of-principle study[51]; future experiments should be conducted that allow the transient expression of the transgenes using non-integrative vectors. In addition, approaches such as that described by Itakura et al[89], who have developed iPSC lines that carry an inducible caspase-9 gene, will ensure a safer application of these cells; this is all the more relevant when considering the vast potential of iPSCs as donor cells in instances where hearing loss is due to a genetic mutation. Survival and differentiation to appropriate cell lineages will also depend on the specific locations reached by the transplanted cells, which will in turn be affected by the route of implantation that is selected. Careful selection of the delivery route when considering donor cell-based therapies will be of paramount importance; thus, for example, although implantation into the scala tympani could be a method of choice when considering the convenience to the patient, this might result in insufficient numbers of exogenous cells reaching the target areas[47,48]. It has been conclusively shown that, independently from the transplanted cell type, the survival and engraftment rates of the exogenous cells are much higher in areas of damage, probably due to the release of relevant factors into the local microenvironment. There are doubts as to whether the observations made on experimental animal models do in fact mimic the clinical situation, and whether transplanted cells would encounter a similar microenvironment in the patient. Identification of the factors that promote the survival and differentiation of transplanted cells in animal models will nevertheless constitute highly valuable information[90]. There are other concerns to the transplantation of exogenous cells, such as the need for immunosuppression when receiving allogeneic donor cells; work by various groups have underlined the immunosuppressive properties of MSCs[22].
Very importantly, a major obstacle to the integration of implanted cells in the damaged OC is its cellular complexity and the fact that this is a highly organized epithelium, refractory to the integration of exogenous cells. This and the fact that formation of ectopic ganglia by exogenous cell-derived neurons results in some recovery of the hearing function make HC replacement a longer-term goal than SGN regeneration, as supported by the promising results that some groups have reported[13,49,91]. Yet, differentiation studies are proving highly valuable at dissecting the signalling pathways involved in inner ear development and their complex interactions. Huge progress has been made, as demonstrated by the generation of inner ear organoid models that are shedding very important information on inner ear development and provide excellent cellular models that may yet be used in future transplantation approaches. Moreover, the availability of inner ear organoids of human origin is of great importance, especially in light of the interspecies variability that has been observed[13,20]. Use of gene edited-hiPSC cell lines to generate organoids provides an extremely useful tool to identify instructive molecules and to study the effect of gene mutations on inner ear development[57,59,61].
With regard to a re-activation of the regenerative program in the adult cochlea, although extremely interesting, this is still a largely unknown area. It has been demonstrated that cells with stem cell-like properties persist in the adult inner ear; characterization of the progenitor population in the vestibular system is proving less difficult, since this tissue maintains some very limited regenerative capacity in adult mammals[92]. However, the identity of progenitor cells in the adult cochlea has not yet been confirmed. Data from the Raphael and the Zine labs[69,71] suggest that there may be a reduced pool of stem cell-like cells in the limbus area of adult cochlea that have been displaced from their location in the OC during the neonatal period and that may be responsive to specific signals such as Atoh1 expression. On the other hand, there are also low numbers of tympanic border cells and LGR5+ cells present in the adult cochlea; these populations are responsive to Wnt sigalling and can give rise to HCs and SCs during postnatal stages. However, their role and properties in the adult cochlea have not been characterized. Very interestingly, new evidence is emerging that point at changes in the epigenetic landscape of LGR5+ cells in the adult cochlea, opening the door to new approaches to reactivate a regenerative programme in these cells[93,94]. It is also important to remember that all the information we have on the various progenitor cell populations has been obtained on animal models. There are only two reports on the presence of LGR5+ cells in the human foetal cochlea[14,95]. Intriguingly, Johnson Chacko et al[95] have described LGR5 expression in cochlear HCs of 12-week-old foetuses, but not in SCs. Although there are doubts concerning the reliability of the currently available LGR5 antibodies, this observation raises the question of whether the data that have accumulated on murine cochlear LGR5+ cells are in agreement with the characteristics of their human counterparts.
The author gratefully acknowledges Dr. Luka Kranjc for his assistance in the elaboration of the figures in this article and funding from the University of Valladolid, Valladolid, Spain (University of Valladolid 2017 Post-doctoral contract Call).
Manuscript source: Invited manuscript
Specialty type: Cell and tissue engineering
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ventura C S-Editor: Wang J L-Editor: A E-Editor: Liu MY
| 1. | Conde de Felipe MM, Feijoo Redondo A, García-Sancho J, Schimmang T, Durán-Alonso MB. Cell- and gene-therapy approaches to inner ear repair. Histol Histopathol. 2011;26:923-940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 2. | Gao X, Tao Y, Lamas V, Huang M, Yeh WH, Pan B, Hu YJ, Hu JH, Thompson DB, Shu Y, Li Y, Wang H, Yang S, Xu Q, Polley DB, Liberman MC, Kong WJ, Holt JR, Chen ZY, Liu DR. Treatment of autosomal dominant hearing loss by in vivo delivery of genome editing agents. Nature. 2018;553:217-221. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 306] [Cited by in RCA: 393] [Article Influence: 49.1] [Reference Citation Analysis (0)] |
| 3. | Lee MY, Park YH. Potential of Gene and Cell Therapy for Inner Ear Hair Cells. Biomed Res Int. 2018;2018:8137614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 4. | Ma Y, Wise AK, Shepherd RK, Richardson RT. New molecular therapies for the treatment of hearing loss. Pharmacol Ther. 2019;200:190-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 5. | Roccio M, Senn P, Heller S. Novel insights into inner ear development and regeneration for targeted hearing loss therapies. Hear Res. 2019;107859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 6. | Wei D, Levic S, Nie L, Gao WQ, Petit C, Jones EG, Yamoah EN. Cells of adult brain germinal zone have properties akin to hair cells and can be used to replace inner ear sensory cells after damage. Proc Natl Acad Sci USA. 2008;105:21000-21005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 7. | Hu Z, Wei D, Johansson CB, Holmström N, Duan M, Frisén J, Ulfendahl M. Survival and neural differentiation of adult neural stem cells transplanted into the mature inner ear. Exp Cell Res. 2005;302:40-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 110] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 8. | Regala C, Duan M, Zou J, Salminen M, Olivius P. Xenografted fetal dorsal root ganglion, embryonic stem cell and adult neural stem cell survival following implantation into the adult vestibulocochlear nerve. Exp Neurol. 2005;193:326-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 53] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 9. | Edin F, Liu W, Boström M, Magnusson PU, Rask-Andersen H. Differentiation of human neural progenitor cell-derived spiral ganglion-like neurons: a time-lapse video study. Acta Otolaryngol. 2014;134:441-447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 10. | Doyle KL, Kazda A, Hort Y, McKay SM, Oleskevich S. Differentiation of adult mouse olfactory precursor cells into hair cells in vitro. Stem Cells. 2007;25:621-627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 11. | Young E, Westerberg B, Yanai A, Gregory-Evans K. The olfactory mucosa: a potential source of stem cells for hearing regeneration. Regen Med. 2018;13:581-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 12. | Xu YP, Shan XD, Liu YY, Pu Y, Wang CY, Tao QL, Deng Y, Cheng Y, Fan JP. Olfactory epithelium neural stem cell implantation restores noise-induced hearing loss in rats. Neurosci Lett. 2016;616:19-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 13. | Chen W, Jongkamonwiwat N, Abbas L, Eshtan SJ, Johnson SL, Kuhn S, Milo M, Thurlow JK, Andrews PW, Marcotti W, Moore HD, Rivolta MN. Restoration of auditory evoked responses by human ES-cell-derived otic progenitors. Nature. 2012;490:278-282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 291] [Cited by in RCA: 271] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 14. | Roccio M, Perny M, Ealy M, Widmer HR, Heller S, Senn P. Molecular characterization and prospective isolation of human fetal cochlear hair cell progenitors. Nat Commun. 2018;9:4027. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 61] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 15. | Rask-Andersen H, Boström M, Gerdin B, Kinnefors A, Nyberg G, Engstrand T, Miller JM, Lindholm D. Regeneration of human auditory nerve. In vitro/in video demonstration of neural progenitor cells in adult human and guinea pig spiral ganglion. Hear Res. 2005;203:180-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 111] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 16. | Senn P, Oshima K, Teo D, Grimm C, Heller S. Robust postmortem survival of murine vestibular and cochlear stem cells. J Assoc Res Otolaryngol. 2007;8:194-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 17. | Kondo T, Johnson SA, Yoder MC, Romand R, Hashino E. Sonic hedgehog and retinoic acid synergistically promote sensory fate specification from bone marrow-derived pluripotent stem cells. Proc Natl Acad Sci USA. 2005;102:4789-4794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 125] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 18. | Jeon SJ, Oshima K, Heller S, Edge AS. Bone marrow mesenchymal stem cells are progenitors in vitro for inner ear hair cells. Mol Cell Neurosci. 2007;34:59-68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 83] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 19. | Boddy SL, Chen W, Romero-Guevara R, Kottam L, Bellantuono I, Rivolta MN. Inner ear progenitor cells can be generated in vitro from human bone marrow mesenchymal stem cells. Regen Med. 2012;7:757-767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 20. | Durán-Alonso MB, Feijoo-Redondo A, Conde de Felipe M, Carnicero E, García AS, García-Sancho J, Rivolta MN, Giráldez F, Schimmang T. Generation of inner ear sensory cells from bone marrow-derived human mesenchymal stem cells. Regen Med. 2012;7:769-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 21. | Bas E, Van De Water TR, Lumbreras V, Rajguru S, Goss G, Hare JM, Goldstein BJ. Adult human nasal mesenchymal-like stem cells restore cochlear spiral ganglion neurons after experimental lesion. Stem Cells Dev. 2014;23:502-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 22. | Ishikawa M, Ohnishi H, Skerleva D, Sakamoto T, Yamamoto N, Hotta A, Ito J, Nakagawa T. Transplantation of neurons derived from human iPS cells cultured on collagen matrix into guinea-pig cochleae. J Tissue Eng Regen Med. 2017;11:1766-1778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 23. | Mittal R, Ocak E, Zhu A, Perdomo MM, Pena SA, Mittal J, Bohorquez J, Eshraghi AA. Effect of Bone Marrow-Derived Mesenchymal Stem Cells on Cochlear Function in an Experimental Rat Model. Anat Rec (Hoboken). 2020;303:487-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 24. | Kondo T, Matsuoka AJ, Shimomura A, Koehler KR, Chan RJ, Miller JM, Srour EF, Hashino E. Wnt signaling promotes neuronal differentiation from mesenchymal stem cells through activation of Tlx3. Stem Cells. 2011;29:836-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 25. | Li H, Roblin G, Liu H, Heller S. Generation of hair cells by stepwise differentiation of embryonic stem cells. Proc Natl Acad Sci USA. 2003;100:13495-13500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 197] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 26. | Bermingham NA, Hassan BA, Price SD, Vollrath MA, Ben-Arie N, Eatock RA, Bellen HJ, Lysakowski A, Zoghbi HY. Math1: an essential gene for the generation of inner ear hair cells. Science. 1999;284:1837-1841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 814] [Cited by in RCA: 841] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 27. | Chen W, Cacciabue-Rivolta DI, Moore HD, Rivolta MN. The human fetal cochlea can be a source for auditory progenitors/stem cells isolation. Hear Res. 2007;233:23-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 28. | Chen W, Johnson SL, Marcotti W, Andrews PW, Moore HD, Rivolta MN. Human fetal auditory stem cells can be expanded in vitro and differentiate into functional auditory neurons and hair cell-like cells. Stem Cells. 2009;27:1196-1204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 29. | Schäck L, Budde S, Lenarz T, Krettek C, Gross G, Windhagen H, Hoffmann A, Warnecke A. Induction of neuronal-like phenotype in human mesenchymal stem cells by overexpression of Neurogenin1 and treatment with neurotrophins. Tissue Cell. 2016;48:524-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | Reyes JH, O'Shea KS, Wys NL, Velkey JM, Prieskorn DM, Wesolowski K, Miller JM, Altschuler RA. Glutamatergic neuronal differentiation of mouse embryonic stem cells after transient expression of neurogenin 1 and treatment with BDNF and GDNF: in vitro and in vivo studies. J Neurosci. 2008;28:12622-12631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 89] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 31. | Czajkowski A, Mounier A, Delacroix L, Malgrange B. Pluripotent stem cell-derived cochlear cells: a challenge in constant progress. Cell Mol Life Sci. 2019;76:627-635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 32. | Hu Z, Ulfendahl M, Olivius NP. Central migration of neuronal tissue and embryonic stem cells following transplantation along the adult auditory nerve. Brain Res. 2004;1026:68-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 33. | Zhu H, Chen J, Guan L, Xiong S, Jiang H. The transplantation of induced pluripotent stem cells into the cochleae of mature mice. Int J Clin Exp Pathol. 2018;11:4423-4430. [PubMed] |
| 34. | Oshima K, Shin K, Diensthuber M, Peng AW, Ricci AJ, Heller S. Mechanosensitive hair cell-like cells from embryonic and induced pluripotent stem cells. Cell. 2010;141:704-716. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 282] [Cited by in RCA: 232] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 35. | Ronaghi M, Nasr M, Ealy M, Durruthy-Durruthy R, Waldhaus J, Diaz GH, Joubert LM, Oshima K, Heller S. Inner ear hair cell-like cells from human embryonic stem cells. Stem Cells Dev. 2014;23:1275-1284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 36. | Ealy M, Ellwanger DC, Kosaric N, Stapper AP, Heller S. Single-cell analysis delineates a trajectory toward the human early otic lineage. Proc Natl Acad Sci USA. 2016;113:8508-8513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 37. | Abboud N, Fontbonne A, Watabe I, Tonetto A, Brezun JM, Feron F, Zine A. Culture conditions have an impact on the maturation of traceable, transplantable mouse embryonic stem cell-derived otic progenitor cells. J Tissue Eng Regen Med. 2017;11:2629-2642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 38. | Lahlou H, Nivet E, Lopez-Juarez A, Fontbonne A, Assou S, Zine A. Enriched Differentiation of Human Otic Sensory Progenitor Cells Derived From Induced Pluripotent Stem Cells. Front Mol Neurosci. 2018;11:452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 39. | Chen J, Hong F, Zhang C, Li L, Wang C, Shi H, Fu Y, Wang J. Differentiation and transplantation of human induced pluripotent stem cell-derived otic epithelial progenitors in mouse cochlea. Stem Cell Res Ther. 2018;9:230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 40. | Lopez-Juarez A, Lahlou H, Ripoll C, Cazals Y, Brezun JM, Wang Q, Edge A, Zine A. Engraftment of Human Stem Cell-Derived Otic Progenitors in the Damaged Cochlea. Mol Ther. 2019;27:1101-1113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 41. | Ouji Y, Ishizaka S, Nakamura-Uchiyama F, Yoshikawa M. In vitro differentiation of mouse embryonic stem cells into inner ear hair cell-like cells using stromal cell conditioned medium. Cell Death Dis. 2012;3:e314. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 42. | Ouji Y, Ishizaka S, Nakamura-Uchiyama F, Wanaka A, Yoshikawa M. Induction of inner ear hair cell-like cells from Math1-transfected mouse ES cells. Cell Death Dis. 2013;4:e700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 43. | Ohnishi H, Skerleva D, Kitajiri S, Sakamoto T, Yamamoto N, Ito J, Nakagawa T. Limited hair cell induction from human induced pluripotent stem cells using a simple stepwise method. Neurosci Lett. 2015;599:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 44. | Costa A, Sanchez-Guardado L, Juniat S, Gale JE, Daudet N, Henrique D. Generation of sensory hair cells by genetic programming with a combination of transcription factors. Development. 2015;142:1948-1959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 94] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 45. | Shi F, Corrales CE, Liberman MC, Edge AS. BMP4 induction of sensory neurons from human embryonic stem cells and reinnervation of sensory epithelium. Eur J Neurosci. 2007;26:3016-3023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 94] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 46. | Corrales CE, Pan L, Li H, Liberman MC, Heller S, Edge AS. Engraftment and differentiation of embryonic stem cell-derived neural progenitor cells in the cochlear nerve trunk: growth of processes into the organ of Corti. J Neurobiol. 2006;66:1489-1500. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 142] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 47. | Coleman B, Hardman J, Coco A, Epp S, de Silva M, Crook J, Shepherd R. Fate of embryonic stem cells transplanted into the deafened mammalian cochlea. Cell Transplant. 2006;15:369-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 84] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 48. | Sekiya T, Kojima K, Matsumoto M, Kim TS, Tamura T, Ito J. Cell transplantation to the auditory nerve and cochlear duct. Exp Neurol. 2006;198:12-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 44] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 49. | Okano T, Nakagawa T, Endo T, Kim TS, Kita T, Tamura T, Matsumoto M, Ohno T, Sakamoto T, Iguchi F, Ito J. Engraftment of embryonic stem cell-derived neurons into the cochlear modiolus. Neuroreport. 2005;16:1919-1922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 43] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 50. | Nishimura K, Nakagawa T, Ono K, Ogita H, Sakamoto T, Yamamoto N, Okita K, Yamanaka S, Ito J. Transplantation of mouse induced pluripotent stem cells into the cochlea. Neuroreport. 2009;20:1250-1254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 51. | Durán-Alonso MB, Lopez Hernandez I, de la Fuente MA, Garcia-Sancho J, Giraldez F, Schimmang T. Transcription factor induced conversion of human fibroblasts towards the hair cell lineage. PLoS One. 2018;13:e0200210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 52. | Koehler KR, Mikosz AM, Molosh AI, Patel D, Hashino E. Generation of inner ear sensory epithelia from pluripotent stem cells in 3D culture. Nature. 2013;500:217-221. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 275] [Cited by in RCA: 297] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 53. | Koehler KR, Nie J, Longworth-Mills E, Liu XP, Lee J, Holt JR, Hashino E. Generation of inner ear organoids containing functional hair cells from human pluripotent stem cells. Nat Biotechnol. 2017;35:583-589. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 171] [Cited by in RCA: 227] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 54. | Perny M, Ting CC, Kleinlogel S, Senn P, Roccio M. Generation of Otic Sensory Neurons from Mouse Embryonic Stem Cells in 3D Culture. Front Cell Neurosci. 2017;11:409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 55. | Jeong M, O'Reilly M, Kirkwood NK, Al-Aama J, Lako M, Kros CJ, Armstrong L. Generating inner ear organoids containing putative cochlear hair cells from human pluripotent stem cells. Cell Death Dis. 2018;9:922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 56. | Mattei C, Lim R, Drury H, Nasr B, Li Z, Tadros MA, D'Abaco GM, Stok KS, Nayagam BA, Dottori M. Generation of Vestibular Tissue-Like Organoids From Human Pluripotent Stem Cells Using the Rotary Cell Culture System. Front Cell Dev Biol. 2019;7:25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 57. | Schaefer SA, Higashi AY, Loomis B, Schrepfer T, Wan G, Corfas G, Dressler GR, Duncan RK. From Otic Induction to Hair Cell Production: Pax2EGFP Cell Line Illuminates Key Stages of Development in Mouse Inner Ear Organoid Model. Stem Cells Dev. 2018;27:237-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 58. | DeJonge RE, Liu XP, Deig CR, Heller S, Koehler KR, Hashino E. Modulation of Wnt Signaling Enhances Inner Ear Organoid Development in 3D Culture. PLoS One. 2016;11:e0162508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 52] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 59. | Hartman BH, Bӧscke R, Ellwanger DC, Keymeulen S, Scheibinger M, Heller S. Fbxo2VHC mouse and embryonic stem cell reporter lines delineate in vitro-generated inner ear sensory epithelia cells and enable otic lineage selection and Cre-recombination. Dev Biol. 2018;443:64-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 60. | Roccio M, Edge ASB. Inner ear organoids: new tools to understand neurosensory cell development, degeneration and regeneration. Development. 2019;146:dev177188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 51] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 61. | Tang PC, Alex AL, Nie J, Lee J, Roth AA, Booth KT, Koehler KR, Hashino E, Nelson RF. Defective Tmprss3-Associated Hair Cell Degeneration in Inner Ear Organoids. Stem Cell Reports. 2019;13:147-162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 62. | Liu XP, Koehler KR, Mikosz AM, Hashino E, Holt JR. Functional development of mechanosensitive hair cells in stem cell-derived organoids parallels native vestibular hair cells. Nat Commun. 2016;7:11508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 75] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 63. | White PM, Doetzlhofer A, Lee YS, Groves AK, Segil N. Mammalian cochlear supporting cells can divide and trans-differentiate into hair cells. Nature. 2006;441:984-987. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 301] [Cited by in RCA: 327] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 64. | Oshima K, Grimm CM, Corrales CE, Senn P, Martinez Monedero R, Géléoc GS, Edge A, Holt JR, Heller S. Differential distribution of stem cells in the auditory and vestibular organs of the inner ear. J Assoc Res Otolaryngol. 2007;8:18-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 241] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 65. | Shi F, Kempfle JS, Edge AS. Wnt-responsive Lgr5-expressing stem cells are hair cell progenitors in the cochlea. J Neurosci. 2012;32:9639-9648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 208] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 66. | Malgrange B, Belachew S, Thiry M, Nguyen L, Rogister B, Alvarez ML, Rigo JM, Van De Water TR, Moonen G, Lefebvre PP. Proliferative generation of mammalian auditory hair cells in culture. Mech Dev. 2002;112:79-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 116] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 67. | Li H, Liu H, Heller S. Pluripotent stem cells from the adult mouse inner ear. Nat Med. 2003;9:1293-1299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 351] [Cited by in RCA: 334] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 68. | Lou X, Zhang Y, Yuan C. Multipotent stem cells from the young rat inner ear. Neurosci Lett. 2007;416:28-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 69. | Smeti I, Savary E, Capelle V, Hugnot JP, Uziel A, Zine A. Expression of candidate markers for stem/progenitor cells in the inner ears of developing and adult GFAP and nestin promoter-GFP transgenic mice. Gene Expr Patterns. 2011;11:22-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 70. | Kawamoto K, Ishimoto S, Minoda R, Brough DE, Raphael Y. Math1 gene transfer generates new cochlear hair cells in mature guinea pigs in vivo. J Neurosci. 2003;23:4395-4400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 336] [Cited by in RCA: 314] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 71. | Izumikawa M, Minoda R, Kawamoto K, Abrashkin KA, Swiderski DL, Dolan DF, Brough DE, Raphael Y. Auditory hair cell replacement and hearing improvement by Atoh1 gene therapy in deaf mammals. Nat Med. 2005;11:271-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 515] [Cited by in RCA: 529] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 72. | Jan TA, Chai R, Sayyid ZN, van Amerongen R, Xia A, Wang T, Sinkkonen ST, Zeng YA, Levin JR, Heller S, Nusse R, Cheng AG. Tympanic border cells are Wnt-responsive and can act as progenitors for postnatal mouse cochlear cells. Development. 2013;140:1196-1206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 73] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 73. | Mellado Lagarde MM, Wan G, Zhang L, Gigliello AR, McInnis JJ, Zhang Y, Bergles D, Zuo J, Corfas G. Spontaneous regeneration of cochlear supporting cells after neonatal ablation ensures hearing in the adult mouse. Proc Natl Acad Sci USA. 2014;111:16919-16924. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 58] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 74. | Bramhall NF, Shi F, Arnold K, Hochedlinger K, Edge AS. Lgr5-positive supporting cells generate new hair cells in the postnatal cochlea. Stem Cell Reports. 2014;2:311-322. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 139] [Cited by in RCA: 188] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 75. | Taniguchi M, Yamamoto N, Nakagawa T, Ogino E, Ito J. Identification of tympanic border cells as slow-cycling cells in the cochlea. PLoS One. 2012;7:e48544. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 76. | Chai R, Xia A, Wang T, Jan TA, Hayashi T, Bermingham-McDonogh O, Cheng AG. Dynamic expression of Lgr5, a Wnt target gene, in the developing and mature mouse cochlea. J Assoc Res Otolaryngol. 2011;12:455-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 128] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 77. | Lenz DR, Gunewardene N, Abdul-Aziz DE, Wang Q, Gibson TM, Edge ASB. Applications of Lgr5-Positive Cochlear Progenitors (LCPs) to the Study of Hair Cell Differentiation. Front Cell Dev Biol. 2019;7:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 78. | Cox BC, Chai R, Lenoir A, Liu Z, Zhang L, Nguyen DH, Chalasani K, Steigelman KA, Fang J, Rubel EW, Cheng AG, Zuo J. Spontaneous hair cell regeneration in the neonatal mouse cochlea in vivo. Development. 2014;141:816-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 240] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 79. | Chai R, Kuo B, Wang T, Liaw EJ, Xia A, Jan TA, Liu Z, Taketo MM, Oghalai JS, Nusse R, Zuo J, Cheng AG. Wnt signaling induces proliferation of sensory precursors in the postnatal mouse cochlea. Proc Natl Acad Sci U S A. 2012;109:8167-8172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 240] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 80. | Shi F, Hu L, Edge AS. Generation of hair cells in neonatal mice by β-catenin overexpression in Lgr5-positive cochlear progenitors. Proc Natl Acad Sci USA. 2013;110:13851-13856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 151] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 81. | Li W, Wu J, Yang J, Sun S, Chai R, Chen ZY, Li H. Notch inhibition induces mitotically generated hair cells in mammalian cochleae via activating the Wnt pathway. Proc Natl Acad Sci USA. 2015;112:166-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 150] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 82. | Ni W, Zeng S, Li W, Chen Y, Zhang S, Tang M, Sun S, Chai R, Li H. Wnt activation followed by Notch inhibition promotes mitotic hair cell regeneration in the postnatal mouse cochlea. Oncotarget. 2016;7:66754-66768. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 65] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 83. | Kuo BR, Baldwin EM, Layman WS, Taketo MM, Zuo J. In Vivo Cochlear Hair Cell Generation and Survival by Coactivation of β-Catenin and Atoh1. J Neurosci. 2015;35:10786-10798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 87] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 84. | Chen Y, Lu X, Guo L, Ni W, Zhang Y, Zhao L, Wu L, Sun S, Zhang S, Tang M, Li W, Chai R, Li H. Hedgehog Signaling Promotes the Proliferation and Subsequent Hair Cell Formation of Progenitor Cells in the Neonatal Mouse Cochlea. Front Mol Neurosci. 2017;10:426. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 85. | McLean WJ, Yin X, Lu L, Lenz DR, McLean D, Langer R, Karp JM, Edge ASB. Clonal Expansion of Lgr5-Positive Cells from Mammalian Cochlea and High-Purity Generation of Sensory Hair Cells. Cell Rep. 2017;18:1917-1929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 156] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 86. | Park YH, Wilson KF, Ueda Y, Tung Wong H, Beyer LA, Swiderski DL, Dolan DF, Raphael Y. Conditioning the cochlea to facilitate survival and integration of exogenous cells into the auditory epithelium. Mol Ther. 2014;22:873-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 87. | Gao J, Wang S, Tang Q, Li X, Zhang Y, Liu W, Gao Z, Yang H, Zhao RC. In Vitro Survival of Human Mesenchymal Stem Cells is Enhanced in Artificial Endolymph with Moderately High Concentrations of Potassium. Stem Cells Dev. 2018;27:658-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 88. | Nishimura K, Nakagawa T, Sakamoto T, Ito J. Fates of murine pluripotent stem cell-derived neural progenitors following transplantation into mouse cochleae. Cell Transplant. 2012;21:763-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 89. | Itakura G, Kawabata S, Ando M, Nishiyama Y, Sugai K, Ozaki M, Iida T, Ookubo T, Kojima K, Kashiwagi R, Yasutake K, Nakauchi H, Miyoshi H, Nagoshi N, Kohyama J, Iwanami A, Matsumoto M, Nakamura M, Okano H. Fail-Safe System against Potential Tumorigenicity after Transplantation of iPSC Derivatives. Stem Cell Reports. 2017;8:673-684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 91] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 90. | He Y, Zhang PZ, Sun D, Mi WJ, Zhang XY, Cui Y, Jiang XW, Mao XB, Qiu JH. Wnt1 from cochlear schwann cells enhances neuronal differentiation of transplanted neural stem cells in a rat spiral ganglion neuron degeneration model. Cell Transplant. 2014;23:747-760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 91. | Huang X, Liu J, Wu W, Hu P, Wang Q. Taurine enhances mouse cochlear neural stem cell transplantation via the cochlear lateral wall for replacement of degenerated spiral ganglion neurons via sonic hedgehog signaling pathway. Cell Tissue Res. 2019;378:49-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 92. | You D, Guo L, Li W, Sun S, Chen Y, Chai R, Li H. Characterization of Wnt and Notch-Responsive Lgr5+ Hair Cell Progenitors in the Striolar Region of the Neonatal Mouse Utricle. Front Mol Neurosci. 2018;11:137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 93. | Samarajeewa A, Lenz DR, Xie L, Chiang H, Kirchner R, Mulvaney JF, Edge ASB, Dabdoub A. Transcriptional response to Wnt activation regulates the regenerative capacity of the mammalian cochlea. Development. 2018;145:dev166579. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 94. | Samarajeewa A, Jacques BE, Dabdoub A. Therapeutic Potential of Wnt and Notch Signaling and Epigenetic Regulation in Mammalian Sensory Hair Cell Regeneration. Mol Ther. 2019;27:904-911. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 56] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 95. | Johnson Chacko L, Sergi C, Eberharter T, Dudas J, Rask-Andersen H, Hoermann R, Fritsch H, Fischer N, Glueckert R, Schrott-Fischer A. Early appearance of key transcription factors influence the spatiotemporal development of the human inner ear. Cell Tissue Res. 2020;379:459-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |