Published online Feb 15, 2003. doi: 10.3748/wjg.v9.i2.334
Revised: October 24, 2002
Accepted: November 4, 2002
Published online: February 15, 2003
AIM: To observe the localization of TRAIL/TRAIR (DR4, DR5, DcR1, DcR2) in the fetal pancreas.
METHODS: Fetal pancreas of 32 wk of pregnancy were obtained from induced abortions, embedded in paraffin, and 4-μm sections were prepared. The localization of TRAIL/TRAILR in fetal pancreas was investigated by fluorescence immunohistochemical method combined with laser scanning confocal microscopy.
RESULTS: TRAIL immunoreactive cells were mainly located on the periphery of the pancreas islets. There were a few DcR1 and DcR2 positive cells whereas there were no immunoreactive cells of DR4 and DR5 in the pancreas islets. In the acini and the ducts of the exocrine pancreas there were no TRAIL/TRAILR immunoreactive cells.
CONCLUSION: This study not only describes the distribution of TRAIL/TRAILR in the fetal pancreas, but also provides a morphological basis for deducing the function of TRAIL/TRAILR in pancreas, suggesting that in normal pancreatic islets, the pancreatic cells are resistant towards apoptosis too.
- Citation: Chen LH, Liu XS, Wang WY, Han WN, Pan BR, Jin BQ. Localization of TRAIL/TRAILR in fetal pancreas. World J Gastroenterol 2003; 9(2): 334-337
- URL: https://www.wjgnet.com/1007-9327/full/v9/i2/334.htm
- DOI: https://dx.doi.org/10.3748/wjg.v9.i2.334
Being an important endocrinology organ, the pancreas plays an important role in many physical and pathological processes[1-11]. The pancreatic islets could secrete not only the classical endocrinology substances such as insulin, glucagons, somatostatin and so on, but also could secrete some neuropeptides and cytokines[12,13].
Tumor necrosis factor related apoptosis inducing ligand (TRAIL)/TRAILR is a group of molecules belong to TNFSF/TNFRSF identified from 1995 and displays very important biological function[14-20]. Despite the widespread interest in TRAIL and its receptors to date, studies on TRAIL/TRAILR are mostly stayed on mRNA level just for the lack of monoclonal antibodies. Fortunately, a series of antibodies against TRAIL/TRAILR system have been prepared and identified successfully by our department and Screaton group recently and these provided a useful way to detect the expression of TRAIL/TRAILR in many organs and tissues[21]. In this experiment, we detected the localization of TRAIL/TRAILR in fetal pancreas.
The mouse anti-human DR4, DR5, DcR1, and DcR2 antibodies and the rabbit anti-human TRAIL antibody were prepared in our department. Biotin conjugated horse anti-rabbit IgG, FITC conjugated goat anti-mouse IgG and Texas red-conjugated streptavidin were purchased from Sigma.
Five fetal pancreas of 32-week pregnancy were surgically obtained. They were washed with physiological saline at 4 °C and a 6-mm3 piece was taken from each before fixed in Bouin's solution overnight. Each piece was embedded in paraffin and 4-μm sections were prepared.
Four-micrometer sections from fetal pancreas of 32-week pregnancy were employed in the fluorescent immunohistochemical analysis of TRAIL/TRAILR. Several dilutions of the antibody were tested to find the optimal staining concentration before the entire series was processed. The staining procedure was carried out as previous reports, without protease treatment[22]. Briefly, the steps included: (1) the sections were deparaffinized in xylene, hydrated in ethanol, and washed in 0.01 mol/L PBS, then pretreated with 30 mL/L normal goat serum for 40 min and rinsed in 0.1 mol/L PBS; (2) incubation at 4 °C for 24 h in the primary antibodies, mouse anti-human DR4, DR5, DcR1 and DcR2 antibody (1:200 dilution, final concentration of 25 mg/L) was performed respectively, and then in rabbit anti-human TRAIL antibody (1:300 dilution in 10 mL/L BSA-PBS); (3) the secondary antibody, biotin-labeled horse anti-rabbit IgG (1:200 dilution), was incubated at room temperature for 1 h; (4) simultaneous incubation with 1:200 FITC conjugated goat anti-mouse IgG and Texas red-conjugated streptavidin (1:1000 dilution) for 30 min. The sections were washed three times for 10 min each after incubation from steps 2 to 4, respectively, and were finally mounted in 50 g/L glycerin. The sections were examined with Bio-Rad 1024 LSCM. The specimens were excited with a laser beam at wavelengths of 568 nm (Texas Red) and 488 nm (FITC) and the emission light was focused through a pinhole aperture. The full field of view was scanned in square image formats of 512x512 pixels.
Primary antibodies were substituted by irrelevant antibodies and normal rabbit or goat serum as specific antibody control. PBS was substituted for primary antibody as negative control. Primary antibody was omitted as blank control.
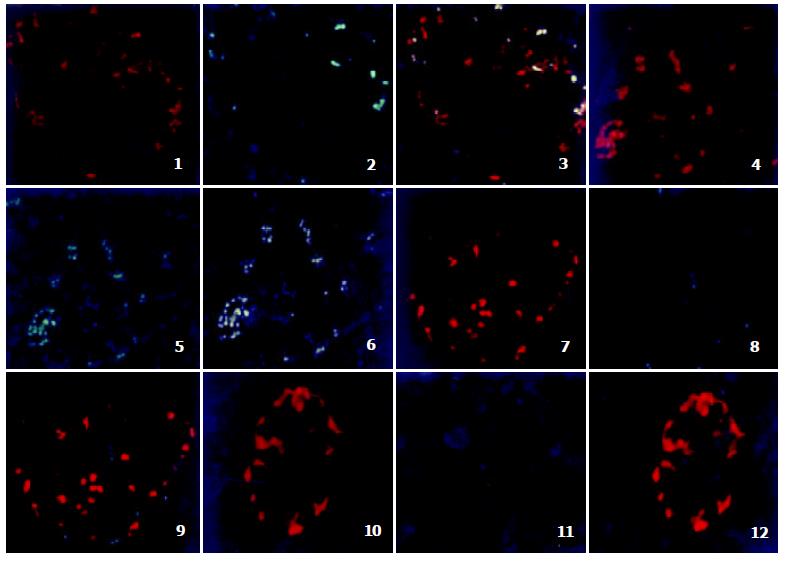
Fluorescent immunohistochemistry showed that TRAIL positive cells located on the periphery of pancreatic islets mainly. In the acini and the ducts of the exocrine pancreas there were no TRAIL immunoreactive cells. The positive cells distributed in disperse or patch pattern. The positive reactivity was mainly distributed in membrane and cytoplasm, while nuclei remained immunonegative (Figure 1 (1,4,7 and 10)).
Figure 1 (2, 5, 8) and 11 showed the localization of DcR1, DcR2, DR4 and DR5 respectively in fetal pancreas. There were a few DcR1 (Figure 1 (2)) and DcR2 (Figure 1 (5)) positive cells and the positive reactivity located in membrane mainly (green). There was no DR4 (Figure 1 (8)) and DR5 (Figure 1 (11))positive cells distributed in fetal pancreas.
Some TRAIL positive cells coexpressed with DcR1 or DcR2, whereas there was no coexpression of TRAIL and DR4, TRAIL and DR5 in fetal pancreas. All DcR1 and DcR2 immunoreactive cells showed TRAIL immunoreactivity (Figure 1 (3, 6, 9 and 12)).
Members of the TNF and TNF superfamilies of proteins are involved in the regulation of many important biological processes, including development, organogenesis, and innate and adaptive immunity[23]. The TNF-related apoptosis-inducing ligand (TRAIL, also known as Apo-2L) is a newly identified TNF superfamily member with high homology to FasL. In the TRAIL/TRAILR system, control over apoptosis relies on differential display of receptors (TRAILR). These include DR4 (TRAIL-R1) and DR5 (TRICK2/TRAIL-R2), which transduce apoptotic signals, as well as DcR1 (TRID/LIT/TRAIL-R3) and DcR2 (TRUNDD/TRAIL-R4), which lack functional death domains and act as decoys. Osteoprotegerin (OPG), a soluble decoy receptor for OPGL/RANKL/TRANCE, which is involved in osteoclast function, has also been reported to bind to TRAIL[24-36].
There were some reports on the distribution of TRAIL/TRAILR in human pancreas and pancreatic cancer. Liao et al[37] found that TRAIL-R3 mRNA and protein expression were generally weak in pancreatic cancers and normal pancreatic tissues. In contrast, TRAIL-R4 mRNA and protein were expressed at moderate to high levels in human pancreatic cancer tissues, but demonstrated weak to negative expression in the normal pancreas by Northern blotting, Western blotting and immunohistochemistry. Satoh et al[38] detected the expression of TRAIL and its receptors other than osteoprotegerin in normal pancreatic tissues using RT-PCR. In our experiment, we found for the first time that in fetal pancreatic islets, there was strong distribution of TRAIL protein. The TRAIL immunoreactive substances mainly located on the membrane and cytoplasm with negative nuclei. Some TRAIL immunoreactive cells also showed DcR1 or DcR2 positive immunstaining, whereas there were no expression of DR4 and DR5 in fetal pancreas. Our results show some consistence with what Liao et al[37] and Satoh et al[38] have found.
We all know that pancreatic cancer is one of the most aggressive cancers, partly due to the general resistance of pancreatic cancer cells towards apoptosis. Ibrahim et al[39] demonstrated that pancreatic carcinoma cells evaded the immune system in two mechanisms. One is the expression of nonfunctional receptors, decoy receptors, and molecules that block cell death, such as bcl2 and bcl-Xl. The other is the expression of apoptosis-inducing ligands, such as TRAIL, that could induce cell death of immune cells. In our experiment, we found that in fetal pancreatic islets, some cells expressed TRAIL. At the same time, DcR1 and DcR2 also colocated with TRAIL in some cells, but there was no expression of DR4 and DR5. Our findings suggest that in fetal pancreatic islets, the pancreatic cells are resistant towards apoptosis too. The characteristics of the fetal pancreas are consistent with those of pancreatic cancer.
It is well known that there are some kinds of cell types in human pancreatic islets such as A cells, B cells, D cells and PP cells. Although we found that TRAIL was located in some endocrine cells, we could not conclude which kind of cell types it existed in. This still needs to be clarified further.
Edited by Zhang JZ
| 1. | Fu XY, Wang HY, Tan L, Liu SQ, Cao HF, Wu MC. Overexpression of p28/gankyrin in human hepatocellular carcinoma and its clinical significance. World J Gastroenterol. 2002;8:638-643. [PubMed] |
| 2. | Kohut M, Nowak A, Nowakowska-Duiawa E, Marek T. Presence and density of common bile duct microlithiasis in acute biliary pancreatitis. World J Gastroenterol. 2002;8:558-561. [PubMed] |
| 3. | Yu Y, Yang WX, Wang H, Zhang WZ, Liu BH, Dong ZY. Characteristics and mechanism of enzyme secretion and increase in [Ca2+]i in Saikosaponin (I) stimulated rat pancreatic acinar cells. World J Gastroenterol. 2002;8:524-527. [PubMed] |
| 4. | Zhou ZG, Chen YD. Influencing factors of pancreatic microcirculatory impairment in acute panceatitis. World J Gastroenterol. 2002;8:406-412. [PubMed] |
| 5. | Liu B, Staren E, Iwamura T, Appert H, Howard J. Taxotere resistance in SUIT Taxotere resistance in pancreatic carcinoma cell line SUIT 2 and its sublines. World J Gastroenterol. 2001;7:855-859. [PubMed] |
| 6. | Ghaneh P, Slavin J, Sutton R, Hartley M, Neoptolemos JP. Adjuvant therapy in pancreatic cancer. World J Gastroenterol. 2001;7:482-489. [PubMed] |
| 7. | Slavin J, Ghaneh P, Sutton R, Hartley M, Rowlands P, Garvey C, Hughes M, Neoptolemos J. Management of necrotizing pancreatitis. World J Gastroenterol. 2001;7:476-481. [PubMed] |
| 8. | Fleischer F, Dabew R, Göke B, Wagner AC. Stress kinase inhibition modulates acute experimental pancreatitis. World J Gastroenterol. 2001;7:259-265. [PubMed] |
| 9. | Guo XZ, Friess H, Shao XD, Liu MP, Xia YT, Xu JH, Buchler MW. KAI1 gene is differently expressed in papillary and pancreatic cancer: influence on metastasis. World J Gastroenterol. 2000;6:866-871. [PubMed] |
| 10. | Pan QS, Fang ZP, Huang FJ. Identification, localization and morphology of APUD cells in gastroenteropancreatic system of stomach-containing teleosts. World J Gastroenterol. 2000;6:842-847. [PubMed] |
| 11. | Yan FM, Chen AS, Hao F, Zhao XP, Gu CH, Zhao LB, Yang DL, Hao LJ. Hepatitis C virus may infect extrahepatic tissues in patients with hepatitis C. World J Gastroenterol. 2000;6:805-811. [PubMed] |
| 12. | Adeghate E, Ponery AS. The role of leucine-enkephalin on insulin and glucagon secretion from pancreatic tissue fragments of normal and diabetic rats. Arch Physiol Biochem. 2001;109:223-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 13. | Yang BM, Demaine AG, Kingsnorth A. Chemokines MCP-1 and RANTES in isolated rat pancreatic acinar cells treated with CCK and ethanol in vitro. Pancreas. 2000;21:22-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 14. | Shetty S, Gladden JB, Henson ES, Hu X, Villanueva J, Haney N, Gibson SB. Tumor necrosis factor-related apoptosis inducing ligand (TRAIL) up-regulates death receptor 5 (DR5) mediated by NFkappaB activation in epithelial derived cell lines. Apoptosis. 2002;7:413-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 76] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 15. | Pettersen I, Figenschau Y, Olsen E, Bakkelund W, Smedsröd B, Sveinbjörnsson B. Tumor necrosis factor-related apoptosis-inducing ligand induces apoptosis in human articular chondrocytes in vitro. Biochem Biophys Res Commun. 2002;296:671-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 16. | Dorothée G, Vergnon I, Menez J, Echchakir H, Grunenwald D, Kubin M, Chouaib S, Mami-Chouaib F. Tumor-infiltrating CD4+ T lymphocytes express APO2 ligand (APO2L)/TRAIL upon specific stimulation with autologous lung carcinoma cells: role of IFN-alpha on APO2L/TRAIL expression and -mediated cytotoxicity. J Immunol. 2002;169:809-817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 55] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 17. | Guan B, Yue P, Lotan R, Sun SY. Evidence that the human death receptor 4 is regulated by activator protein 1. Oncogene. 2002;21:3121-3129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 51] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 18. | Eid MA, Lewis RW, Abdel-Mageed AB, Kumar MV. Reduced response of prostate cancer cells to TRAIL is modulated by NFkappaB-mediated inhibition of caspases and Bid activation. Int J Oncol. 2002;21:111-117. [PubMed] |
| 19. | Xiang H, Fox JA, Totpal K, Aikawa M, Dupree K, Sinicropi D, Lowe J, Escandón E. Enhanced tumor killing by Apo2L/TRAIL and CPT-11 co-treatment is associated with p21 cleavage and differential regulation of Apo2L/TRAIL ligand and its receptors. Oncogene. 2002;21:3611-3619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 39] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 20. | Grataroli R, Vindrieux D, Gougeon A, Benahmed M. Expression of tumor necrosis factor-alpha-related apoptosis-inducing ligand and its receptors in rat testis during development. Biol Reprod. 2002;66:1707-1715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 21. | Simon AK, Williams O, Mongkolsapaya J, Jin B, Xu XN, Walczak H, Screaton GR. Tumor necrosis factor-related apoptosis-inducing ligand in T cell development: sensitivity of human thymocytes. Proc Natl Acad Sci USA. 2001;98:5158-5163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 48] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 22. | Huang XF, Wang CM, Dai XW, Li ZJ, Pan BR, Yu LB, Qian B, Fang L. Expressions of chromogranin A and cathepsin D in human primary hepatocellular carcinoma. World J Gastroenterol. 2000;6:693-698. [PubMed] |
| 23. | Locksley RM, Killeen N, Lenardo MJ. The TNF and TNF receptor superfamilies: integrating mammalian biology. Cell. 2001;104:487-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2679] [Cited by in RCA: 2879] [Article Influence: 120.0] [Reference Citation Analysis (0)] |
| 24. | Evdokiou A, Bouralexis S, Atkins GJ, Chai F, Hay S, Clayer M, Findlay DM. Chemotherapeutic agents sensitize osteogenic sarcoma cells, but not normal human bone cells, to Apo2L/TRAIL-induced apoptosis. Int J Cancer. 2002;99:491-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 106] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 25. | Zhang HG, Xie J, Xu L, Yang P, Xu X, Sun S, Wang Y, Curiel DT, Hsu HC, Mountz JD. Hepatic DR5 induces apoptosis and limits adenovirus gene therapy product expression in the liver. J Virol. 2002;76:5692-5700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 26. | He Q, Lee DI, Rong R, Yu M, Luo X, Klein M, El-Deiry WS, Huang Y, Hussain A, Sheikh MS. Endoplasmic reticulum calcium pool depletion-induced apoptosis is coupled with activation of the death receptor 5 pathway. Oncogene. 2002;21:2623-2633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 75] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 27. | Luciano F, Ricci JE, Herrant M, Bertolotto C, Mari B, Cousin JL, Auberger P. T and B leukemic cell lines exhibit different requirements for cell death: correlation between caspase activation, DFF40/DFF45 expression, DNA fragmentation and apoptosis in T cell lines but not in Burkitt's lymphoma. Leukemia. 2002;16:700-707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 28. | van Noesel MM, van Bezouw S, Salomons GS, Voûte PA, Pieters R, Baylin SB, Herman JG, Versteeg R. Tumor-specific down-regulation of the tumor necrosis factor-related apoptosis-inducing ligand decoy receptors DcR1 and DcR2 is associated with dense promoter hypermethylation. Cancer Res. 2002;62:2157-2161. [PubMed] |
| 29. | Lamothe B, Aggarwal BB. Ectopic expression of Bcl-2 and Bcl-xL inhibits apoptosis induced by TNF-related apoptosis-inducing ligand (TRAIL) through suppression of caspases-8, 7, and 3 and BID cleavage in human acute myelogenous leukemia cell line HL-60. J Interferon Cytokine Res. 2002;22:269-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 46] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 30. | Morrison BH, Bauer JA, Hu J, Grane RW, Ozdemir AM, Chawla-Sarkar M, Gong B, Almasan A, Kalvakolanu DV, Lindner DJ. Inositol hexakisphosphate kinase 2 sensitizes ovarian carcinoma cells to multiple cancer therapeutics. Oncogene. 2002;21:1882-1889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 50] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 31. | Nzeako UC, Guicciardi ME, Yoon JH, Bronk SF, Gores GJ. COX-2 inhibits Fas-mediated apoptosis in cholangiocarcinoma cells. Hepatology. 2002;35:552-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 118] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 32. | Koyama S, Koike N, Adachi S. Expression of TNF-related apoptosis-inducing ligand (TRAIL) and its receptors in gastric carcinoma and tumor-infiltrating lymphocytes: A possible mechanism of immune evasion of the tumor. J Cancer Res Clin Oncol. 2002;128:73-79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 51] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 33. | Legembre P, Moreau P, Daburon S, Moreau JF, Taupin JL. Potentiation of Fas-mediated apoptosis by an engineered glycosylphosphatidylinositol-linked Fas. Cell Death Differ. 2002;9:329-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 34. | Bretz JD, Mezosi E, Giordano TJ, Gauger PG, Thompson NW, Baker JR. Inflammatory cytokine regulation of TRAIL-mediated apoptosis in thyroid epithelial cells. Cell Death Differ. 2002;9:274-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 35. | Park SY, Billiar TR, Seol DW. IFN-gamma inhibition of TRAIL-induced IAP-2 upregulation, a possible mechanism of IFN-gamma-enhanced TRAIL-induced apoptosis. Biochem Biophys Res Commun. 2002;291:233-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 36. | Odoux C, Albers A, Amoscato AA, Lotze MT, Wong MK. TRAIL, FasL and a blocking anti-DR5 antibody augment paclitaxel-induced apoptosis in human non-small-cell lung cancer. Int J Cancer. 2002;97:458-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 37. | Liao Q, Friess H, Kleeff J, Büchler MW. Differential expression of TRAIL-R3 and TRAIL-R4 in human pancreatic cancer. Anticancer Res. 2001;21:3153-3159. [PubMed] |
| 38. | Satoh K, Kaneko K, Hirota M, Masamune A, Satoh A, Shimosegawa T. Tumor necrosis factor-related apoptosis-inducing ligand and its receptor expression and the pathway of apoptosis in human pancreatic cancer. Pancreas. 2001;23:251-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 39. | Ibrahim SM, Ringel J, Schmidt C, Ringel B, Müller P, Koczan D, Thiesen HJ, Löhr M. Pancreatic adenocarcinoma cell lines show variable susceptibility to TRAIL-mediated cell death. Pancreas. 2001;23:72-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 64] [Article Influence: 2.7] [Reference Citation Analysis (0)] |