Published online Dec 15, 1998. doi: 10.3748/wjg.v4.i6.513
Revised: November 20, 1998
Accepted: December 5, 1998
Published online: December 15, 1998
AIM: To recognize the characteristic findings of non-pathologic perfusion defects with CT arterial portography (CTAP) and nonpathologic enhancement found in CT hepatic arteriography (CTHA).
METHONDS: The manifestations of nonpathologic perfusion defects with CTAP and non-pathologic enhancement found in CTHA were analyzed in 50 patients with primary hepatocellular carcinoma.
RESULTS: The false-positive rate of perfusion defects detected in CTAP was 15.1%. The shapes of perfusion defects were peripheral wedge, small, round, and patchy. The occurrence rate of non-pathologic enhancement found in CTHA was 22.0%. The shapes of non-pathologic enhancement were small, round, irregular, and wedge.
CONCLUSION: There was high frequency of non-pathologic perfusion defects detected with CTAP and non-pathologic enhancement found in CTHA. The simultaneous use of both procedures may help decrease the false-positive rate, and increase the veracity of diagnosis for hepatocellular carcinoma.
- Citation: Li L, Wu PH, Lin HG, Li JQ, Mo YX, Zheng L, Lu LX, Ruan CM, Chen L. Findings of non-pathologic perfusion defects by CT arterial portography and non-pathologic enhancement of CT hepatic arteriography. World J Gastroenterol 1998; 4(6): 513-515
- URL: https://www.wjgnet.com/1007-9327/full/v4/i6/513.htm
- DOI: https://dx.doi.org/10.3748/wjg.v4.i6.513
CT arterial portography (CTAP) and CT hepatic arteriography (CTHA) are the most sensitive methods of detecting hepatocellular carcinoma[1,2]. In recent years, there are more reports on non-pathologic perfusion defects of CTAP and non-pathologic enhancement CTHA. To better recognize and understand the characteristic manifestation of non-pathologic perfusion defects with CTAP and non-pathologic enhancement in CTHA, we analyzed the CT images of 50 cases of hepatocellular carcinoma on CTAP and CTHA in our hospital from January 1995 to January 1998.
Fifty patients (44 man, 6 women) with hepatocellular carcinoma were examined with CTAP and CTHA in our department. They ranged in age from 21 to 65 years (mean age, 413 years). AFP was elevated in 41 patients. Cirrhosis occurred in 42 cases. Child’s classification showed 16 cases of Child’s A, 30 cases of Child’s B, and 4 cases of Child¡äs C. The tumors measured 0.2 cm-5.5 cm in size with a mean of 3.6 cm. Multiple nodules were found in 42 cases.
CTAP examinations were performed with incremental scanning of liver in cranial-to-caudal direction with 8-mm or 10-mm collimation on bi-spiral Elscint Twin Flash scanner (Elscint Corp.). CT images were obtained 25 s-35 s after the initiation of transcatheter (5-F) superior mesenteric artery injection of 30 mL-40 mL of non-ionic contrast material. Contrast material was injected at a rate of 3.0 mL/s-3.5 mL/s with an automatic power injector (Medrad, Pittsburgh). Conventional angiography was not performed before CTAP. During the catheterization, contrast material administered before CT scanning was limited to 5 mL-10 mL injected by hand to visualize any aberrant vessels and to facilitate proper catheter placement.
CTHA was done by injecting contrast material into the proper hepatic artery, the common hepatic artery, or the celiac artery. The volume of 20 mL-30 mL of contrast material was injected at a rate of 3.0 mL/s-3.5 mL/s. Consecutive scanning of the liver was started 6 s-8 s after the initiation of injection of contrast material. After CTAP and CTHA examination, 3 mL-15 mL of lipiodol was injected into the hepatic artery, and plain CT scan of liver was performed after two or three weeks (Lipiodol CT, Lp-CT).
CTAP and CTHA images of 50 cases were interpreted prospectively double-blind by two radiologists, and confirmed pathologically combined with lipiodol deposits in Lp-CT, and 6 mo-16 mo follow-up (42 tumors of 18 cases proved by operative pathology; 30 lesions of 15 cases by biopsy, and the other 27 cases by Lp-CT and follow-up). A total of 232 tumors were found, including 214 tumors detected by CTAP (the rate of detectability was 92.2%), 209 detected by CTHA (the rate of detectability was 90.1%), and 220 detected by simultaneous use of both procedures (the rate of detectability was 94.8%). Our study did not reveal a statistically significant difference in the sensitivities of CTAP, CTHA and simultaneous use of both procedures.
A total of 252 perfusion defects were detected with CTAP in the 50 patients, including 38 non-pathologic abnormal perfusion defects in 18 cases with a false positive rate of 15.1%. The shape of perfusion defects was peripheral wedge, small round and patchy. All the non-pathologic perfusion defects were not demonstrated on CTHA images.
A total of 268 enhancement foci were found with CTHA in 50 patients, including 59 non-pathologic enhancement foci in 21 cases with an occurrence rate of 22.0%. The shape of non-pathologic enhancement was small round, irregular and wedge.
In recent years, with the development of spiral CT technique, CTAP and CTHA have become widely used in diagnosis and differential diagnosis of small hepatocellular carcinoma. Compared with non-spiral CT arteriography, spiral CT arteriography has achieved great improvement not only in scanning technique, but also in quality of CT images[1]. We scanned the entire liver consecutively, and obtained excellent images of CTAP and CTHA by the technique of a single breath-hold scanning, with a comparatively small quantity of contrast material (CTAP 30 mL-40 mL, CTHA 20 mL-30 mL). Compared with conventionally enhanced CT (contrast material administrated by venous injection), CTAP and CTHA have a high detectability of 80%-96%[2]. In our 50 patients examined with CTAP and CTHA, the detectability rate of CTAP, CTHA and simultaneous use of both procedures were 92.2%, 90.1% and 94.8% respectively.
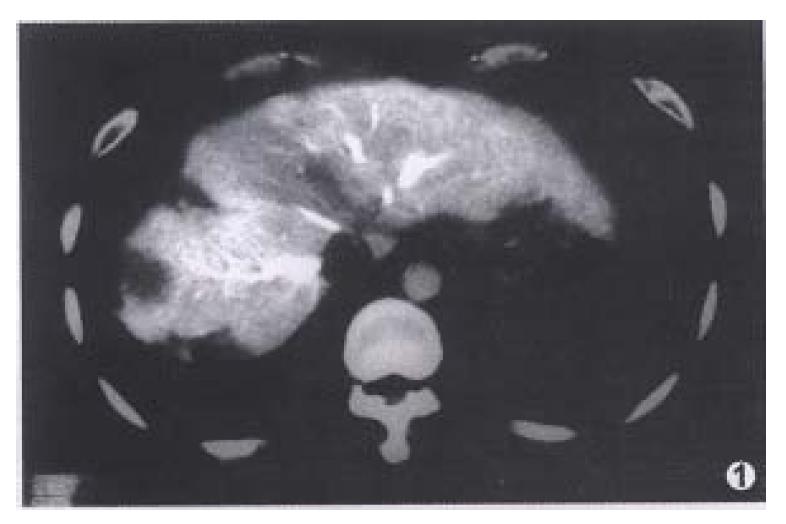
With the widespread use of CTAP and CTHA, there have been more reports on non-pathologic perfusion defects found with CTAP and non-pathologic enhancement with CTHA. The manifestation of pseudolesion in CTAP was noted relatively earlier. Due to aberration of blood supply for some parts of the liver, such as arterio-portal shunt[3], cirrhotic nodule, focal nodular hyperplasia, and focal fatty infiltration, the normal tissue could show up as perfusion defects in CTAP, especially in the medial segment of the left lobe[4]. lrie et al[5] reported that perfusion defects on CTAP are sometimes nonspecific, and suggested that peripheral flat or wedgeshaped perfusion defects indicate benignity. Their study showed that, except for cysts, perfusion defects larger than 1.5 cm may indicate malignancy, and combination with CTHA might be helpful in differentiating malignant from benign perfusion defects with CTAP. Peterson et al[6] reported that peripheral wedge-shaped perfusion defects on CTAP were nearly uniformly benign, and may be used as a criterion of benignity. They hypothesized that the pathogenesis of these peripheral wedge-shaped perfusion defects may represent variations in the normal portal perfusion of the microvasculature of the peripheral sub-capsular liver parenchyma. In our series of 50 cases, the occurrence rate of non-pathologic perfusion defects on CTAP was 15.1%, and peripheral wedge-shaped perfusion defects were the most common (Figure 1).
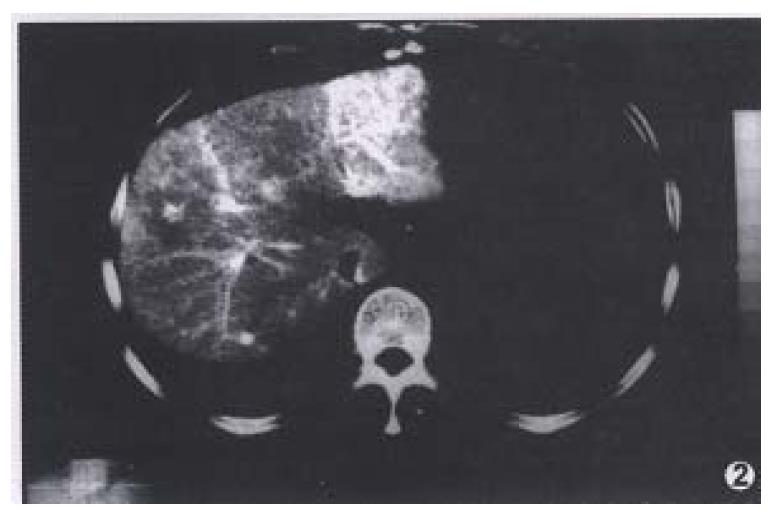
Since the advent of helical CT technique, CTHA became more widely used in the differential diagnosis for hepatocellular carcinoma[7]. Similar to findings of CTAP, pseudo-lesion enhancement of normal liver tissue also occurred in CTHA. Kanematsu et al[8] supposed that local non-pathologic enhancement detected with CTHA might result from the cystic venous drainage or peripheral arterio-portal shunts. They analyzed the frequency, size, location, and shape of local non-pathologic enhancement on CTHA. In a series of 31 patients examined with CTHA, 36 non-pathologic enhancements were found in 14 cases, the occurrence rate being 36.4%. The shapes of non-pathologic enhancement on CTHA appeared round, veriform, irregular, punctate, and wedge-like. Although the shape of the enhancement was nonspecific, Irie et al[5] suggested that rim enhancement indicated malignancy in both cirrhotic and non-cirrhotic liver. In our study, the occurrence rate of non-pathologic enhancement on CTHA was 22.0%, and small rounded peripheral enhancement was the most common (Figure 2). In practice, we discovered that the frequency of non-pathologic enhancement was to some extent, related to the depth of catheterization at which CTHA was performed. There was a relatively higher frequency of non-pathologic enhancement in CTHA formed at catheterization of hepatic proper artery, compared with CTHA formed at catheterization of celiac artery trunk. The reason might be the relatively larger volume of contrast material and higher speed of injection in CTHA performed at catheterization of hepatic proper artery. Thus, we did not recommend CTAP or CTHA alone for interpretation of hepatocellular carcinoma. We suggest that the simultaneous use of both procedures help decrease the false positive rate of CTAP and CTHA, and increase the veracity of diagnosis for hepatocellular carcinoma.
Project supported by “9.5” Natioal Major Project of National Committee of Sciences and Technology, No. 96-907-03-02
| 1. | Hori M, Murakami T, Oi H, Kim T, Takahashi S, Matsushita M, Tomoda K, Narumi Y, Kadowaki K, Nakamura H. Sensitivity in detection of hypervascular hepatocellular carcinoma by helical CT with intra-arterial injection of contrast medium, and by helical CT and MR imaging with intravenous injection of contrast medium. Acta Radiol. 1998;39:144-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 2. | Kanematsu M, Oliver JH, Carr B, Baron RL. Hepatocellular carcinoma: the role of helical biphasic contrast-enhanced CT versus CT during arterial portography. Radiology. 1997;205:75-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 71] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 3. | Tamura S, Kihara Y, Yuki Y, Sugimura H, Shimizu T, Adjei ON, Watanabe K. Pseudo lesions on CTAP secondary to arterio-portal shunts. Clin Imaging. 1997;21:359-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 4. | Matsui O, Kadoya M, Yoshikawa J, Gabata T, Kawamori Y, Ueda K, Nobata K, Takashima T. Posterior aspect of hepatic segment IV: patterns of portal venule branching at helical CT during arterial portography. Radiology. 1997;205:159-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 5. | Irie T, Takeshita K, Wada Y, Kusano S, Terahata S, Tamai S, Hatsuse K, Aoki H, Sugiura Y. CT evaluation of hepatic tumors: comparison of CT with arterial portography, CT with infusion hepatic arteriography, and simultaneous use of both techniques. AJR Am J Roentgenol. 1995;164:1407-1412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 6. | Peterson MS, Baron RL, Dodd GD, Zajko AJ, Oliver JH, Miller WJ, Carr BI, Bron KM, Campbell WL, Sammon JK. Hepatic parenchymal perfusion defects detected with CTAP: imaging-pathologic correlation. Radiology. 1992;185:149-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 76] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 7. | Murakami T, Oi H, Hori M, Kim T, Takahashi S, Tomoda K, Narumi Y, Nakamura H. Helical CT during arterial portography and hepatic arteriography for detecting hypervascular hepatocellular carcinoma. AJR Am J Roentgenol. 1997;169:131-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 85] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 8. | Kanematsu M, Hoshi H, Imaeda T, Yamawaki Y, Mizuno S, Manabe T, Enya M, Yokoyama R. Nonpathological focal enhancements on spiral CT hepatic angiography. Abdom Imaging. 1997;22:55-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |