Published online Sep 28, 2022. doi: 10.3748/wjg.v28.i36.5240
Peer-review started: May 17, 2022
First decision: June 19, 2022
Revised: June 23, 2022
Accepted: September 8, 2022
Article in press: September 8, 2022
Published online: September 28, 2022
Processing time: 128 Days and 19 Hours
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can cause pancreatic damage, both directly to the pancreas via angiotensin-converting enzyme 2 receptors (the transmembrane proteins required for SARS-CoV-2 entry, which are highly expressed by pancreatic cells) and indirectly through locoregional vasculitis and thrombosis. Despite that, there is no clear evidence that SARS-CoV-2 is an etiological agent of acute pancreatitis. Acute pancreatitis in coronavirus disease 2019 (COVID-19) positive patients often recognizes biliary or alcoholic etiology. The prevalence of acute pancreatitis in COVID-19 positive patients is not exactly known. However, COVID-19 positive patients with acute pancreatitis have a higher mortality and an increased risk of intensive care unit admission and necrosis compared to COVID-19 negative patients. Acute respiratory distress syndrome is the most frequent cause of death in COVID-19 positive patients and concomitant acute pancreatitis. In this article, we reported recent evidence on the correlation between COVID-19 infection and acute pancreatitis.
Core Tip: The outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), causing a severe acute respiratory syndrome, has rapidly spread from China all over the world, affecting millions of people. Whereas typical presentations of this infection (such as fever, cough, myalgia, fatigue and pneumonia) are well recognized, several studies report a low incidence of gastrointestinal symptoms. The relationship between SARS-CoV-2 infection and acute pancreatitis is controversial. Acute pancreatitis is not specifically caused by SARS-CoV-2. Furthermore, coronavirus disease 2019 positive patients are more likely to develop severe acute pancreatitis and multiple organ failure.
- Citation: Brisinda G, Chiarello MM, Tropeano G, Altieri G, Puccioni C, Fransvea P, Bianchi V. SARS-CoV-2 and the pancreas: What do we know about acute pancreatitis in COVID-19 positive patients? World J Gastroenterol 2022; 28(36): 5240-5249
- URL: https://www.wjgnet.com/1007-9327/full/v28/i36/5240.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i36.5240
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) secondary to coronavirus disease 2019 (COVID-19) has spread rapidly from China. The disease has affected millions of people[1]. Whereas typical presentations of this infection (such as fever, cough, myalgia, fatigue and pneumonia) are well recognized, few studies reported the incidence of atypical gastrointestinal symptoms[2-5]. COVID-19 is linked to organ damage including lungs, heart and kidneys and can lead to multiple organ failure. Evidence shows that SARS-CoV-2 has a strong tropism for the gastrointestinal tract[6,7]. Moreover, pancreatic injury in COVID-19 has not been common.
Acute pancreatitis is an inflammatory process that originates from the glandular parenchyma, causing damage and/or destruction to the acinar component first and then extending to the surrounding tissues[8-11]. There are several etiological factors contributing to its onset. However, it is possible to ascribe most of the episodes to two main causes, namely gallbladder lithiasis and alcohol abuse. Pharmacological, iatrogenic and viral causes must also be taken into account[12].
The severity of acute pancreatitis does not always correlate with pancreatic structural changes. It is possible to distinguish interstitial pancreatitis when the organ is locally or wholly increased in volume due to the presence of interstitial edema and necrotizing pancreatitis (10%-20% of all cases)[10,13,14]. It is unclear why COVID-19 starts with exclusively gastrointestinal symptoms, albeit in a low percentage of cases. However, acute pancreatic involvement induced by COVID-19 is severe and can develop rapidly. Close monitoring and admission are necessary to offer proper treatment. The following sections of this editorial discuss some of the recent findings and the approaches for a more effective clinical diagnosis and treatment of acute pancreatitis in patients with SARS-CoV-2 infection.
Viral RNA has been found in the stools of COVID-19 patients[1,2,15-17]. High replication of the virus has been documented in both the small and large intestines by electron microscopy studies on tissues derived from biopsies and/or autopsy material[7,18,19]. Additionally, fecal-oral transmission has been documented for SARS-CoV-2[20]. Some molecules are receptors for the virus. These determine a specific tropism for different tissues and organs. SARS-CoV-2 uses its surface envelope called the spike glycoprotein. By means of this protein, SARS-CoV-2 interacts and enters host cells. The virus enters the cell via the angiotensin-converting enzyme 2 (ACE2) receptor. As such, the spike glycoprotein-ACE2 binding is the determinant for virus entry and propagation and for the transmissibility of COVID-19-related disease. ACE2 is expressed in human pancreatic cells and pancreatic islets and highly represented in pancreas microvasculature pericytes[6,21,22]. As ACE2 is internalized by SARS-CoV-2 binding, an imbalance of the RAS peptides can be established with a rise of angiotensin II and a decrease in angiotensin 1-7. The latter exerts anti-thrombogenic, anti-inflammatory and pro-resolving actions[23]. Laboratory abnormalities, suggesting pancreatic injury, have been noted in 8.0%-17.5% of cases in several studies[24,25], with 7.0% displaying significant pancreatic changes on computed tomography.
The etiopathogenesis of pancreatic injury in SARS-CoV-2 patients is still unclear. Both pancreatic ACE2 expression and drugs taken before the hospitalization might be involved[26]. The inflammation that occurs in the intestine causes the release of cytokines and bacteria. Both cytokines and bacteria can reach and enter the lung through the bloodstream. At this level, a direct influence on the immune response and inflammation is observed[27-29]. The gut-liver axis is also strongly influenced by damage to the intestinal mucosa and bacterial imbalance[30]. The host and the microbial metabolites present in the intestine are transferred to the liver through the mesenteric-portal circulation; these affect liver function. The liver releases bile acids and bioactive media into the biliary and systemic circulation in order to transport them to the intestine. This could lead to pancreatic function damage in patients and may also explain the abnormality of pancreatic function indicators in COVID-19 patients. It is important not to consider each COVID-19 patient with increased serum amylase level as affected by acute pancreatitis[31]. Elevated levels of pancreatic enzymes may occur during kidney failure or diarrhea in the course of COVID-19[32]. It could be useful to find some criteria that can guide clinical suspicion.
Available literature is not able to determine whether the tissue damage leading to acute pancreatitis occurs as a result of direct SARS-CoV-2 infection or as a results of systemic multiple organ dysfunction with increased levels of amylase and lipase[33,34]. Only a few COVID-19 positive patients also have acute pancreatitis; the association between the two pathologies is infrequent. Only 10% of COVID-19 positive patients show abdominal symptoms exclusively[35]: Usually, these patients have the most severe forms of COVID-19 infections. Furthermore, it has not been demonstrated that an increased incidence of acute pancreatitis occurred during the COVID-19 pandemic period[36]. SARS-CoV-2 cannot be said to be an etiological agent of acute pancreatitis[33,37]. Acute pancreatitis in COVID-19 positive patients is idiopathic in most cases[37], and there is no sufficient evidence showing that COVID-19 can cause acute pancreatitis or negatively impact its prognosis.
The involvement of the pancreatic gland appears possible, in consideration of the fact that ACE2 receptors are present both on exocrine cells and on pancreatic islets[23]. In addition, the ACE2 receptor is expressed more in the pancreatic tissue than in the lungs. Furthermore, SARS-CoV-2 induces the onset of endotheliitis, which results in ischemic damage and that can also occur in the pancreas[38].
In the COVID PAN study, it was documented that acute pancreatitis is more severe in COVID-19 positive patients than in COVID-19 negative patients. In them, the onset of multiple organ failure can be linked to factors other than acute pancreatitis[39]. A greater severity of acute pancreatitis, an increased risk of necrosis, intensive care unit admission, persistent organ failure and the need for mechanical ventilation were observed in COVID-19 positive patients with acute pancreatitis. In this same study, 30-d overall mortality from acute pancreatitis was statistically higher in COVID-19 positive (14.7%) than in COVID-19 negative patients (2.6%, P < 0.04)[39]. Furthermore, necrosectomy was more likely to be performed in SARS-CoV-2 positive patients, occurring in 5% compared with 1.3% in the control group (P < 0.001).
The predominant organ dysfunction was respiratory failure in the majority of COVID-19 positive patients. In addition, acute pancreatitis with concomitant SARS-CoV-2 was more likely to have poorer outcomes due to double pulmonary damage[40]. Lung involvement is common in severe acute pancreatitis. This involvement can progress to full blown acute respiratory distress syndrome[40]. At present, it could be difficult to stratify the severity of symptoms and the degree of lung involvement. Acute respiratory distress syndrome due to acute pancreatitis can worsen lung injury related to COVID-19 pneumonia. Moreover, changes in the gastrointestinal flora affect the respiratory tract through the common mucosal immune system, the so-called “gut-lung axis”[27,29]. COVID-19 positive patients with gastrointestinal symptoms are more likely to be complicated with acute respiratory distress and pancreatic damage. In these patients the prognosis is poorer. In COVID-19 positive patients with gastrointestinal symptoms, attention should be paid to the patient’s gastrointestinal symptoms in the diagnosis and treatment process. It also appears essential to prevent the transmission of the virus via the fecal-oral route. In a retrospective cohort study, Inamdar et al[37] concluded that pancreatitis should be included in the list of gastrointestinal manifestations of COVID-19.
There are few studies in the literature that investigate the relationship between COVID-19 infection and acute pancreatitis. In their experience, Inamdar et al[37] documented 189 cases of acute pancreatitis in 48012 hospitalized patients (0.39%). Thirty-two (17%) of these 189 patients were COVID-19 positive, with a prevalence of 0.27% of acute pancreatitis among the patients hospitalized for COVID-19. Idiopathic forms were more frequent (69%) in this patient group than in COVID-19 negative patients (21%, P < 0.0001). Wang et al[41] found that of 52 COVID-19 patients enrolled, 17% had pancreatic injury, defined as an abnormal increase of amylase and lipase serum levels. Stephens et al[31] showed that COVID-19 patients had an amylase serum peak not always related with acute pancreatitis. They enrolled 234 patients, 158 of which had a serum amylase level three times greater than the normal upper limit, but only 1.7% of the studied population met the revised criteria of Atlanta for diagnosis of acute pancreatitis[31].
Vaccines are considered one of the most important public health achievements of the last century. Many vaccines have side effects. One of these adverse events is the onset of pancreatitis. In these cases, it is believed that acute pancreatitis is secondary to an immunologically induced phenomenon as demonstrated by reports in the literature after the administration of vaccines against the hepatitis virus or other viruses[42-44]. The vaccine BNT162b1 developed by Pfizer-BioNTech is a nucleoside-modified mRNA that encodes the receptor binding domain of the SARS-CoV-2 spike protein. The vaccine RNA is formulated in lipid nanoparticles for an efficient delivery into cells after injections[45].
In the literature there are several cases reporting acute pancreatitis after mRNA-based vaccine[46-50]. Although the vaccine has been proven to be effective and safe, vaccine-induced side effects have been observed. There are nausea, diarrhea, decreased appetite, abdominal pain, vomiting, heartburn and constipation among the most frequently reported gastrointestinal side effects. Most of the cases describe mild acute pancreatitis, however Ozaka et al[46] and Walter et al[47] reported a single case of necrotizing pancreatitis. According to Pfizer’s data, 1 case of pancreatitis and 1 case of obstructive pancreatitis as adverse reactions were observed during the phase II/clinical trial of the COVID-19 mRNA vaccine. The trial included about 38000 participants, indicating that such a link between vaccination and pancreatitis is a very rare adverse event[51]. Between December 9, 2020 and July 21, 2021, information obtained from the United Kingdom database showed 275820 adverse reaction reports, which included 18 cases of mild acute pancreatitis and 1 case of necrotizing pancreatitis[52]. In France, out of a total of 42523573 doses, the Agence Nationale de Securité du Medicament et des Produits de Santé reported 57 cases of acute pancreatitis[53]. VigiBase, the World Health Organization global database of individual case safety reports, included 298 cases of acute pancreatitis and 17 cases of necrotizing pancreatitis[54]. The Agenzia Italiana del Farmaco reported 497 gastrointestinal adverse events equal to 14.1% of the total observations in 1 year. The document does not specify the number of cases of acute pancreatitis[55]. Although it is difficult to make conclusions about the likelihood of the vaccine being the etiologic factor of pancreatitis, it is essential to continue monitoring it for possible under-reported side effects until we have extensive long-term data available in post-marketing surveillance for long-term and rare side effects. Surveillance is also necessary because there are currently no conclusive data on the severity of pancreatitis in the population of subjects vaccinated against COVID-19 compared to patients who have not undergone vaccination. Preliminary results would document a reduced incidence of severe forms of acute pancreatitis in subjects vaccinated against SARS-CoV-2.
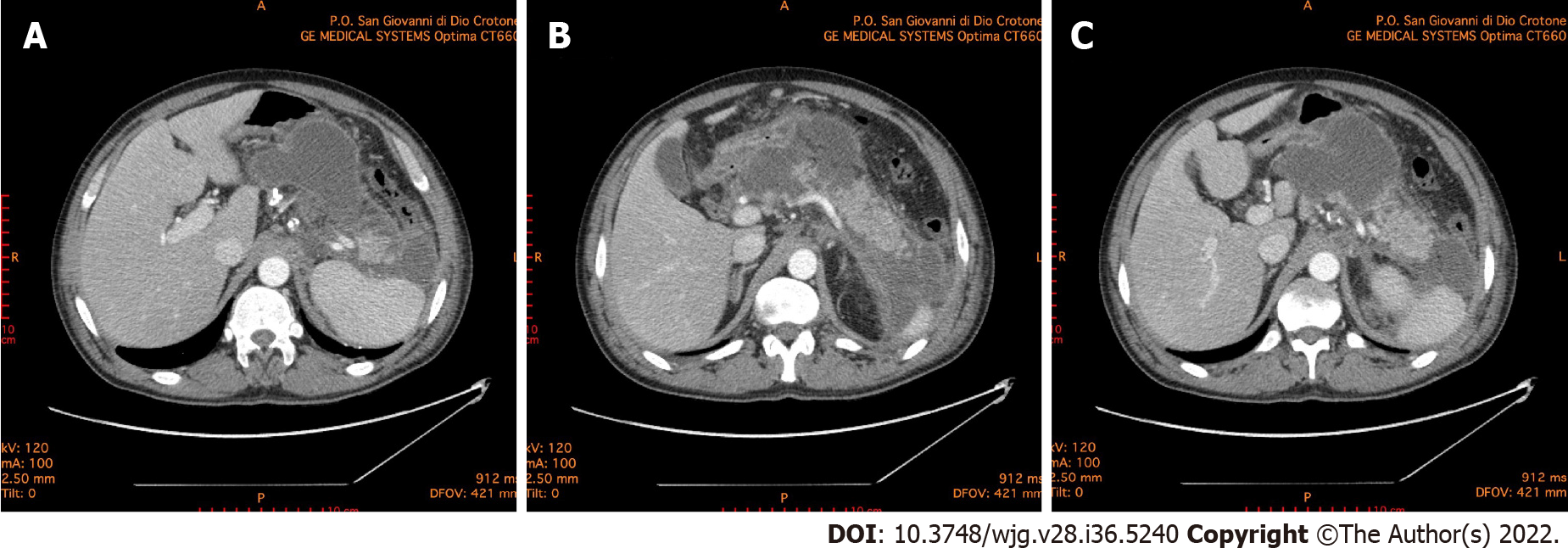
Since pancreatitis in COVID-19 positive patients occurs more frequently in severe forms, treatment must be intensive and prompt. It is crucial to define and stratify the severity of illness in patients with acute pancreatitis because of the extreme range of potential clinical courses due to the wide range of organs and tissues that may become involved. It is also fundamental to identify patients with potentially severe pancreatitis who require a multidisciplinary approach and an earlier and more aggressive treatment[56,57]. Suspected acute pancreatitis can be confirmed with laboratory and instrumental investigations. Increased pancreatic enzymes levels are associated with a poor prognosis in COVID-19 patients. Recent findings show that the increment of pancreatic enzymes are significant in critical COVID-19 patients, but only a few of them progress to acute pancreatitis[58]. The rapid response of C-reactive protein to changes in the inflammatory process intensity has also suggested its use in the management and monitoring of acute pancreatitis. C-reactive protein is not specific, and although it is correlated with severity, it cannot be used to predict clinical evolution. Serum procalcitonin is also one of the parameters used in predicting the development of severe acute pancreatitis. Its continuous increase correlates with a bacterial superinfection of pancreatic necrosis[59]. In all patients with or without SARS-CoV-2 illness contrast-enhanced computed tomography scan is the gold standard for the diagnosis of acute pancreatitis to evaluate both pancreatic and extrapancreatic alterations (Figure 1).
The revised Atlanta classification provides a good clinical distinction between mild, moderate and severe acute pancreatitis, and it is used in most COVID-19 positive patients with acute pancreatitis[8]. Several scoring systems have been developed to predict the severity of acute pancreatitis; however, none of them represents a gold standard. The scoring systems are without clinical relevance because of their low predictive value[56]. The bedside index for severity in acute pancreatitis failed to identify the severity of acute pancreatitis in COVID-19 positive patients[37].
Early fluid resuscitation is recommended in order to improve tissue perfusion[60], and the maintenance of microcirculation may be associated with resolution of multiple organ failure[61], especially in patients with COVID-19 and acute pancreatitis. Enteral nutrition is safe during acute pancreatitis[62]. Moreover, supplementation of enteral nutrition with probiotics may decrease septic complications. In patients with COVID-19 requiring mechanical ventilation, early enteral nutrition is associated with earlier liberation from ventilator support, shorter intensive care unit and hospital stay and decreased cost[63,64].
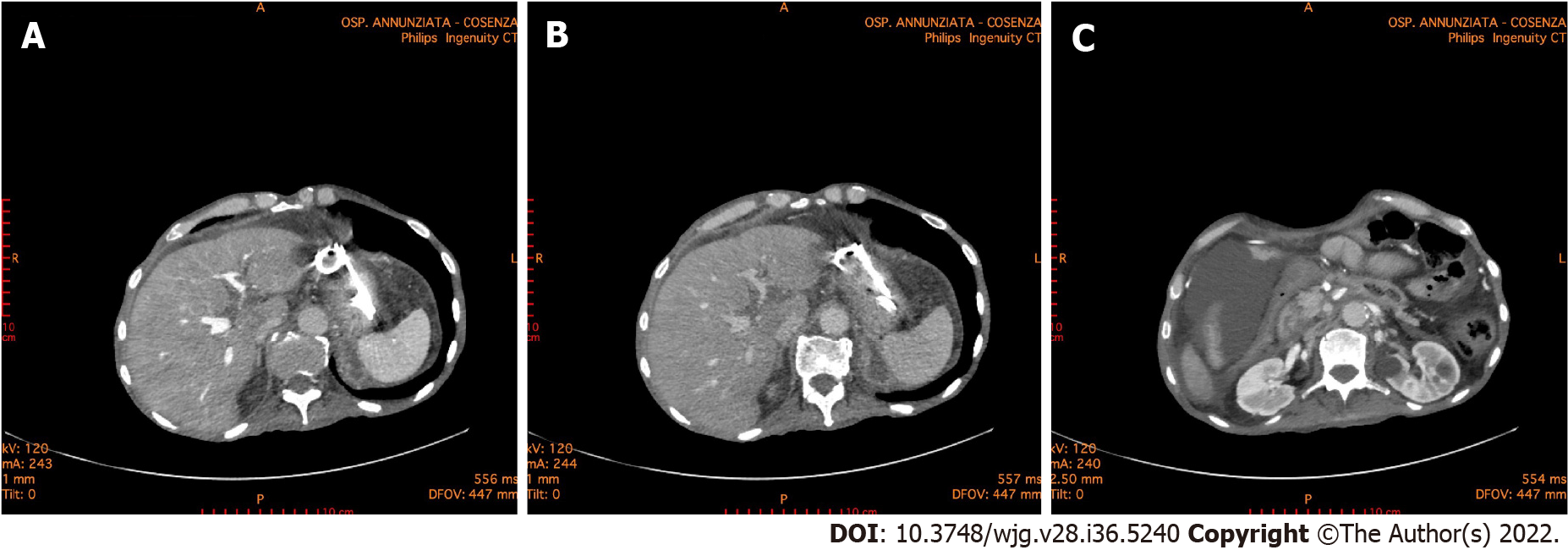
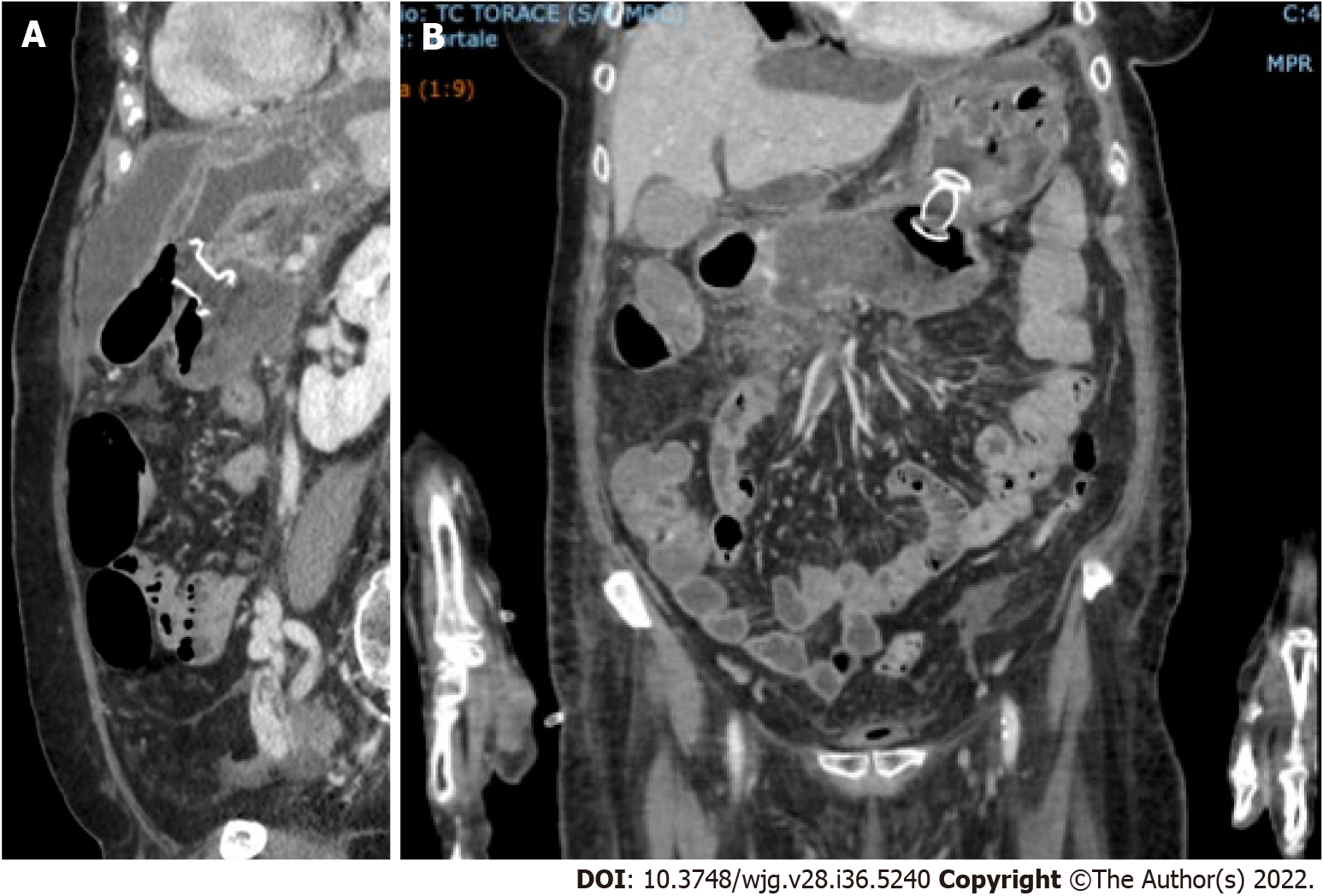
Most patients with sterile necrosis can be managed without invasive treatments. Walled off necrotic collections or pseudocysts may cause mechanical obstruction and a step-up approach is indicated. Percutaneous or transmural endoscopic drainage are both appropriate first-line approaches in managing these patients (Figure 2). However, endoscopic drainage is preferred as it avoids the risk of forming a pancreatocutaneous fistula. In the earlier phase of the pandemic, patients with COVID-19, especially with severe pneumonia and considered highly contagious, did not undergo endoscopic ultrasound or endoscopic treatments. In the later stages, patients with COVID-19 and acute pancreatitis were subjected, with necessary precautions, to percutaneous and endoscopic treatments similar to the COVID-19 negative patients (Figure 3).
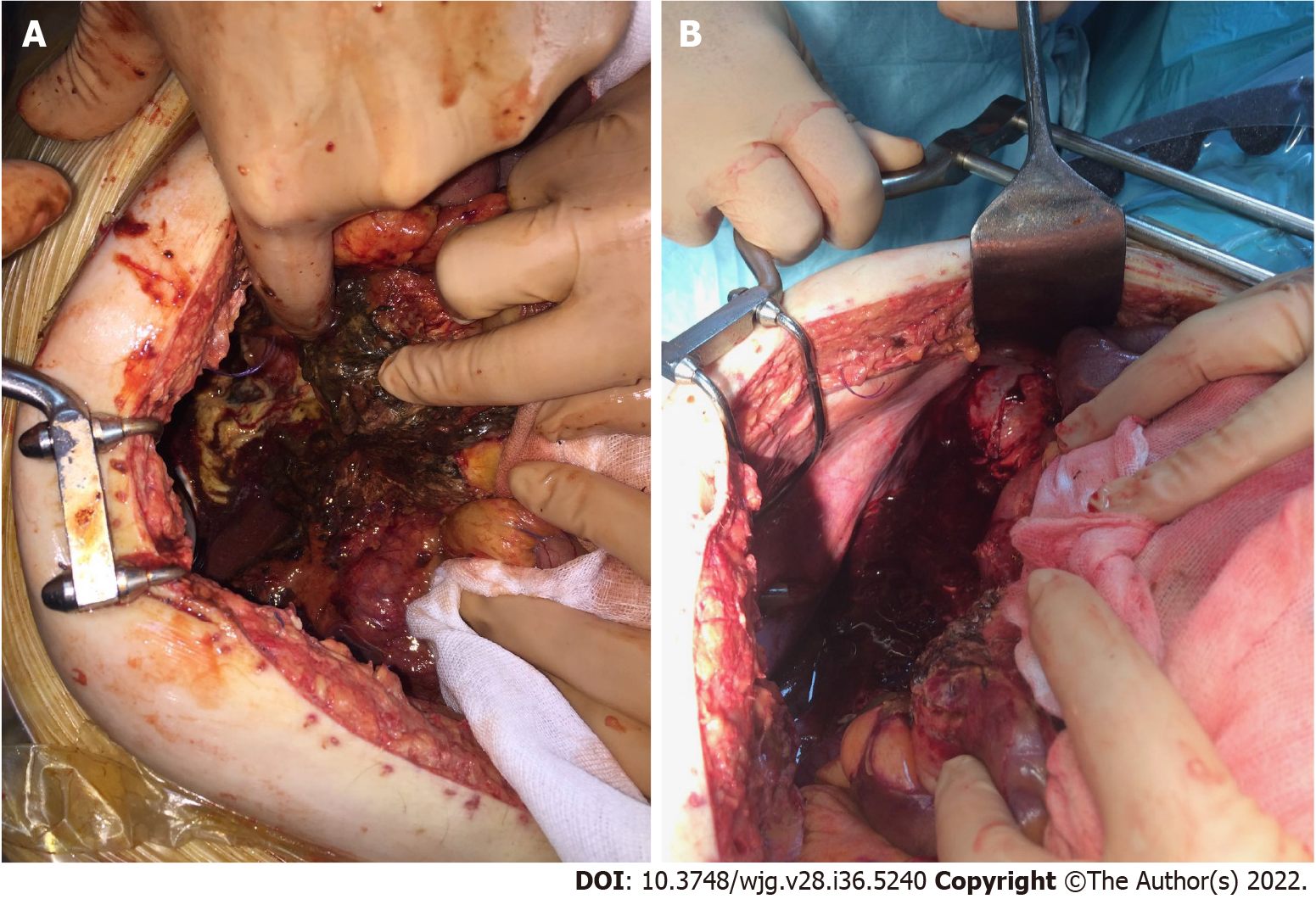
The step-up approach might reduce the rates of complications and death by minimizing surgical trauma in already critically ill patients[65,66]. Currently, there is general agreement that surgery in severe acute pancreatitis should be performed as late as possible[67]. In the management of necrotizing acute pancreatitis, open operative debridement (Figure 4) maintains a role in cases not amenable to less invasive endoscopic and/or laparoscopic procedures. Open operative debridement was performed in patients with COVID-19 related lung symptoms and lesions.
In pandemic times, clinical conditions could be worsened by COVID-19 infection. SARS-CoV-2 cannot be said to be an etiological agent of acute pancreatitis. Acute pancreatitis in COVID-19 positive patients is idiopathic in most cases, and there is no sufficient evidence showing that SARS-CoV-2 can negatively impact prognosis. On the other hand, acute pancreatitis with concomitant SARS-CoV-2 is more likely to have worse outcomes due to double lung damage and greater pancreatic severity. The multiparametric scores could not recognize and stratify the severity of pancreatic diseases and concomitant COVID-19 infection. In these patients, computed tomography is the gold standard for the diagnosis. Management of COVID-19 positive patients with pancreatitis is complex, and it is optimally provided by a multidisciplinary team. Operative treatments should be modulated, preferably, from the least to the most invasive option. Thus, surgical necrosectomy is relegated to the role of “last resort”, remembering that necrotizing pancreatitis is a heterogeneous disease with marked variations in extent and course. This also means that one size treatment does not fit all.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Cai J, China; Gonçalves TN, Portugal S-Editor: Wang JJ L-Editor: Filipodia P-Editor: Wang JJ
| 1. | Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS; China Medical Treatment Expert Group for Covid-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382:1708-1720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19202] [Cited by in RCA: 18859] [Article Influence: 3771.8] [Reference Citation Analysis (7)] |
| 2. | Henry BM, de Oliveira MHS, Benoit J, Lippi G. Gastrointestinal symptoms associated with severity of coronavirus disease 2019 (COVID-19): a pooled analysis. Intern Emerg Med. 2020;15:857-859. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 3. | Nobel YR, Phipps M, Zucker J, Lebwohl B, Wang TC, Sobieszczyk ME, Freedberg DE. Gastrointestinal Symptoms and Coronavirus Disease 2019: A Case-Control Study From the United States. Gastroenterology. 2020;159:373-375.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 182] [Cited by in RCA: 265] [Article Influence: 53.0] [Reference Citation Analysis (0)] |
| 4. | Nayar M, Varghese C, Kanwar A, Siriwardena AK, Haque AR, Awan A, Balakrishnan A, Rawashdeh A, Ivanov B, Parmar C, Halloran CM, Caruana C, Borg CM, Gomez D, Damaskos D, Karavias D, Finch G, Ebied H, Pine JK, Skipworth JRA, Milburn J, Latif J, Apollos J, El Kafsi J, Windsor JA, Roberts K, Wang K, Ravi K, Coats MV, Hollyman M, Phillips M, Okocha M, Wilson MS, Ameer NA, Kumar N, Shah N, Lapolla P, Magee C, Al-Sarireh B, Lunevicius R, Benhmida R, Singhal R, Balachandra S, Demirli Atıcı S, Jaunoo S, Dwerryhouse S, Boyce T, Charalampakis V, Kanakala V, Abbas Z, Tewari N, Pandanaboyana S; COVIDPAN Collborative Group; COVID Pain Collborative Group. SARS-CoV-2 infection is associated with an increased risk of idiopathic acute pancreatitis but not pancreatic exocrine insufficiency or diabetes: long-term results of the COVIDPAN study. Gut. 2022;71:1444-1447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 5. | Troncone E, Salvatori S, Sena G, De Cristofaro E, Alfieri N, Marafini I, Paganelli C, Argirò R, Giannarelli D, Monteleone G, Del Vecchio Blanco G. Low Frequency of Acute Pancreatitis in Hospitalized COVID-19 Patients. Pancreas. 2021;50:393-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 6. | Akarsu C, Karabulut M, Aydin H, Sahbaz NA, Dural AC, Yegul D, Peker KD, Ferahman S, Bulut S, Dönmez T, Asar S, Yasar KK, Adas GT. Association between Acute Pancreatitis and COVID-19: Could Pancreatitis Be the Missing Piece of the Puzzle about Increased Mortality Rates? J Invest Surg. 2022;35:119-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 7. | Cholankeril G, Podboy A, Aivaliotis VI, Pham EA, Spencer SP, Kim D, Ahmed A. Association of Digestive Symptoms and Hospitalization in Patients With SARS-CoV-2 Infection. Am J Gastroenterol. 2020;115:1129-1132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 8. | Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, Tsiotos GG, Vege SS; Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62:102-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4932] [Cited by in RCA: 4315] [Article Influence: 359.6] [Reference Citation Analysis (45)] |
| 9. | Seppänen H, Puolakkainen P. Classification, Severity Assessment, and Prevention of Recurrences in Acute Pancreatitis. Scand J Surg. 2020;109:53-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 10. | Leppäniemi A, Tolonen M, Tarasconi A, Segovia-Lohse H, Gamberini E, Kirkpatrick AW, Ball CG, Parry N, Sartelli M, Wolbrink D, van Goor H, Baiocchi G, Ansaloni L, Biffl W, Coccolini F, Di Saverio S, Kluger Y, Moore EE, Catena F. Executive summary: WSES Guidelines for the management of severe acute pancreatitis. J Trauma Acute Care Surg. 2020;88:888-890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 11. | Brisinda G, Vanella S, Crocco A, Mazzari A, Tomaiuolo P, Santullo F, Grossi U, Crucitti A. Severe acute pancreatitis: advances and insights in assessment of severity and management. Eur J Gastroenterol Hepatol. 2011;23:541-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 12. | Rawla P, Bandaru SS, Vellipuram AR. Review of Infectious Etiology of Acute Pancreatitis. Gastroenterology Res. 2017;10:153-158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 112] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 13. | van Dijk SM, Hallensleben NDL, van Santvoort HC, Fockens P, van Goor H, Bruno MJ, Besselink MG; Dutch Pancreatitis Study Group. Acute pancreatitis: recent advances through randomised trials. Gut. 2017;66:2024-2032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 296] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 14. | Brisinda G, Crocco A, Giustacchini P. Classification of the severity of acute pancreatitis: how much is really needed for a new classification? Ann Surg. 2015;261:e101-e102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 15. | Du M, Cai G, Chen F, Christiani DC, Zhang Z, Wang M. Multiomics Evaluation of Gastrointestinal and Other Clinical Characteristics of COVID-19. Gastroenterology. 2020;158:2298-2301.e7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 112] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 16. | Correia de Sá T, Soares C, Rocha M. Acute pancreatitis and COVID-19: A literature review. World J Gastrointest Surg. 2021;13:574-584. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 27] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 17. | Rubin R. SARS-CoV-2 RNA Can Persist in Stool Months After Respiratory Tract Clears Virus. JAMA. 2022;327:2175-2176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 18. | Hanley B, Naresh KN, Roufosse C, Nicholson AG, Weir J, Cooke GS, Thursz M, Manousou P, Corbett R, Goldin R, Al-Sarraj S, Abdolrasouli A, Swann OC, Baillon L, Penn R, Barclay WS, Viola P, Osborn M. Histopathological findings and viral tropism in UK patients with severe fatal COVID-19: a post-mortem study. Lancet Microbe. 2020;1:e245-e253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 328] [Cited by in RCA: 400] [Article Influence: 80.0] [Reference Citation Analysis (0)] |
| 19. | Chmielik E, Jazowiecka-Rakus J, Dyduch G, Nasierowska-Guttmejer A, Michalowski L, Sochanik A, Ulatowska-Bialas M. COVID-19 Autopsies: A Case Series from Poland. Pathobiology. 2021;88:78-87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 20. | Gu J, Han B, Wang J. COVID-19: Gastrointestinal Manifestations and Potential Fecal-Oral Transmission. Gastroenterology. 2020;158:1518-1519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 954] [Cited by in RCA: 949] [Article Influence: 189.8] [Reference Citation Analysis (1)] |
| 21. | Dalan R, Bornstein SR, El-Armouche A, Rodionov RN, Markov A, Wielockx B, Beuschlein F, Boehm BO. The ACE-2 in COVID-19: Foe or Friend? Horm Metab Res. 2020;52:257-263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 108] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 22. | Shaharuddin SH, Wang V, Santos RS, Gross A, Wang Y, Jawanda H, Zhang Y, Hasan W, Garcia G Jr, Arumugaswami V, Sareen D. Deleterious Effects of SARS-CoV-2 Infection on Human Pancreatic Cells. Front Cell Infect Microbiol. 2021;11:678482. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 23. | Liu F, Long X, Zhang B, Zhang W, Chen X, Zhang Z. ACE2 Expression in Pancreas May Cause Pancreatic Damage After SARS-CoV-2 Infection. Clin Gastroenterol Hepatol. 2020;18:2128-2130.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 343] [Cited by in RCA: 473] [Article Influence: 94.6] [Reference Citation Analysis (0)] |
| 24. | Zhou Z, Zhao N, Shu Y, Han S, Chen B, Shu X. Effect of Gastrointestinal Symptoms in Patients With COVID-19. Gastroenterology. 2020;158:2294-2297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 167] [Article Influence: 33.4] [Reference Citation Analysis (0)] |
| 25. | Aloysius MM, Thatti A, Gupta A, Sharma N, Bansal P, Goyal H. COVID-19 presenting as acute pancreatitis. Pancreatology. 2020;20:1026-1027. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 141] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 26. | Annunziata A, Coppola A, Andreozzi P, Lanza M, Simioli F, Carannante N, Di Somma C, Di Micco P, Fiorentino G. Acute Pancreatitis and COVID-19: A Single-Center Experience. J Multidiscip Healthc. 2021;14:2857-2861. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Aktas B, Aslim B. Gut-lung axis and dysbiosis in COVID-19. Turk J Biol. 2020;44:265-272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 28. | Dang AT, Marsland BJ. Microbes, metabolites, and the gut-lung axis. Mucosal Immunol. 2019;12:843-850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 356] [Cited by in RCA: 632] [Article Influence: 105.3] [Reference Citation Analysis (0)] |
| 29. | Marsland BJ, Trompette A, Gollwitzer ES. The Gut-Lung Axis in Respiratory Disease. Ann Am Thorac Soc. 2015;12 Suppl 2:S150-S156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 373] [Article Influence: 41.4] [Reference Citation Analysis (1)] |
| 30. | Albillos A, de Gottardi A, Rescigno M. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J Hepatol. 2020;72:558-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 542] [Cited by in RCA: 1249] [Article Influence: 249.8] [Reference Citation Analysis (1)] |
| 31. | Stephens JR, Wong JLC, Broomhead R, Stümpfle R, Waheed U, Patel P, Brett SJ, Soni S. Raised serum amylase in patients with COVID-19 may not be associated with pancreatitis. Br J Surg. 2021;108:e152-e153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 32. | Abramczyk U, Nowaczyński M, Słomczyński A, Wojnicz P, Zatyka P, Kuzan A. Consequences of COVID-19 for the Pancreas. Int J Mol Sci. 2022;23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 33. | de-Madaria E, Capurso G. COVID-19 and acute pancreatitis: examining the causality. Nat Rev Gastroenterol Hepatol. 2021;18:3-4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 102] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 34. | Suchman K, Raphael KL, Liu Y, Wee D, Trindade AJ; Northwell COVID-19 Research Consortium. Acute pancreatitis in children hospitalized with COVID-19. Pancreatology. 2021;21:31-33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 35. | Mao R, Qiu Y, He JS, Tan JY, Li XH, Liang J, Shen J, Zhu LR, Chen Y, Iacucci M, Ng SC, Ghosh S, Chen MH. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2020;5:667-678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 741] [Cited by in RCA: 751] [Article Influence: 150.2] [Reference Citation Analysis (0)] |
| 36. | Miró Ò, Llorens P, Jiménez S, Piñera P, Burillo-Putze G, Martín A, Martín-Sánchez FJ, González Del Castillo J; Spanish Investigators in Emergency Situations TeAm (SIESTA) network. Frequency of five unusual presentations in patients with COVID-19: results of the UMC-19-S1. Epidemiol Infect. 2020;148:e189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 37. | Inamdar S, Benias PC, Liu Y, Sejpal DV, Satapathy SK, Trindade AJ; Northwell COVID-19 Research Consortium. Prevalence, Risk Factors, and Outcomes of Hospitalized Patients With Coronavirus Disease 2019 Presenting as Acute Pancreatitis. Gastroenterology. 2020;159:2226-2228.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 103] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 38. | Pons S, Fodil S, Azoulay E, Zafrani L. The vascular endothelium: the cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Crit Care. 2020;24:353. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 360] [Cited by in RCA: 347] [Article Influence: 69.4] [Reference Citation Analysis (0)] |
| 39. | Pandanaboyana S, Moir J, Leeds JS, Oppong K, Kanwar A, Marzouk A, Belgaumkar A, Gupta A, Siriwardena AK, Haque AR, Awan A, Balakrishnan A, Rawashdeh A, Ivanov B, Parmar C, M Halloran C, Caruana C, Borg CM, Gomez D, Damaskos D, Karavias D, Finch G, Ebied H, K Pine J, R A Skipworth J, Milburn J, Latif J, Ratnam Apollos J, El Kafsi J, Windsor JA, Roberts K, Wang K, Ravi K, V Coats M, Hollyman M, Phillips M, Okocha M, Sj Wilson M, A Ameer N, Kumar N, Shah N, Lapolla P, Magee C, Al-Sarireh B, Lunevicius R, Benhmida R, Singhal R, Balachandra S, Demirli Atıcı S, Jaunoo S, Dwerryhouse S, Boyce T, Charalampakis V, Kanakala V, Abbas Z, Nayar M; COVID PAN collaborative group. SARS-CoV-2 infection in acute pancreatitis increases disease severity and 30-day mortality: COVID PAN collaborative study. Gut. 2021;70:1061-1069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 63] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 40. | Chiarello MM, Cariati M, Brisinda G. Assessment of severity of acute pancreatitis in a Sars-CoV-2 pandemia. Br J Surg. 2020;107:e379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 41. | Wang K, Luo J, Tan F, Liu J, Ni Z, Liu D, Tian P, Li W. Acute Pancreatitis as the Initial Manifestation in 2 Cases of COVID-19 in Wuhan, China. Open Forum Infect Dis. 2020;7:ofaa324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 42. | Shlomovitz E, Davies W, Cairns E, Brintnell WC, Goldszmidt M, Dresser GK. Severe necrotizing pancreatitis following combined hepatitis A and B vaccination. CMAJ. 2007;176:339-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 43. | Adler JB, Mazzotta SA, Barkin JS. Pancreatitis caused by measles, mumps, and rubella vaccine. Pancreas. 1991;6:489-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 44. | Haviv YS, Sharkia M, Galun E, Safadi R. Pancreatitis following hepatitis A vaccination. Eur J Med Res. 2000;5:229-230. [PubMed] |
| 45. | Sahin U, Muik A, Derhovanessian E, Vogler I, Kranz LM, Vormehr M, Baum A, Pascal K, Quandt J, Maurus D, Brachtendorf S, Lörks V, Sikorski J, Hilker R, Becker D, Eller AK, Grützner J, Boesler C, Rosenbaum C, Kühnle MC, Luxemburger U, Kemmer-Brück A, Langer D, Bexon M, Bolte S, Karikó K, Palanche T, Fischer B, Schultz A, Shi PY, Fontes-Garfias C, Perez JL, Swanson KA, Loschko J, Scully IL, Cutler M, Kalina W, Kyratsous CA, Cooper D, Dormitzer PR, Jansen KU, Türeci Ö. COVID-19 vaccine BNT162b1 elicits human antibody and TH1 T cell responses. Nature. 2020;586:594-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1039] [Cited by in RCA: 1403] [Article Influence: 280.6] [Reference Citation Analysis (0)] |
| 46. | Ozaka S, Kodera T, Ariki S, Kobayashi T, Murakami K. Acute pancreatitis soon after COVID-19 vaccination: A case report. Medicine (Baltimore). 2022;101:e28471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 47. | Walter T, Connor S, Stedman C, Doogue M. A case of acute necrotising pancreatitis following the second dose of Pfizer-BioNTech COVID-19 mRNA vaccine. Br J Clin Pharmacol. 2022;88:1385-1386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 48. | Meo SA, Bukhari IA, Akram J, Meo AS, Klonoff DC. COVID-19 vaccines: comparison of biological, pharmacological characteristics and adverse effects of Pfizer/BioNTech and Moderna Vaccines. Eur Rev Med Pharmacol Sci. 2021;25:1663-1669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 176] [Reference Citation Analysis (0)] |
| 49. | Rosenblum HG, Hadler SC, Moulia D, Shimabukuro TT, Su JR, Tepper NK, Ess KC, Woo EJ, Mba-Jonas A, Alimchandani M, Nair N, Klein NP, Hanson KE, Markowitz LE, Wharton M, McNally VV, Romero JR, Talbot HK, Lee GM, Daley MF, Mbaeyi SA, Oliver SE. Use of COVID-19 Vaccines After Reports of Adverse Events Among Adult Recipients of Janssen (Johnson & Johnson) and mRNA COVID-19 Vaccines (Pfizer-BioNTech and Moderna): Update from the Advisory Committee on Immunization Practices - United States, July 2021. MMWR Morb Mortal Wkly Rep. 2021;70:1094-1099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 150] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 50. | Parkash O, Sharko A, Farooqi A, Ying GW, Sura P. Acute Pancreatitis: A Possible Side Effect of COVID-19 Vaccine. Cureus. 2021;13:e14741. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 51. | Pfizer. Pfizer-BioNTech COVID-19 vaccine (BNT162, PF-07302048). Vaccines and related biological products advisory committee. [cited 13 April 2022]. Available from: https://www.fda.gov/media/144246/download. |
| 52. | COVID-19 mRNA Pfizer-BioNTech vaccine analysis print. All UK spontaneous reports received between 9 December 2020 and 30 June 2021 for mRNA Pfizer/BioNTech vaccine analysis print. 2021. [cited 13 April 2022]. Available from: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1009453/COVID-19_mRNA_Pfizer-BioNTech_Vaccine_Analysis_Print_DPL_28.07.2021. |
| 53. | ANSM. Point de situation sur la surveillance des vaccins contre la COVID-19. [cited 13 April 2022]. Available from: https://ansm.sante.fr/actualites/point-de-situation-sur-la-surveillance-des-vaccins-contre-la-covid-19-7. |
| 54. | World Health Organization. VigiAccess. [cited 13 April 2022]. Available from: http://www.vigiaccess.org. |
| 55. | FEDAIISF. Rapporto annuale sulla sicurezza dei vaccini anti-COVID-19. [cited 13 April 2022]. Available from: https://www.fedaiisf.it/aifa-rapporto-annuale-sulla-sicurezza-dei-vaccini-anti-covid-19/. |
| 56. | Boxhoorn L, Voermans RP, Bouwense SA, Bruno MJ, Verdonk RC, Boermeester MA, van Santvoort HC, Besselink MG. Acute pancreatitis. Lancet. 2020;396:726-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 276] [Cited by in RCA: 576] [Article Influence: 115.2] [Reference Citation Analysis (0)] |
| 57. | Bulthuis MC, Boxhoorn L, Beudel M, Elbers PWG, Kop MPM, van Wanrooij RLJ, Besselink MG, Voermans RP. Acute pancreatitis in COVID-19 patients: true risk? Scand J Gastroenterol. 2021;56:585-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 58. | Ding P, Song B, Liu X, Fang X, Cai H, Zhang D, Zheng X. Elevated Pancreatic Enzymes in ICU Patients With COVID-19 in Wuhan, China: A Retrospective Study. Front Med (Lausanne). 2021;8:663646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 59. | Huang HL, Nie X, Cai B, Tang JT, He Y, Miao Q, Song HL, Luo TX, Gao BX, Wang LL, Li GX. Procalcitonin levels predict acute kidney injury and prognosis in acute pancreatitis: a prospective study. PLoS One. 2013;8:e82250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 60. | Sarr MG. Early fluid "resuscitation/therapy" in acute pancreatitis: which fluid? Ann Surg. 2013;257:189-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 61. | Bortolotti P, Saulnier F, Colling D, Redheuil A, Preau S. New tools for optimizing fluid resuscitation in acute pancreatitis. World J Gastroenterol. 2014;20:16113-16122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 62. | Arvanitakis M, Ockenga J, Bezmarevic M, Gianotti L, Krznarić Ž, Lobo DN, Löser C, Madl C, Meier R, Phillips M, Rasmussen HH, Van Hooft JE, Bischoff SC. ESPEN guideline on clinical nutrition in acute and chronic pancreatitis. Clin Nutr. 2020;39:612-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 140] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 63. | Haines K, Parker V, Ohnuma T, Krishnamoorthy V, Raghunathan K, Sulo S, Kerr KW, Besecker BY, Cassady BA, Wischmeyer PE. Role of Early Enteral Nutrition in Mechanically Ventilated COVID-19 Patients. Crit Care Explor. 2022;4:e0683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 64. | Compher C, Bingham AL, McCall M, Patel J, Rice TW, Braunschweig C, McKeever L. Guidelines for the provision of nutrition support therapy in the adult critically ill patient: The American Society for Parenteral and Enteral Nutrition. JPEN J Parenter Enteral Nutr. 2022;46:12-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 312] [Article Influence: 78.0] [Reference Citation Analysis (0)] |
| 65. | Besselink MG, van Santvoort HC, Nieuwenhuijs VB, Boermeester MA, Bollen TL, Buskens E, Dejong CH, van Eijck CH, van Goor H, Hofker SS, Lameris JS, van Leeuwen MS, Ploeg RJ, van Ramshorst B, Schaapherder AF, Cuesta MA, Consten EC, Gouma DJ, van der Harst E, Hesselink EJ, Houdijk LP, Karsten TM, van Laarhoven CJ, Pierie JP, Rosman C, Bilgen EJ, Timmer R, van der Tweel I, de Wit RJ, Witteman BJ, Gooszen HG; Dutch Acute Pancreatitis Study Group. Minimally invasive 'step-up approach' vs maximal necrosectomy in patients with acute necrotising pancreatitis (PANTER trial): design and rationale of a randomised controlled multicenter trial [ISRCTN13975868]. BMC Surg. 2006;6:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 126] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 66. | Hollemans RA, Bakker OJ, Boermeester MA, Bollen TL, Bosscha K, Bruno MJ, Buskens E, Dejong CH, van Duijvendijk P, van Eijck CH, Fockens P, van Goor H, van Grevenstein WM, van der Harst E, Heisterkamp J, Hesselink EJ, Hofker S, Houdijk AP, Karsten T, Kruyt PM, van Laarhoven CJ, Laméris JS, van Leeuwen MS, Manusama ER, Molenaar IQ, Nieuwenhuijs VB, van Ramshorst B, Roos D, Rosman C, Schaapherder AF, van der Schelling GP, Timmer R, Verdonk RC, de Wit RJ, Gooszen HG, Besselink MG, van Santvoort HC; Dutch Pancreatitis Study Group. Superiority of Step-up Approach vs Open Necrosectomy in Long-term Follow-up of Patients With Necrotizing Pancreatitis. Gastroenterology. 2019;156:1016-1026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 150] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 67. | Boxhoorn L, van Dijk SM, van Grinsven J, Verdonk RC, Boermeester MA, Bollen TL, Bouwense SAW, Bruno MJ, Cappendijk VC, Dejong CHC, van Duijvendijk P, van Eijck CHJ, Fockens P, Francken MFG, van Goor H, Hadithi M, Hallensleben NDL, Haveman JW, Jacobs MAJM, Jansen JM, Kop MPM, van Lienden KP, Manusama ER, Mieog JSD, Molenaar IQ, Nieuwenhuijs VB, Poen AC, Poley JW, van de Poll M, Quispel R, Römkens TEH, Schwartz MP, Seerden TC, Stommel MWJ, Straathof JWA, Timmerhuis HC, Venneman NG, Voermans RP, van de Vrie W, Witteman BJ, Dijkgraaf MGW, van Santvoort HC, Besselink MG; Dutch Pancreatitis Study Group. Immediate versus Postponed Intervention for Infected Necrotizing Pancreatitis. N Engl J Med. 2021;385:1372-1381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 140] [Article Influence: 35.0] [Reference Citation Analysis (0)] |