Published online May 21, 2021. doi: 10.3748/wjg.v27.i19.2270
Peer-review started: January 24, 2021
First decision: February 22, 2021
Revised: February 22, 2021
Accepted: April 13, 2021
Article in press: April 13, 2021
Published online: May 21, 2021
Processing time: 108 Days and 13.3 Hours
Compelling evidence supports the crucial role of the receptor for advanced glycation end-products (RAGE) axis activation in many clinical entities. Since the beginning of the coronavirus disease 2019 pandemic, there is an increasing concern about the risk and handling of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in inflammatory gastrointestinal disorders, such as inflammatory bowel diseases (IBD). However, clinical data raised during pandemic suggests that IBD patients do not have an increased risk of contracting SARS-CoV-2 infection or develop a more severe course of infection. In the present review, we intend to highlight how two potentially important contributors to the inflammatory response to SARS-CoV-2 infection in IBD patients, the RAGE axis activation as well as the cross-talk with the renin-angiotensin system, are dampened by the high expression of soluble forms of both RAGE and the angiotensin-converting enzyme (ACE) 2. The soluble form of RAGE functions as a decoy for its ligands, and soluble ACE2 seems to be an additionally attenuating contributor to RAGE axis activation, particularly by avoiding the transactivation of the RAGE axis that can be produced by the virus-mediated imbalance of the ACE/angiotensin II/angiotensin II receptor type 1 pathway.
Core Tip: Data raised during the pandemic suggest that inflammatory bowel diseases do not have an increased risk of contracting severe acute respiratory syndrome coronavirus 2 infection or develop a more severe course of infection. These findings are in some way unexpected considering that inflammatory bowel disease is a chronic inflammatory state of the gastrointestinal tract. We herein discuss how the receptor for advanced glycation end-products axis activation as well as the cross-talk with the renin-angiotensin system are dampened by the high expression of soluble forms of both receptor for advanced glycation end-products and angiotensin-converting enzyme 2.
- Citation: Rojas A, Schneider I, Lindner C, Gonzàlez I, Morales MA. Receptor for advanced glycation end-products axis and coronavirus disease 2019 in inflammatory bowel diseases: A dangerous liaison? World J Gastroenterol 2021; 27(19): 2270-2280
- URL: https://www.wjgnet.com/1007-9327/full/v27/i19/2270.htm
- DOI: https://dx.doi.org/10.3748/wjg.v27.i19.2270
At the end of 2019, China reported several cases of severe pneumonia of unknown cause; the coronavirus severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was subsequently identified as the etiological agent[1]. Due to its rapid spread all over the world, the World Health Organization defined coronavirus disease 2019 (COVID-19) as a pandemic on January 30, 2020.
The main symptoms of COVID-19 affect the lower respiratory tract, causing high mortality-rate complications such as acute distress respiratory syndrome[2-6]. However, recent reports reveal that gastrointestinal (GI) manifestations of SARS-CoV-2 infection are common clinical symptoms among patients who develop COVID-19[7-11].
The SARS-CoV-2 uses the cellular transmembrane angiotensin-converting enzyme 2 (ACE2) molecule as the receptor for viral cell entry. Under physiological conditions, epithelial ACE2 is widely expressed in several tissues. However, the expression of epithelial ACE2 in the terminal ileum and colon are amongst the highest in the body, which could explain why COVID-19 patients experience several GI symptoms[12-16].
Consequently, there is an increasing concern about the risk and handling of SARS-CoV-2 infection in inflammatory GI disorders, such as inflammatory bowel disease (IBD). The IBDs are chronic intestinal diseases that comprise Crohn´s disease (CD) and ulcerative colitis, which are characterized by chronic and relapsing intestinal inflammation[17,18]. Thus, since the beginning of the SARS-CoV-2 pandemic, IBD patients were considered a high-risk group for increased severity and adverse outcomes in SARS-CoV-2 infection[19,20].
However, clinical data raised during pandemic suggest that IBD patients do not have an increased risk of contracting SARS-CoV-2 infection or develop a more severe course of infection[21-25]. A compelling body of both clinical and experimental evidence has shed light on the crucial role of the receptor of advanced glycation end-products (RAGE) activation in many chronic inflammatory diseases[26-31]. More recently, the role of RAGE axis activation as a key contributor in the clinical course of SARS-CoV-2 infection has been documented[32].
In the present review, we intend to highlight the role of the RAGE axis activation in the context of SARS-CoV-2 infection and the clinical evolution of the IBD patient.
Firstly described in 1992, the RAGE is a type I single-pass transmembrane protein that can bind advanced glycation-end products (AGEs). This molecule belongs to the immunoglobulin superfamily of cell surface receptors, which is now considered as a pattern recognition receptor and is regarded as a central mediator in chronic inflammatory and immune responses[33-35].
RAGE is usually expressed at low levels in many cell types and tissues, except for the lungs. However, this expression is noticeably increased under inflammatory conditions[36-38].
Besides the transmembrane form of RAGE, several soluble isoforms of this receptor (sRAGE) are generated either by alternative splicing or by the action of membrane associated-proteases, such matrix metalloproteinase-9 (MMP-9), a disintegrin metalloproteases (ADAM)-10, and ADAM-17[39-42]. These soluble variants may function as a decoy receptor for ligands and thus prevent the interaction with the membrane-anchored full-length RAGE. In consequence, a high bioavailability of sRAGE will decreases the inflammatory responses driven by full-length RAGE activation [35,43,44]. Besides AGEs, RAGE can recognize many other ligands including the alarmin high-mobility group box 1 (HMGB1), members of the S100 protein family, glycosaminoglycans, and amyloid β peptides, among many others[35,45].
As a consequence of RAGE engagement by its ligands, multiple signaling pathways are triggered, including reactive oxygen species, p21ras, extracellular signal-regulated protein kinase 1/2 (p44/p42) mitogen-activated protein (MAP) kinases, p38 and stress-activated protein kinases/c-Jun N-terminal kinase mitogen-activated protein kinases, rhoGTPases, phosphoinositol-3 kinase, and the janus kinase/signal transducer and activator of transcription pathway, having crucial downstream inflammatory consequences such as activation of nuclear factor-kappaB (NF-κB), AP-1, and signal transducer and activator of transcription-3[35].
Indeed, the RAGE axis signaling not only triggers pro-inflammatory gene expression but also a positive feed-forward loop, in which the inflammatory stimuli activate NF-κB, which induces RAGE expression, following an enhanced and sustained inflammatory response[35,46-48].
Initially, RAGE axis activation was linked to the complications of diabetes such as macro-and microvascular complications[49,50]. However, a growing body of evidence indicates RAGE as a key molecule involved in many chronic inflammatory diseases[28-30,51].
Many underlying molecular mechanisms are involved in the onset and perpetuation of the disease, particularly those fueling the robust pro-inflammatory signals found in IBD patients[26,52]. Noteworthy, some pieces of evidence reveal an increased expression of RAGE and its ligands on intestinal cells in IBD patients, especially in inflamed areas[53-55]. In this context, it is important to highlight that the release of the RAGE ligand HMGB1 and members of the S100 protein family is increased under inflammation conditions[54-57]. Thus, the engagement of RAGE may play an important role in the maintenance of intestinal injury and inflammatory environment [53-57].
Strikingly, increased levels of both MMP-9 and ADAM17 have been reported in IBD patients[58,59], and both metalloproteases are involved in RAGE shedding, thus increasing the levels of sRAGE, which in turn can modulate the inflammatory responses driven by RAGE axis activation in IBD patients[58]. At present, a compelling body of evidence supports the fact that increased sRAGE levels correlate with a decrease in the RAGE activation-mediated inflammatory responses in many clinical entities[60-63]. In this context, it is important to highlight that CD147 significantly contributes to epithelial inflammation in many clinical entities including IBD[64,65], and it has been recently shown to act as a receptor for SARS-CoV-2[66]. Noteworthy, the inhibition of RAGE activation-mediated inflammatory response leads to a reduced expression of CD147[67].
The renin-angiotensin system (RAS) is a hormonal system regulated by two complementary pathways that mediate opposing effects on inflammation, fibrosis, and cell proliferation[68-70]. Thus, the balance of both pathways determines pro-inflammatory or anti-inflammatory conditions among several systems such as cardiovascular, renal, and respiratory systems[71-74].
The classical pathway mediated via ACE, angiotensin II (Ang II) and its receptor Ang II receptor type 1 (AT1R), triggers activation of pro-inflammatory signals such as oxidative and nitrosative stresses, the induction of cytokines and cell adhesion molecules, as well as the activation of transcription factors such NF-κB[75-78]. On the contrary, the alternative pathway predominantly mediated by ACE2, Ang (1-7) and its receptor Mas (MasR), induces the opposite effects of AT1R activation, being an anti-inflammatory and anti-fibrotic counter regulator of the effects of ACE/Ang II/AT1R[71,75,79,80]. ACE and ACE2 are highly expressed in several tissues such as the lungs, kidneys, and blood vessels. However, the brush border of the ileum and the colon are among the tissues with the highest expression of both enzymes[13-16,81]. Both enzymes can cleave angiotensin, generating different sub-products and regulating the balance between both pathways of the RAS system[79,82,83].
Recent studies suggest high expression of the major components of both RAS pathways across the ileum and colon[81]. In this sense, the gut could be an especially susceptible organ for the imbalance of RAS pathways. Thus, the dysregulation of these components could have potential implications for inflammation and fibrosis for IBD patients[84,85]. Strikingly, several studies have revealed that the intestinal expression of ACE2 is inversely correlated with fibrosis in IBD patients[81,86].
Additionally, Ang (1-7) ameliorates colonic myofibroblast collagen secretion via MasR[81]. Furthermore, angiotensin receptor blockers and ACE inhibitors are reported to decrease mucosal pro-inflammatory cytokines, ameliorate colitis, and were associated with lower rates of complications, surgery, and hospitalization in patients with IBD[87-89].
Normally, ACE2 breaks down Ang II to Ang 1–7 peptide and thus avoiding the activation of the pro-inflammatory pathways of RAS. However, SARS-CoV-2 can hijack ACE2 and use it to gain entry into host cells[12,90]. Noteworthy, high bioavailability of soluble ACE2 has been reported in IBD patients[81,84], mainly ascribed to the increased level of ADAM17 observed in these patients[58,91-93], which in turn may function as a decoy receptor for SARS-CoV-2 and thus avoiding the hijacking of the counterbalancing enzyme.
This is particularly important considering that a novel ligand-independent mechanism for RAGE transactivation has been recently reported to occur following activation of the AT1R by Ang-II, thus leading to NF-κB dependent expression of pro-inflammatory mediators[48].
Contrary to what is expected, considering the pathophysiology of IBD, there is currently no evidence for an increased risk of worse clinical outcomes in patients with IBD in the context of COVID-19[21-25]. The role of the RAGE axis in the patho
However, a counterbalancing element must be added to this scenario: The soluble RAGE. This molecule is generated by alternative splicing or by cleavage of the ectodomain of the membrane-anchored RAGE by the action of both MMP-9 and ADAM17, which are highly expressed in IBD patients[58,59]. Therefore, the high bioavailability of soluble RAGE may dampen RAGE activation, despite the abundance of both receptor and ligands in the inflamed intestinal mucosa of IBD patients.
On the other hand, the high expression of ACE2 in GI tract, especially among IBD patients, makes this tissue a particularly trophic niche for infection with SARS-CoV-2. Furthermore, the ACE2 exhaustion mediated by the entry of SARS-CoV-2 may then induce a robust RAS imbalance in favor of the pro-inflammatory ACE/Ang II/AT1R pathway[95]. These observations suggest that the inflamed gut of IBD patients represents an optimal doorway for SARS-CoV-2 entry, driving poor clinical outcomes in IBD patients who develop COVID-19.
However, this hypothetical scenario also has an important counterbalancing actor, the soluble form of ACE2, which is also increased in patients with IBD due to the shedding of the membrane-anchored ACE2 by ADAM17[58-59]. This is particularly important considering the non-cognate transactivation mechanism described for RAGE because of AT1R activation by Ang II[48], which is dampened by the preservation of membrane-associated ACE2 exhaustion by its soluble form.
A growing body of evidence demonstrates that in IBD patients the use of systemic immunosuppression is not associated with an increased risk of COVID-19 patients with IBD[96-100]. Furthermore, we must also keep in mind that the main objective of pharmacological treatments in IBD is to reduce inflammation levels. In this sense, in addition to interfering with signaling pathways, many drugs used in the current treatments also decrease the expression of RAGE and the bioavailability of some RAGE ligands, particularly the alarmins HMGB1 and S100 protein family members[96,97]. Indeed, several authors remark the possible protective role of IBD therapy against SARS-CoV-2 infection, especially through interfering with cytokine activity observed in the clinical course of COVID-19[98-100].
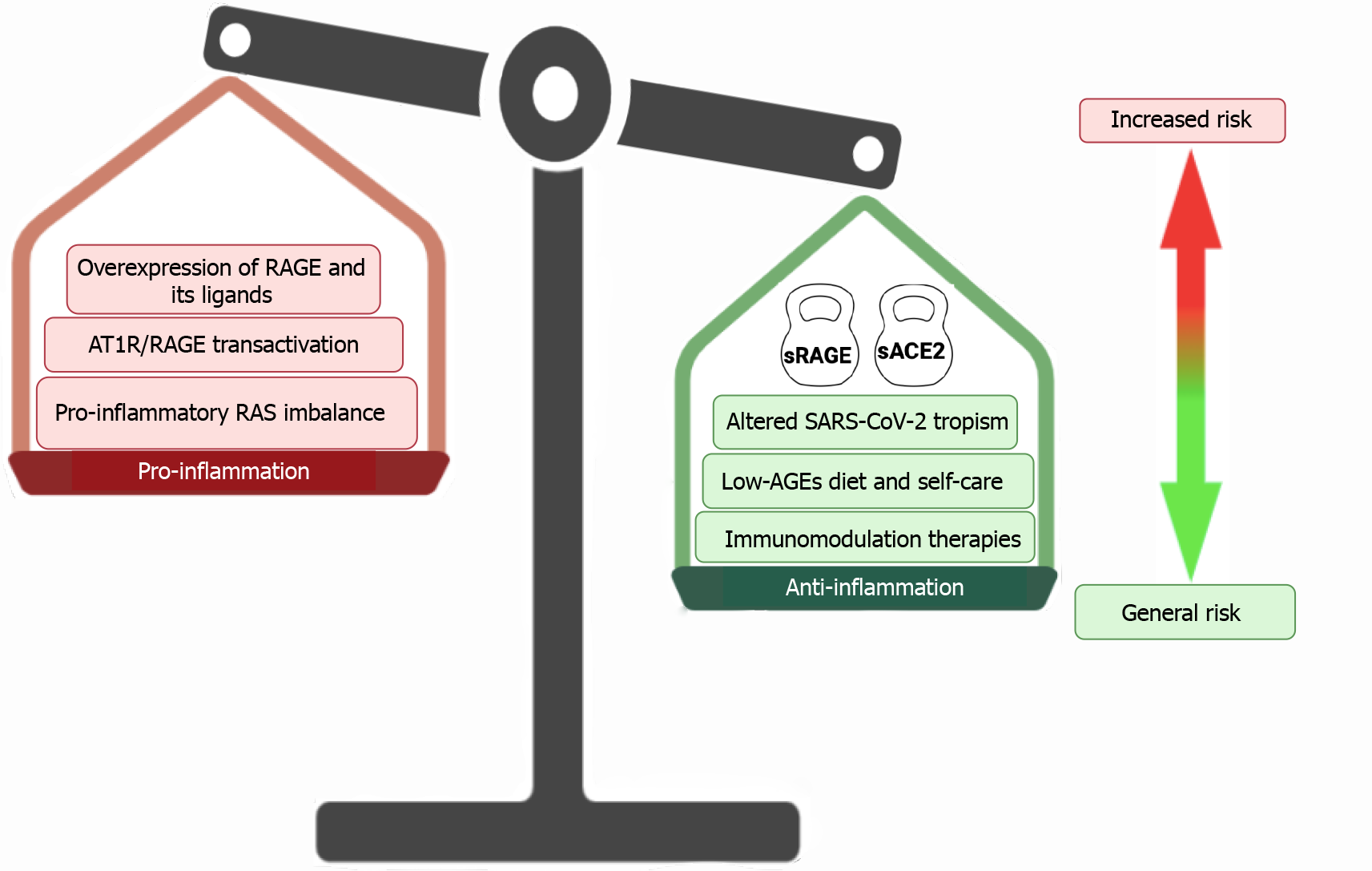
Additionally, the IBD patients have a high self-care standard and follow diets that help them to maintain good nutritional levels and the disease under control[101]. Some of these nutritional regimens are associated with a low-AGE diet, which may contribute to reducing the proinflammatory intestinal milieu mediated by RAGE activation[102] (Figure 1).
The COVID-19 pandemics represent the worst challenge for a century for health systems all over the world. Severity and mortality have been highest in people with underlying morbidities. Therefore, special efforts have been done to understand how SARS-CoV-2 may particularly fuel inflammation in many clinical entities where the chronicity of an inflammatory environment is a relevant part of the pathogenesis of diseases. Based on a particularly inflamed landscape depicted in IBD patients, the activation of the RAGE axis as well the RAS imbalance seem to be crucial contributors to worsen inflammation in the gut. However, data raised during the pandemic suggests that IBD patients have neither an increased risk of contracting SARS-CoV-2 infection nor developing a more severe course of infection.
RAGE axis activation seems to be dampened by the high bioavailability of soluble receptors functioning as a decoy for its ligands. Additionally, soluble ACE2 seems to be another attenuating contributor to RAGE axis activation, particularly by avoiding the transactivation of RAGE axis that can be produced by the virus-mediated imbalance of the ACE/Ang II/ AT1R pathway. Thus, RAGE axis activation in COVID-19 IBD patients does not seem to be a dangerous liaison.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Chile
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Maric I S-Editor: Fan JR L-Editor: Filipodia P-Editor: Ma YJ
| 1. | Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W; China Novel Coronavirus Investigating and Research Team. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020;382:727-733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18987] [Cited by in RCA: 17615] [Article Influence: 3523.0] [Reference Citation Analysis (0)] |
| 2. | Goyal P, Choi JJ, Pinheiro LC, Schenck EJ, Chen R, Jabri A, Satlin MJ, Campion TR Jr, Nahid M, Ringel JB, Hoffman KL, Alshak MN, Li HA, Wehmeyer GT, Rajan M, Reshetnyak E, Hupert N, Horn EM, Martinez FJ, Gulick RM, Safford MM. Clinical Characteristics of Covid-19 in New York City. N Engl J Med. 2020;382:2372-2374. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1644] [Cited by in RCA: 1615] [Article Influence: 323.0] [Reference Citation Analysis (0)] |
| 3. | Hui KPY, Cheung MC, Perera RAPM, Ng KC, Bui CHT, Ho JCW, Ng MMT, Kuok DIT, Shih KC, Tsao SW, Poon LLM, Peiris M, Nicholls JM, Chan MCW. Tropism, replication competence, and innate immune responses of the coronavirus SARS-CoV-2 in human respiratory tract and conjunctiva: an analysis in ex-vivo and in-vitro cultures. Lancet Respir Med. 2020;8:687-695. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 393] [Cited by in RCA: 388] [Article Influence: 77.6] [Reference Citation Analysis (0)] |
| 4. | Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS; China Medical Treatment Expert Group for Covid-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382:1708-1720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19202] [Cited by in RCA: 18852] [Article Influence: 3770.4] [Reference Citation Analysis (7)] |
| 5. | Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35178] [Cited by in RCA: 30077] [Article Influence: 6015.4] [Reference Citation Analysis (3)] |
| 6. | Lai CC, Shih TP, Ko WC, Tang HJ, Hsueh PR. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int J Antimicrob Agents. 2020;55:105924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3736] [Cited by in RCA: 3194] [Article Influence: 638.8] [Reference Citation Analysis (0)] |
| 7. | Zhou Z, Zhao N, Shu Y, Han S, Chen B, Shu X. Effect of Gastrointestinal Symptoms in Patients With COVID-19. Gastroenterology. 2020;158:2294-2297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 166] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 8. | Jin X, Lian JS, Hu JH, Gao J, Zheng L, Zhang YM, Hao SR, Jia HY, Cai H, Zhang XL, Yu GD, Xu KJ, Wang XY, Gu JQ, Zhang SY, Ye CY, Jin CL, Lu YF, Yu X, Yu XP, Huang JR, Xu KL, Ni Q, Yu CB, Zhu B, Li YT, Liu J, Zhao H, Zhang X, Yu L, Guo YZ, Su JW, Tao JJ, Lang GJ, Wu XX, Wu WR, Qv TT, Xiang DR, Yi P, Shi D, Chen Y, Ren Y, Qiu YQ, Li LJ, Sheng J, Yang Y. Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019 (COVID-19) with gastrointestinal symptoms. Gut. 2020;69:1002-1009. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 944] [Cited by in RCA: 868] [Article Influence: 173.6] [Reference Citation Analysis (0)] |
| 9. | Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 2020; 158: 1831-1833. e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1985] [Cited by in RCA: 1993] [Article Influence: 398.6] [Reference Citation Analysis (1)] |
| 10. | Garg M, Christensen B, Lubel JS. Gastrointestinal ACE2, COVID-19 and IBD: Opportunity in the Face of Tragedy? Gastroenterology. 2020;159:1623-1624.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 11. | Grassia R, Testa S, Pan A, Conti CB. SARS-CoV-2 and gastrointestinal tract: The dark side of the pandemic. Dig Liver Dis. 2020;52:700-701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 12. | Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020; 181: 271-280. e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11946] [Cited by in RCA: 14238] [Article Influence: 2847.6] [Reference Citation Analysis (0)] |
| 13. | GTEx Consortium. . The Genotype-Tissue Expression (GTEx) project. Nat Genet. 2013;45:580-585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6464] [Cited by in RCA: 6200] [Article Influence: 516.7] [Reference Citation Analysis (0)] |
| 14. | Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631-637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3643] [Cited by in RCA: 4146] [Article Influence: 197.4] [Reference Citation Analysis (0)] |
| 15. | Harmer D, Gilbert M, Borman R, Clark KL. Quantitative mRNA expression profiling of ACE 2, a novel homologue of angiotensin converting enzyme. FEBS Lett. 2002;532:107-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 585] [Cited by in RCA: 654] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 16. | Sibony M, Gasc JM, Soubrier F, Alhenc-Gelas F, Corvol P. Gene expression and tissue localization of the two isoforms of angiotensin I converting enzyme. Hypertension. 1993;21:827-835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 119] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 17. | Graham DB, Xavier RJ. Pathway paradigms revealed from the genetics of inflammatory bowel disease. Nature. 2020;578:527-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 459] [Cited by in RCA: 463] [Article Influence: 92.6] [Reference Citation Analysis (0)] |
| 18. | Zhang YZ, Li YY. Inflammatory bowel disease: pathogenesis. World J Gastroenterol. 2014;20:91-99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 751] [Cited by in RCA: 1031] [Article Influence: 93.7] [Reference Citation Analysis (18)] |
| 19. | An P, Ji M, Ren H, Su J, Ding NS, Kang J, Yin A, Zhou Q, Shen L, Zhao L, Jiang X, Xiao Y, Tan W, Lv X, Li J, Liu S, Zhou J, Chen H, Xu Y, Liu J, Chen M, Cao J, Zhou Z, Tan S, Yu H, Dong W, Ding Y. Prevention of COVID-19 in patients with inflammatory bowel disease in Wuhan, China. Lancet Gastroenterol Hepatol. 2020;5:525-527. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 77] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 20. | Kennedy NA, Jones GR, Lamb CA, Appleby R, Arnott I, Beattie RM, Bloom S, Brooks AJ, Cooney R, Dart RJ, Edwards C, Fraser A, Gaya DR, Ghosh S, Greveson K, Hansen R, Hart A, Hawthorne AB, Hayee B, Limdi JK, Murray CD, Parkes GC, Parkes M, Patel K, Pollok RC, Powell N, Probert CS, Raine T, Sebastian S, Selinger C, Smith PJ, Stansfield C, Younge L, Lindsay JO, Irving PM, Lees CW. British Society of Gastroenterology guidance for management of inflammatory bowel disease during the COVID-19 pandemic. Gut. 2020;69:984-990. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 209] [Article Influence: 41.8] [Reference Citation Analysis (0)] |
| 21. | Macaluso FS, Orlando A. COVID-19 in patients with inflammatory bowel disease: A systematic review of clinical data. Dig Liver Dis. 2020;52:1222-1227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 22. | Monteleone G, Ardizzone S. Are Patients with Inflammatory Bowel Disease at Increased Risk for Covid-19 Infection? J Crohns Colitis. 2020;14:1334-1336. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 148] [Article Influence: 29.6] [Reference Citation Analysis (0)] |
| 23. | Aziz M, Fatima R, Haghbin H, Lee-Smith W, Nawras A. The Incidence and Outcomes of COVID-19 in IBD Patients: A Rapid Review and Meta-analysis. Inflamm Bowel Dis. 2020;26:e132-e133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 24. | Allocca M, Fiorino G, Zallot C, Furfaro F, Gilardi D, Radice S, Danese S, Peyrin-Biroulet L. Incidence and Patterns of COVID-19 Among Inflammatory Bowel Disease Patients From the Nancy and Milan Cohorts. Clin Gastroenterol Hepatol. 2020;18:2134-2135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 102] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 25. | Neurath MF. COVID-19 and immunomodulation in IBD. Gut. 2020;69:1335-1342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 205] [Cited by in RCA: 216] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 26. | Moura FA, Goulart MOF, Campos SBG, da Paz Martins AS. The Close Interplay of Nitro-Oxidative Stress, Advanced Glycation end Products and Inflammation in Inflammatory Bowel Diseases. Curr Med Chem. 2020;27:2059-2076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 27. | Schmidt AM, Yan SD, Yan SF, Stern DM. The multiligand receptor RAGE as a progression factor amplifying immune and inflammatory responses. J Clin Invest. 2001;108:949-955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 405] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 28. | Bierhaus A, Stern DM, Nawroth PP. RAGE in inflammation: a new therapeutic target? Curr Opin Investig Drugs. 2006;7:985-991. [PubMed] |
| 29. | Sparvero LJ, Asafu-Adjei D, Kang R, Tang D, Amin N, Im J, Rutledge R, Lin B, Amoscato AA, Zeh HJ, Lotze MT. RAGE (Receptor for Advanced Glycation Endproducts), RAGE ligands, and their role in cancer and inflammation. J Transl Med. 2009;7:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 366] [Cited by in RCA: 469] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 30. | Chuah YK, Basir R, Talib H, Tie TH, Nordin N. Receptor for advanced glycation end products and its involvement in inflammatory diseases. Int J Inflam. 2013;2013:403460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 176] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 31. | Chavakis T, Bierhaus A, Nawroth PP. RAGE (receptor for advanced glycation end products): a central player in the inflammatory response. Microbes Infect. 2004;6:1219-1225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 228] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 32. | Yalcin Kehribar D, Cihangiroglu M, Sehmen E, Avci B, Capraz A, Yildirim Bilgin A, Gunaydin C, Ozgen M. The receptor for advanced glycation end product (RAGE) pathway in COVID-19. Biomarkers. 2021;26:114-118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 33. | Schmidt AM, Vianna M, Gerlach M, Brett J, Ryan J, Kao J, Esposito C, Hegarty H, Hurley W, Clauss M. Isolation and characterization of two binding proteins for advanced glycosylation end products from bovine lung which are present on the endothelial cell surface. J Biol Chem. 1992;267:14987-14997. [PubMed] |
| 34. | Yan SF, Ramasamy R, Schmidt AM. Mechanisms of disease: advanced glycation end-products and their receptor in inflammation and diabetes complications. Nat Clin Pract Endocrinol Metab. 2008;4:285-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 325] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 35. | Rojas A, Delgado-López F, González I, Pérez-Castro R, Romero J, Rojas I. The receptor for advanced glycation end-products: a complex signaling scenario for a promiscuous receptor. Cell Signal. 2013;25:609-614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 76] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 36. | Zen K, Chen CX, Chen YT, Wilton R, Liu Y. Receptor for advanced glycation endproducts mediates neutrophil migration across intestinal epithelium. J Immunol. 2007;178:2483-2490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 61] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 37. | González I, Romero J, Rodríguez BL, Pérez-Castro R, Rojas A. The immunobiology of the receptor of advanced glycation end-products: trends and challenges. Immunobiology. 2013;218:790-797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 47] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 38. | Oczypok EA, Perkins TN, Oury TD. All the "RAGE" in lung disease: The receptor for advanced glycation endproducts (RAGE) is a major mediator of pulmonary inflammatory responses. Paediatr Respir Rev. 2017;23:40-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 125] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 39. | Raucci A, Cugusi S, Antonelli A, Barabino SM, Monti L, Bierhaus A, Reiss K, Saftig P, Bianchi ME. A soluble form of the receptor for advanced glycation endproducts (RAGE) is produced by proteolytic cleavage of the membrane-bound form by the sheddase a disintegrin and metalloprotease 10 (ADAM10). FASEB J. 2008;22:3716-3727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 396] [Cited by in RCA: 440] [Article Influence: 25.9] [Reference Citation Analysis (0)] |
| 40. | Zhang L, Bukulin M, Kojro E, Roth A, Metz VV, Fahrenholz F, Nawroth PP, Bierhaus A, Postina R. Receptor for advanced glycation end products is subjected to protein ectodomain shedding by metalloproteinases. J Biol Chem. 2008;283:35507-35516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 224] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 41. | Deuss M, Reiss K, Hartmann D. Part-time alpha-secretases: the functional biology of ADAM 9, 10 and 17. Curr Alzheimer Res. 2008;5:187-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 75] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 42. | Metz VV, Kojro E, Rat D, Postina R. Induction of RAGE shedding by activation of G protein-coupled receptors. PLoS One. 2012;7:e41823. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 43. | Grauen Larsen H, Marinkovic G, Nilsson PM, Nilsson J, Engström G, Melander O, Orho-Melander M, Schiopu A. High Plasma sRAGE (Soluble Receptor for Advanced Glycation End Products) Is Associated With Slower Carotid Intima-Media Thickness Progression and Lower Risk for First-Time Coronary Events and Mortality. Arterioscler Thromb Vasc Biol. 2019;39:925-933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 44. | Geroldi D, Falcone C, Emanuele E. Soluble receptor for advanced glycation end products: from disease marker to potential therapeutic target. Curr Med Chem. 2006;13:1971-1978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 192] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 45. | Bucciarelli LG, Wendt T, Rong L, Lalla E, Hofmann MA, Goova MT, Taguchi A, Yan SF, Yan SD, Stern DM, Schmidt AM. RAGE is a multiligand receptor of the immunoglobulin superfamily: implications for homeostasis and chronic disease. Cell Mol Life Sci. 2002;59:1117-1128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 226] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 46. | Bierhaus A, Schiekofer S, Schwaninger M, Andrassy M, Humpert PM, Chen J, Hong M, Luther T, Henle T, Klöting I, Morcos M, Hofmann M, Tritschler H, Weigle B, Kasper M, Smith M, Perry G, Schmidt AM, Stern DM, Häring HU, Schleicher E, Nawroth PP. Diabetes-associated sustained activation of the transcription factor nuclear factor-kappaB. Diabetes. 2001;50:2792-2808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 626] [Cited by in RCA: 648] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 47. | Hudson BI, Lippman ME. Targeting RAGE Signaling in Inflammatory Disease. Annu Rev Med. 2018;69:349-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 367] [Article Influence: 45.9] [Reference Citation Analysis (0)] |
| 48. | Pickering RJ, Tikellis C, Rosado CJ, Tsorotes D, Dimitropoulos A, Smith M, Huet O, Seeber RM, Abhayawardana R, Johnstone EK, Golledge J, Wang Y, Jandeleit-Dahm KA, Cooper ME, Pfleger KD, Thomas MC. Transactivation of RAGE mediates angiotensin-induced inflammation and atherogenesis. J Clin Invest. 2019;129:406-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 61] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 49. | Ramasamy R, Yan SF, Schmidt AM. The RAGE axis and endothelial dysfunction: maladaptive roles in the diabetic vasculature and beyond. Trends Cardiovasc Med. 2005;15:237-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 66] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 50. | Manigrasso MB, Juranek J, Ramasamy R, Schmidt AM. Unlocking the biology of RAGE in diabetic microvascular complications. Trends Endocrinol Metab. 2014;25:15-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 155] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 51. | Sims GP, Rowe DC, Rietdijk ST, Herbst R, Coyle AJ. HMGB1 and RAGE in inflammation and cancer. Annu Rev Immunol. 2010;28:367-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 956] [Cited by in RCA: 1016] [Article Influence: 67.7] [Reference Citation Analysis (0)] |
| 52. | Yadav V, Varum F, Bravo R, Furrer E, Bojic D, Basit AW. Inflammatory bowel disease: exploring gut pathophysiology for novel therapeutic targets. Transl Res. 2016;176:38-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 135] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 53. | Ciccocioppo R, Vanoli A, Klersy C, Imbesi V, Boccaccio V, Manca R, Betti E, Cangemi GC, Strada E, Besio R, Rossi A, Falcone C, Ardizzone S, Fociani P, Danelli P, Corazza GR. Role of the advanced glycation end products receptor in Crohn's disease inflammation. World J Gastroenterol. 2013;19:8269-8281. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 54. | Hu Z, Wang X, Gong L, Wu G, Peng X, Tang X. Role of high-mobility group box 1 protein in inflammatory bowel disease. Inflamm Res. 2015;64:557-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 55. | Foell D, Kucharzik T, Kraft M, Vogl T, Sorg C, Domschke W, Roth J. Neutrophil derived human S100A12 (EN-RAGE) is strongly expressed during chronic active inflammatory bowel disease. Gut. 2003;52:847-853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 232] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 56. | Yamasaki H, Mitsuyama K, Masuda J, Kuwaki K, Takedatsu H, Sugiyama G, Yamada S, Sata M. Roles of high-mobility group box 1 in murine experimental colitis. Mol Med Rep. 2009;2:23-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 57. | Manolakis AC, Kapsoritakis AN, Tiaka EK, Potamianos SP. Calprotectin, calgranulin C, and other members of the s100 protein family in inflammatory bowel disease. Dig Dis Sci. 2011;56:1601-1611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 74] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 58. | Cesaro A, Abakar-Mahamat A, Brest P, Lassalle S, Selva E, Filippi J, Hébuterne X, Hugot JP, Doglio A, Galland F, Naquet P, Vouret-Craviari V, Mograbi B, Hofman PM. Differential expression and regulation of ADAM17 and TIMP3 in acute inflamed intestinal epithelia. Am J Physiol Gastrointest Liver Physiol. 2009;296:G1332-G1343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 53] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 59. | Meijer MJ, Mieremet-Ooms MA, van der Zon AM, van Duijn W, van Hogezand RA, Sier CF, Hommes DW, Lamers CB, Verspaget HW. Increased mucosal matrix metalloproteinase-1, -2, -3 and -9 activity in patients with inflammatory bowel disease and the relation with Crohn's disease phenotype. Dig Liver Dis. 2007;39:733-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 120] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 60. | Park L, Raman KG, Lee KJ, Lu Y, Ferran LJ Jr, Chow WS, Stern D, Schmidt AM. Suppression of accelerated diabetic atherosclerosis by the soluble receptor for advanced glycation endproducts. Nat Med. 1998;4:1025-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 856] [Cited by in RCA: 859] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 61. | Lalla E, Lamster IB, Feit M, Huang L, Spessot A, Qu W, Kislinger T, Lu Y, Stern DM, Schmidt AM. Blockade of RAGE suppresses periodontitis-associated bone loss in diabetic mice. J Clin Invest. 2000;105:1117-1124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 266] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 62. | Wendt T, Harja E, Bucciarelli L, Qu W, Lu Y, Rong LL, Jenkins DG, Stein G, Schmidt AM, Yan SF. RAGE modulates vascular inflammation and atherosclerosis in a murine model of type 2 diabetes. Atherosclerosis. 2006;185:70-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 170] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 63. | Bucciarelli LG, Wendt T, Qu W, Lu Y, Lalla E, Rong LL, Goova MT, Moser B, Kislinger T, Lee DC, Kashyap Y, Stern DM, Schmidt AM. RAGE blockade stabilizes established atherosclerosis in diabetic apolipoprotein E-null mice. Circulation. 2002;106:2827-2835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 435] [Cited by in RCA: 447] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 64. | Wang H, Ye J, Liu R, Chen G, Zhao J, Huang L, Yang F, Li M, Zhang S, Jingxie, Xiong L, Chen H, Xu Y, Su M, Xie Y, Zheng F, Geng L, Xu W, Gong S. Clinical Significance of CD147 in Children with Inflammatory Bowel Disease. Biomed Res Int. 2020;2020:7647181. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 65. | Xu Z, Liu R, Huang L, Xu Y, Su M, Chen J, Geng L, Xu W, Gong S. CD147 Aggravated Inflammatory Bowel Disease by Triggering NF-kB-Mediated Pyroptosis. Biomed Res Int. 2020;2020:5341247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 66. | Wang K, Chen W, Zhang Z, Deng Y, Lian JQ, Du P, Wei D, Zhang Y, Sun XX, Gong L, Yang X, He L, Zhang L, Yang Z, Geng JJ, Chen R, Zhang H, Wang B, Zhu YM, Nan G, Jiang JL, Li L, Wu J, Lin P, Huang W, Xie L, Zheng ZH, Zhang K, Miao JL, Cui HY, Huang M, Zhang J, Fu L, Yang XM, Zhao Z, Sun S, Gu H, Wang Z, Wang CF, Lu Y, Liu YY, Wang QY, Bian H, Zhu P, Chen ZN. CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal Transduct Target Ther. 2020;5:283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 408] [Cited by in RCA: 744] [Article Influence: 148.8] [Reference Citation Analysis (0)] |
| 67. | Bao W, Min D, Twigg SM, Shackel NA, Warner FJ, Yue DK, McLennan SV. Monocyte CD147 is induced by advanced glycation end products and high glucose concentration: possible role in diabetic complications. Am J Physiol Cell Physiol. 2010;299:C1212-C1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 57] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 68. | Garg M, Angus PW, Burrell LM, Herath C, Gibson PR, Lubel JS. Review article: the pathophysiological roles of the renin-angiotensin system in the gastrointestinal tract. Aliment Pharmacol Ther. 2012;35:414-428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 119] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 69. | Khajah MA, Fateel MM, Ananthalakshmi KV, Luqmani YA. Anti-Inflammatory Action of Angiotensin 1-7 in Experimental Colitis. PLoS One. 2016;11:e0150861. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 83] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 70. | Santos RAS, Oudit GY, Verano-Braga T, Canta G, Steckelings UM, Bader M. The renin-angiotensin system: going beyond the classical paradigms. Am J Physiol Heart Circ Physiol. 2019;316:H958-H970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 211] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 71. | Gaddam RR, Chambers S, Bhatia M. ACE and ACE2 in inflammation: a tale of two enzymes. Inflamm Allergy Drug Targets. 2014;13:224-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 111] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 72. | Mehta PK, Griendling KK. Angiotensin II cell signaling: physiological and pathological effects in the cardiovascular system. Am J Physiol Cell Physiol. 2007;292:C82-C97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1315] [Cited by in RCA: 1458] [Article Influence: 76.7] [Reference Citation Analysis (0)] |
| 73. | Simões E Silva AC, Teixeira MM. ACE inhibition, ACE2 and angiotensin-(1-7) axis in kidney and cardiac inflammation and fibrosis. Pharmacol Res. 2016;107:154-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 171] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 74. | Tan WSD, Liao W, Zhou S, Mei D, Wong WF. Targeting the renin-angiotensin system as novel therapeutic strategy for pulmonary diseases. Curr Opin Pharmacol. 2018;40:9-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 138] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 75. | Fyhrquist F, Saijonmaa O. Renin-angiotensin system revisited. J Intern Med. 2008;264:224-236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 391] [Cited by in RCA: 417] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 76. | Suzuki Y, Ruiz-Ortega M, Lorenzo O, Ruperez M, Esteban V, Egido J. Inflammation and angiotensin II. Int J Biochem Cell Biol. 2003;35:881-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 489] [Cited by in RCA: 508] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 77. | Husain K, Hernandez W, Ansari RA, Ferder L. Inflammation, oxidative stress and renin angiotensin system in atherosclerosis. World J Biol Chem. 2015;6:209-217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 216] [Cited by in RCA: 232] [Article Influence: 23.2] [Reference Citation Analysis (2)] |
| 78. | Capettini LS, Montecucco F, Mach F, Stergiopulos N, Santos RA, da Silva RF. Role of renin-angiotensin system in inflammation, immunity and aging. Curr Pharm Des. 2012;18:963-970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 115] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 79. | Donoghue M, Hsieh F, Baronas E, Godbout K, Gosselin M, Stagliano N, Donovan M, Woolf B, Robison K, Jeyaseelan R, Breitbart RE, Acton S. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ Res. 2000;87:E1-E9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2045] [Cited by in RCA: 2185] [Article Influence: 87.4] [Reference Citation Analysis (0)] |
| 80. | Ruiz-Ortega M, Ruperez M, Esteban V, Rodriguez-Vita J, Sanchez-Lopez E, Egido J. Modulation of angiotensin II effects, A potential novel approach to inflammatory and immune diseases. Curr Med Chem. 2003;2:379-394. [RCA] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 81. | Garg M, Royce SG, Tikellis C, Shallue C, Batu D, Velkoska E, Burrell LM, Patel SK, Beswick L, Jackson A, Britto K, Lukies M, Sluka P, Wardan H, Hirokawa Y, Tan CW, Faux M, Burgess AW, Hosking P, Monagle S, Thomas M, Gibson PR, Lubel J. Imbalance of the renin-angiotensin system may contribute to inflammation and fibrosis in IBD: a novel therapeutic target? Gut. 2020;69:841-851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 154] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 82. | Santos RAS, Sampaio WO, Alzamora AC, Motta-Santos D, Alenina N, Bader M, Campagnole-Santos MJ. The ACE2/Angiotensin-(1-7)/MAS Axis of the Renin-Angiotensin System: Focus on Angiotensin-(1-7). Physiol Rev. 2018;98:505-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 764] [Cited by in RCA: 782] [Article Influence: 111.7] [Reference Citation Analysis (0)] |
| 83. | Patel S, Rauf A, Khan H, Abu-Izneid T. Renin-angiotensin-aldosterone (RAAS): The ubiquitous system for homeostasis and pathologies. Biomed Pharmacother. 2017;94:317-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 452] [Cited by in RCA: 386] [Article Influence: 48.3] [Reference Citation Analysis (0)] |
| 84. | Garg M, Burrell LM, Velkoska E, Griggs K, Angus PW, Gibson PR, Lubel JS. Upregulation of circulating components of the alternative renin-angiotensin system in inflammatory bowel disease: A pilot study. J Renin Angiotensin Aldosterone Syst. 2015;16:559-569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 69] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 85. | Hirasawa K, Sato Y, Hosoda Y, Yamamoto T, Hanai H. Immunohistochemical localization of angiotensin II receptor and local renin-angiotensin system in human colonic mucosa. J Histochem Cytochem. 2002;50:275-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 79] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 86. | Ferreira-Duarte M, Estevinho MM, Duarte-Araújo M, Magro F, Morato M. Unraveling the Role of ACE2, the Binding Receptor for SARS-CoV-2, in Inflammatory Bowel Disease. Inflamm Bowel Dis. 2020;26:1787-1795. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 87. | Jacobs JD, Wagner T, Gulotta G, Liao C, Li YC, Bissonnette M, Pekow J. Impact of Angiotensin II Signaling Blockade on Clinical Outcomes in Patients with Inflammatory Bowel Disease. Dig Dis Sci. 2019;64:1938-1944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 88. | Wengrower D, Zanninelli G, Pappo O, Latella G, Sestieri M, Villanova A, Faitelson Y, Pines M, Goldin E. Prevention of fibrosis in experimental colitis by captopril: the role of tgf-beta1. Inflamm Bowel Dis. 2004;10:536-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 77] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 89. | Mantaka A, Tsoukali E, Fragkaki M, Karmiris K, Viazis N, Mantzaris GJ, Koutroubakis IE. Is there any role of renin-angiotensin system inhibitors in modulating inflammatory bowel disease outcome? Eur J Gastroenterol Hepatol. 2021;33:364-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 90. | Wang Q, Zhang Y, Wu L, Niu S, Song C, Zhang Z, Lu G, Qiao C, Hu Y, Yuen KY, Wang Q, Zhou H, Yan J, Qi J. Structural and Functional Basis of SARS-CoV-2 Entry by Using Human ACE2. Cell 2020; 181: 894-904. e9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2090] [Cited by in RCA: 2206] [Article Influence: 441.2] [Reference Citation Analysis (0)] |
| 91. | Colón AL, Menchén LA, Hurtado O, De Cristóbal J, Lizasoain I, Leza JC, Lorenzo P, Moro MA. Implication of TNF-alpha convertase (TACE/ADAM17) in inducible nitric oxide synthase expression and inflammation in an experimental model of colitis. Cytokine. 2001;16:220-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 53] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 92. | Brynskov J, Foegh P, Pedersen G, Ellervik C, Kirkegaard T, Bingham A, Saermark T. Tumour necrosis factor alpha converting enzyme (TACE) activity in the colonic mucosa of patients with inflammatory bowel disease. Gut. 2002;51:37-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 159] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 93. | He L, Du J, Chen Y, Liu C, Zhou M, Adhikari S, Rubin DT, Pekow J, Li YC. Renin-angiotensin system promotes colonic inflammation by inducing TH17 activation via JAK2/STAT pathway. Am J Physiol Gastrointest Liver Physiol. 2019;316:G774-G784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 94. | De Francesco EM, Vella V, Belfiore A. COVID-19 and Diabetes: The Importance of Controlling RAGE. Front Endocrinol (Lausanne). 2020;11:526. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 95. | Rojas A, Gonzalez I, Morales MA. SARS-CoV-2-mediated inflammatory response in lungs: should we look at RAGE? Inflamm Res. 2020;69:641-643. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 96. | Bezzio C, Saibeni S, Variola A, Allocca M, Massari A, Gerardi V, Casini V, Ricci C, Zingone F, Amato A, Caprioli F, Lenti MV, Viganò C, Ascolani M, Bossa F, Castiglione F, Cortelezzi C, Grossi L, Milla M, Morganti D, Pastorelli L, Ribaldone DG, Sartini A, Soriano A, Manes G, Danese S, Fantini MC, Armuzzi A, Daperno M, Fiorino G; Italian Group for the Study of Inflammatory Bowel Disease (IG-IBD). Outcomes of COVID-19 in 79 patients with IBD in Italy: an IG-IBD study. Gut. 2020;69:1213-1217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 248] [Cited by in RCA: 260] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 97. | Burke KE, Kochar B, Allegretti JR, Winter RW, Lochhead P, Khalili H, Colizzo FP, Hamilton MJ, Chan WW, Ananthakrishnan AN. Immunosuppressive Therapy and Risk of COVID-19 Infection in Patients With Inflammatory Bowel Diseases. Inflamm Bowel Dis. 2021;27:155-161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 54] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 98. | Occhipinti V, Pastorelli L. Challenges in the Care of IBD Patients During the CoViD-19 Pandemic: Report From a "Red Zone" Area in Northern Italy. Inflamm Bowel Dis. 2020;26:793-796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 99. | Tursi A, Angarano G, Monno L, Saracino A, Signorile F, Ricciardi A, Papa A. COVID-19 infection in Crohn's disease under treatment with adalimumab. Gut. 2020;69:1364-1365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 100. | Bezzio C, Pellegrini L, Manes G, Arena I, Picascia D, Della Corte C, Devani M, Schettino M, Saibeni S. Biologic Therapies May Reduce the Risk of COVID-19 in Patients With Inflammatory Bowel Disease. Inflamm Bowel Dis. 2020;26:e107-e109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 101. | Sigall-Boneh R, Levine A, Lomer M, Wierdsma N, Allan P, Fiorino G, Gatti S, Jonkers D, Kierkus J, Katsanos KH, Melgar S, Yuksel ES, Whelan K, Wine E, Gerasimidis K. Research Gaps in Diet and Nutrition in Inflammatory Bowel Disease. A Topical Review by D-ECCO Working Group [Dietitians of ECCO]. J Crohns Colitis. 2017;11:1407-1419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 83] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 102. | Uribarri J, del Castillo MD, de la Maza MP, Filip R, Gugliucci A, Luevano-Contreras C, Macías-Cervantes MH, Markowicz Bastos DH, Medrano A, Menini T, Portero-Otin M, Rojas A, Sampaio GR, Wrobel K, Garay-Sevilla ME. Dietary advanced glycation end products and their role in health and disease. Adv Nutr. 2015;6:461-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 273] [Article Influence: 27.3] [Reference Citation Analysis (0)] |