Published online Oct 28, 2020. doi: 10.3748/wjg.v26.i40.6270
Peer-review started: June 17, 2020
First decision: July 28, 2020
Revised: August 11, 2020
Accepted: September 23, 2020
Article in press: September 23, 2020
Published online: October 28, 2020
Processing time: 132 Days and 11.5 Hours
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, mostly causing respiratory symptoms, is also known to affect the gastrointestinal tract. Several case reports hypothesize that SARS-CoV-2 could be an etiological factor in acute pancreatitis (AP).
To assess all the available evidence in the literature relating to coronavirus disease 2019 (COVID-19) and AP.
We performed a systematic review of the available literature on the topic. The systematic search was conducted on 15 May 2020 on MEDLINE, EMBASE, CENTRAL, Web of Science and Scopus with a search key using the terms “amylase,” “lipase,” “pancr*,” “COVID-19” and synonyms. Due to the low quality and poor comparability of the studies, a meta-analysis was not performed.
Six case reports and two retrospective cohorts were included, containing data on eleven COVID-19 patients with AP. Five patients had AP according to the Atlanta classification. Other publications did not provide sufficient information on the diagnostic criteria. Most cases were considered SARS-CoV-2-induced, while several established etiological factors were not investigated. We were able to identify other possible causes in most of them.
We strongly highlight the need for adherence to the guidelines during a diagnostic and etiological workup, which could alter therapy.
Core Tip: As the severe acute respiratory syndrome coronavirus 2 pandemic spreads, numerous coronavirus disease 2019 patients will be diagnosed with acute pancreatitis (AP). Viral infections are known etiological factors of AP, but taking a look at the available literature several shortcomings of the diagnostic end etiological workups were uncovered, therefore the causative relationship between coronavirus disease 2019 and AP cannot be established. We highlight the fundamental role of guideline adherence in the diagnosis and etiological workup of AP since etiology-specific therapeutic options are available. Identifying underlying etiological factors is the foundation of high-quality patient care in AP.
- Citation: Juhász MF, Ocskay K, Kiss S, Hegyi P, Párniczky A. Insufficient etiological workup of COVID-19-associated acute pancreatitis: A systematic review. World J Gastroenterol 2020; 26(40): 6270-6278
- URL: https://www.wjgnet.com/1007-9327/full/v26/i40/6270.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i40.6270
In 2019, a novel coronavirus emerged in Wuhan, China, causing multiple cases of severe pneumonia and launching the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. The clinical syndrome seen in SARS-CoV-2 infection is called coronavirus disease 2019 (COVID-19). The main clinical symptoms of COVID-19 are fever, cough, myalgia, and fatigue[1]. Pulmonary involvement is the most frequent[2], but systemic dissociation is seen in severe cases. Furthermore, a significant proportion of patients exhibit gastrointestinal symptoms such as diarrhea, vomiting, and abdominal pain. SARS-CoV-2 was also detected in stool specimens[3] and in the cytoplasm of gastric, duodenal, and rectal glandular epithelial cells[4].
Viral infections such as mumps, Coxsackie, hepatitis, and herpes viruses are known causes of pancreatitis[5]. There is a strong possibility that, like other, less common causes of acute pancreatitis (AP), infectious etiology is underdiagnosed on account of the insufficient workup of idiopathic cases and cases where an apparent cause (e.g., alcohol consumption) is already established[6-8].
On the other hand, during a pandemic of such proportions, polymerase chain reaction testing is made widely available. This will of course lead to a proportion of patients with a variety of diseases, including AP, being diagnosed with SARS-CoV-2 infection. Given the right temporal association, even a more experienced practitioner could be led to ponder a cause-effect relationship between COVID-19 and AP. Even more so, taking into account the often-neglected etiological workup of idiopathic cases and the opportunity to aid the scientific and medical communities by providing information on presumed complications of the infection.
This systematic review aims to assess all publications containing COVID-19 AP cases and to determine the plausibility of an association between the two.
This systematic review was registered with PROSPERO as “Pancreas involvement in COVID-19: A systematic review” under registration number CRD42020186426. After completing the systematic search, we decided to deviate from the protocol for the eligibility of studies: We narrowed our focus to AP from the original plan of any pancreatic involvement. We did so because slight pancreatic enzyme elevation in COVID-19 patients, reported by two studies[9,10], has already been discussed by de-Madaria et al[11] and information on pancreatic cancer patients, reported by three studies[12-14] is at this point far too scarce to even discuss its relation with COVID-19 and effect on outcomes. There were no other deviations from the protocol.
Any study, regardless of design, was considered eligible if it contained the original data on at least 1 SARS-CoV-2-infected individual diagnosed with AP. Only human studies were eligible; studies containing solely animal or in vitro data were excluded.
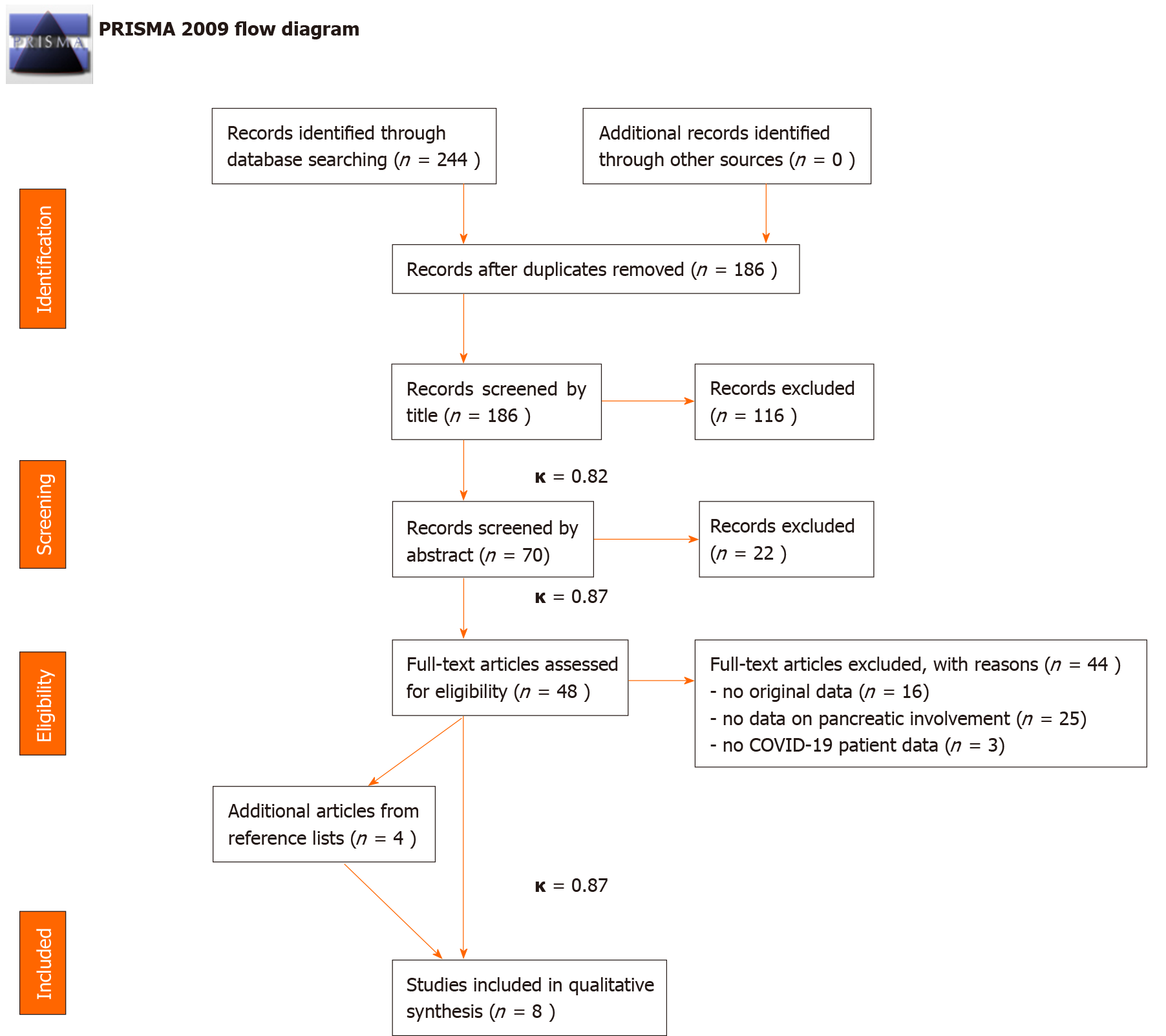
Using the same search key as detailed in the supplementary material (Supplemental 1), the systematic search was conducted in five databases: EMBASE, MEDLINE (via PubMed), CENTRAL, Web of Science, and Scopus. The last systematic search was carried out on May 14, 2020. The search was restricted to 2020, and no other filters were applied. Citations were exported to a reference management program (EndNote X9, Clarivate Analytics). Two independent review authors (Ocskay K and Juhász MF) conducted the selection by title, abstract and full text based on the previously disclosed, predetermined set of rules. After each selection step, Cohen’s kappa coefficient (κ)[15] was calculated. An independent third party (SK) settled any disagreements. Citing articles and references in the studies assessed for eligibility in the full-text phase were reviewed to identify any additional eligible records. Data were extracted from all eligible studies into a standardized Excel sheet designed on the basis of recommendations from the Cochrane Collaboration[16] (for details on data extraction, see Supplemental 2).
The Joanna Briggs Institute Critical Appraisal Checklist for Case Reports[17] was used to assess the risk of bias in case reports, and the Newcastle–Ottawa Scale[18] was used for cohorts (results in Supplemental 3). Due to the design and quality of the included studies, the Grading of Recommendations, Assessment, Development, and Evaluations approach was not used and a very low grade of evidence was auto-matically established.
Only qualitative synthesis was performed; no statistical analysis was carried out.
The details of the systematic search and selection are presented in Figure 1.
In total, six case reports and two retrospective cohort studies were included in this systematic review (Table 1). Information on the diagnostic criteria and etiological factors of AP was collected from the appropriate case reports in Table 2. Of the six cases, five fulfilled the diagnostic criteria for acute pancreatitis[19], and in one case[20] enzyme elevation reached the threshold. However, abdominal pain could not be reported on account of the patient being ventilated and sedated, and no imaging findings were disclosed. A case report by Gou et al[21] was not included in this table, as biliary etiology was determined and COVID-19 symptoms first emerged on day 18 of the patient’s hospital stay; thus, the infection was not assumed as an etiological factor[21].
| Ref. | Study design | Study population | AP, n (%) | Description |
| Aloysius et al[29], United States | Case report | One AP patient with COVID-19 | 1 (100) | 36-year-old obese female presenting with AP. No sign of biliary pathology, denies alcoholism, TG unremarkable |
| Anand et al[44], United Kingdom | Case report | One AP patient with COVID-19 | 1 (100) | A 59-year-old cholecystectomized woman with minimal alcohol consumption, readmitted with abdominal symptoms five days after discharge with doxycycline for co-infection. CT showed signs of AP on a formerly atrophic pancreas |
| Gou et al[21], China | Case report | Four “pancreatic disease” patients with COVID-19 pneumonia | 1 (25) | One female with AP (51), biliary etiology confirmed, showed initial COVID-19 symptoms 18 d after admission |
| Hadi et al[45], Denmark | Case report | Three family members with COVID-19 | 2 (67) | Idiopathic AP in mother (68) and daughter (47), both requiring intensive care and ventilation |
| Hossain et al[23], United States | Retrospective cohort | 119 COVID-19 patients presenting at ER with non-respiratory symptoms | 3/32 (9.4) | Out of the 101 instances where abdominal/pelvic CT was obtained, 32 had acute/significant findings, including three cases of pancreatitis. No more information available on these patients |
| Li et al[22], China | Retrospective cohort | 25 death cases with COVID-19 | 1 (4) | A 56-year-old male patient had AP as an “underlying disease”–it is not clear whether this is from his medical history or was present concomitantly |
| Meireles et al[46], Portugal | Case report | One AP patient with COVID-19 | 1 (100) | 36-year-old female, AP symptoms started on day 11 of disease, US and CT showed no signs of biliary pathology/ischemia. No information on alcohol consumption. Negatively screened for multiple viruses |
| Morrison et al[20], United States | Case report | Two cases of acute hypertriglyceridemia in COVID-19 patients | 1 (50) | Acute hypertriglyceridemia-induced AP after treatment with tocilizumab, ritonavir, lopinavir, ribavirin, hydroxychloroquine, and propofol |
| Ref. | Diagnostic workup | COVID-19 (PCR) | Etiological workup | Quality of case reports | |||||||||||
| Abdominal pain | Enzyme elevation (3 x) | Imaging | Biliary | Alcohol | HTG (> 11.5 mmol/L) | Drug | Hyper-calcemia | Ischemia | Auto-immunity | Viral (except nCoV) | Anatomy | JBI Overall rating ( /8) | Written according to CARE | ||
| Aloysius et al[29], United States | + | + | - | + | ? | - | - | - | ? | ? | ? | ? | - | 3 | No |
| Anand et al[44], United Kingdom | + | ? | + | + | ? | - | ? | + | ? | ? | ? | ? | - | 0 | No |
| Hadi et al[45], Denmark | ? | + | + | + | ? | - | - | ? | - | + | ? | ? | ? | 4 | No |
| + | + | ? | + | ? | ? | ? | + | - | + | ? | ? | ? | 2 | ||
| Meireles et al[46], Portugal | + | + | - | + | ? | - | - | - | - | - | - | - | - | 1 | No |
| Morrison et al[20], United States | ? | + | ? | + | ? | ? | + | + | ? | + | ? | ? | ? | 1 | No |
In a retrospective cohort of COVID-19 mortality cases by Li et al[22], AP is listed as an underlying disease in a single patient without further clarification as to whether it is a past event from the patient’s medical history or it occurred during COVID-19-related hospitalization[22]. Hossain et al[23] noted three cases of AP among 119 patients presenting to the ER with non-respiratory symptoms who turned out to have concomitant SARS-CoV-2 infection[23].
The multiple-hit theory can be implemented in the pathogenesis of AP[24]; therefore, information on possible contributing factors was collected for each case (Table 2). Multiple etiological factors are often responsible for AP[24], but the lack of proper workup often leads to cases being deemed idiopathic or an important factor not being discovered due to the presence of a more convenient diagnosis[6]. In addition to the established etiological factors, various mechanisms have been postulated as the cause of pancreatic damage in COVID-19.
SARS-CoV-2 enters epithelia through the angiotensin-converting enzyme 2[25], which is abundantly expressed in the pancreas[26,27]. SARS-CoV-2 RNA and protein were also shown by in situ hybridization and immunohistochemistry from autopsy samples of infected patients’ pancreas[28]. Aloysius proposed that virus replication may have a direct cytopathic effect or elicit pancreatic cell death as a consequence of the immune response[29]. Furthermore, microvascular injury and thrombosis have been described as a consequence of COVID-19[30,31], which, complicated with shock and gastrointestinal hypoperfusion[32], could also cause pancreatic damage[33].
However, a cause-effect relationship has not been investigated directly so far. Also, before entertaining the possibility of a new virus as a causative agent in cases where no apparent etiological factors are present, other, less frequent causes of AP must be considered. In such cases, the International Association of Pancreatology/American Pancreatic Association (IAP/APA) recommendations should be followed[6,7,19].
For instance, drugs used in treating COVID-19 may cause pancreatic damage directly or indirectly. A patient whose case was presented as idiopathic AP was on a course of doxycycline, which is a drug with a documented probable association with pancreatitis[34]. Several drugs currently used or being considered for COVID-19 might play a role in the pathogenesis of pancreatitis, such as enalapril, asparaginase, estrogens, and steroids[34]. Hypertriglyceridemia, another established etiological factor frequently neglected, can also occur as a consequence of therapy, as in the case described by Morrison et al[20]. Not only tocilizumab[35] but propofol and ritonavir could also have been responsible for the elevation of serum triglyceride levels in this case[36]. Hypertriglyceridemia-associated drug-induced AP was observed[37,38] in association with the following drugs being tested for COVID-19 according to our search on clinicaltrials.gov: lisinopril, asparaginase, estrogens, isotretinoin, steroids, propofol, and ruxolitinib.
In a case reported by Aloysius et al[29], there are no apparent etiological factors present in the description. Even so, the report does not describe any further efforts to identify the seemingly idiopathic etiology, such as performing an endoscopic ultrasonogram. While thoroughly ruled out AP-associated viruses and even screened for antinuclear antibodies, they also did not utilize endoscopic ultrasonogram during the etiology search.
Other than the highlighted problems tied to the etiological workup, we would like to briefly address an issue with the diagnosis. Two studies not included in this review[9,10] labeled patients with serum amylase and/or lipase values higher than the upper limit of normal to possess “pancreatic injury”. As de-Madaria et al[11] pointed out in reflecting on Wang et al[9], the elevation of pancreatic enzyme levels in the blood is not necessarily a consequence of an insult to the pancreas. Possible reasons are the high prevalence of renal impairment and diabetes mellitus, gastroenteritis, and metabolic changes, such as acidosis, or even salivary glandular entry by SARS CoV-2[39-42]. More importantly, a slight elevation in serum amylase and/or lipase levels alone is not established as an indicator of pancreatic damage. The Atlanta diagnostic criteria should be applied when determining the presence of AP[19].
The case reports in our review carry considerable risk of bias and their deviation from the Case Report guideline[43] on reporting methods. As demonstrated, the etiological workup of patients was incomplete, and often COVID-19 was named as the causative agent of AP, while other established factors were also present.
Considering limitations, incomplete reporting of the included studies encompasses a high risk of bias in our analysis[44-46].
To conclude, we strongly emphasize the need for guideline adherence when diagnosing and uncovering the underlying etiological factors of AP, even during a pandemic. As specific therapeutic options[19] are available depending on etiology, neglecting these steps can hinder direct therapy and lower the chances of recovery, while increasing the probability of complications and recurrent episodes.
Since the rapid progression of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic, numerous publications postulated pancreatic involvement. Furthermore, angiotensin-converting enzyme 2 expression -the cellular entry point of the virus- was described in the pancreas.
Multiple etiological factors can be uncovered in a large proportion of acute pancreatitis cases. Therefore, the characterization of SARS-CoV-2 infection as a potential contributing factor was necessary.
Our aim was to review all available clinical evidence on acute pancreatitis cases in coronavirus disease 2019 (COVID-19) patients and to analyze the role of COVID-19 as an etiological factor.
A systematic search was conducted in five databases on 14 May 2020 (registration number CRD42020186426). Record selection and data extraction were carried out by two independent review authors. Studies containing the original data of at least 1 SARS-CoV-2-infected individual diagnosed with acute pancreatitis were considered eligible. The Joanna Briggs Institute Critical Appraisal Checklist for Case Reports and the Newcastle–Ottawa Scale were used for risk of bias assessment.
Eight studies (six case reports and two retrospective cohort studies) were included in this systematic review. All acute pancreatitis cases lacked proper etiological workup, but SARS-CoV-2 infection was confirmed by polymerase chain reaction in all cases. High risk of bias and non-compliance with the Case Report guideline was noted in all case reports.
Guideline adherence is a quality indicator of patient care. We advise all clinicians to conduct proper etiological workup before entertaining the possibility of SARS-CoV-2 as a causative agent of acute pancreatitis.
The potential mechanisms of pancreatic damage in COVID-19 should be investigated utilizing basic research methods and animal models to evaluate a possible causative association between SARS-CoV-2 and AP.
Manuscript source: Unsolicited manuscript
Corresponding Author's Membership in Professional Societies: International Association of Pancreatology, No. 4418.
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Hungary
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Tjora E S-Editor: Zhang L L-Editor:A P-Editor: Ma YJ
| 1. | Li LQ, Huang T, Wang YQ, Wang ZP, Liang Y, Huang TB, Zhang HY, Sun W, Wang Y. COVID-19 patients' clinical characteristics, discharge rate, and fatality rate of meta-analysis. J Med Virol. 2020;92:577-583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 886] [Cited by in RCA: 848] [Article Influence: 169.6] [Reference Citation Analysis (0)] |
| 2. | Conti P, Gallenga CE, Tetè G, Caraffa A, Ronconi G, Younes A, Toniato E, Ross R, Kritas SK. How to reduce the likelihood of coronavirus-19 (CoV-19 or SARS-CoV-2) infection and lung inflammation mediated by IL-1. J Biol Regul Homeost Agents. 2020;34:333-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 75] [Reference Citation Analysis (0)] |
| 3. | Tian Y, Rong L, Nian W, He Y. Review article: gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020;51:843-851. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 594] [Cited by in RCA: 583] [Article Influence: 116.6] [Reference Citation Analysis (0)] |
| 4. | Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology. 2020;158:1831-1833.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1985] [Cited by in RCA: 1995] [Article Influence: 399.0] [Reference Citation Analysis (1)] |
| 5. | Rawla P, Bandaru SS, Vellipuram AR. Review of Infectious Etiology of Acute Pancreatitis. Gastroenterology Res. 2017;10:153-158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 112] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 6. | Zádori N, Párniczky A, Szentesi A, Hegyi P. Insufficient implementation of the IAP/APA guidelines on aetiology in acute pancreatitis: Is there a need for implementation managers in pancreatology? United European Gastroenterol J. 2020;8:246-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 7. | Hallensleben ND, Umans DS, Bouwense SA, Verdonk RC, Romkens TE, Witteman BJ, Schwartz MP, Spanier MB, Laheij R, van Santvoort HC, Besselink MG, van Hooft JE, Bruno MJ; Dutch Pancreatitis Study Group. The diagnostic work-up and outcomes of 'presumed' idiopathic acute pancreatitis: A post-hoc analysis of a multicentre observational cohort. United European Gastroenterol J. 2020;8:340-350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 8. | Mosztbacher D, Hanák L, Farkas N, Szentesi A, Mikó A, Bajor J, Sarlós P, Czimmer J, Vincze Á, Hegyi PJ, ErÅ‘ss B, Takács T, Czakó L, Németh BC, Izbéki F, Halász A, Gajdán L, Hamvas J, Papp M, Földi I, Fehér KE, Varga M, Csefkó K, Török I, Farkas HP, Mickevicius A, Maldonado ER, Sallinen V, Novák J, Ince AT, Galeev S, Bod B, Sümegi J, Pencik P, Dubravcsik Z, Illés D, Gódi S, Kui B, Márta K, Pécsi D, Varjú P, Szakács Z, Darvasi E, Párniczky A, Hegyi P; Hungarian Pancreatic Study Group. Hypertriglyceridemia-induced acute pancreatitis: A prospective, multicenter, international cohort analysis of 716 acute pancreatitis cases. Pancreatology. 2020;20:608-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 86] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 9. | Wang F, Wang H, Fan J, Zhang Y, Wang H, Zhao Q. Pancreatic Injury Patterns in Patients With Coronavirus Disease 19 Pneumonia. Gastroenterology. 2020;159:367-370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 276] [Cited by in RCA: 325] [Article Influence: 65.0] [Reference Citation Analysis (0)] |
| 10. | Liu F, Long X, Zhang B, Zhang W, Chen X, Zhang Z. ACE2 Expression in Pancreas May Cause Pancreatic Damage After SARS-CoV-2 Infection. Clin Gastroenterol Hepatol. 2020;18:2128-2130.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 343] [Cited by in RCA: 473] [Article Influence: 94.6] [Reference Citation Analysis (0)] |
| 11. | de-Madaria E, Siau K, Cárdenas-Jaén K. Increased amylase and lipase in patients with COVID-19 pneumonia: don´t blame the pancreas just yet! Gastroenterology. 2020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 12. | Dane B, Brusca-Augello G, Kim D, Katz DS. Unexpected Findings of Coronavirus Disease (COVID-19) at the Lung Bases on Abdominopelvic CT. AJR Am J Roentgenol. 2020;215:603-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 13. | Luong-Nguyen M, Hermand H, Abdalla S, Cabrit N, Hobeika C, Brouquet A, Goéré D, Sauvanet A. Nosocomial infection with SARS-Cov-2 within Departments of Digestive Surgery. J Visc Surg. 2020;157:S13-S18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 14. | Yu J, Ouyang W, Chua MLK, Xie C. SARS-CoV-2 Transmission in Patients With Cancer at a Tertiary Care Hospital in Wuhan, China. JAMA Oncol. 2020;6:1108-1110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 626] [Cited by in RCA: 776] [Article Influence: 155.2] [Reference Citation Analysis (0)] |
| 15. | McHugh ML. Interrater reliability: the kappa statistic. Biochem Med (Zagreb). 2012;22:276-282. [PubMed] |
| 16. | Li T, Higgins JPT, Deeks JJ. Chapter 5 Collecting data. Cochrane Handbook for Systematic Reviews of Interventions. 2019: 109-141. |
| 17. |
Moola S, Munn Z, Tufanaru C, Aromataris E, Sears K, Sfetcu R, Currie M, Lisy K, Qureshi R, Mattis P, Mu P. Chapter 7: Systematic reviews of etiology and risk. In: Aromataris E, Munn Z (Editors). JBI Manual for Evidence Synthesis. JBI, 2020.
Available from: URL: https://synthesismanual.jbi.global. |
| 18. | Wells G, Shea B, O'Connell D, Peterson j, Welch V, Losos M, Tugwell P. The Newcastle–Ottawa Scale (NOS) for Assessing the Quality of Non-Randomized Studies in Meta-Analysis. 2000. Available from: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. |
| 19. | Working Group IAP/APA Acute Pancreatitis Guidelines. IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013;13:e1-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1080] [Cited by in RCA: 1041] [Article Influence: 86.8] [Reference Citation Analysis (6)] |
| 20. | Morrison AR, Johnson JM, Ramesh M, Bradley P, Jennings J, Smith ZR. Acute hypertriglyceridemia in patients with COVID-19 receiving tocilizumab. J Med Virol. 2020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 69] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 21. | Gou SM, Yin T, Xiong JX, Peng T, Li Y, Wu HS. [Treatment of pancreatic diseases and prevention of infection during outbreak of 2019 coronavirus disease]. Zhonghua Wai Ke Za Zhi. 2020;58:326-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 22. | Li X, Wang L, Yan S, Yang F, Xiang L, Zhu J, Shen B, Gong Z. Clinical characteristics of 25 death cases with COVID-19: A retrospective review of medical records in a single medical center, Wuhan, China. Int J Infect Dis. 2020;94:128-132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 249] [Cited by in RCA: 262] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 23. | Hossain R, Lazarus MS, Roudenko A, Dako F, Mehta V, Alis J, Zalta B, Lei B, Haramati LB, White CS. CT Scans Obtained for Nonpulmonary Indications: Associated Respiratory Findings of COVID-19. Radiology. 2020;296:E173-E179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 24. | Sahin-Tóth M, Hegyi P. Smoking and Drinking Synergize in Pancreatitis: Multiple Hits on Multiple Targets. Gastroenterology. 2017;153:1479-1481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 25. | Dalan R, Bornstein SR, El-Armouche A, Rodionov RN, Markov A, Wielockx B, Beuschlein F, Boehm BO. The ACE-2 in COVID-19: Foe or Friend? Horm Metab Res. 2020;52:257-263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 108] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 26. | Yang JK, Lin SS, Ji XJ, Guo LM. Binding of SARS coronavirus to its receptor damages islets and causes acute diabetes. Acta Diabetol. 2010;47:193-199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 686] [Cited by in RCA: 781] [Article Influence: 52.1] [Reference Citation Analysis (0)] |
| 27. | Fang HJ, Yang JK. Tissue-specific pattern of angiotensin-converting enzyme 2 expression in rat pancreas. J Int Med Res. 2010;38:558-569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 28. | Ding Y, He L, Zhang Q, Huang Z, Che X, Hou J, Wang H, Shen H, Qiu L, Li Z, Geng J, Cai J, Han H, Li X, Kang W, Weng D, Liang P, Jiang S. Organ distribution of severe acute respiratory syndrome (SARS) associated coronavirus (SARS-CoV) in SARS patients: implications for pathogenesis and virus transmission pathways. J Pathol. 2004;203:622-630. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 729] [Cited by in RCA: 789] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 29. | Aloysius MM, Thatti A, Gupta A, Sharma N, Bansal P, Goyal H. COVID-19 presenting as acute pancreatitis. Pancreatology. 2020;20:1026-1027. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 141] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 30. | Magro C, Mulvey JJ, Berlin D, Nuovo G, Salvatore S, Harp J, Baxter-Stoltzfus A, Laurence J. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: A report of five cases. Transl Res. 2020;220:1-13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1635] [Cited by in RCA: 1597] [Article Influence: 319.4] [Reference Citation Analysis (1)] |
| 31. | Yao XH, Li TY, He ZC, Ping YF, Liu HW, Yu SC, Mou HM, Wang LH, Zhang HR, Fu WJ, Luo T, Liu F, Guo QN, Chen C, Xiao HL, Guo HT, Lin S, Xiang DF, Shi Y, Pan GQ, Li QR, Huang X, Cui Y, Liu XZ, Tang W, Pan PF, Huang XQ, Ding YQ, Bian XW. [A pathological report of three COVID-19 cases by minimal invasive autopsies]. Zhonghua Bing Li Xue Za Zhi. 2020;49:411-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 461] [Reference Citation Analysis (0)] |
| 32. | van Haren FM, Sleigh JW, Pickkers P, Van der Hoeven JG. Gastrointestinal perfusion in septic shock. Anaesth Intensive Care. 2007;35:679-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 43] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 33. | Hackert T, Hartwig W, Fritz S, Schneider L, Strobel O, Werner J. Ischemic acute pancreatitis: clinical features of 11 patients and review of the literature. Am J Surg. 2009;197:450-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 41] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 34. | Nitsche C, Maertin S, Scheiber J, Ritter CA, Lerch MM, Mayerle J. Drug-induced pancreatitis. Curr Gastroenterol Rep. 2012;14:131-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 104] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 35. | Flaig T, Douros A, Bronder E, Klimpel A, Kreutz R, Garbe E. Tocilizumab-induced pancreatitis: case report and review of data from the FDA Adverse Event Reporting System. J Clin Pharm Ther. 2016;41:718-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 36. | Rubel AR, Chong PL, Abdullah MS, Asli R, Momin RN, Mani BI, Chong VH. Lipemic serum in patients with Coronavirus Disease 2019 (COVID-19) undergoing treatment. J Med Virol. 2020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 37. | Watson AP, Brunstein CG, Holtan SG. Life-Threatening Hypertriglyceridemia in a Patient on Ruxolitinib and Sirolimus for Chronic Graft-versus-Host Disease. Case Rep Transplant. 2018;2018:4539757. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 38. | Elkhouly MA, Salazar MJ, Simons-Linares CR. Hypertriglyceridemia-Associated Drug-Induced Acute Pancreatitis. Pancreas. 2019;48:22-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 39. | Tositti G, Fabris P, Barnes E, Furlan F, Franzetti M, Stecca C, Pignattari E, Pesavento V, de Lalla F. Pancreatic hyperamylasemia during acute gastroenteritis: incidence and clinical relevance. BMC Infect Dis. 2001;1:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 40. | Steinberg WM, Nauck MA, Zinman B, Daniels GH, Bergenstal RM, Mann JF, Steen Ravn L, Moses AC, Stockner M, Baeres FM, Marso SP, Buse JB; LEADER Trial investigators. LEADER 3--lipase and amylase activity in subjects with type 2 diabetes: baseline data from over 9000 subjects in the LEADER Trial. Pancreas. 2014;43:1223-1231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 55] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 41. | Liu L, Wei Q, Alvarez X, Wang H, Du Y, Zhu H, Jiang H, Zhou J, Lam P, Zhang L, Lackner A, Qin C, Chen Z. Epithelial cells lining salivary gland ducts are early target cells of severe acute respiratory syndrome coronavirus infection in the upper respiratory tracts of rhesus macaques. J Virol. 2011;85:4025-4030. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 259] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 42. | Wang C, Wu H, Ding X, Ji H, Jiao P, Song H, Li S, Du H. Does infection of 2019 novel coronavirus cause acute and/or chronic sialadenitis? Med Hypotheses. 2020;140:109789. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 43. | Riley DS, Barber MS, Kienle GS, Aronson JK, von Schoen-Angerer T, Tugwell P, Kiene H, Helfand M, Altman DG, Sox H, Werthmann PG, Moher D, Rison RA, Shamseer L, Koch CA, Sun GH, Hanaway P, Sudak NL, Kaszkin-Bettag M, Carpenter JE, Gagnier JJ. CARE guidelines for case reports: explanation and elaboration document. J Clin Epidemiol. 2017;89:218-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 741] [Cited by in RCA: 1022] [Article Influence: 127.8] [Reference Citation Analysis (0)] |
| 44. | Anand ER, Major C, Pickering O, Nelson M. Acute pancreatitis in a COVID-19 patient. Br J Surg. 2020;107:e182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 69] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 45. | Hadi A, Werge M, Kristiansen KT, Pedersen UG, Karstensen JG, Novovic S, Gluud LL. Coronavirus Disease-19 (COVID-19) associated with severe acute pancreatitis: Case report on three family members. Pancreatology. 2020;20:665-667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 147] [Cited by in RCA: 169] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 46. | Meireles PA, Bessa F, Gaspar P, Parreira I, Silva VD, Mota C, Alvoeiro L. Acalculous Acute Pancreatitis in a COVID-19 Patient. Eur J Case Rep Intern Med. 2020;7:001710. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |