Published online Oct 21, 2020. doi: 10.3748/wjg.v26.i39.5970
Peer-review started: July 21, 2020
First decision: August 8, 2020
Revised: August 12, 2020
Accepted: September 12, 2020
Article in press: September 12, 2020
Published online: October 21, 2020
Processing time: 91 Days and 22.8 Hours
Acute pancreatitis (AP) is a sudden inflammatory process of the pancreas that may also involve surrounding tissues and/or remote organs. Inflammation and parenchymal cell death are common pathological features of this condition and determinants of disease severity. Polyethylene glycols (PEGs) are non-immunogenic, non-toxic water-soluble polymers widely used in biological, chemical, clinical and pharmaceutical settings.
To evaluate the protective effect of a 35-kDa molecular weight PEG (PEG35) on the pancreatic damage associated to cerulein-induced acute pancreatitis in vivo and in vitro.
Wistar rats were assigned at random to a control group, a cerulein–induced AP group and a PEG35 treatment group. AP was induced by five hourly intraperitoneal injections of cerulein (50 μg/kg/bw), while the control animals received saline solution. PEG35 was administered intraperitoneally 10 minutes before each cerulein injection in a dose of 10 mg/kg. After AP induction, samples of pancreatic tissue and blood were collected for analysis. AR42J pancreatic acinar cells were treated with increasing concentrations of PEG35 prior to exposure with tumor necrosis factor α (TNFα), staurosporine or cerulein. The severity of AP was determined on the basis of plasma levels of lipase, lactate dehydrogenase activity, pancreatic edema and histological changes. To evaluate the extent of the inflammatory response, the gene expression of inflammation-associated markers was determined in the pancreas and in AR42J-treated cells. Inflammation-induced cell death was also measured in models of in vivo and in vitro pancreatic damage.
Administration of PEG35 significantly improved pancreatic damage through reduction on lipase levels and tissue edema in cerulein-induced AP rats. The increased associated inflammatory response caused by cerulein administration was attenuated by a decrease in the gene expression of inflammation-related cytokines and inducible nitric oxide synthase enzyme in the pancreas. In contrast, pancreatic tissue mRNA expression of interleukin 10 was markedly increased. PEG35 treatment also protected against inflammation-induced cell death by attenuating lactate dehydrogenase activity and modulating the pancreatic levels of apoptosis regulator protein BCL-2 in cerulein hyperstimulated rats. Furthermore, the activation of pro-inflammatory markers and inflammation-induced cell death in pancreatic acinar cells treated with TNFα, cerulein or staurosporine was significantly reduced by PEG35 treatment, in a dose-dependent manner.
PEG35 ameliorates pancreatic damage in cerulein-induced AP and AR42J-treated cells through the attenuation of the inflammatory response and associated cell death. PEG35 may be a valuable option in the management of AP.
Core Tip: Acute pancreatitis (AP) is a sudden inflammatory condition of the pancreas with variable involvement of peri-pancreatic tissues and/or remote organ systems. This disease is a major clinical challenge for which no specific pharmacological therapy currently exists. The manuscript describes the protective role of 35-kDa molecular weight polyethylene glycol (PEG35) on cerulein-induced AP. PEG35 treatment was able to lessen the inflammatory process in the pancreas and associated cell death in cerulein-induced AP and in vitro models of pancreatic damage.
- Citation: Ferrero-Andrés A, Panisello-Roselló A, Roselló-Catafau J, Folch-Puy E. Polyethylene glycol 35 ameliorates pancreatic inflammatory response in cerulein-induced acute pancreatitis in rats. World J Gastroenterol 2020; 26(39): 5970-5982
- URL: https://www.wjgnet.com/1007-9327/full/v26/i39/5970.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i39.5970
Acute pancreatitis (AP) is an inflammatory disease of the exocrine pancreas characterized by abnormal intracellular activation of proteolytic enzymes. Parenchymal injury, pancreatic acinar cell death and an intense inflammatory reaction are common pathological features of this condition and determine the severity of the disease[1]. A majority of patients presenting with AP have the mild form of the disease, which is mostly self-limited and consists of the appearance of edema and inflammation of the pancreas[2]. In this group, organ failure and local complications are generally not observed, and the disease usually resolves in the first week. However, between 20% and 30% develop a severe form requiring intensive care unit admission, which is often associated with local and systemic complications and, in some occasions, leads to death[3]. To date, no drug is available to prevent or treat this condition, and any improved clinical outcomes have mostly been due to continuous advancement of various supportive treatments.
Although pancreatic inflammation may be firstly caused by acinar events such as trypsinogen activation, it finally depends on the subsequent stimulation of components of the innate immune system. The initial acinar cell damage triggers the release of pro-inflammatory cytokines and chemokines, leading to increase of microvascular permeability and subsequent formation of interstitial edema[4]. Activation of inflammatory cells then provokes the production of additional cytokines and other mediators that initiate the inflammatory response. These mediators recruit different types of leukocytes (first neutrophils, followed by macrophages, monocytes and lymphocytes) to the pancreas. In parallel to the pro-inflammatory response, an anti-inflammatory response is also released[5]. If the anti-inflammatory response is adequate, the local inflammation resolves at this stage. However, in some cases, an overwhelming pro-inflammatory response drives the migration of inflammatory mediators into systemic circulation, leading to distant organ dysfunction[6].
Polyethylene glycols (PEGs) are hydrophilic polymers comprised of repeating ethylene glycol units[7]. PEGs have several physicochemical properties that make it advantageous in diverse biological, chemical and pharmaceutical settings, especially in view of its low toxicity. For instance, these polymers have been found to exert beneficial effects in several in vivo and in vitro models of cell and tissue injury[8-10].
There are very few studies linking PEGs of different molecular weight with an anti-inflammatory activity. In a model of traumatic inflammation, the intraperitoneal administration of 4-kDa molecular weight PEG prevented the formation of initial adhesions and reduced the leukocytes number in the peritoneal cavity as a consequence of an inflammatory peritoneal reaction[11]. Oral treatment with 4-kDa PEG in experimental colitis reinforced the epithelial barrier function and reduced the inflammation of the colon[12]. Likewise, in two different models of gut-derived sepsis, therapeutic administration of PEG reduced inflammatory cytokine expression and activation of neutrophils[13]. Our group has recently demonstrated an anti-inflammatory role for PEG35 in an experimental model of severe necrotizing AP. In this sense, the therapeutic administration of PEG35 notably alleviated the severity of AP and protected against the associated lung inflammatory response[14].
Based on the protective features of PEGs, we now have evaluated the effects of PEG35 in experimental models of pancreatic damage in vivo and in vitro.
All experimental animal proceedings were conducted according with European Union regulatory standards for experimentation with animals (Directive 2010/63/EU on the Protection of Animals Used for Scientific Purposes). The Ethical Committee for Animal Experimentation (CEEA, University of Barcelona, April 11, 2018, ethic approval number: 211/18) authorized all animal experimentation.
The protocol was designed to minimize pain and discomfort to animals. Adult male Wistar rats (n = 21) weighing 200-250 g were purchased from Charles River (Boston, MA, United States) and accommodated in a controlled environment with free access to standard laboratory pelleted formula (A04; Panlab, Barcelona, Spain) and tap water. Rats were kept in a climate-controlled environment with a 12-h light/12-h dark cycle for one week. For the 12 h prior to the experiment of AP induction, rats were fasted with free access to drinking water.
Rats were randomly selected and assigned to three equal groups: (1) Treated with saline, as controls (n = 7); and (2) Treated with cerulein, to induce AP (CerAP, n = 7) and (3) Treated with cerulein after a PEG35 pretreatment (PEG35 + CerAP, n = 7). Immediately before the first injection of PEG35 or saline, 0.05 mg/kg of buprenorphine was administered as an analgesic. Cerulein (Sigma-Aldrich, St. Louis, MO) was dissolved with phosphate-buffered saline (PBS) and administered intraperitoneally at a supramaximal stimulating concentration of 50 μg/kg/body weight (bw) at 1-h intervals (total of 5 injections); control animals received intraperitoneal saline solution with the same regime. The use of this supramaximal dosage of cerulein induce a transient form of interstitial edematous AP characterized by marked hypera-mylasemia, pancreatic edema and neutrophil infiltration within the pancreas, as well as pancreatic acinar cell vacuolization and necrosis[15].
PEG35 was administered intraperitoneally at a dose of 10 mg/kg, 10 min prior to each cerulein injection. Immediately after the last injection of cerulein or saline, animals were euthanized by intraperitoneal injection of 40-60 mg/kg of sodium pentobarbital, and blood was collected from the vena cava in heparinized syringes. Harvested blood was centrifuged and the obtained plasma was stored at −80 °C until analysis. Four tissue samples from each animal were taken from the head of the pancreas. One portion of each tissue sample was immediately weighed and oven-dried for the wet-to-dry weight ratio calculation. Another portion was fixed in 10% phosphate−buffered formalin for histological analysis. The third portion was frozen and stored at −80 ºC for western blot analysis, and the last portion was saved in RNAlater solution for real-time qRT-PCR analysis.
Pancreas tissue was fixed in 10% phosphate-buffered formalin and then embedded in paraffin. 3-μm thickness sections were mounted on glass slides. Slides were dewaxed and rehydrated and stained with hematoxylin and eosin. Assessment of changes in the tissue was carried out by an experienced pathologist through the examination of different microscopic fields randomly chosen from each experimental group in a blinded manner. Pancreatic tissue sections were evaluated for the severity of pancreatitis based on edema, inflammatory infiltration, parenchymal necrosis, and vacuolation of acinar cells.
The rat pancreatic acinar AR42J cell line was purchased from Sigma (St. Louis, MI, United States). Cells were grown at 37 ºC in RPMI medium supplemented with 100 mL/L fetal bovine serum, 100 U/ml penicillin and 100 μg/mL streptomycin in a humidified atmosphere of 50 mL/L CO2. Acinar cells were plated at a density of 3 × 105/well in 12-well culture plates, or at a density of 2 × 104/well in 96-well plates, and allowed to attach for 24 h or 48 h. Cells were pretreated with PEG35 diluted in PBS, at a concentration of 0.5, 1, 2, 4, or 6% for 30 min prior to treatment with the appropriate stimuli: 2 µmol/L or 4 µmol/L staurosporine, 100 ng/mL TNFα or 10 nM cerulein. All three reagents were purchased from Sigma-Aldrich (St. Louis, MO, United States). Time points of 3 h were used for TNFα treatment, and of 24 h for the remaining stimuli.
Plasma lipase activity levels were determined using a turbidimetric assay kit from Randox (County Antrim, Crumlin, United Kingdom), in accordance with the supplier’s specifications. Briefly, the degradation of triolein by the pancreatic lipase results in lowered turbidity, which was determined in the sample at 340 nm using a microplate reader (iEMS Reader MF; Labsystems, Helsinki, Finland). The activity of the sample was obtained in U/L. All samples were run in duplicate.
Edema formation in the pancreas was evaluated by the determination of the wet-to-dry weight ratio. A portion of the pancreas was weighed. The content of water was measured by calculating the wet-to-dry weight ratio from the initial weight (wet weight) and its weight after incubation in an oven at 60 °C for 48 h (dry weight).
Lactate dehydrogenase (LDH) activity was measured in plasma samples and cell culture supernatants using the Lactate Dehydrogenase Assay Kit (Abcam; Cambridge, United Kingdom). Briefly, LDH reduces NAD to NADH, which interacts with a specific probe to produce colour. Changes in absorbance due to NADH formation were measured at 450 nm at 37 °C using an automated microplate reader (iEMS Reader MF; Labsystems, Helsinki, Finland). Sample activity was expressed in mU/mL. All samples were run in duplicate. The lower limit of detection for ELISA ranged from 14 to 36 mU/mL.
The cell proliferation was determined by measuring metabolic activity of the cells through the reduction of the tetrazolium dye MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] to its insoluble formazan. AR42J cells were seeded in 96-well plates at a density of 2 × 104 cells/well in 100 μL of culture medium with or without the compounds to be tested for 24 h. MTT reagent was added and incubated for 2 h at 37 °C, and the formazan produced in the cells formed dark crystals at the bottom of the wells. Crystal-dissolving solution was added and the absorbance of each sample was quantified at 570 nm using an automated microplate reader (iEMS Reader MF; Labsystems, Helsinki, Finland). All samples were run in duplicate. The absorbance intensity was proportional to the number of viable cells.
Total RNA from the pancreatic tissue and cultured cells was extracted with Nucleozol reagent (Macherey-Nagel, Dueren, Germany) in accordance with the manufacturer’s protocol. RNA concentration and quality were measured with the OD A260/A280 ratio and the OD A260/A230 ratio, respectively. Reverse transcription was performed on a 1 µg RNA sample employing the iScript cDNA Synthesis Kit (Bio-Rad Laboratories, Hercules, CA, United States). PCR amplification was performed using SsoAdvanced™ Universal SYBR® Green Supermix (Bio-Rad Laboratories, Hercules, CA, United States) on a CFX Real-Time PCR Detection System (Bio-Rad Laboratories, Hercules, CA, United States) using 10 µL of amplification mixture containing 50 ng of reverse-transcribed RNA and 250 nmol/L of the corresponding forward and reverse primers.
PCR primers for the detection of interleukin (IL) 6, IL1β, IL10, inducible isoform of nitric oxide synthase (iNOS), or glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were validated primers from BioRad (Hercules, CA, United States). PCR primers for tumor necrosis factor α (TNFα), designed with Primer3.0 plus[16], were: TNFα forward, 5’- ATGGGCTCCCTCTCATCAGT-3’ and reverse, 5’-GCTTG GTGGTTTGCTACGAC-3’. The specificity of amplicon was determined by melting curve analysis. Threshold cycle values were normalized to GAPDH gene expression and the ratio of the relative expression of target genes to GAPDH was calculated by the DCt formula.
Pancreatic tissue was homogenized in ice-cold RIPA buffer. Lysates were then centrifuged at 15000 g for 20 min at 4 °C, and the supernatants were collected. Supernatant protein concentrations were measured using the Bradford protein assay (Bio-Rad Laboratories, Hercules, CA, United States). SDS-PAGE was performed on a 10% gel and proteins were transferred to a polyvinylidene difluoride membrane for blotting.
The following antibodies were used for immunoblotting:rabbit polyclonal cleaved caspase-3 (Asp175) antibody (1:800 dilution, reference #9661) from Cell Signaling, rabbit polyclonal BCL-2 (1: 500 dilution, reference #59348) from Abcam (Cambridge, United Kingdom) and β-actin-HRP conjugated (1: 20000 dilution, reference A3854) from Sigma (Sigma-Aldrich, St. Louis, MO). Bound antibodies were detected using enhanced chemiluminescence (ECL) (Bio-Rad Laboratories, Hercules, CA, United States), and were analyzed using ChemiDoc™ Touch Imaging System (Bio-Rad Laboratories, Hercules, CA, United States). Protein expression of cleaved caspase-3 and BCL-2 were normalized to β-actin for quantification.
All data were exported into Graph Pad Prism 4 (GraphPad Software, Inc.) and presented as means ± SEM. Statistical analyses were carried out by one-way analysis of variance (ANOVA), followed by Tukey’s multiple comparison test to determine the significance between pairs. The minimal level of statistical significance was considered to be < 0.05.
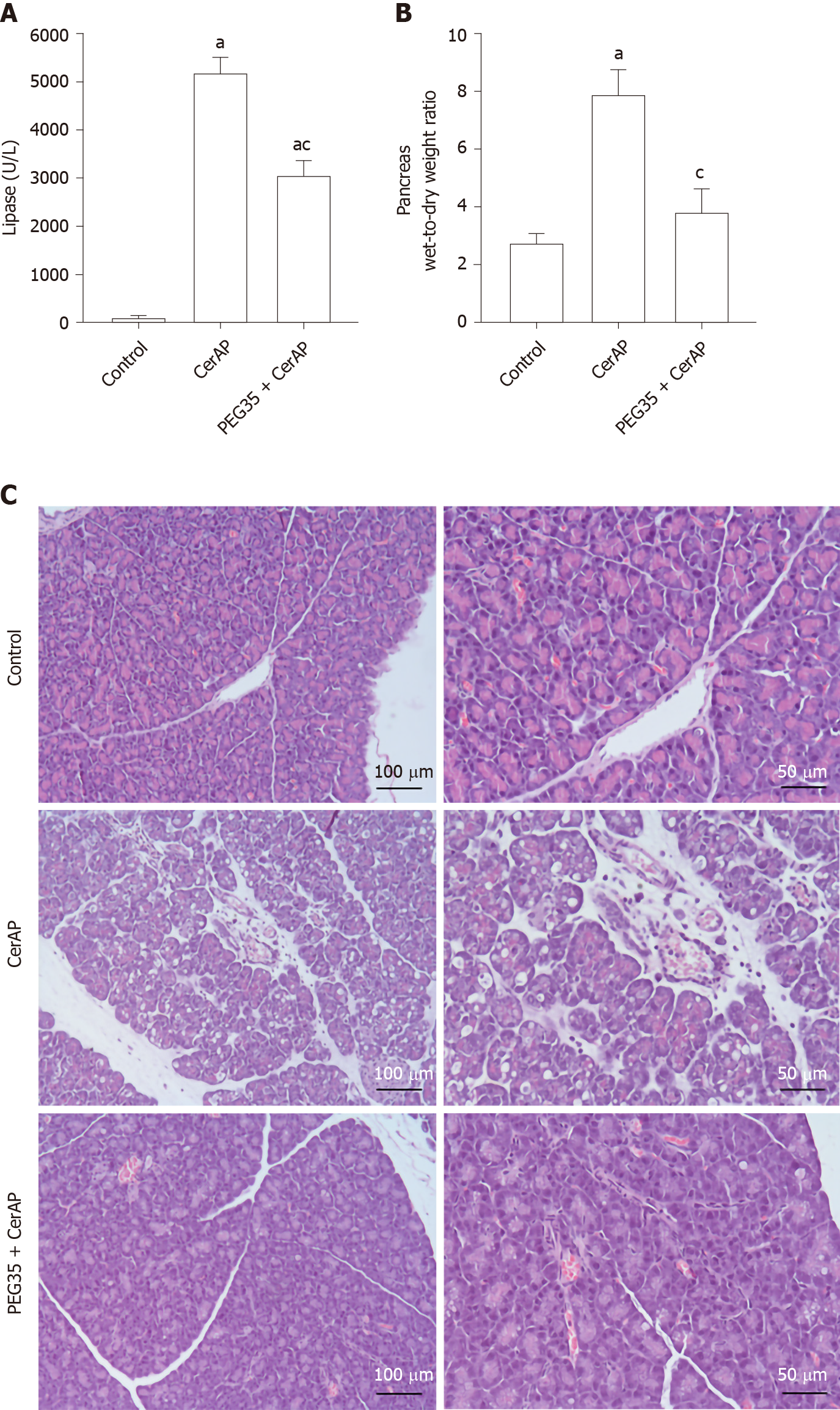
Cerulein-induced AP in rats was associated with significant raised plasma levels of lipase, comparing with the control group, reflecting the degree of pancreatic injury (Figure 1A). Such increase was significantly reduced in rats pre-treated with intravenous PEG35 at 10 mg/kg.
As cerulein-induced pancreatitis is characterized by a progressive interstitial edema development, we analyzed the pancreas wet-to-dry weight ratio (Figure 1B). A significant increase in the pancreas wet-to-dry weight ratio was observed in rats after AP induction with cerulein (7.865 ± 0.86) as compared to control rats (2.76 ± 0.28). However, this increase could be largely prevented by co-treatment with PEG35 in cerulein-treated rats, with a wet-to-dry weight ratio of 3.8 ± 0.85.
Histopathological results showed that cerulein hyperstimulated rats caused an interstitial edematous acute pancreatitis with considerable areas of interstitial edema, local necrosis, infiltrated polymorphonuclear neutrophils and vacuolation of the acinar cells. (Figure 1C). In the PEG35-treated group, there were consistent reductions in these characteristics.
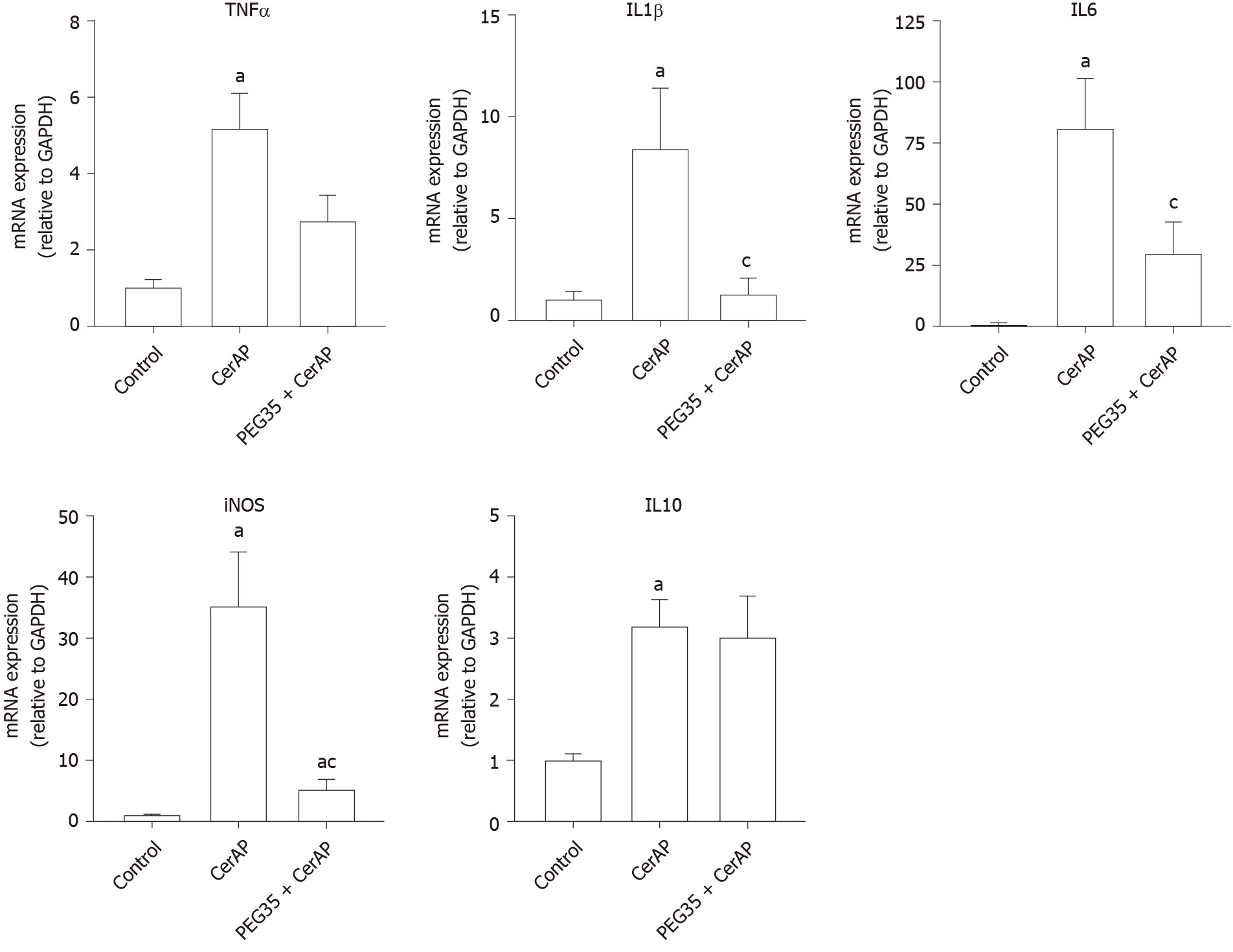
Further, we explored whether PEG35 treatment improves the inflammatory response after cerulein hyperstimulation in rats, by measuring the gene expression of inflammatory mediators in the pancreas. Pancreatic tissue levels of IL6, IL1β, TNFα, IL10 and iNOS increased markedly in rats after AP induction as compared to that of control rats (Figure 2). Notably, PEG35 treatment significantly reduced the AP-induced increases in IL1β, IL6 and iNOS. While TNFα expression levels showed a tendency to decrease, this was not statistically significant. Finally, as expected based on its anti-inflammatory role, the gene expression of the IL10 cytokine was not reduced in PEG35-treated animals.
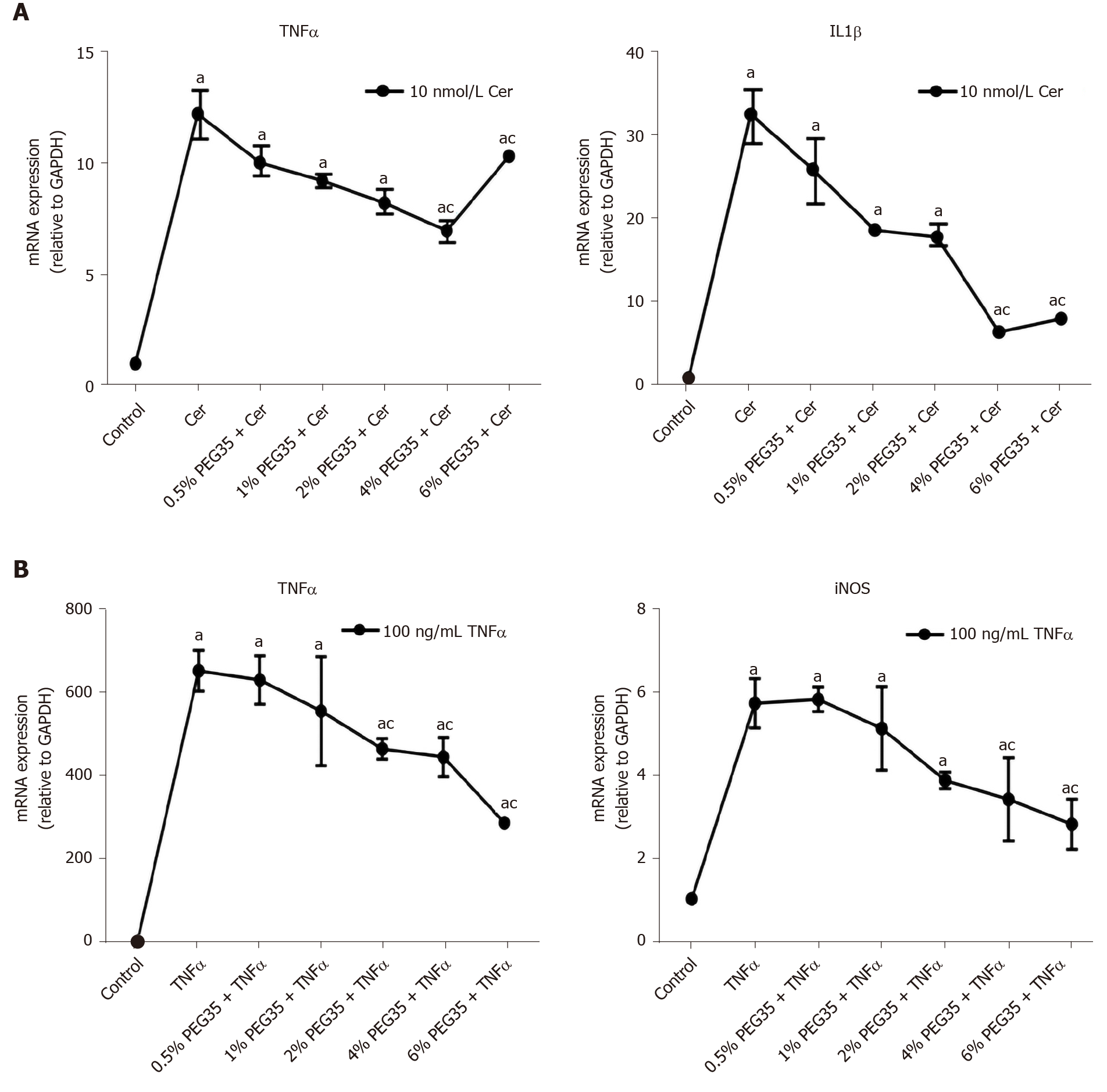
In addition to its in vivo effects, a direct anti-inflammatory effect of PEG35 was also observed in in vitro model. Specifically, in a model of cerulein-induced inflammation in the acinar cells using cultured AR42J cells, PEG35 attenuated the gene expression of the pro-inflammatory IL1β and TNFα in a dose-dependent manner (Figure 3A). Additionally, TNFα-treated cells induced the production of iNOS as well as of TNFα itself, both of which were markedly reduced after the treatment with increasing concentrations of PEG35 (Figure 3B).
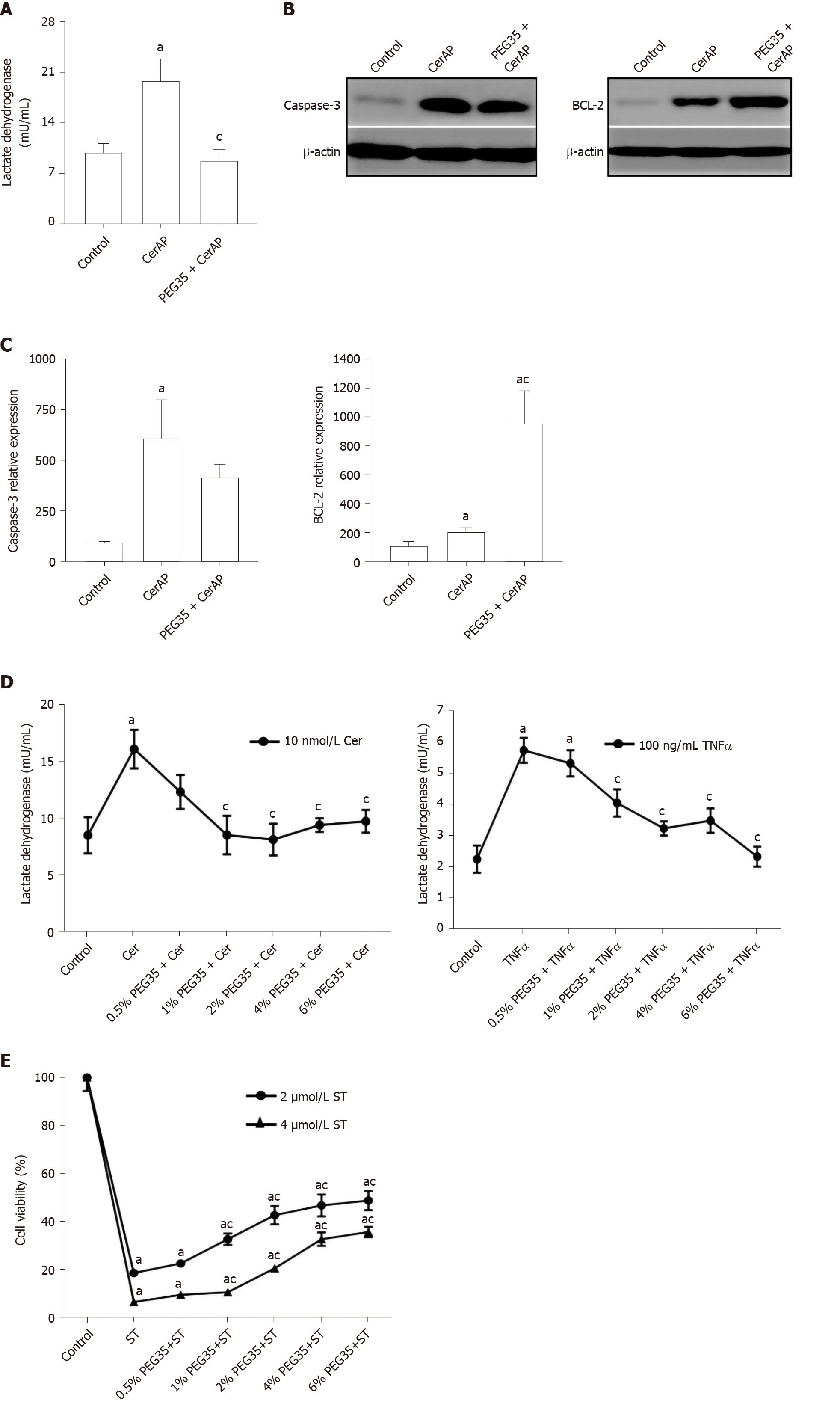
To investigate the potential protective effects of PEG35 on the pancreas, cell death was determined through LDH release and expression of the apoptosis-related proteins BCL-2 and cleaved-caspase-3 by Western blot. Indeed, a significant increment in LDH activity in plasma occurred in cerulein AP-induced animals (Figure 4A). Notably, rats that had PEG35 co-treatment had significantly reduced levels of the LDH necrotic marker.
The pancreatic levels of cleaved caspase-3 and BCL-2 were also markedly higher following cerulein-induced AP as compared to the control group, (Figure 4B and C). The administration of PEG35 promoted a further increase in the levels of anti-apoptotic BCL-2 as compared with cerulein hyperstimulated rats while the reduction in the pro-apoptotic cleaved caspase-3 was not statistically significant.
The decreased LDH activity observed in vivo in PEG35-treated animals led us to examine cell death in in vitro models of inflammation. AR42J cells are a well-established cell model for studying intracellular mechanisms involved in the cell death and inflammatory responses of acute pancreatitis. We therefore analysed whether PEG35 affected the release of LDH in AR42J cells in the presence of the pro-inflammatory stimulus cerulein or TNFα (Figure 4D). Indeed, both cerulein and TNFα-induced cell death were significantly reduced by PEG35 in a dose-dependent manner. Likewise, PEG35 markedly prevented staurosporine-induced AR42J apoptotic cell death in a dose-dependent manner (Figure 4E). These results suggest that PEG35 exerts a protective role against inflammation-induced cell death in vitro and in vivo.
Acute pancreatitis (AP) is an inflammatory disease that can have a mild to severe course. We have recently reported an anti-inflammatory role for PEG35 in a severe necrotizing AP experimental model. To further investigate the effect of this polymer in a milder form of the disease, we used a model of cerulein-induced mild edematous pancreatitis that is mainly characterized by a dysregulation of the production and secretion of digestive enzymes, interstitial edema formation, infiltration of neutrophil and mononuclear cells within the pancreas, cytoplasmic vacuolization and the death of acinar cells[17]. We determined that PEG35 reduced the course of cerulein-induced AP by inhibiting the inflammatory response as well as inflammation-induced cell death. In our study, treating the animals with PEG35 significantly abrogated the severity of cerulein-induced AP, as indicated by the lessened activity of lipase in plasma and edema formation as well as histopathological features of AP in the PEG35-treated animals.
A sudden inflammatory response in the pancreas contributes to the development of AP, primarily through the release of inflammatory cytokines. TNFα has long been considered as one of the initial triggers of the inflammatory cascade in experimental pancreatitis[18]. In this setting, stimulation of acinar cells of the pancreas by TNFα have been reported to cause a direct activation of pancreatic enzymes, contributing to premature protease activation and cell necrosis[19]. Increased accumulation of TNFα promotes the production of other inflammatory cytokines, including IL1β and IL6, which result in the activation of an inflammatory cascade that leads to widespread tissue damage in multiple tissues and organs. Indeed, the levels of TNFα, IL1β and IL6 have been correlated with the severity of AP[20-23]. In the current study, treatment with PEG35 was capable to significantly reduce the AP-induced raises in pro-inflammatory IL1β and IL6. However, no significant effect on TNFα was observed. This fact could be explained by the levels of IL10 found in rats co-treated with cerulein and PEG35, which were similar to those found in those only treated with cerulein. As IL10 plays a fundamental role in the attenuation of the cytokine response during acute inflammation, the significant increase of IL10 found in hyperstimulated rats may contribute to slow TNFα production, with an observed tendency towards a decrease in its expression. Indeed, in an experimental model of cerulein-induced AP, intraperitoneal IL10 administration attenuated TNFα production, which was associated with dramatically lessened pancreatitis severity and mortality[24].
Furthermore, a direct anti-inflammatory effect of PEG35 was observed in cultured AR42J cells. In an in vitro model of cerulein-induced inflammation, PEG35 was able to attenuate the gene expression of pro-inflammatory IL1β and TNFα in a dose-dependent manner. Moreover, PEG35 reduced the levels of TNFα in AR42J cells stimulated with TNFα.
Pro-inflammatory cytokines are known to activate the inducible iNOS and the subsequent production of nitric oxide, thus contributing to the pathophysiology of AP. In fact, the degree of pancreatic inflammation and tissue injury of cerulein-induced AP has been found to be markedly reduced in iNOS-deficient mice[25]. In our study, we observed an increased mRNA expression of iNOS following cerulein hyperstimulation in rats, which underwent a significant reduction after PEG35 treatment. Likewise, PEG35 abrogated TNFα-induced iNOS expression in acinar cells in a concentration-dependent manner. Altogether, these results suggest that PEG35 treatment reduced pancreatic inflammation in pancreatitis by suppressing the expression of pro-inflammatory mediators.
These changes in the inflammatory response brought about by PEG35 treatment were further emphasized by a reduction in pancreatic cell death. PEG35 treatment reduced cell death in cerulein-induced AP rats by lowering plasmatic LDH activity. In addition, the increased release of LDH observed in cerulein and TNFα-treated acinar cells in vitro was reverted upon incubation with increasing concentrations of PEG35.
In the pancreas, inflammation is associated with injured acinar cells that can go through necrosis or apoptosis. Thus, we measured the apoptosis index in pancreatic tissue following cerulein-induced AP. Injured pancreatic tissue induced a significant increase in cleaved caspase-3 and BCL-2 apoptotic proteins as compared to the respective controls. Following treatment with PEG35, anti-apoptotic BCL-2 further increased as compared with cerulein-treated animals while cleaved caspase-3 levels were similar to that found in cerulein hyperstimulated animals. Collectively, these findings suggest that PEG35 has anti-apoptotic and anti-necrotic properties for cerulein-induced pancreatitis.
In conclusion, results from this study reveal a mechanism by which PEG35 exerts anti-inflammatory effects that alleviate experimental cerulein-induced AP, by inhibiting the inflammatory response as well as inflammation-induced cell death. Because of its low toxicity as well as its proven biocompatibility, PEG35 could be used as a new therapeutic tool to resolve the cellular damage associated to mild AP.
Acute pancreatitis (AP) is a common gastrointestinal condition with an increasing incidence worldwide. The course of the disease ranges from a mild, self-limiting condition to a more severe acute illness with a high morbidity and mortality. Our group has previously demonstrated an anti-inflammatory role for a 35-kDa molecular weight polyethylene glycol (PEG35) in an experimental model of severe necrotizing AP. The therapeutic administration of PEG35 notably alleviated the severity of AP and protected against the associated lung inflammatory response, which is the main contributing factor to early death in patients with this condition.
To date, the treatment of AP continues to be supportive as there are no effective pharmacologic therapies available. Polyethylene glycols (PEGs) are neutral polymers widely used in biomedical applications due to its hydrophilic properties combined with a low intrinsic toxicity. In this study, we demonstrated the protective role of PEG35 in a mild form of AP.
To evaluate the effect of PEG35 in experimental models of mild acute pancreatitis in vivo and in vitro.
AP was induced by five hourly intraperitoneal injections of cerulein (50 μg/kg/bw). PEG35 was administered intraperitoneally 10 minutes before each cerulein injection in a dose of 10 mg/kg. After AP induction, samples of pancreatic tissue and blood were collected for analysis. AR42J pancreatic acinar cells were treated with increasing concentrations of PEG35 prior to exposure with tumor necrosis factor α, staurosporine or cerulein. The severity of AP was determined on the basis of plasma levels of lipase, lactate dehydrogenase activity, pancreatic edema and histological changes. To evaluate the extent of the inflammatory response, the gene expression of inflammation-associated markers was determined in the pancreas and in AR42J-treated cells. Inflammation-induced cell death was also measured in both in vivo and in vitro models of pancreatic damage through apoptosis and necrosis-related assays.
PEG35 treatment significantly improved pancreatic damage in cerulein-induced AP in rats through reduction on lipase levels and tissue edema. Furthermore, PEG35 ameliorated the inflammatory response and associated cell death in vivo and in vitro, in treated-acinar cells, by lowering inflammatory-related cytokines and iNOS gene expression, levels of apoptotic markers and the activity of lactate dehydrogenase.
PEG35 ameliorated pancreatic damage in cerulein-induced AP and cultured acinar AR42J-treated cells through the attenuation of the inflammatory response and associated cell death.
Our study provided evidence of a protective role of PEG35 in a mild form of AP suggesting that PEG35 may be a valuable option in the management of clinical AP.
This study was supported by grant from Ministerio de Ciencia e Innovación (PID2019-104130RB-I00) to Emma Folch-Puy. The authors thank Veronica Raker for revising the English text.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Tian H S-Editor: Zhang H L-Editor: A P-Editor: Ma YJ
| 1. | Gukovskaya AS, Gukovsky I, Algül H, Habtezion A. Autophagy, Inflammation, and Immune Dysfunction in the Pathogenesis of Pancreatitis. Gastroenterology. 2017;153:1212-1226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 256] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 2. | Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, Tsiotos GG, Vege SS; Acute Pancreatitis Classification Working Group. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62:102-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4932] [Cited by in RCA: 4328] [Article Influence: 360.7] [Reference Citation Analysis (45)] |
| 3. | van Santvoort HC, Bakker OJ, Bollen TL, Besselink MG, Ahmed Ali U, Schrijver AM, Boermeester MA, van Goor H, Dejong CH, van Eijck CH, van Ramshorst B, Schaapherder AF, van der Harst E, Hofker S, Nieuwenhuijs VB, Brink MA, Kruyt PM, Manusama ER, van der Schelling GP, Karsten T, Hesselink EJ, van Laarhoven CJ, Rosman C, Bosscha K, de Wit RJ, Houdijk AP, Cuesta MA, Wahab PJ, Gooszen HG; Dutch Pancreatitis Study Group. A conservative and minimally invasive approach to necrotizing pancreatitis improves outcome. Gastroenterology. 2011;141:1254-1263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 460] [Cited by in RCA: 466] [Article Influence: 33.3] [Reference Citation Analysis (2)] |
| 4. | Garber A, Frakes C, Arora Z, Chahal P. Mechanisms and Management of Acute Pancreatitis. Gastroenterol Res Pract. 2018;2018:6218798. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 5. | Sendler M, van den Brandt C, Glaubitz J, Wilden A, Golchert J, Weiss FU, Homuth G, De Freitas Chama LL, Mishra N, Mahajan UM, Bossaller L, Völker U, Bröker BM, Mayerle J, Lerch MM. NLRP3 Inflammasome Regulates Development of Systemic Inflammatory Response and Compensatory Anti-Inflammatory Response Syndromes in Mice With Acute Pancreatitis. Gastroenterology. 2020;158:253-269.e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 216] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 6. | Kylänpää ML, Repo H, Puolakkainen PA. Inflammation and immunosuppression in severe acute pancreatitis. World J Gastroenterol. 2010;16:2867-2872. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 124] [Cited by in RCA: 143] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 7. | Pasut G, Panisello A, Folch-Puy E, Lopez A, Castro-Benítez C, Calvo M, Carbonell T, García-Gil A, Adam R, Roselló-Catafau J. Polyethylene glycols: An effective strategy for limiting liver ischemia reperfusion injury. World J Gastroenterol. 2016;22:6501-6508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 49] [Cited by in RCA: 45] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 8. | Chen H, Quick E, Leung G, Hamann K, Fu Y, Cheng JX, Shi R. Polyethylene glycol protects injured neuronal mitochondria. Pathobiology. 2009;76:117-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 9. | Malhotra R, Valuckaite V, Staron ML, Theccanat T, D'Souza KM, Alverdy JC, Akhter SA. High-molecular-weight polyethylene glycol protects cardiac myocytes from hypoxia- and reoxygenation-induced cell death and preserves ventricular function. Am J Physiol Heart Circ Physiol. 2011;300:H1733-H1742. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 10. | Chiang ET, Camp SM, Dudek SM, Brown ME, Usatyuk PV, Zaborina O, Alverdy JC, Garcia JG. Protective effects of high-molecular weight polyethylene glycol (PEG) in human lung endothelial cell barrier regulation: role of actin cytoskeletal rearrangement. Microvasc Res. 2009;77:174-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 11. | Nagelschmidt M, Minor T, Saad S. Polyethylene glycol 4000 attenuates adhesion formation in rats by suppression of peritoneal inflammation and collagen incorporation. Am J Surg. 1998;176:76-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 12. | Videla S, Lugea A, Vilaseca J, Guarner F, Treserra F, Salas A, Crespo E, Medina C, Malagelada JR. Polyethylene glycol enhances colonic barrier function and ameliorates experimental colitis in rats. Int J Colorectal Dis. 2007;22:571-580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 13. | Ackland GL, Gutierrez Del Arroyo A, Yao ST, Stephens RC, Dyson A, Klein NJ, Singer M, Gourine AV. Low-molecular-weight polyethylene glycol improves survival in experimental sepsis. Crit Care Med. 2010;38:629-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 14. | Ferrero-Andrés A, Panisello-Roselló A, Serafín A, Roselló-Catafau J, Folch-Puy E. Polyethylene Glycol 35 (PEG35) Protects against Inflammation in Experimental Acute Necrotizing Pancreatitis and Associated Lung Injury. Int J Mol Sci. 2020;21:917. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 15. | Hyun JJ, Lee HS. Experimental models of pancreatitis. Clin Endosc. 2014;47:212-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 16. | Untergasser A, Cutcutache I, Koressaar T, Ye J, Faircloth BC, Remm M, Rozen SG. Primer3--new capabilities and interfaces. Nucleic Acids Res. 2012;40:e115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5674] [Cited by in RCA: 6218] [Article Influence: 478.3] [Reference Citation Analysis (0)] |
| 17. | Ding SP, Li JC, Jin C. A mouse model of severe acute pancreatitis induced with caerulein and lipopolysaccharide. World J Gastroenterol. 2003;9:584-589. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 87] [Cited by in RCA: 103] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 18. | Surbatovic M, Radakovic S. Tumor necrosis factor-α levels early in severe acute pancreatitis: is there predictive value regarding severity and outcome? J Clin Gastroenterol. 2013;47:637-643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 19. | Sendler M, Dummer A, Weiss FU, Krüger B, Wartmann T, Scharffetter-Kochanek K, van Rooijen N, Malla SR, Aghdassi A, Halangk W, Lerch MM, Mayerle J. Tumour necrosis factor α secretion induces protease activation and acinar cell necrosis in acute experimental pancreatitis in mice. Gut. 2013;62:430-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 152] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 20. | Sathyanarayan G, Garg PK, Prasad H, Tandon RK. Elevated level of interleukin-6 predicts organ failure and severe disease in patients with acute pancreatitis. J Gastroenterol Hepatol. 2007;22:550-554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 79] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 21. | Pooran N, Indaram A, Singh P, Bank S. Cytokines (IL-6, IL-8, TNF): early and reliable predictors of severe acute pancreatitis. J Clin Gastroenterol. 2003;37:263-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 122] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 22. | Silva-Vaz P, Abrantes AM, Castelo-Branco M, Gouveia A, Botelho MF, Tralhão JG. Multifactorial Scores and Biomarkers of Prognosis of Acute Pancreatitis: Applications to Research and Practice. Int J Mol Sci. 2020;21:338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 82] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 23. | Mayer J, Rau B, Gansauge F, Beger HG. Inflammatory mediators in human acute pancreatitis: clinical and pathophysiological implications. Gut. 2000;47:546-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 307] [Article Influence: 12.3] [Reference Citation Analysis (1)] |
| 24. | Rongione AJ, Kusske AM, Kwan K, Ashley SW, Reber HA, McFadden DW. Interleukin 10 reduces the severity of acute pancreatitis in rats. Gastroenterology. 1997;112:960-967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 224] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 25. | Cuzzocrea S, Mazzon E, Dugo L, Serraino I, Centorrino T, Ciccolo A, Van de Loo FA, Britti D, Caputi AP, Thiemermann C. Inducible nitric oxide synthase-deficient mice exhibit resistance to the acute pancreatitis induced by cerulein. Shock. 2002;17:416-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 65] [Article Influence: 2.8] [Reference Citation Analysis (0)] |