Published online Jul 28, 2018. doi: 10.3748/wjg.v24.i28.3071
Peer-review started: April 10, 2018
First decision: April 26, 2018
Revised: May 17, 2018
Accepted: June 27, 2018
Article in press: June 27, 2018
Published online: July 28, 2018
Processing time: 108 Days and 19.4 Hours
Helicobacter pylori (H. pylori) is present in roughly 50% of the human population worldwide and infection levels reach over 70% in developing countries. The infection has classically been associated with different gastro-intestinal diseases, but also with extra gastric diseases. Despite such associations, the bacterium frequently persists in the human host without inducing disease, and it has been suggested that H. pylori may also play a beneficial role in health. To understand how H. pylori can produce such diverse effects in the human host, several studies have focused on understanding the local and systemic effects triggered by this bacterium. One of the main mechanisms by which H. pylori is thought to damage the host is by inducing local and systemic inflammation. However, more recently, studies are beginning to focus on the effects of H. pylori and its metabolism on the gastric and intestinal microbiome. The objective of this review is to discuss how H. pylori has co-evolved with humans, how H. pylori presence is associated with positive and negative effects in human health and how inflammation and/or changes in the microbiome are associated with the observed outcomes.
Core tip: This review focuses on discussing how Helicobacter pylori (H. pylori) has co-evolved with humans, potential mechanisms that may explain both positive and negative correlations in population-based studies between H. pylori infection and the development of several diseases, as well as how inflammation and/or changes in the microbiome might be linked to the respective outcomes. Our analysis of the literature reveals that human infection by H. pylori has a longstanding history, whereby the consequences therefore are extremely complex and not always detrimental to the human host. Thus, future research should focus on determining how potentially beneficial consequences of this interaction could be promoted all the while preventing the disease-causing effects in humans.
- Citation: Bravo D, Hoare A, Soto C, Valenzuela MA, Quest AF. Helicobacter pylori in human health and disease: Mechanisms for local gastric and systemic effects. World J Gastroenterol 2018; 24(28): 3071-3089
- URL: https://www.wjgnet.com/1007-9327/full/v24/i28/3071.htm
- DOI: https://dx.doi.org/10.3748/wjg.v24.i28.3071
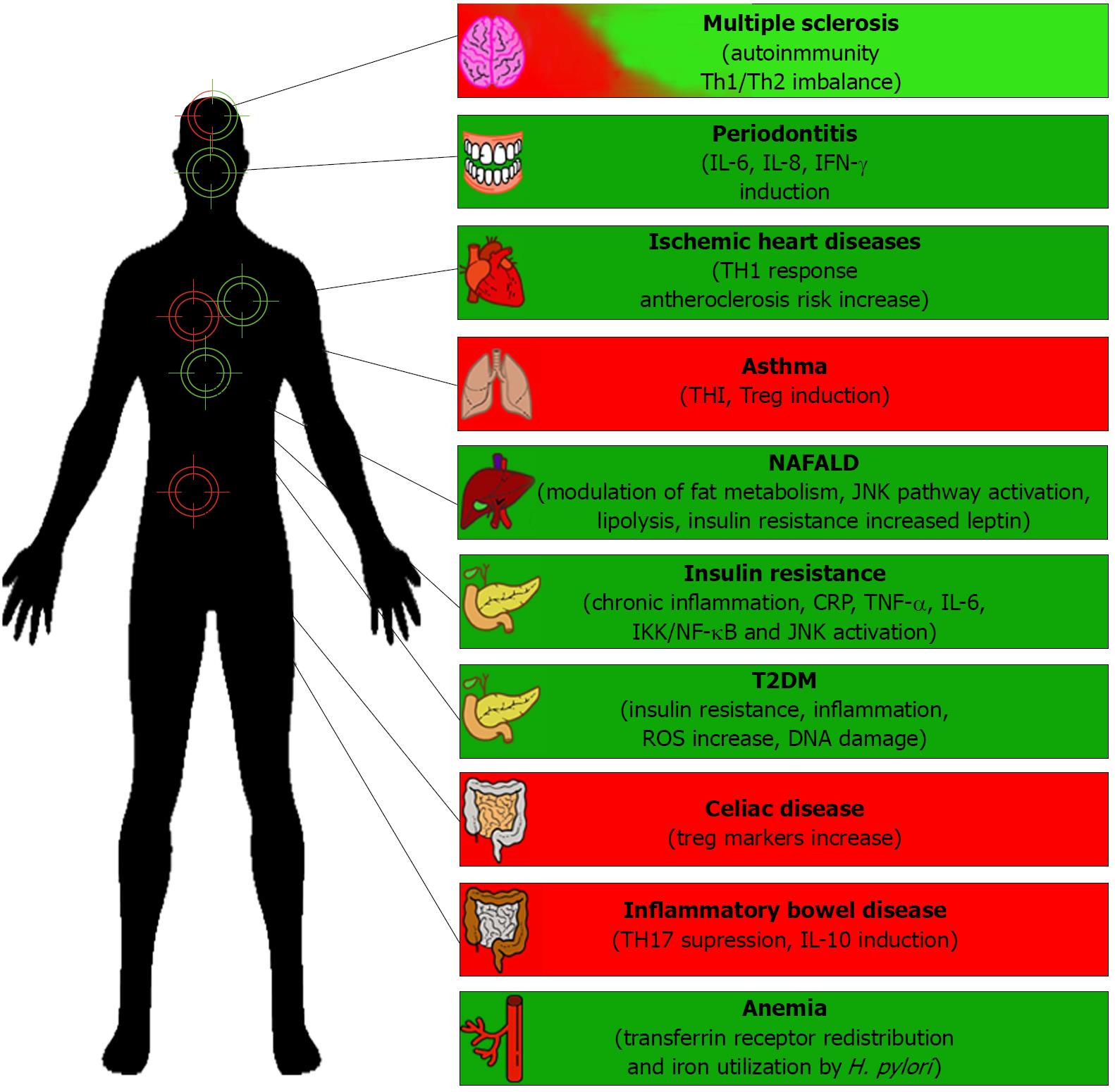
Helicobacter pylori (H. pylori) infects approximately 50% of the human population worldwide and the infection could reach more than 70% in developing countries[1,2] The consequences of infection have been associated with the development of different gastro-intestinal diseases, such as gastric ulcers, gastric cancer, mucosa-associated lymphoid tissue (MALT) lymphoma and biliary tract cancer[3]. Moreover, H. pylori infection has also been associated with extra gastric diseases, such us ischemic heart diseases[4], type 2 diabetes mellitus[5], anemia[6], adverse metabolic traits in obese subjects[7] and insulin resistance[8], to mention but a few.
Despite the existence of such associations, these diseases occur only in a small percentage of infected people, suggesting that the bacteria frequently persists in the human host without inducing any obvious signs of disease, and it has been suggested that H. pylori may also play a beneficial role in human health[9-14]. Indeed, recent studies indicate that the decreasing incidence of H. pylori in the developing world is paralleled by an increase in the incidence of allergies and autoimmune diseases[15]. Furthermore, the absence of H. pylori has been linked to elevated incidence of diseases, such us multiple sclerosis and celiac disease, among others[16-18].
Several studies have focused on understanding the local and systemic effects triggered by this bacterium in order to understand how H. pylori can produce such diverse effects in the human host. One of the best-characterized mechanisms involved in such effects is likely to be the damage to the host induced by local and systemic inflammation[19]. However, more recently, studies are beginning to focus on the effects of H. pylori and its metabolism on the gastric and intestinal microbiome[20-23]. This emerging field of interest could explain, at least in part, the wide variety of effects that are currently attributed to the presence of H. pylori in the human body.
In this review, we discuss such gastric and extra-gastric effects of H. pylori and the possible mechanisms involved.
H. pylori is a Gram-negative bacterium whose presence in the stomach of infected individuals is linked to the development of several gastric diseases, such as chronic gastritis. Although it is estimated that 50% of the world population is infected by H. pylori, only a small percentage of infected patients develop more severe pathologies, such as ulcers (10%-15%) and stomach adenocarcinomas (less than 1%)[1,2], the latter representing 15.4% of the cancers produced by infectious agents worldwide in 2012[24]. These values suggest that while relevant to the development of severe diseases, including gastric cancer, this pathogen could also play other roles in the human host.
It is now well established that H. pylori has been a highly prevalent pathogen in humans for over sixty thousand years and that infection occurs mainly in the intimate family environment or through vertical transmission[25]. These continuous infections and contact with other bacterial strains promoted the existence of a large number of mutations and genetic variability among bacteria due to horizontal transfer of information[26-28]. The emerging differences have been characterized particularly with respect to geographic distribution and such studies have revealed that the observed genomic alterations allow H. pylori to survive in different microenvironments[29,30]. Moreover, these genetic modifications are thought to have lead to the emergence of less virulent strains, which may explain the low percentage of patients affected with serious pathologies, such as adenocarcinomas[31].
The events during human evolution associated with initial acquisition H. pylori are thought to have been the development of agrarian practices, as evidenced by the presence of DNA remnants in the H. pylori genome, such as the vir genes of Agrobacterium tumifaciens[32]. Also population migration is likely to have contributed to the acquisition of genes or genomic islands important for H. pylori virulence. One of them is the cagPAI genomic island, where cagA is one of the most important virulence genes associated with an increase in the activation of pro-inflammatory pathways and the production of pro-inflammatory cytokines in the stomach mucosa[33,34]. According to genomic analyses of H. pylori, European colonization trips to South America may have contributed to the acquisition of cagA by indigenous people living in the Andes, who possessed H. pylori without a functional cagA gene[30,35]. Moreover, other studies analyzing South American populations, such as the Colombians, have determined that the mountain people with greater similarity to the native people of that region have a higher incidence of gastric cancer compared with residents of coastal towns, who were more strongly influenced by the colonization of African migrant populations. Currently, this sector of the population has a low incidence of gastric cancer associated with H. pylori infection[36,37].
However, it has been observed that although in certain populations positive CagA strains are more numerous (as in parts of Eastern Asia), there are specific characteristics that make it unlikely for them to spread to the rest of the world, such as Western countries, with fewer positive CagA strains. These observations suggest that “fitness traits” exist, which aid in the survival of the bacteria in different hosts, in addition to other well-known factors, like difference in the lifestyles, socioeconomic levels and diet of the host population[38]. Japan, for example, has the highest rate of gastric cancer worldwide, associated with the highest presence of CagA (57%); however a lower seroprevalence is observed compared to other populations[39]. In contrast, despite the high prevalence of H. pylori in India, a low rate of gastric cancer is registered (known as the “Indian Enigma”). One of the main hypotheses seeking to explain this enigma is that the higher rate of enteric infections in more poorly developed countries could boost the immune system and limit the consequences of H. pylori infection. In addition, the high diet content of peppers, which represent an important ingredient in the Indian diet, may protect against H. pylori infection[40]. In conjunction, these examples support the hypothesis that a variety of factors contribute to the fitness of H. pylori in different human host populations.
Therefore, bacterial and host fitness are very relevant in H. pylori infection. In particular, host genetics likely affect the progression of pathologies associated with H. pylori infection. Indeed, specific polymorphisms in genes coding for cytokines, such as IL-1β, IL-8, IL-10 and TNF-α, are associated with an increase in the pro-inflammatory responses, greater colonization and infection, as well as an increased risk of gastric cancer[41-45]. Also, polymorphisms in innate immunity genes, such as the toll-like receptor 4 (TLR4), are relevant because TLR4 is implicated as a receptor responsible for H. pylori induced signaling in gastric epithelial cells. Moreover, epigenetic changes due to hypermethylation in the promoter regions of tumor suppressor genes, such as LOX, HAND1 and APC, and the alteration as well as deregulation of microRNAs (miRs) are associated with a higher prevalence of gastric cancer following H. pylori infection[46,47].
Despite clearly representing a human pathogen, evidence is available suggesting that this bacterium could also be considered a commensal bacteria in the human host. This notion is supported by the simple observation that the bacteria is present in so many individuals, yet generates relatively few symptoms or pathologies. This raises the issue as to whether to refer to H. pylori as a commensal or pathogen, because pathologies are likely not only to be associated with specific traits of H. pylori, but also with a series of specific conditions in the human host.
H. pylori has been found as part of the normal oral microbiota and part of the microbiota of the stomach in the absence of inflammation[48,49]. In addition, it has been reported that H. pylori infection is not associated with the onset of the gastric cancer, but rather with its recurrence and chronicity[50]. Moreover, the presence of H. pylori in the stomach microbiota may result in changes in the normal microbiota[21,23].
In this context, it is worth mentioning that several studies have attributed positive effects to H. pylori infection. These include the suppression of bacteria that cause tuberculosis (Mycobacterium tuberculosis), protection against asthma, Crohn’s disease, esophageal reflux, diarrheal diseases, as well as esophageal cancer[9-14]. This controversy has led to the discussion whether eradication of H. pylori is recommendable to help restore the host’s health status, or if alternative strategies should be developed to control virulence of the bacteria, thereby avoiding the appearance of ulcers and adenocarcinomas without eliminating the positive effects that this bacterium may have [51]. With this in mind, it is not surprising that H. pylori is so widely studied and considered a relevant target in many therapies.
H. pylori infection is inversely associated with the development of some diseases, suggesting that the presence of these bacteria may also be beneficial to the host, as is the case for reducing the risk of obesity, childhood asthma, inflammatory bowel disease and celiac disease among others. In some cases the data available strongly support the notion that H. pylori presence is beneficial, while for others convincing data still remains at large (Figure 1 and Table 1).
| Disease | Association | Reference | Type/model of study | Sample size | Statistical analysis | H. pylori detection | Diagnosis of the pathology |
| Asthma | NA | Holster et al[53], 2012 | Cohort study | 545 childrens | Chi-square and t-tests. Univariate and multiple logistic regression analysesa | Serum anti-H. pylori inmunoglobulin G, and CagA by ELISA | Positive diagnosed asthma by a medical questionnaire |
| Positive | Den Hollander[54], 2016 | Cohort study | 3797 childrens | Chi-square test. Multivariate logistic regression analysis. Odd ratios 95% CIb | inmunoglobulin G levels in serum | Positive diagnosed asthma by a medical questionnaire | |
| Inverse | Chen and Blaser[10], 2007 | Cohort study | 7663 adults | Unconditional logistic regression models. Odd ratios 95%CIa | inmunoglobulin G levels in serum | Positive diagnosed asthma by a medical questionnaire | |
| Chen and Blaser[55], 2008 | Case-control study | 7412 individuals | Chi-square and t-testsb | Wampole ELISA | Positive diagnosed asthma by a medical questionnaire | ||
| Sommer et al[57], 1998 | Ex vivo study. Isolated T cells from gastric biopsy | Biopsies from 30 patients | Not mentioned | Histological detection | Endoscopic examination | ||
| Bamford et al[56], 1998 | Ex vivo study. Isolated T cells from gastric biopsy | n patients = 5, n control = 3 | t-testa | Rapid urease test (RUT) or histopathology | - | ||
| Oertli et al[59], 2013 | In vivo study. C57BL/6 mice | 60 mice | Chi-square. Mann Whitney U-test and Kruskal-Wallis testa | CFU from homogenised tissues | - | ||
| De la Pena-Ponce et al[60], 2017 | In vitro study. Airway epithelial cells | Between n=3 to n=10 | One way ANOVAa | CagA detection by western blot | - | ||
| Inflammatory Bowel Disease | Inverse | Higgins et al[61], 2011 | Meta-analysis and In vivo study. C57BL/6 mice | Between n=3 to n=9 | Odd ratios 95%CI. P-value < 0.1. One way ANOVA and t-testa | Not mentioned | - |
| Lord et al[62], 2018 | Cross-sectional study | 704 individuals | Odd ratios 95%CIb | Not mentioned | Not mentioned | ||
| Castano-Rodriguez et al[63], 2017 | Meta analysis | 6130 patients and 74659 controls | Chi-square, t-test, fixed effect model and odd ratios 95%CIb | Histology, culture, rapid urease test, serology and/or urea breath test (UBT) | Not mentioned but differentiated among Crohn's disease, ulcerative colitis, IBD, and unclassified | ||
| Celiac Disease | Inverse | Lebwohl et al[65], 2013 | Cross-sectional study | 136179 individuals | Odd ratios and 95%CIb | Polyclonal inmunochemical stain | Duodenal and gastric biopsies |
| Narang et al[18], 2017 | Cross-sectional study | 324 childrens | Chi-square test or Fisher exact test, and odd ratio and 95%CIa | Giemsa staining and rapid urease test (RUT) | Serum levels of inmunoglobulin A-tissue transglutaminase antibodies (IgA-tTG> 18 U/mL = CD +). Further analysis by upper gastrointestinal endoscopy for biopsies to confirm | ||
| Lucero et al[17], 2017 | Case-control study | 66 patients and 50 controls | Chi-square test or Fisher exact test, and odd ratio and 95%CIb | Rapid urease test (RUT), histological evaluation and PCR | Duodenal histopathology and inmunoglobulin A-tissue transglutaminase (IgA-tTG) serology | ||
| Multiple Sclerosis | Inverse | Yao et al[67], 2016 | Meta analysis | 1553 patients and 1253 controls | Chi-square test, odd ratio and 95%CIb | ELISA, immunofluorescence and latex agglutination tests | Not mentioned |
| Jaruvongvanich.[68], 2016 | Meta analysis | 1902 individuals | Chi-square test, odd ratios, multivariant models and random-effect modelsa | Urea breath test (UBT), rapid urease test, PCR and ELISA | Diagnosed by neurologist using the McDonald criteria (based on clinical presentations, finding on magnetic resonance imaging and cerebrospinal fluid profile) | ||
| Positive | Efthymiou et al[16], 2016 | Cohort study | 129 patients, 49 controls | Two-tailed t-testa | Serum anti-H. pylori, anti-VacA, anti-CagA, anti-Hsp60 ELISA | Not mentioned, but relapsing remitting MS (RRMS) and secondary progressive MS (SPMS) are differentiated | |
| Ischemic Heart Diseases | Positive | Liu et al[4], 2015 | Meta analysis | 5829 patients and ~16000 controls | Fixed and random effect models. Odd ratios and 95%CI. P-value = 0.06 | Not mentioned | Medical records |
| Shmuely et al[3], 2014 | Cohort study | 173 patients and 127 controls | Multivariate analysis. Odd ratios 95%CI. t-test and ANOVAa | Serum anti-H. pylori inmunoglobulin G, and CagA by ELISA | Myocardial perfusion imaging in patients with angina symptoms, chest pain, suspected CAD, cardiac related symptoms or risk stratications in patients with known CAD | ||
| Huang et al[72], 2014 | Retrospective cohort study | 17332 patients and 69328 controls | Chi-square and t-testsa | Not mentioned | Isquemic stroke | ||
| Anemia | Positive | Xu et al[6], 2017 | Retrospective study | 17791 individuals 7804 Hp positive | Chi-square and t-tests. Odd ratios 95%CIa | Serum anti-H. pylori inmunoglobulin G and inmunoglobulin M ELISA | Using haemoglobin level |
| Flores et al[76], 2015 | In vitro study. AGS human gastric adenocarcinoma cell line | n=7 experiments | One way ANOVA or non-parametric t-testa | CFU from homogenised cells | - | ||
| Flores et al[75], 2017 | In vitro study. AGS human gastric adenocarcinoma cell line | n=3 experiments | One way ANOVA or non-parametric t-test a | CFU from homogenised cells | - | ||
| Kato et al[77], 2017 | In vitro study. Isolated H. pylori strains from patients and controls (whole genome sequencing) | 4 patients and 4 controls | t-testa | Biopsy directly inoculated in growth medium | Measuring serum iron and ferritin | ||
| NAFLD | Positive | Chen et al[7], 2017 | Cohort study | 2263 individuals | Chi-square and t-testsa | 13C-labeled urea breath test (UBT) | Using the NALFD criteria suggested by the Chinese Liver Disease Association and Clinical Diagnosis Standards |
| Huang et al[78], 2009 | In vivo study. C57BL/6 mice | n = 20 | Chi-square testa | Gram staining, PCR and urease/catalase reactions | Histopathology and inmunochemical analysis | ||
| Insulin resistance | Positive | Aydemir et al[82], 2005 | Cross-sectional Study | 63 patients | t-testa | Giemsa staining | HOMA-IR |
| Gunji et al[83], 2009 | Cross-sectional Study | 1107 participants (1008 IR- y 99 IR+) | Chi-square and t testsa | Serum anti-H. pylori inmunoglobulin G ELISA | HOMA-IR | ||
| Chen et al[84], 2015 | Cohort study | 811 individuals | Chi-square or Fisher exact testa | Serum anti-H. pylori inmunoglobulin G ELISA | HOMA-IR | ||
| Polyzos et al[86], 2011 | Meta analysis | 2120 participants | t-testa | Gastric mucosa histologic examination for H. pylori presence, gastric mucosa rapid urease test (CLO test), serum H. pylori-specific immunoglobulin G antibody concentration (ELISA), serum H. pylori-specific immunoglobulin G antibody concentration (chemiluminescence) | HOMA-IR | ||
| Yildrim et al[89], 2016 | Cohort study | 41 patients and 27 controls | t-testa | 13C-labeled urea breath test (UBT) and gastroscopy | HOMA-IR | ||
| Upala et al[8], 2016 | Meta analysis | 27544 participants | Chi-square. Odd ratio 95%CIa | Urea breath test (UBT), rapid urease test (RUT), PCR and ELISA | HOMA-IR | ||
| Type 2 Diabetes Mellitus | NA | Anastasios et al[93], 2002 | Cross-sectional study | 67 patients and 105 controls | Chi-square. P-value < 0.05 | Giemsa staining | Previously diagnosed patients |
| Positive | Li et al[5], 2017 | Meta analysis | 57397 participants | Fixed and random effect models. Odd ratios and 95%CIa | 13C or 14C urea breath test, stool antigen test, anti-H. pylori antibody, rapid urease test, histology or biopsy, culture | Not mentioned | |
| Bener et al[91], 2007 | Case-control study | 210 patients | t-testa | Serum anti-H. pylori inmunoglobulin G and inmunoglobulin A ELISA | Using venous blood glucose values or currently taking diabetic medication | ||
| Devrajani et al[92], 2010 | Case-control study | 74 patients and 74 controls | Chi-squarea | Stool H. pylori-antigen detection by Enzyme immunoassay (EIA) | Fasting blood sugar (FBS) level, random blood sugar (RBS) level and hemoglobin A1c | ||
| Aslan et al[95], 2006 | Cross-sectional Study | 103 patients | t-testa | Rapid urease test (RUT) and histopathologic examination | Serum glucose concentration and serum insulin levels | ||
| Nasif et al[96], 2016 | Cross-sectional Study | 100 patients | t-test, Manne Whitney U-testa | Serum anti-H. pylori IgG ELISA | Postprandial glucose level, glycated hemoglobin (HbA1c) and body mass index (BMI). Serum 8-OHdG and Ox-LDL | ||
| Periodontitis | Positive | Sujatha et al[106], 2015 | Cohort study | 40 patients | Fisher exact testa | Rapid urease test, histopathological examination | Periodontal examination |
| Pei et al[192], 2015 | Cross-sectional study | 70 patients ans 70 controls | The ratios were compared using × 2 test and × 2 statistics was adjusted. t-testa | PCR for urease C gene | Probing depth (PD), plaque index (PI) and bleeding index (BI) | ||
| Hu et al[107], 2016 | In vitro study. THP-1 cells | 28 samples from 14 patients | One way ANOVAa | real-time PCR | Probing depth (PD), plaque index (PLI), bleeding index (BI), attachment loss (AL) |
Asthma: Asthma is characterized by a chronic hyper-responsiveness to specific and non-specific stimuli that favor obstruction of the airways, characterized by increased serum immunoglobulin E (IgE) levels combined with infiltration of the lungs by eosinophils, mast cells and activated CD4+ T-cells, a process orchestrated by effector T-helper 2 cells, implying the participation of the cytokines IL-4, IL-13, IL-5 and IL-9 in these events[52]. Several studies have proposed an inverse association between the presence of H. pylori infection and asthma, although this association is still controversial. While Holster et al[53] showed in a cohort of 545 children that there are no significant differences in H. pylori prevalence between children with asthma (7.1% vs 9.4%), others have shown either positive or negative effects. Significantly higher prevalence of asthma was reported in H. pylori positive compared to H. pylori negative children, based on a cohort study of 3759 children[54]. In contrast, studies involving more than 7000 adults[10,55] showed that H. pylori presence and also the CagA protein were inversely correlated with the development of asthma. More recently, Miftahussurur et al[15] reviewed several studies, surveys, cohort studies and meta-analyses in different European counties and in the United States of America (USA), involving a large number of persons. They concluded that there is a significant but weak inverse correlation between H. pylori infection, allergies and asthma, suggesting that H. pylori infection may have a beneficial protective role against development of these diseases [15].
The proposed mechanism involves the bacterial induction of naïve T cells, mainly in T helper 1 (Th1) rather than helper 2 (Th2) subsets[56,57]. On the other hand, it has also been observed that T-regulatory (Treg) cells are increased in the gastric mucosa of H. pylori- infected humans[58]. Moreover, the H. pylori virulence factors γ-glutamyl transpeptidase and VacA, induced Treg cells in the mouse gastric mucosa, resulting in the development of tolerance and a reduction in allergic responses[59]. Also, in a recent study using infant and adult airway epithelial cells infected with H. pylori, IL-8 synthesis increased 4-fold in infant versus adult cultures, suggesting that the infant epithelium elicits a higher immune response than the adult tissue. This mechanism is mediated by the H. pylori type IV secretion system and stimulation of the p38 MAP kinase pathway[60], and VacA was found to potentially also contribute to this mechanism.
Inflammatory bowel disease: Inflammatory bowel disease (IBD) is a chronic inflammatory intestinal disease that develops as the consequence of a deregulated immune response. Interestingly, several studies have sought to establish a relationship between H. pylori infection and IBD. Higgins et al[61] demonstrated the effect of gastric H. pylori colonization on a distant bacterial-host immune system interaction in an experimental model of colitis. Also, H. pylori was shown to suppress the Th17 response to S. Typhimurium infection, but did not alter the Th2 or Treg response. Moreover, the authors showed that the co-infection by H. pylori/S. Typhimurium decreases inflammation in both the cecum and the stomach and that H. pylori infection induces IL-10 in the mesenteric lymph nodes, suggesting an extra-gastric mechanism for immunomodulation. Also, IBD protection is suggested to be linked to the cagA-positive status of the strain[62]. More recently, a meta-analysis performed by Castaño-Rodríguez et al[63] also revealed that H. pylori may exert an immunomodulatory effect and thereby favor the development of IBD.
Celiac disease: Celiac disease (CD) is an autoimmune disease whose prevalence in the USA has increased up to 4-fold in the past 50 years[64]. A cross-sectional study of patients who underwent esophago-gastroduodendoscopy with analysis of gastric and duodenal biopsies during a 4.5-year period showed that H. pylori prevalence was lower in patients with CD (4.4%) than in those without CD[65], indicating an inverse association between CD and H. pylori infection.
In the same context, a recent study including 324 children with confirmed CD, the H. pylori prevalence was compared with a reference group of non-celiac children referred for endoscopy. The results showed that the prevalence of H. pylori in patients without CD was significantly higher[18], indicating that CD and gastric H. pylori infection are inversely correlated.
The mechanistic link between H. pylori infection and CD remains to be elucidated. However, recently, Lucero et al[17] demonstrated that infection by CagA positive H. pylori induced Treg markers and that this may be protective against CD progression.
Multiple sclerosis: Emerging evidence suggests that H. pylori may also be inversely associated with neurodegenerative diseases. In this context, several studies have sought to establish an association between H. pylori infection and multiple sclerosis (MS), a chronic autoimmune, inflammatory and neurodegenerative disorder of the central nervous system[66].
In a meta-analysis of nine studies involving 2806 cases (1553 patients with MS and 1253 controls), Yao et al[67] found that the prevalence of H. pylori infection in MS patients was lower than that in control groups. Another meta-analysis of six observational studies involving 1902 participants showed also a statistically significant lower prevalence of H. pylori infection in patients with MS[68].
In spite of these studies, Efthymiou et al[16] in a cohort study of 129 patients and 49 controls, showed that anti-H. pylori antibody titers were higher in 129 MS patients than in 48 healthy controls. Additionally, anti-H. pylori hsp 60 seropositivity correlated with age at disease onset, suggesting a possible role of this factor in the pathogenesis of MS[16].
In this context, it has been proposed that the inflammatory mediators induced by H. pylori infection might impact on the nervous system and induce damage[69]. Additionally, circulating pro-inflammatory cytokines, such as IL-17, and reactive oxygen species (ROS) can reach the CNS and induce damage[70]. Moreover, recent reports implicate the Galectin-3 receptor, a leptin receptor that is stimulated by H. pylori, in inducing a pro-inflammatory response via TLRs. Activation of these receptors in the CNS, triggers an inflammatory response mediated by interferon (IFN)-γ and TNF-α that is associated with neuro-pathophysiological changes [70].
Despite these observations suggesting a protective role for H. pylori against several diseases, there is a large body of literature associating H. pylori infection with the development of gastric diseases, such as peptic ulcer diseases, gastric adenocarcinoma, MALT lymphoma and biliary tract[71]. Moreover, the positive correlations between H. pylori and disease conditions have also been noted for extra-intestinal diseases, such as dermatological diseases, heart diseases, obesity, anemia, insulin resistance and non-alcoholic fatty liver disease, among others (Figure 1 and Table 1). In most of these cases, disease development is associated with the chronic inflammatory response that the infection triggers in the host.
Ischemic heart diseases:H. pylori has been suggested to contribute to the development of coronary heart diseases (CAD). In a meta-analysis of 26 studies, including more than 20000 patients, Liu et al[4] observed a significant association between H. pylori infection and the risk of myocardial infarction. In the same context, Shmuely et al[3] in a cohort study of 173 patients and 127 controls, observed that H. pylori infection was significantly higher in CAD-positive patients than in CAD-negative subjects, suggesting a positive correlation between H. pylori seropositivity and CAD. In addition, in a retrospective cohort study by Huang et al[72], involving 17332 patients with H. pylori infection and 69328 randomly selected age- and gender-matched controls, a more specific association between chronic H. pylori infection and ischemic stroke was observed since patients diagnosed with H. pylori infection exhibited a higher incidence rate of ischemic stroke. Despite such observations suggesting that the presence of H. pylori favors the development of heart disease, the mechanisms involved remain to be determined. However, because chronic inflammation is believed to be associated with an increased risk of atherosclerosis[73], this may in an indirect manner explain the augmented risk of heart disease associated with H. pylori infection.
Anemia: An association between H. pylori infection and iron deficiency was proposed based on studies showing that for individuals with idiopathic iron deficiency anemia of unknown origin and no evidence of bleeding due to lesions, iron deficiency anemia was no longer observed in any of the follow-up examinations following eradication of H. pylori[74]. In a recent retrospective cohort study, Xu et al[6] evaluated the relationship between anemia and H. pylori infection in 17791 subjects. They observed a higher probability for anemia in H. pylori positive populations coincident with lower hemoglobin levels.
Recent studies suggest that the mechanism involves changes in the intracellular iron distribution associated with the uptake and trafficking of H. pylori through the cells[75], since H. pylori uptake by gastric cells is associated with an increase in total cellular iron content and its homeostasis depends on the transferrin receptor[76]. Indeed, a study by Flores et al[75] showed that H. pylori infection is associated with an increase in the total intracellular iron levels, redistribution of the transferrin receptor from the cell cytosol to the cell surface, and increased levels of ferritin. Moreover, Kato et al[77] showed that the SabA gene is highly expressed in bacterial isolates from iron deficient anemia patients, suggesting a role for this virulence factor in the development of anemia.
Non-alcoholic fatty liver disease: Non-alcoholic fatty acid disease (NAFLD) has also been suggested to be associated with H. pylori infection. In this context, a large number of reports, including cross-sectional studies, case reports and randomized-controlled studies have revealed a strong association between NAFLD and H. pylori infection[7]. In addition, it has been demonstrated in an animal model of H. pylori infection that the orally inoculated bacterium can reach the liver and cause hepatitis[78].
H. pylori infection may induce NAFLD by producing chronic systemic inflammation, increasing the levels of inflammatory cytokines, such as IL-6 and TNF-α, and activating NF-κB pathway, which induce insulin resistance (IR)[79]. The mechanisms of the pathogenesis of H. pylori-related inflammation in NAFLD involve directly reducing hepatocyte glycogen levels via a JNK signaling pathway[80], which in turn can down-regulate the expression of key genes involved in glucose metabolism and accelerating lipolysis[81], thereby contributing indirectly to the development of IR. In addition, H. pylori may also induce white adipose tissue to release leptin, and then promote liver stearoyl- CoA desaturase, favoring the accumulation of fat deposits in the liver tissue[79].
Insulin resistance: Several studies have revealed a strong association between H. pylori and IR. In 2005, Aydemir et al[82] confirmed the existence of a positive correlation between chronic H. pylori infection and IR by showing that the homeostasis model assessment (HOMA-IR) of H. pylori positive subjects was significantly higher compared with H. pylori negative individuals. In another large cross-sectional study including 1,107 subjects, H. pylori seropositivity was significantly higher for patients with IR (HOMA-IR ≥ 2.5)[83]. Subsequent studies also confirmed the causal relationship between H. pylori and IR[84-86].
Mechanistically, H. pylori-induced IR may be caused indirectly by chronic inflammation or directly by activating certain signaling pathways. Several reports have confirmed that chronic inflammation is important for IR onset[87] and H. pylori-mediated chronic inflammation may increase the expression of C-reactive protein, TNF-α, and IL-6[88,89]. These cytokines activate IKK/NF-κB and JNK pathways, which may trigger IR by increasing insulin receptor phosphorylation on serine[81] or by inhibition of insulin receptor substrate-1 phosphorylation on tyrosine residues[90].
Despite such evidence, a recent systematic review revealed that H. pylori eradication does not improve insulin resistance, but may increase body weight (BW) and the body mass index (BMI), suggesting that further studies are needed to clarify the effect of H. pylori eradication on metabolism [8].
Type 2 diabetes mellitus: The association between Type 2 diabetes mellitus (T2DM) and H. pylori infection is controversial. Some studies indicate that the prevalence of H. pylori is higher in diabetic compared with non-diabetic patients[91,92], while others indicate that there are no differences between those groups[93]. Nevertheless, more recently, He et al[94] discussed the possibility that this controversy is likely due to inconsistencies in the methods used to define H. pylori positivity, diabetic status and the reduced sample sizes, among other limitations. More recently, a meta-analysis including 57397 individuals showed that there is significantly higher prevalence of H. pylori infection in diabetic type 2 patients as compared with healthy individuals[5].
The possible mechanisms linking H. pylori to diabetes include alterations in IR signaling, inflammation, accumulation of ROS and oxidative DNA damage in the gastric mucosa. It has been reported that ROS levels and oxidative DNA damage increase due to neutrophil infiltration in H. pylori-infected patients[95]. Moreover, a recent study performed in 100 patients showed increased serum levels of oxidative DNA damage (8-OHdG) and oxidized low-density lipoprotein in T2DM patients positive for H. pylori infection[96].
Periodontitis: Periodontitis is characterized by the accumulation of bacterial plaque at the gingival margin, which induces an inflammatory response that leads to destruction of the connective tissue attachments to teeth, alveolar bone resorption and tooth loss[97]. The recent Global Burden of Disease Study (1990-2010) indicates that severe periodontitis is the 6th most prevalent disease worldwide, with an overall prevalence of 11.2%, although mild forms of this disease may reach over 90% of the population in developing countries[98]. Remarkably, periodontitis has also been linked to an increased risk in developing atherosclerosis, diabetes, rheumatoid arthritis and cancer[99-103]
Moreover, the oral cavity might represent a reservoir for H. pylori[104]. In a recent cross-sectional study that included 70 patients and 70 controls it was reported that the presence of H. pylori in dental plaque correlates with periodontitis and that the correlation appears to be better in severe forms of the disease [105]. Additionally, a study involving 40 patients (32 periodontitis and 8 controls) showed that periodontal disease positively correlates with gastric and oral H. pylori (P < 0.005)[106]. In this study, in spite of the small number of controls, it was shown that 70% of periodontitis patients have biopsies positive for H. pylori. Also, it is important to mention that 81% of periodontitis patients were positive for H. pylori in oral plaques.
Recently, Hu et al[107] demonstrated that H. pylori infected patients have worse periodontal parameters than non-infected individuals, suggesting that infection correlates the progression of the disease. This study also showed that the presence of periodontitis-associated bacteria was significantly higher in subjects with H. pylori infection than those without H. pylori infection. Moreover, the expression of inflammatory molecules, such as IL-8, IL-6 and IFN-γ significantly increased after H. pylori infection. Interestingly, those effects were associated with the presence of CagA[107].
H. pylori infection triggers several adaptative cellular mechanisms in host cells that may favor gastric cancer development and progression[108]. However, whether the disease develops or not and the final outcome are thought to depend largely on the extent of inflammation promoted by the bacteria in the host during the pre-neoplastic process[109]. Indeed, chronic inflammation is a common causative event associated with the development of several types of cancer[110,111] and, as was demonstrated in animal models, H. pylori infection is considered the major factor responsible for gastric epithelial damage and deregulation of signaling leading to irreversible epigenetic changes in the gastric mucosa, a consistent hallmark observed during the gastric carcinogenic cascade[19,109]. Initially, H. pylori-related studies focused on identifying the virulence factors implicated in these processes. Those factors epidemiologically associated with a higher risk of developing gastric cancer were tested in vitro and shown to induce signaling pathways associated with exacerbated inflammatory responses. For example, strains harboring particular vacA and cagA allele variants were found to induce elevated inflammatory responses in infected cells[112]. However, this seemingly simple scenario rapidly transited to a more complex one when epidemiological data revealed that in certain ubiquitously infected populations no correlation with elevated gastric cancer incidence was detected, as is the case for the so-called African enigma[113] where only a minor percentage of infected patients progress to develop cancer. To date, experimental studies have clarified that final disease outcome depends not only on the contribution of certain bacterial virulence factors, but also on host susceptibility, diet and environmental factors[114]. Here, it is important to mention that co-existence with the bacteria is not only to be viewed negatively (see previous and following chapters), bearing in mind that since the prevalence of the infection has declined in developed countries over the last decades, several disorders have emerged as a consequence of the lack of exposure to H. pylori[115]. Human beings have co-evolved with the bacterium and gastric as well as extra-gastric physiology has been conditioned to such association[116]. In order to persist in the gastric niche, H. pylori have evolved mechanisms necessary to evade and to attenuate the innate and adaptive immune systems by several mechanisms, including those implicated in evasion of recognition by pattern recognition receptors, inhibition of phagocytic killing, inhibition of killing by ROS and nitric oxide, among others[117]. Particularly, antigenic phase variation, modulation of adhesion molecules, immune inhibition by VacA protein and lipopolysaccharide (LPS) have been widely described[118]. Moreover, additional mitigating local mechanisms mediated by bacterial enzymes exist. For instance, it was recently reported that the expression of a cholesterol-α-glucosyltransferase reduced cholesterol levels in gastric epithelial cells, blocking IFN-γ signaling, a classical Th1 cytokine[119]. Notably, this enzyme is present in most Helicobacter species and cholesteryl α-glucosides are also involved in resistance to antibiotics[120], interference with phagosome trafficking[121], H. pylori type IV secretion system function[122] and immune evasion by inhibiting T-cell activation[123]. On the other hand, H. pylori superoxide dismutase (SOD) has been shown to suppress the production of pro-inflammatory cytokines during in vivo infection by reducing oxidative stress. Thus, SOD from H. pylori can inhibit the production of pro-inflammatory cytokines during in vivo infection[124]
Additionally, H. pylori deregulates adaptive immune responses by interfering with antigen presentation and modulation of T-cell responses[117,118]. Eradication of H. pylori has revealed the importance of this modulation of the immune response in preventing the development of extra-gastric immune and inflammatory disorders, such as gastroesophageal reflux disease, childhood asthma and allergy, as well as metabolic disorders[15,52,54]. Although in most of the cases correlations are derived from cross-sectional studies, the most experimentally validated preventive association is the appearance of childhood asthma[115,125]. Moreover, innate immune responses are also involved, given that bronchial epithelial cells, mast cells, basophils, natural killer T cells and dendritic cells (DC) also produce inflammatory mediators[52]. This harmful effector response is modulated by CD24+CD25+ regulatory cells (Treg) present in the lung, which secrete anti-inflammatory cytokines, such as IL-10 and transforming growth factor beta (TGF-β), preventing or modulating the Th2 responses to allergens[126]. Tregs of healthy individuals shift allergen-specific immune responses toward tolerance, thereby preventing the development of asthma and other allergic disorders[52]. Also in animal models of infection, the importance of dendritic cells in H. pylori-specific adaptive immune responses was noted. Particularly tolerance induction[52,127], Treg skewing and Th17 suppression observed in mice occurred in a cagA- and vacA-independent manner[128]. Chronic exposure to H. pylori impairs dendritic cell function and inhibits Th1 development[129]. Also, H. pylori-mediated protection was linked to IL-10-secretion by peripheral blood Treg cells[130].
The importance of these immune cells has been validated in experimental animal models of infection or induced asthma. For instance, blocking CD24+CD25+ Treg cells by a CD25-neutralizing antibody abrogated Treg cell tolerance promoted by H. pylori infection and enhanced pulmonary inflammation following albumin induced asthma[131]. Also, KO animals for IL-10, TGF-β or animals depleted of Tregs, develop gastritis to an elevated extent in response to H. pylori infection. Although these animals are able to clear the infection, pre-neoplastic lesions develop since Th1 responses predominate[52]. Together, these results indicate that tolerance rather than immunity protects against H. pylori-induced gastric pre-neoplastic lesions[132]. Accordingly, decreased Treg cell function is associated with an increase in peptic ulcer development upon H. pylori infection[133].
A currently unresolved question is how H. pylori promotes tolerance in distant organs such as the lung? A recent study showed that systemic and mucosal pre-administration of recombinant neutrophil-activating protein prevented ovalbumin-induced allergic asthma in mice, indicating that secreted virulence factors may be responsible[134]. What determines the balance between tolerance or elimination of an exacerbated Th1 type chronic inflammation could be explained in part by host hyperreactivity to allergens or bacterial infection. In this respect, pro-inflammatory cytokine polymorphisms are generally thought to participate in the genesis of gastric and other types of cancer[109] and interleukin family cytokines, like IL-1β and IL-18, have emerged as central mediators of mucosal inflammation[135]. On the other hand, virulence factors can determine the type of response. For instance, blocking the TLR4 in a mouse model using a specific antibody prior to H. pylori infection, was shown to reduce the number of T-cell effectors (Th1 and Th17) and diminish the immune response[136]. In agreement with this observation, activation of TLR4 signaling was reported to be associated with gastric cancer progression by inducing mitochondrial ROS production[137]. Moreover, type I H. pylori (cag PAI+ and vacuolating toxin A+, VacA+) LPS exhibited a stimulatory effect on TLR4 signaling followed by mitogen oxidase 1 activation in cultured gastric pit cells through the lipid A portion of LPS[138]. In a similar study, LPS from some H. pylori strains were shown to act as TLR4 antagonists, which may contribute to more beneficial clinical outcomes of H. pylori infection in host individuals[139]. Additionally, H. pylori LPS from type I, but not type II strains, promotes cytotoxicity in cultured gastric mucosal cells[140]. Conversely, TLR2 mediates H. pylori-induced tolerogenic immune responses[141] and TLR9 signaling has anti-inflammatory effects during the early phase of H. pylori-induced gastritis in mice[142]. Also, additional virulence factors have been implicated in immune response suppression or tolerance, such as the suppression of dendritic cells by OipA in vitro[143], promotion of immune tolerance by VacA-mediated inhibition of T-cell proliferation and antigen-presentation[59]. Also, bacterial gamma-glutamyl transpeptidase[59] and outer membrane vesicles inhibit T-cell responses[144,145]. Furthermore, H. pylori infection has been associated inversely with IBD. Experimental immuno-regulatory properties of the H. pylori genome and particularly the immuno-regulatory sequence TTTAGGG was demonstrated to down-regulate dendritic cell-mediated production of pro-inflammatory cytokines both in an in vitro and in vivo model[146].
As discussed above, H. pylori infection has been associated both positively and negatively with the development of gastric and non-gastric diseases. Disease development is often linked to chronic inflammatory responses induced by H. pylori, as was discussed in the previous section. However, it is now becoming increasingly clear that H. pylori also induces changes in the host by altering the microbiome. This aspect will be covered in the following paragraphs.
The harsh gastric environment is thought to represent a key limitation to the complexity of the stomach microbiota[147]. This assumption, together with limitations imposed by culture-dependent strategies for bacterial identification, has historically leaded to an underestimation of the biodiversity in the stomach. In this context, the gastric microbiota was initially considered to include only a very select group of taxa, including mainly Veillonella spp., Lactobacillus spp., and Clostridium spp., besides -of course- H. pylori[148-151]. Nonetheless, with the development of more sophisticated 16S rRNA-based bacterial identification techniques, we now have gleaned deeper insight to the complexity of the gastric microbiome. Accordingly, an increasing number of publications describe greater ecosystem diversity in the stomach and, importantly, correlate the presence of H. pylori with variations in the composition of the microbiome[23,48,152-156]. Bik et al[152] identified taxa, such as Caulobacter, Actinobacillus, Corynebacterium, Rothia, Gemella, Leptotrichia, Porphyromonas, Capnocytophaga, TM7, Flexistipes, and Deinococcus in the normal microbiome. Alternatively, Li et al[155] showed that the most common genera in gastric biopsies from both normal and non-H. pylori gastritis individuals were Prevotella, Neisseria, Haemophilus, and Porphyromonas. Later, Delgado et al[153] also identified Propionibacterium, Lactobacillus and Streptococcus as dominant genera in healthy samples.
Regarding the effect of H. pylori on the gastric microbiome, there is still some controversy. No effect on either diversity and/or evenness in community members between H. pylori-positive vs H. pylori-negative samples were observed at the phylum level[152]. Likewise, in a mouse model of H. pylori infection, neither acute nor chronic H. pylori infection altered the murine gastric microbiota[157]. Similarly, others described that, although when present H. pylori dominates the microbiome, only minor differences in community structure were observed in stomach biopsies from H. pylori-positive and negative subjects[158]. However, others have shown that the presence of H. pylori dramatically reduces the diversity of the gastric microbiota[20,21,23] and modifies the microbiome by increasing the relative abundance of Proteobacteria, Spirochetes and Acidobacteria, while decreasing Actinobacteria, Bacteroidetes and Firmicutes[156]. Similar results were reported by Thorell et al[30], who performed a meta-transcriptomic analysis and reported higher levels of Firmicutes, Bacteroidetes, and Actinobacteria in subjects with low levels of H. pylori. Such discrepancies might be due to inter-subject variations, since the gastric microbiome seems to be sensitive to exogenous factors, such as diet and lifestyle, as has been shown by the analysis of monozygotic twins[159].
Interestingly, while the experimental inoculation of H. pylori into an established community in rhesus monkeys, did not affect the community membership or structure[160], pre-infection of mice with H. pylori did alter the microbiota structure in the stomach[22]. Therefore, the time-point in life when H. pylori is acquired is another aspect to be considered in the discrepancies reported for H. pylori-associated microbiome variations. Differences in both diversity and community composition were also observed in the stomachs of H. pylori-infected vs H. pylori-negative children and also in comparison to adults, regardless of the H. pylori status[161]. Thus, early acquisition of the bacterium is likely to shape the microbiome by inducing local modifications in the stomach environment. One of these effects is driven by the production of ammonia and bicarbonate from urea[162,163]. Such compounds may serve as substrates for other bacteria[164], in addition to altering the stomach pH[162,163], which facilitates the colonization by other species, such as nitrogen-reducing bacteria[165]. Moreover, H. pylori-induced increases in the stomach pH favor the migration to the stomach of some bacterial taxa that are usually restricted to the intestinal tract (Bacteroides and Clostridia) in mice[166]. Interestingly, the effect of H. pylori on acid secretion depends on the pattern of gastritis that is induced[167]. In predominantly antral gastritis, the production of gastric acid is increased (hyperchlorhydria)[168], while in predominantly corpus gastritis, acid production decreases (hypochlorhydria)[169]. Thus, microbiome shifts may differ in both cases. In fact, hyperchlorhydria increases microbial diversity in the stomach[170] and it has been implicated in the development and progression of cancer (reviewed by Espinoza et al 2018[171]). Additionally, the viscosity of the gastric mucus layer decreases when the pH increases[172], making it easier for other microorganisms to colonize the epithelium. Finally, H. pylori can directly alter the mucus barrier by modulating the expression of stomach mucins[173].
As illustrated above, all these environmental modifications in the stomach may impact on the local microbiome, as well as induce changes in the entire gastrointestinal tract, since these are dynamic compartments between which fluids are exchanged and therefore microbes can easily migrate from one gastrointestinal segment to another[174]. Some of these H. pylori-mediated downstream effects in other compartments include impairment in the absorption of iron and vitamin B12 in the intestine[175,176], and alterations in carbohydrate and amino acid metabolism of the host[177]. Interestingly, besides the direct effect of H. pylori in the stomach/intestine, also the immune response triggered by the bacterium could affect the local microbiome, as well as bacterial populations at more distal sites in the human body.
Regarding the effect of H. pylori-mediated immune mediators in the microbiome, there are some contradictory reports. No statistically significant differences in the microbiota were found in CagA-positive (n = 10) compared to CagA-negative (n = 10) biopsies from human subjects[178]. Therefore, the increased production of pro-inflammatory cytokines mediated by CagA appears not to have an effect on the microbiome in this model. Nonetheless, as the sample cohort was small in that study, this question probably needs to be re-evaluated in larger groups of samples. In contrast, in a transgenic Drosophila model of CagA expression, CagA was sufficient to alter midgut host microbiota[179]. Additionally, a series of reports demonstrated differences in the intestinal microbiome related to the presence of H. pylori both in humans[180] and mice[22]. Moreover Schulz et al[158] correlated the presence of H. pylori in human individuals with modifications in the microbiome of the duodenum and the oral cavity. More specifically, Heimesaat et al[181] demonstrated that chronic infection of Mongolian gerbils with H. pylori resulted in changes in some specific genera, including increased abundance in the large intestine of Akkermansia, which is involved in mucus degradation. These changes were accompanied by variations in the expression of immunity-related genes in both the stomach and the lung, with stronger effects in the former. The authors speculated that early community shifts could reflect changes in the niche microenvironment (e.g., altered gastric pH), while later shifts might be driven by the cumulative changes in the immune/inflammatory response triggered by H. pylori. These effects could also be observed at distant sites in the host organism and be driven by other members of the Helicobacter genus. For instance, natural colonization of the mouse digestive tract with Helicobacter hepaticus leads to a shift in gut microbiota, which generates subclinical inflammation and a drastic impairment of the control of Mycobacterium tuberculosis growth by the immune system[182].
It is likely that H. pylori might affect mucosal diseases at distant sites via its effects on immune cells that traffic through lymphatic vessels. H. pylori also induces a shift in the immune response toward an induction of Treg cells, mediated by VacA and GGT[59,161,183,184]. Treg cell responses are important in differentiating between self and foreign antigens, i.e., immunological tolerance. This effect could be involved in H. pylori persistence in the stomach, and also could contribute to suppressing gastric inflammation, which may explain reduced gastric disease severity in H. pylori-positive children compared to H. pylori-positive adults[161]. Such effects have been linked to alterations in the stomach physiology and its microbiota, as well as to progression of extra-gastric diseases, such as asthma[59], celiac disease[17], ischemic heart diseases, insulin resistance, Type 2 diabetes mellitus, periodontal diseases, among others (see previous sections and references listed in Table 1).
Interestingly, as was stated above, H. pylori is part of a complex microbiota in the stomach and its presence has been linked to modifications in the microbiome at other sites in the body. Therefore, it is reasonable to speculate that other community members could also contribute to changes observed following H. pylori infection of the host. Indeed, direct bacteria-bacteria interaction was described by Khosravi et al[185] between H. pylori and Streptococcus mitis, in which H. pylori transit from spiral to coccoid-shaped cells that are more resistant to stressing conditions. The exact effect of this conversion on disease progression remains to be elucidated. Also, antimicrobial molecules produced by Lactobacillus spp have been shown to be active against H. pylori strains[186-189]. Therefore, bidirectional communication and modulation between H. pylori and other community members occur, and the combination of all these interactions will be reflected in the host health status. The question as to how the microbiota shapes the immune system and how this affects its response to some diseases has been broadly discussed in the literature (see for instance, Hooper et al[190] 2012). Therefore, modifications in the microbiome induced by early acquisition of H. pylori[161] may also determine the host immune status and, as a consequence, the development of a number of systemic diseases. This is relevant, since Rolig et al[191] reported that when the microbiota is altered by antibiotic treatment, the H. pylori-triggered inflammation is reduced.
In summary, H. pylori has a strong effect in the stomach microenvironment and also on the host immunological status, leading to shifts in the microbiome at different sites of the body. These shifts are involved not only in the pathogenesis of gastric diseases, but also in some of the non-gastric H. pylori-related diseases that were discussed in this review. The question as to whether the bacterium is acquired early on or later in life is a key point when analyzing such effects, as H. pylori has been reported to co-evolve with its host, shaping the immune system and consequently, the microbiome. Therefore, a better understanding of the exact mechanisms involved in such effects, as well as the H. pylori-induced microbiome shifts that may be related to the development of specific diseases, will likely be useful to predict and hopefully prevent H. pylori-associated diseases. Ideally, one would aspire to achieving this goal while at the same time preserving the beneficial effects that H. pylori-host co-habitation appears also to offer.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Chile
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
P- Reviewer: Caruso R, Martini F S- Editor: Wang JL L- Editor: A E- Editor: Yin SY
| 1. | de Martel C, Ferlay J, Franceschi S, Vignat J, Bray F, Forman D, Plummer M. Global burden of cancers attributable to infections in 2008: a review and synthetic analysis. Lancet Oncol. 2012;13:607-615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1583] [Cited by in RCA: 1714] [Article Influence: 131.8] [Reference Citation Analysis (1)] |
| 2. | Suerbaum S, Michetti P. Helicobacter pylori infection. N Engl J Med. 2002;347:1175-1186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1848] [Cited by in RCA: 1906] [Article Influence: 82.9] [Reference Citation Analysis (3)] |
| 3. | Shmuely H, Wattad M, Solodky A, Yahav J, Samra Z, Zafrir N. Association of Helicobacter pylori with coronary artery disease and myocardial infarction assessed by myocardial perfusion imaging. Isr Med Assoc J. 2014;16:341-346. [PubMed] |
| 4. | Liu J, Wang F, Shi S. Helicobacter pylori Infection Increase the Risk of Myocardial Infarction: A Meta-Analysis of 26 Studies Involving more than 20,000 Participants. Helicobacter. 2015;20:176-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 54] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 5. | Li JZ, Li JY, Wu TF, Xu JH, Huang CZ, Cheng D, Chen QK, Yu T. Helicobacter pylori Infection Is Associated with Type 2 Diabetes, Not Type 1 Diabetes: An Updated Meta-Analysis. Gastroenterol Res Pract. 2017;2017:5715403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 6. | Xu MY, Cao B, Yuan BS, Yin J, Liu L, Lu QB. Association of anaemia with Helicobacter pylori infection: a retrospective study. Sci Rep. 2017;7:13434. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 7. | Chen CX, Mao YS, Foster P, Zhu ZW, Du J, Guo CY. Possible association between Helicobacter pylori infection and nonalcoholic fatty liver disease. Appl Physiol Nutr Metab. 2017;42:295-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 8. | Upala S, Sanguankeo A, Saleem SA, Jaruvongvanich V. Effects of Helicobacter pylori eradication on insulin resistance and metabolic parameters: a systematic review and meta-analysis. Eur J Gastroenterol Hepatol. 2017;29:153-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 9. | Bach JF. The effect of infections on susceptibility to autoimmune and allergic diseases. N Engl J Med. 2002;347:911-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1881] [Cited by in RCA: 1789] [Article Influence: 77.8] [Reference Citation Analysis (0)] |
| 10. | Chen Y, Blaser MJ. Inverse associations of Helicobacter pylori with asthma and allergy. Arch Intern Med. 2007;167:821-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 282] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 11. | Cohen D, Shoham O, Orr N, Muhsen K. An inverse and independent association between Helicobacter pylori infection and the incidence of shigellosis and other diarrheal diseases. Clin Infect Dis. 2012;54:e35-e42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 12. | Islami F, Kamangar F. Helicobacter pylori and esophageal cancer risk: a meta-analysis. Cancer Prev Res (Phila). 2008;1:329-338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 257] [Cited by in RCA: 250] [Article Influence: 14.7] [Reference Citation Analysis (1)] |
| 13. | Perry S, de Jong BC, Solnick JV, de la Luz Sanchez M, Yang S, Lin PL, Hansen LM, Talat N, Hill PC, Hussain R. Infection with Helicobacter pylori is associated with protection against tuberculosis. PLoS One. 2010;5:e8804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 124] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 14. | Sonnenberg A, Dellon ES, Turner KO, Genta RM. The influence of Helicobacter pylori on the ethnic distribution of esophageal eosinophilia. Helicobacter. 2017;22:e12370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 15. | Miftahussurur M, Nusi IA, Graham DY, Yamaoka Y. Helicobacter, Hygiene, Atopy, and Asthma. Front Microbiol. 2017;8:1034. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 16. | Efthymiou G, Dardiotis E, Liaskos C, Marou E, Tsimourtou V, Scheper T, Meyer W, Daponte A, Sakkas LI, Hadjigeorgiou G. Anti-hsp60 antibody responses based on Helicobacter pylori in patients with multiple sclerosis: (ir)Relevance to disease pathogenesis. J Neuroimmunol. 2016;298:19-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 17. | Lucero Y, Oyarzún A, O’Ryan M, Quera R, Espinosa N, Valenzuela R, Simian D, Alcalde E, Arce C, Farfán MJ. Helicobacter pylori cagA+ Is Associated with Milder Duodenal Histological Changes in Chilean Celiac Patients. Front Cell Infect Microbiol. 2017;7:376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 18. | Narang M, Puri AS, Sachdeva S, Singh J, Kumar A, Saran RK. Celiac disease and Helicobacter pylori infection in children: Is there any Association? J Gastroenterol Hepatol. 2017;32:1178-1182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 19. | Niwa T, Toyoda T, Tsukamoto T, Mori A, Tatematsu M, Ushijima T. Prevention of Helicobacter pylori-induced gastric cancers in gerbils by a DNA demethylating agent. Cancer Prev Res (Phila). 2013;6:263-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 59] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 20. | Andersson AF, Lindberg M, Jakobsson H, Bäckhed F, Nyrén P, Engstrand L. Comparative analysis of human gut microbiota by barcoded pyrosequencing. PLoS One. 2008;3:e2836. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 743] [Cited by in RCA: 741] [Article Influence: 43.6] [Reference Citation Analysis (0)] |
| 21. | Das A, Pereira V, Saxena S, Ghosh TS, Anbumani D, Bag S, Das B, Nair GB, Abraham P, Mande SS. Gastric microbiome of Indian patients with Helicobacter pylori infection, and their interaction networks. Sci Rep. 2017;7:15438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 22. | Kienesberger S, Cox LM, Livanos A, Zhang XS, Chung J, Perez-Perez GI, Gorkiewicz G, Zechner EL, Blaser MJ. Gastric Helicobacter pylori Infection Affects Local and Distant Microbial Populations and Host Responses. Cell Rep. 2016;14:1395-1407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 120] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 23. | Llorca L, Pérez-Pérez G, Urruzuno P, Martinez MJ, Iizumi T, Gao Z, Sohn J, Chung J, Cox L, Simón-Soro A. Characterization of the Gastric Microbiota in a Pediatric Population According to Helicobacter pylori Status. Pediatr Infect Dis J. 2017;36:173-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 75] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 24. | Plummer M, de Martel C, Vignat J, Ferlay J, Bray F, Franceschi S. Global burden of cancers attributable to infections in 2012: a synthetic analysis. Lancet Glob Health. 2016;4:e609-e616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1068] [Cited by in RCA: 1016] [Article Influence: 112.9] [Reference Citation Analysis (0)] |
| 25. | Linz B, Balloux F, Moodley Y, Manica A, Liu H, Roumagnac P, Falush D, Stamer C, Prugnolle F, van der Merwe SW. An African origin for the intimate association between humans and Helicobacter pylori. Nature. 2007;445:915-918. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 720] [Cited by in RCA: 636] [Article Influence: 35.3] [Reference Citation Analysis (3)] |
| 26. | Achtman M, Azuma T, Berg DE, Ito Y, Morelli G, Pan ZJ, Suerbaum S, Thompson SA, van der Ende A, van Doorn LJ. Recombination and clonal groupings within Helicobacter pylori from different geographical regions. Mol Microbiol. 1999;32:459-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 270] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 27. | Morelli G, Didelot X, Kusecek B, Schwarz S, Bahlawane C, Falush D, Suerbaum S, Achtman M. Microevolution of Helicobacter pylori during prolonged infection of single hosts and within families. PLoS Genet. 2010;6:e1001036. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 120] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 28. | Schwarz S, Morelli G, Kusecek B, Manica A, Balloux F, Owen RJ, Graham DY, van der Merwe S, Achtman M, Suerbaum S. Horizontal versus familial transmission of Helicobacter pylori. PLoS Pathog. 2008;4:e1000180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 108] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 29. | Falush D, Wirth T, Linz B, Pritchard JK, Stephens M, Kidd M, Blaser MJ, Graham DY, Vacher S, Perez-Perez GI. Traces of human migrations in Helicobacter pylori populations. Science. 2003;299:1582-1585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 738] [Cited by in RCA: 707] [Article Influence: 32.1] [Reference Citation Analysis (1)] |
| 30. | Thorell K, Yahara K, Berthenet E, Lawson DJ, Mikhail J, Kato I, Mendez A, Rizzato C, Bravo MM, Suzuki R. Correction: Rapid evolution of distinct Helicobacter pylori subpopulations in the Americas. PLoS Genet. 2017;13:e1006730. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 31. | Agnew P, Koella JC. Virulence, parasite mode of transmission, and host fluctuating asymmetry. Proc Biol Sci. 1997;264:9-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 73] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 32. | Ahmed N, Tenguria S, Nandanwar N. Helicobacter pylori--a seasoned pathogen by any other name. Gut Pathog. 2009;1:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 33. | Hatakeyama M. [Oncogenic mechanism of Helicobacter pylori]. Nihon Rinsho Meneki Gakkai Kaishi. 2008;31:132-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 34. | Atherton JC, Tham KT, Peek RM Jr, Cover TL, Blaser MJ. Density of Helicobacter pylori infection in vivo as assessed by quantitative culture and histology. J Infect Dis. 1996;174:552-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 125] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 35. | Devi SM, Ahmed I, Khan AA, Rahman SA, Alvi A, Sechi LA, Ahmed N. Genomes of Helicobacter pylori from native Peruvians suggest admixture of ancestral and modern lineages and reveal a western type cag-pathogenicity island. BMC Genomics. 2006;7:191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 39] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 36. | Kodaman N, Pazos A, Schneider BG, Piazuelo MB, Mera R, Sobota RS, Sicinschi LA, Shaffer CL, Romero-Gallo J, de Sablet T. Human and Helicobacter pylori coevolution shapes the risk of gastric disease. Proc Natl Acad Sci USA. 2014;111:1455-1460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 180] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 37. | Gutiérrez-Escobar AJ, Trujillo E, Acevedo O, Bravo MM. Phylogenomics of Colombian Helicobacter pylori isolates. Gut Pathog. 2017;9:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 38. | Duncan SS, Valk PL, McClain MS, Shaffer CL, Metcalf JA, Bordenstein SR, Cover TL. Comparative genomic analysis of East Asian and non-Asian Helicobacter pylori strains identifies rapidly evolving genes. PLoS One. 2013;8:e55120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 39. | Kauser F, Khan AA, Hussain MA, Carroll IM, Ahmad N, Tiwari S, Shouche Y, Das B, Alam M, Ali SM. The cag pathogenicity island of Helicobacter pylori is disrupted in the majority of patient isolates from different human populations. J Clin Microbiol. 2004;42:5302-5308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 72] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 40. | Mhaskar RS, Ricardo I, Azliyati A, Laxminarayan R, Amol B, Santosh W, Boo K. Assessment of risk factors of helicobacter pylori infection and peptic ulcer disease. J Glob Infect Dis. 2013;5:60-67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 41. | El-Omar EM, Carrington M, Chow WH, McColl KE, Bream JH, Young HA, Herrera J, Lissowska J, Yuan CC, Rothman N. Interleukin-1 polymorphisms associated with increased risk of gastric cancer. Nature. 2000;404:398-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1690] [Cited by in RCA: 1674] [Article Influence: 67.0] [Reference Citation Analysis (0)] |
| 42. | El-Omar EM, Carrington M, Chow WH, McColl KE, Bream JH, Young HA, Herrera J, Lissowska J, Yuan CC, Rothman N. The role of interleukin-1 polymorphisms in the pathogenesis of gastric cancer. Nature. 2001;412:99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 160] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 43. | El-Omar EM, Rabkin CS, Gammon MD, Vaughan TL, Risch HA, Schoenberg JB, Stanford JL, Mayne ST, Goedert J, Blot WJ. Increased risk of noncardia gastric cancer associated with proinflammatory cytokine gene polymorphisms. Gastroenterology. 2003;124:1193-1201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 672] [Cited by in RCA: 676] [Article Influence: 30.7] [Reference Citation Analysis (0)] |
| 44. | Figueiredo C, Machado JC, Pharoah P, Seruca R, Sousa S, Carvalho R, Capelinha AF, Quint W, Caldas C, van Doorn LJ. Helicobacter pylori and interleukin 1 genotyping: an opportunity to identify high-risk individuals for gastric carcinoma. J Natl Cancer Inst. 2002;94:1680-1687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 463] [Cited by in RCA: 471] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 45. | Garza-González E, Bosques-Padilla FJ, El-Omar E, Hold G, Tijerina-Menchaca R, Maldonado-Garza HJ, Pérez-Pérez GI. Role of the polymorphic IL-1B, IL-1RN and TNF-A genes in distal gastric cancer in Mexico. Int J Cancer. 2005;114:237-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 99] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 46. | Ando T, Yoshida T, Enomoto S, Asada K, Tatematsu M, Ichinose M, Sugiyama T, Ushijima T. DNA methylation of microRNA genes in gastric mucosae of gastric cancer patients: its possible involvement in the formation of epigenetic field defect. Int J Cancer. 2009;124:2367-2374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 221] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 47. | Shin CM, Kim N, Jung Y, Park JH, Kang GH, Kim JS, Jung HC, Song IS. Role of Helicobacter pylori infection in aberrant DNA methylation along multistep gastric carcinogenesis. Cancer Sci. 2010;101:1337-1346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 54] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 48. | Monstein HJ, Tiveljung A, Kraft CH, Borch K, Jonasson J. Profiling of bacterial flora in gastric biopsies from patients with Helicobacter pylori-associated gastritis and histologically normal control individuals by temperature gradient gel electrophoresis and 16S rDNA sequence analysis. J Med Microbiol. 2000;49:817-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 87] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 49. | Song Q, Lange T, Spahr A, Adler G, Bode G. Characteristic distribution pattern of Helicobacter pylori in dental plaque and saliva detected with nested PCR. J Med Microbiol. 2000;49:349-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 111] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 50. | Pest P, Zárate J, Varsky C, Man F, Schraier M. Helicobacter pylori in recently-diagnosed versus chronic duodenal ulcer. Acta Gastroenterol Latinoam. 1996;26:273-276. [PubMed] |
| 51. | Mishra S. Is Helicobacter pylori good or bad? Eur J Clin Microbiol Infect Dis. 2013;32:301-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 52. | Arnold IC, Hitzler I, Müller A. The immunomodulatory properties of Helicobacter pylori confer protection against allergic and chronic inflammatory disorders. Front Cell Infect Microbiol. 2012;2:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 101] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 53. | Holster IL, Vila AM, Caudri D, den Hoed CM, Perez-Perez GI, Blaser MJ, de Jongste JC, Kuipers EJ. The impact of Helicobacter pylori on atopic disorders in childhood. Helicobacter. 2012;17:232-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 54. | den Hollander WJ, Sonnenschein-van der Voort AM, Holster IL, de Jongste JC, Jaddoe VW, Hofman A, Perez-Perez GI, Moll HA, Blaser MJ, Duijts L. Helicobacter pylori in children with asthmatic conditions at school age, and their mothers. Aliment Pharmacol Ther. 2016;43:933-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 55. | Chen Y, Blaser MJ. Helicobacter pylori colonization is inversely associated with childhood asthma. J Infect Dis. 2008;198:553-560. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 291] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 56. | Bamford KB, Fan X, Crowe SE, Leary JF, Gourley WK, Luthra GK, Brooks EG, Graham DY, Reyes VE, Ernst PB. Lymphocytes in the human gastric mucosa during Helicobacter pylori have a T helper cell 1 phenotype. Gastroenterology. 1998;114:482-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 429] [Cited by in RCA: 425] [Article Influence: 15.7] [Reference Citation Analysis (1)] |
| 57. | Sommer F, Faller G, Konturek P, Kirchner T, Hahn EG, Zeus J, Röllinghoff M, Lohoff M. Antrum- and corpus mucosa-infiltrating CD4(+) lymphocytes in Helicobacter pylori gastritis display a Th1 phenotype. Infect Immun. 1998;66:5543-5546. [PubMed] |
| 58. | Lundgren A, Trollmo C, Edebo A, Svennerholm AM, Lundin BS. Helicobacter pylori-specific CD4+ T cells home to and accumulate in the human Helicobacter pylori-infected gastric mucosa. Infect Immun. 2005;73:5612-5619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 74] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 59. | Oertli M, Noben M, Engler DB, Semper RP, Reuter S, Maxeiner J, Gerhard M, Taube C, Müller A. Helicobacter pylori γ-glutamyl transpeptidase and vacuolating cytotoxin promote gastric persistence and immune tolerance. Proc Natl Acad Sci USA. 2013;110:3047-3052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 186] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 60. | Dela Pena-Ponce MG, Jimenez MT, Hansen LM, Solnick JV, Miller LA. The Helicobacter pylori type IV secretion system promotes IL-8 synthesis in a model of pediatric airway epithelium via p38 MAP kinase. PLoS One. 2017;12:e0183324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 61. | Higgins PD, Johnson LA, Luther J, Zhang M, Sauder KL, Blanco LP, Kao JY. Prior Helicobacter pylori infection ameliorates Salmonella typhimurium-induced colitis: mucosal crosstalk between stomach and distal intestine. Inflamm Bowel Dis. 2011;17:1398-1408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 64] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 62. | Lord AR, Simms LA, Hanigan K, Sullivan R, Hobson P, Radford-Smith GL. Protective effects of Helicobacter pylori for IBD are related to the cagA-positive strain. Gut. 2018;67:393-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 63. | Castaño-Rodríguez N, Kaakoush NO, Lee WS, Mitchell HM. Dual role of Helicobacter and Campylobacter species in IBD: a systematic review and meta-analysis. Gut. 2017;66:235-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 165] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 64. | Rubio-Tapia A, Kyle RA, Kaplan EL, Johnson DR, Page W, Erdtmann F, Brantner TL, Kim WR, Phelps TK, Lahr BD. Increased prevalence and mortality in undiagnosed celiac disease. Gastroenterology. 2009;137:88-93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 583] [Cited by in RCA: 519] [Article Influence: 32.4] [Reference Citation Analysis (1)] |
| 65. | Lebwohl B, Blaser MJ, Ludvigsson JF, Green PH, Rundle A, Sonnenberg A, Genta RM. Decreased risk of celiac disease in patients with Helicobacter pylori colonization. Am J Epidemiol. 2013;178:1721-1730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 98] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 66. | Grigoriadis N, van Pesch V; ParadigMS Group. A basic overview of multiple sclerosis immunopathology. Eur J Neurol. 2015;22 Suppl 2:3-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 126] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 67. | Yao G, Wang P, Luo XD, Yu TM, Harris RA, Zhang XM. Meta-analysis of association between Helicobacter pylori infection and multiple sclerosis. Neurosci Lett. 2016;620:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 68. | Jaruvongvanich V, Sanguankeo A, Jaruvongvanich S, Upala S. Association between Helicobacter pylori infection and multiple sclerosis: A systematic review and meta-analysis. Mult Scler Relat Disord. 2016;7:92-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 69. | Alvarez-Arellano L, Maldonado-Bernal C. Helicobacter pylori and neurological diseases: Married by the laws of inflammation. World J Gastrointest Pathophysiol. 2014;5:400-404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 65] [Cited by in RCA: 72] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 70. | Boziki M, Polyzos SA, Deretzi G, Kazakos E, Katsinelos P, Doulberis M, Kotronis G, Giartza-Taxidou E, Laskaridis L, Tzivras D. A potential impact of Helicobacter pylori-related galectin-3 in neurodegeneration. Neurochem Int. 2018;113:137-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 71. | Testerman TL, Morris J. Beyond the stomach: an updated view of Helicobacter pylori pathogenesis, diagnosis, and treatment. World J Gastroenterol. 2014;20:12781-12808. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 188] [Cited by in RCA: 214] [Article Influence: 19.5] [Reference Citation Analysis (3)] |
| 72. | Huang WS, Tseng CH, Lin CL, Tsai CH, Kao CH. Helicobacter pylori infection increases subsequent ischemic stroke risk: a nationwide population-based retrospective cohort study. QJM. 2014;107:969-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 73. | Sharma V, Aggarwal A. Helicobacter pylori: Does it add to risk of coronary artery disease. World J Cardiol. 2015;7:19-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 74. | Konno M, Muraoka S, Takahashi M, Imai T. Iron-deficiency anemia associated with Helicobacter pylori gastritis. J Pediatr Gastroenterol Nutr. 2000;31:52-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 74] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 75. | Flores SE, Aitchison A, Day AS, Keenan JI. Helicobacter pylori infection perturbs iron homeostasis in gastric epithelial cells. PLoS One. 2017;12:e0184026. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 76. | Flores SE, Day AS, Keenan JI. Measurement of total iron in Helicobacter pylori-infected gastric epithelial cells. Biometals. 2015;28:143-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 77. | Kato S, Osaki T, Kamiya S, Zhang XS, Blaser MJ. Helicobacter pylori sabA gene is associated with iron deficiency anemia in childhood and adolescence. PLoS One. 2017;12:e0184046. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 78. | Huang Y, Tian XF, Fan XG, Fu CY, Zhu C. The pathological effect of Helicobacter pylori infection on liver tissues in mice. Clin Microbiol Infect. 2009;15:843-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 79. | Cheng DD, He C, Ai HH, Huang Y, Lu NH. The Possible Role of Helicobacter pylori Infection in Non-alcoholic Fatty Liver Disease. Front Microbiol. 2017;8:743. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 80. | Li L, He Q, Huang X, Man Y, Zhou Y, Wang S, Wang J, Li J. NOX3-derived reactive oxygen species promote TNF-alpha-induced reductions in hepatocyte glycogen levels via a JNK pathway. FEBS Lett. 2010;584:995-1000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 81. | Hotamisligil GS, Johnson RS, Distel RJ, Ellis R, Papaioannou VE, Spiegelman BM. Uncoupling of obesity from insulin resistance through a targeted mutation in aP2, the adipocyte fatty acid binding protein. Science. 1996;274:1377-1379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 621] [Cited by in RCA: 607] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 82. | Aydemir S, Bayraktaroglu T, Sert M, Sokmen C, Atmaca H, Mungan G, Gun BD, Borazan A, Ustundag Y. The effect of Helicobacter pylori on insulin resistance. Dig Dis Sci. 2005;50:2090-2093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 66] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 83. | Gunji T, Matsuhashi N, Sato H, Fujibayashi K, Okumura M, Sasabe N, Urabe A. Helicobacter pylori infection significantly increases insulin resistance in the asymptomatic Japanese population. Helicobacter. 2009;14:144-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 94] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 84. | Chen LW, Chien CY, Yang KJ, Kuo SF, Chen CH, Chien RN. Helicobacter pylori Infection Increases Insulin Resistance and Metabolic Syndrome in Residents Younger than 50 Years Old: A Community-Based Study. PLoS One. 2015;10:e0128671. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 85. | Polyzos SA, Kountouras J, Zavos C, Deretzi G. Helicobacter pylori Infection and insulin resistance. Helicobacter. 2013;18:165-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 86. | Polyzos SA, Kountouras J, Zavos C, Deretzi G. The association between Helicobacter pylori infection and insulin resistance: a systematic review. Helicobacter. 2011;16:79-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 159] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 87. | Hossain IA, Akter S, Bhuiyan FR, Shah MR, Rahman MK, Ali L. Subclinical inflammation in relation to insulin resistance in prediabetic subjects with nonalcoholic fatty liver disease. BMC Res Notes. 2016;9:266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 88. | Tsai S, Clemente-Casares X, Revelo XS, Winer S, Winer DA. Are obesity-related insulin resistance and type 2 diabetes autoimmune diseases? Diabetes. 2015;64:1886-1897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 64] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 89. | Yildirim Z, Bozkurt B, Ozol D, Armutcu F, Akgedik R, Karamanli H, Kizilirmak D, İkizek M. Increased Exhaled 8-Isoprostane and Interleukin-6 in Patients with Helicobacter pylori Infection. Helicobacter. 2016;21:389-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 90. | Dandona P, Aljada A, Bandyopadhyay A. Inflammation: the link between insulin resistance, obesity and diabetes. Trends Immunol. 2004;25:4-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1432] [Cited by in RCA: 1519] [Article Influence: 72.3] [Reference Citation Analysis (0)] |
| 91. | Bener A, Micallef R, Afifi M, Derbala M, Al-Mulla HM, Usmani MA. Association between type 2 diabetes mellitus and Helicobacter pylori infection. Turk J Gastroenterol. 2007;18:225-229. [PubMed] |
| 92. | Devrajani BR, Shah SZ, Soomro AA, Devrajani T. Type 2 diabetes mellitus: A risk factor for Helicobacter pylori infection: A hospital based case-control study. Int J Diabetes Dev Ctries. 2010;30:22-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 93. | Anastasios R, Goritsas C, Papamihail C, Trigidou R, Garzonis P, Ferti A. Helicobacter pylori infection in diabetic patients: prevalence and endoscopic findings. Eur J Intern Med. 2002;13:376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 50] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 94. | He C, Yang Z, Lu NH. Helicobacter pylori infection and diabetes: is it a myth or fact? World J Gastroenterol. 2014;20:4607-4617. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 54] [Cited by in RCA: 69] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 95. | Aslan M, Horoz M, Nazligul Y, Bolukbas C, Bolukbas FF, Selek S, Celik H, Erel O. Insulin resistance in H pylori infection and its association with oxidative stress. World J Gastroenterol. 2006;12:6865-6868. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 33] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 96. | Nasif WA, Mukhtar MH, Nour Eldein MM, Ashgar SS. Oxidative DNA damage and oxidized low density lipoprotein in Type II diabetes mellitus among patients with Helicobacter pylori infection. Diabetol Metab Syndr. 2016;8:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 97. | Hernández M, Dutzan N, García-Sesnich J, Abusleme L, Dezerega A, Silva N, González FE, Vernal R, Sorsa T, Gamonal J. Host-pathogen interactions in progressive chronic periodontitis. J Dent Res. 2011;90:1164-1170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 128] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 98. | Gamonal J, Mendoza C, Espinoza I, Muñoz A, Urzúa I, Aranda W, Carvajal P, Arteaga O. Clinical attachment loss in Chilean adult population: First Chilean National Dental Examination Survey. J Periodontol. 2010;81:1403-1410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 99. | Atanasova KR, Yilmaz O. Looking in the Porphyromonas gingivalis cabinet of curiosities: the microbium, the host and cancer association. Mol Oral Microbiol. 2014;29:55-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 100] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 100. | Genco RJ, Van Dyke TE. Prevention: Reducing the risk of CVD in patients with periodontitis. Nat Rev Cardiol. 2010;7:479-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 137] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 101. | Han YW, Houcken W, Loos BG, Schenkein HA, Tezal M. Periodontal disease, atherosclerosis, adverse pregnancy outcomes, and head-and-neck cancer. Adv Dent Res. 2014;26:47-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 102. | Lundberg K, Wegner N, Yucel-Lindberg T, Venables PJ. Periodontitis in RA-the citrullinated enolase connection. Nat Rev Rheumatol. 2010;6:727-730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 255] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 103. | Petersen PE, Ogawa H. Strengthening the prevention of periodontal disease: the WHO approach. J Periodontol. 2005;76:2187-2193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 289] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 104. | Al Sayed A, Anand PS, Kamath KP, Patil S, Preethanath RS, Anil S. Oral Cavity as an Extragastric Reservoir of Helicobacter pylori. ISRN Gastroenterol. 2014;2014:261369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 105. | Zheng P, Zhou W. Relation between periodontitis and helicobacter pylori infection. Int J Clin Exp Med. 2015;8:16741-16744. [PubMed] |
| 106. | Sujatha S, Jalihal UM, Sharma S. Association between periodontal disease and oral and gastric Helicobacter pylori infection. Indian J Gastroenterol. 2015;34:343-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 107. | Hu Z, Zhang Y, Li Z, Yu Y, Kang W, Han Y, Geng X, Ge S, Sun Y. Effect of Helicobacter pylori infection on chronic periodontitis by the change of microecology and inflammation. Oncotarget. 2016;7:66700-66712. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 108. | Díaz P, Valenzuela Valderrama M, Bravo J, Quest AFG. Helicobacter pylori and Gastric Cancer: Adaptive Cellular Mechanisms Involved in Disease Progression. Front Microbiol. 2018;9:5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 156] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 109. | Valenzuela MA, Canales J, Corvalán AH, Quest AF. Helicobacter pylori-induced inflammation and epigenetic changes during gastric carcinogenesis. World J Gastroenterol. 2015;21:12742-12756. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 72] [Cited by in RCA: 87] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 110. | Taniguchi K, Karin M. NF-κB, inflammation, immunity and cancer: coming of age. Nat Rev Immunol. 2018;18:309-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1132] [Cited by in RCA: 1911] [Article Influence: 273.0] [Reference Citation Analysis (0)] |
| 111. | Kanda Y, Osaki M, Okada F. Chemopreventive Strategies for Inflammation-Related Carcinogenesis: Current Status and Future Direction. Int J Mol Sci. 2017;18:pii: E867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 112. | Nejati S, Karkhah A, Darvish H, Validi M, Ebrahimpour S, Nouri HR. Influence of Helicobacter pylori virulence factors CagA and VacA on pathogenesis of gastrointestinal disorders. Microb Pathog. 2018;117:43-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (1)] |
| 113. | Sitaraman R. Allergies, Helicobacter pylori and the continental enigmas. Front Microbiol. 2015;6:578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 114. | Zabaleta J. Multifactorial etiology of gastric cancer. Methods Mol Biol. 2012;863:411-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 107] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 115. | Robinson K. Helicobacter pylori-Mediated Protection against Extra-Gastric Immune and Inflammatory Disorders: The Evidence and Controversies. Diseases. 2015;3:34-55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 116. | Atherton JC, Blaser MJ. Coadaptation of Helicobacter pylori and humans: ancient history, modern implications. J Clin Invest. 2009;119:2475-2487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 367] [Cited by in RCA: 393] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 117. | Lina TT, Alzahrani S, Gonzalez J, Pinchuk IV, Beswick EJ, Reyes VE. Immune evasion strategies used by Helicobacter pylori. World J Gastroenterol. 2014;20:12753-12766. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 72] [Cited by in RCA: 97] [Article Influence: 8.8] [Reference Citation Analysis (2)] |
| 118. | Mejías-Luque R, Gerhard M. Immune Evasion Strategies and Persistence of Helicobacter pylori. Curr Top Microbiol Immunol. 2017;400:53-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 119. | Morey P, Pfannkuch L, Pang E, Boccellato F, Sigal M, Imai-Matsushima A, Dyer V, Koch M, Mollenkopf HJ, Schlaermann P. Helicobacter pylori Depletes Cholesterol in Gastric Glands to Prevent Interferon Gamma Signaling and Escape the Inflammatory Response. Gastroenterology. 2018;154:1391-1404.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 96] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 120. | McGee DJ, George AE, Trainor EA, Horton KE, Hildebrandt E, Testerman TL. Cholesterol enhances Helicobacter pylori resistance to antibiotics and LL-37. Antimicrob Agents Chemother. 2011;55:2897-2904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 121. | Du SY, Wang HJ, Cheng HH, Chen SD, Wang LH, Wang WC. Cholesterol glucosylation by Helicobacter pylori delays internalization and arrests phagosome maturation in macrophages. J Microbiol Immunol Infect. 2016;49:636-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 122. | Wang HJ, Cheng WC, Cheng HH, Lai CH, Wang WC. Helicobacter pylori cholesteryl glucosides interfere with host membrane phase and affect type IV secretion system function during infection in AGS cells. Mol Microbiol. 2012;83:67-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 123. | Wunder C, Churin Y, Winau F, Warnecke D, Vieth M, Lindner B, Zähringer U, Mollenkopf HJ, Heinz E, Meyer TF. Cholesterol glucosylation promotes immune evasion by Helicobacter pylori. Nat Med. 2006;12:1030-1038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 218] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 124. | Stent A, Every AL, Chionh YT, Ng GZ, Sutton P. Superoxide dismutase from Helicobacter pylori suppresses the production of pro-inflammatory cytokines during in vivo infection. Helicobacter. 2018;23:e12459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 125. | van Wijck Y, de Kleijn S, John-Schuster G, Mertens TCJ, Hiemstra PS, Müller A, Smits HH, Taube C. Therapeutic Application of an Extract of Helicobacter pylori Ameliorates the Development of Allergic Airway Disease. J Immunol. 2018;200:1570-1579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 126. | Robinson DS, Hamid Q, Ying S, Tsicopoulos A, Barkans J, Bentley AM, Corrigan C, Durham SR, Kay AB. Predominant TH2-like bronchoalveolar T-lymphocyte population in atopic asthma. N Engl J Med. 1992;326:298-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2058] [Cited by in RCA: 2041] [Article Influence: 61.8] [Reference Citation Analysis (0)] |
| 127. | Zhang M, Berndt BE, Eaton KA, Rathinavelu S, Pierzchala A, Kao JY. Helicobacter pylori-pulsed dendritic cells induce H. pylori-specific immunity in mice. Helicobacter. 2008;13:200-208. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 128. | Kao JY, Zhang M, Miller MJ, Mills JC, Wang B, Liu M, Eaton KA, Zou W, Berndt BE, Cole TS. Helicobacter pylori immune escape is mediated by dendritic cell-induced Treg skewing and Th17 suppression in mice. Gastroenterology. 2010;138:1046-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 251] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 129. | Mitchell P, Germain C, Fiori PL, Khamri W, Foster GR, Ghosh S, Lechler RI, Bamford KB, Lombardi G. Chronic exposure to Helicobacter pylori impairs dendritic cell function and inhibits Th1 development. Infect Immun. 2007;75:810-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 78] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 130. | Hussain K, Letley DP, Greenaway AB, Kenefeck R, Winter JA, Tomlinson W, Rhead J, Staples E, Kaneko K, Atherton JC. Helicobacter pylori-Mediated Protection from Allergy Is Associated with IL-10-Secreting Peripheral Blood Regulatory T Cells. Front Immunol. 2016;7:71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 131. | Arnold IC, Dehzad N, Reuter S, Martin H, Becher B, Taube C, Müller A. Helicobacter pylori infection prevents allergic asthma in mouse models through the induction of regulatory T cells. J Clin Invest. 2011;121:3088-3093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 326] [Cited by in RCA: 358] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 132. | Arnold IC, Lee JY, Amieva MR, Roers A, Flavell RA, Sparwasser T, Müller A. Tolerance rather than immunity protects from Helicobacter pylori-induced gastric preneoplasia. Gastroenterology. 2011;140:199-209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 242] [Cited by in RCA: 234] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 133. | Bagheri N, Shirzad H, Elahi S, Azadegan-Dehkordi F, Rahimian G, Shafigh M, Rashidii R, Sarafnejad A, Rafieian-Kopaei M, Faridani R. Downregulated regulatory T cell function is associated with increased peptic ulcer in Helicobacter pylori-infection. Microb Pathog. 2017;110:165-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 134. | Zhou S, Huang Y, Liang B, Dong H, Yao S, Chen Y, Xie Y, Long Y, Gong S, Zhou Z. Systemic and mucosal pre-administration of recombinant Helicobacter pylori neutrophil-activating protein prevents ovalbumin-induced allergic asthma in mice. FEMS Microbiol Lett. 2017;364:pii: fnw288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 135. | Tran LS, Chonwerawong M, Ferrero RL. Regulation and functions of inflammasome-mediated cytokines in Helicobacter pylori infection. Microbes Infect. 2017;19:449-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 136. | Gong Y, Tao L, Jing L, Liu D, Hu S, Liu W, Zhou N, Xie Y. Association of TLR4 and Treg in Helicobacter pylori Colonization and Inflammation in Mice. PLoS One. 2016;11:e0149629. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 137. | Yuan X, Zhou Y, Wang W, Li J, Xie G, Zhao Y, Xu D, Shen L. Activation of TLR4 signaling promotes gastric cancer progression by inducing mitochondrial ROS production. Cell Death Dis. 2013;4:e794. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 98] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 138. | Kawahara T, Teshima S, Oka A, Sugiyama T, Kishi K, Rokutan K. Type I Helicobacter pylori lipopolysaccharide stimulates toll-like receptor 4 and activates mitogen oxidase 1 in gastric pit cells. Infect Immun. 2001;69:4382-4389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 147] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 139. | Lepper PM, Triantafilou M, Schumann C, Schneider EM, Triantafilou K. Lipopolysaccharides from Helicobacter pylori can act as antagonists for Toll-like receptor 4. Cell Microbiol. 2005;7:519-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 76] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 140. | Kawahara T, Kuwano Y, Teshima-Kondo S, Sugiyama T, Kawai T, Nikawa T, Kishi K, Rokutan K. Helicobacter pylori lipopolysaccharide from type I, but not type II strains, stimulates apoptosis of cultured gastric mucosal cells. J Med Invest. 2001;48:167-174. [PubMed] |
| 141. | Sun X, Zhang M, El-Zataari M, Owyang SY, Eaton KA, Liu M, Chang YM, Zou W, Kao JY. TLR2 mediates Helicobacter pylori-induced tolerogenic immune response in mice. PLoS One. 2013;8:e74595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 142. | Otani K, Tanigawa T, Watanabe T, Nadatani Y, Sogawa M, Yamagami H, Shiba M, Watanabe K, Tominaga K, Fujiwara Y. Toll-like receptor 9 signaling has anti-inflammatory effects on the early phase of Helicobacter pylori-induced gastritis. Biochem Biophys Res Commun. 2012;426:342-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 143. | Teymournejad O, Mobarez AM, Hassan ZM, Moazzeni SM, Ahmadabad HN. In vitro suppression of dendritic cells by Helicobacter pylori OipA. Helicobacter. 2014;19:136-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 144. | Hock BD, McKenzie JL, Keenan JI. Helicobacter pylori outer membrane vesicles inhibit human T cell responses via induction of monocyte COX-2 expression. Pathog Dis. 2017;75:ftx034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 145. | Winter J, Letley D, Rhead J, Atherton J, Robinson K. Helicobacter pylori membrane vesicles stimulate innate pro- and anti-inflammatory responses and induce apoptosis in Jurkat T cells. Infect Immun. 2014;82:1372-1381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 146. | Owyang SY, Luther J, Owyang CC, Zhang M, Kao JY. Helicobacter pylori DNA’s anti-inflammatory effect on experimental colitis. Gut Microbes. 2012;3:168-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 147. | Engstrand L, Lindberg M. Helicobacter pylori and the gastric microbiota. Best Pract Res Clin Gastroenterol. 2013;27:39-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 115] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 148. | Kato S, Fujimura S, Kimura K, Nishio T, Hamada S, Minoura T, Oda M. Non-Helicobacter bacterial flora rarely develops in the gastric mucosal layer of children. Dig Dis Sci. 2006;51:641-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 149. | Sanduleanu S, Jonkers D, De Bruine A, Hameeteman W, Stockbrügger RW. Non-Helicobacter pylori bacterial flora during acid-suppressive therapy: differential findings in gastric juice and gastric mucosa. Aliment Pharmacol Ther. 2001;15:379-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 133] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 150. | Sharma BK, Santana IA, Wood EC, Walt RP, Pereira M, Noone P, Smith PL, Walters CL, Pounder RE. Intragastric bacterial activity and nitrosation before, during, and after treatment with omeprazole. Br Med J (Clin Res Ed). 1984;289:717-719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 151] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 151. | Zilberstein B, Quintanilha AG, Santos MA, Pajecki D, Moura EG, Alves PR, Maluf Filho F, de Souza JA, Gama-Rodrigues J. Digestive tract microbiota in healthy volunteers. Clinics (Sao Paulo). 2007;62:47-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 115] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 152. | Bik EM, Eckburg PB, Gill SR, Nelson KE, Purdom EA, Francois F, Perez-Perez G, Blaser MJ, Relman DA. Molecular analysis of the bacterial microbiota in the human stomach. Proc Natl Acad Sci USA. 2006;103:732-737. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 730] [Cited by in RCA: 775] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 153. | Delgado S, Cabrera-Rubio R, Mira A, Suárez A, Mayo B. Microbiological survey of the human gastric ecosystem using culturing and pyrosequencing methods. Microb Ecol. 2013;65:763-772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 160] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 154. | Dicksved J, Lindberg M, Rosenquist M, Enroth H, Jansson JK, Engstrand L. Molecular characterization of the stomach microbiota in patients with gastric cancer and in controls. J Med Microbiol. 2009;58:509-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 242] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 155. | Li XX, Wong GL, To KF, Wong VW, Lai LH, Chow DK, Lau JY, Sung JJ, Ding C. Bacterial microbiota profiling in gastritis without Helicobacter pylori infection or non-steroidal anti-inflammatory drug use. PLoS One. 2009;4:e7985. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 189] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 156. | Maldonado-Contreras A, Goldfarb KC, Godoy-Vitorino F, Karaoz U, Contreras M, Blaser MJ, Brodie EL, Dominguez-Bello MG. Structure of the human gastric bacterial community in relation to Helicobacter pylori status. ISME J. 2011;5:574-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 235] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 157. | Tan MP, Kaparakis M, Galic M, Pedersen J, Pearse M, Wijburg OL, Janssen PH, Strugnell RA. Chronic Helicobacter pylori infection does not significantly alter the microbiota of the murine stomach. Appl Environ Microbiol. 2007;73:1010-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 158. | Schulz C, Schütte K, Koch N, Vilchez-Vargas R, Wos-Oxley ML, Oxley APA, Vital M, Malfertheiner P, Pieper DH. The active bacterial assemblages of the upper GI tract in individuals with and without Helicobacter infection. Gut. 2018;67:216-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 144] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 159. | Dong Q, Xin Y, Wang L, Meng X, Yu X, Lu L, Xuan S. Characterization of Gastric Microbiota in Twins. Curr Microbiol. 2017;74:224-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 160. | Martin ME, Bhatnagar S, George MD, Paster BJ, Canfield DR, Eisen JA, Solnick JV. The impact of Helicobacter pylori infection on the gastric microbiota of the rhesus macaque. PLoS One. 2013;8:e76375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 161. | Brawner KM, Kumar R, Serrano CA, Ptacek T, Lefkowitz E, Morrow CD, Zhi D, Kyanam-Kabir-Baig KR, Smythies LE, Harris PR. Helicobacter pylori infection is associated with an altered gastric microbiota in children. Mucosal Immunol. 2017;10:1169-1177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 70] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 162. | Bauerfeind P, Garner R, Dunn BE, Mobley HL. Synthesis and activity of Helicobacter pylori urease and catalase at low pH. Gut. 1997;40:25-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 168] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 163. | Wen Y, Feng J, Scott DR, Marcus EA, Sachs G. The HP0165-HP0166 two-component system (ArsRS) regulates acid-induced expression of HP1186 alpha-carbonic anhydrase in Helicobacter pylori by activating the pH-dependent promoter. J Bacteriol. 2007;189:2426-2434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 49] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 164. | Williams CL, Preston T, Hossack M, Slater C, McColl KE. Helicobacter pylori utilises urea for amino acid synthesis. FEMS Immunol Med Microbiol. 1996;13:87-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 65] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 165. | Ziebarth D, Spiegelhalder B, Bartsch H. N-nitrosation of medicinal drugs catalysed by bacteria from human saliva and gastro-intestinal tract, including Helicobacter pylori. Carcinogenesis. 1997;18:383-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 64] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 166. | Aebischer T, Fischer A, Walduck A, Schlötelburg C, Lindig M, Schreiber S, Meyer TF, Bereswill S, Göbel UB. Vaccination prevents Helicobacter pylori-induced alterations of the gastric flora in mice. FEMS Immunol Med Microbiol. 2006;46:221-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 47] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 167. | Parsons BN, Ijaz UZ, D’Amore R, Burkitt MD, Eccles R, Lenzi L, Duckworth CA, Moore AR, Tiszlavicz L, Varro A. Comparison of the human gastric microbiota in hypochlorhydric states arising as a result of Helicobacter pylori-induced atrophic gastritis, autoimmune atrophic gastritis and proton pump inhibitor use. PLoS Pathog. 2017;13:e1006653. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 149] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 168. | Hansson LE, Nyrén O, Hsing AW, Bergström R, Josefsson S, Chow WH, Fraumeni JF Jr, Adami HO. The risk of stomach cancer in patients with gastric or duodenal ulcer disease. N Engl J Med. 1996;335:242-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 430] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 169. | Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N, Schlemper RJ. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3126] [Cited by in RCA: 3181] [Article Influence: 132.5] [Reference Citation Analysis (0)] |
| 170. | Guerre J, Vedel G, Gaudric M, Paul G, Cornuau J. Bacterial flora in gastric juice taken at endoscopy in 93 normal subjects. Pathol Biol (Paris). 1986;34:57-60. [PubMed] |
| 171. | Espinoza JL, Matsumoto A, Tanaka H, Matsumura I. Gastric microbiota: An emerging player in Helicobacter pylori-induced gastric malignancies. Cancer Lett. 2018;414:147-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 79] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 172. | Goddard AF, Spiller RC. The effect of omeprazole on gastric juice viscosity, pH and bacterial counts. Aliment Pharmacol Ther. 1996;10:105-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 173. | Navabi N, Johansson ME, Raghavan S, Lindén SK. Helicobacter pylori infection impairs the mucin production rate and turnover in the murine gastric mucosa. Infect Immun. 2013;81:829-837. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 68] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 174. | Manson JM, Rauch M, Gilmore MS. The commensal microbiology of the gastrointestinal tract. Adv Exp Med Biol. 2008;635:15-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 100] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 175. | Akcam M, Ozdem S, Yilmaz A, Gultekin M, Artan R. Serum ferritin, vitamin B(12), folate, and zinc levels in children infected with Helicobacter pylori. Dig Dis Sci. 2007;52:405-410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 176. | Capurso G, Marignani M, Delle Fave G, Annibale B. Iron-deficiency anemia in premenopausal women: why not consider atrophic body gastritis and Helicobacter pylori role? Am J Gastroenterol. 1999;94:3084-3085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 177. | Gao XX, Ge HM, Zheng WF, Tan RX. NMR-based metabonomics for detection of Helicobacter pylori infection in gerbils: which is more descriptive. Helicobacter. 2008;13:103-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 178. | Klymiuk I, Bilgilier C, Stadlmann A, Thannesberger J, Kastner MT, Högenauer C, Püspök A, Biowski-Frotz S, Schrutka-Kölbl C, Thallinger GG. The Human Gastric Microbiome Is Predicated upon Infection with Helicobacter pylori. Front Microbiol. 2017;8:2508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 78] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 179. | Jones TA, Hernandez DZ, Wong ZC, Wandler AM, Guillemin K. The bacterial virulence factor CagA induces microbial dysbiosis that contributes to excessive epithelial cell proliferation in the Drosophila gut. PLoS Pathog. 2017;13:e1006631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 180. | Yap TW, Gan HM, Lee YP, Leow AH, Azmi AN, Francois F, Perez-Perez GI, Loke MF, Goh KL, Vadivelu J. Helicobacter pylori Eradication Causes Perturbation of the Human Gut Microbiome in Young Adults. PLoS One. 2016;11:e0151893. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 104] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 181. | Heimesaat MM, Fischer A, Plickert R, Wiedemann T, Loddenkemper C, Göbel UB, Bereswill S, Rieder G. Helicobacter pylori induced gastric immunopathology is associated with distinct microbiota changes in the large intestines of long-term infected Mongolian gerbils. PLoS One. 2014;9:e100362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 73] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 182. | Majlessi L, Sayes F, Bureau JF, Pawlik A, Michel V, Jouvion G, Huerre M, Severgnini M, Consolandi C, Peano C. Colonization with Helicobacter is concomitant with modified gut microbiota and drastic failure of the immune control of Mycobacterium tuberculosis. Mucosal Immunol. 2017;10:1178-1189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 183. | Gebert B, Fischer W, Weiss E, Hoffmann R, Haas R. Helicobacter pylori vacuolating cytotoxin inhibits T lymphocyte activation. Science. 2003;301:1099-1102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 404] [Cited by in RCA: 409] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 184. | Wüstner S, Mejías-Luque R, Koch MF, Rath E, Vieth M, Sieber SA, Haller D, Gerhard M. Helicobacter pylori γ-glutamyltranspeptidase impairs T-lymphocyte function by compromising metabolic adaption through inhibition of cMyc and IRF4 expression. Cell Microbiol. 2015;17:51-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 185. | Khosravi Y, Loke MF, Goh KL, Vadivelu J. Proteomics Analysis Revealed that Crosstalk between Helicobacter pylori and Streptococcus mitis May Enhance Bacterial Survival and Reduces Carcinogenesis. Front Microbiol. 2016;7:1462. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 186. | Boyanova L, Gergova G, Markovska R, Yordanov D, Mitov I. Bacteriocin-like inhibitory activities of seven Lactobacillus delbrueckii subsp. bulgaricus strains against antibiotic susceptible and resistant Helicobacter pylori strains. Lett Appl Microbiol. 2017;65:469-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 187. | Salas-Jara MJ, Sanhueza EA, Retamal-Díaz A, González C, Urrutia H, García A. Probiotic Lactobacillus fermentum UCO-979C biofilm formation on AGS and Caco-2 cells and Helicobacter pylori inhibition. Biofouling. 2016;32:1245-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 188. | Zheng PX, Fang HY, Yang HB, Tien NY, Wang MC, Wu JJ. Lactobacillus pentosus strain LPS16 produces lactic acid, inhibiting multidrug-resistant Helicobacter pylori. J Microbiol Immunol Infect. 2016;49:168-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 189. | Elitsur Y, Neace C, Werthammer MC, Triest WE. Prevalence of CagA, VacA antibodies in symptomatic and asymptomatic children with Helicobacter pylori infection. Helicobacter. 1999;4:100-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 190. | Hooper LV, Littman DR, Macpherson AJ. Interactions between the microbiota and the immune system. Science. 2012;336:1268-1273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3003] [Cited by in RCA: 2985] [Article Influence: 229.6] [Reference Citation Analysis (0)] |
| 191. | Rolig AS, Cech C, Ahler E, Carter JE, Ottemann KM. The degree of Helicobacter pylori-triggered inflammation is manipulated by preinfection host microbiota. Infect Immun. 2013;81:1382-1389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 192. | Pei X, Ouyang X, He L, Cao C, Luan Q, Suda R. A 4-year prospective study of the progression of periodontal disease in a rural Chinese population. J Dent. 2015;43:192-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |