Published online Mar 14, 2018. doi: 10.3748/wjg.v24.i10.1063
Peer-review started: December 15, 2017
First decision: December 27, 2017
Revised: February 2, 2018
Accepted: February 9, 2018
Article in press: February 9, 2018
Published online: March 14, 2018
Processing time: 88 Days and 15 Hours
Iron deficiency without anemia and iron deficiency anemia are common and frequently overlooked complications of inflammatory bowel disease. Despite the frequency and impact of iron deficiency in inflammatory bowel disease, there are gaps in our understanding about its incidence, prevalence and natural history and, consequently, patients may be undertreated. Medical registries have a key role in collecting data on the disease’s natural history, the safety and effectiveness of drugs in routine clinical practice, and the quality of care delivered by healthcare services. Even though iron deficiency impacts inflammatory bowel disease patients and healthcare systems substantially, none of the established European inflammatory bowel disease registries systematically collects information on iron parameters and related outcomes. Collection of robust iron parameter data from patient registries is one way to heighten awareness about the importance of iron deficiency in this disease and to generate data to improve the quality of patient care, patient outcomes, and thus quality of life. This objective could be achieved through collection of specific laboratory, clinical, and patient-reported measurements that could be incorporated into existing registries. This review describes the status of current European inflammatory bowel disease registries and the data they generate, in order to highlight their potential role in collecting iron data, to discuss how such information gathering could contribute to our understanding of iron deficiency anemia, and to provide practical information in regard to the incorporation of accumulated iron parameter data into registries.
Core tip: Despite its prevalence, iron deficiency is often overlooked in inflammatory bowel disease (IBD). More data are required to fully understand the epidemiology, treatment, and quality of care around iron deficiency in IBD. We suggest that IBD registries are ideally positioned to collect these data from routine clinical practice. We discuss the laboratory, clinical, and patient-reported data that could be collected, and review how best to incorporate collection of these data into existing registries.
- Citation: Halfvarson J, Cummings F, Grip O, Savoye G. Inflammatory bowel disease registries for collection of patient iron parameters in Europe. World J Gastroenterol 2018; 24(10): 1063-1071
- URL: https://www.wjgnet.com/1007-9327/full/v24/i10/1063.htm
- DOI: https://dx.doi.org/10.3748/wjg.v24.i10.1063
The current healthcare landscape relies heavily on controlled trials to generate evidence to support regulatory approval of new drugs and inform treatment of patients[1-4]. While scientifically rigorous, the applicability to everyday clinical situations might be questionable, since it does not necessarily include a cohort that is representative of the real-world patient population[5]. In contrast, registries have a key role in acquiring, maintaining, analyzing and, ultimately, publishing data that reveal how drugs are used in routine clinical practice. Medical registries can survey the demographics of patients with a particular medical condition, collect data on the use of a particular pharmaceutical product, class of products or use of a particular medical device, or be used to evaluate the quality of care delivered. Consequently, the use of national registries to generate real-world data is of great value, particularly in complex diseases such as inflammatory bowel disease (IBD), namely Crohn’s disease and ulcerative colitis.
Iron deficiency anemia (IDA) is ranked as one of the world’s ten leading causes of years lived with disability[6] and can manifest in a broad range of symptoms, including chronic fatigue, increased susceptibility to infection, tachycardia, sleep disturbance, impaired cognitive function, hair loss, and changes to skin and nails[7]. Iron deficiency without anemia (ID) and IDA are two of the most common systemic complications of IBD; estimates of prevalence indicate that ID occurs in 60%-80% of people with IBD, with the prevalence of anemia ranging from 16% to 74%[8].
For patients with IBD, IDA can be caused by a variety of factors[8] and affect a range of physical, personal, and social parameters, impairing work productivity and ultimately compounding the diminished quality of life (QoL) associated with IBD alone[9]. The economic burden of IBD is elevated by the presence of IDA; a retrospective review of healthcare claims data in the US estimated the direct treatment costs for each IBD patient to be substantially higher for those with anemia versus those without[10].
The natural history of IDA in IBD is not fully understood and its occurrence remains underestimated[11], so patients are potentially undertreated. The gaps in our understanding require comprehensive longitudinal data collection-an important source of which may be the collection of iron parameter data from patient registries. The objectives of this review article are to describe the status of current European IBD registries and the data they generate, to highlight the potential role of IBD registries in collecting IDA data and contributing to our understanding of IDA, and to provide practical information in regard to the incorporation of collated iron parameter data into IBD registries.
A literature search was conducted to identify IBD registries that are currently collecting data in Europe, using the PubMed online database and internet searches. The search term “inflammatory bowel disease registry” was used and results were reviewed to identify articles meeting the inclusion criteria. The reference lists of any article meeting the inclusion criteria were reviewed manually to identify additional relevant publications. Where articles were identified, the name of the registry was used as a search term in the Google internet search engine in an attempt to find further relevant information. Where a web page did not provide information in English, translation software was used to determine if the information was relevant. For inclusion in this review, studies were required to have met the following criteria, in regard to the registry: (1) Being disease-specific with information on IBD; (2) being located in a European country; and (3) currently collecting data. Registries that were specific to treatment, and not disease, were excluded. Details of the registries identified are shown in Table 1. Note that the list of identified registries presented here may not be exhaustive, as small local or regional registries may not be visible to such a search, and this work is not intended to be a complete systematic review.
| Name | Country | Year established | Description and aim | IBD Study population | Data collected |
| Competence Network IBD Registry[27] | Germany | 2015 | Scientists, physicians, clinics, research institutes, and industry with an interest in research to improve the care of patients with IBD | Approximately 50000 (anticipated) | IBD frequency and course; comorbidity incidence; efficacy of treatment; predictive parameters; subgroup characterization; outpatient vs inpatient costing; guideline implementation; service delivery |
| ENEIDA[28,29] | Spain | 2006 | Promotion of clinical and genetic studies in IBD, epidemiology of IBD, clinical outcomes, and drug safety in IBD treatment | > 11000 | Patient demographics, disease classification, treatment outcomes and safety, phenotype, and family history of IBD |
| EPIMAD[18,30-34] | Northern France | 1988 | To provide reliable data on the epidemiology of IBD to healthcare authorities, provide data to search for a cause of disease, and describe in a population-based setting the natural history and real-life management of IBD | All IBD patients in region (approximately 20000 patients) | Patient demographics and disease data |
| SWIBREG[35] | Sweden | 2005 | To serve as a decision support tool in everyday life, assessing disease activity and quality of life | > 40000 (adult and pediatric) | Patient demographics and disease data, drug history, treatment and disease outcomes, surgical interventions, patient-reported outcomes. Capacity to enter information about comorbidities |
| SIBDC[12] | Switzerland | 2005 | To build understanding of the consequences of IBD on the physical, mental, and social conditions of IBD patients | Voluntary national database of > 2500 patients | Patient demographics, disease and treatment information. Patient blood samples are kept in a biobank providing a databank of genetic and disease information |
| UK IBD Registry[13] | United Kingdom | 2013 | To drive improvement in patient care and access to care across the United Kingdom, inform commissioning and service design, improve understanding of long, term outcomes in IBD, provide local, regional, and national data in order to better define the pattern of ulcerative colitis and Crohn’s disease, and support IBD research | Approximately 20000 (adult and pediatric) | Patient demographics, disease data, any surgical interventions, drug history, treatment and disease outcomes, disease activity scores and patient-reported outcomes |
| Pediatric registries | |||||
| CEDATA[36] | Austria and Germany | 2004 | Developed and operated by the working group of chronic inflammatory bowel diseases of the Society for Pediatric Gastroenterology and Nutrition; aims to improve the care of children and adolescents with IBD. | Approximately 1000 | Patient demographics, disease and treatment data |
| SPIRIT-IBD[37] | Spain | 1996 | To understand the incidence and prevalence of IBD in pediatric patients | Approximately 2000 | Patient demographics, disease and treatment data |
| EUROKIDS[38,39] | Europe and Israel | 2004 | To audit the diagnostic workup of pediatric IBD patients and to accurately describe disease in newly diagnosed pediatric IBD patients | Approximately 4000 | Patient demographics, disease and treatment data |
Items common to the IBD registries include: number of patients with IBD in the region covered, information on clinical characteristics, medication, treatment effectiveness, and service delivery. Some information is registry specific, such as biobanking of blood samples for genotyping, as within the Swiss cohort[12]. It is notable that, despite the prevalence of ID/IDA among IBD patients, none of the registries identified explicitly were collecting information on iron parameters and related outcomes in a systematic manner at the time of this review.
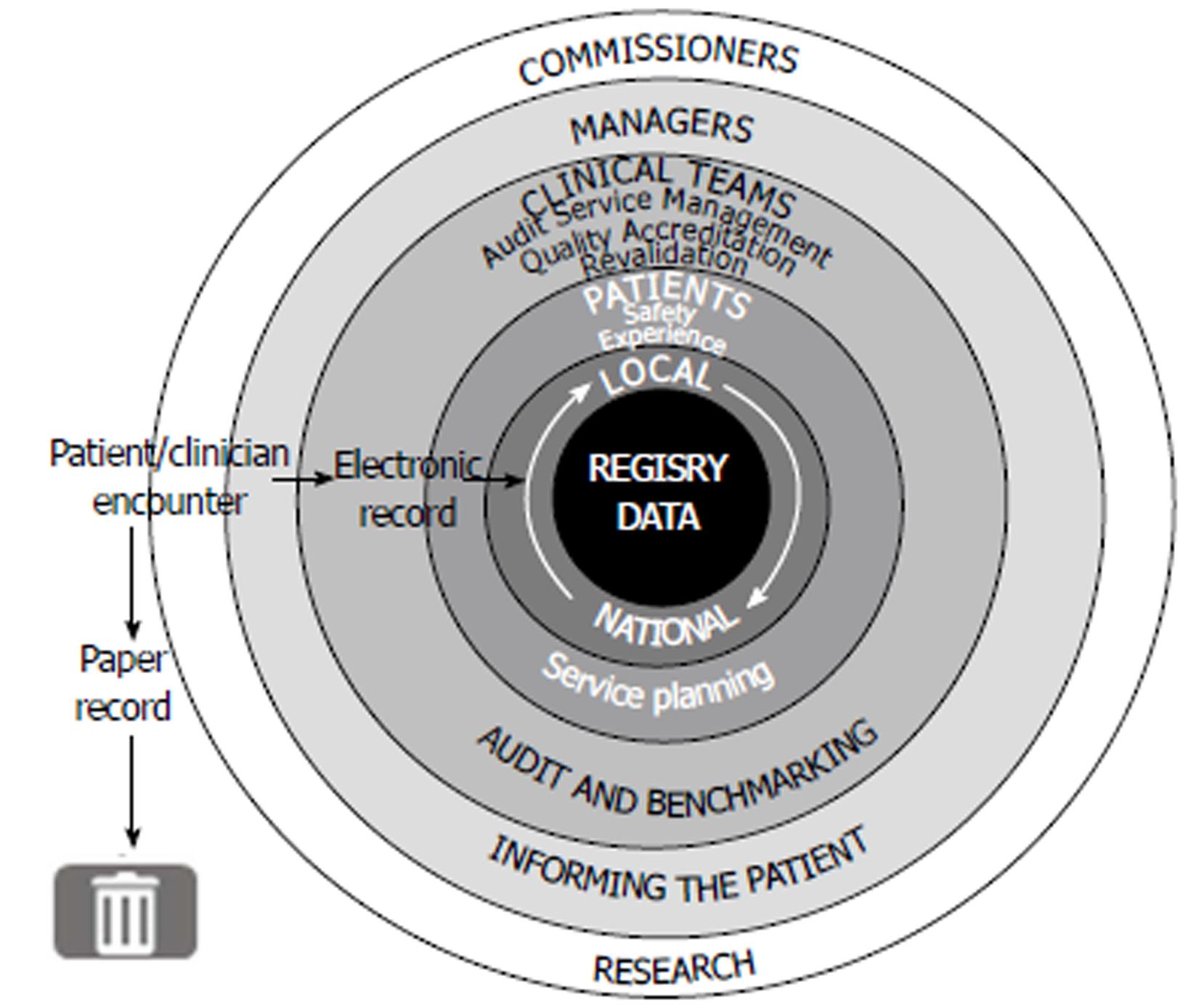
The approach to data collection and verification varies among registries. For example, in EPIMAD (Registre des Maladies Inflammatoires Chroniques de l’Intestin du Nord Ouest de la France), data are collected by interviewer practitioners when possible cases of IBD are identified, and a diagnosis of IBD is verified by two expert practitioners. Each patient can be followed, if necessary, for 2 years until a final diagnosis is confirmed. In contrast, the UK IBD Registry utilizes electronic data-entry systems, such that data are collected in real time at the point of care (Figure 1). Some of the data-entry tools allow for the upload of laboratory data directly into the system, such that, after negotiations with the relevant providers, iron parameters could be added to the data collected with relative ease[13]. Similarly, the Swedish Quality Registry for Inflammatory Bowel Disease (SWIBREG) - which is notable in being well established and in covering half of the Swedish IBD population-can be linked to other national registers to evaluate etiology, prognosis, and outcomes of different treatment modalities[14].
Despite the coverage of these registries, the initial research questions used, the configuration of data-collection systems, and available resources (both financial and personnel) all impose limits on the data they can gather. Information about medications to treat conditions concomitant to IBD, such as ID and IDA, may not be captured. Similarly, the collection and inclusion of patient-reported outcome (PRO) data in existing IBD registries is not a trivial undertaking. Given the importance of ID-related symptoms, the collection and analysis of PRO questionnaires is important and should be considered.
Although national healthcare databases collect and store data from blood tests on anemias, ID, and the drugs used in their treatment, they do not seem to be an appropriate way to collect data on IDA in IBD. In general, such databases are optimized to collect high-level administrative data, and do not describe IBD patients adequately to offer the level of medical detail needed to research the natural history of disease. This is also the case for databases owned by agencies involved in reimbursement of medical treatment. From this, a clear need for prospective and specific collection of iron parameter data can be established, which may be achieved through the adaptation of existing IBD registries.
Even though IDA and ID are common in IBD, and exert a substantial burden on patients and healthcare systems[8-10], none of the established European IBD registries systematically collects information on iron parameters and related outcomes. Addressing this shortfall in data collection requires examination of the primary objectives of the registries in question, and consideration of parameters that could answer further research questions around this objective. The parameters collected would be determined by the type of registry (condition-, product-, or service-based) and should serve its primary research objective; suggestions are listed in Table 2.
| Laboratory measurements |
| Hemoglobin level |
| Serum ferritin level |
| Transferrin saturation level |
| Soluble transferrin receptor level |
| Hepcidin |
| Mean corpuscular volume |
| Mean corpuscular hemoglobin |
| C-reactive protein level |
| Clinical observations |
| Identification of ID/IDA-related symptoms |
| Duration of possible ID/IDA-related symptoms |
| Number of, duration of, and reasons for healthcare visits |
| Medication usage |
| Patient-reported outcomes |
| Quality-of-life questionnaires |
| Fatigue questionnaires |
| Work productivity and activity questionnaires |
| Treatment decisions |
| Iron supplement (oral or IV) |
| IBD medication |
| Surgical procedures |
A condition-based registry designed to monitor epidemiology of IBD could also collect data to provide snapshots of the prevalence and severity of IDA. Longitudinal analyses of IDA duration and disease course, with the potential to identify commonly associated factors-including ID-related burden occurring during pregnancy-could also be included. To chronicle the natural history of IDA, it is necessary to collect longitudinal information on clinical, biochemical and endoscopic disease activity; data from imaging techniques such as magnetic resonance imaging should ideally be included. In line with the current European Crohn’s and Colitis Organisation (ECCO) consensus on the diagnosis and management of ID with and without anemias in IBD[15], a basic dataset may comprise hemoglobin levels, transferrin saturation and serum ferritin levels, and perhaps mean corpuscular volume and mean corpuscular hemoglobin. Measuring C-reactive protein would inform the interpretation of serum ferritin levels, which may be elevated by inflammation. Recording the levels of individual iron parameters (rather than a diagnosis of anemia) would offer a more detailed view of ID and the capacity to investigate concepts such as absolute and functional ID[15]. To further understand the impact that ID and IDA have on disease course and its influence on the response to treatment the registry would need to collect medication history and validated disease activity scores/PROs that evaluate fatigue, QoL, and impaired productivity. Fatigue is a key symptom of ID (and of IBD) and can be quantified using a validated scoring system such as the functional assessment of chronic illness therapy-fatigue (FACIT-F)-Fatigue questionnaire[16]. There are several tools available to measure QoL in patients with IBD or, more recently, disability[17,18], although these do not necessarily distinguish between the burden of ID/IDA and IBD.
A product-based registry (focused on IBD treatment) could be used to monitor use of iron supplements. Information on prescribed iron replacement therapy, including doses, may be recorded as either oral or intravenous, to illustrate any impact of a different choice of iron administration route.
A service-based registry would typically have a different dataset, including information on the use of healthcare (including hospitalizations and surgery), and the resource utilization attributable to ID and IDA in IBD. Collection of data on screening, diagnosis, and monitoring of ID and IDA in IBD could offer a perspective on aspects of quality of care, with reference to the recommendations of the European Consensus on the Diagnosis and Management of Iron Deficiency and Anaemia in Inflammatory Bowel Diseases[15]. If an economic perspective is deemed valuable, observations on impaired work productivity and activity can be collected, as well as information on absences from work or full-time education, healthcare attendance, need for surgery, and medication adherence and persistence.
A registry would ideally use appropriate digital and online technologies to make it easier for sites to contribute data, including enabling data entry at point of care and a facility for patients to submit disease-related information. Incentives for teams to use these technologies might include the opportunity to deliver data-driven care to patients. Patients may also be given access to presentations of these data to facilitate supported self-management, and improve patient participation and empowerment. An example of success in this area is Improve Care Now-a network of people from 95 care centers in the United States, Qatar, and United Kingdom that has the objective of improving the health, care, and costs for children and adolescents with IBD[19]. The centers involved in this initiative collect data about treatment and performance in a standardized format; the tools for best practice in the treatment of these patients, with the related evidence, is shared within the network, leading to substantial improvements in patient outcomes and better patient safety[20].
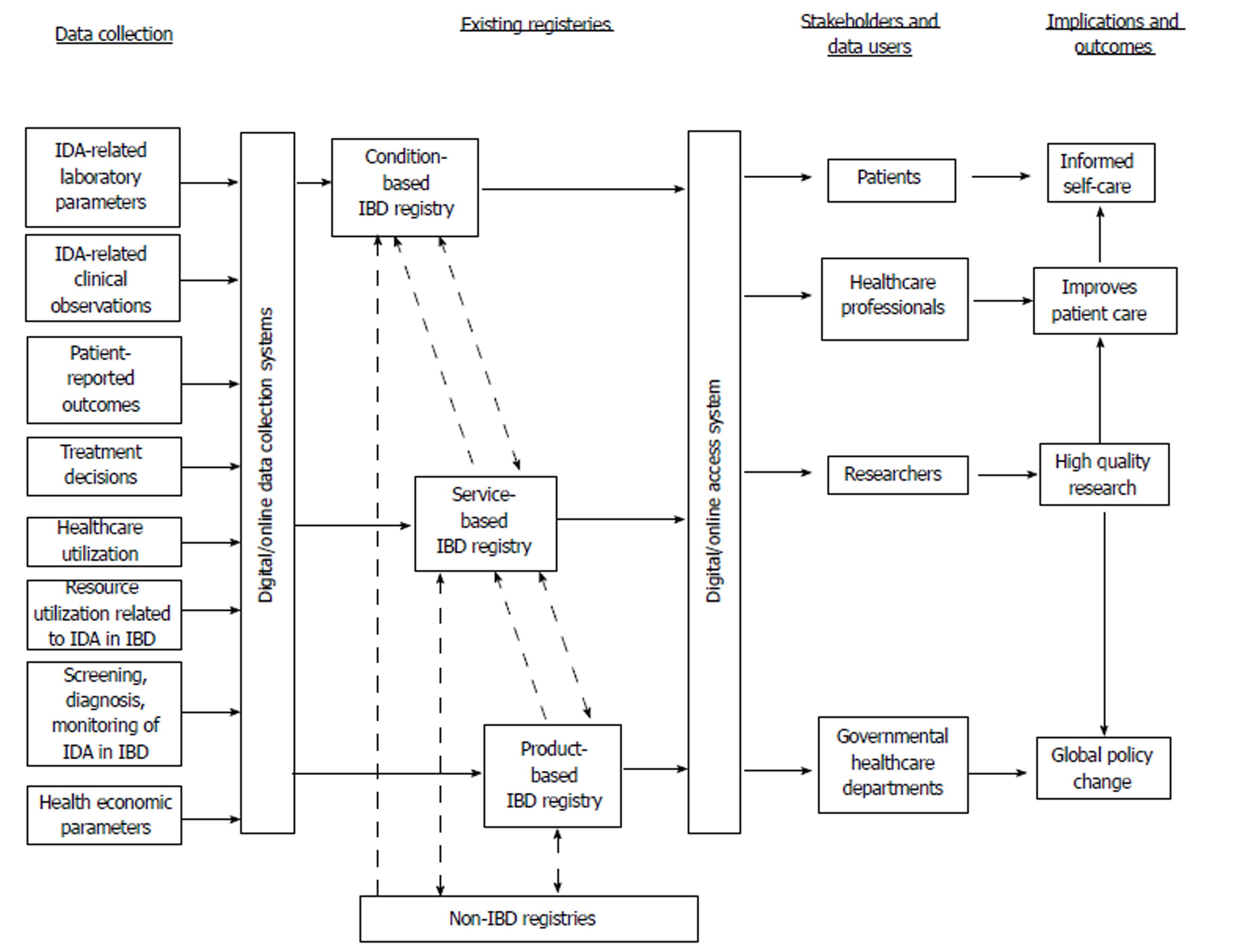
Cross-registry interoperability and integration, to facilitate sharing of data with existing healthcare systems and resources, would be highly beneficial; Figure 2 illustrates a potential model for how data could move between registries and shows the potential outcomes for stakeholders. Collection and incorporation of unique pseudoanonymized national personal identifiers of patients enables connections to be made between registries. IDA is prevalent in patients with renal disease, heart disease, and cancer, and can affect cognitive development/function and resistance to infection[21,22]. Correlation between disease registries in IBD, oncology, heart failure, and mental health could offer completely new perspectives on improving the multidisciplinary care of patients with ID. Further connections could be made with drug prescription databases with the intention of correlating disease outcomes with specific treatment information. Linkage of an IBD registry with national service-based registries could show the frequency of outpatient visits, inpatient care, surgery, and co-existing disease, based on the international classification of disease codes. Linkage with mortality databases would provide an additional view of disease outcomes. A potential barrier to sharing of data is the governance issues around information. Any such sharing should comply with the registry’s ethical requirements and national laws covering data protection. If there are restrictions on exporting personal data from the country, steps must be taken to anonymize and aggregate the information. It is also important to understand exactly what is of interest to a collaborating registry, and to identify and characterize all variables collected, as these may vary between registries[14].
The current IBD registries are positioned well to collect iron parameter data and link these with IBD-specific data. Although it is beyond the scope of this article to provide detailed guidance on adapting a registry to collect a new set of variables, there are several factors that should be considered, in regard to the prospective collection of iron parameters in an IBD registry. Initially, it is important to consider the change in scope of a registry, in terms of its expected redefined purpose, objectives, and overall outputs. While it may be tempting to maximize the opportunities for data collection, this must be balanced against the importance of collecting high-quality, reliable data with no significant increased burden associated with their entry to the database. Limiting the scope of a registry ensures that specific objectives and well-defined research questions can be established in order to specify what outputs should be sought. Defining and confirming the scope of any changes with all stakeholders will optimize the work needed as the changes to the registry are implemented. When the objectives have been clearly established, the data to be collected can then be determined. The data collection method should suit the data type. The practicalities of incorporating collection of iron parameters into an existing IBD registry could present a challenge, as it would depend on the healthcare system in which the registry operates. Ideally, the addition of new parameters to a registry should not generate substantially more work for sites contributing to the registry. With the introduction of electronic medical records and network integration it is theoretically possible for iron parameter data from medical laboratories to be automatically added to a medical record and uploaded to a registry database. This would greatly minimize the resources needed for data entry, avoiding the review and entry of information from unstructured case notes, and circumventing double entry of data. However, there are currently barriers to this approach. Firstly, retrieval of data from laboratory service providers requires negotiation, financial commitment, and often improvement of IT facilities. This problem is magnified if a country has multiple service providers, each of which is presenting data in a proprietary format, thus requiring a large amount of interface development work. Secondly, the use of electronic medical records is still not widely used, and standards of interoperability are yet to be established. The functioning of a registry could be complicated if data source inputs present information in a range of formats that have to be interpreted before they can be used. Many commercial providers offer digital and online interfaces to facilitate data capture in a registry; however, there can be great variation between them in terms of financial cost, ease-of-use, and features (not all of which may be necessary or relevant to an individual registry)[23]. Indeed, the quality and capabilities of different software used to manage data in clinical studies and registries can vary greatly, raising barriers in interoperability and evaluation of data across studies[24]. Open-source software offers an alternative that is both low-cost and can be modified by researchers to suit specific requirements[24]. A key role for national and international registries would be to define data submission frameworks that enable the entry of data from a range of different sources.
The rise in health technologies that are accessible from personal mobile devices is likely to have an important role in collection of PROs. Electronic PRO data collection has many advantages relating to easy, accurate, and complete collection of data that is potentially of great value to investigators and healthcare systems[25]. In the context of a registry, as opposed to a clinical trial, it may make sense for a patient to be able to complete PRO information using their own mobile device, rather than attending an appointment. Although this adds a level of convenience for both patient and healthcare team, a number of barriers still remain including data security, patient compliance with data collection, and what to do if a patient does not possess a suitable device[25]. Nevertheless, ePROs show promise; indeed, pilot studies of an electronic data submission method implemented by the SWIBREG registry have proved successful and its launch is imminent in some regions in Sweden. A UK study to collect ePROs in an elective orthopedic setting showed great promise, gathering reliable data but also highlighting that the ePRO system may not be adequate for some patients[26].
The incorporation of iron parameter data into European IBD registries offers great opportunities to improve the care of IBD patients. Firstly, highlighting the prevalence of IDA in the IBD population would show the unmet need in this patient group in countries across Europe. It is notable that the ECCO guidelines for diagnosis and management of ID and IDA in IBD recommend that all patients with IBD should be assessed for the presence of anemia[15]. Robust data are necessary to heighten awareness of the importance of IDA in IBD and to lobby healthcare systems to improve access to the full range of iron supplementation therapies. Secondly, longitudinal analysis of iron parameters and treatment could offer a perspective to assess some aspects of the quality of care that patients receive, including the management of IDA and ID, as well as the numbers of patients who receive optimal treatment. Thirdly, the inclusion of QoL information and PROs would offer perspectives on how optimal treatment could change the burden of IDA on the patient, as well as potentially providing economic arguments for the use of iron supplementation. Finally, the information collected could inform service planning at the local and national level, including on how resources should be managed to provide improved access to nursing support or access to infusion suites. Periodic assessment of the data collected in national registries would also provide information needed for clinical audits and quality improvement programs.
Beyond the local and national level, the collection and comparison of iron parameters by multiple registries would allow the relationship between IBD and IDA to be examined from different perspectives, stemming from each registry’s specific remit, leading to the generation of new research questions and ultimately improving patient care. A further possibility would be the pooling of data between national registries to identify and study any differences in patterns of disease, and to compare practices in healthcare service delivery. A very large dataset should also able to highlight any rare effects that might not be evident from a single national registry. This initiative would, of course, require agreement on the collection of a basic dataset and standardization of methodologies, as well as addressing how data should be anonymized and aggregated to comply with the legal aspects of sharing data across national borders.
Iron deficiency and IDA are common and often overlooked complications of IBD. Historically, ID and IDA have been accepted as part of IBD, and not as an issue to be addressed and treated independently. The prevailing attitude was that treatment of IBD would lead to the resolution of anemia, so organized research efforts to understand ID in patients with IBD were minimal. In recent years, however, opinion has shifted, with a growing acknowledgement that ID/IDA is an issue to be managed concomitantly with IBD. A recent consensus statement provides up-to-date guidance on the diagnosis, treatment and prevention of ID/IDA in IBD[15]. Despite the frequency and impact of ID and IDA in IBD, there are gaps in our understanding about its incidence, prevalence, natural history, and particularly its impact on patients. Due to similarities between symptoms of IBD and ID/IDA, the identification of ID/IDA-specific symptoms in the IBD population is necessary. There is, therefore, a clear need to collect robust, structured data around ID/IDA in IBD, its treatment, and on related service delivery. Despite potential limitations (possible selection bias and potential variability in data quality), it has been suggested that ‘real-world’ data have a greater applicability to the routine clinical setting than the findings of controlled trials[5]. Current digital technologies and networking capabilities raise the prospect of an acquisition and analysis of real-world data that is (and should be) easier than ever before. With appropriate planning, information governance structures, adaptation, and negotiation, registries are ideally placed to collect this information in a routine clinical practice setting. The direct input by patients of self-reported data can capture and quantify symptoms such as fatigue and cognitive impairment, which can otherwise be difficult to evaluate. If multiple registries were to commence collection of ID/IDA data in IBD as a standard, there would be an opportunity to compare and pool data collected from multiple sources. Although this endeavor would be challenging, the ultimate benefit to the care of patients with IBD would be substantial.
Vifor Pharma provided funding for an initial meeting of authors at which the content of this article was discussed. Under the direction of the authors, AXON Communications provided writing and editorial, assistance in formatting, proofreading, copy editing and fact checking, funded by Vifor Pharma.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Sweden
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Gassler N, Gazouli M, Sergi CM S- Editor: Gong ZM L- Editor:A E- Editor: Ma YJ
| 1. | Gionchetti P, Dignass A, Danese S, Magro Dias FJ, Rogler G, Lakatos PL, Adamina M, Ardizzone S, Buskens CJ, Sebastian S. 3rd European Evidence-based Consensus on the Diagnosis and Management of Crohn’s Disease 2016: Part 2: Surgical Management and Special Situations. J Crohns Colitis. 2017;11:135-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 594] [Cited by in RCA: 531] [Article Influence: 66.4] [Reference Citation Analysis (0)] |
| 2. | Gomollón F, Dignass A, Annese V, Tilg H, Van Assche G, Lindsay JO, Peyrin-Biroulet L, Cullen GJ, Daperno M, Kucharzik T. 3rd European Evidence-based Consensus on the Diagnosis and Management of Crohn’s Disease 2016: Part 1: Diagnosis and Medical Management. J Crohns Colitis. 2017;11:3-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1585] [Cited by in RCA: 1448] [Article Influence: 181.0] [Reference Citation Analysis (0)] |
| 3. | Harbord M, Eliakim R, Bettenworth D, Karmiris K, Katsanos K, Kopylov U, Kucharzik T, Molnár T, Raine T, Sebastian S. Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 2: Current Management. J Crohns Colitis. 2017;11:769-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 938] [Cited by in RCA: 875] [Article Influence: 109.4] [Reference Citation Analysis (0)] |
| 4. | Magro F, Gionchetti P, Eliakim R, Ardizzone S, Armuzzi A, Barreiro-de Acosta M, Burisch J, Gecse KB, Hart AL, Hindryckx P. Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 1: Definitions, Diagnosis, Extra-intestinal Manifestations, Pregnancy, Cancer Surveillance, Surgery, and Ileo-anal Pouch Disorders. J Crohns Colitis. 2017;11:649-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1446] [Cited by in RCA: 1298] [Article Influence: 162.3] [Reference Citation Analysis (0)] |
| 5. | Silverman SL. From randomized controlled trials to observational studies. Am J Med. 2009;122:114-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 239] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 6. | GBD 2015 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388:1545-1602. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5206] [Cited by in RCA: 4834] [Article Influence: 537.1] [Reference Citation Analysis (0)] |
| 7. | Ghosh K. Non haematological effects of iron deficiency - a perspective. Indian J Med Sci. 2006;60:30-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 8. | Stein J, Dignass AU. Management of iron deficiency anemia in inflammatory bowel disease - a practical approach. Ann Gastroenterol. 2013;26:104-113. [PubMed] |
| 9. | Danese S, Hoffman C, Vel S, Greco M, Szabo H, Wilson B, Avedano L. Anaemia from a patient perspective in inflammatory bowel disease: results from the European Federation of Crohn’s and Ulcerative Colitis Association’s online survey. Eur J Gastroenterol Hepatol. 2014;26:1385-1391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 10. | Ershler WB, Chen K, Reyes EB, Dubois R. Economic burden of patients with anemia in selected diseases. Value Health. 2005;8:629-638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 102] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 11. | Stein J, Bager P, Befrits R, Gasche C, Gudehus M, Lerebours E, Magro F, Mearin F, Mitchell D, Oldenburg B. Anaemia management in patients with inflammatory bowel disease: routine practice across nine European countries. Eur J Gastroenterol Hepatol. 2013;25:1456-1463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 12. | Swiss IBD Cohort (SIBDC). Zurich: Department of Gastroenterology and Hepatology, Department of Internal Medicine, University Hospital of Zurich. [cited July 31, 2017]. 2017. Available from: http://www.ibdcohort.ch/. |
| 13. | The UK IBD Registry. London: British Society of Gastroenterology; 2017 [cited July 31, 2017]. Available from: http://ibdregistry.org.uk/. |
| 14. | Brooke H, Holzmann M, Olén O, Talbäck M, Feychting M, Berglund A, Ludvigsson J, Ljung R. Enhancing evidence based medicine: Twelve tips for conducting register-based research. MedEdPublish. 2016;5:43. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 15. | Dignass AU, Gasche C, Bettenworth D, Birgegård G, Danese S, Gisbert JP, Gomollon F, Iqbal T, Katsanos K, Koutroubakis I. European consensus on the diagnosis and management of iron deficiency and anaemia in inflammatory bowel diseases. J Crohns Colitis. 2015;9:211-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 449] [Cited by in RCA: 410] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 16. | Tinsley A, Macklin EA, Korzenik JR, Sands BE. Validation of the functional assessment of chronic illness therapy-fatigue (FACIT-F) in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2011;34:1328-1336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 107] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 17. | Alrubaiy L, Rikaby I, Dodds P, Hutchings HA, Williams JG. Systematic review of health-related quality of life measures for inflammatory bowel disease. J Crohns Colitis. 2015;9:284-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 60] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 18. | Gower-Rousseau C, Sarter H, Savoye G, Tavernier N, Fumery M, Sandborn WJ, Feagan BG, Duhamel A, Guillon-Dellac N, Colombel JF, Peyrin-Biroulet L; and the International Programme to Develop New Indexes for Crohn’s Disease (IPNIC) group; International Programme to Develop New Indexes for Crohn’s Disease (IPNIC) group. Validation of the Inflammatory Bowel Disease Disability Index in a population-based cohort. Gut. 2017;66:588-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 126] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 19. | Improve Care Now [internet]. 2017 [cited July 31, 2017]. Available from: http://www.improvecarenow.org/. |
| 20. | Crandall WV, Margolis PA, Kappelman MD, King EC, Pratt JM, Boyle BM, Duffy LF, Grunow JE, Kim SC, Leibowitz I. Improved outcomes in a quality improvement collaborative for pediatric inflammatory bowel disease. Pediatrics. 2012;129:e1030-e1041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 165] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 21. | Abbaspour N, Hurrell R, Kelishadi R. Review on iron and its importance for human health. J Res Med Sci. 2014;19:164-174. [PubMed] |
| 22. | Peyrin-Biroulet L, Williet N, Cacoub P. Guidelines on the diagnosis and treatment of iron deficiency across indications: a systematic review. Am J Clin Nutr. 2015;102:1585-1594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 199] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 23. | Shah J, Rajgor D, Pradhan S, McCready M, Zaveri A, Pietrobon R. Electronic Data Capture for Registries and Clinical Trials in Orthopaedic Surgery: Open Source versus Commercial Systems. Clin Orthop Relat Res. 2010;468:2664-2671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 51] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 24. | Müller J, Heiss KI, Oberhoffer R. Implementation of an open adoption research data management system for clinical studies. BMC Res Notes. 2017;10:252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 25. | Coons SJ, Eremenco S, Lundy JJ, O’Donohoe P, O’Gorman H, Malizia W. Capturing Patient-Reported Outcome (PRO) Data Electronically: The Past, Present, and Promise of ePRO Measurement in Clinical Trials. Patient. 2015;8:301-309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 144] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 26. | Malhotra K, Buraimoh O, Thornton J, Cullen N, Singh D, Goldberg AJ. Electronic capture of patient-reported and clinician-reported outcome measures in an elective orthopaedic setting: a retrospective cohort analysis. BMJ Open. 2016;6:e011975. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 27. | Competence Network Inflammatory Bowel Disease. Call for tenders for the Project: Long-term Observation of Patients with Chronic Inflammatory Bowel Disease: Establishment of a Registry [information document]. [cited July 31, 2017]. Kiel: Competence Network Inflammatory Bowel Disease 2013; Available from: http://www.kompetenznetz-ced.de/Aktuelle_Meldung/items/ausschreibung-zur-einrichtung-eines-nationalen-ced-registers-91.html?file=tl_files/Downloads/PDF/Presse/2014/Ausschreibung_Langzeitregister_KN%20CED.pdf. |
| 28. | Grupo Espanol de Trabajo en Enfermdad de Crohn y Colitis Ulcerosa [Internet]. Bilbao: Grupo Espanol de Trabajo en Enfermdad de Crohn y Colitis Ulcerosa [internet]; 2016 Características y objetivos de ENEIDA [Features and objectives of ENEIDA]. [cited July 31, 2017]. Available from: http://geteccu.org/eneida/caracteristicas-y-objetivos-de-eneida. |
| 29. | Andreu M, Márquez L, Domènech E, Gisbert JP, García V, Marín-Jiménez I, Peñalva M, Gomollón F, Calvet X, Merino O, Garcia-Planella E, Vázquez-Romero N, Esteve M, Nos P, Gutiérrez A, Vera I, Cabriada JL, Martín MD, Cañas-Ventura A, Panés J; Spanish GETECCU group (ENEIDA project). Disease severity in familial cases of IBD. J Crohns Colitis. 2014;8:234-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 30. | Charpentier C, Salleron J, Savoye G, Fumery M, Merle V, Laberenne JE, Vasseur F, Dupas JL, Cortot A, Dauchet L. Natural history of elderly-onset inflammatory bowel disease: a population-based cohort study. Gut. 2014;63:423-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 259] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 31. | Chouraki V, Savoye G, Dauchet L, Vernier-Massouille G, Dupas JL, Merle V, Laberenne JE, Salomez JL, Lerebours E, Turck D. The changing pattern of Crohn’s disease incidence in northern France: a continuing increase in the 10- to 19-year-old age bracket (1988-2007). Aliment Pharmacol Ther. 2011;33:1133-1142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 131] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 32. | Gower-Rousseau C, Savoye G, Colombel JF, Peyrin-Biroulet L. Are we improving disease outcomes in IBD? A view from the epidemiology side. Gut. 2014;63:1529-1530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 33. | Gower-Rousseau C, Vasseur F, Fumery M, Savoye G, Salleron J, Dauchet L, Turck D, Cortot A, Peyrin-Biroulet L, Colombel JF. Epidemiology of inflammatory bowel diseases: new insights from a French population-based registry (EPIMAD). Dig Liver Dis. 2013;45:89-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 145] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 34. | Penninck E, Fumery M, Armengol-Debeir L, Sarter H, Savoye G, Turck D, Pineton de Chambrun G, Vasseur F, Dupas JL, Lerebours E. Postoperative Complications in Pediatric Inflammatory Bowel Disease: A Population-based Study. Inflamm Bowel Dis. 2016;22:127-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 35. | Jakobsson GL, Sternegård E, Olén O, Myrelid P, Ljung R, Strid H, Halfvarson J, Ludvigsson JF. Validating inflammatory bowel disease (IBD) in the Swedish National Patient Register and the Swedish Quality Register for IBD (SWIBREG). Scand J Gastroenterol. 2016;52:216-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 151] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 36. | Gesellschaft für Pädiatrische Gastroenterologie und Ernährung[internet]. Berlin: Gesellschaft für Pädiatrische Gastroenterologie und Ernährung; 2017 CEDATA-GPGE. [cited July 31, 2017]. Available from: http://www.gpge.de/cedata-gpge/. |
| 37. | Martín-de-Carpi J, Rodríguez A, Ramos E, Jiménez S, Martínez-Gómez MJ, Medina E; SPIRIT-IBD Working Group of Sociedad Española de Gastroenterología, Hepatología y Nutricion Pediátrica. Increasing incidence of pediatric inflammatory bowel disease in Spain (1996-2009): the SPIRIT Registry. Inflamm Bowel Dis. 2013;19:73-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 95] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 38. | European Society for Paediatric Gastroenterology, Hepatology and Nutrition[internet]. 2017 Paediatric and adolescent inflammatory bowel diseases [cited July 31, 2017]. Available from: http://www.espghan.org/about-espghan/committees/gastroenterology/working-groups/paediatric-and-adolescent-ibd/. |
| 39. | de Bie C. Pediatric Inflammatory Bowel Disease: from diagnosis to transition. PhD thesis, Erasmus Universiteit Rotterdam [internet];. 2012 [cited July 31, 2017]. Available from: https://repub.eur.nl/pub/37166/120907_Bie,%20Charlotte%20Irene%20de.pdf. |