Published online Nov 7, 2016. doi: 10.3748/wjg.v22.i41.9104
Peer-review started: July 12, 2016
First decision: July 29, 2016
Revised: August 25, 2016
Accepted: September 28, 2016
Article in press: September 28, 2016
Published online: November 7, 2016
Processing time: 120 Days and 21.9 Hours
To study the innate immune function in ulcerative colitis (UC) patients who fail to respond to anti-tumor necrosis factor (TNF) therapy.
Effects of anti-TNF therapy, inflammation and medications on innate immune function were assessed by measuring peripheral blood mononuclear cell (PBMC) cytokine expression from 18 inflammatory bowel disease patients pre- and 3 mo post-anti-TNF therapy. Toll-like receptor (TLR) expression and cytokine production post TLR stimulation was assessed in UC “responders” (n = 12) and “non-responders” (n = 12) and compared to healthy controls (n = 12). Erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) levels were measured in blood to assess disease severity/activity and inflammation. Pro-inflammatory (TNF, IL-1β, IL-6), immuno-regulatory (IL-10), Th1 (IL-12, IFNγ) and Th2 (IL-9, IL-13, IL-17A) cytokine expression was measured with enzyme-linked immunosorbent assay while TLR cellular composition and intracellular signalling was assessed with FACS.
Prior to anti-TNF therapy, responders and non-responders had similar level of disease severity and activity. PBMC’s ability to respond to TLR stimulation was not affected by TNF therapy, patient’s severity of the disease and inflammation or their medication use. At baseline, non-responders had elevated innate but not adaptive immune responses compared to responders (P < 0.05). Following TLR stimulation, non-responders had consistently reduced innate cytokine responses to all TLRs compared to healthy controls (P < 0.01) and diminished TNF (P < 0.001) and IL-1β (P < 0.01) production compared to responders. This innate immune dysfunction was associated with reduced number of circulating plasmacytoid dendritic cells (pDCs) (P < 0.01) but increased number of CD4+ regulatory T cells (Tregs) (P = 0.03) as well as intracellular accumulation of IRAK4 in non-responders following TLR-2, -4 and -7 activation (P < 0.001).
Reduced innate immunity in non-responders may explain reduced efficacy to anti-TNF therapy. These serological markers may prove useful in predicting the outcome of costly anti-TNF therapy.
Core tip: Anti-tumor necrosis factor (TNF) therapy is effective in approximately 60% of ulcerative colitis (UC) patients. Currently we do not know which patients are likely to benefit from this costly treatment. Here we show that differences in innate immune function [measured by patients response to toll-like-receptor (TLR), TLR agonists] exist between UC responders and non-responders. Differences exist in (1) content of immune and regulatory cells in their blood; (2) capacity of their cells to produce cytokines; and (3) in their signalling following TLR activation. Serological measure of TLR function may prove to be a useful tool in clinic to predict patient’s response to anti-TNF treatment.
- Citation: Baird AC, Mallon D, Radford-Smith G, Boyer J, Piche T, Prescott SL, Lawrance IC, Tulic MK. Dysregulation of innate immunity in ulcerative colitis patients who fail anti-tumor necrosis factor therapy. World J Gastroenterol 2016; 22(41): 9104-9116
- URL: https://www.wjgnet.com/1007-9327/full/v22/i41/9104.htm
- DOI: https://dx.doi.org/10.3748/wjg.v22.i41.9104
Inflammatory bowel diseases (IBDs), including Crohn’s disease (CD) and ulcerative colitis (UC) are life-long, immunologically-mediated disorders that are increasing in frequency[1,2]. One of the main pro-inflammatory cytokines involved in ongoing and uncontrolled inflammation in IBD is tumor necrosis factor alpha (TNFα). Although the use of anti-TNF therapy (Infliximab® and Adalimumab®) has revolutionized the treatment of the disease[3-6], one third of patients fail to respond and significant proportion loose sensitivity or become steroid dependent. In the past, serologic and faecal[7] as well as genetic[8,9] markers have been used to predict response to anti-TNF therapy, however these are often not effective or extremely expensive. Our lack of understanding why certain patients respond to anti-TNF therapy and others don’t hinders our progress in predicting which patients are likely to benefit from this costly treatment.
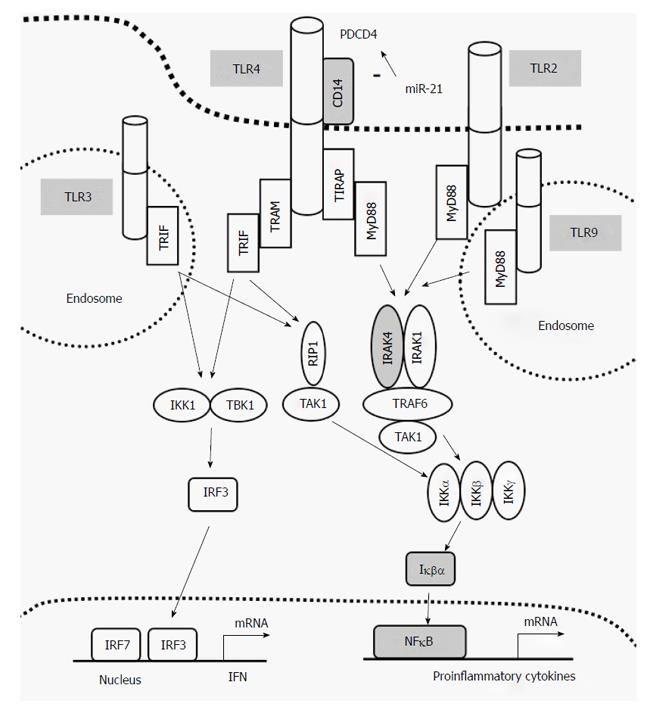
Inflammation in IBD is thought to result from inappropriate activation of the innate immune system by intestinal luminal antigens or a defect in its signaling regulation in genetically susceptible individuals[1]. Toll-like receptors (TLR 1-10) are crucial activators of innate immunity. All TLRs signal through MyD88-dependent pathway except TLR3. TLR4 can signal through both MyD88-dependent and MyD88-independent pathways but requires CD14 (Figure 1). IRAK4 plays a critical role in initiating nuclear factor kappa B (NFκB) intracellular signalling pathway and therefore production of pro-inflammatory cytokines. The role of TLRs in IBD is mounting[10-12]; polymorphisms in TLR genes are associated with increased risk of IBD[13-15] and genes regulating TNF signalling and TNF production have been shown to be important predictors of anti-TNF therapy[13]. Together these finding suggest a strong pathogenic association between the TLRs and IBD.
Previously we have demonstrated that measurement of early innate immune function in peripheral blood of children (TLR responses during their first 5 years of life) identified striking differences in the developmental of their innate immune responses and profile of these responses were a good indicator of their subsequent risk of development of allergic disease[16]. As the inflammatory extra-intestinal manifestations in UC suggest an immunological component not isolated to the intestine, the same methodology was used to investigate the global innate immune function in UC patients to determine if differences can explain the heterogeneity in their clinical responses to anti-TNF therapy.
This study investigated the interaction between the TLR activity in UC patients responsive or non-responsive to anti-TNF therapy to determine if TLR levels, activity and/or TLR signalling pathways correlate with patient’s response to anti-TNF therapy. It was hypothesised that there are inherent differences in innate immune function between responders and non-responders which may explain differences in their clinical effectiveness of treatment. These novel results extend our understanding of intestinal inflammation pathogenesis and implications of innate immunity in UC patients’ response to anti-TNF therapy.
This study was conducted as a prospective and retrospective observational study. The former, to determine whether inflammation levels, medication use, patient demographics, surgery and anti-TNF therapy itself influenced patient outcome (response or non-response to TNF therapy), and the latter to determine if there were differences in the underlying mechanisms responsible for TLR recognition and innate immune response. To address these aims, pro-inflammatory cytokine levels, TLR expression, TLR signalling and cell populations were analysed from isolated PBMCs pre- and post-anti-TNF therapy and; (1) correlated back to inflammation levels, medication use, patient demographics, surgery and anti-TNF therapy itself, and (2) were then compared between responders and non-responders.
IBD patients (n = 42) and healthy controls (n = 12) were recruited from Centre for Inflammatory Bowel Diseases, Fremantle Hospital, Perth, Australia. The diagnosis of CD and UC was made based upon clinical, endoscopic, histopathological and radiological findings, and classified by the “Montreal classification”. Patient demographic data included data of birth, age at diagnosis, age at time of study, timing of anti-TNF therapy, concurrent immunosuppressive medications, surgeries, family and smoking history. To examine the effects of disease, anti-TNF therapy, inflammation and medication use on innate immunity, blood was taken from 18 IBD patients (13 with CD and 5 with UC) prior (pre-anti-TNF) and 3 mo after anti-TNF therapy commenced (post-anti-TNF). To study immune responses in responder and non-responder UC patients, blood was collected from separate 24 UC patients and compared to 12 healthy controls. UC patients achieving clinical remission, defined by a Colitis Activity Index (CAI) ≤ 4, and normal C reactive protein (CRP) ≤ 10 mg/L, were considered responders (Rs, n = 12), whilst those who failed to respond with a reduction in CAI of < 4 points and a consistently elevated CRP as non-responders (NRs, n = 12).
Sixty millilitres of peripheral blood was collected and peripheral blood mononuclear cells (PBMCs) were isolated by Ficoll-Hypaque gradient centrifugation and cryopreserved (7.5% DMSO) at -80 °C for future use.
PBMCs were cultured alone or with various TLR agonists including lipoteichoic acid (LTA 1 μg/mL, TLR2 ligand), Poly I:C (50 μg/mL, TLR3 ligand), E. coli lipopolysaccharide (LPS 10 ng/mL, TLR4 ligand), Flagellin (1 μg/mL, TLR5 ligand), Imiquimod (10 μg/mL, TLR7 ligand), Gardiquimod (10 μg/mL, TLR8 ligand) or CpG oligonucleotide (CpG 3 μg/mL, TLR9 ligand); all purchased from InvivoGen, CA, United States. All cultures were plated in duplicate in 96-well round-bottom plates in 250 μL RPMI (Gibco, Life Technology, Grand Island, NY, United States) supplemented with 10% foetal calf serum (Australia Biosearch, Australia) and incubated at 37 °C with 5% CO2 for 24 h (LTA, Poly I:C, LPS and Flagellin) or 48 h (Imiquimod, Gardiquimod or CpG). The supernatants were then removed and stored at -20 °C until cytokine analysis.
Cytokines [TNFα, interferon γ (IFNγ), interleukin (IL)-1β, IL-6, IL-9, IL-10, IL-12, IL-13 and IL-17A] were measured from culture supernatants. Multiplex beads for the Bio-Plex® multiplex system (Life Sciences, Bio-Rad Laboratories Pty, Ltd., Vic, Australia) were diluted 1:2 in bead diluents and the 9plex bead assay according to the manufacturer’s protocol using a Luminex®200 Bead array with Xmap® multiplexing technology located at the Centre of Microscopy, characterisation and Analysis (CMCA), UWA, Australia. The limit of detection was 3 pg/mL for all cytokines. Data was analysed using the xPONENT 4.2 for MAGPIX software (Luminex Corporation, Austin, TX, United States).
PBMC cells were stained with monoclonal antibodies to identify macrophages/monocytes [Mφ] (HLADR+CD14+), natural killer cells (CD16+CD56+), myeloid (Lin1-HLADR+CD123-CD11c+) and plasmacytoid (Lin1-HLADR+CD123+CD11c-) dendritic cells (DC), effector T cells (CD4+ or CD8+), T regulatory cells (CD4+CD25+CD127- or CD8+CD25+CD127-), memory T cells (CD45RO+) and naïve T cells (CD45RA+CD4+) (Supplement Table 1). Isotype-matched antibodies were used as controls (Supplement Table 1) and assessed by FACS analysis. For the analysis of TLR and CD14 receptor levels, unstimulated and stimulated PBMCs were stained with TLR2 (1:20 dilution, PE; eBiosciences, San Diego, CA, United States), TLR4 (1:50 dilution, APC; eBiosciences), TLR9 (1:20 dilution, APC; BD Pharmingen, San Diego, CA, United States) and CD14 (1:20 dilution, FITC; eBiosciences) prior to fixing according to manufacturer’s instructions. Isotype-matched antibodies were used as controls.
To address differences in MyD88-dependent signalling in unstimulated and stimulated PBMCs, PBMCs were cultured alone or with TLR2, TLR3, TLR4, TLR7 TLR9 agonists as previously described for 15 min at 37 °C with 5% CO2. Cells were fixed and permeabilized according to manufacturer’s instructions (BD Biosciences, San Diego, CA, United States) and stained for phosphorylated NFκB (pNFκB) (1:5 dilution, AF488; BD Biosciences), total IRAK4 (1:5 dilution, PE; BD Biosciences) and total Iκβα (1:5 dilution, AF647; BD Biosciences). Stained cells were captured using the FACScanto II bench top flow cytometer (BD Biosciences) at the CMCA, UWA, Australia and analysed using FlowJO v7.6.3 research software (Tree Star Inc. Oregon, United States).
Significance between groups at 95% confidence level was determined by paired and Mann-Whitney non-parametric unpaired t tests, using Graphpad Prism 4.0 software package (Graphpad, San Diego, CA, United States). Results were expressed as median geometric mean with 95% confidence interval, fold-change from basal ± SD, mean percentage of total cell population ± SD or mean fluorescence intensity (MFI) ± SD. Correlation between medications and patient’s response to anti-TNF therapy was determined by multiple regression analyses using SPSS version 14.0 software package for Windows PC (IBM, Armonk, NY, United States). Statistical significance was considered as P < 0.05.
Clinical data comparison of patient population: To determine if innate immune response were altered by the use of immuno-suppressants and anti-inflammatory medications, disease type or the level of inflammation measured by erythrocyte sedimentation rate (ESR), C-reactive protein (CRP) levels, CD activity index (CDAI) and partial Mayo score pre-anti-TNF therapy, the clinical data of 18 IBD patients were compared (Tables 1 and 2).
| Characteristics | n or mean ± SD (range) |
| Male:female | 7:11 |
| Age at diagnosis(yr) | 26.6 ± 11.3 (12-56) |
| Disease duration | 8.4 ± 7.9 (0-30) |
| Montreal classification | |
| CD | 13 |
| UC | 5 |
| Age at diagnosis (yr) | |
| A1 ( ≤ 16) | 1 |
| A2 (17-40) | 14 |
| A3 (> 40) | 3 |
| Smoking status | |
| Never smoked | 7 |
| Ex-smoker | 5 |
| Current smoker | 6 |
| Anti-TNF response | |
| Responder | 13 |
| Non-responder | 5 |
| Responders (n = 13) (n or mean ± SEM) | Non-responders (n = 5) (n or mean ± SEM) | P value | |
| Immuno-suppressants | |||
| Thiopurine | 10 | 4 | NS |
| Tacrolimus | 0 | 0 | |
| Prednisone | 6 | 4 | NS |
| Prednisone/thiopurine | 5 | 2 | NS |
| Methotrexate | 0 | 0 | |
| 5-ASA | 1 | 2 | |
| Corticosteroids | 9 | 2 | NS |
| ESR | 23.9 ± 4.8 (n = 11) | 9.6 ± 2.2 (n = 4) | NS |
| CRP | 25.8 ± 6.8 (n = 13) | 9.2 ± 3.7 (n = 4) | NS |
| Partial Mayo1 (out of 9 - UC patients only) | 8 ± 1.3 (n = 3) | 8 (n = 3) | NS |
| CDAI (CD patients only) | 321.9 ± 35.4 (n = 9) | 385 ± 55 (n = 3) | NS |
Prior to anti-TNF therapy, clinical data of responders and non-responders demonstrated no significant differences in the number of patients on individual or combined immune-suppressants, methotrexate, 5-ASA or corticosteroids (Table 2). Although the ESR and CRP levels appeared higher in responders, they were not significantly different to non-responders suggesting similar level of disease severity/activity between the two groups. This suggests that whether a patient responds or not to anti-TNF therapy is not predicted by their ESR, CRP, CDAI or partial Mayo score, nor their medication use. As expected, the partial Mayo scores were significantly higher in non-responders (7.3 ± 0.6, n = 3) than in responders (0.5 ± 0.5, n = 4) post-anti-TNF therapy (P = 0.004), as were the CDAI scores (198 ± 42, n = 2 vs 80.1 ± 21.7, n = 9 respectively, P = 0.04). This was also true of the CRP levels (23 ± 1.9, n = 5 vs 7.4 ± 3.4, n = 13 respectively, P = 0.01) (data not shown). Considering CD and UC patients separately, all patients suffered from moderately-severe inflammation and there were no significant differences in medications, ESR or CRP levels pre anti-TNF therapy (Table 3). At the time of the second blood draw (3 mo post-anti-TNF induction therapy), two thirds of the patients had gone into remission with anti-TNF therapy (CDAI < 150, CAI ≤ 4 and CRP ≤ 10 mg/L) and had ceased steroid therapy. Five (n = 5) did not respond to anti-TNF therapy, three (n = 3) continued on steroid therapy (1 responder and 2 non-responders P > 0.05) and two (n = 2) underwent surgery with cessation of immunomodulation.
| CD (n = 13) (n or mean ± SEM) | UC (n = 5) (n or mean ± SEM) | P value | |
| Immunosuppressant | |||
| Thiopurine | 10 | 4 | NS |
| Tacrolimus | 0 | 0 | |
| Prednisone | 6 | 4 | NS |
| Prednisone/thiopurine | 3 | 3 | |
| Methotrexate | 0 | 0 | |
| 5-ASA | 2 | 3 | NS |
| ESR | 17.4 ± 3.3 (n = 11) | 27.6 ± 11.8 (n = 4) | NS |
| CRP | 19.5 ± 6.3 (n = 13) | 29.8 ± 12 (n = 4) | NS |
| Partial Mayo1 (out of 9 - UC patients only) | - | 8.5 ± 0.9 (n = 4) | |
| CDAI (CD patients only) | 337 ± 29.8 (n = 12) | - |
Basal and stimulated PBMC cytokine production pre- and post-anti-TNF therapy: To investigate whether medication use, anti-TNF therapy, disease type or inflammation affected baseline PBMC function, isolated PBMCs were cultured in vitro and basal and stimulated supernatant cytokine levels measured. Basal innate (TNF, IL-1β, IL-6), immunoregulatory (IL-10), Th1 (IL-12 and IFNγ) and Th2 (IL-9, IL-13 and IL-17A) cytokine expression was similar pre- and post-anti-TNF therapy suggesting it was not affected by treatment (Supplementary Figure 1).
To determine if anti-TNF therapy affected the PBMC’s ability to recognise and respond to stimulation, TNF production pre- and post-anti-TNF therapy was measured post TLR activation. TNF production was unaffected or even higher (P = 0.03 for TLR4 stimulation) post-TNF therapy compared to production pre-therapy (Supplementary Figure 2) suggesting that anti-TNF therapy does not reduce the PBMC’s ability to recognise, or responds to TLR activation. None of the other cytokine levels were affected post therapy (data not shown). TNF production post TLR3 and TLR4 activation was approximately 10-fold higher than stimulation of other TLRs.
As the use of anti-TNF therapy for the treatment of UC is associated with higher rates of primary and secondary non-responses than in CD patients, we next set out to study innate immune responses in UC patients who respond or do not respond to treatment.
Demographic and clinical data comparison of UC patient cohort: PBMCs from 24 UC patients (n = 12 responders and n = 12 non-responders to anti-TNF therapy), and 12 healthy controls were isolated. Bloods were taken post-anti-TNF therapy, with the median time being 208 (12-468) wk for responders and 156 (12-364) wk for non-responders (P = 0.52). No significant demographic differences were detected between the UC populations except that responders had longer disease duration (P = 0.01) (Table 4). None of the controls were on any medications or suffering from any infections or inflammatory conditions. There was no correlation between any of the medications (alone or in combination) and patient’s response to anti-TNF therapy. At time of blood draw (post-anti-TNF therapy) all responders were in clinical remission (CAI ≤ 4 and CRP ≤ 10 mg/L) with 5 patients on maintenance anti-TNF therapy, 8 on thiopurine and 4 on 5-ASA. Of the non-responders, 3 had blood drawn following recovery from colectomy from uncontrolled UC, 1 was in remission on tacrolimus and 4 had ongoing inflammation with 6 taking oral corticosteroids and all receiving thiopurines (Table 5).
| UC non-responders (NR) (n = 12) | UC responders (R) (n = 12) | P value | Controls | |
| NR vs R | (n = 12) | |||
| Male:female | 6:6 | 4:8 | NS | 4:8 |
| Mean age at diagnosis (yr) (SD/range) | 27.5 (12.2/15-59) | 24.5 (8.8/10-36) | NS | N/A |
| Mean age at assessment (yr) (SD/range) | 33.6 (14/20-67) | 38.6 (14.2/21-64) | NS | 33.4 (13/18-52) |
| Mean Disease duration (yr) (SD/range) | 6.9 (2.7/13.1/20-67) | 17.5 (10.6/4-37) | 0.01 | N/A |
| Median time of blood draw post-therapy (wk) (range) | 156 (12-364) | 208 (12-468) | NS | N/A |
| Montreal classification, n | N/A | |||
| Age at diagnosis (yr) | ||||
| A1 - ≤ 16 | 1 | 1 | NS | |
| A2 - 17-40 | 9 | 10 | NS | |
| A3 - > 40 | 2 | 1 | NS | |
| Disease location | ||||
| E1 - Proctitis | 2 | 0 | NS | |
| E2 - Left sided | 1 | 4 | NS | |
| E3 - Extensive | 9 | 8 | NS | |
| Smoking status (n) | ||||
| Never smoked | 8 | 8 | NS | 8 |
| Ex-smoker | 3 | 4 | NS | 2 |
| Current smoker | 1 | 0 | NS | 2 |
| Non-responder (n = 12) | Responder (n = 12) | P value | |
| (n or mean ± SEM) | (n or mean ± SEM) | ||
| Pre-anti-TNF therapy | |||
| Immunosuppressant | |||
| Thiopurine | 7 | 6 | NS |
| Tacrolimus | 2 | 0 | NS |
| Prednisone | 9 | 7 | NS |
| Prednisone/thiopurine | 6 | 2 | NS |
| Methotrexate | 1 | 0 | |
| 5-ASA | 6 | 5 | NS |
| ESR | 42.5 ± 12.9 (n = 7) | 33.1 ± 10.4 (n = 7) | NS |
| CRP | 36.3 ± 10.1 (n = 11) | 27.4 ± 7.4 (n = 12) | NS |
| Partial Mayo1 | 7.3 ± 0.5 (n = 7) | 7.5 ± 0.2 (n = 11) | NS |
| Post-anti-TNF therapy | |||
| Immunosuppressant | |||
| Thiopurine | 12 | 8 | NS |
| Tacrolimus | 4 | 0 | 0.045 |
| Prednisone | 6 | 0 | 0.007 |
| Prednisone/thiopurine | 0 | 0 | |
| Methotrexate | 0 | 0 | |
| 5-ASA | 6 | 4 | NS |
| ESR | 22.9 ± 7.9 (n = 4) | 13.5 ± 4.7 (n = 9) | NS |
| CRP | 51.2 ± 14.6 (n = 12) | 4.6 ± 1.2 (n = 12) | 0.0043 |
| Partial Mayo1 | 7.6 ± 0.4 (n = 12) | 0.3 ± 0.2 (n = 12) | < 0.0001 |
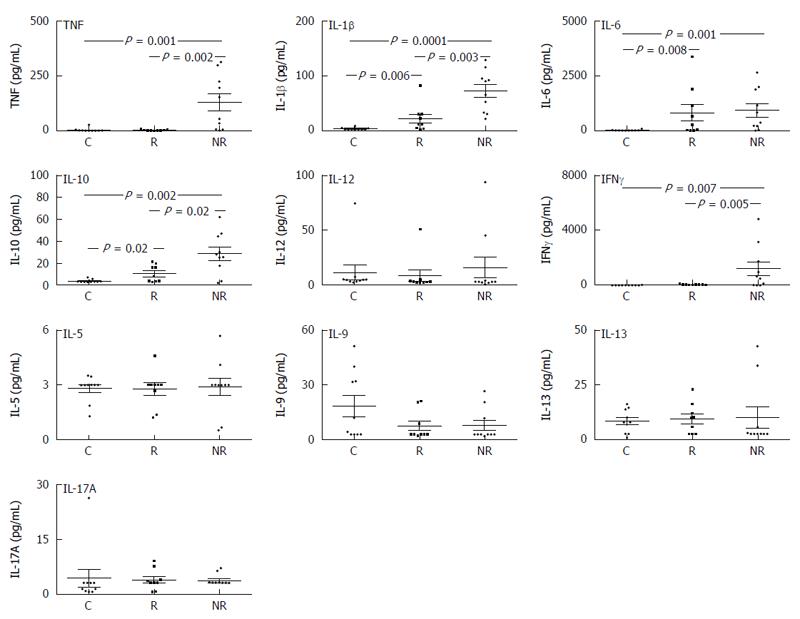
Basal cytokine production in UC responders and non-responders: PBMCs from UC patients (responders and non-responders) had significantly greater basal IL-1β, IL-6 and IL-10 levels compared to healthy controls (Figure 2). Non-responders had significantly increased TNF, IL-1β and IL-10 compared to responders. There were no differences in basal IL-12 production between UC groups compared to controls or production of any of the Th2 cytokines measured (IL-5, IL-9, IL-13 or IL-17A) (Figure 2). The Th1 cytokine IFNγ was significantly elevated in non-responders compared to responders and controls (Figure 2).
Differences in stimulated cytokine production by UC responders and non-responders: In general, responders had similar TNF, IL-1β, IL-6 and IL-10 responses to healthy controls following TLR stimulation; exceptions being increased TLR9-induced TNF in responders (Figure 3A), and reduced TLR7-induced IL-1β (Figure 3B) as well as reduced TLR-3, -5 and -7 induced IL-6 responses (P = 0.04) (Figure 3C). In contrast, TNF, IL-1β, IL-6 and IL-10 responses to all TLR agonists were significantly lower in non-responders compared to healthy controls (P < 0.01) (Figure 3A-D). Non-responders had significantly lower TNF and IL-1β production to all TLRs compared to responders (Figure 3A and B) as well as reduced TLR9-induced IL-6 (Figure 3C) and TLR-3, -4, -8 and -9-induced IL-10 (Figure 3D).
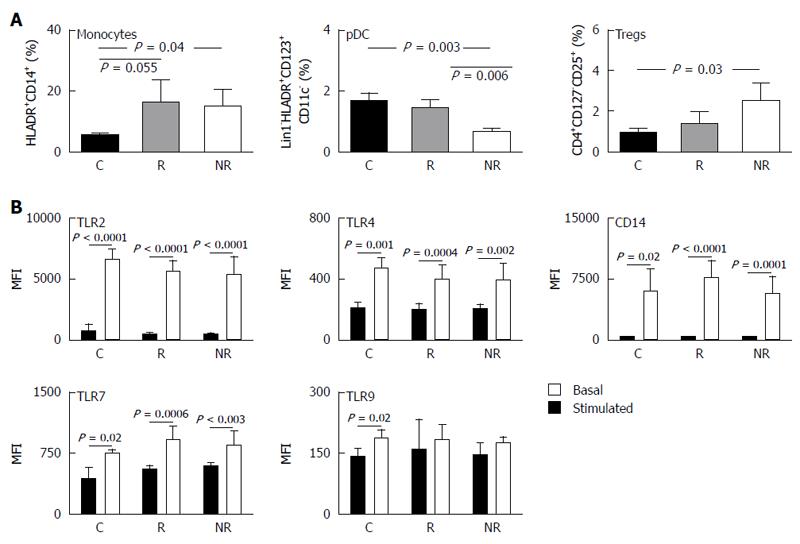
Isolated PBMCs are a mixed cell population and differences in cytokine production are likely to be attributed to differences in distribution of cellular populations. Characterization of cellular populations in the three study groups have shown both UC subgroups to have higher percentage of monocytes in circulating blood compared to controls (Figure 4A). Non-responders had a significantly lower plasmacytoid DC (pDC) frequency compared to responders and controls (P < 0.01). This decrease in pDCs was associated with increased percentage of CD4+ regulatory T cell (Tregs) compared to controls (P = 0.03) (Figure 4A). Increased Tregs was of borderline significance in responders compared to controls (P = 0.09; Figure 4A). We found no difference in memory, naïve, CD4+, CD8+ effector T cells, CD8+ Treg cells, NK cells or myeloid DC (mDC) between the groups (data not shown).
As pDC frequency varied between responders and non-responders, the level of basal and stimulated TLR-2, TLR-4, TLR-7, TLR-9 and CD14 expression levels were assessed. The baseline expression of TLRs and CD14 were similar between the three groups and was increased to similar levels in each of the groups following stimulation (Figure 4B). No differences were observed in basal or stimulated TLR/CD14 expression levels between the groups suggesting that the percentage pDC did not impact expression of TLRs or CD14.
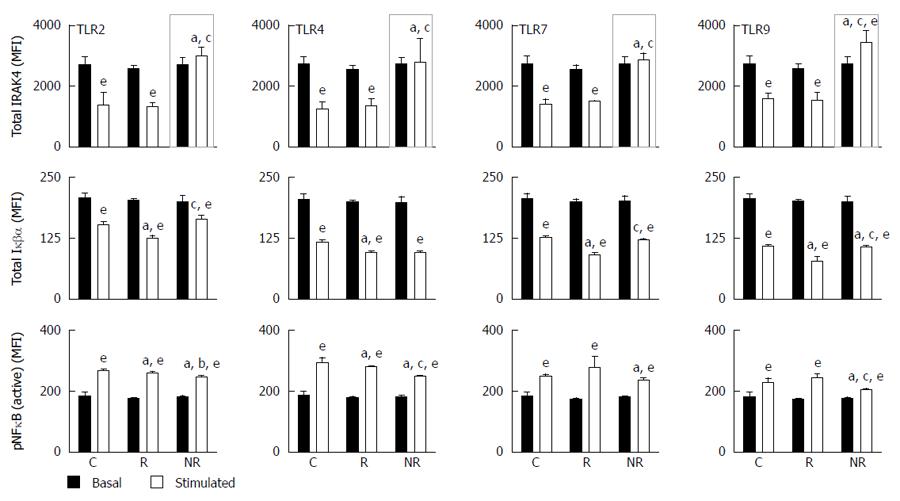
MyD88-dependent signalling was assessed following TLR-2, -3, -4, -7 and -9 stimulation of PBMCs by measuring total IRAK4, total Iκβα and phosphorylated NFκB (pNFκB) activity. TLR-3 was used as a control as TLR3-mediated signalling is independent of MyD88, IRAK4 and IRAK1[17]. Total IRAK4 levels did not change upon TLR-3 activation and no differences in protein levels between the 3 groups were identified (data not shown). When comparing basal to stimulated total IRAK4 levels; responders and controls had similar response profiles, that is, significantly lower total IRAK4 levels upon TLR stimulation (Figure 5), whilst non-responders failed to reduce total IRAK4 following TLR-2, -4, and -7 stimulation (Figure 5). Total IRAK4 was significantly increased in non-responders following TLR-9 activation compared to basal levels (P = 0.03, Figure 5). This suggests that in the non-responders, the degradation/inhibition of IRAK4 may be dysregulated resulting in its aberrant accumulation. Whilst total Iκβα were significantly decreased in all groups following stimulation (P < 0.02 for all), pNFκB were significantly increased in all groups (P < 0.02 for all; Figure 5).
Non-communicable disease including cardiovascular, metabolic, IBD and allergic diseases are now surpassing infectious disease accounting for more than 60% of all global deaths[18]. The IBDs are incurable, disabling life-long conditions. Albeit expensive, anti-TNF therapy is an effective treatment for approximately 60% UC patients; however, the mechanisms responsible for lack of responses to treatment are unknown. Here we have shown, for the first time, clear differences in innate immune function in peripheral blood of responders and non-responders UC patients given anti-TNF therapy. We have demonstrated that whilst, in general, responders have similar innate cytokine responses to healthy controls, non-responders have diminished innate responses to all TLR agonists compared to controls and reduced TNF and IL-1β responses compared to responders. These results suggest dysregulation of innate immunity in non-responders and may explain heterogeneity in clinical effectiveness of anti-TNF treatment in UC patients. Individuals innate immune function may prove be a useful tool to predict cost effective application of this treatment.
As there were no significant differences in medication use, baseline ESR, CRP and inflammation levels (as indicated by partial Mayo scores and CDAIs) between responders and non-responders prior to anti-TNF induction (part 1 of study), this strengthens the hypothesis that the differences are intrinsic and that there is an inherent difference within the innate immune response of these two cohorts, i.e., their PBMCs function differently in response to TLR stimulation. This was clearly demonstrated in non-responders having significantly higher basal Th1 cytokine production compared to responders. Having higher Th1 but not Th2 cytokine production was also an indicator that the problem lays within the innate and not the adaptive immune response. By using both UC and CD patients in the initial experiments, it demonstrated that these intrinsic differences may be inherent in both UC and CD patients, and the ability to be able to target and treat patients based on their innate immune response may be applied to both of the IBD cohorts.
When specifically looking at differences between the UC responders and non-responders, it was noted that the non-responders had a significantly higher constitutive or basal cytokine production than responders and controls, and smaller fold change in cytokine production upon TLR stimulation. High basal cytokine levels and the lack of fluctuation in cytokine production upon TLR stimulation suggests that the mechanisms involved in the negative regulation of TLR signalling may be impaired. Indeed, low levels of immunoregulatory cytokine IL-10 seen in non-responders compared to responders following TLR activation (Figure 3) may explain why inflammation cannot be controlled with anti-TNF therapy in these patients.
We demonstrated that non-responders had a significantly lower number of pDCs in their peripheral blood compared to responders and to healthy controls. Others have shown that UC and CD patients experience a significant drop in their peripheral pDC populations during acute inflammation and significant increase in numbers within the intestinal mucosa[19]. The decreased pDC frequency we see in non-responders may result from continuous migration of peripheral pDCs into the intestinal mucosa whereupon they mature, activate and contribute to gut inflammation, thus resulting in an elevated basal Th1 cytokine profile which is characteristic of this population. The maturational status of the peripheral pDCs in non-responders may also be of importance, as healthy individuals display an immature pDC phenotype which normally induces T cell unresponsiveness[20], whilst IBD patients have a lack of immature peripheral pDCs which would perpetuate inflammation[21]. Further investigation into the distribution of pDCs in the peripheral blood and the intestinal mucosa, and their maturational status in the UC subgroups is required. The increased frequency of CD4+ Treg cells in non-responders could suggest a problem with Treg homing to the mesenteric lymph nodes and lamina propria to inhibit pathogenic T effector cells during inflammation via direct contact with cD11c+ dendritic cells[22], thus leading to ongoing inflammation. Consistent with the increase in pro-inflammatory cytokines, monocyte frequency was significantly greater in both UC subgroups compared to controls, and no differences were observed in naïve, memory or CD8+ effector or CD8+ Treg cell frequency, which again supports the concept that the differences in immunologic responses between the UC subgroups lie within the innate immune system.
Downstream of the TLRs, we saw accumulation of total IRAK4 in non-responders upon stimulation, particularly following TLR-9 activation. IRAK4 is a key signalling component in the innate immune response[23] and IRAK4 deficiencies have been implicated in IBD[24]. We know that IBD patients who do not respond to anti-TNF therapy maintain an increased expression of pro-inflammatory cytokines[25]. In our non-responder population, this is associated with IRAK4 accumulation and we may speculate such accumulation may lead to prolonged activation of the signalling pathway resulting in sustained and excessive pro-inflammatory cytokine production seen in UC patients. Our signalling data shows non-responders to have normal IRAK4 kinase activity however, other mechanisms which may contribute to its accumulation such as defects in IRAK4 degradation or inhibition remains to be tested. It’s been previously shown that IL-10 can induce IRAK4 ubiquitination and proteasomal degradation[26,27]. Our results support reduced ability of non-responders to induce IRAK4 ubiquitination due to their reduced capacity to produce IL-10 following TLR stimulation. Alternatively, IRAK4 activity is inhibited by cleavage into its inactive form[26]. Cleavage occurs by an NFκB-induced protease resulting in a smaller molecular weight protein (32 kDa) that can also be recognised by anti-IRAK4 antibodies[26]. As IRAK1 phosphorylation and NFκB activation precedes IRAK4 cleavage, this suggests that this may be part of a negative feedback inhibition loop[26].
There is no doubt that anti-TNF therapy can be effective in UC but only in some patients. The ability to predict patient’s response to anti-TNF therapy would allow for more targeted therapy with better cost-effectiveness. Here we provide evidence which suggests that heterogeneity in the innate immune function between UC patients may give us an important insight into their subsequent responses to future anti-TNF therapy. This would be particularly beneficial for the patient with acute severe colitis requiring rescue therapy when a choice must be made between cyclosporine or anti-TNF therapy. It is important to acknowledge that one of the potential limitations of this study is the relatively small and a mixed population of patients (CD and UC) used. Moreover, it is known that pathogenesis of CD and UC are different and disease can be more severe in the elderly[28]. In our cohort, results could not be explained by differences in age between groups and our functional innate differences between responders and non-responders have been performed in UC patients only. Our data offers promise for serological measure of innate immune function in UC patients as a potential application in clinic to predict response to costly anti-TNF therapy. These data remain to be confirmed in a larger cohort of not only UC but also CD patients. With better prediction of the response to therapy, targeted patient treatment may be possible in the future, resulting in improved efficacy and cost-effectiveness of treatment for all IBD patients.
The authors would like to thank Jillian Philpott, Debra Marr and Karen Martin for the collection of patient consent and blood for this study, and Frances Lloyd for the isolation of PBMCs.
Monoclonal antibodies against tumour necrosis factor [anti-tumour necrosis factor (TNF) therapy] can be used to treat patients with ulcerative colitis (UC) who are no longer responding to corticosteroids. Anti-TNF treatment is expensive and 30%-40% of patients do not respond.
Unravelling the mechanisms involved in lack of response to anti-TNF is paramount for prediction of response to treatment.
Here the authors show, for the first time, that differences in innate immune function exist between UC patients who respond to anti-TNF therapy and those that don’t. Both quantitative (difference in presence of inflammatory cells in their peripheral blood) and qualitative (production of cytokines and signalling capacity following activation of innate immune pathways) differences exist between responders and non-responders.
Measurement of innate immune function in the blood of UC patients (their response to TLR agonists) may be a useful tool in predicting patient’s response to anti-TNF treatment. With improved prediction of the response to therapy, targeted and individualised patient treatment may be possible in future, resulting in improved efficacy and cost-effectiveness.
Innate immune function is measured by cellular response to toll-like receptor (TLR) stimulation. To understand which TLRs are implicated in lack of response to anti-TNF treatment, we examined peripheral blood mononuclear cells responses to a wide range of TLR agonists and closely examined the TLR signalling pathway molecules.
In the presented article the authors aimed to predict anti-TNF response in IBD patients by means of alterations in immune functions. There are two parts of the study. The effects of disease, treatment and inflammation on innate immunity were evaluated in 18 patients. In the second part, the differences between responders and non-responders were evaluated in 24 patients. The study adds new knowledge to the current literature.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: France
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Daniel F, Garcia-Olmo D, Ozen H, Pellicano R S- Editor: Gong ZM L- Editor: A E- Editor: Wang CH
| 1. | de Souza HS, Fiocchi C. Immunopathogenesis of IBD: current state of the art. Nat Rev Gastroenterol Hepatol. 2016;13:13-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1185] [Cited by in RCA: 1112] [Article Influence: 123.6] [Reference Citation Analysis (1)] |
| 2. | Molodecky NA, Soon IS, Rabi DM, Ghali WA, Ferris M, Chernoff G, Benchimol EI, Panaccione R, Ghosh S, Barkema HW. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012;142:46-54.e42; quiz e30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3789] [Cited by in RCA: 3519] [Article Influence: 270.7] [Reference Citation Analysis (5)] |
| 3. | Reinisch W, Sandborn WJ, Hommes DW, D’Haens G, Hanauer S, Schreiber S, Panaccione R, Fedorak RN, Tighe MB, Huang B. Adalimumab for induction of clinical remission in moderately to severely active ulcerative colitis: results of a randomised controlled trial. Gut. 2011;60:780-787. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 588] [Cited by in RCA: 670] [Article Influence: 47.9] [Reference Citation Analysis (0)] |
| 4. | Rutgeerts P, Sandborn WJ, Feagan BG, Reinisch W, Olson A, Johanns J, Travers S, Rachmilewitz D, Hanauer SB, Lichtenstein GR. Infliximab for induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2005;353:2462-2476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2744] [Cited by in RCA: 2878] [Article Influence: 143.9] [Reference Citation Analysis (2)] |
| 5. | Rutgeerts P, Van Assche G, Vermeire S. Optimizing anti-TNF treatment in inflammatory bowel disease. Gastroenterology. 2004;126:1593-1610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 281] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 6. | Schreiber S, Khaliq-Kareemi M, Lawrance IC, Thomsen OØ, Hanauer SB, McColm J, Bloomfield R, Sandborn WJ. Maintenance therapy with certolizumab pegol for Crohn’s disease. N Engl J Med. 2007;357:239-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 758] [Cited by in RCA: 729] [Article Influence: 40.5] [Reference Citation Analysis (0)] |
| 7. | Dubinsky MC. Serologic and laboratory markers in prediction of the disease course in inflammatory bowel disease. World J Gastroenterol. 2010;16:2604-2608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 13] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 8. | Dubinsky MC, Mei L, Friedman M, Dhere T, Haritunians T, Hakonarson H, Kim C, Glessner J, Targan SR, McGovern DP. Genome wide association (GWA) predictors of anti-TNFalpha therapeutic responsiveness in pediatric inflammatory bowel disease. Inflamm Bowel Dis. 2010;16:1357-1366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 111] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 9. | Arijs I, Li K, Toedter G, Quintens R, Van Lommel L, Van Steen K, Leemans P, De Hertogh G, Lemaire K, Ferrante M. Mucosal gene signatures to predict response to infliximab in patients with ulcerative colitis. Gut. 2009;58:1612-1619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 271] [Cited by in RCA: 304] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 10. | Fernandes P, MacSharry J, Darby T, Fanning A, Shanahan F, Houston A, Brint E. Differential expression of key regulators of Toll-like receptors in ulcerative colitis and Crohn’s disease: a role for Tollip and peroxisome proliferator-activated receptor gamma? Clin Exp Immunol. 2016;183:358-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 11. | Wu H, Li XM, Wang JR, Gan WJ, Jiang FQ, Liu Y, Zhang XD, He XS, Zhao YY, Lu XX. NUR77 exerts a protective effect against inflammatory bowel disease by negatively regulating the TRAF6/TLR-IL-1R signalling axis. J Pathol. 2016;238:457-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 12. | Yadav V, Varum F, Bravo R, Furrer E, Bojic D, Basit AW. Inflammatory bowel disease: exploring gut pathophysiology for novel therapeutic targets. Transl Res. 2016;176:38-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 135] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 13. | Bank S, Andersen PS, Burisch J, Pedersen N, Roug S, Galsgaard J, Turino SY, Brodersen JB, Rashid S, Rasmussen BK. Associations between functional polymorphisms in the NFκB signaling pathway and response to anti-TNF treatment in Danish patients with inflammatory bowel disease. Pharmacogenomics J. 2014;14:526-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 116] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 14. | De Jager PL, Franchimont D, Waliszewska A, Bitton A, Cohen A, Langelier D, Belaiche J, Vermeire S, Farwell L, Goris A. The role of the Toll receptor pathway in susceptibility to inflammatory bowel diseases. Genes Immun. 2007;8:387-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 107] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 15. | Kim EJ, Chung WC, Lee KM, Paik CN, Jung SH, Lee BI, Chae HS, Choi KY. Association between toll-like receptors/CD14 gene polymorphisms and inflammatory bowel disease in Korean population. J Korean Med Sci. 2012;27:72-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 16. | Tulic MK, Hodder M, Forsberg A, McCarthy S, Richman T, D’Vaz N, van den Biggelaar AH, Thornton CA, Prescott SL. Differences in innate immune function between allergic and nonallergic children: new insights into immune ontogeny. J Allergy Clin Immunol. 2011;127:470-478.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 125] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 17. | Jiang Z, Mak TW, Sen G, Li X. Toll-like receptor 3-mediated activation of NF-kappaB and IRF3 diverges at Toll-IL-1 receptor domain-containing adapter inducing IFN-beta. Proc Natl Acad Sci USA. 2004;101:3533-3538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 308] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 18. | Prescott SL. Disease prevention in the age of convergence - the need for a wider, long ranging and collaborative vision. Allergol Int. 2014;63:11-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 19. | Ben-Horin S, Chowers Y. Tailoring anti-TNF therapy in IBD: drug levels and disease activity. Nat Rev Gastroenterol Hepatol. 2014;11:243-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 163] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 20. | Hawiger D, Inaba K, Dorsett Y, Guo M, Mahnke K, Rivera M, Ravetch JV, Steinman RM, Nussenzweig MC. Dendritic cells induce peripheral T cell unresponsiveness under steady state conditions in vivo. J Exp Med. 2001;194:769-779. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1424] [Cited by in RCA: 1375] [Article Influence: 57.3] [Reference Citation Analysis (0)] |
| 21. | Baumgart DC, Metzke D, Schmitz J, Scheffold A, Sturm A, Wiedenmann B, Dignass AU. Patients with active inflammatory bowel disease lack immature peripheral blood plasmacytoid and myeloid dendritic cells. Gut. 2005;54:228-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 105] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 22. | Mottet C, Uhlig HH, Powrie F. Cutting edge: cure of colitis by CD4+CD25+ regulatory T cells. J Immunol. 2003;170:3939-3943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 717] [Cited by in RCA: 754] [Article Influence: 34.3] [Reference Citation Analysis (0)] |
| 23. | Lye E, Mirtsos C, Suzuki N, Suzuki S, Yeh WC. The role of interleukin 1 receptor-associated kinase-4 (IRAK-4) kinase activity in IRAK-4-mediated signaling. J Biol Chem. 2004;279:40653-40658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 105] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 24. | Staschke KA, Dong S, Saha J, Zhao J, Brooks NA, Hepburn DL, Xia J, Gulen MF, Kang Z, Altuntas CZ. IRAK4 kinase activity is required for Th17 differentiation and Th17-mediated disease. J Immunol. 2009;183:568-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 48] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | Leal RF, Planell N, Kajekar R, Lozano JJ, Ordás I, Dotti I, Esteller M, Masamunt MC, Parmar H, Ricart E. Identification of inflammatory mediators in patients with Crohn’s disease unresponsive to anti-TNFα therapy. Gut. 2015;64:233-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 110] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 26. | Hatao F, Muroi M, Hiki N, Ogawa T, Mimura Y, Kaminishi M, Tanamoto K. Prolonged Toll-like receptor stimulation leads to down-regulation of IRAK-4 protein. J Leukoc Biol. 2004;76:904-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 33] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Chang J, Kunkel SL, Chang CH. Negative regulation of MyD88-dependent signaling by IL-10 in dendritic cells. Proc Natl Acad Sci USA. 2009;106:18327-18332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 93] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 28. | Ardesia M, Villanacci V, Fries W. The aged gut in inflammatory bowel diseases. Minerva Gastroenterol Dietol. 2015;61:235-247. [PubMed] |