Published online Jun 7, 2016. doi: 10.3748/wjg.v22.i21.5042
Peer-review started: December 4, 2015
First decision: February 18, 2016
Revised: February 24, 2016
Accepted: March 14, 2016
Article in press: March 14, 2016
Published online: June 7, 2016
Processing time: 178 Days and 20.2 Hours
AIM: To compare the effect of transarterial chemoembolization (TACE) plus GRGDSP (Gly-Arg-Gly-Asp-Ser-Pro, integrin-inhibitor) loaded nanoparticles with TACE alone or TACE + GRGDSP in a rat model of liver tumor.
METHODS: Morris hepatoma 3924A tumors were implanted in the livers of 30 ACI rats. The ACI rats were divided randomly into three groups (10 animals each). Tumor volume before treatment (V1) was examined by magnetic resonance imaging (MRI), and then, after laparotomy and placement of a PE-10 catheter into the hepatic artery, the following interventional protocols were performed: TACE (mitomycin C + lipiodol + degradable starch microspheres) + GRGDSP loaded nanoparticles for group A; TACE + GRGDSP for group B (control group 1); TACE alone for group C (control group 2). Tumor volume (V2) was assessed by MRI and the mean ratio of the post-treatment to pretreatment tumor volumes (V2/V1) was calculated. Immunohistochemical analysis was performed to assess the quantification of matrix metalloprotein 9 (MMP-9) and vascular endothelial growth factor (VEGF) positive tumor cells in each treatment group.
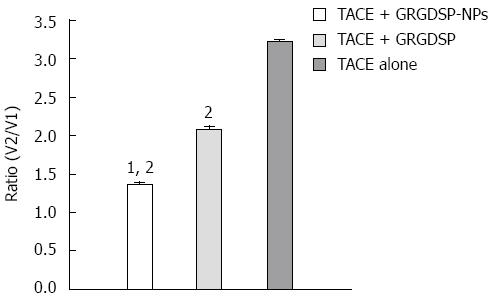
RESULTS: The mean tumor growth ratios (V2/V1) were 1.3649 ± 0.1194 in group A, 2.0770 ± 0.1595 in group B, and 3.2148 ± 0.1075 in group C. Compared with groups B and C, group A showed a significant reduction in tumor volume. Lower expression of MMP-9 and VEGF in hepatocellular carcinoma was observed in group A than in groups B and C. The angiogenesis of tumor was evaluated using anti-VEGF antibodies, and the metastasis of tumor was assessed using anti-MMP-9 antibody. MMP-9 and VEGF were expressed in all specimens. The immunoexpression of these proteins was confirmed by the presence of red cytoplasmic staining in tumor cells. Lower expression of MMP-9 and VEGF in hepatocellular carcinoma was observed in group A than in groups B and C.
CONCLUSION: Transarterial administration of integrin inhibitor loaded nanoparticles combined with TACE evidently retards tumor growth and intrahepatic metastases compared with TACE alone or TACE plus integrin inhibitor in an animal model of hepatocellular carcinoma.
Core tip: Our experimental study was designed to reduce tumor progression and recurrence through a combination of transarterial administration of GRGDSP (integrin-inhibitor) loaded nanoparticles plus transarterial chemoembolization (TACE) in an animal model of liver tumor. Our data showed that the combined biological and interventional treatment is a safe and effective therapy compared with TACE alone or TACE plus GRGDSP. The combined multimodal targeting therapies exhibit tremendous advantages over conventional interventional therapy alone.
- Citation: Qian J, Oppermann E, Tran A, Imlau U, Qian K, Vogl TJ. Transarterial administration of integrin inhibitor loaded nanoparticles combined with transarterial chemoembolization for treating hepatocellular carcinoma in a rat model. World J Gastroenterol 2016; 22(21): 5042-5049
- URL: https://www.wjgnet.com/1007-9327/full/v22/i21/5042.htm
- DOI: https://dx.doi.org/10.3748/wjg.v22.i21.5042
Hepatocellular carcinoma (HCC) is one of the most common malignances worldwide and it has a poor prognosis due to its rapid infiltration, liver cirrhosis and metastases. Surgical resection and liver transplantation are regarded as potentially curative therapies for patients with HCC[1,2]. However, most patients are not suitable candidates for surgical approaches because of liver dysfunction, extrahepatic metastases, lack of donor organs and high recurrence rates. Currently, transarterial chemoembolization (TACE), percutaneous ethanol injection, radiofrequency ablation, microwave coagulation therapy, laser induced thermotherapy and cryotherapy are important components for minimally invasive therapy in patients with cirrhosis and unresectable primary or metastatic liver tumors[3-5]. TACE has been shown to reduce systemic toxicity and increase local effects and thus improve the therapeutic results[6]. However, the long-term survival rate of patients has not been substantiated in randomized clinical studies, mainly due to the tumor recurrence and metastases after treatment[5]. While it is well known that tumor metastasis is a multifactorial process, one key to tumor cell infiltration and metastasis is integrin-mediated adhesion of tumor cells to the normal basement membrane.
Integrin is a king of receptor molecules on the surface of cells, and the basic function of which is to mediate the intercellular adherence or adherence between cells and extracellular matrix (ECM). Integrin expressed by tumor cells and host cells can promote the progress of metastatic dissemination. Recently, studies of anti-integrin therapies are drawing more and more attention to the treatments that protect against recurrence and metastasis of tumors[7]. It was demonstrated that transarterial infusion of GRGDSP (Gly-Arg-Gly-Asp-Ser-Pro integrin-inhibitor which includes RGD-peptide) combined with TACE noticeably inhibited the growth of liver tumor in Wistar rats[8].
It is well known that the nanoparticles considered as drug carriers in the targeting treatment can change the drug distribution in the body, besides the benefit feature of slow drug release. Nanoparticles can be combined with different kinds of drugs or ligands for targeted drug delivery[9,10]. The nanoparticle-therapies have the potential to enhance the effect for inhibiting tumor proliferation and angiogenesis[11,12]. It was reported that the therapeutic effect of chemotherapeutic drug on liver tumor could be noticeably enhanced by the administration of nanoparticles via the hepatic artery. The rats that received Adriamycin loaded nanoparticles acquired obvious inhibition on tumor growth, as well as prolonged their survival[11,12]. However, to our knowledge, there have been no experimental or clinical reports on the therapeutic effectiveness of TACE combined with integrin inhibitor-loaded nanoparticles for treatment of HCC. Thus, the purpose of our study was to assess the effect of TACE combined with GRGDSP loaded nanoparticles, compared with TACE alone or TACE plus GRGDSP for treating HCC in an animal model.
Morris hepatoma 3924A tumors, poorly differentiated HCC, was used in this study. The hepatoma cells were obtained from the German Cancer Research Center in Heidelberg. Thirty male ACI rats (200-220 g) were obtained from Harlan Winkelmann (Borchen, Germany). The experiments were performed in accordance with the German government and the institutional animal research review board. All the experiments were carried out under intraperitoneal anesthesia with ketamine hydrochloride (100 mg/kg), xylazin hydrochloride (15 mg/kg), and atropine sulfate (0.1 mg/kg).
Tumor implantation (day 0) was performed according to the method described by Yang et al[13] with slight modification[14]. The tumor tissue was recovered from an animal 12 d after subcutaneous implantation (5 × 106 tumor cells) and cut into small cubes (ca. 2 mm). The left lateral lobe of the liver of the recipient rat was exposed through a subxiphoid abdominal incision and a small subcapsular incision was made. The tumor fragment was gently embedded into the pocket and the abdominal wall was subsequently closed.
GRGDSP loaded nanoparticles were kindly provided by School of Life Science and Technology, Huazhong University of Science and Technology (Wuhan, China). GRGDSP loaded nanoparticles were synthesized using the method of Yang et al[15] with slight modifications. Superparamagnetic iron oxide (SPIO) was used as RGD (Arg-Gly-Asp) nanocarriers. The size and size distribution of the final product were determined by photon correlation spectroscopy (PCS) with a nano-ZS90 laser particle analyzer (Malvern Instruments Corp., United Kingdom). The mean diameter of particles was 107 nm, and the drug loading ratio was 50%.
A dose of 0.25 mg GRGDSP loaded nanoparticles was suspended in 0.6 mL OF 0.9% NaCl for 10 min before administration.
A dose of 0.1 mg mitomycin, 0.1 mL lipiodol and 5.0 mg degradable starch microspheres was administered into the hepatic artery of the rats in the experiment.
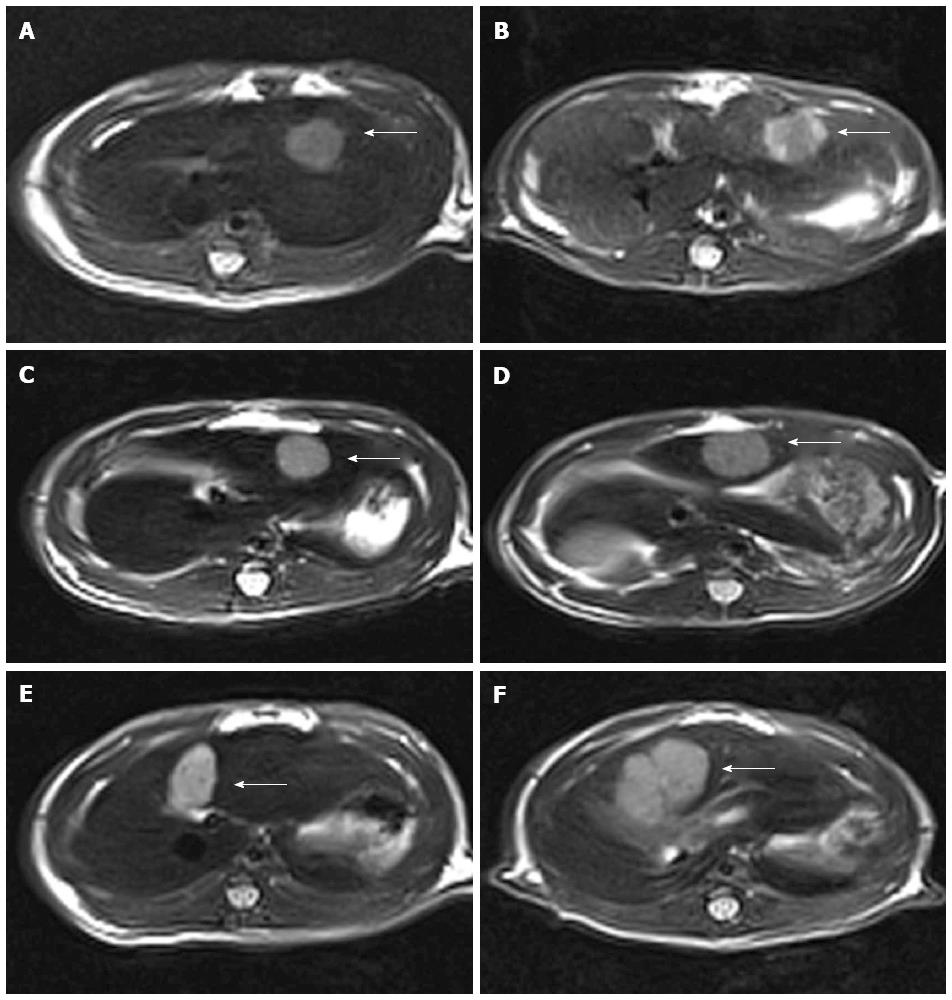
One day before and 12 d after the interventional therapy, MRI was performed with a 3.0 Tesla Magnetom superconducting system (Siemens; Erlangen, Germany) using a wrist coil. MR images of the liver were acquired in the transverse plane using a T2-weighted turbo spin-echo sequence with the following imaging parameters: TR/TE, 3870/80 ms; slice thickness, 2 mm; matrix, 192 × 256. The tumor volume was evaluated in T2-weighted images according to the ellipsoid volume formula[16]: V = 0.5 × d1 × d22 (d1 = maximum diameter of the tumor; d2 = minimum diameter perpendicular to d1).
A second laparotomy was performed 1 d after MRI examination for interventional treatment. A PE-10 polyethylene catheter (inner diameter 0.28 mm, outer diameter 0.61 mm, Wenzel, Heidelberg, Germany) was used for catheterization under a microscope. The catheter was inserted retrogradely into the gastroduodenal artery and pushed to the common hepatic artery. The following therapeutic agents were injected through the catheter to the hepatic artery by sandwich technique (sequential injection of lipiodol + mitomycin + GRGDSP loaded nanoparticles or GRGDSP + degradable starch microspheres):
Group A (TACE + GRGDSP loaded nanoparticles; n = 10): 0.1 mg mitomycin + 0.1 mL lipiodol + 5.0 mg degradable starch microspheres + 0.25 mg GRGDSP loaded nanoparticles.
Group B (control group 1, TACE + GRGDSP; n = 10): 0.1 mg mitomycin + 0.1 mL lipiodol + 5.0 mg degradable starch microspheres + 0.25 mg GRGDSP (2.5 mg/mL, Jingmei Biological, Wuhan, China).
Group C (control group 2, TACE alone; n = 10): 0.1 mg mitomycin + 0.1 mL lipiodol + 5.0 mg degradable starch microspheres.
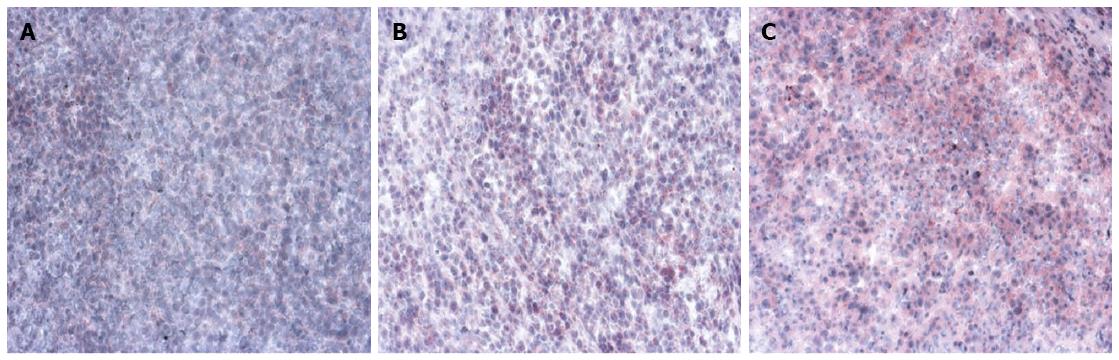
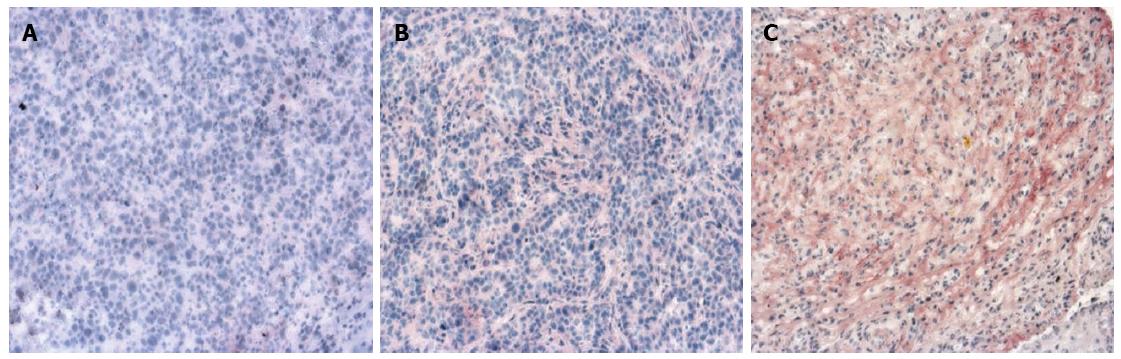
All rats were sacrificed after the MRI examination by intravenous administration of overdose sodium pentobarbital. Liver samples were embedded and frozen in Tissue-Tek and 5 μm cryosections were generated. Sections were fixed in 100% acetone and endogenous peroxidase activity was blocked with 0.6% H2O2/MeOH followed by incubation with anti-MMP-9 rabbit polyclonal antibody (Cell Signaling Technology Inc., MA, United States) and/or anti-VEGF rabbit polyclonal antibody (Santa Cruz Biotechnology Inc., United States) overnight at 4 °C. Sections were then incubated with an anti-rabbit alkaline phosphatase supervision polymer system (DCS Innovative Diagnostik-Systeme, Hamburg, Germany), and endogenous alkaline phosphatase was inhibited by 1 mmol/L levamisole present in the substrate. Sections were subsequently counterstained with hematoxylin and mounted in Kaisers Glycerol Gelatin (Merck, Darmstadt, Germany). To evaluate the expression of MMP-9 and VEGF, all slides were examined and scored by two independent pathologists who were blinded to the animal data. Stained cells were counted in 10 microscopic fields (× 100) per slide in tumor area and the average was calculated. Slides were evaluated in a semiquantitative method relating to the percentage staining of the cells and were scored as follows: 0 (No staining); 1 (0%-5%); 2 (6%-25%); 3 (26%-50%); 4 (51%-75%); and 5 (76%-100%).
The mean tumor growth ratio (V2/V1) by MRI from each group and the significance of differences were analyzed using the statistical software Prism (version 3.02, La Jolla, CA, United States).
Immunohistochemical staining of MMP-9 and VEGF was evaluated using descriptive and semiquantitative methods. Statistical analyses were performed using Prism (version 3.02, La Jolla, CA, United States).
Comparisons between groups were made using the Bonferroni test. Differences with a P-value less than 0.05 were considered statistically significant.
Tumor implantation was successful in all of the rats. Most tumors appeared homogeneous and were hypointense on T1-weighted images and hyperintense on T2-weighted images prior to treatment, but inhomogeneous after treatment. The mean growth ratios of tumors [V2 (postreatment)/V1 (pretreatment)] were 1.3649 ± 0.1194 in group A, 2.0770 ± 0.1595 in group B, and 3.2148 + 0.1075 in group C. Compared to groups B and C, group A (TACE + GRGDSP loaded nanoparticles) showed a significant reduction of tumor growth (P < 0.01) in the period of observation by Bonferroni test (Figures 1 and 2).
The angiogenesis of tumor was evaluated using anti-VEGF antibody, and the metastasis of tumor was assessed using anti-MMP-9 antibody. MMP-9 and VEGF were expressed in all specimens. The immunoexpression of these proteins was confirmed by the presence of red cytoplasmic staining in tumor cells (Figures 3 and 4). Lower expression of MMP-9 and VEGF in HCC were observed in the group A than in groups B and C (controls) (P < 0.01) (Tables 1 and 2).
| Group | TACE + GRGDSP | TACE + GRGDSP loaded nanoparticles | P value | |||||||||||
| Score | 0 | 1 | 2 | 3 | 4 | 5 | 0 | 1 | 2 | 3 | 4 | 5 | ||
| VEGF | Tumor | 3 | 13 | 28 | 20 | 20 | 16 | 29 | 5 | 18 | 20 | 18 | 10 | 0.000 |
| MMP-9 | Tumor | 0 | 1 | 17 | 18 | 42 | 22 | 0 | 6 | 36 | 41 | 12 | 5 | 0.000 |
| Group | TACE | TACE + GRGDSP loaded nanoparticles | P value | |||||||||||
| Score | 0 | 1 | 2 | 3 | 4 | 5 | 0 | 1 | 2 | 3 | 4 | 5 | ||
| VEGF | Tumor | 0 | 2 | 10 | 11 | 16 | 61 | 29 | 5 | 18 | 20 | 18 | 10 | 0.000 |
| MMP-9 | Tumor | 0 | 6 | 4 | 0 | 9 | 81 | 0 | 6 | 36 | 41 | 12 | 5 | 0.000 |
HCC is one of the most common malignancies with very high morbidity and mortality. TACE is a widely used palliative treatment for patients with unresectable HCC[4]. However, it has not led to significant improvements in the long-term survival rates, because of postoperative metastasis and recurrence of tumors[8]. Local infiltration and metastasis of tumors are a complicated process which is influenced by many factors.
The mechanism of adhesion molecules was reported to play an important role in the regulation of cellular migration, proliferation and apoptosis[17,18]. Integrin receptors are abnormally expressed on the surface of tumor cells, where they perform the basic function of mediating intercellular and cell-extracellular matrix (ECM) adherence. The adhesive function of integrins works by identifying the specific RGD sequence in the ligand (one part of ECM) and the links to it. RGD peptide is a kind of extrinsic peptide, which can competitively bind to integrin and inhibit binding with the RGD sequence of the ECM. The integrin-mediated adherence between tumor cells and ECM can be decreased by RGD peptide, and the inhibitory action is dose dependent. Furthermore, degradation of ECM caused by MMP-9 can be inhibited. Binding of RGD to integrin receptor αvβ3, which is abnormally expressed in the endothelial cells of tumor blood vessels, may prevent blood vessel formation and infiltrating[17,19-21]. Therefore, RGD peptide can be regarded as a broad-spectrum antagonist of integrin. As a synthetic linear RGD peptide, GRGDSP (Gly-Arg-Gly-Asp-Ser-Pro) could inhibit the adherence of tumor cells to endothelial cells of blood vessels and limit its metastasis[22-24]. Tsuchiya et al[20] have found that intravenous administration of synthetic RGD pseudo-peptide (FC-336) could inhibit intrahepatic metastasis compared with control group (P < 0.05). Typically, a previous study has demonstrated that transarterial infusion of GRGDSP combined with TACE noticeably inhibited the growth of hepatic carcinoma and intrahepatic metastasis in Walker-256 rats[8]. Recently, nanobiotechnology has many advantages for improving drug delivery by the following approaches[10]. First, particle size can be reduced to nanometer size range to increase the surface area, thereby increasing the rate of dissolution. Second, nanoparticles can improve the absorption of insoluble compounds and macromolecules, enhance the bioavailability and release rates, and therefore reduce the amount of dose required and side effects. Finally, nanoparticles can be combined with ligands for targeted drug delivery. Nanotechnology is particularly useful for delivery of biological therapies. Nanotechnology will enable design and delivery of more effective drugs with increased efficacy and reduced toxicity. Wang et al[25] have showed that the nanoparticles coupled with RGD-peptide and doxorubicin represent high efficacy in inducing apoptosis in specific malignant cancer cells. Iwasaki et al[9] demonstrated that nanoparticles can be intravenously administrated for delivery of therapeutic genes with anti-tumor activity into human liver tumors. It was also reported that the therapeutic effect of adriamycin on liver malignancy can be significantly enhanced by its nanoparticle formulation and administration via the hepatic artery[11,12]. However, up to date no study has reported on the therapeutic effect of nanoparticles combined with TACE for treating HCC in vivo or in clinic. Thus, our experimental study was designed to reduce tumor progression and recurrence by combination of transarterial administration of GRGDSP loaded nanoparticles with TACE by using sandwich technique in an animal model of HCC. Our experimental results showed that transarterial administration of GRGDSP loaded nanoparticles + TACE can significantly inhibit the growth of hepatic tumor and intrahepatic metastases. Lower expression of MMP-9 and VEGF in HCC was observed in the group A (TACE + GRGDSP loaded-nanoparticles) than in group B (TACE + GRGDSP) and group C (TACE alone). The invasive progression and metastases of tumor cells in group A were noticeably inhibited compared with the control groups.
For application in TACE, lipiodol not only occludes the small arteries supplying the tumor, but can also be used as the carriers bringing the anticancer drugs to the tumor. Lipiodol can deliver cytotoxic agents directly into tumor cells and endothelial cells, enter into the microcirculation of the tumor and block the blood flow[26-28]. Anticancer drugs administered through TACE can escape first-pass metabolism and have a prolonged half-life[29]. Moreover, the currently synthesized GRGDSP loaded nanoparticles have a mean diameter of 107 nm. It was documented that the passive targeting ability of the nanoparticles depends on the vessel microstructures of target organs. The nanoparticles with a diameter ranging from 20 to 300 nm have also the ability to directly enter the hepatocytes[30].
In conclusion, encouraging results were obtained by combining transarterial administration of integrin inhibitor loaded nanoparticles with TACE for treating HCC in rats in comparison with control groups, and may prove valuable to human application as a therapeutic approach for the treatment of HCC. The combined multimodal targeting therapies reveal their enormous advantages as compared with conventional interventional therapy alone. However, detailed therapeutic mechanisms, therapeutic indications, monitoring and side effects of these combined therapies remain unclear and require more randomized experimental studies.
The authors would like to thank all of those who participated in this study, and would like to thank professor Yan-Hong Zhu and Xiang-Liang Yang, The School of Life Science and Technology, Huazhong University of Science & Technology, Wuhan 430074, Hubei Province, China, for their selfless help to the preparation of the nanoparticles.
Transarterial chemoembolization (TACE) was introduced as one of the most common forms of interventional therapy but its therapeutic effect combined with gene therapy remains to be elucidated.
Hepatocellular carcinoma (HCC) is one of the most commonly occurring tumors worldwide and TACE was introduced as an effective treatment in patients with unresectable HCC. Integrins expressed by tumor cells and host cells can contribute directly to the control and progress of metastatic dissemination. The authors have previously demonstrated the encouraging results of interventional therapy of TACE plus GRGDSP compared with TACE or GRGDSP alone. Transarterial infusion of integrin inhibitor (GRGDSP) loaded nanoparticles plus TACE may be a safe and effective therapy targeting metastatic dissemination of tumor cells.
This study for the first time evaluates the effects of TACE plus GRGDSP loaded nanoparticles compared with TACE alone or TACE plus GRGDSP for treating HCC in an animal model. Its results indicate that transarterial administration of GRGDSP loaded nanoparticles combined with TACE evidently retards tumor growth and intrahepatic metastases compared with TACE alone or TACE plus GRGDSP in rats.
Integrin inhibitor loaded nanoparticles combined with TACE might be used as a new therapeutic approach for the treatment of HCC and inhibition of intrahepatic metastasis after TACE.
Integrin is a receptor molecule on the surface of cells, and its basic function is to mediate the intercellular adherence or adherence between cells and extracellular matrix. GRGDSP (Gly-Arg-Gly-Asp-Ser-Pro, integrin-inhibitor) can prevent the adhesion of tumor cells and endothelial cells of blood vessels, and also inhibit the metastasis of tumor cells.
The manuscript investigated the effect of combined administration of TACE and GRGDSP-conjugated nanoparticles in a rat model of HCC. The study appears to be well performed and the manuscript is well written. This study is more a pilot study rather than an exhaustive study, but is worth publishing. Subsequent studies should be performed to localize the nanoparticles shortly after the administration and to investigate the distribution of the nanoparticles in the liver.
P- Reviewer: Piiper A S- Editor: Gong ZM L- Editor: Wang TQ E- Editor: Zhang DN
| 1. | Chen X, Liu HP, Li M, Qiao L. Advances in non-surgical management of primary liver cancer. World J Gastroenterol. 2014;20:16630-16638. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 43] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 2. | Bellissimo F, Pinzone MR, Cacopardo B, Nunnari G. Diagnostic and therapeutic management of hepatocellular carcinoma. World J Gastroenterol. 2015;21:12003-12021. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 51] [Cited by in RCA: 54] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 3. | Li D, Kang J, Madoff DC. Locally ablative therapies for primary and metastatic liver cancer. Expert Rev Anticancer Ther. 2014;14:931-945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 4. | Rou WS, Lee BS, Moon HS, Lee ES, Kim SH, Lee HY. Risk factors and therapeutic results of early local recurrence after transcatheter arterial chemoembolization. World J Gastroenterol. 2014;20:6995-7004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 5. | Qian J, Feng GS, Vogl T. Combined interventional therapies of hepatocellular carcinoma. World J Gastroenterol. 2003;9:1885-1891. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 61] [Cited by in RCA: 56] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 6. | Llovet JM, Real MI, Montaña X, Planas R, Coll S, Aponte J, Ayuso C, Sala M, Muchart J, Solà R. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet. 2002;359:1734-1739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2502] [Cited by in RCA: 2611] [Article Influence: 113.5] [Reference Citation Analysis (0)] |
| 7. | Cai W, Chen X. Anti-angiogenic cancer therapy based on integrin alphavbeta3 antagonism. Anticancer Agents Med Chem. 2006;6:407-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 179] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 8. | Qian J, Yin J, Liang H, Wang Y, Feng G. Experimental study on transarterial administration of GRGDSP combined with transarterial chemoembolization in rats with hepatic carcinoma. Cardiovasc Intervent Radiol. 2008;31:377-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 9. | Iwasaki Y, Ueda M, Yamada T, Kondo A, Seno M, Tanizawa K, Kuroda S, Sakamoto M, Kitajima M. Gene therapy of liver tumors with human liver-specific nanoparticles. Cancer Gene Ther. 2007;14:74-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 10. | Jain KK. Nanomedicine: application of nanobiotechnology in medical practice. Med Princ Pract. 2008;17:89-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 133] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 11. | Chen JH, Ling R, Yao Q, Wang L, Ma Z, Li Y, Wang Z, Xu H. Enhanced antitumor efficacy on hepatoma-bearing rats with adriamycin-loaded nanoparticles administered into hepatic artery. World J Gastroenterol. 2004;10:1989-1991. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 10] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (2)] |
| 12. | Chen JH, Wang L, Ling R, Li Y, Wang Z, Yao Q, Ma Z. Body distribution of nanoparticle-containing adriamycin injected into the hepatic artery of hepatoma-bearing rats. Dig Dis Sci. 2004;49:1170-1173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | Yang R, Rescorla FJ, Reilly CR, Faught PR, Sanghvi NT, Lumeng L, Franklin TD, Grosfeld JL. A reproducible rat liver cancer model for experimental therapy: introducing a technique of intrahepatic tumor implantation. J Surg Res. 1992;52:193-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 14. | Qian J, Truebenbach J, Graepler F, Pereira P, Huppert P, Eul T, Wiemann G, Claussen C. Application of poly-lactide-co-glycolide-microspheres in the transarterial chemoembolization in an animal model of hepatocellular carcinoma. World J Gastroenterol. 2003;9:94-98. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 30] [Cited by in RCA: 27] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 15. | Yang X, Hong H, Grailer JJ, Rowland IJ, Javadi A, Hurley SA, Xiao Y, Yang Y, Zhang Y, Nickles RJ. cRGD-functionalized, DOX-conjugated, and 64Cu-labeled superparamagnetic iron oxide nanoparticles for targeted anticancer drug delivery and PET/MR imaging. Biomaterials. 2011;32:4151-4160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 377] [Cited by in RCA: 306] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 16. | Carlsson G, Gullberg B, Hafström L. Estimation of liver tumor volume using different formulas - an experimental study in rats. J Cancer Res Clin Oncol. 1983;105:20-23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 223] [Cited by in RCA: 225] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 17. | Hynes RO. Integrins: bidirectional, allosteric signaling machines. Cell. 2002;110:673-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6323] [Cited by in RCA: 6571] [Article Influence: 285.7] [Reference Citation Analysis (0)] |
| 18. | Hynes RO. Integrins: versatility, modulation, and signaling in cell adhesion. Cell. 1992;69:11-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7430] [Cited by in RCA: 7404] [Article Influence: 224.4] [Reference Citation Analysis (0)] |
| 19. | Jia JB, Zhuang PY, Sun HC, Zhang JB, Zhang W, Zhu XD, Xiong YQ, Xu HX, Tang ZY. Protein expression profiling of vascular endothelial growth factor and its receptors identifies subclasses of hepatocellular carcinoma and predicts survival. J Cancer Res Clin Oncol. 2009;135:847-854. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 20. | Tsuchiya Y, Sawada S, Tsukada K, Saiki I. A new pseudo-peptide of Arg-Gly-Asp (RGD) inhibits intrahepatic metastasis of orthotopically implanted murine hepatocellular carcinoma. Int J Oncol. 2002;20:319-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 21. | Hall H, Djonov V, Ehrbar M, Hoechli M, Hubbell JA. Heterophilic interactions between cell adhesion molecule L1 and alphavbeta3-integrin induce HUVEC process extension in vitro and angiogenesis in vivo. Angiogenesis. 2004;7:213-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 22. | Sheu JR, Lin CH, Peng HC, Huang TF. Triflavin, an Arg-Gly-Asp-containing peptide, inhibits the adhesion of tumor cells to matrix proteins via binding to multiple integrin receptors expressed on human hepatoma cells. Proc Soc Exp Biol Med. 1996;213:71-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 23. | Takayama T, Suzuki N, Narukawa M, Goldberg HA, Otsuka K, Ito K. Enamel matrix derivative is a potent inhibitor of breast cancer cell attachment to bone. Life Sci. 2005;76:1211-1221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 24. | Joshi P, Chung CY, Aukhil I, Erickson HP. Endothelial cells adhere to the RGD domain and the fibrinogen-like terminal knob of tenascin. J Cell Sci. 1993;106:389-400. [PubMed] |
| 25. | Wang Z, Chui WK, Ho PC. Design of a multifunctional PLGA nanoparticulate drug delivery system: evaluation of its physicochemical properties and anticancer activity to malignant cancer cells. Pharm Res. 2009;26:1162-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 76] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 26. | Chen MS, Li JQ, Zhang YQ, Lu LX, Zhang WZ, Yuan YF, Guo YP, Lin XJ, Li GH. High-dose iodized oil transcatheter arterial chemoembolization for patients with large hepatocellular carcinoma. World J Gastroenterol. 2002;8:74-78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 74] [Cited by in RCA: 79] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 27. | Savastano S, Miotto D, Casarrubea G, Teso S, Chiesura-Corona M, Feltrin GP. Transcatheter arterial chemoembolization for hepatocellular carcinoma in patients with Child’s grade A or B cirrhosis: a multivariate analysis of prognostic factors. J Clin Gastroenterol. 1999;28:334-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 54] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 28. | Bhattacharya S, Dhillon AP, Winslet MC, Davidson BR, Shukla N, Gupta SD, Al-Mufti R, Hobbs KE. Human liver cancer cells and endothelial cells incorporate iodised oil. Br J Cancer. 1996;73:877-881. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 36] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 29. | Kalva SP, Iqbal SI, Yeddula K, Blaszkowsky LS, Akbar A, Wicky S, Zhu AX. Transarterial chemoembolization with Doxorubicin-eluting microspheres for inoperable hepatocellular carcinoma. Gastrointest Cancer Res. 2011;4:2-8. [PubMed] |
| 30. | Shen LF, Zhang YD, Shen HJ, Zeng S, Wang X, Wang C, Le Y, Shen H. Liver targeting and the delayed drug release of the nanoparticles of adriamycin polybutylcyanoacrylate in mice. Chin Med J (Engl). 2006;119:1287-1293. [PubMed] |