Published online Nov 7, 2015. doi: 10.3748/wjg.v21.i41.11862
Peer-review started: June 14, 2015
First decision: July 14, 2015
Revised: August 7, 2015
Accepted: September 30, 2015
Article in press: September 30, 2015
Published online: November 7, 2015
Processing time: 146 Days and 1.6 Hours
AIM: To evaluate ATP-binding cassette (ABC) transporters in colonic pathophysiology as they had recently been related to colorectal cancer (CRC) development.
METHODS: Literature search was conducted on PubMed using combinations of the following terms: ABC transporters, ATP binding cassette transporter proteins, inflammatory bowel disease, ulcerative, colitis, Crohns disease, colorectal cancer, colitis, intestinal inflammation, intestinal carcinogenesis, ABCB1/P-glycoprotein (P-gp/CD243/MDR1), ABCC2/multidrug resistance protein 2 (MRP2) and ABCG2/breast cancer resistance protein (BCRP), Abcb1/Mdr1a, abcc2/Mrp2, abcg2/Bcrp, knock-out mice, tight junction, membrane lipid function.
RESULTS: Recently, human studies reported that changes in the levels of ABC transporters were early events in the adenoma-carcinoma sequence leading to CRC. A link between ABCB1, high fat diet and gut microbes in relation to colitis was suggested by the animal studies. The finding that colitis was preceded by altered gut bacterial composition suggests that deletion of Abcb1 leads to fundamental changes of host-microbiota interaction. Also, high fat diet increases the frequency and severity of colitis in specific pathogen-free Abcb1 KO mice. The Abcb1 KO mice might thus serve as a model in which diet/environmental factors and microbes may be controlled and investigated in relation to intestinal inflammation. Potential molecular mechanisms include defective transport of inflammatory mediators and/or phospholipid translocation from one side to the other of the cell membrane lipid bilayer by ABC transporters affecting inflammatory response and/or function of tight junctions, phagocytosis and vesicle trafficking. Also, diet and microbes give rise to molecules which are potential substrates for the ABC transporters and which may additionally affect ABC transporter function through nuclear receptors and transcriptional regulation. Another critical role of ABCB1 was suggested by the finding that ABCB1 expression identifies a subpopulation of pro-inflammatory Th17 cells which were resistant to treatment with glucocorticoids. The evidence for the involvement of ABCC2 and ABCG2 in colonic pathophysiology was weak.
CONCLUSION: ABCB1, diet, and gut microbes mutually interact in colonic inflammation, a well-known risk factor for CRC. Further insight may be translated into preventive and treatment strategies.
Core tip: Recently, human studies reported that changes in the levels of ATP-binding cassette (ABC) transporters were early events in the adenoma-carcinoma sequence leading to colorectal cancer. A link between ABCB1, high fat diet and gut microbes in relation to colitis was suggested by the animal studies. The Abcb1 KO mice might thus serve as a model in which diet/environmental factors and microbes may be controlled and investigated in relation to intestinal inflammation. Such strategy may provide insight which can be translated into preventive and treatment strategies to benefit the patients.
- Citation: Andersen V, Svenningsen K, Knudsen LA, Hansen AK, Holmskov U, Stensballe A, Vogel U. Novel understanding of ABC transporters ABCB1/MDR/P-glycoprotein, ABCC2/MRP2, and ABCG2/BCRP in colorectal pathophysiology. World J Gastroenterol 2015; 21(41): 11862-11876
- URL: https://www.wjgnet.com/1007-9327/full/v21/i41/11862.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i41.11862
Colorectal cancer (CRC) constitutes the third most common cancer in the world and the second leading cause of cancer-related deaths. The number of cases is increasing and has been estimated to raise from 1.4 million cases in 2012 to 2.4 million cases in 2035 worldwide[1]. Early detection of CRC is important as early treatment has been associated with improved outcomes and saved lives[2]. Therefore, population screening programs have been initiated in a number of countries such as the United Kingdom, Australia, Holland and Denmark[3-6]. The fecal occult blood test (FOBT) is the most widely used for population screening[7] and individuals with a positive FOBT are referred for an endoscopic investigation of the colonic mucosa thereby enabling the sampling of biopsies from the colonic mucosa.
Recently, a major part of research had focused on improving prognosis and treatment selection in CRC[8-10]. Another approach could be to prevent the development of cancer in subgroups of patients with high risk, i.e., secondary prevention. Thus, the molecular evaluation of the (unaffected) colonic mucosa from the patients undergoing an endoscopic evaluation could potentially stratify the patients according to their risk of developing CRC. Our recent findings indicate that even healthy looking mucosa as determined by histology may contain a significantly elevated level of immune response proteins[11]. Biomarkers potentially predicting the disease risk among selected patient groups could improve the efficiency of the screening programs and patient care. Furthermore, they have the potential to dramatically alter the established patient care pathways as follow-up of the patients may be tailored according to their individual risk and thereby the organization and use of resources of the health care system.
CRC develops in the colonic mucosa which is highly affected by the metabolic activities in the intestinal lumen. The dietary items reaching the colon are digested by the commensal bacteria giving rise to various substrates which may prevent, initiate or promote colorectal cancer development[12]. Thus, in order to understand the processes leading to CRC we need to take into account the delicate interactions between dietary intake, activity of the commensal bacteria and host factors.
We recently reported that low ABCB1 and ABCG2 gene transcription levels and high ABCC2 levels are early events in the colorectal adenoma-carcinoma sequence[13,14] suggesting that changes in expression levels of the ATP binding cassette (ABC) transporter proteins [EC 3.6.3.44] precede cancer development. In addition, inflammatory bowel disease (IBD) may be a risk factor for the development of CRC[8]. Therefore, we wanted to discuss the current understanding of how these ABC transporters may affect intestinal inflammation and carcinogenesis, how they may potentially interact with the environment such as diet and gut microbes, and whether this knowledge may be utilized for improved treatment care strategies.
Literature search was conducted on PubMed using combinations of the following terms: ABC transporters, ATP binding cassette transporter proteins, inflammatory bowel disease, ulcerative, colitis, Crohn’s disease, colorectal cancer, colitis, intestinal inflammation, intestinal carcinogenesis, ABCB1/P-glycoprotein (P-gp/CD243/MDR1), ABCC2/multidrug resistance protein 2 (MRP2) and ABCG2/breast cancer resistance protein (BCRP), Abcb1/Mdr1a, abcc2/Mrp2, abcg2/Bcrp, knock-out mice, tight junction, membrane lipid function.
The large family of ABC transporter proteins is highly conserved through evolution and extensive sequence and protein homology is shared between numerous bacterial and eukaryotic ABC transport proteins[15]. The ABC proteins are found in the cell membranes and intracellular organelles and the ABC family members exert multiple different functions depending on the cellular context[16].
The ABCB1, ABCC2, and ABCG2 transporters, encoded by ABCB1, ABCC2, and ABCG2, respectively, are located in the apical cell membrane of epithelial and endothelial interfaces within the intestine, testis, kidneys, liver, brain, and placenta[17-20]. Thereby, they exert barrier functions influencing absorption, distribution, excretion, and toxicology (ADME-Tox) of exogenous substrates with potential impact on inflammation and carcinogenesis[21-25]. ABCB1 and ABCG2 transporters have also been identified on haematological cells[20,26,27]. Whereas ABCB1 has been extensively studied in relation to the gastrointestinal system[28], less is known for ABCC2 and ABCG2[29].
No monogenic diseases have been identified involving ABCB1 and ABCG2[30,31], but several different mutations in ABCC2 have been observed in patients with Dubin-Johnson syndrome, an autosomal recessive disorder characterized by conjugated hyperbilirubinemia[32].
Nuclear receptors such as aryl hydrocarbon receptor (AHR), pregnane x receptor (PXR, NR1I2), vitamin D receptor (VDR, NR1I1), and constitutive androstane/activated receptor (NR1I3) are activated by a wide variety of exogenous and endogenous factors including diet, heavy metals, gut microbes, carcinogens and inflammation[33,34] (reviewed in[35]). These nuclear receptors may be involved in the transcriptional regulation of ABC transporters[34,36-40] as are the transcription factors nuclear factor kappa B (NF-κB), activator protein 1 (AP-1)[41], and Wnt signaling transcription factor TCF4[42]. Furthermore, ABCB1 undergoes several posttranslational modifications (PTMs)[43,44] which have been shown to affect the stability of ABCB1 and/or substrate transport specificities[45]. ABCB1 is a 170-180 kDa glycoprotein with N-linked glycosylation at residues Asp91, Asp94 and Asp99. ABCB1 and ABCC2 have two ATP-binding sites and two six-transmembrane domains in a symmetric structure whereas ABCG2 is a half-transporter and have one ATP binding site and one six-transmembrane domain.
ABC transporter substrates include many diverse endogenous and exogenous molecules including amino acids, peptides, metabolites, vitamins, fatty acids, steroids, phospholipids, conjugated organic anions, and dietary and environmental carcinogens, pesticides, metals, metalloids, lipid peroxidation products and drugs[22-24]. Substrate overlap has been reported between the ABCB1, ABCC2, ABCG2, and especially between ABCC2 and the basolaterally located ABCC1[23,29]. Specific substrates and their potential role in ABC transporter related gut inflammation will be discussed later in this review.
CRC is a heterogeneous disease complex with environmental, genetic and host factors involved in the aetiology[46,47]. Inflammation is a risk factor for CRC[48-50] and accordingly, a subset of patients with IBD[51,52] [with the two main forms ulcerative colitis (UC) and Crohn’s disease (CD)] characterised by long-term and extensive colitis are at high risk of CRC[53,54]. The incidences of both CRC and IBD are rising[1,55], which point to important roles of environment factors.
The intestinal mucosa is by far the body’s largest surface exposed to and interacting with environmental factors. The intestinal epithelium and the mucus form a barrier against luminal antigens and invading microbes[56,57]. Microbial sensing by intestinal epithelium cells and local innate lymphoid cells (ILCs) through pattern recognition receptors (PRR) leads to secretion of pro-inflammatory cytokines such as tumour necrosis factor-α (TNF-α), interferon-γ (INF-γ), interleukin 6 (IL-6), and IL-17[58,59], cytokines which have been related to IBD and CRC[60]. Activation of PRR stimulates autophagocytic networks[61,62]. Also, activation of the innate immune system may result in activation of the adaptive immune response with T cell involvement; Th1, Th2 and Th17 cells characterised by secretion of their signature cytokines INF-γ, IL-4, IL-17, respectively, whereas Tregs (and to a lesser degree Th2), in contrast, are characterised by their production of the anti-inflammatory cytokines IL-10 and transforming growth factor β (TGF-β)[63,64]. The role of the Th17-associated cytokines in animal models of colitis[65], IBD[66] and CRC[67] have been in focus the recent years and it has been suggested that Th17 cells may have evolved to combat bacterial and fungal infections via orchestration of the neutrophil inflammatory response[63]. However, this seems to be a simplistic view[68] and more T cell subsets with as yet unclarified functions in IBD and CRC have been identified these years[69-71].
Englund et al[72] found significantly lower levels of both ABCB1 and ABCG2 mRNA in colon and rectal biopsies from 16 patients with active UC compared to healthy individuals whereas the levels did not differ between UC patients in remission and healthy controls (Table 1). The authors also reported lower ABCB1 and ABCG2 levels in colon from patients with active inflammation compared with controls[72]. Langmann et al[40] reported low levels of ABCB1 and ABCC2 mRNA in biopsies from colon adjacent to inflammation from patients with UC compared to the levels in controls. In contrast, Deuring et al[73] reported similar levels of ABCG2 mRNA in intestinal biopsies from healthy individuals, patients in remission and patients with active inflammation but dramatically reduced levels of ABCG2 in IBD patients with active inflammation when compared to patients in remission or healthy controls using quantitative immunohistochemistry (Table 1). These observations suggest that the low levels of ABCG2 observed in inflamed colon were caused by posttranscriptional processes[73]. The study also found inflamed colon to contain high levels of the endoplasmic reticulum (ER)-stress marker GRP78 and in vitro they found nitric oxide induced ER-stress to impair ABCG2 function[73]. The authors therefore suggested that incorrect protein folding caused by inflammation-induced ER dysfunction may lead to low levels of ABCG2 in inflamed colon of IBD patients[73,74].
| Controls | Inactive disease | Active disease | Ref. | ||||||||
| Colon | Colon | P value | Rectum | P value | Colon | P value | Rectum | P value | |||
| Gene | ABCB11 | 1 (ref) | NA | NS | NA | 22% | < 0.001 | 34% | < 0.01 | [72] | |
| ABCC21 | 1 (ref) | NA | NS | NA | NA | NS | NA | NS | [72] | ||
| ABCG21 | 1 (ref) | NA | NS | NA | 11% | < 0.001 | 16% | < 0.001 | [72] | ||
| Array | ABCB12 | 287 | -1.5 | [40] | |||||||
| ABCC22 | 81 | -8.6 | [40] | ||||||||
| Protein | ABCG23 | 100 (9/9) | 80 (53/67) | 24 (13/54) | 0.01 | [73] | |||||
The role of ABC transporters has also been investigated in relation to CRC (Table 2). As previously mentioned, low levels of ABCB1 in colon was found to be an early event that preceded malignancy[13]. Similarly, in another study using the same cohort low levels of ABCG2 and high levels of ABCC2 mRNA were found in both colon adenomas and carcinomas compared to morphological normal tissue surrounding the cancer tissue, and compared to levels in tissue from healthy individuals[14]. Taken together, the studies suggest that changed expression levels of the ABC transport proteins may be early events in the development of IBD and CRC.
| Unaffected tissue | P value1 | Adenomas/carcinomas | P value1 | P value2 | Ref. | |
| ABCB1 | [13] | |||||
| Healthy individuals | 0.012 ± 0.008 | |||||
| Mild/moderate dysplasia cases | 0.009 ± 0.004 | NS | 0.005 ± 0.004 | < 0.050 | < 0.001 | |
| Severe dysplasia cases | 0.009 ± 0.030 | NS | 0.003 ± 0.002 | < 0.050 | < 0.001 | |
| Cancer patients | 0.009 ± 0.014 (distant) | < 0.05 | 0.003 ± 0.005 | < 0.001 | < 0.001 | |
| 0.007 ± 0.009 (adjacent) | < 0.05 | < 0.010 | ||||
| ABCC2 | [14] | |||||
| Healthy individuals | 5.35 ± 3.24 | |||||
| Mild moderate dysplasia cases | 4.62 ± 4.79 | 0.081 | 6.68 ± 6.77 | 0.87 | 0.037 | |
| Severe dysplasia cases | 6.66 ± 8.47 | 0.880 | 10.18 ± 11.52 | 0.27 | 0.240 | |
| Cancer patients | 28.06 ± 68.84 (distant) | 0.036 | 87.50 ± 270.21 | 0.0046 | 0.0037 | |
| 11.44 ± 25.58 (adjacent) | 0.690 | < 0.0001 | ||||
| ABCG2 | [14] | |||||
| Healthy individuals | 718.06 ± 761.24 | |||||
| Mild moderate dysplasia | 732.85 ± 2305.28 | 0.550 | 56.02 ± 118.42 | < 0.0001 | < 0.0001 | |
| Severe dysplasia | 448.02 ± 195.34 | 0.840 | 76.31 ± 102.63 | < 0.0001 | < 0.0001 | |
| Cancer patients | 6679 ± 58353 (distant) | 0.080 | 98.41 ± 476.36 | < 0.0001 | < 0.0001 | |
| 1302 ± 10090 (adjacent) | 0.011 | < 0.0001 |
Genetically determined variation in ABC transporters has been investigated in relation to risk of developing IBD[75-79] and CRC[80-82] with varying results[83-85]. In particular the polymorphisms ABCB1 C1236T, G2677T/A, and C3435T have been investigated. These polymorphisms are in linkage disequilibrium. Haplotype frequencies vary among ethnic groups and the CGC and TTT haplotypes are frequent among Caucasians[86]. The synonymous C3435T polymorphism was reported to cause changes in protein folding due to ribosome stalling caused by impaired interaction between the tRNA and the chaperone protein that aids the folding process at the ribosome[86] which resulted in altered transporter function[87]. A recent meta-analysis found that the ABCB1 C3435T polymorphism (rs1045642) was associated with risk of UC, but not with CD[84]. In relation to CRC, a large case-control analysis of a Czech and two German cohorts of 4677 cases in total found no indications of a strong role of ABCB1 in CRC[88] which was in accordance with a meta-analysis (not including the above study)[85]. A prospective study based on a Danish cohort found that two ABCB1 polymorphisms, including the C3435T polymorphism, were associated with CRC risk[82]. Furthermore, these two polymorphisms were found to interact with meat intake in relation to risk of CRC. Only few studies of ABCC2 and ABCG2 polymorphisms as risk factors for IBD and CRC have been performed. No strong indications that genetic variation in ABCC2 or ABCG2 per see is associated with IBD or CRC were found[80,81].
The Abcb1/Mdr1a knock-out (Mdr1a KO) mouse, in which the gene corresponding to the human intestinal ABCB1 gene has been deleted[89,90], has been utilized as an animal model of colitis[91-95]. The colitis is characterized by histological changes and high levels of the cytokines INF-γ, TNF-α, IL-1β, IL-6 and IL-17 thus resembling the findings in UC patients. The classical study by Panwala et al[91] reported that a proportion of Mdr1a KO mice developed colitis when exposed to commensal gut bacteria. The development of spontaneous colitis was prevented if the mice were maintained germfree. Also, spontaneous colitis and active inflammation was resolved by oral treatment with a mixture of streptomycin, neomycin, bacitracin, and amphotericin. These findings highlight an important role of bacteria in the initiation and perpetuation of colitis in the Mdr1a KO mouse[91]. Since then, the finding that lack of Mdr1a confers risk of colitis has been replicated by others[94-98]. Furthermore, a proportion of the Mdr1a KO mice dual-infected with Helicobacter species (H.bilis and H. hepaticus) developed dysplasia[99].
One study found redused in the diversity and total number of bacteria in mdr1a KO mice compared to wildtype mice. These alterations were found to precede and associate with the development of inflammation[95]. Another study reported changes in colonic gene expression which also preceded disease development[98]. High expression of INF-γ was found in histologically normal colonic tissue from Mdr1a KO mice and the change preceded a high expression of the inflammatory cytokines IL-1β, IL-6, TNF-α, increased colonic permeability, and histologically determined colon inflammation[98]. Yet, another study found a high level of the pro-inflammatory cytokine IL-17 in colon from the Mdr1a KO mice model[92]. INF-γ expression has been associated with reduced intestinal barrier function due to effects on tight junction proteins[96]. Also, one study suggested that impaired intestinal barrier function contributed to the development of colitis in Mdr1a KO mice. In this study, high permeability of FITC-dextran (4.4 kDa) and horseradish peroxidase (44 kDa) was found in colon tissue mounted in Ussing chambers and in vivo, high bacterial translocation to lymphoid tissue including increased trabecular infiltrate with neutrophils were found[94]. These changes were observed prior to onset of colitis. Furthermore, decreased phosphorylation of tight junction proteins including occludin was observed[94]. Thus, inflammation and the following high INF-γ expression may contribute to the loss of barrier function which has been observed in the Abcb1 KO mice.
High fat diet-induced obesity increases the frequency and severity of colitis in the mdr1a KO mice[100]. Wildtype mice feeding either high-fat diet or low fat diet did not develop colitis[100]. In contrast, specific pathogen free Mdr1a KO mice fed high fat diet had a higher frequency and more severe colitis compared to those who were fed a low fat diet[100]. Although the microbiota was not investigated in this study, the authors concluded that the diet and potential diet-induced changes in microbiota was not sufficient to induce colitis in the mice but that additional host genetic factors are required before the high fat diet is a risk factor for colitis[100].
Impaired immune system may also be involved in the aetiology of colitis in the Mdr1a KO mice model. In mice, regulatory T cells (Tregs) characterised by the expression of the transcription factor Foxp3[101] are considered to down-regulate effector T cells that react to microbial or other gastrointestinal antigens. In the study by Tanner et al[97], they also found that there appeared to be fewer Tregs present in intestine from mdr1a KO mice and that these Tregs were unable to effectively suppress TNF-α induced colitis. These results are in accordance with the notion that inflammation primarily is initiated by the innate immune system.
In contradiction to the findings in the Mdr1a KO mice model, Abcc2/Mrp2 KO and Abcg2/Bcrp1 KO mice were found to be phenotypically normal under standard housing conditions[102,103].
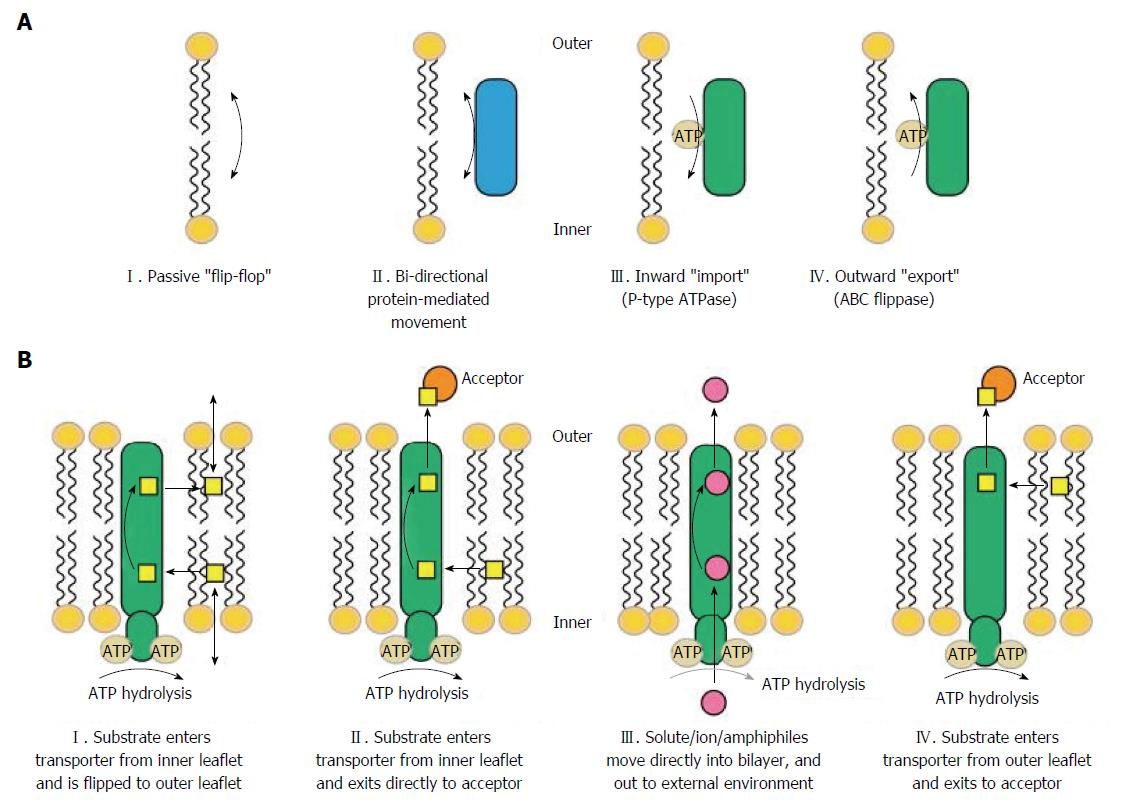
Cellular processes such as phagocytosis, apoptosis, cytokine release, vesicle formation and tight junction function require cell membrane budding and curvature and therefore, different composition of the inner and outer side of the lipid bilayer forming the cell membrane (Figure 1)[104]. Translocation of phospholipids between the two sides of the lipid bilayer within the cell membrane is therefore important for generating such differences. ABCB1, ABCC2, and ABCG2 have been found to translocate various phospholipid membrane components; cholesterol, sphingomyelin, and other glycosphingolipids suggesting that ABC transporters are important for regulating the budding of the membrane function[15,16,105,106]. Furthermore, the cellular processes also require cell cytoskeleton anchoring through specialised domains[107]. ABCB1 has been found to be associated with such domains[106,108,109]. Other phospholipid transporters such as scramblases, P4-ATPases and additional members of the ABC transporter family, are reviewed in[15].
In vitro studies of rat kidney and Sertoli cells support the involvement of ABC transporters in tight junction function and apoptosis[110,111]. At the Sertoli cell blood-testis barrier, ABCB1 was found to co-localise with occluding, claudin-11 and junction adhesion molecule A[110]. Knockdown of Abcb1 (Abcb1a and Abcb1b) by RNAi in rat Sertoli cell cultures led to a decline of claudin-11, internalisation and degradation of occluding, and disruption of tight junction barrier function[110]. Another study found that ABCB1 decreased apoptosis by decreasing the availability of a precursor of ceramide[111], an intracellular signalling molecule involved in apoptosis induced by TNF-α and other apoptotic stimuli[106,108]. However, the functions of the ABC transporters may be tissue specific and therefore the results may not apply for intestinal conditions.
Figure 1 shows mechanisms of substrate recognition and transport by ABC transporters[16]. An in vitro study by Pawlik et al[112] on cultured peripheral blood mononuclear cells PBMC from healthy individuals found that stimulation with phytohaemagglutinin (PHA) leads to secretion of IL-2, IL-4, IL-6, IL-10, INF-γ, and TNF-α[112]. Furthermore, secretion of IL-2, IL-4, INF-γ, and TNF-α was inhibited by anti-MDR1 specific antibody whereas secretion of IL-6 and IL-10 was unaffected. In a similar study, blockade of ABCC1 by anti-MRP1 specific antibodies led to reversible abrogated cytokine secretion of IL-10, TNF-α, IL-4 and INF-γ[113]. However, another study using splenocytes from Mdr1a KO mice found that IL-2, IL-4, IL-10, and INF-γ secretion was independent of ABCB1. The authors suggested that ABCB1 may not be required for secretion of these cytokines because they contain a signal sequence designating the cytokines for secretion from the cells[114]. Yet, a further in vitro study by Pawlik et al[115] on cultured PBMC, this time from 72 healthy ABCB1 genotyped individuals was conducted. The cultured cells were stimulated with PHA and cytokines were measured in the supernatant. The authors found significantly lower concentration of IL-2, IL-4, INF-γ, and TNF-α, and unchanged concentration of IL-6 and IL-10 in cultured cells from individuals with ABCB1 C3435T TT genotypes compared to CC genotypes[115]. Also, ABCB1 blockade by the antagonist PSC833 resulted in impaired IL-12 secretion by antigen presenting cells from peripheral blood from healthy human volunteers suggesting that functional ABCB1 is required for IL-12 secretion in these cells[116]. As previously mentioned, cytokines and chemokines are important modulators of intestinal inflammation and carcinogenesis[108,117]. Additionally, ABCB1, ABCC2, and ABCG2 also transport bioactive lipids[15,16,105]. The levels of the ABCB1 substrate platelet-activating factor[117-119] have been found to be high in intestinal mucosa from CD patients[120]. PAF has been reported to regulate the function of tight junctions[121] and to activate human neutrophils to extrusion of neutrophil extracellular traps (NETs) mediating extracellular capture and killing of bacteria[122,123]. Also, ABCB1 has been reported to transport steroids, mineralocorticoids, androgens and oestrogens[106]. Interestingly, the ABC substrate testosterone was found to be a key mediator of autoimmune responses in the non-obese diabetic mouse model of type 1 diabetes[124]. Whether a similar phenomenon contributes to the observed male preponderance in Mdr1a KO IBD mouse model has not been studied as far as we know[94]. ABCG2 transport the anti-inflammatory butyrate, a product of bacterial digestion of dietary fibres, and phytoestrogen from vegetables[125,126]. ABCC2 has been reported to transport the pro-inflammatory signalling molecules leukotriene (LT) B4 and LTC4 involved in dendritic cell migration and CRC, and, furthermore, various diet- and smoke-derived carcinogens[127-131]. Sulfasalazine and 5-aminosalicylic acid (5-ASA, mesalazine) are used for treatment and prevention of UC flares[132]. ABCG2 is regarded as being the main transporter of sulfasalazine[133,134] and ABCG2 activity has been suggested as having impact on sulfasalazine treatment efficacy in patients with rheumatoid arthritis (RA)[135,136].
One study utilised ABCB1 expression to identify human Th17 cells with a unique pro-inflammatory transcriptional signature[20]. This novel subset of Th17 cells, MDR1-positive Th17 cells, was identified by fluorescence activated cell sorting (FACS) analysis of PBMC from healthy individuals. Compared to MDR1-negative Th17 cells, the MDR1-positive Th17 cells were characterized by a high production of pro-inflammatory Th1 (INF-γ) and Th17 (IL-17A, IL-17F, and IL-22) cytokines and low levels of anti-inflammatory cytokines such as IL-10 upon stimulation[20]. In contrast to the MDR1-negative T cells, the MDR1-positive T cells were resistant to treatment with glucocorticoids. Thus, MDR1-positive T cells from healthy humans were enriched two- to three-fold during culturing of peripheral blood memory T cells in the presence of glucocorticoids[20]. Furthermore, in a small study of 3-5 CD patients, MDR1-positive Th17 cells (assessed as percent of the total number of memory cells) were enriched both in non-inflamed and inflamed gut tissue compared to blood levels[20]. High mRNA levels of IFN-γ, IL23R, and TNF were found in MDR1-positive Th17 cells compared to MDR1-negative Th17 cells following FACS-sorting of mononuclear cells from gut tissue from two CD patients[20].
The ABC transport proteins may confer a link between the environment and intestinal inflammation and potentially intestinal carcinogenesis via intestinal inflammation[48-50,137,138] . Diet affects risk of CRC[1], the course[139-143] and risk of IBD [144-148] (reviewed in[149-153]). Diet affects gut microbial composition[154,155] and both diet and intestinal microbes affect intestinal inflammation[156,157] and carcinogenesis[12,158-161].
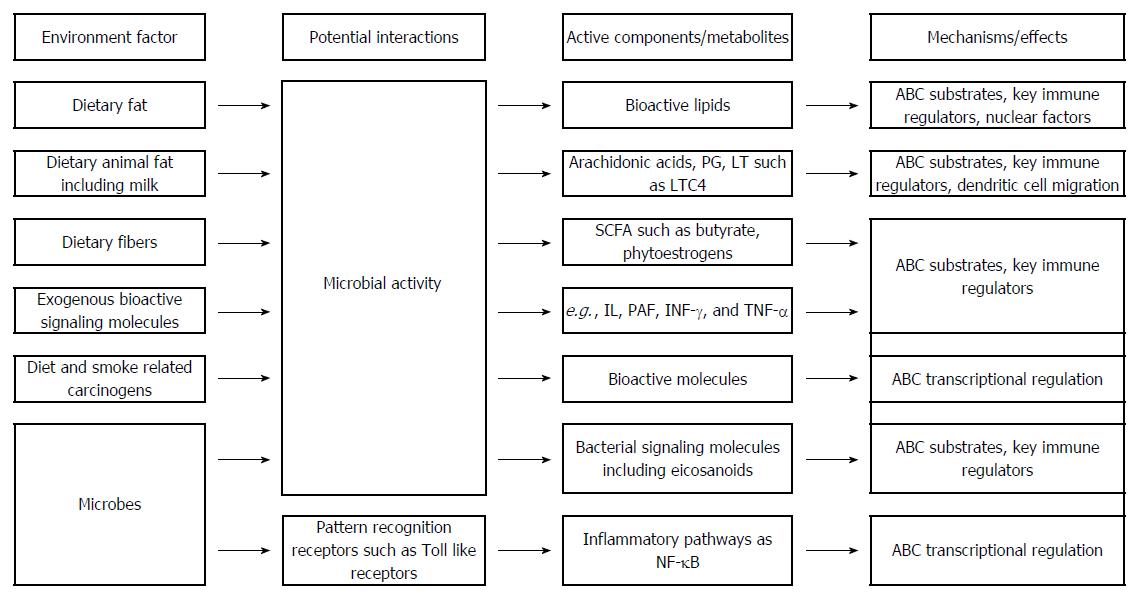
A link between ABCB1, diet and the gut microbes in relation to colitis is suggested by the animal studies. High fat diet increases the frequency and severity of colitis in specific pathogen-free Abcb1 KO mice[100]. Undigested dietary items reaching the colon are digested by commensal bacteria thereby providing the host with valuable energy, essential vitamins, fatty acids etc. Dietary fibre from grains, fruit and vegetables is converted into short-chain fatty acids (SCFA) which represent important key regulators of the immune system[12]. The gut microbiome in active IBD is characterised by decreased microbial diversity with a decreased number of Firmicutes[162]. Low abundance of the Clostridium and Bacteroides species which preferentially produce butyrate and other SCFA may result in low production of SCFA[163]. High intake of meat which is a rich source of sulphur may lead to the formation of hydrogen sulphide by bacterial fermentation[12] which, at least theoretically, may be aggravated by high intake of milk fat which was found to favour the presence of the sulphate-reducing bacteria Bilophila wadsworthia in mice[157]. Also, intake of animal fat may give rise to arachidonic acid which is converted into e.g., prostaglandins and leukotrienes[12]. Some of these molecules are ABC transporter substrates including dietary pro- and anti-inflammatory molecules, bioactive lipids, and bacterial derived molecules[125,126]. Figure 2 shows potential mechanisms of the involvement of ABC transporters in inflammation. In addition, diet and other environmental factors may impact the transcriptional regulation of ABC transporters through effects on nuclear receptors and transcription factors leading to changes of the ABC transporter activity thereby affecting IBD and CRC. The ABC transporters may impact IBD and CRC through their transport of various substrates thereby affecting underlying biological mechanisms involved in intestinal inflammation (Figures 1 and 2).
ABC transporter polymorphisms have been evaluated in relation to development of IBD and CRC with inconsistent results. These studies are based on the hypothesis that genetic variations are associated with functional changes in ABC activity and/or specificity. It has been suggested that genetic diversity of the ABCB1 gene among various ethnicities may contribute to the varying results in candidate gene studies[164,165]. In addition, ABCB1 polymorphisms may only be associated with risk of CRC in populations with a relevant dietary exposure[166]. This aspect may be exemplified by the finding of an interaction between meat intake and the gene NFKB1 encoding NFκB p50 in a Danish cohort[137]. This interaction may explain the finding that the NFKB1 polymorphism was associated with risk of CRC in a Swedish cohort but not in a Chinese cohort[167]. Meat intake are higher in Denmark and Sweden compared to China[168]. Therefore, NFKB1 was identified as a risk gene in the Danish and Swedish high meat intake cohorts but not in the Chinese low meat intake cohort. A detailed assessment of the diet seems to be important for assessing the roles of ABC polymorphisms. Thus, future studies should focus on studying large cohorts with well-defined and relevant prospectively sampled environmental exposures in order to identify underlying IBD and CRC disease mechanisms.
Due to the many confounding parameters, potential causality cannot be evaluated through molecular epidemiological studies. Studies using animal models, where a range of parameters can be controlled are therefore needed for establishing causality. Germfree mice do not develop colitis. Although germfree mice are not exposed for living bacteria they will meet dietary derived microbial antigens which could activate PRR in the mucosa and induce inflammation. Inflammation, however, has not been observed in the germfree mice. Moreover, colitis can be prevented by antibiotics in conventionally housed, specific pathogen-free, mice. These findings suggest that microbial derived antigens are not sufficient to trigger colitis but that living microbes are needed and may thus point to potential mechanisms such as microbial derived metabolites, signalling peptides and extracellular vesicles[169,170]. Indeed, gut microbial derived metabolites were found to affect the balance between pro- and anti-inflammatory cells in mice[171]. These metabolites may be absorbed into the blood and thereby affect distant organs. Gut microbes have been reported to affect the immune system, in particular the Th17 pathway, in various autoimmune mouse models[172-176]. Some studies, but not all[177], indicate a similar mechanism in humans which might also associate with human autoimmunity[178-180]. Also, bacterially derived fatty acids and other relevant metabolites should be investigated in the Abcb1 KO mice like it has been done in male C57BL/6 (B6) mice[171]. The Abcb1 KO mice might provide a model, in which the interplay of environment factors, diet, and microbes can be controlled and investigated. Due to important differences of human and murine immune systems, the translational value of results obtained from the mouse model need also to be evaluated through human data.
The finding that presence of ABCB1 on immune cells could be used to identify pro-inflammatory Th17 cells may have important clinical implications as glucocorticoids are a mainstay in the treatment of serious flares of IBD[181] and since a large proportion (20%-30%) of patients are resistant to glucocorticoid treatment[182]. Thus, high ABCB1 mediated drug efflux may lead to decreased intracellular drug concentrations in target cells [183,184] and thereby confer glucocorticoid treatment resistance. Likewise, ABCG2 activity may affect efficacy of treatment with sulfasalazine. Further evaluation of the roles of ABC transporters in treatment response in IBD is warranted.
In conclusion, results from animal and human studies indicate that ABCB1, diet, and gut microbes mutually interact in colonic inflammation. Diet and microbes may give rise to molecules which are substrates for the ABC transporters and may additionally affect ABC transporter function through e.g., nuclear receptors and transcriptional regulation. The Abcb1 KO mice might provide a model in which these factors can be controlled and investigated. Such strategy may provide insight which can be translated into preventive and treatment strategies to benefit the patients. The evidence for the involvement of ABCC2 and ABCG2 in colitis was weak.
Staff at the Libraries, Regional Hospital Viborg and Hospital of Southern Denmark, are thanked for help.
Colorectal cancer (CRC) constitutes the third most common cancer in the world and the second leading cause of cancer-related deaths. The number of cases is increasing and has been estimated to raise from 1.4 million cases in 2012 to 2.4 million cases in 2035 worldwide. Early detection of CRC is important as early treatment has been associated with improved outcomes and saved lives. Therefore, population screening programs have been initiated in a number of countries such as the United Kingdom, Australia, Holland and Denmark. The fecal occult blood test (FOBT) is the most widely used for population screening and individuals with a positive FOBT are referred for an endoscopic investigation of the colonic mucosa thereby enabling the sampling of biopsies from the colonic mucosa.
Recently, a major part of research had focused on improving prognosis and treatment selection in CRC. Another approach could be to prevent the development of cancer in subgroups of patients with high risk, i.e., secondary prevention. Thus, the molecular evaluation of the (unaffected) colonic mucosa from the patients undergoing an endoscopic evaluation could potentially stratify the patients according to their risk of developing CRC. Recently, human studies by authors reported that changes in the levels of ABC transporters were early events in the adenoma-carcinoma sequence leading to CRC. These findings indicate that even healthy looking mucosa as determined by histology may contain a significantly elevated level of immune response proteins.
The authors recently reported that low ABCB1 and ABCG2 gene transcription levels and high ABCC2 levels are early events in the colorectal adenoma-carcinoma sequence suggesting that changes in expression levels of the ATP binding cassette (ABC) transporter proteins [EC 3.6.3.44] precede cancer development. In addition, inflammatory bowel disease (IBD) may be a risk factor for the development of CRC. Therefore, the authors wanted to discuss the current understanding of how these ABC transporters may affect intestinal inflammation and carcinogenesis, how they may potentially interact with the environment such as diet and gut microbes, and whether this knowledge may be utilized for improved treatment care strategies. A link between ABCB1, high fat diet and gut microbes in relation to colitis was suggested by the animal studies. The Abcb1 KO mice might thus serve as a model in which diet/environmental factors and microbes may be controlled and investigated in relation to intestinal inflammation. Such strategy may provide insight which can be translated into preventive and treatment strategies to benefit the patients.
Biomarkers potentially predicting the disease risk among selected patient groups could improve the efficiency of the screening programs and patient care. Furthermore, they have the potential to dramatically alter the established patient care pathways as follow-up of the patients may be tailored according to their individual risk and thereby the organization and use of resources of the health care system.
Congratulations to the authors for their review on ABC transporters ABCB1/MDR/P-glycoprotein, ABCC2/MRP2, and ABCG2/BCRP in colorectal pathophysiology. It is certain that this paper will be very inspiring in this field. Personally recommend it to be accepted.
P- Reviewer: Aytac E, Ciccone MM S- Editor: Yu J L- Editor: A E- Editor: Ma S
| 1. | WCRF. World Cancer Research Fund International. Available from: http://www.wcrf.org/ 2014. |
| 2. | UEG. Colorectal cancer in Europe. Available from: http://www.ueg.eu/press/crceurope/ 2014. |
| 3. | CRC screening UK. Available from: http://www.cancerscreening.nhs.uk/bowel/.. |
| 4. | CRC screening Australia. Available from: http://www.bowelcanceraustralia.org/. |
| 5. | CRC screening the Nederlands. Available from: http://www.rivm.nl/Onderwerpen/B/Bevolkingsonderzoek_darmkanker.. |
| 6. | CRC screening DK. Available from: http://www.sundhed.dk/borger/sundhedsjournal-og-registreringer/tilmeldinger/screeningsprogrammer/tarmkraeftscreening/. |
| 7. | Huddy JR, Ni MZ, Markar SR, Hanna GB. Point-of-care testing in the diagnosis of gastrointestinal cancers: current technology and future directions. World J Gastroenterol. 2015;21:4111-4120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 18] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 8. | Duffy MJ, Lamerz R, Haglund C, Nicolini A, Kalousová M, Holubec L, Sturgeon C. Tumor markers in colorectal cancer, gastric cancer and gastrointestinal stromal cancers: European group on tumor markers 2014 guidelines update. Int J Cancer. 2014;134:2513-2522. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 253] [Cited by in RCA: 248] [Article Influence: 22.5] [Reference Citation Analysis (1)] |
| 9. | Koch C, Trojan J. Established and Potential Predictive Biomarkers in Gastrointestinal Cancer--c-Kit, Her2, Ras and Beyond. Digestion. 2015;91:294-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Luo HY, Xu RH. Predictive and prognostic biomarkers with therapeutic targets in advanced colorectal cancer. World J Gastroenterol. 2014;20:3858-3874. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 35] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 11. | Bennike TB, Carlsen TG, Ellingsen T, Bonderup OK, Glerup H, Bøgsted M, Christiansen G, Birkelund S, Stensballe A, Andersen V. Neutrophil Extracellular Traps in Ulcerative Colitis: A Proteome Analysis of Intestinal Biopsies. Inflamm Bowel Dis. 2015;21:2052-2067. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 139] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 12. | Andersen V, Vogel U. Dietary fibres and meat in relation to Colorectal Cancer. Norske Gastroenterologisk Forening-nytt. 2014;34-36. |
| 13. | Andersen V, Vogel U, Godiksen S, Frenzel FB, Sæbø M, Hamfjord J, Kure E, Vogel LK. Low ABCB1 gene expression is an early event in colorectal carcinogenesis. PLoS One. 2013;8:e72119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 14. | Andersen V, Vogel LK, Kopp TI, Sæbø M, Nonboe AW, Hamfjord J, Kure EH, Vogel U. High ABCC2 and low ABCG2 gene expression are early events in the colorectal adenoma-carcinoma sequence. PLoS One. 2015;10:e0119255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 15. | Coleman JA, Quazi F, Molday RS. Mammalian P4-ATPases and ABC transporters and their role in phospholipid transport. Biochim Biophys Acta. 2013;1831:555-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 115] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 16. | Tarling EJ, de Aguiar Vallim TQ, Edwards PA. Role of ABC transporters in lipid transport and human disease. Trends Endocrinol Metab. 2013;24:342-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 221] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 17. | Mack JT, Beljanski V, Tew KD, Townsend DM. The ATP-binding cassette transporter ABCA2 as a mediator of intracellular trafficking. Biomed Pharmacother. 2006;60:587-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 56] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 18. | Wessler JD, Grip LT, Mendell J, Giugliano RP. The P-glycoprotein transport system and cardiovascular drugs. J Am Coll Cardiol. 2013;61:2495-2502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 254] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 19. | Staud F, Cerveny L, Ceckova M. Pharmacotherapy in pregnancy; effect of ABC and SLC transporters on drug transport across the placenta and fetal drug exposure. J Drug Target. 2012;20:736-763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 90] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 20. | Ramesh R, Kozhaya L, McKevitt K, Djuretic IM, Carlson TJ, Quintero MA, McCauley JL, Abreu MT, Unutmaz D, Sundrud MS. Pro-inflammatory human Th17 cells selectively express P-glycoprotein and are refractory to glucocorticoids. J Exp Med. 2014;211:89-104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 314] [Cited by in RCA: 372] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 21. | Fardel O, Lecureur V, Guillouzo A. The P-glycoprotein multidrug transporter. Gen Pharmacol. 1996;27:1283-1291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 387] [Cited by in RCA: 385] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 22. | Ambudkar SV, Dey S, Hrycyna CA, Ramachandra M, Pastan I, Gottesman MM. Biochemical, cellular, and pharmacological aspects of the multidrug transporter. Annu Rev Pharmacol Toxicol. 1999;39:361-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1547] [Cited by in RCA: 1519] [Article Influence: 58.4] [Reference Citation Analysis (0)] |
| 23. | Leslie EM, Deeley RG, Cole SP. Multidrug resistance proteins: role of P-glycoprotein, MRP1, MRP2, and BCRP (ABCG2) in tissue defense. Toxicol Appl Pharmacol. 2005;204:216-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 996] [Cited by in RCA: 1034] [Article Influence: 51.7] [Reference Citation Analysis (0)] |
| 24. | Abu-Qare AW, Elmasry E, Abou-Donia MB. A role for P-glycoprotein in environmental toxicology. J Toxicol Environ Health B Crit Rev. 2003;6:279-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 60] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 25. | Schinkel AH, Jonker JW. Mammalian drug efflux transporters of the ATP binding cassette (ABC) family: an overview. Adv Drug Deliv Rev. 2003;55:3-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1057] [Cited by in RCA: 1062] [Article Influence: 48.3] [Reference Citation Analysis (0)] |
| 26. | Sarkadi B, Homolya L, Szakács G, Váradi A. Human multidrug resistance ABCB and ABCG transporters: participation in a chemoimmunity defense system. Physiol Rev. 2006;86:1179-1236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 522] [Cited by in RCA: 558] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 27. | Chaudhary PM, Roninson IB. Expression and activity of P-glycoprotein, a multidrug efflux pump, in human hematopoietic stem cells. Cell. 1991;66:85-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 686] [Cited by in RCA: 669] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 28. | Ho GT, Moodie FM, Satsangi J. Multidrug resistance 1 gene (P-glycoprotein 170): an important determinant in gastrointestinal disease? Gut. 2003;52:759-766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 150] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 29. | Haimeur A, Conseil G, Deeley RG, Cole SP. The MRP-related and BCRP/ABCG2 multidrug resistance proteins: biology, substrate specificity and regulation. Curr Drug Metab. 2004;5:21-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 389] [Cited by in RCA: 364] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 30. | Stefková J, Poledne R, Hubácek JA. ATP-binding cassette (ABC) transporters in human metabolism and diseases. Physiol Res. 2004;53:235-243. [PubMed] |
| 31. | Speca S, Giusti I, Rieder F, Latella G. Cellular and molecular mechanisms of intestinal fibrosis. World J Gastroenterol. 2012;18:3635-3661. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 177] [Cited by in RCA: 203] [Article Influence: 15.6] [Reference Citation Analysis (2)] |
| 32. | Basseri RJ, Basseri B, Papadakis KA. Dysplasia and cancer in inflammatory bowel disease. Expert Rev Gastroenterol Hepatol. 2011;5:59-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 153] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 33. | Satsu H, Hiura Y, Mochizuki K, Hamada M, Shimizu M. Activation of pregnane X receptor and induction of MDR1 by dietary phytochemicals. J Agric Food Chem. 2008;56:5366-5373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 61] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 34. | Tolson AH, Wang H. Regulation of drug-metabolizing enzymes by xenobiotic receptors: PXR and CAR. Adv Drug Deliv Rev. 2010;62:1238-1249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 269] [Cited by in RCA: 288] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 35. | Wang K, Wan YJ. Nuclear receptors and inflammatory diseases. Exp Biol Med (Maywood). 2008;233:496-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 60] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 36. | Albermann N, Schmitz-Winnenthal FH, Z’graggen K, Volk C, Hoffmann MM, Haefeli WE, Weiss J. Expression of the drug transporters MDR1/ABCB1, MRP1/ABCC1, MRP2/ABCC2, BCRP/ABCG2, and PXR in peripheral blood mononuclear cells and their relationship with the expression in intestine and liver. Biochem Pharmacol. 2005;70:949-958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 220] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 37. | Tachibana S, Yoshinari K, Chikada T, Toriyabe T, Nagata K, Yamazoe Y. Involvement of Vitamin D receptor in the intestinal induction of human ABCB1. Drug Metab Dispos. 2009;37:1604-1610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 38. | Wang X, Hawkins BT, Miller DS. Aryl hydrocarbon receptor-mediated up-regulation of ATP-driven xenobiotic efflux transporters at the blood-brain barrier. FASEB J. 2011;25:644-652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 82] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 39. | Blokzijl H, Vander Borght S, Bok LI, Libbrecht L, Geuken M, van den Heuvel FA, Dijkstra G, Roskams TA, Moshage H, Jansen PL. Decreased P-glycoprotein (P-gp/MDR1) expression in inflamed human intestinal epithelium is independent of PXR protein levels. Inflamm Bowel Dis. 2007;13:710-720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 109] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 40. | Langmann T, Moehle C, Mauerer R, Scharl M, Liebisch G, Zahn A, Stremmel W, Schmitz G. Loss of detoxification in inflammatory bowel disease: dysregulation of pregnane X receptor target genes. Gastroenterology. 2004;127:26-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 259] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 41. | Miller DS. Regulation of P-glycoprotein and other ABC drug transporters at the blood-brain barrier. Trends Pharmacol Sci. 2010;31:246-254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 280] [Cited by in RCA: 254] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 42. | Chakraborty PK, Lee WK, Molitor M, Wolff NA, Thévenod F. Cadmium induces Wnt signaling to upregulate proliferation and survival genes in sub-confluent kidney proximal tubule cells. Mol Cancer. 2010;9:102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 43. | Chambers TC, Pohl J, Glass DB, Kuo JF. Phosphorylation by protein kinase C and cyclic AMP-dependent protein kinase of synthetic peptides derived from the linker region of human P-glycoprotein. Biochem J. 1994;299:309-315. [PubMed] |
| 44. | Xie Y, Burcu M, Linn DE, Qiu Y, Baer MR. Pim-1 kinase protects P-glycoprotein from degradation and enables its glycosylation and cell surface expression. Mol Pharmacol. 2010;78:310-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 78] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 45. | Begley GS, Horvath AR, Taylor JC, Higgins CF. Cytoplasmic domains of the transporter associated with antigen processing and P-glycoprotein interact with subunits of the proteasome. Mol Immunol. 2005;42:137-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 46. | Goel A, Boland CR. Recent insights into the pathogenesis of colorectal cancer. Curr Opin Gastroenterol. 2010;26:47-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 58] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 47. | Ogino S, Chan AT, Fuchs CS, Giovannucci E. Molecular pathological epidemiology of colorectal neoplasia: an emerging transdisciplinary and interdisciplinary field. Gut. 2011;60:397-411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 430] [Cited by in RCA: 451] [Article Influence: 32.2] [Reference Citation Analysis (0)] |
| 48. | Collett A, Higgs NB, Gironella M, Zeef LA, Hayes A, Salmo E, Haboubi N, Iovanna JL, Carlson GL, Warhurst G. Early molecular and functional changes in colonic epithelium that precede increased gut permeability during colitis development in mdr1a(-/-) mice. Inflamm Bowel Dis. 2008;14:620-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 49. | McConnell BB, Yang VW. The Role of Inflammation in the Pathogenesis of Colorectal Cancer. Curr Colorectal Cancer Rep. 2009;5:69-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 50. | Rhodes JM, Campbell BJ. Inflammation and colorectal cancer: IBD-associated and sporadic cancer compared. Trends Mol Med. 2002;8:10-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 241] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 51. | Baumgart DC, Carding SR. Inflammatory bowel disease: cause and immunobiology. Lancet. 2007;369:1627-1640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1299] [Cited by in RCA: 1506] [Article Influence: 83.7] [Reference Citation Analysis (2)] |
| 52. | Strober W, Fuss I, Mannon P. The fundamental basis of inflammatory bowel disease. J Clin Invest. 2007;117:514-521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 992] [Cited by in RCA: 1001] [Article Influence: 55.6] [Reference Citation Analysis (0)] |
| 53. | Jess T, Horváth-Puhó E, Fallingborg J, Rasmussen HH, Jacobsen BA. Cancer risk in inflammatory bowel disease according to patient phenotype and treatment: a Danish population-based cohort study. Am J Gastroenterol. 2013;108:1869-1876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 188] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 54. | Van Assche G, Dignass A, Bokemeyer B, Danese S, Gionchetti P, Moser G, Beaugerie L, Gomollón F, Häuser W, Herrlinger K. Second European evidence-based consensus on the diagnosis and management of ulcerative colitis part 3: special situations. J Crohns Colitis. 2013;7:1-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 602] [Cited by in RCA: 640] [Article Influence: 49.2] [Reference Citation Analysis (0)] |
| 55. | Molodecky NA, Soon IS, Rabi DM, Ghali WA, Ferris M, Chernoff G, Benchimol EI, Panaccione R, Ghosh S, Barkema HW. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012;142:46-54.e42; quiz e30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3789] [Cited by in RCA: 3522] [Article Influence: 270.9] [Reference Citation Analysis (5)] |
| 56. | Denson LA. The role of the innate and adaptive immune system in pediatric inflammatory bowel disease. Inflamm Bowel Dis. 2013;19:2011-2020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 283] [Cited by in RCA: 340] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 57. | Marchiando AM, Shen L, Graham WV, Edelblum KL, Duckworth CA, Guan Y, Montrose MH, Turner JR, Watson AJ. The epithelial barrier is maintained by in vivo tight junction expansion during pathologic intestinal epithelial shedding. Gastroenterology. 2011;140:1208-1218.e1-2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 230] [Cited by in RCA: 218] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 58. | Maloy KJ, Powrie F. Intestinal homeostasis and its breakdown in inflammatory bowel disease. Nature. 2011;474:298-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1218] [Cited by in RCA: 1443] [Article Influence: 103.1] [Reference Citation Analysis (0)] |
| 59. | Franchi L, Muñoz-Planillo R, Núñez G. Sensing and reacting to microbes through the inflammasomes. Nat Immunol. 2012;13:325-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 735] [Cited by in RCA: 834] [Article Influence: 64.2] [Reference Citation Analysis (0)] |
| 60. | Chen GY, Núñez G. Inflammasomes in intestinal inflammation and cancer. Gastroenterology. 2011;141:1986-1999. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 126] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 61. | Delgado M, Singh S, De Haro S, Master S, Ponpuak M, Dinkins C, Ornatowski W, Vergne I, Deretic V. Autophagy and pattern recognition receptors in innate immunity. Immunol Rev. 2009;227:189-202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 139] [Cited by in RCA: 135] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 62. | Delgado ME, Dyck L, Laussmann MA, Rehm M. Modulation of apoptosis sensitivity through the interplay with autophagic and proteasomal degradation pathways. Cell Death Dis. 2014;5:e1011. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 63. | Turner JR. Intestinal mucosal barrier function in health and disease. Nat Rev Immunol. 2009;9:799-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 274] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 64. | MacDonald TT, Monteleone I, Fantini MC, Monteleone G. Regulation of homeostasis and inflammation in the intestine. Gastroenterology. 2011;140:1768-1775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 206] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 65. | Wedebye Schmidt EG, Larsen HL, Kristensen NN, Poulsen SS, Lynge Pedersen AM, Claesson MH, Pedersen AE. TH17 cell induction and effects of IL-17A and IL-17F blockade in experimental colitis. Inflamm Bowel Dis. 2013;19:1567-1576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 75] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 66. | Rovedatti L, Kudo T, Biancheri P, Sarra M, Knowles CH, Rampton DS, Corazza GR, Monteleone G, Di Sabatino A, Macdonald TT. Differential regulation of interleukin 17 and interferon gamma production in inflammatory bowel disease. Gut. 2009;58:1629-1636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 283] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 67. | Xie Z, Qu Y, Leng Y, Sun W, Ma S, Wei J, Hu J, Zhang X. Human colon carcinogenesis is associated with increased interleukin-17-driven inflammatory responses. Drug Des Devel Ther. 2015;9:1679-1689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 68. | Busman-Sahay KO, Walrath T, Huber S, O’Connor W. Cytokine crowdsourcing: multicellular production of TH17-associated cytokines. J Leukoc Biol. 2015;97:499-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 69. | Ueno A, Jijon H, Chan R, Ford K, Hirota C, Kaplan GG, Beck PL, Iacucci M, Fort Gasia M, Barkema HW. Increased prevalence of circulating novel IL-17 secreting Foxp3 expressing CD4+ T cells and defective suppressive function of circulating Foxp3+ regulatory cells support plasticity between Th17 and regulatory T cells in inflammatory bowel disease patients. Inflamm Bowel Dis. 2013;19:2522-2534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 172] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 70. | ten Hove T, Drillenburg P, Wijnholds J, Te Velde AA, van Deventer SJ. Differential susceptibility of multidrug resistance protein-1 deficient mice to DSS and TNBS-induced colitis. Dig Dis Sci. 2002;47:2056-2063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 72] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 71. | Sarrabayrouse G, Bossard C, Chauvin JM, Jarry A, Meurette G, Quévrain E, Bridonneau C, Preisser L, Asehnoune K, Labarrière N. CD4CD8αα lymphocytes, a novel human regulatory T cell subset induced by colonic bacteria and deficient in patients with inflammatory bowel disease. PLoS Biol. 2014;12:e1001833. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 107] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 72. | Englund G, Jacobson A, Rorsman F, Artursson P, Kindmark A, Rönnblom A. Efflux transporters in ulcerative colitis: decreased expression of BCRP (ABCG2) and Pgp (ABCB1). Inflamm Bowel Dis. 2007;13:291-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 110] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 73. | Deuring JJ, de Haar C, Koelewijn CL, Kuipers EJ, Peppelenbosch MP, van der Woude CJ. Absence of ABCG2-mediated mucosal detoxification in patients with active inflammatory bowel disease is due to impeded protein folding. Biochem J. 2012;441:87-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 74. | Deuring JJ, Peppelenbosch MP, Kuipers EJ, van der Woude CJ, de Haar C. Impeded protein folding and function in active inflammatory bowel disease. Biochem Soc Trans. 2011;39:1107-1111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 75. | Østergaard M, Ernst A, Labouriau R, Dagiliené E, Krarup HB, Christensen M, Thorsgaard N, Jacobsen BA, Tage-Jensen U, Overvad K. Cyclooxygenase-2, multidrug resistance 1, and breast cancer resistance protein gene polymorphisms and inflammatory bowel disease in the Danish population. Scand J Gastroenterol. 2009;44:65-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 76. | Ho GT, Soranzo N, Nimmo ER, Tenesa A, Goldstein DB, Satsangi J. ABCB1/MDR1 gene determines susceptibility and phenotype in ulcerative colitis: discrimination of critical variants using a gene-wide haplotype tagging approach. Hum Mol Genet. 2006;15:797-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 93] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 77. | Ho GT, Nimmo ER, Tenesa A, Fennell J, Drummond H, Mowat C, Arnott ID, Satsangi J. Allelic variations of the multidrug resistance gene determine susceptibility and disease behavior in ulcerative colitis. Gastroenterology. 2005;128:288-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 132] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 78. | Brant SR, Panhuysen CI, Nicolae D, Reddy DM, Bonen DK, Karaliukas R, Zhang L, Swanson E, Datta LW, Moran T. MDR1 Ala893 polymorphism is associated with inflammatory bowel disease. Am J Hum Genet. 2003;73:1282-1292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 172] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 79. | Schwab M, Schaeffeler E, Marx C, Fromm MF, Kaskas B, Metzler J, Stange E, Herfarth H, Schoelmerich J, Gregor M. Association between the C3435T MDR1 gene polymorphism and susceptibility for ulcerative colitis. Gastroenterology. 2003;124:26-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 219] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 80. | Andersen V, Egeberg R, Tjønneland A, Vogel U. ABCC2 transporter gene polymorphisms, diet and risk of colorectal cancer: a Danish prospective cohort study. Scand J Gastroenterol. 2012;47:572-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 81. | Campa D, Pardini B, Naccarati A, Vodickova L, Novotny J, Försti A, Hemminki K, Barale R, Vodicka P, Canzian F. A gene-wide investigation on polymorphisms in the ABCG2/BRCP transporter and susceptibility to colorectal cancer. Mutat Res. 2008;645:56-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 82. | Andersen V, Ostergaard M, Christensen J, Overvad K, Tjønneland A, Vogel U. Polymorphisms in the xenobiotic transporter Multidrug Resistance 1 (MDR1) and interaction with meat intake in relation to risk of colorectal cancer in a Danish prospective case-cohort study. BMC Cancer. 2009;9:407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 80] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 83. | Annese V, Valvano MR, Palmieri O, Latiano A, Bossa F, Andriulli A. Multidrug resistance 1 gene in inflammatory bowel disease: a meta-analysis. World J Gastroenterol. 2006;12:3636-3644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 97] [Cited by in RCA: 96] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 84. | Wang LH, Song YB, Zheng WL, Jiang L, Ma WL. The association between polymorphisms in the MDR1 gene and risk of cancer: a systematic review and pooled analysis of 52 case-control studies. Cancer Cell Int. 2013;13:46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 85. | He T, Mo A, Zhang K, Liu L. ABCB1/MDR1 gene polymorphism and colorectal cancer risk: a meta-analysis of case-control studies. Colorectal Dis. 2013;15:12-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 86. | Fung KL, Gottesman MM. A synonymous polymorphism in a common MDR1 (ABCB1) haplotype shapes protein function. Biochim Biophys Acta. 2009;1794:860-871. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 256] [Cited by in RCA: 261] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 87. | Fung KL, Pan J, Ohnuma S, Lund PE, Pixley JN, Kimchi-Sarfaty C, Ambudkar SV, Gottesman MM. MDR1 synonymous polymorphisms alter transporter specificity and protein stability in a stable epithelial monolayer. Cancer Res. 2014;74:598-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 98] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 88. | Campa D, Sainz J, Pardini B, Vodickova L, Naccarati A, Rudolph A, Novotny J, Försti A, Buch S, von Schönfels W. A comprehensive investigation on common polymorphisms in the MDR1/ABCB1 transporter gene and susceptibility to colorectal cancer. PLoS One. 2012;7:e32784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 89. | Borst P, Schinkel AH. P-glycoprotein ABCB1: a major player in drug handling by mammals. J Clin Invest. 2013;123:4131-4133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 123] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 90. | Cui YJ, Cheng X, Weaver YM, Klaassen CD. Tissue distribution, gender-divergent expression, ontogeny, and chemical induction of multidrug resistance transporter genes (Mdr1a, Mdr1b, Mdr2) in mice. Drug Metab Dispos. 2009;37:203-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 92] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 91. | Panwala CM, Jones JC, Viney JL. A novel model of inflammatory bowel disease: mice deficient for the multiple drug resistance gene, mdr1a, spontaneously develop colitis. J Immunol. 1998;161:5733-5744. [PubMed] |
| 92. | Staley EM, Schoeb TR, Lorenz RG. Differential susceptibility of P-glycoprotein deficient mice to colitis induction by environmental insults. Inflamm Bowel Dis. 2009;15:684-696. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 93. | Staley EM, Yarbrough VR, Schoeb TR, Daft JG, Tanner SM, Steverson D, Lorenz RG. Murine P-glycoprotein deficiency alters intestinal injury repair and blunts lipopolysaccharide-induced radioprotection. Radiat Res. 2012;178:207-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 94. | Pastorelli L, De Salvo C, Mercado JR, Vecchi M, Pizarro TT. Central role of the gut epithelial barrier in the pathogenesis of chronic intestinal inflammation: lessons learned from animal models and human genetics. Front Immunol. 2013;4:280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 112] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 95. | Nones K, Knoch B, Dommels YE, Paturi G, Butts C, McNabb WC, Roy NC. Multidrug resistance gene deficient (mdr1a-/-) mice have an altered caecal microbiota that precedes the onset of intestinal inflammation. J Appl Microbiol. 2009;107:557-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 96. | Staley EM, Dimmitt RA, Schoeb TR, Tanner SM, Lorenz RG. Critical role for P-glycoprotein expression in hematopoietic cells in the FVB.Mdr1a(-/-) model of colitis. J Pediatr Gastroenterol Nutr. 2011;53:666-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 97. | Tanner SM, Staley EM, Lorenz RG. Altered generation of induced regulatory T cells in the FVB.mdr1a-/- mouse model of colitis. Mucosal Immunol. 2013;6:309-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 98. | Low D, Nguyen DD, Mizoguchi E. Animal models of ulcerative colitis and their application in drug research. Drug Des Devel Ther. 2013;7:1341-1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 40] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 99. | Maggio-Price L, Bielefeldt-Ohmann H, Treuting P, Iritani BM, Zeng W, Nicks A, Tsang M, Shows D, Morrissey P, Viney JL. Dual infection with Helicobacter bilis and Helicobacter hepaticus in p-glycoprotein-deficient mdr1a-/- mice results in colitis that progresses to dysplasia. Am J Pathol. 2005;166:1793-1806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 83] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 100. | Paik J, Fierce Y, Treuting PM, Brabb T, Maggio-Price L. High-fat diet-induced obesity exacerbates inflammatory bowel disease in genetically susceptible Mdr1a-/- male mice. J Nutr. 2013;143:1240-1247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 77] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 101. | Ellinghaus D, Zhang H, Zeissig S, Lipinski S, Till A, Jiang T, Stade B, Bromberg Y, Ellinghaus E, Keller A. Association between variants of PRDM1 and NDP52 and Crohn’s disease, based on exome sequencing and functional studies. Gastroenterology. 2013;145:339-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 130] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 102. | Jonker JW, Buitelaar M, Wagenaar E, Van Der Valk MA, Scheffer GL, Scheper RJ, Plosch T, Kuipers F, Elferink RP, Rosing H. The breast cancer resistance protein protects against a major chlorophyll-derived dietary phototoxin and protoporphyria. Proc Natl Acad Sci USA. 2002;99:15649-15654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 616] [Cited by in RCA: 629] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 103. | Kruh GD, Belinsky MG, Gallo JM, Lee K. Physiological and pharmacological functions of Mrp2, Mrp3 and Mrp4 as determined from recent studies on gene-disrupted mice. Cancer Metastasis Rev. 2007;26:5-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 59] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 104. | Goñi FM. The basic structure and dynamics of cell membranes: an update of the Singer-Nicolson model. Biochim Biophys Acta. 2014;1838:1467-1476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 236] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 105. | Quazi F, Molday RS. Lipid transport by mammalian ABC proteins. Essays Biochem. 2011;50:265-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 79] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 106. | Aye IL, Singh AT, Keelan JA. Transport of lipids by ABC proteins: interactions and implications for cellular toxicity, viability and function. Chem Biol Interact. 2009;180:327-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 101] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 107. | McDaniel K, Correa R, Zhou T, Johnson C, Francis H, Glaser S, Venter J, Alpini G, Meng F. Functional role of microvesicles in gastrointestinal malignancies. Ann Transl Med. 2013;1:4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 108. | Johnstone RW, Ruefli AA, Smyth MJ. Multiple physiological functions for multidrug transporter P-glycoprotein? Trends Biochem Sci. 2000;25:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 221] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 109. | Klappe K, Hummel I, Hoekstra D, Kok JW. Lipid dependence of ABC transporter localization and function. Chem Phys Lipids. 2009;161:57-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 91] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 110. | Su L, Mruk DD, Lui WY, Lee WM, Cheng CY. P-glycoprotein regulates blood-testis barrier dynamics via its effects on the occludin/zonula occludens 1 (ZO-1) protein complex mediated by focal adhesion kinase (FAK). Proc Natl Acad Sci USA. 2011;108:19623-19628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 111. | Dimeloe S, Frick C, Fischer M, Gubser PM, Razik L, Bantug GR, Ravon M, Langenkamp A, Hess C. Human regulatory T cells lack the cyclophosphamide-extruding transporter ABCB1 and are more susceptible to cyclophosphamide-induced apoptosis. Eur J Immunol. 2014;44:3614-3620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 44] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 112. | Pawlik A, Baśkiewicz-Masiuk M, Machaliński B, Safranow K, Gawrońska-Szklarz B. Involvement of P-glycoprotein in the release of cytokines from peripheral blood mononuclear cells treated with methotrexate and dexamethasone. J Pharm Pharmacol. 2005;57:1421-1425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 113. | Veldhoen M, Brucklacher-Waldert V. Dietary influences on intestinal immunity. Nat Rev Immunol. 2012;12:696-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 114. | Gollapudi S, Kim C, Gupta S. P-glycoprotein (encoded by multidrug resistance genes) is not required for interleukin-2 secretion in mice and humans. Genes Immun. 2000;1:371-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 115. | Pawlik A, Baskiewicz-Masiuk M, Machalinski B, Kurzawski M, Gawronska-Szklarz B. Involvement of C3435T and G2677T multidrug resistance gene polymorphisms in release of cytokines from peripheral blood mononuclear cells treated with methotrexate and dexamethasone. Eur J Pharmacol. 2005;528:27-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 116. | Pendse SS, Behjati S, Schatton T, Izawa A, Sayegh MH, Frank MH. P-glycoprotein functions as a differentiation switch in antigen presenting cell maturation. Am J Transplant. 2006;6:2884-2893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 117. | Raggers RJ, Vogels I, van Meer G. Multidrug-resistance P-glycoprotein (MDR1) secretes platelet-activating factor. Biochem J. 2001;357:859-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 118. | Papafili A, Hill MR, Brull DJ, McAnulty RJ, Marshall RP, Humphries SE, Laurent GJ. Common promoter variant in cyclooxygenase-2 represses gene expression: evidence of role in acute-phase inflammatory response. Arterioscler Thromb Vasc Biol. 2002;22:1631-1636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 259] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 119. | Bosch I, Dunussi-Joannopoulos K, Wu RL, Furlong ST, Croop J. Phosphatidylcholine and phosphatidylethanolamine behave as substrates of the human MDR1 P-glycoprotein. Biochemistry. 1997;36:5685-5694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 112] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 120. | Sobhani I, Hochlaf S, Denizot Y, Vissuzaine C, Rene E, Benveniste J, Lewin MM, Mignon M. Raised concentrations of platelet activating factor in colonic mucosa of Crohn’s disease patients. Gut. 1992;33:1220-1225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 45] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 121. | Xu LF, Teng X, Guo J, Sun M. Protective effect of intestinal trefoil factor on injury of intestinal epithelial tight junction induced by platelet activating factor. Inflammation. 2012;35:308-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 122. | Kaplan MJ, Radic M. Neutrophil extracellular traps: double-edged swords of innate immunity. J Immunol. 2012;189:2689-2695. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 123. | Yost CC, Weyrich AS, Zimmerman GA. The platelet activating factor (PAF) signaling cascade in systemic inflammatory responses. Biochimie. 2010;92:692-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 801] [Cited by in RCA: 883] [Article Influence: 67.9] [Reference Citation Analysis (0)] |
| 124. | Flak MB, Neves JF, Blumberg RS. Immunology. Welcome to the microgenderome. Science. 2013;339:1044-1045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1230] [Cited by in RCA: 1465] [Article Influence: 122.1] [Reference Citation Analysis (0)] |
| 125. | van de Wetering K, Sapthu S. ABCG2 functions as a general phytoestrogen sulfate transporter in vivo. FASEB J. 2012;26:4014-4024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 126. | Gonçalves P, Gregório I, Martel F. The short-chain fatty acid butyrate is a substrate of breast cancer resistance protein. Am J Physiol Cell Physiol. 2011;301:C984-C994. [PubMed] |
| 127. | Randolph GJ. Dendritic cell migration to lymph nodes: cytokines, chemokines, and lipid mediators. Semin Immunol. 2001;13:267-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 144] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 128. | Dietrich CG, de Waart DR, Ottenhoff R, Bootsma AH, van Gennip AH, Elferink RP. Mrp2-deficiency in the rat impairs biliary and intestinal excretion and influences metabolism and disposition of the food-derived carcinogen 2-amino-1-methyl-6-phenylimidazo. Carcinogenesis. 2001;22:805-811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 129. | Deeley RG, Cole SP. Substrate recognition and transport by multidrug resistance protein 1 (ABCC1). FEBS Lett. 2006;580:1103-1111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 211] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 130. | Jedlitschky G, Keppler D. Transport of leukotriene C4 and structurally related conjugates. Vitam Horm. 2002;64:153-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 131. | Wang D, Dubois RN. Eicosanoids and cancer. Nat Rev Cancer. 2010;10:181-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1494] [Cited by in RCA: 1405] [Article Influence: 93.7] [Reference Citation Analysis (0)] |
| 132. | Harris MS, Lichtenstein GR. Review article: delivery and efficacy of topical 5-aminosalicylic acid (mesalazine) therapy in the treatment of ulcerative colitis. Aliment Pharmacol Ther. 2011;33:996-1009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 133. | Urquhart BL, Ware JA, Tirona RG, Ho RH, Leake BF, Schwarz UI, Zaher H, Palandra J, Gregor JC, Dresser GK. Breast cancer resistance protein (ABCG2) and drug disposition: intestinal expression, polymorphisms and sulfasalazine as an in vivo probe. Pharmacogenet Genomics. 2008;18:439-448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 98] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 134. | Zaher H, Khan AA, Palandra J, Brayman TG, Yu L, Ware JA. Breast cancer resistance protein (Bcrp/abcg2) is a major determinant of sulfasalazine absorption and elimination in the mouse. Mol Pharm. 2006;3:55-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 120] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 135. | van der Heijden J, de Jong MC, Dijkmans BA, Lems WF, Oerlemans R, Kathmann I, Schalkwijk CG, Scheffer GL, Scheper RJ, Jansen G. Development of sulfasalazine resistance in human T cells induces expression of the multidrug resistance transporter ABCG2 (BCRP) and augmented production of TNFalpha. Ann Rheum Dis. 2004;63:138-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 65] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 136. | Furst DE. Acquired resistance of human T cells to sulfasalazine. Ann Rheum Dis. 2004;63:115-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 137. | Andersen V, Christensen J, Overvad K, Tjønneland A, Vogel U. Polymorphisms in NFkB, PXR, LXR and risk of colorectal cancer in a prospective study of Danes. BMC Cancer. 2010;10:484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 138. | Kopp TI, Andersen V, Tjonneland A, Vogel U. Polymorphisms in NFKB1 and TLR4 and interaction with dietary and life style factors in relation to colorectal cancer in a Danish prospective case-cohort study. PLoS One. 2015;10:e0116394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 139. | Herfarth HH, Martin CF, Sandler RS, Kappelman MD, Long MD. Prevalence of a gluten-free diet and improvement of clinical symptoms in patients with inflammatory bowel diseases. Inflamm Bowel Dis. 2014;20:1194-1197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 115] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 140. | Cohen AB, Lee D, Long MD, Kappelman MD, Martin CF, Sandler RS, Lewis JD. Dietary patterns and self-reported associations of diet with symptoms of inflammatory bowel disease. Dig Dis Sci. 2013;58:1322-1328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 141. | Jowett SL, Seal CJ, Pearce MS, Phillips E, Gregory W, Barton JR, Welfare MR. Influence of dietary factors on the clinical course of ulcerative colitis: a prospective cohort study. Gut. 2004;53:1479-1484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 330] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 142. | Gearry RB, Irving PM, Barrett JS, Nathan DM, Shepherd SJ, Gibson PR. Reduction of dietary poorly absorbed short-chain carbohydrates (FODMAPs) improves abdominal symptoms in patients with inflammatory bowel disease-a pilot study. J Crohns Colitis. 2009;3:8-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 225] [Article Influence: 14.1] [Reference Citation Analysis (1)] |
| 143. | Kyaw MH, Moshkovska T, Mayberry J. A prospective, randomized, controlled, exploratory study of comprehensive dietary advice in ulcerative colitis: impact on disease activity and quality of life. Eur J Gastroenterol Hepatol. 2014;26:910-917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 144. | Chan SS, Luben R, Olsen A, Tjonneland A, Kaaks R, Lindgren S, Grip O, Bergmann MM, Boeing H, Hallmans G. Association between high dietary intake of the n-3 polyunsaturated fatty acid docosahexaenoic acid and reduced risk of Crohn’s disease. Aliment Pharmacol Ther. 2014;39:834-842. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 145. | Iskandar H, Greer JB, Schraut WH, Regueiro MD, Davis PL, Hartman DJ, Siegel CA, Herfarth HH, Williams ED, Schwartz MB. IBD LIVE case series-case 1: smoking, a controversial but effective treatment for ulcerative colitis. Inflamm Bowel Dis. 2014;20:1696-1701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 129] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 146. | de Silva PS, Luben R, Shrestha SS, Khaw KT, Hart AR. Dietary arachidonic and oleic acid intake in ulcerative colitis etiology: a prospective cohort study using 7-day food diaries. Eur J Gastroenterol Hepatol. 2014;26:11-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 147. | Yamamoto T, Shiraki M, Nakahigashi M, Umegae S, Matsumoto K. Enteral nutrition to suppress postoperative Crohn’s disease recurrence: a five-year prospective cohort study. Int J Colorectal Dis. 2013;28:335-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 148. | John S, Luben R, Shrestha SS, Welch A, Khaw KT, Hart AR. Dietary n-3 polyunsaturated fatty acids and the aetiology of ulcerative colitis: a UK prospective cohort study. Eur J Gastroenterol Hepatol. 2010;22:602-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 130] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 149. | Spooren CE, Pierik MJ, Zeegers MP, Feskens EJ, Masclee AA, Jonkers DM. Review article: the association of diet with onset and relapse in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2013;38:1172-1187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 84] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 150. | Richman E, Rhodes JM. Review article: evidence-based dietary advice for patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2013;38:1156-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 78] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 151. | Andersen V, Olsen A, Carbonnel F, Tjønneland A, Vogel U. Diet and risk of inflammatory bowel disease. Dig Liver Dis. 2012;44:185-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 92] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 152. | Hou JK, Lee D, Lewis J. Diet and inflammatory bowel disease: review of patient-targeted recommendations. Clin Gastroenterol Hepatol. 2014;12:1592-1600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 150] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 153. | Charlebois A, Rosenfeld G, Bressler B. The Impact of Dietary Interventions on the Symptoms of Inflammatory Bowel Disease: A Systematic Review. Crit Rev Food Sci Nutr. 2015;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 51] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 154. | Flint HJ, Scott KP, Louis P, Duncan SH. The role of the gut microbiota in nutrition and health. Nat Rev Gastroenterol Hepatol. 2012;9:577-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1207] [Cited by in RCA: 1403] [Article Influence: 107.9] [Reference Citation Analysis (0)] |
| 155. | Walker AW, Ince J, Duncan SH, Webster LM, Holtrop G, Ze X, Brown D, Stares MD, Scott P, Bergerat A. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J. 2011;5:220-230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1347] [Cited by in RCA: 1181] [Article Influence: 84.4] [Reference Citation Analysis (0)] |
| 156. | Abraham C, Medzhitov R. Interactions between the host innate immune system and microbes in inflammatory bowel disease. Gastroenterology. 2011;140:1729-1737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 423] [Cited by in RCA: 405] [Article Influence: 28.9] [Reference Citation Analysis (1)] |
| 157. | Devkota S, Wang Y, Musch MW, Leone V, Fehlner-Peach H, Nadimpalli A, Antonopoulos DA, Jabri B, Chang EB. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10-/- mice. Nature. 2012;487:104-108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1442] [Cited by in RCA: 1414] [Article Influence: 108.8] [Reference Citation Analysis (0)] |
| 158. | Andersen V, Holst R, Kopp TI, Tjønneland A, Vogel U. Interactions between diet, lifestyle and IL10, IL1B, and PTGS2/COX-2 gene polymorphisms in relation to risk of colorectal cancer in a prospective Danish case-cohort study. PLoS One. 2013;8:e78366. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 51] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 159. | Arthur JC, Perez-Chanona E, Mühlbauer M, Tomkovich S, Uronis JM, Fan TJ, Campbell BJ, Abujamel T, Dogan B, Rogers AB. Intestinal inflammation targets cancer-inducing activity of the microbiota. Science. 2012;338:120-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1374] [Cited by in RCA: 1660] [Article Influence: 127.7] [Reference Citation Analysis (1)] |
| 160. | Tjalsma H, Boleij A, Marchesi JR, Dutilh BE. A bacterial driver-passenger model for colorectal cancer: beyond the usual suspects. Nat Rev Microbiol. 2012;10:575-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 161. | Zintzaras E. Is there evidence to claim or deny association between variants of the multidrug resistance gene (MDR1 or ABCB1) and inflammatory bowel disease? Inflamm Bowel Dis. 2012;18:562-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 484] [Cited by in RCA: 664] [Article Influence: 51.1] [Reference Citation Analysis (0)] |
| 162. | Manichanh C, Rigottier-Gois L, Bonnaud E, Gloux K, Pelletier E, Frangeul L, Nalin R, Jarrin C, Chardon P, Marteau P. Reduced diversity of faecal microbiota in Crohn’s disease revealed by a metagenomic approach. Gut. 2006;55:205-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1633] [Cited by in RCA: 1675] [Article Influence: 88.2] [Reference Citation Analysis (0)] |
| 163. | Goldsmith JR, Sartor RB. The role of diet on intestinal microbiota metabolism: downstream impacts on host immune function and health, and therapeutic implications. J Gastroenterol. 2014;49:785-798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 155] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 164. | Woodahl EL, Ho RJ. The role of MDR1 genetic polymorphisms in interindividual variability in P-glycoprotein expression and function. Curr Drug Metab. 2004;5:11-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 87] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 165. | Goldstein DB, Hirschhorn JN. In genetic control of disease, does ‘race’ matter? Nat Genet. 2004;36:1243-1244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 166. | Andersen V, Holst R, Vogel U. Systematic review: diet-gene interactions and the risk of colorectal cancer. Aliment Pharmacol Ther. 2013;37:383-391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 167. | Lewander A, Butchi AK, Gao J, He LJ, Lindblom A, Arbman G, Carstensen J, Zhang ZY, Sun XF. Polymorphism in the promoter region of the NFKB1 gene increases the risk of sporadic colorectal cancer in Swedish but not in Chinese populations. Scand J Gastroenterol. 2007;42:1332-1338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 48] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 168. | World Resources Institute Earth Trends The environmental information portal. 2009. Available from: http://earthtrends.wri.org/searchable_db/index.php?theme=8&variable_ID=193&action=select_countries. |
| 169. | Kang CS, Ban M, Choi EJ, Moon HG, Jeon JS, Kim DK, Park SK, Jeon SG, Roh TY, Myung SJ. Extracellular vesicles derived from gut microbiota, especially Akkermansia muciniphila, protect the progression of dextran sulfate sodium-induced colitis. PLoS One. 2013;8:e76520. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 313] [Cited by in RCA: 403] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 170. | Bufe B, Schumann T, Kappl R, Bogeski I, Kummerow C, Podgórska M, Smola S, Hoth M, Zufall F. Recognition of bacterial signal peptides by mammalian formyl peptide receptors: a new mechanism for sensing pathogens. J Biol Chem. 2015;290:7369-7387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 82] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 171. | Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, deRoos P, Liu H, Cross JR, Pfeffer K, Coffer PJ. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature. 2013;504:451-455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2516] [Cited by in RCA: 3417] [Article Influence: 284.8] [Reference Citation Analysis (0)] |
| 172. | Joscelyn J, Kasper LH. Digesting the emerging role for the gut microbiome in central nervous system demyelination. Mult Scler. 2014;20:1553-1559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 173. | Kriegel MA, Sefik E, Hill JA, Wu HJ, Benoist C, Mathis D. Naturally transmitted segmented filamentous bacteria segregate with diabetes protection in nonobese diabetic mice. Proc Natl Acad Sci USA. 2011;108:11548-11553. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 355] [Cited by in RCA: 342] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 174. | Wu HJ, Ivanov II, Darce J, Hattori K, Shima T, Umesaki Y, Littman DR, Benoist C, Mathis D. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity. 2010;32:815-827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1330] [Cited by in RCA: 1236] [Article Influence: 82.4] [Reference Citation Analysis (0)] |
| 175. | Lee YK, Menezes JS, Umesaki Y, Mazmanian SK. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc Natl Acad Sci USA. 2011;108 Suppl 1:4615-4622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 881] [Cited by in RCA: 1023] [Article Influence: 68.2] [Reference Citation Analysis (0)] |
| 176. | Ivanov II, Atarashi K, Manel N, Brodie EL, Shima T, Karaoz U, Wei D, Goldfarb KC, Santee CA, Lynch SV. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell. 2009;139:485-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3529] [Cited by in RCA: 3516] [Article Influence: 219.8] [Reference Citation Analysis (0)] |
| 177. | Yamada H, Nakashima Y, Okazaki K, Mawatari T, Fukushi JI, Kaibara N, Hori A, Iwamoto Y, Yoshikai Y. Th1 but not Th17 cells predominate in the joints of patients with rheumatoid arthritis. Ann Rheum Dis. 2008;67:1299-1304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 202] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 178. | Sarkar S, Fox DA. Targeting IL-17 and Th17 cells in rheumatoid arthritis. Rheum Dis Clin North Am. 2010;36:345-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 64] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 179. | Kemppinen AK, Kaprio J, Palotie A, Saarela J. Systematic review of genome-wide expression studies in multiple sclerosis. BMJ Open. 2011;1:e000053. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 77] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 180. | Shen W, Durum SK. Synergy of IL-23 and Th17 cytokines: new light on inflammatory bowel disease. Neurochem Res. 2010;35:940-946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 181. | Baumgart DC, Sandborn WJ. Inflammatory bowel disease: clinical aspects and established and evolving therapies. Lancet. 2007;369:1641-1657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1247] [Cited by in RCA: 1353] [Article Influence: 75.2] [Reference Citation Analysis (0)] |
| 182. | Huebner C, Browning BL, Petermann I, Han DY, Philpott M, Barclay M, Gearry R, McCulloch A, Demmers P, Ferguson LR. Genetic analysis of MDR1 and inflammatory bowel disease reveals protective effect of heterozygous variants for ulcerative colitis. Inflamm Bowel Dis. 2009;15:1784-1793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 171] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 183. | Farrell RJ, Murphy A, Long A, Donnelly S, Cherikuri A, O’Toole D, Mahmud N, Keeling PW, Weir DG, Kelleher D. High multidrug resistance (P-glycoprotein 170) expression in inflammatory bowel disease patients who fail medical therapy. Gastroenterology. 2000;118:279-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 196] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 184. | Hirano T, Onda K, Toma T, Miyaoka M, Moriyasu F, Oka K. MDR1 mRNA expressions in peripheral blood mononuclear cells of patients with ulcerative colitis in relation to glucocorticoid administration. J Clin Pharmacol. 2004;44:481-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 1.3] [Reference Citation Analysis (0)] |