Published online Sep 7, 2015. doi: 10.3748/wjg.v21.i33.9803
Peer-review started: March 5, 2015
First decision: April 13, 2015
Revised: April 21, 2015
Accepted: June 10, 2015
Article in press: June 10, 2015
Published online: September 7, 2015
Processing time: 188 Days and 13.8 Hours
Lymph node status is considered a key prognostic and predictive factor in patients with gastric cancer (GC). Although there is a practical approach to the intraoperative detection of sentinel lymph nodes (SLNs), such a procedure is not included in the European surgical protocol. In this report, we present a practical approach to SLN mapping in a representative case with early gastric cancer (EGC). A 74-year-old female was hospitalized with an endoscopically observed, superficially ulcerated tumor located in the antral region. Subtotal gastrectomy with D2 lymphadenectomy and SLN mapping was performed by injecting methylene blue dye into the peritumoral submucosal layer. An incidentally detected blue-stained lymph node located along the middle colic artery was also removed. This was detected 40 min after injection of the methylene blue. Histopathologic examination showed a pT1b-staged well-differentiated HER-2-negative adenocarcinoma. All of the 41 LNs located at the first, third, and fifth station of the regional LN compartments were found to be free of tumor cells. The only lymph node with metastasis was located along the middle colic artery and was considered a non-regional lymph node. This incidentally identified skip metastasis indicated stage IV GC. A classic chemotherapy regimen was given, and no recurrences were observed six months after surgery. In this representative case, low-cost SLN mapping, with a longer intraoperative waiting time, totally changed the stage of the tumor in a patient with EGC.
Core tip: The aim of this paper was to report an unusual case of early gastric cancer in which sentinel node mapping totally changed the tumor staging and therapeutic protocol.
- Citation: Bara Jr T, Gurzu S, Jung I, Kadar Z, Sugimura H, Bara T. Single skip metastasis in sentinel lymph node: In an early gastric cancer. World J Gastroenterol 2015; 21(33): 9803-9807
- URL: https://www.wjgnet.com/1007-9327/full/v21/i33/9803.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i33.9803
The term “early gastric cancer (EGC)” is used for gastric carcinomas with invasion limited to mucosa/muscularis mucosae (pT1a stage) or submucosal layer of the stomach (pT1b). Although the 5-year survival rate in EGC is reported to be about 90%, recurrences and metastases in the lymph nodes, liver, and even bones can occur[1]. For proper therapeutic management, careful preoperative evaluation of the lymph node status, tumor size and depth of infiltration is mandatory for accurate preoperative staging. Based on the international well-defined guidelines, with some national particularities, tumor resection consists of endoscopic mucosal resection (EMR), endoscopic submucosal dissection (ESD) or surgical excision[1-3]. The EMR is recommended for pT1a-staged non-ulcerated/non-metastatic differentiated carcinomas with diameter less than 2 cm. In ulcerated pT1a-differentiated carcinomas, EMR is indicated for tumors smaller than 1 cm[1-3]. ESD is recommended for non-metastatic differentiated carcinomas larger than 2 cm, with early invasion in the submucosa (pT1b), that do not involve associated angiolymphatic invasion. In larger tumors, poorly cohesive carcinomas (that are associated with a higher risk for metastases), metastatic cases (pN1-3 or M1), and advanced carcinomas (pT 2-4), gastrectomy is recommended; sub-total gastrectomy in carcinomas with at least 5 cm free margin up to the gastro-esophageal junction (8 cm in poorly cohesive carcinomas) and radical gastrectomy in the other cases[1-4].
In Eastern Europe, gastrectomy with D1/D2 lymphadenectomy is still considered the gold standard, even in EGC. Although there is a debate regarding D1 vs D2 lymphadenectomy, based on the duration of surgery and patient’s quality of life, studies performed in dedicated centers showed that the morbidity and mortality rate was approximately 0.5% and 2%, respectively, in D2, and not significantly different from D1. Based on these facts, recent guidelines require at least 15 regional lymph nodes for accurate tumor staging, and indicate D2 dissection as the standard procedure, in surgically-removed tumors[1-4]. For the correct identification of high-risk lymph nodes, sentinel node mapping may be helpful[3].
Moreover, in EGC, patients’ quality of life seems to be mainly influenced by the extent of resection and not the extent of lymphadenectomy. Independently, based on tumor stage, the morbidity and mortality of gastrectomy is high, ranging from 10% to 20%. These aspects highlight the necessity for changing therapeutic management, to reduce postoperative morbidity. The number of cases in which EMR/ESD is performed instead of surgery has increased. However, in low-income countries this change is not yet possible due to financial status, which does not allow a proper preoperative evaluation that includes endoscopic and computed tomography examinations of the thoracic and abdominal cavity, and does not facilitate proper screening programs. In these countries, gastrectomy with proper lymph node excision remains the standard, even in EGC. Taking into account the fact that the incidence of lymph node metastasis in EGC is 3%-24%[1,4], the introduction of an inexpensive and reliable system of preoperative evaluation of lymph node status is mandatory.
Although it is not universally accepted, due to the high rate of false negative results in intraoperatively examined nodes[5], the detection of sentinel lymph nodes (SLNs) is considered useful because the gastric lymphatic drainage can have aberrant flow[6] with identified sentinel nodes outside the lymphatic basin[5]. The rate of false-negative results for intraoperatively-examined lymph nodes depends on the clinical experience of the surgeon and the intraoperative method of detection (i.e., blue dye, indocyanine green), but is also related to the fact that only one plane-frozen sections can be performed to examine the nodes in a short-time and to retain tissue for further paraffin-embedding; micro-metastases cannot be detected intraoperatively. Even in large centers, intraoperative false-negative results were reported in more than 40% of cases; in paraffin-embedded sections, postoperatively, the rate was 14%[5]. Postoperatively, this rate also depends heavily on the method of SLN analysis (i.e., IHC, RT-qPCR), the number of intraoperatively examined lymph nodes and the number of sections per examined node.
In this paper, we present a representative case in which the accurate detection of SLN totally changed the postoperative therapeutic protocol in a patient with EGC.
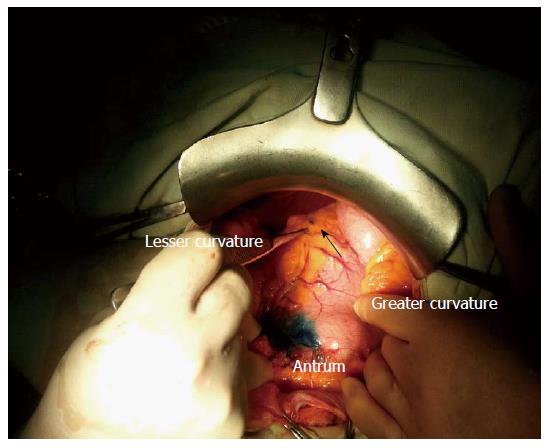
A 74-year-old previously healthy female was hospitalized due to an episode of hematemesis. Upper endoscopy detected 3 cm × 2.5 cm ulcerated tumor located in the antrum. Histopathologic examination of the biopsy specimen showed a well-differentiated adenocarcinoma limited to the gastric submucosa; it was decided to perform subtotal gastrectomy with D2 lymphadenectomy and intraoperative SLN mapping by injecting 2 mL of methylene blue dye into the submucosal layer of the stomach, in the neighborhood of the tumor, circumferentially at four cardinal points (Figure 1). This injection was carried out before mobilization of the stomach to avoid alteration of lymphatic drainage[5]. A signed written consent for the surgical intervention was obtained from the patient.
Following submucosal injection of methylene blue (Figure 1), we waited 10 min and then removed the first two blue-stained nodes, which were located at the 5th station (lesser curvature) and considered SLNs (Figure 1); both nodes were submitted for frozen tissue section and found to be free of tumor.
Standard subtotal gastrectomy and D2 lymphadenectomy were carried out. However, during the second phase of surgery (40 min after injection of methylene blue), two blue-stained lymph nodes were incidentally detected at the level of the middle colic artery, considered the 15th lymph node station; these were removed, together with seven other non-stained neighboring nodes. Finally, lymphadenectomy which included lymph node stations 1, 3, 4, 5, and 6 (lymph node compartment I); stations 7, 8, 9, and 12 (compartment II); and lymph node stations 15 (compartment IV), respectively, was performed (Table 1).
| Lymph node compartment | Total number of lymph nodes | Total number of blue nodes | Total numberof positive blue nodes | |
| Detected intraoperatively by surgeon (SLN) | Detected postoperatively by pathologist | |||
| I | 19 | 2 | 5 | - |
| II | 16 | - | 3 | - |
| IV | 7 | 2 | - | 1 |
| Total | 42 | 4 | 8 | 1 |
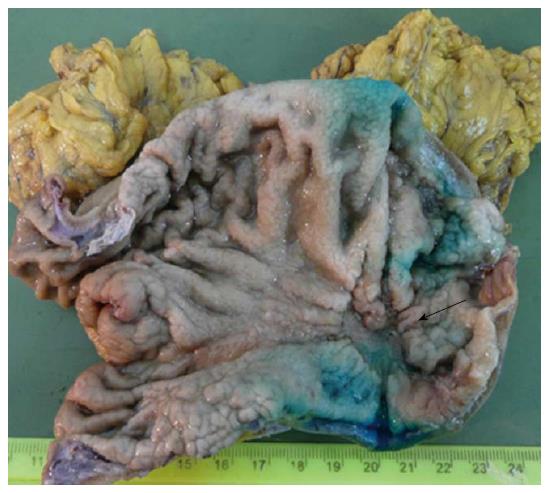
Gross examination of the surgical specimen showed a 35 mm × 20 mm ulcerated tumor of the antrum and lesser curvature with a maximum thickness of 10 mm (Figure 2), which was microscopically shown to be a pT1b-staged well-differentiated adenocarcinoma. The deepest level of infiltration was the middle third of the gastric submucosa.
With regard to the lymph nodes, a total of 41 lymph nodes were identified, including those of the middle colic artery. Of the 41 nodes, with the exception of the 4 intraoperatively detected SLNs (2 from station 5 and 2 from station 15), another 8 nodes were stained blue (4 from station 3, 1 from station 6, and 3 from station 7). Of the 41 nodes, 40 were free of tumor cells. The only positive node was one of the two blue-stained nodes that were incidentally seen intraoperatively at the level of the middle colic artery; this node was considered a skip metastasis (Table 1). Because this lymph node station was far from the primary tumor, this was considered to be a distant metastasis[7], and the final tumor stage was pT1N0M1 (stage IV). There were no tumor implants in the omentum, no invasion in blood vessels, and no intraoperatively detected liver metastasis. All the resection margins were free of tumor.
The immunohistochemical and molecular examinations showed a p53/CDH1-wild type-tumor that displayed positivity for E-cadherin, keratin 7 and 20, and a cytoplasmic pattern of maspin; p53 and Ki67 nuclear expression was seen in over 70% of the tumor cells. HER-2 was not expressed by the tumor cells.
In terms of tumor proximity, an unusual proliferation of extremely large vessels with thick or thinner walls was noted in the mucosa and submucosa, which corresponded to Dieulafoy’s lesion. Due to HER-2 negativity, trastuzumab was not included in the postoperative oncologic therapy. A classic chemotherapy regimen was recommended. The postoperative evolution was favorable; no recurrences or metastases were reported six months after surgery.
Data regarding the concept of SLN in surgical practice were first presented by Morton et al[8] in 1992; since then, SLN detection has become a standard of care in patients with melanoma and is used in cancers of other organs, such as the breast and colon[9,10].
With regard to gastric cancer, the application of SLN was first carried out in 2001 by Hiratsuka et al[11]’s team, who used indocyanine green in T1 and T2 cases, with a success rate of 99%. Later, the same team reported a high rate of false-negative results in T1 cases, using indocyanine green[5]. To increase the sensitivity, a double-marker method that used radioactive colloids and blue dye was recommended for EGC[6,12]. In the most recent report, non-exposed endoscopic wall-inversion surgery with sentinel node basin dissection was suggested as the standard of care in patients with EGC and a high risk of node metastases[13].
In the present case, the intraoperative examination of SLNs also revealed a preliminary false-negative result. However, in contrast to other studies, in which SLNs are removed first (10-15 min after injection of the dye) and then gastrectomy and lymphadenectomy are performed[5], we waited for the intraoperative histopathologic report before carrying out the gastrectomy. During this waiting period, the methylene blue dye migrated to the skip metastatic nodes located along the middle colic artery.
This case suggests that, due to aberrant drainage of the lymphatic fluid and a high rate of skip metastases, which is approximately 5% in EGC and 17% in advanced GC[14], SLN mapping can influence the tumor staging protocol. Increasing the period of waiting can increase the number of detected nodes and the sensitivity of SLN mapping. Although most of the skip metastases were detected in the lymph nodes located in stations 7 and 8 (compartment II) and in the para-aortic station[4], especially in tumors located in the lesser curvature and distal stomach[15], the nodes of the middle colic artery should also be checked intraoperatively and removed if there is any suspicion of metastatic involvement. To date, no data regarding the impact of middle colic artery node removal with standard D2 lymphadenectomy have been published.
Special attention should also be paid to gastric stump cancer and cases with vascular malformations, such as Dieulafoy lesions, which can also be associated with aberrant lymphatic drainage[16]. Because most gastric vascular malformations are undiagnosed, hematemesis may be a valuable clinical sign, especially when only one episode occurs, without any other symptoms, such as in the current case.
In the case under study, which showed several indicators of node metastases risk, such as tumor size larger than 3 cm, ulceration, location in the distal stomach, involvement of the submucosa, and associated vascular malformation, the correct staging and therapeutic management were carried out as a result of a long waiting time in SLN mapping and the detection of a single skip metastasis in an extra-regional lymph node. In view of the low cost and the absence of associated side effects in GC, we suggest that all blue stained nodes, whether identified in vivo or ex vivo, should be considered as SLNs.
A 74-year-old female with an early gastric cancer and skip metastasis.
Hematemesis and ulcerated tumor of the stomach, at upper endoscopy.
Chronic peptic ulcer, gastric metastasis.
Due to the diagnosis of adenocarcinoma in the gastric biopsy specimen, gastrectomy was performed without supplementary investigations.
Examination of the surgical specimen from the stomach revealed a well-differentiated adenocarcinoma with invasion in the middle third of the submucosal layer.
Classic chemotherapy. HER-2 negativity did not allow therapy with trastuzumab.
No reported cases of EGC and skip metastasis at the level of the middle colic artery.
Skip metastasis is a “jump” or discontinuous nodal metastasis that occurs as a result of aberrant lymphatic flow.
This case report highlights the aberrant behavior of EGC. An inexpensive method of sentinel lymph node mapping could modify the therapeutic guideline (the type of lymphadenectomy) in patients with EGC.
This article showed the benefits of the proper surgeon-pathologist team work. It shows the inexpensive way of a proper diagnosis, evaluation and therapy of EGCs with aberrant behavior.
P- Reviewer: Jagric T S- Editor: Yu J L- Editor: Webster JR E- Editor: Zhang DN
| 1. | Kobayashi M, Okabayashi T, Sano T, Araki K. Metastatic bone cancer as a recurrence of early gastric cancer -- characteristics and possible mechanisms. World J Gastroenterol. 2005;11:5587-5591. [PubMed] |
| 2. | Yada T, Yokoi C, Uemura N. The current state of diagnosis and treatment for early gastric cancer. Diagn Ther Endosc. 2013;2013:241320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 3. | Waddell T, Verheij M, Allum W, Cunningham D, Cervantes A, Arnold D. Gastric cancer: ESMO-ESSO-ESTRO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24 Suppl 6:vi57-vi63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 219] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 4. | Gotoda T, Yanagisawa A, Sasako M, Ono H, Nakanishi Y, Shimoda T, Kato Y. Incidence of lymph node metastasis from early gastric cancer: estimation with a large number of cases at two large centers. Gastric Cancer. 2000;3:219-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1308] [Cited by in RCA: 1325] [Article Influence: 53.0] [Reference Citation Analysis (0)] |
| 5. | Miyashiro I, Hiratsuka M, Sasako M, Sano T, Mizusawa J, Nakamura K, Nashimoto A, Tsuburaya A, Fukushima N. High false-negative proportion of intraoperative histological examination as a serious problem for clinical application of sentinel node biopsy for early gastric cancer: final results of the Japan Clinical Oncology Group multicenter trial JCOG0302. Gastric Cancer. 2014;17:316-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 113] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 6. | Kitagawa Y, Takeuchi H, Takagi Y, Natsugoe S, Terashima M, Murakami N, Fujimura T, Tsujimoto H, Hayashi H, Yoshimizu N. Sentinel node mapping for gastric cancer: a prospective multicenter trial in Japan. J Clin Oncol. 2013;31:3704-3710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 232] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 7. | Edge SB, Nyrd DR, Compton CC, Fritz AG, Greene FL, Trotti A, editors . Stomach. 7th ed. Berlin: Springer 2010; 145-152. |
| 8. | Morton DL, Wen DR, Wong JH, Economou JS, Cagle LA, Storm FK, Foshag LJ, Cochran AJ. Technical details of intraoperative lymphatic mapping for early stage melanoma. Arch Surg. 1992;127:392-399. [PubMed] |
| 9. | Bara T, Bara T, Bancu S, Egyed IZ, Gurzu S, Bancu L, Azamfirei L, Feher AM. [Sentinel lymph node mapping in colorectal cancer]. Chirurgia (Bucur). 2011;106:195-198. [PubMed] |
| 10. | Gurzu S, Jung I, Bara T, Bara T, Szentirmay Z, Azamfirei L, Tóth E, Turcu M, Egyed-Zsigmond E. Practical value of the complex analysis of sentinel lymph nodes in colorectal carcinomas. Rom J Morphol Embryol. 2011;52:593-598. [PubMed] |
| 11. | Hiratsuka M, Miyashiro I, Ishikawa O, Furukawa H, Motomura K, Ohigashi H, Kameyama M, Sasaki Y, Kabuto T, Ishiguro S. Application of sentinel node biopsy to gastric cancer surgery. Surgery. 2001;129:335-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 191] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 12. | Takeuchi H, Kitagawa Y. Sentinel node navigation surgery in patients with early gastric cancer. Dig Surg. 2013;30:104-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 13. | Goto O, Takeuchi H, Kawakubo H, Sasaki M, Matsuda T, Matsuda S, Kigasawa Y, Kadota Y, Fujimoto A, Ochiai Y. First case of non-exposed endoscopic wall-inversion surgery with sentinel node basin dissection for early gastric cancer. Gastric Cancer. 2015;18:434-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 72] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 14. | Su Z, Shu K, Zheng M, Sun X, Fang Z, Wang G. Sentinel lymph node and skip metastases in gastric cancer: a prospective study. Hepatogastroenterology. 2013;60:1513-1518. [PubMed] |
| 15. | Lee JH, Lee HJ, Kong SH, Park do J, Lee HS, Kim WH, Kim HH, Yang HK. Analysis of the lymphatic stream to predict sentinel nodes in gastric cancer patients. Ann Surg Oncol. 2014;21:1090-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 16. | Gurzu S, Copotoiu C, Azamfirei L, Jung I. A Caliber Persistent Artery (Dieulafoy’s Lesion) which is Associated with an Early-Stage Gastric Stump Cancer Following a Distal Gastrectomy. J Clin Diagn Res. 2013;7:1717-1719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |