Published online Sep 7, 2015. doi: 10.3748/wjg.v21.i33.9707
Peer-review started: March 19, 2015
First decision: May 18, 2015
Revised: June 2, 2015
Accepted: July 15, 2015
Article in press: July 15, 2015
Published online: September 7, 2015
Processing time: 173 Days and 1.8 Hours
Despite advances in diagnosis and treatment, gastric cancer remains one of the most common malignant tumors worldwide, and early diagnosis remains a challenge. The lack of effective methods to detect these tumors early is a major factor contributing to the high mortality in patients with gastric cancer, who are typically diagnosed at an advanced stage. Additionally, the early detection of metastases and the curative treatment of gastric cancer are difficult to achieve, and the detailed mechanisms remain to be fully elucidated. Thus, the identification of valuable predictive biomarkers and therapeutic targets to improve the prognosis of patients with gastric cancer is becoming increasingly important. Contactin 1 (CNTN1), a cell adhesion molecule, is a glycosylphosphatidylinositol-anchored neuronal membrane protein that plays an important role in cancer progression. The expression of CNTN1 is upregulated in primary lesions, and its expression level correlates with tumor metastasis in cancer patients. The current evidence reveals that the functions of CNTN1 in the development and progression of cancer likely promote the invasion and metastasis of cancer cells via the VEGFC/FLT4 axis, the RHOA-dependent pathway, the Notch signaling pathway and the epithelial-mesenchymal transition progression. Therefore, CNTN1 may be a novel biomarker and a possible therapeutic target in cancer treatment in the near future.
Core tip: Gastric cancer remains a major public health issue, and the investigation of therapeutic targets and biomarkers is of great importance. The accumulated evidence demonstrates that contactin 1 (CNTN1) plays a crucial role in cancer progression. In this manuscript, the role of CNTN1 in cancer is discussed and the mechanisms through which CNTN1 mediates the invasion and metastasis of cancer cells are summarized.
- Citation: Chen DH, Yu JW, Jiang BJ. Contactin 1: A potential therapeutic target and biomarker in gastric cancer. World J Gastroenterol 2015; 21(33): 9707-9716
- URL: https://www.wjgnet.com/1007-9327/full/v21/i33/9707.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i33.9707
Gastric cancer (GC) remains a global cancer burden, with a total of 9896000 new GC cases and 738000 deaths in 2008, accounting for 8% of the total cancer cases and 10% of the total cancer-related deaths[1], which makes GC the fifth most common malignancy and the third leading cause of death in the world[1,2]. More than 70% of GC cases occur in developing countries, and half of the total number of GC cases around the world occur in East Asia (mainly China)[3]. Although various factors, including early detection strategies[4,5] and improvements in living standards[6,7], have contributed to the observed decrease in the incidence of GC, it remains one of the most frequently occurring cancers, and the early diagnosis and effective treatment of GC remain challenging. Early in the disease course, patients with GC experience nonspecific symptoms, and most GC patients are diagnosed with advanced GC due to a lack of early-stage symptoms; late-stage diagnoses are generally too late for effective treatment, resulting in a five-year survival rate of only approximately 20%[8].
At present, the effects of conventional chemotherapy, radiotherapy and immunotherapy are generally not satisfactory, and surgery remains the main choice for the treatment of patients with GC if R0, or at least R1, resection can be achieved. Despite developments in oncology theory, surgical technique, operative skill and chemotherapy in recent decades, metastasis and post-surgery recurrence are critical obstacles to the curative treatment of GC in patients with metastatic cancer[9]. Thus, a comprehensive investigation of the mechanisms of initiation and progression of GC is crucial for the identification of novel, sensitive and specific biomarkers for early diagnosis and for the detection of potential therapeutic targets. In recent years, several novel targets and biomarkers for GC such as long noncoding RNAs[10], promoter methylated microRNAs[11], circulating microRNAs[12], and the EZH2[13], SPARC[14], EPHA2[15,16] and CNTN1 genes[17,18] that are involved in signaling pathways have been identified, and these may have potential prognostic value and application as novel therapeutic targets.
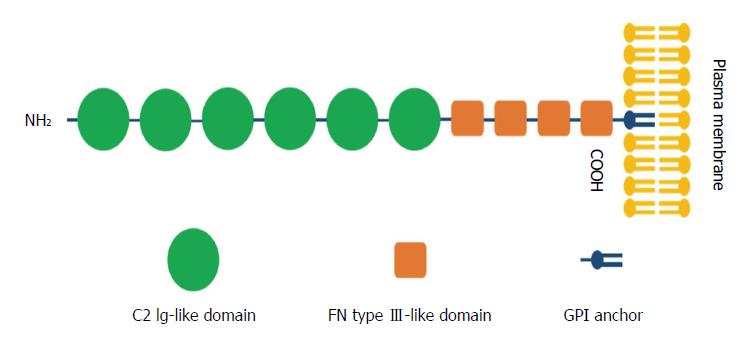
CNTNs are a subpopulation of molecules belonging to the immunoglobulin (Ig) superfamily, which contains six members: CNTN1, TAG-1/CNTN2, BIG-1/CNTN3, BIG-2/CNTN4, NB-2/CNTN5 and NB-3/CNTN6[19,20]. CNTN1, as the first identified member of the CNTN family of CAMs[21], was mapped to the chromosome 12q11-q12 region and exhibits a common structure of six C2 Ig-like repeats, four fibronectin type III (FNIII) domains, and a glycosylphosphatidylinositol (GPI)-linkage at the carboxy-terminus that anchors the protein to the extracellular plasma membrane[22] (Figure 1).
Previous investigations involving limited organs and diseases have revealed that CNTN1 is highly expressed in the human brain and neuronal tissues and is of vital importance for nervous system development. The gene plays a critical role in neuronal and glial development and differentiation, in myelination and synaptogenesis, and in fasciculation[19,23-27]. Indeed, CNTN1 mediates neuron-glial interactions mainly through binding with elements [tenascin-C, tenascin-R, and receptor protein tyrosine phosphatase β (RPTPβ)/phosphagen] in the extracellular matrix, leading to axonal growth and fasciculation[21]. Interestingly, since the discovery of specific interactions between CNTN1 and PTPRZ[20,28], it is widely accepted that the transmembrane form of PTPRZ expressed on glial cells interacts in trans with CNTN1 expressed on the surface of axons, leading to neurite outgrowth and glial adhesion[29,30]. Unlike the general conception, the most recent study concluded that CNTN1 expressed on the surface of oligodendrocyte precursor cells (OPCs) interacts with a soluble form of PTPRZ to modulate the proliferation of OPCs[31]. Other major functions of CNTN1 include serving as a ligand to the Notch receptor, thereby influencing oligodendrocyte maturation[27], and interacting in cis with receptor protein tyrosine phosphatase α to transduce extracellular signals to tyrosine kinase FYN[32], thereby regulating cell mobility[33]. Furthermore, the CNTN1 expression level has been found to decrease as patients exhibit age-related declines in memory[34], to progressively increase during postnatal life[35] and to enhance neurogenesis, synaptic plasticity and memory[36], suggesting that it may be involved in memory processes, consistent with the results of studies investigating its participation in some forms of synaptic plasticity and in neurotransmitter release[37]. In addition to the roles described above, CNTN1 has been correlated with devastating brain disorders such as autism and schizophrenia[38].
Despite the increasing number of studies focusing on the regulatory roles of CNTN1 in the nervous system, little attention has been paid to its function outside of the nervous system. Strikingly, several recent studies have demonstrated that CNTN1 also plays a key role in diseases not related to the nervous system, and its most notable function is its participation in cancer progression in cancers such as esophageal squamous cell carcinoma (ESCC)[39], GC[40], lung adenocarcinoma[17,41], oral squamous cell carcinoma (OSCC)[42], hepatocellular carcinoma[43] and prostate cancer[44], which is in agreement with accumulating evidence supporting the finding that certain members of the Ig superfamily promote the invasion and metastasis of cancer[45,46]. Moreover, the 12q11-q12 chromosomal region, the location of CNTN1, is a breakpoint region in certain types of cancer, further demonstrating that CNTN1 plays a potential role in tumor formation and/or advancement. Su et al[17,41] first described CNTN1 as a metastasis-promoting oncogenic gene.
In a study of the crucial regulatory genes responsible for the invasion and metastasis of cancer cells, Su et al[17] unexpectedly discovered, through genome-wide cDNA microarray analysis, that CNTN1, which is detected in primary lung cancer, plays an essential role in lung cancer metastasis. These researchers demonstrated that the expression level of CNTN1, which is differentially expressed in tumor tissues, is positively correlated with the tumor stage and lymph node metastasis and negatively correlated with the prognosis of patients with lung adenocarcinoma. Similarly, reduced CNTN1 expression has been found to result in impairments in the ability of lung adenocarcinoma cells to invade Matrigel in vitro, the polymerization of filamentous-actin and the formation of focal adhesion structures. Additionally, the knockdown of CNTN1 in lung cancer cells specifically inhibits their ability to metastasize, but not their proliferation in vitro, and abolishes their metastatic capacity to increase survival but not the formation of subcutaneously transplanted tumors in an animal model, suggesting that CNTN1 plays a crucial role in the invasion and metastasis of lung cancer cells[17]. In support of this finding, Su et al[41] also demonstrated that the upregulation of CNTN1, induced by the VEGFC/FLT4 axis through the activation of the SRC/p38 MAPK-mediated CEBPA-dependent signaling pathway, may enhance the invasive capacity of different types of cancer cells (e.g., lung, cervical and breast cancers). Consistent with these reports, the tobacco carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) has been reported to enhance the invasiveness of lung cancer cells by elevating the expression level of CNTN1, which is regarded as a downstream effector of NNK via the α7 nicotinic acetylcholine receptor (α7 nAChR) downstream of the AKT and extracellular signal-regulated kinase (ERK) signaling pathway[47].
Since the discovery of the CNTN1-mediated invasion and metastasis of lung cancer cells, it has become increasingly evident that CNTN1 is also of critical importance in the induction of the invasion and metastasis of other cancer cells. Recent studies have revealed that the knockdown of FLT4 in human GC MKN45 cells using a short hairpin RNA lentiviral vector contributes to the downregulation of the downstream molecule CNTN1, indicating the possible involvement of CNTN1 in the invasion and metastasis of human GC cells[40]. Furthermore, CNTN1 is considered a recurrently mutated cell adhesion gene. Moreover, cell adhesion is the most highly enriched biological process among the mutated genes in the GC exomes[48]. More importantly, the mRNA and protein expression levels of CNTN1 are increased in primary lesions compared with adjacent normal gastric mucosal tissue[18,49]. The expression level of CNTN1 in primary lesions is significantly related to VEGFC or FLT4 expression; positively correlated with lymphatic invasion, lymph node metastasis and the TNM stage of patients with GC; and inversely associated with the prognosis of these patients. Moreover, patients with CNTN1-positive expression show a higher lymphatic vessel density (LVD)[18]. Taken together, the results show that CNTN1 may facilitate the invasion and metastasis of GC cells and may be a valuable indicator of poor prognosis in patients with GC.
In addition to these observations associated with GC, previous studies have demonstrated CNTN1 expression in upper gastrointestinal cancers and have noted its involvement in esophageal cancer[50] and ESCC[39] and OSCC[42]. As previously reported, the expression of CNTN1 is closely correlated with that of VEGFC because VEGFC can induce the recruitment of CEBPA to bind with the CNTN1 promoter[41]. Interestingly, the enhancement of the migration of esophageal cancer cells, which is attributable to a corresponding increase in VEGFC expression, is significantly reversed through a reduction in CNTN1 expression[50], suggesting that CNTN1 may play a key role in the VEGFC-induced migration of esophageal cancer cells. Moreover, the mRNA and protein expression levels of CNTN1 have been found to be elevated in ESCC tissues by real-time PCR and immunohistochemistry and to be significantly correlated with the ESCC stage, lymph node metastasis and lymphatic invasion, indicating the participation of CNTN1 in esophageal cancer progression[39]. Therefore, inhibitors against CNTN1 may be a promising therapy for ESCC. Similarly, CNTN1 is overexpressed in patients with OSCC. High expression of CNTN1 is markedly correlated with regional lymph node metastasis in patients with OSCC. CNTN1 expression is markedly associated with the survival time of patients with OSCC. The knockdown of CNTN1 expression decreases the invasion potential of OSCC cells, but CNTN1 ablation exerts no effect on the proliferation of OSCC cells, confirming that CNTN1 promotes the malignant progression of OSCC through an exclusive activation of the metastatic potential. Thus, CNTN1 may be a useful predictor of prognostic outcome in patients with OSCC[42].
Studies on the identification of new genes associated with melanoma have suggested that CNTN1, as an activator of Notch signaling, has been overlooked as a key factor in the progression of melanoma and should be investigated in greater depth[51]. The deregulation of CNTN1 mRNA expression has also been demonstrated in endometrial adenocarcinoma (EAC). The expression level of CNTN1 is higher in late-stage than early-stage EAC[52]. Additionally, CNTN1 has been reported to be involved in human astrocytic gliomas[53], glioblastoma[54], hepatocellular carcinoma[43] and prostate cancer[44].
Recent data have indicated that several genetic and epigenetic changes have an influence on patient prognosis and survival[55-57]. The field of “molecular pathological epidemiology (MPE)”, addressing the basic heterogeneity of disease processes, has emerged as an interdisciplinary integration of “molecular pathology” and “epidemiology” that aims to understand the interplay among etiological factors, cellular molecular characteristics and disease evolution. The holistic MPE approach can be performed to assess the interactive effects of environmental influences and disease molecular signatures on disease progression and enables us to obtain novel pathogenic insights regarding causality[58]. In conclusion, the gene encoding CNTN1, a factor that potentially accelerates cancer progression, should be further investigated with the ultimate aim of developing an effective cancer treatment.
Despite mounting evidence supporting the influence of CNTN1 on cancer metastasis, insights into the underlying mechanisms responsible for this process remain to be discovered for the development of targeted therapy. At present, several studies have shown that a variety of signaling pathways are involved in CNTN1-mediated cell functions. CNTN1 is a GPI-anchored neuronal membrane protein that functions as a neural cell adhesion molecule (NCAM) and has been observed to interact with other cell-surface proteins that are thought to participate in a variety of signaling pathways and cell functions in the nervous system[20,25,31]. As previously reported, CNTN1 interacts in trans with RPTPβ to facilitate neurite outgrowth[29] and in cis with RPTPα to transduce extracellular signals to FYN kinase[32], a member of the SRC kinase family that modulates cell mobility[33,59]. In addition, the involvement of Notch signaling pathways in CNTN1-mediated cell function has been demonstrated. Nonetheless, the mechanism through which CNTN1 promotes the metastasis of cancer cells remains unclear.
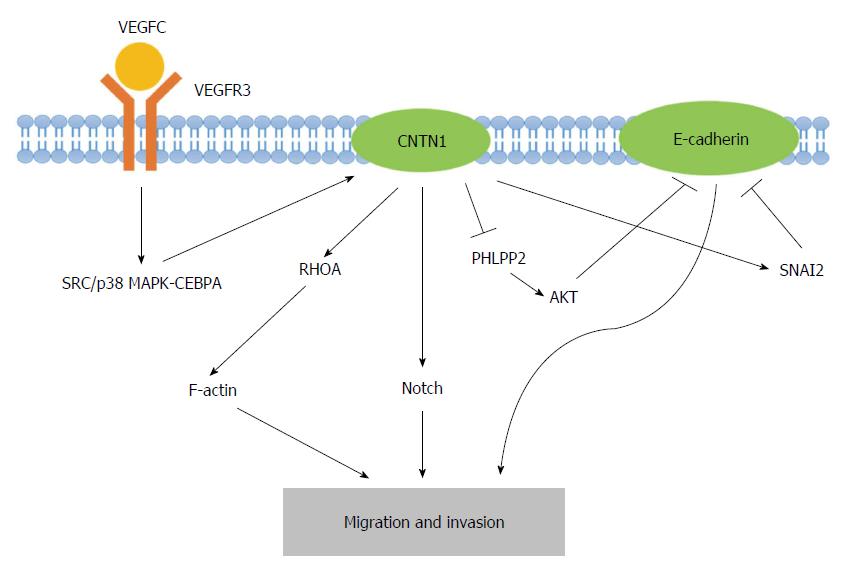
Recently, the VEGFC/FLT4 axis, which stimulates the upregulation of CNTN1 expression, has emerged as a central feature of CNTN1-induced cancer cell metastasis[41]. Su et al[41] concluded that the expression of VEGFC/FLT4 correlates with the stage and lymph node metastasis of cancer and the survival of cancer patients and that the VEGFC/FLT4 axis promotes the migration and invasion of cancer cells. Investigations into the functional linkages between CNTN1 and VEGFC/FLT4-induced cell invasion revealed that CNTN1 is involved in cancer cell invasiveness and acts as a downstream effector of the VEGFC/FLT4 signaling pathway, in which SRC/p38 MAPK-mediated CEBPA signaling is required for VEGFC/FLT4-mediated CNTN1 expression. Furthermore, rearrangements of F-actin-containing microfilament bundles have been found in VEGFC/FLT4-mediated CNTN1 expression, indicating that the VEGFC/FLT4 axis regulates cancer cell invasion via CNTN1-dependent F-actin (a member of the actin cytoskeleton) rearrangement in cancer cells[41]. Indeed, F-actin is regularly polymerized and depolymerized in migratory cells and is required for cell motility[60]. Furthermore, RHO genes, including CDC42, RAC1, and RHOA, are best characterized by their effects on the cytoskeleton and cell adhesion. Interestingly, the activity of the small GTP-binding protein RHOA has also been demonstrated to be involved in CNTN1-induced F-actin polymerization and cell invasion[17]. In addition to the VEGFC/FLT4 axis and the RHOA-dependent pathway, Notch signaling may play a partial role in CNTN1-mediated cancer cell invasion. In melanoma, the activation of Notch signaling appears to be of vital importance throughout tumor progression. For example, the activation of Notch1 in primary melanoma cells leads to a more metastatic phenotype[61]. Indeed, CNTN1, an activator of Notch, has been identified as a novel melanoma-associated biomarker[51]. Hence, the interaction between CNTN1 and Notch signaling may be a crucial factor in melanoma progression.
In addition to signaling, other factors play important roles in CNTN1-mediated cancer cell metastasis. It has been revealed that CNTN1 promotes cancer cell invasion through the inhibition of CDH1 (E-cadherin) expression[49]. The downregulation of E-cadherin is largely achieved through transcriptional regulatory mechanisms, including SNAIL, SNAI2, TWIST, ZEB1 and SIP1[62]. CNTN1 may indirectly inhibit E-cadherin through the upregulation of transcriptional factor SNAI2 rather than SNAIL. In addition, CNTN1 may decrease E-cadherin at the gene level as well as the transcriptional level. The mechanism through which the inhibition of the PH domain and leucine-rich repeat protein phosphatase 2 (PHLPP2), instead of phosphatase and tensin homolog (PTEN), results in AKT activation explains how CNTN1 reduces E-cadherin expression[63]. Furthermore, as previously demonstrated, the loss of E-cadherin is a major contributor to the invasion and metastasis of cancers[64]. The epithelial-mesenchymal transition (EMT) of cancer cells may provide a novel perspective for the theory of cancer stem cells (CSCs) and stem cell research in the relevant field of cancer progression. Thus, CNTN1 represses E-cadherin expression in cancer cells through the acquisition of a more invasive phenotype. Overall, CNTN1 induces the migration and invasion of cancer cells through a combination of the above-described mechanisms (Figure 2).
The origin of cancer has long been debated. The concept of clonal evolution, which explains how a set of clone cells accumulate mutations over a period of time to result in damage to normal cells and ultimately cancer, remains widely accepted as explaining the origin of cancer[65]. The theory of CSCs, so-called “tumor-initiating cells”, was first proposed in 1994[66], explaining how a small subpopulation of cells with self-renewal and pluripotency capabilities can explain why some cancer patients will likely relapse with metastatic and/or recurrent cancer even though the primary neoplasm has been completely resected. Mounting lines of evidence have indicated that CSCs play a crucial role in cancer recurrence and metastasis and exhibit resistance to chemotherapy and radiotherapy[67]. The presence of human CSCs has been confirmed in solid tumors, including breast, colon, pancreas, prostate[68] and stomach cancers[69,70]. However, the maintenance of CSCs can largely depend on their microenvironment (the stem cell niche), which can protect CSCs from extrinsic stimuli. Overall, the CSC niche is composed of proteins, cellular elements (e.g., macrophages, fibroblasts, and endothelial cells), and structural moieties (e.g., E-cadherin) that interact with each other to protect CSCs. In addition, several signaling pathways [e.g., WNT, Notch, TAB2 (TGF-β) and NF-κB] participate in the maintenance of the undifferentiated state of CSCs[65,71]. Therefore, in the current theory, the CSC niche is a prerequisite for carcinogenesis[71]; thus, the therapeutic targeting of this niche is of great interest.
Similarly, it has been widely acknowledged that CAMs play an important role in cancer progression as well as cancer cell invasion and metastasis[72]. Consistent with the identification of several signaling pathways that trigger EMT involved in CSC development[73], researchers have demonstrated that the EMT of cancer cells can result in the properties of migration, invasion and CSCs[74,75]. Loss of E-cadherin expression is regarded as the hallmark of the invasive phase of cancer. Moreover, the reversible switch of the EMT, which is termed the mesenchymal-to-epithelial transition (MET), is considered to participate in the establishment and stabilization of distant metastases in the target organ by allowing cancerous cells to regain an epithelial identity and thereby regain their proliferating ability[76].
Similarly, the EMT participates in the progression of GC and endows GC cells with the properties of migration and invasion, thus promoting cancer metastasis. This multi-step process includes the loss of cellular adhesion molecules, promoting cell adhesion to the extracellular matrix (ECM), the degradation of the ECM and the motility of tumor cells. Additionally, the CSC characteristics acquired by the EMT endow GC cells with the abilities to self-renew and to develop drug resistance and apoptotic resistance[62].
In recent years, circulating tumor cells (CTCs), which are defined as tumor cells originating from either the primary tumor or metastatic tissues, have been discovered in most epithelial tumors and increase the risk of regional recurrence as well as distant metastasis. Stem cell properties and the EMT are commonly observed in CTCs, and the phenotype of CTCs is regarded as a more attractive predictor of prognosis than solely the number of CTCs in the blood[77]. Indeed, only a small population of CTCs with some properties of stem cells, which are called circulating tumor stem cells (CTSCs), can migrate to other targeted organs/tissues for the development of secondary tumors[78]. Strikingly, in addition to their clinical importance as an independent predictive biomarker of poor prognosis in patients with GC[79], evaluating the number of CTCs in therapy may greatly improve the management of patients, and recent developments of novel technology have enabled the detection of CTCs in cancer patients[80]. Moreover, a recent study revealed that CTSCs provide a more important and specific value for the prognosis of in patients with GC[81]. Nevertheless, further investigations of the molecular mechanisms of CTCs and CTSCs are urgently required to discover potential therapeutic targets for the prevention of micrometastasis and affecting the migration and TNM classification in GC[82], thereby improving the prognosis of cancer patients.
In recent decades, improvements in strategies for the detection, treatment and care of patients have contributed to a marked decrease in the mortality of GC worldwide. It is clear that upper gastrointestinal endoscopy is beneficial for improvements in the earlier diagnosis of GC. Magnifying endoscopy with narrow-band imaging (NBI), which is the result of technological innovations, has been reported to be more reliable for the early diagnosis of GC[83,84], and endoscopic submucosal dissection (ESD) is a preferred option compared with endoscopic mucosal resection (EMR)[85]. For advanced GC, surgery is usually regarded as the only curative treatment, whereas perioperatively adjuvant chemotherapy can to some extent improve the prognosis of resectable GC[86,87]. Advancements in surgical techniques, such as laparoscopic surgery and robot-assisted gastrectomy[88,89], are beneficial to the outcome of patients. Despite these findings, limited success has been achieved in the prevention of advanced GC, and most patients with GC are diagnosed at an advanced stage worldwide due mainly to the absence of adequate and appropriate screening that would identify gastric cancer in early stages, causing the optimal window for curative surgery to be missed. However, tumor biomarkers, such as the auxiliary analysis of disease-related indicators, have drawn increased attention over the years. Investigations of the mechanisms of invasion and metastasis of GC, which may provide key targets for new drug development, are of vital importance. Additionally, the identification of predictive and prognostic biomarkers can better provide targeted therapy for ideal patients.
Notable achievements have been made in molecularly targeted therapy. The use of the anti-ERBB2 (HER2) antibody trastuzumab and the anti-KDR (VEGFR2) antibody ramucirumab as supplementary chemotherapy for patients overexpressing HER2 or VEGFR2 receptor has led to significant gains in overall survival compared with chemotherapy alone[86,90]. In recent years, the potential molecular target CNTN1 has been the focus of investigations on its role in cancer metastasis. To the best of our knowledge, elevated CNTN1 expression levels in primary lesions are correlated with lymph node metastasis and the prognosis of patients with GC[18]. Thus, CNTN1 may be considered a potentially promising prognostic indicator and therapeutic target.
With regard to VEGF-C expression, previous studies have revealed a significant difference between positive and negative lymph node metastasis in patients with GC. VEGFC/FLT4 plays an important role in the development of GC, and GC patients overexpressing VEGFC/FLT4 in the primary lesion are more likely to present with lymph node metastasis[91]. Recent investigations have demonstrated that CNTN1 not only correlates with VEGFC, FLT4, lymph node metastasis and prognosis in patients with GC[18] but also acts as a downstream effector of the VEGFC/FLT4 axis to mediate GC cell invasion[40]. In addition to these findings, CNTN1 seems to promote the migration and invasion of GC cells via EMT alteration probably induced by inhibition of SNAI2[49]. As a consequence, CNTN1 may be a novel prognostic biomarker and potential therapeutic target in patients with GC. Despite this finding, the mechanism through which CNTN1 modulates GC cell invasion and metastasis remains incompletely explained. Further investigations are therefore required to obtain an in-depth characterization of its role and the mechanisms underlying the invasion and metastasis of GC.
CNTN1, a GPI-anchored adhesion molecule, was recently found to be overexpressed in various cancer tissues, including lung cancer, melanoma, OSCC, esophageal cancer, and GC. CNTN1 has been shown to not only promote the invasion and metastasis of cancer cells but also correlate with tumor metastasis in cancer patients. The VEGFC/FLT4 axis, Notch signaling and loss of E-cadherin via the inhibition of SNAI2 or the activation of AKT may contribute to the CNTN1-facilitated invasion and metastasis of cancer cells. Similarly, the expression level of CNTN1 is correlated with lymph node metastasis and the prognosis of patients with GC and positively associated with the invasion of GC cells. An in-depth understanding of the functional aspects of CNTN1 and the full mechanisms underlying CNTN1-induced invasion that result in a potentially applicable treatment for patients with GC is urgently needed. In conclusion, CNTN1 may be useful as a potential therapeutic target and biomarker in cancer in the future.
P- Reviewer: Georgoulias V, Goral V, Ogino S S- Editor: Yu J L- Editor: Wang TQ E- Editor: Ma S
| 1. | Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23762] [Cited by in RCA: 25543] [Article Influence: 1824.5] [Reference Citation Analysis (7)] |
| 2. | Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359-E386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20108] [Cited by in RCA: 20516] [Article Influence: 2051.6] [Reference Citation Analysis (20)] |
| 3. | Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893-2917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11128] [Cited by in RCA: 11837] [Article Influence: 845.5] [Reference Citation Analysis (4)] |
| 4. | Kaltenbach T, Sano Y, Friedland S, Soetikno R. American Gastroenterological Association (AGA) Institute technology assessment on image-enhanced endoscopy. Gastroenterology. 2008;134:327-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 128] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 5. | Lee KJ, Inoue M, Otani T, Iwasaki M, Sasazuki S, Tsugane S. Gastric cancer screening and subsequent risk of gastric cancer: a large-scale population-based cohort study, with a 13-year follow-up in Japan. Int J Cancer. 2006;118:2315-2321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 148] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 6. | Tkachenko MA, Zhannat NZ, Erman LV, Blashenkova EL, Isachenko SV, Isachenko OB, Graham DY, Malaty HM. Dramatic changes in the prevalence of Helicobacter pylori infection during childhood: a 10-year follow-up study in Russia. J Pediatr Gastroenterol Nutr. 2007;45:428-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 69] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 7. | Bertuccio P, Chatenoud L, Levi F, Praud D, Ferlay J, Negri E, Malvezzi M, La Vecchia C. Recent patterns in gastric cancer: a global overview. Int J Cancer. 2009;125:666-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 484] [Article Influence: 30.3] [Reference Citation Analysis (1)] |
| 8. | Jemal A, Tiwari RC, Murray T, Ghafoor A, Samuels A, Ward E, Feuer EJ, Thun MJ; American Cancer Society. Cancer statistics, 2004. CA Cancer J Clin. 2004;54:8-29. [PubMed] |
| 9. | Catalano V, Labianca R, Beretta GD, Gatta G, de Braud F, Van Cutsem E. Gastric cancer. Crit Rev Oncol Hematol. 2009;71:127-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 324] [Article Influence: 20.3] [Reference Citation Analysis (1)] |
| 10. | Fang XY, Pan HF, Leng RX, Ye DQ. Long noncoding RNAs: novel insights into gastric cancer. Cancer Lett. 2015;356:357-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 166] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 11. | Guo X, Xia J, Yan J. Promoter methylated microRNAs: potential therapeutic targets in gastric cancer. Mol Med Rep. 2015;11:759-765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | Tsujiura M, Ichikawa D, Komatsu S, Shiozaki A, Takeshita H, Kosuga T, Konishi H, Morimura R, Deguchi K, Fujiwara H. Circulating microRNAs in plasma of patients with gastric cancers. Br J Cancer. 2010;102:1174-1179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 449] [Cited by in RCA: 509] [Article Influence: 33.9] [Reference Citation Analysis (0)] |
| 13. | Matsukawa Y, Semba S, Kato H, Ito A, Yanagihara K, Yokozaki H. Expression of the enhancer of zeste homolog 2 is correlated with poor prognosis in human gastric cancer. Cancer Sci. 2006;97:484-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 176] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 14. | Zhao ZS, Wang YY, Chu YQ, Ye ZY, Tao HQ. SPARC is associated with gastric cancer progression and poor survival of patients. Clin Cancer Res. 2010;16:260-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 108] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 15. | Hou F, Yuan W, Huang J, Qian L, Chen Z, Ge J, Wu S, Chen J, Wang J, Chen Z. Overexpression of EphA2 correlates with epithelial-mesenchymal transition-related proteins in gastric cancer and their prognostic importance for postoperative patients. Med Oncol. 2012;29:2691-2700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 41] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 16. | Huang J, Xiao D, Li G, Ma J, Chen P, Yuan W, Hou F, Ge J, Zhong M, Tang Y. EphA2 promotes epithelial-mesenchymal transition through the Wnt/β-catenin pathway in gastric cancer cells. Oncogene. 2014;33:2737-2747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 172] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 17. | Su JL, Yang CY, Shih JY, Wei LH, Hsieh CY, Jeng YM, Wang MY, Yang PC, Kuo ML. Knockdown of contactin-1 expression suppresses invasion and metastasis of lung adenocarcinoma. Cancer Res. 2006;66:2553-2561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 56] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 18. | Yu JW, Wu SH, Lu RQ, Wu JG, Ni XC, Zhou GC, Jiang HG, Zheng LH, Li XQ, Du GY. Expression and significances of contactin-1 in human gastric cancer. Gastroenterol Res Pract. 2013;2013:210205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 19. | Bizzoca A, Corsi P, Gennarini G. The mouse F3/contactin glycoprotein: structural features, functional properties and developmental significance of its regulated expression. Cell Adh Migr. 2009;3:53-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 20. | Bouyain S, Watkins DJ. The protein tyrosine phosphatases PTPRZ and PTPRG bind to distinct members of the contactin family of neural recognition molecules. Proc Natl Acad Sci USA. 2010;107:2443-2448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 104] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 21. | Shimoda Y, Watanabe K. Contactins: emerging key roles in the development and function of the nervous system. Cell Adh Migr. 2009;3:64-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 128] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 22. | Berglund EO, Ranscht B. Molecular cloning and in situ localization of the human contactin gene (CNTN1) on chromosome 12q11-q12. Genomics. 1994;21:571-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 63] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 23. | Bizzoca A, Corsi P, Polizzi A, Pinto MF, Xenaki D, Furley AJ, Gennarini G. F3/Contactin acts as a modulator of neurogenesis during cerebral cortex development. Dev Biol. 2012;365:133-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 24. | Bizzoca A, Virgintino D, Lorusso L, Buttiglione M, Yoshida L, Polizzi A, Tattoli M, Cagiano R, Rossi F, Kozlov S. Transgenic mice expressing F3/contactin from the TAG-1 promoter exhibit developmentally regulated changes in the differentiation of cerebellar neurons. Development. 2003;130:29-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 71] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 25. | Çolakoğlu G, Bergstrom-Tyrberg U, Berglund EO, Ranscht B. Contactin-1 regulates myelination and nodal/paranodal domain organization in the central nervous system. Proc Natl Acad Sci USA. 2014;111:E394-E403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 89] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 26. | Dityatev A, Bukalo O, Schachner M. Modulation of synaptic transmission and plasticity by cell adhesion and repulsion molecules. Neuron Glia Biol. 2008;4:197-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 70] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 27. | Hu QD, Ang BT, Karsak M, Hu WP, Cui XY, Duka T, Takeda Y, Chia W, Sankar N, Ng YK. F3/contactin acts as a functional ligand for Notch during oligodendrocyte maturation. Cell. 2003;115:163-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 289] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 28. | Peles E, Nativ M, Campbell PL, Sakurai T, Martinez R, Lev S, Clary DO, Schilling J, Barnea G, Plowman GD. The carbonic anhydrase domain of receptor tyrosine phosphatase beta is a functional ligand for the axonal cell recognition molecule contactin. Cell. 1995;82:251-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 315] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 29. | Sakurai T, Lustig M, Nativ M, Hemperly JJ, Schlessinger J, Peles E, Grumet M. Induction of neurite outgrowth through contactin and Nr-CAM by extracellular regions of glial receptor tyrosine phosphatase beta. J Cell Biol. 1997;136:907-918. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 145] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 30. | Parent AS, Mungenast AE, Lomniczi A, Sandau US, Peles E, Bosch MA, Rønnekleiv OK, Ojeda SR. A contactin-receptor-like protein tyrosine phosphatase beta complex mediates adhesive communication between astroglial cells and gonadotrophin-releasing hormone neurones. J Neuroendocrinol. 2007;19:847-859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 31. | Lamprianou S, Chatzopoulou E, Thomas JL, Bouyain S, Harroch S. A complex between contactin-1 and the protein tyrosine phosphatase PTPRZ controls the development of oligodendrocyte precursor cells. Proc Natl Acad Sci USA. 2011;108:17498-17503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 32. | Zeng L, D’Alessandri L, Kalousek MB, Vaughan L, Pallen CJ. Protein tyrosine phosphatase alpha (PTPalpha) and contactin form a novel neuronal receptor complex linked to the intracellular tyrosine kinase fyn. J Cell Biol. 1999;147:707-714. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 93] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 33. | Umemori H, Sato S, Yagi T, Aizawa S, Yamamoto T. Initial events of myelination involve Fyn tyrosine kinase signalling. Nature. 1994;367:572-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 294] [Cited by in RCA: 315] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 34. | Shimazaki K, Hosoya H, Takeda Y, Kobayashi S, Watanabe K. Age-related decline of F3/contactin in rat hippocampus. Neurosci Lett. 1998;245:117-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 35. | Hosoya H, Shimazaki K, Kobayashi S, Takahashi H, Shirasawa T, Takenawa T, Watanabe K. Developmental expression of the neural adhesion molecule F3 in the rat brain. Neurosci Lett. 1995;186:83-86. [PubMed] |
| 36. | Puzzo D, Bizzoca A, Privitera L, Furnari D, Giunta S, Girolamo F, Pinto M, Gennarini G, Palmeri A. F3/Contactin promotes hippocampal neurogenesis, synaptic plasticity, and memory in adult mice. Hippocampus. 2013;23:1367-1382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 48] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 37. | Milanese C, Fiumara F, Bizzoca A, Giachello C, Leitinger G, Gennarini G, Montarolo PG, Ghirardi M. F3/contactin-related proteins in Helix pomatia nervous tissue (HCRPs): distribution and function in neurite growth and neurotransmitter release. J Neurosci Res. 2008;86:821-831. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 38. | Burbach JP, van der Zwaag B. Contact in the genetics of autism and schizophrenia. Trends Neurosci. 2009;32:69-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 111] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 39. | Liu P, Chen S, Wu W, Liu B, Shen W, Wang F, He X, Zhang S. Contactin-1 (CNTN-1) overexpression is correlated with advanced clinical stage and lymph node metastasis in oesophageal squamous cell carcinomas. Jpn J Clin Oncol. 2012;42:612-618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 40. | Qin XJ, Dai DJ, Gao ZG, Huan JL, Zhu L. Effect of lentivirus-mediated shRNA targeting VEGFR-3 on proliferation, apoptosis and invasion of gastric cancer cells. Int J Mol Med. 2011;28:761-768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 41. | Su JL, Yang PC, Shih JY, Yang CY, Wei LH, Hsieh CY, Chou CH, Jeng YM, Wang MY, Chang KJ. The VEGF-C/Flt-4 axis promotes invasion and metastasis of cancer cells. Cancer Cell. 2006;9:209-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 320] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 42. | Wu HM, Cao W, Ye D, Ren GX, Wu YN, Guo W. Contactin 1 (CNTN1) expression associates with regional lymph node metastasis and is a novel predictor of prognosis in patients with oral squamous cell carcinoma. Mol Med Rep. 2012;6:265-270. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 43. | Tsai KH, Hsien HH, Chen LM, Ting WJ, Yang YS, Kuo CH, Tsai CH, Tsai FJ, Tsai HJ, Huang CY. Rhubarb inhibits hepatocellular carcinoma cell metastasis via GSK-3-β activation to enhance protein degradation and attenuate nuclear translocation of β-catenin. Food Chem. 2013;138:278-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 44. | Vinarskaja A, Yamanaka M, Ingenwerth M, Schulz WA. DNA Methylation and the HOXC6 Paradox in Prostate Cancer. Cancers (Basel). 2011;3:3714-3725. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 45. | Prag S, Lepekhin EA, Kolkova K, Hartmann-Petersen R, Kawa A, Walmod PS, Belman V, Gallagher HC, Berezin V, Bock E. NCAM regulates cell motility. J Cell Sci. 2002;115:283-292. [PubMed] |
| 46. | Lehembre F, Yilmaz M, Wicki A, Schomber T, Strittmatter K, Ziegler D, Kren A, Went P, Derksen PW, Berns A. NCAM-induced focal adhesion assembly: a functional switch upon loss of E-cadherin. EMBO J. 2008;27:2603-2615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 143] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 47. | Hung YH, Hung WC. 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) enhances invasiveness of lung cancer cells by up-regulating contactin-1 via the alpha7 nicotinic acetylcholine receptor/ERK signaling pathway. Chem Biol Interact. 2009;179:154-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 48. | Zang ZJ, Cutcutache I, Poon SL, Zhang SL, McPherson JR, Tao J, Rajasegaran V, Heng HL, Deng N, Gan A. Exome sequencing of gastric adenocarcinoma identifies recurrent somatic mutations in cell adhesion and chromatin remodeling genes. Nat Genet. 2012;44:570-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 512] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 49. | Chen H, Yu JW, Wu JG, Wang SL, Jiang BJ. Significances of contactin-1 expression in human gastric cancer and knockdown of contactin-1 expression inhibits invasion and metastasis of MKN45 gastric cancer cells. J Cancer Res Clin Oncol. 2015;Epub ahead of print. [PubMed] |
| 50. | Liu P, Zhou J, Zhu H, Xie L, Wang F, Liu B, Shen W, Ye W, Xiang B, Zhu X. VEGF-C promotes the development of esophageal cancer via regulating CNTN-1 expression. Cytokine. 2011;55:8-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 51. | Mauerer A, Roesch A, Hafner C, Stempfl T, Wild P, Meyer S, Landthaler M, Vogt T. Identification of new genes associated with melanoma. Exp Dermatol. 2011;20:502-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 52. | Mhawech-Fauceglia P, Wang D, Kesterson J, Clark K, Monhollen L, Odunsi K, Lele S, Liu S. Microarray analysis reveals distinct gene expression profiles among different tumor histology, stage and disease outcomes in endometrial adenocarcinoma. PLoS One. 2010;5:e15415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 53. | Eckerich C, Zapf S, Ulbricht U, Müller S, Fillbrandt R, Westphal M, Lamszus K. Contactin is expressed in human astrocytic gliomas and mediates repulsive effects. Glia. 2006;53:1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 54. | Nord H, Hartmann C, Andersson R, Menzel U, Pfeifer S, Piotrowski A, Bogdan A, Kloc W, Sandgren J, Olofsson T. Characterization of novel and complex genomic aberrations in glioblastoma using a 32K BAC array. Neuro Oncol. 2009;11:803-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 55. | Colussi D, Brandi G, Bazzoli F, Ricciardiello L. Molecular pathways involved in colorectal cancer: implications for disease behavior and prevention. Int J Mol Sci. 2013;14:16365-16385. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 279] [Cited by in RCA: 331] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 56. | Bardhan K, Liu K. Epigenetics and colorectal cancer pathogenesis. Cancers (Basel). 2013;5:676-713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 152] [Cited by in RCA: 188] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 57. | Zoratto F, Rossi L, Verrico M, Papa A, Basso E, Zullo A, Tomao L, Romiti A, Lo Russo G, Tomao S. Focus on genetic and epigenetic events of colorectal cancer pathogenesis: implications for molecular diagnosis. Tumour Biol. 2014;35:6195-6206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 88] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 58. | Ogino S, Lochhead P, Chan AT, Nishihara R, Cho E, Wolpin BM, Meyerhardt JA, Meissner A, Schernhammer ES, Fuchs CS. Molecular pathological epidemiology of epigenetics: emerging integrative science to analyze environment, host, and disease. Mod Pathol. 2013;26:465-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 167] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 59. | Huang J, Asawa T, Takato T, Sakai R. Cooperative roles of Fyn and cortactin in cell migration of metastatic murine melanoma. J Biol Chem. 2003;278:48367-48376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 54] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 60. | Cooper JA. The role of actin polymerization in cell motility. Annu Rev Physiol. 1991;53:585-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 233] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 61. | Pinnix CC, Lee JT, Liu ZJ, McDaid R, Balint K, Beverly LJ, Brafford PA, Xiao M, Himes B, Zabierowski SE. Active Notch1 confers a transformed phenotype to primary human melanocytes. Cancer Res. 2009;69:5312-5320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 94] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 62. | Peng Z, Wang CX, Fang EH, Wang GB, Tong Q. Role of epithelial-mesenchymal transition in gastric cancer initiation and progression. World J Gastroenterol. 2014;20:5403-5410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 139] [Cited by in RCA: 166] [Article Influence: 15.1] [Reference Citation Analysis (1)] |
| 63. | Yan J, Wong N, Hung C, Chen WX, Tang D. Contactin-1 reduces E-cadherin expression via activating AKT in lung cancer. PLoS One. 2013;8:e65463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 64. | Onder TT, Gupta PB, Mani SA, Yang J, Lander ES, Weinberg RA. Loss of E-cadherin promotes metastasis via multiple downstream transcriptional pathways. Cancer Res. 2008;68:3645-3654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1038] [Cited by in RCA: 1158] [Article Influence: 68.1] [Reference Citation Analysis (0)] |
| 65. | Boral D, Nie D. Cancer stem cells and niche mircoenvironments. Front Biosci (Elite Ed). 2012;4:2502-2514. [PubMed] |
| 66. | Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J, Minden M, Paterson B, Caligiuri MA, Dick JE. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature. 1994;367:645-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3316] [Cited by in RCA: 3388] [Article Influence: 109.3] [Reference Citation Analysis (0)] |
| 67. | Yu Z, Pestell TG, Lisanti MP, Pestell RG. Cancer stem cells. Int J Biochem Cell Biol. 2012;44:2144-2151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 396] [Cited by in RCA: 498] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 68. | Ailles LE, Weissman IL. Cancer stem cells in solid tumors. Curr Opin Biotechnol. 2007;18:460-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 363] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 69. | Takaishi S, Okumura T, Tu S, Wang SS, Shibata W, Vigneshwaran R, Gordon SA, Shimada Y, Wang TC. Identification of gastric cancer stem cells using the cell surface marker CD44. Stem Cells. 2009;27:1006-1020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 820] [Cited by in RCA: 817] [Article Influence: 51.1] [Reference Citation Analysis (0)] |
| 70. | Zhang C, Li C, He F, Cai Y, Yang H. Identification of CD44+CD24+ gastric cancer stem cells. J Cancer Res Clin Oncol. 2011;137:1679-1686. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 160] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 71. | Barcellos-Hoff MH, Lyden D, Wang TC. The evolution of the cancer niche during multistage carcinogenesis. Nat Rev Cancer. 2013;13:511-518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 193] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 72. | Bendas G, Borsig L. Cancer cell adhesion and metastasis: selectins, integrins, and the inhibitory potential of heparins. Int J Cell Biol. 2012;2012:676731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 287] [Cited by in RCA: 340] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 73. | Gradilone A, Raimondi C, Nicolazzo C, Petracca A, Gandini O, Vincenzi B, Naso G, Aglianò AM, Cortesi E, Gazzaniga P. Circulating tumour cells lacking cytokeratin in breast cancer: the importance of being mesenchymal. J Cell Mol Med. 2011;15:1066-1070. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 74. | Floor S, van Staveren WC, Larsimont D, Dumont JE, Maenhaut C. Cancer cells in epithelial-to-mesenchymal transition and tumor-propagating-cancer stem cells: distinct, overlapping or same populations. Oncogene. 2011;30:4609-4621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 148] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 75. | Scheel C, Weinberg RA. Phenotypic plasticity and epithelial-mesenchymal transitions in cancer and normal stem cells? Int J Cancer. 2011;129:2310-2314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 171] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 76. | Yang J, Weinberg RA. Epithelial-mesenchymal transition: at the crossroads of development and tumor metastasis. Dev Cell. 2008;14:818-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2157] [Cited by in RCA: 2338] [Article Influence: 137.5] [Reference Citation Analysis (0)] |
| 77. | Tinhofer I, Saki M, Niehr F, Keilholz U, Budach V. Cancer stem cell characteristics of circulating tumor cells. Int J Radiat Biol. 2014;90:622-627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 78. | Książkiewicz M, Markiewicz A, Zaczek AJ. Epithelial-mesenchymal transition: a hallmark in metastasis formation linking circulating tumor cells and cancer stem cells. Pathobiology. 2012;79:195-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 143] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 79. | Illert B, Fein M, Otto C, Cording F, Stehle D, Thiede A, Timmermann W. Disseminated tumor cells in the blood of patients with gastric cancer are an independent predictive marker of poor prognosis. Scand J Gastroenterol. 2005;40:843-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 80. | Zhang Y, Li J, Cao L, Xu W, Yin Z. Circulating tumor cells in hepatocellular carcinoma: detection techniques, clinical implications, and future perspectives. Semin Oncol. 2012;39:449-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 81. | Li M, Zhang B, Zhang Z, Liu X, Qi X, Zhao J, Jiang Y, Zhai H, Ji Y, Luo D. Stem cell-like circulating tumor cells indicate poor prognosis in gastric cancer. Biomed Res Int. 2014;2014:981261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 55] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 82. | Yu JW, Wu JG, Tajima Y, Li XQ, Du GY, Zheng LH, Zhang B, Ni XC, Jiang BJ. Study on lymph node metastasis correlated to lymphangiogenesis, lymphatic vessel invasion, and lymph node micrometastasis in gastric cancer. J Surg Res. 2011;168:188-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 83. | Ezoe Y, Muto M, Uedo N, Doyama H, Yao K, Oda I, Kaneko K, Kawahara Y, Yokoi C, Sugiura Y. Magnifying narrowband imaging is more accurate than conventional white-light imaging in diagnosis of gastric mucosal cancer. Gastroenterology. 2011;141:2017-2025.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 283] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 84. | Nagahama T, Yao K, Maki S, Yasaka M, Takaki Y, Matsui T, Tanabe H, Iwashita A, Ota A. Usefulness of magnifying endoscopy with narrow-band imaging for determining the horizontal extent of early gastric cancer when there is an unclear margin by chromoendoscopy (with video). Gastrointest Endosc. 2011;74:1259-1267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 145] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 85. | Oka S, Tanaka S, Kaneko I, Mouri R, Hirata M, Kawamura T, Yoshihara M, Chayama K. Advantage of endoscopic submucosal dissection compared with EMR for early gastric cancer. Gastrointest Endosc. 2006;64:877-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 487] [Cited by in RCA: 527] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 86. | Orditura M, Galizia G, Sforza V, Gambardella V, Fabozzi A, Laterza MM, Andreozzi F, Ventriglia J, Savastano B, Mabilia A. Treatment of gastric cancer. World J Gastroenterol. 2014;20:1635-1649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 461] [Cited by in RCA: 483] [Article Influence: 43.9] [Reference Citation Analysis (5)] |
| 87. | Pasini F, Fraccon AP, DE Manzoni G. The role of chemotherapy in metastatic gastric cancer. Anticancer Res. 2011;31:3543-3554. [PubMed] |
| 88. | Coratti A, Annecchiarico M, Di Marino M, Gentile E, Coratti F, Giulianotti PC. Robot-assisted gastrectomy for gastric cancer: current status and technical considerations. World J Surg. 2013;37:2771-2781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 89. | Uyama I, Suda K, Satoh S. Laparoscopic surgery for advanced gastric cancer: current status and future perspectives. J Gastric Cancer. 2013;13:19-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 90. | Smyth EC, Cunningham D. Targeted therapy for gastric cancer. Curr Treat Options Oncol. 2012;13:377-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 54] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 91. | Liu XE, Sun XD, Wu JM. Expression and significance of VEGF-C and FLT-4 in gastric cancer. World J Gastroenterol. 2004;10:352-355. [PubMed] |