Published online Jan 21, 2015. doi: 10.3748/wjg.v21.i3.944
Peer-review started: June 16, 2014
First decision: July 21, 2014
Revised: August 11, 2014
Accepted: September 18, 2014
Article in press: September 19, 2014
Published online: January 21, 2015
Processing time: 218 Days and 7.5 Hours
AIM: To evaluate the efficacy of centralized culture and possible influencing factors.
METHODS: From January 2010 to July 2012, 66452 patients with suspected Helicobacter pylori (H. pylori) infection from 26 hospitals in Zhejiang and Jiangsu Provinces in China underwent gastrointestinal endoscopy. Gastric mucosal biopsies were taken from the antrum for culture. These biopsies were transported under natural environmental temperature to the central laboratory in Hangzhou city and divided into three groups based on their transport time: 5, 24 and 48 h. The culture results were reported after 72 h and the positive culture rates were analyzed by a χ2 test. An additional 5736 biopsies from H. pylori-positive patients (5646 rapid urease test-positive and 90 14C-urease breath test-positive) were also cultured for quality control in the central laboratory setting.
RESULTS: The positive culture rate was 31.66% (21036/66452) for the patient samples and 71.72% (4114/5736) for the H. pylori-positive quality control specimens. In the 5 h transport group, the positive culture rate was 30.99% (3865/12471), and 32.84% (14960/45553) in the 24 h transport group. In contrast, the positive culture rate declined significantly in the 48 h transport group (26.25%; P < 0.001). During transportation, the average natural temperature increased from 4.67 to 29.14 °C, while the positive culture rate declined from 36.67% (1462/3987) to 24.12% (1799/7459). When the temperature exceeded 24 °C, the positive culture rate decreased significantly, especially in the 48 h transport group (23.17%).
CONCLUSION: Transportation of specimens within 24 h and below 24 °C is reasonable and acceptable for centralized culture of multicenter H. pylori samples.
Core tip: This is the first large-scale study on the centralized culture of Helicobacter pylori in a large number of clinical samples from multiple centers. The efficacy of centralized culture and possible influencing factors were evaluated. The results confirm the feasibility of establishing a culture center for individualized medical use. The findings of this study can be promisingly applied in clinical and public health practice.
-
Citation: Gong YN, Li YM, Yang NM, Li HZ, Guo F, Lin L, Wang QY, Zhang JK, Ji ZZ, Mao JB, Mao JL, Shi ZC, Tang WH, Zhu XJ, Shao W, Zhang XF, Wang XH, Tong YF, Jiang MZ, Chen GL, Wang ZY, Tu HM, Jiang GF, Wu JS, Chen XP, Ding QL, Ouyang H, Jin FZ, Xu YL, Zhang JZ. Centralized isolation of
Helicobacter pylori from multiple centers and transport condition influences. World J Gastroenterol 2015; 21(3): 944-952 - URL: https://www.wjgnet.com/1007-9327/full/v21/i3/944.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i3.944
Helicobacter pylori (H. pylori), one of the most common human pathogens, can lead to gastric ulcers, gastritis, gastric cancer, and mucosa-associated lymphoid tumors[1]. According to the Maastricht IV and Chinese Consensus Report, two antibiotics combined with one proton-pump inhibitor are recommended as standard first-line treatment to eradicate H. pylori[2,3]. However, in recent years, the success rates have declined below 80% in most European and Asian countries[4-6]. There are many reasons accounting for eradication failure, such as poor patient compliance, low gastric pH, and resistant bacteria. In fact, the main reason for the decline is the increasing H. pylori resistance to the antibiotics used[7,8]. In China, the resistance to clarithromycin, a key antibiotic in the triple therapy, has reached above 20% and should not be used in anti-H.pylori therapy without a susceptibility test[2,9-11].
Accumulated evidence suggests that culture-susceptibility tests may improve the eradication rate of H. pylori[12-17]. However, H. pylori is a rather fastidious bacterium at culture, especially when a low bacterial load is present[18]. The ideal situation for culture-susceptibility test is not available in many clinical settings, since most endoscopic units do not have direct access to a microbiology laboratory. Therefore, gastric biopsy specimens should be transported to a central laboratory for H. pylori culture. During transportation, time and temperature could influence the survivability of H. pylori and these issues are still in debate[19-23]. Some investigators emphasized the need for rapid transport at a low temperature[21]. Others demonstrated that H. pylori could survive at room temperature for 24 h without loss of the ability to recover[22]. The transport of samples at natural temperature is required for a routine clinical application system. In order to assess the factors, transportation[24] and isolation tests of thousands of H. pylori strains were performed in our lab, but the complexity of large-scale clinical application could not be accurately represented. The feasibility of centralized isolation of H. pylori in a large number of clinical samples from multiple centers and influencing factors need to be tested for actual practice.
A central laboratory for H. pylori isolation was set up in Zhiyuan Medical Inspection Institute Co., Ltd., in Hangzhou city, which has provided a personalized treatment strategy for H. pylori eradication in recent years. In order to evaluate the efficacy of centralized culture and possible influencing factors, we conducted research in the central laboratory to analyze positive culture rates of a large number of clinical samples collected from 26 hospitals in nine cities.
The sampling was conducted in Zhejiang Province and Jiangsu Province, China. Between January 2010 and July 2012, consecutive participants with suspected H. pylori infection underwent gastrointestinal endoscopy. The gastric mucosal biopsies were taken from the greater curvature of gastric antrum using sterile disposable biopsy forceps. Additionally, in order to assess the positive rate of H. pylori culture, specimens from H. pylori-positive patients (5646 rapid urease test-positive and 90 14C-urease breath test-positive) were also collected as a control. The H. pylori-positive samples were all collected from Wenzhou city, including The First Affiliated Hospital of Wenzhou Medical College and The First People’s Hospital of Pingyang.
This study was approved by the Ethics Committee of National Institute for Communicable Disease Control and Prevention, Chinese Center for Disease Control and Prevention and the Ethics Committee of Zhiyuan Medical Inspection Institute Co., Ltd., and written informed consent was obtained from all patients.
The gastric mucosal sample from each patient was stored in sterile tube that contained 1 mL brain-heart infusion with 20% glycerol and kept under 4 °C before transportation. All the fresh biopsy specimens were transported at external temperature to Zhiyuan Medical Inspection Institute Co., Ltd., for centralized isolation. The 66452 specimens were divided into three independent groups based on transport time: 5, 24 and 48 h. The first group (5 h; n = 12471) was collected from Hangzhou city between 9 am and 12 pm and cultured at 2 pm on the same day. The second group (24 h; n = 45553) was collected from other cities hundreds of kilometers away from Hangzhou between 9 am and 12 pm and sent to Hangzhou for culture at 9 am the next day. The third group (48 h; n = 8428) were collected from Jinhua city and transported to Hangzhou for culture 48 h later because of the limitations of local hospital conditions. The H. pylori-positive control samples were transported the same as the 24 h group.
The samples were ground/broken and cultivated on Columbia agar plates (Oxoid of Thermo Fisher Scientific Inc., Waltham, MA, United States) supplemented with 5% defibrinated sheep blood, 3 μg/mL synergist, 2.5 μg/mL vancomycin, 2 μg/mL amphotericin B, and 2 μg/mL bacillosporin B under microaerophilic conditions (5% O2, 10% CO2, 85% N2) for 72 h. Translucent colonies (0.5-2.0 mm) from the original agar plates were selected for gram staining and urease, oxidase and catalase tests. Colonies with curved gram-negative rods resembling Helicobacter spp and positive in three enzyme tests were identified as H. pylori.
The information concerning daily measured maximum, minimum and average temperatures in each city related with this study in Zhejiang Province from January 1, 2010 to December 31, 2012 was supplied by the Meteorological Bureau of Zhejiang Province. The average daily temperature was the average value of temperatures at four time points each day (2 am, 8 am, 2 pm and 8 pm). In our investigation, we used the regional level meteorological data from six cities (Hangzhou, Jiaxing, Jinhua, Taizhou, Wenzhou, and Zhoushan) to calculate the monthly maximum, minimum and average temperatures and draw the temperature change curves.
The results of isolation and identification of H. pylori for all specimens were reported 72 h after culture and all calculations were performed using SPSS 18.0 software (SPSS Inc., Chicago, IL, United States). The positive culture rates were assessed with a χ2 test. A P≤ 0.05 was considered as statistically significant.
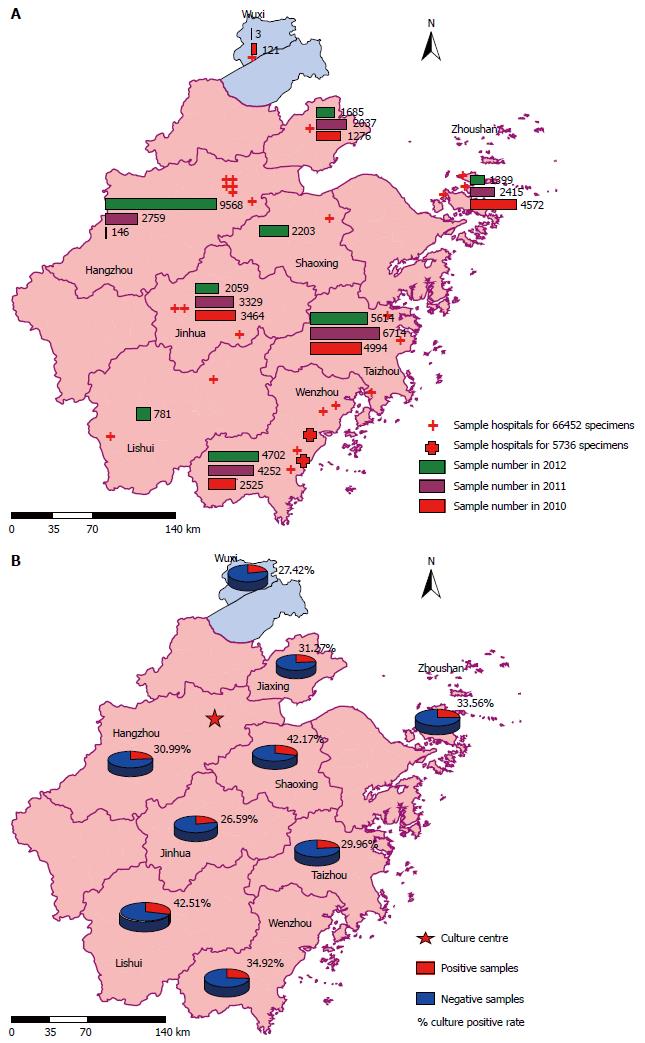
The random 66452 samples were collected from 24 hospitals in nine cities: Hangzhou (n = 6), Jiaxing (n = 1), Jinhua (n = 3), Shaoxing (n = 1), Taizhou (n = 3), Wenzhou (n = 4), Zhoushan (n = 3), Lishui (n = 2) and Wuxi (n = 1). The 5736 H. pylori-positive specimens were collected from another two hospitals in Wenzhou city. Eight of the 26 sampling hospitals are Class 2A hospitals, which are primarily county-level hospitals providing medical service to the communities. The other hospitals belong to Class 3A, which are regional hospitals and possess the highest medical level in Chinese hospital grading. The distribution and numbers of samples in each year and city are shown in Figure 1A.
Of the 66452 specimens, 31.66% (21036/66452) were H. pylori-positive. The positive culture rates of specimens in 2010, 2011 and 2012 were 28.64% (4897/17098), 30.28% (6513/21509) and 34.57% (9626/27845), respectively. The differences showed statistical significance (P < 0.001). The positive culture rate for Class 2A hospitals was 31.96% (8103/25350) and that of Class 3A hospitals was 31.47% (12933/41102).
The positive culture rates of samples from nine cities varied from 26.59% (2354/8852) in Jinhua city to 42.51% (332/781) in Lishui city (Figure 1B). The positive culture rate of Hangzhou city, which was the culture center, was 30.99% (3866/12473).
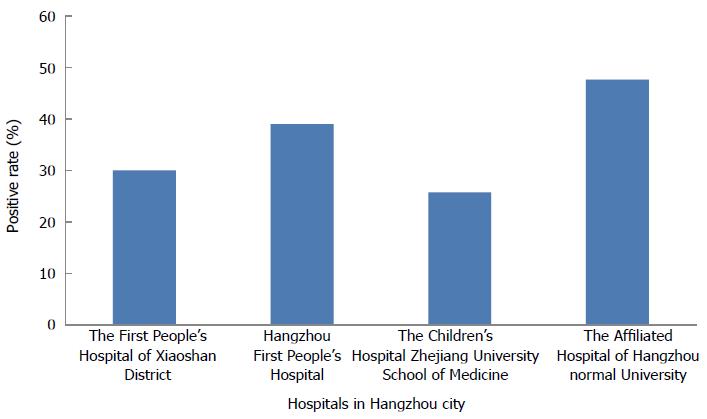
Even in the same city, the positive culture rates of different sampling hospitals were different. The culture positive rates from four hospitals in Hangzhou city were 29.97% (3316/11064), 38.95% (298/705), 25.73% (79/307) and 47.66% (61/128) (Figure 2).
The positive culture rates from the 5, 24, and 48 h transport groups were 30.99% (3865/12471), 32.84% (14960/45553) and 26.25% (2211/8428), respectively. The positive culture rate of the 48 h transport group was significantly lower than the other two groups (P < 0.001). The positive culture rate from the 48 h group was significantly lower than the 24 h group in samples from the same city, (26.25% vs 33.73%, P < 0.001).
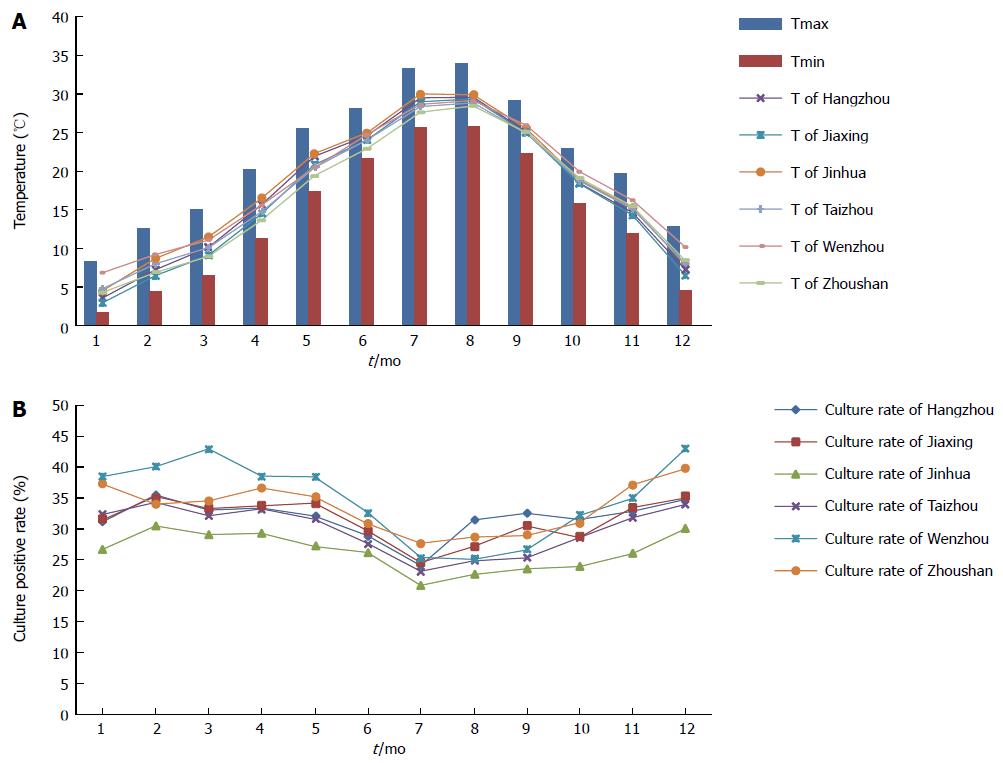
The average natural temperature in Zhejiang province varied within one year from 4.67 to 29.14 °C. The average temperatures in January, May, June and July were 4.66, 20.85, 24.24 and 28.85 °C, respectively. The change curves of average temperatures were similar in six cities, and the monthly average temperature, average maximum and minimum temperatures of the six cities during this study are shown in Figure 3A.
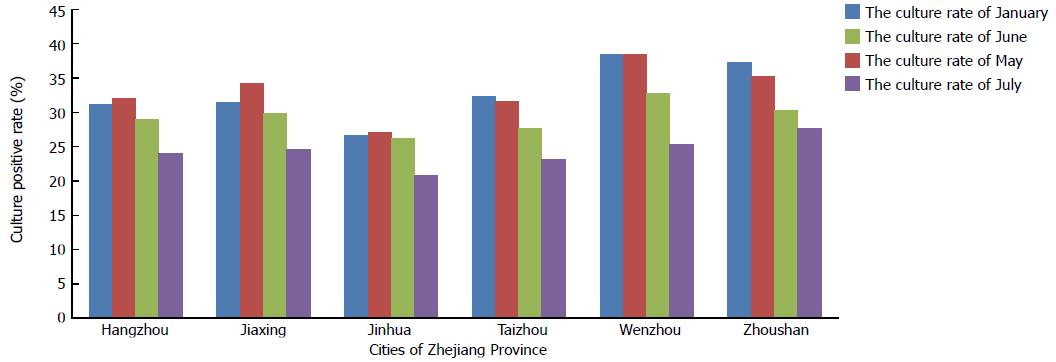
As the temperature increased, the total positive culture rate declined from 36.67% (1462/3987) in December to 24.12% (1799/7459) in July. Although the average temperature elevated significantly from January to May, the positive culture rates were similar. When the average temperature exceeded 24 °C in June and July, the positive culture rates declined significantly (P < 0.001), with the lowest positive culture rate in July (24.12%; 1799/7459). This phenomenon was particularly evident in Wenzhou city whose positive culture rates changed from 38.44% (316/822) in January to 25.38% (389/1533) in July. The positive culture rates in the other months changed slightly even when the temperatures changed significantly (Figure 3B). Positive culture rates of six cities in Zhejiang Province in January, May, June and July are shown in Figure 4.
Of the H. pylori-positive specimens, the isolation of H. pylori was successful in 71.72% (4114/5736) of cases, demonstrating the positive predictive value of H. pylori isolation in the central cultural platform.
Isolation of all samples was conducted in the same laboratory to eliminate the intrinsic variability from multiple laboratories. The advantage of our study is that it contains largest sample size to date, involving a wide range of demographic characteristics, geographic areas and hospitals from various medical levels (nearly all the gastrointestinal endoscopic tests were done in Class 2A and 3A hospitals in China). Compared with a previous study[25], the limitations of small sample size and collection of data from different laboratories were certainly avoided in this study, and more reliable information for the centralized culture of H. pylori was provided.
Previous studies reported H. pylori-positive isolation rates ranging from 75% to 94%[25,26]. Considering the differences between the H. pylori cultures used for clinical study and those for individual medical service, as well as the amount of samples and the time-efficient characteristics (a limited time of about seven days for clinical report was usually set in the latter), the positive culture rate of 71.72% found in this study is acceptable.
In China, the total infectious rate of H. pylori was relatively high, reaching 40%-60% in adults. In this study, the positive culture rate of the suspected H. pylori infection group was 31.66%. Combined with the culture rate of positive control samples, a 44% infectious rate was calculated, in agreement with the reported data, indicating the positive culture rate of random samples was reasonable. In addition, the cost of culture and sensitivity tests for six antibiotics is approximately $18, whereas the urease breath test costs $29 in Zhejiang province. The random samples were collected without any H. pylori detection, leading to a lower cost (51.25% reduced) and economic burden in this population compared with the detection strategy.
Factors influencing the isolation rates during transportation have been discussed by previous studies[18,23,27,28]. Use of dry ice and need for storage at a constant temperature of 4 °C were recommended, but they cannot be put into actual practice in the H. pylori isolation for individualized medical need in a large number of cases because dry ice is too costly and constant 4 °C preservation condition is hard to maintain. Despite the appearance as a necessary condition for the optimal diagnosis of Helicobacter infections via bacteriologic methods in a small-sample study[29], transport on ice was not recommended, as repeated freezing and thawing is more harmful for the successful isolation of H. pylori. Thus, in our study, transportation of biopsy specimens under ambient temperature was performed. Our results indicate that the samples were maintained without influencing the culture rates within 24 h, especially when the mean temperature was lower than 24 °C. A prolonged or delayed transport time and higher natural temperature (daily average temperature above 24 °C) could significantly influence the positive culture rates, probably due to contamination and overgrowth of other bacteria. Based on these factors, the proper position of the culture center should be taken into account, and measures to lower ambient temperature should be taken in summer.
Besides the two strong influencing factors, other factors such as sampling and operation levels should also be considered, since variability in the positive culture rates was observed among the four hospitals in Hangzhou city that shared the same sample population and transport conditions. In one study[30], low isolation rates were obtained from failed eradication therapy patients. The patients from Class 2A and 3A hospitals might have different backgrounds of antibiotics use. In this study, a novel design was to compare the culture rates between Class 2A and Class 3A hospitals. The results show no obvious differences, indicating the medical levels of hospitals can be ignored. In addition, the positive culture rates significantly increased over three years, probably due to the improved sampling skills and experience of the laboratory personnel. This indicates that the positive culture rate may increase, even within an H. pylori-isolation platform for individualized medical need in a large number of cases.
There were also some limitations in this study. First, we only compared the positive culture rates of four hospitals in Hangzhou city. The other two hospitals were excluded, as the number of samples was small and the sampling was not consecutive. Second, we only selected six cities to assess the temperature effects on positive culture rates, because the samples collected in Lishui, Wuxi and Shaoxing were missed for some months, and the meteorological data was incomplete. Even with these exclusions, the selected data accounted for the majority and was sufficient to compare these differences. Third, although the H. pylori-positive group was available to assess the culture results, there was no direct comparison or control group in this study for all samples, such as a non-culture based test, and the positive culture rates in this study were considered acceptable.
In general, this was the first large-scale study on centralized culture of H. pylori, confirming the feasibility of establishing a culture center for individualized medical use. We believe the findings of this study can be promisingly applied in clinical and public health practice. The resistance to clarithromycin often poses a challenge in clinical design. For example, as the eradication rate of two couplet therapeutics decreased ten years ago, triple therapy was recommended, thus doctors had to choose quadruple or sequential therapy in failed cases. However, this cycle is likely to continue, and the eradication rate of quadruple or sequential therapies will be reduced to unacceptable levels. Establishment of a personalized treatment strategy may potentially resolve the public health problem and reduce economic burden on the patients and the community, especially in those populations with high resistance.
The centralized culture of Helicobacter pylori (H. pylori) in a large number of clinical samples from multiple centers was established. The efficacy of centralized culture and possible influencing factors was evaluated.
Antibiotic resistance is a worldwide problem that prevents H. pylori eradication. Personalized treatment strategy based on culture and antimicrobial susceptibility tests is one of the most promising ways to solve the problem.
Previous studies have discussed factors influencing H. pylori isolation rates during transportation. This is the first large-scale study on centralized culture of H. pylori, confirming the feasibility of establishing a culture center for individualized medical use.
The findings of this study can be promisingly applied in clinical and public health practice. Establishment of a personalized treatment strategy may potentially resolve the public health problem and reduce economic burden on the patients and the community, especially in those populations with high resistance.
Clarithromycin is one of the core antibiotics of a triple regimen for H. pylori eradication. If the clarithromycin resistance rate of H. pylori increases to 15%-20% in a population, this antibiotic should not be used without a susceptibility test.
In this study, the authors evaluated the efficacy of centralized culture and discussed the possible influencing factors. It reveals that centralized culture of multicenter samples is feasible. The results of this study can be promisingly applied in individualized medical use and clinical H. pylori eradication.
P- Reviewer: Buzas GM, Yula E S- Editor: Qi Y L- Editor: AmEditor E- Editor: Zhang DN
| 1. | Parsonnet J. Helicobacter pylori: the size of the problem. Gut. 1998;43 Suppl 1:S6-S9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 76] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 2. | Malfertheiner P, Megraud F, O’Morain CA, Atherton J, Axon AT, Bazzoli F, Gensini GF, Gisbert JP, Graham DY, Rokkas T. Management of Helicobacter pylori infection--the Maastricht IV/ Florence Consensus Report. Gut. 2012;61:646-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1719] [Cited by in RCA: 1591] [Article Influence: 122.4] [Reference Citation Analysis (5)] |
| 3. | Hu FL, Hu PJ, Liu WZ, De Wang J, Lv NH, Xiao SD, Zhang WD, Cheng H, Xie Y. Third Chinese National Consensus Report on the management of Helicobacter pylori infection. J Dig Dis. 2008;9:178-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 4. | Altintas E, Sezgin O, Ulu O, Aydin O, Camdeviren H. Maastricht II treatment scheme and efficacy of different proton pump inhibitors in eradicating Helicobacter pylori. World J Gastroenterol. 2004;10:1656-1658. [PubMed] |
| 5. | Gumurdulu Y, Serin E, Ozer B, Kayaselcuk F, Ozsahin K, Cosar AM, Gursoy M, Gur G, Yilmaz U, Boyacioglu S. Low eradication rate of Helicobacter pylori with triple 7-14 days and quadriple therapy in Turkey. World J Gastroenterol. 2004;10:668-671. [PubMed] |
| 6. | Suzuki H, Nishizawa T, Hibi T. Helicobacter pylori eradication therapy. Future Microbiol. 2010;5:639-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 7. | Mégraud F, Lamouliatte H. Review article: the treatment of refractory Helicobacter pylori infection. Aliment Pharmacol Ther. 2003;17:1333-1343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 149] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 8. | Mégraud F. H pylori antibiotic resistance: prevalence, importance, and advances in testing. Gut. 2004;53:1374-1384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 607] [Cited by in RCA: 681] [Article Influence: 32.4] [Reference Citation Analysis (2)] |
| 9. | Gao W, Cheng H, Hu F, Li J, Wang L, Yang G, Xu L, Zheng X. The evolution of Helicobacter pylori antibiotics resistance over 10 years in Beijing, China. Helicobacter. 2010;15:460-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 111] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 10. | Su P, Li Y, Li H, Zhang J, Lin L, Wang Q, Guo F, Ji Z, Mao J, Tang W. Antibiotic resistance of Helicobacter pylori isolated in the Southeast Coastal Region of China. Helicobacter. 2013;18:274-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 136] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 11. | Malfertheiner P, Megraud F, O’Morain C, Bazzoli F, El-Omar E, Graham D, Hunt R, Rokkas T, Vakil N, Kuipers EJ. Current concepts in the management of Helicobacter pylori infection: the Maastricht III Consensus Report. Gut. 2007;56:772-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1396] [Cited by in RCA: 1352] [Article Influence: 75.1] [Reference Citation Analysis (1)] |
| 12. | Dore MP, Leandro G, Realdi G, Sepulveda AR, Graham DY. Effect of pretreatment antibiotic resistance to metronidazole and clarithromycin on outcome of Helicobacter pylori therapy: a meta-analytical approach. Dig Dis Sci. 2000;45:68-76. [PubMed] |
| 13. | Realdi G, Dore MP, Piana A, Atzei A, Carta M, Cugia L, Manca A, Are BM, Massarelli G, Mura I. Pretreatment antibiotic resistance in Helicobacter pylori infection: results of three randomized controlled studies. Helicobacter. 1999;4:106-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 74] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 14. | Glupczynski Y, Mégraud F, Lopez-Brea M, Andersen LP. European multicentre survey of in vitro antimicrobial resistance in Helicobacter pylori. Eur J Clin Microbiol Infect Dis. 2001;20:820-823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 194] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 15. | Wang G, Zhao Q, Li S. Study of drug sensitivity test in Helicobacter pylori eradication therapy. J Clin Intern Med. 2008;25:474-477. |
| 16. | Romano M, Iovene MR, Montella F, Vitale LM, De S :imone T, Del Vecchio Blanco C. Pretreatment antimicrobial-susceptibility testing in the eradication of H. pylori infection. Am J Gastroenterol. 2000;95:3317-3318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 17. | Romano M, Marmo R, Cuomo A, De Simone T, Mucherino C, Iovene MR, Montella F, Tufano MA, Del Vecchio Blanco C, Nardone G. Pretreatment antimicrobial susceptibility testing is cost saving in the eradication of Helicobacter pylori. Clin Gastroenterol Hepatol. 2003;1:273-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 64] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 18. | Soltesz V, Zeeberg B, Wadström T. Optimal survival of Helicobacter pylori under various transport conditions. J Clin Microbiol. 1992;30:1453-1456. [PubMed] |
| 19. | Yuen B, Zbinden R, Fried M, Bauerfeind P, Bernardi M. Cultural recovery and determination of antimicrobial susceptibility in Helicobacter pylori by using commercial transport and isolation media. Infection. 2005;33:77-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 20. | Xia HX, Keane CT, O’Morain CA. Determination of the optimal transport system for Helicobacter pylori cultures. J Med Microbiol. 1993;39:334-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 21. | Roosendaal R, Kuipers EJ, Peña AS, de Graaff J. Recovery of Helicobacter pylori from gastric biopsy specimens is not dependent on the transport medium used. J Clin Microbiol. 1995;33:2798-2800. [PubMed] |
| 22. | Heep M, Scheibl K, Degrell A, Lehn N. Transport and storage of fresh and frozen gastric biopsy specimens for optimal recovery of Helicobacter pylori. J Clin Microbiol. 1999;37:3764-3766. [PubMed] |
| 23. | Siu LK, Leung WK, Cheng AF, Sung JY, Ling TK, Ling JM, Ng EK, Lau JY, Chung SC. Evaluation of a selective transport medium for gastric biopsy specimens to be cultured for Helicobacter pylori. J Clin Microbiol. 1998;36:3048-3050. [PubMed] |
| 24. | Xia HX, Keane CT, Chen J, Zhang J, Walsh EJ, Moran AP, Hua JS, Megraud F, O’Morain CA. Transportation of Helicobacter pylori cultures by optimal systems. J Clin Microbiol. 1994;32:3075-3077. [PubMed] |
| 25. | Grove DI, McLeay RA, Byron KE, Koutsouridis G. Isolation of Helicobacter pylori after transport from a regional laboratory of gastric biopsy specimens in saline, Portagerm pylori or cultured on chocolate agar. Pathology. 2001;33:362-364. [PubMed] |
| 26. | Debongnie JC, Delmee M, Mainguet P, Beyaert C, Haot J, Legros G. Cytology: a simple, rapid, sensitive method in the diagnosis of Helicobacter pylori. Am J Gastroenterol. 1992;87:20-23. [PubMed] |
| 27. | van der Hulst RW, Verheul SB, Weel JF, Gerrits Y, ten Kate FJ, Dankert J, Tytgat GN. Effect of specimen collection techniques, transport media, and incubation of cultures on the detection rate of Helicobacter pylori. Eur J Clin Microbiol Infect Dis. 1996;15:211-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 28. | Veenendaal RA, Lichtendahl-Bernards AT, Peña AS, Endtz HP, van Boven CP, Lamers CB. Effect of transport medium and transportation time on culture of Helicobacter pylori from gastric biopsy specimens. J Clin Pathol. 1993;46:561-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 29. | Meunier O, Walter P, Chamouard P, Piemont Y, Monteil H. [Isolation of Helicobacter pylori: necessity of control of transport conditions]. Pathol Biol (Paris). 1997;45:82-85. [PubMed] |
| 30. | Savarino V, Zentilin P, Pivari M, Bisso G, Raffaella Mele M, Bilardi C, Borro P, Dulbecco P, Tessieri L, Mansi C. The impact of antibiotic resistance on the efficacy of three 7-day regimens against Helicobacter pylori. Aliment Pharmacol Ther. 2000;14:893-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 1.6] [Reference Citation Analysis (0)] |