Published online Jun 21, 2015. doi: 10.3748/wjg.v21.i23.7142
Peer-review started: January 28, 2015
First decision: March 10, 2015
Revised: March 21, 2015
Accepted: April 17, 2015
Article in press: April 17, 2015
Published online: June 21, 2015
Processing time: 143 Days and 12.5 Hours
Intestinal Ca2+ absorption is a crucial physiological process for maintaining bone mineralization and Ca2+ homeostasis. It occurs through the transcellular and paracellular pathways. The first route comprises 3 steps: the entrance of Ca2+ across the brush border membranes (BBM) of enterocytes through epithelial Ca2+ channels TRPV6, TRPV5, and Cav1.3; Ca2+ movement from the BBM to the basolateral membranes by binding proteins with high Ca2+ affinity (such as CB9k); and Ca2+ extrusion into the blood. Plasma membrane Ca2+ ATPase (PMCA1b) and sodium calcium exchanger (NCX1) are mainly involved in the exit of Ca2+ from enterocytes. A novel molecule, the 4.1R protein, seems to be a partner of PMCA1b, since both molecules co-localize and interact. The paracellular pathway consists of Ca2+ transport through transmembrane proteins of tight junction structures, such as claudins 2, 12, and 15. There is evidence of crosstalk between the transcellular and paracellular pathways in intestinal Ca2+ transport. When intestinal oxidative stress is triggered, there is a decrease in the expression of several molecules of both pathways that inhibit intestinal Ca2+ absorption. Normalization of redox status in the intestine with drugs such as quercetin, ursodeoxycholic acid, or melatonin return intestinal Ca2+ transport to control values. Calcitriol [1,25(OH)2D3] is the major controlling hormone of intestinal Ca2+ transport. It increases the gene and protein expression of most of the molecules involved in both pathways. PTH, thyroid hormones, estrogens, prolactin, growth hormone, and glucocorticoids apparently also regulate Ca2+ transport by direct action, indirect mechanism mediated by the increase of renal 1,25(OH)2D3 production, or both. Different physiological conditions, such as growth, pregnancy, lactation, and aging, adjust intestinal Ca2+ absorption according to Ca2+ demands. Better knowledge of the molecular details of intestinal Ca2+ absorption could lead to the development of nutritional and medical strategies for optimizing the efficiency of intestinal Ca2+ absorption and preventing osteoporosis and other pathologies related to Ca2+ metabolism.
Core tip: Intestinal Ca2+ absorption occurs through transcellular and paracellular pathways. Apparently, novel proteins, such as Cav1.3 and 4.1R, are involved in the Ca2+ transcellular pathway. Proteins involved in tight junction structures, such as claudins 2, 12, and 15, participate in the paracellular pathway. There is evidence of crosstalk between the transcellular and paracellular pathways. Better knowledge of the molecular details of intestinal Ca2+ absorption could lead to the development of nutritional and medical strategies for optimizing the efficiency of intestinal Ca2+ absorption and preventing osteoporosis and other pathologies related to Ca2+ metabolism.
- Citation: Diaz de Barboza G, Guizzardi S, Tolosa de Talamoni N. Molecular aspects of intestinal calcium absorption. World J Gastroenterol 2015; 21(23): 7142-7154
- URL: https://www.wjgnet.com/1007-9327/full/v21/i23/7142.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i23.7142
The Ca2+ ion plays an important role during human life. The accumulation of Ca2+ in the first decades of life allows for the achievement of optimal peak bone mass and, later in life, the maintenance of bone mass. It is a key constituent of many essential physiological processes, from intracellular signals to the mineralization of bone[1]. It is well known that Ca2+ is involved in nerve impulse transmission, muscle contraction, blood coagulation, secretory activity, cell death, cell differentiation, immune response, and enzyme activation[2]. The dysregulation of Ca2+ homeostasis is associated with bone disorders, metabolic diseases[3], and an increase in the risk of epithelial cancers[4].
The intestine, kidney, bone, and parathyroid glands work together to maintain serum Ca2+ within a narrow range. Intestinal Ca2+ absorption is a crucial process for the maintenance of Ca2+ balance and bone health. It occurs through two main mechanisms: transcellular, metabolically driven transport, and a passive non-saturable route called the paracellular pathway[5]. Both pathways are regulated by hormones, nutrients, and other factors. There has been much research work dedicated to ascertaining the regulation mechanisms of these factors due to their high relevance in the prevention of osteoporosis and other pathologies related to Ca2+ metabolism.
The transcellular pathway implicates Ca2+ movement from the mucosal-to-serosal side of the intestinal barrier that occurs against a concentration gradient. It is an active saturable process that predominates in the duodenum and jejunum, and is regulated by nutritional and physiological factors, mainly vitamin D. The paracellular mechanism is a non-saturable and passive transport that occurs across the majority of the intestine and is a linear function of luminal Ca2+ concentration[6]. Recently, it has been demonstrated that Ca2+ transport through the paracellular shunt is also significantly regulated[1].
The total Ca2+ absorbed depends on the amount of Ca2+ consumed, the sojourn time in the various segments of the small and large intestine, and the amount of soluble Ca2+ available for absorption, which is mainly determined by the pH in each segment. The acidic environment of the stomach dissolves calcium salts into Ca2+ ions. However, the pH of stomach is less relevant than that of the other segments, as Ca2+ is absorbed in the small and large intestine. The average pH is 7.3 in the small intestine and 6.6 in the colon. When pH increases, Ca solubility decreases. Nevertheless, the duodenum seems to be the site with the maximum solubility of Ca, as the average pH is 6.0, which is the lowest in the entire gut[7].
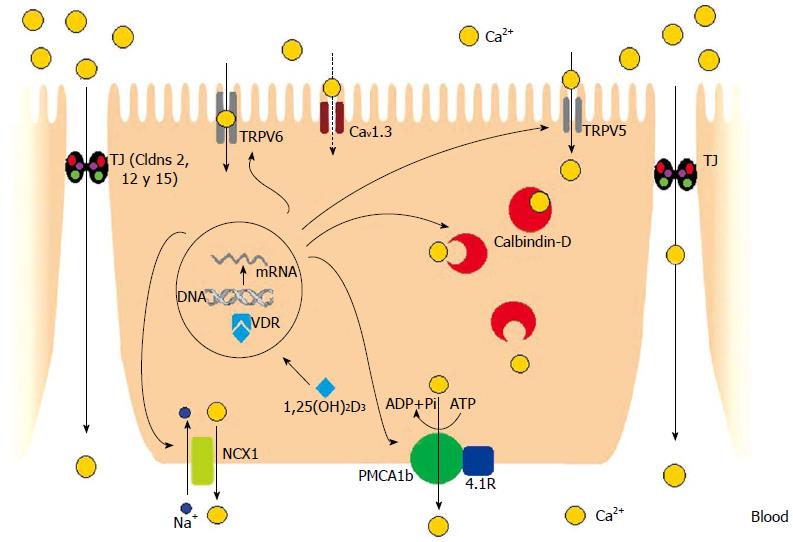
The transcellular pathway of intestinal Ca2+ absorption comprises 3 steps: the entrance of Ca2+ across the brush border membranes (BBM) of enterocytes through epithelial Ca2+ channels; Ca2+ movement from the BBM to the basolateral membranes (BLM) by binding to proteins with high Ca2+ affinity [(calbindins (CB)]; and Ca2+ extrusion into the blood via plasma membrane Ca2+-ATPase (PMCA1b/Ca2+ pump) and the Na+/Ca2+ exchanger (NCX1)[5].
Two epithelial Ca2+ channels seem to be involved with Ca2+ entry to enterocytes: the epithelial Ca2+ channel transient receptor potential vanilloid 6 (TRPV6; previously named ECaC2 and CaT1) and TRPV5 (previously named ECaC1 and CaT2). Both molecules are co-expressed in the human kidney and intestine, but TRPV6 is highly expressed in the intestine and TRPV5 is the major isoform in the kidney. High levels of TRPV6 have been detected in the duodenum and colon of humans, rats, and mice[8-10]. Although TRPV6 is one of the key players in human intestinal Ca2+ absorption, its precise role needs to be investigated. In TRPV6-/- mice, a considerable amount of Ca2+ transport still occurs, which suggests that some other channels or molecules contribute significantly to intestinal Ca2+ absorption[11]. TRPV6 and TRPV5 are also present in other organs, such as the pancreas, prostate, mammary glands, sweat, and salivary glands[11]. Both channels can be combined to form heterotetrameric channel complexes with different properties[12]. They originate from two genes juxtaposed on human chromosome 7q35, have 75% homology, but differ in the N and C terminal tails. They are regulated by calcitriol, estrogen, and dietary Ca2+. However, the inactivation of both by intracellular Ca2+ shows different kinetics, and the response to ruthenium red is also different. Ruthenium red is a potent blocker of epithelial Ca2+ channel activity. However, TRPV6 has a 100-fold lower affinity for ruthenium red (IC50 9 ± 1 μm) than TRPV5 (IC50 121 ± 13 nm)[12]. In vitro studies demonstrate that a negatively charged amino acid (D) within the putative pore region of mouse TRPV6 (position 541 in mice; position 542 in humans) is critical for Ca2+ permeation of the channel. Woudenberg-Vrenken et al[13] analyzed the role of TRPV6 in transepithelial Ca2+ transport in vivo by using a TRPV6D541A/D541A knock-in mouse model. TRPV6D541A/D541A mice showed significantly impaired intestinal Ca2+ uptake compared with wild-type mice, and duodenal TRPV5 expression was increased, but insufficient to correct the diminished Ca2+ absorption. Since intestinal Ca2+ absorption was not totally abolished in the TRPV6D541A/D541A mice, the authors suggest that other transport mechanisms, either paracellular or an as yet unidentified transcellular transport mechanism, were functioning.
Cav1.3 is an L-type channel, located in the apical membrane, capable of active, transcellular Ca2+ absorption in the intestine. It has been hypothesized that TRPV6 and Cav1.3 have complementary roles in Ca2+ entry. TRPV6 plays a dominant role under the polarizing conditions between meals. Overnight or during starvation, the BBM repolarize and the intestine gradually atrophies, so the Ca2+ lost into the lumen by desquamation must be recovered. TRPV6 is activated by apical membrane repolarization and upregulated by vitamin D to avoid massive loss of Ca2+ from the body. In contrast, Cav1.3 plays a dominant role under depolarizing conditions, such as during digestion, mainly when diet and Ca2+ are plentiful. Glucose, amino acids, and peptides may activate Cav1.3 and inhibit TRPV6. The general distribution of TRPV6 and Cav1.3 is in line with the aforementioned hypothesis. TRPV6 levels are higher in the duodenum, which has a polarizing environment and decreases through the jejunum to the ileum. In contrast, Cav1.3 levels are low in the duodenum, but high from the proximal jejunum to the mid-ileum. The interplay of TRPV6 and Cav1.3 maintains the tight control of free Ca2+ concentration in the extracellular space at any time of day. TRPV6 and Cav1.3 would have independent and complementary actions through activation by repolarization or depolarization between digestive periods or during digestion, respectively[14].
TRPV6 transcripts were detected in the duodenum, but not in the ileum of human intestinal biopsies. The duodenal expression of TRPV6 in men was vitamin D dependent, whereas in elderly women TRPV6 and vitamin D receptor (VDR) expressions were low and not vitamin D dependent. This might explain, at least in part, the lower intestinal Ca2+ absorption in elderly post-menopausal women[15]. In mice, the basal levels of TRPV6 protein in the duodenum, ileum, and colon have been found in the rank order of duodenum > colon (72% of duodenum) > ileum (25% of duodenum)[16].
Traditionally, calbindins (CB) were proteins considered to be responsible for carrying Ca2+ from the apical side of the enterocyte to the BLM of the cell. CB9k (human gene symbol: S100G) is present in the intestine of mammals and CB28k (Mr ≈ 29 kDa; human gene symbol: CALB1) in the intestine of avian species[17]. CB9k is the smallest protein with four alpha-helical regions, which form an EF-hand pair consisting of a canonical and non-canonical/pseudo EF-hand domain, joined by a linker region. Two Ca2+ ions bind the EF-hand domains with positive cooperativity[18]. CB28k has six EF-hand domains, but only the four medium/high affinity sites are considered Ca2+-specific[19].
CBs not only carry Ca2+ from the apical side to the BLM of enterocytes, but also buffer Ca2+ maintaining intracellular Ca2+ concentrations below 10-7 mol/L, which prevents premature cell death by apoptosis. Excess Ca2+ resulting from a downregulation of CBs may trigger apoptosis in the epithelial cells[20]. It is known that a high concentration of free Ca2+ provokes apoptosis in many different cell types. CB28k is able to inhibit apoptosis in osteoblastic cells[21] and in germ cells from Robertsonian mice[22,23]. In the kidney, CB28k acts as a dynamic buffer which regulates Ca2+ concentration in the vicinity to the TRPV5 pore by direct association with the channel[24]. The possibility that these mechanisms occur in the intestine and other tissues with important fluctuations of intracellular Ca2+ should be explored.
The regulation of human intestinal CB9k is not completely elucidated. CB9k is actively expressed in enterocytes, which are the predominant cells of the duodenal mucosa, and its expression decreases along the gastrointestinal tract until finally reaching undetectable levels in the distal ileum and large intestine[25]. It is quite intriguing that intestinal CB9k expression increases with age in the bulb and 2nd portion of the duodenum, whereas the plasma Ca2+ levels decrease[26]. In rodents, CB9k is regulated at the transcriptional and post-transcriptional levels by 1,25-dihydroxyvitamin D3 [1,25(OH)2D3 (calcitriol)], with the hormonal form derived from vitamin D[27-29]. It has been observed in mice that TRPV6 and CB9k are similarly regulated. Both proteins are induced at weaning or under low Ca diet and after 1,25(OH)2D3 administration. It is important to note that the induction of these proteins occur before the peak of intestinal Ca2+ absorption[30]. Christakos et al[31] have suggested that CB9k and TRPV6 are associated, with the function of CB9k being to facilitate TRPV6-mediated Ca2+ influx by preventing Ca2+ channel inactivation, but this requires further evidence to be proven.
Hwang et al[32] have demonstrated that an ablation of CB9k alters the expression of paracellular tight junction (TJ) genes. The compensatory expression of paracellular TJ genes in the duodenum was associated with transcellular CB9k, but not CB28k. This crosstalk between the transcellular and paracellular pathways might partially explain the variety of gut responses to the absorption of Ca2+ under different pathophysiological conditions.
Ca2+ extrusion from enterocytes is performed by two proteins: PMCA1 and NCX1. PMCA1 was first detected in erythrocyte membranes and found to have a high affinity for Ca2+[33]. There are four PMCA isoforms (PMCA1 to 4) that are in turn divided into several subtypes by alternative splicing. In mammals, four different genes encode PMCA, which in humans are located on four different chromosomes. For the four genes, two splice sites have been characterized, which are positioned either close to or within the regulatory regions of the pump, giving rise to many spliced isoforms[34]. PMCA1 is known as the housekeeping isoform because its mRNA is found in all tissues, but this concept is now questionable due to several factors involved in its regulation[35]. The predominant form in the intestine is the isoform PMCA1b, the expression and activity of which is higher in enterocytes from the villus tip in comparison with those from the villus crypt[36]. This finding agrees with the idea that mature enterocytes have the greatest capacity for transcellular Ca2+ movement. In chick intestine, vitamin D deficiency decreases the expression and activity of PMCA1b, which can be partially reversed by a single large dose of cholecalciferol[37].
PMCAs can be activated by Ca2+/calmodulin, acidic phospholipids, and serine/threonine phosphorylation, and can interact with numerous molecules[38]. The novel protein 4.1R seems to be a partner of PMCA1b, which could have a crucial role in the transcellular Ca2+ pathway. The protein 4.1R was first identified in the erythrocyte membrane skeleton, and is expressed in the epithelia of the intestine and other epithelia. So far, its physiological function is not well known. Liu et al[39] found that 4.1R co-localizes with PMCA1b. They also found that 4.1R KO mice exhibit impairment in intestinal Ca2+ absorption and decreased expression of PMCA1b in enterocytes. The association between PMCA1b and 4.1R involves the membrane-binding domain of 4.1R, as well as the second intracellular loop and C-terminus of PMCA1b. The finding that the protein 4.1R binds to PMCA1b suggests that protein 4.1R may regulate the function of PMCA1b and, consequently, intestinal Ca2+ absorption.
PMCA1b is the main protein involved in the exit of Ca2+ from enterocytes[40]. In contrast, NCX1 is only responsible for about 20% of Ca2+ extrusion from the intestine to plasma[41]. Because of that, this exchanger has received little attention, with some reviews ignoring it as another molecule involved in the exit of Ca2+ from the intestine. In contrast, NCX1 is critical for Ca² regulation in cardiac muscle, vascular smooth muscle, and nerve fibers, and most of the literature about NCX1 refers to these organs[42-44]. NCX electrogenically exchanges Na+ and Ca2+ across the plasma membrane, depending on membrane potential and ion gradients[45]. This exchanger has a stoichiometry of 3 Na+:1 Ca2+, and can operate in either a forward mode (Ca2+ extrusion) or in a reversed mode (Ca2+ entry), which depends on the Na+ and Ca2+ gradients and the potential across the plasma membrane[46,47]. The expression and activity of NCX1 are quite similar between mature and immature enterocytes from chick duodenum, but are slightly higher in the villus tip cells[36].
The movement of molecules and ions through this pathway is regulated by the TJ, which are specialized membrane domains mostly positioned in the apical region of enterocytes. TJ are intercellular structures where plasma membranes of adjacent cells have very close contact. These junctions are composed of transmembrane proteins, cytoskeleton components, and cytoplasmic plaques[48]. The transmembrane proteins of TJ structures are synthesized in adjacent cells and include occludin (Ocln) and claudins (Cldns). These proteins close intercellular junctions and restrict the free movement of materials through the paracellular space. Cldn 2, 12, and 15 are responsible for transporting Ca2+ in the intestine[49,50]. Cldn 1 and Cldn 5 have clear sealing functions that might also affect Ca2+ transport, as they influence general paracellular permeability[51,52]. The involvement of Ocln in intestinal Ca2+ absorption remains to be determined. Ocln is a tetraspan transmembrane protein, but its precise function is not well established[32]. Cytoplasmic plaques, such as zona occludens (ZO) proteins, contain a binding domain for transmembrane proteins[53]. ZO-1, a cytoplasmic protein with the ability to bind with both occludin and claudins[54], is associated with the structure and formation of the TJ, and possibly with paracellular ion transport[55]. Although it is not clear whether ZO-1 has a physiological role in intestinal Ca2+ transport, the increase in ZO-1 expression in the intestine of rats with 21-d chronic metabolic acidosis suggests that this protein might be implicated in intestinal Ca2+ absorption[56].
Ca2+ transport through the TJ is a passive process which depends on the concentration and electric gradient across the epithelium. The process is non-saturable transport that predominates in the jejunum and ileum when Ca2+ intake is adequate or high[57]. This route becomes important when Ca2+ intake is high, because the sojourn time in the intestine is short and there is a downregulation of proteins involved in the transcellular pathway[58] (Figure 1).
As previously mentioned, it seems that there is crosstalk between the transcellular and paracellular pathways involved in intestinal Ca2+ absorption. The expression of most TJ genes in the duodenum was significantly increased in CB9k KO mice compared to wild-type animals under a normal diet. A deficiency in dietary Ca2+ or vitamin D decreased TJ gene expression in CB9k KO mice. The data indicate that the expression of paracellular TJ genes is regulated by transcellular CB proteins, which suggests that active and passive Ca2+ transport pathways may function cooperatively[32]. More research work is necessary to clarify the network of transcellular and paracellular pathways for intestinal Ca2+ absorption.
Intestinal redox status is critical for both transcellular and paracellular pathways of intestinal Ca2+ absorption. Xiao et al[59] have demonstrated that a duodenal oxidation state induced by a high fat diet could significantly downregulate the expression of CB9k, PMCA1b, and NCX, as well as inhibiting intestinal calcium absorption. We have recently demonstrated that type I diabetes mellitus transiently inhibits intestinal Ca2+ absorption. Inhibition is accompanied by oxidative stress, which alters the gene and protein expression of molecules involved in the transcellular and paracellular pathways. When insulin is administered, the duodenal redox state returns to the control values, while intestinal Ca2+ absorption normalizes[60]. Similarly, oxidant drugs such as menadione, DL-buthionine-S,R-sulfoximine, and sodium deoxycholate decrease intestinal glutathione content, thereby affecting mainly the gene and protein expression of molecules involved in the transcellular pathway of intestinal Ca2+ absorption. Normalization of redox status with drugs such as quercetin, ursodeoxycholic acid, and melatonin returns intestinal Ca2+ transport to control values[61-65].
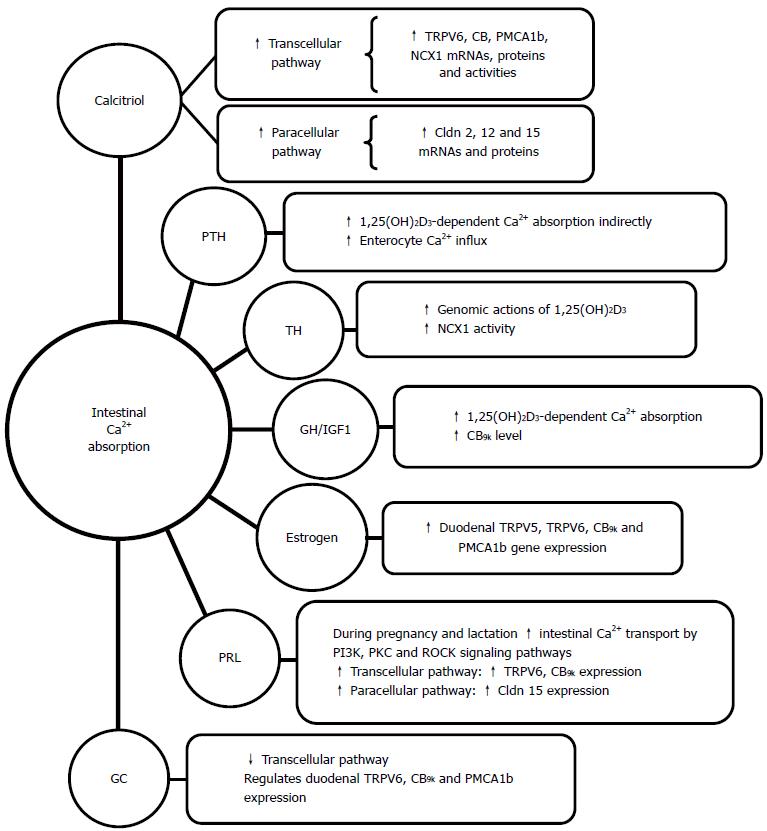
1,25 (OH)2D3 is the major controlling hormone of intestinal Ca2+ absorption. It causes changes in the structure and function of enterocytes[66,37], which enhance Ca2+ transport across the intestine. The action of calcitriol is mediated by genomic and non-genomic mechanisms after binding VDR. This receptor, located mainly in the nucleus, is a transcription factor that mediates the cellular effects of vitamin D by binding the vitamin D response elements of target genes[67]. The critical role of VDR and its ligand in intestinal Ca2+ absorption was confirmed in VDR KO mice in the third week of life. At birth, VDR KO mice are indistinguishable from their normal littermates. Alterations in growth and mineral ion homeostasis begin later[68], which is consistent with the observation that intestinal Ca2+ absorption is vitamin D independent in rodents in the first weeks of life[69]. Ten-week-old VDR KO mice showed a dramatic decrease in duodenal Ca2+ absorption, which was associated with impaired expression of TRPV6, TRPV5, and CB9k[70]. Beyond VDR function in intestinal Ca2+ absorption, a recent proteomic approach has revealed that VDR is also an important factor for controlling cell proliferation, migration, and stress response in the small intestine[71].
Most studies related to the effect of calcitriol on intestinal Ca2+ absorption have been focused on the transcellular Ca2+ pathway. All molecules presumably involved in this route are increased by calcitriol in experimental animals, and even in humans[72-75].
Cyp24a1 is a major VDR-responsive gene that metabolizes 1,25(OH)2D3 into 1,24,25-trihydroxyvitamin D3 and 25(OH)D3 into 24,25-dihydroxyvitamin D3[76], while Cyp27b1 is the gene involved in the synthesis of 1,25(OH)2D3, which is mainly located in the kidney[77], but is also expressed in other tissues, such as the intestine[78] and parathyroid gland[79]. Both genes are regulated by 1,25(OH)2D3 levels. Their temporal profiles and those from other VDR responsive genes were analyzed in the intestine and other tissues of mice after single and multiple dosing of 1,25(OH)2D3. Due to the lipophilic nature of the compound, a rapid distribution of 1,25(OH)2D3 into tissues was observed, regardless of variation in VDR abundance in different tissues. The maximal induction of VDR target genes such as TRPV6 and Cyp24a1 mRNA expression in the intestine were similar after single vs multiple dosing, with a peak between 3 and 9 h post-injection, whereas the peak of 1,25(OH)2D3 concentration in the ileum occurred at 0.5-1 h. This lag time was the result of the time required for translocation of the VDR into the nucleus in order to heterodimerize with the RXR to initiate the transcription. An increase in ileal VDR levels was also observed, as well as an attenuation of serum PTH and a decrease in renal Cyp27b1 expression after a time delay in VDR activation. The data revealed that exogenous 1,25(OH)2D3 enters the intestine, rapidly equilibrates, and then VDR target genes respond quickly. Consequently, plasma Ca2+ levels increase as a result of enhanced intestinal Ca2+ absorption[16].
1,25(OH)2D3-enhanced Ca2+ transport in mice was reported to be inhibited by fibroblast growth factor-23 (FGF-23), as well as Ca2+ transport in colon cancer Caco-2 cells. FGF-23 produced an abolishment of enhanced transcellular active Ca2+ fluxes and a modest downregulation of the paracellular Ca2+ route[80].
VDR null mice adapt to pregnancy by the upregulation of duodenal TRPV6 and intestinal Ca2+ absorption. These mice lactate normally and fully restore bone mineral content after weaning. Therefore, VDR seems not to be required for skeletal adaptation during pregnancy, lactation, and after weaning[81]. In the elderly, there is a decrease in intestinal Ca2+ absorption, and thus higher Ca2+ intake is needed. Increasing Ca2+ intake via dairy products and Ca2+-fortified food is a much better option than supplements. It has been estimated a 30% reduction in fractures for elderly individuals is possible by using the simple and inexpensive strategy of a daily vitamin D intake of 800 IU, together with a total Ca2+ intake of 1000 mg/d[82].
The paracellular pathway of intestinal Ca2+ absorption has been demonstrated to also be increased by 1,25(OH)2D3, predominantly in the jejunum and ileum[83]. It has been found that 1,25(OH)2D3 significantly enhanced Cldn- 2 and Cldn-12 mRNA levels in colon cancer Caco-2 cells. The mRNA and protein levels for these proteins were lower at 12 wk in the jejunum of VDR KO mice in comparison with wild-type mice, and siRNA against these Cldns diminished Ca2+ permeability in Caco-2 cells[84]. Cadherin-17 and aquaporin-8 have been reported to be downregulated by 1,25(OH)2D3 in the intestine[85,86]. Cadherin-17 is involved in cell-to-cell contact, and its decrease might in turn increase intestinal permeability. A decrease in channel aquaporin-8 might influence TJ selectivity towards cations. The data indicate that Ca2+ movement through TJ is regulated and supports the regulation of the paracellular Ca2+ transport route by 1,25(OH)2D3[31].
PTH acts indirectly on intestinal Ca2+ absorption by the stimulation of renal CYP27B1 and, therefore, increases 1,25(OH)2D3-dependent Ca2+ absorption. A direct effect of PTH on intestinal Ca2+ absorption has not been demonstrated, but some direct effects of PTH on Ca2+ uptake by enterocytes from rat duodenum were reported. PTH/PTHrP receptors have been localized in intestinal epithelial cells along the villus[87]. It has been suggested that an in vivo model would need to be generated with targeted deletion of intestinal PTH receptor 1 in order to test if PTH directly affects intestinal Ca2+ absorption[88].
With regard to thyroid hormones, it has been reported that they produce a cooperative effect with vitamin D for intestinal Ca2+ transport. Apparently, thyroid hormones increase the genomic action of 1,25(OH)2D3 in the intestine[89]. Kumar et al[90] demonstrated that hyperthyroid rats show higher Ca2+ uptake and Ca2+ efflux from enterocytes than hypothyroid rats. They have also observed that NCX1 activity was highly increased by thyroid hormones, presumably via the cAMP-mediated pathway. Orihuela[91] has analyzed the effect of different statuses of thyroid hormones on the inhibitory effect of aluminum (Al) on intestinal Ca2+ absorption by using a rodent model. Mucosa-to-serosa Ca2+ fluxes in Al-exposed rats declined as thyroid hormones levels increased, thereby showing a trend opposite to that seen in non-Al-treated control rats.
Growth hormone (GH) has a major role in linear bone growth and bone Ca2+ deposition during childhood and adolescence. GH has proliferative effects upon the intestinal epithelium[92], and can also stimulate intestinal Ca2+ absorption, which would occur indirectly by increasing serum 1,25(OH)2D3 concentration[93].
However, it has also been shown that GH treatment increases intestinal Ca2+ absorption and duodenal CB9k levels in aged rats without increasing serum 1,25(OH)2D3 levels[94]. In adult men, Ca2+ absorption has been shown to be positively correlated with IGF-1, and age-related declines in IGF-1 have a negative impact on Ca2+ absorption that could not be explained by a decrease in serum 1,25(OH)2D3[95].
Most estrogen studies related to intestinal Ca2+ absorption were performed in ovariectomized (OVX) animals. This ablation significantly decreases endogenous estrogen, but not totally, since adrenal androgens can be aromatized to estrogen[96]. An estradiol replacement in OVX rats has been reported to increase intestinal Ca2+ absorption without stimulation of circulating 1,25(OH)2D3 levels[97]. van Abel et al[98] found increased duodenal gene expression of TRPV5, TRPV6, CB9k, and PMCA1b in OVX rats treated with estradiol. They used Cyp27b1 KO mice to analyze the calcitriol dependency of the stimulatory effects of estradiol on intestinal Ca2+ absorption, and found that estradiol treatment increased mRNA levels of duodenal TRPV6. Cell culture studies suggest that estrogen corrects the decline in the efficiency of intestinal Ca2+ absorption at the onset of menopause[99], but the mechanisms that underlie this effect remain unknown. Estrogen receptor alpha (ERα) KO mice showed a decrease in duodenal TRPV6 mRNA expression, while CB9k, PMCA1b, and VDR levels were not modified. Therefore, it seems that the genomic effects of estrogen on mice are mainly mediated by ERα[100]. In addition to estrogen, prolactin, a hormone that is elevated during pregnancy and lactation, has been shown to stimulate active intestinal Ca2+ transport in vitamin D deficient rats[101]. A direct effect of prolactin on active duodenal Ca2+ transport was shown[102]. Some data indicate that prolactin can regulate intestinal TRPV6 and cooperate with 1,25(OH)2D3 in regulating TRPV6 and CB9k. Prolactin also has a direct effect on the transcription of the Cyp27b1 gene, thus enhancing CYP27B1 protein expression and increasing levels of 1,25(OH)2D3 during lactation when there is an increased Ca2+ requirement for the neonate[103]. It has been suggested that prolactin also has an effect on the paracellular pathway of intestinal Ca2+ absorption through an upregulation of Cldn 15[104]. Some authors indicate that there is enough evidence that prolactin could be considered the cardinal calciotropic hormone in pregnancy and lactation[105].
Although reduced intestinal Ca2+ absorption seems to be part of the pathogenesis of glucocorticoid-induced osteoporosis[106], the mechanisms triggered by GCs in the intestine remain unclear. Short-term GC treatment in young animals does not alter the expression of genes involved in intestinal Ca2+ absorption, such as TRPV6, CB9k and PMCA1b[107], but sustained dexamethasone suppresses mouse duodenal CB9k expression[108]. Kim et al[109] found that gene regulation in the intestine by dexamethasone is complex in mice. It provokes an increase of duodenal TRPV6, CB9k, and PMCA1b 24 h after administration, which was followed by a decrease in a 5-d treatment. Ten days of prednisolone treatment decreases rat intestinal Ca2+ absorption through a diminished expression of the active Ca2+ transporters, which is independent of 1,25(OH)2D3[110] (Figure 2).
Intestinal Ca2+ absorption changes according to the physiological conditions of individuals. When needs are high and/or dietary Ca2+ is low, intestinal Ca2+ absorption becomes more efficient. Growth, pregnancy, lactation, dietary Ca2+ deficiency, and high physical activity enhance the Ca2+ demands that promote intestinal Ca2+ absorption. During pregnancy, Ca2+ absorption is higher than before conception or after delivery. The enhancement occurs in early-to-mid pregnancy, and precedes the increased Ca2+ demand from the fetus for skeletal growth. This alteration in Ca2+ absorption during pregnancy may be due to increased serum calcitriol, with little alteration in serum PTH or calcitonin[111].
The maternal adaptation for the enhancement of intestinal Ca2+ absorption in pregnancy and lactation is very important for fetal development and lactogenesis. Intestinal Ca2+ absorption in pregnant adolescents has been shown to be higher in the third trimester of pregnancy than in the early postpartum period[112].
Vitamin D seems to play an important role during pregnancy. Yamagishi et al[113] found in pregnant rats that vitamin D deficiency produces severe hypocalcemia due to reduced intestinal Ca2+ absorption and elevated fetal demand for cation. It has been shown in mice that serum calcitriol was enhanced five-fold during pregnancy, whereas vitamin D binding protein levels were unchanged. A 30-fold higher expression of Cyp27b1 in maternal kidneys vs placenta suggests that the increase in calcitriol comes from the kidneys. Apparently, PTH is not required to upregulate Cyp27b1 expression during pregnancy[114]. A custom-designed cDNA microarray validated by quantitative real time PCR has found in rats that several duodenal transporters, such as TRPV6, are upregulated during pregnancy[115]. In addition, when the plasma estradiol levels are increased in pregnancy, intestinal CB9k gene expression is concomitantly enhanced, which suggests that the CB9k gene is involved in the compensatory induction of other Ca2+ transporter genes in duodenal epithelial cells[116]. Zhu et al[117] have found that both PMCA1 and CB mRNA levels were increased 2- to 3-fold in rats at 21 d of gestation. These levels of PMCA1 and CB mRNA remained elevated at 7 d of lactation.
Recently, in an animal model of preeclampsia, disturbance of Ca2+ metabolism in the placenta, intestine, and kidney has been observed. A decrease in Ca2+ transporting genes (TRPV5, TPRV6, PMCA1, and CB9k) has been detected in all these organs. In the duodenum, there was a slight recovery after calcium supplementation, whereas, in the kidney, these alterations were reverted to the control levels by the supplements[118].
The mechanisms by which intestinal Ca2+ absorption is induced in lactation are not quite clear. One possible adaptive mechanism includes hypertrophy and hyperplasia of the intestinal villi, as shown in lactating mammals such as rodents, pigs, and sheep[119,120]. Wongdee et al[121] have also demonstrated that intestinal hypertrophy in lactating rats is associated with an upregulation of Cldn 15 protein expression. It has been shown in rats that the suckling-induced transient prolactin surge increases intestinal Ca2+ transport during lactation, an effect that occurs through signaling pathways involving phosphoinositide 3-kinase C (PI3K), as well as two serine/threonine kinases, such as protein kinase C (PKC) and RhoA-associated coiled-coil-forming kinase (ROCK)[122].
In newborn rats, intestinal Ca2+ absorption is largely passive non-saturable, and not dependent on calcitriol[123]. This lack of responsiveness to calcitriol at earlier stages is explained by the undetectable levels of VDR within enterocytes at 7 and 14 d after birth[69]. Human data indicate that, in neonates, intestinal absorption is initially a passive process, which is favored by milk lactose. Later, it becomes a calcitriol dependent active process, but the hormone’s role can be bypassed by high dietary Ca2+ content or parenteral Ca2+ administration[124].
Ca2+ supplied in human milk during infancy is primarily derived from maternal bone, which is rapidly replenished during and after weaning[125]. The optimization of Ca2+ intake is crucial in adolescents to maximize calcium retention, acquire a good peak of bone mass, and prevent osteoporosis later in life. At early puberty, there is an association between an increase in both Ca2+ absorption and bone Ca2+ deposition. In girls, bone Ca2+ deposition reaches a maximum shortly before menarche, with the deposition rate being approximately five times that of adulthood. After menarche, bone Ca2+ deposition, as well as intestinal Ca2+ absorption, gradually declines[125]. In boys aged 11-14 years on their usual diets, it has been shown that adolescents absorbed 31% of their dietary Ca2+ intake and retained 20% of their total Ca2+ intake, but their dietary Ca2+ intake failed to meet recommended values[126]. During puberty, polymorphisms of the Fok I site in the VDR gene have been significantly associated with Ca2+ absorption and bone mineral density[127].
Aging has been associated with lower intestinal Ca2+ absorption[128], with an additional decrease at the time of menopause that is reversible with estrogen therapy[129]. This decrease might occur by a declination in serum calcitriol levels and a resistance to the actions of calcitriol in the intestine[130]. It has been reported that low levels of VDR in mouse heterozygotes for the VDR gene KO produce a resistance of intestinal Ca2+ absorption to 1,25(OH)2D3. This resistance seems to be generated by the low translation of CB9k, which is mediated by binding VDR with the ligand[131]. A post hoc analysis of dual isotope studies in post-menopausal women has identified associations of several factors with intestinal Ca2+ absorption. The data indicate that age, 1,25(OH)2D3, and dietary calcium and fat are associated with Ca2+ absorption, whereas serum 25(OH)D levels are not. The authors claim that this study has unique findings, as the dietary intake of kilocalories, carbohydrates, and potassium are also associated with intestinal Ca2+ absorption, suggesting that beyond the traditional focus on Ca2+ and vitamin D, some other factors also influence intestinal Ca2+ absorption in post-menopausal women[129].
Since Ca2+ ions are involved in most physiological processes, it becomes important to know the molecular details of intestinal absorption of cation, as the intestine is the only entrance for Ca2+ to the organism. Absorption occurs through two different pathways, transcellular and paracellular, which apparently interact with each other depending on physiological conditions. Calcitriol [1,25(OH)2D3] is the major regulating hormone, which clearly operates through VDR signaling, but the exact mechanism by which Ca2+ moves from the lumen to the serosa remains elusive. The role of other hormones, such as PTH, GC, GH, estrogens, thyroid hormones, and prolactin, is under investigation due to controversial data. Some of them act indirectly through the regulation of renal 1,25(OH)2D3 production, others by direct action, and still others by both methods. Different physiological conditions, such as growth, pregnancy, lactation, and aging, significantly alter intestinal Ca2+ absorption, according to Ca2+ requirements. Further research should be carried out to improve the current knowledge concerning the regulation of intestinal Ca2+ absorption in order to develop nutritional or medical strategies to optimize the efficiency of intestinal Ca2+ absorption and prevent osteoporosis and other pathologies related to Ca2+ metabolism.
P- Reviewer: Caviglia RD, Daniel F, Serafino A, Tovo CT, Wang DS S- Editor: Qi Y L- Editor: Rutherford A E- Editor: Liu XM
| 1. | Alexander RT, Rievaj J, Dimke H. Paracellular calcium transport across renal and intestinal epithelia. Biochem Cell Biol. 2014;92:467-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 2. | Mori M, Tanifuji S, Mochida S. Kinetic organization of Ca2+ signals that regulate synaptic release efficacy in sympathetic neurons. Mol Pharmacol. 2014;86:297-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 3. | Bartlett PJ, Gaspers LD, Pierobon N, Thomas AP. Calcium-dependent regulation of glucose homeostasis in the liver. Cell Calcium. 2014;55:306-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 4. | Crosara Teixeira M, Braghiroli MI, Sabbaga J, Hoff PM. Primary prevention of colorectal cancer: myth or reality? World J Gastroenterol. 2014;20:15060-15069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 34] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 5. | Pérez AV, Picotto G, Carpentieri AR, Rivoira MA, Peralta López ME, Tolosa de Talamoni NG. Minireview on regulation of intestinal calcium absorption. Emphasis on molecular mechanisms of transcellular pathway. Digestion. 2008;77:22-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 106] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 6. | Wasserman RH. Vitamin D and the dual processes of intestinal calcium absorption. J Nutr. 2004;134:3137-3139. [PubMed] |
| 7. | van der Velde RY, Brouwers JR, Geusens PP, Lems WF, van den Bergh JP. Calcium and vitamin D supplementation: state of the art for daily practice. Food Nutr Res. 2014;58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 8. | Karbach U, Feldmeier H. The cecum is the site with the highest calcium absorption in rat intestine. Dig Dis Sci. 1993;38:1815-1824. [PubMed] |
| 9. | Nijenhuis T, Hoenderop JG, van der Kemp AW, Bindels RJ. Localization and regulation of the epithelial Ca2+ channel TRPV6 in the kidney. J Am Soc Nephrol. 2003;14:2731-2740. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 148] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 10. | Peng JB, Chen XZ, Berger UV, Vassilev PM, Tsukaguchi H, Brown EM, Hediger MA. Molecular cloning and characterization of a channel-like transporter mediating intestinal calcium absorption. J Biol Chem. 1999;274:22739-22746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 489] [Cited by in RCA: 445] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 11. | Suzuki Y, Kovacs CS, Takanaga H, Peng JB, Landowski CP, Hediger MA. Calcium channel TRPV6 is involved in murine maternal-fetal calcium transport. J Bone Miner Res. 2008;23:1249-1256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 81] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 12. | Hoenderop JG, Vennekens R, Müller D, Prenen J, Droogmans G, Bindels RJ, Nilius B. Function and expression of the epithelial Ca(2+) channel family: comparison of mammalian ECaC1 and 2. J Physiol. 2001;537:747-761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 79] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 13. | Woudenberg-Vrenken TE, Lameris AL, Weißgerber P, Olausson J, Flockerzi V, Bindels RJ, Freichel M, Hoenderop JG. Functional TRPV6 channels are crucial for transepithelial Ca2+ absorption. Am J Physiol Gastrointest Liver Physiol. 2012;303:G879-G885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 51] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 14. | Kellett GL. Alternative perspective on intestinal calcium absorption: proposed complementary actions of Ca(v)1.3 and TRPV6. Nutr Rev. 2011;69:347-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 69] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 15. | Walters JR, Balesaria S, Chavele KM, Taylor V, Berry JL, Khair U, Barley NF, van Heel DA, Field J, Hayat JO. Calcium channel TRPV6 expression in human duodenum: different relationships to the vitamin D system and aging in men and women. J Bone Miner Res. 2006;21:1770-1777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 16. | Chow EC, Quach HP, Vieth R, Pang KS. Temporal changes in tissue 1α,25-dihydroxyvitamin D3, vitamin D receptor target genes, and calcium and PTH levels after 1,25(OH)2D3 treatment in mice. Am J Physiol Endocrinol Metab. 2013;304:E977-E989. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 50] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 17. | Tolosa de Talamoni N, Perez A, Alisio A. Effect of cholecalciferol on intestinal epithelial cells. Trends in Comparative Biochemistry and Physiology. Trivandrum: Research Trends 1998; 179-185. |
| 18. | Schwaller B. Cytosolic Ca2+ buffers. Cold Spring Harb Perspect Biol. 2010;2:a004051. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 290] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 19. | Nägerl UV, Novo D, Mody I, Vergara JL. Binding kinetics of calbindin-D(28k) determined by flash photolysis of caged Ca(2+). Biophys J. 2000;79:3009-3018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 154] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 20. | Choi KJ, Cho DS, Kim JY, Kim BJ, Lee KM, Kim SH, Kim DK, Kim SH, Park HS. Ca-induced Ca Release from Internal Stores in INS-1 Rat Insulinoma Cells. Korean J Physiol Pharmacol. 2011;15:53-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 21. | Bellido T, Huening M, Raval-Pandya M, Manolagas SC, Christakos S. Calbindin-D28k is expressed in osteoblastic cells and suppresses their apoptosis by inhibiting caspase-3 activity. J Biol Chem. 2000;275:26328-26332. [PubMed] |
| 22. | Merico V, de Barboza GD, Vasco C, Ponce R, Rodriguez V, Garagna S, Tolosa de Talamoni N. A mitochondrial mechanism is involved in apoptosis of Robertsonian mouse male germ cells. Reproduction. 2008;135:797-804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 23. | Rodriguez V, Diaz de Barboza G, Ponce R, Merico V, Garagna S, Tolosa de Talamoni N. Spermatocyte apoptosis, which involves both intrinsic and extrinsic pathways, explains the sterility of Graomys griseoflavus x Graomys centralis male hybrids. Reprod Fertil Dev. 2010;22:478-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 24. | Lambers TT, Mahieu F, Oancea E, Hoofd L, de Lange F, Mensenkamp AR, Voets T, Nilius B, Clapham DE, Hoenderop JG. Calbindin-D28K dynamically controls TRPV5-mediated Ca2+ transport. EMBO J. 2006;25:2978-2988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 100] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 25. | Walters JR, Howard A, Lowery LJ, Mawer EB, Legon S. Expression of genes involved in calcium absorption in human duodenum. Eur J Clin Invest. 1999;29:214-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 26. | Choi JH, Lee CH, Chung DW, Hwang IK, Won MH, Seong JK, Yoon YS, Lee IS. Age-related changes of calbindin D-28k-immunoreactive neurons in the myenteric plexus of gerbil duodenum. J Vet Med Sci. 2008;70:343-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 27. | Roche C, Bellaton C, Pansu D, Miller A, Bronner F. Localization of vitamin D-dependent active Ca2+ transport in rat duodenum and relation to CaBP. Am J Physiol. 1986;251:G314-G320. [PubMed] |
| 28. | Wasserman RH, Fullmer CS. On the molecular mechanism of intestinal calcium transport. Adv Exp Med Biol. 1989;249:45-65. [PubMed] |
| 29. | Darwish HM, DeLuca HF. Identification of a 1,25-dihydroxyvitamin D3-response element in the 5’-flanking region of the rat calbindin D-9k gene. Proc Natl Acad Sci USA. 1992;89:603-607. [PubMed] |
| 30. | Song Y, Peng X, Porta A, Takanaga H, Peng JB, Hediger MA, Fleet JC, Christakos S. Calcium transporter 1 and epithelial calcium channel messenger ribonucleic acid are differentially regulated by 1,25 dihydroxyvitamin D3 in the intestine and kidney of mice. Endocrinology. 2003;144:3885-3894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 171] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 31. | Christakos S, Lieben L, Masuyama R, Carmeliet G. Vitamin D endocrine system and the intestine. Bonekey Rep. 2014;3:496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 32. | Hwang I, Yang H, Kang HS, Ahn C, Hong EJ, An BS, Jeung EB. Alteration of tight junction gene expression by calcium- and vitamin D-deficient diet in the duodenum of calbindin-null mice. Int J Mol Sci. 2013;14:22997-23010. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 33. | Schatzmann HJ. ATP-dependent Ca++-extrusion from human red cells. Experientia. 1966;22:364-365. [PubMed] |
| 34. | Strehler EE, Zacharias DA. Role of alternative splicing in generating isoform diversity among plasma membrane calcium pumps. Physiol Rev. 2001;81:21-50. [PubMed] |
| 35. | Brini M, Calì T, Ottolini D, Carafoli E. The plasma membrane calcium pump in health and disease. FEBS J. 2013;280:5385-5397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 126] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 36. | Centeno VA, Díaz de Barboza GE, Marchionatti AM, Alisio AE, Dallorso ME, Nasif R, Tolosa de Talamoni NG. Dietary calcium deficiency increases Ca2+ uptake and Ca2+ extrusion mechanisms in chick enterocytes. Comp Biochem Physiol A Mol Integr Physiol. 2004;139:133-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 37. | Alisio A, Cañas F, de Bronia DH, Pereira R, Tolosa de Talamoni N. Effect of vitamin D deficiency on lipid composition and calcium transport in basolateral membrane vesicles from chick intestine. Biochem Mol Biol Int. 1997;42:339-347. [PubMed] |
| 38. | Di Leva F, Domi T, Fedrizzi L, Lim D, Carafoli E. The plasma membrane Ca2+ ATPase of animal cells: structure, function and regulation. Arch Biochem Biophys. 2008;476:65-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 199] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 39. | Liu C, Weng H, Chen L, Yang S, Wang H, Debnath G, Guo X, Wu L, Mohandas N, An X. Impaired intestinal calcium absorption in protein 4.1R-deficient mice due to altered expression of plasma membrane calcium ATPase 1b (PMCA1b). J Biol Chem. 2013;288:11407-11415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 40. | Freeman TC, Howard A, Bentsen BS, Legon S, Walters JR. Cellular and regional expression of transcripts of the plasma membrane calcium pump PMCA1 in rabbit intestine. Am J Physiol. 1995;269:G126-G131. [PubMed] |
| 41. | Ghijsen WE, De Jong MD, Van Os CH. Kinetic properties of Na+/Ca2+ exchange in basolateral plasma membranes of rat small intestine. Biochim Biophys Acta. 1983;730:85-94. [PubMed] |
| 42. | Kofuji P, Hadley RW, Kieval RS, Lederer WJ, Schulze DH. Expression of the Na-Ca exchanger in diverse tissues: a study using the cloned human cardiac Na-Ca exchanger. Am J Physiol. 1992;263:C1241-C1249. [PubMed] |
| 43. | Iwamoto T, Kita S, Katsuragi T. Salt-sensitive hypertension, Na+/Ca2+ exchanger, and vascular smooth muscle. Trends Cardiovasc Med. 2005;15:273-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 44. | Khananshvili D. Sodium-calcium exchangers (NCX): molecular hallmarks underlying the tissue-specific and systemic functions. Pflugers Arch. 2014;466:43-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 94] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 45. | Nishiyama K, Azuma YT, Kita S, Azuma N, Hayashi S, Nakajima H, Iwamoto T, Takeuchi T. Na-/Ca²+ exchanger 1/2 double-heterozygote knockout mice display increased nitric oxide component and altered colonic motility. J Pharmacol Sci. 2013;123:235-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 46. | Blaustein MP, Lederer WJ. Sodium/calcium exchange: its physiological implications. Physiol Rev. 1999;79:763-854. [PubMed] |
| 47. | Ottolia M, Philipson KD. NCX1: mechanism of transport. Adv Exp Med Biol. 2013;961:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 48. | Gumbiner BM. Cell adhesion: the molecular basis of tissue architecture and morphogenesis. Cell. 1996;84:345-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2618] [Cited by in RCA: 2557] [Article Influence: 88.2] [Reference Citation Analysis (0)] |
| 49. | Fujita H, Chiba H, Yokozaki H, Sakai N, Sugimoto K, Wada T, Kojima T, Yamashita T, Sawada N. Differential expression and subcellular localization of claudin-7, -8, -12, -13, and -15 along the mouse intestine. J Histochem Cytochem. 2006;54:933-944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 190] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 50. | Inai T, Sengoku A, Guan X, Hirose E, Iida H, Shibata Y. Heterogeneity in expression and subcellular localization of tight junction proteins, claudin-10 and -15, examined by RT-PCR and immunofluorescence microscopy. Arch Histol Cytol. 2005;68:349-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 55] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 51. | Inai T, Kobayashi J, Shibata Y. Claudin-1 contributes to the epithelial barrier function in MDCK cells. Eur J Cell Biol. 1999;78:849-855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 208] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 52. | Amasheh S, Schmidt T, Mahn M, Florian P, Mankertz J, Tavalali S, Gitter AH, Schulzke JD, Fromm M. Contribution of claudin-5 to barrier properties in tight junctions of epithelial cells. Cell Tissue Res. 2005;321:89-96. [PubMed] |
| 53. | Guillemot L, Paschoud S, Pulimeno P, Foglia A, Citi S. The cytoplasmic plaque of tight junctions: a scaffolding and signalling center. Biochim Biophys Acta. 2008;1778:601-613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 149] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 54. | Itoh M, Furuse M, Morita K, Kubota K, Saitou M, Tsukita S. Direct binding of three tight junction-associated MAGUKs, ZO-1, ZO-2, and ZO-3, with the COOH termini of claudins. J Cell Biol. 1999;147:1351-1363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 829] [Cited by in RCA: 854] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 55. | Umeda K, Matsui T, Nakayama M, Furuse K, Sasaki H, Furuse M, Tsukita S. Establishment and characterization of cultured epithelial cells lacking expression of ZO-1. J Biol Chem. 2004;279:44785-44794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 213] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 56. | Charoenphandhu N, Tudpor K, Pulsook N, Krishnamra N. Chronic metabolic acidosis stimulated transcellular and solvent drag-induced calcium transport in the duodenum of female rats. Am J Physiol Gastrointest Liver Physiol. 2006;291:G446-G455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 58. | Bronner F. Mechanisms of intestinal calcium absorption. J Cell Biochem. 2003;88:387-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 177] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 59. | Xiao Y, Cui J, Shi YH, Sun J, Wang ZP, Le GW. Effects of duodenal redox status on calcium absorption and related genes expression in high-fat diet-fed mice. Nutrition. 2010;26:1188-1194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 60. | Rivoira M, Rodríguez V, López MP, Tolosa de Talamoni N. Time dependent changes in the intestinal Ca(2+) absorption in rats with type I diabetes mellitus are associated with alterations in the intestinal redox state. Biochim Biophys Acta. 2015;1852:386-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 61. | Tolosa de Talamoni N, Marchionatti A, Baudino V, Alisio A. Glutathione plays a role in the chick intestinal calcium absorption. Comp Biochem Physiol A Physiol. 1996;115:127-132. [PubMed] |
| 62. | Marchionatti AM, Díaz de Barboza GE, Centeno VA, Alisio AE, Tolosa de Talamoni NG. Effects of a single dose of menadione on the intestinal calcium absorption and associated variables. J Nutr Biochem. 2003;14:466-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 63. | Marchionatti AM, Pacciaroni A, Tolosa de Talamoni NG. Effects of quercetin and menadione on intestinal calcium absorption and the underlying mechanisms. Comp Biochem Physiol A Mol Integr Physiol. 2013;164:215-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 64. | Carpentieri A, Marchionatti A, Areco V, Perez A, Centeno V, Tolosa de Talamoni N. Antioxidant and antiapoptotic properties of melatonin restore intestinal calcium absorption altered by menadione. Mol Cell Biochem. 2014;387:197-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 65. | Rodríguez V, Rivoira M, Marchionatti A, Pérez A, Tolosa de Talamoni N. Ursodeoxycholic and deoxycholic acids: A good and a bad bile acid for intestinal calcium absorption. Arch Biochem Biophys. 2013;540:19-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 66. | Tolosa de Talamoni N, Morero R, Cañas F. Vitamin D3 administration increases the membrane fluidity of intestinal mitochondria. Biochem Int. 1989;19:701-707. [PubMed] |
| 67. | Haussler MR, Whitfield GK, Kaneko I, Haussler CA, Hsieh D, Hsieh JC, Jurutka PW. Molecular mechanisms of vitamin D action. Calcif Tissue Int. 2013;92:77-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 486] [Cited by in RCA: 508] [Article Influence: 42.3] [Reference Citation Analysis (0)] |
| 68. | Li YC, Pirro AE, Amling M, Delling G, Baron R, Bronson R, Demay MB. Targeted ablation of the vitamin D receptor: an animal model of vitamin D-dependent rickets type II with alopecia. Proc Natl Acad Sci USA. 1997;94:9831-9835. [PubMed] |
| 69. | Halloran BP, DeLuca HF. Appearance of the intestinal cytosolic receptor for 1,25-dihydroxyvitamin D3 during neonatal development in the rat. J Biol Chem. 1981;256:7338-7342. [PubMed] |
| 70. | Van Cromphaut SJ, Dewerchin M, Hoenderop JG, Stockmans I, Van Herck E, Kato S, Bindels RJ, Collen D, Carmeliet P, Bouillon R. Duodenal calcium absorption in vitamin D receptor-knockout mice: functional and molecular aspects. Proc Natl Acad Sci USA. 2001;98:13324-13329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 464] [Cited by in RCA: 404] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 71. | Kühne H, Schutkowski A, Weinholz S, Cordes C, Schierhorn A, Schulz K, König B, Stangl GI. Vitamin D receptor regulates intestinal proteins involved in cell proliferation, migration and stress response. Lipids Health Dis. 2014;13:51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 72. | Meyer J, Fullmer CS, Wasserman RH, Komm BS, Haussler MR. Dietary restriction of calcium, phosphorus, and vitamin D elicits differential regulation of the mRNAs for avian intestinal calbindin-D28k and the 1,25-dihydroxyvitamin D3 receptor. J Bone Miner Res. 1992;7:441-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 34] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 73. | Wasserman RH, Smith CA, Brindak ME, De Talamoni N, Fullmer CS, Penniston JT, Kumar R. Vitamin D and mineral deficiencies increase the plasma membrane calcium pump of chicken intestine. Gastroenterology. 1992;102:886-894. [PubMed] |
| 74. | Centeno V, Picotto G, Pérez A, Alisio A, Tolosa de Talamoni N. Intestinal Na(+)/Ca(2+) exchanger protein and gene expression are regulated by 1,25(OH)(2)D(3) in vitamin D-deficient chicks. Arch Biochem Biophys. 2011;509:191-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 75. | Balesaria S, Sangha S, Walters JR. Human duodenum responses to vitamin D metabolites of TRPV6 and other genes involved in calcium absorption. Am J Physiol Gastrointest Liver Physiol. 2009;297:G1193-G1197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 62] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 76. | Jones G, Strugnell SA, DeLuca HF. Current understanding of the molecular actions of vitamin D. Physiol Rev. 1998;78:1193-1231. [PubMed] |
| 77. | Fahkri H, Zhang B, Fajol A, Hernando N, Elvira B, Mannheim JG, Pichler BJ, Daniel C, Amann K, Hirao A. Checkpoint kinase Chk2 controls renal Cyp27b1 expression, calcitriol formation, and calcium-phosphate metabolism. Pflugers Arch. 2014;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 78. | Cross HS, Nittke T, Kallay E. Colonic vitamin D metabolism: implications for the pathogenesis of inflammatory bowel disease and colorectal cancer. Mol Cell Endocrinol. 2011;347:70-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 79. | Ritter CS, Haughey BH, Armbrecht HJ, Brown AJ. Distribution and regulation of the 25-hydroxyvitamin D3 1α-hydroxylase in human parathyroid glands. J Steroid Biochem Mol Biol. 2012;130:73-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 80. | Khuituan P, Wongdee K, Jantarajit W, Suntornsaratoon P, Krishnamra N, Charoenphandhu N. Fibroblast growth factor-23 negates 1,25(OH)2D3-induced intestinal calcium transport by reducing the transcellular and paracellular calcium fluxes. Arch Biochem Biophys. 2013;536:46-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 81. | Fudge NJ, Kovacs CS. Pregnancy up-regulates intestinal calcium absorption and skeletal mineralization independently of the vitamin D receptor. Endocrinology. 2010;151:886-895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 88] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 82. | Gallagher JC. Vitamin D and aging. Endocrinol Metab Clin North Am. 2013;42:319-332. [PubMed] |
| 83. | Karbach U. Paracellular calcium transport across the small intestine. J Nutr. 1992;122:672-677. [PubMed] |
| 84. | Fujita H, Sugimoto K, Inatomi S, Maeda T, Osanai M, Uchiyama Y, Yamamoto Y, Wada T, Kojima T, Yokozaki H. Tight junction proteins claudin-2 and -12 are critical for vitamin D-dependent Ca2+ absorption between enterocytes. Mol Biol Cell. 2008;19:1912-1921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 321] [Cited by in RCA: 333] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 85. | Benn BS, Ajibade D, Porta A, Dhawan P, Hediger M, Peng JB, Jiang Y, Oh GT, Jeung EB, Lieben L. Active intestinal calcium transport in the absence of transient receptor potential vanilloid type 6 and calbindin-D9k. Endocrinology. 2008;149:3196-3205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 203] [Cited by in RCA: 157] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 86. | Kutuzova GD, Deluca HF. Gene expression profiles in rat intestine identify pathways for 1,25-dihydroxyvitamin D(3) stimulated calcium absorption and clarify its immunomodulatory properties. Arch Biochem Biophys. 2004;432:152-166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 120] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 87. | Gentili C, Morelli S, de Boland AR. Characterization of PTH/PTHrP receptor in rat duodenum: effects of ageing. J Cell Biochem. 2003;88:1157-1167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 88. | Fleet JC, Schoch RD. Molecular mechanisms for regulation of intestinal calcium absorption by vitamin D and other factors. Crit Rev Clin Lab Sci. 2010;47:181-195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 187] [Cited by in RCA: 148] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 89. | Cross HS, Debiec H, Peterlik M. Thyroid hormone enhances the genomic action of calcitriol in the small intestine. Prog Clin Biol Res. 1990;332:163-180. [PubMed] |
| 90. | Kumar V, Prasad R. Thyroid hormones stimulate calcium transport systems in rat intestine. Biochim Biophys Acta. 2003;1639:185-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 91. | Orihuela D. Inhibitory effect of aluminium on calcium absorption in small intestine of rats with different thyroid hormone status. J Inorg Biochem. 2009;103:1542-1547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 93. | Zoidis E, Gosteli-Peter M, Ghirlanda-Keller C, Meinel L, Zapf J, Schmid C. IGF-I and GH stimulate Phex mRNA expression in lungs and bones and 1,25-dihydroxyvitamin D(3) production in hypophysectomized rats. Eur J Endocrinol. 2002;146:97-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 28] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 94. | Fleet JC, Bruns ME, Hock JM, Wood RJ. Growth hormone and parathyroid hormone stimulate intestinal calcium absorption in aged female rats. Endocrinology. 1994;134:1755-1760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 95. | Fatayerji D, Mawer EB, Eastell R. The role of insulin-like growth factor I in age-related changes in calcium homeostasis in men. J Clin Endocrinol Metab. 2000;85:4657-4662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 96. | Bouillon R, Carmeliet G, Van Cromphaut S. Intestinal calcium absorption: lessons from knockout mice and men. Vitamin D. San Diego: Academic Press 2005; 429-452. |
| 97. | O’Loughlin PD, Morris HA. Oestrogen deficiency impairs intestinal calcium absorption in the rat. J Physiol. 1998;511:313-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 69] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 98. | van Abel M, Hoenderop JG, van der Kemp AW, van Leeuwen JP, Bindels RJ. Regulation of the epithelial Ca2+ channels in small intestine as studied by quantitative mRNA detection. Am J Physiol Gastrointest Liver Physiol. 2003;285:G78-G85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 118] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 99. | Cotter AA, Cashman KD. Effect of 17beta-oestradiol on transepithelial calcium transport in human intestinal-like Caco-2 cells and its interactions with 1,25-dihydroxycholecalciferol and 9-cis retinoic acid. Eur J Nutr. 2006;45:234-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 100. | Campbell-Thompson M, Lynch IJ, Bhardwaj B. Expression of estrogen receptor (ER) subtypes and ERbeta isoforms in colon cancer. Cancer Res. 2001;61:632-640. [PubMed] |
| 101. | Pahuja DN, DeLuca HF. Stimulation of intestinal calcium transport and bone calcium mobilization by prolactin in vitamin D-deficient rats. Science. 1981;214:1038-1039. [PubMed] |
| 102. | Charoenphandhu N, Limlomwongse L, Krishnamra N. Prolactin directly stimulates transcellular active calcium transport in the duodenum of female rats. Can J Physiol Pharmacol. 2001;79:430-438. [PubMed] |
| 103. | Ajibade DV, Dhawan P, Fechner AJ, Meyer MB, Pike JW, Christakos S. Evidence for a role of prolactin in calcium homeostasis: regulation of intestinal transient receptor potential vanilloid type 6, intestinal calcium absorption, and the 25-hydroxyvitamin D(3) 1alpha hydroxylase gene by prolactin. Endocrinology. 2010;151:2974-2984. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 58] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 104. | Charoenphandhu N, Nakkrasae LI, Kraidith K, Teerapornpuntakit J, Thongchote K, Thongon N, Krishnamra N. Two-step stimulation of intestinal Ca(2+) absorption during lactation by long-term prolactin exposure and suckling-induced prolactin surge. Am J Physiol Endocrinol Metab. 2009;297:E609-E619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 66] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 105. | Charoenphandhu N, Wongdee K, Krishnamra N. Is prolactin the cardinal calciotropic maternal hormone? Trends Endocrinol Metab. 2010;21:395-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 106. | Reid IR. Glucocorticoid osteoporosis--mechanisms and management. Eur J Endocrinol. 1997;137:209-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 172] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 107. | Van Cromphaut SJ, Stockmans I, Torrekens S, Van Herck E, Carmeliet G, Bouillon R. Duodenal calcium absorption in dexamethasone-treated mice: functional and molecular aspects. Arch Biochem Biophys. 2007;460:300-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 108. | Lee GS, Choi KC, Jeung EB. Glucocorticoids differentially regulate expression of duodenal and renal calbindin-D9k through glucocorticoid receptor-mediated pathway in mouse model. Am J Physiol Endocrinol Metab. 2006;290:E299-E307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 40] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 109. | Kim MH, Lee GS, Jung EM, Choi KC, Jeung EB. The negative effect of dexamethasone on calcium-processing gene expressions is associated with a glucocorticoid-induced calcium-absorbing disorder. Life Sci. 2009;85:146-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 110. | Huybers S, Naber TH, Bindels RJ, Hoenderop JG. Prednisolone-induced Ca2+ malabsorption is caused by diminished expression of the epithelial Ca2+ channel TRPV6. Am J Physiol Gastrointest Liver Physiol. 2007;292:G92-G97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 89] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 111. | Prentice A. Maternal calcium metabolism and bone mineral status. Am J Clin Nutr. 2000;71:1312S-1316S. [PubMed] |
| 112. | O’Brien KO, Nathanson MS, Mancini J, Witter FR. Calcium absorption is significantly higher in adolescents during pregnancy than in the early postpartum period. Am J Clin Nutr. 2003;78:1188-1193. [PubMed] |
| 113. | Yamagishi N, Sassa H, Sato R, Taniguchi K, Okura N, Sato S, Naito Y. Calcium metabolism of pregnant rats fed a vitamin D-depleted diet. J Vet Med Sci. 2007;69:441-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 114. | Kirby BJ, Ma Y, Martin HM, Buckle Favaro KL, Karaplis AC, Kovacs CS. Upregulation of calcitriol during pregnancy and skeletal recovery after lactation do not require parathyroid hormone. J Bone Miner Res. 2013;28:1987-2000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 59] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 115. | Teerapornpuntakit J, Klanchui A, Karoonuthaisiri N, Wongdee K, Charoenphandhu N. Expression of transcripts related to intestinal ion and nutrient absorption in pregnant and lactating rats as determined by custom-designed cDNA microarray. Mol Cell Biochem. 2014;391:103-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 116. | Hong EJ, Jeung EB. Biological significance of calbindin-D9k within duodenal epithelium. Int J Mol Sci. 2013;14:23330-23340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 117. | Zhu Y, Goff JP, Reinhardt TA, Horst RL. Pregnancy and lactation increase vitamin D-dependent intestinal membrane calcium adenosine triphosphatase and calcium binding protein messenger ribonucleic acid expression. Endocrinology. 1998;139:3520-3524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 118. | Yang H, Ahn C, Jeung EB. Differential expression of calcium transport genes caused by COMT inhibition in the duodenum, kidney and placenta of pregnant mice. Mol Cell Endocrinol. 2015;401:45-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 119. | Pelletier G, de Passillé AM, Bernier-Cardou M, Morisset J. Influence of pregnancy, lactation, litter size and diet energy density on the stomach and intestine of sows. J Nutr. 1987;117:1759-1766. [PubMed] |
| 120. | Hammond KA. Adaptation of the maternal intestine during lactation. J Mammary Gland Biol Neoplasia. 1997;2:243-252. [PubMed] |
| 121. | Wongdee K, Teerapornpuntakit J, Siangpro C, Chaipai S, Charoenphandhu N. Duodenal villous hypertrophy and upregulation of claudin-15 protein expression in lactating rats. J Mol Histol. 2013;44:103-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 122. | Wongdee K, Charoenphandhu N. Regulation of epithelial calcium transport by prolactin: from fish to mammals. Gen Comp Endocrinol. 2013;181:235-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 123. | Halloran BP, DeLuca HF. Calcium transport in small intestine during early development: role of vitamin D. Am J Physiol. 1980;239:G473-G479. [PubMed] |
| 124. | Kovacs CS. Bone development and mineral homeostasis in the fetus and neonate: roles of the calciotropic and phosphotropic hormones. Physiol Rev. 2014;94:1143-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 153] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 125. | Abrams SA. Calcium turnover and nutrition through the life cycle. Proc Nutr Soc. 2001;60:283-289. [PubMed] |
| 126. | Seiquer I, López-Frías M, Muñoz-Hoyos A, Galdó G, Delgado-Andrade C, Mesías M, Navarro MP. Dietary calcium utilization among a group of Spanish boys aged 11-14 years on their usual diets. J Physiol Biochem. 2006;62:9-16. [PubMed] |
| 127. | Abrams SA, Griffin IJ, Hawthorne KM, Chen Z, Gunn SK, Wilde M, Darlington G, Shypailo RJ, Ellis KJ. Vitamin D receptor Fok1 polymorphisms affect calcium absorption, kinetics, and bone mineralization rates during puberty. J Bone Miner Res. 2005;20:945-953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 91] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 128. | Nordin BE, Need AG, Morris HA, O’Loughlin PD, Horowitz M. Effect of age on calcium absorption in postmenopausal women. Am J Clin Nutr. 2004;80:998-1002. [PubMed] |
| 129. | Ramsubeik K, Keuler NS, Davis LA, Hansen KE. Factors associated with calcium absorption in postmenopausal women: a post hoc analysis of dual-isotope studies. J Acad Nutr Diet. 2014;114:761-767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 130. | Ebeling PR, Sandgren ME, DiMagno EP, Lane AW, DeLuca HF, Riggs BL. Evidence of an age-related decrease in intestinal responsiveness to vitamin D: relationship between serum 1,25-dihydroxyvitamin D3 and intestinal vitamin D receptor concentrations in normal women. J Clin Endocrinol Metab. 1992;75:176-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 36] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 131. | Song Y, Fleet JC. Intestinal resistance to 1,25 dihydroxyvitamin D in mice heterozygous for the vitamin D receptor knockout allele. Endocrinology. 2007;148:1396-1402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 1.7] [Reference Citation Analysis (0)] |