Published online Jan 7, 2015. doi: 10.3748/wjg.v21.i1.124
Peer-review started: July 13, 2014
First decision: August 6, 2014
Revised: September 16, 2014
Accepted: November 7, 2014
Article in press: November 11, 2014
Published online: January 7, 2015
Processing time: 178 Days and 10.1 Hours
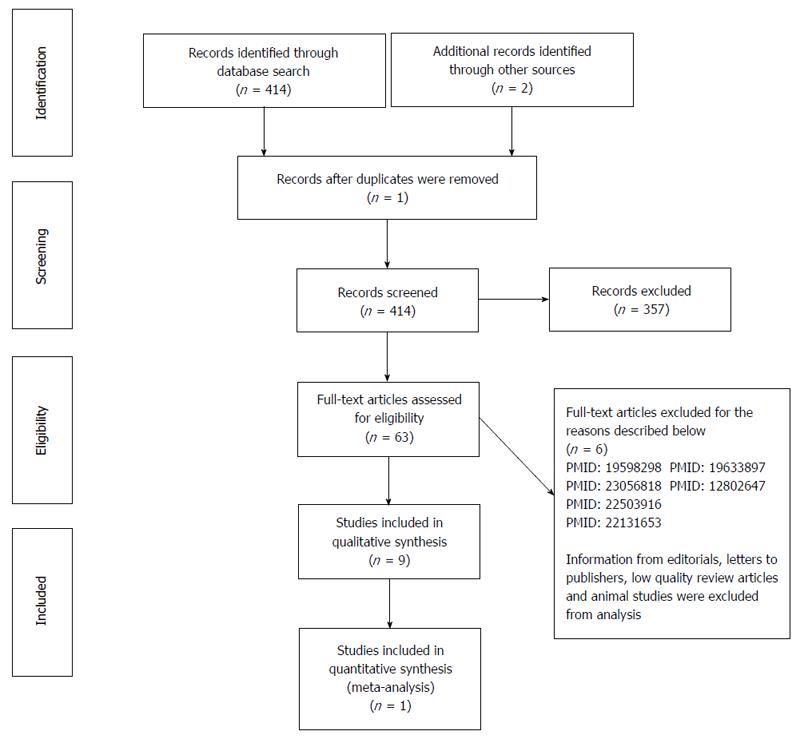
A review was carried out in Medline, LILACS and the Cochrane Library. Our database search strategy included the following terms: “hydatid cyst”, “liver”, “management”, “meta-analysis” and “randomized controlled trial”. No language limits were used in the literature search. The latest electronic search date was the 7th of January 2014. Inclusion and exclusion criteria: all relevant studies on the assessment of therapeutic methods for hydatid cysts of the liver were considered for analysis. Information from editorials, letters to publishers, low quality review articles and studies done on animals were excluded from analysis. Additionally, well-structured abstracts from relevant articles were selected and accepted for analysis. Standardized forms were designed for data extraction; two investigators entered the data on patient demographics, methodology, recurrence of HC, mean cyst size and number of cysts per group. Four hundred and fourteen articles were identified using the previously described search strategy. After applying the inclusion and exclusion criteria detailed above, 57 articles were selected for final analysis: one meta-analysis, 9 randomized clinical trials, 5 non-randomized comparative prospective studies, 7 non-comparative prospective studies, and 34 retrospective studies (12 comparative and 22 non-comparative). Our results indicate that antihelminthic treatment alone is not the ideal treatment for liver hydatid cysts. More studies in the literature support the effectiveness of radical treatment compared with conservative treatment. Conservative surgery with omentoplasty is effective in preventing postoperative complications. A laparoscopic approach is safe in some situations. Percutaneous drainage with albendazole therapy is a safe and effective alternative treatment for hydatid cysts of the liver. Radical surgery with pre- and post-operative administration of albendazole is the best treatment option for liver hydatid cysts due to low recurrence and complication rates.
Core tip: Hepatic hydatid disease still exists worldwide. Because of human migration, surgeons in developed countries are encountering patients with hepatic hydatid disease more frequently but may be unfamiliar with the treatment options for this condition. This review article is particularly important because there are no current updates of evidence-based practices for the treatment of hydatid cysts of the liver. Thus, a careful search of the published literature pertaining to this disorder was carried out.
- Citation: Gomez i Gavara C, López-Andújar R, Belda Ibáñez T, Ramia Ángel JM, Moya Herraiz &, Orbis Castellanos F, Pareja Ibars E, San Juan Rodríguez F. Review of the treatment of liver hydatid cysts. World J Gastroenterol 2015; 21(1): 124-131
- URL: https://www.wjgnet.com/1007-9327/full/v21/i1/124.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i1.124
Hydatid cyst (HC), or hydatidosis, is a global parasitic zoonosis[1] that is endemic to countries in the Mediterranean, Turkey, Australia, North Africa, Australia, New Zealand, South America, the Philippines, Northern China and the Indian subcontinent. Like other cestoda, Echinococcus has intermediate and definitive hosts. Canidae, especially dogs, are the definitive hosts; ruminants may also host the organism. Sheep and goats are intermediate hosts, although E. granulosus can also be hosted by a wide range of vertebrates. The diagnosis of non-complicated HC of the liver is based on clinical suspicion. Ultrasonography and computed tomography are the most important diagnostic tools and are helpful for identifying any associated complications and for planning treatment[2].
Although there is abundant literature on hydatidosis, few randomized controlled trials (RCT) have been conducted. The literature consists mainly of retrospective studies, albeit with larger sample sizes than the prospective studies. The problem with these reports is that although many have been published about liver hydatid cyst treatment, most present conflicting results.
Treatment of HC of the liver can include medical therapy, percutaneous drainage, or surgical intervention (via a conventional or laparoscopic approach).
The aim of this review was to provide evidence-based answers to questions about the effectiveness of chemotherapy, percutaneous drainage and surgery in the treatment of HC of the liver.
Contraindications to surgery include complex or widespread injury, advanced patient age, pregnancy, comorbidities, multiple cysts that are difficult to access, partially inactive or calcified liver cysts, or patient refusal of surgery[2-4].
The use of benzimidazole compound derivatives, albendazole (ALB) and mebendazole (MBZ), has been studied in 7 RCTs (Figure 1 and Table 1).
| RCT | Year | Treatment | Endpoint | Size | Number of cysts per group | Mean cyst size (cm) | P < 0.05 | Jadad Score |
| Khuroo et al[34] | 1993 | ALB flow diagram s PD vs ALB+ PD | Echo P | 33 | 10, 12, 11 | 9.2 ± 4.4; 10.8 ± 3; 8.8 ± 4.5 | Yes | 2 |
| ALB + PD > ALB alone | ||||||||
| Gil-Grande et al[7] | 1993 | ALB 1-3 mo vs PLB + S | Viability | 55 | 18, 18, 19 | 10.4 ± 4; 8.91± 4; 10.5 ± 5 | Yes | 5 |
| ALB 3 mo > ALB 1 mo > no ALB | ||||||||
| Khuroo et al[35] | 1997 | ALB + PD vs S alone | Echo P | 50 | 25, 25 | 8 ± 3; 9.1 ± 3 | Yes | 3 |
| ALB + PD > S alone | ||||||||
| Dziri et al[54] | 1999 | OP vs no OP | Deep abscess | 115 | 58, 57 | 50; 502 | Yes | 2 |
| OP > no OP | ||||||||
| Franchi et al[6] | 1999 | ALB vs MBZ | Echo P | 448 | 178, 440 | - | Yes | 2 |
| ALB > MBZ | ||||||||
| Keshmiri et al[8] | 2001 | ALB vs PLB | Echo P | 29 | 49, 191 | 74.01 ± 130.79; 198.13 ± 403.711 | Yes | 4 |
| ALB > PLB | ||||||||
| Bildik et al[9] | 2007 | ALB 1-3 mo vs no ALB + S | Viability | 84 | - | - | Yes | 1 |
| ALB 3 mo > no ALB | ||||||||
| Yüksel et al[15] | 2008 | Radical S vs conservative | Early recurrence; cavity complications | 32 | 15, 17 | 7.8; 8.1 | Yes | 3 |
| Radical > Conservative | ||||||||
| Shams-Ul-Bari et al[29] | 2011 | ALB + S vs S alone | Viability | 72 | 36, 36 | - | Yes | 3 |
| ALB + S > S alone |
From 1984 to 1986, the World Health Organization (WHO) carried out two multicenter trials in Europe to compare ALB and MBZ. Both drugs had similar efficacy, but MBZ required higher doses and a different treatment duration[5]. In the RCT conducted by Franchi et al[6] (Table 1) that compared the effectiveness of ALB and MBZ, treatment with ALB was objectively superior.
From a review of the RCTs, we can conclude that ALB is superior to placebo (Gil-Grande et al[7] and Keshmiri et al[8]) (Table 1). Compared with a shorter treatment course, three months of oral treatment appears to have better outcomes in terms of cyst degeneration (based on imaging) and cure rates. However, in 5 of the 7 clinical trials published, the cure rate for HC treated with ALB alone was below 60%. Only trials that included 3 mo of ALB treatment and surgery had cure rates > 90%. Based on the results of published RCTs, benzimidazole derivatives alone cannot fully eliminate cysts.
Radical surgery for liver HC refers to pericystectomy and liver resection, whereas conservative surgery involves the removal of the cyst content and sterilization of the residual cavity, together with partial cyst resection.
There are several non-comparative retrospective studies concluding that radical surgery is both safe and effective[9-12].
A comparative retrospective study by Aydin et al[13] of 242 patients described significantly higher morbidity and recurrence rates in patients who underwent conservative surgery (11% vs 3%; 24% vs 3%).
In another comparative retrospective study by Tagliacozzo et al[14] that included 454 patients, two hundred and fourteen underwent conservative surgery (external drainage, marsupialization or omentoplasty), while the remaining 240 were treated with radical surgery. Morbidity and recurrence rates were significantly higher in the group that underwent conservative surgery.
Only one randomized study[15], involving 32 patients, compared radical surgery and conservative surgery. The authors concluded that conservative surgery leads to a significantly higher early recurrence rate (P = 0.045) compared to radical surgery, as well as a higher rate of complications in the residual cyst cavity (P = 0.011).
Surgery can be curative if the parasite is removed completely. Frequently, surgical procedures are inconsistent and comprise liver resection or the opening of the parasitic cysts and subsequent removal of the parasite. Conservative procedures are safe and less complex than radical surgery, even though the risk of associated morbidity may be higher due to the presence of the residual cavity.
A variety of techniques[16] have been described (omentoplasty, introflexion, capitonage, external drainage or synthetic fibrin) to prevent postoperative complications caused by the presence of the residual cavity following conservative surgery .
A comparative retrospective study of 304 patients by Balik et al[17] concluded that external drainage had a significantly higher rate of complications (e.g., infection of the residual cavity and biliary fistulization) than omentoplasty or capitonage.
Another comparative study by Mentes et al[18] found that omentoplasty had fewer complications and a shorter hospital stay than introflexion or a combination of the two procedures.
A Greek team, led by Vagianos et al[19], published a study evaluating the result of adding omentoplasty to conservative surgery (cystectomy and drainage). Objectively, their results did not show any improvement when omentoplasty was combined with external drainage. However, the drawback of this study was that the omentoplasty was an added procedure and, as such, did not compare the effectiveness of drains vs omentoplasty.
According to the RCT by Dziri et al[16] and retrospective studies by Utkan et al[20] and Reza Mousavi et al[21], omentoplasty alone leads to fewer complications than external drainage (Table 1).
Finally, the use of fibrin glue has been assessed in non-randomized studies both for radical (Cois et al[22]) and conservative surgery (Hofstetter et al[23]), and has been shown not to play a significant role.
In conclusion, omentoplasty is effective in preventing complications from conservative surgery in the treatment of liver HC.
The use of a laparoscopic approach for managing liver HC was first documented in 1992[24]. The advantages of a laparoscopic approach compared to open surgery are a shorter hospital stay, a lower incidence of wound infection and less postoperative pain. The disadvantages are difficulties in accessing cysts in certain locations, an increased risk of cyst content spillage, and difficulty in aspirating cysts with viscous contents[16].
No randomized study has evaluated a laparoscopic approach to the treatment of liver HC, and most published studies are non-comparative.
Katkhouda et al[25] and Descottes et al[26] published results on patients with benign solid lesions or liver cysts and concluded that the laparoscopic approach was safe for selected patients. However, in his study of 43 patients, Katkhouda advised against using this approach for HC (n = 6) due to the high incidence of postoperative complications observed. The most important literature studies involving laparoscopy of liver HC are shown in Table 2. All the studies reported have observed that a laparoscopic approach is safe for the treatment of HC, with objectively low conversion rates and no mortality cases.
| Ref. | Year | Study design | Size | Surgical modality | Conversion1 | Complicationrate2 | Recurrencerate | Follow-up(mo) |
| Emel´ianov et al[56] | 2000 | Prospective | 37 | Partial cystectomy | 3% | 10% | - | |
| Non-comparative | ||||||||
| Khuory et al[57] | 2000 | Prospective | 108 | Marsupialization3 | 11% | 3% | 3.6% | 4-54 |
| Non-comparative | ||||||||
| Bickel et al[58] | 2001 | Prospective | 31 | Puncture + parasite neutralization + evacuation | 3% | 16% | 0% | 9-97 |
| Non-comparative | ||||||||
| Polat[59] | 2012 | Retrospective | Open n = 12 | Cystectomy | 13% | 0% | 24 | |
| Comparative | Lap4n = 7 | 0% | 14% | 0% | ||||
| Zaharie et al[60] | 2013 | Retrospective | Open n = 271 | Partial cystectomy | 14% | 0% | 6-40 | |
| Comparative | Lap4n = 62 | 4.85% | 0% | 0% | 6-32 |
In conclusion, a laparoscopic surgical approach for liver HC is safe.
A non-randomized prospective study of 70 patients by Aktan et al[27], which compared preoperative administration of ALB for three weeks vs no ALB treatment, found a significantly higher number of non-viable liver cysts in patients treated with ALB.
Türkçapar et al[28] published a non-comparative prospective study in which ALB was administered one month before surgery and then again 2 mo after surgery to 25 patients. In two cases, drug treatment had to be suspended due to abnormal liver enzymes. In the remaining 23 patients, no recurrences were observed, with an average follow-up period of 29 mo.
In a RCT of 72 patients by Shams-Ul-Bari et al[29], patients were randomized to two groups: group A received ALB for 12 mo before and after surgery, whereas group B went straight to surgery. The recurrence rate in the surgery-alone group was 16%, whereas in the group treated with ALB, no recurrences were detected (P < 0.01) at an average follow-up of 5 years.
Another randomized study by the same group (Arif et al[30]) divided 64 patients into 4 groups: surgery alone, treatment with ALB and then surgery, ALB followed by surgery and postoperative ALB, or surgery followed by ALB. Lower recurrence rates were observed in patients treated with ALB (4.16% vs 18.75%).
Combined treatment with ALB + surgery leads to a lower risk of recurrence than surgery alone.
Two non-comparative prospective studies[31,32] of 34 and 87 patients treated with percutaneous drainage concluded that it is a safe and effective treatment that can be used in patients with contraindications to surgery and for types I-III disease, according to Gharbi et al[33]. A randomized study involving 33 patients supported the effectiveness of treatment with oral ALB alone, percutaneous drainage/ALB/puncture, aspiration, injection, re-aspiration (PAIR), or combining PAIR. Oral ALB showed the best results in terms of cyst size reduction (P < 0.05) when ALB and PAIR were combined[34].
The results of these studies suggest that percutaneous drainage combined with ALB is a safe and effective treatment for liver HC.
A randomized study of 50 patients published by Khuroo et al[35] that compared treatment with ALB and percutaneous drainage vs surgery alone (cystectomy and cavity drainage) concluded that percutaneous treatment with ALB is a safe and effective treatment for uncomplicated HC and is associated with a shorter hospital stay.
In a meta-analysis of 21 studies, Smego et al[36] compared 769 patients who received ALB/MBZ treatment and PAIR to 952 patients treated with surgery alone. They concluded that PAIR in conjunction with ALB/MBZ is more effective than surgery and is associated with lower rates of morbidity and mortality, decreased recurrence risk and a shorter hospital stay. However, these results were obtained considering surgical interventions, conservative or radical approaches in the same group.
A comparative retrospective study by Gupta et al[37] compared 128 patients. The rate of biliary fistulae and residual cavity abscesses was significantly higher in the group treated with conservative surgery (P = 0.032 and P = 0.001, respectively).
PAIR may be a promising treatment for liver HC, but there is not sufficient evidence to support its use as a standard procedure for patients with uncomplicated cysts[38]. There is a need for well-designed clinical trials to confirm these results.
Age (> 40 years), cyst size (> 10 cm), the number of cysts (> 3), complicated HC and conservative surgery have been identified as high-risk factors in HC patients in 2 retrospective studies, as these factors significantly increase morbidity and mortality rates[39-41].
Intrabiliary rupture is the most common complication of hepatic HC; however, it is unusual, occurring in only 3%-17% of cases[42].
A retrospective study of 63 patients with HC who received preoperative CT scans concluded that the presence of certain radiological signs (pericystic ducts and exophytic components) is a contraindication to non-surgical management because of the danger of biliary obstruction.
There are very few studies evaluating the treatment of intrabiliary rupture. A retrospective study[43] of 80 patients revealed that morbidity rates following choledochoduodenostomy or T-tube drainage of the common bile duct were 40% and 18%, respectively. A multicenter study[44] reported a morbidity rate of 49%, 11% and 62% for suture, transfistulary drainage and direct fistulization, respectively.
Endoscopic retrograde cholangiopancreatography (ERCP) defines the cystobiliary relationship to guide management decisions during the pre- and postoperative periods. Preoperative endoscopic sphincterotomy may decrease the incidence of postoperative external fistula from 11.1% to 7.6%[45]. During the postoperative period, ERCP may provide an opportunity to manage postoperative external biliary fistulae[46].
All studies[47-53] available in the literature agreed with the strategy of conducting common bile duct exploration with intraoperative cholangiography and choledoscopy; when the biliary tract is cleared of all cystic content, T tube drainage is sufficient treatment. ERCP may be useful in decreasing and managing the incidence of postoperative external fistulae.
This review offers evidence-based answers to questions about the effective treatment of liver HC. The limitations of this review are due to the different surgical modalities used in the published articles and to the fact that some studies are not comparable.
MBZ was the first drug used to treat hydatid disease. Later, ALB was introduced because of its superior absorption properties.
In an RCT conducted by Gil-Grande et al[7], all patients underwent surgery: 18 patients received no ALB treatment, 18 received ALB (10 mg/kg daily) for 1 mo, and 19 received the drug for approximately 3 mo. Eight cysts (50%) in the control group, 13 (72%) in the group who received ALB 1 for month, and 16 (94%) for the group who received ALB for 3 mo were non-viable (P = 0.015). Similar results were obtained in a study performed by Keshmiri et al[8], in which 29 patients with 240 cysts were randomized to receive ALB (3 mo) or placebo. The best results were obtained when ALB treatment accompanied surgery.
Multiple retrospective studies[10-14] have been published comparing radical vs conservative surgery. In spite of its small sample size, the only randomized trial, conducted by Yüksel et al[15], found that conservative surgery was associated with significantly higher recurrence and morbidity rates. These results are consistent with previous retrospective studies.
Some situations require conservative surgery, e.g., for cysts located close to major biliovascular channels. In an RCT conducted by Dziri et al[54], 115 consecutive patients were randomly assigned to treatment with omentoplasty (OP; n = 58) or not (NO; n = 57). Fewer deep abdominal complications occurred in the OP (10%) than in the NO (23%) group, as well as fewer deep abdominal abscesses (0% vs 11%, P < 0.03). Omentoplasty is the most effective option for preventing postoperative complications following conservative surgery.
There is no RCT comparing open vs laparoscopic treatment for hydatid disease of the liver. Nevertheless, a paired study by Zaharie et al[60], with comparable groups and a sample size of 62, showed that a laparoscopic approach is safe for the treatment of HC in almost all segments of the liver.
Percutaneous drainage combined with ALB therapy is better than monotherapy, and additionally, it is safe and effective for the treatment of liver HC; however, complications are possible. Khuroo et al[34] reported that complications associated with drainage included cyst infections in 2 patients, fever in 3, rupture of the cyst into biliary structures in 1, and skin rash in 2, although these complications were managed successfully without any mortality.
PAIR treatment seems to be promising, but in the published literature it has been compared principally with conservative surgery and not with radical surgery. Only one retrospective study, by Gupta et al[37], compared 128 patients with liver HC who were treated with PAIR/PAIR-drainage (n = 52), radical / excisional surgery (n = 61) and conservative surgery (n = 33). In ten patients, the PAIR procedure was stopped due to aspiration of either bile or pultaceous material after the initial puncture. These patients subsequently underwent surgery. Presently, there is no evidence to support PAIR as a standard treatment and further well-designed studies are necessary prior to recommending it.
Finally, there is little available evidence concerning the treatment of complicated HC of the liver and disseminated echinococcal cysts, though it is unquestioned. By defining the cystobiliary relationship, ERCP helps to guide management decisions during the pre- and postoperative periods.
This review offers evidence-based answers to questions about the effective treatment of liver HC. The limitations of this review are due to the different surgical modalities used in the published articles and to the fact that some studies are not comparable.
Antihelminthic treatment is superior to placebo but is not the ideal treatment for HC of the liver when used alone. The level of evidence for this recommendation is 2a, and the recommendation is grade B[55]. The majority of published studies conclude that radical surgery is a better option than conservative treatment, with a level of evidence of 2b and recommendation grade B[55]. Omentoplasty associated with conservative surgical treatment is effective in preventing postoperative complications, with a level of evidence of 2b and recommendation grade B[55]. Further studies are necessary to evaluate recurrence rates following laparoscopic surgery for liver HC, with a level of evidence of 4 and recommendation grade C[55]. Combined treatment with ALB + surgery leads to a lower risk of recurrence compared with surgery alone, with a level of evidence of 2 and recommendation grade C[55].
P- Reviewer: Borzio M, El-Bendary M, Shi Z, Yonem O S- Editor: Qi Y L- Editor: A E- Editor: Wang CH
| 1. | Buttenschoen K, Carli Buttenschoen D. Echinococcus granulosus infection: the challenge of surgical treatment. Langenbecks Arch Surg. 2003;388:218-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 70] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 2. | Sayek I, Onat D. Diagnosis and treatment of uncomplicated hydatid cyst of the liver. World J Surg. 2001;25:21-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 119] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 3. | Junghanss T, da Silva AM, Horton J, Chiodini PL, Brunetti E. Clinical management of cystic echinococcosis: state of the art, problems, and perspectives. Am J Trop Med Hyg. 2008;79:301-311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 4. | Perdomo R, Alvarez C, Monti J, Ferreira C, Chiesa A, Carbó A, Alvez R, Grauert R, Stern D, Carmona C. Principles of the surgical approach in human liver cystic echinococcosis. Acta Trop. 1997;64:109-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Davis A, Dixon H, Pawlowski ZS. Multicentre clinical trials of benzimidazole-carbamates in human cystic echinococcosis (phase 2). Bull World Health Organ. 1989;67:503-508. [PubMed] |
| 6. | Franchi C, Di Vico B, Teggi A. Long-term evaluation of patients with hydatidosis treated with benzimidazole carbamates. Clin Infect Dis. 1999;29:304-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 113] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 7. | Gil-Grande LA, Rodriguez-Caabeiro F, Prieto JG, Sánchez-Ruano JJ, Brasa C, Aguilar L, García-Hoz F, Casado N, Bárcena R, Alvarez AI. Randomised controlled trial of efficacy of albendazole in intra-abdominal hydatid disease. Lancet. 1993;342:1269-1272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 148] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 8. | Keshmiri M, Baharvahdat H, Fattahi SH, Davachi B, Dabiri RH, Baradaran H, Rajabzadeh F. Albendazole versus placebo in treatment of echinococcosis. Trans R Soc Trop Med Hyg. 2001;95:190-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 53] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 9. | Bildik N, Cevik A, Altintaş M, Ekinci H, Canberk M, Gülmen M. Efficacy of preoperative albendazole use according to months in hydatid cyst of the liver. J Clin Gastroenterol. 2007;41:312-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 10. | Berrada S, Essadki B, Zerouali NO. [Hydatid cyst of the liver. Treatment by resection of the cyst wall. Our experience apropos of a series of 495 cases]. Ann Chir. 1993;47:510-512. [PubMed] |
| 11. | Alfieri S, Doglietto GB, Pacelli F, Costamagna G, Carriero C, Mutignani M, Liberatori M, Crucitti F. Radical surgery for liver hydatid disease: a study of 89 consecutive patients. Hepatogastroenterology. 1997;44:496-500. [PubMed] |
| 12. | Alonso Casado O, Moreno González E, Loinaz Segurola C, Gimeno Calvo A, González Pinto I, Pérez Saborido B, Paseiro Crespo G, Ortiz Johansson C. Results of 22 years of experience in radical surgical treatment of hepatic hydatid cysts. Hepatogastroenterology. 2001;48:235-243. [PubMed] |
| 13. | Aydin U, Yazici P, Onen Z, Ozsoy M, Zeytunlu M, Kiliç M, Coker A. The optimal treatment of hydatid cyst of the liver: radical surgery with a significant reduced risk of recurrence. Turk J Gastroenterol. 2008;19:33-39. [PubMed] |
| 14. | Tagliacozzo S, Miccini M, Amore Bonapasta S, Gregori M, Tocchi A. Surgical treatment of hydatid disease of the liver: 25 years of experience. Am J Surg. 2011;201:797-804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 47] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 15. | Yüksel O, Akyürek N, Sahin T, Salman B, Azili C, Bostanci H. Efficacy of radical surgery in preventing early local recurrence and cavity-related complications in hydatic liver disease. J Gastrointest Surg. 2008;12:483-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 45] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 16. | Dziri C, Haouet K, Fingerhut A. Treatment of hydatid cyst of the liver: where is the evidence? World J Surg. 2004;28:731-736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 133] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 17. | Balik AA, Başoğlu M, Celebi F, Oren D, Polat KY, Atamanalp SS, Akçay MN. Surgical treatment of hydatid disease of the liver: review of 304 cases. Arch Surg. 1999;134:166-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 79] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 18. | Mentes A, Yüzer Y, Ozbal O, Coker A, Ilter T, Musoğlu A. Omentoplasty versus introflexion for hydatid liver cysts. J R Coll Surg Edinb. 1993;38:82-85. [PubMed] |
| 19. | Vagianos CE, Karavias DD, Kakkos SK, Vagenas CA, Androulakis JA. Conservative surgery in the treatment of hepatic hydatidosis. Eur J Surg. 1995;161:415-420. [PubMed] |
| 20. | Utkan NZ, Cantürk NZ, Gönüllü N, Yildirir C, Dülger M. Surgical experience of hydatid disease of the liver: omentoplasty or capitonnage versus tube drainage. Hepatogastroenterology. 2001;48:203-207. [PubMed] |
| 21. | Reza Mousavi S, Khoshnevis J, Kharazm P. Surgical treatment of hydatid cyst of the liver: drainage versus omentoplasty. Ann Hepatol. 2005;4:272-274. [PubMed] |
| 22. | Cois A, Iasiello G, Nardello O, Mattana A, Uccheddu A, Cagetti M. [Human fibrin glue in the treatment of residual parenchymal surface after total pericystectomy for hepatic echinococcus]. Ann Ital Chir. 1997;68:701-706; discussion 706-709. [PubMed] |
| 23. | Hofstetter C, Segovia E, Vara-Thorbeck R. Treatment of uncomplicated hydatid cyst of the liver by closed marsupialization and fibrin glue obliteration. World J Surg. 2004;28:173-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 24. | Katkhouda N, Fabiani P, Benizri E, Mouiel J. Laser resection of a liver hydatid cyst under videolaparoscopy. Br J Surg. 1992;79:560-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 81] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 25. | Katkhouda N, Hurwitz M, Gugenheim J, Mavor E, Mason RJ, Waldrep DJ, Rivera RT, Chandra M, Campos GM, Offerman S. Laparoscopic management of benign solid and cystic lesions of the liver. Ann Surg. 1999;229:460-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 127] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 26. | Descottes B, Glineur D, Lachachi F, Valleix D, Paineau J, Hamy A, Morino M, Bismuth H, Castaing D, Savier E. Laparoscopic liver resection of benign liver tumors. Surg Endosc. 2003;17:23-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 176] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 27. | Aktan AO, Yalin R. Preoperative albendazole treatment for liver hydatid disease decreases the viability of the cyst. Eur J Gastroenterol Hepatol. 1996;8:877-879. [PubMed] |
| 28. | Türkçapar AG, Ersöz S, Güngör C, Aydinuraz K, Yerdel MA, Aras N. Surgical treatment of hepatic hydatidosis combined with perioperative treatment with albendazole. Eur J Surg. 1997;163:923-928. [PubMed] |
| 29. | Shams-Ul-Bari NA, Arif SH, Malik AA, Khaja AR, Dass TA, Naikoo ZA. Role of albendazole in the management of hydatid cyst liver. Saudi J Gastroenterol. 2011;17:343-347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 67] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 30. | Arif SH, Shams-Ul-Bari NA, Zargar SA, Wani MA, Tabassum R, Hussain Z, Baba AA, Lone RA. Albendazole as an adjuvant to the standard surgical management of hydatid cyst liver. Int J Surg. 2008;6:448-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 95] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 31. | Ormeci N, Soykan I, Bektas A, Sanoğlu M, Palabiyikoğlu M, Hadi Yasa M, Dökmeci A, Uzunalimoğlu O. A new percutaneous approach for the treatment of hydatid cysts of the liver. Am J Gastroenterol. 2001;96:2225-2230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 32. | Peláez V, Kugler C, Correa D, Del Carpio M, Guangiroli M, Molina J, Marcos B, Lopez E. PAIR as percutaneous treatment of hydatid liver cysts. Acta Trop. 2000;75:197-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 38] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 33. | Gharbi HA, Hassine W, Brauner MW, Dupuch K. Ultrasound examination of the hydatic liver. Radiology. 1981;139:459-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 528] [Cited by in RCA: 487] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 34. | Khuroo MS, Dar MY, Yattoo GN, Zargar SA, Javaid G, Khan BA, Boda MI. Percutaneous drainage versus albendazole therapy in hepatic hydatidosis: a prospective, randomized study. Gastroenterology. 1993;104:1452-1459. [PubMed] |
| 35. | Khuroo MS, Wani NA, Javid G, Khan BA, Yattoo GN, Shah AH, Jeelani SG. Percutaneous drainage compared with surgery for hepatic hydatid cysts. N Engl J Med. 1997;337:881-887. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 194] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 36. | Smego RA, Bhatti S, Khaliq AA, Beg MA. Percutaneous aspiration-injection-reaspiration drainage plus albendazole or mebendazole for hepatic cystic echinococcosis: a meta-analysis. Clin Infect Dis. 2003;37:1073-1083. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 138] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 37. | Gupta N, Javed A, Puri S, Jain S, Singh S, Agarwal AK. Hepatic hydatid: PAIR, drain or resect? J Gastrointest Surg. 2011;15:1829-1836. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 49] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 38. | Nasseri-Moghaddam S, Abrishami A, Taefi A, Malekzadeh R. Percutaneous needle aspiration, injection, and re-aspiration with or without benzimidazole coverage for uncomplicated hepatic hydatid cysts. Cochrane Database Syst Rev. 2011;CD003623. [PubMed] |
| 39. | Daradkeh S, El-Muhtaseb H, Farah G, Sroujieh AS, Abu-Khalaf M. Predictors of morbidity and mortality in the surgical management of hydatid cyst of the liver. Langenbecks Arch Surg. 2007;392:35-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 59] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 40. | El Malki HO, El Mejdoubi Y, Souadka A, Mohsine R, Ifrine L, Abouqal R, Belkouchi A. Predictive factors of deep abdominal complications after operation for hydatid cyst of the liver: 15 years of experience with 672 patients. J Am Coll Surg. 2008;206:629-637. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 48] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 41. | Kilic M, Yoldas O, Koc M, Keskek M, Karakose N, Ertan T, Gocmen E, Tez M. Can biliary-cyst communication be predicted before surgery for hepatic hydatid disease: does size matter? Am J Surg. 2008;196:732-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 45] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 42. | Kumar R, Reddy SN, Thulkar S. Intrabiliary rupture of hydatid cyst: diagnosis with MRI and hepatobiliary isotope study. Br J Radiol. 2002;75:271-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 43. | Elbir O, Gundogdu H, Caglikulekci M, Kayaalp C, Atalay F, Savkilioglu M, Seven C. Surgical treatment of intrabiliary rupture of hydatid cysts of liver: comparison of choledochoduodenostomy with T-tube drainage. Dig Surg. 2001;18:289-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 44. | Zaouche A, Haouet K, Jouini M, El Hachaichi A, Dziri C. Management of liver hydatid cysts with a large biliocystic fistula: multicenter retrospective study. Tunisian Surgical Association. World J Surg. 2001;25:28-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 81] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 45. | Galati G, Sterpetti AV, Caputo M, Adduci M, Lucandri G, Brozzetti S, Bolognese A, Cavallaro A. Endoscopic retrograde cholangiography for intrabiliary rupture of hydatid cyst. Am J Surg. 2006;191:206-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 46. | Ozaslan E, Bayraktar Y. Endoscopic therapy in the management of hepatobiliary hydatid disease. J Clin Gastroenterol. 2002;35:160-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 44] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 47. | Sharma BC, Agarwal N, Garg S, Kumar R, Sarin SK. Endoscopic management of liver abscesses and cysts that communicate with intrahepatic bile ducts. Endoscopy. 2006;38:249-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 48. | Bedirli A, Sakrak O, Sozuer EM, Kerek M, Ince O. Surgical management of spontaneous intrabiliary rupture of hydatid liver cysts. Surg Today. 2002;32:594-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 49. | Köksal N, Müftüoglu T, Günerhan Y, Uzun MA, Kurt R. Management of intrabiliary ruptured hydatid disease of the liver. Hepatogastroenterology. 2001;48:1094-1096. [PubMed] |
| 50. | Daali M, Fakir Y, Hssaida R, Hajji A, Hda A. [Hydatid cysts of the liver opening in the biliary tract. Report of 64 cases]. Ann Chir. 2001;126:242-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 51. | Taçyildiz I, Aldemir M, Aban N, Keles C. Diagnosis and surgical treatment of intrabiliary ruptured hydatid disease of the liver. S Afr J Surg. 2004;42:43-46. [PubMed] |
| 52. | Chautems R, Bühler LH, Gold B, Giostra E, Poletti P, Chilcott M, Morel P, Mentha G. Surgical management and long-term outcome of complicated liver hydatid cysts caused by Echinococcus granulosus. Surgery. 2005;137:312-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 49] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 53. | Jabbour N, Shirazi SK, Genyk Y, Mateo R, Pak E, Cosenza DC, Peyré CG, Selby RR. Surgical management of complicated hydatid disease of the liver. Am Surg. 2002;68:984-988. [PubMed] |
| 54. | Dziri C, Paquet JC, Hay JM, Fingerhut A, Msika S, Zeitoun G, Sastre B, Khalfallah T. Omentoplasty in the prevention of deep abdominal complications after surgery for hydatid disease of the liver: a multicenter, prospective, randomized trial. French Associations for Surgical Research. J Am Coll Surg. 1999;188:281-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 55. | Available from: http://www.cebm.net/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/. |
| 56. | Emel’ianov SI, Khamidov MA. [Laparoscopic treatment of hydatid liver cysts]. Khirurgiia (Mosk). 2000;32-34. [PubMed] |
| 57. | Khoury G, Abiad F, Geagea T, Nabout G, Jabbour S. Laparoscopic treatment of hydatid cysts of the liver and spleen. Surg Endosc. 2000;14:243-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 60] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 58. | Bickel A, Loberant N, Singer-Jordan J, Goldfeld M, Daud G, Eitan A. The laparoscopic approach to abdominal hydatid cysts: a prospective nonselective study using the isolated hypobaric technique. Arch Surg. 2001;136:789-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 47] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 59. | Polat FR. Hydatid cyst: open or laparoscopic approach? A retrospective analysis. Surg Laparosc Endosc Percutan Tech. 2012;22:264-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 60. | Zaharie F, Bartos D, Mocan L, Zaharie R, Iancu C, Tomus C. Open or laparoscopic treatment for hydatid disease of the liver? A 10-year single-institution experience. Surg Endosc. 2013;27:2110-2116. [PubMed] |