Published online Feb 21, 2014. doi: 10.3748/wjg.v20.i7.1846
Revised: November 3, 2013
Accepted: November 18, 2013
Published online: February 21, 2014
Processing time: 191 Days and 3.2 Hours
AIM: To evaluate the applicability of AIMS65 scores in predicting outcomes of peptic ulcer bleeding.
METHODS: This was a retrospective study in a single center between January 2006 and December 2011. We enrolled 522 patients with upper gastrointestinal haemorrhage who visited the emergency room. High-risk patients were regarded as those who had re-bleeding within 30 d from the first endoscopy as well as those who died within 30 d of visiting the Emergency room. A total of 149 patients with peptic ulcer bleeding were analysed, and the AIMS65 score was used to retrospectively predict the high-risk patients.
RESULTS: A total of 149 patients with peptic ulcer bleeding were analysed. The poor outcome group comprised 28 patients [male: 23 (82.1%) vs female: 5 (10.7%)] while the good outcome group included 121 patients [male: 93 (76.9%) vs female: 28 (23.1%)]. The mean age in each group was not significantly different. The mean serum albumin levels in the poor outcome group were slightly lower than those in the good outcome group (P = 0.072). For the prediction of poor outcome, the AIMS65 score had a sensitivity of 35.5% (95%CI: 27.0-44.8) and a specificity of 82.1% (95%CI: 63.1-93.9) at a score of 0. The AIMS65 score was insufficient for predicting outcomes in peptic ulcer bleeding (area under curve = 0.571; 95%CI: 0.49-0.65).
CONCLUSION: The AIMS65 score may therefore not be suitable for predicting clinical outcomes in peptic ulcer bleeding. Low albumin levels may be a risk factor associated with high mortality in peptic ulcer bleeding.
Core tip: AIMS65 score is a novel simple score for predicting outcomes for acute upper gastrointestinal bleeding (UGIB). However, this scoring system is based on analyses of data from a mixed patient population with both variceal and non-variceal UGIB. The present study focused on the effectiveness of the AIMS65 score in predicting outcomes of peptic ulcer bleeding. This retrospective single-centre study, which included 149 patients, revealed that the AIMS65 score may not be suitable for predicting outcomes in peptic ulcer bleeding. Further, low albumin levels may be a risk factor associated with high mortality in peptic ulcer bleeding.
- Citation: Jung SH, Oh JH, Lee HY, Jeong JW, Go SE, You CR, Jeon EJ, Choi SW. Is the AIMS65 score useful in predicting outcomes in peptic ulcer bleeding? World J Gastroenterol 2014; 20(7): 1846-1851
- URL: https://www.wjgnet.com/1007-9327/full/v20/i7/1846.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i7.1846
Acute upper gastrointestinal (GI) bleeding is a common emergency associated with high morbidity and medical expense. The yearly incidence of acute upper GI bleeding is 50-150 per 100000 of the population, with a mortality rate of 10%-14%[1]. A major cause of acute upper gastrointestinal bleeding is peptic ulcer bleeding[2]. Endoscopic treatment and acid suppression with proton -pump inhibitors are most important in the management of peptic ulcer bleeding and these treatments have been reduced mortality[2-4]. Despite recent advances in endoscopic and pharmacological management, non-variceal upper gastrointestinal bleeding (NVUGIB) is still associated with considerable mortality and morbidity[5]. The recently published International Consensus Recommendations on the management of patients with non-variceal upper GI bleeding recommend “early risk stratification”, by using validated prognostic scales[1]. Several prognostic indices are available, including the Rockall[6] and Baylor[7] scores; however, these include clinical and endoscopic components and are therefore unsuitable for pre-endoscopic triage. The Glasgow-Blatchford score[8], which may be used for pre-endoscopic triage, compares favourably with the pre-endoscopic component of the Rockall score[9,10]. However, it has not been adopted in routine clinical practice, because of its limitations: it is weighted and assigns points to elements in the patient’s medical history, some of which lack a clear definition[11]. Recently, AIMS65-a new simple risk score for acute upper gastrointestinal bleeding-has been developed and validated[12-14]. The 5 parameters of AIMS65 are as follows: albumin levels, international normalized ratio (prothrombin time), altered mental status, systolic blood pressure, and age > 65 years. However, the role and utility of this for peptic ulcer bleeding has not yet been clarified since this scoring system was based on analysis of data from a mixed patient population, with acute upper GI bleeding that included both variceal and non-variceal UGIB. We considered whether this score would be useful in patients with peptic ulcer bleeding since the parameters evaluated in AIMS65, such as albumin and INR, appear to be associated with variceal bleeding. Therefore, in the present study, we aimed to evaluate the applicability of the AIMS65 score in predicting outcomes of peptic ulcer bleeding.
This study was performed in St. Paul’s Hospital, Catholic Medical Center, South Korea. This retrospective analysis included patients enrolled consecutively between January 2006 and December 2011. The study was reviewed and approved by the institutional review board. Patients were considered eligible for inclusion if they were over 18 years of age and had visited the emergency room (ER) for any upper GI bleeding symptoms, including melena, haematemesis and/or haematochezia. Of these, only patients who underwent endoscopy were included in the analysis. Exclusion criteria for the study were as follows: patients who did not undergo upper GI endoscopy; presence of variceal bleeding, bleeding ulcer from the anastomosis following gastrectomy, bleeding due to stomach cancer, obscure GI bleeding, Mallory-Weiss syndrome or angiodysplastic bleeding; and inability to follow up after 30 d from visiting the ER (determined from patient charts).
The variables examined included demographic factors (age and sex), vital signs (pulse, systolic blood pressure, diastolic blood pressure, temperature and respiratory rate), mental status, results of laboratory tests and underlying co-morbid conditions. Altered mental status was defined as physician-charted findings of “disoriented”, “stupor”, or “coma”. Vital signs, mental status and laboratory test results on the day of admission, including routine chemistry and haematology, were recorded.
High-risk patients were defined as those who suffered re-bleeding within 30 d of the first endoscopy along with those who died within 30 d from visiting the ER. Re-bleeding was characterized as fresh haematemesis and/or melena associated with the development of shock (pulse > 100 beats/min, systolic blood pressure < 100 mmHg), or a reduction in haemoglobin concentration greater than 2 g/dL over 24 h[15]. Re-bleeding also included cases requiring repeat endoscopy, surgical intervention or any interventional radiology procedure. Patient charts and/or electronic patient records were used to evaluate 30-d mortality. All high-risk patients were included in the “poor outcome” group.
Regarding the AIMS65 score, the following 5 factors were included: serum albumin < 3.0 g/dL, INR > 1.5, altered mental status, systolic blood pressure ≤ 90 mmHg, and age > 65 years. Each risk factor carries 1 point. Mortality risk can be differentiated as low (AIMS65 0-1 risk factors) or high (AIMS65 2-5 risk factors)[12]. We investigated whether the AIMS65 scores could predict patients with poor outcomes.
Categorical data are presented as mean ± SD. The χ2 test or Fisher’s exact test were applied to evaluate categorical variables. The t-test was used to evaluate continuous variables. Differences between good and poor outcomes were assessed using the χ2 test and t-tests. Both univariate and multivariate analyses were performed [SAS system for Windows (release 9.2; SAS Institute, Cary, NC, United States)]. P < 0.05 was considered statistically significant. The score for the area under curve (AUC) was suggested through the receiver operating curve (ROC) using the cut-off value of the AIMS65 score (MedCalc ver. 11.2.1.0).
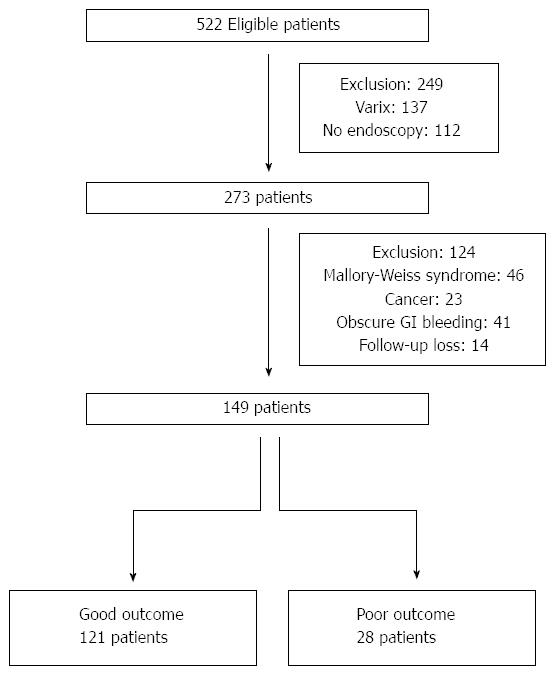
We reviewed 522 patients over the age of 18 years who visited the ER of St. Paul’s Hospital, Catholic Medical Center, South Korea, for complaints of upper GI bleeding, including haematemesis, melena, and/or haematochezia. Overall, we excluded 373 patients from the study for the following reasons: 112 patients did not undergo endoscopy, 206 patients were diagnosed with gastrointestinal bleeding from causes other than peptic ulcer (variceal bleeding, 137; Mallory-Weiss syndrome, 46; gastric cancer bleeding or bleeding ulcer during gastrectomy, 23; and other causes such as obscure GI bleeding or angiodysplastic bleeding, 41), and 14 patients were lost to follow-up. Thus, 149 patients with peptic ulcer bleeding were included in the final analysis (Figure 1).
The poor outcome group comprised 28 patients [male: 23 (82.1%) vs female: 5 (10.7%)] while the good outcome group included 121 patients [male: 93 (76.9%) vs female: 28 (23.1%)]. The mean age in each group was not significantly different (good outcome group vs poor outcome group; 66.4 ± 13.0 vs 62.9 ± 15.9; P = 0.216). The serum albumin level in the poor outcome group was slightly lower than that in the good outcome group; however, this difference was not statistically significant (Table 1). The poor outcome group included the following outcomes: repeat endoscopy (n = 24), operation (n = 3), and death (n = 1).
| Total (n = 149) | Good outcome (n = 121) | Poor outcome (n = 28) | P value | ||
| Diagnosis | Gastric ulcer | 117 (78.5) | 92 (76.0) | 25 (89.3) | 0.124 |
| Duodenal ulcer | 32 (21.5) | 29 (24.0) | 3 (10.7) | ||
| Sex | Male | 116 (77.9) | 93 (76.9) | 23 (82.1) | 0.544 |
| Female | 33 (22.1) | 28 (23.1) | 5 (17.9) | ||
| Age | mean ± SD | 62.9 ± 15.9 | 62.1 ± 16.4 | 66.3 ± 13.0 | 0.216 |
| < 65 yr | 72 (48.3) | 62 (51.2) | 10 (35.7) | 0.139 | |
| ≥ 65 yr | 77 (51.7) | 59 (48.8) | 18 (64.3) | ||
| Systolic BP | mean ± SD | 110.9 ± 22.6 | 109.8 ± 22.7 | 115.4 ± 22.4 | 0.243 |
| ≤ 90 | 36 (24.2) | 31 (25.6) | 5 (17.9) | 0.387 | |
| > 90 | 113 (75.8) | 90 (74.4) | 23 (82.1) | ||
| Albumin | mean ± SD | 3.3 ± 0.6 | 3.4 ± 0.6 | 3.1 ± 0.6 | 0.072 |
| < 3.0 | 47 (31.5) | 35 (28.9) | 12 (42.9) | 0.153 | |
| ≥ 3.0 | 102 (68.5) | 86 (71.1) | 16 (57.1) | ||
| INR (PT) | mean ± SD | 1.2 ± 0.8 | 1.2 ± 0.8 | 1.2 ± 0.2 | 0.537 |
| ≤ 1.5 | 138 (92.6) | 113 (93.4) | 25 (89.3) | 0.4341 | |
| > 1.5 | 11 (7.4) | 8 (6.6) | 3 (10.7) | ||
| Mental status | alert | 144 (96.6) | 117 (96.7) | 27 (96.4) | 0.999 |
| drowsy, coma | 5 (3.4) | 4 (3.3) | 1 (3.6) | ||
| AIMS65 score | 0 | 48 (32.2) | 43 (35.5) | 5 (17.9) | 0.2721 |
| 1 | 49 (32.9) | 37 (30.6) | 12 (42.9) | ||
| 2 | 34 (22.8) | 26 (21.5) | 8 (28.6) | ||
| 3 | 15 (10.1) | 13 (10.7) | 2 (7.1) | ||
| 4 | 3 (2.0) | 2 (1.7) | 1 (3.6) | ||
| < 2 (0–1) | 97 (65.1) | 80 (66.1) | 17 (60.7) | 0.589 | |
| ≥ 2 (2–5) | 52 (34.9) | 41 (33.9) | 11 (39.3) | ||
| < 1 | 48 (32.2) | 43 (35.5) | 5 (17.9) | 0.071 | |
| ≥ 1 | 101 (67.8) | 78 (64.5) | 23 (82.1) |
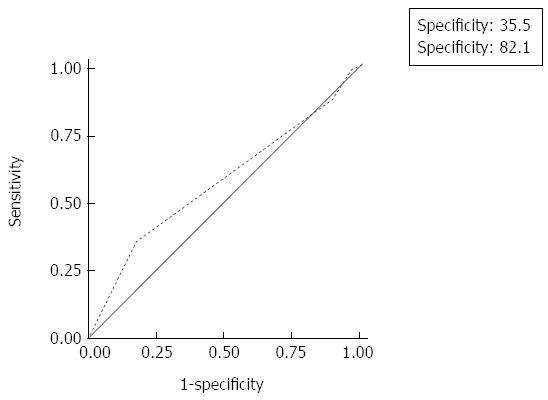
With regard to the AIMS65 score, 43 (35.5%) patients from the good outcome group and 5 (17.9%) patients from the poor outcome group scored 0 (P = 0.071). For the prediction of poor outcome, the AIMS65 score had a sensitivity of 35.5% (95%CI: 27.0-44.8) and a specificity of 82.1% (95%CI: 63.1-93.9) at a score of 0. Sensitivity and specificity were also suboptimal at higher decision thresholds (≤ 1, ≤ 2, and ≤ 3) (Table 2). The AIMS65 score was thus insufficient in predicting outcomes in peptic ulcer bleeding (AUC = 0.571; 95%CI: 0.49-0.65) (Figure 2).
| AIMS65 score cut-off point | Sensitivity | Specificity | Positive predictive value | Negative predictive value | Area under the ROC curve |
| ≤ 0 | 35.5% | 82.1% | 89.6 | 22.8 | 0.571 |
| ≤ 1 | 66.1% | 39.3% | 82.5 | 21.2 | (SE: 0.054 |
| ≤ 2 | 87.6% | 10.7% | 80.9 | 16.7 | 95%CI: 0.49-0.65) |
| ≤ 3 | 98.4% | 3.6% | 81.5 | 33.3 |
Peptic ulcer bleeding is the most common cause of acute non-variceal upper GI bleeding with high mortality, especially in older patients[16-18]. It is widely accepted that endoscopy should be performed as soon as possible, i.e., within 24 h of presentation at the hospital, and it is recommended that validated prognostic scales are applied to such patients for optimal management[1,19]. These strategies make it possible to identify high-risk lesions, such as active haemorrhage, non-bleeding visible vessels or non-bleeding adherent clots, and apply endoscopic therapy to these for improved prognosis.
The most consistently reported predictors of mortality and re-bleeding in NVUGIB have been age, number of co-morbid conditions and haemodynamic instability[20-23]. Several prognostic scales have been developed; however, these are not often adopted in routine clinical practice because of their complexity. In comparison, the AIMS65 score, which accurately predicts in-hospital mortality and length of stay, is a very simple risk score predicting outcomes in patients with acute upper GI bleeding[12]. Two recent reports confirmed the applicability of AIMS65 in acute upper GI bleeding patients, including bleeding of variceal and non-variceal origin[12,13]. However, whether the AIMS65 score is applicable for predicting outcomes in patients of non-variceal GI bleeding remains uncertain, since 2 of the 5 risk factors in AIMS65 scores are generally accepted as poor prognostic factors of liver cirrhosis, i.e. serum albumin < 3.0 g/dL and INR > 1.5. Therefore, the AIMS65 score might be useful for predicting outcomes in variceal GI bleeding but not in non-variceal GI bleeding. Our present results revealed a disappointing ROC value for the AIMS65 score, indicating that the AIMS65 score was not particularly useful for predicting poor outcomes in patients with peptic ulcer bleeding.
Interestingly, the mean serum albumin level in the poor outcomes group was slightly lower than that in the good outcomes group although this difference was not statistically significant (P = 0.072). This may have been caused by the inclusion of patients with co-morbidities other than liver cirrhosis in the poor outcomes group. On the other hand, low serum albumin levels may be a single prognostic factor predicting outcomes in patients with peptic ulcer bleeding. Two recent studies have demonstrated that serum albumin level ≤ 3 g/dL or < 2.6 g/dL are associated with the in-hospital mortality in patients with non-variceal GI bleeding[24,25]. In terms of INR, systemic review has shown that the INR does not predict re-bleeding among NVUGIB patients[26]. However, INR ≥ 1.5 has been shown to be independently associated with in-hospital mortality in upper GI bleeding in the UK[27]. More research is needed to clarify whether the albumin level and INR can indeed predict outcomes in patients with non-variceal GI bleeding.
This study has certain limitations. First, this is a retrospective single-centre study. Second, we enrolled only patients who underwent endoscopy and excluded patients who refused endoscopy or were discharged by the emergency department. In addition, patients with bleeding due to stress ulcers in the ICU were excluded because this was considered to be related to other co-morbidities rather than peptic ulcer disease specifically. These exclusions may create a bias. Third, it is possible that the small sample size especially that for the poor outcome group, could affect the results of this study. However, the current study is the first to examine the applicability of the AIMS65 score in patients with peptic ulcer bleeding taking re-bleeding into consideration, which was not evaluated previously in their study[12]. Fourth, ethnic differences between Western population and Asian may have affected our results. Although the Blatchford score and Rockall score are useful for predict prognoses in Western populations, a recent study demonstrated that in Asians, only the Blatchford score was appropriate for predicting low-risk patients who do not need therapeutic endoscopy[28].
In conclusion, the AIMS65 score may not be suitable for predicting outcomes in patients with peptic ulcer bleeding. A low albumin level may be a risk factor associated with high mortality in patients with peptic ulcer bleeding. However, further studies are necessary to validate the role of the AIMS65 score in variceal and non-variceal GI bleeding and its usefulness in identifying high-risk patients needing endoscopic therapy.
Acute upper gastrointestinal bleeding is a common emergency associated with high morbidity and medical expenses. Several prognostic indices are clinically available, including the Rockall score, Baylor score, and the Glasgow-Blatchford score. Recently, AIMS65 has been proposed as a new simple risk score.
Acute upper gastrointestinal bleeding remains widely prevalent, since anti-platelet medications, such as aspirin, are currently commonly used. Despite recent advances in endoscopic and pharmacological management, non-variceal upper gastrointestinal bleeding (UGIB) continues to be associated with considerable mortality and morbidity. It is important to accurately select high-risk patients with UGIB since these patients need emergent endoscopy.
This study focused on the effectiveness of the AIMS65 score in predicting outcomes of peptic ulcer bleeding. The AIMS65 score is based on analysis of data from a mixed patient population with both variceal and non-variceal UGIB. The present study showed that the AIMS65 score appeared to be unsuitable for predicting outcomes in patients with peptic ulcer bleeding in an Asian population.
Low albumin levels may be a risk factor associated with high mortality in patients with peptic ulcer bleeding. Further studies are necessary to validate the role of the AIMS65 score in variceal and non-variceal gastrointestinal (GI) bleeding and its usefulness in identifying high-risk patients needing endoscopic therapy.
This is an interesting manuscript studying whether a validated score for predicting outcomes in patients with upper GI bleeding is useful for evaluating patients with bleeding peptic ulcers. Although there are several limitations, the negative results merit publication. A large-scale multi-centric trial across Asia would provide the evidence for supporting these findings.
P- Reviewers: Basoli A, Mayol J, Reddy DN S- Editor: Ma YJ L- Editor: A E- Editor: Liu XM
| 1. | Marshall JK, Collins SM, Gafni A. Prediction of resource utilization and case cost for acute nonvariceal upper gastrointestinal hemorrhage at a Canadian community hospital. Am J Gastroenterol. 1999;94:1841-1846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 2. | Lau JY, Barkun A, Fan DM, Kuipers EJ, Yang YS, Chan FK. Challenges in the management of acute peptic ulcer bleeding. Lancet. 2013;381:2033-2043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 151] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 3. | Sreedharan A, Martin J, Leontiadis GI, Dorward S, Howden CW, Forman D, Moayyedi P. Proton pump inhibitor treatment initiated prior to endoscopic diagnosis in upper gastrointestinal bleeding. Cochrane Database Syst Rev. 2010;CD005415. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 4. | Barkun AN, Martel M, Toubouti Y, Rahme E, Bardou M. Endoscopic hemostasis in peptic ulcer bleeding for patients with high-risk lesions: a series of meta-analyses. Gastrointest Endosc. 2009;69:786-799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 117] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 5. | Loffroy RF, Abualsaud BA, Lin MD, Rao PP. Recent advances in endovascular techniques for management of acute nonvariceal upper gastrointestinal bleeding. World J Gastrointest Surg. 2011;3:89-100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 47] [Cited by in RCA: 44] [Article Influence: 3.1] [Reference Citation Analysis (2)] |
| 6. | Rockall TA, Logan RF, Devlin HB, Northfield TC. Risk assessment after acute upper gastrointestinal haemorrhage. Gut. 1996;38:316-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 888] [Cited by in RCA: 897] [Article Influence: 30.9] [Reference Citation Analysis (0)] |
| 7. | Saeed ZA, Winchester CB, Michaletz PA, Woods KL, Graham DY. A scoring system to predict rebleeding after endoscopic therapy of nonvariceal upper gastrointestinal hemorrhage, with a comparison of heat probe and ethanol injection. Am J Gastroenterol. 1993;88:1842-1849. [PubMed] |
| 8. | Blatchford O, Murray WR, Blatchford M. A risk score to predict need for treatment for upper-gastrointestinal haemorrhage. Lancet. 2000;356:1318-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 666] [Cited by in RCA: 684] [Article Influence: 27.4] [Reference Citation Analysis (1)] |
| 9. | Ananthakrishnan AN, McGinley EL, Saeian K. Outcomes of weekend admissions for upper gastrointestinal hemorrhage: a nationwide analysis. Clin Gastroenterol Hepatol. 2009;7:296-302e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 124] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 10. | Chen IC, Hung MS, Chiu TF, Chen JC, Hsiao CT. Risk scoring systems to predict need for clinical intervention for patients with nonvariceal upper gastrointestinal tract bleeding. Am J Emerg Med. 2007;25:774-779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 115] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 11. | Mungan Z. An observational European study on clinical outcomes associated with current management strategies for non-variceal upper gastrointestinal bleeding (ENERGIB-Turkey). Turk J Gastroenterol. 2012;23:463-477. [PubMed] |
| 12. | Saltzman JR, Tabak YP, Hyett BH, Sun X, Travis AC, Johannes RS. A simple risk score accurately predicts in-hospital mortality, length of stay, and cost in acute upper GI bleeding. Gastrointest Endosc. 2011;74:1215-1224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 337] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 13. | Hyett BH, Abougergi MS, Charpentier JP, Kumar NL, Brozovic S, Claggett BL, Travis AC, Saltzman JR. The AIMS65 score compared with the Glasgow-Blatchford score in predicting outcomes in upper GI bleeding. Gastrointest Endosc. 2013;77:551-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 101] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 14. | Chandra S. AIMS65 score predicts short-term mortality but not the need for intervention in acute upper GI bleeding. Gastrointest Endosc. 2013;78:381-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 15. | Manta R, Galloro G, Mangiavillano B, Conigliaro R, Pasquale L, Arezzo A, Masci E, Bassotti G, Frazzoni M. Over-the-scope clip (OTSC) represents an effective endoscopic treatment for acute GI bleeding after failure of conventional techniques. Surg Endosc. 2013;27:3162-3164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 86] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 16. | Barkun AN, Bardou M, Kuipers EJ, Sung J, Hunt RH, Martel M, Sinclair P. International consensus recommendations on the management of patients with nonvariceal upper gastrointestinal bleeding. Ann Intern Med. 2010;152:101-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 754] [Cited by in RCA: 710] [Article Influence: 47.3] [Reference Citation Analysis (1)] |
| 17. | El Ouali S, Barkun AN, Wyse J, Romagnuolo J, Sung JJ, Gralnek IM, Bardou M, Martel M. Is routine second-look endoscopy effective after endoscopic hemostasis in acute peptic ulcer bleeding? A meta-analysis. Gastrointest Endosc. 2012;76:283-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 18. | Gralnek IM, Barkun AN, Bardou M. Management of acute bleeding from a peptic ulcer. N Engl J Med. 2008;359:928-937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 213] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 19. | Lim LG, Ho KY, Chan YH, Teoh PL, Khor CJ, Lim LL, Rajnakova A, Ong TZ, Yeoh KG. Urgent endoscopy is associated with lower mortality in high-risk but not low-risk nonvariceal upper gastrointestinal bleeding. Endoscopy. 2011;43:300-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 125] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 20. | Barkun A, Bardou M, Marshall JK. Consensus recommendations for managing patients with nonvariceal upper gastrointestinal bleeding. Ann Intern Med. 2003;139:843-857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 365] [Cited by in RCA: 331] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 21. | Lahiff C, Shields W, Cretu I, Mahmud N, McKiernan S, Norris S, Silke B, Reynolds JV, O’Toole D. Upper gastrointestinal bleeding: predictors of risk in a mixed patient group including variceal and nonvariceal haemorrhage. Eur J Gastroenterol Hepatol. 2012;24:149-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 22. | Lanas A, Aabakken L, Fonseca J, Mungan ZA, Papatheodoridis GV, Piessevaux H, Cipolletta L, Nuevo J, Tafalla M. Clinical predictors of poor outcomes among patients with nonvariceal upper gastrointestinal bleeding in Europe. Aliment Pharmacol Ther. 2011;33:1225-1233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 23. | Marmo R, Koch M, Cipolletta L, Capurso L, Pera A, Bianco MA, Rocca R, Dezi A, Fasoli R, Brunati S. Predictive factors of mortality from nonvariceal upper gastrointestinal hemorrhage: a multicenter study. Am J Gastroenterol. 2008;103:1639-1647; quiz 1648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 113] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 24. | Weng SC, Shu KH, Tarng DC, Tang YJ, Cheng CH, Chen CH, Yu TM, Chuang YW, Huang ST, Sheu WH. In-hospital mortality risk estimation in patients with acute nonvariceal upper gastrointestinal bleeding undergoing hemodialysis: a retrospective cohort study. Ren Fail. 2013;35:243-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 25. | González-González JA, Vázquez-Elizondo G, García-Compeán D, Gaytán-Torres JO, Flores-Rendón ÁR, Jáquez-Quintana JO, Garza-Galindo AA, Cárdenas-Sandoval MG, Maldonado-Garza HJ. Predictors of in-hospital mortality in patients with non-variceal upper gastrointestinal bleeding. Rev Esp Enferm Dig. 2011;103:196-203. [PubMed] |
| 26. | Shingina A, Barkun AN, Razzaghi A, Martel M, Bardou M, Gralnek I. Systematic review: the presenting international normalised ratio (INR) as a predictor of outcome in patients with upper nonvariceal gastrointestinal bleeding. Aliment Pharmacol Ther. 2011;33:1010-1018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 49] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 27. | Jairath V, Kahan BC, Stanworth SJ, Logan RF, Hearnshaw SA, Travis SP, Palmer KR, Murphy MF. Prevalence, management, and outcomes of patients with coagulopathy after acute nonvariceal upper gastrointestinal bleeding in the United Kingdom. Transfusion. 2013;53:1069-1076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 37] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 28. | Pang SH, Ching JY, Lau JY, Sung JJ, Graham DY, Chan FK. Comparing the Blatchford and pre-endoscopic Rockall score in predicting the need for endoscopic therapy in patients with upper GI hemorrhage. Gastrointest Endosc. 2010;71:1134-1140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 108] [Article Influence: 7.2] [Reference Citation Analysis (0)] |