Published online Feb 14, 2014. doi: 10.3748/wjg.v20.i6.1450
Revised: December 21, 2013
Accepted: January 3, 2014
Published online: February 14, 2014
Processing time: 129 Days and 18.2 Hours
Helicobacter pylori (H. pylori) is a successful pathogen that can persist in the stomach of an infected person for their entire life. It provokes chronic gastric inflammation that leads to the development of serious gastric diseases such as peptic ulcers, gastric cancer and Mucosa associated lymphoid tissue lymphoma. It is known that these ailments can be avoided if the infection by the bacteria can be prevented or eradicated. Currently, numerous antibiotic-based therapies are available. However, these therapies have several inherent problems, including the appearance of resistance to the antibiotics used and associated adverse effects, the risk of re-infection and the high cost of antibiotic therapy. The delay in developing a vaccine to prevent or eradicate the infection has furthered research into new therapeutic approaches. This review summarises the most relevant recent studies on vaccine development and new treatments using natural resources such as plants, probiotics and nutraceuticals. In addition, novel alternatives based on microorganisms, peptides, polysaccharides, and intragastric violet light irradiation are presented. Alternative therapies have not been effective in eradicating the bacteria but have been shown to maintain low bacterial levels. Nevertheless, some of them are useful in preventing the adverse effects of antibiotics, modulating the immune response, gastroprotection, and the general promotion of health. Therefore, those agents can be used as adjuvants of allopathic anti-H. pylori eradication therapy.
Core tip:Helicobacter pylori (H. pylori) plays a role in several gastric diseases. Current anti-H. pylori therapy fails in more than 20% of cases, primarily due to antimicrobial resistance and patient non-adherence. This situation has encouraged the search for other approaches to control H. pylori infection. This review discusses advances in the development of an H. pylori vaccine and new treatments based on plants, probiotics, nutraceuticals, microorganisms, peptides and intragastric violet light irradiation. Alternative therapies have not been effective in eradicating the bacteria in vivo but are promising as complementary treatments diseases associated with H. pylori infection.
-
Citation: Ayala G, Escobedo-Hinojosa WI, Cruz-Herrera CFL, Romero I. Exploring alternative treatments for
Helicobacter pylori infection. World J Gastroenterol 2014; 20(6): 1450-1469 - URL: https://www.wjgnet.com/1007-9327/full/v20/i6/1450.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i6.1450
Helicobacter pylori (H. pylori) infection is an important public health problem in several parts of the world. Because this pathogen is associated with various gastric diseases, ranging from mild discomfort, such as superficial gastritis, to severe ailments, such as chronic atrophic gastritis, gastric cancer or peptic (gastric or duodenal) ulcer, there is much interest in understanding how infection with H. pylori could be prevented.
H. pylori has virulence factors that are necessary to colonise the acid environment of the stomach and to survive in it; these factors are expressed in all isolates[1]. Of these, the most remarkable are urease and the adhesins. Urease metabolises urea into ammonia and carbon dioxide, and it contributes to the neutralisation of gastric acid[2]. In addition, urease is strongly immunogenic and chemotactic for phagocytes[3], and it promotes the production of the proinflammatory cytokines interleukin (IL)-1β, IL-6 and IL-8, as well as tumoural necrosis factor-alpha (TNF-α)[4,5]. H. pylori adheres specifically to the epithelial cells of the gastric mucosa by means of the adhesins. Of these, the most studied are BabA and SabA, which are external membrane proteins that link to the blood group antigens Lewis-b and Lewis-x, respectively[6,7]. The gene babA is polymorphic, occurring as babA1 and babA2[8]. Strains containing babA2 are associated with a higher risk of peptic ulcer, intestinal metaplasia and gastric cancer. Moreover, babA2+ strains generally display the most cytotoxic vacA genotype (s1/m1) and they are cagA+, which further increases the risk of peptic ulcer, intestinal metaplasia and gastric cancer[9,10]. Other membrane proteins that function as adhesins have been reported, including AlpA, AlpB, HopZ and HopH, also called outer inflammatory protein A (OipA) due to its association with the increased secretion of IL-8 by epithelial cells in vitro and with intense gastric inflammation in vivo[11-14]. Nevertheless, only approximately 20% of the H. pylori population in the stomach adheres to the epithelial cells, whereas the rest is found in the mucosal layer[15].
The neutrophil-activating protein of H. pylori (HP-NAP) allows the bacteria to capture iron, which is essential for its growth[16]. HP-NAP is particularly important in the pathogenesis of the infection because it induces adhesion and chemotaxis of mononuclear and polymorphonuclear phagocytes[17]. HP-NAP activates the NADPH oxidase enzyme, which is involved in the production of reactive species of oxygen (ROS)[18]. It also stimulates the production of IL-12 and IL-23 by neutrophils and monocytes; these cytokines promote inflammation[19]. Because of its properties, HP-NAP increases the degree of gastric mucosa inflammation and the continuous harm to gastric cells caused by ROS.
There are other important enzymes that protect H. pylori against the ROS produced through internal metabolic processes as well as by neutrophils and macrophages during inflammation[20]. Among these are superoxide dismutase (Hp-SOD), which catalyses the dismutation of the superoxide radical into hydrogen peroxide and molecular oxygen, and catalase (KatA), which converts hydrogen peroxide into water and oxygen. Another important anti-ROS enzyme is alkyl hydroperoxidase (AhpC), which is a highly abundant and conserved protein in H. pylori[21]. This enzyme is a member of the peroxiredoxin family that has been shown to be capable of reducing different peroxides, but its major function under physiological conditions is to reduce organic hydroperoxides into non-toxic alcohols[22].
Several H. pylori virulence factors have been associated with gastric carcinogenesis. The most important of these include vacuolating cytotoxin (VacA) and the cytotoxin-associated gen A (CagA) protein. Both are especially relevant for the pathology of the infection by H. pylori because strains that produce them have been more frequently isolated from patients with gastric cancer[23,24].
VacA is one of the most important factors determining the virulence of the pathogenic strains of H. pylori. It was first characterised by its ability to cause cell vacuolisation in tissue culture cells. VacA creates pores in the membrane of the host cells, allowing the exit of chlorine and bicarbonate ions, pyruvate and urea[25,26]. VacA causes the release of iron, nickel, sugars and amino acids through the tight junctions of the gastric epithelium without affecting their integrity[27]. It has been reported that VacA interferes with the process of antigen presentation in vitro[28], induces apoptosis in epithelial cells[29] and inhibits the activation and proliferation of T and B cells in vitro[30,31]. The functions of VacA suggest that its initial activity consists in providing nutrients for the establishment of the infection. Afterwards, it contributes to the persistence and chronicity of the infection by inhibiting immune cells and altering the balance of cellular turnover; it also increases cellular proliferation and allows the persistence of mutation-carrying cells, thus contributing to the carcinogenic process.
The H. pylori CagA protein is a 120- to 140-kDa protein produced by approximately 60% of clinical isolates. The cagA gene is located at the end of the cytotoxin-associated genes pathogenicity island (cagPAI)[32]. This region contains 32 genes, some of which encode for multiple structural components of the type IV secretion system (T4SS). The T4SS is an external structure that can be visualised microscopically as pilus-like structures protruding from the bacterial membrane[33]. In addition to CagA, there are other cagPAI products that are equally important in the pathogenesis processes associated with H. pylori. Among these is the CagL protein, which is found at the tip of the pilus and serves as an adhesine, which binds to host cell integrin[34] to trigger the translocation of CagA into the cell[35]. Additionally, CagL interacts with the α5β1 integrin to induce IL-8 production and the nucleotide-binding oligomerisation domain-containing 1 (NOD1) signalling[36] independently of CagA translocation. Once CagA is translocated into gastric epithelial cells, it carries out functions that depend on both the phosphorylated and the non-phosphorylated forms of CagA[37]. Phosphorylation-dependent functions include the dispersion and elongation of cells, affecting their proliferation and adhesion, as well as the organisation of the cytoskeleton[38-40]. Phosphorylation-independent functions include: interruption of the tight adhesive junctions, loss of cellular polarity, proinflammatory and mitogenic responses, degradation of the extracellular matrix, and induction of cell cycle progression[41-45]. The result of these processes is the destabilisation of the gastric epithelium, which potentially contributes to the pathogenesis of H. pylori in vivo. Moreover, these findings point to the preferential survival of H. pylori-infected cells and to the oncogenic role of the bacteria.
A gene associated with the development of duodenal ulcer (dupA) has been reported. The dupA gene is the most frequent gene in strains isolated from patients with duodenal ulcer. It is associated with the development of duodenal ulcers, as well as with neutrophil infiltration in the antrum and higher levels IL-8[46]. However, other investigations have not found these associations, so more research work is needed to confirm this finding[47,48].
The development of innate immunity depends on the host’s recognition of microbial pathogens through pathogen-associated molecular patterns (PAMPs), such as lipopolysaccharide (LPS), peptidoglycan, lipoproteins, flagellins or double-stranded RNA[49]. These PAMPs are highly conserved molecular structures that are recognised by Toll-like receptors (TLRs)[50] or by NOD-like receptors (NLRs)[51]. H. pylori LPS and flagellin are poor activators of TLR-4 and TLR-5, respectively. H. pylori LPS has a weak endotoxic activity compared to Salmonella typhimurium LPS[52] and is a weaker inducer of pro-inflammatory cytokines than Escherichia coli LPS[53]. H. pylori flagellin is weakly recognised by TLR-5 and it is not pro-inflammatory[54,55]. H. pylori evades an initial innate response by preventing intense immune activation; thus, it can colonise the gastric mucosa. The H. pylori peptidoglycan is delivered into cells through the T4SS[56] and the outer membrane vesicles[57]. Once translocated, the peptidoglycan is recognised by NLR NOD-1, resulting in the translocation of nuclear factor (NF)-κB to the nucleus and the activation of the immune response genes[56,58].
The hallmark of the interaction between H. pylori and the host’s immune system is the persistence of the infection for years, leading to a chronic inflammation of the gastric mucosa. Once the H. pylori infection is established, both cellular and humoural adaptive immunities are developed: naive T helper (Th) CD4+ cells differentiate into Th effector cells (cellular response), and B cells that produce specific antibodies are activated (humoural response)[59]. However, there is evidence indicating that B cells and antibodies are dispensable for H. pylori control[60,61], whereas Th1 and Th17 effector T cell subsets and their cytokines are essential for the control of the infection[62-64]. Th1 cells produce the pro-inflammatory cytokines gamma interferon (IFN-γ) and tumour necrosis factor α and β that stimulate innate and T-cell immune responses[65]. Th17 cells are a recently identified class of effector T cells that produce pro-inflammatory cytokine IL-17. This interleukin stimulates fibroblasts, endothelial and epithelial cells, and gastric and lamina propria mononuclear cells to produce a diversity of cytokines and chemokines; this process results in neutrophil infiltration that contributes to H. pylori-associated inflammation[66]. Despite the local and systemic response against the infection, H. pylori can subvert and/or modulate the adaptive immunity perpetuating the infection and chronic inflammation[67]. In a small proportion of infected individuals, this chronic inflammation leads to the development of gastric cancer.
Regulatory T cells (Treg cells) contribute to peripheral immunologic tolerance by suppressing activated T cells, natural killer cells, B cells and dendritic cells[68]. Treg cells are induced by IL-18 in response to H. pylori; they regulate the effector T cells to prevent excessive activation and promote a persistent infection[69]. Treg cells from the gastric mucosa of H. pylori-associated gastric cancer patients could down-modulate the T cell response against H. pylori in vitro[70]. Furthermore, high levels of suppressive cytokine IL-10[71] and a Treg cell-mediated reduction of T cell transendothelial migration[72] from H. pylori-associated gastric cancer patients has been detected. Anti-tumour responses are characterised by cell-mediated immunity. In the mouse model, Treg cells can suppress an effective immune response against tumours[73,74]. It seems that the suppressive functions of the Treg cells may contribute to the reduced anti-tumour responses and thus to tumour progression in H. pylori-associated gastric cancer patients.
In some H. pylori-infected individuals, acid secretion is higher than normal. The acid flows into the duodenum, leading to gastric metaplasia. H. pylori cannot colonise a normal duodenum; it preferentially colonises areas of duodenal gastric metaplasia[75]. The numbers of CD4+ FOXP3+ T cells are increased in areas of gastric metaplasia in the duodenum of H. pylori-infected ulcer patients[76]. Interestingly, there is evidence showing reduced cytokine production in the duodenal epithelium of duodenal ulcer patients[77]. These findings suggest that a down-regulation of the immune response, possibly by Treg cells, allows a higher bacterial density in the duodenum that, together with the high secretion of acid, plays a role in the development of H. pylori-associated duodenal ulcer[78].
In most individuals, the H. pylori infection can continue throughout life as an asymptomatic condition. Unfortunately, its persistence in the stomach causes chronic gastric inflammation and tissue damage, leading to alterations that could evolve to severe gastric diseases such as peptic ulcers, gastric cancer or mucosa associated lymphoid tissue lymphoma. Therefore, eradication appears to offer the most direct approach to reducing the enormous human and economic consequences of H. pylori infection[79,80].
In general, several international guidelines for treating patients diagnosed with H. pylori infections are consistent with the use of triple therapy as the first-line treatment. This treatment consists of the administration of a proton pump inhibitor (PPI), clarithromycin, and amoxicillin for 7-14 d[81-83]. However, H. pylori eradication treatments following this regimen produce cure rates lower than 80%, mainly due to an increase in clarithromycin resistance[84,85]. As a result, other regimens (second-line therapies) have been proposed[85]. These treatments usually consist of a PPI in combination with two or three antibiotics, among which amoxicillin, clarithromycin, metronidazole, and tetracycline are included. To overcome the antimicrobial resistance problem and to increase the cure rates of initial treatments, new drug combinations are being developed from existing formulas. The use= of a four-drug treatment (i.e., either sequential, concomitant or bismuth-containing) has been recommended. Sequential treatment consists of a dual therapy (a PPI plus amoxicillin) for 5 d, followed by a 5-d triple therapy with a PPI plus clarithromycin and tinidazole or metronidazole to complete a 10-d treatment. Concomitant therapy consists of four drugs (a PPI, clarithromycin, metronidazole/tinidazole and amoxicillin) given twice a day for 3-7 d. Bismuth-containing quadruple therapy consists of a bismuth salt, tetracycline HCl, metronidazole/tinidazole, and a PPI given three or four times a day for 7-14 d[86].
European guidelines recommend culture before the selection of a third-line treatment based on the microbial antibiotic sensitivity. After two eradication failures, H. pylori isolates are often resistant to both metronidazole and clarithromycin. The alternative candidates for third-line therapy are quinolones, tetracycline, rifabutin and furazolidone; high-dose PPI/amoxicillin therapy might also be promising[87].
The main reasons for treatment failure are antimicrobial resistance and patient non-adherence. The lack of treatment compliance by the patient is a basic factor that explains the low rates of bacterial eradication. The cause is the complexity of the therapy, which involves at least three drugs, administered in repeated doses for a long time. Consequently, there are side effects, which, coupled with a lack of immediate improvement, discourage the patient to continue with the therapy. The high cost of anti-H. pylori treatments is another drawback. Finally, the recurrence of H. pylori infection after successful eradication also represents a problem in terms of the efficiency of therapies, especially in developing countries.
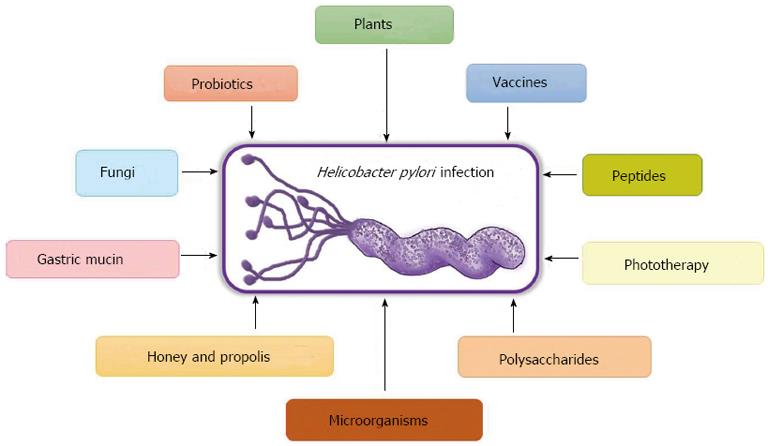
Taking into account the problems inherent to anti-H. pylori therapies in clinical practice, new therapeutic approaches to obtain a vaccine against the bacteria and to find new molecules with antibiotic activity or adjuvants that help patients to comply with the therapies have emerged. In this review we summarise the most relevant recent research on alternative treatments to the current therapy against H. pylori (Figure 1).
The need for a vaccine is especially evident in those countries with a high prevalence of the infection; with increased resistance to the antibiotics used to treat it, which might increase the recurrence rate of the infection; and with high morbidity and mortality rates caused by H. pylori infection-associated pathologies[85]. The high cost of treatment for diseases associated with this infection (i.e., gastric cancer and peptic ulcer) make vaccine development a cost-effective alternative. Thus, effective vaccines that could prevent and/or cure the infection or at least modify the host-pathogen interactions in a manner that prevents disease progression are desirable.
The continued efforts to obtain a vaccine against H. pylori have been focused on finding the best delivery route, as well as adjuvants and antigens that favour the induction of protective immunity. Because H. pylori is an extracellular mucosal pathogen, most of the vaccines that have been developed are based on mucosal immunisation. Several routes of mucosal vaccination and different antigens in combination with mucosal immune adjuvants, such as the cholera toxin (CT) or the E. coli heat-labile toxin (LT), have been tested[88]. In animal models, both CT and LT are strong adjuvants that induce protection, defined as a significant reduction in the bacterial load; they also induce protection against a challenge with H. pylori. However, these adjuvants have adverse effects in humans, which prevent their clinical application. The best alternative for human studies is aluminium hydroxide, which is already approved by the United States Food and Drug Administration (FDA)[89]. The introduction of live attenuated Salmonella expressing H. pylori urease A and B subunits[90,91] is an alternative strategy that does not require adjuvants, but studies in humans[92,93] did not produce satisfactory results.
Different antigens have been shown to be effective in inducing the immune response to the infection. Urease (UreA and UreB), CagA, VacA, neutrophil-activating protein (NapA) and catalase are recognised by antibodies[94,95]; therefore, these antigens are the best candidates for vaccine formulations. Among these antigens, urease is highly expressed by all H. pylori strains, which makes it a good candidate for a vaccine formulation. Although the initial results were disappointing, the combination of urease with other antigens such as HpaA (essential surface protein) conferred better protection[96]. The search for novel antigens has led to an interesting new development: a vaccine comprising genetically fused HspA, UreB and HpaA to create a multivalent vaccine[97]. When this trivalent fusion vaccine was tested in BALB/c mice, it showed significant protection against H. pylori infection, which was associated with H. pylori-specific antibody production and the Th1/Th2-type immune responses. Another innovative development consists of a system where the measles virus (MV) is used as a vector for the expression of the H. pylori NapA. It was demonstrated that attenuated MV strains expressing the NapA antigen induce a significant humoural and cellular immune response against both the MV and the NapA antigen[98]. Other novel antigens include antioxidant enzymes, superoxide dismutase[99] and alkyl hydroperoxide reductase[100], which are essential for H. pylori’s survival in vivo. When they are used as vaccines, the immune response generated against them could also have an impact on bacterial survival and thus favour elimination. Recent research reports from human studies show that a vaccine formulation comprising CagA, VacA and NapA, in combination with aluminium hydroxide as an adjuvant and delivered by intramuscular injection, elicits antigen-specific humoural and cellular responses against CagA and VacA; specific T cells against CagA and VacA were observed, and they were detectable 24 mo after the primary vaccination, suggesting T cell memory[89].
There is evidence that cellular immunity is required for H. pylori clearance and that an initial humoural response fails to prevent infection and colonisation[60,101]. Studies in animal models have found that certain antigens and adjuvant combinations for immunisation can induce protective immunity[102-104], although the protective mechanism remains unclear. From these studies, it has also been revealed that the immune response induced by H. pylori contains a significant component of Treg cells that may limit both the inflammatory and the immune mechanisms in the gastric mucosa, which might play an important role in clearing the infection. These immunomodulatory mechanisms used by H. pylori to persist in its gastric niche are crucial to prevent sterilising immunity[61]. Therefore, successful vaccine formulations and vaccination strategies must overcome the immunomodulatory response induced by H. pylori[105,106].
Although some promising results have been obtained in animal models, pointing to the feasibility of developing a secure and effective vaccine for human use[107], more clinical trials are needed to establish whether the positive results obtained in animal models can be reproduced in humans. In addition, other aspects require investigation, to provide clues regarding vaccine formulations, antigens, adjuvants, and delivery systems.
Plants and spices have been used since ancient times as therapeutic agents, and their uses have been passed down from generation to generation. Today, traditional medicine systems based on the use of plants are especially recognised in developing countries, where the availability of medical services and the accessibility to allopathic treatments is limited. Natural products, specifically those of plant origin, are potential sources for the discovery and development of new effective agents against infections[108].
There are hundreds of scientific publications worldwide that describe specifically the antibiotic activity of herbal products against H. pylori. A search (plant anti H. pylori) in the PubMed database from 1991 to August 31, 2013, lists over 300 entries, including plant extracts, plant compounds, and plant processed products. In 1991, the anti-H. pylori effect of 13 Malagasy medicinal plants was reported[109]. Since then, several investigations of medicinal plants of a specific region or country have been undertaken. Examples include studies of the anti-H. pylori activity of Chinese[110], Mexican[111-113], Iranian[114,115], Taiwanese[116], African[117,118] and Greek[119] herbal medicines. The largest group of reports refers to the activity of an individual plant, either for medicinal or dietary use.
In addition to screening for active regional medicinal plants, research efforts have also focused on the bioguided isolation and identification of pure compounds, as well as on chemical modification of lead compounds in order to obtain more active molecules against H. pylori. From these phytochemical studies, many compounds with remarkable anti-H. pylori effect in vitro have been identified[120,121]. However, very few of them retain their in vivo activity. Examples include catechins, particularly epigallocatechin gallate[122], the terpene plaunotol[123], 1-methyl-2-[(Z)-8-tridecenyl]-4-(1H)-quinolone and 1-methyl-2-[(Z)-7-tridecenyl]-4-(1H)-quinolone[124], the flavonoid kaempferol, the alkaloid tryptanthrin[125], sulphoraphane[126,127], 4-vinyl-2,6-dimethoxyphenol, canolol[128], and β-artecyclopropylmether, an artemisinin derivative[129].
More integral investigations include other pharmacological aspects of the plants and extracts in addition to their anti-H. pylori activity; for instance, anti-secretory, antioxidant, gastroprotective, anti-inflammatory, anti-haemorrhagic, and gastric ulcer resolution activities[130-133].
Studies that have attempted to elucidate the mechanism of action of the plant extracts or compounds on H. pylori are available. For example, some of these studies have examined their effect of various compounds on bacterial virulence factors such as urease[134], adherence[135-138], motility, vacuolisation[139,140] or on some key metabolic enzymes[135,141].
Below, we describe examples of the research conducted on plants or plant products, emphasising those in which its anti-H. pylori effectiveness in vivo was studied.
Garlic, Allium sativum, is invaluable not only as an essential flavouring element in food but also for its therapeutic properties. These effects are attributable to specific oil- and water-soluble organosulphur compounds such as thiosulphinates, which are responsible for the typical odour and flavour of garlic. The interest in studying the therapeutic properties of garlic on H. pylori-related diseases arose when an inverse relationship between garlic consumption and the incidence of gastric cancer was reported[142].
The in vitro anti-H. pylori activity of extracts and compounds obtained from garlic[143-147] has been extensively documented, although a few studies reported negative results[138,148]. An aqueous garlic extract, standardised for its thiosulphinate concentration, had a minimum inhibitory concentration (MIC) of 40 μg/mL, and for other garlic compounds (allicin, ajoenes, vinyldithiins, thiosulphinates), the MIC values were approximately 10 to 25 μg/mL[144,147,149]. However, in the few in vivo studies that have been carried out, the viability of the bacterium was not affected. For example, the inclusion of sliced garlic in the diet of patients did not result in a reduction of the infection as assessed by measuring H. pylori infection by the urease breath test[150]. Treatment with a 4 mg garlic oil capsule, administered four times a day for 14 d, also gave negative results[151]. Similar results were obtained when a 4% garlic extract was administered daily for 6 wk to Mongolian gerbils with H. pylori-induced gastritis, but in this case, a reduction in gastritis was observed[152].
The Shandong Intervention Trial began in Linqu County, Shandong, China (a rural region with a high incidence of gastric cancer) in 1995, with the aim of assessing interventions to reduce the prevalence of advanced precancerous gastric lesions. The individuals selected (3365 subjects) were randomly assigned to three interventions or placebos. The 3 interventions included a 2-wk course of amoxicillin and omeprazole (H. pylori treatment); the second group was treated for 7 years with an oral supplementation of a mixture of garlic extract and steam-distilled garlic oil, and the third group was treated for 7 years with vitamin supplementation. The trial demonstrated that amoxicillin and omeprazole significantly reduced the prevalence and the average of the histological severity of precancerous gastric lesions, whereas no statistically significant favourable effects were observed for garlic or vitamin supplements[153].
The results of a 14.7-year follow-up for gastric cancer incidence and cause-specific mortality among the subjects in the Shandong trial showed that the treatment with amoxicillin and omeprazole resulted in a statistically significant 39% reduction in gastric cancer incidence. A similar but non-statistically significant decline was observed for gastric cancer mortality. Neither garlic nor vitamin long-term supplementation was associated with a statistically significant decrease in gastric cancer incidence and mortality[154].
In another case-control study conducted to evaluate the effects of dietary and life-style habits of patients diagnosed with gastric cancer in Turkey, it was found that frequent consumption of garlic did not result in a lower gastric cancer risk[155].
The traditional medicinal value of broccoli is due to its anticancer, antiviral and antibacterial properties. Two pilot trials were conducted to evaluate the effect of short-term broccoli consumption (alone or in combination with yoghurt) on H. pylori eradication in infected volunteers. In one group, a temporally associated eradication of H. pylori infection was observed[156]; in the other group, the treatment was ineffective[157].
An epidemiological investigation of the relationship between broccoli consumption and chronic atrophic gastritis was conducted in 438 male employees, aged 39 to 60, at a Japanese steel company. Chronic atrophic gastritis was serologically determined by measuring pepsinogen I and the pepsinogen I/pepsinogen II ratio (biomarkers of gastric inflammation). Broccoli consumption (weekly frequency) in the diet was monitored by using a 31-item food frequency questionnaire. Unexpectedly, the results showed that broccoli consumption once or more weekly significantly increased the risk for chronic atrophic gastritis[158].
Isothiocyanate sulphoraphane (SF), an abundant compound in broccoli sprouts, has been identified as a potent bacteriostatic agent against H. pylori (in 48 tested strains, MIC media = 2 μg/mL); moreover, brief exposure to SF eliminated intracellular H. pylori from a human epithelial cell line (HEp-2). SF was effective in eradicating H. pylori from human gastric xenografts in nude mice[159].
Another in vivo approach was used to evaluate the efficacy of fresh broccoli sprouts in reducing H. pylori infection and its sequelae in an H. pylori-infected mouse model, as well as in infected human volunteers[127]. Oral treatment with SF-rich broccoli sprouts of C57BL mice infected with H. pylori and maintained on a high-salt diet (7.5% NaCl has been shown to exaggerate H. pylori-induced gastritis in mice) reduced gastric bacterial colonisation, attenuated mucosal expression of tumour necrosis factor-α and interleukin-1β, mitigated corpus inflammation, and prevented the expression of high salt-induced gastric corpus atrophy.
In the human trial, 48 H. pylori-infected volunteers were instructed to consume broccoli sprouts (70 g/d; containing 420 μmol of SF precursor) for 8 wk. The treatment decreased urease levels as measured by the urea breath test, H. pylori stool antigens, and serum pepsinogens I and II. Nevertheless, all values recovered to their original levels 2 mo after treatment discontinuation, indicating that broccoli sprout treatment does reduce H. pylori colonisation but does not completely eradicate it. In summary, evidence from mice and humans suggests that SF may have two mechanisms: a direct effect on H. pylori (bacteriostatic), leading to a reduction in gastritis, and an indirect effect by inducing the host cytoprotective response.
Green tea is one of the most widely consumed beverages worldwide, and it has been shown to inhibit the growth of Helicobacter spp. Remarkable antibiotic activity of green tea against H. pylori and H. felis has been demonstrated in vitro[160]. In in vivo models, green tea administration also gave encouraging results. In one case, green tea decreased the number of bacteria and the inflammatory score of H. pylori-infected C57BL/6J mice, but the greatest impact on these two parameters was obtained when mice received green tea prior to infection[160]. In another assay, infected Mongolian gerbils received green tea extract in drinking water (500, 1000 and 2000 ppm) for 6 wk; interestingly, gastritis and the prevalence of H. pylori-infected animals were suppressed in a dose-dependent manner[161].
Catechins, the main antioxidant compounds of green tea, also showed antibacterial activity against H. pylori in vitro and in vivo. Epigallocatechin gallate had the strongest activity, with an MIC = 8 μg/mL for 50% of the tested strains[122]. In infected Mongolian gerbils, the effect of catechins in the diet (2%) for 2 wk was associated with a low rate of bacterial eradication (36%), but with significant decreases in mucosal haemorrhage and erosion[122]. The authors suggest that combining catechins with a proton pump inhibitor and the inclusion of a drug delivery system that prolongs the gastric-transit time would improve the efficacy of catechins.
Green tea extract greatly inhibited H. pylori urease, with an IC50 value of 13 μg/mL. Catechins were identified as the active compounds, and the hydroxyl group at the 5’-position appeared to be important for urease inhibition[161]. Moreover, polyphenols present in green tea inhibited the vacuolisation effect induced by H. pylori VacA toxin[140].
The antibacterial activity of red wine against H. pylori has been tested. It has been proposed that resveratrol is, at least in part, the compound responsible for the activity (MIC range of 6.25-25 μg/mL)[162,163]. Moreover, resveratrol and red wine showed an inhibitory effect on H. pylori urease activity[163].
Red wine inhibits ion and urea conduction in vitro, as well as cell vacuolisation induced by VacA. These inhibitory effects were attributed to its polyphenol content, because these compounds inhibit the VacA channel[140].
It has been shown that administration of a red wine and green tea mixture, or a polyphenol mixture (tannic acid and n-propyl gallate) to H. pylori infected mice, or to mice treated with VacA toxin, limited gastric epithelium damage but did not significantly affect bacterial colonisation. These results suggest that VacA inhibition plays a role in this protective effect[139,164].
These data indicate that polyphenols or polyphenol-rich foods or beverages, such as green tea and red wine, may limit some of the symptomatology related to H. pylori infection.
Glycyrrhiza glabra Linn, commonly known as liquorice, is thought to be a useful treatment for peptic ulcers in traditional systems such as Indian, Chinese and Kampo medicine. Several studies have evaluated in vitro the antibiotic effect of liquorice and some of its metabolites on H. pylori[138,165,166]. Recently, a flavonoid rich extract of G. glabra, GutGard®, was studied to determine its anti-H. pylori activity and to elucidate its possible mechanism of action on the bacteria[135]. GutGard® had an MIC = 32-64 μg/mL, and glabridin, the major flavonoid present in the extract, had a more potent activity against H. pylori. The mechanism by which GutGard® exerts its antibiotic action could include inhibition of protein synthesis, DNA gyrase or dihydrofolate reductase. However, the adhesion of H. pylori to the human gastric adenocarcinoma cell line (AGS) was not significantly affected by GutGard®.
To assess the in vivo effect of GutGard on H. pylori colonisation, its effect was evaluated in two animal models[167]. After treating infected Mongolian gerbils (once daily 6 times/wk, for 8 wk) with 15, 30 and 60 mg/kg of the extract, a decrease in bacterial load was observed and the gastric mucosa of the animals did not present significant signs of inflammation. These results were more evident at the higher doses, with an 83% decrement on H. pylori infection. To test the efficiency of the extract for suppressing H. pylori colonisation in the early stages, C57BL/6 mice were infected and treated in the same way as the gerbils but with a 25 mg/kg GutGard dose for 3 wk. The results showed that GutGard treatment significantly reduced the ability of H. pylori to colonise the gastric mucosa. In conclusion, GutGard possesses significant in vivo anti-H. pylori properties and could be a natural resource to control bacterial associated gastric diseases. Clinical trials are required to test its effectiveness.
Honey is widely known for its antibacterial properties. The attributed antibacterial mechanisms are: an osmotic effect due to its sugar content, its hydrogen peroxide content (produced by the glucose oxidase added by the bee), its acidity, and other substances derived from flowers. Honey has been studied for its anti-H. pylori activity in vitro[168-171].
Manuka honey comes from one flower source. This honey has been shown to possess bacteriostatic properties against H. pylori at a 50 mL/L concentration[172].
Osato et al[169], in an effort to determine the role of osmotic effects and hydrogen peroxide in the inhibitory activity of honey in vitro, used control solutions containing glucose, fructose, combined glucose/fructose, and catalase. It was concluded that the anti-H. pylori activity was not related to the presence of hydrogen peroxide in the honey samples. However, the osmotic effect was shown to be the most important parameter for killing H. pylori at concentrations ≥ 150 mL/L.
In an in vitro assay, 8 commercial honey brands sold in Muscat, Oman, were tested for anti-H. pylori activity by a surface diffusion method and in combination with amoxicillin or clarithromycin. The results demonstrated that all of them had anti-H. pylori activity, but no synergy was observed, either with honey and clarithromycin or honey and amoxicillin[170]. These data suggest that a triple regimen with these honeys could help to eliminate the bacteria.
Recently, in an effort to find the active compounds of honey, two studies were performed. In the first one, 3 honeys from different regions of South Africa were tested for anti-H. pylori activity. “Pure Honey” presented the maximal inhibitory effect (73.3%) at 750 mL/L, so it was extracted with different organic solvents (n-hexane, diethyl ether, chloroform or ethyl acetate). All extracts demonstrated anti-H. pylori activity at concentrations ≥ 10%, but the chloroform extract had the lowest MIC95 value, ranging from 0.156-500 mL/L depending on the strain[173]. This suggests that all extracts could contain compounds that are inhibitory for the bacteria.
In the second work, a bioguided fractionation of a hexane extract from Golden Crest Honey was undertaken. The highest antibacterial activity was exhibited by fraction GCF3, with an MIC = 5 mg/mL[174]. This value is very high compared to commercial antibiotics (for amoxicillin, the MIC = 0.015 to 0.12 μg/mL for ATCC 43504, according to Clinical and Laboratory Standards Institute guidelines).
Finally, in the only clinical trial made with honey, 12 non-diabetic patients, positive for rapid urease and 14C urea breath tests, but with normal gastroscopies, were recruited. Six of them were treated with a tablespoon of Manuka honey four times a day for 2 wk and 6 were treated with honey and omeprazole (20 mg) twice a day for the same period. Four weeks after the completion of treatment, the twelve patients remained positive for H. pylori as demonstrated by 14C urea breath tests[175].
Honey has demonstrated anti-H. pylori activity, but more research must be conducted using animal models and in clinical trials to assess its efficiency as an alternative or complementary H. pylori therapy.
Propolis is a resinous mixture collected by honeybees from various plant sources to reinforce the structural stability of the hive and is thought to be a natural antibiotic. The exact composition of propolis depends on its botanical origin, but it has a high content of phenolic compounds. It has been reported that 30% ethanolic extracts of propolis have considerable in vitro inhibitory effect on the growth of several H. pylori clinical isolates, as assessed by agar-well diffusion, agar dilution, and disc diffusion methods[176]. A collection of phenolic compounds derived from propolis were evaluated for enzyme inhibition against H. pylori peptide deformylase (HpPDF). This enzyme catalyses the removal of formyl groups from the N-terminus of nascent polypeptide chains, which is essential for H. pylori survival, and is considered as a promising drug target for anti-H. pylori therapy. The results showed that caffeic acid phenethyl ester (CAPE), one of the main medicinal components of propolis, is a competitive inhibitor of HpPDF, with an IC50 = 4.02 μmol/L. Furthermore, absorption spectra and crystal structure characterisation revealed that is different from most well-known PDF inhibitors. CAPE blocks the substrate entrance to the active site, but has neither a chelative interaction with HpPDF, nor does it disturb metal-dependent catalysis[177].
In another in vitro study, 25 identified constituents of Brazilian propolis were tested for anti-H. pylori activity; 50% of them were active. The labdane type diterpenes and some prenylated phenolic compounds were the most active, with an MIC = 0.13 mg/mL[178]. A clinical trial evaluating a twenty drops/day therapy of a 4% alcoholic preparation of Brazilian propolis in 18 H. pylori positive patients showed that the use of green propolis preparation did not succeed in suppressing or eradicating H. pylori, as determined by a urea breath test at 3 and 40 d after the end of therapy[179].
According to the Food and Agriculture Organization and the World Health Organization[180], probiotics are “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host”. Interest in probiotic activity against H. pylori and its possible inclusion in the eradication therapy has increased because it represents a low-cost, large-scale alternative solution to prevent or decrease H. pylori colonisation.
There are numerous studies on probiotics; the scope of these investigations includes evaluations in vitro and in vivo in animal models, as well as in clinical trials. In fact, the knowledge that has been generated about probiotics and H. pylori deserves a separate review. We only report here a brief note about probiotics, highlighting some important findings.
Among probiotics, Bifidobacterium is one of the favourite genera used for the prevention of gastrointestinal infection, and it is commonly incorporated in fermented dairy products or food supplements. Bifidobacterium exerts an in vitro anti-H. pylori effect and inhibits adhesion to the mucosa by competition[181]. Several studies have demonstrated a direct relationship between the addition of potential probiotic strains and the in vitro inhibition of H. pylori growth. Lactobacillus acidophilus[182], Lactobacillus casei strain Shirota[183], Bacillus subtilis[184], and Weissella confusa[185], among others, have an antagonistic effect on H. pylori.
Several mechanisms have been hypothesised based on in vitro studies of host intestinal epithelial or immune cell responses to probiotic strains. Probiotic bacteria can inhibit H. pylori by either immunological or non-immunological mechanisms. In addition, distinct probiotic strains may generate divergent immune responses, depending on the host’s immune status[186]. Various probiotics have been shown to exert favourable effects in animal models of H. pylori infection. The main outcomes were a prophylactic effect against H. pylori, a reduction in bacterial colonisation, and alleviation of H. pylori-associated gastric inflammation[187]. The direct role of probiotics in the treatment of gastrointestinal infections is increasingly being documented as an alternative or a complement to antibiotics, with the potential to decrease the use of antibiotics or reduce their side effects. Patel et al[188] recently reviewed the in vivo clinical trials studying the effect of probiotics on H. pylori infection. They reported 12 human studies investigating the efficacy of antibiotic and probiotic combinations, and 16 studies using probiotics alone as an alternative to antibiotics for the infection treatment. The results indicated that in the majority of the cases, an improvement in H. pylori gastritis and a reduction in bacterial colonisation were associated with probiotics administration, and in any case, eradication could be completely attained. It also appeared that the use of probiotics was helpful to reduce the adverse effects associated with antibiotics. Long-term intakes of products containing probiotic strains may be beneficial in reducing the risk of H. pylori-associated complications.
Very potent compounds against H. pylori have been isolated from the actinomycete Pseudonocardia sp. CL38489 and from the basidiomycete Phanerochaete velutina CL6387. In Pseudonocardia sp. CL38489, 8 novel quinolones have been obtained with MIC values up to 0.1 ng/mL[189]. Phthalide compounds isolated from P. velutina were active against H. pylori. One of the most potent was CJ-12,954, with an MIC and a MBC value of 5 ng/mL[190]. Both types of compounds appear to be specific for H. pylori because they did not show antimicrobial activity when tested against a panel of other bacteria. Unfortunately, no more research on H. pylori and these compounds has been performed.
Recently, the anti-H. pylori activity of 14 basidiomycetes used in traditional Chinese medicine was screened by the agar diffusion method. The MIC values of 12 mushroom ethanol extracts were < 3 mg/mL. The best results were obtained for ethyl acetate fractions of Hericium erinaceus against 9 clinical isolates of H. pylori, with MIC values ranging between 62.5-250 μg/mL, and a MBC value of 200 μg/mL for the strain ATCC 43504[191].
In another finding, the triterpenoid methylantcinate B (MAB) isolated from the medicinal Chinese mushroom Antrodia camphorata displayed anti-H. pylori activity and inhibited H. pylori-associated inflammation in human gastric epithelial AGS cells by inhibiting adhesion, invasion, NF-κB activation, and release of IL-8[192]. The mechanism by which MAB inhibits H. pylori-induced inflammation in AGS cells may rely on attenuating CagA function. It is known that CagA interacts with membrane cholesterol provoking inflammation and that MAB competes with this interaction[193]. These reports suggest that it is possible that the pharmaceutical mechanism of secondary metabolites from mushrooms could be directly exerted on the bacteria and by immunomodulation.
Finally, a clinical study was performed using Tremella mesenterica, which reportedly has immunomodulatory activities. Fifty-two patients diagnosed with H. pylori infection were treated with 2 g/daily of submerged cultivated T. mesenterica mycelium for 10 d. The treatment was not effective at eradicating H. pylori, as determined by the urea breath test, whether it was administered in the presence or absence of omeprazole. Nevertheless, fewer adverse effects and a significant symptomatic relief were found among treated patients, so the authors suggest that studies should be conducted with a different administration scheme to obtain better results[194].
A promising substance in the fermentation broth of Streptomyces sp. strain HC-21 was isolated and identified as indolmycin (TAK-083). This antibiotic inhibits bacterial tryptophanyl-tRNA synthetase, but because it did not have enough activity against other common pathogenic bacteria, its study was abandoned. Nevertheless, indolmycin has a highly selective and potent anti-H. pylori activity, with an MIC90≤ 0.031 μg/mL, four-fold more potent than the currently available anti-H. pylori agents[195]. Additionally, indolmycin completely cleared H. pylori in experimentally infected Mongolian gerbils at a dose of 10 mg/kg. Therefore, this antibiotic was considered as a candidate for the treatment of H. pylori infection. Nevertheless, Vecchione and Sello[196] found a gene encoding an indolmycin-resistant isoform of tryptophanyl-tRNA synthetase. Overexpression of this gene in an indolmycin-sensitive strain increased the indolmycin MIC 60-fold. The authors speculated that homologs of this antibiotic-resistant gene could be found in other bacteria.
Antimicrobial peptides are cationic molecules. Although their precise mechanism of action is not yet defined, it is thought that they interact and lyse bacterial membranes. Many organisms produce peptides as part of their defence systems against invasive microorganisms. Amphibian skin glands are rich resources for antimicrobial peptides. Magainin 2[197] and odorranain-HP[198], obtained from Xenopus laevis and Odorrana graham, respectively, are antimicrobial peptides with a good anti-H. pylori activity. The antibacterial activities of normal and reversed magainin 2 synthetic analogues have been tested against two strains of H. pylori. Analogue MSI-78A had the strongest activity against H. pylori, with an MIC = 8 and 16 μg/mL for ATCC 43526 and ATCC 43579, respectively. The MIC values were similar to those against E. coli and S. aureus[197]. Later, the same group reported a new derivative of (±)-6 benzyl-1-(3-carboxylpropyl) indane that is more selective for H. pylori (MIC = 32 μg/mL) than for E. coli and S. aureus[199]. These results showed that with chemical modifications, it could be possible to obtain novel agents with more selective and stronger activity against the bacteria.
In another study, two peptides (S3, S5) obtained by enzymatic hydrolysis of seed proteins from pea (Pisum sativum) showed anti-adhesive properties. These peptides have their effect by interacting with H. pylori adhesin BabA, one of the outer membrane proteins involved in the adhesion of the bacteria to gastric epithelial cells[136]. This means that bioactive peptides from pea protein could be used in prophylaxis against H. pylori infection.
Antimicrobial peptides produced in the gastrointestinal tract are recognised as components of innate immunity against microorganisms. One family of antimicrobial peptides, the defensins, is produced by mucosal epithelial cells and by neutrophils. The cathelicidins comprise another group of mammalian antimicrobial proteins. The single known human cathelicidin is a cationic antibacterial protein of 18 kDa (hCAP18), whose C-terminal 37 amino acid peptide is termed LL-37.
Bajaj-Elliott et al[200] evaluated the role of β-defensins in the innate immune response of the gastric epithelium to H. pylori infection by measuring mRNA expression and regulation of human β-defensins 1 and 2 (hBD1, hBD2) in AGS and MKN (both, human gastric adenocarcinoma cell lines)7 cell lines, as well as in biopsies obtained from patients with histologically proven active gastritis (H. pylori positive). They found an increased expression of both defensins in vitro and in vivo in the presence of the bacteria.
A similar study examined the role of LL-37/hCAP18 and found that infection with H. pylori up-regulated the production of LL-37/hCAP18 by the gastric epithelium and increased LL-37 concentrations in gastric secretions from infected patients[201]. Unlike the previous study, they did not find a change in hBD1 mRNA levels when chronic mucosal inflammation was observed. They also determined the bactericidal activity of the peptides. LL-37 killed H. pylori strains SD4 and SD14 with an EC50 = 1.4 μmol/L in 3 h. On the other hand, hBD1 was capable of killing H. pylori SD4 and SD14 at 16 μmol/L, whereas hBD2 was not, and the combination of defensins had synergistic activity to kill bacteria. The difference in the bactericidal activity suggests that other factors besides the cationic nature of the peptides (e.g., differences in the membrane composition or structure of H. pylori) are involved in the bioactivity of these antimicrobial peptides.
Although these pilot studies require confirmation, the modulation of human antimicrobial peptide expression may be useful to improve control of the infection. It has been demonstrated that tomato defensins exert a broad antimicrobial spectrum. Based on a representative member of the tomato defensins family, the γ-motif of the peptide was chemically synthesised. It exhibited potent antibacterial activity against Gram-positive (MIC = 40 μg/mL) and Gram-negative bacteria, including H. pylori (MIC = 15 μg/mL)[202]. This activity could be due to a strong electrostatic interaction between the cationic nature of the peptide and the anionic bacterial membranes. In addition, the peptide down-regulated the level of proinflammatory cytokines and this effect was comparable with well-known anti-inflammatory drugs. Thus, the peptide displays 2 roles, acting against pathogens and reducing inflammation. These findings represent an alternative source for a new treatment of H. pylori illnesses.
The progression of diseases such as peptic ulcer or gastric carcinoma is restricted by secreted mucins in the deeper portion of the gastric mucosa. This effect is partly due to the expression of 1,4-N-acetylglucosamine residues attached to the mucin (MUC6). 1,4-N-acetylglucosamine residues inhibit cholesterol α-glucosyltransferase, the enzyme responsible for the first step in the biosynthesis of the unusual H. pylori major cell wall component, cholesteryl-α-D-glucopyranoside. This inhibitory activity is thus regarded as a natural antibiotic. Because cholesterol α-glucosyltransferase is unique to Helicobacter species[203], it is feasible that by inhibiting the enzyme, H. pylori could be eradicated without causing diarrhoea or other symptoms that arise due to adverse effects exerted on the local microbiota of the digestive tract. Moreover, cholesterol α-glucosyltransferase represents an entirely new target for drug development.
H. pylori is mainly found within the gastric mucus layer and attached to the epithelial cell surface. The carbohydrate structures present on the gastric mucosal surface include secreted and membrane bound mucins. It is known that H. pylori interacts with the secreted gel-forming mucin MUC5AC[204], but to establish the infection, adherence to gastric epithelial cells is required. Both interactions (with mucin and cells) occur via lectin-like molecules and specific carbohydrate structure recognition. Therefore, an approach using polysaccharides to block the interaction between H. pylori and the host might prevent infection. Under this principle, the likelihood of developing bacterial resistance is unlikely.
Several crude and purified polysaccharides from different sources have been isolated, analysed and examined for their effects against H. pylori. Because algae and microalgae possess high concentrations of polysaccharides with several biological activities, their anti-adhesive properties have been explored. Fucoidan, a sulphated polysaccharide mainly found in the edible brown algae Cladosiphon okamuranus Tokida, had an inhibitory effect on H. pylori attachment to porcine gastric mucin in vitro, and reduced H. pylori-induced gastritis and the prevalence of infection in Mongolian gerbils[205]. In another in vitro study, polysaccharides derived from Spirulina and Chlorella (two commercially available dietary microalgae), prevented H. pylori from binding to porcine gastric mucin at low pH without killing the bacteria and AGS cells. Moreover, Spirulina polysaccharides administered 3 times per week for 4 wk before infection with H. pylori reduced by > 90% the bacterial density in mice, demonstrating its effectiveness as a carbohydrate-based anti-adhesive treatment[206].
Plant polysaccharides have also been studied for their anti-adhesive properties. An acidic pectin-type polysaccharide from green tea (Camellia sinensis) had a selective inhibitory activity on the adhesion of H. pylori to AGS cells, without effects against commensal bacteria. The anti-adhesive activity obtained with C. sinensis was better than the that of Artemisia capillaris and Panax ginseng acidic polysaccharides[207]. Liquorice root is another plant that may act against H. pylori. An aqueous extract, and a derived polysaccharide fraction, inhibited the adhesion of H. pylori to human gastric tissue by interacting with bacterial adhesines and not with binding sites on the epithelial cells[208]. Black currant (Ribes nigrum L.) seeds also possess acidic polysaccharides with anti-adhesive properties. High molecular weight galactans are responsible for the activity, by interacting with H. pylori adhesins[209].
In general, polysaccharides do not inhibit bacterial growth in vitro, but their anti-adhesive properties could be very valuable to prevent or to treat H. pylori infection, or even to prevent reinfection after antibiotic eradication therapy. Because the sources of these compounds are easily available, carbohydrate-based anti-adhesive treatment could represent a low cost and safe alternative.
H. pylori accumulates photoactive porphyrins, making the organism susceptible to inactivation by light. A controlled, prospective pilot trial of eighteen adults with H. pylori infection was conducted using a novel light source consisting of laser diodes and diffusing fibres to deliver 408-nm illumination to the whole stomach at escalating total fluences. The results showed an important reduction in bacterial load in the antrum (> 97%), followed by the body (> 95%) and the fundus (> 86%). There was a correlation between logarithmic reduction and initial bacterial load in the antrum. No dose response was observed with increasing illumination times. Nevertheless, the urea breath test results indicated that the bacteria repopulated within a few days after illumination. Although none of the patients achieved complete and sustained eradication of H. pylori with this therapy, the results of this study are promising. Intra-gastric ultraviolet light phototherapy is feasible and safe and may represent a novel approach for the eradication of H. pylori, particularly in patients who have failed standard antibiotic treatment[210].
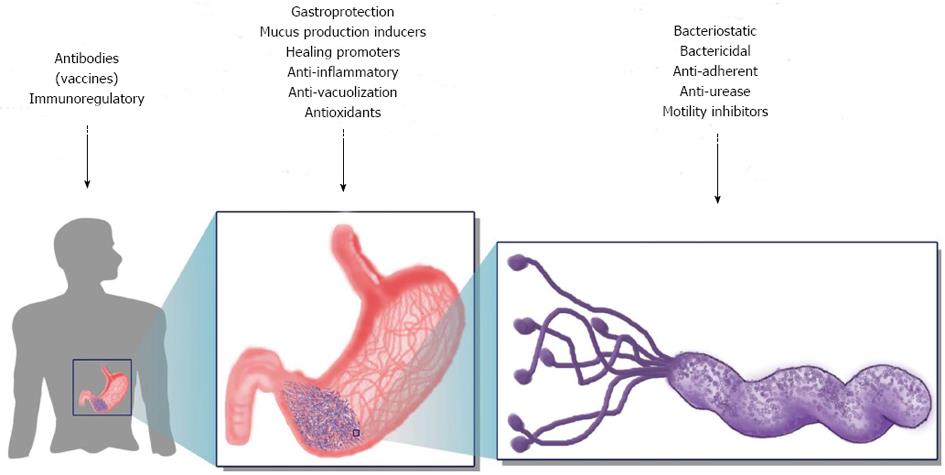
The serious gastric diseases associated with H. pylori infection still constitute a public health threat in different parts of the world. Since the difficulties created by standard therapies (i.e., triple, quadruple) to treat H. pylori-associated diseases have been recognised, several alternative options that focus on prevention and eradication have been proposed. Because the transmission route is not well defined, prevention can be achieved by blocking the colonisation pathways or through prophylactic vaccination. Antibiotics are the only existing choice for eradication, so efforts should be directed toward finding novel anti-H. pylori agents. The current state of research in terms of alternative methods is focused on vaccines, phytotherapy, probiotic-based diets, and nutraceutical agents. The mechanisms and targets used in these alternatives strategies are summarised in Figure 2.
Although large number of studies have been carried out, to date, neither a prophylactic nor a therapeutic vaccine to prevent or clear the infection in humans has been described. Three factors that have delayed the development of an effective vaccine have been identified: (1) type of antigen; (2) need for an adjuvant; and (3) inappropriate host immune response. (1) Of the new developments, the use of multivalent antigens has shown promise. They can target different elements: the enzyme pathways that are vital for bacterial survival and that are common to all strains, the bacterial determinants of pathogenesis, or the factors involved in the induction of the inflammatory immune response; (2) regarding adjuvants, native or attenuated bacterial toxins are the most commonly used; however, their use in humans has been restricted due to their side effects. To date, the only FDA-approved human adjuvant for parenteral vaccines is aluminium hydroxide. A promising alternative for human use could be to employ live attenuated Salmonella bacteria expressing multiple H. pylori antigens, since as these bacteria do not require an adjuvant. Some data have shown that the combination of mucosal and systemic immunisation can enhance long-term protection against the H. pylori infection; and (3) the failure of the human host response to eliminate the H. pylori infection is determined by several factors. First, failure can be explained by poor activation of Toll-like receptors during the innate response. Some bacterial factors, such as VacA, inhibit the adaptive response by blocking antigen presentation and inhibiting T-cell proliferation. Additionally, the induction of Treg cells, which suppress the Th17-cell response, enables the persistence of bacteria.
On the other hand, the few clinical trials that have been carried out have succeeded in terms of neither immunisation nor bacterial clearance. Phase I/II clinical studies are needed before a safe and effective vaccine against H. pylori can be obtained. In conclusion, to date, no commercial vaccine is available, and substantial work is required to develop a promising one.
In view of this lack of effective treatment and prevention options, other alternatives, including natural products, such as probiotics and nutraceuticals, have been studied, due to their low cost and relative safety (people have consumed them for a long time). However, the research that has focused primarily on bacterial eradication has had an important limitation, i.e., the lack of correlation between in vitro susceptibility and in vivo efficacy; only a few studies have also demonstrated in vivo efficacy. On the other hand, in those cases where the in vivo treatments were successful, either the number of individuals (animals or patients) involved was not statistically significant or only a specific population was analysed. All in all, the results are not completely satisfactory because in most cases H. pylori eradication is not achieved. Nevertheless, a reduction in the pathological outcome of the illness and a good resolution of the symptomatology is accomplished. In addition, when alternative treatments are combined with allopathic ones, side effects are reduced and the success rate is increased.
There is an inverse relationship between the low rates of H. pylori eradication and the appearance of side effects associated with the current therapies. The most common adverse effects observed in patients treated for H. pylori eradication include abdominal discomfort, diarrhoea, nausea, vomiting, headache and weakness; furthermore, these symptoms have an impact on treatment compliance. The inclusion of alternative treatments in the anti-H. pylori scheme would enhance both the effectiveness of the therapy and the resolution of the pathology. Moreover, eradication rates would increase, and the development of bacterial resistance could be avoided.
Although some of the treatments based on natural products include instructions for their use, there are no precise indications as to how these products could be integrated into a structured health program. It is clear that further efforts are required to generate more information for the establishment of new options for the treatment of H. pylori-related diseases.
A high percentage of H. pylori-infected individuals remain asymptomatic, but they are still at risk of developing the pathologies associated with H. pylori. However, the eradication of H. pylori in the case of asymptomatic patients is not recommended. Alternative therapies have proved to be useful in maintaining low bacterial levels, controlling inflammation, modulating the immune response, inhibiting adherence to the gastric epithelium, and neutralising some of the bacterial virulence factors such as the urease enzyme and the vacuolating toxin. Based on these results, the inclusion of natural products in the diet of asymptomatic patients could reduce the risk, as well as the development, of an unfavourable outcome of the infection.
In conclusion, there are no results indicating that any alternative treatment can truly eradicate H. pylori. In contrast, most of the findings have demonstrated that some agents exhibit good anti-inflammatory, immunomodulatory and gastro-protective activities, which as a whole favour the resolution of gastric damage despite the fact that H. pylori is not completely eliminated. Therefore, those agents can be used as adjuvants of allopathic anti-H. pylori eradication therapy, but not as a monotherapy. As long as a vaccine or new antibiotics remain unavailable, the synergism of allopathic and alternative treatments is most likely the best choice.
Another option would be to combine alternative treatments with no or few adverse effects: using one with high anti-H. pylori activity and another offering resolution of gastric damage. Finally, in coming years, the development of new proposals for the eradication of H. pylori, such as phototherapy and the use of antimicrobial peptides and mucins, will be followed with great interest.
P- Reviewers: Lee SH, Puntel RL, Zhang XW S- Editor: Cui XM L- Editor: A E- Editor: Wu HL
| 1. | Clyne M, Dolan B, Reeves EP. Bacterial factors that mediate colonization of the stomach and virulence of Helicobacter pylori. FEMS Microbiol Lett. 2007;268:135-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 2. | Dunn BE, Campbell GP, Perez-Perez GI, Blaser MJ. Purification and characterization of urease from Helicobacter pylori. J Biol Chem. 1990;265:9464-9469. [PubMed] |
| 3. | Eaton KA, Krakowka S. Effect of gastric pH on urease-dependent colonization of gnotobiotic piglets by Helicobacter pylori. Infect Immun. 1994;62:3604-3607. [PubMed] |
| 4. | Harris PR, Mobley HL, Perez-Perez GI, Blaser MJ, Smith PD. Helicobacter pylori urease is a potent stimulus of mononuclear phagocyte activation and inflammatory cytokine production. Gastroenterology. 1996;111:419-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 160] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 5. | Koch KS, Leffert HL. Hypothesis: Targeted Ikkβ deletion upregulates MIF signaling responsiveness and MHC class II expression in mouse hepatocytes. Hepat Med. 2010;2010:39-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 77] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 6. | Ilver D, Arnqvist A, Ogren J, Frick IM, Kersulyte D, Incecik ET, Berg DE, Covacci A, Engstrand L, Borén T. Helicobacter pylori adhesin binding fucosylated histo-blood group antigens revealed by retagging. Science. 1998;279:373-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 872] [Cited by in RCA: 842] [Article Influence: 31.2] [Reference Citation Analysis (1)] |
| 7. | Mahdavi J, Sondén B, Hurtig M, Olfat FO, Forsberg L, Roche N, Angstrom J, Larsson T, Teneberg S, Karlsson KA. Helicobacter pylori SabA adhesin in persistent infection and chronic inflammation. Science. 2002;297:573-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 639] [Cited by in RCA: 665] [Article Influence: 28.9] [Reference Citation Analysis (0)] |
| 8. | Israel DA, Peek RM. Pathogenesis of Helicobacter pylori-induced gastric inflammation. Aliment Pharmacol Ther. 2001;15:1271-1290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 107] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 9. | Gerhard M, Lehn N, Neumayer N, Borén T, Rad R, Schepp W, Miehlke S, Classen M, Prinz C. Clinical relevance of the Helicobacter pylori gene for blood-group antigen-binding adhesin. Proc Natl Acad Sci USA. 1999;96:12778-12783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 431] [Cited by in RCA: 434] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 10. | Prinz C, Schöniger M, Rad R, Becker I, Keiditsch E, Wagenpfeil S, Classen M, Rösch T, Schepp W, Gerhard M. Key importance of the Helicobacter pylori adherence factor blood group antigen binding adhesin during chronic gastric inflammation. Cancer Res. 2001;61:1903-1909. [PubMed] |
| 11. | Senkovich OA, Yin J, Ekshyyan V, Conant C, Traylor J, Adegboyega P, McGee DJ, Rhoads RE, Slepenkov S, Testerman TL. Helicobacter pylori AlpA and AlpB bind host laminin and influence gastric inflammation in gerbils. Infect Immun. 2011;79:3106-3116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 87] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 12. | Peck B, Ortkamp M, Diehl KD, Hundt E, Knapp B. Conservation, localization and expression of HopZ, a protein involved in adhesion of Helicobacter pylori. Nucleic Acids Res. 1999;27:3325-3333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 132] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 13. | Yamaoka Y, Kwon DH, Graham DY. A M(r) 34,000 proinflammatory outer membrane protein (oipA) of Helicobacter pylori. Proc Natl Acad Sci USA. 2000;97:7533-7538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 292] [Cited by in RCA: 318] [Article Influence: 12.7] [Reference Citation Analysis (2)] |
| 14. | Yamaoka Y, Kikuchi S, el-Zimaity HM, Gutierrez O, Osato MS, Graham DY. Importance of Helicobacter pylori oipA in clinical presentation, gastric inflammation, and mucosal interleukin 8 production. Gastroenterology. 2002;123:414-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 229] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 15. | Hessey SJ, Spencer J, Wyatt JI, Sobala G, Rathbone BJ, Axon AT, Dixon MF. Bacterial adhesion and disease activity in Helicobacter associated chronic gastritis. Gut. 1990;31:134-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 207] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 16. | Tonello F, Dundon WG, Satin B, Molinari M, Tognon G, Grandi G, Del Giudice G, Rappuoli R, Montecucco C. The Helicobacter pylori neutrophil-activating protein is an iron-binding protein with dodecameric structure. Mol Microbiol. 1999;34:238-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 132] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 17. | Satin B, Del Giudice G, Della Bianca V, Dusi S, Laudanna C, Tonello F, Kelleher D, Rappuoli R, Montecucco C, Rossi F. The neutrophil-activating protein (HP-NAP) of Helicobacter pylori is a protective antigen and a major virulence factor. J Exp Med. 2000;191:1467-1476. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 236] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 18. | Polenghi A, Bossi F, Fischetti F, Durigutto P, Cabrelle A, Tamassia N, Cassatella MA, Montecucco C, Tedesco F, de Bernard M. The neutrophil-activating protein of Helicobacter pylori crosses endothelia to promote neutrophil adhesion in vivo. J Immunol. 2007;178:1312-1320. [PubMed] |
| 19. | Amedei A, Cappon A, Codolo G, Cabrelle A, Polenghi A, Benagiano M, Tasca E, Azzurri A, D’Elios MM, Del Prete G. The neutrophil-activating protein of Helicobacter pylori promotes Th1 immune responses. J Clin Invest. 2006;116:1092-1101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 251] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 20. | Stent A, Every AL, Sutton P. Helicobacter pylori defense against oxidative attack. Am J Physiol Gastrointest Liver Physiol. 2012;302:G579-G587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 21. | Lundström AM, Sundaeus V, Bölin I. The 26-kilodalton, AhpC homologue, of Helicobacter pylori is also produced by other Helicobacter species. Helicobacter. 2001;6:44-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 22. | Wang G, Conover RC, Benoit S, Olczak AA, Olson JW, Johnson MK, Maier RJ. Role of a bacterial organic hydroperoxide detoxification system in preventing catalase inactivation. J Biol Chem. 2004;279:51908-51914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 47] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 23. | Amieva MR, El-Omar EM. Host-bacterial interactions in Helicobacter pylori infection. Gastroenterology. 2008;134:306-323. [PubMed] |
| 24. | Parsonnet J, Friedman GD, Orentreich N, Vogelman H. Risk for gastric cancer in people with CagA positive or CagA negative Helicobacter pylori infection. Gut. 1997;40:297-301. [PubMed] |
| 25. | Iwamoto H, Czajkowsky DM, Cover TL, Szabo G, Shao Z. VacA from Helicobacter pylori: a hexameric chloride channel. FEBS Lett. 1999;450:101-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 102] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 26. | Tombola F, Morbiato L, Del Giudice G, Rappuoli R, Zoratti M, Papini E. The Helicobacter pylori VacA toxin is a urea permease that promotes urea diffusion across epithelia. J Clin Invest. 2001;108:929-937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 27. | Papini E, Satin B, Norais N, de Bernard M, Telford JL, Rappuoli R, Montecucco C. Selective increase of the permeability of polarized epithelial cell monolayers by Helicobacter pylori vacuolating toxin. J Clin Invest. 1998;102:813-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 185] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 28. | Molinari M, Salio M, Galli C, Norais N, Rappuoli R, Lanzavecchia A, Montecucco C. Selective inhibition of Ii-dependent antigen presentation by Helicobacter pylori toxin VacA. J Exp Med. 1998;187:135-140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 226] [Cited by in RCA: 208] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 29. | Yamasaki E, Wada A, Kumatori A, Nakagawa I, Funao J, Nakayama M, Hisatsune J, Kimura M, Moss J, Hirayama T. Helicobacter pylori vacuolating cytotoxin induces activation of the proapoptotic proteins Bax and Bak, leading to cytochrome c release and cell death, independent of vacuolation. J Biol Chem. 2006;281:11250-11259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 120] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 30. | Torres VJ, VanCompernolle SE, Sundrud MS, Unutmaz D, Cover TL. Helicobacter pylori vacuolating cytotoxin inhibits activation-induced proliferation of human T and B lymphocyte subsets. J Immunol. 2007;179:5433-5440. [PubMed] |
| 31. | Sewald X, Gebert-Vogl B, Prassl S, Barwig I, Weiss E, Fabbri M, Osicka R, Schiemann M, Busch DH, Semmrich M. Integrin subunit CD18 Is the T-lymphocyte receptor for the Helicobacter pylori vacuolating cytotoxin. Cell Host Microbe. 2008;3:20-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 90] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 32. | Censini S, Lange C, Xiang Z, Crabtree JE, Ghiara P, Borodovsky M, Rappuoli R, Covacci A. cag, a pathogenicity island of Helicobacter pylori, encodes type I-specific and disease-associated virulence factors. Proc Natl Acad Sci USA. 1996;93:14648-14653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1375] [Cited by in RCA: 1392] [Article Influence: 48.0] [Reference Citation Analysis (0)] |
| 33. | Rohde M, Püls J, Buhrdorf R, Fischer W, Haas R. A novel sheathed surface organelle of the Helicobacter pylori cag type IV secretion system. Mol Microbiol. 2003;49:219-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 202] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 34. | Kwok T, Zabler D, Urman S, Rohde M, Hartig R, Wessler S, Misselwitz R, Berger J, Sewald N, König W. Helicobacter exploits integrin for type IV secretion and kinase activation. Nature. 2007;449:862-866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 473] [Cited by in RCA: 508] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 35. | Conradi J, Tegtmeyer N, Woźna M, Wissbrock M, Michalek C, Gagell C, Cover TL, Frank R, Sewald N, Backert S. An RGD helper sequence in CagL of Helicobacter pylori assists in interactions with integrins and injection of CagA. Front Cell Infect Microbiol. 2012;2:70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 36. | Gorrell RJ, Guan J, Xin Y, Tafreshi MA, Hutton ML, McGuckin MA, Ferrero RL, Kwok T. A novel NOD1- and CagA-independent pathway of interleukin-8 induction mediated by the Helicobacter pylori type IV secretion system. Cell Microbiol. 2012;Oct 26; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 77] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 37. | Wroblewski LE, Peek RM, Wilson KT. Helicobacter pylori and gastric cancer: factors that modulate disease risk. Clin Microbiol Rev. 2010;23:713-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 817] [Cited by in RCA: 1010] [Article Influence: 67.3] [Reference Citation Analysis (1)] |
| 38. | Suzuki M, Mimuro H, Suzuki T, Park M, Yamamoto T, Sasakawa C. Interaction of CagA with Crk plays an important role in Helicobacter pylori-induced loss of gastric epithelial cell adhesion. J Exp Med. 2005;202:1235-1247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 185] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 39. | Selbach M, Moese S, Hurwitz R, Hauck CR, Meyer TF, Backert S. The Helicobacter pylori CagA protein induces cortactin dephosphorylation and actin rearrangement by c-Src inactivation. EMBO J. 2003;22:515-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 190] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 40. | Churin Y, Al-Ghoul L, Kepp O, Meyer TF, Birchmeier W, Naumann M. Helicobacter pylori CagA protein targets the c-Met receptor and enhances the motogenic response. J Cell Biol. 2003;161:249-255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 283] [Cited by in RCA: 293] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 41. | Amieva MR, Vogelmann R, Covacci A, Tompkins LS, Nelson WJ, Falkow S. Disruption of the epithelial apical-junctional complex by Helicobacter pylori CagA. Science. 2003;300:1430-1434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 598] [Cited by in RCA: 583] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 42. | Saadat I, Higashi H, Obuse C, Umeda M, Murata-Kamiya N, Saito Y, Lu H, Ohnishi N, Azuma T, Suzuki A. Helicobacter pylori CagA targets PAR1/MARK kinase to disrupt epithelial cell polarity. Nature. 2007;447:330-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 351] [Cited by in RCA: 395] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 43. | Murata-Kamiya N, Kurashima Y, Teishikata Y, Yamahashi Y, Saito Y, Higashi H, Aburatani H, Akiyama T, Peek RM, Azuma T. Helicobacter pylori CagA interacts with E-cadherin and deregulates the beta-catenin signal that promotes intestinal transdifferentiation in gastric epithelial cells. Oncogene. 2007;26:4617-4626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 361] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 44. | Brandt S, Kwok T, Hartig R, König W, Backert S. NF-kappaB activation and potentiation of proinflammatory responses by the Helicobacter pylori CagA protein. Proc Natl Acad Sci USA. 2005;102:9300-9305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 394] [Cited by in RCA: 420] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 45. | Pillinger MH, Marjanovic N, Kim SY, Lee YC, Scher JU, Roper J, Abeles AM, Izmirly PI, Axelrod M, Pillinger MY. Helicobacter pylori stimulates gastric epithelial cell MMP-1 secretion via CagA-dependent and -independent ERK activation. J Biol Chem. 2007;282:18722-18731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 52] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 46. | Arachchi HS, Kalra V, Lal B, Bhatia V, Baba CS, Chakravarthy S, Rohatgi S, Sarma PM, Mishra V, Das B. Prevalence of duodenal ulcer-promoting gene (dupA) of Helicobacter pylori in patients with duodenal ulcer in North Indian population. Helicobacter. 2007;12:591-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 54] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 47. | Douraghi M, Mohammadi M, Oghalaie A, Abdirad A, Mohagheghi MA, Hosseini ME, Zeraati H, Ghasemi A, Esmaieli M, Mohajerani N. dupA as a risk determinant in Helicobacter pylori infection. J Med Microbiol. 2008;57:554-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 45] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 48. | Argent RH, Burette A, Miendje Deyi VY, Atherton JC. The presence of dupA in Helicobacter pylori is not significantly associated with duodenal ulceration in Belgium, South Africa, China, or North America. Clin Infect Dis. 2007;45:1204-1206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 76] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 49. | Ishii KJ, Koyama S, Nakagawa A, Coban C, Akira S. Host innate immune receptors and beyond: making sense of microbial infections. Cell Host Microbe. 2008;3:352-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 367] [Cited by in RCA: 383] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 50. | Takeda K, Akira S. Toll receptors and pathogen resistance. Cell Microbiol. 2003;5:143-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 212] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 51. | Brodsky IE, Monack D. NLR-mediated control of inflammasome assembly in the host response against bacterial pathogens. Semin Immunol. 2009;21:199-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 108] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 52. | Muotiala A, Helander IM, Pyhälä L, Kosunen TU, Moran AP. Low biological activity of Helicobacter pylori lipopolysaccharide. Infect Immun. 1992;60:1714-1716. [PubMed] |
| 53. | Bliss CM, Golenbock DT, Keates S, Linevsky JK, Kelly CP. Helicobacter pylori lipopolysaccharide binds to CD14 and stimulates release of interleukin-8, epithelial neutrophil-activating peptide 78, and monocyte chemotactic protein 1 by human monocytes. Infect Immun. 1998;66:5357-5363. [PubMed] |
| 54. | Gewirtz AT, Yu Y, Krishna US, Israel DA, Lyons SL, Peek RM. Helicobacter pylori flagellin evades toll-like receptor 5-mediated innate immunity. J Infect Dis. 2004;189:1914-1920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 214] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 55. | Lee SK, Stack A, Katzowitsch E, Aizawa SI, Suerbaum S, Josenhans C. Helicobacter pylori flagellins have very low intrinsic activity to stimulate human gastric epithelial cells via TLR5. Microbes Infect. 2003;5:1345-1356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 135] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 56. | Viala J, Chaput C, Boneca IG, Cardona A, Girardin SE, Moran AP, Athman R, Mémet S, Huerre MR, Coyle AJ. Nod1 responds to peptidoglycan delivered by the Helicobacter pylori cag pathogenicity island. Nat Immunol. 2004;5:1166-1174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 906] [Cited by in RCA: 926] [Article Influence: 44.1] [Reference Citation Analysis (0)] |
| 57. | Kaparakis M, Turnbull L, Carneiro L, Firth S, Coleman HA, Parkington HC, Le Bourhis L, Karrar A, Viala J, Mak J. Bacterial membrane vesicles deliver peptidoglycan to NOD1 in epithelial cells. Cell Microbiol. 2010;12:372-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 342] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 58. | Kim YG, Park JH, Shaw MH, Franchi L, Inohara N, Núñez G. The cytosolic sensors Nod1 and Nod2 are critical for bacterial recognition and host defense after exposure to Toll-like receptor ligands. Immunity. 2008;28:246-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 216] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 59. | Wilson KT, Crabtree JE. Immunology of Helicobacter pylori: insights into the failure of the immune response and perspectives on vaccine studies. Gastroenterology. 2007;133:288-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 191] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 60. | Ermak TH, Giannasca PJ, Nichols R, Myers GA, Nedrud J, Weltzin R, Lee CK, Kleanthous H, Monath TP. Immunization of mice with urease vaccine affords protection against Helicobacter pylori infection in the absence of antibodies and is mediated by MHC class II-restricted responses. J Exp Med. 1998;188:2277-2288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 329] [Cited by in RCA: 321] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 61. | Hitzler I, Oertli M, Becher B, Agger EM, Müller A. Dendritic cells prevent rather than promote immunity conferred by a Helicobacter vaccine using a mycobacterial adjuvant. Gastroenterology. 2011;141:186-196, 196.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 62. | Akhiani AA, Pappo J, Kabok Z, Schön K, Gao W, Franzén LE, Lycke N. Protection against Helicobacter pylori infection following immunization is IL-12-dependent and mediated by Th1 cells. J Immunol. 2002;169:6977-6984. [PubMed] |
| 63. | Sayi A, Kohler E, Hitzler I, Arnold I, Schwendener R, Rehrauer H, Müller A. The CD4+ T cell-mediated IFN-gamma response to Helicobacter infection is essential for clearance and determines gastric cancer risk. J Immunol. 2009;182:7085-7101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 140] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 64. | Velin D, Favre L, Bernasconi E, Bachmann D, Pythoud C, Saiji E, Bouzourene H, Michetti P. Interleukin-17 is a critical mediator of vaccine-induced reduction of Helicobacter infection in the mouse model. Gastroenterology. 2009;136:2237-2246.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 110] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 65. | Wan YY, Flavell RA. How diverse--CD4 effector T cells and their functions. J Mol Cell Biol. 2009;1:20-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 142] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 66. | Kabir S. The role of interleukin-17 in the Helicobacter pylori induced infection and immunity. Helicobacter. 2011;16:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 71] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 67. | Salama NR, Hartung ML, Müller A. Life in the human stomach: persistence strategies of the bacterial pathogen Helicobacter pylori. Nat Rev Microbiol. 2013;11:385-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 405] [Cited by in RCA: 477] [Article Influence: 39.8] [Reference Citation Analysis (2)] |
| 68. | O’Keeffe J, Moran AP. Conventional, regulatory, and unconventional T cells in the immunologic response to Helicobacter pylori. Helicobacter. 2008;13:1-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 37] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 69. | Oertli M, Sundquist M, Hitzler I, Engler DB, Arnold IC, Reuter S, Maxeiner J, Hansson M, Taube C, Quiding-Järbrink M. DC-derived IL-18 drives Treg differentiation, murine Helicobacter pylori-specific immune tolerance, and asthma protection. J Clin Invest. 2012;122:1082-1096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 247] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 70. | Enarsson K, Lundgren A, Kindlund B, Hermansson M, Roncador G, Banham AH, Lundin BS, Quiding-Järbrink M. Function and recruitment of mucosal regulatory T cells in human chronic Helicobacter pylori infection and gastric adenocarcinoma. Clin Immunol. 2006;121:358-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 79] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 71. | Lundin BS, Enarsson K, Kindlund B, Lundgren A, Johnsson E, Quiding-Järbrink M, Svennerholm AM. The local and systemic T-cell response to Helicobacter pylori in gastric cancer patients is characterised by production of interleukin-10. Clin Immunol. 2007;125:205-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 72. | Enarsson K, Lundin BS, Johnsson E, Brezicka T, Quiding-Järbrink M. CD4+ CD25high regulatory T cells reduce T cell transendothelial migration in cancer patients. Eur J Immunol. 2007;37:282-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 73. | Li X, Kostareli E, Suffner J, Garbi N, Hämmerling GJ. Efficient Treg depletion induces T-cell infiltration and rejection of large tumors. Eur J Immunol. 2010;40:3325-3335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 109] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 74. | Teng MW, Ngiow SF, von Scheidt B, McLaughlin N, Sparwasser T, Smyth MJ. Conditional regulatory T-cell depletion releases adaptive immunity preventing carcinogenesis and suppressing established tumor growth. Cancer Res. 2010;70:7800-7809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 164] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 75. | Hamlet A, Thoreson AC, Nilsson O, Svennerholm AM, Olbe L. Duodenal Helicobacter pylori infection differs in cagA genotype between asymptomatic subjects and patients with duodenal ulcers. Gastroenterology. 1999;116:259-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 109] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 76. | Kindlund B, Sjöling A, Hansson M, Edebo A, Hansson LE, Sjövall H, Svennerholm AM, Lundin BS. FOXP3-expressing CD4(+) T-cell numbers increase in areas of duodenal gastric metaplasia and are associated to CD4(+) T-cell aggregates in the duodenum of Helicobacter pylori-infected duodenal ulcer patients. Helicobacter. 2009;14:192-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 77. | Strömberg E, Edebo A, Svennerholm AM, Lindholm C. Decreased epithelial cytokine responses in the duodenal mucosa of Helicobacter pylori-infected duodenal ulcer patients. Clin Diagn Lab Immunol. 2003;10:116-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 78. | Khulusi S, Badve S, Patel P, Lloyd R, Marrero JM, Finlayson C, Mendall MA, Northfield TC. Pathogenesis of gastric metaplasia of the human duodenum: role of Helicobacter pylori, gastric acid, and ulceration. Gastroenterology. 1996;110:452-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 59] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 79. | Cheung TK, Wong BC. Treatment of Helicobacter pylori and prevention of gastric cancer. J Dig Dis. 2008;9:8-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 80. | Pimanov SI, Makarenko EV, Voropaeva AV, Matveenko ME, Voropaev EV. Helicobacter pylori eradication improves gastric histology and decreases serum gastrin, pepsinogen I and pepsinogen II levels in patients with duodenal ulcer. J Gastroenterol Hepatol. 2008;23:1666-1671. [PubMed] |
| 81. | Malfertheiner P, Megraud F, O’Morain C, Bazzoli F, El-Omar E, Graham D, Hunt R, Rokkas T, Vakil N, Kuipers EJ. Current concepts in the management of Helicobacter pylori infection: the Maastricht III Consensus Report. Gut. 2007;56:772-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1396] [Cited by in RCA: 1349] [Article Influence: 74.9] [Reference Citation Analysis (1)] |
| 82. | Coelho LG, León-Barúa R, Quigley EM. Latin-American Consensus Conference on Helicobacter pylori infection. Latin-American National Gastroenterological Societies affiliated with the Inter-American Association of Gastroenterology (AIGE). Am J Gastroenterol. 2000;95:2688-2691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 68] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 83. | Chey WD, Wong BC. American College of Gastroenterology guideline on the management of Helicobacter pylori infection. Am J Gastroenterol. 2007;102:1808-1825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 819] [Cited by in RCA: 829] [Article Influence: 46.1] [Reference Citation Analysis (3)] |
| 84. | Malfertheiner P, Selgrad M. Helicobacter pylori infection and current clinical areas of contention. Curr Opin Gastroenterol. 2010;26:618-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 85. | Graham DY, Fischbach L. Helicobacter pylori treatment in the era of increasing antibiotic resistance. Gut. 2010;59:1143-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 652] [Cited by in RCA: 735] [Article Influence: 49.0] [Reference Citation Analysis (1)] |
| 86. | Graham DY, Shiotani A. New concepts of resistance in the treatment of Helicobacter pylori infections. Nat Clin Pract Gastroenterol Hepatol. 2008;5:321-331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 270] [Cited by in RCA: 281] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 87. | Suzuki H, Nishizawa T, Hibi T. Helicobacter pylori eradication therapy. Future Microbiol. 2010;5:639-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 88. | Kabir S. The current status of Helicobacter pylori vaccines: a review. Helicobacter. 2007;12:89-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 45] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 89. | Malfertheiner P, Schultze V, Rosenkranz B, Kaufmann SH, Ulrichs T, Novicki D, Norelli F, Contorni M, Peppoloni S, Berti D. Safety and immunogenicity of an intramuscular Helicobacter pylori vaccine in noninfected volunteers: a phase I study. Gastroenterology. 2008;135:787-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 136] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 90. | DiPetrillo MD, Tibbetts T, Kleanthous H, Killeen KP, Hohmann EL. Safety and immunogenicity of phoP/phoQ-deleted Salmonella typhi expressing Helicobacter pylori urease in adult volunteers. Vaccine. 1999;18:449-459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 120] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 91. | Bumann D, Metzger WG, Mansouri E, Palme O, Wendland M, Hurwitz R, Haas G, Aebischer T, von Specht BU, Meyer TF. Safety and immunogenicity of live recombinant Salmonella enterica serovar Typhi Ty21a expressing urease A and B from Helicobacter pylori in human volunteers. Vaccine. 2001;20:845-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 80] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 92. | Metzger WG, Mansouri E, Kronawitter M, Diescher S, Soerensen M, Hurwitz R, Bumann D, Aebischer T, Von Specht BU, Meyer TF. Impact of vector-priming on the immunogenicity of a live recombinant Salmonella enterica serovar typhi Ty21a vaccine expressing urease A and B from Helicobacter pylori in human volunteers. Vaccine. 2004;22:2273-2277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 60] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 93. | Aebischer T, Bumann D, Epple HJ, Metzger W, Schneider T, Cherepnev G, Walduck AK, Kunkel D, Moos V, Loddenkemper C. Correlation of T cell response and bacterial clearance in human volunteers challenged with Helicobacter pylori revealed by randomised controlled vaccination with Ty21a-based Salmonella vaccines. Gut. 2008;57:1065-1072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 103] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 94. | Yan J, Mao YF, Shao ZX. Frequencies of the expression of main protein antigens from Helicobacter pylori isolates and production of specific serum antibodies in infected patients. World J Gastroenterol. 2005;11:421-425. [PubMed] |
| 95. | Lepper PM, Möricke A, Vogt K, Bode G, Trautmann M. Comparison of different criteria for interpretation of immunoglobulin G immunoblotting results for diagnosis of Helicobacter pylori infection. Clin Diagn Lab Immunol. 2004;11:569-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 96. | Flach CF, Svensson N, Blomquist M, Ekman A, Raghavan S, Holmgren J. A truncated form of HpaA is a promising antigen for use in a vaccine against Helicobacter pylori. Vaccine. 2011;29:1235-1241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 49] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 97. | Wang L, Liu XF, Yun S, Yuan XP, Mao XH, Wu C, Zhang WJ, Liu KY, Guo G, Lu DS. Protection against Helicobacter pylori infection by a trivalent fusion vaccine based on a fragment of urease B-UreB414. J Microbiol. 2010;48:223-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 98. | Iankov ID, Haralambieva IH, Galanis E. Immunogenicity of attenuated measles virus engineered to express Helicobacter pylori neutrophil-activating protein. Vaccine. 2011;29:1710-1720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 99. | Every AL, Stent A, Moloney MB, Ng GZ, Skene CD, Edwards SJ, Sutton P. Evaluation of superoxide dismutase from Helicobacter pylori as a protective vaccine antigen. Vaccine. 2011;29:1514-1518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 100. | Stent A, Every AL, Ng GZ, Chionh YT, Ong LS, Edwards SJ, Sutton P. Helicobacter pylori thiolperoxidase as a protective antigen in single- and multi-component vaccines. Vaccine. 2012;30:7214-7220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 101. | Blanchard TG, Eisenberg JC, Matsumoto Y. Clearance of Helicobacter pylori infection through immunization: the site of T cell activation contributes to vaccine efficacy. Vaccine. 2004;22:888-897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 102. | Rossi G, Ruggiero P, Peppoloni S, Pancotto L, Fortuna D, Lauretti L, Volpini G, Mancianti S, Corazza M, Taccini E. Therapeutic vaccination against Helicobacter pylori in the beagle dog experimental model: safety, immunogenicity, and efficacy. Infect Immun. 2004;72:3252-3259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 74] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 103. | Jeremy AH, Du Y, Dixon MF, Robinson PA, Crabtree JE. Protection against Helicobacter pylori infection in the Mongolian gerbil after prophylactic vaccination. Microbes Infect. 2006;8:340-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 104. | Chen J, Lin M, Li N, Lin L, She F. Therapeutic vaccination with Salmonella-delivered codon-optimized outer inflammatory protein DNA vaccine enhances protection in Helicobacter pylori infected mice. Vaccine. 2012;30:5310-5315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 105. | Lundgren A, Suri-Payer E, Enarsson K, Svennerholm AM, Lundin BS. Helicobacter pylori-specific CD4+ CD25high regulatory T cells suppress memory T-cell responses to H. pylori in infected individuals. Infect Immun. 2003;71:1755-1762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 245] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 106. | Raghavan S, Suri-Payer E, Holmgren J. Antigen-specific in vitro suppression of murine Helicobacter pylori-reactive immunopathological T cells by CD4CD25 regulatory T cells. Scand J Immunol. 2004;60:82-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 66] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 107. | Czinn SJ, Blanchard T. Vaccinating against Helicobacter pylori infection. Nat Rev Gastroenterol Hepatol. 2011;8:133-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 83] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 108. | Al Mofleh IA. Spices, herbal xenobiotics and the stomach: friends or foes? World J Gastroenterol. 2010;16:2710-2719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 56] [Cited by in RCA: 52] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 109. | Cassel-Beraud AM, Le Jan J, Mouden JC, Andriantsoa M, Andriantsiferana R. Preliminary study of the prevalence of Helicobacter pylori in Tananarive, Madagascar and the antibacterial activity in vitro of 13 Malagasy medicinal plants on this germ. Arch Inst Pasteur Madagascar. 1991;59:9-23. [PubMed] |
| 110. | Higuchi K, Arakawa T, Ando K, Fujiwara Y, Uchida T, Kuroki T. Eradication of Helicobacter pylori with a Chinese herbal medicine without emergence of resistant colonies. Am J Gastroenterol. 1999;94:1419-1420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 111. | Ankli A, Heinrich M, Bork P, Wolfram L, Bauerfeind P, Brun R, Schmid C, Weiss C, Bruggisser R, Gertsch J. Yucatec Mayan medicinal plants: evaluation based on indigenous uses. J Ethnopharmacol. 2002;79:43-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 36] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 112. | Castillo-Juárez I, González V, Jaime-Aguilar H, Martínez G, Linares E, Bye R, Romero I. Anti-Helicobacter pylori activity of plants used in Mexican traditional medicine for gastrointestinal disorders. J Ethnopharmacol. 2009;122:402-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 87] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 113. | Robles-Zepeda RE, Velázquez-Contreras CA, Garibay-Escobar A, Gálvez-Ruiz JC, Ruiz-Bustos E. Antimicrobial activity of Northwestern Mexican plants against Helicobacter pylori. J Med Food. 2011;14:1280-1283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 114. | Nariman F, Eftekhar F, Habibi Z, Falsafi T. Anti-Helicobacter pylori activities of six Iranian plants. Helicobacter. 2004;9:146-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 38] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 115. | Hajimahmoodi M, Shams-Ardakani M, Saniee P, Siavoshi F, Mehrabani M, Hosseinzadeh H, Foroumadi P, Safavi M, Khanavi M, Akbarzadeh T. In vitro antibacterial activity of some Iranian medicinal plant extracts against Helicobacter pylori. Nat Prod Res. 2011;25:1059-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 116. | Wang YC, Huang TL. Screening of anti-Helicobacter pylori herbs deriving from Taiwanese folk medicinal plants. FEMS Immunol Med Microbiol. 2005;43:295-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 80] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 117. | Fabry W, Okemo P, Ansorg R. Activity of east African medicinal plants against Helicobacter pylori. Chemotherapy. 1996;42:315-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 118. | Ndip RN, Malange Tarkang AE, Mbullah SM, Luma HN, Malongue A, Ndip LM, Nyongbela K, Wirmum C, Efange SM. In vitro anti-Helicobacter pylori activity of extracts of selected medicinal plants from North West Cameroon. J Ethnopharmacol. 2007;114:452-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 56] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 119. | Stamatis G, Kyriazopoulos P, Golegou S, Basayiannis A, Skaltsas S, Skaltsa H. In vitro anti-Helicobacter pylori activity of Greek herbal medicines. J Ethnopharmacol. 2003;88:175-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 66] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 120. | Kawase M, Motohashi N. Plant-derived leading compounds for eradication of Helicobacter pylori. Curr Med Chem-Anti-Infective Agents. 2004;3:89-100. [RCA] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 121. | Castillo-Juárez I, Romero I. Plantas con actividad anti-Helicobacter pylori: una revisión. Bol Soc Bot Mex. 2007;80:35-62. |
| 122. | Mabe K, Yamada M, Oguni I, Takahashi T. In vitro and in vivo activities of tea catechins against Helicobacter pylori. Antimicrob Agents Chemother. 1999;43:1788-1791. [PubMed] |
| 123. | Koga T, Inoue H, Ishii C, Okazaki Y, Domon H, Utsui Y. Effect of plaunotol in combination with clarithromycin or amoxicillin on Helicobacter pylori in vitro and in vivo. J Antimicrob Chemother. 2002;50:133-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 124. | Tominaga K, Higuchi K, Hamasaki N, Hamaguchi M, Takashima T, Tanigawa T, Watanabe T, Fujiwara Y, Tezuka Y, Nagaoka T. In vivo action of novel alkyl methyl quinolone alkaloids against Helicobacter pylori. J Antimicrob Chemother. 2002;50:547-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 125. | Kataoka M, Hirata K, Kunikata T, Ushio S, Iwaki K, Ohashi K, Ikeda M, Kurimoto M. Antibacterial action of tryptanthrin and kaempferol, isolated from the indigo plant (Polygonum tinctorium Lour.), against Helicobacter pylori-infected Mongolian gerbils. J Gastroenterol. 2001;36:5-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 104] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 126. | Haristoy X, Angioi-Duprez K, Duprez A, Lozniewski A. Efficacy of sulforaphane in eradicating Helicobacter pylori in human gastric xenografts implanted in nude mice. Antimicrob Agents Chemother. 2003;47:3982-3984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 63] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 127. | Yanaka A. Sulforaphane enhances protection and repair of gastric mucosa against oxidative stress in vitro, and demonstrates anti-inflammatory effects on Helicobacter pylori-infected gastric mucosae in mice and human subjects. Curr Pharm Des. 2011;17:1532-1540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 42] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 128. | Cao X, Tsukamoto T, Seki T, Tanaka H, Morimura S, Cao L, Mizoshita T, Ban H, Toyoda T, Maeda H. 4-Vinyl-2,6-dimethoxyphenol (canolol) suppresses oxidative stress and gastric carcinogenesis in Helicobacter pylori-infected carcinogen-treated Mongolian gerbils. Int J Cancer. 2008;122:1445-1454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 129. | Goswami S, Bhakuni RS, Chinniah A, Pal A, Kar SK, Das PK. Anti-Helicobacter pylori potential of artemisinin and its derivatives. Antimicrob Agents Chemother. 2012;56:4594-4607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 130. | Lima ZP, dos Santos Rde C, Torres TU, Sannomiya M, Rodrigues CM, dos Santos LC, Pellizzon CH, Rocha LR, Vilegas W, Souza Brito AR. Byrsonima fagifolia: an integrative study to validate the gastroprotective, healing, antidiarrheal, antimicrobial and mutagenic action. J Ethnopharmacol. 2008;120:149-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 131. | Mazzolin LP, Nasser AL, Moraes TM, Santos RC, Nishijima CM, Santos FV, Varanda EA, Bauab TM, da Rocha LR, Di Stasi LC. Qualea parviflora Mart.: an integrative study to validate the gastroprotective, antidiarrheal, antihemorragic and mutagenic action. J Ethnopharmacol. 2010;127:508-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 132. | Moleiro FC, Andreo MA, Santos Rde C, Moraes Tde M, Rodrigues CM, Carli CB, Lopes FC, Pellizzon CH, Carlos IZ, Bauab TM. Mouririelliptica: validation of gastroprotective, healing and anti-Helicobacter pylori effects. J Ethnopharmacol. 2009;123:359-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 133. | Escobedo-Hinojosa WI, Del Carpio JD, Palacios-Espinosa JF, Romero I. Contribution to the ethnopharmacological and anti-Helicobacter pylori knowledge of Cyrtocarpa procera Kunth (Anacardiaceae). J Ethnopharmacol. 2012;143:363-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 134. | Amin M, Anwar F, Naz F, Mehmood T, Saari N. Anti-Helicobacter pylori and urease inhibition activities of some traditional medicinal plants. Molecules. 2013;18:2135-2149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 135. | Asha MK, Debraj D, Prashanth D, Edwin JR, Srikanth HS, Muruganantham N, Dethe SM, Anirban B, Jaya B, Deepak M. In vitro anti-Helicobacter pylori activity of a flavonoid rich extract of Glycyrrhiza glabra and its probable mechanisms of action. J Ethnopharmacol. 2013;145:581-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 66] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 136. | Niehues M, Euler M, Georgi G, Mank M, Stahl B, Hensel A. Peptides from Pisum sativum L. enzymatic protein digest with anti-adhesive activity against Helicobacter pylori: structure-activity and inhibitory activity against BabA, SabA, HpaA and a fibronectin-binding adhesin. Mol Nutr Food Res. 2010;54:1851-1861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 137. | Wittschier N, Faller G, Hensel A. An extract of Pelargonium sidoides (EPs 7630) inhibits in situ adhesion of Helicobacter pylori to human stomach. Phytomedicine. 2007;14:285-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 138. | O’Mahony R, Al-Khtheeri H, Weerasekera D, Fernando N, Vaira D, Holton J, Basset C. Bactericidal and anti-adhesive properties of culinary and medicinal plants against Helicobacter pylori. World J Gastroenterol. 2005;11:7499-7507. [PubMed] |
| 139. | Ruggiero P, Tombola F, Rossi G, Pancotto L, Lauretti L, Del Giudice G, Zoratti M. Polyphenols reduce gastritis induced by Helicobacter pylori infection or VacA toxin administration in mice. Antimicrob Agents Chemother. 2006;50:2550-2552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 140. | Tombola F, Campello S, De Luca L, Ruggiero P, Del Giudice G, Papini E, Zoratti M. Plant polyphenols inhibit VacA, a toxin secreted by the gastric pathogen Helicobacter pylori. FEBS Lett. 2003;543:184-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 71] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 141. | Liu JS, Cheng WC, Wang HJ, Chen YC, Wang WC. Structure-based inhibitor discovery of Helicobacter pylori dehydroquinate synthase. Biochem Biophys Res Commun. 2008;373:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 142. | Steinmetz KA, Potter JD. Vegetables, fruit, and cancer prevention: a review. J Am Diet Assoc. 1996;96:1027-1039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1437] [Cited by in RCA: 1200] [Article Influence: 41.4] [Reference Citation Analysis (0)] |
| 143. | Cellini L, Di Campli E, Masulli M, Di Bartolomeo S, Allocati N. Inhibition of Helicobacter pylori by garlic extract (Allium sativum). FEMS Immunol Med Microbiol. 1996;13:273-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 120] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 144. | Sivam GP, Lampe JW, Ulness B, Swanzy SR, Potter JD. Helicobacter pylori--in vitro susceptibility to garlic (Allium sativum) extract. Nutr Cancer. 1997;27:118-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 70] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 145. | Chung JG, Chen GW, Wu LT, Chang HL, Lin JG, Yeh CC, Wang TF. Effects of garlic compounds diallyl sulfide and diallyl disulfide on arylamine N-acetyltransferase activity in strains of Helicobacter pylori from peptic ulcer patients. Am J Chin Med. 1998;26:353-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 35] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 146. | Cañizares P, Gracia I, Gómez LA, Martín de Argila C, Boixeda D, García A, de Rafael L. Allyl-thiosulfinates, the bacteriostatic compounds of garlic against Helicobacter pylori. Biotechnol Prog. 2004;20:397-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 147. | Mahady GB, Matsuura H, Pendland SL. Allixin, a phytoalexin from garlic, inhibits the growth of Helicobacter pylori in vitro. Am J Gastroenterol. 2001;96:3454-3455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 148. | Tabak M, Armon R, Potasman I, Neeman I. In vitro inhibition of Helicobacter pylori by extracts of thyme. J Appl Bacteriol. 1996;80:667-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 44] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 149. | Ohta R, Yamada N, Kaneko H, Ishikawa K, Fukuda H, Fujino T, Suzuki A. In vitro inhibition of the growth of Helicobacter pylori by oil-macerated garlic constituents. Antimicrob Agents Chemother. 1999;43:1811-1812. [PubMed] |
| 150. | Graham DY, Anderson SY, Lang T. Garlic or jalapeño peppers for treatment of Helicobacter pylori infection. Am J Gastroenterol. 1999;94:1200-1202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 45] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 151. | McNulty CA, Wilson MP, Havinga W, Johnston B, O’Gara EA, Maslin DJ. A pilot study to determine the effectiveness of garlic oil capsules in the treatment of dyspeptic patients with Helicobacter pylori. Helicobacter. 2001;6:249-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 152. | Iimuro M, Shibata H, Kawamori T, Matsumoto T, Arakawa T, Sugimura T, Wakabayashi K. Suppressive effects of garlic extract on Helicobacter pylori-induced gastritis in Mongolian gerbils. Cancer Lett. 2002;187:61-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 52] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 153. | You WC, Brown LM, Zhang L, Li JY, Jin ML, Chang YS, Ma JL, Pan KF, Liu WD, Hu Y. Randomized double-blind factorial trial of three treatments to reduce the prevalence of precancerous gastric lesions. J Natl Cancer Inst. 2006;98:974-983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 322] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 154. | Ma JL, Zhang L, Brown LM, Li JY, Shen L, Pan KF, Liu WD, Hu Y, Han ZX, Crystal-Mansour S. Fifteen-year effects of Helicobacter pylori, garlic, and vitamin treatments on gastric cancer incidence and mortality. J Natl Cancer Inst. 2012;104:488-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 353] [Article Influence: 27.2] [Reference Citation Analysis (1)] |
| 155. | Yassibaş E, Arslan P, Yalçin S. Evaluation of dietary and life-style habits of patients with gastric cancer: a case-control study in Turkey. Asian Pac J Cancer Prev. 2012;13:2291-2297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 156. | Galan MV, Kishan AA, Silverman AL. Oral broccoli sprouts for the treatment of Helicobacter pylori infection: a preliminary report. Dig Dis Sci. 2004;49:1088-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 64] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 157. | Opekun AR, Yeh CW, Opekun JL, Graham DY. In vivo tests of natural therapy, Tibetan yogurt or fresh broccoli, for Helicobacter pylori infection. Methods Find Exp Clin Pharmacol. 2005;27:327-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 158. | Sato K, Kawakami N, Ohtsu T, Tsutsumi A, Miyazaki S, Masumoto T, Horie S, Haratani T, Kobayashi F, Araki S. Broccoli consumption and chronic atrophic gastritis among Japanese males: an epidemiological investigation. Acta Med Okayama. 2004;58:127-133. [PubMed] |
| 159. | Fahey JW, Haristoy X, Dolan PM, Kensler TW, Scholtus I, Stephenson KK, Talalay P, Lozniewski A. Sulforaphane inhibits extracellular, intracellular, and antibiotic-resistant strains of Helicobacter pylori and prevents benzo[a]pyrene-induced stomach tumors. Proc Natl Acad Sci USA. 2002;99:7610-7615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 595] [Cited by in RCA: 521] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 160. | Stoicov C, Saffari R, Houghton J. Green tea inhibits Helicobacter growth in vivo and in vitro. Int J Antimicrob Agents. 2009;33:473-478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 161. | Matsubara S, Shibata H, Ishikawa F, Yokokura T, Takahashi M, Sugimura T, Wakabayashi K. Suppression of Helicobacter pylori-induced gastritis by green tea extract in Mongolian gerbils. Biochem Biophys Res Commun. 2003;310:715-719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 108] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 162. | Mahady GB, Pendland SL, Chadwick LR. Resveratrol and red wine extracts inhibit the growth of CagA+ strains of Helicobacter pylori in vitro. Am J Gastroenterol. 2003;98:1440-1441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 64] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 163. | Paulo L, Oleastro M, Gallardo E, Queiroz JA, Domingues F. Anti-Helicobacter pylori and urease inhibitory activities of resveratrol and red wine. Food Res Int. 2011;44:964-969. [RCA] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 52] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 164. | Ruggiero P, Rossi G, Tombola F, Pancotto L, Lauretti L, Del Giudice G, Zoratti M. Red wine and green tea reduce H pylori- or VacA-induced gastritis in a mouse model. World J Gastroenterol. 2007;13:349-354. [PubMed] |
| 165. | Fukai T, Marumo A, Kaitou K, Kanda T, Terada S, Nomura T. Anti-Helicobacter pylori flavonoids from licorice extract. Life Sci. 2002;71:1449-1463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 246] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 166. | Krausse R, Bielenberg J, Blaschek W, Ullmann U. In vitro anti-Helicobacter pylori activity of Extractum liquiritiae, glycyrrhizin and its metabolites. J Antimicrob Chemother. 2004;54:243-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 71] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 167. | Kim JM, Zheng HM, Lee BY, Lee WK, Lee DH. Anti-Helicobacter pylori Properties of Gut Gard. Prev Nutr Food Sci. 2013;18:104-110. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 168. | Ali AT, Chowdhury MN, al Humayyd MS. Inhibitory effect of natural honey on Helicobacter pylori. Trop Gastroenterol. 1991;12:139-143. [PubMed] |
| 169. | Osato MS, Reddy SG, Graham DY. Osmotic effect of honey on growth and viability of Helicobacter pylori. Dig Dis Sci. 1999;44:462-464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 170. | Nzeako BC, Al-Namaani F. The antibacterial activity of honey on Helicobacter pylori. Sultan Qaboos Univ Med J. 2006;6:71-76. [PubMed] |
| 171. | Ndip RN, Malange Takang AE, Echakachi CM, Malongue A, Akoachere JF, Ndip LM, Luma HN. In-vitro antimicrobial activity of selected honeys on clinical isolates of Helicobacter pylori. Afr Health Sci. 2007;7:228-232. [PubMed] |
| 172. | al Somal N, Coley KE, Molan PC, Hancock BM. Susceptibility of Helicobacter pylori to the antibacterial activity of manuka honey. J R Soc Med. 1994;87:9-12. [PubMed] |
| 173. | Manyi-Loh CE, Clarke AM, Munzhelele T, Green E, Mkwetshana NF, Ndip RN. Selected South African honeys and their extracts possess in vitro anti-Helicobacter pylori activity. Arch Med Res. 2010;41:324-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 174. | Manyi-Loh CE, Clarke AM, Ndip RN. Detection of phytoconstituents in column fractions of n-hexane extract of Goldcrest honey exhibiting anti-Helicobacter pylori activity. Arch Med Res. 2012;43:197-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 175. | McGovern DP, Abbas SZ, Vivian G, Dalton HR. Manuka honey against Helicobacter pylori. J R Soc Med. 1999;92:439. [PubMed] |
| 176. | Boyanova L, Gergova G, Nikolov R, Derejian S, Lazarova E, Katsarov N, Mitov I, Krastev Z. Activity of Bulgarian propolis against 94 Helicobacter pylori strains in vitro by agar-well diffusion, agar dilution and disc diffusion methods. J Med Microbiol. 2005;54:481-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 96] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 177. | Cui K, Lu W, Zhu L, Shen X, Huang J. Caffeic acid phenethyl ester (CAPE), an active component of propolis, inhibits Helicobacter pylori peptide deformylase activity. Biochem Biophys Res Commun. 2013;435:289-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 178. | Banskota AH, Tezuka Y, Adnyana IK, Ishii E, Midorikawa K, Matsushige K, Kadota S. Hepatoprotective and anti-Helicobacter pylori activities of constituents from Brazilian propolis. Phytomedicine. 2001;8:16-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 123] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 179. | Coelho LG, Bastos EM, Resende CC, Paula e Silva CM, Sanches BS, de Castro FJ, Moretzsohn LD, Vieira WL, Trindade OR. Brazilian green propolis on Helicobacter pylori infection. a pilot clinical study. Helicobacter. 2007;12:572-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 180. | Health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria. Report of a joint FAO/WHO expert consultation on evaluation of health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria 2001. FAO/WHO, Cordoba, Argentina. . |
| 181. | Chenoll E, Casinos B, Bataller E, Astals P, Echevarría J, Iglesias JR, Balbarie P, Ramón D, Genovés S. Novel probiotic Bifidobacterium bifidum CECT 7366 strain active against the pathogenic bacterium Helicobacter pylori. Appl Environ Microbiol. 2011;77:1335-1343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 94] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 182. | Lorca GL, Wadström T, Valdez GF, Ljungh A. Lactobacillus acidophilus autolysins inhibit Helicobacter pylori in vitro. Curr Microbiol. 2001;42:39-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 85] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 183. | Sgouras D, Maragkoudakis P, Petraki K, Martinez-Gonzalez B, Eriotou E, Michopoulos S, Kalantzopoulos G, Tsakalidou E, Mentis A. In vitro and in vivo inhibition of Helicobacter pylori by Lactobacillus casei strain Shirota. Appl Environ Microbiol. 2004;70:518-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 208] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 184. | Pinchuk IV, Bressollier P, Verneuil B, Fenet B, Sorokulova IB, Mégraud F, Urdaci MC. In vitro anti-Helicobacter pylori activity of the probiotic strain Bacillus subtilis 3 is due to secretion of antibiotics. Antimicrob Agents Chemother. 2001;45:3156-3161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 150] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 185. | Nam H, Ha M, Bae O, Lee Y. Effect of Weissella confusa strain PL9001 on the adherence and growth of Helicobacter pylori. Appl Environ Microbiol. 2002;68:4642-4645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 75] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 186. | Haller D, Bode C, Hammes WP, Pfeifer AM, Schiffrin EJ, Blum S. Non-pathogenic bacteria elicit a differential cytokine response by intestinal epithelial cell/leucocyte co-cultures. Gut. 2000;47:79-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 315] [Article Influence: 12.6] [Reference Citation Analysis (1)] |
| 187. | Johnson-Henry KC, Mitchell DJ, Avitzur Y, Galindo-Mata E, Jones NL, Sherman PM. Probiotics reduce bacterial colonization and gastric inflammation in H. pylori-infected mice. Dig Dis Sci. 2004;49:1095-1102. [PubMed] |
| 188. | Patel A, Shah N, Prajapati JB. Clinical appliance of probiotics in the treatment of Helicobacter pylori infection-A brief review. J Microbiol Immunol Infect. 2013;Jun 8; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 189. | Dekker KA, Inagaki T, Gootz TD, Huang LH, Kojima Y, Kohlbrenner WE, Matsunaga Y, McGuirk PR, Nomura E, Sakakibara T. New quinolone compounds from Pseudonocardia sp. with selective and potent anti-Helicobacter pylori activity: taxonomy of producing strain, fermentation, isolation, structural elucidation and biological activities. J Antibiot (Tokyo). 1998;51:145-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 49] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 190. | Dekker KA, Inagaki T, Gootz TD, Kaneda K, Nomura E, Sakakibara T, Sakemi S, Sugie Y, Yamauchi Y, Yoshikawa N. CJ-12,954 and its congeners, new anti-Helicobacter pylori compounds produced by Phanerochaete velutina: fermentation, isolation, structural elucidation and biological activities. J Antibiot (Tokyo). 1997;50:833-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 43] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 191. | Shang X, Tan Q, Liu R, Yu K, Li P, Zhao GP. In vitro anti-Helicobacter pylori effects of medicinal mushroom extracts, with special emphasis on the Lion’s Mane mushroom, Hericium erinaceus (higher Basidiomycetes). Int J Med Mushrooms. 2013;15:165-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 192. | Geethangili M, Fang SH, Lai CH, Rao YK, Lien HM, Tzeng YM. Inhibitory effect of Antrodia camphorata constituents on the Helicobacter pylori-associated gastric inflammation. Food Chemistry. 2010;119:149-153. [RCA] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 193. | Lin CJ, Rao YK, Hung CL, Feng CL, Lane HY, Tzeng DT, Hsu PN, Lai CH, Tzeng YM. Inhibition of Helicobacter pylori CagA-Induced Pathogenesis by Methylantcinate B from Antrodia camphorata. Evid Based Complement Alternat Med. 2013;2013:682418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 194. | Lachter J, Yampolsky Y, Gafni-Schieber R, Wasser SP. Yellow brain culinary-medicinal mushroom, Tremella mesenterica Ritz.: Fr. (higher Basidiomycetes), is subjectively but not objectively effective for eradication of Helicobacter pylori: a prospective controlled trial. Int J Med Mushrooms. 2012;14:55-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 195. | Kanamaru T, Nakano Y, Toyoda Y, Miyagawa KI, Tada M, Kaisho T, Nakao M. In vitro and in vivo antibacterial activities of TAK-083, an agent for treatment of Helicobacter pylori infection. Antimicrob Agents Chemother. 2001;45:2455-2459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 57] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 196. | Vecchione JJ, Sello JK. A novel tryptophanyl-tRNA synthetase gene confers high-level resistance to indolmycin. Antimicrob Agents Chemother. 2009;53:3972-3980. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 197. | Iwahori A, Hirota Y, Sampe R, Miyano S, Takahashi N, Sasatsu M, Kondo I, Numao N. On the antibacterial activity of normal and reversed magainin 2 analogs against Helicobacter pylori. Biol Pharm Bull. 1997;20:805-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 198. | Chen L, Li Y, Li J, Xu X, Lai R, Zou Q. An antimicrobial peptide with antimicrobial activity against Helicobacter pylori. Peptides. 2007;28:1527-1531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 199. | Numao N, Hirota Y, Iwahori A, Sasatsu M, Kondo I, Takao K, Kobayashi S. Antibacterial activity of (+/-) 6-benzyl-1-(3-carboxypropyl) indane; a possible way to identify leading novel anti-H. pylori agents. Biol Pharm Bull. 1997;20:1204-1207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 200. | Bajaj-Elliott M, Fedeli P, Smith GV, Domizio P, Maher L, Ali RS, Quinn AG, Farthing MJ. Modulation of host antimicrobial peptide (beta-defensins 1 and 2) expression during gastritis. Gut. 2002;51:356-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 82] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 201. | Hase K, Murakami M, Iimura M, Cole SP, Horibe Y, Ohtake T, Obonyo M, Gallo RL, Eckmann L, Kagnoff MF. Expression of LL-37 by human gastric epithelial cells as a potential host defense mechanism against Helicobacter pylori. Gastroenterology. 2003;125:1613-1625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 165] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 202. | Rigano MM, Romanelli A, Fulgione A, Nocerino N, D’Agostino N, Avitabile C, Frusciante L, Barone A, Capuano F, Capparelli R. A novel synthetic peptide from a tomato defensin exhibits antibacterial activities against Helicobacter pylori. J Pept Sci. 2012;Nov 4; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 203. | Lee H, Kobayashi M, Wang P, Nakayama J, Seeberger PH, Fukuda M. Expression cloning of cholesterol alpha-glucosyltransferase, a unique enzyme that can be inhibited by natural antibiotic gastric mucin O-glycans, from Helicobacter pylori. Biochem Biophys Res Commun. 2006;349:1235-1241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 204. | Lindén SK, Wickström C, Lindell G, Gilshenan K, Carlstedt I. Four modes of adhesion are used during Helicobacter pylori binding to human mucins in the oral and gastric niches. Helicobacter. 2008;13:81-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 80] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 205. | Shibata H, Iimuro M, Uchiya N, Kawamori T, Nagaoka M, Ueyama S, Hashimoto S, Yokokura T, Sugimura T, Wakabayashi K. Preventive effects of Cladosiphon fucoidan against Helicobacter pylori infection in Mongolian gerbils. Helicobacter. 2003;8:59-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 85] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 206. | Loke MF, Lui SY, Ng BL, Gong M, Ho B. Antiadhesive property of microalgal polysaccharide extract on the binding of Helicobacter pylori to gastric mucin. FEMS Immunol Med Microbiol. 2007;50:231-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 207. | Lee JH, Shim JS, Lee JS, Kim JK, Yang IS, Chung MS, Kim KH. Inhibition of pathogenic bacterial adhesion by acidic polysaccharide from green tea (Camellia sinensis). J Agric Food Chem. 2006;54:8717-8723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 77] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 208. | Wittschier N, Faller G, Hensel A. Aqueous extracts and polysaccharides from liquorice roots (Glycyrrhiza glabra L.) inhibit adhesion of Helicobacter pylori to human gastric mucosa. J Ethnopharmacol. 2009;125:218-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 119] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 209. | Lengsfeld C, Deters A, Faller G, Hensel A. High molecular weight polysaccharides from black currant seeds inhibit adhesion of Helicobacter pylori to human gastric mucosa. Planta Med. 2004;70:620-626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 50] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 210. | Lembo AJ, Ganz RA, Sheth S, Cave D, Kelly C, Levin P, Kazlas PT, Baldwin PC, Lindmark WR, McGrath JR. Treatment of Helicobacter pylori infection with intra-gastric violet light phototherapy: a pilot clinical trial. Lasers Surg Med. 2009;41:337-344. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |