Published online Nov 21, 2014. doi: 10.3748/wjg.v20.i43.16095
Revised: May 21, 2014
Accepted: July 15, 2014
Published online: November 21, 2014
Processing time: 246 Days and 17.2 Hours
Fermented foods have been consumed for centuries across many geographical locales and have traditionally been considered healthy foods, partly because of the live microbes contained in them. The concept of “probiotics” further requires that the microbes be defined and their health effects be demonstrated through human intervention studies or other suitable investigations before marketing with corresponding health messages. Here, we review recommendations for fermented foods and probiotics in several countries outside the EU, focusing on food-based dietary guidelines. We emphasize recommendations on yoghurt and probiotics made by expert bodies. We found that dietary guidelines commonly advocate the consumption of yoghurt or similar products, but specific comments on probiotics are rare. Further, we reviewed guidelines from clinical associations. In general, they acknowledge the beneficial effects of probiotics, but often suggest the need for further research. This is true despite good quality evidence supporting the role of probiotics for certain health effects, such as prevention of eczema in infants, management of side effects from antibiotics and alleviation of functional bowel symptoms. Additional research to support future dietary recommendations should focus on determining effect size, identifying responders and non-responders, clarifying strain-specificity of effects and confirming mechanisms.
Core tip: Probiotics are by definition beneficial to health. Benefits can be nutritional, clinical, medical and treatment-related. Nutritional benefits of probiotics and fermented foods have been recognized in varying degrees in European Union dietary recommendations. This review summarizes the nutritional and therapeutic recommendations for probiotics and fermented foods outside European Union countries.
- Citation: Ebner S, Smug LN, Kneifel W, Salminen SJ, Sanders ME. Probiotics in dietary guidelines and clinical recommendations outside the European Union. World J Gastroenterol 2014; 20(43): 16095-16100
- URL: https://www.wjgnet.com/1007-9327/full/v20/i43/16095.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i43.16095
Food-based dietary guidelines may include beneficial microbes or dairy products as recommended foods for specific dietary purposes. In Europe, for instance, five countries include yoghurt and/or beneficial microbes in their dietary guidelines[1,2] Beneficial microbes have been used in food for centuries. In particular, the use of fermented milk has been described in several historical documents. The term “yoghurt” was defined in 1925 and specifies a typical milk fermented with Streptococcus thermophilus and Lactobacillus delbrueckii subsp. bulgaricus[1]. Fermented foods have a long history of safe use. The European Union further reviews microorganisms and compiles a list known as the “Qualified Presumption of Safety” (QPS) list, which delineates biological agents deemed safe for use in foods, such as bacteria, yeasts, filamentous fungi and viruses for food and feed additives[2]. The International Dairy Federation lists 62 genera and 264 species used in mainly in dairy products, but also in other food matrices[3].
Probiotics are defined as “live microorganisms which when administered in adequate amounts confer a health benefit on the host”[4]. Elie Metchnikoff introduced the concept of probiotic microorganisms, from the Greek “pro bios” (for life), in 1907. The term “probiotics” was proposed in several sources: by Ferdinand Vergin in 1954 for “active substances that are essential for a healthy development of life”[1], by Werner Kollath in 1953[5] and was used by Lilly and Stillwell in 1965[6]. Several Lactobacillus and Bifidobacterium species have been investigated intensely, and certain strains of Saccharomyces, Enterococcus, Lactococcus, Propionibacterium and Streptococcus species have been considered as microorganisms with probiotic properties[7].
A large collection of bacteria, yeasts and filamentous fungi with a history of safe use is being used for the fermentation of foods. In general, the primary aim is to produce healthy and easily digestible foods, which have an enhanced shelf life and also contain live microorganisms. Specific microbes have been reported to exert general beneficial effects on human health, such as competing with pathogens, enhancing the gut barrier against pathogens, modifying digestion of nutrients and by modulating the gut-associated lymphoid tissue[8]. Microbes fulfilling the requirements set up by the WHO/FAO (2002) can further be considered to be probiotics-as beneficial as microbes in fermented foods generally, but with additional documented health effects. Intervention trials are the best for establishing causality; however, well-controlled observational studies can also provide evidence of probiotic function[2,3].
The biological effects of probiotics are strain specific, thus it is not possible to extrapolate failure or success of one strain to another, even for taxonomically closely related strains, unless a common efficacy mechanism functions in both strains[9]. Nevertheless, numerous meta-analyses provide evidence that multiple probiotics may function similarly with respect to certain benefits to health.
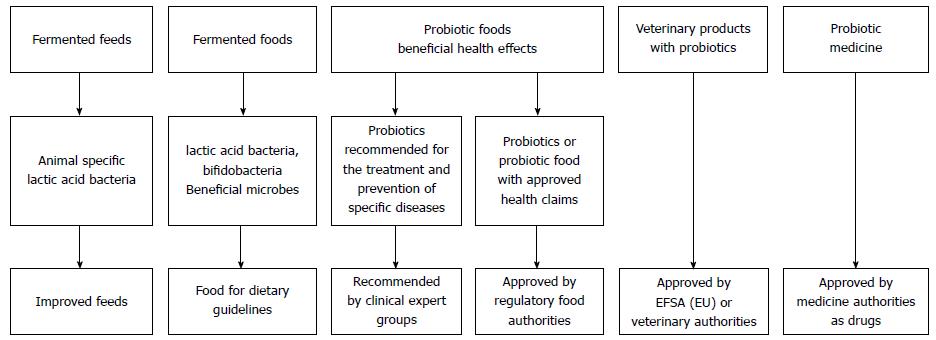
An overview of the possible regulatory pathways for fermented foods and probiotics is given in Figure 1. Areas of intensive research with specific probiotics comprise acute gastroenteritis or diarrhea in various forms, such as acute infectious diarrhea[10,11]; antibiotic associated diarrhea and its side effects[12,13] or traveller’s diarrhea[14]; irritable bowel syndrome symptoms[15,16]; inflammatory bowel disease[14,17]; Helicobacter pylori infections[18]; and oral health[19], including reducing the risk of caries and gingivitis.
Nutritional recommendations vary from country to country, because the nutrient intake and the priority in the selection of principal nutrients may depend on available foods and food preferences. In the EU member states, the main food groups within national dietary guidelines do not differ considerably, but the differences in the types of foods within the groups and the recommended amounts deviate substantially. To date, there are no harmonized guidelines on an EU level because of a lack of representative consumption data[20]. Similar challenges are present in other parts of the world.
In an earlier study, the nutrition guidelines given by governments or government-related expert organizations of 13 EU member states (Austria, Denmark, Estonia, Finland, France, Germany, Ireland, Italy, Poland, Spain, Sweden, the Netherlands and the United Kingdom) plus Switzerland, were reviewed, with the particular aim of comparing the dietary guidelines concerning yoghurt and probiotic products[21]. All countries included yoghurt as part of a healthy diet. Interestingly, none of countries mentioned yoghurt as an alternative for people with lactose intolerance, even though there is an approved “function claim” on live cultures in yoghurt or fermented milk to “improve lactose digestion of the product in individuals who have difficulty digesting lactose” in the European Union[22]. Yoghurt is easily digestible, it normally contains less lactose and supports further lactose hydrolysis via bacterial lactase activity. This information is important for countries with populations commonly unable to digest lactose. Moreover, only five of the countries considered in the survey explicitly recommended yoghurt because of live bacteria or even probiotic properties, even though accumulating evidence supports the benefits of ingestion of live cultures[23]. In this paper, we extend our research approach to ten countries outside the European Union (Table 1). Different approaches were used to search the websites of the responsible organizations for dietary guidelines. There, the search was conducted by targeting “nutrition guidelines” or “dietary guidelines” and “yoghurt” or probiotics. Any statements recommending yoghurt, probiotics or probiotic yoghurt were assessed.
| Country | Organization | Document | Yoghurt recommendation | Probiotic recommendation |
| New Zealand | Ministry of Health | Part 1: New Zealand Food and Nutrition Guidelines[24] | Milk and milk products, including yogurt. Provide energy, protein, fats (mostly saturated), vitamins (riboflavin, B12, A) and minerals (calcium, iodine, phosphorus, zinc). They are important for children and young people to ensure optimal bone health. Reduced or low fat particularly, milk and milk products are the best choices because these foods include less saturated fat, and often more protein and calcium than high-fat alternatives | Not mentioned |
| Canada | Agriculture and Agri-Food Canada; Health Canada | Probiotics- understanding them will lead to greater use[25]; Canada's Food Guide[26] | Yoghurt (as lower fat milk alternative; milk per se as source of Vitamin D) | Approved by Health Canada as Functional Food with Added Probiotics[27]: Activia® with Bifidobacterium (animalis) lactis DN-173 010; DanActive® with Lactobacillus casei DN-114 001; Yoptimal® and iÖGO ProbioTM with Bifidobacterium lactis BB-12 and Lactobacillus acidophilus LA-5) |
| United States | United States Department of Agriculture | Dietary Guidelines for Americans, 2010[28] | All milks, including yogurts, frozen yogurts and cheeses. Most choices should be fat-free or low fat. Milk and milk products contribute many nutrients, such as calcium, vitamin D (for products fortified with vita-min D), and potassium, to the diet. Moderate evidence shows that intake of milk and milk products is linked to improved bone health, especially in children and adolescents. Moderate evidence also indicates that intake of milk and milk products is associated with a reduced risk of cardiovascular disease and type 2 diabetes and with lower blood pressure in adults. Choosing fat-free or low-fat milk and milk products provides the same nutrients with less solid fat, and thus, fewer calories. In addition, selecting more milk group intake as fat-free or low-fat fluid milk or yogurt rather than as cheese can increase intake of potassium, vitamin A, and vitamin D and decrease intake of sodium, cholesterol, and saturated fatty acids | Not mentioned |
| India | National Institute of Nutrition | Dietary Guidelines for Indians[29] | Recommended as curd | Not mentioned |
| Australia | The Department of Health and Ageing; The Australian Nutrition Foundation; | The Australian Guide to Healthy Eating[30] Australian Dietary Guidelines[31] | Milks, including buttermilk, yoghurt (all yoghurts including reduced fat or full cream-without added sugar), soy yoghurt (calcium fortified), and cheese Milk, cheese and yoghurt provide calcium in a readily absorbable and convenient form. They have various health benefits and are a good source of many nutrients, including calcium, protein, iodine, vitamin A, vitamin D, riboflavin, vitamin B12 and zinc. Avoiding dairy foods and not making suitable alternative choices, such as the ones recommended in this food group, can affect your long-term health | Not mentioned |
| Brazil | The Ministry of Health | The Food Guide[32] | Milk is an important source of vitamin B2 and main source of calcium in the feed; dairy products such as yoghurt and cottage cheese have the same profile | Not mentioned |
| China | The Ministry of Health | China's Dietary Guidelines (2007)[33], Chinese Dietary Guidelines[34] | Yogurt, as source of calcium | Not mentioned |
| Israel | Ministry of Health | The Food Pyramid[35] | Yogurt as source of calcium in the group of protein-rich foods | Not mentioned |
| Japan | Health and Agriculture ministries | The Food Guide[36] | Yoghurt as a source of calcium | Not mentioned |
| Mexico | Department of Nutrition and Health Promotion | The Plate of Good Eating[37] | Recommended | Not mentioned |
| Thailand | Ministry of Public Health[38] | Mentioned in general along with milk | A list of approved microorganisms for food use provided, but no recommendation made. | |
| Turkey | Ministry of Health | Basic Food Groups[39] | Yogurt, cheeses contain important nutrients: proteins, calcium, phosphorus, vitamin B2 (riboflavin) and vitamin B12; eating yogurt and drinking ayran (buttermilk) help in treatment of diarrhea | Not mentioned |
Several clinical organizations have assessed probiotics and probiotic foods for their evidence-based health effects. Such evaluations have resulted in clinical recommendations made by medical organizations that suggested specific well-defined probiotics for specific clinical conditions, such as treatment and prevention of acute gastroenteritis, necrotizing enterocolitis, or antibiotic associated diarrhea or, more broadly, to supplement infant formula to make it resemble the composition and microbial content of human milk[40-41]. Recently, other studies suggested that the benefits of probiotics can positively impact healthcare costs. This nutritional economics approach applies both to general community health care and to specialist care in hospitals[42,43] (Table 2).
| Scientific society | Area of application | Recommendations |
| National Institute for Health and Care | Diarrhoea and vomiting in children under 5: NICE guideline[44] | Probiotic specificity for each target should be considered available studies report benefits in reduced duration of diarrhea or stool frequency BUT: published studies have methodological limitations - in specific evaluated probiotics and treatment regimes Many of the studies were conducted in developing countries - response to probiotic therapy may differ Good quality randomized controlled trials should be conducted in the United Kingdom |
| Excellence (NICE) European Society of Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) and European Society for Paediatric Infectious Diseases (ESPID) | Evidence-based Guidelines for the Management of Acute Gastroenteritis in Children in Europe[39] | Selected probiotics may reduce the duration and intensity of symptoms Oral rehydration solution with Lactobacillus GG may be beneficial in children with acute gastroenteritis; however, because of insufficient evidence, routine use is not recommended Lactobacillus GG and Saccharomyces boulardii showed benefits in the management of diarrhea but evidence of a lack of risk of antibiotic resistance transfer is required |
| European Society of Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) | Supplementation of infant formula with probiotics and/or prebiotics[45] | Probiotic-supplemented formula: no safety concerns with regard to growth and adverse effects for healthy infants Administration of probiotic-supplemented infant formula during early life does not result in any consistent clinical effects Administration of a few probiotics supplemented to infant or follow-on formulae and given beyond early infancy may have clinical benefits; however there is too much uncertainty to draw reliable conclusions No extrapolation of safety and clinical effects of one probiotic microbe to another ones Generally there is a lack of data on the long-term effects of the administration of formula supplemented with probiotics; would be of particular importance Routine use of probiotic-supplemented formula in infants is not recommended by the Committee |
Probiotics and fermented foods, specifically fermented milks, may benefit human health in many ways. Fermented foods are usually easily digestible. They provide live beneficial microorganisms to the human diet and have a long history of safe use. There are many products with the label “probiotic”. However, such products often do not meet minimum criteria, such as defined content, appropriate viable count at end of shelf life and suitable evidence of health benefit. Therefore, a recent panel of International Scientific Association of Probiotics and Prebiotics recommended that the term “probiotic” should be used only for products that deliver live microorganisms with a suitable viable count of well-defined strains with a reasonable expectation of delivering benefits for the wellbeing of the host[46]. Evidence from well-conducted observational studies and numerous randomized, controlled trials supports their potential contribution to human health. Their role in dietary guidelines of many nations has been illustrated; however, a deeper understanding of probiotic health benefits needs to be analyzed from the already existing human studies to identify opportunities to develop evidence-based dietary and clinical recommendations for probiotics.
P- Reviewer: de Medina FS, El-Nezami H, Howarth GS, Marotta F, Rishi P S- Editor: Qi Y L- Editor: Stewart G E- Editor: Wang CH
| 1. | Caramia G, Silvi S. Probiotics: From the Ancient Wisdom to the Actual Therapeutical and Nutraceutical Perspective. Springer Netherlands: Probiotic Bacteria and Enteric Infections 2011; 3. |
| 2. | European Food Safety Authority. Scientific Opinion on the maintenance of the list of QPS biological agents intentionally added to food and feed (2012 update). EFSA J. 2012;10:3020. [RCA] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 3. | Bourdichon F, Casaregola S, Farrokh C, Frisvad JC, Gerds ML, Hammes WP, Harnett J, Huys G, Laulund S, Ouwehand A. Food fermentations: microorganisms with technological beneficial use. Int J Food Microbiol. 2012;154:87-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 398] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 4. | Food and Agriculture Organization/World Health Organization. Health and Nutritional Properties of Probiotics in Food including Powder Milk with Live Lactic Acid Bacteria[Internet]. 2001[cited. 2014;Feb 20]; Available from: ftp://ftp.fao.org/docrep/fao/009/a0512e/a0512e00.pdf. |
| 5. | Kollath W. The increase of the diseases of civilization and their prevention. Munch Med Wochenschr. 1953;95:1260-1262. [PubMed] |
| 6. | Lilly DM, Stillwell RH. Probiotics: Growth-promoting factors produced by microorganisms. Science. 1965;147:747-748. [PubMed] |
| 7. | Fung WY, Lye HS, Lim TJ, Kuan CY, Liong MT. Roles of Probiotic on Gut Health. Probiotics. Berlin Heidelberg: Springer 2011; 139-165. |
| 8. | Brownawell AM, Caers W, Gibson GR, Kendall CW, Lewis KD, Ringel Y, Slavin JL. Prebiotics and the health benefits of fiber: current regulatory status, future research, and goals. J Nutr. 2012;142:962-974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 124] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 9. | Rijkers GT, de Vos WM, Brummer RJ, Morelli L, Corthier G, Marteau P. Health benefits and health claims of probiotics: bridging science and marketing. Br J Nutr. 2011;106:1291-1296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 123] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 10. | de Vrese M, Marteau PR. Probiotics and prebiotics: effects on diarrhea. J Nutr. 2007;137:803S-811S. [PubMed] |
| 11. | Hajela N, Nair GB, Ganguly NK. Are probiotics a feasible intervention for prevention of diarrhoea in the developing world? Gut Pathog. 2010;2:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 12. | McFarland LV. Evidence-based review of probiotics for antibiotic-associated diarrhea and Clostridium difficile infections. Anaerobe. 2009;15:274-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 112] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 13. | Santosa S, Farnworth E, Jones PJ. Probiotics and their potential health claims. Nutr Rev. 2006;64:265-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | Culligan EP, Hill C, Sleator RD. Future Prospects of Probiotics as Therapeutics Against Enteric Disorders. Springer Netherlands: Probiotic Bacteria and Enteric Infections 2011; 445-464. |
| 15. | Ringel Y, Quigley EM, Lin HC. Using Probiotics in Gastrointestinal Disorders. Am J Gastroenterol Suppl. 2012;1:34-40. [RCA] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 16. | Whelan K, Quigley EM. Probiotics in the management of irritable bowel syndrome and inflammatory bowel disease. Curr Opin Gastroenterol. 2013;29:184-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 128] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 17. | Naidoo K, Gordon M, Fagbemi AO, Thomas AG, Akobeng AK. Probiotics for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2011;CD007443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 64] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 18. | Ahmad K, Fatemeh F, Mehri N, Maryam S. Probiotics for the treatment of pediatric helicobacter pylori infection: a randomized double blind clinical trial. Iran J Pediatr. 2013;23:79-84. [PubMed] |
| 19. | Banas JA, Popp ET. Recovery of Viable Bacteria from Probiotic Products that Target Oral Health. Probiotics Antimicrob Proteins. 2013;5:227-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 20. | European Food Safety Authority. Scientific Opinion on establishing Food-Based Dietary Guidelines. EFSA J. 2010;8:1460. [RCA] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 21. | Smug LN, Salminen S, Sanders ME, Ebner S. Yoghurt and probiotic bacteria in dietary guidelines of the member states of the European Union. Benef Microbes. 2014;5:61-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 22. | European Commission. EU Register of nutrition and health claims made on foods [Internet]. Available from: http://ec.europa.eu/nuhclaims/. |
| 23. | Sanders ME, Lenoir-Wijnkoop I, Salminen S, Merenstein DJ, Gibson GR, Petschow BW, Nieuwdorp M, Tancredi DJ, Cifelli CJ, Jacques P. Probiotics and prebiotics: prospects for public health and nutritional recommendations. Ann N Y Acad Sci. 2014;1309:19-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 25. | Agriculture and Agri-Food Canada. Probiotics- understanding them will lead to greater use[Internet]. 2013[cited. 2013;Dec 27] Available from: http://www5.agr.gc.ca/eng/science-and-innovation/science-publications-and-resources/agri-science-e-zine/agri-science-e-zine-agri-food-food-innovation/?id=1368651096741#a3. |
| 26. | Health Canada. Canada’s Food Guide. 2011[cited. 2013;Dec 27] Available from: http://www.hc-sc.gc.ca/fn-an/alt_formats/hpfb-dgpsa/pdf/food-guide-aliment/view_eatwell_vue_bienmang-eng.pdf. |
| 28. | United States Department of Agriculture. Dietary Guidelines for Americans, 2010[Internet]. 2010[cited. 2013;Dec 27] Available from: http://www.cnpp.usda.gov/DGAs2010-PolicyDocument.htm. |
| 29. | National Institute of Nutrition. Dietary Guidelines for Indians[Internet]. 2010[cited. 2013;Dec 27] Available from: http://ninindia.org/DietaryguidelinesforIndians-Finaldraft.pdf. |
| 30. | The Department of Health and Ageing. The Australian Guide to Healthy Eating[Internet]. 2013[cited. 2013;Dec 27] Available from: http://www.health.gov.au/internet/publications/publishing.nsf/Content/nhsc-guidelines~aus-guide-healthy-eating. |
| 31. | The Australian Nutrition Foundation. Australian Dietary Guidelines[Internet]. 2013[cited. 2014;Mar 5] Available from: http://www.nutritionaustralia.org/national/resource/australian-dietary-guidelines-2013. |
| 32. | The Ministry of Health. The Food Guide[Internet]. 2005[cited. 2013;Dec 27] Available from: http://dtr2001.saude.gov.br/editora/produtos/livros/pdf/05_1109_M.pdf. |
| 33. | The Ministry of Health. China’s Dietary Guidelines (2007) [Internet]. 2007[cited. 2013;Dec 27] Available from: http://www.gov.cn/xwfb/2008-01/15/content_858517.htm. |
| 34. | LinXia Food and Drug Administration. Chinese Dietary Guidelines [Internet]. 2012[cited. 2013;Dec 27] Available from: http://www.lxzfda.gov.cn/news_view.asp?news_id=126869. |
| 35. | Ministry of Health. The Food Pyramid[Internet]. 2013[cited. 2013;Dec 27] Available from: http://www.health.gov.il/Subjects/FoodAndNutrition/Nutrition/Adequate_nutrition/Pages/food_pyramid.aspx. |
| 36. | Health and Agriculture ministries. The Food Guide[Internet]. 2007[cited. 2013;Dec 27] Available from: http://www.maff.go.jp/j/balance_guide/. |
| 37. | Department of Nutrition and Health Promotion. The Plate of Good Eating[Internet]. 2003[cited. 2013;Dec 27] Available from: http://www.fns.org.mx/index.php?IdContenido=46. |
| 38. | Ministry of Public Health. Ministry Webpage [Internet]. 2014[cited. 2014;Mar 5] Available from: http://eng.moph.go.th/. |
| 39. | Ministry of Health. Basic Food Groups[Internet]. 2013[cited. 2013;Dec 27] Available from: http://www.beslenme.gov.tr/index.php?lang=tr&page=188. |
| 40. | World Gastroenterology Organisation. WGO Practice Guideline - Probiotics and Prebiotics 2011[Internet]. 2011[cited. 2014;Feb 26] Available from: http://www.worldgastroenterology.org/probiotics-prebiotics.html. |
| 41. | Alfaleh K, Anabrees J, Bassler D, Al-Kharfi T. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Cochrane Database Syst Rev. 2011;CD005496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 109] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 42. | Vandenplas Y, De Hert S. Cost/benefit of synbiotics in acute infectious gastroenteritis: spend to save. Benef Microbes. 2012;3:189-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 43. | Kamdeu Fansi AA, Guertin JR, LeLorier J. Savings from the use of a probiotic formula in the prophylaxis of antibiotic-associated diarrhea. J Med Econ. 2012;15:53-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 44. | National Institute for Health and Care Excellence. Diarrhoea and vomiting in children - Diarrhoea and vomiting caused by gastroenteritis: diagnosis, assessment and management in children younger than 5 years[Internet]. NICE Clin. Guidel. 842009[cited 2013 Feb 27]. 2013; Available from: http://guidance.nice.org.uk/cg84. |
| 45. | Braegger C, Chmielewska A, Decsi T, Kolacek S, Mihatsch W, Moreno L, Pieścik M, Puntis J, Shamir R, Szajewska H. Supplementation of infant formula with probiotics and/or prebiotics: a systematic review and comment by the ESPGHAN committee on nutrition. J Pediatr Gastroenterol Nutr. 2011;52:238-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 327] [Cited by in RCA: 254] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 46. | Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, Morelli L, Canani RB, Flint HJ, Salminen S. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11:506-514. [PubMed] |