Published online Sep 28, 2014. doi: 10.3748/wjg.v20.i36.12934
Revised: March 30, 2014
Accepted: May 28, 2014
Published online: September 28, 2014
Processing time: 349 Days and 23.1 Hours
The excess consumption of alcohol is associated with alcoholic liver diseases (ALD). ALD is a major healthcare problem, personal and social burden, and significant reason for economic loss worldwide. The ALD spectrum includes alcoholic fatty liver, alcoholic hepatitis, cirrhosis, and the development of hepatocellular carcinoma. The diagnosis of ALD is based on a combination of clinical features, including a history of significant alcohol intake, evidence of liver disease, and laboratory findings. Abstinence is the most important treatment for ALD and the treatment plan varies according to the stage of the disease. Various treatments including abstinence, nutritional therapy, pharmacological therapy, psychotherapy, and surgery are currently available. For severe alcoholic hepatitis, corticosteroid or pentoxifylline are recommended based on the guidelines. In addition, new therapeutic targets are being under investigation.
Core tip: Abstinence is the most important treatment for alcoholic liver diseases and the treatment plan varies according to the stage of the disease. Various treatments including abstinence, nutritional therapy, pharmacological therapy, psychotherapy, and surgery are currently available. For severe alcoholic hepatitis, corticosteroid or pentoxifylline are recommended based on the guidelines.
- Citation: Suk KT, Kim MY, Baik SK. Alcoholic liver disease: Treatment. World J Gastroenterol 2014; 20(36): 12934-12944
- URL: https://www.wjgnet.com/1007-9327/full/v20/i36/12934.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i36.12934
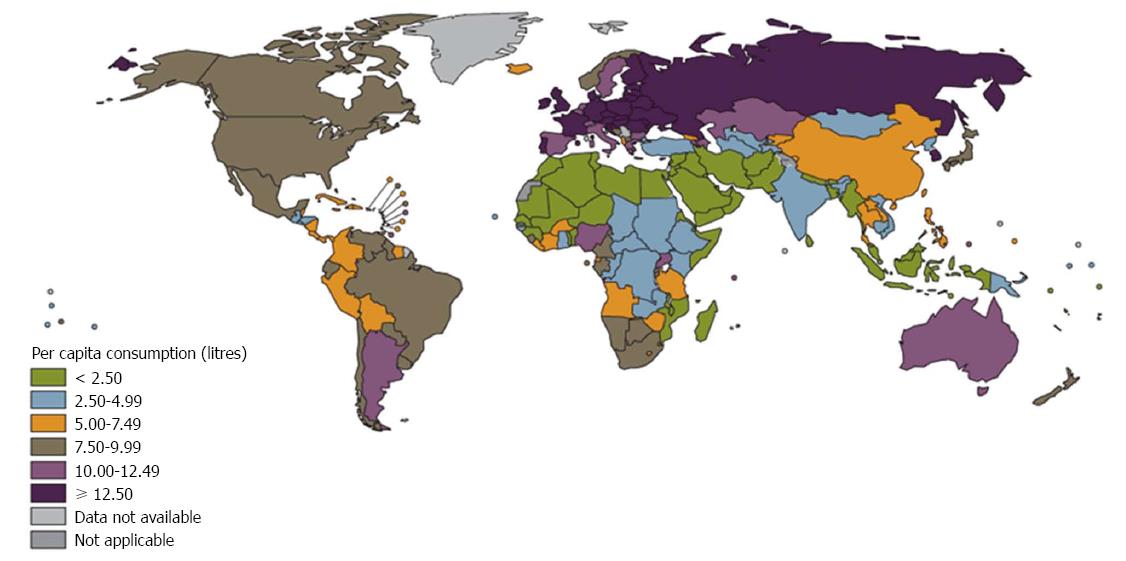
Alcohol is consumed worldwide and has long been identified as a major risk factor for all liver diseases (Figure 1)[1]. Alcoholic liver disease (ALD) is the third most common cause of chronic liver disease, and it has a global burden of mortality[2]. According to a study of the United States, the incidence of chronic liver disease was 72.3 per 100000 among which 24% had chronic liver disease due to alcohol[3]. Globally, alcohol-attributable liver cirrhosis was responsible for 493300 deaths in 2010 (156900 female deaths and 336400 male deaths)[2]. The number of alcohol-related deaths remains high at 2.5 million deaths annually, constituting 4% of all deaths worldwide[4] and death from ALD constitute approximately 25% of deaths due to alcohol consumption[5].
Many factors affect the development of alcoholic liver injury including the dose, duration, and type of alcohol consumption; drinking patterns; sex; ethnicity; and associated risk factors including obesity, viral disease, and genetic factors[6]. The histological spectrum of ALD varies from simple fatty liver to cirrhosis, including the development of hepatocellular carcinoma[7].
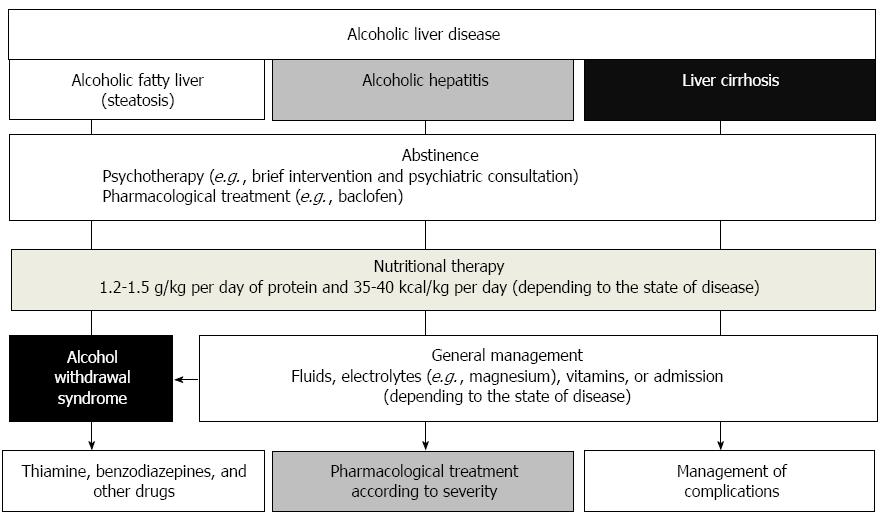
ALD is preventable and reversible by timely treatment. However, ALD is often asymptomatic in the early stages and can only be identified by laboratory findings. Screening and treatment for alcohol-use disorders is the first approach for treating ALD[8]. ALD treatment varies depending on the stage of the disease[6,8] (Figure 2). Abstinence is the most important therapeutic intervention for patients with ALD. Various treatments such as abstinence, nutritional therapy, pharmacological therapy, psychotherapy, and surgery are currently available for the spectrums of ALD[6]. In particular, in patients with severe alcoholic hepatitis, steroid or pentoxifylline can be used according to recommendations of the guidelines. In patients with cirrhosis, portal hypertension and complications such as bleeding, encephalopathy, or ascites, should be treated according to the treatment guidelines[9,10]. However, this medical treatment for ALD has not significantly changed over the past 40 years. The slow therapeutic advance in the field of ALD has resulted from the lack of experimental models of ALD and from difficulties in performing clinical trials in patients with ALD. This review focuses on the current management of ALD and suggests new therapeutic target in the management of ALD.
Immediate abstinence is the most important treatment option for patients with ALD[11,12]. Continued drinking is associated with the eventual development of cirrhosis in approximately 20% of individuals[13]. Abstinence improves the survival and prognosis of patients with ALD and prevents progression to liver cirrhosis through histologic improvement and reduction in portal pressure[14,15]. Alcoholic steatosis can be reversed after abstinence for several weeks[16] and multivariate analysis showed that persistent alcohol intake was an independent poor prognostic marker[12].
With respect to pharmacological treatment, some medications such as baclofen, acamprosate, and naltrexone have been used to encourage abstinence in patients with alcohol use disorder[17,18]. However, few data are available regarding the use of pharmacological agents in the treatment of ALD, because agents such as naltrexone or acamprosate undergo extensive liver metabolism; thus drug-related liver damage is possible[19]. More studies that provide evidence of the efficacy and safety of these agents in the treatment of ALD are needed.
Baclofen is a GABAB receptor agonist that is used as an anti-spasticity medication to treat neurological disorder[20]. Some reports have recently suggested that baclofen may be valuable to the treatment of alcohol dependence[21-23]. Acamprosate reduces withdrawal and cravings for alcohol. In a meta-analysis, acamprosate had a significant beneficial effect on enhancing abstinence in recently detoxified, alcohol-dependent patients[24]. Naltrexone decreases excessive drinking and the recurrence rate and increasing the abstinence duration in patients with alcohol dependence[25,26].
With respect to psychotherapy for abstinence, brief intervention is an effective method that includes motivational interviewing and counseling over a limited time span[27]. A meta-analysis found evidence for the positive impact of brief interventions on alcohol consumption and alcohol-related morbidity and mortality[28,29]. It is necessary for ALD patients to consult with psychologist about psychological and social support for abstinence.
Malnutrition is a major complication of ALD and this has been studied, especially in patients with alcoholic hepatitis. Malnutrition worsens clinical outcome in ALD, and nutritional support improves nutritional status and may improve clinical outcome.
The mechanisms for malnutrition are multifactorial. Poor intake, anorexia, vomiting, mal-digestion, iatrogenic causes, metabolic disturbance, or mal-absorption might cause malnutrition in patients with ALD[30]. Mendenhall et al[31] suggested that most patients with ALD are malnourished. In this study, patients had almost 50% of energy intake from alcohol. Thus, while calorie intake was frequently not inadequate, there was often deficient intake of protein and critical nutrients.
The severity of liver disease generally correlated with the severity of malnutrition[32]. Patients who voluntarily consumed > 3000 kcal/d had virtually no mortality whereas those consuming < 1000 kcal/d had > 80% 6-mo mortality[33]. Moreover, the degree of malnutrition correlated with the development of serious complications such as encephalopathy, ascites, and hepatorenal syndrome[34].
Early intervention with nutrition therapy may improve treatment response, alleviate symptoms, and improve the quality and quantity of life[35,36]. In one trial, nutritional supplementation through a feeding tube significantly improved liver function in inpatients with ALD as assessed by serum bilirubin levels and antipyrine clearance, compared to inpatients who ate a hospital diet[37]. In patients with severe hepatitis, the parenteral nutrition group showed an overall mortality rate that was comparable to that of the steroid treatment group[38].
A symptom-based and supportive approach is necessary to achieve appropriate nutritional therapy in patients with ALD[39]. Nutrition goals include providing adequate calories, protein, and nutrients to support hepatocyte regeneration within the existing metabolic alterations of liver disease[40]. The use of branched chain amino acids (34 g/d) has been shown to decrease the number of hospitalizations due to complications of liver cirrhosis[41,42]. Depending to the status of ALD, 1.2-1.5 g/kg per day of protein and 35-40 kcal/kg per day should be supplied in addition to medical treatment[43]. Vitamin A, thiamine, vitamin B12, folic acid, pyridoxine, vitamin D, magnesium, selenium, and zinc may be administered to patients with ALD along with nutritional therapy[44,45].
Enteral nutrition is desired over parenteral nutrition because of cost, risk of sepsis of the parenteral nutrition line, preservation of the integrity of the gut mucosa, and prevention of bacterial translocation and multiple organ failure[46]. Moreover, total parenteral nutrition can, in some instances, cause liver disease as one of its complications.
Alcohol withdrawal syndrome is characterized by the symptoms and signs that occur 6-24 h following the cessation of alcohol consumption among those who habitually drink excessively. The indications for admission are severe alcohol withdrawal syndrome, high levels of recent drinking, a history of withdrawal seizures or delirium tremens, the co-occurrence of a serious medical or psychiatric illness, or failure to recover in outpatient treatment[47].
Long-acting benzodiazepines [chlordiazepoxide (per oral, 25-100 mg every 4-6 h) or diazepam (per oral/intravenous/intramuscular, 5-10 mg every 6-8 h)] are recommended for the prevention of seizures, and an intermediate-acting benzodiazepine [lorazepam (per oral/intravenous/intramuscular, 1-4 mg every 4-8 h)] is recommended for patients with severe alcohol withdrawal syndrome, advanced age, recent head trauma, liver failure, respiratory failure, other serious medical comorbidities, or obesity[47-52]. Carbamazepine (per oral, 200 mg every 6-8 h) can be used as an alternative to benzodiazepine and is effective in seizure prevention[47]. Haloperidol (per oral/intramuscular, 0.5-5 mg every 8-12 h) can be used carefully as an adjunctive therapy only in cases of agitation or psychotic symptoms such as hallucinations that are not controlled by benzodiazepine[47-52]. Thiamine should be administered to all patients with alcohol withdrawal syndrome (100-300 mg/d) and maintained for 2-3 mo following the resolution of their withdrawal symptoms[53]. Thiamine should be given before administering fluids containing glucose.
Corticosteroids (prednisolone 40 mg/d for 28 d followed by tapering over 2-4 wk) are the mainstay of treatment for severe alcoholic hepatitis (modified discriminant function ≥ 32 or presence of encephalopathy). The modified discriminant function can be calculated by the equation of 4.6 × [prothrombin timepatient - prothrombin timecontrol (s)] + serum total bilirubin (mg/dL). Typically, in patients with severe alcoholic hepatitis, mortality rate within 28 d was 30%-50%.
The mechanism is thought to decrease inflammation by reducing the transcription of pro-inflammatory cytokines such as tumor necrosis factor (TNF)-α and interleukin (IL)-8. The efficacy of corticosteroids in alcoholic hepatitis has been evaluated in 13 randomized controlled studies and 4 meta-analyses since 1971[54,55]. However, there have been conflicting results[56,57]. Although there are mixed results from various studies, corticosteroids are overall currently considered to have survival benefit in patients with severe alcoholic hepatitis. Unfortunately, alcoholic hepatitis is unresponsive to corticosteroid treatment in approximately 40% of patients (Table 1).
| Target | Method | Characteristic | |
| Abstinence | Stop drinking | Baclofen, acamprosate, naltrexone, and psychotherapy | The role of pharmacologic agents in maintaining abstinence is unclear. |
| Nutritional support | Correct malnutrition | 1.2-1.5 g/kg per day of protein and 35-40 kcal/kg per day | Vitamin A, thiamine, vitamin B12, folic acid, pyridoxine, vitamin D, magnesium, selenium, and zinc may be administered. |
| Corticosteroid | Decrease inflammation | Prednisolone 40 mg/d for 28 d followed by tapering over 2-4 wk | Increase in the serum bilirubin and Lille score > 0.45 after 1 wk of therapy are associated with worse outcome |
| Pentoxifylline | Ablate cytokines | 400 mg 3 times/d for 28 d | Protective effect against hepatorenal syndrome |
| Infliximab | Ablate TNF-α | Not confirmed | Further studies are needed |
| Etanercept | Ablate TNF-α | ||
| N-acetylcysteine | Anti-oxidant | ||
| Metadoxine | Anti-oxidant | ||
| Propylthiouracil | Reduce ischemic damage | ||
| Colchicine | Anti-fibrosis | ||
| PL | Anti-fibrosis | ||
| AIIR blocker | Anti-fibrosis | ||
| Silymarin | Anti-oxidant and anti-fibrosis |
To predict the prognosis following corticosteroid treatment, early changes in bilirubin levels and the Lille model were introduced[58,59]. Early changes in bilirubin levels are defined as a bilirubin level that is lower at 7 d than that on the first day of treatment[59]. Lille’s score (http://www.lillemodel.com), which is a more complicated but comprehensive evaluation method of the treatment response, is measured at 7 d after the start of treatment[58].
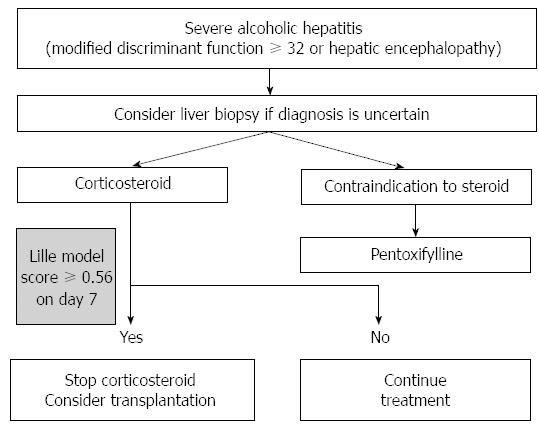
A score of > 0.45 suggests that a patient is not responding to corticosteroid therapy and a lower survival rate at 6 mo of 25%, which is lower than that of other reported more lower scores[58]. Recent studies have shown that classification as complete responders (Lille score ≤ 0.16), partial responders (Lille score 0.16-0.56), and null responders (Lille score ≥ 0.56) is associated with the 28-d survival rate[60]. From the finding that non-responders to corticosteroid are associated with the development of infection and a lower survival rate, discontinuing corticosteroid is generally recommended in non-responders (Figure 3)[61].
Pentoxifylline (400 mg 3 times/d for 28 d) is considered as an alternative to corticosteroid treatment in patients with severe alcoholic hepatitis[62]. The exact mechanism is not understood. However, pentoxifylline inhibits phosphodiesterase and increases the intracellular adenosine 3’,5’-cyclic monophosphate level inducing inhibition of cytokine expression and macrophage inflammatory protein-1a[63]. Pentoxifylline failed to decrease the serum TNF-α level in the treated group in another study[62]. The protective effect against hepatorenal syndrome appears to be the main mechanism of action against alcoholic hepatitis according to a randomized controlled study[62]. However, in a meta-analysis and systematic review, pentoxifylline failed to improve the survival rate[64-66]. In a study comparing the efficacy of pentoxifylline and prednisolone in severe alcoholic hepatitis, pentoxifylline showed superior efficacy in terms of mortality[67]. However, this study enrolled only 68 patients and the survival rates were 35.29% vs 14.71% (pentoxifylline vs prednisolone) which were relatively lower than that in previous studies[67]. Studies about combination treatment with pentoxifylline and corticosteroid have indicated no additional survival benefit[66,68]. Additionally, as the salvage treatment in non-responders to corticosteroid, pentoxifylline showed no additional benefit[69]. Overall, pentoxifylline is an efficient alternative to corticosteroid in severe alcoholic hepatitis, especially if there are contraindications to corticosteroid use (Figure 3). However, more confirmative studies are needed in the future.
Anti-TNF-α agents have been developed to block TNF-α, a major cytokine involved in alcoholic hepatitis. Two anti-TNF-α agents have been investigated as therapy for alcoholic hepatitis, infliximab and etanercept. In a pilot study, combination therapy with infliximab and steroids was reported to be effective[70]. However, the trial was stopped early by the independent data and safety monitoring board because of a significant excess of severe infections and an insignificant increase in the deaths in the infliximab cohort (Table 1)[71].
Etanercept showed an increase in the short-term survival of patients in a pilot study[72]. However, a subsequent randomized, placebo-controlled trial revealed a worse 6-mo survival rate in the group treated with etanercept than in the placebo group[73]. Therefore, anti-TNF-α agents are currently not recommended for treating alcoholic hepatitis.
N-acetylcysteine: The combination of N-acetylcysteine, an antioxidant, with prednisolone has been studied. This study enrolled 174 patients who were randomly assigned to receive either prednisolone 40 mg/d for 28 d along with intravenous N-acetylcysteine for 5 d or prednisolone 40 mg/d for 28 d alone. Combination therapy improved the 1-mo survival in patients with alcoholic hepatitis. However, the 6-mo survival, which was the primary endpoint of the study, did not improve[74,75]. Further research is needed to evaluate the efficacy of N-acetylcysteine.
Metadoxine: Metadoxine, a combination of 2 antioxidants (pyridoxine and pyrrolidone), is potentially a useful drug in the treatment of ALD. In a large randomized controlled trial, there was significant improvement in the liver function tests in both groups. The percentage of patients with persistent hepatic steatosis as assessed by ultrasound, was also significantly lower in the metadoxine group (28% vs 70%)[76]. These positive effects were more noticeable in patients who abstained from alcohol compared to those who continued to drink. However, the clinical implications are still unclear; therefore, metadoxine is not recommended for the treatment of ALD. Further studies are required to better explain the significance of these observations on other clinical endpoints.
Propylthiouracil: Propylthiouracil has also been evaluated for the treatment of acute hepatitis. In some studies, PTU improved the mortality rate by suppressing hypermetabolic activation[77,78]. However, in a Cochrane meta-analysis of 6 studies with 710 ALD patients that compared propylthiouracil versus placebo, there was no clear improvement in the liver histology or liver-related or overall mortality rate[79,80].
Colchicine: Colchicine affects hepatic fibrogenesis, including the inhibition of collagen production, the enhancement of collagenase activity, and the interference with collagen transcellular trafficking. In addition, colchicine also has favorable effects on cytokine production associated with fibroblast proliferation. In clinical studies involving patients with alcoholic liver cirrhosis, colchicine showed anti-inflammatory and anti-fibrotic effects[81,82], and had a positive effect on survival[83]. However, controlled trials later had conflicting results[84].
Polyunsaturated lecithin: Polyunsaturated lecithin is extracted from soybeans and is a constituent of cell membranes[85]. Polyunsaturated lecithin appears to improve histology and reduce the activation of hepatic stellate cells in baboons with alcoholic liver injury[86]. However, polyenylphosphatidylcholine did not show a clear association with the progression of liver fibrosis in a follow-up randomized controlled study[87].
Angiotensin II receptor blocker: It has been reported in one randomized controlled trial that combination therapy with candesartan and ursodeoxycholic acid shows greater histologic improvement compared to monotherapy with ursodeoxycholic acid[88].
Silymarin: Silymarin, a milk thistle extract with anti-oxidative and anti-fibrotic properties, has been evaluated in many studies as a potential treatment agent for ALD[89,90]. Although one study reported that silymarin contributes to improved survival, this result has not been confirmed for ALD patients in meta-analyses such as the Cochrane review[89,91].
ALD is one of the most common indications for liver transplantation in North America and Europe[92,93]. The survival rate of liver transplantation in ALD is comparable with other causes[94-97]. Indications for transplantation in ALD are identical to those in other end-stage liver diseases. Patients with severe alcoholic hepatitis who do not respond to corticosteroids or pentoxifylline have a mortality rate of 50% to 75% at 6 mo[98].
In a review of 22 studies on ALD, relapse ranged from 3% to 49% with graft dysfunction and death ranging from 0% to 27% and 0% to 6.5%, respectively[99]. Therefore, it is essential to accurately identify patients before transplantation who are likely to relapse to harmful drinking after receiving the transplant. Six months of abstinence prior to liver transplantation is considered a necessary step[97,100-104]. Recidivism following liver transplantation is a common occurrence, which occurs at a rate of approximately 10%-52%. Alcohol consumption following liver transplantation causes histologic damage in the liver, including liver fibrosis[96,105,106].
Most European and North American liver transplantation centers do not consider patients with severe alcoholic hepatitis as candidates for liver transplantation because they do not fulfill the criterion of abstinence for 6 mo prior to liver transplantation[6,100]. However, a prospective, multi-center study showed an increase in the survival rate with liver transplantation in patients with severe alcoholic hepatitis who are not responsive to medical treatment[107]. Therefore, liver transplantation may be considered in the specific group of patients whose severe alcoholic hepatitis has failed medical treatment. However, improvement in long-term survival must be verified in future studies.
Similar to those who have received liver transplantation for other causes, patients who have received liver transplantation due to ALD show a high incidence of de novo cancer in other areas of the body[92,94,108-111]. These de novo cancers are associated with an increase in mortality following transplantation. The immunosuppressive drugs that are used post-transplantation may be related to the onset of new cancers. Compared to other causes, liver transplantation due to ALD is associated with a strikingly high rate of cardiovascular complications[92].
Recent studies have suggested that stem cell therapy may reduce liver inflammation, and subsequently improve fibrosis, which could be a promising strategy for patients with liver cirrhosis[112]. Mesenchymal stem cell (MSC) directly inhibit the activation of hepatic stellate cells via MSC-derived cytokines and may also induce hepatic stellate cells apoptosis via the Fas/FasL pathway[113]. MSCs have been reported to contribute to the direct production of new hepatocytes as well as to stimulate proliferation of endogenous hepatocytes[114,115]. In a pilot study of 12 patients with alcoholic cirrhosis[116], bone marrow-derived MSC used as therapy in alcoholic cirrhosis induces a histological and quantitative improvement for hepatic fibrosis. Further evidence is needed and a 2-phase multicenter trial in humans is now in process. In addition, a significant improvement in Child-Pugh score and albumin was reported in 9 cirrhotic patients given a portal vein infusion of autologous bone-marrow derived stem cells[117]. Better liver function after MSC therapy was also documented in patients with cirrhosis[118]. However, in others studies, infusion of autologous, expanded, mobilized, adult bone-marrow derived stem cells did benefit patients with ALD[119,120]. Therefore, further evidences about the pathogenic and therapeutic roles of bone-derived stem cells are needed.
In the stem cell therapy, unsolved issues in protocols require further investigation, such as the optimal type of transfused MSCs, the optimal therapeutic timing, the most effective number of stem cells, the best route of administration. The long-term clinical benefits and safety of stem cell-based therapies should be confirmed in a large-sized randomized controlled trial.
In the future, with a safe, controllable, and feasible recommendation, the clinical application of stem cells for the treatment of patients with ALD will be further warranted.
New treatments or strategies are required to improve the survival of patients with ALD. Recent translational work using human liver tissue has been informative in identifying some potential therapeutic targets for ALD, such as CXC chemokines, IL-22/signal transducer and activator of transcription 3, TNF receptor superfamily, complement, osteopontin, gut microbiota and lipopolysaccharide (LPS), endocannabinoids, and inflammasomes[121]. Hepatic expression of CXC chemokines, CXC family of chemokines includes IL-8 and Gro-α; these usually attract neutrophils, is increased and correlates with survival time and the degree of portal hypertension[122]. IL-22 might be used to treat patients with ALD because of its antioxidant, antiapoptotic, antisteatotic, proliferative, and antimicrobial effects[123]. Complement activation, apoptosis, and osteopontin up-regulation are prominent feature of ALD[124,125].
Gut-derived microbial LPS, a component of the outer wall of gram-negative bacteria, has been known as a central role in the pathogenesis of ALD[126,127]. Alcohol has been known to disrupt the gut barrier function, consequently, promotes the translocation of microbial LPS from the lumen of the intestines to the portal vein, where it travels to the liver. In the Kupffer cell, LPS binds to CD14, which combines with toll-like receptor 4, ultimately activating multiple pro-inflammatory cytokine genes[128]. Therefore, probiotics, prebiotics, antibiotics, or transplantation of gut-microbiota have been proposed as possible treatments for ALD by ablating the increase in LPS or repopulating the gut.
Ethanol consumption causes epigenetic changes that contribute to alcohol-induced liver damage. Exposure to ethanol or its metabolite acetate up-regulates histone acetylation in macrophages, contributing to the up-regulation of several pro-inflammatory cytokines that could promote alcoholic hepatitis[129]. Therefore, epigenetic modifications can be new therapeutic target.
ALD is a broad term that encompasses a spectrum of phenotypes ranging from simple fatty liver to alcoholic hepatitis, cirrhosis, and hepatocellular carcinoma. The mechanisms underlying the development of these different disease stages are not fully understood. The standard treatment for ALD, which includes abstinence, nutritional therapy, pharmacological therapy, psychotherapy, and surgery, has not changed in the last 40 years. Novel pathophysiology-oriented therapies are needed for patients with ALD in the future.
P- Reviewer: Buzas GM, Cai KY, Fernandez-Rodriguez CM, Tiribelli C S- Editor: Ma YJ L- Editor: A E- Editor: Zhang DN
| 1. | World Health Organization. Global status report on alcohol and health 2011. Available from: http://www.who.int/substance_abuse/publications/global_alcohol_report/en/. |
| 2. | Rehm J, Samokhvalov AV, Shield KD. Global burden of alcoholic liver diseases. J Hepatol. 2013;59:160-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 475] [Cited by in RCA: 538] [Article Influence: 44.8] [Reference Citation Analysis (0)] |
| 3. | Kim WR, Brown RS, Terrault NA, El-Serag H. Burden of liver disease in the United States: summary of a workshop. Hepatology. 2002;36:227-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 425] [Cited by in RCA: 411] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 4. | Room R, Babor T, Rehm J. Alcohol and public health. Lancet. 2005;365:519-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 591] [Cited by in RCA: 712] [Article Influence: 35.6] [Reference Citation Analysis (0)] |
| 5. | Shaw JJ, Shah SA. Rising incidence and demographics of hepatocellular carcinoma in the USA: what does it mean? Expert Rev Gastroenterol Hepatol. 2011;5:365-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 6. | O’Shea RS, Dasarathy S, McCullough AJ. Alcoholic liver disease. Hepatology. 2010;51:307-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 837] [Cited by in RCA: 852] [Article Influence: 56.8] [Reference Citation Analysis (2)] |
| 7. | MacSween RN, Burt AD. Histologic spectrum of alcoholic liver disease. Semin Liver Dis. 1986;6:221-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 163] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 8. | European Association for the Study of Liver. EASL clinical practical guidelines: management of alcoholic liver disease. J Hepatol. 2012;57:399-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 454] [Cited by in RCA: 455] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 9. | Runyon BA. Introduction to the revised American Association for the Study of Liver Diseases Practice Guideline management of adult patients with ascites due to cirrhosis 2012. Hepatology. 2013;57:1651-1653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 514] [Cited by in RCA: 519] [Article Influence: 43.3] [Reference Citation Analysis (1)] |
| 10. | Suk KT, Baik SK, Yoon JH, Cheong JY, Paik YH, Lee CH, Kim YS, Lee JW, Kim DJ, Cho SW. Revision and update on clinical practice guideline for liver cirrhosis. Korean J Hepatol. 2012;18:1-21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 106] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 11. | Borowsky SA, Strome S, Lott E. Continued heavy drinking and survival in alcoholic cirrhotics. Gastroenterology. 1981;80:1405-1409. [PubMed] |
| 12. | Pessione F, Ramond MJ, Peters L, Pham BN, Batel P, Rueff B, Valla DC. Five-year survival predictive factors in patients with excessive alcohol intake and cirrhosis. Effect of alcoholic hepatitis, smoking and abstinence. Liver Int. 2003;23:45-53. [PubMed] |
| 13. | Morgan MY. The prognosis and outcome of alcoholic liver disease. Alcohol Alcohol Suppl. 1994;2:335-343. [PubMed] |
| 14. | Veldt BJ, Lainé F, Guillygomarc’h A, Lauvin L, Boudjema K, Messner M, Brissot P, Deugnier Y, Moirand R. Indication of liver transplantation in severe alcoholic liver cirrhosis: quantitative evaluation and optimal timing. J Hepatol. 2002;36:93-98. [PubMed] |
| 15. | Luca A, García-Pagán JC, Bosch J, Feu F, Caballería J, Groszmann RJ, Rodés J. Effects of ethanol consumption on hepatic hemodynamics in patients with alcoholic cirrhosis. Gastroenterology. 1997;112:1284-1289. [PubMed] |
| 16. | Teli MR, Day CP, Burt AD, Bennett MK, James OF. Determinants of progression to cirrhosis or fibrosis in pure alcoholic fatty liver. Lancet. 1995;346:987-990. [PubMed] |
| 17. | Mason BJ, Lehert P. Acamprosate for alcohol dependence: a sex-specific meta-analysis based on individual patient data. Alcohol Clin Exp Res. 2012;36:497-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 60] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 18. | Roozen HG, de Waart R, van der Windt DA, van den Brink W, de Jong CA, Kerkhof AJ. A systematic review of the effectiveness of naltrexone in the maintenance treatment of opioid and alcohol dependence. Eur Neuropsychopharmacol. 2006;16:311-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 57] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 19. | Liu J, Wang L. Baclofen for alcohol withdrawal. Cochrane Database Syst Rev. 2011;CD008502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 20. | Davidoff RA. Antispasticity drugs: mechanisms of action. Ann Neurol. 1985;17:107-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 218] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 21. | Agabio R, Marras P, Addolorato G, Carpiniello B, Gessa GL. Baclofen suppresses alcohol intake and craving for alcohol in a schizophrenic alcohol-dependent patient: a case report. J Clin Psychopharmacol. 2007;27:319-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Addolorato G, Leggio L, Ferrulli A, Cardone S, Vonghia L, Mirijello A, Abenavoli L, D’Angelo C, Caputo F, Zambon A. Effectiveness and safety of baclofen for maintenance of alcohol abstinence in alcohol-dependent patients with liver cirrhosis: randomised, double-blind controlled study. Lancet. 2007;370:1915-1922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 483] [Cited by in RCA: 472] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 23. | Stallings W, Schrader S. Baclofen as prophylaxis and treatment for alcohol withdrawal: a retrospective chart review. J Okla State Med Assoc. 2007;100:354-360. [PubMed] |
| 24. | Mann K, Lehert P, Morgan MY. The efficacy of acamprosate in the maintenance of abstinence in alcohol-dependent individuals: results of a meta-analysis. Alcohol Clin Exp Res. 2004;28:51-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 206] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 25. | Rösner S, Hackl-Herrwerth A, Leucht S, Vecchi S, Srisurapanont M, Soyka M. Opioid antagonists for alcohol dependence. Cochrane Database Syst Rev. 2010;CD001867. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 210] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 26. | Soyka M, Rösner S. Opioid antagonists for pharmacological treatment of alcohol dependence - a critical review. Curr Drug Abuse Rev. 2008;1:280-291. [PubMed] |
| 27. | Bien TH, Miller WR, Tonigan JS. Brief interventions for alcohol problems: a review. Addiction. 1993;88:315-335. [PubMed] |
| 28. | Vasilaki EI, Hosier SG, Cox WM. The efficacy of motivational interviewing as a brief intervention for excessive drinking: a meta-analytic review. Alcohol Alcohol. 2006;41:328-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 365] [Cited by in RCA: 336] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 29. | Kaner EF, Dickinson HO, Beyer F, Pienaar E, Schlesinger C, Campbell F, Saunders JB, Burnand B, Heather N. The effectiveness of brief alcohol interventions in primary care settings: a systematic review. Drug Alcohol Rev. 2009;28:301-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 300] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 30. | Singal AK, Charlton MR. Nutrition in alcoholic liver disease. Clin Liver Dis. 2012;16:805-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 31. | Mendenhall CL, Anderson S, Weesner RE, Goldberg SJ, Crolic KA. Protein-calorie malnutrition associated with alcoholic hepatitis. Veterans Administration Cooperative Study Group on Alcoholic Hepatitis. Am J Med. 1984;76:211-222. [PubMed] |
| 32. | Mendenhall CL, Moritz TE, Roselle GA, Morgan TR, Nemchausky BA, Tamburro CH, Schiff ER, McClain CJ, Marsano LS, Allen JI. A study of oral nutritional support with oxandrolone in malnourished patients with alcoholic hepatitis: results of a Department of Veterans Affairs cooperative study. Hepatology. 1993;17:564-576. [PubMed] |
| 33. | Mendenhall C, Roselle GA, Gartside P, Moritz T. Relationship of protein calorie malnutrition to alcoholic liver disease: a reexamination of data from two Veterans Administration Cooperative Studies. Alcohol Clin Exp Res. 1995;19:635-641. [PubMed] |
| 34. | Lee S, Jin Y, Kee C, Chang Y. Nutritional Status in Alcohol- and Virus-Related Liver Cirrhosis. Korean J Hepatol. 2000;6:59-72. |
| 35. | Stickel F, Hoehn B, Schuppan D, Seitz HK. Review article: Nutritional therapy in alcoholic liver disease. Aliment Pharmacol Ther. 2003;18:357-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 82] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 36. | Henkel AS, Buchman AL. Nutritional support in patients with chronic liver disease. Nat Clin Pract Gastroenterol Hepatol. 2006;3:202-209. [PubMed] [DOI] [Full Text] |
| 37. | Kearns PJ, Young H, Garcia G, Blaschke T, O’Hanlon G, Rinki M, Sucher K, Gregory P. Accelerated improvement of alcoholic liver disease with enteral nutrition. Gastroenterology. 1992;102:200-205. [PubMed] |
| 38. | Cabré E, Rodríguez-Iglesias P, Caballería J, Quer JC, Sánchez-Lombraña JL, Parés A, Papo M, Planas R, Gassull MA. Short- and long-term outcome of severe alcohol-induced hepatitis treated with steroids or enteral nutrition: a multicenter randomized trial. Hepatology. 2000;32:36-42. [PubMed] |
| 39. | Campillo B, Richardet JP, Scherman E, Bories PN. Evaluation of nutritional practice in hospitalized cirrhotic patients: results of a prospective study. Nutrition. 2003;19:515-521. [PubMed] |
| 40. | Hirsch S, de la Maza MP, Gattás V, Barrera G, Petermann M, Gotteland M, Muñoz C, Lopez M, Bunout D. Nutritional support in alcoholic cirrhotic patients improves host defenses. J Am Coll Nutr. 1999;18:434-441. [PubMed] |
| 41. | Marchesini G, Dioguardi FS, Bianchi GP, Zoli M, Bellati G, Roffi L, Martines D, Abbiati R. Long-term oral branched-chain amino acid treatment in chronic hepatic encephalopathy. A randomized double-blind casein-controlled trial. The Italian Multicenter Study Group. J Hepatol. 1990;11:92-101. [PubMed] |
| 42. | Hirsch S, Bunout D, de la Maza P, Iturriaga H, Petermann M, Icazar G, Gattas V, Ugarte G. Controlled trial on nutrition supplementation in outpatients with symptomatic alcoholic cirrhosis. JPEN J Parenter Enteral Nutr. 1993;17:119-124. [PubMed] |
| 43. | Plauth M, Cabré E, Riggio O, Assis-Camilo M, Pirlich M, Kondrup J, Ferenci P, Holm E, Vom Dahl S, Müller MJ. ESPEN Guidelines on Enteral Nutrition: Liver disease. Clin Nutr. 2006;25:285-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 511] [Cited by in RCA: 404] [Article Influence: 21.3] [Reference Citation Analysis (1)] |
| 44. | Plauth M, Cabré E, Campillo B, Kondrup J, Marchesini G, Schütz T, Shenkin A, Wendon J. ESPEN Guidelines on Parenteral Nutrition: hepatology. Clin Nutr. 2009;28:436-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 145] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 45. | DiCecco SR, Francisco-Ziller N. Nutrition in alcoholic liver disease. Nutr Clin Pract. 2006;21:245-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 46. | McClain CJ, Barve SS, Barve A, Marsano L. Alcoholic liver disease and malnutrition. Alcohol Clin Exp Res. 2011;35:815-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 47. | Korean Association for the Study of the Liver (KASL). KASL clinical practice guidelines: management of alcoholic liver disease. Clin Mol Hepatol. 2013;19:216-254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 48. | Mayo-Smith MF, Beecher LH, Fischer TL, Gorelick DA, Guillaume JL, Hill A, Jara G, Kasser C, Melbourne J. Management of alcohol withdrawal delirium. An evidence-based practice guideline. Arch Intern Med. 2004;164:1405-1412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 238] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 49. | Lankarani KB, Ghaffarpasand F, Mahmoodi M, Lotfi M, Zamiri N, Heydari ST, Fallahzadeh MK, Maharlouei N, Babaeinejad M, Mehravar S. Non alcoholic fatty liver disease in southern Iran: a population based study. Hepat Mon. 2013;13:e9248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 63] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 50. | Saitz R, O’Malley SS. Pharmacotherapies for alcohol abuse. Withdrawal and treatment. Med Clin North Am. 1997;81:881-907. [PubMed] |
| 51. | Ghamar Chehreh ME, Vahedi M, Pourhoseingholi MA, Ashtari S, Khedmat H, Amin M, Zali MR, Alavian SM. Estimation of diagnosis and treatment costs of non-alcoholic Fatty liver disease: a two-year observation. Hepat Mon. 2013;13:e7382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 52. | Jung JH, Kim HS. The inhibitory effect of black soybean on hepatic cholesterol accumulation in high cholesterol and high fat diet-induced non-alcoholic fatty liver disease. Food Chem Toxicol. 2013;60:404-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 53. | Day E, Bentham P, Callaghan R, Kuruvilla T, George S. Thiamine for Wernicke-Korsakoff Syndrome in people at risk from alcohol abuse. Cochrane Database Syst Rev. 2004;CD004033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 54. | Morgan MY. The treatment of alcoholic hepatitis. Alcohol Alcohol. 1996;31:117-134. [PubMed] |
| 55. | Forrest E, Mellor J, Stanton L, Bowers M, Ryder P, Austin A, Day C, Gleeson D, O’Grady J, Masson S. Steroids or pentoxifylline for alcoholic hepatitis (STOPAH): study protocol for a randomised controlled trial. Trials. 2013;14:262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 56] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 56. | Rambaldi A, Saconato HH, Christensen E, Thorlund K, Wetterslev J, Gluud C. Systematic review: glucocorticosteroids for alcoholic hepatitis--a Cochrane Hepato-Biliary Group systematic review with meta-analyses and trial sequential analyses of randomized clinical trials. Aliment Pharmacol Ther. 2008;27:1167-1178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 132] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 57. | Mathurin P, Mendenhall CL, Carithers RL, Ramond MJ, Maddrey WC, Garstide P, Rueff B, Naveau S, Chaput JC, Poynard T. Corticosteroids improve short-term survival in patients with severe alcoholic hepatitis (AH): individual data analysis of the last three randomized placebo controlled double blind trials of corticosteroids in severe AH. J Hepatol. 2002;36:480-487. [PubMed] |
| 58. | Louvet A, Naveau S, Abdelnour M, Ramond MJ, Diaz E, Fartoux L, Dharancy S, Texier F, Hollebecque A, Serfaty L. The Lille model: a new tool for therapeutic strategy in patients with severe alcoholic hepatitis treated with steroids. Hepatology. 2007;45:1348-1354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 479] [Cited by in RCA: 512] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 59. | Mathurin P, Abdelnour M, Ramond MJ, Carbonell N, Fartoux L, Serfaty L, Valla D, Poupon R, Chaput JC, Naveau S. Early change in bilirubin levels is an important prognostic factor in severe alcoholic hepatitis treated with prednisolone. Hepatology. 2003;38:1363-1369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 59] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 60. | Mathurin P, O’Grady J, Carithers RL, Phillips M, Louvet A, Mendenhall CL, Ramond MJ, Naveau S, Maddrey WC, Morgan TR. Corticosteroids improve short-term survival in patients with severe alcoholic hepatitis: meta-analysis of individual patient data. Gut. 2011;60:255-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 292] [Cited by in RCA: 305] [Article Influence: 21.8] [Reference Citation Analysis (1)] |
| 61. | Louvet A, Wartel F, Castel H, Dharancy S, Hollebecque A, Canva-Delcambre V, Deltenre P, Mathurin P. Infection in patients with severe alcoholic hepatitis treated with steroids: early response to therapy is the key factor. Gastroenterology. 2009;137:541-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 233] [Cited by in RCA: 272] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 62. | Akriviadis E, Botla R, Briggs W, Han S, Reynolds T, Shakil O. Pentoxifylline improves short-term survival in severe acute alcoholic hepatitis: a double-blind, placebo-controlled trial. Gastroenterology. 2000;119:1637-1648. [PubMed] |
| 63. | Doherty GM, Jensen JC, Alexander HR, Buresh CM, Norton JA. Pentoxifylline suppression of tumor necrosis factor gene transcription. Surgery. 1991;110:192-198. [PubMed] |
| 64. | Whitfield K, Rambaldi A, Wetterslev J, Gluud C. Pentoxifylline for alcoholic hepatitis. Cochrane Database Syst Rev. 2009;CD007339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 65. | Parker R, Armstrong MJ, Corbett C, Rowe IA, Houlihan DD. Systematic review: pentoxifylline for the treatment of severe alcoholic hepatitis. Aliment Pharmacol Ther. 2013;37:845-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 71] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 66. | Mathurin P, Louvet A, Duhamel A, Nahon P, Carbonell N, Boursier J, Anty R, Diaz E, Thabut D, Moirand R. Prednisolone with vs without pentoxifylline and survival of patients with severe alcoholic hepatitis: a randomized clinical trial. JAMA. 2013;310:1033-1041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 146] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 67. | De BK, Gangopadhyay S, Dutta D, Baksi SD, Pani A, Ghosh P. Pentoxifylline versus prednisolone for severe alcoholic hepatitis: a randomized controlled trial. World J Gastroenterol. 2009;15:1613-1619. [PubMed] |
| 68. | Sidhu SS, Goyal O, Singla P, Gupta D, Sood A, Chhina RS, Soni RK. Corticosteroid plus pentoxifylline is not better than corticosteroid alone for improving survival in severe alcoholic hepatitis (COPE trial). Dig Dis Sci. 2012;57:1664-1671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 69. | Louvet A, Diaz E, Dharancy S, Coevoet H, Texier F, Thévenot T, Deltenre P, Canva V, Plane C, Mathurin P. Early switch to pentoxifylline in patients with severe alcoholic hepatitis is inefficient in non-responders to corticosteroids. J Hepatol. 2008;48:465-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 118] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 70. | Spahr L, Rubbia-Brandt L, Frossard JL, Giostra E, Rougemont AL, Pugin J, Fischer M, Egger H, Hadengue A. Combination of steroids with infliximab or placebo in severe alcoholic hepatitis: a randomized controlled pilot study. J Hepatol. 2002;37:448-455. [PubMed] |
| 71. | Naveau S, Chollet-Martin S, Dharancy S, Mathurin P, Jouet P, Piquet MA, Davion T, Oberti F, Broët P, Emilie D. A double-blind randomized controlled trial of infliximab associated with prednisolone in acute alcoholic hepatitis. Hepatology. 2004;39:1390-1397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 322] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 72. | Menon KV, Stadheim L, Kamath PS, Wiesner RH, Gores GJ, Peine CJ, Shah V. A pilot study of the safety and tolerability of etanercept in patients with alcoholic hepatitis. Am J Gastroenterol. 2004;99:255-260. [PubMed] |
| 73. | Boetticher NC, Peine CJ, Kwo P, Abrams GA, Patel T, Aqel B, Boardman L, Gores GJ, Harmsen WS, McClain CJ. A randomized, double-blinded, placebo-controlled multicenter trial of etanercept in the treatment of alcoholic hepatitis. Gastroenterology. 2008;135:1953-1960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 265] [Cited by in RCA: 255] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 74. | Moreno C, Langlet P, Hittelet A, Lasser L, Degré D, Evrard S, Colle I, Lemmers A, Devière J, Le Moine O. Enteral nutrition with or without N-acetylcysteine in the treatment of severe acute alcoholic hepatitis: a randomized multicenter controlled trial. J Hepatol. 2010;53:1117-1122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 80] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 75. | Nguyen-Khac E, Thevenot T, Piquet MA, Benferhat S, Goria O, Chatelain D, Tramier B, Dewaele F, Ghrib S, Rudler M. Glucocorticoids plus N-acetylcysteine in severe alcoholic hepatitis. N Engl J Med. 2011;365:1781-1789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 294] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 76. | Caballería J, Parés A, Brú C, Mercader J, García Plaza A, Caballería L, Clemente G, Rodrigo L, Rodés J. Metadoxine accelerates fatty liver recovery in alcoholic patients: results of a randomized double-blind, placebo-control trial. Spanish Group for the Study of Alcoholic Fatty Liver. J Hepatol. 1998;28:54-60. [PubMed] |
| 77. | Mezey E. Commentary on the hypermetabolic state and the role of oxygen in alcohol-induced liver injury. Recent Dev Alcohol. 1984;2:135-141. [PubMed] |
| 78. | Orrego H, Blake JE, Blendis LM, Compton KV, Israel Y. Long-term treatment of alcoholic liver disease with propylthiouracil. N Engl J Med. 1987;317:1421-1427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 120] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 79. | Rambaldi A, Gluud C. Propylthiouracil for alcoholic liver disease. Cochrane Database Syst Rev. 2002;CD002800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 80. | Fede G, Germani G, Gluud C, Gurusamy KS, Burroughs AK. Propylthiouracil for alcoholic liver disease. Cochrane Database Syst Rev. 2011;CD002800. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 81. | Morgan TR, Weiss DG, Nemchausky B, Schiff ER, Anand B, Simon F, Kidao J, Cecil B, Mendenhall CL, Nelson D. Colchicine treatment of alcoholic cirrhosis: a randomized, placebo-controlled clinical trial of patient survival. Gastroenterology. 2005;128:882-890. [PubMed] |
| 82. | Kershenobich D, Uribe M, Suárez GI, Mata JM, Pérez-Tamayo R, Rojkind M. Treatment of cirrhosis with colchicine. A double-blind randomized trial. Gastroenterology. 1979;77:532-536. [PubMed] |
| 83. | Kershenobich D, Vargas F, Garcia-Tsao G, Perez Tamayo R, Gent M, Rojkind M. Colchicine in the treatment of cirrhosis of the liver. N Engl J Med. 1988;318:1709-1713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 210] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 84. | Rambaldi A, Gluud C. Colchicine for alcoholic and non-alcoholic liver fibrosis and cirrhosis. Cochrane Database Syst Rev. 2005;CD002148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 30] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 85. | Li J, Kim CI, Leo MA, Mak KM, Rojkind M, Lieber CS. Polyunsaturated lecithin prevents acetaldehyde-mediated hepatic collagen accumulation by stimulating collagenase activity in cultured lipocytes. Hepatology. 1992;15:373-381. [PubMed] |
| 86. | Lieber CS, Robins SJ, Li J, DeCarli LM, Mak KM, Fasulo JM, Leo MA. Phosphatidylcholine protects against fibrosis and cirrhosis in the baboon. Gastroenterology. 1994;106:152-159. [PubMed] |
| 87. | Lieber CS, Weiss DG, Groszmann R, Paronetto F, Schenker S. II. Veterans Affairs Cooperative Study of polyenylphosphatidylcholine in alcoholic liver disease. Alcohol Clin Exp Res. 2003;27:1765-1772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 87] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 88. | Kim MY, Cho MY, Baik SK, Jeong PH, Suk KT, Jang YO, Yea CJ, Kim JW, Kim HS, Kwon SO. Beneficial effects of candesartan, an angiotensin-blocking agent, on compensated alcoholic liver fibrosis - a randomized open-label controlled study. Liver Int. 2012;32:977-987. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 58] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 89. | Rambaldi A, Jacobs BP, Iaquinto G, Gluud C. Milk thistle for alcoholic and/or hepatitis B or C virus liver diseases. Cochrane Database Syst Rev. 2005;CD003620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 90. | Ferenci P, Dragosics B, Dittrich H, Frank H, Benda L, Lochs H, Meryn S, Base W, Schneider B. Randomized controlled trial of silymarin treatment in patients with cirrhosis of the liver. J Hepatol. 1989;9:105-113. [PubMed] |
| 91. | Rambaldi A, Jacobs BP, Iaquinto G, Gluud C. Milk thistle for alcoholic and/or hepatitis B or C liver diseases--a systematic cochrane hepato-biliary group review with meta-analyses of randomized clinical trials. Am J Gastroenterol. 2005;100:2583-2591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 66] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 92. | Burra P, Senzolo M, Adam R, Delvart V, Karam V, Germani G, Neuberger J. Liver transplantation for alcoholic liver disease in Europe: a study from the ELTR (European Liver Transplant Registry). Am J Transplant. 2010;10:138-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 248] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 93. | Waki K, Tamura S, Sugawara Y, Yamashiki N, Kadowaki T, Kokudo N. An analysis of the OPTN/UNOS Liver Transplant Registry. Clin Transpl. 2009;55-64. [PubMed] |
| 94. | Dumortier J, Guillaud O, Adham M, Boucaud C, Delafosse B, Bouffard Y, Paliard P, Scoazec JY, Boillot O. Negative impact of de novo malignancies rather than alcohol relapse on survival after liver transplantation for alcoholic cirrhosis: a retrospective analysis of 305 patients in a single center. Am J Gastroenterol. 2007;102:1032-1041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 81] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 95. | Mackie J, Groves K, Hoyle A, Garcia C, Garcia R, Gunson B, Neuberger J. Orthotopic liver transplantation for alcoholic liver disease: a retrospective analysis of survival, recidivism, and risk factors predisposing to recidivism. Liver Transpl. 2001;7:418-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 119] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 96. | Burra P, Mioni D, Cecchetto A, Cillo U, Zanus G, Fagiuoli S, Naccarato R, Martines D. Histological features after liver transplantation in alcoholic cirrhotics. J Hepatol. 2001;34:716-722. [PubMed] |
| 97. | Tomé S, Martinez-Rey C, González-Quintela A, Gude F, Brage A, Otero E, Abdulkader I, Forteza J, Bustamante M, Varo E. Influence of superimposed alcoholic hepatitis on the outcome of liver transplantation for end-stage alcoholic liver disease. J Hepatol. 2002;36:793-798. [PubMed] |
| 98. | Dureja P, Lucey MR. The place of liver transplantation in the treatment of severe alcoholic hepatitis. J Hepatol. 2010;52:759-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 45] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 99. | McCallum S, Masterton G. Liver transplantation for alcoholic liver disease: a systematic review of psychosocial selection criteria. Alcohol Alcohol. 2006;41:358-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 82] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 100. | Lucey MR, Brown KA, Everson GT, Fung JJ, Gish R, Keeffe EB, Kneteman NM, Lake JR, Martin P, McDiarmid SV. Minimal criteria for placement of adults on the liver transplant waiting list: a report of a national conference organized by the American Society of Transplant Physicians and the American Association for the Study of Liver Diseases. Liver Transpl Surg. 1997;3:628-637. [PubMed] |
| 101. | Everhart JE, Beresford TP. Liver transplantation for alcoholic liver disease: a survey of transplantation programs in the United States. Liver Transpl Surg. 1997;3:220-226. [PubMed] |
| 102. | Pfitzmann R, Schwenzer J, Rayes N, Seehofer D, Neuhaus R, Nüssler NC. Long-term survival and predictors of relapse after orthotopic liver transplantation for alcoholic liver disease. Liver Transpl. 2007;13:197-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 209] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 103. | Perney P, Bismuth M, Sigaud H, Picot MC, Jacquet E, Puche P, Jaber S, Rigole H, Navarro F, Eledjam JJ. Are preoperative patterns of alcohol consumption predictive of relapse after liver transplantation for alcoholic liver disease? Transpl Int. 2005;18:1292-1297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 52] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 104. | De Gottardi A, Spahr L, Gelez P, Morard I, Mentha G, Guillaud O, Majno P, Morel P, Hadengue A, Paliard P. A simple score for predicting alcohol relapse after liver transplantation: results from 387 patients over 15 years. Arch Intern Med. 2007;167:1183-1188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 154] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 105. | Pageaux GP, Bismuth M, Perney P, Costes V, Jaber S, Possoz P, Fabre JM, Navarro F, Blanc P, Domergue J. Alcohol relapse after liver transplantation for alcoholic liver disease: does it matter? J Hepatol. 2003;38:629-634. [PubMed] |
| 106. | Tang H, Boulton R, Gunson B, Hubscher S, Neuberger J. Patterns of alcohol consumption after liver transplantation. Gut. 1998;43:140-145. [PubMed] |
| 107. | Mathurin P, Moreno C, Samuel D, Dumortier J, Salleron J, Durand F, Castel H, Duhamel A, Pageaux GP, Leroy V. Early liver transplantation for severe alcoholic hepatitis. N Engl J Med. 2011;365:1790-1800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 616] [Cited by in RCA: 658] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 108. | Vanlemmens C, Di Martino V, Milan C, Messner M, Minello A, Duvoux C, Poynard T, Perarnau JM, Piquet MA, Pageaux GP. Immediate listing for liver transplantation versus standard care for Child-Pugh stage B alcoholic cirrhosis: a randomized trial. Ann Intern Med. 2009;150:153-161. [PubMed] |
| 109. | Zanus G, Carraro A, Vitale A, Gringeri E, D’Amico F, Valmasoni M, D’Amico FE, Brolese A, Boccagni P, Neri D. Alcohol abuse and de novo tumors in liver transplantation. Transplant Proc. 2009;41:1310-1312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 110. | Haagsma EB, Hagens VE, Schaapveld M, van den Berg AP, de Vries EG, Klompmaker IJ, Slooff MJ, Jansen PL. Increased cancer risk after liver transplantation: a population-based study. J Hepatol. 2001;34:84-91. [PubMed] |
| 111. | Park HW, Hwang S, Ahn CS, Kim KH, Moon DB, Ha TY, Song GW, Jung DH, Park GC, Namgoong JM. De novo malignancies after liver transplantation: incidence comparison with the Korean cancer registry. Transplant Proc. 2012;44:802-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 112. | Zhang Z, Wang FS. Stem cell therapies for liver failure and cirrhosis. J Hepatol. 2013;59:183-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 75] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 113. | Akiyama K, Chen C, Wang D, Xu X, Qu C, Yamaza T, Cai T, Chen W, Sun L, Shi S. Mesenchymal-stem-cell-induced immunoregulation involves FAS-ligand-/FAS-mediated T cell apoptosis. Cell Stem Cell. 2012;10:544-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 486] [Cited by in RCA: 570] [Article Influence: 43.8] [Reference Citation Analysis (0)] |
| 114. | Aurich H, Sgodda M, Kaltwasser P, Vetter M, Weise A, Liehr T, Brulport M, Hengstler JG, Dollinger MM, Fleig WE. Hepatocyte differentiation of mesenchymal stem cells from human adipose tissue in vitro promotes hepatic integration in vivo. Gut. 2009;58:570-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 258] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 115. | Kuo TK, Hung SP, Chuang CH, Chen CT, Shih YR, Fang SC, Yang VW, Lee OK. Stem cell therapy for liver disease: parameters governing the success of using bone marrow mesenchymal stem cells. Gastroenterology. 2008;134:2111-221, 2111-221. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 358] [Cited by in RCA: 354] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 116. | Jang YO, Kim YJ, Baik SK, Kim MY, Eom YW, Cho MY, Park HJ, Park SY, Kim BR, Kim JW. Histological improvement following administration of autologous bone marrow-derived mesenchymal stem cells for alcoholic cirrhosis: a pilot study. Liver Int. 2014;34:33-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 133] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 117. | Terai S, Ishikawa T, Omori K, Aoyama K, Marumoto Y, Urata Y, Yokoyama Y, Uchida K, Yamasaki T, Fujii Y. Improved liver function in patients with liver cirrhosis after autologous bone marrow cell infusion therapy. Stem Cells. 2006;24:2292-2298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 363] [Cited by in RCA: 349] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 118. | Ismail A, Fouad O, Abdelnasser A, Chowdhury A, Selim A. Stem cell therapy improves the outcome of liver resection in cirrhotics. J Gastrointest Cancer. 2010;41:17-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 119. | Pai M, Zacharoulis D, Milicevic MN, Helmy S, Jiao LR, Levicar N, Tait P, Scott M, Marley SB, Jestice K. Autologous infusion of expanded mobilized adult bone marrow-derived CD34+ cells into patients with alcoholic liver cirrhosis. Am J Gastroenterol. 2008;103:1952-1958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 153] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 120. | Spahr L, Lambert JF, Rubbia-Brandt L, Chalandon Y, Frossard JL, Giostra E, Hadengue A. Granulocyte-colony stimulating factor induces proliferation of hepatic progenitors in alcoholic steatohepatitis: a randomized trial. Hepatology. 2008;48:221-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 177] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 121. | Orman ES, Odena G, Bataller R. Alcoholic liver disease: pathogenesis, management, and novel targets for therapy. J Gastroenterol Hepatol. 2013;28 Suppl 1:77-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 179] [Article Influence: 14.9] [Reference Citation Analysis (2)] |
| 122. | Dominguez M, Miquel R, Colmenero J, Moreno M, García-Pagán JC, Bosch J, Arroyo V, Ginès P, Caballería J, Bataller R. Hepatic expression of CXC chemokines predicts portal hypertension and survival in patients with alcoholic hepatitis. Gastroenterology. 2009;136:1639-1650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 190] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 123. | Ki SH, Park O, Zheng M, Morales-Ibanez O, Kolls JK, Bataller R, Gao B. Interleukin-22 treatment ameliorates alcoholic liver injury in a murine model of chronic-binge ethanol feeding: role of signal transducer and activator of transcription 3. Hepatology. 2010;52:1291-1300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 364] [Cited by in RCA: 367] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 124. | Pritchard MT, McMullen MR, Stavitsky AB, Cohen JI, Lin F, Medof ME, Nagy LE. Differential contributions of C3, C5, and decay-accelerating factor to ethanol-induced fatty liver in mice. Gastroenterology. 2007;132:1117-1126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 124] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 125. | Seth D, Gorrell MD, Cordoba S, McCaughan GW, Haber PS. Intrahepatic gene expression in human alcoholic hepatitis. J Hepatol. 2006;45:306-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 126. | Szabo G, Bala S. Alcoholic liver disease and the gut-liver axis. World J Gastroenterol. 2010;16:1321-1329. [PubMed] |
| 127. | Seo YS, Shah VH. The role of gut-liver axis in the pathogenesis of liver cirrhosis and portal hypertension. Clin Mol Hepatol. 2012;18:337-346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 109] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 128. | Lucey MR, Mathurin P, Morgan TR. Alcoholic hepatitis. N Engl J Med. 2009;360:2758-2769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 659] [Cited by in RCA: 693] [Article Influence: 43.3] [Reference Citation Analysis (0)] |
| 129. | Kendrick SF, O’Boyle G, Mann J, Zeybel M, Palmer J, Jones DE, Day CP. Acetate, the key modulator of inflammatory responses in acute alcoholic hepatitis. Hepatology. 2010;51:1988-1997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 136] [Article Influence: 9.1] [Reference Citation Analysis (0)] |