Published online Sep 21, 2014. doi: 10.3748/wjg.v20.i35.12391
Revised: January 30, 2014
Accepted: May 29, 2014
Published online: September 21, 2014
Processing time: 288 Days and 1.4 Hours
Colorectal cancer (CRC) is one of the commonest cancers with 1.2 million new cases diagnosed each year in the world. It remains the fourth most common cause of cancer-related mortality in the world and accounts for > 600000 cancer-related deaths each year. There have been significant advances in treatment of metastatic CRC in last decade or so, due to availability of new active targeted agents and more aggressive approach towards the management of CRC, particularly with liver-only-metastases; however, these drugs work best when combined with conventional chemotherapy agents. Despite these advances, there is a lack of biomarkers to inform us about the accurate management of the patients with metastatic CRC. It is therefore imperative to carefully select the patients with comprehensive multi-disciplinary team input in order to optimise the management of these patients. In this review we will discuss various treatment options available in management of colorectal liver metastases with potential guidance on how and when to choose these options along with consideration on future directions in management of this disease.
Core tip: Colorectal cancer is one of the commonest cancers in the world. The management of metastatic colorectal cancer has changed significantly in last decade or so; primarily based on better understanding of the molecular complexity of colorectal cancer coupled with aggressive approach in management of colorectal liver metastases. Colorectal liver metastasises were once considered as incurable disease but with better treatment options and valuable input from multi-disciplinary teams, this disease can be cured in a proportion of patients. This review takes into account various clinical scenarios and their complexity that the clinicians may face during management of this disease.
- Citation: Khan K, Wale A, Brown G, Chau I. Colorectal cancer with liver metastases: Neoadjuvant chemotherapy, surgical resection first or palliation alone? World J Gastroenterol 2014; 20(35): 12391-12406
- URL: https://www.wjgnet.com/1007-9327/full/v20/i35/12391.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i35.12391
Colorectal cancer (CRC) is a leading cause of cancer-related mortality in the world with over 1.2 million new cases diagnosed each year along with > 600000 death per year[1,2]. Curative surgery is the mainstay of treatment for early stage CRC with pathological staging being considered as the most important predictor of post-operative outcome. Majority of the patients could have relatively good outcomes with 5-year survival ranging from 50%-90%[3] depending upon the initial stage of the disease and other prognostic factors; however, despite significant improvements in screening and management of CRC in recent times, the 5-year survival rate for patients with metastatic CRC (mCRC) remains poor[4]. In these patients, the prognosis is closely related to the location and extent of distant metastatic disease.
In patients with unresectable mCRC, improvement in survival from 12 mo with fluorouracil therapy to approximately 2 years has been observed with combinations with oxaliplatin (FOLFOX) or it’s pro-drug capecitabine (CAPOX), or with irinotecan (FOLFIRI)[5-8]. Recent randomised control trials suggested that median overall survival (OS) of over 30 mo can now be achieved[9,10] in patients with mCRC. Approximately 50% of the patients tend to have involvement of liver during the course of their disease[11,12] and a proportion of them with colorectal liver-only metastases (CLM) can undergo liver metastasectomy; this could result in significant improvement in their outcomes. Many patients however develop liver metastases which are unresectable. Earlier reports suggested that with oxaliplatin-based chemotherapy, 13% of patients with initially non-resectable liver metastases could have their disease resected after chemotherapy[13]. Five- and ten-year overall survival rates of 33% and 27% have been observed respectively in this cohort of patients[14]. Moreover, approximately 15% of patients never developed recurrent disease, thus they were considered “cured”[15]. Furthermore, liver resection rate after chemotherapy correlates significantly with radiological objective response rate (ORR)[16] and in addition resected patients enjoy significantly prolonged survival compared to those who can’t undergo resection[17].
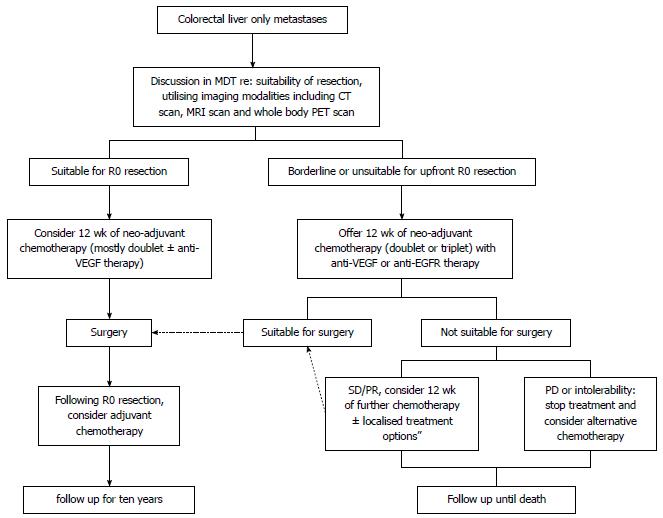
The treatment options for CLM include (Figure 1): surgical resection, systemic chemotherapy, localised treatment options: radiofrequency ablation (RFA), selective internal radiotherapy (SIRT), chemoembolization, other local ablative methods.
In this review we will discuss the most common treatment options with particular emphasis upon the role of systemic therapy in CLM, either deemed resectable or un-resectable at the initial presentation. We will also discuss the key molecular pathways and emerging areas of interest that may provide new opportunities for therapeutic intervention in this context.
If CLM are amenable to surgery, then this can be offered with curative intent. Post metastasectomy the 5-year survival rates vary between 27%-58% and 10-year survival rates are 15%-17%[18-29]; Post-operative complication rates are higher in patients undergoing simultaneous hepatic and colonic resections of the synchronous tumours. However, in most high volume surgical centres with experience in CLM metastasectomy, surgical mortality rates are reported to be less than 5%[30,31]. The modern approach to resectability of CLM has largely changed with emphasis of most MDT decisions in favour of offering surgery to the patients, in the absence of extra-hepatic disease. Due to its clear impact on overall outcome, surgical resection is the treatment of choice, where possible; however, these decisions should be made after comprehensive discussion in the regional hepato-biliary MDT.
Resectability criteria and prognostic factor: Although there is published literature on various treatment approaches, universal agreement on the resectability criteria in the context of CLM, remains not well defined. The distinction between resectable and non-resectable CLM varies from institute to institute and this was highlighted in the large CELIM study which identified 64.5% discordance within a board of surgeons when they were blinded to baseline or post-treatment scans and patient clinical outcomes; this included 6.8% disagreement in critical decisions of resectability vs non-resectability[32]. Some authors have defined risk-scoring systems to guide the MDTs about the patient selection, but most MDTs rely on clinical assessment and local surgical expertise rather than the set scoring criteria. This reflects on the heterogeneity of patient population treated within the context of different clinical trials and lack of reliable data to inform us about the prognostic factors being examined in these trials.
One of the commonly used scoring system to help patient selection was described by Fong and colleagues[33], where a retrospective review of 1001 patients after hepatic resection for mCRC was conducted. In this study clinical, pathological and outcome data were collected. Five year and 10-year survival rates were 37% and 22% respectively. The authors identified seven independent factors including positive margins, extra-hepatic disease, node-positive primary, disease-free survival from primary to metastatic disease, number of hepatic tumour > 1, largest tumour > 5 cm and carcinoembryonic antigen > 200 ng/mL which were found to be predictive of survival. The last five of the above factors were combined to form a prognostic score, with each factor assigned one point. The combined score was found to be highly predictive of the outcome. Patients with < 2 score had a favourable outcome; those with three, four or five criteria were recommended to be considered for adjuvant chemotherapy trials in view of high risk and poor outcomes. Patients with score of 5 had 5-year survival of 14% with a median survival of 22 mo compared to 44% and 51 mo in patients with one risk factor. This score however only was validated for patients with upfront resection and doesn’t include patients who underwent preoperative chemotherapy; its validity in modern treatment paradigm is therefore often limited. Following that many scoring systems have been developed but none of them have been validated and thus have limited utility in clinical practice[34,35].
The limitations for hepatic resection in general are considered to be, based on one of the following: (1) no upfront R0 resection of all hepatic lesions possible; (2) less than 30% estimated residual liver after resection; (3) disease in contact with major vessels of the remnant liver (vessels remaining after potential hepatectomy); (4) significant co-morbidities excluding surgery; and/or (5) presence of extra-hepatic disease.
Detection of number and location of metastases: Based on several retrospective analyses, in the past, number of metastatic sites was considered as one of the major contraindications to hepatic resection in CLM but with modern surgical techniques and more encouraging role of preoperative chemotherapy, patients could still be deemed resectable with multiple CLM; although several studies have identified the prognostic value of number of metastatic sites at initial presentation[36,37]. Furthermore, bilobar involvement previously deterred the surgeons from offering curative hepatic resection but these concepts have been challenged by several other groups[28,38-42]. Additionally, one of the classic contraindication to surgery in CLM is considered to be portal node metastases due to an old perception that these patients were at higher risk of developing systemic disease; however, this concept has again been challenged in recent times, with several authors recognising that although this could be considered a poor predictor of outcome but can not be considered an absolute contraindication to hepatectomy[43,44]. Similarly, the presence of limited extra-hepatic metastatic disease is no longer considered an absolute contraindication to surgery; however, the outcomes may not be as favourable as with CLM or thoracic metastases alone[45-47].
Importance of imaging modalities: The above discussion highlights the importance of having detailed baseline information about the actual extent of the disease in order to make best possible decisions for the patients. Modern techniques of imaging including contrast-enhanced computed tomography (CT), contrast enhanced magnetic resonance imaging (MRI) scan of the liver and whole body positron emission tomography (PET) with computed tomography (PET-CT) have made significant contributions in assessment of resectability of CLM. However, PET-CT is limited in the diagnosis of liver metastases smaller than 1cm[48]. A large meta-analysis[49] including 39 studies with 3391 patients examined the sensitivity estimates of CT, MRI and fludeoxyglucose FDG PET on per-lesion basis. All included patients had histologically proven CRC along with CLM. The study showed 74.4%, 80.3% and 81.4% sensitivity respectively; however on per-patient basis the sensitivity was 83.6%, 88.2% and 94.1% with three modalities respectively. Per-patient sensitivity of CT was found to be lower than FDG PET (P = 0.025). For lesions smaller than 10 mm, the sensitivity estimates for MR imaging were better than CT scan; no difference was seen for lesion above 10 mm. The study authors concluded that the MRI scan was better imaging modality for detection of liver metastases; however, limited data were available on FDG PET.
Given the sensitivity of MRI scan of the liver is proven to be superior to CT scan, even in the presence of new generation CT scanners using triple phase imaging with contrast administration, multiple imaging modalities including both CT and MRI scans are often used in evaluation of CLM. Additionally, the apparent diffusion coefficient (ADC), derived from diffusion-weighted MRI (DW-MRI), may provide information on predicting response to chemotherapy[50,51]. Our group performed a recent retrospective series including 102 patients, who underwent pre-treatment DW-MRI; we found that the CLM with higher pre-treatment mean ADC were associated with poorer response to chemotherapy but this didn’t co-relate with the outcome of the patients. This technique can nevertheless be utilised in clinical setting especially in patients undergoing neo-adjuvant chemotherapy[52].
FDG-PET scan is considered to be more sensitive investigation than CT for detection of metastatic disease and thus can have an impact in the clinical management of CLM patients. A pooled analysis on six previously reported articles showed that the sensitivity and specificity of FDG PET for hepatic disease were 79.9% and 92.3% compared to 82.7% and 84.1% with CT scan; for extra-hepatic disease those were 91.2% and 98.4% with PET scan compared to 60.9% and 91.1% with CT respectively. This had a significant impact on clinical management in 25% of the cases[53]. Subsequent studies showed that the use of PET as an adjunct to CT could be associated with alteration in patient’s management in one-third of the patients[54].
These data indicate that treatment approach in CLM could be significantly influenced by the extent and resectability of disease; therefore, multiple imaging modalities should be considered, in order to optimise the management of patients with CLM and indeed in order to avoid un-necessary surgical morbidity and mortality.
Neo-adjuvant chemotherapy for clearly R0 resectable CLM: Surgical resection is regarded as the standard of care for patients with CLM, but relapse is common. Patients with potentially resectable CLM can therefore be considered for neoadjuvant chemotherapy. Recent data, however suggests that the initial advantage seen in progression free survival (PFS) for patients receiving additional systemic treatment may not actually translate into a meaningful OS advantage[55]. EORTC 40983[56] is the largest randomised double blinded trial examining the role of neo-adjuvant chemotherapy in this context. This trial recruited 364 patients with 1:1 randomisation to FOLFOX4 [fluorouracil (5-FU)/leucovorin and oxaliplatin] six cycles before and six cycles after surgery (n = 182) vs upfront surgery alone (n = 182). Patients assigned to chemotherapy arm had an ORR of 36% with four patients achieving a complete response (CR). Eleven patients progressed, of which 8 were no longer resectable. Eighty-three percent patients were resected after a median of 6 cycles of preoperative chemotherapy compared to 84% in the surgery group. The absolute increase in progression free survival at 3 years for patients undergoing liver resection after chemotherapy was 9.2%. Reversible complications occurred more frequently in the chemotherapy group (25% vs 16%); however, no impact on post-operative mortality was observed. Five year OS improvement of 4.1% with median OS of 64 mo (vs 55 mo) in favour of chemotherapy arm was observed; however, this was not statistically significant. It is however worth-noting that the trial was not powered to detect OS benefit. Furthermore a recent retrospective study analysing 466 patients with CLM, concluded that there was no significant difference observed between the outcomes of the patients receiving preoperative neoadjuvant chemotherapy compared with those who didn’t receive neo-adjuvant treatment[57]. This study had however serious limitations including the retrospective nature of the study, imbalanced sample size between the two groups and heterogeneity of the patient population treated in the study.
Although these data show that the benefit for upfront chemotherapy in patients with CLM remains controversial; decision about scope of systemic therapy must be made on case-to-case basis with consideration of tumour biology, previous response and PFS on chemotherapy in the event of metachronous CLM, other co-morbid conditions and local surgical expertise.
Conversion chemotherapy for borderline CLM: Most patients with CLM may present with extensive hepatic disease which may not be amenable to surgery. These patients require systemic chemotherapy, either given with palliative intent or as neo-adjuvant treatment to downsize their tumours in order to make them operable. This treatment is referred to as, “conversion therapy (Figure 2)”. Three conventional cytotoxic drugs are available for treatment of mCRC: fluoropyrimidines, oxaliplatin and irinotecan. The exposure to all there drugs during the course of patients illness correlates strongly with median OS of the patients with mCRC. Sequential use of these drugs can sometimes be justified in order to reduce toxic effects and eventual exposure to all of these drugs. The major caveats however to this approach include: not exploiting the synergistic potential of the drugs, lack of intrinsic anti-tumour activity of oxaliplatin as monotherapy and tendency of some patients to deteriorate prior to receiving all of these drugs.
In patients where downsizing the tumour to optimise the chances of surgery is an option, one of the aims of conversion chemotherapy should be to achieve the highest possible ORR. The association of better ORR with improved resection rates has been previously demonstrated[16]. It is also been reported that 12%-33% of the patients who were treated with doublet chemotherapy regimen and had initially un-resectable CLM were able to achieve significant response so that they could undergo surgery[13,58-60]. In addition, epidermal growth factor receptor (EGFR) targeted therapy along with two-drug regimen resulted in one of the highest ORR for CLM in patients with no mutations in Kirsten sarcoma virus (K-RAS) genes[32]. Also, CAPOX along with vascular endothelial growth factor (VEGF) targeted therapy with bevacizumab has been shown to result in similar ORR for CLM[61]. Both aforementioned studies demonstrated similar R0/R1 liver resection rates after conversion chemotherapy. Ye et al[62] performed one of the first randomised studies of chemotherapy with or without cetuximab in patients with unresectable CLM. This study did confirm the general belief that chemotherapy plus cetuximab led to significantly higher ORR, liver resection rate, PFS and OS. The study therefore provided considerable weight to support the use of chemotherapy plus cetuximab as a standard treatment option in the K-RAS wild type (WT) disease. Nevertheless, as the primary endpoint of this study was the conversion rate to liver resection, the number of patients included was relatively small. The survival outcomes were only secondary endpoints and this could not be deemed to be definitive that chemotherapy plus cetuximab would improve OS, especially given the short follow up in this study. Therefore there remains a need for a phase III study to address this question. One of the major strengths of this study however, was the multi-disciplinary approach in the management of patients with CLM. The authors chose the criteria of unresectability which would be in line with modern international practice and given that this study was conducted in a single institution, it was likely that the decision-making process of resectability was uniform and consistent both among patients as well as over time. It was however unclear both from the paper and the protocol of the study as to what pre-treatment evaluations were undertaken. United Kingdom-based phase III study which randomised patients with KRAS WT CLM between surgery and surgery with chemotherapy along with cetuximab failed to show any survival advantage in favour of cetuximab plus chemotherapy arm[63]. Although the final results of this study are still awaited, the proposed reasons for failure include heterogeneity of selected patient population (patients could represent either resectable or un-resectable CLM with no restriction to number of liver lesions) and use of capecitabine and oxaliplatin in number of patients in the study, which may not have synergised well with cetuximab, as previously demonstrated in the COIN study[64].
In recent times, the triplet FOLFOXIRI was investigated as first-line treatment in un-selected patients with mCRC and showed higher ORR (66% vs 41%, P = 0.0002), and better secondary metastasectomy rate (15% vs 6%, P = 0.003) when compared to FOLFIRI[65]. PFS (9.8 mo vs 6.9 mo) and OS (22.6 mo vs 16.7 mo) were also both significantly better and in favour of FOLFIXIRI. This trial however reported benefits in mCRC with all comers (with extra-hepatic disease) and thus the results couldn’t be interpreted in patients with CLM alone. Recently data from a phase II study reported 5-year survival of 42% in patients who were converted by FOLFOXIRI to undergo liver resection[66]. In another phase II study, the addition of bevacizumab to FOLFOXIRI[67] resulted in ORR of 80% and R0 resection rate of 40% in patients with CLM. The ORR to FOLFOXIRI plus bevacizumab did not appear to be influenced by K-RAS or B-RAF mutational status of the tumour. More recently, a randomised phase III study showed a significant improvement of PFS in patients receiving FOLFOXIRI + bevacizumab compared to those who received FOLFIRI + bevacizumab[68]. Likewise, the addition of another EGFR targeting drug, panitumumab to FOLFOXIRI resulted in ORR of 89%, with 43% patients undergoing secondary for metastatic disease and R0 resection in 35% of the patients with K-RAS WT patients was achieved in this single-arm phase II study[69]. All these studies strengthen the need for conducting a randomised phase III trial evaluating the role of triplet chemotherapy with targeted therapies in order to have more robust data to support the initial clinical findings.
With the current evidence, we support the view of using doublet chemotherapy with bevacizumab or cetuximab in K-RAS WT patients and that of using doublet chemotherapy with bevacizumab in K-RAS mutant patients; however, where possible, triplet chemotherapy along with targeted agents should be considered.
Challenges associated with neo-adjuvant chemotherapy for CLM: Although neo-adjuvant chemotherapy has the potential to convert initially un-resectable disease into resectable one for some patients, the frequency with which this happens is still relatively low. There are some important considerations in adapting this approach which are summarised below: (1) one of the common concerns about the neo-adjuvant chemotherapy in initially resectable disease is the associated risk of progression of the disease due to delay in surgery. In the EORTC 40983 study, 7% of the patients experienced progressive disease during the time of pre-operative chemotherapy leading to un-resectability; half of them however had new lesions[56]. Some old reports indicate poor prognosis in such patients partly attributable to the inability to offer these patients surgical resection[70]; this concept has however been challenged in recent times with others suggesting that the progression of disease rather reflects upon the aggressive disease biology and may in fact prevent unnecessary surgical morbidity or indeed mortality[14,71,72]; (2) with the recent advancements in cytotoxic and targeted therapy regimens, and aggressive management of CRC, radiological CR to the treatment can be observed in a proportion of patients. This could lead to disappearance of initially observed liver metastases and can pose a potential challenge for physicians to form an appropriate treatment strategy. Many published series have reported the increasing incidence and management of disappearing liver metastases (DLM) with variety of chemotherapy regimens with out without hepatic arterial infusion (HIA)[73-81]. The rate of DLM has varied from 6.5% to 36%, partly due to the fact that CR is largely dependent on the quality of preoperative imaging. It is important to recognise that CR on imaging doesn’t always mean complete pathological response. The true CR in the context of CLM could be best defined as either complete pathological response or no recurrence of the CLM, if let in situ[82]. The rate of reported pathological response in the CLM setting is largely variable, ranging from 9% to 58%[73,76,77,81,83]. This could be attributed to several heterogeneous factors including the use of HIA, number and size of metastatic sites, disease biology, pre-operative imaging techniques and the number of cycles and duration of neo-adjuvant chemotherapy. When CLM are left in situ, after achieving CR on imaging, the recurrence rates have varied from 38% to 74%[74,76]; however, this had no major impact on the OS of the patients when the CLM were left in situ as the patients were offered re-resection or other localised treatments like RFA at the time of recurrence[79]. The above discussion on DLM highlights the importance of baseline and pre-operative imaging and the fact that there are currently no standard guidelines to manage DLM. All the imaging modalities including DW-MRI scan and PET scan should be utilised in order to be certain about the nature of response to neo-adjuvant therapy; in the event of definite CR, we adopt “watch and wait” policy in our institute with frequent clinical, and imaging monitoring of these patients on 3-monthly basis during the first three years followed by 6-monthly imaging by CT scan for another two years. We suggest that for patients where DLM are left in situ, there should be a low threshold for repeating the imaging on any rise of tumour markers or in the event of clinical symptoms; (3) there are growing numbers of reports suggesting liver toxicity as results of neo-adjuvant chemotherapy. Whilst, oxaliplatin-based backbone is associated with risk of increased vascular lesions, irinotecan-based combinations can cause steatohepatitis[84,85]. In addition chemotherapy carries risk of systemic toxicities including post-operative bleeding, neutropenia and associated infections, pot-operative complications and poorer functional reserve in the presence of vascular lesions. It is however re-assuring that the impact of these complications on surgical mortality is insignificant; however, the impact on co-morbidities remains controversial in view of conflicting evidence from different reports[85,86]. In addition, bevacizumab which is commonly used along with chemotherapy back-bone can theoretically increase the risk of thrombo-embolic events including stroke and arterial thromboembolism, bowel perforation and wound healing. Nevertheless, our own experience and those of others using chemotherapy in conjunction with bevacizumab is satisfactory and doesn’t seem to influence the liver regeneration after surgery[61,87]. Furthermore, some retrospective data suggests that addition of bevacizumab to oxaliplatin-based regimen may actually reduce the risk of oxaliplatin induced liver toxicity[88-90]; and (4) we therefore suggest that all the patients should be individually assessed for the risk of potential complications with neo-adjuvant therapy; however, this on its own shouldn’t deter the physicians from adopting the approach of upfront systemic treatment. Because of the half-life is bevacizumab (20 d), at least 3-wk interval between the last dose of chemotherapy and surgery should be considered; thus the last treatment may be offered without bevacizumab. The choice of chemotherapy regimen in relation to toxicities remains controversial and may depend on the local guidelines and physicians discretion.
Post-operative chemotherapy: Post-operative adjuvant chemotherapy has well-defined role in high-risk patients with CRC; however the data for adjuvant therapy in stage IV disease is still limited. Two randomised phase III studies evaluated the role of adjuvant treatment in this setting and their pooled analysis[91] the French FFCD 9002[92] and EORTC trials (n = 278 patients) showed median disease free survival (DFS) of 28 mo vs 19 mo (P = 0.058) and OS of 62 mo vs 47 mo in favour of chemotherapy (P = 0.095). Both trials used the same chemotherapy regimen of 5-FU and leucovorin, administered for five days consecutively in a 4-wk cycle for 6 mo; however, both closed pre-maturely due to poor accrual. The improvement in DFS and OS was deemed statistically insignificant but could represent clinically meaningful advantage.
The major criticism on these trials was the choice of contemporary regimen which can be more toxic and possibly not active enough to reflect any significant advantage. Post-operative treatment with irinotecan in addition to 5-FU in a multicenter phase III trial showed no additional benefit compared to 5-FU alone. Although there was a non-significant 3-mo increase in DFS (24.7 mo vs 21.6 mo), the 3-year survival rates with combination were strikingly similar (73% vs 72%) to single agent therapy[93]. These results however need to be interpreted with caution as at least three previous adjuvant trials utilising FOLFIRI in stage II/III disease have failed to demonstrate any DFS benefit[94-96].
The role of adjuvant chemotherapy in this setting remains controversial partly due to the poor accrual and pre-mature closure of clinical trials designed to address this question; however, based on the benefit demonstrated in several clinical trials in high-risk CRC patients, consideration should be given to adjuvant therapy particularly if the patient gained benefit from neo-adjuvant therapy.
RFA is a local ablative method which can be carried out using an open, laparoscopic or percutaneous technique; the choice of procedure is often operator choice as there is not any current evidence to support a superior approach. Lesions along the inferior edge of the liver and multiple lesions are difficult to treat with RFA. Lesions located close to large vessels (> 1 cm) may be incompletely treated as the proximity of a large vessel means optimal tissue heating cannot be achieved, so called the “heat sink effect”[97]. In these cases other treatment methods may be more appropriate.
RFA is performed under local or general anaesthesia; a high frequency alternating current is applied through needle electrodes which are placed under imaging guidance, either with CT or ultrasound. Heat is generated which results in localised coagulative necrosis and causes destruction of the tumour [National Institute of Clinical Excellence (NICE), radiofrequency ablation for colorectal liver metastases 2009]. RFA is a relatively well-tolerated procedure; however the reported complications range from 6%-9% with mortality of 0%-2%[98]. These include liver abscess, pleural effusions, pneumothorax, acute renal insufficiency and hypoxemia.
The NICE guidelines recommend that RFA is used to treat CLM in patients who are unfit or unsuitable for surgery, for those with recurrent disease or increasingly as an adjunct to hepatic resection as a curative treatment option (NICE, radiofrequency ablation for colorectal liver metastases 2009). In addition RFA may be used repeatedly to achieve local tumour control.
The CLOCC study was a prospective trial which examined the use of RFA in patients with CLM. This randomised phase II study aimed to establish the added benefits of using RFA in addition to chemotherapy in patients with unresectable liver metastases but no extrahepatic disease[99]. One hundred and nineteen patients were recruited in the study; of those 59 patients had chemotherapy alone, 60 had chemotherapy and RFA. Median PFS was 16.8 mo in the RFA + chemotherapy group, compared to 9.9 mo in the chemotherapy alone group (P = 0.025). Thirty-month OS was 61.7% in the RFA + chemotherapy arm, and 57.6% in the chemotherapy alone arm. The authors concluded that the benefit of RFA in the treatment of liver metastases was uncertain and further work was required. Otherwise, the published data on efficacy of RFA in CRC mostly comprises of retrospective series and requires cautious interpretation. Minami et al[100] performed a review of local tumour progression and OS after RFA and found variable local recurrence rates after RFA (ranging from 8.8% to 40%) and described that 5-year OS ranged from 20% to 48.5%. The authors also reviewed the survival rates after RFA in comparison to survival after hepatic resection. For the majority of published papers OS after RFA was significantly less than survival after hepatic resection[25,101,102]. The authors summarised that this difference may be due to the selection bias as RFA was mostly used in patients who are unsuitable for hepatic resection and thus may represent aggressive disease biology.
The variable and high local tumour progression rates may limit the use of RFA. Some authors have therefore tried to identify factors which affect tumour progression; to date blood vessel proximity, the size of the metastasis and the size of the radiofrequency ablation margin have been shown to affect local recurrence rates in small series[24,103,104]. Although further work is required to validate these findings, we suggest that these factors should be carefully considered when offering RFA to the patients with CLM.
SIRT: SIRT with or without chemotherapy can be used to treat patients with CLM alone, or patients with limited extrahepatic disease. Localised high dose radiation is applied to the metastases by the embolisation of small vessels around the metastases. It is indicated for patients who are unsuitable for treatment with surgery or ablation. As with local ablation the treatment can be repeated (NICE IPG401 selective internal radiation therapy for non-resectable colorectal metastases in the liver 2011).
To date only small randomised controlled trials have been performed but some have been able to show improved median survival when SIRT was used with systemic chemotherapy compared to systemic chemotherapy alone[105] and improved tumour response when SIRT was used with hepatic artery chemotherapy (HAC) compared to HAC alone[106].
SIRT may be potentially beneficial for the treatment of unresectable CLM but more research is required to demonstrate its efficacy and to establish the potential use of SIRT in treatment naive liver metastases (NICE, 2011).
HIA CT: HIA chemotherapy with or without systemic therapy can be used to downstage the tumour size or following metastasectomy; based on the biological rationale that liver metastases primarily derive their blood supply from the hepatic artery. Some studies have shown encouraging results with HIA following metastasectomy; however, sample sizes have often been small[107,108]. A large randomised trial aiming to address the question of post-operative HIA was closed pre-maturely due to poor accrual[109], while another study was closed prematurely on grounds of worst outcome[110]. The combination of systemic chemotherapy with HIA has been evaluated in a phase I study[111]. This study recruited 49 patients. Patients were treated with HIA plus systemic oxaliplatin and irinotecan. ORR was 92% with 8% CR and 84% PR. 43% of the patients were able to undergo resection of the tumour, with free margins in 19. Median OS for the entire cohort was 40 mo.
Although these results were promising, and also some other studies utilising this approach with various chemotherapy regimens showed improvement in the rate of hepatic recurrence and recurrence-free survival[112]; OS benefit was not gained with this approach[112,113]. HIA has not so far been evaluated in large randomised clinical trials and is not routinely used in many parts of the world in management of CLM.
Other local ablative methods: Microwave ablation and cryotherapy are other ablative techniques which are used to treat CLM. These also suffer from problems of high local progression rates and the lack of prospective data. Pathak et al[114] performed a systematic review of the use of ablative methods for CLM. They found variable local progression rates and OS for RFA as Minami et al[100]. Twenty six cryotherapy studies were included showing, local progression rates of 12%-39% and mean 5-year overall survival of 17%. Thirteen microwave studies, also showed variable local progression rates (5%-13%) and mean 5- year OS of 16%.
Local ablative techniques are increasingly used for patients with CLM, in both a curative and palliative setting. RFA, cryotherapy and microwave ablation have the potential to improve survival for patients with CLM; however these techniques are hampered by variable local progression rates and survival rates are lower than those for curative resection. Evaluation in large randomised trials is required to further clarify the emerging role of local ablation.
The presence of both synchronous liver and lung metastases doesn’t necessarily represent an absolute contraindication to surgery, as long as complete resection of all metastatic sites is possible. One of the earlier studies reported on the outcome of a series of 165 patients with mCRC with 5 and 10 year survival rates of 39.6% and 37.2% respectively in un-selected population. Those patients who underwent hepatic metastasectomy (n = 26) had 10 year survival of 34.1%; 21 patients had two or three thoracotomies for recurrent CRC and for those the reported 5 year survival was 52.1 mo from the time of second thoracotomy. Patients with hilar or mediastinal lymph nodes had the worst outcome with 5-year survival of 6.2%[115]. Another study including 63 patients with either lung only metastases (group 1, n = 45) or synchronous lung and liver metastases (group 2, n = 18), treated aggressively with surgery showed no significant difference in survival at median follow-up of 24 and 27 mo respectively in the two groups[45]. More recently, Gonzalez et al[116] conducted retrospective analysis of 27 consecutive patients who underwent lung resection after previous hepatic metastasectomy for CRC. They reported 3 and 5-year survivals of 56% and 39% respectively with a median survival of 46 mo. Likewise, another recent study reported on the outcome of sixty-five patients who underwent surgery for liver and lung metastases from CRC. In 33 cases, the first distant metastasis was diagnosed synchronously with the primary tumour; for the remaining, median time interval between primary tumour and first distant metastasis was 18 mo (5-69 mo). Five- and 10-year survival rates for all patients were 57% and 15% from diagnosis of the primary tumour, 37% and 14% from resection of the first metastasis and 20% and 15% from resection of the second metastasis. After complete resection, 5- and 10-year survival rates improved to 61% and 18%, 43% and 17%, and 25% and 19%, in the three scenarios respectively. Negative margins (P = 0.002), the absence of pulmonary involvement in synchronous metastases (P = 0.0003) and single metastases in both organs (P = 0.036) were associated with a better prognosis[117]. Some other studies also illustrated the importance of offering surgical resection in this context, where possible[118].
Although these data are largely based on retrospective experience of various institutes, considering the difficulty in designing prospective studies to evaluate outcome of patients with limited extra-hepatic disease in the presence of CLM, and based on our own experience[119,120], we feel that aggressive surgical management of CLM patients should still be considered where synchronous or metachronous limited extra-hepatic disease can be potentially resected.
Up to 70% of the patients, who had hepatic resection for CLM, could present with recurrent disease and the most frequent site of recurrence is liver[19,121,122]. In the presence of controlled primary disease, recurrent hepatic metastases could be a result of the development of occult tumours not detected at the time of initial resection or with more recent evidence, could represent metastases from the previous hepatic secondary[123,124]. Lung is the second most common site of metastatic disease from CRC; however, the frequency of isolated lung metastases with CRC is 1%-5%. Lung metastases tend to be more commonly associated with rectal rather than colon primary[125]. This perhaps suggests that majority of pulmonary metastatic disease grows from the CLM[126,127].
Regardless of the pathophysiology, re-resection of oligo-metastatic disease, where possible has been the only treatment modality that has shown improvement in long-term survival[46,128-130]; in the absence of randomised controlled trials, these data are valuable and demonstrate the safety of the aggressive surgical approach[131-136]. There is however some conflicting evidence questioning the efficacy of re-resection[137]; nevertheless, most reported series show promise with achievement of better long-term survival[138,139].
We suggest offering surgical resection in the event of recurrence; however, careful consideration should be given to the disease biology, relapse-free interval from the last resection, extent of metastatic disease and co-morbidity status of the patients.
The complex stepwise process of CRC tumorigenesis was first proposed by Vogelstein two decades ago[140]. Over the years, our understanding about the heterogeneity in CRC tumorigenesis has improved exponentially. Traditionally, CRC is biologically divided into those with microsatellite instability (MSI) which is frequently associated with CpG island methylator phenotype and hyper-mutation and those with microsatellite stable but chromosomally unstable disease[141]. Other investigators[142,143] unfolded the critically important genes and pathways important to CRC tumorigenesis; those included WNT, RAS-RAF-MAP, P53 and DNA mismatch-repair pathways[144]. In 2012, the cancer genome atlas network published results of molecular characterization performed in CRC[145]. Genome-scale analysis comprising of whole exome sequencing, DNA copy number, promoter methylation, messenger RNA and microRNA expression were performed on 276 samples with CRC. This consortium highlighted the complex molecular biology of CRC; 16% of CRC were found to have hypermutation (75% with expected high MSI and 25% with somatic mis-match repair gene). Excluding the hypermutated patients, 24 genes were mutated in other patients; of those APC, TP53, SMAD4, PIK3CA were previously established relevant mutations in the context of CRC. Additionally, ARID1A, SOX9 and FAM123B were also found to be frequent mutations, which were not previously defined as common mutations in CRC. WNT signalling pathway was found to be altered in 93% of all tumours[145]. All these data highlight the genetic complexity of CRC and associated significant challenges in management of this disease. At the same time, this however, allows a wide window of opportunity for further research in order to form effective and more robust strategies in targeting these pathways.
Intra-tumour heterogeneity is a well-established phenomenon which can contribute to treatment failures and drug resistance in various malignancies[123,146-151]. Other important consideration is the heterogeneity of mutations between the primary and the secondary tumours. This is particularly important in the context of CRC, as treatment plan could vary significantly based on the RAS status of the tumour. Several studies have examined the heterogeneity of CRC and any existence of discordance in the mutations between primary and secondary tumour with variable results. Baldus et al[152] found that heterogeneity between primary and secondary tumours in CRC samples was present in 10% for KRAS and 5% with PIK3CA mutations. A large systemic review included 21 studies, examining the concordance of KRAS between the primary CRC and metastases. This study found an overall concordance rate of 93% (range 76%-100%), 98% and 68% in KRAS, BRAF and PTEN loss respectively[153]. It is however noteworthy that KRAS mutation has always been considered as one of the early events in CRC tumorigenesis[140,154]; nevertheless, the small rates of discordance may have an impact on the choice of treatment as KRAS/RAS is currently the only validated biomarker in management of CRC. The molecular profiling of primary and metastatic disease in modern era will perhaps further unfold the late events causing heterogeneity of the primary tumour followed by progression of one clone, resulting in metastases[142]. Given that CLM patients can potentially live longer than they used to live a decade ago, the development of late mutations leading to drug resistance will become more relevant in the context of systemic management of CLM.
A variety of drugs are under evaluation in management of CRC. They can be broadly divided into growth factor receptor inhibitors targeting EGFR and VEGF pathways, RAS pathway inhibitors and PI3K-AKT-mTOR pathway inhibitors. There is also growing interest in targeting WNT pathway due to the frequent aberration of this pathway in CRC. At the same time, there remain challenges in aberrant compensatory pathways, which could lead to resistance to these targeted therapies. Additionally, the druggability of these targets is yet under evaluation. The two new agents approved by United States Food and Drug Administration are regorafenib and aflibercept[155-158]. These new drugs are likely to be investigated in clinical trial setting in the management of CLM, particularly in patients with disease, which is deemed borderline or un-resectable at the outset. There are several other new drugs under evaluation in phase I setting as single agent and as combinations[159-161]; these may become part of armoury of CRC management in future.
CRC is one of the commonest cancers with high incidence and mortality associated with metastatic disease. Vast majority of patients with metastatic disease may present with CLM; of those some have CLM alone or in the presence of limited extra-hepatic disease. Surgical resection of CLM, where possible remains the most effective treatment to achieve long-term cure; however, optimal patient selection with utility of available imaging techniques and comprehensive multi-disciplinary involvement is paramount to avoiding un-necessary surgical complications and achieving long-term cure.
Where R0 resection is possible, perioperative chemotherapy should be considered for three months preoperativey followed by another three months of postoperative chemotherapy.
For patients where R0 resection is not possible, aggressive preoperative chemotherapy approach with EGFR or VEGFR targeted therapy along with chemotherapy backbone of FOLFOX or FOLFIRI should be considered. Where possible, either in the context of clinical trials or local availability of drugs, triplet chemotherapy should be encouraged as this can potentially increase the chances of resectability by optimising the response to the treatment. For patients who present with synchronous CLM, the optimal timing for resection of primary and the metastatic disease is uncertain. One-stage surgery with resection of both primary and the secondary can be considered, where surgical expertise allow that; otherwise resection of CLM could be performed 6-8 wk after resection of the primary or vice versa. The role of localised treatments as an adjunct to systemic therapy and surgery is rather uncertain but these approaches can be considered on case-to-case basis.
P- Reviewer: Chen Y, Maurel J, Plataniotis G S- Editor: Gou SX L- Editor: A E- Editor: Liu XM
| 1. | Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9215] [Cited by in RCA: 9856] [Article Influence: 821.3] [Reference Citation Analysis (4)] |
| 2. | Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893-2917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11128] [Cited by in RCA: 11837] [Article Influence: 845.5] [Reference Citation Analysis (4)] |
| 3. | Compton CC. Colorectal carcinoma: diagnostic, prognostic, and molecular features. Mod Pathol. 2003;16:376-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 278] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 4. | Meyerhardt JA, Mayer RJ. Systemic therapy for colorectal cancer. N Engl J Med. 2005;352:476-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 859] [Cited by in RCA: 859] [Article Influence: 43.0] [Reference Citation Analysis (0)] |
| 5. | Masi G, Vasile E, Loupakis F, Cupini S, Fornaro L, Baldi G, Salvatore L, Cremolini C, Stasi I, Brunetti I. Randomized trial of two induction chemotherapy regimens in metastatic colorectal cancer: an updated analysis. J Natl Cancer Inst. 2011;103:21-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 139] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 6. | O'Neil BH, Goldberg RM. Innovations in chemotherapy for metastatic colorectal cancer: an update of recent clinical trials. Oncologist. 2008;13:1074-1083. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 7. | Sanoff HK, Sargent DJ, Campbell ME, Morton RF, Fuchs CS, Ramanathan RK, Williamson SK, Findlay BP, Pitot HC, Goldberg RM. Five-year data and prognostic factor analysis of oxaliplatin and irinotecan combinations for advanced colorectal cancer: N9741. J Clin Oncol. 2008;26:5721-5727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 239] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 8. | Cunningham D, Atkin W, Lenz HJ, Lynch HT, Minsky B, Nordlinger B, Starling N. Colorectal cancer. Lancet. 2010;375:1030-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1208] [Cited by in RCA: 1178] [Article Influence: 78.5] [Reference Citation Analysis (0)] |
| 9. | Alfredo Falcone CC, Gianluca Masi, Sara Lonardi, Vittorina Zagonel, Lisa Salvatore, Patrizia Trenta, Gianluca Tomasello, Monica Ronzoni, Libero Ciuffreda, Alberto Zaniboni. FOLFOXIRI/bevacizumab (bev) vs FOLFIRI/bev as first-line treatment in unresectable metastatic colorectal cancer (mCRC) patients (pts): Results of the phase III TRIBE trial by GONO group. Chicago, IL: ASCO 2013; . |
| 10. | Heinemann LFvW V, Decker T, Kiani A, Vehling-Kaiser U, Al Batran S, Heintges T, Lerchenmüller C, Kahl C, Seipelt G, Kullmann F, Stauch M, Scheithauer W, Hielscher J, Scholz M, Müller S, Schaefer B, Modest DP, Jung A, Stintzing S. Randomized comparison of FOLFIRI plus cetuximab vs FOLFIRI plus bevacizumab as first-line treatment of KRAS wild-type metastatic colorectal cancer. German AIO study KRK-0306: (FIRE-3) 2013; . |
| 11. | Faivre J, Manfredi S, Bouvier AM. [Epidemiology of colorectal cancer liver metastases]. Bull Acad Natl Med. 2003;187:815-822; discussion 822-823. [PubMed] |
| 12. | van der Pool AE, Damhuis RA, Ijzermans JN, de Wilt JH, Eggermont AM, Kranse R, Verhoef C. Trends in incidence, treatment and survival of patients with stage IV colorectal cancer: a population-based series. Colorectal Dis. 2012;14:56-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 177] [Article Influence: 13.6] [Reference Citation Analysis (2)] |
| 13. | Adam R, Delvart V, Pascal G, Valeanu A, Castaing D, Azoulay D, Giacchetti S, Paule B, Kunstlinger F, Ghémard O. Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: a model to predict long-term survival. Ann Surg. 2004;240:644-657; discussion 657-658. [PubMed] |
| 14. | Adam R, Wicherts DA, de Haas RJ, Ciacio O, Lévi F, Paule B, Ducreux M, Azoulay D, Bismuth H, Castaing D. Patients with initially unresectable colorectal liver metastases: is there a possibility of cure? J Clin Oncol. 2009;27:1829-1835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 422] [Cited by in RCA: 410] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 15. | Adam R, Aloia TA. Is hepatic resection justified after chemotherapy in patients with colorectal liver metastases and lymph node involvement? J Clin Oncol. 2009;27:1343-1345; author reply 1345. [PubMed] |
| 16. | Folprecht G, Grothey A, Alberts S, Raab HR, Köhne CH. Neoadjuvant treatment of unresectable colorectal liver metastases: correlation between tumour response and resection rates. Ann Oncol. 2005;16:1311-1319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 480] [Cited by in RCA: 471] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 17. | Folprecht G, Bechstein WO. [Neoadjuvant therapy concepts for liver metastases]. Chirurg. 2011;82:989-994. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 18. | Tomlinson JS, Jarnagin WR, DeMatteo RP, Fong Y, Kornprat P, Gonen M, Kemeny N, Brennan MF, Blumgart LH, D’Angelica M. Actual 10-year survival after resection of colorectal liver metastases defines cure. J Clin Oncol. 2007;25:4575-4580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 845] [Cited by in RCA: 895] [Article Influence: 49.7] [Reference Citation Analysis (0)] |
| 19. | Hughes KS, Simon R, Songhorabodi S, Adson MA, Ilstrup DM, Fortner JG, Maclean BJ, Foster JH, Daly JM, Fitzherbert D. Resection of the liver for colorectal carcinoma metastases: a multi-institutional study of patterns of recurrence. Surgery. 1986;100:278-284. [PubMed] |
| 20. | Scheele J, Stangl R, Schmidt K, Altendorf-Hofmann A. [Recurrent tumor after R0 resection of colorectal liver metastases. Incidence, resectability and prognosis]. Chirurg. 1995;66:965-973. [PubMed] |
| 21. | Nordlinger B, Guiguet M, Vaillant JC, Balladur P, Boudjema K, Bachellier P, Jaeck D. Surgical resection of colorectal carcinoma metastases to the liver. A prognostic scoring system to improve case selection, based on 1568 patients. Association Française de Chirurgie. Cancer. 1996;77:1254-1262. [PubMed] |
| 22. | Jamison RL, Donohue JH, Nagorney DM, Rosen CB, Harmsen WS, Ilstrup DM. Hepatic resection for metastatic colorectal cancer results in cure for some patients. Arch Surg. 1997;132:505-510; discussion 511. [PubMed] |
| 23. | Iwatsuki S, Dvorchik I, Madariaga JR, Marsh JW, Dodson F, Bonham AC, Geller DA, Gayowski TJ, Fung JJ, Starzl TE. Hepatic resection for metastatic colorectal adenocarcinoma: a proposal of a prognostic scoring system. J Am Coll Surg. 1999;189:291-299. [PubMed] |
| 24. | Choti MA, Sitzmann JV, Tiburi MF, Sumetchotimetha W, Rangsin R, Schulick RD, Lillemoe KD, Yeo CJ, Cameron JL. Trends in long-term survival following liver resection for hepatic colorectal metastases. Ann Surg. 2002;235:759-766. [PubMed] |
| 25. | Abdalla EK, Vauthey JN, Ellis LM, Ellis V, Pollock R, Broglio KR, Hess K, Curley SA. Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg. 2004;239:818-825; discussion 825-827. [PubMed] |
| 26. | Wei AC, Greig PD, Grant D, Taylor B, Langer B, Gallinger S. Survival after hepatic resection for colorectal metastases: a 10-year experience. Ann Surg Oncol. 2006;13:668-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 269] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 27. | Rees M, Tekkis PP, Welsh FK, O’Rourke T, John TG. Evaluation of long-term survival after hepatic resection for metastatic colorectal cancer: a multifactorial model of 929 patients. Ann Surg. 2008;247:125-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 777] [Cited by in RCA: 810] [Article Influence: 47.6] [Reference Citation Analysis (0)] |
| 28. | de Jong MC, Pulitano C, Ribero D, Strub J, Mentha G, Schulick RD, Choti MA, Aldrighetti L, Capussotti L, Pawlik TM. Rates and patterns of recurrence following curative intent surgery for colorectal liver metastasis: an international multi-institutional analysis of 1669 patients. Ann Surg. 2009;250:440-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 534] [Cited by in RCA: 590] [Article Influence: 36.9] [Reference Citation Analysis (0)] |
| 29. | Morris EJ, Forman D, Thomas JD, Quirke P, Taylor EF, Fairley L, Cottier B, Poston G. Surgical management and outcomes of colorectal cancer liver metastases. Br J Surg. 2010;97:1110-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 294] [Cited by in RCA: 281] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 30. | Fernandez FG, Drebin JA, Linehan DC, Dehdashti F, Siegel BA, Strasberg SM. Five-year survival after resection of hepatic metastases from colorectal cancer in patients screened by positron emission tomography with F-18 fluorodeoxyglucose (FDG-PET). Ann Surg. 2004;240:438-447; discussion 447-450. [PubMed] |
| 31. | Cummings LC, Payes JD, Cooper GS. Survival after hepatic resection in metastatic colorectal cancer: a population-based study. Cancer. 2007;109:718-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 242] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 32. | Folprecht G, Gruenberger T, Bechstein WO, Raab HR, Lordick F, Hartmann JT, Lang H, Frilling A, Stoehlmacher J, Weitz J. Tumour response and secondary resectability of colorectal liver metastases following neoadjuvant chemotherapy with cetuximab: the CELIM randomised phase 2 trial. Lancet Oncol. 2010;11:38-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 704] [Cited by in RCA: 712] [Article Influence: 44.5] [Reference Citation Analysis (0)] |
| 33. | Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg. 1999;230:309-318; discussion 318-321. [PubMed] |
| 34. | Feroci F, Fong Y. Use of clinical score to stage and predict outcome of hepatic resection of metastatic colorectal cancer. J Surg Oncol. 2010;102:914-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 35. | Maithel SK, Gönen M, Ito H, Dematteo RP, Allen PJ, Fong Y, Blumgart LH, Jarnagin WR, D’Angelica MI. Improving the clinical risk score: an analysis of molecular biomarkers in the era of modern chemotherapy for resectable hepatic colorectal cancer metastases. Surgery. 2012;151:162-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 36. | Yamaguchi T, Mori T, Takahashi K, Matsumoto H, Miyamoto H, Kato T. A new classification system for liver metastases from colorectal cancer in Japanese multicenter analysis. Hepatogastroenterology. 2008;55:173-178. [PubMed] |
| 37. | Malik HZ, Hamady ZZ, Adair R, Finch R, Al-Mukhtar A, Toogood GJ, Prasad KR, Lodge JP. Prognostic influence of multiple hepatic metastases from colorectal cancer. Eur J Surg Oncol. 2007;33:468-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 38. | Bolton JS, Fuhrman GM. Survival after resection of multiple bilobar hepatic metastases from colorectal carcinoma. Ann Surg. 2000;231:743-751. [PubMed] |
| 39. | Weber SM, Jarnagin WR, DeMatteo RP, Blumgart LH, Fong Y. Survival after resection of multiple hepatic colorectal metastases. Ann Surg Oncol. 2000;7:643-650. [PubMed] |
| 40. | Pawlik TM, Abdalla EK, Ellis LM, Vauthey JN, Curley SA. Debunking dogma: surgery for four or more colorectal liver metastases is justified. J Gastrointest Surg. 2006;10:240-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 74] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 41. | Kornprat P, Jarnagin WR, Gonen M, DeMatteo RP, Fong Y, Blumgart LH, D’Angelica M. Outcome after hepatectomy for multiple (four or more) colorectal metastases in the era of effective chemotherapy. Ann Surg Oncol. 2007;14:1151-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 114] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 42. | Brouquet A, Abdalla EK, Kopetz S, Garrett CR, Overman MJ, Eng C, Andreou A, Loyer EM, Madoff DC, Curley SA. High survival rate after two-stage resection of advanced colorectal liver metastases: response-based selection and complete resection define outcome. J Clin Oncol. 2011;29:1083-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 276] [Cited by in RCA: 303] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 43. | Pulitanò C, Bodingbauer M, Aldrighetti L, Choti MA, Castillo F, Schulick RD, Gruenberger T, Pawlik TM. Colorectal liver metastasis in the setting of lymph node metastasis: defining the benefit of surgical resection. Ann Surg Oncol. 2012;19:435-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 44. | Oussoultzoglou E, Romain B, Panaro F, Rosso E, Pessaux P, Bachellier P, Jaeck D. Long-term survival after liver resection for colorectal liver metastases in patients with hepatic pedicle lymph nodes involvement in the era of new chemotherapy regimens. Ann Surg. 2009;249:879-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 45. | Patel NA, Keenan RJ, Medich DS, Woo Y, Celebrezze J, Santucci T, Maley R, Landreneau RL, Roh MS. The presence of colorectal hepatic metastases does not preclude pulmonary metastasectomy. Am Surg. 2003;69:1047-1053; discussion 1053. [PubMed] |
| 46. | Mise Y, Imamura H, Hashimoto T, Seyama Y, Aoki T, Hasegawa K, Beck Y, Sugawara Y, Makuuchi M, Nakajima J. Cohort study of the survival benefit of resection for recurrent hepatic and/or pulmonary metastases after primary hepatectomy for colorectal metastases. Ann Surg. 2010;251:902-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 47. | Gonzalez M, Poncet A, Combescure C, Robert J, Ris HB, Gervaz P. Risk factors for survival after lung metastasectomy in colorectal cancer patients: a systematic review and meta-analysis. Ann Surg Oncol. 2013;20:572-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 290] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 48. | Kong G, Jackson C, Koh DM, Lewington V, Sharma B, Brown G, Cunningham D, Cook GJ. The use of 18F-FDG PET/CT in colorectal liver metastases--comparison with CT and liver MRI. Eur J Nucl Med Mol Imaging. 2008;35:1323-1329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 96] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 49. | Niekel MC, Bipat S, Stoker J. Diagnostic imaging of colorectal liver metastases with CT, MR imaging, FDG PET, and/or FDG PET/CT: a meta-analysis of prospective studies including patients who have not previously undergone treatment. Radiology. 2010;257:674-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 393] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 50. | Koh DM, Scurr E, Collins D, Kanber B, Norman A, Leach MO, Husband JE. Predicting response of colorectal hepatic metastasis: value of pretreatment apparent diffusion coefficients. AJR Am J Roentgenol. 2007;188:1001-1008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 284] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 51. | Cui Y, Zhang XP, Sun YS, Tang L, Shen L. Apparent diffusion coefficient: potential imaging biomarker for prediction and early detection of response to chemotherapy in hepatic metastases. Radiology. 2008;248:894-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 294] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 52. | Tam HH, Collins DJ, Brown G, Chau I, Cunningham D, Leach MO, Koh DM. The role of pre-treatment diffusion-weighted MRI in predicting long-term outcome of colorectal liver metastasis. Br J Radiol. 2013;86:20130281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 53. | Wiering B, Krabbe PF, Jager GJ, Oyen WJ, Ruers TJ. The impact of fluor-18-deoxyglucose-positron emission tomography in the management of colorectal liver metastases. Cancer. 2005;104:2658-2670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 173] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 54. | Engledow AH, Skipworth JR, Pakzad F, Imber C, Ell PJ, Groves AM. The role of 18FDG PET/CT in the management of colorectal liver metastases. HPB (Oxford). 2012;14:20-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 55. | Jones RP, Malik HZ, Fenwick SW, Poston GJ. Perioperative chemotherapy for resectable colorectal liver metastases: where now? Eur J Surg Oncol. 2013;39:807-811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 56. | Nordlinger B, Sorbye H, Glimelius B, Poston GJ, Schlag PM, Rougier P, Bechstein WO, Primrose JN, Walpole ET, Finch-Jones M. Perioperative chemotherapy with FOLFOX4 and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC Intergroup trial 40983): a randomised controlled trial. Lancet. 2008;371:1007-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1478] [Cited by in RCA: 1444] [Article Influence: 84.9] [Reference Citation Analysis (0)] |
| 57. | Zhu D, Zhong Y, Wei Y, Ye L, Lin Q, Ren L, Ye Q, Liu T, Xu J, Qin X. Effect of neoadjuvant chemotherapy in patients with resectable colorectal liver metastases. PLoS One. 2014;9:e86543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 58. | Ott R, Wein A, Hohenberger W. [Liver metastases--primary or multimodal therapy?]. Chirurg. 2001;72:887-897. [PubMed] |
| 59. | Delaunoit T, Alberts SR, Sargent DJ, Green E, Goldberg RM, Krook J, Fuchs C, Ramanathan RK, Williamson SK, Morton RF. Chemotherapy permits resection of metastatic colorectal cancer: experience from Intergroup N9741. Ann Oncol. 2005;16:425-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 120] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 60. | Barone C, Nuzzo G, Cassano A, Basso M, Schinzari G, Giuliante F, D’Argento E, Trigila N, Astone A, Pozzo C. Final analysis of colorectal cancer patients treated with irinotecan and 5-fluorouracil plus folinic acid neoadjuvant chemotherapy for unresectable liver metastases. Br J Cancer. 2007;97:1035-1039. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 92] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 61. | Wong R, Cunningham D, Barbachano Y, Saffery C, Valle J, Hickish T, Mudan S, Brown G, Khan A, Wotherspoon A. A multicentre study of capecitabine, oxaliplatin plus bevacizumab as perioperative treatment of patients with poor-risk colorectal liver-only metastases not selected for upfront resection. Ann Oncol. 2011;22:2042-2048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 156] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 62. | Ye LC, Liu TS, Ren L, Wei Y, Zhu DX, Zai SY, Ye QH, Yu Y, Xu B, Qin XY. Randomized controlled trial of cetuximab plus chemotherapy for patients with KRAS wild-type unresectable colorectal liver-limited metastases. J Clin Oncol. 2013;31:1931-1938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 312] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 64. | Maughan TS, Adams RA, Smith CG, Meade AM, Seymour MT, Wilson RH, Idziaszczyk S, Harris R, Fisher D, Kenny SL. Addition of cetuximab to oxaliplatin-based first-line combination chemotherapy for treatment of advanced colorectal cancer: results of the randomised phase 3 MRC COIN trial. Lancet. 2011;377:2103-2114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 766] [Cited by in RCA: 763] [Article Influence: 54.5] [Reference Citation Analysis (2)] |
| 65. | Falcone A, Ricci S, Brunetti I, Pfanner E, Allegrini G, Barbara C, Crinò L, Benedetti G, Evangelista W, Fanchini L. Phase III trial of infusional fluorouracil, leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) compared with infusional fluorouracil, leucovorin, and irinotecan (FOLFIRI) as first-line treatment for metastatic colorectal cancer: the Gruppo Oncologico Nord Ovest. J Clin Oncol. 2007;25:1670-1676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 830] [Cited by in RCA: 888] [Article Influence: 49.3] [Reference Citation Analysis (0)] |
| 66. | Masi G, Loupakis F, Pollina L, Vasile E, Cupini S, Ricci S, Brunetti IM, Ferraldeschi R, Naso G, Filipponi F. Long-term outcome of initially unresectable metastatic colorectal cancer patients treated with 5-fluorouracil/leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) followed by radical surgery of metastases. Ann Surg. 2009;249:420-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 177] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 67. | Masi G, Loupakis F, Salvatore L, Fornaro L, Cremolini C, Cupini S, Ciarlo A, Del Monte F, Cortesi E, Amoroso D. Bevacizumab with FOLFOXIRI (irinotecan, oxaliplatin, fluorouracil, and folinate) as first-line treatment for metastatic colorectal cancer: a phase 2 trial. Lancet Oncol. 2010;11:845-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 202] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 68. | Lisa Salvatore FL, Chiara Cremolini, Marta Schirripa, Gianluca Masi, Carlotta Antoniotti, Lorenzo Fornaro, Elisa Sensi, Cristiana Lupi, Francesca Bergamo, Sara Lonardi, Vittorina Zagonel, Gabriella Fontanini, Alfredo Falcone. FOLFOXIRI plus bevacizumab as first-line treatment of BRAF-mutant metastatic colorectal cancer patients. San Francisco: ASCO 2013; . |
| 69. | Fornaro L, Lonardi S, Masi G, Loupakis F, Bergamo F, Salvatore L, Cremolini C, Schirripa M, Vivaldi C, Aprile G. FOLFOXIRI in combination with panitumumab as first-line treatment in quadruple wild-type (KRAS, NRAS, HRAS, BRAF) metastatic colorectal cancer patients: a phase II trial by the Gruppo Oncologico Nord Ovest (GONO). Ann Oncol. 2013;24:2062-2067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 79] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 70. | Vogt P, Raab R, Ringe B, Pichlmayr R. Resection of synchronous liver metastases from colorectal cancer. World J Surg. 1991;15:62-67. [PubMed] |
| 71. | Lambert LA, Colacchio TA, Barth RJ. Interval hepatic resection of colorectal metastases improves patient selection*. Curr Surg. 2000;57:504. [PubMed] |
| 72. | Stein A, Schmoll HJ. Systemic treatment of liver metastases from colorectal cancer. Ther Adv Med Oncol. 2013;5:193-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 73. | Benoist S, Brouquet A, Penna C, Julié C, El Hajjam M, Chagnon S, Mitry E, Rougier P, Nordlinger B. Complete response of colorectal liver metastases after chemotherapy: does it mean cure? J Clin Oncol. 2006;24:3939-3945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 385] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 74. | Elias D, Goere D, Boige V, Kohneh-Sharhi N, Malka D, Tomasic G, Dromain C, Ducreux M. Outcome of posthepatectomy-missing colorectal liver metastases after complete response to chemotherapy: impact of adjuvant intra-arterial hepatic oxaliplatin. Ann Surg Oncol. 2007;14:3188-3194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 95] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 75. | Fiorentini G, Del Conte A, De Simone M, Guadagni S, Mambrini A, D’Alessandro M, Aliberti C, Rossi G, Cantore M. Complete response of colorectal liver metastases after intra-arterial chemotherapy. Tumori. 2008;94:489-492. [PubMed] |
| 76. | Tanaka K, Takakura H, Takeda K, Matsuo K, Nagano Y, Endo I. Importance of complete pathologic response to prehepatectomy chemotherapy in treating colorectal cancer metastases. Ann Surg. 2009;250:935-942. [PubMed] |
| 77. | Auer RC, White RR, Kemeny NE, Schwartz LH, Shia J, Blumgart LH, Dematteo RP, Fong Y, Jarnagin WR, D’Angelica MI. Predictors of a true complete response among disappearing liver metastases from colorectal cancer after chemotherapy. Cancer. 2010;116:1502-1509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 138] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 78. | Carpenter S, Fong Y. Management of disappearing colorectal hepatic metastases. Adv Surg. 2010;44:269-279. [PubMed] |
| 79. | van Vledder MG, de Jong MC, Pawlik TM, Schulick RD, Diaz LA, Choti MA. Disappearing colorectal liver metastases after chemotherapy: should we be concerned? J Gastrointest Surg. 2010;14:1691-1700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 98] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 80. | Ono T, Ishida H, Kumamoto K, Okada N, Ishibashi K. Outcome in disappearing colorectal cancer liver metastases during oxaliplatin-based chemotherapy. Oncol Lett. 2012;4:905-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 81. | Ferrero A, Langella S, Russolillo N, Vigano’ L, Lo Tesoriere R, Capussotti L. Intraoperative detection of disappearing colorectal liver metastases as a predictor of residual disease. J Gastrointest Surg. 2012;16:806-814. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 82. | Gaujoux S, Goéré D, Dumont F, Souadka A, Dromain C, Ducreux M, Elias D. Complete radiological response of colorectal liver metastases after chemotherapy: what can we expect? Dig Surg. 2011;28:114-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 83. | Bischof DA, Clary BM, Maithel SK, Pawlik TM. Surgical management of disappearing colorectal liver metastases. Br J Surg. 2013;100:1414-1420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 84. | Nordlinger B, Van Cutsem E, Gruenberger T, Glimelius B, Poston G, Rougier P, Sobrero A, Ychou M. Combination of surgery and chemotherapy and the role of targeted agents in the treatment of patients with colorectal liver metastases: recommendations from an expert panel. Ann Oncol. 2009;20:985-992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 267] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 85. | Vauthey JN, Pawlik TM, Ribero D, Wu TT, Zorzi D, Hoff PM, Xiong HQ, Eng C, Lauwers GY, Mino-Kenudson M. Chemotherapy regimen predicts steatohepatitis and an increase in 90-day mortality after surgery for hepatic colorectal metastases. J Clin Oncol. 2006;24:2065-2072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 970] [Cited by in RCA: 953] [Article Influence: 50.2] [Reference Citation Analysis (0)] |
| 86. | Nakano H, Oussoultzoglou E, Rosso E, Casnedi S, Chenard-Neu MP, Dufour P, Bachellier P, Jaeck D. Sinusoidal injury increases morbidity after major hepatectomy in patients with colorectal liver metastases receiving preoperative chemotherapy. Ann Surg. 2008;247:118-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 367] [Cited by in RCA: 351] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 87. | Gruenberger B, Tamandl D, Schueller J, Scheithauer W, Zielinski C, Herbst F, Gruenberger T. Bevacizumab, capecitabine, and oxaliplatin as neoadjuvant therapy for patients with potentially curable metastatic colorectal cancer. J Clin Oncol. 2008;26:1830-1835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 318] [Cited by in RCA: 340] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 88. | Robinson SM, Wilson CH, Burt AD, Manas DM, White SA. Chemotherapy-associated liver injury in patients with colorectal liver metastases: a systematic review and meta-analysis. Ann Surg Oncol. 2012;19:4287-4299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 158] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 89. | van der Pool AE, Marsman HA, Verheij J, Ten Kate FJ, Eggermont AM, Ijzermans JN, Verhoef C. Effect of bevacizumab added preoperatively to oxaliplatin on liver injury and complications after resection of colorectal liver metastases. J Surg Oncol. 2012;106:892-897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 90. | Ribero D, Wang H, Donadon M, Zorzi D, Thomas MB, Eng C, Chang DZ, Curley SA, Abdalla EK, Ellis LM. Bevacizumab improves pathologic response and protects against hepatic injury in patients treated with oxaliplatin-based chemotherapy for colorectal liver metastases. Cancer. 2007;110:2761-2767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 281] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 91. | Mitry E, Fields AL, Bleiberg H, Labianca R, Portier G, Tu D, Nitti D, Torri V, Elias D, O’Callaghan C. Adjuvant chemotherapy after potentially curative resection of metastases from colorectal cancer: a pooled analysis of two randomized trials. J Clin Oncol. 2008;26:4906-4911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 407] [Cited by in RCA: 411] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 92. | Portier G, Elias D, Bouche O, Rougier P, Bosset JF, Saric J, Belghiti J, Piedbois P, Guimbaud R, Nordlinger B. Multicenter randomized trial of adjuvant fluorouracil and folinic acid compared with surgery alone after resection of colorectal liver metastases: FFCD ACHBTH AURC 9002 trial. J Clin Oncol. 2006;24:4976-4982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 414] [Cited by in RCA: 414] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 93. | Ychou M, Hohenberger W, Thezenas S, Navarro M, Maurel J, Bokemeyer C, Shacham-Shmueli E, Rivera F, Kwok-Keung Choi C, Santoro A. A randomized phase III study comparing adjuvant 5-fluorouracil/folinic acid with FOLFIRI in patients following complete resection of liver metastases from colorectal cancer. Ann Oncol. 2009;20:1964-1970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 196] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 94. | Ychou M, Raoul JL, Douillard JY, Gourgou-Bourgade S, Bugat R, Mineur L, Viret F, Becouarn Y, Bouché O, Gamelin E. A phase III randomised trial of LV5FU2 + irinotecan versus LV5FU2 alone in adjuvant high-risk colon cancer (FNCLCC Accord02/FFCD9802). Ann Oncol. 2009;20:674-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 140] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 95. | Van Cutsem E, Labianca R, Bodoky G, Barone C, Aranda E, Nordlinger B, Topham C, Tabernero J, André T, Sobrero AF. Randomized phase III trial comparing biweekly infusional fluorouracil/leucovorin alone or with irinotecan in the adjuvant treatment of stage III colon cancer: PETACC-3. J Clin Oncol. 2009;27:3117-3125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 351] [Cited by in RCA: 331] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 96. | Saltz LB, Niedzwiecki D, Hollis D, Goldberg RM, Hantel A, Thomas JP, Fields AL, Mayer RJ. Irinotecan fluorouracil plus leucovorin is not superior to fluorouracil plus leucovorin alone as adjuvant treatment for stage III colon cancer: results of CALGB 89803. J Clin Oncol. 2007;25:3456-3461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 331] [Cited by in RCA: 314] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 97. | Goldberg SN, Grassi CJ, Cardella JF, Charboneau JW, Dodd GD, Dupuy DE, Gervais DA, Gillams AR, Kane RA, Lee FT. Image-guided tumor ablation: standardization of terminology and reporting criteria. J Vasc Interv Radiol. 2009;20:S377-S390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 338] [Cited by in RCA: 325] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 98. | Wong SL, Mangu PB, Choti MA, Crocenzi TS, Dodd GD, Dorfman GS, Eng C, Fong Y, Giusti AF, Lu D. American Society of Clinical Oncology 2009 clinical evidence review on radiofrequency ablation of hepatic metastases from colorectal cancer. J Clin Oncol. 2010;28:493-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 318] [Cited by in RCA: 304] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 99. | Ruers TPC, Coevorden F, Rinkes B, Ledermann G, Poston G, Bechstein W, Lentz M, Nordlinger B. Final results of the EORTC intergroup randomized study 40004 (CLOCC) evaluating the benefit of radiofrequency ablation (RFA) combined with chemotherapy for unresectable colorectal liver metastases (CRC LM). ASCO. Chicago 2010. J Clin Oncol. 2010;28:15s. |
| 100. | Minami Y, Kudo M. Radiofrequency ablation of liver metastases from colorectal cancer: a literature review. Gut Liver. 2013;7:1-6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 62] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 101. | Park IJ, Kim HC, Yu CS, Kim PN, Won HJ, Kim JC. Radiofrequency ablation for metachronous liver metastasis from colorectal cancer after curative surgery. Ann Surg Oncol. 2008;15:227-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 74] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 102. | Aloia TA, Vauthey JN, Loyer EM, Ribero D, Pawlik TM, Wei SH, Curley SA, Zorzi D, Abdalla EK. Solitary colorectal liver metastasis: resection determines outcome. Arch Surg. 2006;141:460-466; discussion 466-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 326] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 103. | Ambiru S, Miyazaki M, Isono T, Ito H, Nakagawa K, Shimizu H, Kusashio K, Furuya S, Nakajima N. Hepatic resection for colorectal metastases: analysis of prognostic factors. Dis Colon Rectum. 1999;42:632-639. [PubMed] |
| 104. | Bakalakos EA, Burak WE, Young DC, Martin EW. Is carcino-embryonic antigen useful in the follow-up management of patients with colorectal liver metastases? Am J Surg. 1999;177:2-6. [PubMed] |
| 105. | Van Hazel G, Blackwell A, Anderson J, Price D, Moroz P, Bower G, Cardaci G, Gray B. Randomised phase 2 trial of SIR-Spheres plus fluorouracil/leucovorin chemotherapy versus fluorouracil/leucovorin chemotherapy alone in advanced colorectal cancer. J Surg Oncol. 2004;88:78-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 343] [Cited by in RCA: 308] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 106. | Gray B, Van Hazel G, Hope M, Burton M, Moroz P, Anderson J, Gebski V. Randomised trial of SIR-Spheres plus chemotherapy vs. chemotherapy alone for treating patients with liver metastases from primary large bowel cancer. Ann Oncol. 2001;12:1711-1720. [PubMed] |
| 107. | Lygidakis NJ, Ziras N, Parissis J. Resection versus resection combined with adjuvant pre- and post-operative chemotherapy--immunotherapy for metastatic colorectal liver cancer. A new look at an old problem. Hepatogastroenterology. 1995;42:155-161. [PubMed] |
| 108. | Asahara T, Kikkawa M, Okajima M, Ojima Y, Toyota K, Nakahara H, Katayama K, Itamoto T, Marubayashi S, One E. Studies of postoperative transarterial infusion chemotherapy for liver metastasis of colorectal carcinoma after hepatectomy. Hepatogastroenterology. 1998;45:805-811. [PubMed] |
| 109. | Kemeny MM, Goldberg DA, Browning S, Metter GE, Miner PJ, Terz JJ. Experience with continuous regional chemotherapy and hepatic resection as treatment of hepatic metastases from colorectal primaries. A prospective randomized study. Cancer. 1985;55:1265-1270. [PubMed] |
| 110. | Lorenz M, Müller HH, Schramm H, Gassel HJ, Rau HG, Ridwelski K, Hauss J, Stieger R, Jauch KW, Bechstein WO. Randomized trial of surgery versus surgery followed by adjuvant hepatic arterial infusion with 5-fluorouracil and folinic acid for liver metastases of colorectal cancer. German Cooperative on Liver Metastases (Arbeitsgruppe Lebermetastasen). Ann Surg. 1998;228:756-762. [PubMed] |
| 111. | Kemeny NE, Melendez FD, Capanu M, Paty PB, Fong Y, Schwartz LH, Jarnagin WR, Patel D, D‘Angelica M. Conversion to resectability using hepatic artery infusion plus systemic chemotherapy for the treatment of unresectable liver metastases from colorectal carcinoma. J Clin Oncol. 2009;27:3465-3471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 221] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 112. | Kemeny MM, Adak S, Gray B, Macdonald JS, Smith T, Lipsitz S, Sigurdson ER, O‘Dwyer PJ, Benson AB. Combined-modality treatment for resectable metastatic colorectal carcinoma to the liver: surgical resection of hepatic metastases in combination with continuous infusion of chemotherapy--an intergroup study. J Clin Oncol. 2002;20:1499-1505. [PubMed] |
| 113. | Bolton JS, O’Connell MJ, Mahoney MR, Farr GH, Fitch TR, Maples WJ, Nagorney DM, Rubin J, Fuloria J, Steen PD. Hepatic arterial infusion and systemic chemotherapy after multiple metastasectomy in patients with colorectal carcinoma metastatic to the liver: a North Central Cancer Treatment Group (NCCTG) phase II study, 92-46-52. Clin Colorectal Cancer. 2012;11:31-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 114. | Pathak S, Jones R, Tang JM, Parmar C, Fenwick S, Malik H, Poston G. Ablative therapies for colorectal liver metastases: a systematic review. Colorectal Dis. 2011;13:e252-e265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 122] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 115. | Saito Y, Omiya H, Kohno K, Kobayashi T, Itoi K, Teramachi M, Sasaki M, Suzuki H, Takao H, Nakade M. Pulmonary metastasectomy for 165 patients with colorectal carcinoma: A prognostic assessment. J Thorac Cardiovasc Surg. 2002;124:1007-1013. [PubMed] |
| 116. | Gonzalez M, Robert JH, Halkic N, Mentha G, Roth A, Perneger T, Ris HB, Gervaz P. Survival after lung metastasectomy in colorectal cancer patients with previously resected liver metastases. World J Surg. 2012;36:386-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 117. | Schüle S, Dittmar Y, Knösel T, Krieg P, Albrecht R, Settmacher U, Altendorf-Hofmann A. Long-term results and prognostic factors after resection of hepatic and pulmonary metastases of colorectal cancer. Int J Colorectal Dis. 2013;28:537-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 118. | Sourrouille I, Mordant P, Maggiori L, Dokmak S, Lesèche G, Panis Y, Belghiti J, Castier Y. Long-term survival after hepatic and pulmonary resection of colorectal cancer metastases. J Surg Oncol. 2013;108:220-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 119. | Negri F, Musolino A, Cunningham D, Pastorino U, Ladas G, Norman AR. Retrospective study of resection of pulmonary metastases in patients with advanced colorectal cancer: the development of a preoperative chemotherapy strategy. Clin Colorectal Cancer. 2004;4:101-106. [PubMed] |
| 120. | Hawkes EA, Ladas G, Cunningham D, Nicholson AG, Wassilew K, Barbachano Y, Ratnayake G, Rao S, Chau I. Peri-operative chemotherapy in the management of resectable colorectal cancer pulmonary metastases. BMC Cancer. 2012;12:326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 121. | Bozzetti F, Doci R, Bignami P, Morabito A, Gennari L. Patterns of failure following surgical resection of colorectal cancer liver metastases. Rationale for a multimodal approach. Ann Surg. 1987;205:264-270. [PubMed] |
| 122. | Hohenberger P, Schlag P, Schwarz V, Herfarth C. Tumor recurrence and options for further treatment after resection of liver metastases in patients with colorectal cancer. J Surg Oncol. 1990;44:245-251. [PubMed] |
| 123. | Gerlinger M, Rowan AJ, Horswell S, Larkin J, Endesfelder D, Gronroos E, Martinez P, Matthews N, Stewart A, Tarpey P. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012;366:883-892. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6102] [Cited by in RCA: 5961] [Article Influence: 458.5] [Reference Citation Analysis (0)] |
| 124. | Kim MJ, Lee HS, Kim JH, Kim YJ, Kwon JH, Lee JO, Bang SM, Park KU, Kim DW, Kang SB. Different metastatic pattern according to the KRAS mutational status and site-specific discordance of KRAS status in patients with colorectal cancer. BMC Cancer. 2012;12:347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 117] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 125. | Tan KK, Lopes Gde L, Sim R. How uncommon are isolated lung metastases in colorectal cancer? A review from database of 754 patients over 4 years. J Gastrointest Surg. 2009;13:642-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 104] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 126. | Kobayashi K, Kawamura M, Ishihara T. Surgical treatment for both pulmonary and hepatic metastases from colorectal cancer. J Thorac Cardiovasc Surg. 1999;118:1090-1096. [PubMed] |
| 127. | Miller G, Biernacki P, Kemeny NE, Gonen M, Downey R, Jarnagin WR, D’Angelica M, Fong Y, Blumgart LH, DeMatteo RP. Outcomes after resection of synchronous or metachronous hepatic and pulmonary colorectal metastases. J Am Coll Surg. 2007;205:231-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 148] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 128. | Nordlinger B, Vaillant JC, Guiguet M, Balladur P, Paris F, Bachellier P, Jaeck D. Survival benefit of repeat liver resections for recurrent colorectal metastases: 143 cases. Association Francaise de Chirurgie. J Clin Oncol. 1994;12:1491-1496. [PubMed] |
| 129. | Adam R, Bismuth H, Castaing D, Waechter F, Navarro F, Abascal A, Majno P, Engerran L. Repeat hepatectomy for colorectal liver metastases. Ann Surg. 1997;225:51-60; discussion 60-62. [PubMed] |
| 130. | Imamura H, Kawasaki S, Miyagawa S, Ikegami T, Kitamura H, Shimada R. Aggressive surgical approach to recurrent tumors after hepatectomy for metastatic spread of colorectal cancer to the liver. Surgery. 2000;127:528-535. [PubMed] |
| 131. | Petrowsky H, Gonen M, Jarnagin W, Lorenz M, DeMatteo R, Heinrich S, Encke A, Blumgart L, Fong Y. Second liver resections are safe and effective treatment for recurrent hepatic metastases from colorectal cancer: a bi-institutional analysis. Ann Surg. 2002;235:863-871. [PubMed] |
| 132. | Nagakura S, Shirai Y, Suda T, Hatakeyama K. Multiple repeat resections of intra- and extrahepatic recurrences in patients undergoing initial hepatectomy for colorectal carcinoma metastases. World J Surg. 2002;26:141-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 133. | Tanaka K, Shimada H, Ohta M, Togo S, Saitou S, Yamaguchi S, Endo I, Sekido H. Procedures of choice for resection of primary and recurrent liver metastases from colorectal cancer. World J Surg. 2004;28:482-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 32] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 134. | Pessaux P, Lermite E, Brehant O, Tuech JJ, Lorimier G, Arnaud JP. Repeat hepatectomy for recurrent colorectal liver metastases. J Surg Oncol. 2006;93:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 60] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 135. | Ishiguro S, Akasu T, Fujimoto Y, Yamamoto J, Sakamoto Y, Sano T, Shimada K, Kosuge T, Yamamoto S, Fujita S. Second hepatectomy for recurrent colorectal liver metastasis: analysis of preoperative prognostic factors. Ann Surg Oncol. 2006;13:1579-1587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 136. | Sa Cunha A, Laurent C, Rault A, Couderc P, Rullier E, Saric J. A second liver resection due to recurrent colorectal liver metastases. Arch Surg. 2007;142:1144-1149; discussion 1150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 54] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 137. | Jones NB, McNally ME, Malhotra L, Abdel-Misih S, Martin EW, Bloomston M, Schmidt CR. Repeat hepatectomy for metastatic colorectal cancer is safe but marginally effective. Ann Surg Oncol. 2012;19:2224-2229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 138. | Adair RA, Young AL, Cockbain AJ, Malde D, Prasad KR, Lodge JP, Toogood GJ. Repeat hepatic resection for colorectal liver metastases. Br J Surg. 2012;99:1278-1283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 139. | Wicherts DA, de Haas RJ, Salloum C, Andreani P, Pascal G, Sotirov D, Adam R, Castaing D, Azoulay D. Repeat hepatectomy for recurrent colorectal metastases. Br J Surg. 2013;100:808-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 117] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 140. | Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990;61:759-767. [PubMed] |
| 141. | Fearon ER. Molecular genetics of colorectal cancer. Annu Rev Pathol. 2011;6:479-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1163] [Cited by in RCA: 1271] [Article Influence: 90.8] [Reference Citation Analysis (1)] |
| 142. | Bass AJ, Lawrence MS, Brace LE, Ramos AH, Drier Y, Cibulskis K, Sougnez C, Voet D, Saksena G, Sivachenko A. Genomic sequencing of colorectal adenocarcinomas identifies a recurrent VTI1A-TCF7L2 fusion. Nat Genet. 2011;43:964-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 256] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 143. | Sjöblom T, Jones S, Wood LD, Parsons DW, Lin J, Barber TD, Mandelker D, Leary RJ, Ptak J, Silliman N. The consensus coding sequences of human breast and colorectal cancers. Science. 2006;314:268-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2562] [Cited by in RCA: 2543] [Article Influence: 133.8] [Reference Citation Analysis (0)] |
| 144. | Hewish M, Lord CJ, Martin SA, Cunningham D, Ashworth A. Mismatch repair deficient colorectal cancer in the era of personalized treatment. Nat Rev Clin Oncol. 2010;7:197-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 175] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 145. | Cancer Genome Atlas Network. Comprehensive molecular characterization of human colon and rectal cancer. Nature. 2012;487:330-337. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6773] [Cited by in RCA: 6673] [Article Influence: 513.3] [Reference Citation Analysis (0)] |
| 146. | Lee AJ, Swanton C. Tumour heterogeneity and drug resistance: personalising cancer medicine through functional genomics. Biochem Pharmacol. 2012;83:1013-1020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 51] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 147. | Jones S, Zhang X, Parsons DW, Lin JC, Leary RJ, Angenendt P, Mankoo P, Carter H, Kamiyama H, Jimeno A. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science. 2008;321:1801-1806. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3216] [Cited by in RCA: 3027] [Article Influence: 178.1] [Reference Citation Analysis (0)] |
| 148. | Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P, Mankoo P, Carter H, Siu IM, Gallia GL. An integrated genomic analysis of human glioblastoma multiforme. Science. 2008;321:1807-1812. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4786] [Cited by in RCA: 4468] [Article Influence: 262.8] [Reference Citation Analysis (0)] |
| 149. | Leary RJ, Lin JC, Cummins J, Boca S, Wood LD, Parsons DW, Jones S, Sjöblom T, Park BH, Parsons R. Integrated analysis of homozygous deletions, focal amplifications, and sequence alterations in breast and colorectal cancers. Proc Natl Acad Sci USA. 2008;105:16224-16229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 248] [Cited by in RCA: 223] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 150. | Cancer Genome Atlas Research Network. Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609-615. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6439] [Cited by in RCA: 6154] [Article Influence: 439.6] [Reference Citation Analysis (0)] |
| 151. | Mardis ER, Ding L, Dooling DJ, Larson DE, McLellan MD, Chen K, Koboldt DC, Fulton RS, Delehaunty KD, McGrath SD. Recurring mutations found by sequencing an acute myeloid leukemia genome. N Engl J Med. 2009;361:1058-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1728] [Cited by in RCA: 1781] [Article Influence: 111.3] [Reference Citation Analysis (0)] |
| 152. | Baldus SE, Schaefer KL, Engers R, Hartleb D, Stoecklein NH, Gabbert HE. Prevalence and heterogeneity of KRAS, BRAF, and PIK3CA mutations in primary colorectal adenocarcinomas and their corresponding metastases. Clin Cancer Res. 2010;16:790-799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 340] [Cited by in RCA: 358] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 153. | Baas JM, Krens LL, Guchelaar HJ, Morreau H, Gelderblom H. Concordance of predictive markers for EGFR inhibitors in primary tumors and metastases in colorectal cancer: a review. Oncologist. 2011;16:1239-1249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 74] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 154. | Vogelstein B, Fearon ER, Hamilton SR, Kern SE, Preisinger AC, Leppert M, Nakamura Y, White R, Smits AM, Bos JL. Genetic alterations during colorectal-tumor development. N Engl J Med. 1988;319:525-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4616] [Cited by in RCA: 4464] [Article Influence: 120.6] [Reference Citation Analysis (0)] |
| 155. | Chu E. An update on the current and emerging targeted agents in metastatic colorectal cancer. Clin Colorectal Cancer. 2012;11:1-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 107] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 156. | Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:303-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2194] [Cited by in RCA: 2118] [Article Influence: 176.5] [Reference Citation Analysis (0)] |
| 157. | Bagnasco L, Piras D, Parodi S, Bauer I, Zoppoli G, Patrone F, Ballestrero A. Role of angiogenesis inhibitors in colorectal cancer: sensitive and insensitive tumors. Curr Cancer Drug Targets. 2012;12:303-315. [PubMed] |
| 158. | Van Cutsem E, Tabernero J, Lakomy R, Prenen H, Prausová J, Macarulla T, Ruff P, van Hazel GA, Moiseyenko V, Ferry D. Addition of aflibercept to fluorouracil, leucovorin, and irinotecan improves survival in a phase III randomized trial in patients with metastatic colorectal cancer previously treated with an oxaliplatin-based regimen. J Clin Oncol. 2012;30:3499-3506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 933] [Cited by in RCA: 1026] [Article Influence: 78.9] [Reference Citation Analysis (0)] |
| 159. | Khan KH, Yap TA, Yan L, Cunningham D. Targeting the PI3K-AKT-mTOR signaling network in cancer. Chin J Cancer. 2013;32:253-265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 165] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 160. | Yap TA, Omlin A, de Bono JS. Development of therapeutic combinations targeting major cancer signaling pathways. J Clin Oncol. 2013;31:1592-1605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 233] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 161. | Curtin JC. Novel drug discovery opportunities for colorectal cancer. Expert Opin Drug Discov. 2013;8:1153-1164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |