Published online Jan 21, 2014. doi: 10.3748/wjg.v20.i3.654
Revised: November 18, 2013
Accepted: January 2, 2014
Published online: January 21, 2014
Processing time: 127 Days and 17.4 Hours
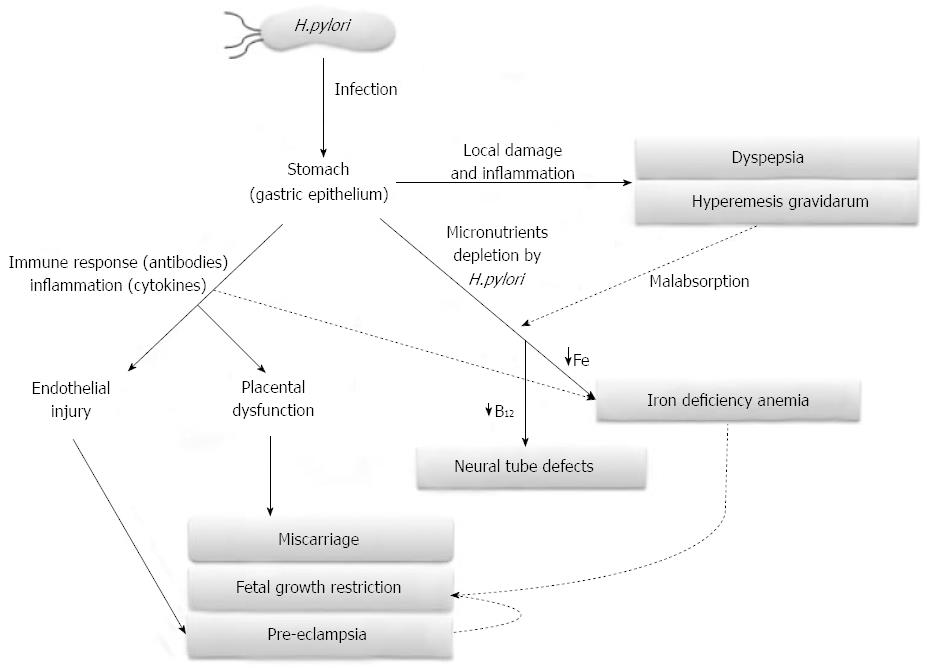
Helicobacter pylori (H. pylori) infection is investigated in gastric diseases even during pregnancy. In particular, this Gram-negative bacterium seems to be associated with hyperemesis gravidarum, a severe form of nausea and vomiting during pregnancy. During the last decade, the relationship among H. pylori and several extra-gastric diseases strongly emerged in literature. The correlation among H. pylori infection and pregnancy-related disorders was mainly focused on iron deficiency anemia, thrombocytopenia, fetal malformations, miscarriage, pre-eclampsia and fetal growth restriction. H. pylori infection may have a role in the pathogenesis of various pregnancy-related disorders through different mechanisms: depletion of micronutrients (iron and vitamin B12) in maternal anemia and fetal neural tube defects; local or systemic induction of pro-inflammatory cytokines release and oxidative stress in gastrointestinal disorders and pre-eclampsia; cross-reaction between specific anti-H. pylori antibodies and antigens localized in placental tissue and endothelial cells (pre-eclampsia, fetal growth restriction, miscarriage). Since H. pylori infection is most likely acquired before pregnancy, it is widely believed that hormonal and immunological changes occurring during pregnancy could activate latent H. pylori with a negative impact not only on maternal health (nutritional deficiency, organ injury, death), but also on the fetus (insufficient growth, malformation, death) and sometime consequences can be observed later in life. Another important issue addressed by investigators was to determine whether it is possible to transmit H. pylori infection from mother to child and whether maternal anti-H. pylori antibodies could prevent infant’s infection. Studies on novel diagnostic and therapeutic methods for H. pylori are no less important, since these are particularly sensitive topics in pregnancy conditions. It could be interesting to study the possible correlation between H. pylori infection and other pregnancy-related diseases of unknown etiology, such as gestational diabetes mellitus, obstetric cholestasis and spontaneous preterm delivery. Since H. pylori infection is treatable, the demonstration of its causative role in pregnancy-related disorders will have important social-economic implications.
Core tip:Helicobacter pylori (H. pylori) infection in pregnancy is not only associated with gastrointestinal disorders such as hyperemesis gravidarum, but also with iron deficiency anemia, fetal malformations, miscarriage, pre-eclampsia and fetal growth restriction. These pregnancy related-disorders are potentially life-threatening for both mother and fetus/neonate. Another important issue that has been addressed in literature was the question of whether it is possible to transmit H. pylori infection from mother to child and whether maternal anti-H. pylori antibodies could prevent infant’s infection. Indeed, if H. pylori is actually a causal factor, the public health implications would be important since the infection is treatable.
-
Citation: Cardaropoli S, Rolfo A, Todros T.
Helicobacter pylori and pregnancy-related disorders. World J Gastroenterol 2014; 20(3): 654-664 - URL: https://www.wjgnet.com/1007-9327/full/v20/i3/654.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i3.654
Helicobacter pylori (H. pylori) infection affects approximately one half of the world population and it is more prevalent in developing countries[1,2]. This microorganism colonizes the stomach. Typically, it is acquired during childhood and causes asymptomatic chronic infection[2]. A small portion of H. pylori infected subjects develop peptic ulcers and gastric carcinoma, usually during late adulthood[2].
H. pylori pathogenicity depends on several strain-specific factors. Some H. pylori strains express specific genes conferring pro-inflammatory, cytotoxic and vacuolating properties which could enhance the in vivo pathogenicity[3]. Virulence factors such as urease and flagella are present in all strains and they are pivotal for pathogenesis and colonization[4]. Adhesins, such as Outer inflammatory protein and Sialic acid-binding adhesin, facilitate bacterial attachment to the host epithelium and often induce its inflammatory response[5,6]. H. pylori-strains can also express Cytotoxin-associated antigen A (CagA) and Vacuolating cytotoxin A (VacA), the most investigated cytotoxins among H. pylori virulence factors. CagA is directly injected into the cytoplasm of epithelial cells, affecting cell morphology, proliferation and apoptosis[7]. H. pylori strains carrying CagA have been associated with both duodenal ulcer and gastric cancer[8], and infection with CagA-positive strain is generally associated to higher levels of inflammatory mediators compared to CagA negative strains[3]. VacA is a proteic pore-forming toxin crucial to promote and maintain bacterial colonization[9]. It disrupts cell polarity, promotes epithelial cells apoptosis and inhibits T cell proliferation and effector function[10]. Interestingly, combined seropositivity for both CagA and VacA directly correlates with elevated morbidity[11-13].
During the past decades, several reports indicated a correlation between H. pylori infection and various extra-gastric disorders[14]. Such manifestations include ischemic heart disease, diabetes mellitus, idiopathic thrombocytopenia, urticaria, and sideropenic anemia[14]. Lanciers et al[15] (1999) found a significantly increased incidence of pregnant subjects with high H. pylori IgM (marker for recently acquired infection) compared to non pregnant women. These Authors suggested that pregnancy itself may increase the susceptibility to H. pylori infection[15]. This is probably due to the fact that there are immunologic adaptations in pregnancy to ensure maternal tolerance towards the semi-allogeneic fetus. In general, pregnancy is characterized by a decreased cell-mediated cytotoxic immune response with preservation of humoral and innate immunity[16].
Nowadays, no follow-up study was conducted to describe the complete immune response against H. pylori infection during pregnancy. Most studies on the correlation between H. pylori infection and pregnancy-related disorders were cross-sectional investigations where H. pylori positivity was detected during pregnancy or soon after delivery. H. pylori infection was tested before conception only in one prospective study, where early pregnancy loss was associated with maternal H. pylori CagA-strains seropositivity before intra-cytoplasmic sperm injection[17]. Indeed, it is not possible to definitely conclude whether pregnancy-related complications are correlated to H. pylori infection acquired before or during pregnancy.
The prevalence of H. pylori infection in pregnant women varies according to geographic area, socioeconomic conditions and method used to detect H. pylori infection. For example, the prevalence of H. pylori infection among pregnant women is about 20%-30% in most European countries[18-20], Japan[21] and Australia[22], while it is 50%-70% in Turkey[23,24], Mexico and in Texas, United States[25,26], more than 80% in Egypt[27] and Gambia[28]. Furthermore, inadequate sanitation practices, low social class and crowded or high-density living conditions seem to be related to a higher prevalence of H. pylori infection. These observations suggest that poor hygiene and crowded conditions may facilitate transmission of infection among family members and they are consistent with data on intra-familial and institutional clustering of H. pylori[29,30].
The first investigations on pregnancy focused their attention mainly on the relationship between hyperemesis gravidarum and H. pylori infection. Next, researchers turned their attention to other pregnancy-related disorders, such as iron deficiency anemia, thrombocytopenia, fetal growth defects and malformations, miscarriage and, more recently, to pre-eclampsia. Another important issue that has been addressed was the question of whether it is possible to transmit H. pylori infection from mother to child and whether maternal anti-H. pylori antibodies could prevent infant’s infection. Finally, investigations on diagnostic and therapeutic methods for H. pylori are no less important, since they are particularly sensitive topics in pregnancy-related conditions. Herein, we reviewed the up-to-date literature about H. pylori and pregnancy.
Mild to moderate dyspepsia is commonly associated with nausea and vomiting and complicates about 50% of all pregnancies and it diminishes women’s life quality and social functions during early pregnancy[31]. In most women, these symptoms resolve by fluid and vitamin supplementation as well as dietary modification. About 0.3%-2% of pregnant women suffer from Hyperemesis Gravidarum (HG) characterized by severe and protracted vomiting that often results in dehydration, electrolyte imbalance, ketonemia, ketonuria, and weight loss[31-34]. Dehydration and acid base disturbances may lead to renal and hepatic injury[35]. Patients who manifest continuous weight loss and electrolyte disturbances may be at risk for growth restriction, fetal anomalies and decreased neonatal birth weight[36].
The onset of gastrointestinal symptoms is always during the first trimester, but HG may persist throughout gestation. The etiology of HG, which still remains unknown, seems to be multifactorial and may be the final result of various unrelated conditions. Indeed, treatment is performed on a symptomatic basis[35]. In particular, psychological causes, gastrointestinal tract dysfunctions, endocrine factors (i.e., elevated human chorionic gonadotropin and estrogen), genetic incompatibility, immunological factors and nutritional deficiencies have been considered part of the pathologic mechanism underlying HG. However, no single theory seems to provide an adequate explanation for HG[33,35].
Significant positive association between HG and H. pylori infection has been demonstrated by several case-control studies[37-42], and in a systematic review of 14 case-control studies, Golberg et al[32] (2007) found higher prevalence of HG in H. pylori-infected pregnant women than uninfected ones (pooled OR = 4.45; 95%CI: 2.31-8.54). In contrast, most of the studies aimed to determine the link between H. pylori and dyspepsia failed to show a significant correlation between the clinical symptoms of the disease and H. pylori infection[43,44]. Only two studies investigated the relationship between CagA-positive H. pylori strains and gastrointestinal problems in pregnancy. Noyan et al[45] (2004) found a significant association between CagA-seropositivity and dyspepsia in pregnancy, though H. pylori-seroprevalence resulted slightly but not significantly higher in pregnant women with dyspeptic complaints (74.6%) compared to the controls (63.8%). Xia et al[46] (2004) demonstrated that the infection rates of both H. pylori and CagA-positive strains are significantly higher in HG patients (88.9% and 78.1%, respectively) than in asymptomatic pregnant women (45.0% and 31.3%, respectively) (P < 0.01 for both).
Despite a high seropositive rate in pregnant women with severe gastrointestinal symptoms during early pregnancy, no correlation was found between seropositivity and clinical symptoms or their duration[47,48]. Shirin and colleagues (2004) reported an association between H. pylori and mild vomiting during early pregnancy but not with gastrointestinal symptoms later in pregnancy[49]. Studies performed on endoscopic biopsies of gastric mucosa demonstrated that the severity of gastrointestinal symptoms in early pregnancy may be associated with the density of H. pylori in the gastric epithelium[50]. Additionally, two case reports showed that H. pylori eradication treatment reduces the severity of HG[51,52].
In contrast, several studies found no relationship between HG and H. pylori[24,53-55]. These contradictory findings are probably due to the fact that a universally accepted HG definition does not exist, thus indicating a high heterogeneity of the study population.
It has been proposed that a reduction of gastric acid production during early pregnancy as a result of increased accumulation of woman’s body fluid, steroid hormone changes, and immunologic tolerance could lead the activation of latent H. pylori infection, which can exacerbate nausea and vomiting symptoms[42].
Iron deficiency is the most common nutritional deficiency in the world and results in impairment of immune, cognitive and reproductive functions, as well as decreased work performance[56]. Iron deficiency anemia (IDA) affects more than a billion people worldwide and contributes to up to 40 percent of maternal deaths in the developing countries[57]. In a typical singleton pregnancy, the average daily demand for iron is approximately 4.4 mg. A supplementation is needed when diet alone cannot supply this amount of iron, but despite iron supplementation, many women continue to remain anemic[58].
Muhsen et al[59] (2013) recommended the investigation of H. pylori infection as a potential factor that might play a role in the occurrence of anemia in children and pregnant women. Furthermore, eradication of H. pylori infection has been recommended for patients with unexplained IDA[60,61]. These recommendations are based on several studies that found a relationship between H. pylori and IDA. In a systematic review and meta-analysis of 12 case reports and series, 19 observational epidemiologic studies and six interventional trials, Muhsen and Cohen (2008) found higher prevalence of IDA in H. pylori-infected subjects than uninfected ones[62]. Several IDA mechanisms have been hypothesized in H. pylori infection, some of which are decreased mucosal iron absorption capacity due to low gastric pH, reduction of stomach vitamin C levels, bacterium-host competition for dietary iron supply, lactoferrin mediated iron sequestration by gastric H. pylori, increased hepatocytes hepcidin release in response to IL-6 production associated with H. pylori gastritis[56,62-67].
In pregnant women, H. pylori infection has been found to be associated with IDA[19,68-70]. Weyermann et al[19] (2005) found lower haemoglobin (Hb) levels at the beginning of pregnancy in H. pylori infected mothers vs noninfected (-0.25 g/dL; 95%CI: -0.49--0.003) and a more unfavourable change in Hb level during course of pregnancy (-0.14 g/dL; 95%CI: -0.38-0.10). In a cross-sectional study, out of 117 pregnant women, 27 had anemia and all of the anemic patients were shown to be H. pylori infected, and with a high chance of fetal growth restriction[68]. In a small prospective study aimed to confirm the association between H. pylori infection and HG, it was found that infected pregnant women with HG have higher prevalence of IDA compared to symptomatic uninfected patients[70]. In a small randomized double-blind placebo controlled trial, high prevalence of H. pylori infection was seen in pregnant women suffering from IDA and eradication of the infection by triple drug therapy during third trimester enhanced the response to oral iron folic acid supplementation[69].
Pre-eclampsia (PE) is a pregnancy-related syndrome characterized by new onset hypertension and proteinuria after 20 wk of gestation in a previously normotensive woman. PE affects about 2%-8% of all pregnancies and remains one of the main causes of either maternal or fetal mortality and morbidity worldwide[71]. Despite PE has been object of intense investigation, its etio-pathogenetic mechanisms are still poorly understood. This difficulty is certainly due to the fact that PE is a syndrome where similar symptoms could origin from different pathogenic pathways. PE is characterized by a generalized vascular dysfunction and an excessive maternal inflammatory response. Furthermore, it is possible to recognize two different forms of PE: “placental PE”, characterized by abnormal placentation and feto-placental compromise, and “maternal PE”, where etio-pathogenic mechanisms do not directly involve placenta and the fetus but they are of exclusive maternal origin[72,73].
Several evidences suggest that subclinical infections could play a role in the onset of PE[74,75]. The association between H. pylori seropositivity and PE was found for the first time by our group[76]. We showed that H. pylori seropositivity frequency is higher in mothers with PE (51.1%) compared with women with uneventful pregnancy (31.9%) (OR = 2.67; 95%CI: 1.08-6.57; P = 0.033)[76]. Afterwards, other two case-control studies reported a significantly higher H. pylori seropositivity rate in PE patients compared to controls[77,78]. UstUn et al[77] (2010) reported a significantly higher positivity for IgA anti-H. pylori in patients with PE compared with controls (P = 0.034), and Aksoy and colleagues found a H. pylori seropositivity rate of 81% in the pre-eclampsia group, and of 60% in normal pregnant women (OR = 2.86; 95%CI: 1.05-7.82; P = 0.036)[78]. We also showed a strong association between the onset of PE and CagA-positive H. pylori strains infection, which are more virulent and therefore more likely to elicit the generalized inflammation and the subsequent vascular damage typical of PE[76]. Recently, we found that CagA/VacA dual seropositivity is specifically associated with PE and, in particular, with “placental PE”[79]. Interestingly, Franceschi et al[80] (2012) demonstrated that antibodies against the H. pylori virulence factor CagA cross-react in vitro with placental tissue reducing its invasiveness ability and it is well known that these antibodies recognize antigens localized on the surface of endothelial cells[81]. Therefore infection with CagA-positive strains could contribute not only to the exacerbated maternal inflammatory response leading to all forms of PE but also to the abnormal placentation typical of “placental PE”.
H. pylori could be involved in the pathogenesis of PE mainly by inducing inflammation and oxidative stress and consequently generalized endothelial dysfunction. In fact, it was observed that H. pylori seropositive PE subjects are characterized by a more severe inflammatory status compared to the inflammatory response characterizing normal pregnancy, since pre-eclamptic women showed higher levels of C-reactive protein, tumour necrosis factor (TNF)-alpha and maternal leukocytes counts[77,79]. Interestingly, pre-eclamptic patients, had higher H. pylori seropositivity rate and serum malondialdehyde levels, a common marker of lipid peroxidation, compared with healthy pregnant. Furthermore, the subgroup of seropositive PE mothers had higher serum levels of total cholesterol and low-density lipoprotein (LDL)-C compared to seronegative PE women. The Authors hypothesized that H. pylori infection may be a contributory factor in atherosclerosis in PE cases later in life[78]. Prospective cohort studies are required to confirm this hypothesis. However, such studies would be difficult to be conducted, since large cohorts of pregnant women would be needed to detect significance and prospective studies are limited in their ability to evaluate uncommon outcomes such as PE.
Fetal growth restriction (FGR) is defined as failure of the fetus to achieve its genetically determined growth potential[82,83]. FGR may be due to either feto-placental or maternal causes and 3%-10% of infants suffer from growth restriction. Feto-placental causes include infections and other placental pathologies. Known maternal causes of FGR include vascular disorders (chronic hypertension, pre-eclampsia or diabetes with vasculopathy), poor maternal weight gain, smoking, alcohol, cocaine, advanced maternal age and previous poor pregnancy outcome[84].
Eslick et al[22] (2002) observed for the first time an association between H. pylori infection and low birth weight, in particular they showed that intrauterine growth restriction was more common in H. pylori seropositive women (13.5%) than in seronegative mothers (6.0%) (OR = 2.41; 95%CI: 1.14-5.08; P = 0.018). Furthermore, it has been reported that H. pylori infected mice showed a decrease in implantation rates, and their offspring were of low birth-weight[85]. However, in another experimental mice model study these results were not confirmed[86].
H. pylori may be linked with an increase in symptoms including dyspepsia, nausea or vomiting[42,52], because of underlying undiagnosed peptic ulcer disease, which in turn may affect maternal gastric absorption and therefore impair fetal growth. Also maternal anemia associated to H. pylori infection may lead to FGR. In fact, Mulayim et al[68] (2008) observed that pregnant women with H. pylori infection delivered neonates with a significantly lower birth-weight compared to mothers without the infection. However, in this study FGR could be due to maternal anemia since all anemic pregnant women were in the H. pylori positive group. As previously underlined, it was recently demonstrated that anti-CagA antibodies cross-react in vitro with placental tissue reducing its invasiveness ability[80] and the consequent abnormal placentation could lead to FGR. However, in our study on H. pylori virulence factors we demonstrated a strong association between H. pylori infection and FGR in pre-eclamptic pregnancies, while there was no association between H. pylori and idiopathic FGR[79].
H. pylori infection has been linked to other few disease states in pregnancy but there are still small amount of data supporting these premises.
Miscarriage or spontaneous abortion, occurring in 15% of pregnancies, is defined as an unintended termination of pregnancy resulting in fetal death prior to 23 wk of gestation[87]. Among non-chromosomal causes of fetal loss, infections have a minor relevance compared to other etiologic factors. However Rossi et al[85] (2004) observed a higher number of fetal resorption in H. pylori infected pregnant mice compared to non-infected controls. Hajishafiha et al[17] found an association between H. pylori CagA-strains maternal infection and early pregnancy loss in patients undergoing intra-cytoplasmic sperm injection. Recently we found a significantly higher percentage of H. pylori seropositive women among primigravidae with a miscarriage compared to controls, while the presence of maternal serum antibodies against H. pylori did not appear to be associated with recurrent miscarriage[20]. These findings suggest a relationship between H. pylori infection and implantation/placentation failure, possibly due to a cross-reaction between antibodies against H. pylori and placental tissue[80].
Several studies reported that serum/plasma vitamin B12 and folate levels are lower in subjects with H. pylori infection compared to uninfected persons[88,89]. Moreover, several investigations indicated that vitamin B12 and folate levels improve after H. pylori eradication[90,91]. Two case-control studies in a Mexican-American and in Iranian population reported that H. pylori could play a role in neural tube defect (NTD) causation by reducing folate and vitamin B12 concentrations. They showed that H. pylori seropositivity in pregnant women can increase the risk of occurrence of NTDs in newborns, since seropositivity was more frequent among mothers of newborns with NTDs than controls[25,92]. However in both studies the differences were not significant.
Thrombocytopenia, although often innocuous[93], could have dangerous complications during pregnancy. Pregnancies affected by extremely low platelets, often with immune (idiopathic) thrombocytopenic purpura (ITP), require frequent careful monitoring during prenatal visits, especially once entering the third trimester in preparation for delivery[94]. Furthermore, a recent retrospective study showed that ITP was an independent risk factor for both perinatal mortality and preterm delivery[95].
Association between H. pylori and thrombocytopenia has been demonstrated in a non-pregnant population[96-98]. The etiology of thrombocytopenia may be due to cross-molecular mimicry between specific H. pylori protein (CagA) and platelet antigens[99], however no relationship was found between H. pylori infection and platelet count during pregnancy[68,79,100,101].
It would be interesting to confirm the above mentioned findings and to investigate the possible correlation among H. pylori infection and other pregnancy-related diseases of unknown etiology, such as gestational diabetes mellitus, obstetric cholestasis and spontaneous preterm delivery. In fact, H. pylori infection seems to be associated to diabetes mellitus[102] and hepatobiliary diseases in the general population[103] and it is well known that bacterial infections increase the risk of spontaneous preterm delivery[104].
Children of H. pylori infected mothers seem to have a higher risk of acquiring H. pylori[27,105]. However, experimental animal models suggested that vertical infection during the prenatal period or delivery procedure is unlikely to be route of mother-to-child transmission of the infection. It is possible that H. pylori is acquired through breast-feeding, contaminated saliva and fecal-oral transmission during co-habitation[21,86,106]. Indeed, for the general population, the most common way of transmission is from person to person by either oral-oral route (through vomitus or possibly saliva) or fecal-oral route. The person-to-person way of transmission is supported by the higher incidence of infection among institutionalized children and adults and the clustering of H. pylori infection within families. Moreover, detection of H. pylori DNA in vomitus, saliva, dental plaque, gastric juice, and feces further supports this concept. Waterborne transmission, probably due to fecal contamination, may be an important source of infection, especially in those world’s areas in which untreated water is common[29,30].
Furthermore, in our previous study on pre-eclampsia and H. pylori we indirectly demonstrated the absence of vertical transmission in humans, since we never found the presence of H. pylori DNA in placentae of H. pylori positive patients[76].
It is widely established that specific anti-H. pylori IgG antibodies are transplacentally transferred from mothers to fetuses[18] and a close correlation between maternal and cord specific IgG levels was demonstrated[21,28]. These passively acquired antibodies decline over the first 3-4 mo of life[18,28]. Some researchers have suggested that maternal IgG may protect against H. pylori colonization[18,107] and this is supported by work in murine models[108]. Other investigators found no evidence of a protective role for passively acquired maternal antibodies in infants at high risk of early H. pylori colonization[28].
It was also suggested that IgA antibodies in maternal milk confer passive protection against early human H. pylori colonization[109-111]. However, in a previous study the relationship between breastfeeding and H. pylori was investigated in 946 preschool children and their mothers with C-urea breath test. H. pylori prevalence was higher in breastfed children compared with children who were never breastfed. The Authors concluded that breastfeeding was not protective against H. pylori[112].
The current diagnostic methods include invasive and non-invasive tests. Invasive tests involve an upper gastrointestinal endoscopy with gastric mucosal biopsy and rapid urease activity detection, histology, microbiological culture, or polymerase chain reaction assays. Although mucosal biopsy and histopathologic examination of specimens for the presence of H. pylori and/or gastritis is considered the gold standard for the diagnosis of H. pylori infection, invasive tests are not well tolerated by patients and may be a source of ethical problems. Gastroscopy can be performed in pregnant patients, but only when it is strictly necessary[113].
The non invasive methods are more widely accepted in the prenatal period and include serum antibody detection, carbon-labeled urea breath tests, and stool antigen detection.
Serologic and stool antigen tests are the first choice for H. pylori infection diagnosis in pregnancy, since they are easy to perform and low-cost non invasive diagnostic tests. Serologic tests are usually based on the detection of specific anti-H. pylori IgG antibodies in the patients’ sera by immuno-enzymatic assay. Measurement of IgG antibodies against H. pylori reveals an immune response that could represent either a current infection or a previous exposure, since IgG antibodies disappear only several months after eradication of the microorganism[114].
The stool antigen test is an enzymatic immunoassay that detects the active presence of H. pylori antigen in human feces. Stool antigen test is preferred to determine the H. pylori status after eradication[115].
Urea breath tests are not commonly used during pregnancy, despite they are reliable and noninvasive diagnostic test. In fact, it is demonstrated that 13C-urea breath test, using the stable isotope 13C as tracer, is not radioactive and safe also in children and pregnancy. Therefore, it could be used as a valuable non-invasive semi-quantitative diagnostic tool for the assessment of gastric bacterial H. pylori infection. The urea breath test is recommended for test-and-treat strategies and suitable for control after eradication therapy and in epidemiological or pharmacological studies[116]. Despite the excellent sensitivity and specificity of these tests, they are expensive and require specific instrumentation and specialized staff.
Furthermore, it was stated that ionizing radiation dose involved in 14C-urea breath test is extremely low, much lower than the radiation dose adsorbed from natural sources, a thousand times lower than the amount of fetal radiation considered to be teratogenic, therefore in the event of inadvertent exposure during pregnancy, the pregnant women should be reassured[117].
There are multiple options for H. pylori infection treatment. The association of a proton-pump inhibitor and two antibiotics for 1 or 2 wk gives the best eradication rates in non pregnant subjects. Currently, there are no guidelines to treat H. pylori infection during pregnancy and the optimal therapy in pregnancy remains uncertain[118]. Hayakawa et al[119] treated four women with hyperemesis gravidarum by a combination of penicillin and erythromycin, leading to alleviation of symptoms thus demonstrating the possible effectiveness of this specific H. pylori treatment. This hypothesis is supported in four additional case reports that showed similar symptom relief after antibiotic treatment[47,51,52,120].
Several investigators have evaluated the safety of individual drugs, including proton pump inhibitors used in the anti H. pylori drug therapy in pregnant women. A recent meta-analysis reported that the use of proton pump inhibitors during first-trimester does not seem to be associated with increased risk of spontaneous abortion, preterm delivery or major congenital birth defects[121]. Nevertheless, some experts recommend that H. pylori eradication should be deferred until after pregnancy and lactation[122].
It must be considered that treatment of H. pylori infection has a low successful rate, with 35%-85% of infections being cleared, reaching the lowest values in some European countries[123]. The gradual but steady occurrence of antibiotic-resistant strains represents a major obstacle in the treatment of H. pylori infection. Pharmacogenomics-based approaches seem to increase the cure rates, but re-infection also remains problematic. In fact, it is well known that eradication of H. pylori infections with antimicrobial agents in adults does not induce immunity against re-infection. In general, low annual recurrence rates were observed in developed countries (up to 2% for both adults and children), but high recurrence rates (> 10%) were observed in developing countries[124]. There is no clear evidence that pregnancy predisposes to de novo H. pylori infection.
In view of these evidences, new approaches need to be considered for treatment of this disease, such as design of effective vaccines. Especially in case of pregnancy related diseases, it would be preferable to prevent H. pylori infection consequences, thus avoiding pharmacologic therapies during pregnancy. Recently, several clinical trials and animal studies have been focused on generating H. pylori recombinant vaccines useful to eradicate and protect against the infection; however a safe and effective H. pylori vaccine has not yet been developed for use in humans[123].
Therefore, if H. pylori infection will be confirmed as an important risk factor for pregnancy complications, we suggest the conventional H. pylori eradication, namely triple therapy, should ideally be obtained several months before conception in order to reach seronegativity. This approach would avoid cross-reaction between anti-H. pylori antibodies and host tissue antigens, waiting for the discovery of novel effective vaccines.
H. pylori infection was investigated not only in association with gastrointestinal manifestations during pregnancy but also with other severe pregnancy-related disorders. H. pylori infection may have a role in the pathogenesis of these disorders through different mechanisms: depletion of micronutrients (iron and vitamin B12) in the case of maternal anemia and fetal neural tube defects; local and systemic induction of pro-inflammatory cytokines release and oxidative stress in gastrointestinal disorders and pre-eclampsia; cross-reaction between specific anti-H. pylori antibodies and antigens localized in placental and endothelial cells (pre-eclampsia, fetal growth restriction, miscarriage) (Figure 1). Since H. pylori infection is most likely acquired before pregnancy, it is believed that hormonal and immunological changes occurring during pregnancy can activate latent H. pylori infection and this could have an impact not only on the mother health (nutritional deficiency, organ injury, death), but also on her child (insufficient growth, malformation, death) and sometime consequences can be observed later in life.
H. pylori mother to child transmission does not appear to occur during pregnancy or delivery. Furthermore, it was demonstrated that specific antibodies against this microorganism are transferred to the fetus/infant both transplacentally and by means of maternal milk. However, it is not clear whether maternal antibodies are able to protect the children against H. pylori colonization.
Currently, clinicians choose a non-invasive diagnostic method for H. pylori infection and prefer to treat the infection out of pregnancy. If H. pylori will be confirmed as causal and/or contributing factor of major pregnancy-related disorders, it will have important positive implications for the public health system since the infection is treatable. It is likely that pre-pregnancy diagnosis and preventive H. pylori eradication would reduce the incidence of some of these complications. More data are needed to understand if screening for H. pylori infection could be effective in preventing pregnancy disorders. The design of an effective vaccine will be even more useful in order to avoid drug resistance and re-infection problems.
P- Reviewers: Shi ZJ, Wang CC, Wang FZ S- Editor: Cui XM L- Editor: A E- Editor: Liu XM
| 1. | Malaty HM. Epidemiology of Helicobacter pylori infection. Best Pract Res Clin Gastroenterol. 2007;21:205-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 165] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 2. | Suerbaum S, Michetti P. Helicobacter pylori infection. N Engl J Med. 2002;347:1175-1186. [PubMed] [DOI] [Full Text] |
| 3. | Graham DY, Yamaoka Y. Disease-specific Helicobacter pylori virulence factors: the unfulfilled promise. Helicobacter. 2000;5 Suppl 1:S3-9; discussion S27-31. [PubMed] |
| 4. | Harris PR, Mobley HL, Perez-Perez GI, Blaser MJ, Smith PD. Helicobacter pylori urease is a potent stimulus of mononuclear phagocyte activation and inflammatory cytokine production. Gastroenterology. 1996;111:419-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 160] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 5. | Yamaoka Y, Kwon DH, Graham DY. A M(r) 34,000 proinflammatory outer membrane protein (oipA) of Helicobacter pylori. Proc Natl Acad Sci USA. 2000;97:7533-7538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 292] [Cited by in RCA: 318] [Article Influence: 12.7] [Reference Citation Analysis (2)] |
| 6. | Mahdavi J, Sondén B, Hurtig M, Olfat FO, Forsberg L, Roche N, Angstrom J, Larsson T, Teneberg S, Karlsson KA. Helicobacter pylori SabA adhesin in persistent infection and chronic inflammation. Science. 2002;297:573-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 639] [Cited by in RCA: 665] [Article Influence: 28.9] [Reference Citation Analysis (0)] |
| 7. | Blaser MJ, Atherton JC. Helicobacter pylori persistence: biology and disease. J Clin Invest. 2004;113:321-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 631] [Cited by in RCA: 621] [Article Influence: 29.6] [Reference Citation Analysis (1)] |
| 8. | Graham DY, Yamaoka Y. H. pylori and cagA: relationships with gastric cancer, duodenal ulcer, and reflux esophagitis and its complications. Helicobacter. 1998;3:145-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 124] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 9. | Cover TL, Blanke SR. Helicobacter pylori VacA, a paradigm for toxin multifunctionality. Nat Rev Microbiol. 2005;3:320-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 372] [Cited by in RCA: 411] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 10. | Palframan SL, Kwok T, Gabriel K. Vacuolating cytotoxin A (VacA), a key toxin for Helicobacter pylori pathogenesis. Front Cell Infect Microbiol. 2012;2:92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 199] [Article Influence: 15.3] [Reference Citation Analysis (2)] |
| 11. | Blaser MJ, Perez-Perez GI, Kleanthous H, Cover TL, Peek RM, Chyou PH, Stemmermann GN, Nomura A. Infection with Helicobacter pylori strains possessing cagA is associated with an increased risk of developing adenocarcinoma of the stomach. Cancer Res. 1995;55:2111-2115. [PubMed] |
| 12. | Kuipers EJ, Pérez-Pérez GI, Meuwissen SG, Blaser MJ. Helicobacter pylori and atrophic gastritis: importance of the cagA status. J Natl Cancer Inst. 1995;87:1777-1780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 412] [Cited by in RCA: 395] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 13. | Van Doorn LJ, Figueiredo C, Mégraud F, Pena S, Midolo P, Queiroz DM, Carneiro F, Vanderborght B, Pegado MD, Sanna R. Geographic distribution of vacA allelic types of Helicobacter pylori. Gastroenterology. 1999;116:823-830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 299] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 14. | Banić M, Franceschi F, Babić Z, Gasbarrini A. Extragastric manifestations of Helicobacter pylori infection. Helicobacter. 2012;17 Suppl 1:49-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 62] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 15. | Lanciers S, Despinasse B, Mehta DI, Blecker U. Increased susceptibility to Helicobacter pylori infection in pregnancy. Infect Dis Obstet Gynecol. 1999;7:195-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 16. | Chang J, Streitman D. Physiologic adaptations to pregnancy. Neurol Clin. 2012;30:781-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 72] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 17. | Hajishafiha M, Ghasemi-Rad M, Memari A, Naji S, Mladkova N, Saeedi V. Effect of Helicobacter pylori infection on pregnancy rates and early pregnancy loss after intracytoplasmic sperm injection. Int J Womens Health. 2011;3:329-335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 18. | Blecker U, Lanciers S, Keppens E, Vandenplas Y. Evolution of Helicobacter pylori positivity in infants born from positive mothers. J Pediatr Gastroenterol Nutr. 1994;19:87-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 19. | Weyermann M, Rothenbacher D, Gayer L, Bode G, Adler G, Grab D, Flock F, Brenner H. Role of Helicobacter pylori infection in iron deficiency during pregnancy. Am J Obstet Gynecol. 2005;192:548-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 20. | Cardaropoli S, Piazzese A, Piccoli E, Rolfo A, Todros T. Is Helicobacter pylori infection a risk factor for miscarriage? Placenta. 2013;34:A37-A38. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 21. | Kitagawa M, Natori M, Katoh M, Sugimoto K, Omi H, Akiyama Y, Sago H. Maternal transmission of Helicobacter pylori in the perinatal period. J Obstet Gynaecol Res. 2001;27:225-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Eslick GD, Yan P, Xia HH, Murray H, Spurrett B, Talley NJ. Foetal intrauterine growth restrictions with Helicobacter pylori infection. Aliment Pharmacol Ther. 2002;16:1677-1682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 23. | Karaer A, Ozkan O, Ozer S, Bayir B, Kilic S, Babur C, Danişman N. Gastrointestinal symptoms and Helicobacter pylori infection in early pregnancy. A seroepidemiologic study. Gynecol Obstet Invest. 2008;66:44-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 24. | Berker B, Soylemez F, Cengiz SD, Kose SK. Serologic assay of Helicobacter pylori infection. Is it useful in hyperemesis gravidarum? J Reprod Med. 2003;48:809-812. [PubMed] |
| 25. | Felkner M, Suarez L, Liszka B, Brender JD, Canfield M. Neural tube defects, micronutrient deficiencies, and Helicobacter pylori: a new hypothesis. Birth Defects Res A Clin Mol Teratol. 2007;79:617-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 26. | Goodman KJ, O’Rourke K, Day RS, Wang C, Redlinger T, Campos A, de la Rosa JM. Helicobacter pylori infection in pregnant women from a U.S.-Mexico border population. J Immigr Health. 2003;5:99-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Bassily S, Frenck RW, Mohareb EW, Wierzba T, Savarino S, Hall E, Kotkat A, Naficy A, Hyams KC, Clemens J. Seroprevalence of Helicobacter pylori among Egyptian newborns and their mothers: a preliminary report. Am J Trop Med Hyg. 1999;61:37-40. [PubMed] |
| 28. | Bunn JE, Thomas JE, Harding M, Coward WA, Weaver LT. Placental acquisition of maternal specific IgG and Helicobacter pylori colonization in infancy. Helicobacter. 2003;8:568-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 29. | Brown LM. Helicobacter pylori: epidemiology and routes of transmission. Epidemiol Rev. 2000;22:283-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 620] [Cited by in RCA: 623] [Article Influence: 24.9] [Reference Citation Analysis (0)] |
| 30. | Goodman KJ, Cockburn M. The role of epidemiology in understanding the health effects of Helicobacter pylori. Epidemiology. 2001;12:266-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 49] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 31. | Broussard CN, Richter JE. Nausea and vomiting of pregnancy. Gastroenterol Clin North Am. 1998;27:123-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 95] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 32. | Golberg D, Szilagyi A, Graves L. Hyperemesis gravidarum and Helicobacter pylori infection: a systematic review. Obstet Gynecol. 2007;110:695-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 113] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 33. | Verberg MF, Gillott DJ, Al-Fardan N, Grudzinskas JG. Hyperemesis gravidarum, a literature review. Hum Reprod Update. 2005;11:527-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 254] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 34. | Hod M, Orvieto R, Kaplan B, Friedman S, Ovadia J. Hyperemesis gravidarum. A review. J Reprod Med. 1994;39:605-612. [PubMed] |
| 35. | Eliakim R, Abulafia O, Sherer DM. Hyperemesis gravidarum: a current review. Am J Perinatol. 2000;17:207-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 123] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 36. | Gross S, Librach C, Cecutti A. Maternal weight loss associated with hyperemesis gravidarum: a predictor of fetal outcome. Am J Obstet Gynecol. 1989;160:906-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 100] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 37. | Guven MA, Ertas IE, Coskun A, Ciragil P. Serologic and stool antigen assay of Helicobacter pylori infection in hyperemesis gravidarum: which test is useful during early pregnancy? Taiwan J Obstet Gynecol. 2011;50:37-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 38. | Frigo P, Lang C, Reisenberger K, Kölbl H, Hirschl AM. Hyperemesis gravidarum associated with Helicobacter pylori seropositivity. Obstet Gynecol. 1998;91:615-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 40] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 39. | Kazerooni T, Taallom M, Ghaderi AA. Helicobacter pylori seropositivity in patients with hyperemesis gravidarum. Int J Gynaecol Obstet. 2002;79:217-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 40. | Salimi-Khayati A, Sharami H, Mansour-Ghanaei F, Sadri S, Fallah MS. Helicobacter pylori aeropositivity and the incidence of hyperemesis gravidarum. Med Sci Monit. 2003;9:CR12-CR15. [PubMed] |
| 41. | Cevrioglu AS, Altindis M, Yilmazer M, Fenkci IV, Ellidokuz E, Kose S. Efficient and non-invasive method for investigating Helicobacter pylori in gravida with hyperemesis gravidarum: Helicobacter pylori stool antigen test. J Obstet Gynaecol Res. 2004;30:136-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 42. | Koçak I, Akcan Y, Ustün C, Demirel C, Cengiz L, Yanik FF. Helicobacter pylori seropositivity in patients with hyperemesis gravidarum. Int J Gynaecol Obstet. 1999;66:251-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 43. | Tanriverdi HA, Ustundag Y, Tekin IO, Barut A. Dyspeptic complaints after 20 weeks of gestation are not related to Helicobacter pylori seropositivity. Med Sci Monit. 2005;11:CR445-CR448. [PubMed] |
| 44. | McKenna D, Watson P, Dornan J. Helicobacter pylori infection and dyspepsia in pregnancy. Obstet Gynecol. 2003;102:845-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 45. | Noyan V, Apan TZ, Yucel A, Sagsoz N. Cytotoxin associated gene A-positive Helicobacter pylori strains in dyspeptic pregnant women. Eur J Obstet Gynecol Reprod Biol. 2004;116:186-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 46. | Xia LB, Yang J, Li AB, Tang SH, Xie QZ, Cheng D. Relationship between hyperemesis gravidarum and Helicobacter pylori seropositivity. Chin Med J (Engl). 2004;117:301-302. [PubMed] |
| 47. | Wu CY, Tseng JJ, Chou MM, Lin SK, Poon SK, Chen GH. Correlation between Helicobacter pylori infection and gastrointestinal symptoms in pregnancy. Adv Ther. 2000;17:152-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 48. | Erdem A, Arslan M, Erdem M, Yildirim G, Himmetoğlu O. Detection of Helicobacter pylori seropositivity in hyperemesis gravidarum and correlation with symptoms. Am J Perinatol. 2002;19:87-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 49. | Shirin H, Sadan O, Shevah O, Bruck R, Boaz M, Moss SF, Everon S, Glezerman M, Avni Y. Positive serology for Helicobacter pylori and vomiting in the pregnancy. Arch Gynecol Obstet. 2004;270:10-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 50. | Bagis T, Gumurdulu Y, Kayaselcuk F, Yilmaz ES, Killicadag E, Tarim E. Endoscopy in hyperemesis gravidarum and Helicobacter pylori infection. Int J Gynaecol Obstet. 2002;79:105-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 37] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 51. | El Younis CM, Abulafia O, Sherer DM. Rapid marked response of severe hyperemesis gravidarum to oral erythromycin. Am J Perinatol. 1998;15:533-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 52. | Jacoby EB, Porter KB. Helicobacter pylori infection and persistent hyperemesis gravidarum. Am J Perinatol. 1999;16:85-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 53. | Jacobson GF, Autry AM, Somer-Shely TL, Pieper KL, Kirby RS. Helicobacter pylori seropositivity and hyperemesis gravidarum. J Reprod Med. 2003;48:578-582. [PubMed] |
| 54. | Karadeniz RS, Ozdegirmenci O, Altay MM, Solaroglu A, Dilbaz S, Hizel N, Haberal A. Helicobacter pylori seropositivity and stool antigen in patients with hyperemesis gravidarum. Infect Dis Obstet Gynecol. 2006;2006:73073. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 55. | Aytac S, Türkay C, Kanbay M. Helicobacter pylori stool antigen assay in hyperemesis gravidarum: a risk factor for hyperemesis gravidarum or not? Dig Dis Sci. 2007;52:2840-2843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 56. | Cardenas VM, Mulla ZD, Ortiz M, Graham DY. Iron deficiency and Helicobacter pylori infection in the United States. Am J Epidemiol. 2006;163:127-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 146] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 57. | Brabin BJ, Hakimi M, Pelletier D. An analysis of anemia and pregnancy-related maternal mortality. J Nutr. 2001;131:604S-614S; discussion 614S-615S. [PubMed] |
| 58. | Milman N, Bergholt T, Byg KE, Eriksen L, Graudal N. Iron status and iron balance during pregnancy. A critical reappraisal of iron supplementation. Acta Obstet Gynecol Scand. 1999;78:749-757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 43] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 59. | Muhsen K, Cohen D. Helicobacter pylori infection and anemia. Am J Trop Med Hyg. 2013;89:398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 60. | Malfertheiner P, Megraud F, O’Morain C, Bazzoli F, El-Omar E, Graham D, Hunt R, Rokkas T, Vakil N, Kuipers EJ. Current concepts in the management of Helicobacter pylori infection: the Maastricht III Consensus Report. Gut. 2007;56:772-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1396] [Cited by in RCA: 1349] [Article Influence: 74.9] [Reference Citation Analysis (1)] |
| 61. | Caselli M, Zullo A, Maconi G, Parente F, Alvisi V, Casetti T, Sorrentino D, Gasbarrini G. “Cervia II Working Group Report 2006”: guidelines on diagnosis and treatment of Helicobacter pylori infection in Italy. Dig Liver Dis. 2007;39:782-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 82] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 62. | Muhsen K, Cohen D. Helicobacter pylori infection and iron stores: a systematic review and meta-analysis. Helicobacter. 2008;13:323-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 134] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 63. | Choe YH, Oh YJ, Lee NG, Imoto I, Adachi Y, Toyoda N, Gabazza EC. Lactoferrin sequestration and its contribution to iron-deficiency anemia in Helicobacter pylori-infected gastric mucosa. J Gastroenterol Hepatol. 2003;18:980-985. [PubMed] |
| 64. | DuBois S, Kearney DJ. Iron-deficiency anemia and Helicobacter pylori infection: a review of the evidence. Am J Gastroenterol. 2005;100:453-459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 140] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 65. | Annibale B, Capurso G, Lahner E, Passi S, Ricci R, Maggio F, Delle Fave G. Concomitant alterations in intragastric pH and ascorbic acid concentration in patients with Helicobacter pylori gastritis and associated iron deficiency anaemia. Gut. 2003;52:496-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 122] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 66. | Pellicano R, Rizzetto M. Is hepcidin the bridge linking Helicobacter pylori and anemia of chronic infection? A research proposal. Panminerva Med. 2004;46:165-169. [PubMed] |
| 67. | Ciacci C, Sabbatini F, Cavallaro R, Castiglione F, Di Bella S, Iovino P, Palumbo A, Tortora R, Amoruso D, Mazzacca G. Helicobacter pylori impairs iron absorption in infected individuals. Dig Liver Dis. 2004;36:455-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 64] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 68. | Mulayim B, Celik NY, Yanik FF. Helicobacter pylori infection detected by 14C-urea breath test is associated with iron deficiency anemia in pregnant women. J Obstet Gynaecol Res. 2008;34:980-985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 69. | Malik R, Guleria K, Kaur I, Sikka M, Radhakrishnan G. Effect of Helicobacter pylori eradication therapy in iron deficiency anaemia of pregnancy - a pilot study. Indian J Med Res. 2011;134:224-231. [PubMed] |
| 70. | Bezircioǧlu I, Elveren HB, Baloǧlu A, Biçer M. The positivity of Helicobacter pylori Stool Antigen in patients with Hyperemesis gravidarum. J Turkish German Gynecol Associ. 2011;12:71-74. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 71. | Duley L. The global impact of pre-eclampsia and eclampsia. Semin Perinatol. 2009;33:130-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1596] [Cited by in RCA: 1765] [Article Influence: 110.3] [Reference Citation Analysis (0)] |
| 72. | Todros T, Vasario E, Cardaropoli S. Preeclampsia as an infectious disease. E. xp Rev Obstetr Gynecol. 2007;2:735-741. [RCA] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 73. | Redman CW, Sargent IL. Latest advances in understanding preeclampsia. Science. 2005;308:1592-1594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1791] [Cited by in RCA: 1881] [Article Influence: 94.1] [Reference Citation Analysis (0)] |
| 74. | Conde-Agudelo A, Villar J, Lindheimer M. Maternal infection and risk of preeclampsia: systematic review and metaanalysis. Am J Obstet Gynecol. 2008;198:7-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 210] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 75. | Rustveld LO, Kelsey SF, Sharma R. Association between maternal infections and preeclampsia: a systematic review of epidemiologic studies. Matern Child Health J. 2008;12:223-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 86] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 76. | Ponzetto A, Cardaropoli S, Piccoli E, Rolfo A, Gennero L, Kanduc D, Todros T. Pre-eclampsia is associated with Helicobacter pylori seropositivity in Italy. J Hypertens. 2006;24:2445-2449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 77. | UstUn Y, Engin-UstUn Y, Ozkaplan E, Otlu B, Sait TekerekoGlu M. Association of Helicobacter pylori infection with systemic inflammation in preeclampsia. J Matern Fetal Neonatal Med. 2010;23: 311-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 78. | Aksoy H, Ozkan A, Aktas F, Borekci B. Helicobacter pylori seropositivity and its relationship with serum malondialdehyde and lipid profile in preeclampsia. J Clin Lab Anal. 2009;23:219-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 79. | Cardaropoli S, Rolfo A, Piazzese A, Ponzetto A, Todros T. Helicobacter pylori’s virulence and infection persistence define pre-eclampsia complicated by fetal growth retardation. World J Gastroenterol. 2011;17:5156-5165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 43] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 80. | Franceschi F, Di Simone N, D’Ippolito S, Castellani R, Di Nicuolo F, Gasbarrini G, Yamaoka Y, Todros T, Scambia G, Gasbarrini A. Antibodies anti-CagA cross-react with trophoblast cells: a risk factor for pre-eclampsia? Helicobacter. 2012;17:426-434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 81. | Franceschi F, Niccoli G, Ferrante G, Gasbarrini A, Baldi A, Candelli M, Feroce F, Saulnier N, Conte M, Roccarina D. CagA antigen of Helicobacter pylori and coronary instability: insight from a clinico-pathological study and a meta-analysis of 4241 cases. Atherosclerosis. 2009;202:535-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 80] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 82. | Cetin I, Foidart JM, Miozzo M, Raun T, Jansson T, Tsatsaris V, Reik W, Cross J, Hauguel-de-Mouzon S, Illsley N. Fetal growth restriction: a workshop report. Placenta. 2004;25:753-757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 73] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 83. | Pollack RN, Divon MY. Intrauterine growth retardation: definition, classification, and etiology. Clin Obstet Gynecol. 1992;35:99-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 153] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 84. | Lin CC, Santolaya-Forgas J. Current concepts of fetal growth restriction: part I. Causes, classification, and pathophysiology. Obstet Gynecol. 1998;92:1044-1055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 59] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 85. | Rossi G, Romagnoli S, Lauretti L, Pancotto L, Taccini E, Rappuoli R, Del Giudice G, Ruggiero P. Helicobacter pylori infection negatively influences pregnancy outcome in a mouse model. Helicobacter. 2004;9:152-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 86. | Gøbel R, Symonds EL, Butler RN, Tran CD. Association between Helicobacter pylori infection in mothers and birth weight. Dig Dis Sci. 2007;52:3049-3053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 87. | Brown S. Miscarriage and its associations. Semin Reprod Med. 2008;26:391-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 77] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 88. | Akcam M, Ozdem S, Yilmaz A, Gultekin M, Artan R. Serum ferritin, vitamin B(12), folate, and zinc levels in children infected with Helicobacter pylori. Dig Dis Sci. 2007;52:405-410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 89. | Serin E, Gümürdülü Y, Ozer B, Kayaselçuk F, Yilmaz U, Koçak R. Impact of Helicobacter pylori on the development of vitamin B12 deficiency in the absence of gastric atrophy. Helicobacter. 2002;7:337-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 48] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 90. | Avcu N, Avcu F, Beyan C, Ural AU, Kaptan K, Ozyurt M, Nevruz O, Yalçin A. The relationship between gastric-oral Helicobacter pylori and oral hygiene in patients with vitamin B12-deficiency anemia. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2001;92:166-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 91. | Ozer B, Serin E, Gumurdulu Y, Kayaselcuk F, Anarat R, Gur G, Kul K, Guclu M, Boyacioglu S. Helicobacter pylori eradication lowers serum homocysteine level in patients without gastric atrophy. World J Gastroenterol. 2005;11:2764-2767. [PubMed] |
| 92. | Golalipour MJ, Sedehi M, Qorbani M. Does maternal Helicobacter pylori infection increase the risk of occurrence of neural tube defects in newborns in Northern Iran? Neurosciences (Riyadh). 2012;17:219-225. [PubMed] |
| 93. | Sukenik-Halevy R, Ellis MH, Fejgin MD. Management of immune thrombocytopenic purpura in pregnancy. Obstet Gynecol Surv. 2008;63:182-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 94. | George JN, Woolf SH, Raskob GE, Wasser JS, Aledort LM, Ballem PJ, Blanchette VS, Bussel JB, Cines DB, Kelton JG. Idiopathic thrombocytopenic purpura: a practice guideline developed by explicit methods for the American Society of Hematology. Blood. 1996;88:3-40. [PubMed] |
| 95. | Belkin A, Levy A, Sheiner E. Perinatal outcomes and complications of pregnancy in women with immune thrombocytopenic purpura. J Matern Fetal Neonatal Med. 2009;22:1081-1085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 96. | Kohda K, Kuga T, Kogawa K, Kanisawa Y, Koike K, Kuroiwa G, Hirayama Y, Sato Y, Niitsu Y. Effect of Helicobacter pylori eradication on platelet recovery in Japanese patients with chronic idiopathic thrombocytopenic purpura and secondary autoimmune thrombocytopenic purpura. Br J Haematol. 2002;118:584-588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 98] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 97. | Hino M, Yamane T, Park K, Takubo T, Ohta K, Kitagawa S, Higuchi K, Arakawa T. Platelet recovery after eradication of Helicobacter pylori in patients with idiopathic thrombocytopenic purpura. Ann Hematol. 2003;82:30-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 63] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 98. | Suzuki T, Matsushima M, Masui A, Watanabe K, Takagi A, Ogawa Y, Shirai T, Mine T. Effect of Helicobacter pylori eradication in patients with chronic idiopathic thrombocytopenic purpura-a randomized controlled trial. Am J Gastroenterol. 2005;100:1265-1270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 92] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 99. | Yamaoka Y, Kodama T, Gutierrez O, Kim JG, Kashima K, Graham DY. Relationship between Helicobacter pylori iceA, cagA, and vacA status and clinical outcome: studies in four different countries. J Clin Microbiol. 1999;37:2274-2279. [PubMed] |
| 100. | Fukui O, Shimoya K, Shimizu T, Fukuda H, Wasada K, Murata Y. Helicobacter pylori infection and platelet counts during pregnancy. Int J Gynaecol Obstet. 2005;89:26-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 101. | Epstein A, Wing DA, Ouzounian JG, Miller DA, Lee RH. Helicobacter pylori and thrombocytopenia in the pregnant hispanic population. J Matern Fetal Neonatal Med. 2012;25:2588-2590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 102. | Zhou X, Zhang C, Wu J, Zhang G. Association between Helicobacter pylori infection and diabetes mellitus: a meta-analysis of observational studies. Diabetes Res Clin Pract. 2013;99:200-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 97] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 103. | Roubaud Baudron C, Franceschi F, Salles N, Gasbarrini A. Extragastric diseases and Helicobacter pylori. Helicobacter. 2013;18 Suppl 1:44-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 104. | Goldenberg RL, Culhane JF, Iams JD, Romero R. Epidemiology and causes of preterm birth. Lancet. 2008;371:75-84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4905] [Cited by in RCA: 5177] [Article Influence: 304.5] [Reference Citation Analysis (0)] |
| 105. | Yang YJ, Sheu BS, Lee SC, Yang HB, Wu JJ. Children of Helicobacter pylori-infected dyspeptic mothers are predisposed to H. pylori acquisition with subsequent iron deficiency and growth retardation. Helicobacter. 2005;10:249-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 42] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 106. | Lee JU, Kim O. Natural maternal transmission of H. pylori in Mongolian gerbils. World J Gastroenterol. 2006;12:5663-5667. [PubMed] |
| 107. | Gold BD, Khanna B, Huang LM, Lee CY, Banatvala N. Helicobacter pylori acquisition in infancy after decline of maternal passive immunity. Pediatr Res. 1997;41:641-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 59] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 108. | Blanchard TG, Czinn SJ, Maurer R, Thomas WD, Soman G, Nedrud JG. Urease-specific monoclonal antibodies prevent Helicobacter felis infection in mice. Infect Immun. 1995;63:1394-1399. [PubMed] |
| 109. | Thomas JE, Austin S, Dale A, McClean P, Harding M, Coward WA, Weaver LT. Protection by human milk IgA against Helicobacter pylori infection in infancy. Lancet. 1993;342:121. [PubMed] |
| 110. | Bhuiyan TR, Saha A, Lundgren A, Qadri F, Svennerholm AM. Immune responses to Helicobacter pylori infection in Bangladeshi children during their first two years of life and the association between maternal antibodies and onset of infection. J Infect Dis. 2010;202:1676-1684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 111. | Weyermann M, Borowski C, Bode G, Gürbüz B, Adler G, Brenner H, Rothenbacher D. Helicobacter pylori-specific immune response in maternal serum, cord blood, and human milk among mothers with and without current Helicobacter pylori infection. Pediatr Res. 2005;58:897-902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 112. | Rothenbacher D, Bode G, Brenner H. History of breastfeeding and Helicobacter pylori infection in pre-school children: results of a population-based study from Germany. Int J Epidemiol. 2002;31:632-637. [PubMed] |
| 113. | Winbery SL, Blaho KE. Dyspepsia in pregnancy. Obstet Gynecol Clin North Am. 2001;28:333-350. [PubMed] |
| 114. | Feldman M, Cryer B, Lee E, Peterson WL. Role of seroconversion in confirming cure of Helicobacter pylori infection. JAMA. 1998;280:363-365. [PubMed] |
| 115. | Gisbert JP, de la Morena F, Abraira V. Accuracy of monoclonal stool antigen test for the diagnosis of H. pylori infection: a systematic review and meta-analysis. Am J Gastroenterol. 2006;101:1921-1930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 187] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 116. | Braden B. Methods and functions: Breath tests. Best Pract Res Clin Gastroenterol. 2009;23:337-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 65] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 117. | Bentur Y, Matsui D, Koren G. Safety of 14C-UBT for diagnosis of Helicobacter pylori infection in pregnancy. Can Fam Physician. 2009;55:479-480. [PubMed] |
| 118. | Bazzoli F, Pozzato P, Rokkas T. Helicobacter pylori: the challenge in therapy. Helicobacter. 2002;7 Suppl 1:43-49. [PubMed] |
| 119. | Hayakawa S, Nakajima N, Karasaki-Suzuki M, Yoshinaga H, Arakawa Y, Satoh K, Yamamoto T. Frequent presence of Helicobacter pylori genome in the saliva of patients with hyperemesis gravidarum. Am J Perinatol. 2000;17:243-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 120. | Strachan BK, Jokhi RP, Filshie GM. Persistent hyperemesis gravidarum and Helicobacter pylori. J Obstet Gynaecol. 2000;20:427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 121. | Gill SK, O’Brien L, Einarson TR, Koren G. The safety of proton pump inhibitors (PPIs) in pregnancy: a meta-analysis. Am J Gastroenterol. 2009;104:1541-1545; quiz 1540, 1546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 94] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 122. | Mahadevan U. Gastrointestinal medications in pregnancy. Best Pract Res Clin Gastroenterol. 2007;21:849-877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 1.5] [Reference Citation Analysis (2)] |
| 123. | Every AL. Key host-pathogen interactions for designing novel interventions against Helicobacter pylori. Trends Microbiol. 2013;21:253-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 124. | Zhang YY, Xia HH, Zhuang ZH, Zhong J. Review article: ‘true’ re-infection of Helicobacter pylori after successful eradication--worldwide annual rates, risk factors and clinical implications. Aliment Pharmacol Ther. 2009;29:145-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 2.3] [Reference Citation Analysis (0)] |