Published online Aug 7, 2014. doi: 10.3748/wjg.v20.i29.10128
Revised: January 29, 2014
Accepted: April 8, 2014
Published online: August 7, 2014
Processing time: 236 Days and 9.9 Hours
AIM: To determine the clinical effectiveness of intense psychological support to physical activity (PA) in nonalcoholic fatty liver disease (NAFLD), compared with cognitive-behavioral treatment (CBT).
METHODS: Twenty-two NAFLD cases received support to exercise, tailored to their motivational needs (PA group). The effects on body weight, physical fitness [6-min walk test, VO2max and the PA-rating (PA-R) questionnaire] and body fat (fatty liver indices and visceral adiposity index) were compared with data obtained in 44 NAFLD subjects enrolled in a CBT program for weight loss, after adjustment for propensity score, calculated on baseline data. Measurements were performed at baseline, at 4-mo and one-year follow-up. Changes in anthropometric, biochemical and PA parameters were tested by repeated measurement ANOVA. Outcome results were tested by logistic regression analysis.
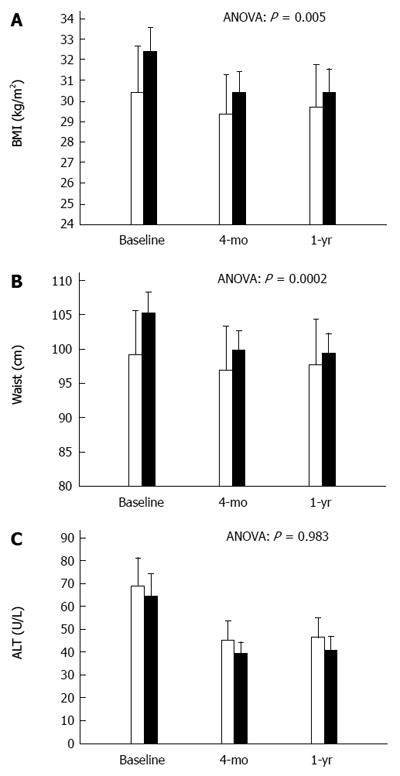
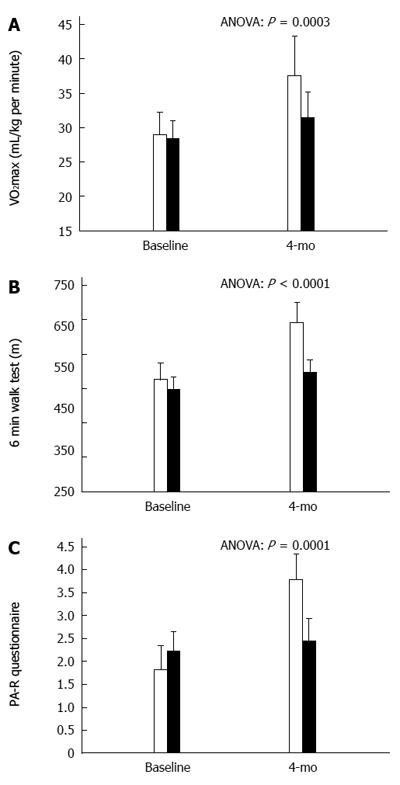
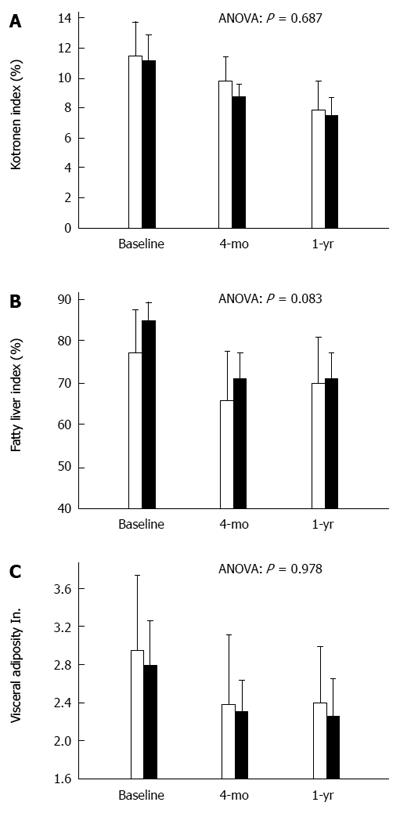
RESULTS: At the end of the intensive program, BMI was less significantly reduced in the PA group (-1.09 ± 1.68 kg/m2vs -2.04 ± 1.42 kg/m2 in the CBT group, P = 0.019) and the difference was maintained at 1-year follow-up (-0.73 ± 1.63 vs -1.95 ± 1.88, P = 0.012) (ANOVA, P = 0.005). PA-R was similar at baseline, when only 14% of cases in PA and 36% in CBT (P = 0.120) recorded values ≥ 3. At 4 mo, a PA-R ≥ 3 was registered in 91% of PA and 46% of CBT, respectively (P < 0.001) and PA-R ≥ 5 (up to 3 h/wk of moderate-to-heavy intensity physical activity) was registered in 41% of PA and only 9% of CBT group (P < 0.007). The 6-min walk test increased by 139 ± 26 m in PA and by only 43 ± 38 m in CBT (P < 0.001) and VO2max by 8.2 ± 3.8 mL/kg per minute and 3.3 ± 2.7 mL/kg per minute, respectively (P < 0.002). After adjustment for propensity, weight loss > 7% was significantly associated with CBT group at one year (OR = 6.21; 95%CI: 1.23-31.30), whereas PA-R > 3 was associated with PA group (10.31; 2.02-52.63). Liver enzymes decreased to values within normal limits in 36% of PA cases and 61% of CBT (P < 0.070). Estimated liver fat (Kotronen index) fell below the fatty liver threshold in 36% of PA and 34% and CBT cases at one-year (not different). Also the fatty liver index and the visceral adiposity index improved to a similar extent.
CONCLUSION: Intensive psychological counseling for PA produces hepatic effects not different from standard CBT, improving physical fitness and liver fat independent of weight loss. Strategies promoting exercise are worth and effective in motivated patients, particularly in lean NAFLD patients where large weight loss cannot be systematically pursued.
Core tip: The adherence to healthy diet is usually considered as part of effective treatment by patients with nonalcoholic fatty liver disease (NAFLD), whereas motivation to habitual physical activity is more difficult to pursue. We recently developed a specific psychological support program to regular physical activity to be proposed to NAFLD patients, particularly to those who failed or refused the classical weight loss approach. In a clinical audit, we compared the preliminary results obtained by the physical activity program with the data achieved in a group treated by the standard nutritional counseling, after adjusting for propensity score. The results indicate that physical activity may be implemented in motivated patients, with good results on physical fitness, liver enzymes and liver fat.
- Citation: Montesi L, Caselli C, Centis E, Nuccitelli C, Moscatiello S, Suppini A, Marchesini G. Physical activity support or weight loss counseling for nonalcoholic fatty liver disease? World J Gastroenterol 2014; 20(29): 10128-10136
- URL: https://www.wjgnet.com/1007-9327/full/v20/i29/10128.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i29.10128
Programs aimed at modifying unhealthy behaviors are mandatory in patients with nonalcoholic fatty liver disease (NAFLD) to reduce the risk of disease progression. Both epidemiological studies and several controlled clinical trials, either randomized[1] or non-randomized[2], have consistently demonstrated that weight loss and exercise reduce liver enzyme levels and have beneficial effects on a few histological outcomes: steatosis and necroinflammation are reduced, whereas minor or no effects on fibrosis are shown[3].
A recent metaanalysis of exercise trials has confirmed that physical activity per se is beneficial in NAFLD[4]. The effects of exercise may be independent of weight loss[5], but making NAFLD patients move is difficult. Although the majority of cases are well aware of the importance of physical activity, motivation to exercising is much lower than motivation to dieting[6]. NAFLD cases have scarce readiness to physical activity change, and 50% of cases were classified in either the precontemplation or contemplation stage of change[6], i.e., refractory to increase exercise.
Strategies to improve adherence to exercise have been described in the area of obesity[7] and in other metabolic diseases[8]. In the NAFLD area, physical activity counselling may be particularly difficult due the high prevalence of fatigue as the leading symptom[9], but the relatively young age of most NAFLD patients may favor this approach compared to dieting. In addition, physical activity, coupled with healthy food choices, remains the only option for the limited number of lean NAFLD patients who nonetheless have features of the metabolic syndrome[10]. Also in this area we need to move from the traditional prescriptive approach to an individualized, multidisciplinary, empowerment-based intervention, tailored on patients’ preferences[11].
We tested the effectiveness of a patient-tailored motivational and psychological support to physical activity on clinical and laboratory parameters associated with NAFLD. The results were compared with the results of a classical cognitive-behavior therapy (CBT) for weight loss, already active in our department, after matching by a propensity score approach.
The study was carried out in outpatients attending our tertiary-level unit for the diagnosis and treatment of NAFLD. The diagnosis was based on clinical, ultrasonographic and laboratory values, after exclusion of any other cause of chronic liver disease (hepatitis B and C virus, autoimmune disease) and alcohol intake not exceeding 20 g/d (confirmed by relatives).
After baseline assessment, patients were invited to choose between our standard, group-based program for weight loss or a pilot program, based on an intensive motivational and psychological support to exercise (experimental physical activity group - PA group), chaired by a psychologist. The reasons for preferring the activity group were either previous failures with the dietary approach (in other settings) or job constraints that made it impossible to participate to fixed group sessions. In the PA group, individual sessions of one hour were carried out every two weeks, to stimulate patients to increase their physical activity on the basis of monitored data (walking steps measured by a pedometer) and fitness tests (see below). Sessions were tailored to the motivational needs of each participant. After an initial assessment of patients’ readiness to change their physical activity level, based on the model of behavior change[12], a motivational approach was used to favor a more active lifestyle[13]. Instead of the common prescriptive approach, subjects were helped identify their preferred leisure-time activities (walking or riding the bike) and then supported in developing and implementing a weekly schedule. The sessions focused on how to deal with common psychological barriers to physical activity and on the importance of self-monitoring. Well-defined, reasonable goals were set during the sessions and the supervision of the weekly schedule was used to provide feedback and increase motivation and self-efficacy. The final goal was a habitual physical activity workload > 20 METs/h per week (corresponding to 3-h/wk moderate-intense physical activity). In addition, patients received one-hour counseling on healthy diet, based on an in-house developed manual, containing limited basic information on portion size, based on the nutrition pyramid, and examples to reduce food intake by a fixed amount of calories (maximum calorie reduction, 500 kcal/d). The program lasted 3-4 mo, depending on progress and the adoption of healthier lifestyles.
Subjects enrolled into this experimental program (n = 22) were compared with 44 consecutive NAFLD patients enrolled into a CBT program for weight loss during the same period, operative in our Unit since 2005[2]. Briefly, it consists of 13 weekly group sessions (15-20 subjects), 120 min each, chaired by physicians, dieticians, psychologists, education experts supported by a residential manual based on the principles of LEARN program for weight control[2]. Only one session is specifically devoted to exercise and its barriers.
In both groups, all measurements were repeated at the end of the intensive program (4 mo after the enrolling visit), as well after one year (excluding fitness values). Subjects with diabetes maintained their medications (metformin) throughout the study period.
The study was carried out as part of the clinical activity of the metabolic Unit and was approved by the Senior Staff Committee, an institution regulating non-interventional studies. At entry, all cases signed an informed consent to clinical studies.
Body weight, height, waist circumference and blood pressure were measured as previously reported[14]. Body mass index (BMI) is weight (kg) divided by squared height (m).
Plasma glucose, total and HDL-cholesterol, triglycerides and liver enzymes (aspartate and alanine aminotransferase (AST and ALT) and gamma-glutamyl-transpeptidase (GGT)) were measured by routine assays. Insulin resistance was calculated according to the homeostasis model assessment (HOMA) method[15].
Hepatic steatosis was quantitatively estimated by means of an algorithm (Kotronen index), based on clinical and biochemical parameters (presence of metabolic syndrome and type 2 diabetes, fasting insulin, AST and AST/ALT ratio), validated against proton magnetic resonance (fatty liver threshold, 5.56%)[16]. We also calculated the Fatty Liver Index (FLI)[17] and the Visceral Adipose Index (VAI)[18]. FLI measures the probability of significant steatosis; values < 30 rule out and values ≥ 60 rule in fatty liver. VAI is an indicator of visceral fat associated with cardiometabolic risk, with cut points derived from a Caucasian population[19].
Physical activity tests were performed at baseline and at the end of the intensive program. The time spent in exercise was graded according to the physical activity rating (PA-R) questionnaire[20]. Scores > 1 indicate regular participation in recreation or modest physical activity; scores > 3 indicate regular participation in moderate-to-intense physical activity (running or jogging, swimming, cycling, running in place) or any aerobic activity > 1 h/wk; scores > 5 extend aerobic activity up to 3 h/wk.
Physical fitness was tested by oxygen consumption (VO2max) on a treadmill [RunRace, Technogym, Gambettola (FC)], during four 5-min consecutive periods of exercise at graded heart rate (55%, 60%, 65% and 70% of maximal theoretical heart rate). VO2max was extrapolated by regression to maximum heart rate.
The 6-min walk test (the distance an individual is able to walk over 6 min on a flat surface) was used as a test of exercise capacity[21]. The subjects were allowed to self-pace and walk forth and back along a corridor inside the Unit.
Data were extracted from the database using specific subroutines and were analyzed by intention-to-treat, independently of the number of attained lifestyle sessions, using StatView 5.0™ (SAS Institute Inc., Cary, NC) and SPSS v11.0.4 (SPSS Inc., Chicago, IL). Unpaired t-test (2-tail), χ2-contingency test and Fisher’s exact test were used, whenever appropriate, to test baseline differences. The time-course of biochemical and clinical data were analyzed by repeated-measures, time x treatment ANOVA.
To adjust for baseline differences between groups, a propensity score approach was used. The propensity score was calculated by logistic regression analysis. The propensity score defines the conditional probability to be enrolled into to the self-adjusted PA program, compared to CBT, given a set of clinical and biochemical baseline values (BMI, ALT, individual features of MS) and personal characteristics (age, sex, marital status, educational level and occupation, Table 1). These last variables constitute potential barriers to treatment and are linked to the time constraints and to the economic burden of attending the program.
| PA Group(n = 22) | CBT Group(n = 44) | P value1 | |
| Male gender | 77% | 64% | 0.400 |
| Age (yr) | 48.2 ± 14.6 | 52.1 ± 10.5 | 0.208 |
| Education (primary/secondary/vocational/degree) | 9%/18%/59%/14% | 13%/18%/39%/30% | 0.213 |
| Civil status (single/married/widowed or divorced) | 45%/45%/9% | 70%/23%/7% | 0.241 |
| Profession (student/housewife/employee/self-employed/retired) | 13%/0%/55%/9%/23% | 2%/11%/48%/16%/23% | 0.309 |
| Body mass index (kg/m2) | 30.4 ± 5.0 | 32.4 ± 3.9 | 0.089 |
| Waist circumference (cm) | 99.0 ± 14.9 | 105.1 ± 10.4 | 0.058 |
| Blood glucose (mg/dL) | 114.0 ± 32.9 | 106.9 ± 26.1 | 0.343 |
| Insulin (μU/mL) | 19.1 ± 10.7 | 19.8 ± 7.6 | 0.773 |
| HOMA-IR | 5.36% ± 3.17% | 5.26% ± 2.45% | 0.889 |
| Total cholesterol (mg/dL) | 222 ± 47 | 229 ± 46 | 0.598 |
| HDL-cholesterol (mg/dL) | 49.4 ± 11.9 | 47.2 ± 9.4 | 0.419 |
| Triglycerides (mg/dL) | 211 ± 101 | 182 ± 82 | 0.214 |
| Systolic pressure (mmHg) | 128.3 ± 10.0 | 132.7 ± 12.8 | 0.169 |
| Diastolic pressure (mmHg) | 84.6 ± 8.3 | 84.8 ± 8.2 | 0.953 |
| Alanine aminotransferase (mU/mL) | 68.5 ± 28.5 | 64.1 ± 32.2 | 0.585 |
| Gamma-glutamyltransferases (mU/mL) | 76.0 ± 30.8 | 70.5 ± 35.4 | 0.534 |
| Diabetes | 33% | 30% | 0.979 |
| Hypertension | 50% | 39% | 0.614 |
| Treated with statins/fibrates | 5% | 18% | 0.310 |
| Kotronen index | 11.5% ± 5.3% | 11.2% ± 5.7% | 0.841 |
| Fatty liver index | 77.0% ± 23.7% | 84.7% ± 14.8% | 0.119 |
| Visceral adiposity index | 2.94 ± 1.83 | 2.79 ± 1.54 | 0.740 |
The propensity scores were used to adjust for baseline values in logistics regression analysis in three different models, having weight loss ≥ 7% initial body weight, PA-R ≥ 3 and ALT normalization as dependent variables. Two upper limits of ALT were considered: (1) the standard values used in our Departments (40 U/L); and (2) an updated definition of healthy range (< 31 U/L men; < 19 U/L women) proposed in 2002[22]. In the ALT model, the odds ratios were also corrected for changes in BMI and VO2max during the observation period.
As a clinical audit testing the feasibility and effectiveness of the psychological support program, no sample size calculation was carried out.
Data in text and in tables are given as mean ± SD or as percent. Given that 3 sets of data were analyzed, the significance level was set at P < 0.015.
The two groups were well balanced at baseline (Table 1). At the end of the intensive program, BMI was less reduced in the PA group (-1.09 ± 1.68 kg/m2vs -2.04 ± 1.42 kg/m2 in the CBT group, P = 0.019) and the difference was maintained at 1-year follow-up (-0.73 ± 1.63 vs -1.95 ± 1.88, P = 0.012) (time x treatment ANOVA; P = 0.005; Figure 1). Similar differences were observed in waist circumference (ANOVA, P = 0.0002). No systematic differences were observed in the time course of laboratory parameters and blood pressure. In the whole population, at 1-year follow-up glucose decreased by 8.6 mg/dL (P = 0.005), insulin by 4.1 U/L (P = 0.003), cholesterol by 13.6 mg/dL (P = 0.010), triglycerides by 28 mg/dL (P = 0.009), HOMA by 1.45% (P < 0.001), whereas HDL-cholesterol increased by 2.4 mg/dL (P = 0.011).
Both treatments similarly reduced ALT, both at 4-mo (PA group, -23.5 ± 26.1 U/L; CBT group, -24.6 ± 27.3 U/L) and at 1-year follow up (-22.3 ± 30.2 U/L and -23.3 ± 31.8 U/L, respectively).
No differences in PA-R were present at baseline (Figure 2), when only 14% of cases in PA and 36% in CBT (P = 0.120) recorded values ≥ 3. At 4 mo, a PA-R ≥ 3 was registered in 91% of PA and 46% of CBT, respectively (P < 0.001) and PA-R ≥ 5 (up to 3 h/wk of moderate-to-heavy intensity physical activity) was registered in 41% of PA and only 9% of CBT group (P < 0.007). The 6-min walk test increased by 139 ± 26 m in PA and by only 43 ± 38 m in CBT (P < 0.001) and VO2max by 8.2 ± 3.8 mL/kg per minute and 3.3 ± 2.7 mL/kg per minute, respectively (P < 0.002).
After adjustment for propensity score, the probability of weight loss > 7% was associated with CBT at 4 mo (OR = 2.52; 95%CI: 0.60-10.53) and at one year (OR = 6.21; 95%CI: 1.23-31.30). By contrast, the probability of PA-R > 3 at 4 mo was significantly associated with the PA group (OR = 10.31; 95%CI: 2.02-52.63). PA-R ≥ 5 was registered in 40% of PA and only 9% of CBT group (P = 0.017).
ALT levels within normal limits were recorded in 41% of cases in the PA group and 57% in the CBT at 4 mo (P = 0.298) and 36% and 61% at 1-year, respectively (P = 0.070). When the updated reference values of ALT were tested[22], only 4 cases in PA (18%) and 10 in CBT (23%) had ALT values within normal limits (P = 0.759).
The probability to achieve normal ALT at 4 mo was independent of program, after adjustment for propensity and changes in BMI and VO2max (CBT vs PA: OR = 1.35; 95%CI: 0.10-19.05), with a similar trend in favor of CBT also at one-year (OR = 1.76; 95%CI: 0.51-6.03). The use of updated ALT limit did not change the results.
No differences were present between groups in surrogate indices of fatty liver at baseline (Figure 3). The Kotronen index significantly decreased in both groups at one year (to 7.8% ± 4.4% and 7.5% ± 3.9% in PA and CBT, respectively; P < 0.001 for both), when liver triglycerides were estimated below the critical value of 5.56%, indicative of fatty liver, in 36% and 34% of cases (not different).
FLI decreased by 11.7% ± 13.9% in PA and by 13.7% ± 11.2% in CBT at 4 mo (P = 0.544) and by 7.5% ± 14.0% and 13.9% ± 11.4% at 1 year (P = 0.053), respectively. VAI decreased by -0.56 ± 1.36 vs -0.50 ± 1.42 at 4 mo (P = 0.884) and by -0.53 ± 1.32 vs -0.55 ± 1.32, at one year (P = 0.956) in PA and CBT, respectively.
The treatment produced a shift in the grading of fatty liver towards “indeterminate” (30% to 60% liver fat) in both PA (P = 0.007) and CBT (P < 0.001), as well as a down-shift in the VAI values across the range of cardiovascular risk (PA, P = 0.026; CBT, P = 0.001).
An intensive psychological counseling for physical activity reduces liver enzymes levels and surrogate markers of liver and visceral fat by an extent similar to that achieved by standard CBT, improving physical fitness independently of weight loss. These results are maintained at 1-year follow up and support the concept that specific activities aimed at promoting exercise are definitely worth implementing in motivated patients. A strategy combining physical activity and weight loss is likely to produce additional effects on liver fat and hepatocellular necrosis.
The difference in weight loss between the two programs was largely expected. The PA group received a very brief counseling on healthy diet (no more than one hour), compared with the extensive CBT training, where nutritional education was coupled with calorie counting, food diaries and behavioral strategies to reduce compulsive and emotional eating[2]. Weight loss at one year averaged 6% in CBT, a figure lower than the desirable 7% loss promoted by lifestyle programs[1,23,24], but sufficient to improve the cardiovascular risk profile. This weight loss target was neither expected nor reached in the PA group, where intervention did not focus on calorie restriction.
Several studies measured the combined effects of dietary-induced weight loss and increased physical activity, whereas the relative role of the two components has been less extensively studied. In older adults randomized to diet, exercise or both, body weight decreased on diet and on combined treatment, without any change in the exercise group[25]. In our experience, the effects of intervention on physical fitness mirrored those on weight loss. Both the 6-min walk test and VO2max increased on average by 25% in the PA group, significantly more than the modest 10% improvement observed with CBT, probably related to weight loss. This translated into decreased cardiovascular risk, as reported in other settings[26], where a dose-response relationship between the frequency and intensity of aerobic training (walking) and the increase in VO2max was recorded[27].
The study was specifically aimed at measuring the effects on the liver, although no attempt was made to investigate the possible molecular mechanism(s) responsible of weight loss and physical activity, respectively, which remain purely speculative. The effects of lifestyle modifications on NAFLD are well demonstrated[2], also on histology[1], but the specific effects of physical activity have been less clearly determined[28]. In subjects with type 2 diabetes, adding physical activity to diet decreased hepatocellular lipid content, measured by magnetic resonance spectroscopy (MRS) in a short-term study[29], and data were confirmed in a 6-mo trial[30]; aerobic and endurance exercise were both effective[31,32], and also an 8-wk resistance exercise in sedentary adults significantly reduced liver fat[33]. In a recent NAFLD study, regular aerobic exercise (30 min/d, at least 5 d/wk) decreased insulin resistance, liver enzymes and a second liver biopsy showed improved steatosis and necroinflammation in a subset of cases[34]. We confirmed the effects of physical activity on liver enzymes, similar to those observed with CBT. The surrogate indices of liver fat and visceral adiposity were all reduced, although not all cases reached the desired physical activity target. In particular, the estimated liver fat (Kotronen index) at baseline was similar to what measured in other NAFLD studies by 1H-MRS[16,35], and decreased by 30%-40% following both programs, as it occurred with a pure dietary approach[35]. Similarly, the FLI, that recently received external validation[36] and proved to correlate with the SteatoTest[37], improved with both programs.
The strength of this study is the novelty of the psychological support program, as well as the careful comparison between the two interventions by propensity score, to adjust for multiple baseline differences[38]. Both treatments were totally non-directive; subjects were invited to change their lifestyle according to individual preferences and progressively implemented either the physical activity program or the weight loss program following tailored, agreed decisions along the principles of behavior therapy.
The study has however limits. First, it was not randomized; this policy was selected to minimize dropout and to favor participation to lifestyle changes, a prerequisite to success, as demonstrated by the high rates of success in both groups (physical fitness and weight loss, respectively). Second, the study was carried out in a NAFLD group, attending a tertiary unit. The results might be different in the community, with less motivated patients. Third, the study used surrogate markers of liver fat; no attempt was made to prove the effectiveness on histological ground, already demonstrated in other studies[1,34]. In order to minimize the possible effects of concomitant drug treatment, all subjects were maintained on statin, antihypertensive and glucose-lowering agents in the course of the study, and no cases were being treated with anti-oxidants or hepatoprotective agents during the study period. However, this does not exclude a possible influence of external factors or concomitant diseases on liver enzymes and, according to accepted guidelines[39,40], the present results require histological confirmation in a prospective randomized trial. Fourth, the interaction between patients and therapists was very different (26 h as group sessions in CBT, only 6-8 h as individual sessions in PA), due to the different strategy used for the psychological support and the two interventions were not mutually exclusive; physical activity was also counseled in the CBT group (to a much lower extent), whereas the PA group received general counseling for healthy diet. Finally, the external validity of our experience might be limited by costs and difficulties in the organization of a psychological support in different settings. As previously suggested[11], liver units might find support in diabetes and obesity units, more prone to behavioral and psychological approaches, or in sports and recreational associations, where groups of leisure-time physical activity might be implemented.
In conclusion, in motivated individuals and in the presence of adequate support, physical activity may be proposed as a possible alternative to dieting to improve the clinical profile of NAFLD patients, considering that the beneficial effects largely extend outside liver disease to cardiovascular disease and well-being. A physical activity program might be particularly useful in the subgroup of lean NAFLD[10] and in males, more prone to physical activity, to correct the features of the metabolic syndrome with minor effects on body weight. It remains to be determined whether the results are maintained after the one-year follow-up and/or how much continuous support is needed.
In the behavioral treatment of nonalcoholic fatty liver disease (NAFLD), the role of nutritional counseling is well established, whereas physical activity remains more difficult to implement, because of stronger barriers from patients’ side and lower competence in therapists.
Motivation to habitual physical activity and its final metabolic effects on the liver remain areas of intense research.
A psychological support program may be effective to stimulate motivated patients to regular physical activity, to an extent promoting significant effects on visceral and liver fat. The final results on the liver are similar to those achieved by standard counseling to healthy lifestyle, largely focusing on reduced calorie intake and weight loss.
A physical activity program might be particularly useful in the subgroup of lean NAFLD individuals, to correct the features of the metabolic syndrome with minor effects on body weight. To reach this purpose, liver units might find support in diabetes and obesity units, more prone to behavioral and psychological approaches, or in sports and recreational associations, where groups of leisure-time physical activity might be implemented.
NAFLD: A liver disease (from pure steatosis to fibrosis and cirrhosis) of metabolic origin, mimicking the effects of alcohol on the liver, in the absence of significant alcohol intake.
The authors show that an intensive psychological counseling for physical activity reduces liver enzymes levels and surrogate markers of liver and visceral fat by an extent similar to those achieved by standard cognitive-behavioral therapy, improving physical fitness independently of weight loss. The issue, in general terms, is very interesting and has implications in terms of clinical impact. The methods are appropriate. The manuscript is well written and references up to date.
P- Reviewer: Carulli L, Loguercio C, Montecucco F, Muhammad A S- Editor: Ma YJ L- Editor: A E- Editor: Liu XM
| 1. | Promrat K, Kleiner DE, Niemeier HM, Jackvony E, Kearns M, Wands JR, Fava JL, Wing RR. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology. 2010;51:121-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 973] [Cited by in RCA: 971] [Article Influence: 64.7] [Reference Citation Analysis (1)] |
| 2. | Moscatiello S, Di Luzio R, Bugianesi E, Suppini A, Hickman IJ, Di Domizio S, Dalle Grave R, Marchesini G. Cognitive-behavioral treatment of nonalcoholic Fatty liver disease: a propensity score-adjusted observational study. Obesity (Silver Spring). 2011;19:763-770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 40] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 3. | Zelber-Sagi S, Ratziu V, Oren R. Nutrition and physical activity in NAFLD: an overview of the epidemiological evidence. World J Gastroenterol. 2011;17:3377-3389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 210] [Cited by in RCA: 224] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 4. | Keating SE, Hackett DA, George J, Johnson NA. Exercise and non-alcoholic fatty liver disease: a systematic review and meta-analysis. J Hepatol. 2012;57:157-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 400] [Cited by in RCA: 372] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 5. | St George A, Bauman A, Johnston A, Farrell G, Chey T, George J. Independent effects of physical activity in patients with nonalcoholic fatty liver disease. Hepatology. 2009;50:68-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 221] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 6. | Centis E, Moscatiello S, Bugianesi E, Bellentani S, Fracanzani AL, Calugi S, Petta S, Dalle Grave R, Marchesini G. Stage of change and motivation to healthier lifestyle in non-alcoholic fatty liver disease. J Hepatol. 2013;58:771-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 71] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 7. | Dalle Grave R, Calugi S, Centis E, El Ghoch M, Marchesini G. Cognitive-behavioral strategies to increase the adherence to exercise in the management of obesity. J Obes. 2011;2011:348293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 147] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 8. | Montesi L, Moscatiello S, Malavolti M, Marzocchi R, Marchesini G. Physical activity for the prevention and treatment of metabolic disorders. Intern Emerg Med. 2013;8:655-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 9. | Newton JL, Jones DE, Henderson E, Kane L, Wilton K, Burt AD, Day CP. Fatigue in non-alcoholic fatty liver disease (NAFLD) is significant and associates with inactivity and excessive daytime sleepiness but not with liver disease severity or insulin resistance. Gut. 2008;57:807-813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 145] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 10. | Younossi ZM, Stepanova M, Negro F, Hallaji S, Younossi Y, Lam B, Srishord M. Nonalcoholic fatty liver disease in lean individuals in the United States. Medicine (Baltimore). 2012;91:319-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 359] [Cited by in RCA: 400] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 11. | Bellentani S, Dalle Grave R, Suppini A, Marchesini G. Behavior therapy for nonalcoholic fatty liver disease: The need for a multidisciplinary approach. Hepatology. 2008;47:746-754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 177] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 12. | Prochaska JO, DiClemente CC. Transtheoretical therapy: toward a more integrative model of change. Psychotherapy. 1982;19:276-288. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2004] [Cited by in RCA: 2100] [Article Influence: 48.8] [Reference Citation Analysis (0)] |
| 13. | Miller WR, Rollnick S. Motivational Interviewing. New York: The Guilford Press 2002; . |
| 14. | Marchesini G, Bugianesi E, Forlani G, Cerrelli F, Lenzi M, Manini R, Natale S, Vanni E, Villanova N, Melchionda N. Nonalcoholic fatty liver, steatohepatitis, and the metabolic syndrome. Hepatology. 2003;37:917-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1907] [Cited by in RCA: 1917] [Article Influence: 87.1] [Reference Citation Analysis (0)] |
| 15. | Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412-419. [PubMed] |
| 16. | Kotronen A, Peltonen M, Hakkarainen A, Sevastianova K, Bergholm R, Johansson LM, Lundbom N, Rissanen A, Ridderstråle M, Groop L. Prediction of non-alcoholic fatty liver disease and liver fat using metabolic and genetic factors. Gastroenterology. 2009;137:865-872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 490] [Cited by in RCA: 600] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 17. | Bedogni G, Bellentani S, Miglioli L, Masutti F, Passalacqua M, Castiglione A, Tiribelli C. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006;6:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1238] [Cited by in RCA: 2029] [Article Influence: 106.8] [Reference Citation Analysis (0)] |
| 18. | Amato MC, Giordano C, Galia M, Criscimanna A, Vitabile S, Midiri M, Galluzzo A. Visceral Adiposity Index: a reliable indicator of visceral fat function associated with cardiometabolic risk. Diabetes Care. 2010;33:920-922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 778] [Cited by in RCA: 1153] [Article Influence: 76.9] [Reference Citation Analysis (0)] |
| 19. | Amato MC, Giordano C, Pitrone M, Galluzzo A. Cut-off points of the visceral adiposity index (VAI) identifying a visceral adipose dysfunction associated with cardiometabolic risk in a Caucasian Sicilian population. Lipids Health Dis. 2011;10:183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 163] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 20. | Jackson AS, Blair SN, Mahar MT, Wier LT, Ross RM, Stuteville JE. Prediction of functional aerobic capacity without exercise testing. Med Sci Sports Exerc. 1990;22:863-870. [PubMed] |
| 21. | Guyatt GH, Thompson PJ, Berman LB, Sullivan MJ, Townsend M, Jones NL, Pugsley SO. How should we measure function in patients with chronic heart and lung disease? J Chronic Dis. 1985;38:517-524. [PubMed] |
| 22. | Prati D, Taioli E, Zanella A, Della Torre E, Butelli S, Del Vecchio E, Vianello L, Zanuso F, Mozzi F, Milani S. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann Intern Med. 2002;137:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1025] [Cited by in RCA: 1049] [Article Influence: 45.6] [Reference Citation Analysis (4)] |
| 23. | Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346:393-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13206] [Cited by in RCA: 12413] [Article Influence: 539.7] [Reference Citation Analysis (1)] |
| 24. | Wing RR, Lang W, Wadden TA, Safford M, Knowler WC, Bertoni AG, Hill JO, Brancati FL, Peters A, Wagenknecht L. Benefits of modest weight loss in improving cardiovascular risk factors in overweight and obese individuals with type 2 diabetes. Diabetes Care. 2011;34:1481-1486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1082] [Cited by in RCA: 1246] [Article Influence: 89.0] [Reference Citation Analysis (0)] |
| 25. | Villareal DT, Chode S, Parimi N, Sinacore DR, Hilton T, Armamento-Villareal R, Napoli N, Qualls C, Shah K. Weight loss, exercise, or both and physical function in obese older adults. N Engl J Med. 2011;364:1218-1229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 710] [Cited by in RCA: 758] [Article Influence: 54.1] [Reference Citation Analysis (0)] |
| 26. | Goodpaster BH, Delany JP, Otto AD, Kuller L, Vockley J, South-Paul JE, Thomas SB, Brown J, McTigue K, Hames KC. Effects of diet and physical activity interventions on weight loss and cardiometabolic risk factors in severely obese adults: a randomized trial. JAMA. 2010;304:1795-1802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 403] [Cited by in RCA: 364] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 27. | Duncan GE, Anton SD, Sydeman SJ, Newton RL, Corsica JA, Durning PE, Ketterson TU, Martin AD, Limacher MC, Perri MG. Prescribing exercise at varied levels of intensity and frequency: a randomized trial. Arch Intern Med. 2005;165:2362-2369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 115] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 28. | Johnson NA, Keating SE, George J. Exercise and the liver: implications for therapy in fatty liver disorders. Semin Liver Dis. 2012;32:65-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 29. | Tamura Y, Tanaka Y, Sato F, Choi JB, Watada H, Niwa M, Kinoshita J, Ooka A, Kumashiro N, Igarashi Y. Effects of diet and exercise on muscle and liver intracellular lipid contents and insulin sensitivity in type 2 diabetic patients. J Clin Endocrinol Metab. 2005;90:3191-3196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 234] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 30. | Larson-Meyer DE, Newcomer BR, Heilbronn LK, Volaufova J, Smith SR, Alfonso AJ, Lefevre M, Rood JC, Williamson DA, Ravussin E. Effect of 6-month calorie restriction and exercise on serum and liver lipids and markers of liver function. Obesity (Silver Spring). 2008;16:1355-1362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 152] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 31. | Devries MC, Samjoo IA, Hamadeh MJ, Tarnopolsky MA. Effect of endurance exercise on hepatic lipid content, enzymes, and adiposity in men and women. Obesity (Silver Spring). 2008;16:2281-2288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 62] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 32. | van der Heijden GJ, Wang ZJ, Chu ZD, Sauer PJ, Haymond MW, Rodriguez LM, Sunehag AL. A 12-week aerobic exercise program reduces hepatic fat accumulation and insulin resistance in obese, Hispanic adolescents. Obesity (Silver Spring). 2010;18:384-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 170] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 33. | Hallsworth K, Fattakhova G, Hollingsworth KG, Thoma C, Moore S, Taylor R, Day CP, Trenell MI. Resistance exercise reduces liver fat and its mediators in non-alcoholic fatty liver disease independent of weight loss. Gut. 2011;60:1278-1283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 388] [Cited by in RCA: 353] [Article Influence: 25.2] [Reference Citation Analysis (2)] |
| 34. | Bhat G, Baba CS, Pandey A, Kumari N, Choudhuri G. Life style modification improves insulin resistance and liver histology in patients with non-alcoholic fatty liver disease. World J Hepatol. 2012;4:209-217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 35. | Jiralerspong S, Kim ES, Dong W, Feng L, Hortobagyi GN, Giordano SH. Obesity, diabetes, and survival outcomes in a large cohort of early-stage breast cancer patients. Ann Oncol. 2013;24:2506-2514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 136] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 36. | Koehler EM, Schouten JN, Hansen BE, Hofman A, Stricker BH, Janssen HL. External validation of the fatty liver index for identifying nonalcoholic fatty liver disease in a population-based study. Clin Gastroenterol Hepatol. 2013;11:1201-1204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 243] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 37. | Zelber-Sagi S, Webb M, Assy N, Blendis L, Yeshua H, Leshno M, Ratziu V, Halpern Z, Oren R, Santo E. Comparison of fatty liver index with noninvasive methods for steatosis detection and quantification. World J Gastroenterol. 2013;19:57-64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 87] [Cited by in RCA: 108] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 38. | Rubin DB. The design versus the analysis of observational studies for causal effects: parallels with the design of randomized trials. Stat Med. 2007;26:20-36. [PubMed] |
| 39. | Ratziu V, Bellentani S, Cortez-Pinto H, Day C, Marchesini G. A position statement on NAFLD/NASH based on the EASL 2009 special conference. J Hepatol. 2010;53:372-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 723] [Cited by in RCA: 789] [Article Influence: 52.6] [Reference Citation Analysis (1)] |
| 40. | Chalasani N, Younossi Z, Lavine JE, Diehl AM, Brunt EM, Cusi K, Charlton M, Sanyal AJ. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Gastroenterological Association, American Association for the Study of Liver Diseases, and American College of Gastroenterology. Gastroenterology. 2012;142:1592-1609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1226] [Cited by in RCA: 1355] [Article Influence: 104.2] [Reference Citation Analysis (4)] |