Published online Jan 14, 2014. doi: 10.3748/wjg.v20.i2.384
Revised: November 5, 2013
Accepted: November 12, 2013
Published online: January 14, 2014
Processing time: 114 Days and 10.8 Hours
Irritable bowel syndrome (IBS) is a common gastrointestinal disorder that is generally considered to be functional because there appears to be no associated anatomical defect. Stress and psychological factors are thought to play an important role in IBS. The gut neuroendocrine system (NES), which regulates all functions of the gastrointestinal tract, consists of endocrine cells that are scattered among the epithelial cells of the mucosa, and the enteric nervous system. Although it is capable of operating independently from the central nervous system (CNS), the gut NES is connected to and modulated by the CNS. This review presents evidence for the presence of an anatomical defect in IBS patients, namely in the gastrointestinal endocrine cells. These cells have specialized microvilli that project into the lumen and function as sensors for the luminal content and respond to luminal stimuli by releasing hormones into the lamina propria, which starts a chain reaction that progresses throughout the entire NES. The changes in the gastrointestinal endocrine cells observed in IBS patients are highly consistent with the other abnormalities reported in IBS patients, such as visceral hypersensitivity, dysmotility, and abnormal secretion.
Core tip: This review presents recent observations in irritable bowel syndrome (IBS) patients that point toward the existence of an anatomical defect in the gastrointestinal endocrine cells. It includes also an argument that IBS is an organic disorder and that the abnormalities in the gastrointestinal endocrine cells can explain the visceral hypersensitivity, dysmotility and abnormal secretion reported in these patients.
- Citation: El-Salhy M, Gundersen D, Gilja OH, Hatlebakk JG, Hausken T. Is irritable bowel syndrome an organic disorder? World J Gastroenterol 2014; 20(2): 384-400
- URL: https://www.wjgnet.com/1007-9327/full/v20/i2/384.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i2.384
Irritable bowel syndrome (IBS) is a common chronic gastrointestinal disorder with a reported prevalence of 5%-20% and an incidence of about 200 per 100000 of the world population[1-29]. Patients with IBS suffer from recurrent abdominal pain/discomfort and altered bowel habit, which vary in both degree and time pattern between patients: from tolerable to severe, and from daily symptoms to intervals of weeks/months, respectively[3,11,30-40]. IBS is more common in females than males, and in patients younger than 50 years of age[3,11,14,15,19,21,26,30,31,33-40].
IBS is not associated with the development of serious disease or with an excessive mortality rate[41,42]. However, it considerably reduces the sufferer’s quality of life, interfering with their education, working ability and social life. Moreover, IBS represents an economic burden to both patients and society[23,43-49], since IBS patients visit their doctors more frequently, undergo more diagnostic tests, consume more medications, miss more workdays, are less productive at work, and are hospitalized more frequently than those without IBS[28,32,39,50-53].
There are no biomarkers for the diagnosis of IBS[54,55], which is instead based on the assessment of symptoms such as the Rome III criteria[56,57]. IBS patients are subgrouped according to differences in the predominant bowel symptoms as IBS-diarrhea (D), IBS-constipation (C), IBS-both diarrhea and constipation (M), and nonsubtyped IBS (patients with insufficient abnormality of stool consistency to meet the criteria for IBS-C, -D, or -M)[56,57]. The Rome criteria were introduced to facilitate positive diagnoses, reduce the costs due to unnecessary testing, and guide treatment; however, they fall short on these expectations and are generally neglected in clinical practice by both general practitioners and gastroenterologists[54,58-64].
IBS is considered to be a functional disorder in the absence of a known anatomic defect[65], the pathophysiology of which is incompletely understood. The pathogenesis appears to be multifactorial, with several factors suggested to play a role in the process, such as psychological factors, genetic factors, low-grade chronic intestinal inflammation, an abnormal gut neuroendocrine system (NES) and/or altered signaling in this system, dietary factors, and intestinal flora[66].
IBS patients can be roughly divided into two subsets: sporadic (nonspecific) and postinfectious (PI-IBS)/inflammatory bowel disease (IBD)-associated (IBD-IBS)[66]. Sporadic IBS includes patients who have had symptoms for a long time and without any associated events, in particular gastrointestinal or other infections. PI-IBS is defined as a sudden onset of IBS symptoms following gastroenteritis in individuals who have previously had no gastrointestinal complaints, and IBD-IBS is defined in IBD patients in remission who display IBS symptoms[66]. PI-IBS constitutes about 6%-17% of patients with IBS[67], and IBD-IBS occurs in 33%-46% of ulcerative colitis patients and 42%-60% of those with Crohn’s disease[68-72].
This article summarizes the published findings on abnormalities in the gut neuroendocrine cells, discusses them in view of the currently known facts about IBS, and presents an argument for IBS being an organic gastrointestinal disorder.
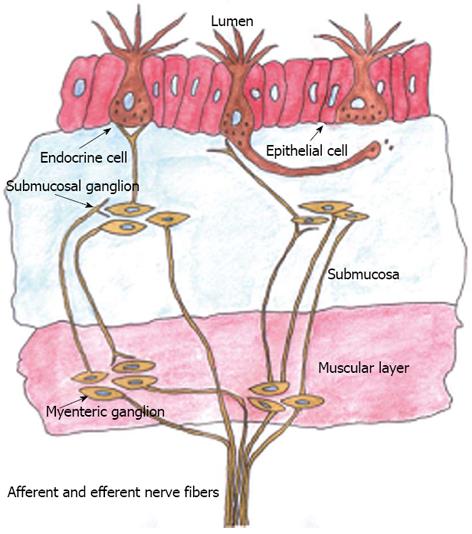
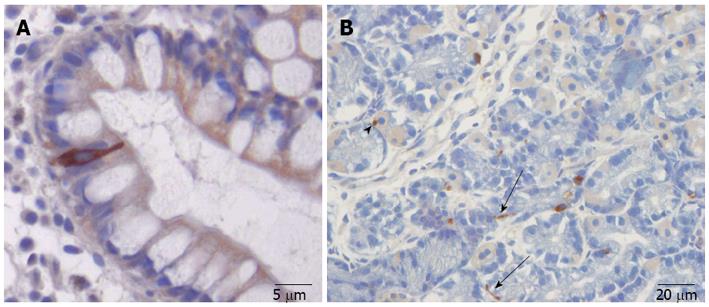
The gut contains a large number of endocrine cells that are dispersed among the epithelial cells of the gut mucosa in all of the gut segments except for the esophagus[66,73-78]. These cells constitute the largest endocrine organ in the body and comprise about 1% of all epithelial cells in the gastrointestinal tract, where they are separated from one another by epithelial cells[73,74,79-81]. These cells have specialized microvilli that project into the lumen and function as sensors for the gut contents and respond to luminal stimuli by releasing hormones that, in general, target other parts of the digestive system (Figure 1)[82-94]. There are at least 15 different populations of endocrine cells in the gastrointestinal tract[60,73-76]. Some of them [including somatostatin and peptide YY (PYY) cells] have long slender cytoplasmic processes that project toward neighboring cells, increasing their paracrine effects (Figure 2)[95]. The distribution, functions, and modes of action of the most important endocrine/paracrine cells are given in Table 1[60,75,76,96-108].
| Cell content | Localization | Source of release | Actions |
| Serotonin (5-HT) | EC cells in the stomach, large and small intestines | Noradrenalin; acetylcholine; acidification and intraluminal pressure | Inhibits gastric emptying and stimulates colonic motility; accelerates small- and large-intestine transit activates the submucosal sensory branch of the enteric nervous system that conveys sensation from the gut to the central nervous system |
| Histamine | EC-like cells in the gastric oxyntic mucosa | Vagus nerve stimulation and inhibited by somatostatin | Stimulates gastric acid secretion |
| Somatostatin | The stomach, and large and small intestines | Mixed meal and acidification of the stomach | Inhibits intestinal contraction; inhibits gut exocrine and neuroendocrine secretion |
| Ghrelin | Gastric oxyntic mucosa | Protein and fat ingestion; suppressed by carbohydrate ingestion | Increases appetite and food intake; stimulates gastric and intestinal motility |
| Gastrin | Gastric antral mucosa | Intraluminal peptides; amino acids; calcium; amines; low pH and prostaglandins. Somatostatin inhibits release | Stimulates gastric acid secretion and histamine release; trophic action on gastric mucosa; stimulates contraction of the LES and antrum |
| CCK | Small intestine, especially the duodenum | Intraluminal protein and fat and inhibited by somatostatin | Inhibits gastric emptying; stimulates gallbladder contraction and intestinal motility; stimulates pancreatic exocrine secretion and growth; regulates food intake |
| Secretin | Small intestine, especially the duodenum | Acidification and inhibited by somatostatin | Stimulates pancreatic bicarbonate and fluid secretion; inhibits gastric emptying; inhibits contractile activity of the small and large intestines |
| GIP | Small intestine, especially the duodenum | Intraluminal glucose; amino acids and fat | Incretin action; inhibits gastric acid secretion |
| Motilin | Small intestine, especially the jejunum | Protein and fat ingestion | Induces phase-III migrating motor complex; stimulates gastric emptying; stimulates contraction of the LES |
| Neurotensin | Small intestine | Fat | Stimulates pancreatic section; inhibits gastric secretion; delays gastric emptying; stimulates colon motility |
| PYY | Terminal ileum and large intestine | Protein-rich meals | Delays gastric emptying; inhibits gastric and pancreatic secretion, stimulates the absorption of water and electrolytes; major mediator of the ileal brake |
| PP | Terminal ileum and large intestine | Protein-rich meals | Inhibits pancreatic secretion; stimulates gastric acid secretion; relaxes the gallbladder; stimulates motility of the stomach and small intestine |
| Enteroglucagon (oxyntomodulin) | Terminal ileum and large intestine | Intraluminal carbohydrates and fat | Inhibits gastric and pancreatic secretion; reduces gastric motility; has some incretin effect |
| Chromogranin | All gastrointestinal tract segments | Ingestion of a meal | Induces formation, sorting, and release of secretory granules of the gut endocrine/paracrine cells; an inflammatory mediator |
Some of the different endocrine cell types are located in specific areas of the gut, while others (primarily somatostatin and serotonin cells) are found throughout the gut[73,74,76]. They secrete one or more signaling substances into the lamina propria, where they exert their action locally on nearby structures (autocrine/paracrine mode) and/or via an endocrine mode of action (by circulating in the blood to reach distant targets)[109]. These cells interact in an integrated manner with each other and with the enteric nervous system (ENS) and the afferent and efferent nerve fibers of the central nervous system (CNS), in particular the autonomic nervous system[60,76,99,110]. All of the cell types in the crypt/villus originate from pluripotent stem cells of endodermal origin[73,74,111]. Each intestinal crypt contains four to six stem cells that differentiate into all epithelial cell types including enterocytes, goblet cells, Paneth cells, and endocrine cells[112-125]. These cells regulate several functions of the gastrointestinal tract, including sensation, motility, secretion, absorption, local immune defense, and food intake (by affecting the appetite)[60,73,74,76,110].
Several abnormalities have been reported in all segments of the gastrointestinal tract of patients with IBS. As mentioned above, the endocrine cells exert their effects in part locally; however for some of them the endocrine mode of action is difficult to elucidate[99]. One example of this is the serotonin cells. The serotonin that they secrete is taken up into the blood and carried by platelets as they circulate through the gut[126-129]. Thus, the circulating serotonin is locked within the dense granules of the platelets, without any possibility of being delivered to distant targets. Therefore, summarizing and discussing abnormalities in the endocrine cells are considered separately herein relative to the various segments of the gastrointestinal tract.
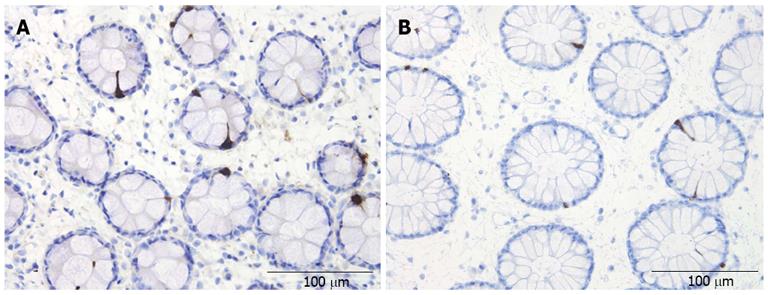
Abnormal endocrine/paracrine cells have been found in the stomach (Figure 3), proximal small intestine (duodenum), distal small intestine (ileum), colon (Figure 4), and rectum of patients with sporadic IBS[130-141]. These abnormalities manifest mostly as changes in the densities of these cells (i.e., an anatomical defect). The abnormalities in the different endocrine cells in the various segments of the gastrointestinal tract of patients with sporadic IBS are summarized in Table 2. In addition to the abnormalities observed in the endocrine cells, there are alterations in the levels of serotonin transporter (SERT), which appear to be increased in the ileum and decreased in the rectum of IBS patients[130,141,142].
| Gastrointestinal segment | Endocrine cell type | IBS-D | IBS-M | IBS-C |
| Stomach | ||||
| Oxyntic mucosa | Ghrelin | High | Normal | Low |
| Serotonin | High | Normal | Low | |
| Somatostatin | Low | Low | High | |
| Chromogranin A | Normal | Normal | High | |
| Antrum | Serotonin | Normal | Low | High |
| Gastrin | High | High | High | |
| Somatostatin | Low | Low | Low | |
| Chromogranin A | Normal | Low | High | |
| Small intestine | ||||
| Duodenum | Serotonin | Normal | - | Normal |
| CCK | Low | - | Normal | |
| Secretin | Low | - | Normal | |
| GIP | Low | - | Low | |
| Somatostatin | Low | - | Low | |
| Chromogranin A | Low | - | Low | |
| Ileum | Serotonin | Low | Low | Low |
| PYY | Normal | Normal | High | |
| Chromogranin A | Low | Low | Low | |
| Large intestine | ||||
| Colon | Serotonin | Low | - | Low |
| PYY | Low | - | Low | |
| Chromogranin A | Low | - | Low | |
| Rectum | Serotonin | Normal | - | Normal |
| PYY | Low | - | Low | |
| Enteroglucagon | Low | - | Low | |
| Somatostatin | High | - | High | |
| Chromogranin A | Normal | - | Normal | |
Similar to sporadic IBS, abnormal endocrine/paracrine cell densities have been found in both PI-IBS and IBD-IBS. However, the nature of these abnormalities is different from those in sporadic IBS (Table 3)[141,143-150].
| Gastrointestinal segment | Endocrine cell type | PI-IBS | IBD-IBS |
| Small intestine | |||
| Duodenum | Serotonin | High | - |
| CCK | High | - | |
| Large intestine | |||
| Serotonin | High | High/low | |
| PYY | High | Low | |
| PP | - | Low | |
| Enteroglucagon | - | High | |
The mechanisms regulated by gastrointestinal endocrine cells include gut sensation, motility, and secretion. IBS patients exhibit visceral hypersensitivity, disturbed gastrointestinal motility, and abnormal gut secretion[65,107,151-153]. The degree to which the abnormalities in these cells observed in IBS patients contribute to these disturbed functions is discussed to below.
Visceral hypersensitivity has been demonstrated in the colorectal segment of IBS patients[154-161]. Hypersensitivity has also been reported in the esophagus, stomach, and small intestine[162-166]. However, visceral hypersensitivity is not present in all IBS patients, and a large prospective study found that only 20% of IBS patients showed hypersensitivity[167]. Furthermore, visceral hypersensitivity does not seem to be a panintestinal disorder[165]; IBS patients only appear to exhibit rectal hypersensitivity[159]. Whether the severity of abdominal pain is correlated with colorectal hypersensitivity in IBS remains a matter of controversy[154,156,168-172].
As mentioned above, serotonin cells have specialized microvilli that project into the gut lumen and act as sensors for the gut contents, and in particular for increased pressure. Serotonin is released in a regulated and calcium-dependent manner from serotonin cells into the surrounding tissues in response to luminal stimuli[173,174]. It activates the sensory branch of the ENS, which is localized in the submucosal plexus in the submucosa, and the myenteric plexus in between the smooth muscle fibers. These sensory branches convey sensation from the gut to the CNS through the sympathetic and parasympathetic nervous systems (Figure 1)[175-177]. The pain stimuli activate the cerebral cortex through the thalamus and permit the recognition of visceral pain[65,177]. Some studies have found IBS patients to be tolerant of somatic pain, and hence the hypersensitivity is confined to the viscera[158,165,178], while other studies found IBS patients to have a lower tolerance to somatic pain than healthy subjects[162,179,180]. Azpiroz et al[167] postulated that the exclusive visceral hypersensitivity experienced by some IBS patients could be attributable to abnormalities at the level of the gut, spinal cord, or brain, whereas patients with both visceral and somatic hypersensitivities have a disturbance above the gut level. Those authors also argued that a peripheral mechanism is involved in the visceral hypersensitivity in IBS.
The data presented in Table 2 suggest that none of the abnormalities in the gut endocrine cells could possibly contribute to the development of the visceral hypersensitivity seen in some sporadic IBS patients. However, it has been reported that SERT levels are increased in the ileum and reduced in the rectum of these patients[130,141,142]. The gut mucosa has a high SERT-producing capacity, since all of the epithelial cells lining the luminal surface of the gut express SERT[142,181]. A reduction in SERT results in impaired intracellular uptake and degradation in the gut epithelial cells, consequently increasing the availability of serotonin within the gut mucosa[182,183]. Considering that the serotonin cell density in sporadic IBS does not differ from that of a healthy subject, a decrease in SERT would markedly increase the amount of serotonin available at its receptors[141,142]. An increase in serotonin at the 5-hydroxytryptamine (5-HT)3 receptors of the ENS sensory neurons would activate the sensory nerves, which would then transmit nociceptive information to the nervous system[99]. Conversely, duodenal and rectal serotonin cell densities are high in PI-IBS patients, possibly contributing to the development of visceral hypersensitivity.
Dysmotility has been reported to occur in all segments of the gastrointestinal tract of patients with IBS, but mostly in the small and large intestines[151,153]. Some studies found lower pressures in the lower esophageal sphincter and abnormal esophageal contractions in IBS patients[184,185]; however, such esophageal motility abnormalities were not confirmed in other studies[186,187]. In addition, some authors have reported abnormal gastric emptying in patients with IBS[153,188-193], while others did not find any such abnormality in these patients[161,194-197]. It therefore seems that abnormalities of gastric emptying do not occur in all IBS patients. Furthermore, while IBS-C patients often exhibit delayed gastric emptying, rapid gastric emptying is found in IBS-D patients[153,189].
The ghrelin cell density in the gastric oxyntic mucosa is low and the serotonin cell density in the antrum of the stomach is high in IBS-C patients, while in IBS-D patients the ghrelin cell density in the gastric oxyntic mucosa is high and the densities of cholecystokinin (CCK) and secretin cells are reduced in the duodenum[132]. Ghrelin is a peptide hormone that was first isolated from the stomach, and originates mostly from endocrine cells in the oxyntic mucosa of the stomach, although small amounts are expressed in the small intestine, large intestine, and the arcuate nucleus of the hypothalamus[198-200]. Ghrelin has several functions, and plays a role in regulating the release of growth hormone from the pituitary gland, which increases appetite and feeding, and also plays a major role in energy metabolism[201-204]. Furthermore, ghrelin has been found to accelerate gastric and small- and large-intestine motility[83,110,205-214]. Serotonin acts on 5-HT1p receptors, which are located on a subset of inhibitory motor neurons of the myenteric plexus, relaxing the stomach via a nitrergic pathway and delaying gastric emptying[98,215-217]. CCK relaxes the proximal stomach in order to increase its reservoir capacity, and inhibits gastric emptying[218-220]. Secretin also inhibits gastric emptying[76,221]. It is therefore conceivable that low gastric ghrelin and high serotonin contribute to the slow gastric emptying in IBS-C, while the high gastric ghrelin and low intestinal CCK and secretin contribute to the rapid gastric emptying in IBS-D.
Several studies have found small-bowel transit to be delayed in IBS-C and accelerated in IBS-D[195,222-226]. However, a study from the Mayo clinic found no overall association between these IBS subgroups[161]. Studies on the motor patterns of the small bowel in IBS yielded contradictory results, which is probably due to marked inter- and intraindividual variations of small-intestine motor patterns[194,227-243]. As mentioned above, ghrelin cell density is low in the gastric oxyntic mucosa and PYY cell density is high in the ileum of IBS-C patients. Since ghrelin stimulates small-intestine motility and PYY stimulates the absorption of water and electrolytes, and is a major regulator of the ileal brake[244-249]. Moreover, it inhibits prostaglandin E2 and vasoactive intestinal polypeptide (which stimulate intestinal fluid secretion)[250-252], it is possible that the abnormalities in gastric ghrelin and ileal PYY contribute to the slow small-intestine transit in IBS-C. Secretin inhibits the contractile activity of the small intestine, and so the high ghrelin cell density and low duodenal secretin cell density may play a role in the rapid small-intestine transit in IBS-D.
It has been reported by some that colorectal transit is delayed in IBS-C and accelerated in IBS-D[153,222,223,253-258]. However, others have found that the colorectal transit time does not differ between IBS patients and controls[225,253,254]. The myoelectric and motor patterns of the large intestine of IBS patients have been investigated by several studies, which have yielded contradictory results[196,254-279]. In IBS-C, ghrelin cell density in the gastric oxyntic mucosa is low and ileal PYY cell density is high. Given that ghrelin stimulates intestinal motility and PYY stimulates the ileal-break, these abnormalities in ghrelin and PYY may promote the delayed colorectal transit observed in some IBS-C patients. CCK and secretin inhibit intestinal motility, and the cell densities of both are low in the duodenum of IBS-D patients. These factors together with a high gastric ghrelin cell density may contribute to the development of the accelerated colorectal transit seen in IBS-D patients.
In PI-IBS, the serotonin cell densities are high both in the small and large intestines, and CCK cell density is high in the small intestine. Serotonin primarily targets the mucosal projections of the intrinsic primary afferent neurons, which initiate peristaltic and secretory reflexes[156-161,175,280-289]. As mentioned above, CCK stimulates intestinal motility; thus, high serotonin and CCK levels could be responsible for the diarrhea seen in PI-IBS.
Few studies have investigated gastrointestinal secretion in IBS patients. Enhanced intestinal secretion in response to bile acid perfusion in the ileum has been reported in these patients[290]. Increased reactivity of the secretory component of the migrating motor complex has been observed in the small intestine of IBS patients, and especially in those with IBS-D[291]. Among the abnormalities in the gut endocrine cells in IBS patients listed in Table 2, the low duodenal CCK and secretin observed in IBS-D, and the high ileal PYY cell density observed in IBS-C are particularly interesting with respect to gut secretions. CCK stimulates the secretion of digestive enzymes from pancreatic exocrine glands, and secretin stimulates pancreatic bicarbonate and fluid secretions[218,219]. The secretion of pancreatic bicarbonate increases the pH of the gut contents, which are highly acidic after leaving the stomach, and PYY stimulates the absorption of water and electrolytes[76]. This change in pH is essential for lipid digestion, since pancreatic lipase is irreversibly inactivated below pH 4.0[218]. It is tempting to speculate that IBS-D patients could suffer from fat maldigestion and a functional pancreatic insufficiency. Indeed, pancreatic enzyme substitution and a low-fat diet have been applied in clinical practice for these patients, with some success[292]. Moreover, an increase in PYY in the ileum of IBS-C patients may result in increased absorption of water from the feces, resulting in hard feces that worsen their to obstipation (Figure 4).
In conclusion, there are sufficient grounds to suspect that the abnormalities in the gastrointestinal endocrine cells play a role in the development of visceral hypersensitivity, gastrointestinal dysmotility, and abnormal gastrointestinal secretion.
It has long been considered that IBS is caused by psychological stress and/or brain dysfunction, and it is overrepresented in patients with psychiatric illness/and or sexually and/or physically abused individuals. During the past decade there has been rapid progress in our understanding of IBS, and there is accumulating evidence of a biological etiology for this condition. Research to establish effective treatments for IBS have been intensified, and societal attitudes toward IBS patients are slowly changing.
This review presents evidence for an anatomic defect in IBS patients, namely the gastrointestinal endocrine cells. However, the data presented on the gastrointestinal endocrine cells in sporadic IBS were obtained by only two research groups. Further studies performed by other researchers involving different patient cohorts are needed before these observations can be confirmed. Conversely, while the data for PI-IBS were reported by several research groups from different countries and related to different patient cohorts, studies on PI-IBS have focused mainly on serotonin and are mostly restricted to the rectum. Further studies of other endocrine cells in different segments of the gastrointestinal tract are needed in PI-IBS. It should be noted that the gastrointestinal endocrine cells interact in an integrated manner with each other and the ENS, and together constitute the so-called neuroendocrine regulatory system of the gut[76,293-295]. It is thus possible that IBS patients have an abnormality in the ENS, in addition to those in the endocrine cells. However, investigating the ENS is very difficult since it would require whole-wall biopsy sampling under laparoscopic control, which represents a risk for both patients and controls. Regardless of the ethical issues this raises, it is unlikely that either patients or healthy subjects would voluntarily submit to laparoscopy and whole-wall biopsy sampling.
The abnormalities in the gut endocrine cells differ between sporadic IBS and PI-IBS/IBD-IBS, and their etiologies also appear to be different. Familial aggregation, twin, and genetic studies provide evidence for a genetic predisposition in sporadic IBS[296-306], and these patients describe their symptoms as commencing in childhood, suggesting the presence of genetically defective gastrointestinal endocrine cells. However, gastrointestinal mucosal cells - including the endocrine cells - have a rapid turnover, and it is also possible that factors related to luminal content such as diet or bacterial flora can provoke an increase or decrease in the endocrine cell population.
The etiology of the gastrointestinal endocrine cell abnormalities in PI-IBS and IBD-IBS appears to differ from that of sporadic IBS. Patients who develop PI-IBS and IBD-IBS likely have a genetic predisposition (host related) as well as other factors, such as infecting-organism-associated risk factors[307-315]. Following infection, these patients develop a low-grade inflammation that manifests as an increased intraepithelial and mucosal infiltration of lymphocytes and mast cells[143-149,316]. There is some evidence that inflammation and immune cells affect the gastrointestinal endocrine cells[317]. The secretion of serotonin by enterochromaffin (EC) cells can be enhanced or attenuated by the secretory products of immune cells, such as CD4+ T, and also modulates the immune response[126,317]. The EC cells are in contact with or very close to CD3+ and CD20+ lymphocytes, and several serotonergic receptors have been identified in lymphocytes, monocytes, macrophages, and dendritic cells[318]. Therefore, it is conceivable that the abnormalities in the gastrointestinal endocrine cells in PI-IBS and IBD-IBS are caused by endocrine/immune interactions (i.e., the endocrine/immune axis), which are in turn caused by low-grade inflammation in predisposed individuals[319,320].
The gut NES comprises the gastrointestinal endocrine cells and the ENS[76]. This regulatory system controls all gastrointestinal functions independently from the CNS[76,156]. However, the gut NES and the CNS are connected, and the CNS modulates the gastrointestinal functions through this connection[152]. Thus, a defect in the gut NES should be suspected in patients with IBS[76,294]. The gastrointestinal endocrine cells serve as chemical and mechanical transducers for afferent projections to the ENS, and subsequently to the CNS[294,321]. The present review describes evidence in the literature of an anatomic defect in the NES in IBS, namely defective gastrointestinal endocrine cells. Therefore, in line with some other gastroenterologists, we consider it highly likely that IBS is an organic disorder[107].
The endocrine cells interact in an integrated manner with each other. It is possible that the abnormality in many endocrine cells of the gut seen in IBS is caused by a defect in one or more endocrine cell types, which in turn results in changes in the other endocrine cell types.
P- Reviewers: Akiho H, Prasad KK S- Editor: Gou SX L- Editor: A E- Editor: Wu HL
| 1. | Quigley EM, Locke GR, Mueller-Lissner S, Paulo LG, Tytgat GN, Helfrich I, Schaefer E. Prevalence and management of abdominal cramping and pain: a multinational survey. Aliment Pharmacol Ther. 2006;24:411-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 2. | Vandvik PO, Lydersen S, Farup PG. Prevalence, comorbidity and impact of irritable bowel syndrome in Norway. Scand J Gastroenterol. 2006;41:650-656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 102] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 3. | Drossman DA, Li Z, Andruzzi E, Temple RD, Talley NJ, Thompson WG, Whitehead WE, Janssens J, Funch-Jensen P, Corazziari E. U.S. householder survey of functional gastrointestinal disorders. Prevalence, sociodemography, and health impact. Dig Dis Sci. 1993;38:1569-1580. [PubMed] |
| 4. | Saito YA, Schoenfeld P, Locke GR. The epidemiology of irritable bowel syndrome in North America: a systematic review. Am J Gastroenterol. 2002;97:1910-1915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 157] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 5. | Thompson WG, Irvine EJ, Pare P, Ferrazzi S, Rance L. Functional gastrointestinal disorders in Canada: first population-based survey using Rome II criteria with suggestions for improving the questionnaire. Dig Dis Sci. 2002;47:225-235. [PubMed] |
| 6. | Zhao Y, Zou D, Wang R, Ma X, Yan X, Man X, Gao L, Fang J, Yan H, Kang X. Dyspepsia and irritable bowel syndrome in China: a population-based endoscopy study of prevalence and impact. Aliment Pharmacol Ther. 2010;32:562-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 65] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 7. | Boyce PM, Koloski NA, Talley NJ. Irritable bowel syndrome according to varying diagnostic criteria: are the new Rome II criteria unnecessarily restrictive for research and practice? Am J Gastroenterol. 2000;95:3176-3183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 153] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 8. | Barbezat G, Poulton R, Milne B, Howell S, Fawcett JP, Talley N. Prevalence and correlates of irritable bowel symptoms in a New Zealand birth cohort. N Z Med J. 2002;115:U220. [PubMed] |
| 9. | Mearin F, Badía X, Balboa A, Baró E, Caldwell E, Cucala M, Díaz-Rubio M, Fueyo A, Ponce J, Roset M. Irritable bowel syndrome prevalence varies enormously depending on the employed diagnostic criteria: comparison of Rome II versus previous criteria in a general population. Scand J Gastroenterol. 2001;36:1155-1161. [PubMed] |
| 10. | Boekema PJ, van Dam van Isselt EF, Bots ML, Smout AJ. Functional bowel symptoms in a general Dutch population and associations with common stimulants. Neth J Med. 2001;59:23-30. [PubMed] |
| 11. | Agréus L, Svärdsudd K, Nyrén O, Tibblin G. Irritable bowel syndrome and dyspepsia in the general population: overlap and lack of stability over time. Gastroenterology. 1995;109:671-680. [PubMed] |
| 12. | Hillilä MT, Färkkilä MA. Prevalence of irritable bowel syndrome according to different diagnostic criteria in a non-selected adult population. Aliment Pharmacol Ther. 2004;20:339-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 102] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 13. | Kay L, Jørgensen T, Jensen KH. The epidemiology of irritable bowel syndrome in a random population: prevalence, incidence, natural history and risk factors. J Intern Med. 1994;236:23-30. [PubMed] |
| 14. | Hoseini-Asl MK, Amra B. Prevalence of irritable bowel syndrome in Shahrekord, Iran. Indian J Gastroenterol. 2003;22:215-216. [PubMed] |
| 15. | Karaman N, Türkay C, Yönem O. Irritable bowel syndrome prevalence in city center of Sivas. Turk J Gastroenterol. 2003;14:128-131. [PubMed] |
| 16. | Celebi S, Acik Y, Deveci SE, Bahcecioglu IH, Ayar A, Demir A, Durukan P. Epidemiological features of irritable bowel syndrome in a Turkish urban society. J Gastroenterol Hepatol. 2004;19:738-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 17. | Masud MA, Hasan M, Khan AK. Irritable bowel syndrome in a rural community in Bangladesh: prevalence, symptoms pattern, and health care seeking behavior. Am J Gastroenterol. 2001;96:1547-1552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 83] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 18. | Huerta I, Valdovinos MA, Schmulson M. Irritable bowel syndrome in Mexico. Dig Dis. 2001;19:251-257. [PubMed] |
| 19. | Kwan AC, Hu WH, Chan YK, Yeung YW, Lai TS, Yuen H. Prevalence of irritable bowel syndrome in Hong Kong. J Gastroenterol Hepatol. 2002;17:1180-1186. [PubMed] |
| 20. | Lau EM, Chan FK, Ziea ET, Chan CS, Wu JC, Sung JJ. Epidemiology of irritable bowel syndrome in Chinese. Dig Dis Sci. 2002;47:2621-2624. [PubMed] |
| 21. | Schlemper RJ, van der Werf SD, Vandenbroucke JP, Biemond I, Lamers CB. Peptic ulcer, non-ulcer dyspepsia and irritable bowel syndrome in The Netherlands and Japan. Scand J Gastroenterol Suppl. 1993;200:33-41. [PubMed] |
| 22. | Ho KY, Kang JY, Seow A. Prevalence of gastrointestinal symptoms in a multiracial Asian population, with particular reference to reflux-type symptoms. Am J Gastroenterol. 1998;93:1816-1822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 206] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 23. | Xiong LS, Chen MH, Chen HX, Xu AG, Wang WA, Hu PJ. A population-based epidemiologic study of irritable bowel syndrome in South China: stratified randomized study by cluster sampling. Aliment Pharmacol Ther. 2004;19:1217-1224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 97] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 24. | Gwee KA, Wee S, Wong ML, Png DJ. The prevalence, symptom characteristics, and impact of irritable bowel syndrome in an asian urban community. Am J Gastroenterol. 2004;99:924-931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 142] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 25. | Rajendra S, Alahuddin S. Prevalence of irritable bowel syndrome in a multi-ethnic Asian population. Aliment Pharmacol Ther. 2004;19:704-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 26. | Jafri W, Yakoob J, Jafri N, Islam M, Ali QM. Irritable bowel syndrome and health seeking behaviour in different communities of Pakistan. J Pak Med Assoc. 2007;57:285-287. [PubMed] |
| 27. | Jafri W, Yakoob J, Jafri N, Islam M, Ali QM. Frequency of irritable bowel syndrome in college students. J Ayub Med Coll Abbottabad. 2005;17:9-11. [PubMed] |
| 28. | Boivin M. Socioeconomic impact of irritable bowel syndrome in Canada. Can J Gastroenterol. 2001;15 Suppl B:8B-11B. [PubMed] |
| 29. | Locke GR, Yawn BP, Wollan PC, Melton LJ, Lydick E, Talley NJ. Incidence of a clinical diagnosis of the irritable bowel syndrome in a United States population. Aliment Pharmacol Ther. 2004;19:1025-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 51] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 30. | Thompson WG, Heaton KW. Functional bowel disorders in apparently healthy people. Gastroenterology. 1980;79:283-288. [PubMed] |
| 31. | Kennedy TM, Jones RH, Hungin AP, O’flanagan H, Kelly P. Irritable bowel syndrome, gastro-oesophageal reflux, and bronchial hyper-responsiveness in the general population. Gut. 1998;43:770-774. [PubMed] |
| 32. | Talley NJ, Gabriel SE, Harmsen WS, Zinsmeister AR, Evans RW. Medical costs in community subjects with irritable bowel syndrome. Gastroenterology. 1995;109:1736-1741. [PubMed] |
| 33. | Hungin AP, Whorwell PJ, Tack J, Mearin F. The prevalence, patterns and impact of irritable bowel syndrome: an international survey of 40,000 subjects. Aliment Pharmacol Ther. 2003;17:643-650. [PubMed] |
| 34. | Jones R, Lydeard S. Irritable bowel syndrome in the general population. BMJ. 1992;304:87-90. [PubMed] |
| 35. | Bordie AK. Functional disorders of the colon. J Indian Med Assoc. 1972;58:451-456. [PubMed] |
| 36. | O’Keefe EA, Talley NJ, Zinsmeister AR, Jacobsen SJ. Bowel disorders impair functional status and quality of life in the elderly: a population-based study. J Gerontol A Biol Sci Med Sci. 1995;50:M184-M189. [PubMed] |
| 37. | Everhart JE, Renault PF. Irritable bowel syndrome in office-based practice in the United States. Gastroenterology. 1991;100:998-1005. [PubMed] |
| 38. | Wilson S, Roberts L, Roalfe A, Bridge P, Singh S. Prevalence of irritable bowel syndrome: a community survey. Br J Gen Pract. 2004;54:495-502. [PubMed] |
| 39. | Harvey RF, Salih SY, Read AE. Organic and functional disorders in 2000 gastroenterology outpatients. Lancet. 1983;1:632-634. [PubMed] |
| 40. | Spiegel BM. The burden of IBS: looking at metrics. Curr Gastroenterol Rep. 2009;11:265-269. [PubMed] |
| 41. | Harvey RF, Mauad EC, Brown AM. Prognosis in the irritable bowel syndrome: a 5-year prospective study. Lancet. 1987;1:963-965. [PubMed] |
| 42. | Sloth H, Jørgensen LS. Chronic non-organic upper abdominal pain: diagnostic safety and prognosis of gastrointestinal and non-intestinal symptoms. A 5- to 7-year follow-up study. Scand J Gastroenterol. 1988;23:1275-1280. [PubMed] |
| 43. | Gralnek IM, Hays RD, Kilbourne AM, Chang L, Mayer EA. Racial differences in the impact of irritable bowel syndrome on health-related quality of life. J Clin Gastroenterol. 2004;38:782-789. [PubMed] |
| 44. | Gralnek IM, Hays RD, Kilbourne A, Naliboff B, Mayer EA. The impact of irritable bowel syndrome on health-related quality of life. Gastroenterology. 2000;119:654-660. [PubMed] |
| 45. | Whitehead WE, Burnett CK, Cook EW, Taub E. Impact of irritable bowel syndrome on quality of life. Dig Dis Sci. 1996;41:2248-2253. [PubMed] |
| 46. | Pace F, Molteni P, Bollani S, Sarzi-Puttini P, Stockbrügger R, Bianchi Porro G, Drossman DA. Inflammatory bowel disease versus irritable bowel syndrome: a hospital-based, case-control study of disease impact on quality of life. Scand J Gastroenterol. 2003;38:1031-1038. [PubMed] |
| 47. | Drossman DA, Morris CB, Schneck S, Hu YJ, Norton NJ, Norton WF, Weinland SR, Dalton C, Leserman J, Bangdiwala SI. International survey of patients with IBS: symptom features and their severity, health status, treatments, and risk taking to achieve clinical benefit. J Clin Gastroenterol. 2009;43:541-550. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 177] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 48. | Coffin B, Dapoigny M, Cloarec D, Comet D, Dyard F. Relationship between severity of symptoms and quality of life in 858 patients with irritable bowel syndrome. Gastroenterol Clin Biol. 2004;28:11-15. [PubMed] |
| 49. | Li FX, Patten SB, Hilsden RJ, Sutherland LR. Irritable bowel syndrome and health-related quality of life: a population-based study in Calgary, Alberta. Can J Gastroenterol. 2003;17:259-263. [PubMed] |
| 50. | Sandler RS, Everhart JE, Donowitz M, Adams E, Cronin K, Goodman C, Gemmen E, Shah S, Avdic A, Rubin R. The burden of selected digestive diseases in the United States. Gastroenterology. 2002;122:1500-1511. [PubMed] |
| 51. | Peery AF, Dellon ES, Lund J, Crockett SD, McGowan CE, Bulsiewicz WJ, Gangarosa LM, Thiny MT, Stizenberg K, Morgan DR. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology. 2012;143:1179-87.e1-3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1355] [Cited by in RCA: 1467] [Article Influence: 112.8] [Reference Citation Analysis (1)] |
| 52. | Spanier JA, Howden CW, Jones MP. A systematic review of alternative therapies in the irritable bowel syndrome. Arch Intern Med. 2003;163:265-274. [PubMed] |
| 53. | Gaburri M, Bassotti G, Bacci G, Cinti A, Bosso R, Ceccarelli P, Paolocci N, Pelli MA, Morelli A. Functional gut disorders and health care seeking behavior in an Italian non-patient population. Recenti Prog Med. 1989;80:241-244. [PubMed] |
| 54. | Barbara G, Stanghellini V. Biomarkers in IBS: when will they replace symptoms for diagnosis and management? Gut. 2009;58:1571-1575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 32] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 55. | Spiller RC. Potential biomarkers. Gastroenterol Clin North Am. 2011;40:121-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 56. | Longstreth GF, Thompson WG, Chey WD, Houghton LA, Mearin F, Spiller RC. Functional bowel disorders. Gastroenterology. 2006;130:1480-1491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3413] [Cited by in RCA: 3383] [Article Influence: 178.1] [Reference Citation Analysis (1)] |
| 57. | Spiller R, Aziz Q, Creed F, Emmanuel A, Houghton L, Hungin P, Jones R, Kumar D, Rubin G, Trudgill N. Guidelines on the irritable bowel syndrome: mechanisms and practical management. Gut. 2007;56:1770-1798. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 609] [Cited by in RCA: 538] [Article Influence: 29.9] [Reference Citation Analysis (1)] |
| 58. | Quigley EM. The ‘con’ case. The Rome process and functional gastrointestinal disorders: the barbarians are at the gate! Neurogastroenterol Motil. 2007;19:793-797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 59. | Camilleri M. Do the Symptom-Based, Rome Criteria of Irritable Bowel Syndrome Lead to Better Diagnosis and Treatment Outcomes? The Con Argument. Clin Gastroenterol Hepatol. 2009;8:129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 60. | El-Salhy M. Irritable bowel syndrome: diagnosis and pathogenesis. World J Gastroenterol. 2012;18:5151-5163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 77] [Cited by in RCA: 125] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 61. | Gladman LM, Gorard DA. General practitioner and hospital specialist attitudes to functional gastrointestinal disorders. Aliment Pharmacol Ther. 2003;17:651-654. [PubMed] |
| 62. | Lea R, Hopkins V, Hastleton J, Houghton LA, Whorwell PJ. Diagnostic criteria for irritable bowel syndrome: utility and applicability in clinical practice. Digestion. 2004;70:210-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 45] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 63. | Thompson WG, Heaton KW, Smyth GT, Smyth C. Irritable bowel syndrome in general practice: prevalence, characteristics, and referral. Gut. 2000;46:78-82. [PubMed] |
| 64. | Corsetti M, Tack J. Are symptom-based diagnostic criteria for irritable bowel syndrome useful in clinical practice? Digestion. 2004;70:207-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 65. | Karantanos T, Markoutsaki T, Gazouli M, Anagnou NP, Karamanolis DG. Current insights in to the pathophysiology of Irritable Bowel Syndrome. Gut Pathog. 2010;2:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 66. | El-Salhy M, Gundersen D, Hatlebakk JG, Hausken T. Irritable bowel syndrome: diagnosis, pathogenesis and treatment options. New York: Nova Science Publishers, Inc 2012; . |
| 67. | Longstreth GF, Hawkey CJ, Mayer EA, Jones RH, Naesdal J, Wilson IK, Peacock RA, Wiklund IK. Characteristics of patients with irritable bowel syndrome recruited from three sources: implications for clinical trials. Aliment Pharmacol Ther. 2001;15:959-964. [PubMed] |
| 68. | Isgar B, Harman M, Kaye MD, Whorwell PJ. Symptoms of irritable bowel syndrome in ulcerative colitis in remission. Gut. 1983;24:190-192. [PubMed] |
| 69. | Ansari R, Attari F, Razjouyan H, Etemadi A, Amjadi H, Merat S, Malekzadeh R. Ulcerative colitis and irritable bowel syndrome: relationships with quality of life. Eur J Gastroenterol Hepatol. 2008;20:46-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 56] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 70. | Simrén M, Axelsson J, Gillberg R, Abrahamsson H, Svedlund J, Björnsson ES. Quality of life in inflammatory bowel disease in remission: the impact of IBS-like symptoms and associated psychological factors. Am J Gastroenterol. 2002;97:389-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 250] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 71. | Minderhoud IM, Oldenburg B, Wismeijer JA, van Berge Henegouwen GP, Smout AJ. IBS-like symptoms in patients with inflammatory bowel disease in remission; relationships with quality of life and coping behavior. Dig Dis Sci. 2004;49:469-474. [PubMed] |
| 72. | Keohane J, O’Mahony C, O’Mahony L, O’Mahony S, Quigley EM, Shanahan F. Irritable bowel syndrome-type symptoms in patients with inflammatory bowel disease: a real association or reflection of occult inflammation? Am J Gastroenterol. 2010;105:1788, 1789-194; quiz 1795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 173] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 73. | May CL, Kaestner KH. Gut endocrine cell development. Mol Cell Endocrinol. 2010;323:70-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 86] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 74. | Gunawardene AR, Corfe BM, Staton CA. Classification and functions of enteroendocrine cells of the lower gastrointestinal tract. Int J Exp Pathol. 2011;92:219-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 201] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 75. | El-Salhy M, Ostgaard H, Gundersen D, Hatlebakk JG, Hausken T. The role of diet in the pathogenesis and management of irritable bowel syndrome (Review). Int J Mol Med. 2012;29:723-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 76. | El-Salhy M, Seim I, Chopin L, Gundersen D, Hatlebakk JG, Hausken T. Irritable bowel syndrome: the role of gut neuroendocrine peptides. Front Biosci (Elite Ed). 2012;4:2783-2800. [PubMed] |
| 77. | Tanaka-Shintani M, Watanabe M. Immunohistochemical study of enterochromaffin-like cell in human gastric mucosa. Pathol Int. 2007;57:572-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 78. | Lönroth H, Håkanson R, Lundell L, Sundler F. Histamine containing endocrine cells in the human stomach. Gut. 1990;31:383-388. [PubMed] |
| 79. | Buffa R, Capella C, Fontana P, Usellini L, Solcia E. Types of endocrine cells in the human colon and rectum. Cell Tissue Res. 1978;192:227-240. [PubMed] |
| 80. | Schonhoff SE, Giel-Moloney M, Leiter AB. Minireview: Development and differentiation of gut endocrine cells. Endocrinology. 2004;145:2639-2644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 236] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 81. | Sjölund K, Sandén G, Håkanson R, Sundler F. Endocrine cells in human intestine: an immunocytochemical study. Gastroenterology. 1983;85:1120-1130. [PubMed] |
| 82. | Sandström O, El-Salhy M. Ageing and endocrine cells of human duodenum. Mech Ageing Dev. 1999;108:39-48. [PubMed] |
| 83. | El-Salhy M. Ghrelin in gastrointestinal diseases and disorders: a possible role in the pathophysiology and clinical implications (review). Int J Mol Med. 2009;24:727-732. [PubMed] |
| 84. | Tolhurst G, Reimann F, Gribble FM. Intestinal sensing of nutrients. Handb Exp Pharmacol. 2012;309-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 68] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 85. | Lee J, Cummings BP, Martin E, Sharp JW, Graham JL, Stanhope KL, Havel PJ, Raybould HE. Glucose sensing by gut endocrine cells and activation of the vagal afferent pathway is impaired in a rodent model of type 2 diabetes mellitus. Am J Physiol Regul Integr Comp Physiol. 2012;302:R657-R666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 64] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 86. | Parker HE, Reimann F, Gribble FM. Molecular mechanisms underlying nutrient-stimulated incretin secretion. Expert Rev Mol Med. 2010;12:e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 107] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 87. | Raybould HE. Nutrient sensing in the gastrointestinal tract: possible role for nutrient transporters. J Physiol Biochem. 2008;64:349-356. [PubMed] |
| 88. | San Gabriel A, Nakamura E, Uneyama H, Torii K. Taste, visceral information and exocrine reflexes with glutamate through umami receptors. J Med Invest. 2009;56 Suppl:209-217. [PubMed] |
| 89. | Rudholm T, Wallin B, Theodorsson E, Näslund E, Hellström PM. Release of regulatory gut peptides somatostatin, neurotensin and vasoactive intestinal peptide by acid and hyperosmolal solutions in the intestine in conscious rats. Regul Pept. 2009;152:8-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 90. | Sternini C, Anselmi L, Rozengurt E. Enteroendocrine cells: a site of ‘taste’ in gastrointestinal chemosensing. Curr Opin Endocrinol Diabetes Obes. 2008;15:73-78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 306] [Cited by in RCA: 278] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 91. | Sternini C. Taste receptors in the gastrointestinal tract. IV. Functional implications of bitter taste receptors in gastrointestinal chemosensing. Am J Physiol Gastrointest Liver Physiol. 2007;292:G457-G461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 90] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 92. | Buchan AM. Nutrient Tasting and Signaling Mechanisms in the Gut III. Endocrine cell recognition of luminal nutrients. Am J Physiol. 1999;277:G1103-G1107. [PubMed] |
| 93. | Montero-Hadjadje M, Elias S, Chevalier L, Benard M, Tanguy Y, Turquier V, Galas L, Yon L, Malagon MM, Driouich A. Chromogranin A promotes peptide hormone sorting to mobile granules in constitutively and regulated secreting cells: role of conserved N- and C-terminal peptides. J Biol Chem. 2009;284:12420-12431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 58] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 94. | Shooshtarizadeh P, Zhang D, Chich JF, Gasnier C, Schneider F, Haïkel Y, Aunis D, Metz-Boutigue MH. The antimicrobial peptides derived from chromogranin/secretogranin family, new actors of innate immunity. Regul Pept. 2010;165:102-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 96. | Mawe GM, Coates MD, Moses PL. Review article: intestinal serotonin signalling in irritable bowel syndrome. Aliment Pharmacol Ther. 2006;23:1067-1076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 159] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 97. | Wade PR, Chen J, Jaffe B, Kassem IS, Blakely RD, Gershon MD. Localization and function of a 5-HT transporter in crypt epithelia of the gastrointestinal tract. J Neurosci. 1996;16:2352-2364. [PubMed] |
| 98. | Gershon MD, Tack J. The serotonin signaling system: from basic understanding to drug development for functional GI disorders. Gastroenterology. 2007;132:397-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 988] [Cited by in RCA: 1131] [Article Influence: 62.8] [Reference Citation Analysis (0)] |
| 99. | Gershon MD. 5-Hydroxytryptamine (serotonin) in the gastrointestinal tract. Curr Opin Endocrinol Diabetes Obes. 2013;20:14-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 457] [Cited by in RCA: 456] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 100. | Gershon MD. Serotonin is a sword and a shield of the bowel: serotonin plays offense and defense. Trans Am Clin Climatol Assoc. 2012;123:268-280; discussion 280. [PubMed] |
| 101. | El-Salhy M, Mazzawi T, Gundersen D, Hatlebakk JG, Hausken T. The role of peptide YY in gastrointestinal diseases and disorders (review). Int J Mol Med. 2013;31:275-282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 102. | Dubrasquet M, Bataille D, Gespach C. Oxyntomodulin (glucagon-37 or bioactive enteroglucagon): a potent inhibitor of pentagastrin-stimulated acid secretion in rats. Biosci Rep. 1982;2:391-395. [PubMed] |
| 103. | Schjoldager BT, Baldissera FG, Mortensen PE, Holst JJ, Christiansen J. Oxyntomodulin: a potential hormone from the distal gut. Pharmacokinetics and effects on gastric acid and insulin secretion in man. Eur J Clin Invest. 1988;18:499-503. [PubMed] |
| 104. | Schjoldager B, Mortensen PE, Myhre J, Christiansen J, Holst JJ. Oxyntomodulin from distal gut. Role in regulation of gastric and pancreatic functions. Dig Dis Sci. 1989;34:1411-1419. [PubMed] |
| 105. | Dakin CL, Small CJ, Batterham RL, Neary NM, Cohen MA, Patterson M, Ghatei MA, Bloom SR. Peripheral oxyntomodulin reduces food intake and body weight gain in rats. Endocrinology. 2004;145:2687-2695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 230] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 106. | Wynne K, Park AJ, Small CJ, Patterson M, Ellis SM, Murphy KG, Wren AM, Frost GS, Meeran K, Ghatei MA. Subcutaneous oxyntomodulin reduces body weight in overweight and obese subjects: a double-blind, randomized, controlled trial. Diabetes. 2005;54:2390-2395. [PubMed] |
| 107. | Camilleri M. Peripheral mechanisms in irritable bowel syndrome. N Engl J Med. 2012;367:1626-1635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 229] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 108. | Jianu CS, Fossmark R, Syversen U, Hauso Ø, Waldum HL. A meal test improves the specificity of chromogranin A as a marker of neuroendocrine neoplasia. Tumour Biol. 2010;31:373-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 109. | Rindi G, Inzani F, Solcia E. Pathology of gastrointestinal disorders. Endocrinol Metab Clin North Am. 2010;39:713-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 110. | Seim I, El-Salhy M, Hausken T, Gundersen D, Chopin L. Ghrelin and the brain-gut axis as a pharmacological target for appetite control. Curr Pharm Des. 2012;18:768-775. [PubMed] |
| 111. | Lee CS, Kaestner KH. Clinical endocrinology and metabolism. Development of gut endocrine cells. Best Pract Res Clin Endocrinol Metab. 2004;18:453-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 112. | Cardoso WV, Lü J. Regulation of early lung morphogenesis: questions, facts and controversies. Development. 2006;133:1611-1624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 423] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 113. | Darlington GJ. Molecular mechanisms of liver development and differentiation. Curr Opin Cell Biol. 1999;11:678-682. [PubMed] |
| 114. | Fausto N, Campbell JS, Riehle KJ. Liver regeneration. Hepatology. 2006;43:S45-S53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1126] [Cited by in RCA: 1205] [Article Influence: 63.4] [Reference Citation Analysis (0)] |
| 115. | Rawlins EL, Hogan BL. Ciliated epithelial cell lifespan in the mouse trachea and lung. Am J Physiol Lung Cell Mol Physiol. 2008;295:L231-L234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 224] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 116. | Zaret KS. Regulatory phases of early liver development: paradigms of organogenesis. Nat Rev Genet. 2002;3:499-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 385] [Cited by in RCA: 356] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 117. | Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters PJ. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature. 2007;449:1003-1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3854] [Cited by in RCA: 4334] [Article Influence: 240.8] [Reference Citation Analysis (0)] |
| 118. | Barker N, van de Wetering M, Clevers H. The intestinal stem cell. Genes Dev. 2008;22:1856-1864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 482] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 119. | Barker N, Clevers H. Tracking down the stem cells of the intestine: strategies to identify adult stem cells. Gastroenterology. 2007;133:1755-1760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 113] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 120. | Cheng H, Leblond CP. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. V. Unitarian Theory of the origin of the four epithelial cell types. Am J Anat. 1974;141:537-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1049] [Cited by in RCA: 1039] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 121. | Fontaine J, Le Lièvre C, Le Douarin NM. What is the developmental fate of the neural crest cells which migrate into the pancreas in the avian embryo? Gen Comp Endocrinol. 1977;33:394-404. [PubMed] |
| 122. | Le Douarin NM, Teillet MA. The migration of neural crest cells to the wall of the digestive tract in avian embryo. J Embryol Exp Morphol. 1973;30:31-48. [PubMed] |
| 123. | Rawdon BB, Andrew A. Origin and differentiation of gut endocrine cells. Histol Histopathol. 1993;8:567-580. [PubMed] |
| 124. | Hoffman J, Kuhnert F, Davis CR, Kuo CJ. Wnts as essential growth factors for the adult small intestine and colon. Cell Cycle. 2004;3:554-557. [PubMed] |
| 125. | Korinek V, Barker N, Moerer P, van Donselaar E, Huls G, Peters PJ, Clevers H. Depletion of epithelial stem-cell compartments in the small intestine of mice lacking Tcf-4. Nat Genet. 1998;19:379-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1207] [Cited by in RCA: 1204] [Article Influence: 44.6] [Reference Citation Analysis (0)] |
| 126. | Ghia JE, Li N, Wang H, Collins M, Deng Y, El-Sharkawy RT, Côté F, Mallet J, Khan WI. Serotonin has a key role in pathogenesis of experimental colitis. Gastroenterology. 2009;137:1649-1660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 305] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 127. | Walther DJ, Peter JU, Bashammakh S, Hörtnagl H, Voits M, Fink H, Bader M. Synthesis of serotonin by a second tryptophan hydroxylase isoform. Science. 2003;299:76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1104] [Cited by in RCA: 1150] [Article Influence: 52.3] [Reference Citation Analysis (0)] |
| 128. | Gutknecht L, Kriegebaum C, Waider J, Schmitt A, Lesch KP. Spatio-temporal expression of tryptophan hydroxylase isoforms in murine and human brain: convergent data from Tph2 knockout mice. Eur Neuropsychopharmacol. 2009;19:266-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 126] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 129. | Neal KB, Parry LJ, Bornstein JC. Strain-specific genetics, anatomy and function of enteric neural serotonergic pathways in inbred mice. J Physiol. 2009;587:567-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 106] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 130. | Wendelbo I, Mazzawi T, El-Salhy M. Increased serotonin transporter immunoreactivity intensity in the ileum of patients with irritable bowel disease. Mol Med Rep. 2014;9:180-184; Epub 2013 Nov 8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 131. | El-Salhy M, Wendelbo IH, Gundersen D. Reduced chromogranin A cell density in the ileum of patients with irritable bowel syndrome. Mol Med Rep. 2013;7:1241-1244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 132. | El-Salhy M, Vaali K, Dizdar V, Hausken T. Abnormal small-intestinal endocrine cells in patients with irritable bowel syndrome. Dig Dis Sci. 2010;55:3508-3513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 133. | El-Salhy M, Mazzawi T, Gundersen D, Hausken T. Chromogranin A cell density in the rectum of patients with irritable bowel syndrome. Mol Med Rep. 2012;6:1223-1225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 134. | El-Salhy M, Lomholt-Beck B, Hausken T. Chromogranin A as a possible tool in the diagnosis of irritable bowel syndrome. Scand J Gastroenterol. 2010;45:1435-1439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 58] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 135. | El-Salhy M, Lillebø E, Reinemo A, Salmelid L. Ghrelin in patients with irritable bowel syndrome. Int J Mol Med. 2009;23:703-707. [PubMed] |
| 136. | El-Salhy M, Gundersen D, Ostgaard H, Lomholt-Beck B, Hatlebakk JG, Hausken T. Low densities of serotonin and peptide YY cells in the colon of patients with irritable bowel syndrome. Dig Dis Sci. 2012;57:873-878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 54] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 137. | El-Salhy M, Gundersen D, Hatlebakk JG, Hausken T. Chromogranin A cell density as a diagnostic marker for lymphocytic colitis. Dig Dis Sci. 2012;57:3154-3159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 138. | Sjölund K, Ekman R, Wierup N. Covariation of plasma ghrelin and motilin in irritable bowel syndrome. Peptides. 2010;31:1109-1112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 139. | Wang SH, Dong L, Luo JY, Gong J, Li L, Lu XL, Han SP. Decreased expression of serotonin in the jejunum and increased numbers of mast cells in the terminal ileum in patients with irritable bowel syndrome. World J Gastroenterol. 2007;13:6041-6047. [PubMed] |
| 140. | Park JH, Rhee PL, Kim G, Lee JH, Kim YH, Kim JJ, Rhee JC, Song SY. Enteroendocrine cell counts correlate with visceral hypersensitivity in patients with diarrhoea-predominant irritable bowel syndrome. Neurogastroenterol Motil. 2006;18:539-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 63] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 141. | Coates MD, Mahoney CR, Linden DR, Sampson JE, Chen J, Blaszyk H, Crowell MD, Sharkey KA, Gershon MD, Mawe GM. Molecular defects in mucosal serotonin content and decreased serotonin reuptake transporter in ulcerative colitis and irritable bowel syndrome. Gastroenterology. 2004;126:1657-1664. [PubMed] |
| 142. | El-Salhy M, Wendelbo I, Gundersen D. Serotonin and serotonin transporter in the rectum of patients with irritable bowel disease. Mol Med Rep. 2013;8:451-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 143. | Wang LH, Fang XC, Pan GZ. Bacillary dysentery as a causative factor of irritable bowel syndrome and its pathogenesis. Gut. 2004;53:1096-1101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 277] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 144. | Dizdar V, Spiller R, Singh G, Hanevik K, Gilja OH, El-Salhy M, Hausken T. Relative importance of abnormalities of CCK and 5-HT (serotonin) in Giardia-induced post-infectious irritable bowel syndrome and functional dyspepsia. Aliment Pharmacol Ther. 2010;31:883-891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 38] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 145. | Dunlop SP, Jenkins D, Neal KR, Spiller RC. Relative importance of enterochromaffin cell hyperplasia, anxiety, and depression in postinfectious IBS. Gastroenterology. 2003;125:1651-1659. [PubMed] |
| 146. | Lee KJ, Kim YB, Kim JH, Kwon HC, Kim DK, Cho SW. The alteration of enterochromaffin cell, mast cell, and lamina propria T lymphocyte numbers in irritable bowel syndrome and its relationship with psychological factors. J Gastroenterol Hepatol. 2008;23:1689-1694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 130] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 147. | Spiller RC, Jenkins D, Thornley JP, Hebden JM, Wright T, Skinner M, Neal KR. Increased rectal mucosal enteroendocrine cells, T lymphocytes, and increased gut permeability following acute Campylobacter enteritis and in post-dysenteric irritable bowel syndrome. Gut. 2000;47:804-811. [PubMed] |
| 148. | Kim HS, Lim JH, Park H, Lee SI. Increased immunoendocrine cells in intestinal mucosa of postinfectious irritable bowel syndrome patients 3 years after acute Shigella infection--an observation in a small case control study. Yonsei Med J. 2010;51:45-51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 68] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 149. | Dunlop SP, Coleman NS, Blackshaw E, Perkins AC, Singh G, Marsden CA, Spiller RC. Abnormalities of 5-hydroxytryptamine metabolism in irritable bowel syndrome. Clin Gastroenterol Hepatol. 2005;3:349-357. [PubMed] |
| 150. | El-Salhy M, Danielsson A, Stenling R, Grimelius L. Colonic endocrine cells in inflammatory bowel disease. J Intern Med. 1997;242:413-419. [PubMed] |
| 151. | Gunnarsson J, Simrén M. Peripheral factors in the pathophysiology of irritable bowel syndrome. Dig Liver Dis. 2009;41:788-793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 38] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 152. | Delgado-Aros S, Camilleri M. Visceral hypersensitivity. J Clin Gastroenterol. 2005;39:S194-203; discussion S210. [PubMed] |
| 153. | Lee OY. Asian motility studies in irritable bowel syndrome. J Neurogastroenterol Motil. 2010;16:120-130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 154. | Posserud I, Syrous A, Lindström L, Tack J, Abrahamsson H, Simrén M. Altered rectal perception in irritable bowel syndrome is associated with symptom severity. Gastroenterology. 2007;133:1113-1123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 202] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 155. | Ritchie J. Pain from distension of the pelvic colon by inflating a balloon in the irritable colon syndrome. Gut. 1973;14:125-132. [PubMed] |
| 156. | Mertz H, Naliboff B, Munakata J, Niazi N, Mayer EA. Altered rectal perception is a biological marker of patients with irritable bowel syndrome. Gastroenterology. 1995;109:40-52. [PubMed] |
| 157. | Whitehead WE, Palsson OS. Is rectal pain sensitivity a biological marker for irritable bowel syndrome: psychological influences on pain perception. Gastroenterology. 1998;115:1263-1271. [PubMed] |
| 158. | Whitehead WE, Holtkotter B, Enck P, Hoelzl R, Holmes KD, Anthony J, Shabsin HS, Schuster MM. Tolerance for rectosigmoid distention in irritable bowel syndrome. Gastroenterology. 1990;98:1187-1192. [PubMed] |
| 159. | Bouin M, Plourde V, Boivin M, Riberdy M, Lupien F, Laganière M, Verrier P, Poitras P. Rectal distention testing in patients with irritable bowel syndrome: sensitivity, specificity, and predictive values of pain sensory thresholds. Gastroenterology. 2002;122:1771-1777. [PubMed] |
| 160. | Bradette M, Delvaux M, Staumont G, Fioramonti J, Bueno L, Frexinos J. Evaluation of colonic sensory thresholds in IBS patients using a barostat. Definition of optimal conditions and comparison with healthy subjects. Dig Dis Sci. 1994;39:449-457. [PubMed] |
| 161. | Camilleri M, McKinzie S, Busciglio I, Low PA, Sweetser S, Burton D, Baxter K, Ryks M, Zinsmeister AR. Prospective study of motor, sensory, psychologic, and autonomic functions in patients with irritable bowel syndrome. Clin Gastroenterol Hepatol. 2008;6:772-781. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 197] [Cited by in RCA: 188] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 162. | Costantini M, Sturniolo GC, Zaninotto G, D’Incà R, Polo R, Naccarato R, Ancona E. Altered esophageal pain threshold in irritable bowel syndrome. Dig Dis Sci. 1993;38:206-212. [PubMed] |
| 163. | Trimble KC, Farouk R, Pryde A, Douglas S, Heading RC. Heightened visceral sensation in functional gastrointestinal disease is not site-specific. Evidence for a generalized disorder of gut sensitivity. Dig Dis Sci. 1995;40:1607-1613. [PubMed] |
| 164. | Zighelboim J, Talley NJ, Phillips SF, Harmsen WS, Zinsmeister AR. Visceral perception in irritable bowel syndrome. Rectal and gastric responses to distension and serotonin type 3 antagonism. Dig Dis Sci. 1995;40:819-827. [PubMed] |
| 165. | Accarino AM, Azpiroz F, Malagelada JR. Selective dysfunction of mechanosensitive intestinal afferents in irritable bowel syndrome. Gastroenterology. 1995;108:636-643. [PubMed] |
| 166. | Kanazawa M, Hongo M, Fukudo S. Visceral hypersensitivity in irritable bowel syndrome. J Gastroenterol Hepatol. 2011;26 Suppl 3:119-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 85] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 167. | Azpiroz F, Bouin M, Camilleri M, Mayer EA, Poitras P, Serra J, Spiller RC. Mechanisms of hypersensitivity in IBS and functional disorders. Neurogastroenterol Motil. 2007;19:62-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 267] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 168. | Kanazawa M, Palsson OS, Thiwan SI, Turner MJ, van Tilburg MA, Gangarosa LM, Chitkara DK, Fukudo S, Drossman DA, Whitehead WE. Contributions of pain sensitivity and colonic motility to IBS symptom severity and predominant bowel habits. Am J Gastroenterol. 2008;103:2550-2561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 114] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 169. | van der Veek PP, Van Rood YR, Masclee AA. Symptom severity but not psychopathology predicts visceral hypersensitivity in irritable bowel syndrome. Clin Gastroenterol Hepatol. 2008;6:321-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 89] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 170. | Kuiken SD, Lindeboom R, Tytgat GN, Boeckxstaens GE. Relationship between symptoms and hypersensitivity to rectal distension in patients with irritable bowel syndrome. Aliment Pharmacol Ther. 2005;22:157-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 79] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 171. | Lee KJ, Kim JH, Cho SW. Relationship of underlying abnormalities in rectal sensitivity and compliance to distension with symptoms in irritable bowel syndrome. Digestion. 2006;73:133-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 172. | Sabate JM, Veyrac M, Mion F, Siproudhis L, Ducrotte P, Zerbib F, Grimaud JC, Dapoigny M, Dyard F, Coffin B. Relationship between rectal sensitivity, symptoms intensity and quality of life in patients with irritable bowel syndrome. Aliment Pharmacol Ther. 2008;28:484-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 173. | Gershon MD. Review article: roles played by 5-hydroxytryptamine in the physiology of the bowel. Aliment Pharmacol Ther. 1999;13 Suppl 2:15-30. [PubMed] |
| 174. | Lundgren O. Enteric nerves and diarrhoea. Pharmacol Toxicol. 2002;90:109-120. [PubMed] |
| 175. | Gershon MD. Plasticity in serotonin control mechanisms in the gut. Curr Opin Pharmacol. 2003;3:600-607. [PubMed] |
| 176. | Kellum JM, Albuquerque FC, Stoner MC, Harris RP. Stroking human jejunal mucosa induces 5-HT release and Cl- secretion via afferent neurons and 5-HT4 receptors. Am J Physiol. 1999;277:G515-G520. [PubMed] |
| 177. | Bueno L, Fioramonti J. Visceral perception: inflammatory and non-inflammatory mediators. Gut. 2002;51 Suppl 1:i19-i23. [PubMed] |
| 178. | Cook IJ, van Eeden A, Collins SM. Patients with irritable bowel syndrome have greater pain tolerance than normal subjects. Gastroenterology. 1987;93:727-733. [PubMed] |
| 179. | Bouin M, Meunier P, Riberdy-Poitras M, Poitras P. Pain hypersensitivity in patients with functional gastrointestinal disorders: a gastrointestinal-specific defect or a general systemic condition? Dig Dis Sci. 2001;46:2542-2548. [PubMed] |
| 180. | Moriarty KJ, Dawson AM. Functional abdominal pain: further evidence that whole gut is affected. Br Med J (Clin Res Ed). 1982;284:1670-1672. [PubMed] |
| 181. | Chen JX, Pan H, Rothman TP, Wade PR, Gershon MD. Guinea pig 5-HT transporter: cloning, expression, distribution, and function in intestinal sensory reception. Am J Physiol. 1998;275:G433-G448. [PubMed] |
| 182. | Keating C, Beyak M, Foley S, Singh G, Marsden C, Spiller R, Grundy D. Afferent hypersensitivity in a mouse model of post-inflammatory gut dysfunction: role of altered serotonin metabolism. J Physiol. 2008;586:4517-4530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 69] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 183. | Coleman NS, Foley S, Dunlop SP, Wheatcroft J, Blackshaw E, Perkins AC, Singh G, Marsden CA, Holmes GK, Spiller RC. Abnormalities of serotonin metabolism and their relation to symptoms in untreated celiac disease. Clin Gastroenterol Hepatol. 2006;4:874-881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 77] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 184. | Whorwell PJ, Clouter C, Smith CL. Oesophageal motility in the irritable bowel syndrome. Br Med J (Clin Res Ed). 1981;282:1101-1102. [PubMed] |
| 185. | Clouse RE, Eckert TC. Gastrointestinal symptoms of patients with esophageal contraction abnormalities. Dig Dis Sci. 1986;31:236-240. [PubMed] |
| 186. | Soffer EE, Scalabrini P, Pope CE, Wingate DL. Effect of stress on oesophageal motor function in normal subjects and in patients with the irritable bowel syndrome. Gut. 1988;29:1591-1594. [PubMed] |
| 187. | Lind CD. Motility disorders in the irritable bowel syndrome. Gastroenterol Clin North Am. 1991;20:279-295. [PubMed] |
| 188. | van Wijk HJ, Smout AJ, Akkermans LM, Roelofs JM, ten Thije OJ. Gastric emptying and dyspeptic symptoms in the irritable bowel syndrome. Scand J Gastroenterol. 1992;27:99-102. [PubMed] |
| 189. | Charles F, Phillips SF, Camilleri M, Thomforde GM. Rapid gastric emptying in patients with functional diarrhea. Mayo Clin Proc. 1997;72:323-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 190. | Caballero-Plasencia AM, Valenzuela-Barranco M, Herrerías-Gutiérrez JM, Esteban-Carretero JM. Altered gastric emptying in patients with irritable bowel syndrome. Eur J Nucl Med. 1999;26:404-409. [PubMed] |
| 191. | Portincasa P, Moschetta A, Baldassarre G, Altomare DF, Palasciano G. Pan-enteric dysmotility, impaired quality of life and alexithymia in a large group of patients meeting ROME II criteria for irritable bowel syndrome. World J Gastroenterol. 2003;9:2293-2299. [PubMed] |
| 192. | Stanghellini V, Tosetti C, Barbara G, De Giorgio R, Cogliandro L, Cogliandro R, Corinaldesi R. Dyspeptic symptoms and gastric emptying in the irritable bowel syndrome. Am J Gastroenterol. 2002;97:2738-2743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 72] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 193. | Leahy A, Besherdas K, Clayman C, Mason I, Epstein O. Abnormalities of the electrogastrogram in functional gastrointestinal disorders. Am J Gastroenterol. 1999;94:1023-1028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 71] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 194. | Evans PR, Bak YT, Shuter B, Hoschl R, Kellow JE. Gastroparesis and small bowel dysmotility in irritable bowel syndrome. Dig Dis Sci. 1997;42:2087-2093. [PubMed] |
| 195. | Nielsen OH, Gjørup T, Christensen FN. Gastric emptying rate and small bowel transit time in patients with irritable bowel syndrome determined with 99mTc-labeled pellets and scintigraphy. Dig Dis Sci. 1986;31:1287-1291. [PubMed] |
| 196. | Narducci F, Bassotti G, Granata MT, Pelli MA, Gaburri M, Palumbo R, Morelli A. Colonic motility and gastric emptying in patients with irritable bowel syndrome. Effect of pretreatment with octylonium bromide. Dig Dis Sci. 1986;31:241-246. [PubMed] |
| 197. | Acharya U, Waite N, Howlett P, Tanner AR, Smith CL. Failure to demonstrate altered gastric emptying in irritable bowel syndrome. Dig Dis Sci. 1983;28:889-892. [PubMed] |
| 198. | Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature. 1999;402:656-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5961] [Cited by in RCA: 5889] [Article Influence: 226.5] [Reference Citation Analysis (0)] |
| 199. | Date Y, Kojima M, Hosoda H, Sawaguchi A, Mondal MS, Suganuma T, Matsukura S, Kangawa K, Nakazato M. Ghrelin, a novel growth hormone-releasing acylated peptide, is synthesized in a distinct endocrine cell type in the gastrointestinal tracts of rats and humans. Endocrinology. 2000;141:4255-4261. [PubMed] |
| 200. | Hataya Y, Akamizu T, Takaya K, Kanamoto N, Ariyasu H, Saijo M, Moriyama K, Shimatsu A, Kojima M, Kangawa K. A low dose of ghrelin stimulates growth hormone (GH) release synergistically with GH-releasing hormone in humans. J Clin Endocrinol Metab. 2001;86:4552. [PubMed] |
| 201. | Masuda Y, Tanaka T, Inomata N, Ohnuma N, Tanaka S, Itoh Z, Hosoda H, Kojima M, Kangawa K. Ghrelin stimulates gastric acid secretion and motility in rats. Biochem Biophys Res Commun. 2000;276:905-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 622] [Cited by in RCA: 619] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 202. | Fujino K, Inui A, Asakawa A, Kihara N, Fujimura M, Fujimiya M. Ghrelin induces fasted motor activity of the gastrointestinal tract in conscious fed rats. J Physiol. 2003;550:227-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 257] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 203. | Wren AM, Seal LJ, Cohen MA, Brynes AE, Frost GS, Murphy KG, Dhillo WS, Ghatei MA, Bloom SR. Ghrelin enhances appetite and increases food intake in humans. J Clin Endocrinol Metab. 2001;86:5992. [PubMed] |
| 204. | Hosoda H, Kojima M, Kangawa K. Ghrelin and the regulation of food intake and energy balance. Mol Interv. 2002;2:494-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 73] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 205. | Asakawa A, Ataka K, Fujino K, Chen CY, Kato I, Fujimiya M, Inui A. Ghrelin family of peptides and gut motility. J Gastroenterol Hepatol. 2011;26 Suppl 3:73-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 206. | Dornonville de la Cour C, Lindström E, Norlén P, Håkanson R. Ghrelin stimulates gastric emptying but is without effect on acid secretion and gastric endocrine cells. Regul Pept. 2004;120:23-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 115] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 207. | Fukuda H, Mizuta Y, Isomoto H, Takeshima F, Ohnita K, Ohba K, Omagari K, Taniyama K, Kohno S. Ghrelin enhances gastric motility through direct stimulation of intrinsic neural pathways and capsaicin-sensitive afferent neurones in rats. Scand J Gastroenterol. 2004;39:1209-1214. [PubMed] |
| 208. | Levin F, Edholm T, Schmidt PT, Grybäck P, Jacobsson H, Degerblad M, Höybye C, Holst JJ, Rehfeld JF, Hellström PM. Ghrelin stimulates gastric emptying and hunger in normal-weight humans. J Clin Endocrinol Metab. 2006;91:3296-3302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 217] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 209. | Edholm T, Levin F, Hellström PM, Schmidt PT. Ghrelin stimulates motility in the small intestine of rats through intrinsic cholinergic neurons. Regul Pept. 2004;121:25-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 103] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 210. | Tack J, Depoortere I, Bisschops R, Delporte C, Coulie B, Meulemans A, Janssens J, Peeters T. Influence of ghrelin on interdigestive gastrointestinal motility in humans. Gut. 2006;55:327-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 263] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 211. | Ariga H, Tsukamoto K, Chen C, Mantyh C, Pappas TN, Takahashi T. Endogenous acyl ghrelin is involved in mediating spontaneous phase III-like contractions of the rat stomach. Neurogastroenterol Motil. 2007;19:675-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 85] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 212. | Ariga H, Nakade Y, Tsukamoto K, Imai K, Chen C, Mantyh C, Pappas TN, Takahashi T. Ghrelin accelerates gastric emptying via early manifestation of antro-pyloric coordination in conscious rats. Regul Pept. 2008;146:112-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 42] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 213. | Tümer C, Oflazoğlu HD, Obay BD, Kelle M, Taşdemir E. Effect of ghrelin on gastric myoelectric activity and gastric emptying in rats. Regul Pept. 2008;146:26-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 35] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 214. | Tebbe JJ, Mronga S, Tebbe CG, Ortmann E, Arnold R, Schäfer MK. Ghrelin-induced stimulation of colonic propulsion is dependent on hypothalamic neuropeptide Y1- and corticotrophin-releasing factor 1 receptor activation. J Neuroendocrinol. 2005;17:570-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 215. | Tack JF, Janssens J, Vantrappen G, Wood JD. Actions of 5-hydroxytryptamine on myenteric neurons in guinea pig gastric antrum. Am J Physiol. 1992;263:G838-G846. [PubMed] |
| 216. | Michel K, Sann H, Schaaf C, Schemann M. Subpopulations of gastric myenteric neurons are differentially activated via distinct serotonin receptors: projection, neurochemical coding, and functional implications. J Neurosci. 1997;17:8009-8017. [PubMed] |
| 217. | Tack J, Coulie B, Wilmer A, Andrioli A, Janssens J. Influence of sumatriptan on gastric fundus tone and on the perception of gastric distension in man. Gut. 2000;46:468-473. [PubMed] |
| 218. | Camilleri M. Integrated upper gastrointestinal response to food intake. Gastroenterology. 2006;131:640-658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 159] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 219. | Lal S, McLaughlin J, Barlow J, D’Amato M, Giacovelli G, Varro A, Dockray GJ, Thompson DG. Cholecystokinin pathways modulate sensations induced by gastric distension in humans. Am J Physiol Gastrointest Liver Physiol. 2004;287:G72-G79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 58] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 220. | Moran TH, Ladenheim EE, Schwartz GJ. Within-meal gut feedback signaling. Int J Obes Relat Metab Disord. 2001;25 Suppl 5:S39-S41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 66] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 221. | Chey WY, Chang TM. Secretin, 100 years later. J Gastroenterol. 2003;38:1025-1035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 79] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 222. | Cann PA, Read NW, Brown C, Hobson N, Holdsworth CD. Irritable bowel syndrome: relationship of disorders in the transit of a single solid meal to symptom patterns. Gut. 1983;24:405-411. [PubMed] |
| 223. | Sadik R, Stotzer PO, Simrén M, Abrahamsson H. Gastrointestinal transit abnormalities are frequently detected in patients with unexplained GI symptoms at a tertiary centre. Neurogastroenterol Motil. 2008;20:197-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 68] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 224. | Jian R, Najean Y, Bernier JJ. Measurement of intestinal progression of a meal and its residues in normal subjects and patients with functional diarrhoea by a dual isotope technique. Gut. 1984;25:728-731. [PubMed] |
| 225. | Corbett CL, Thomas S, Read NW, Hobson N, Bergman I, Holdsworth CD. Electrochemical detector for breath hydrogen determination: measurement of small bowel transit time in normal subjects and patients with the irritable bowel syndrome. Gut. 1981;22:836-840. [PubMed] |
| 226. | Lu CL, Chen CY, Chang FY, Lee SD. Characteristics of small bowel motility in patients with irritable bowel syndrome and normal humans: an Oriental study. Clin Sci (Lond). 1998;95:165-169. [PubMed] |
| 227. | Kellow JE, Phillips SF, Miller LJ, Zinsmeister AR. Dysmotility of the small intestine in irritable bowel syndrome. Gut. 1988;29:1236-1243. [PubMed] |
| 228. | Kellow JE, Phillips SF. Altered small bowel motility in irritable bowel syndrome is correlated with symptoms. Gastroenterology. 1987;92:1885-1893. [PubMed] |
| 229. | Kellow JE, Gill RC, Wingate DL. Prolonged ambulant recordings of small bowel motility demonstrate abnormalities in the irritable bowel syndrome. Gastroenterology. 1990;98:1208-1218. [PubMed] |
| 230. | Quigley EM, Donovan JP, Lane MJ, Gallagher TF. Antroduodenal manometry. Usefulness and limitations as an outpatient study. Dig Dis Sci. 1992;37:20-28. [PubMed] |
| 231. | Quigley EM. Intestinal manometry--technical advances, clinical limitations. Dig Dis Sci. 1992;37:10-13. [PubMed] |
| 232. | Wang SH, Dong L, Luo JY, Li L, Zhu YL, Wang XQ, Zou BC, Gong J. [A research of migrating motor complex in patients with irritable bowel syndrome]. Zhonghua Neike Zazhi. 2009;48:106-110. [PubMed] |
| 233. | Gorard DA, Libby GW, Farthing MJ. Ambulatory small intestinal motility in ‘diarrhoea’ predominant irritable bowel syndrome. Gut. 1994;35:203-210. [PubMed] |
| 234. | Zhao JH, Dong L, Hao XQ. [Small intestine motility and gastrointestinal hormone levels in irritable bowel syndrome]. Nanfang Yike Daxue Xuebao. 2007;27:1492-1495. [PubMed] |
| 235. | Kellow JE, Eckersley CM, Jones MP. Enhanced perception of physiological intestinal motility in the irritable bowel syndrome. Gastroenterology. 1991;101:1621-1627. [PubMed] |
| 236. | Thompson DG, Laidlow JM, Wingate DL. Abnormal small-bowel motility demonstrated by radiotelemetry in a patient with irritable colon. Lancet. 1979;2:1321-1323. [PubMed] |
| 237. | Kingham JG, Bown R, Colson R, Clark ML. Jejunal motility in patients with functional abdominal pain. Gut. 1984;25:375-380. [PubMed] |
| 238. | Schmidt T, Pfeiffer A, Kaess H. Abnormal intestinal motility in irritable bowel syndrome. Gastroenterology. 1996;111:1400-1401. [PubMed] |
| 239. | Schmidt T, Hackelsberger N, Widmer R, Meisel C, Pfeiffer A, Kaess H. Ambulatory 24-hour jejunal motility in diarrhea-predominant irritable bowel syndrome. Scand J Gastroenterol. 1996;31:581-589. [PubMed] |
| 240. | Hellström PM, Näslund E, Edholm T, Schmidt PT, Kristensen J, Theodorsson E, Holst JJ, Efendic S. GLP-1 suppresses gastrointestinal motility and inhibits the migrating motor complex in healthy subjects and patients with irritable bowel syndrome. Neurogastroenterol Motil. 2008;20:649-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 109] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 241. | Simrén M, Castedal M, Svedlund J, Abrahamsson H, Björnsson E. Abnormal propagation pattern of duodenal pressure waves in the irritable bowel syndrome (IBS) [correction of (IBD)]. Dig Dis Sci. 2000;45:2151-2161. [PubMed] |
| 242. | Stanghellini V, Ghidini C, Maccarini MR, Paparo GF, Corinaldesi R, Barbara L. Fasting and postprandial gastrointestinal motility in ulcer and non-ulcer dyspepsia. Gut. 1992;33:184-190. [PubMed] |
| 243. | Björnsson ES, Abrahamsson H. Interdigestive gastroduodenal manometry in humans. Indication of duodenal phase III as a retroperistaltic pump. Acta Physiol Scand. 1995;153:221-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 244. | Maljaars PW, Keszthelyi D, Masclee AA. An ileal brake-through? Am J Clin Nutr. 2010;92:467-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 245. | Van Citters GW, Lin HC. Ileal brake: neuropeptidergic control of intestinal transit. Curr Gastroenterol Rep. 2006;8:367-373. [PubMed] |
| 246. | Lin HC, Zhao XT, Wang L, Wong H. Fat-induced ileal brake in the dog depends on peptide YY. Gastroenterology. 1996;110:1491-1495. [PubMed] |
| 247. | Pironi L, Stanghellini V, Miglioli M, Corinaldesi R, De Giorgio R, Ruggeri E, Tosetti C, Poggioli G, Morselli Labate AM, Monetti N. Fat-induced ileal brake in humans: a dose-dependent phenomenon correlated to the plasma levels of peptide YY. Gastroenterology. 1993;105:733-739. [PubMed] |
| 248. | Spiller RC, Trotman IF, Adrian TE, Bloom SR, Misiewicz JJ, Silk DB. Further characterisation of the ‘ileal brake’ reflex in man--effect of ileal infusion of partial digests of fat, protein, and starch on jejunal motility and release of neurotensin, enteroglucagon, and peptide YY. Gut. 1988;29:1042-1051. [PubMed] |
| 249. | Spiller RC, Trotman IF, Higgins BE, Ghatei MA, Grimble GK, Lee YC, Bloom SR, Misiewicz JJ, Silk DB. The ileal brake--inhibition of jejunal motility after ileal fat perfusion in man. Gut. 1984;25:365-374. [PubMed] |
| 250. | Goumain M, Voisin T, Lorinet AM, Ducroc R, Tsocas A, Rozé C, Rouet-Benzineb P, Herzog H, Balasubramaniam A, Laburthe M. The peptide YY-preferring receptor mediating inhibition of small intestinal secretion is a peripheral Y(2) receptor: pharmacological evidence and molecular cloning. Mol Pharmacol. 2001;60:124-134. [PubMed] |
| 251. | Souli A, Chariot J, Voisin T, Presset O, Tsocas A, Balasubramaniam A, Laburthe M, Rozé C. Several receptors mediate the antisecretory effect of peptide YY, neuropeptide Y, and pancreatic polypeptide on VIP-induced fluid secretion in the rat jejunum in vivo. Peptides. 1997;18:551-557. [PubMed] |
| 252. | Whang EE, Hines OJ, Reeve JR, Grandt D, Moser JA, Bilchik AJ, Zinner MJ, McFadden DW, Ashley SW. Antisecretory mechanisms of peptide YY in rat distal colon. Dig Dis Sci. 1997;42:1121-1127. [PubMed] |
| 253. | Konturek SJ, Konturek PC, Brzozowska I, Pawlik M, Sliwowski Z, Cześnikiewicz-Guzik M, Kwiecień S, Brzozowski T, Bubenik GA, Pawlik WW. Localization and biological activities of melatonin in intact and diseased gastrointestinal tract (GIT). J Physiol Pharmacol. 2007;58:381-405. [PubMed] |
| 254. | Thor PJ, Krolczyk G, Gil K, Zurowski D, Nowak L. Melatonin and serotonin effects on gastrointestinal motility. J Physiol Pharmacol. 2007;58 Suppl 6:97-103. [PubMed] |
| 255. | Lu WZ, Song GH, Gwee KA, Ho KY. The effects of melatonin on colonic transit time in normal controls and IBS patients. Dig Dis Sci. 2009;54:1087-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 256. | Charles F, Camilleri M, Phillips SF, Thomforde GM, Forstrom LA. Scintigraphy of the whole gut: clinical evaluation of transit disorders. Mayo Clin Proc. 1995;70:113-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 257. | van der Sijp JR, Kamm MA, Nightingale JM, Britton KE, Mather SJ, Morris GP, Akkermans LM, Lennard-Jones JE. Radioisotope determination of regional colonic transit in severe constipation: comparison with radio opaque markers. Gut. 1993;34:402-408. [PubMed] |
| 258. | Vassallo M, Camilleri M, Phillips SF, Brown ML, Chapman NJ, Thomforde GM. Transit through the proximal colon influences stool weight in the irritable bowel syndrome. Gastroenterology. 1992;102:102-108. [PubMed] |
| 259. | Snape WJ, Carlson GM, Cohen S. Colonic myoelectric activity in the irritable bowel syndrome. Gastroenterology. 1976;70:326-330. [PubMed] |
| 260. | Snape WJ. Myoelectric and motor activity of the colon in normal and abnormal states. Scand J Gastroenterol Suppl. 1984;96:55-60. [PubMed] |
| 261. | Sarna S, Latimer P, Campbell D, Waterfall WE. Effect of stress, meal and neostigmine on rectosigmoid electrical control activity (ECA) in normals and in irritable bowel syndrome patients. Dig Dis Sci. 1982;27:582-591. [PubMed] |
| 262. | Latimer P, Sarna S, Campbell D, Latimer M, Waterfall W, Daniel EE. Colonic motor and myoelectrical activity: a comparative study of normal subjects, psychoneurotic patients, and patients with irritable bowel syndrome. Gastroenterology. 1981;80:893-901. [PubMed] |
| 263. | Welgan P, Meshkinpour H, Ma L. Role of anger in antral motor activity in irritable bowel syndrome. Dig Dis Sci. 2000;45:248-251. [PubMed] |
| 264. | Welgan P, Meshkinpour H, Hoehler F. The effect of stress on colon motor and electrical activity in irritable bowel syndrome. Psychosom Med. 1985;47:139-149. [PubMed] |
| 265. | Katschinski M, Lederer P, Ellermann A, Ganzleben R, Lux G, Arnold R. Myoelectric and manometric patterns of human rectosigmoid colon in irritable bowel syndrome and diverticulosis. Scand J Gastroenterol. 1990;25:761-768. [PubMed] |
| 266. | Chey WY, Jin HO, Lee MH, Sun SW, Lee KY. Colonic motility abnormality in patients with irritable bowel syndrome exhibiting abdominal pain and diarrhea. Am J Gastroenterol. 2001;96:1499-1506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 244] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 267. | Vassallo MJ, Camilleri M, Phillips SF, Steadman CJ, Talley NJ, Hanson RB, Haddad AC. Colonic tone and motility in patients with irritable bowel syndrome. Mayo Clin Proc. 1992;67:725-731. [PubMed] |
| 268. | Corsetti M, Ogliari C, Marino B, Basilisco G. Perceptual sensitivity and response bias during rectal distension in patients with irritable bowel syndrome. Neurogastroenterol Motil. 2005;17:541-547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 269. | Corsetti M, Cesana B, Bhoori S, Basilisco G. Rectal hyperreactivity to distention in patients with irritable bowel syndrome: role of distention rate. Clin Gastroenterol Hepatol. 2004;2:49-56. [PubMed] |
| 270. | Bassotti G, de Roberto G, Chistolini F, Sietchiping-Nzepa F, Morelli O, Morelli A. Twenty-four-hour manometric study of colonic propulsive activity in patients with diarrhea due to inflammatory (ulcerative colitis) and non-inflammatory (irritable bowel syndrome) conditions. Int J Colorectal Dis. 2004;19:493-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 38] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 271. | Bassotti G, Sietchiping-Nzepa F, De Roberto G, Chistolini F, Morelli A. Colonic regular contractile frequency patterns in irritable bowel syndrome: the ‘spastic colon’ revisited. Eur J Gastroenterol Hepatol. 2004;16:613-617. [PubMed] |
| 272. | Clemens CH, Samsom M, Van Berge Henegouwen GP, Smout AJ. Abnormalities of left colonic motility in ambulant nonconstipated patients with irritable bowel syndrome. Dig Dis Sci. 2003;48:74-82. [PubMed] |
| 273. | Clemens CH, Samsom M, Roelofs JM, van Berge Henegouwen GP, Smout AJ. Association between pain episodes and high amplitude propagated pressure waves in patients with irritable bowel syndrome. Am J Gastroenterol. 2003;98:1838-1843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 274. | Rogers J, Henry MM, Misiewicz JJ. Increased segmental activity and intraluminal pressures in the sigmoid colon of patients with the irritable bowel syndrome. Gut. 1989;30:634-641. [PubMed] |
| 275. | Simrén M, Abrahamsson H, Björnsson ES. An exaggerated sensory component of the gastrocolonic response in patients with irritable bowel syndrome. Gut. 2001;48:20-27. [PubMed] |
| 276. | Di Stefano M, Miceli E, Missanelli A, Mazzocchi S, Corazza GR. Meal induced rectosigmoid tone modification: a low caloric meal accurately separates functional and organic gastrointestinal disease patients. Gut. 2006;55:1409-1414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 277. | Park JH, Baek YH, Park DI, Kim HJ, Cho YK, Sohn CI, Jeon WK, Kim BI, Rhee PL. Analysis of rectal dynamic and static compliances in patients with irritable bowel syndrome. Int J Colorectal Dis. 2008;23:659-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 278. | Steens J, Van Der Schaar PJ, Penning C, Brussee J, Masclee AA. Compliance, tone and sensitivity of the rectum in different subtypes of irritable bowel syndrome. Neurogastroenterol Motil. 2002;14:241-247. [PubMed] |
| 279. | Kwan CL, Davis KD, Mikula K, Diamant NE. Abnormal rectal motor physiology in patients with irritable bowel syndrome. Neurogastroenterol Motil. 2004;16:251-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 280. | Hillsley K, Kirkup AJ, Grundy D. Direct and indirect actions of 5-hydroxytryptamine on the discharge of mesenteric afferent fibres innervating the rat jejunum. J Physiol. 1998;506:551-561. [PubMed] |
| 281. | Hillsley K, Grundy D. Sensitivity to 5-hydroxytryptamine in different afferent subpopulations within mesenteric nerves supplying the rat jejunum. J Physiol. 1998;509:717-727. [PubMed] |
| 282. | Grundy D, Hillsley K, Kirkup AJ, Richards W. Mesenteric afferent sensitivity to cholecystokinin and 5-hydroxytryptamine. Dtsch Tierarztl Wochenschr. 1998;105:466-468. [PubMed] |
| 283. | Blackshaw LA, Grundy D. Effects of 5-hydroxytryptamine on discharge of vagal mucosal afferent fibres from the upper gastrointestinal tract of the ferret. J Auton Nerv Syst. 1993;45:41-50. [PubMed] |
| 284. | Blackshaw LA, Grundy D. Effects of 5-hydroxytryptamine (5-HT) on the discharge of vagal mechanoreceptors and motility in the upper gastrointestinal tract of the ferret. J Auton Nerv Syst. 1993;45:51-59. [PubMed] |
| 285. | Kirchgessner AL, Liu MT, Tamir H, Gershon MD. Identification and localization of 5-HT1P receptors in the guinea pig pancreas. Am J Physiol. 1992;262:G553-G566. [PubMed] |
| 286. | Kirchgessner AL, Liu MT, Gershon MD. In situ identification and visualization of neurons that mediate enteric and enteropancreatic reflexes. J Comp Neurol. 1996;371:270-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 287. | Gershon MD, Wade PR, Kirchgessner AL, Tamir H. 5-HT receptor subtypes outside the central nervous system. Roles in the physiology of the gut. Neuropsychopharmacology. 1990;3:385-395. [PubMed] |
| 288. | Pan H, Gershon MD. Activation of intrinsic afferent pathways in submucosal ganglia of the guinea pig small intestine. J Neurosci. 2000;20:3295-3309. [PubMed] |
| 289. | Cooke HJ, Sidhu M, Wang YZ. 5-HT activates neural reflexes regulating secretion in the guinea-pig colon. Neurogastroenterol Motil. 1997;9:181-186. [PubMed] |
| 290. | Oddsson E, Rask-Madsen J, Krag E. A secretory epithelium of the small intestine with increased sensitivity to bile acids in irritable bowel syndrome associated with diarrhoea. Scand J Gastroenterol. 1978;13:408-416. [PubMed] |
| 291. | Larsson MH, Simrén M, Thomas EA, Bornstein JC, Lindström E, Sjövall H. Elevated motility-related transmucosal potential difference in the upper small intestine in the irritable bowel syndrome. Neurogastroenterol Motil. 2007;19:812-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 292. | Money ME, Camilleri M. Review: Management of postprandial diarrhea syndrome. Am J Med. 2012;125:538-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 293. | Camilleri M. Peripheral mechanisms in irritable bowel syndrome. N Engl J Med. 2013;368:578-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 294. | Camilleri M, Coulie B, Tack JF. Visceral hypersensitivity: facts, speculations, and challenges. Gut. 2001;48:125-131. [PubMed] |
| 295. | De Giorgio R, Camilleri M. Human enteric neuropathies: morphology and molecular pathology. Neurogastroenterol Motil. 2004;16:515-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 154] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 296. | Hotoleanu C, Popp R, Trifa AP, Nedelcu L, Dumitrascu DL. Genetic determination of irritable bowel syndrome. World J Gastroenterol. 2008;14:6636-6640. [PubMed] |
| 297. | Whorwell PJ, McCallum M, Creed FH, Roberts CT. Non-colonic features of irritable bowel syndrome. Gut. 1986;27:37-40. [PubMed] |
| 298. | Locke GR, Zinsmeister AR, Talley NJ, Fett SL, Melton LJ. Familial association in adults with functional gastrointestinal disorders. Mayo Clin Proc. 2000;75:907-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 180] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 299. | Kalantar JS, Locke GR, Zinsmeister AR, Beighley CM, Talley NJ. Familial aggregation of irritable bowel syndrome: a prospective study. Gut. 2003;52:1703-1707. [PubMed] |
| 300. | Kanazawa M, Endo Y, Whitehead WE, Kano M, Hongo M, Fukudo S. Patients and nonconsulters with irritable bowel syndrome reporting a parental history of bowel problems have more impaired psychological distress. Dig Dis Sci. 2004;49:1046-1053. [PubMed] |
| 301. | Morris-Yates A, Talley NJ, Boyce PM, Nandurkar S, Andrews G. Evidence of a genetic contribution to functional bowel disorder. Am J Gastroenterol. 1998;93:1311-1317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 167] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 302. | Levy RL, Jones KR, Whitehead WE, Feld SI, Talley NJ, Corey LA. Irritable bowel syndrome in twins: heredity and social learning both contribute to etiology. Gastroenterology. 2001;121:799-804. [PubMed] |
| 303. | Lembo A, Zaman M, Jones M, Talley NJ. Influence of genetics on irritable bowel syndrome, gastro-oesophageal reflux and dyspepsia: a twin study. Aliment Pharmacol Ther. 2007;25:1343-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 93] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 304. | Wojczynski MK, North KE, Pedersen NL, Sullivan PF. Irritable bowel syndrome: a co-twin control analysis. Am J Gastroenterol. 2007;102:2220-2229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 305. | Bengtson MB, Rønning T, Vatn MH, Harris JR. Irritable bowel syndrome in twins: genes and environment. Gut. 2006;55:1754-1759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 137] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 306. | Mohammed I, Cherkas LF, Riley SA, Spector TD, Trudgill NJ. Genetic influences in irritable bowel syndrome: a twin study. Am J Gastroenterol. 2005;100:1340-1344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 72] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 307. | Spiller R, Garsed K. Infection, inflammation, and the irritable bowel syndrome. Dig Liver Dis. 2009;41:844-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 97] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 308. | Spiller R, Garsed K. Postinfectious irritable bowel syndrome. Gastroenterology. 2009;136:1979-1988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 489] [Cited by in RCA: 482] [Article Influence: 30.1] [Reference Citation Analysis (1)] |
| 309. | Spiller RC. Irritable bowel syndrome: bacteria and inflammation--clinical relevance now. Curr Treat Options Gastroenterol. 2007;10:312-321. [PubMed] |
| 310. | Spiller RC. Role of infection in irritable bowel syndrome. J Gastroenterol. 2007;42 Suppl 17:41-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 124] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 311. | Sarna SK. Lessons Learnt from Post-Infectious IBS. Front Physiol. 2011;2:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 312. | Gwee KA. Post-Infectious Irritable Bowel Syndrome, an Inflammation-Immunological Model with Relevance for Other IBS and Functional Dyspepsia. J Neurogastroenterol Motil. 2010;16:30-34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 56] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 313. | Ghoshal UC, Park H, Gwee KA. Bugs and irritable bowel syndrome: The good, the bad and the ugly. J Gastroenterol Hepatol. 2010;25:244-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 58] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 314. | Halvorson HA, Schlett CD, Riddle MS. Postinfectious irritable bowel syndrome--a meta-analysis. Am J Gastroenterol. 2006;101:1894-1899; quiz 1942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 239] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 315. | Spiller R. Serotonin, inflammation, and IBS: fitting the jigsaw together? J Pediatr Gastroenterol Nutr. 2007;45 Suppl 2:S115-S119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 316. | Wheatcroft J, Wakelin D, Smith A, Mahoney CR, Mawe G, Spiller R. Enterochromaffin cell hyperplasia and decreased serotonin transporter in a mouse model of postinfectious bowel dysfunction. Neurogastroenterol Motil. 2005;17:863-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 151] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 317. | Khan WI, Ghia JE. Gut hormones: emerging role in immune activation and inflammation. Clin Exp Immunol. 2010;161:19-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 134] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 318. | Yang GB, Lackner AA. Proximity between 5-HT secreting enteroendocrine cells and lymphocytes in the gut mucosa of rhesus macaques (Macaca mulatta) is suggestive of a role for enterochromaffin cell 5-HT in mucosal immunity. J Neuroimmunol. 2004;146:46-49. [PubMed] |
| 319. | Akiho H, Ihara E, Nakamura K. Low-grade inflammation plays a pivotal role in gastrointestinal dysfunction in irritable bowel syndrome. World J Gastrointest Pathophysiol. 2010;1:97-105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 63] [Cited by in RCA: 79] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 320. | Wouters MM, Boeckxstaens GE. Neuroimmune mechanisms in functional bowel disorders. Neth J Med. 2011;69:55-61. [PubMed] |
| 321. | Moran GW, Leslie FC, Levison SE, Worthington J, McLaughlin JT. Enteroendocrine cells: neglected players in gastrointestinal disorders? Therap Adv Gastroenterol. 2008;1:51-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |