Published online Sep 15, 1996. doi: 10.3748/wjg.v2.i3.158
Revised: August 6, 1996
Accepted: August 27, 1996
Published online: September 15, 1996
AIM: To study the safety and efficacy of hepatic arterial embolization (HAE) with Bletilla striata powders containing traditional Chinese herbs in the treatment of primary hepatic carcinoma (PHC).
METHODS: From May 1990 to September 1993, 106 patients with PHC were treated by HAE with different types of Bletilla striata powders (n = 56) or Gelfoam powders (n = 50) under clearly specified conditions. We analyzed the effects and complications associated with these two types of treatment.
RESULTS: The Bletilla striata powders produced extensive and permanent proximal embolization of the hepatic artery, and stimulated the formation of collateral circulation. Treatment could be stopped for as long as 6-12 mo, and there was obvious evidence of tumor necrosis and shrinkage. The patient survival rates at 1, 2, and 3 years were 81.9%, 44.9%, and 33.6%, respectively, and the mean survival time without a serious complication was 19.8 mo. Patients in the Bletilla striata group displayed better clinical effects from their treatment when compared with patients in the Gelfoam group.
CONCLUSION: Bletilla striata powders are superior to Gelfoam powders when used for angioembolus in patients with hepatic carcinoma.
- Citation: Feng GS, Kramann B, Zheng CS, Zhou RM, Liang B, Zhang YF. Comparative study on the effects of hepatic arterial embolization with Bletilla striata or Gelfoam in treatment of primary hepatic carcinoma. World J Gastroenterol 1996; 2(3): 158-160
- URL: https://www.wjgnet.com/1007-9327/full/v2/i3/158.htm
- DOI: https://dx.doi.org/10.3748/wjg.v2.i3.158
From May 1990 to September 1993, 106 patients with primary hepatic carcinoma (PHC) were treated by hepatic arterial embolization (HAE). We objectively compared the effects of permanent HAE produced with Bletilla striata with the effects of temporary HAE produced with Gelfoam.
This study enrolled 166 patients (93 males and 13 females; mean age = 46 years) who were clinically diagnosed as PHC. The patients were randomly assigned to a Bletill striata group (BS group) and a Gelfoam group (GF group). The general medical conditions of the patients and their respective methods of treatment are shown in Table 1. Both groups of patients received hepatic arterial chemotherapy with Adriamycin (ADM; 50 mg) and carboplatin (CAP; 500 mg). A peripheral angioembolus was created by administering an emulsion consisting of 40% Lipiodol (LP) and 10 mg mitomycin C (MMC). The femoral artery was percutaneously punctured using Seldinger’s method, and a catheter was inserted into the segmental or lobar artery of the hepatic tumor. Following transcatheter intra-arterial infusion of the chemotherapeutic agents and the LP-MMC emulsion, 2-3 mL of mixed Bletilla striata powders (300 mg) and 5 mL of 76% meglumini diatrizoatis composita were slowly infused to produce proximal HAE. If the normal hepatic segmental and lobar arteries were not available for catheterization, proximal HAE was performed using Gelfoam powder.
| Subjects | GF group | BS group |
| Number | 50 | 56 |
| Clinical stage; cases (%) | ||
| I | 3 (6.0) | 4 (7.1) |
| II | 42 (84.0) | 46 (82.2) |
| III | 5 (10.0) | 6 (10.7) |
| Pathologic type; cases (%) | ||
| Massive | 38 (76.0) | 43 (76.8) |
| Multinodular | 12 (24.0) | 13 (23.2) |
| Proximal angioembolus | GF powders | BS powders |
| Total number of treatments | 203 | 151 |
| Mean number of treatments | 4.06 | 2.7 |
| Mean intermission between treatments (mo) | 3-6 | 6-12 |
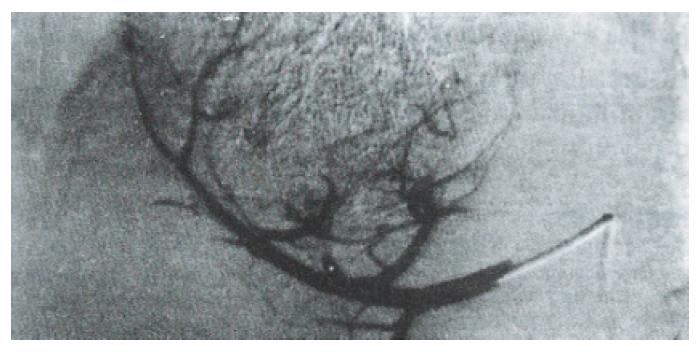
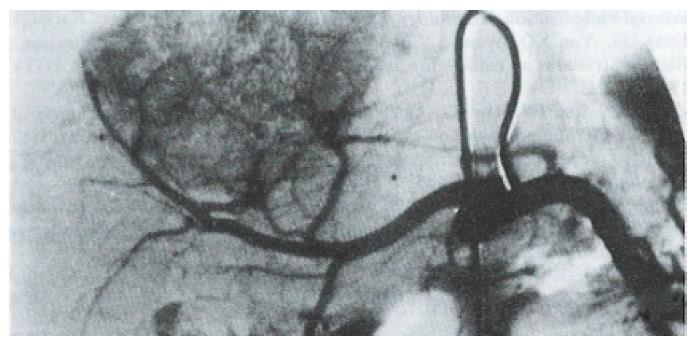
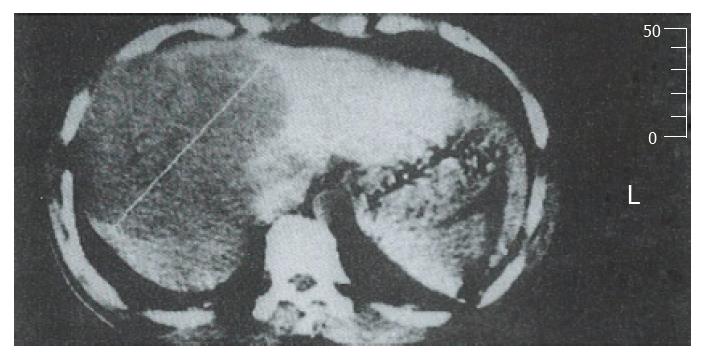
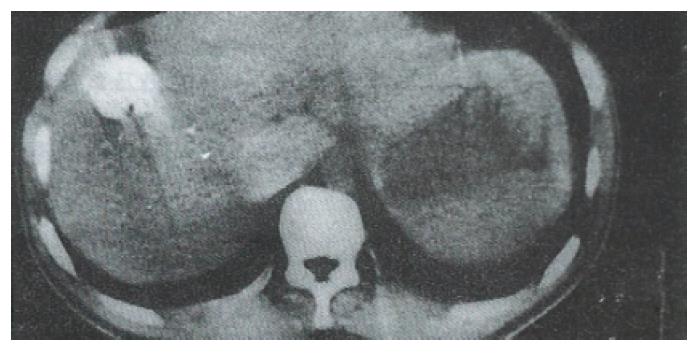
At three mo after HAE, 92% of patients in the Gelfoam group showed recanalization of the embolized artery and formation of collateral circulation (Figure 1 and Figure 2). In contrast, none of the patients in the Bletilla striata group showed recanalization, even at 3 yr after embolization. Collateral circulation was rarely formed, and when it did, it required > 6 mo to develop. Additionally, there was no collateral blood supply to the tumor in ~ 50% of the patients at one year after their embolization procedure (Figure 3 and Figure 4).
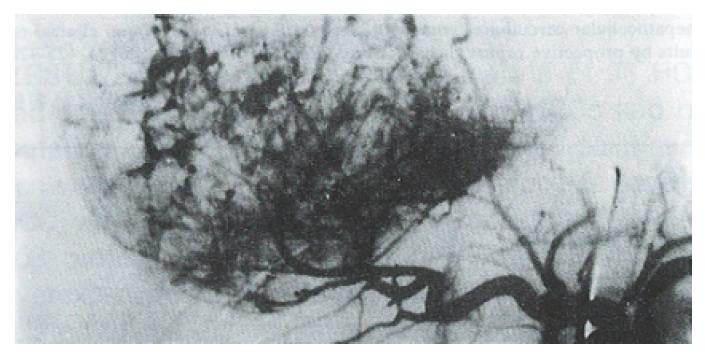
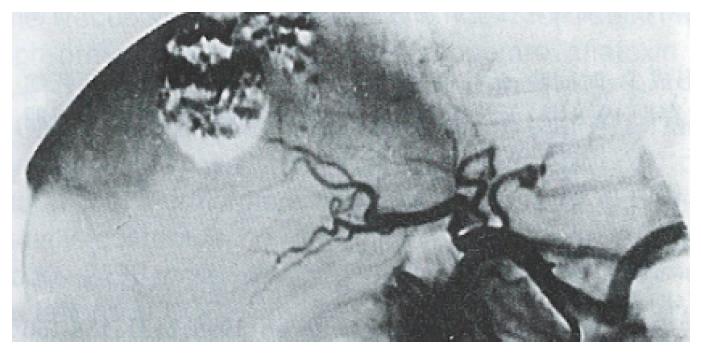
In the Bletilla striata group, 36.2%, 55.8%, and 71.4% of patients showed signs of tumor shrinkage at 3, 6, and 12 mo, respectively, after embolization. The corresponding rates in the Gelform group were 25.0%, 46.4%, and 64.3%, respectively. In the Bletilla striata group, tumor necrosis was most obvious at 3 mo; at which time, the hepatic tumor tissue had sunken due to obvious fibrosis and lipiodol retention in the sites (Figure 5 and Figure 6). In some patients, the tumor became calcified one year later.
Specimens obtained during second surgical resections showed no surviving tumor cells in 2 of 3 cases in the Bletilla striata group, while 2 cases in the Gelform group had surviving tumor cells.
After HAE, hepatic pain, fever, and damage to hepatic function were all more severe in the Blatilla striata group when compared to the Gelfoam group. In both groups, pain lasted for 2-5 d and fever for 3-20 d, and the differences between groups were not statistically significant.
The survival rates and survival times in the two groups of patients are shown in Table 2.
The use of HAE in treatment of PHC has attracted a great deal of attention as a novel methodology[1,2]. Bletilla striata powders display the following characteristics when used in treatment of PHC: (1) They produce permanent proximal embolization of the hepatic artery. Besides mechanically blocking blood vessels, they also induce extensive coagulation[3]. Embolization occurs several seconds after the powder’s application, and a vascular cast appears. (2) Bletilla striata powders can be custom produced at an appropriate concentration for use in combination with a contrast medium, and to meet the requirements of a particular embolization procedure. Bletilla striata powders not only embolize the main trunk of an artery, but also block tumor vessel beds that are not completely filled with Lipiodol. As a result, they effectively prevent the establishment of collateral circulation. However, this viewpoint is not consistent with some research conclusions which state that extensive embolization of a hepatic carcinoma artery may induce the formation of collateral circulation[4,5]. The reason that tumors become revascularized and develop collateral circulation after HAE with Gelfoam powders may be that Lipiodol and Gelfoam powders only block small tumor vessels and blood supply arteries respectively, while mid-sized vessels that are not embolized allow the tumor to establish collateral circulation with surrounding hepatic segmental or lobar arteries. As a result, HAE with Gelfoam does not produce complete necrosis of the tumor, and these patients need to be re-embolized after a short time period[6]. (3) Both the retention and elimination times of Lipiodol in the tumor area were better in the Bletilla striata group when compared with those parameters in the Gelfoam group. This may be because the arterial blood flow washes out the intratumor Lipiodol as intra-arterial Gelfoam powders are being absorbed[7]. And (4) When performing HAE with Bletilla striata powders, the catheter must be inserted into the peripheral branch of the hepatic artery to avoid penetrating the normal hepatic parenchyma. If this is not possible, Gelfoam powders must be used when creating a proximal angioembolus. Additionally, any backflow of the angioembolus must be avoided in order to prevent a misembolization.
In view of the characteristics of Bletilla striata, we believe that a permanent HAE produced with Bletilla strata powders in treatment of PHC not only produces a better clinical effect, but also reduces the economic burden and psychological pressure experienced by patients.
Original title:
S- Editor: Yang ZD L- Editor: Filipodia E- Editor: Li RF
| 1. | Gou JY, Huang ZC, Yan D, Hu GD. Intraarterial infusion chemotherapy and embolization with mixture of lipiodol and anticancer drug in the treatment of hepatic carcinoma. J of Clinical Radiology. 1987;6:281-283. |
| 2. | Matsui O, Kadoya M, Yoshikawa J, Gabata T, Arai K, Demachi H, Miyayama S, Takashima T, Unoura M, Kogayashi K. Small hepatocellular carcinoma: treatment with subsegmental transcatheter arterial embolization. Radiology. 1993;188:79-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 312] [Cited by in RCA: 311] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 3. | Fen GS. [Animal experiment and clinical application of Bletilla striata as an embolizing agent]. Zhonghua Fangshexue Zazhi. 1985;19:193-196. [PubMed] |
| 4. | Zhao LZ, Luo PF, Hu JQ, Guan YH, Chen XM, Liang CH. The significance of CT angiography and radiography in guiding the interventional treatment for hepatocellular carcinoma and prediction of prognosis. Zhonghua Fangshexue Zazhi. 1993;27:450-453. |
| 5. | Cheng YD, Qian XG, Zhang ZG, Jin JS, Shen BX. Analysis of survival time of hepatic carcinoma treated by intraarterial infusion chemotherapy and embolization. Zhonghua Xiaohua Zazhi. 1991;11:304-305. |
| 6. | Koehler RE, Korobkin M, Lewis F. Arteriographic demonstration of collateral arterial supply to the liver after hepatic artery ligation. Radiology. 1975;117:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 108] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 7. | Von Huppert PE, Gei Bler F, Duda SH, Lewis F. Chemoembolization of hepatocellular carcinoma: computed tomography appearances and clinical results by prospective repeat therapy study. Roentgenstr. 1994;106:425-428. |