Published online Nov 28, 2013. doi: 10.3748/wjg.v19.i44.7972
Revised: August 15, 2013
Accepted: October 17, 2013
Published online: November 28, 2013
Processing time: 165 Days and 7.6 Hours
Alcoholism and cirrhosis, which are two of the most serious health problems worldwide, have a broad spectrum of clinical outcomes. Both diseases are influenced by genetic susceptibility and cultural traits that differ globally but are specific for each population. In contrast to other regions around the world, Mexicans present the highest drinking score and a high mortality rate for alcoholic liver disease with an intermediate category level of per capita alcohol consumption. Mexico has a unique history of alcohol consumption that is linked to profound anthropological and social aspects. The Mexican population has an admixture genome inherited from different races, Caucasian, Amerindian and African, with a heterogeneous distribution within the country. Thus, genes related to alcohol addiction, such as dopamine receptor D2 in the brain, or liver alcohol-metabolizing enzymes, such as alcohol dehydrogenase class I polypeptide B, cytochrome P450 2E1 and aldehyde dehydrogenase class 2, may vary from one individual to another. Furthermore, they may be inherited as risk or non-risk haplogroups that confer susceptibility or resistance either to alcohol addiction or abusive alcohol consumption and possibly liver disease. Thus, in this era of genomics, personalized medicine will benefit patients if it is directed according to individual or population-based data. Additional association studies will be required to establish novel strategies for the prevention, care and treatment of liver disease in Mexico and worldwide.
Core tip: Alcoholism and liver disease are leading global health problems. However, the severity and outcome of liver disease appear to vary between individuals and populations. In the present review, we analyze the general scope of alcohol consumption and its relationship with the pattern of drinking score in different countries. We focus on the development of alcoholism in Mexico, which has a strong historical background, and emphasize the need to understand the genetic and environmental factors affecting each population or geographical region of the world.
- Citation: Roman S, Zepeda-Carrillo EA, Moreno-Luna LE, Panduro A. Alcoholism and liver disease in Mexico: Genetic and environmental factors. World J Gastroenterol 2013; 19(44): 7972-7982
- URL: https://www.wjgnet.com/1007-9327/full/v19/i44/7972.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i44.7972
The human history of alcohol consumption has been documented for several thousand years[1]. Alcohol was undoubtedly the result of a fortuitous coincidence that occurred when fruits, grains and flower stalks were fermented for a long time. People may have begun to experience pleasure and happiness after tasting alcoholic beverages[1,2].
Alcoholic beverages are obtained from different sources, depending on the region of the world. Traditionally, must and wines are produced from grapes of the Middle East and Europe, whisky is made from various grains and sake is obtained from rice in Asia. In Mexico, “pulque” was introduced first, followed by tequila, which are made from the maguey and agave plants, respectively[3,4].
Historically, alcohol-based beverages have served as a source of needed nutrients and have been widely used for their medicinal, antiseptic and analgesic properties. However, during the last century, alcohol abuse has increased in several countries, thereby augmenting the rate of accidents and liver diseases. The range of liver diseases secondary to alcohol consumption is extensive, including acute alcoholic hepatitis, alcoholic liver disease (ALD), cirrhosis and hepatocellular carcinoma[5]. Different factors may affect the development of alcoholic liver damage, including the dose, duration and type of alcohol consumption, drinking patterns, gender and ethnicity[1,6-8]. Other associated risk factors include obesity, iron overload, concomitant viral hepatitis infection[1,7,9] and genetic factors[7,8]. Nonetheless, the degree of the association among alcohol consumption, morbidity and mortality due to ALD varies among individuals and populations worldwide. Alcohol consumption and ALD are linked to specific genetic and environmental factors that are prevalent in each population. However, which factors and how they are involved in both alcohol addiction and the adaptation of hepatic genes capable of metabolizing large amounts of ethanol without developing liver disease are challenging questions.
In this comprehensive review, we revisit the information on the worldwide consumption of alcohol and patterns of drinking associated with liver disease, emphasizing the history of alcoholism in Mexico and the differences in the genetic and environmental backgrounds with respect to alcoholism and liver disease among the different countries, with a focus on the genetic factors involved in alcohol dependence and alcohol abuse as well as liver-metabolizing enzymes.
The World Health Organization (WHO) published the total adult per capita alcohol consumption (liters of pure alcohol consumption/year) by distinct geographical regions of the world[10]. Three primary categories, high (10-12 L and > 12 L), intermediate (7.5-9.99 L and 5-7.49 L) and low (2.5-4.99 L and < 2.5 L), were created to compare alcohol consumption among different countries.
The countries with the highest alcohol consumption are located primarily in Europe (Czech Republic, United Kingdom, Ireland, Germany, France, Portugal and the Russian Federation) but also in other regions, such as South Korea, Australia, Nigeria, Uganda and Argentina. The intermediate category includes countries located in the Americas, such as the United States, Canada, Mexico, Chile, Brazil and Colombia, a few African countries, such as Cameroon, South Africa, Namibia and Botswana, and Norway in Europe. The low alcohol consumption category includes several countries within the Eastern Mediterranean region and Asia, generally representing those countries where religious beliefs prohibit alcohol consumption.
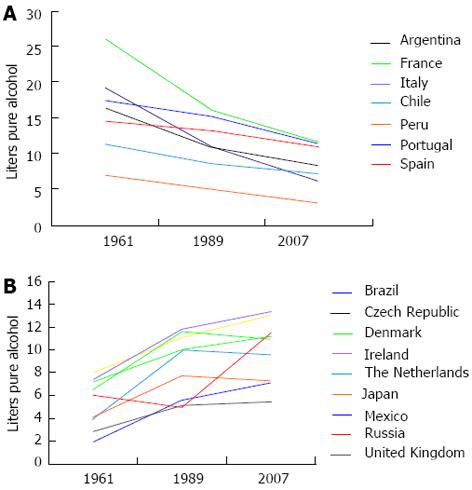
However, there have been different trends in the last 50 years regarding alcohol consumption in countries worldwide. Although several countries have increased alcohol consumption, others have decreased alcohol consumption (Figure 1). Furthermore, since 2008, the WHO has been in the process of drafting a global strategy to reduce the harmful use of alcohol[11]. These observations led us to analyze the effectiveness of these strategies to avoid or decrease alcohol consumption and to improve the understanding of the biological and social events involved in the drinking habits of alcohol in Mexico compared with other regions of the world.
To address these concerns, we examined the mortality related to ALD within several countries. Interestingly, there is a discrepancy between mortality related to ALD and the per-capita alcohol consumption[12]; e.g., Mexico is one of the countries with the highest mortality rate due to ALD but is not included among the countries with the highest alcohol consumption[12]. However, global comparisons among different populations are limited because not all countries report mortality related to ALD[12].
The pattern of drinking score is a composite scale that ranges from 1 to 5 and focuses primarily on the degree of risk associated with how the alcohol is consumed rather than the amount of alcohol consumed. To build this scale, the following indicators are used: quantity of alcohol consumed by occasion, festive drinking, proportion of drinking events that involve becoming drunk, proportion of drinkers who drink daily, drinking with meals and drinking in public places[13].
Unlike alcohol consumption, which is measured by the amount of pure alcohol per capita/year, the pattern of drinking score is closely related to ALD. For example, the countries with the highest pattern of drinking score are Kazakhstan, Mexico, the Russian Federation, South Africa and Ukraine, and the countries with a lower-risk pattern of drinking are Portugal, Spain, France, Italy and Germany[13].
Taken together, alcohol consumption indicators, mortality rates and pattern of drinking scores, which all may contribute to ALD, are heterogeneous worldwide[12,13]. Thus, because ALD is a multifactorial problem, researchers should consider the anthropological and historical aspects prevalent among the different societies.
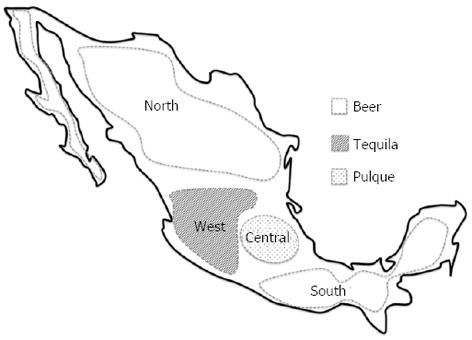
To understand the interaction between the evolutionary and genetic changes associated with specific environments, it is necessary to know when and how these events occurred among the different populations. In the case of Mexico, the establishment of a sedentary lifestyle required approximately 5000 years[14,15]. During this period, the Mesoamericans began the domestication of the well-known staple foods of Mexico, such as maize (Zea mays L.), beans (Phaseolus spp), squash and pumpkin (Curcubita spp) and chili (Capsicum spp). This process was accompanied by the discovery and consumption of fermented alcoholic beverages made from a number of endemic agave plants (Agave spp). The origin of alcoholic beverages, as described by the Aztecs, was a mythical love story between two deities, “Mayahuel” and “Quetzalcóatl” (Figure 2)[16-18].
The core of the mature agave plant produces a honey water, or “aguamiel”, rich in amino acids and proteins[19], which once fermented, produces the traditional alcoholic beverage. The Nahuas in their native language named the former “iztac octli” and the latter “octli poliuhqui”[20]. However, when the Spaniards arrived on the continent, “octli poliuhqui” was phonetically derived as the term “pulque”[20,21].
The “octli” but not “octli poliuhqui” served as nourishment for the elderly and sick and for women after childbirth[22]. The “octli” and perhaps the “octli poliuhqui” were given to all family members, including babies and children, in public ceremonies[17]. The “octli poliuhqui” was also used for medicinal purposes as an antidepressant or as an anesthetic before human sacrifice[16,18].
Additionally, the early Mexicans were familiar with the effects of the abuse of “octli poliuhqui”; thus, excessive drinking was strictly prohibited by law primarily during the religious holidays, and a death penalty was implemented[17]. The Aztec rulers often declared that the abuse of “octli poliuhqui” was the source and beginning of all evil and all ruin[17]. Unfortunately, these laws were not reinforced after the 15th century, granting a tolerance of the abusive consumption of alcoholic beverages during the colonial period[4,23].
The rich history of the consumption of “pulque” by the Mexicans over many centuries is an essential component of the framework that is required to understand the relationship among the Mexican genome, alcoholism and liver disease at present.
Mexico is one of the leading countries with a high mortality rate due to liver diseases in the world[10]. The National Health Secretariat reported an average of 25000 cases of cirrhosis per year from 2000 to 2010[24]. The primary etiologies of cirrhosis are alcohol, followed by hepatitis C infection and non-alcoholic steatohepatitis[25-27].
For Mexico, the WHO reported that the amount of alcohol consumed is 8.4 L of pure alcohol per capita among individuals older than 15 years of age, which corresponds to an intermediate category as previously described[10]. However, if this parameter is applied only to drinkers, alcohol consumption increases to 27.1 L, which is similar to what had been reported in countries with the highest levels of alcohol consumption per capita[10].
However, the pattern of drinking score shows a better scope of alcoholism among the Mexican population. By examining the amount of alcohol consumed by occasion, we observed that alcohol consumption occurs primarily during the weekends[28], unlike in Europe where they drink wine almost daily, at lunch or dinner.
Hepatologists may advise their patients not to drink any alcoholic beverage to maintain a healthy liver. However, a large proportion of adults around the world drink alcoholic beverages[1,5,6]. Thus, the recommendation to avoid liver damage is that the amount of alcohol consumed should be equal or less than 2 drinks per occasion (20-40 g ethanol), not more than 4 drinks per day and not more than 10 to 12 drinks per week, allowing the liver to rest at least 1 or 2 d[1,5,6]. Furthermore, it has been suggested that the number of alcoholic drinks should be less in women than in men because women have a higher risk for developing ALD[1,5,6].
However, each weekend, approximately 30 million Mexicans have been estimated to consume more than five drinks per occasion (more than 80 g of ethanol), with another 10 million consuming at least one alcoholic drink daily. However, alcohol abuse has been detected in 5 million people with a strong dependence on alcohol[28].
The average Mexican begins consuming alcohol before the age of 18 years perhaps because of a strong cultural influence. Studies conducted in the western region of Mexico have shown that 61.4% of the 12- to 17-year-old have already begun to drink alcohol[27]. The primary types of alcoholic beverages consumed in Mexico are beer, tequila and “pulque”, and other distilled beverages are consumed in a lower proportion[27-30]. However, the distribution of alcoholic beverage preferences is heterogeneous. Thus, in central Mexico, “pulque” is preferred, in contrast to tequila in the west or beer in the northern and southern parts of the country (Figure 3). These preferences are associated with the historical cultural background of each region and may be related to the mortality caused by cirrhosis. The mortality rate in central Mexico is greater than 30/100000, followed by the north at less than 10/100000 and the west at less than 5/100000[30,31].
In western Mexico, young people begin to drink beer either during the weekend or at any social or religious event, such as weddings, coming-of-age parties and christenings. After the initiation of alcohol use, the number of beers consumed per occasion over the weekend ranges from 4 to 6 (80-100 g); this number gradually increases to 20-24 beers/355 mL each (300-360 g of alcohol)/occasion per person over a period of approximately 10 years. The second stage involves the combination of beer with tequila or any other distilled beverage during a period of 8 to 10 years. During this stage, the amount of alcohol consumed ranges from 380 to 640 g daily. In the third stage, alcohol dependence is severe, and patients may or may not present with cirrhosis. By this time, they drink an average of 510 g of alcohol per day (450-720 g)[26,32-34].
The time between the initiation of alcohol use and the diagnosis of cirrhosis is 23 to 30 years[26,35]. However, we have identified two distinct age peaks of clinical cirrhosis. In the first group, patients are young, approximately 30 years old, and a plausible genetic predisposition to liver cirrhosis has been proposed to be involved. In the second group, the average age is approximately 45 years[33]. Compared to other countries, Mexico, according to our findings, may have the youngest people with alcoholic cirrhosis in the world. Apparently, the Apo E2[33] and FABP2[36] gene polymorphisms may be involved in the early onset of ALD among the Mexican population.
The majority of patients with ALD seek medical attention in the advanced stages of the disease with a Child-Pugh score of C and multiple complications, such as encephalopathy, variceal bleeding, infections and ascites[25,27,35]. These clinical characteristics are present in the two primary age groups of patients with alcoholic cirrhosis[33]. Furthermore, the patients with alcoholic cirrhosis continue to drink high amounts of alcohol after diagnosis and may die earlier in life due to clinical complications[25]. This observation may be one of the foremost reasons why hepatocellular carcinoma is rare in Mexico compared with other regions of the world[37,38], in conjunction with other environmental factors[39].
ALD has been associated with nutritional deficiencies and malnutrition worldwide[40]. However, preliminary data from a reference center in western Mexico have shown that obesity is also present. Among 90 patients, 17% of the alcoholic cirrhotic patients were malnourished, whereas overweight and obesity were detected in 33% of the patients, with another 50% of normal weight[35]. These data are consistent with the fact that Mexico has the highest prevalence of obesity[41], thus adding a new risk factor for liver disease. Furthermore, in this group of patients, 34% of the patients had drug addictions, which is an increasing social and health problem[35].
Thus, the combination of alcoholism, obesity, drugs and, in several cases, viral hepatitis B or C, leads us to explore specific strategies for treatments and prevention programs to detect cirrhosis at early stages of the disease.
In recent decades, researchers have been using various strategies to identify genes that may be associated with alcohol dependence or alcohol abuse. Studies based on candidate genes[42-45] or linkage disequilibrium were followed by the advances in genotyping that have resulted in the widespread use of genome-wide association studies[46,47]. Previous studies in families, twins and adoption studies have shown that approximately 40%-60% of the variance in the risk for developing alcoholism can be explained by genetic factors[43-47]. However, the interactions between genes and several environmental factors have led experts in the field to identify at least two types of alcoholism: (1) a more severe, more genetic and early-onset type of alcoholism; and (2) a less severe, more environmental and late-onset type of alcoholism[48-52].
Regarding the role of genetic factors in the susceptibility to alcohol dependence and alcohol abuse, research has primarily aimed to study the expression of brain and liver genes. For example, the major brain genes that modulate the neuroadaptative mechanism that translates alcohol stimuli into pleasure, anxiety or cravings are opioid receptor mu 1[53-55], catechol-O-methyltransferase[56], γ-aminobutyric acid receptor A[57,58], 5-hydroxytryptamine (serotonin) receptor adenylate cyclase-coupled[59,60], cholinergic receptor muscarinic 2[61,62], vesicular monoamine transporter 2[63-65] and dopamine receptor D2[48,66-68].
In the liver, several alcohol dehydrogenase (ADH) enzymes, primarily alcohol dehydrogenase class I polypeptide B (ADH1B)[52], cytochrome P450 2E1 (CYP2E1)[69] and aldehyde dehydrogenase class 2 (ALDH2)[52,70], and other minor ADHs, such as ADH1C[71] and ADH4[72], have been related to alcohol metabolism and alcoholism. The three major enzyme genes express variants with different catalytic activities (Vmax) and Michaelis constants (Km); thus, their ability to metabolize substrates is variable.
The combination of the allelic profile of these brain and liver genes may affect the risk of or protection against alcohol dependence or alcohol abuse as well as the amount of alcohol metabolized in the liver and the susceptibility to liver damage. Variances in the distribution of these gene polymorphisms may mark phenotypic differences among populations for the aforementioned features. Hence, for this review, the biological functions of dopamine receptor D2 (DRD2), ADH1B, CYP2E1 and ALDH2 are briefly described, and their global allelic frequencies are compared, including those reported for the Mexican population.
Alcohol has a stimulatory effect on the dopaminergic neurons of the ventral tegmental area. Dopamine is captured by DRD2 in these neurons in the nucleus accumbens, causing a pleasant effect that is integrated into the mesolimbic system[48,67,68].
The DRD2 Taq I A1 polymorphism consists of a T/C nucleotide substitution (rs1800497) that alters the Taq I restriction site located 10541 bp downstream of the termination codon. Several studies have investigated the association of this gene polymorphism with alcohol dependence. Taq I A1 allele carriers reportedly have lower amounts of DRD2 receptors than the Taq I A2 carriers[73]. Thus, A1 allele patients require higher amounts of alcohol to achieve the desired effect[66-68,74-76]. In additional studies, the association between the A1 allele Taq I and alcohol use disorders has been corroborated in some but not in others. However, in several meta-analyses, a significant association between Caucasian A1 allele carriers and alcohol addiction has been found[48,77].
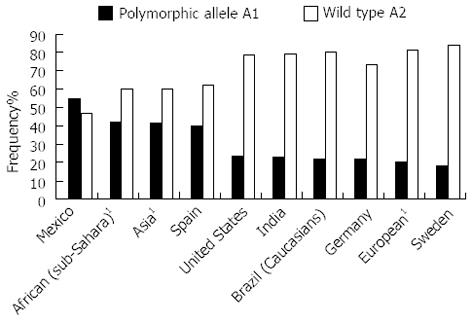
The allelic distribution of DRD2 displays a wide range of frequencies worldwide[74-79], but the highest prevalence of the A1 allele is found among the Amerindian Pima (83%) and Mayas (71%) from Mexico[80,81] (Figure 4).
The ADH1B gene has a polymorphic site, resulting in the Arg47His substitution (rs1229984). The A2 allele (ADH1B His47) confers a 100-fold higher catalytic activity to the ADH1B enzyme than the A1 allele (ADH1B Arg47). The A2 allele carriers have a higher ethanol oxidation capacity than the A1 carriers. However, the A2 carriers have a higher acetaldehyde production that leads to an alcohol-flushing response that has been considered to be protective.
The protective effect of the A2 allele against alcohol dependence is well known in the East Asian population[82]. A study conducted in a cohort of pregnant women from England demonstrated that the A2 carriers consumed less alcohol before pregnancy, had less incidents of binge drinking during pregnancy and were abstainers during the first trimester of gestation[83]. However, although this allele apparently reduces the risk of alcohol dependence, it may confer a substantial risk of chronic liver disease, especially among heavy drinkers[84].
In contrast, A1 allele carriers do not present with the rapid production of acetaldehyde, which eliminates the alcohol-flushing response. A study conducted in a large cohort of individuals from Copenhagen revealed that A1 allele homozygotes drank more alcohol and had a higher risk for developing alcohol dependence[85]. The A1 allele has also been associated with alcohol dependence in several other studies[86-88].
The CYP2E1 gene, which encodes the enzyme that oxidizes ethanol in the microsomal oxidation system, is essential in the pathophysiology of ALD. Alcohol consumption induces the expression of CYP2E1, which is directly involved in the primary oxidation pathway of this substrate, displacing the enzymatic activity of ADH1B (Km: 8-10 mmol/L for CYP2E1 vs 0.2-2.0 mmol/L for ADH1B)[69,89].
The C2 allele, which is less common, has a C/T transition at nucleotide position -1019 within the 5’ terminal regulatory region. The C2 allele is associated with a 100-fold higher transcription activity, higher protein concentration and increased enzyme activity, which lead to a faster rate of alcohol oxidation[90,91].
The C2 allele carriers have been demonstrated to consume excessive amounts of alcohol, which may be caused by the high transcriptional activity of CYP2E1. C2 allele carriers metabolize ethanol (alcohol) to acetaldehyde at a higher rate. Acetaldehyde is a highly toxic and mutagenic metabolite that increases oxidative stress by producing reactive oxygen species and lipid peroxides, such as 4-hydroxy-2,3-nonenal, 4-hydroxy-2,3-alkenals and malondialdehyde[92]. An association between an increased risk for ALD and alcoholic cirrhosis has been reported among the carriers of the C2 allele[93-95], several of whom belong to the mestizo population of western Mexico[96].
The ALDH2 gene encodes the primary mitochondrial isoform enzyme that oxidizes acetaldehyde to acetate in the liver[70,97]. The C/G transition in exon 12 of ALDH2 causes an amino acid substitution of glutamic acid for lysine at position 487 (ALDH2 Glu487Lys, rs671). The A2 allele (ALDH2 Lys487) has little or null enzymatic activity. This deficiency leads to the accumulation of acetaldehyde and consequently provokes a flushing response, which discourages alcohol drinking[97]. Because flushing is an undesirable symptom, it confers relative protection against abusive alcohol consumption. An association between A2 allele carriers and a lower risk for alcohol dependence and reduced alcohol use has been reported[98].
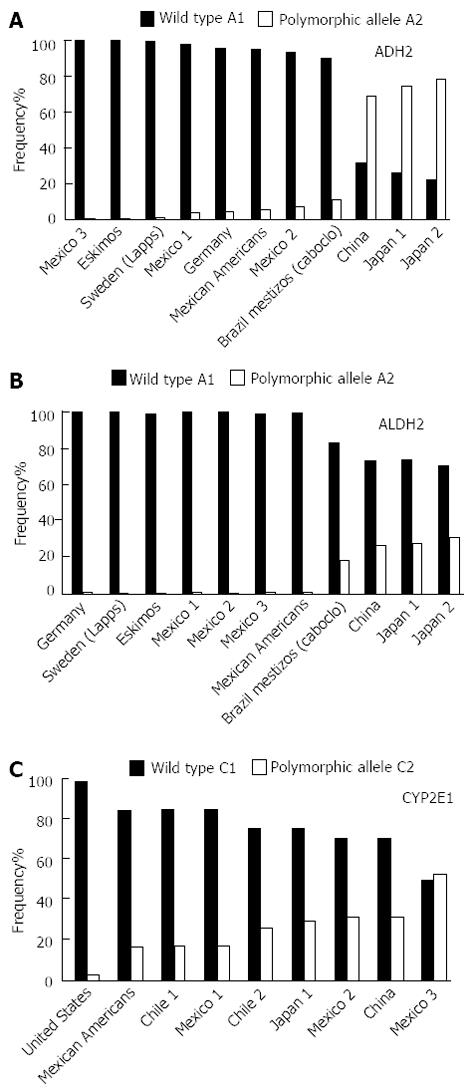
With regard to the distribution of the polymorphisms of the liver alcohol-metabolizing genes, all present contrasting frequencies among different population groups (Figure 5A-C). Among Asians, the ADH1B gene displays the highest frequency of the A2 protective allele, with 78% in Japan and 69% in China. In contrast, the lowest frequency for the A2 allele was detected in Germany and Mexico, at 4% and 3%, respectively (Figure 5A)[91,99-105], wherein the frequency of the A1 allele associated with alcohol dependence was much higher.
The C2 allele for the CYP2E1 gene has a frequency of approximately 30% in Japan and China and 2% in the United States. In Chile and Mexico, the frequency is 16% among the mestizo population (Figure 5B)[91,93,94,105-108]. Interestingly, among the Amerindians of western Mexico, such as the “Huichol” people, this gene polymorphism shows a prevalence of 50%, which is the highest rate reported to date[91].
The highest frequency of the A2 allele for the ALDH gene has been reported in China (29%) and Japan (26%). In Germany, Sweden and Mexico, its frequency is extremely low or absent[91,99-105], which could explain, to some extent, the high amount of alcohol consumption that has been reported in those countries.
We could speculate that the selective evolution of both the brain and liver genes was not necessarily directed only by the exposure to alcohol. For example, liver cytochrome genes metabolize a large variety of xenobiotics, whereas those expressed in the brain fulfill the addiction criteria. However, an alternative point of view is to consider these genes as part of a general survival mechanism. Thus, the basic biological necessities of life, such as food (sugars, e.g., glucose) or sexual reproduction, are ensured and rewarded by pleasure; however, these necessities may not be driven by pleasure exclusively.
At the end of the last century, we began to understand how liver genes are involved in the metabolism of ethanol and how cerebral genes are related to addictions. Additional genetic studies, including genome-wide association studies, will corroborate the association of specific alleles with alcoholism and ALD. The next step may be a personalized medicine strategy for the prevention, diagnosis and treatment of liver diseases. However, as aforementioned in this review, genes and environmental factors are involved in the development of ALD, which requires an in-depth analysis of the different populations. Therefore, the data found in several regions of the world may not correlate to populations from different geographic areas.
Mexicans are an admixture population that has inherited specific alleles from different races, predominantly Caucasian, Amerindian and African[109,110]. Based on the current data on allelic frequencies in different countries, the Mexican population has a particular genetic profile that may explain the epidemiological and clinical manifestations of alcohol-related liver diseases. Thus, the expectation that the different allelic variants of the aforementioned genes (DRD2, ADH2, CYP2E1 and ALDH2) will express themselves individually is plausible. However, considering these alleles as a haplogroup may generate risk or non-risk phenotypes related to liver disease, as well as to proneness towards or resistance against the high intake of alcohol. Haplogroups that could confer a non-risk phenotype for alcoholism and liver damage could be DRD2*A2, ADH2*A2, CYP2E1*C1, ALDH2*A2 and DRD2*A2, ADH2*A1, CYP2E1*C1, ALDH2*A2 because they are related to non-addiction plus flushing by the accumulation of acetaldehyde, exhibiting a protective effect. The haplogroups that could confer risk phenotypes for alcoholism and liver damage could be DRD2*A1, ADH2*A1, CYP2E1*C1, ALDH2*A1 and DRD2*A1, ADH2*A1, CYP2E1*C2, ALDH2*A1 because these are related to addiction plus the efficient metabolism of alcohol but exposure to acetaldehyde. This observation may explain why some patients who consume heavy amounts of alcohol per day (> 80 g/d) for more than 20 years do not have liver damage, whereas others with a less than or equal to consumption level and less exposure suffer liver damage and even die from cirrhosis or its complications[1,6,7]. However, additional studies are required to demonstrate the association between these hypothetical allelic profiles and the clinical outcomes of alcohol-dependent patients in Mexico and worldwide.
P- Reviewers: Luo XG, Pan Q, Romani A S- Editor: Gou SX L- Editor: A E- Editor: Wu HL
| 1. | O’Shea RS, Dasarathy S, McCullough AJ. Alcoholic liver disease. Hepatology. 2010;51:307-328. [PubMed] |
| 2. | Campollo O. El alcoholismo en México. Anuario de Investigación en Adicciones. 2009;10:96-106. |
| 3. | Belmont AD; Sandoval OF. Los orígenes prehispanicos del tradicional pulque. Alcoholismo beneficios y efectos deletéreos del etanol. México: Piensa 1997; 99. |
| 4. | Zamora RL. Mestizaje y El Tequila. Sincronía Año V (18). Available from: http//sincronia.cucsh.udg.mx/mestiz.htm. Accessed June 15, 2013. |
| 5. | Mann RE, Smart RG, Govoni R. The epidemiology of alcoholic liver disease. Alcohol Res Health. 2003;27:209-219. [PubMed] |
| 6. | Bellentani S, Saccoccio G, Costa G, Tiribelli C, Manenti F, Sodde M, Saveria Crocè L, Sasso F, Pozzato G, Cristianini G. Drinking habits as cofactors of risk for alcohol induced liver damage. The Dionysos Study Group. Gut. 1997;41:845-850. [PubMed] |
| 7. | Monzoni A, Masutti F, Saccoccio G, Bellentani S, Tiribelli C, Giacca M. Genetic determinants of ethanol-induced liver damage. Mol Med. 2001;7:255-262. [PubMed] |
| 8. | Douds AC, Cox MA, Iqbal TH, Cooper BT. Ethnic differences in cirrhosis of the liver in a British city: alcoholic cirrhosis in South Asian men. Alcohol Alcohol. 2003;38:148-150. [PubMed] |
| 9. | Tsochatzis EA, Bosch J, Burroughs AK. New therapeutic paradigm for patients with cirrhosis. Hepatology. 2012;56:1983-1992. [PubMed] |
| 10. | World Health Organization. Global Health Observatory (GHO), Global Information System on Alcohol and Health (GISAH) 2011. Available from: http://www.who.int/gho/alcohol/en/. |
| 11. | World Health Organization, Management of substance abuse. Global strategy to reduce the harmful use of alcohol 2010. Available from: http://www.who.int/substance_abuse/activities/gsrhua/en/index.html. |
| 12. | World Health Organization. Global Health Observatory Data Repository: Statistical Information System (WHOSIS) 2004. Available from: http://www.who.int/whosis/en/. |
| 13. | World Health Organization. Global Health Observatory (GHO) Global Information System on Alcohol and Health (GISAH): Patterns of consumption 2013. Available from: http://www.who.int/gho/alcohol/consumption_patterns/drinking_ score_patterns/en/index.htm. |
| 14. | Matos Moctezuma E. La agricultura en Mesoamérica. Arqueología Mexicana. 2013;19:29-36. |
| 15. | Montúfar A. Domesticación y cultivo de plantas alimenticias de México. Arqueología Mexicana. 2013;19:42-47. |
| 16. | González Torres Y. Los Dioses. El sacrificio humano entre los Mexicas. 2nd ed. México: D.F 1985; 141-161. |
| 17. | De Sahagun B. Historia general de las cosas de la Nueva España. Mexico: Editorial Porrúa 2006; 1-1061. |
| 18. | De Sahagun B. Fiestas y supersticiones de los antiguos mexicanos en la “Historia general” de Sahagun (Spanish Edition). Pilar Maynez, México: Fondo de la Cultura Económica 2006; 13-199. |
| 19. | Morales de León J, Camacho ME, Bourges H. Amino acid composition of some Mexican foods. Arch Latinoam Nutr. 2005;55:172-186. [PubMed] |
| 20. | García Escamilla E, Silva Galeana Enrique. Diccionario del Nahuatl en el español de México. 1st ed. México: Universidad Autónoma de México 2007; 8-440. |
| 21. | Robelo Cecilio A. Nombres geográficos indígenas del Estado de Mexico, (Estudio crítico etimológico), edición facsimilar de la de 1900. Toluca: Biblioteca Enciclopédica del Estado de Mexico 1974; 1-250. |
| 22. | Dávalos Hurtado E. Alimentos básicos e inventiva culinaria del mexicano. Serie: Peculiaridades Mexicanas. Mexico: D.F 2000; 5-62. |
| 23. | Alarcón-Segovia D, Bourges-Rodriguez H. La alimentación de los mexicanos. 1st ed. El Colegio Nacional, México: D.F 2002; 3-173. |
| 24. | Sistema Nacional de Información en Salud. Secretaria de Salud. Available from: http://www.sinais.salud.gob.mx/publicaciones/index.html. |
| 25. | Panduro A, Maldonado-Gonzalez M, Fierro NA, Roman S. Distribution of HBV genotypes F and H in Mexico and Central America. Antivir Ther. 2013;18:475-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 26. | Bastidas-Ramirez BE, Nuño-Gonzalez P, Vivas-Arceo C, Sánchez-Orozco LV, Panduro A. Albumin mRNA in peripheral white blood cells of cirrhotic patients with a superimposed alcoholic hepatitis is associated to fatal outcome. Hepatol Res. 2002;24:265. [PubMed] |
| 27. | Campollo O, Valencia-Salinas JJ, Berumen-Arellano A, Pérez-Aranda MA, Panduro-Cerda A, Segura-Ortega J. Epidemiological characteristics of liver cirrhosis at the Hospital Civil of Guadalajara. Salud Publica Mex. 1997;39:195-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 28. | Available from: http://www.conadic.salud.gob.mx/pdfs/ena08/ ENA08_NACIONAL.pdf. |
| 29. | Medina-Mora ME, Villatoro-Velázquez JA, Fleiz-Bautista C, Téllez-Rojo MM, Mendoza-Alvarado LR, Romero-Martínez M, Gutiérrez-Reyes JP, Castro-Tinoco M, Hernández-Ávila M, Tena-Tamayo CAlvear-Sevilla C, Guisa-Cruz V. Reporte de Alcohol. Encuesta Nacional de Adicciones 2011 (National Survey on Addictions, 2011). Available from: http://portal.salud.gob.mx/sites/salud/descargas/pdf/ENA_2011_ALCOHOL.pdf. |
| 30. | Narro-Robles J, Gutiérrez-Avila JH, López-Cervantes M, Borges G, Rosovsky H. Liver cirrhosis mortality in Mexico. I. Relevant epidemiological characteristics. Salud Publica Mex. 1992;34:378-387. [PubMed] |
| 31. | Narro-Robles J, Gutiérrez-Avila JH, López-Cervantes M, Borges G, Rosovsky H. Liver cirrhosis mortality in Mexico. II. Excess mortality and pulque consumption. Salud Publica Mex. 1992;34:388-405. [PubMed] |
| 32. | Campollo O, Martínez MD, Valencia JJ, Segura-Ortega J. Drinking patterns and beverage preferences of liver cirrhosis patients in Mexico. Subst Use Misuse. 2001;36:387-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 33. | Hernández-Nazará ZH, Ruiz-Madrigal B, Martínez-López E, Roman S, Panduro A. Association of the epsilon 2 allele of APOE gene to hypertriglyceridemia and to early-onset alcoholic cirrhosis. Alcohol Clin Exp Res. 2008;32:559-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 34. | Nuño-González P, Ruíz-Madrigal B, Bastidas-Ramírez BE, Martínez-López E, Segura JE, Panduro A. Expression of apolipoprotein AI mRNA in peripheral white blood cells of patients with alcoholic liver disease. Biochim Biophys Acta. 2005;1740:350-356. [PubMed] |
| 35. | Segura JE, Tinoco-Mar BA, Ramos ME, Fafutis-Morris M, Moreno-Luna LE. The impact of overweight and obesity in patients with alcoholic liver disease. Hepatol Int. 2013;7:S43. |
| 36. | Salguero ML, Leon RE, Santos A, Roman S, Segura-Ortega JE, Panduro A. The role of FABP2 gene polymorphism in alcoholic cirrhosis. Hepatol Res. 2005;33:306-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 37. | Pujol FH, Roman S, Panduro A, Navas MC, Lampe E. Hepatocellular carcinoma in Latin America. Hepatocellular carcinoma: a global challenge. New York: Nova Science Publishers 2012; 56-68. |
| 38. | Roman S, Panduro A, Aguilar-Gutierrez Y, Maldonado M, Vazquez-Vandyck M, Martinez-Lopez E, Ruiz-Madrigal B, Hernandez-Nazara Z. A low steady HBsAg seroprevalence is associated with a low incidence of HBV-related liver cirrhosis and hepatocellular carcinoma in Mexico: a systematic review. Hepatol Int. 2009;3:343-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 39] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 39. | Roman S, Fierro NA, Moreno-Luna LE, Panduro A. Hepatitis B virus genotype H and environmental factors associated to the low prevalence of hepatocellular carcinoma in Mexico. J Cancer Ther. 2013;4:367-376. [RCA] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 40. | Singal AK, Charlton MR. Nutrition in alcoholic liver disease. Clin Liver Dis. 2012;16:805-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 41. | Rojas-Martínez R, Aguilar-Salinas CA, Jiménez-Corona A, Gómez-Pérez FJ, Barquera S, Lazcano-Ponce E. Prevalence of obesity and metabolic syndrome components in Mexican adults without type 2 diabetes or hypertension. Salud Publica Mex. 2012;54:7-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 42. | Schuckit MA. Genetics of the risk for alcoholism. Am J Addict. 2000;9:103-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 96] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 43. | Goldman D, Oroszi G, Ducci F. The genetics of addictions: uncovering the genes. Nat Rev Genet. 2005;6:521-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 670] [Cited by in RCA: 650] [Article Influence: 32.5] [Reference Citation Analysis (0)] |
| 44. | Edenberg HJ, Foroud T. The genetics of alcoholism: identifying specific genes through family studies. Addict Biol. 2006;11:386-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 143] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 45. | Edenberg HJ, Foroud T. Genetics and alcoholism. Nat Rev Gastroenterol Hepatol. 2013;10:487-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 121] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 46. | Hill SY, Shen S, Zezza N, Hoffman EK, Perlin M, Allan W. A genome wide search for alcoholism susceptibility genes. Am J Med Genet B Neuropsychiatr Genet. 2004;128B:102-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 81] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 47. | Yan J, Aliev F, Webb BT, Kendler KS, Williamson VS, Edenberg HJ, Agrawal A, Kos MZ, Almasy L, Nurnberger JI Jr. Using genetic information from candidate gene and genome-wide association studies in risk prediction for alcohol dependence. Addict Biol. 2013;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 48. | Noble EP. Addiction and its reward process through polymorphisms of the D2 dopamine receptor gene: a review. Eur Psychiatry. 2000;15:79-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 184] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 49. | Flatscher-Bader T, van der Brug M, Hwang JW, Gochee PA, Matsumoto I, Niwa S, Wilce PA. Alcohol-responsive genes in the frontal cortex and nucleus accumbens of human alcoholics. J Neurochem. 2005;93:359-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 129] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 50. | Spanagel R, Bartsch D, Brors B, Dahmen N, Deussing J, Eils R, Ende G, Gallinat J, Gebicke-Haerter P, Heinz A, Kiefer F, Jäger W, Mann K, Matthäus F, Nöthen M, Rietschel M, Sartorius A, Schütz G, Sommer WH, Sprengel R, Walter H, Wichmann E, Wienker T, Wurst W, Zimmer A. An integrated genome research network for studying the genetics of alcohol addiction. Addict Biol. 2010;15:369-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 47] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 51. | Buscemi L, Turchi C. An overview of the genetic susceptibility to alcoholism. Med Sci Law. 2011;51 Suppl 1:S2-S6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 52. | Kimura M, Higuchi S. Genetics of alcohol dependence. Psychiatry Clin Neurosci. 2011;65:213-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 53. | Ray LA, Barr CS, Blendy JA, Oslin D, Goldman D, Anton RF. The role of the Asn40Asp polymorphism of the mu opioid receptor gene (OPRM1) on alcoholism etiology and treatment: a critical review. Alcohol Clin Exp Res. 2012;36:385-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 73] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 54. | Chen D, Liu L, Xiao Y, Peng Y, Yang C, Wang Z. Ethnic-specific meta-analyses of association between the OPRM1 A118G polymorphism and alcohol dependence among Asians and Caucasians. Drug Alcohol Depend. 2012;123:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 55. | Koller G, Zill P, Rujescu D, Ridinger M, Pogarell O, Fehr C, Wodarz N, Bondy B, Soyka M, Preuss UW. Possible association between OPRM1 genetic variance at the 118 locus and alcohol dependence in a large treatment sample: relationship to alcohol dependence symptoms. Alcohol Clin Exp Res. 2012;36:1230-1236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 56. | Schellekens AF, Franke B, Ellenbroek B, Cools A, de Jong CA, Buitelaar JK, Verkes RJ. Reduced dopamine receptor sensitivity as an intermediate phenotype in alcohol dependence and the role of the COMT Val158Met and DRD2 Taq1A genotypes. Arch Gen Psychiatry. 2012;69:339-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 57. | Edenberg HJ, Dick DM, Xuei X, Tian H, Almasy L, Bauer LO, Crowe RR, Goate A, Hesselbrock V, Jones K. Variations in GABRA2, encoding the alpha 2 subunit of the GABA(A) receptor, are associated with alcohol dependence and with brain oscillations. Am J Hum Genet. 2004;74:705-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 498] [Cited by in RCA: 482] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 58. | Zintzaras E. Gamma-aminobutyric acid A receptor, α-2 (GABRA2) variants as individual markers for alcoholism: a meta-analysis. Psychiatr Genet. 2012;22:189-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 59. | Lovenberg TW, Baron BM, de Lecea L, Miller JD, Prosser RA, Rea MA, Foye PE, Racke M, Slone AL, Siegel BW. A novel adenylyl cyclase-activating serotonin receptor (5-HT7) implicated in the regulation of mammalian circadian rhythms. Neuron. 1993;11:449-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 548] [Cited by in RCA: 541] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 60. | Zlojutro M, Manz N, Rangaswamy M, Xuei X, Flury-Wetherill L, Koller D, Bierut LJ, Goate A, Hesselbrock V, Kuperman S. Genome-wide association study of theta band event-related oscillations identifies serotonin receptor gene HTR7 influencing risk of alcohol dependence. Am J Med Genet B Neuropsychiatr Genet. 2011;156B:44-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 61. | Bonner TI, Buckley NJ, Young AC, Brann MR. Identification of a family of muscarinic acetylcholine receptor genes. Science. 1987;237:527-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1135] [Cited by in RCA: 1129] [Article Influence: 29.7] [Reference Citation Analysis (0)] |
| 62. | Hendershot CS, Bryan AD, Ewing SW, Claus ED, Hutchison KE. Preliminary evidence for associations of CHRM2 with substance use and disinhibition in adolescence. J Abnorm Child Psychol. 2011;39:671-681. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 63. | Peter D, Finn JP, Klisak I, Liu Y, Kojis T, Heinzmann C, Roghani A, Sparkes RS, Edwards RH. Chromosomal localization of the human vesicular amine transporter genes. Genomics. 1993;18:720-723. [PubMed] |
| 64. | Fehr C, Sommerlad D, Sander T, Anghelescu I, Dahmen N, Szegedi A, Mueller C, Zill P, Soyka M, Preuss UW. Association of VMAT2 gene polymorphisms with alcohol dependence. J Neural Transm. 2013;120:1161-1169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 65. | Schwab SG, Franke PE, Hoefgen B, Guttenthaler V, Lichtermann D, Trixler M, Knapp M, Maier W, Wildenauer DB. Association of DNA polymorphisms in the synaptic vesicular amine transporter gene (SLC18A2) with alcohol and nicotine dependence. Neuropsychopharmacology. 2005;30:2263-2268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 34] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 66. | Heinz A, Siessmeier T, Wrase J, Hermann D, Klein S, Grüsser SM, Flor H, Braus DF, Buchholz HG, Gründer G. Correlation between dopamine D(2) receptors in the ventral striatum and central processing of alcohol cues and craving. Am J Psychiatry. 2004;161:1783-1789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 256] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 67. | Barnard ND, Noble EP, Ritchie T, Cohen J, Jenkins DJ, Turner-McGrievy G, Gloede L, Green AA, Ferdowsian H. D2 dopamine receptor Taq1A polymorphism, body weight, and dietary intake in type 2 diabetes. Nutrition. 2009;25:58-65. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 62] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 68. | Gilpin NW, Koob GF. Neurobiology of Alcohol Dependence: Focus on Motivational Mechanisms. Alcohol Res Health. 2008;31:185-195. [PubMed] |
| 69. | Zakhari S. Overview: how is alcohol metabolized by the body? Alcohol Res Health. 2006;29:245-254. [PubMed] |
| 70. | Pautassi RM, Camarini R, Quadros IM, Miczek KA, Israel Y. Genetic and environmental influences on ethanol consumption: perspectives from preclinical research. Alcohol Clin Exp Res. 2010;34:976-987. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 71. | Li D, Zhao H, Gelernter J. Further clarification of the contribution of the ADH1C gene to vulnerability of alcoholism and selected liver diseases. Hum Genet. 2012;131:1361-1374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 72. | Preuss UW, Ridinger M, Rujescu D, Samochowiec J, Fehr C, Wurst FM, Koller G, Bondy B, Wodarz N, Debniak T. Association of ADH4 genetic variants with alcohol dependence risk and related phenotypes: results from a larger multicenter association study. Addict Biol. 2011;16:323-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 73. | Volkow ND, Wang GJ, Fowler JS, Logan J, Hitzemann R, Ding YS, Pappas N, Shea C, Piscani K. Decreases in dopamine receptors but not in dopamine transporters in alcoholics. Alcohol Clin Exp Res. 1996;20:1594-1598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 393] [Cited by in RCA: 365] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 74. | Bau CH, Almeida S, Hutz MH. The TaqI A1 allele of the dopamine D2 receptor gene and alcoholism in Brazil: association and interaction with stress and harm avoidance on severity prediction. Am J Med Genet. 2000;96:302-306. [PubMed] |
| 75. | Berggren U, Fahlke C, Berglund KJ, Wadell K, Zetterberg H, Blennow K, Thelle D, Balldin J. Dopamine D2 receptor genotype is associated with increased mortality at a 10-year follow-up of alcohol-dependent individuals. Alcohol Alcohol. 2010;45:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 76. | Haberstick BC, Timberlake D, Smolen A, Sakai JT, Hopfer CJ, Corley RP, Young SE, Stallings MC, Huizinga D, Menard S. Between- and within-family association test of the dopamine receptor D2 TaqIA polymorphism and alcohol abuse and dependence in a general population sample of adults. J Stud Alcohol Drugs. 2007;68:362-370. [PubMed] |
| 77. | Munafò MR, Matheson IJ, Flint J. Association of the DRD2 gene Taq1A polymorphism and alcoholism: a meta-analysis of case-control studies and evidence of publication bias. Mol Psychiatry. 2007;12:454-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 169] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 78. | Sweetlove MA, Lötter MG, Roodt JP, Badenhorst PN, Kotzé HF, Heyns AD. Blood platelet kinetics in normal subjects modelled by compartmental analysis. Eur J Nucl Med. 1992;19:1023-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 70] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 79. | Prasad P, Ambekar A, Vaswani M. Dopamine D2 receptor polymorphisms and susceptibility to alcohol dependence in Indian males: a preliminary study. BMC Med Genet. 2010;11:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 80. | Rajeevan H, Osier MV, Cheung KH, Deng H, Druskin L, Heinzen R, Kidd JR, Stein S, Pakstis AJ, Tosches NP. ALFRED: the ALelle FREquency Database. Update. Nucleic Acids Res. 2003;31:270-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 70] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 81. | HapMap I The International HapMap Project. Nature. 2003;426:789-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4437] [Cited by in RCA: 4316] [Article Influence: 205.5] [Reference Citation Analysis (0)] |
| 82. | Higuchi S, Kono H. Early diagnosis and treatment of alcoholism: the Japanese experience. Alcohol Alcohol. 1994;29:363-373. [PubMed] |
| 83. | Zuccolo L, Fitz-Simon N, Gray R, Ring SM, Sayal K, Smith GD, Lewis SJ. A non-synonymous variant in ADH1B is strongly associated with prenatal alcohol use in a European sample of pregnant women. Hum Mol Genet. 2009;18:4457-4466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 47] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 84. | Toth R, Pocsai Z, Fiatal S, Szeles G, Kardos L, Petrovski B, McKee M, Adany R. ADH1B*2 allele is protective against alcoholism but not chronic liver disease in the Hungarian population. Addiction. 2010;105:891-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 85. | Tolstrup JS, Nordestgaard BG, Rasmussen S, Tybjaerg-Hansen A, Grønbaek M. Alcoholism and alcohol drinking habits predicted from alcohol dehydrogenase genes. Pharmacogenomics J. 2008;8:220-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 57] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 86. | Whitfield JB. Alcohol dehydrogenase and alcohol dependence: variation in genotype-associated risk between populations. Am J Hum Genet. 2002;71:1247-150; author reply 1247-150;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 75] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 87. | Macgregor S, Lind PA, Bucholz KK, Hansell NK, Madden PA, Richter MM, Montgomery GW, Martin NG, Heath AC, Whitfield JB. Associations of ADH and ALDH2 gene variation with self report alcohol reactions, consumption and dependence: an integrated analysis. Hum Mol Genet. 2009;18:580-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 160] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 88. | van Beek JH, Willemsen G, de Moor MH, Hottenga JJ, Boomsma DI. Associations between ADH gene variants and alcohol phenotypes in Dutch adults. Twin Res Hum Genet. 2010;13:30-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 89. | Lieber CS. Cytochrome P-4502E1: its physiological and pathological role. Physiol Rev. 1997;77:517-544. [PubMed] |
| 90. | Hayashi S, Watanabe J, Kawajiri K. Genetic polymorphisms in the 5’-flanking region change transcriptional regulation of the human cytochrome P450IIE1 gene. J Biochem. 1991;110:559-565. [PubMed] |
| 91. | Gordillo-Bastidas E, Panduro A, Gordillo-Bastidas D, Zepeda-Carrillo EA, García-Bañuelos JJ, Muñoz-Valle JF, Bastidas-Ramírez BE. Polymorphisms of alcohol metabolizing enzymes in indigenous Mexican population: unusual high frequency of CYP2E1*c2 allele. Alcohol Clin Exp Res. 2010;34:142-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 92. | Abdelmegeed MA, Banerjee A, Yoo SH, Jang S, Gonzalez FJ, Song BJ. Critical role of cytochrome P450 2E1 (CYP2E1) in the development of high fat-induced non-alcoholic steatohepatitis. J Hepatol. 2012;57:860-866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 198] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 93. | Grove J, Brown AS, Daly AK, Bassendine MF, James OF, Day CP. The RsaI polymorphism of CYP2E1 and susceptibility to alcoholic liver disease in Caucasians: effect on age of presentation and dependence on alcohol dehydrogenase genotype. Pharmacogenetics. 1998;8:335-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 64] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 94. | Iwahashi K, Ameno S, Ameno K, Okada N, Kinoshita H, Sakae Y, Nakamura K, Watanabe M, Ijiri I, Harada S. Relationship between alcoholism and CYP2E1 C/D polymorphism. Neuropsychobiology. 1998;38:218-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 95. | Pirmohamed M, Kitteringham NR, Quest LJ, Allott RL, Green VJ, Gilmore IT, Park BK. Genetic polymorphism of cytochrome P4502E1 and risk of alcoholic liver disease in Caucasians. Pharmacogenetics. 1995;5:351-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 48] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 96. | García-Bañuelos J, Panduro A, Gordillo-Bastidas D, Gordillo-Bastidas E, Muñoz-Valle JF, Gurrola-Díaz CM, Sánchez-Enríquez S, Ruiz-Madrigal B, Bastidas-Ramírez BE. Genetic polymorphisms of genes coding to alcohol-metabolizing enzymes in western Mexicans: association of CYP2E1*c2/CYP2E1*5B allele with cirrhosis and liver function. Alcohol Clin Exp Res. 2012;36:425-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 97. | Mulligan CJ, Robin RW, Osier MV, Sambuughin N, Goldfarb LG, Kittles RA, Hesselbrock D, Goldman D, Long JC. Allelic variation at alcohol metabolism genes ( ADH1B, ADH1C, ALDH2) and alcohol dependence in an American Indian population. Hum Genet. 2003;113:325-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 70] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 98. | Irons DE, Iacono WG, Oetting WS, McGue M. Developmental trajectory and environmental moderation of the effect of ALDH2 polymorphism on alcohol use. Alcohol Clin Exp Res. 2012;36:1882-1891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 99. | Matsuo K, Wakai K, Hirose K, Ito H, Saito T, Suzuki T, Kato T, Hirai T, Kanemitsu Y, Hamajima H. A gene-gene interaction between ALDH2 Glu487Lys and ADH2 His47Arg polymorphisms regarding the risk of colorectal cancer in Japan. Carcinogenesis. 2006;27:1018-1023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 100. | Goedde HW, Agarwal DP, Fritze G, Meier-Tackmann D, Singh S, Beckmann G, Bhatia K, Chen LZ, Fang B, Lisker R. Distribution of ADH2 and ALDH2 genotypes in different populations. Hum Genet. 1992;88:344-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 384] [Cited by in RCA: 373] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 101. | Yang SJ, Wang HY, Li XQ, Du HZ, Zheng CJ, Chen HG, Mu XY, Yang CX. Genetic polymorphisms of ADH2 and ALDH2 association with esophageal cancer risk in southwest China. World J Gastroenterol. 2007;13:5760-5764. [PubMed] |
| 102. | Konishi T, Smith JL, Lin KM, Wan YJ. Influence of genetic admixture on polymorphisms of alcohol-metabolizing enzymes: analyses of mutations on the CYP2E1, ADH2, ADH3 and ALDH2 genes in a Mexican-American population living in the Los Angeles area. Alcohol Alcohol. 2003;38:93-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 103. | Saito K, Yokoyama T, Yoshiike N, Date C, Yamamoto A, Muramatsu M, Tanaka H. Do the ethanol metabolizing enzymes modify the relationship between alcohol consumption and blood pressure? J Hypertens. 2003;21:1097-1105. [PubMed] |
| 104. | Montano Loza AJ, Ramirez Iglesias MT, Perez Diaz I, Cruz Castellanos S, Garcia Andrade C, Medina Mora ME, Robles Díaz G, Kershenobich D, Gutierrez Reyes G. Association of alcohol-metabolizing genes with alcoholism in a Mexican Indian (Otomi) population. Alcohol. 2006;39:73-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 105. | Mendoza-Cantú A, Castorena-Torres F, Bermudez M, Martínez-Hernández R, Ortega A, Salinas JE, Albores A. Genotype and allele frequencies of polymorphic cytochromes P450 CYP1A2 and CYP2E1 in Mexicans. Cell Biochem Funct. 2004;22:29-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 106. | Quiñones L, Lucas D, Godoy J, Cáceres D, Berthou F, Varela N, Lee K, Acevedo C, Martínez L, Aguilera AM. CYP1A1, CYP2E1 and GSTM1 genetic polymorphisms. The effect of single and combined genotypes on lung cancer susceptibility in Chilean people. Cancer Lett. 2001;174:35-44. [PubMed] |
| 107. | Muñoz S, Vollrath V, Vallejos MP, Miquel JF, Covarrubias C, Raddatz A, Chianale J. Genetic polymorphisms of CYP2D6, CYP1A1 and CYP2E1 in the South-Amerindian population of Chile. Pharmacogenetics. 1998;8:343-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 108. | Wan YJ, Poland RE, Lin KM. Genetic polymorphism of CYP2E1, ADH2, and ALDH2 in Mexican-Americans. Genet Test. 1998;2:79-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 109. | 109Aceves D, Ruiz B, Nuño P, Roman S, Zepeda E, Panduro A. Heterogeneity of apolipoprotein E polymorphism in different Mexican populations. Hum Biol. 2006;78:65-75. [PubMed] |
| 110. | Rangel-Villalobos H, Salazar-Flores J, Dondiego R, Anaya-Palafox M, Nuño-Arana I, Canseco-Ávila LM, Rubi-Castellanos R. South to North increasing gradient of paternal European ancestry throughout the Mexican territory: Evidence of Y-linked short tandem repeats. Forensic Sci Int Genet. 2009;2:448-450. [RCA] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |