Published online Jun 14, 2013. doi: 10.3748/wjg.v19.i22.3415
Revised: February 22, 2013
Accepted: April 13, 2013
Published online: June 14, 2013
Processing time: 181 Days and 7 Hours
AIM: To investigate the disruptions of interstitial cells of Cajal (ICC) in the remaining bowel in rats after massive small bowel resection (mSBR).
METHODS: Thirty male Sprague-Dawley rats fitting entry criteria were divided randomly into three experimental groups (n = 10 each): Group A rats underwent bowel transection and re-anastomosis (sham) and tissue samples were harvested at day 7 post-surgery. Group B and C rats underwent 80% small bowel resection with tissue harvested from Group B rats at day 7 post-surgery, and from Group C rats at day 14 post-surgery. The distribution of ICC at the site of the residual small bowel was evaluated by immunohistochemical analysis of small intestine samples. The ultrastructural changes of ICC in the remnant ileum of model rats 7 and 14 d after mSBR were analyzed by transmission electron microscopy. Intracellular recordings of slow wave oscillations were used to evaluate electrical pacemaking. The protein expression of c-kit, ICC phenotypic markers, and membrane-bound stem cell factor (mSCF) in intestinal smooth muscle of each group were detected by Western blotting.
RESULTS: After mSBR, immunohistochemical analysis indicated that the number of c-kit-positive cells was dramatically decreased in Group B rats compared with sham tissues. Significant ultrastructural changes in ICC with associated smooth muscle hypertrophy were also observed. Disordered spontaneous rhythmic contractions with reduced amplitude (8.5 ± 1.4 mV vs 24.8 ± 1.3 mV, P = 0.037) and increased slow wave frequency (39.5 ± 2.1 cycles/min vs 33.0 ± 1.3 cycles/min, P = 0.044) were found in the residual intestinal smooth muscle 7 d post mSBR. The contractile function and electrical activity of intestinal circular smooth muscle returned to normal levels at 14 d post mSBR (amplitude, 14.9 ± 1.6 mV vs 24.8 ± 1.3 mV; frequency, 30.7 ± 1.7 cycles/min vs 33.0 ± 1.3 cycles/min). The expression of Mscf and c-kit protein was decreased at 7 d (P = 0.026), but gradually returned to normal levels at 14 d. The ICC and associated neural networks were disrupted, which was associated with the phenotype alterations of ICC.
CONCLUSION: Massive small bowel resection in rats triggered damage to ICC networks and decreased the number of ICC leading to disordered intestinal rhythmicity. The mSCF/c-kit signaling pathway plays a role in the regulation and maintenance of ICC phenotypes.
Core tip: Several gastrointestinal motility diseases are associated with altered numbers of interstitial cells of Cajal (ICC). Short bowel syndrome is also characterized by disordered intestinal motility immediately after surgery. We have investigated the alterations in numbers and functional changes of ICC that occur as a result of short bowel syndrome. In summary, our study showed modifications of the ultrastructure morphology of ICC, altered numbers of ICC and subsequent altered electrophysiological functional activity in the ileum after massive small bowel resection. However, the association between motility disorders and the changes of ICC should be further evaluated.
- Citation: Chen J, Du L, Xiao YT, Cai W. Disruption of interstitial cells of Cajal networks after massive small bowel resection. World J Gastroenterol 2013; 19(22): 3415-3422
- URL: https://www.wjgnet.com/1007-9327/full/v19/i22/3415.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i22.3415
Short bowel syndrome (SBS) is characterized by disordered intestinal motility immediately after surgery[1]. After massive small bowel resection (mSBR), the remaining bowel undergoes a compensatory process termed adaptation. During the process of adaptation, the small intestine smooth muscle cells undergo dramatic changes in gross morphology and ultrastructure. Adaptation is associated with the hypertrophy of smooth muscle, which is a physiological response to the increased functional requirement placed on the residual small bowel[2]. In addition, hypertrophy of smooth muscle tissue produces distinct motility disorders in the intestinal remnant, resulting in malabsorption and loss of nutrients because of diarrhea. Therefore, there is a critical need for further investigation of the mechanisms regulating resection-induced adaptation and potential targets that could be developed as therapeutic strategies. In a previous study, we found that resection-induced intestinal adaptation in a rat SBR model involved both the mucosal and intestinal smooth muscle layers. However, the mechanisms regulating adaptation remain unclear[3].
Interstitial cells of Cajal (ICC) reside in the tunica muscularis of the gastrointestinal tract[4]. ICC play a crucial role in gastrointestinal motility in concert with the enteric nervous system, which is composed of both the myenteric (inter-muscular) plexus and the submucosal plexus[5]. ICC are present in organs containing smooth muscle tissue and are pacemaker cells that provide the basal electrical rhythm, which controls peristalsis in the gastrointestinal tract[6]. After receiving inputs from motor neurons, ICC generate and propagate electrical activity[7,8]. ICC are specialized cells in the gastrointestinal tract smooth muscle organs that express the c-kit receptor tyrosine kinase[9]. ICC can be identified by the expression of CD117 (c-kit), which is a membrane receptor with tyrosine kinase activity[5,10,11].
Intracellular signaling via c-kit plays a key role in the development and maintenance of the ICC phenotype and functional activity of ICC in the gastrointestinal tract[7,12]. Maintenance of the ICC phenotype requires membrane-bound stem cell factor (mSCF) produced locally within the tunica muscularis[13-16]. To date, remarkably few studies have investigated the functional changes of ICC that occur as a result of SBS. We hypothesize that the disruption to ICC activity involves the downregulation of the mSCF/c-kit signaling pathway following massive mSBR. In the current study, alterations in ICC phenotype and pacemaker activity were evaluated in a model of mSBR.
Thirty male Sprague-Dawley rats weighing 250-300 g were obtained from the Experimental Animal Center of Shanghai Jiaotong University School of Medicine. All animals were housed in metabolic cages with free access to food and water and acclimated to their environment for 5 d before experimentation. Animals were maintained under standardized temperature, humidity and 12 h light-dark cycles. The rats were divided randomly into three experimental groups (n = 10 each): Group A rats underwent bowel transection and re-anastomosis (sham); Groups B and C rats underwent 80% small bowel resection; Group A and Group B (SBS1W) bowel tissues were harvested at day 7 post-surgery; Group C (SBS2W) bowel tissues were harvested at day 14 post-surgery.
Animals were fasted for 16 h prior to laparotomy, and intestinal surgery was performed the following morning, as previously described[17]. All operative procedures were performed under anesthesia by intraperitoneal (ip) injection of pentobarbital sodium (30 mg/mL), which was administered at doses of 33-40 mg/kg body weight. Briefly, during surgery, the abdomen was opened by a midline incision and the ligament of Treitz and the ileal-cecal junction was identified and marked. For SBR rats, enterectomy was performed by removing approximately 80% of the small intestine, leaving approximately 10 cm of the terminal ileum and 5 cm of the proximal jejunum, which were anastomosed. For sham-operated control rats, the laparotomy and all surgical manipulations were the same as above, but the resection procedure was not carried out. All animals received fluid resuscitation by ip injection of saline (10 mL 0.9% NaCl) before the abdominal wall was closed and surgery concluded. After recovery, rats were transferred back to individual cages and given water ad libitum overnight, after which their regular diet was reinstated.
All experimental protocols were approved by the local Animal Care Committee and conformed to the Guide for the Care and Use of Laboratory Animals published by the Science and Technology Commission of the People’s Republic of China (STCC Publication No. 2, revised 1988).
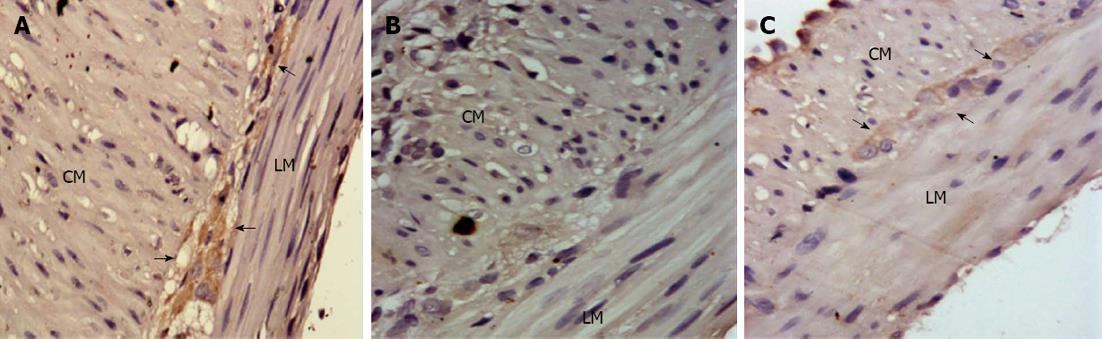
Immunohistochemical analysis was performed on tissue samples of smooth muscle at the same site of the ileum from both control and mSBR rats. The bowel was opened along the mesenteric border and the lumen contents were washed away with Krebs Ringer buffer. Segments of the bowel were pinned to the base of a Sylgard silicone elastomer dish and the mucosa was removed by sharp dissection. After dissection, the tunica muscularis were embedded in Tissue-Tek® OCT compound medium (Sakura Finetek United States, Inc., Torrance, CA, United States). The embedded tunica muscularis was cut at a thickness of 30 μm with a freezing microtome (Leica Microsystems GmbH, Wetzlar, Germany) and stored at -20 °C until use. The sections were fixed with iced acetone for 10 min. After endogenous peroxidase activity was quenched with 3% hydrogen peroxide, the sections were preincubated in 10% goat serum/0.2% Triton X-100/0.1 mol/L phosphate buffered saline (PBS) for 1 h at room temperature. The sections were incubated with antibodies against c-kit protein (1:50 dilution, rabbit polyclonal c-kit antibody, Santa Cruz Technologies, Santa Cruz, CA, United States) in 0.1 mol/L PBS containing 1% goat serum for 2 h. The sections were then incubated for 1 h at room temperature with biotinylated anti-rabbit immunoglobulin G (1:200; Vector Labs, Burlingame, CA, United States). Positive staining was visualized using an avidin-biotin-peroxidase complex system (Vectastain ABC Elite Kit, Vector Labs). The degree of expression of c-kit antibody was analyzed using image analysis software (Zeiss).
Tissue samples from intestinal smooth muscle at the same site of the ileum from both control and mSBR rats were fixed with 3% glutaraldehyde at room temperature. After fixation, the tissues were washed overnight in 0.1 mol/L sodium cacodylate buffer [6% sucrose and 1.25 mmol/L CaCl2 (pH 7.4)] at 4 °C and postfixed with 1% osmium tetroxide in 0.05 mol/L sodium cacodylate buffer (pH 7.4) at 4 °C for 2 h. The tissues were stained with saturated uranyl acetate for 3.5 h at room temperature, dehydrated in graded alcohol and embedded in Eponate 12 resin (Ted Pella, Inc., United States). The tissue was sectioned parallel and transverse to the long axis of the circular muscle layer. At suitable sites, 3 μm sections were cut and stained with 2% toluidine blue. After examination of the toluidine blue stained sections, ultrathin sections of selected areas were obtained with the ultramicrotome using a diamond knife, mounted on 200-mesh grids, and stained with uranyl acetate and lead citrate. The grids were observed with a JEM1200EX electron microscope.
Following euthanasia, a 50 mm mid-segment of the remaining small bowel was removed and placed in Krebs solution (mmol/L; NaCl 117, KCl 4.7, NaHCO3 25, KH2PO4 1.2, MgSO4 1.2, D-glucose 11, CaCl2 2.6). The bowel was opened along the mesenteric border, and the luminal contents were washed away with Krebs solution. Segments of the bowel were pinned to the base of a Sylgard silicone elastomeric dish (Dow Corning, Midland, MI, United States), and the mucosa was removed by sharp dissection[18]. Strips of smooth muscle tissue (8 mm × 4 mm) were cut parallel to the longitudinal muscle oral to the site of the occlusion clips. The muscle was placed in a recording chamber with the submucosal aspect of the muscle facing upwards at 37 °C in an atmosphere of 95% O2 and 5% CO2. Cells were impaled with KCl-filled glass microelectrodes with resistances of 50-90 MΩ. Electrical responses were recorded and amplified through a high input impedance amplifier (SYS-773 Duo 773 Electrometer, WPI, United States). Experiments were performed in the presence of nifedipine (1 μmol/L; Sigma, St Louis, MO, United States) in order to reduce contractions and facilitate the extended period of cell impalement. Slow waves in mouse intestine have been previously shown to be unaffected by nifedipine[19].
Smooth muscle tissues were lysed in radioimmunoprecipitation assay buffer [25 mmol/L Tris-HCl pH 7.6, 150 mmol/L NaCl, 1% NP-40, 1% sodium deoxycholate, 0.1% sodium dodecyl sulfate (SDS)] for the detection of protein expression levels. The Bradford method (Pierce, Rockford, IL, United States) was used to determine protein concentrations. Tissue lysates (20 μg of total protein per lane) were subjected to electrophoretic separation by 10% SDS-polyacrylamide gel electrophoresis and transferred to nitrocellulose membranes (Hybond, GE Healthcare Biosciences, Pittsburgh, PA, United States). Nonspecific binding was reduced by incubation of the membrane in 5% milk. Western blots were performed using antibodies directed against c-kit (1:200 dilution, rabbit polyclonal c-kit antibody, Santa Cruz Technologies, Santa Cruz, CA, United States), murine stem cell factor (mSCF) (1:200 dilution, mouse monoclonal SCF antibody, Santa Cruz Technologies), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (1:600 dilution, rabbit monoclonal antibody, CWbiotech company, Beijing, China). Alkaline phosphatase conjugated secondary antibodies (CWbiotech Company Beijing, China) were used to detect protein bands. TIFF images were captured by Adobe Photoshop and analyzed by Quantity one image software (NIH, MA, United States).
The results are presented as mean ± SE. Statistical differences between the groups were determined using a one-way analysis of variance with the SigmaStat program (SPSS, United States). P < 0.05 was considered statistically significant.
Representative pictures of c-kit immunopositivity at the myenteric plexus are shown in Figure 1. In sham-operated rats, c-kit-positive cells were predominantly present at the myenteric plexus level. However, the number and density of c-kit immunopositive cells were significantly decreased in SBS1W rats (P < 0.05) compared with sham tissues. However, after 2 wk, the number and density of ICC were clearly increased and approached sham levels, as indicated by c-kit positive cells (Figure 1).
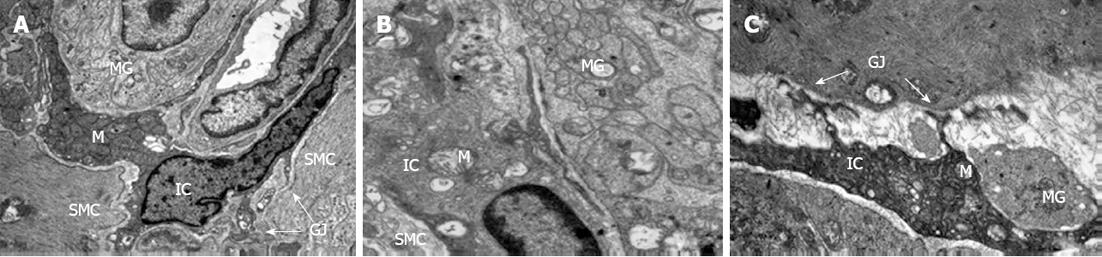
In sham rats, ultrastructural features of ICC in the small intestine were characterized by a less electron-dense cytoplasm and abundant mitochondria in sham rats (Figure 2A). However, the basal lamina or caveolae were not present. Intermediate filaments and thin filaments were apparent as thin processes. Along the length of overlapping processes, ICC predominantly formed large gap junctions between adjoining cells (Figure 2A). In certain areas, slender cytoplasmic processes of ICC were in close contact with a varicosity of the myenteric ganglion. Another part of the same cellular process was connected to a muscle cell via a gap junction, suggesting a functional relationship between these cell types.
Altered ICC ultrastructure was observed in the small intestine of SBS1W rats including clear cytoplasm, sparse mitochondria, scarce smooth endoplasmic reticulum, and a reduced number of contacts between nerves and ICC (Figure 2B). The ultrastructural abnormalities of ICC in the small intestine of SBS2W rats exhibited some improvement compared to SBS1W rats, but the ultrastructural morphology had not returned to normal as observed in the sham-operated rats (Figure 2C).
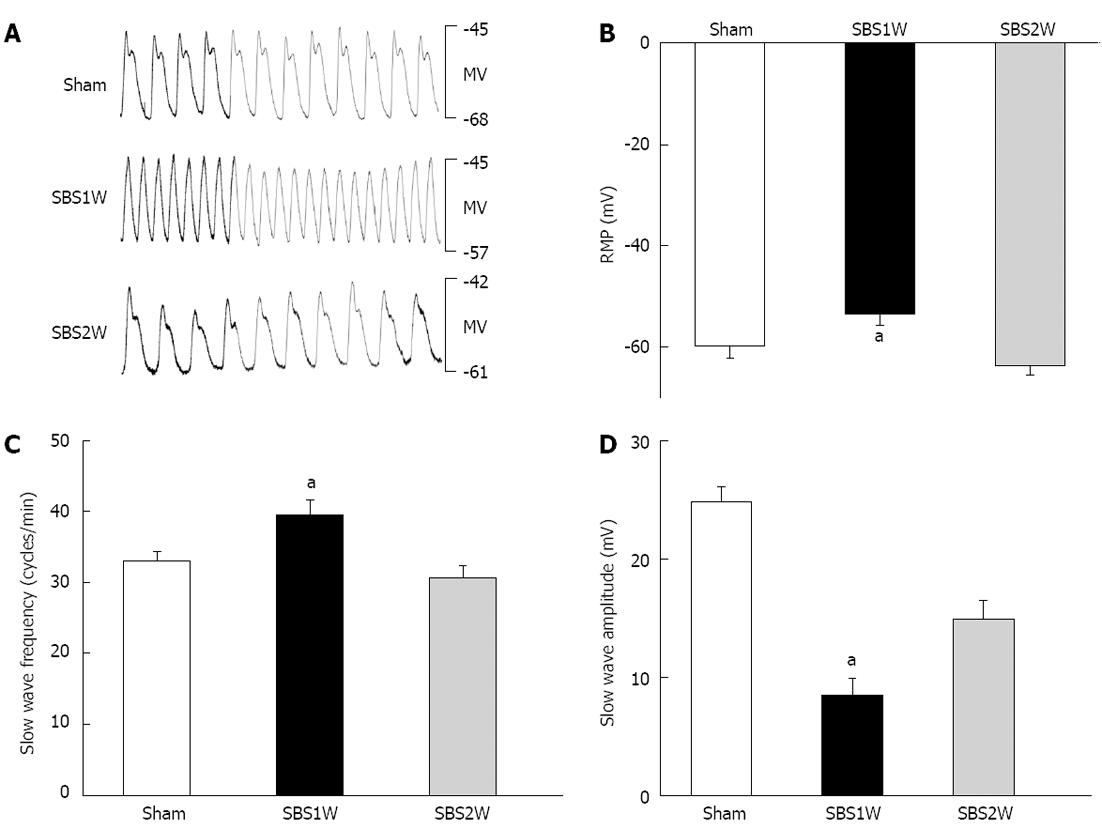
Electrophysiological studies were performed on circular muscles of the ileum in the remaining small bowel at day 7 or 14 following mSBR. The second component of slow waves was absent in SBS1W rats (Figure 3A). Circular muscle cells from the ileum of sham-operated rats (n = 7) exhibited resting membrane potentials (RMP) averaging -63.7 ± 1.8 mV and slow waves of 24.8 ± 1.3 mV in amplitude and a frequency of 33 ± 1.3 cycles/min. The electrical activities of ileum circular muscle cells were markedly different in animals 7 d after mSBR. The slow waves had considerably reduced RMP and amplitude and increased in frequency (RMP, -53.5 ± 2.1 mV; amplitude, 8.5 ± 1.4 mV; frequency, 39.5 ± 2.1 cycles/min; Figure 3B-D, respectively; P < 0.05) compared with sham-operated tissues. However, 14 d after mSBR, the electrical activity of the circular layer showed no significant changes (P > 0.05) in RMP, amplitude and frequency (RMP, -59.8 ± 2.3 mV; slow wave amplitude, 14.87 ± 1.6 mV; frequency, 30.7 ± 1.7 cycles/min) compared with sham-operated tissues.
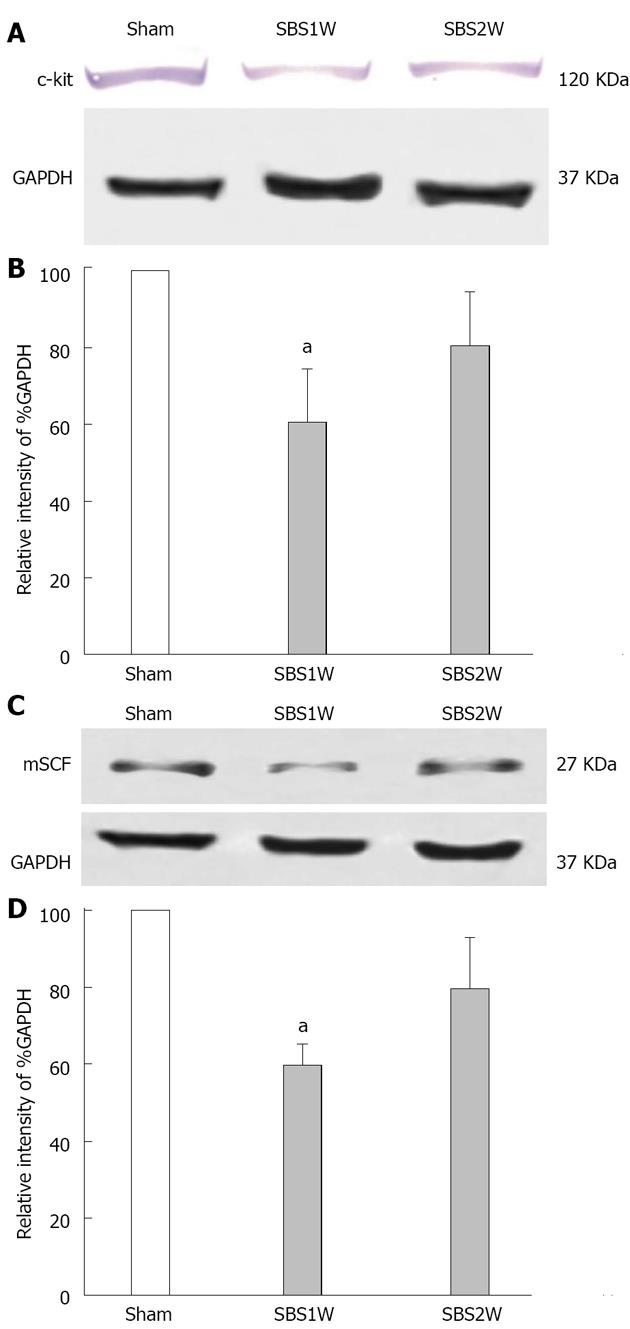
In order to investigate the roles of mSCF and c-kit in the recovery process of the electrophysiological function of smooth muscle, we determined the expression of these two proteins in mSBR and sham rat intestinal smooth muscle tissues by Western blot (Figure 4A and C). The protein expression levels of mSCF and c-kit were normalized to the internal control GAPDH (Figure 4B and D). The protein expression levels of mSCF and c-kit were significantly decreased with mSBR in the SBS1W group (n = 4; P < 0.05). The protein expression levels returned to levels found in sham-operated rats by day 14 after mSBR, which coincided with the recovery of the electrical activities of ileal circular muscle cells.
After mSBR, adaptive alterations in the function of the remaining bowel are often accompanied by disorders of intestinal motility. However, the mechanisms regulating the adaptation and motility disorders are not clear. A number of factors have been implicated in the pathogenesis of intestinal dysfunction, including changes in the number, density and ultrastructural morphology of ICC[11,20-22]. Alterations in the normal function of ICC have been reported in many intestinal disorders. In our previous studies, the unexpected finding of contractile dysfunction after mSBR prompted the investigation of whether intestinal dysfunction after mSBR was also mediated by ICC depletion[3].
ICC are found between and within smooth muscle layers of the gastrointestinal tract from the esophagus to the internal anal sphincter[23,24]. ICC specifically express the proto-oncogene c-kit that encodes a receptor tyrosine kinase. c-kit expression can be clearly detected with the immunohistochemistry method and is a valuable tool for determining ICC structure, localization and distribution of cell networks.
Two separate functional groups of ICC exist in the lumen of the gastrointestinal tract: myenteric ICC (ICC-MY) and intramuscular ICC (ICC-IM). Networks of ICC-MY are located within the intermuscular space at the level of the myenteric plexus between the circular and longitudinal muscle layers. The plexus of ICC-MY, like the sino-atrial node, is the dominant pacemaker center that triggers the generation of slow waves, which are essential for orderly segmenting and peristaltic contractions in the tunica muscularis[25,26]. A second population of cells, ICC-IM, is localized within the muscle layers of the gastrointestinal tract and is innervated preferentially by enteric motor nerves. ICC-IM are closely associated with not only enteric motor nerves but also vagal afferent nerves. Elongated ICC-IM mediate efferent inputs to smooth muscle cells and the pacemaker apparatus, as well as relay afferent mechanical signals[27,28].
We hypothesize that ICC depletion is central to the pathogenesis of many intestinal disorders. ICC are reduced or otherwise dysfunctional in several gastrointestinal tract dysmotilities, including achalasia, diabetic and idiopathic gastroparesis, mechanical ileus, intestinal pseudo-obstructions, slow-transit constipation, inflammations and malformations[29-31]. Rolle et al[30] reported that intestinal neuronal dysplasia was associated with loss or deficiency of ICC networks in the neonatal period. In addition, Chang et al[32] reported that electrical slow waves were significantly disrupted and accompanied by disruption of ICC function and network in the obstructed ileum of mice.
In the current study, an absence or reduction in the number of ICCs was observed in the remaining bowel of an animal model of SBS. Utilizing intracellular recording of smooth muscle cells in isolated segments of the remnant ileal tissues, a reduction of rhythmic contractions and disruption of electrical slow waves were associated with the disruption of ICC and their network in the remnant ileum at 7 d in SBS rats. Slow waves are composed of two components: one produced by electrogenic propagation of driving potentials from the ICC-MY and the other formed by slow potentials from the ICC-IM[32,33]. In the current study, the frequency of slow waves changed in response to RMP changes as depolarization increased the frequency. Slow waves detected in SBS1W rats were lacking the secondary wave component compared with those in sham-operated and SBS2W rats. These differences suggested that alterations in the function of ICC account for the disruption of slow waves. This finding supports the concept that when ICC and their network were disrupted from the SBS ileum, the amplitude of slow waves decreased and the shape of slow waves and neural responses were lost due to the loss of ICC[12,32].
Furthermore, c-kit signaling is essential for normal development of ICC and is required for long-term ICC survival and function[7,8]. This was first demonstrated by the blockade of c-kit signaling postnatally with an antagonistic anti-kit antibody. Inhibition of c-kit signaling resulted in a severe anomaly of gut motility with depletion of c-kit-positive ICC-MY cells in the myenteric regions of the small intestine[33]. Albertí et al[34] also reported a deficient population of ICC in Ws/Ws rats harboring mutations in c-kit. In the gastrointestinal tract, c-kit expression on the cell surface of ICC is activated by mSCF produced by surrounding smooth muscle cells. Maintenance of ICC requires mSCF produced locally within the tunica muscularis. Another study reported a reduction in the content of mSCF in the stomach of non-obese diabetic mice[35]. These mice also exhibited marked depletion of ICC and a reduction in expression level of mSCF to one-third of that in normal mice. These studies support the hypothesis that reduced mSCF/c-kit signaling may underlie the decrease in ICC numbers and its network in obstructed mice[36].
In our study, there was a reduction in mSCF/c-kit in the remnant ileum of SBS1W rats. The correlation between the decrease in ICC numbers and reduction in mSCF/c-kit protein expression levels suggest that reduced mSCF/c-kit in the gastrointestinal tract may be involved in the disruption of ICC function in the remnant ileum of rats with SBS. Although previous work has shown that inflammatory diseases can induce the disruption of ICC and their network, the underlying mechanism by which the expression of mSCF in intestinal smooth muscle is decreased during SBS remains unclear. Additional studies are warranted to determine the role of mSCF and c-kit in the disruption of ICC.
In conclusion, the current study showed modifications of the ultrastructure morphology of ICC, phenotypic changes in ICC and subsequent altered electrophysiological functional activity in the ileum after mSBR, which may be associated with the mechanical alterations. In addition, the association between motility disorders after mSBR and the changes in ICC should be further evaluated.
Motility disorders are a prevalent condition observed in the residual small bowel after massive small bowel resection (mSBR). A number of factors have been implicated in the pathogenesis of intestinal dysfunction. A potential role for interstitial cells of Cajal (ICC) networks in intestinal disorders after mSBR has not previously been reported.
ICC are organized in distinct networks and serve several different functions. ICC generate the electrical slow wave and also set the smooth muscle membrane potential, are mechanosensors and modulate neuronal input to smooth muscle. Moreover, ICC play a protective role in gastrointestinal motility, although disruption of ICC networks are certainly not the only reason for intestinal dysfunction in some cases.
The authors show that disruption of ICC networks is associated with intestinal dysfunction in rats after mSBR. This role of the ICC has not previously been reported and identifies a potential new therapeutic target intestinal disorders after mSBR.
A greater understanding of mechanism of intestinal dysfunction after mSBR will help in utilizing its diverse effects more efficiently. Pharmacotherapy (such as glucagon-like peptide-2, which is supposed to improve the function of intestinal ileum in rats with short bowel syndrome) holds promise as an adjuvant treatment modality for short bowel syndrome.
Phenotypic changes in ICC refers to changes in the structure and function of ICC.
The authors investigate an attractive subject of Cajal network involvement in intestinal dysfunction after small bowel resection, using an experimental rat model. Experimental setup and methodology are appropriate and the study hypothesis and overall manuscript are well presented.
P- Reviewer Chatzaki E S- Editor Gou SX L- Editor Cant MR E- Editor Zhang DN
| 1. | Kinoshita K, Hori M, Fujisawa M, Sato K, Ohama T, Momotani E, Ozaki H. Role of TNF-alpha in muscularis inflammation and motility disorder in a TNBS-induced colitis model: clues from TNF-alpha-deficient mice. Neurogastroenterol Motil. 2006;18:578-588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 49] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 2. | Martin CA, Bernabe KQ, Taylor JA, Nair R, Paul RJ, Guo J, Erwin CR, Warner BW. Resection-induced intestinal adaptation and the role of enteric smooth muscle. J Pediatr Surg. 2008;43:1011-1017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 3. | Chen J, Wen J, Cai W. Smooth muscle adaptation and recovery of contractility after massive small bowel resection in rats. Exp Biol Med (Maywood). 2012;237:578-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 4. | Ordög T, Redelman D, Horváth VJ, Miller LJ, Horowitz B, Sanders KM. Quantitative analysis by flow cytometry of interstitial cells of Cajal, pacemakers, and mediators of neurotransmission in the gastrointestinal tract. Cytometry A. 2004;62:139-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Takaki M. Gut pacemaker cells: the interstitial cells of Cajal (ICC). J Smooth Muscle Res. 2003;39:137-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 131] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 6. | Huizinga JD, Lammers WJ. Gut peristalsis is governed by a multitude of cooperating mechanisms. Am J Physiol Gastrointest Liver Physiol. 2009;296:G1-G8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 257] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 7. | Sanders KM, Ward SM. Kit mutants and gastrointestinal physiology. J Physiol. 2007;578:33-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 83] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 8. | Huizinga JD, Thuneberg L, Klüppel M, Malysz J, Mikkelsen HB, Bernstein A. W/kit gene required for interstitial cells of Cajal and for intestinal pacemaker activity. Nature. 1995;373:347-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1032] [Cited by in RCA: 1074] [Article Influence: 35.8] [Reference Citation Analysis (0)] |
| 9. | Sanders KM. A case for interstitial cells of Cajal as pacemakers and mediators of neurotransmission in the gastrointestinal tract. Gastroenterology. 1996;111:492-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 765] [Cited by in RCA: 747] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 10. | Ward SM. Interstitial cells of Cajal in enteric neurotransmission. Gut. 2000;47 Suppl 4:iv40-iv43; discussion iv52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 11. | Long QL, Fang DC, Shi HT, Luo YH. Gastro-electric dysrhythm and lack of gastric interstitial cells of cajal. World J Gastroenterol. 2004;10:1227-1230. [PubMed] |
| 12. | Won KJ, Suzuki T, Hori M, Ozaki H. Motility disorder in experimentally obstructed intestine: relationship between muscularis inflammation and disruption of the ICC network. Neurogastroenterol Motil. 2006;18:53-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 63] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 13. | Lorincz A, Redelman D, Horváth VJ, Bardsley MR, Chen H, Ordög T. Progenitors of interstitial cells of cajal in the postnatal murine stomach. Gastroenterology. 2008;134:1083-1093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 134] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 14. | Horváth VJ, Vittal H, Lörincz A, Chen H, Almeida-Porada G, Redelman D, Ordög T. Reduced stem cell factor links smooth myopathy and loss of interstitial cells of cajal in murine diabetic gastroparesis. Gastroenterology. 2006;130:759-770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 15. | Wang XY, Albertí E, White EJ, Mikkelsen HB, Larsen JO, Jiménez M, Huizinga JD. Igf1r+/CD34+ immature ICC are putative adult progenitor cells, identified ultrastructurally as fibroblast-like ICC in Ws/Ws rat colon. J Cell Mol Med. 2009;13:3528-3540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 16. | He CL, Soffer EE, Ferris CD, Walsh RM, Szurszewski JH, Farrugia G. Loss of interstitial cells of cajal and inhibitory innervation in insulin-dependent diabetes. Gastroenterology. 2001;121:427-434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 237] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 17. | Washizawa N, Gu LH, Gu L, Openo KP, Jones DP, Ziegler TR. Comparative effects of glucagon-like peptide-2 (GLP-2), growth hormone (GH), and keratinocyte growth factor (KGF) on markers of gut adaptation after massive small bowel resection in rats. JPEN J Parenter Enteral Nutr. 2004;28:399-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 47] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 18. | Chen J, Chen H, Sanders KM, Perrino BA. Regulation of SRF/CArG-dependent gene transcription during chronic partial obstruction of murine small intestine. Neurogastroenterol Motil. 2008;20:829-842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 19. | Suzuki H, Hirst GD. Regenerative potentials evoked in circular smooth muscle of the antral region of guinea-pig stomach. J Physiol. 1999;517:563-573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 121] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 20. | Hudson N, Mayhew I, Pearson G. A reduction in interstitial cells of Cajal in horses with equine dysautonomia (grass sickness). Auton Neurosci. 2001;92:37-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 21. | Rolle U, Piotrowska AP, Nemeth L, Puri P. Altered distribution of interstitial cells of Cajal in Hirschsprung disease. Arch Pathol Lab Med. 2002;126:928-933. [PubMed] |
| 22. | Zárate N, Mearin F, Wang XY, Hewlett B, Huizinga JD, Malagelada JR. Severe idiopathic gastroparesis due to neuronal and interstitial cells of Cajal degeneration: pathological findings and management. Gut. 2003;52:966-970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 97] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 23. | Daniel EE, Posey-Daniel V. Neuromuscular structures in opossum esophagus: role of interstitial cells of Cajal. Am J Physiol. 1984;246:G305-G315. [PubMed] |
| 24. | Hagger R, Gharaie S, Finlayson C, Kumar D. Distribution of the interstitial cells of Cajal in the human anorectum. J Auton Nerv Syst. 1998;73:75-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 43] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 25. | Ward SM, Beckett EA, Wang X, Baker F, Khoyi M, Sanders KM. Interstitial cells of Cajal mediate cholinergic neurotransmission from enteric motor neurons. J Neurosci. 2000;20:1393-1403. [PubMed] |
| 26. | Sanders KM, Koh SD, Ward SM. Interstitial cells of cajal as pacemakers in the gastrointestinal tract. Annu Rev Physiol. 2006;68:307-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 447] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 27. | Ward SM, Sanders KM. Interstitial cells of Cajal: primary targets of enteric motor innervation. Anat Rec. 2001;262:125-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 28. | Fox EA, Phillips RJ, Martinson FA, Baronowsky EA, Powley TL. C-Kit mutant mice have a selective loss of vagal intramuscular mechanoreceptors in the forestomach. Anat Embryol (Berl). 2001;204:11-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 29. | Burns AJ. Disorders of interstitial cells of Cajal. J Pediatr Gastroenterol Nutr. 2007;45 Suppl 2:S103-S106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 30. | Rolle U, Piaseczna-Piotrowska A, Puri P. Interstitial cells of Cajal in the normal gut and in intestinal motility disorders of childhood. Pediatr Surg Int. 2007;23:1139-1152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 35] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 31. | Farrugia G. Interstitial cells of Cajal in health and disease. Neurogastroenterol Motil. 2008;20 Suppl 1:54-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 256] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 32. | Chang IY, Glasgow NJ, Takayama I, Horiguchi K, Sanders KM, Ward SM. Loss of interstitial cells of Cajal and development of electrical dysfunction in murine small bowel obstruction. J Physiol. 2001;536:555-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 166] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 33. | Torihashi S, Nishi K, Tokutomi Y, Nishi T, Ward S, Sanders KM. Blockade of kit signaling induces transdifferentiation of interstitial cells of cajal to a smooth muscle phenotype. Gastroenterology. 1999;117:140-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 227] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 34. | Albertí E, Mikkelsen HB, Wang XY, Díaz M, Larsen JO, Huizinga JD, Jiménez M. Pacemaker activity and inhibitory neurotransmission in the colon of Ws/Ws mutant rats. Am J Physiol Gastrointest Liver Physiol. 2007;292:G1499-G1510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 56] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 35. | Takeda M, Takayama I, Terada N, Baba T, Ward SM, Ohno S, Fujino MA. Immunoelectron-microscopic study of Kit-expressing cells in the jejunum of wildtype and Ws/Ws rats. Cell Tissue Res. 2001;304:21-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 21] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 36. | Guo X, Huang X, Wu YS, Liu DH, Lu HL, Kim YC, Xu WX. Down-regulation of hydrogen sulfide biosynthesis accompanies murine interstitial cells of Cajal dysfunction in partial ileal obstruction. PLoS One. 2012;7:e48249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |